
3rd Quarter 2023 Financial Results & Corporate Update November 6, 2023

This Slide Presentation Includes Forward-Looking Statements 2 This presentation contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995, as amended, including, but not limited to, statements concerning: BioNTech's expected revenues and net profit related to sales of BioNTech's COVID-19 vaccine, referred to as COMIRNATY® where approved for use under full or conditional marketing authorization, in territories controlled by BioNTech's collaboration partners, particularly for those figures that are derived from preliminary estimates provided by BioNTech's partners; the rate and degree of market acceptance of BioNTech's COVID-19 vaccine and, if approved, BioNTech's investigational medicines; expectations regarding anticipated changes in COVID-19 vaccine demand, including changes to the ordering environment, seasonality and expected regulatory recommendations to adapt vaccines to address new variants or sublineages; the initiation, timing, progress, results, and cost of BioNTech's research and development programs, including those relating to additional formulations of BioNTech's COVID-19 vaccine, and BioNTech's current and future preclinical studies and clinical trials, including statements regarding the timing of initiation and completion of studies or trials and related preparatory work and the availability of results; our expectations with respect to our intellectual property; the impact of the Company’s collaboration and licensing agreements; the development of sustainable vaccine production and supply solutions and the nature and feasibility of these solutions; and BioNTech's estimates of commercial and other revenues, cost of sales, research and development expenses, sales and marketing expenses, general and administrative expenses, capital expenditures, income taxes, net profit, cash, cash equivalents and security investments, shares outstanding and cash outflows and share consideration. In some cases, forward-looking statements can be identified by terminology such as “will,” “may,” “should,” “expects,” “intends,” “plans,” “aims,” “anticipates,” “believes,” “estimates,” “predicts,” “potential,” “continue,” or the negative of these terms or other comparable terminology, although not all forward-looking statements contain these words. The forward-looking statements in this presentation are neither promises nor guarantees, and you should not place undue reliance on these forward-looking statements because they involve known and unknown risks, uncertainties, and other factors, many of which are beyond BioNTech’s control, and which could cause actual results to differ materially from those expressed or implied by these forward-looking statements. These risks and uncertainties include, but are not limited to: BioNTech's pricing and coverage negotiations with governmental authorities, private health insurers and other third-party payors after BioNTech's initial sales to national governments; the future commercial demand and medical need for initial or booster doses of a COVID-19 vaccine; competition from other COVID-19 vaccines or related to BioNTech's other product candidates, including those with different mechanisms of action and different manufacturing and distribution constraints, on the basis of, among other things, efficacy, cost, convenience of storage and distribution, breadth of approved use, side-effect profile and durability of immune response; the timing of and BioNTech's ability to obtain and maintain regulatory approval for BioNTech's product candidates; the ability of BioNTech’s COVID-19 vaccines to prevent COVID-19 caused by emerging virus variants; BioNTech's and its counterparties’ ability to manage and source necessary energy resources; BioNTech's ability to identify research opportunities and discover and develop investigational medicines; the ability and willingness of BioNTech's third-party collaborators to continue research and development activities relating to BioNTech's development candidates and investigational medicines; the impact of the COVID-19 pandemic on BioNTech's development programs, supply chain, collaborators and financial performance; unforeseen safety issues and claims for potential personal injury or death arising from the use of BioNTech's COVID-19 vaccine and other products and product candidates developed or manufactured by BioNTech; BioNTech's and its collaborators’ ability to commercialize and market BioNTech's COVID-19 vaccine and, if approved, its product candidates; BioNTech's ability to manage its development and expansion; regulatory developments in the United States and other countries; BioNTech's ability to effectively scale BioNTech's production capabilities and manufacture BioNTech's products, including BioNTech's target COVID-19 vaccine production levels, and BioNTech's product candidates; risks relating to the global financial system and markets; and other factors not known to BioNTech at this time. You should review the risks and uncertainties described under the heading “Risk Factors” in BioNTech’s Report on Form 6-K for the period ended September 30, 2023 and in subsequent filings made by BioNTech with the SEC, which are available on the SEC’s website at https://www.sec.gov/. Except as required by law, BioNTech disclaims any intention or responsibility for updating or revising any forward-looking statements contained in this presentation in the event of new information, future developments or otherwise. These forward-looking statements are based on BioNTech’s current expectations and speak only as of the date hereof.

Financial Results Jens Holstein, Chief Financial Officer3 Strategic Outlook Ryan Richardson, Chief Strategy Officer4 Pipeline Update Özlem Türeci, Chief Medical Officer2 3rd Quarter 2023 Highlights Ugur Sahin, Chief Executive Officer1

1 3rd Quarter 2023 Highlights Ugur Sahin, Chief Executive Officer

Clinical data updates Strategic Priorities and Achievements in Q3 2023 and Post Period 5 1. Partnered with Pfizer; 2. Partnered with DualityBio; 3. Partnered with Genmab; 4. Partnered with OncoC4; 5. Partnered with Regeneron; 6. Partnered with CEPI. Initiated Trial COVID-19 franchise1 Infectious diseases Sustain leadership in COVID-19 vaccines Advance next-gen vaccines Initiate and accelerate clinical programs for diseases of unmet medical need Q3 Achievements Successful launch of XBB.1.5-adapted monovalent COVID-19 vaccine for 2023/2024 season 2023 Strategic Priorities Immuno-oncology Advance oncology pipeline across multiple solid tumors Initiate multiple trials with registrational potential Clinical data updates BNT1666 (Mpox): First-in-humanESMO BNT325/DB-13052 BNT314/GEN10593 BNT211 BNT221 SITC BNT316/ONC-3924 (gotistobart) BNT221 BNT116 ESGO BNT323/DB-13032 Initiated trials BNT323/DB-13032: Ph3 BNT324/DB-13112: Ph1/2 BNT311/GEN10463: Ph2 BNT314//GEN10593: First-in-human BNT1165: Ph2 Partnerships DualityBio MediLink Biotheus COVID-19-Influenza combo Partnership Coalition for Epidemic Preparedness Innovations Peer reviewed papers Murdoch et al, Infect Dis Ther 2023 Muik et al, Cell Rep 2023 Arieta et al, Cell 2023 Beguir et al, Comput Biol Med 2023 Peer reviewed papers Mackensen et al, Nature Med 2023 Bähr-Mahmud et al, Oncoimmunol 2023 Simon et al, J. Transl Med 2023

