
4th Quarter 2023 Financial Results & Corporate Update March 20, 2024 Exhibit 99.1
This Slide Presentation Includes Forward-Looking Statements 2 This presentation contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995, as amended, including, but not limited to, statements concerning: BioNTech's expected revenues and net profit related to sales of BioNTech's COVID-19 vaccine, referred to as COMIRNATY® where approved for use under full or conditional marketing authorization, in territories controlled by BioNTech's collaboration partners, particularly for those figures that are derived from preliminary estimates provided by BioNTech's partners; the rate and degree of market acceptance of BioNTech's COVID-19 vaccine and, if approved, BioNTech's investigational medicines; expectations regarding anticipated changes in COVID-19 vaccine demand, including changes to the ordering environment, seasonality and expected regulatory recommendations to adapt vaccines to address new variants or sublineages; the initiation, timing, progress, results, and cost of BioNTech's research and development programs, including those relating to additional formulations of BioNTech's COVID-19 vaccine, and BioNTech's current and future preclinical studies and clinical trials, including statements regarding the timing of initiation and completion of studies or trials and related preparatory work and the availability of results; our expectations with respect to our intellectual property; the impact of the Company’s collaboration and licensing agreements; the development of sustainable vaccine production and supply solutions and the nature and feasibility of these solutions; and BioNTech's estimates of commercial and other revenues, cost of sales, research and development expenses, sales and marketing expenses, general and administrative expenses, capital expenditures, income taxes, net profit, cash, cash equivalents and security investments, shares outstanding and cash outflows and share consideration. In some cases, forward-looking statements can be identified by terminology such as “will,” “may,” “should,” “expects,” “intends,” “plans,” “aims,” “anticipates,” “believes,” “estimates,” “predicts,” “potential,” “continue,” or the negative of these terms or other comparable terminology, although not all forward-looking statements contain these words. The forward-looking statements in this presentation are neither promises nor guarantees, and you should not place undue reliance on these forward-looking statements because they involve known and unknown risks, uncertainties, and other factors, many of which are beyond BioNTech’s control, and which could cause actual results to differ materially from those expressed or implied by these forward-looking statements. These risks and uncertainties include, but are not limited to: BioNTech's pricing and coverage negotiations with governmental authorities, private health insurers and other third-party payors after BioNTech's initial sales to national governments; the future commercial demand and medical need for initial or booster doses of a COVID-19 vaccine; competition from other COVID-19 vaccines or related to BioNTech's other product candidates, including those with different mechanisms of action and different manufacturing and distribution constraints, on the basis of, among other things, efficacy, cost, convenience of storage and distribution, breadth of approved use, side-effect profile and durability of immune response; the timing of and BioNTech's ability to obtain and maintain regulatory approval for BioNTech's product candidates; the ability of BioNTech’s COVID-19 vaccines to prevent COVID-19 caused by emerging virus variants; BioNTech's and its counterparties’ ability to manage and source necessary energy resources; BioNTech's ability to identify research opportunities and discover and develop investigational medicines; the ability and willingness of BioNTech's third-party collaborators to continue research and development activities relating to BioNTech's development candidates and investigational medicines; the impact of the COVID-19 pandemic on BioNTech's development programs, supply chain, collaborators and financial performance; unforeseen safety issues and claims for potential personal injury or death arising from the use of BioNTech's COVID-19 vaccine and other products and product candidates developed or manufactured by BioNTech; BioNTech's and its collaborators’ ability to commercialize and market BioNTech's COVID-19 vaccine and, if approved, its product candidates; BioNTech's ability to manage its development and expansion; regulatory developments in the United States and other countries; BioNTech's ability to effectively scale BioNTech's production capabilities and manufacture BioNTech's products, including BioNTech's target COVID-19 vaccine production levels, and BioNTech's product candidates; risks relating to the global financial system and markets; and other factors not known to BioNTech at this time. You should review the risks and uncertainties described under the heading “Risk Factors” in BioNTech’s Report on Form 20-F for the period ended December 31, 2023 and in subsequent filings made by BioNTech with the SEC, which are available on the SEC’s website at https://www.sec.gov/. Except as required by law, BioNTech disclaims any intention or responsibility for updating or revising any forward-looking statements contained in this presentation in the event of new information, future developments or otherwise. These forward-looking statements are based on BioNTech’s current expectations and speak only as of the date hereof.

Financial Results Jens Holstein, Chief Financial Officer3 Strategic Outlook Ryan Richardson, Chief Strategy Officer4 Pipeline Update Özlem Türeci, Co-founder & Chief Medical Officer2 4th Quarter and FY 2023 Highlights Ugur Sahin, Co-founder & Chief Executive Officer1

1 4th Quarter and FY 2023 Highlights Ugur Sahin, Founder & Chief Executive Officer

Our Vision: Harnessing the Power of the Immune System to Fight Human Disease 5 AI = artificial intelligence. Innovative precision medicine pipeline targeting multiple product approvals in oncology in the coming years Multi-product immunotherapy pioneer addressing medical need worldwide Sustainable respiratory vaccine business Powered by breakthrough science, disruptive technologies & AI BioNTech’s key objectives for the next phase Elevating success beyond our historical achievement

Developing an Innovative Pipeline Focused on Oncology and Infectious Disease 6 1. Partnered with DualityBio; 2. Partnered with OncoC4; 3. Partnered with Regeneron; 4. Partnered with Genentech, member of Roche Group; 5. Partnered with Genmab; 6. Partnered with MediLink Therapeutics; 7. Partnered with Biotheus; 8. Partnered with Pfizer; 9. In collaboration with Bill & Melinda Gates Foundation; 10. Partnered with the Coalition for Epidemic Preparedness Innovations (CEPI). NSCLC = non-small cell lung cancer; PDAC = pancreatic ductal adenocarcinoma; CRC = colorectal cancer; HPV = human papilloma virus; HNSCC = head and neck squamous cell carcinoma; Mpox = monkey pox. Oncology Infectious Disease 7 clinical