Strong Global Distribution COMIRNATY1 Q3 Highlights 1. Partnered with Pfizer. 2.COMIRNATY approved for prevention of COVID-19 as a single dose for individuals 5 years of age and older and as a 3-dose series in individuals 6 months through 4 years of age. 3. COMIRNATY may be administered as a booster in people aged 12 years and older who have received at least a primary vaccination course against COVID-19. EMA = European Medicines Agency; FDA = Food and Drug Administration; sBLA = supplemental Biologics License Application; EUA = Emergency Use Authorization. First-to-market Omicron XBB.1.5-adapted monovalent vaccine Distributed to over 40 countries and regions worldwide EU: EMA Full Marketing Authorization for ages 6 months and older2 US: FDA sBLA for individuals 12+ years and EUA for ages 6 months to 11 years old3 Other select regions with approval: UK, Japan, Canada, Australia, Singapore and South Korea Rapid Regulatory Advancement 6
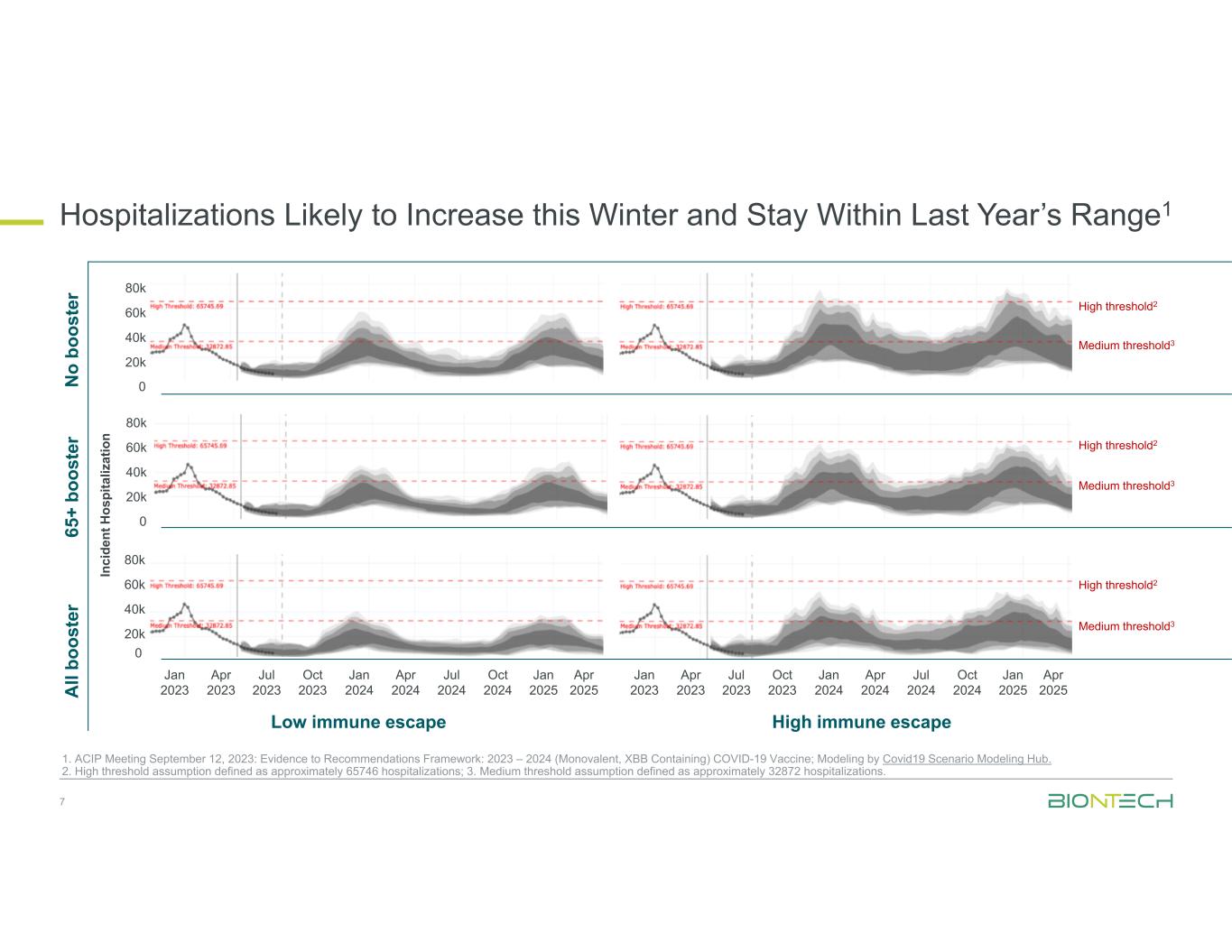
0 20k 40k 60k 80k 0 20k 40k 60k 80k 0 20k 40k 60k 80k Jan 2023 Apr 2023 Jul 2023 Oct 2023 Jan 2024 Apr 2024 Jul 2024 Oct 2024 Jan 2025 Apr 2025 Jan 2023 Apr 2023 Jul 2023 Oct 2023 Jan 2024 Apr 2024 Jul 2024 Oct 2024 Jan 2025 Apr 2025 Hospitalizations Likely to Increase this Winter and Stay Within Last Year’s Range1 7 1. ACIP Meeting September 12, 2023: Evidence to Recommendations Framework: 2023 – 2024 (Monovalent, XBB Containing) COVID-19 Vaccine; Modeling by Covid19 Scenario Modeling Hub. 2. High threshold assumption defined as approximately 65746 hospitalizations; 3. Medium threshold assumption defined as approximately 32872 hospitalizations. In ci de nt H os pi ta liz at io n N o bo os te r A ll bo os te r 65 + bo os te r High immune escapeLow immune escape High threshold2 Medium threshold3 High threshold2 Medium threshold3 High threshold2 Medium threshold3