trials started across platforms 3 first-in-human trials started: Tuberculosis9 Shingles8 Mpox10 22 clinical stage programs 7 clinical stage programs Rigorous pipeline prioritization guided by clinical data and unmet medical need Ph3: BNT323/DB-13031 l BNT316/ONC-3922 Ph2: BNT1163 l autogene cevumeran/BNT1224 Ph1/2: BNT324/DB-13111 l BNT314/GEN10595 Ongoing mid- to late- stage trials: NSCLC BNT316/ONC-3922 l BNT311/GEN10465 l BNT1163 Endometrial cancer BNT323/DB-13031 Breast cancer BNT323/DB-13031 PDAC autogene cevumeran/BNT1224 CRC autogene cevumeran/BNT1224 HPV+ HNSCC BNT113 Melanoma autogene cevumeran/BNT1224 l BNT111 6 clinical assets in-licensed Antibody-drug conjugates: BNT323/DB-13031 BNT324/DB-13111 BNT325/DB-13051 BNT326/YL2026 Antibodies: BNT316/ ONC-3922 PM80027 Clinical and scientific execution in 2023Our pipeline
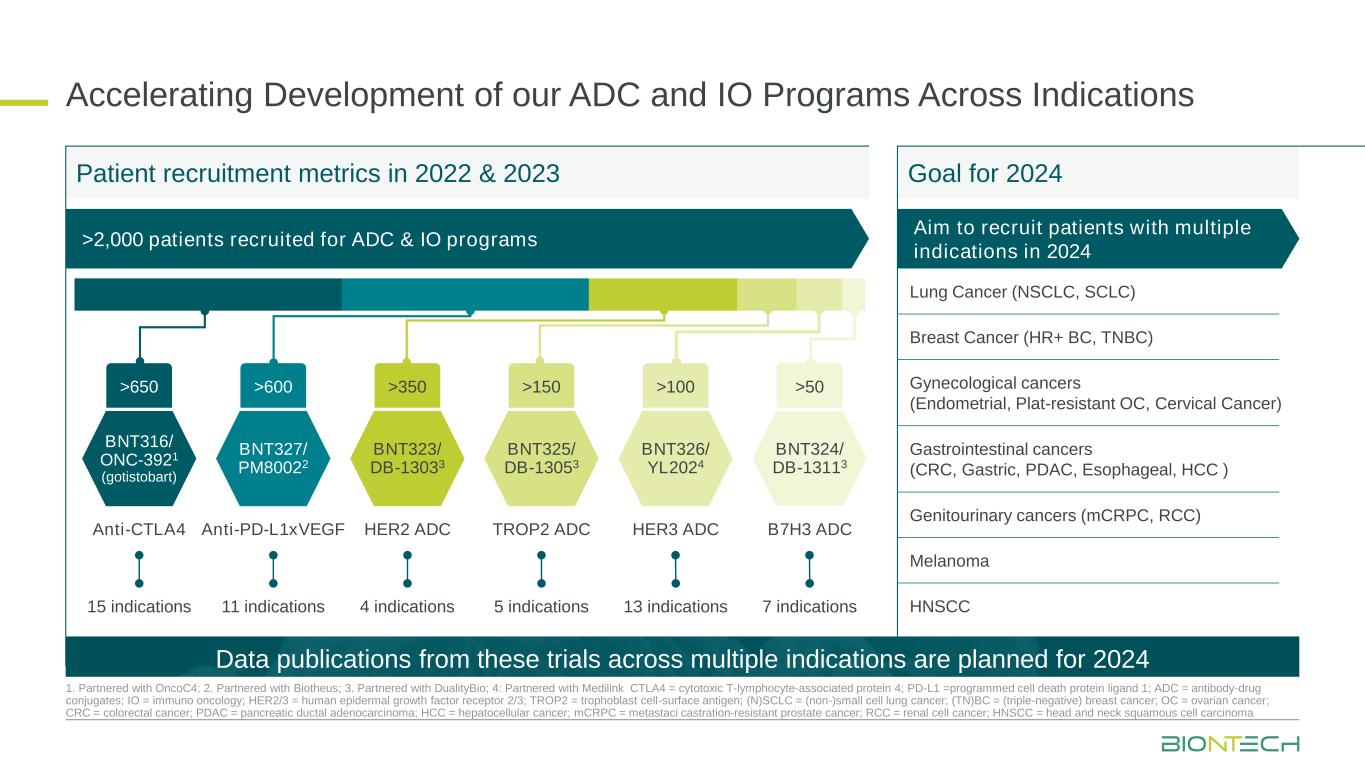
Aim to recruit patients with multiple indications in 2024 >2,000 patients recruited for ADC & IO programs Accelerating Development of our ADC and IO Programs Across Indications 1. Partnered with OncoC4; 2. Partnered with Biotheus; 3. Partnered with DualityBio; 4: Partnered with Medilink CTLA4 = cytotoxic T-lymphocyte-associated protein 4; PD-L1 =programmed cell death protein ligand 1; ADC = antibody-drug conjugates; IO = immuno oncology; HER2/3 = human epidermal growth factor receptor 2/3; TROP2 = trophoblast cell-surface antigen; (N)SCLC = (non-)small cell lung cancer; (TN)BC = (triple-negative) breast cancer; OC = ovarian cancer; CRC = colorectal cancer; PDAC = pancreatic ductal adenocarcinoma; HCC = hepatocellular cancer; mCRPC = metastaci castration-resistant prostate cancer; RCC = renal cell cancer; HNSCC = head and neck squamous cell carcinoma Patient recruitment metrics in 2022 & 2023 Goal for 2024 Lung Cancer (NSCLC, SCLC) Breast Cancer (HR+ BC, TNBC) Gynecological cancers (Endometrial, Plat-resistant OC, Cervical Cancer) Gastrointestinal cancers (CRC, Gastric, PDAC, Esophageal, HCC ) Genitourinary cancers (mCRPC, RCC) Melanoma HNSCC BNT316/ ONC-3921 (gotistobart) Anti-CTLA4 >650 15 indications BNT327/ PM80022 Anti-PD-L1xVEGF >600 11 indications BNT323/ DB-13033 HER2 ADC >350 4 indications BNT325/ DB-13053 TROP2 ADC >150 5 indications BNT326/ YL2024 HER3 ADC >100 13 indications BNT324/ DB-13113 B7H3 ADC >50 7 indications Data publications from these trials across multiple indications are planned for 2024

Corporate Execution in 2023 and Post-Period 1. Consists of €11,663.7 million cash and cash equivalents and €5,989.7 million security investments as of December 31, 2023 AI = artificial intelligence; ADC = antibody drug conjugate; IO = immune oncology; CAR = chimeric antigen receptor 8 Strong cash position ~€ 17.7 bn total cash plus security investments1 Continued progress towards building a multi-product, AI-powered, patient-centric company embedded in the biotech ecosystem Acquired InstaDeep Strategic alliance with AutolusIn-licensed 6 new clinical stage candidates Integrating capabilities in super- computing, AI research and generative AI into various processes Adding new ADCs and next-generation IO antibodies Advancing CAR-T programs towards potential commercialization