Probability of developing long COVID in vaccinated vs non-vaccinated individuals Statistically Significant Reduction in Developing Long COVID after mRNA Vaccination 8 Prevalence of ongoing symptoms lasting at least 3 months after COVID-19 infection by age, regardless of COVID status, U.S. 1 0.8 1.9 6.8 9 7.4 4.2 0.2 0.3 0.8 2.7 4.7 3.8 2.3 0 2 4 6 8 10 0 - 5 years 6 - 11 years 12 - 17 years 18 - 34 years 35 - 49 years 50 - 64 years ≥ 65 years Ever Current Pr ev al en ce (% ) Vaccinated Non-vaccinated 1 – Pr ob ab ili ty o f L C D ia gn os is 10–20% of SARS-CoV-2 infected people may develop long COVID1,2 Long COVID also affects children and may lead to long term effects on development3 Post COVID-19 conditions are associated with increased healthcare utilization4 COVID-19 vaccination reduces post COVID-19 conditions among children and adults5,6 0.96 0.97 0.98 0.99 1.00 45 100 150 200 250 300 Time from COVID index, day Inverse probability of treatment weighting adjusted Kaplan–Meier Curves Brannock et al, Nature Comm. 2023CDC ACIP September 12, 2023 1. Davis, H.E., et al. Nat Rev Microbiol 2023; 2. Editorial, The Lancet Vol 401 2023; 3. Villapol, S. et al Sci Rep Nat Res 2022; 4. Katz GM et al. JAMA Health Forum 2023; 5. Saydah, Centers for Disease Control Advisory Committee on Immunization Practices, September 12, 2023; 6. Brannock et al, Nature Comm. 2023. LC: Long Covid.

XBB.1.5-adapted vaccine Effective against multiple variants of concern5 Long-term health consequences Accumulating evidence demonstrates that COVID-19 vaccination may reduce long COVID conditions4 Continuous evolution Ongoing antigenic evolution of SARS-CoV-21,2 Risk remains high For severe COVID-19 in vulnerable populations3 Long-Term Need for Annually Adapted Vaccines Anticipated 9 1. World Health Organization tracking SARS-CoV-2 variant www.who.int/en/activities/tracking-SARS-CoV-2-variants accessed October 30, 2023; 2. GISAID https://gisaid.org/ accessed October 30, 2023; 3. FDA Briefing Document Vaccines and Related Biological Products Advisory Committee Meeting June 15, 2023; 4 Brannock et al, Nature Comm. 2023; 5. Stankov M. V. et al., medRxiv pre-print, accessed October 5, 2023. Annual and/or SEASONAL VACCINATION with variant-adapted vaccines expected for the foreseeable future

Pipeline Update Özlem Türeci, Chief Medical Officer

Infectious Disease Pipeline: Achievements in Q3 2023 and Post Period 11 1. Partnered with Pfizer; 2. Collaboration with University of Pennsylvania; 3. Collaboration with Bill & Melinda Gates Foundation; 4. Partnered with CEPI; HSV = herpes simplex virus; qFlu = quadrivalent influenza. Please find current product information for Comirnaty at https://www.ema.europa.eu/en/documents/product-information/comirnaty-epar-product-information_en.pdf and https://www.fda.gov/media/151707/download. CommercialPhase 3Phase 2Phase 1/2Phase 1Product CandidateIndication COMIRNATY® COVID-191 BNT162b2 Original/Omicron BA.4/5-adapted bivalent BNT162b2 Original/Omicron BA.1-adapted bivalent LaunchedBNT162b2 Omicron XBB.1.5. BNT162b2 + BNT162b4 T-cell string BNT162b6/7 Stabilized spike antigen DataBNT162b2 BA.4/5-adapted bivalent + BNT161qFlu COVID-19 - Influenza combination1 PlannedBNT162b2 BA.4/5-adapted bivalent + BNT161 qFlu BNT161Influenza1 BNT167Shingles1 BNT163HSV2 BNT164Tuberculosis3 BNT165Malaria NewBNT166Mpox4 Infectious Disease programs partnered with Pfizer Other Infectious Disease programs ✓ ✓ ✓
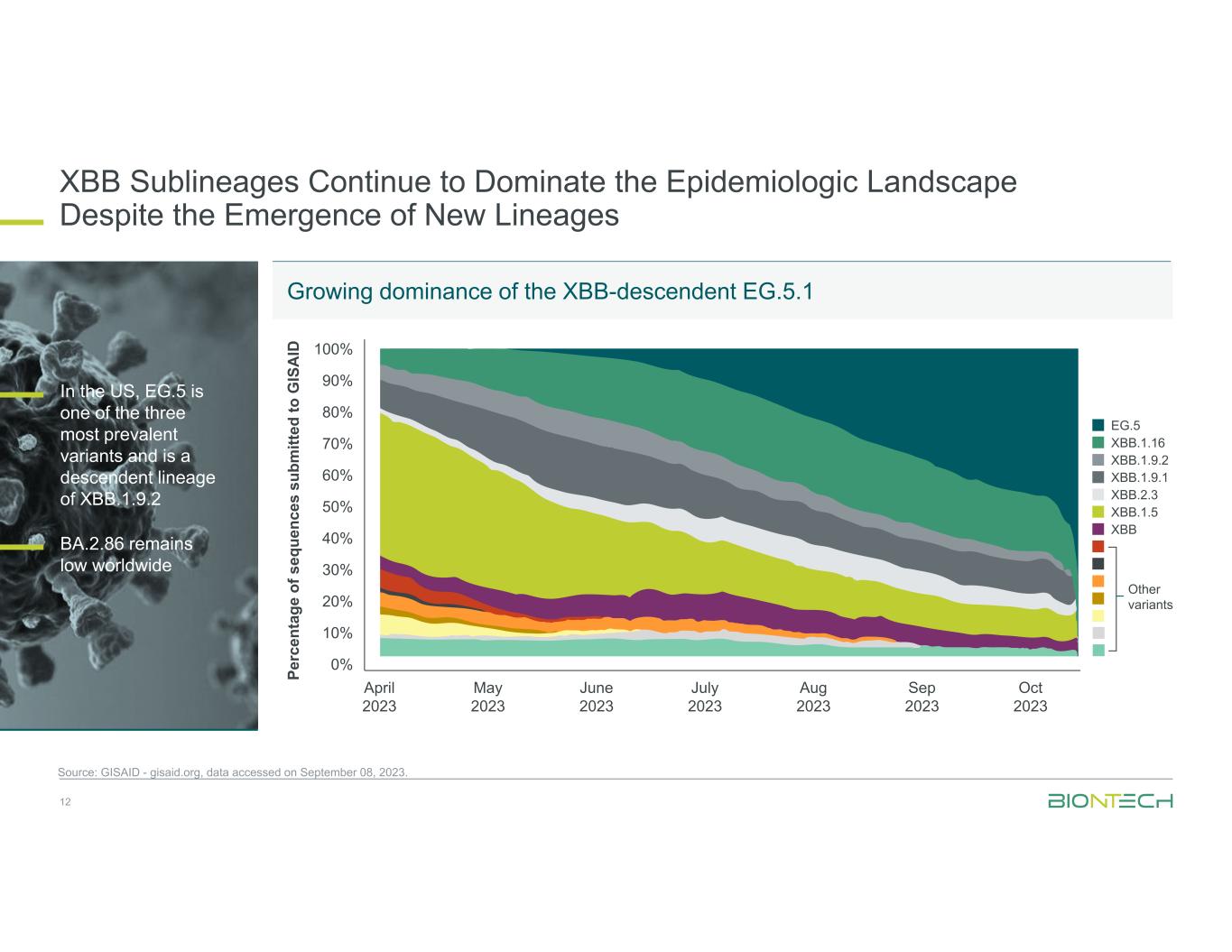
XBB Sublineages Continue to Dominate the Epidemiologic Landscape Despite the Emergence of New Lineages 12 Source: GISAID - gisaid.org, data accessed on September 08, 2023. Growing dominance of the XBB-descendent EG.5.1 In the US, EG.5 is one of the three most prevalent variants and is a descendent lineage of XBB.1.9.2 BA.2.86 remains low worldwide April 2023 100% 0%Pe rc en ta ge o f s eq ue nc es s ub m itt ed to G IS A ID 90% 80% 70% 60% 50% 40% 30% 20% 10% May 2023 June 2023 July 2023 Aug 2023 Sep 2023 Oct 2023 EG.5 XBB.1.16 XBB.1.9.2 XBB.1.9.1 XBB.2.3 XBB.1.5 XBB Other variants

Monovalent XBB.1.5 Adapted Vaccine Elicits Higher nAbs Responses against VoCs Compared to the Bivalent BA.4/5 Adapted Vaccine in Preclinical Models 13 Source: Data were generated by the same pseudovirus neutralization assay and from sera of same mouse study that generated data that were presented at VRBPAC (Vaccines and Related Biological Products Advisory Committee) June 15, 2023 Meeting, https://www.fda.gov/media/169541/download). NAbs = neutralizing antibodies; VOCs= variants of concern. XBB.1.5-adapted monovalent vaccine elicits potent neutralization against various sublineages: XBB.1.5, EG.5.1, BA.2.86, XBB.1.16 and XBB.2.3 Pre-treatment: Primary series of monovalent BNT162b2 Original vaccine and a 3rd dose of Original + BA 4/5 bivalent vaccine. 4th dose: Original+BA.4/5 bivalent vaccine or a monovalent XBB.1.5 vaccine. Experimental design (mice) 3-fold increase 5-fold increase 7-fold increase 50 % N eu tr al iz at io n tit er 105 104 103 102 101 Vaccine group LOD Original + BA.4/5 XBB.1.5 XBB.1.5 EG.5.1 BA.2.86 440 572 422 2043 4069 1249 ~5-fold increase ~4-fold increase ~5-fold increase 50 % N eu tr al iz at io n tit er 106 104 103 102 101 Vaccine group LOD Original + BA.4/5 XBB.1.5 XBB.1.5 XBB.1.16 XBB.2.3 444 733 621 1800 3766 3020 105 Modjarrad, CDC ACIP September 12, 2023 Swanson, CDC VRPAC June 15, 2023
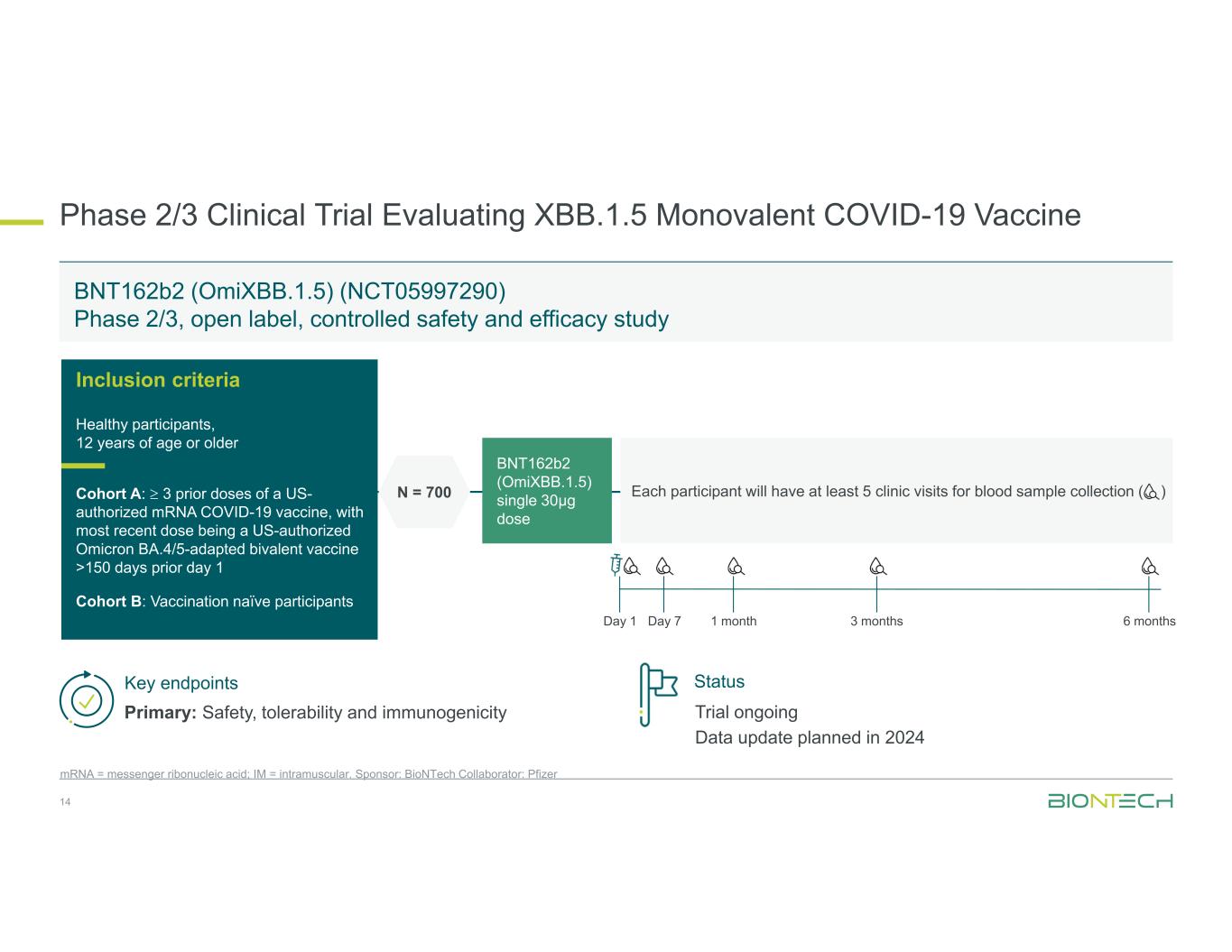
Phase 2/3 Clinical Trial Evaluating XBB.1.5 Monovalent COVID-19 Vaccine 14 mRNA = messenger ribonucleic acid; IM = intramuscular. Sponsor: BioNTech Collaborator: Pfizer BNT162b2 (OmiXBB.1.5) (NCT05997290) Phase 2/3, open label, controlled safety and efficacy study Inclusion criteria Healthy participants, 12 years of age or older Cohort A: 3 prior doses of a US- authorized mRNA COVID-19 vaccine, with most recent dose being a US-authorized Omicron BA.4/5-adapted bivalent vaccine >150 days prior day 1 Cohort B: Vaccination naïve participants BNT162b2 (OmiXBB.1.5) single 30µg dose Each participant will have at least 5 clinic visits for blood sample collection ( )N = 700 Key endpoints Primary: Safety, tolerability and immunogenicity Day 1 Day 7 1 month 3 months 6 months Status Trial ongoing Data update planned in 2024
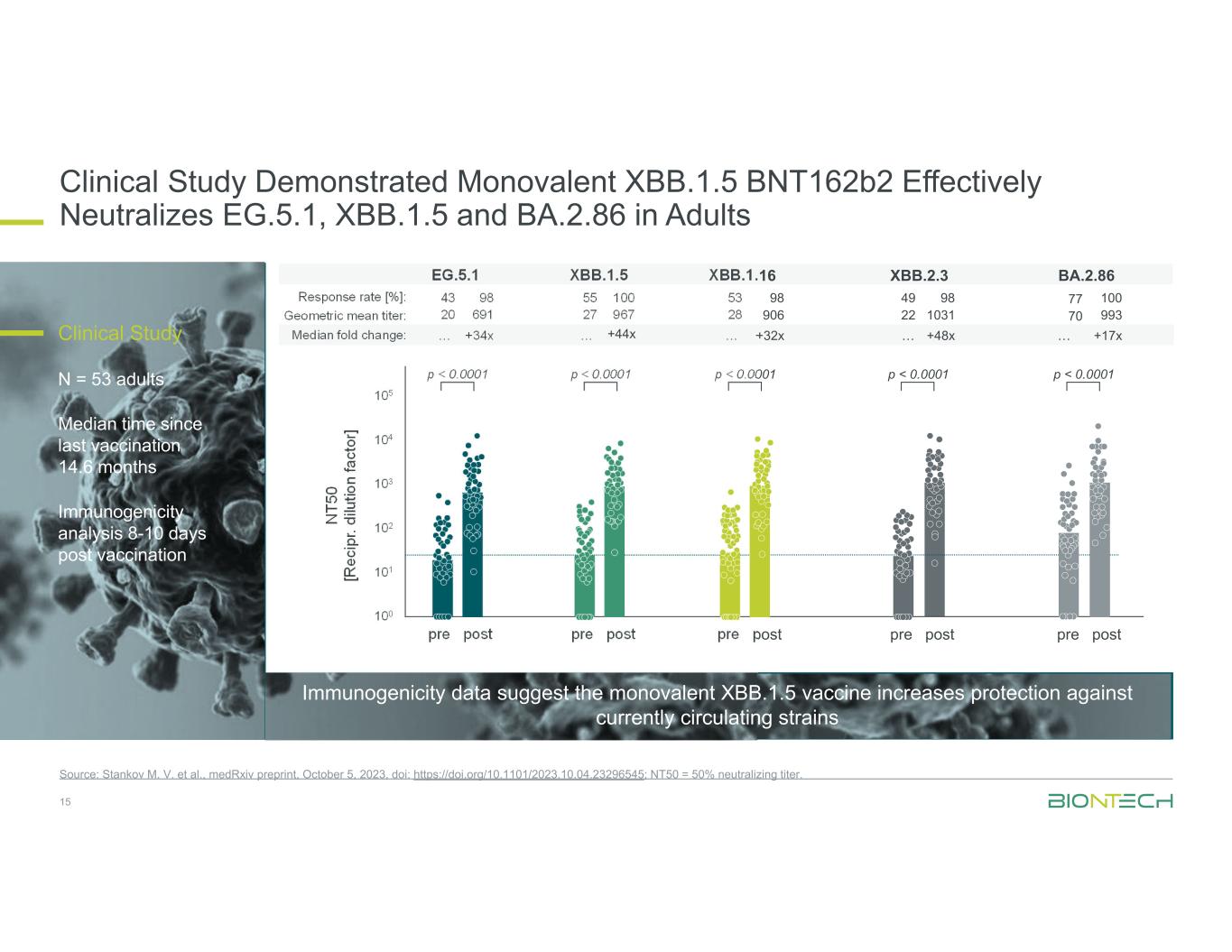
Clinical Study Demonstrated Monovalent XBB.1.5 BNT162b2 Effectively Neutralizes EG.5.1, XBB.1.5 and BA.2.86 in Adults 15 Source: Stankov M. V. et al., medRxiv preprint, October 5, 2023, doi: https://doi.org/10.1101/2023.10.04.23296545; NT50 = 50% neutralizing titer. N T5 0 [R ec ip r. di lu tio n fa ct or ] 105 104 103 102 101 100 pre post pre post pre post pre post pre post p < 0.0001 p < 0.0001 p < 0.0001 p < 0.0001 p < 0.0001 Median fold change: Geometric mean titer: Response rate [%]: EG.5.1 43 98 20 691 … +34x XBB.1.5 55 100 27 967 … XBB.1.16 53 98 28 906 … +32x XBB.2.3 49 98 22 1031 … +48x BA.2.86 77 100 70 993 … +17x+44xClinical Study N = 53 adults Median time since last vaccination 14.6 months Immunogenicity analysis 8-10 days post vaccination Immunogenicity data suggest the monovalent XBB.1.5 vaccine increases protection against currently circulating strains