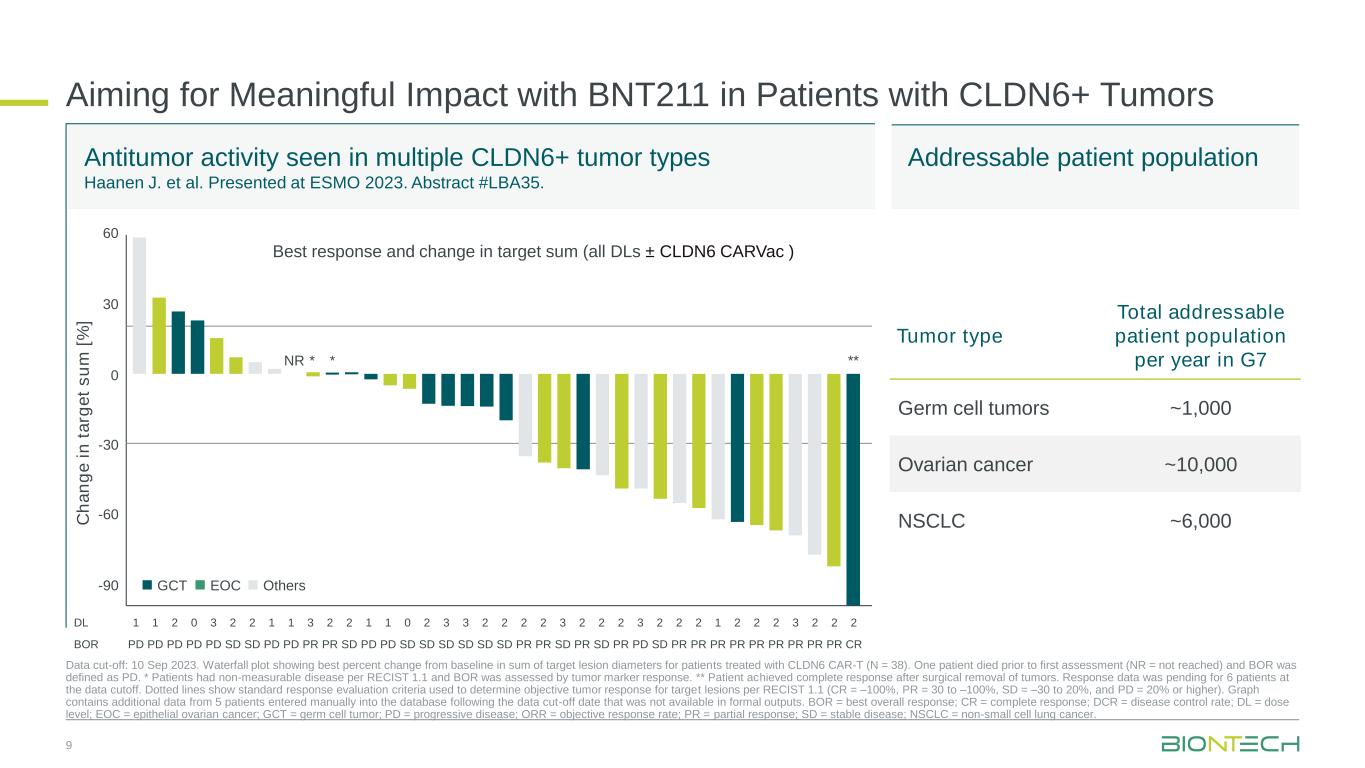
Aiming for Meaningful Impact with BNT211 in Patients with CLDN6+ Tumors Data cut-off: 10 Sep 2023. Waterfall plot showing best percent change from baseline in sum of target lesion diameters for patients treated with CLDN6 CAR-T (N = 38). One patient died prior to first assessment (NR = not reached) and BOR was defined as PD. * Patients had non-measurable disease per RECIST 1.1 and BOR was assessed by tumor marker response. ** Patient achieved complete response after surgical removal of tumors. Response data was pending for 6 patients at the data cutoff. Dotted lines show standard response evaluation criteria used to determine objective tumor response for target lesions per RECIST 1.1 (CR = –100%, PR = 30 to –100%, SD = –30 to 20%, and PD = 20% or higher). Graph contains additional data from 5 patients entered manually into the database following the data cut-off date that was not available in formal outputs. BOR = best overall response; CR = complete response; DCR = disease control rate; DL = dose level; EOC = epithelial ovarian cancer; GCT = germ cell tumor; PD = progressive disease; ORR = objective response rate; PR = partial response; SD = stable disease; NSCLC = non-small cell lung cancer. C h a n g e i n t a rg e t s u m [ % ] 60 30 0 -30 -60 -90 GCT EOC Others NR * * DL 1 1 2 0 3 2 2 1 1 3 2 2 1 1 0 2 3 3 2 2 2 2 3 2 2 2 3 2 2 2 1 2 2 2 3 2 2 2 BOR PD PD PD PD PD SD SD PD PD PR PR SD PD PD