Oncology Pipeline: Achievements in Q3 2023 and Post Period Events 16 1. Partnered with Genentech, member of Roche Group; 2. Partnered with Regeneron; 3. Partnered with Genmab; 4. Partnered with OncoC4; 5. Partnered with DualityBio; 6. Partnered with MediLink Therapeutics. *Two phase 1/2 clinical trials in patients with solid tumors are ongoing in combination with immune checkpoint inhibitor +/- chemotherapy. NSCLC = non-small cell lung cancer; mCRPC = metastatic castration resistant prostate cancer; LPC = localized prostate cancer; HPV = human papillomavirus; PDAC = pancreatic ductal adenocarcinoma; CRC = colorectal cancer; CLDN = claudin; IL = interleukin; 1L = first line; R/R = relapsed/refractory; HER2/HER3 = human epidermal growth factor 2/3; sLeA = sialyl-Lewis A antigen; TROP2 = tumor-associated calcium transducer 2. BNT211 (CLDN6) Multiple solid tumors BNT311/GEN10463 (PD-L1x4-1BB) Multiple solid tumors BNT411 (TLR7) Multiple solid tumors BNT311/GEN10463 (PD-L1x4-1BB) R/R met. NSCLC, +/- pembrolizumab BNT312/GEN10423 * (CD40x4-1BB) Multiple solid tumors BNT313/GEN10533 (CD27) Multiple solid tumors BNT316/ONC-392 (gotistobart)4 (CTLA-4) Multiple solid tumors BNT1122 mCRPC & high risk LPC BNT151 (IL-2 variant) Multiple solid tumors BNT142 Multiple CLDN6-pos. adv. solid tumors BNT325/DB-13055 (TROP2) Multiple solid tumors BNT316/ONC-392 (gotistobart)4 (CTLA-4) anti-PD-1/PD-L1 experienced NSCLC BNT323/DB-13035 (HER2) Multiple solid tumors BNT1112 aPD(L)1-R/R melanoma, + cemiplimab BNT113 1L rec./met. HPV16+ PDL1+ head and neck cancer, + pembrolizumab Autogene cevumeran/BNT1221 1L adv. melanoma, + pembrolizumab Autogene cevumeran/BNT1221 Adj. ctDNA+ Stage II or III CRC BNT1162 1L adv. PD-L1 50% NSCLC, + cemiplimab Autogene cevumeran/BNT1221 Adj. PDAC, + atezolizumab + mFOLFIRINOX BNT324/DB-13115 Multiple solid tumors Phase 1 Phase 1/2 Phase 2 Phase 3 NEW BNT323/DB-13035 (HER2) HR+, HER2 low met. breast cancer PLANNED BNT116 Adv. NSCLC BNT152 + BNT153 (IL-7, IL-2) Multiple solid tumors BNT221 Refractory metastatic melanoma BNT321 (sLeA) Metastatic PDAC BNT322/GEN10563 Multiple solid tumors Autogene cevumeran/BNT1221 Multiple solid tumors NEW BNT314//GEN10593 (EpCAMx4-1BB) Multiple solid tumors PLANNED BNT316/ONC-392 (gotistobart)4 (CTLA-4) Plat.-R. ovarian cancer, + pembrolizumab Clinical data announced in Q3 and subsequently BNT326/YL2026 (HER3) Multiple solid tumors NEW BNT316/ONC-392 (gotistobart)4 mCRPC, + radiotherapy PLANNED BNT311/GEN10463 (PD-L1x4-1BB) 2L endometrial cancer, + pembrolizumab NEW

Multiple Clinical Data Readouts at Major Medical Conferences in 2H 2023 17 1. Partnered with DualityBio. ADC = antibody drug conjugate; HER2 = human epidermal growth factor 2; EC = endometrial cancer; TEAE = treatment emergent adverse event; ILD = interstitial lung disease; NSCLC = non-small cell lung cancer; ORR = objective response rate, DCR = disease control rate, DL2 = dose level 2; r/r met: resistant/refractory metastatic DLT = dose limiting toxicities. Cancer vaccine (FixVac)NEO-STIM Cell therapyCAR T-cell therapyADCADCPlatform BNT116 Advanced NSCLC BNT221 r/r met Melanoma BNT211 CLDN6+ Solid Tumors BNT325/DB-13051 Multiple Solid Tumors BNT323/DB-13031 Multiple Solid TumorsProgram FIH Ph1 • Tolerable safety profile both in monotherapy or combination • No DLTs • BNT116 + cemiplimab active in heavily pre- treated lung cancer patients Ph1 • Tumor shrinkage for 4/9 patients • Manageable safety profile with no DLTs • Polyclonal neoantigen-specific T cells response detected post-infusion Ph1/2 • Encouraging signs of activity in 13 / 22 patients at DL2: • ORR 59% • DCR 95% • Patients with CARVac improved CAR-T persistence in higher dose group • Dose-dependent increase in adverse events Ph1/2 • Encouraging efficacy signals in metastatic NSCLC patients: • unconfirmed ORR 46.2% • unconfirmed DCR 92.3% • TEAEs were manageable in lower dose levels Ph1/2 • Antitumor activity in heavily pretreated HER2-expressing EC patients: • unconfirmed ORR 58.8% • unconfirmed DCR 94.1% • No TEAEs leading to dose discontinuation • No ILD occurred Data update

3 Financial Results Jens Holstein, Chief Financial Officer

YTD 2023 Key Financial Highlights1 19 1. Financial information is prepared and presented in Euros and numbers are rounded to millions and billions of Euros in accordance with standard commercial practice. Inventory write-downs and other charges from Pfizer reduced BioNTech’s revenues by €615.4 million for the nine months ended September 30, 2023. 2. BioNTech’s profit share is estimated based on preliminary data shared between Pfizer and BioNTech as further described in the Annual Report on Form 20-F for the year ended December 31, 2022 as well as the Quarterly Report as of and for the three and nine months ended September 30, 2023, filed as an exhibit to BioNTech’s Current Report on Form 6-K filed on November 6, 2023. 3. Consists of cash and cash equivalents of €13,495.8 million and security investments of €3,471.8 million, as of September 30, 2023. The payment settling our gross profit share for the second quarter of 2023 (as defined by the contract) in the amount of €565.0 million was received from our collaboration partner subsequent to the end of the reporting period as of October 16, 2023. Profit before tax €1.94 €0.5bn €2.3 bn €17.0 bn Total revenues2 Diluted EPS Total cash plus security investments3
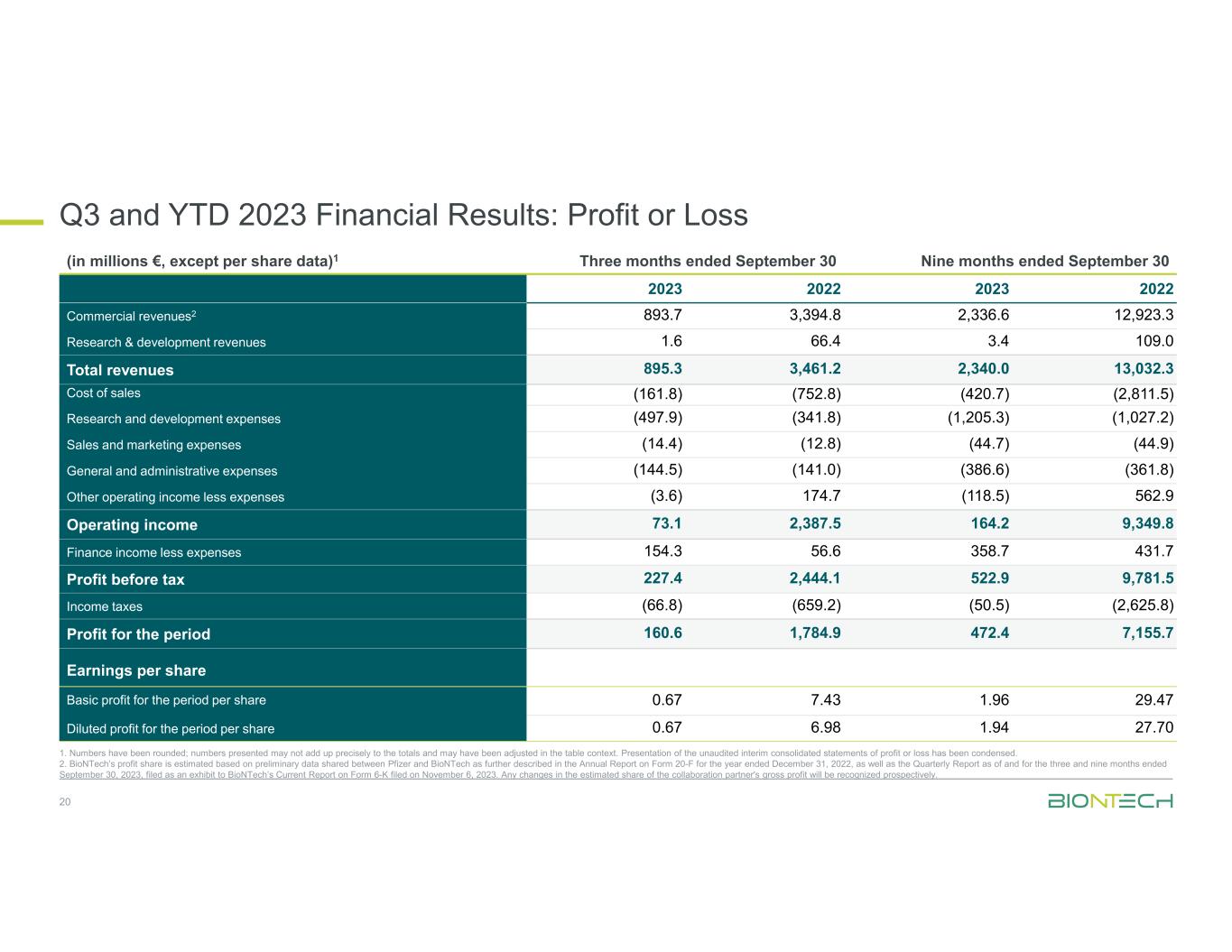
Q3 and YTD 2023 Financial Results: Profit or Loss 20 1. Numbers have been rounded; numbers presented may not add up precisely to the totals and may have been adjusted in the table context. Presentation of the unaudited interim consolidated statements of profit or loss has been condensed. 2. BioNTech’s profit share is estimated based on preliminary data shared between Pfizer and BioNTech as further described in the Annual Report on Form 20-F for the year ended December 31, 2022, as well as the Quarterly Report as of and for the three and nine months ended September 30, 2023, filed as an exhibit to BioNTech’s Current Report on Form 6-K filed on November 6, 2023. Any changes in the estimated share of the collaboration partner's gross profit will be recognized prospectively. Nine months ended September 30Three months ended September 30(in millions €, except per share data)1 2022202320222023 12,923.32,336.63,394.8893.7Commercial revenues2 109.03.466.41.6Research & development revenues 13,032.32,340.03,461.2895.3Total revenues (2,811.5)(420.7)(752.8)(161.8)Cost of sales (1,027.2)(1,205.3)(341.8)(497.9)Research and development expenses (44.9)(44.7)(12.8)(14.4)Sales and marketing expenses (361.8)(386.6)(141.0)(144.5)General and administrative expenses 562.9(118.5)174.7(3.6)Other operating income less expenses 9,349.8164.22,387.573.1Operating income 431.7358.756.6154.3Finance income less expenses 9,781.5522.92,444.1227.4Profit before tax (2,625.8)(50.5)(659.2)(66.8)Income taxes 7,155.7472.41,784.9160.6Profit for the period Earnings per share 29.471.967.430.67Basic profit for the period per share 27.701.946.980.67Diluted profit for the period per share

Implications of Inventory Write-Downs and Other Charges related to COMIRNATY 21 1. Inventory write-downs and other charges, jointly referred to as charges, were mainly triggered by market demand shifting from pandemic into endemic setting as well as the companies following the guidance from regulatory authorities on strain changes leading to the introduction and marketing of updated COVID-19 vaccines better matched to circulating sublineages. 2. Including all effects derived from the information received from BioNTech’s collaboration partner. 3. Inventories and provisions for production capacities derived from contracts with Contract Manufacturing Organizations (CMOs) that became redundant are measured reflecting contractual compensation payments under the collaboration agreements. Manufacturing variances are reflected as transfer price adjustment impacting sales to the collaboration partner and the share of the collaboration partner’s gross profit respectively once assessed as highly probable to occur. Amounts are only cash-effectively shared with the partner once materialized. Please refer to the Annual Report on Form 20-F for the year ended December 31, 2022, as well as the Quarterly Report as of and for the three and nine months ended September 30, 2023, filed as an exhibit to BioNTech’s Current Report on Form 6-K filed on November 6, 2023. Any changes to the assessment will be recognized prospectively. Write-downs / other charges by Pfizer Identified on the collaboration partner’s side Write-downs / other charges by BioNTech Identified on BioNTech’s side Initially announced estimate Up to €0.9 bn Actual YTD 2023 effect recorded €0.6 bn2 (thereof €0.5 bn in Q3 2023) BioNTech already reflected a large portion of write-downs / other charges in 2022 financial results and a smaller portion in 2023 Charges1 mainly reflect that market demand has shifted from pandemic to endemic, flu-like setting; Under the gross profit-sharing agreement charges are borne by both collaboration partners Reducing Pfizer’s gross profit, hence BioNTech’s revenues Reducing BioNTech’s gross profit3

2023 Financial Year Guidance Updated1 22 ~ €1 bn*~ €4 bn~ €5 bnEstimated BioNTech COVID-19 vaccine revenuesCOVID-19 vaccine revenues for FY 2023 ~ €600 m€1,800 – 2,000 m€2,400 – 2,600 mR&D expenses2 Planned FY 2023 expenses and capex ~ €75 m€600 – 650 m€650 – 750 mSG&A expenses ~ €300 m€200 – 300 m€500 – 600 mCapital expenditure for operating activities3 ~ 6 %points~ 21%~ 27%BioNTech Group estimated annual cash effective income tax rate4 Estimated FY 2023 tax assumptions Updated guidance Nov 2023 Initial guidance Mar 2023 Delta midpoint illustration5 1. Numbers reflect current base case projections and are calculated based on constant currency rates. Excluding external risks that are not yet known and/or quantifiable, including, but not limited to, the effects of ongoing and/or future legal disputes or related activity. 2. Numbers include effects identified from additional in-licensing arrangements, collaborations or potential M&A transactions to the extent disclosed and will be updated as needed. 3. Numbers exclude potential effects caused by or driven from in-licensing arrangements, collaborations or M&A transactions. 4. Numbers exclude potential effects caused by or driven from share-based payment settlements in the course of 2023. 5. For simplicity, midpoint in guidance ranges is applied when comparing initial 2023 guidance with Q3 2023 guidance update. 6. €0.6 billion related to Pfizer's recently-announced write-downs and other charges. Other factors influencing the COVID-19 vaccine revenues guidance include, but are not limited to, BioNTech’s and Pfizer’s lower revenue expectations for the full 2023 financial year, which take into account delays in the expected timing of regulatory approvals. * reflecting €0.6 bn6 Pfizer charges

4 Strategic Outlook Ryan Richardson, Chief Strategy Officer

Strategic Priorities and Outlook 1. Partnered with Pfizer. COVID-191 Immuno-oncology Infectious diseases Sustain market leadership in COVID-19 vaccines Advance next-gen vaccines Advance oncology pipeline across multiple solid tumors Grow pipeline of trials with registrational potential Initiate and accelerate clinical programs for high medical need indications Phase 3 COVID-19/Influenza trial due to start in the coming months Initiate trials with registrational potential New trial starts Data readouts to drive decision- making and future trial starts Continued development of next- generation vaccine constructs

Rollout Of Our XBB.1.5 Variant Adapted Vaccine1 25 1. As of October 2, 2023. 2. Company assessment 3. CDC Survey published on October 28, 2023: https://www.cdc.gov/vaccines/acip/meetings/downloads/slides-2023-10-25-26/02-COVID-Stokley-508.pdf. # of countries distributed to market share US/EU/JP (%)2 60 /90/90 40+ ~ 50retail channel in the US (%) ~30% of US population have received or plan to receive an XBB.1.5 variant-adapted vaccine3 7.1 24.6 30.6 37.6 Up to date with 2023-24 COVID-19 vaccine (%) Definitely will get vaccinated (%) Probably will get vaccinated or unsure (%) Probably or definitely will not get vaccinated (%) Maintaining COVID-19 market leadership with variant- adapted vaccine rollout Potential for continued variant-adapted vaccine uptake2

COVID-19 Vaccine Market Potential 2024 and 2025 Growth Drivers 26 • Manufacturing base reset to serve endemic market • Shift to commercialization model in some key markets • Expect continued shift to single dose vials and pre-filled syringes • Potential for increased vaccine uptake from combination and next-gen vaccines Leveraging partner’s infrastructure to maintain a lean cost base, the COVID-19 business should remain profitable for BioNTech 2024 2025 Variant adapted vaccines Combination vaccines Next-gen vaccines

Strategic Outlook for Oncology 27 1. New trial either through initiation or via in-licensing; 2. Partnered with OncoC4; 3. Partnered with DualityBio; 4. Partnered with Medilink Therapeutics; 5. Partnered with Biotheus; 6. Partnered with Regeneron; 7. Partnered with Genentech, member of Roche Group; 8. Partnered with Genmab; 9: BNT323/DB-1303 phase 3 has been initiated with First-Patient-Dosed expected soon; Ab: antibody; ADC: antibody drug conjugate; biAbs: bispecific antibodies; CLDN6 = claudin 6. New oncology clinical trials started year-to-date1: Plans to start additional late-stage trials for select assets in the next 18 months Phase 3 clinical trials initiated in 20239Currently in phase 2 clinical trials BNT326/YL2024 HER3 ADC BNT3162 anti-CTLA-4 Ab BNT3118 PD-L1x4-1BB biAbs BNT1166 FixVac BNT3128 CD40x4-1BB biAbs BNT3233 HER2 ADC BNT211 CLDN6 CAR-T PM80025 PD-L1 x VEGF biAbs BNT1227 iNest Pipeline progress achieved in 2023 Aim to deliver multiple oncology product approvals from 2026 onwards 4 Phase 2 2 Phase 3 5 Phase 1/2

Register to attend online using the QR code Tomorrow, November 7, 2023 9:00 a.m. – 1:00 p.m. ET 3:00 p.m. – 7:00 p.m. CET Innovation Series

Thank you