SD SD SD SD SD SD PR PR SD PR SD PR PD SD PR PR PR PR PR PR PR PR PR CR ** Best response and change in target sum (all DLs ± CLDN6 CARVac ) Antitumor activity seen in multiple CLDN6+ tumor types Haanen J. et al. Presented at ESMO 2023. Abstract #LBA35. 9 Addressable patient population Tumor type Total addressable patient population per year in G7 Germ cell tumors ~1,000 Ovarian cancer ~10,000 NSCLC ~6,000
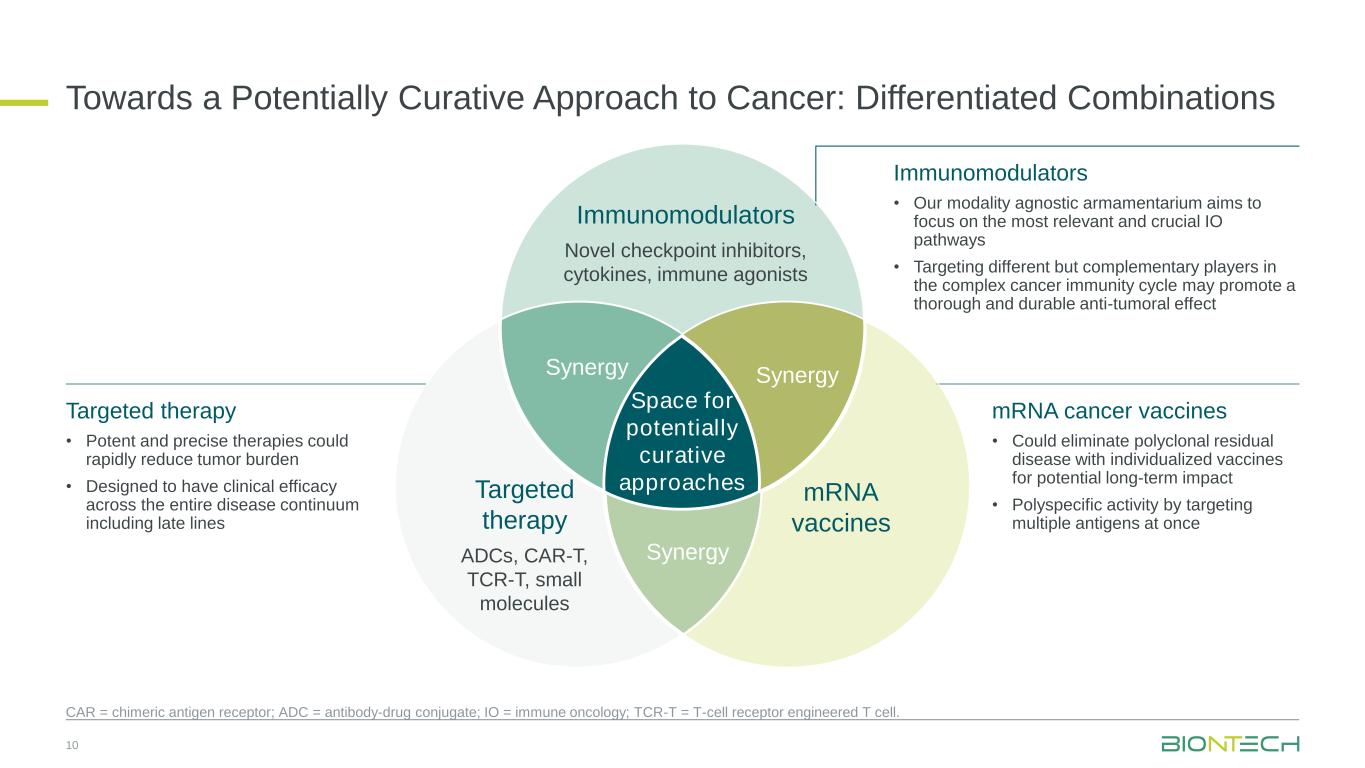
Towards a Potentially Curative Approach to Cancer: Differentiated Combinations Space for potentially curative approaches Immunomodulators Novel checkpoint inhibitors, cytokines, immune agonists mRNA vaccines Targeted therapy ADCs, CAR-T, TCR-T, small molecules SynergySynergy Synergy 10 CAR = chimeric antigen receptor; ADC = antibody-drug conjugate; IO = immune oncology; TCR-T = T-cell receptor engineered T cell. Immunomodulators • Our modality agnostic armamentarium aims to focus on the most relevant and crucial IO pathways • Targeting different but complementary players in the complex cancer immunity cycle may promote a thorough and durable anti-tumoral effect mRNA cancer vaccines • Could eliminate polyclonal residual disease with individualized vaccines for potential long-term impact • Polyspecific activity by targeting multiple antigens at once Targeted therapy • Potent and precise therapies could rapidly reduce tumor burden • Designed to have clinical efficacy across the entire disease continuum including late lines

Pipeline Update Özlem Türeci, Co-Founder & Chief Medical Officer
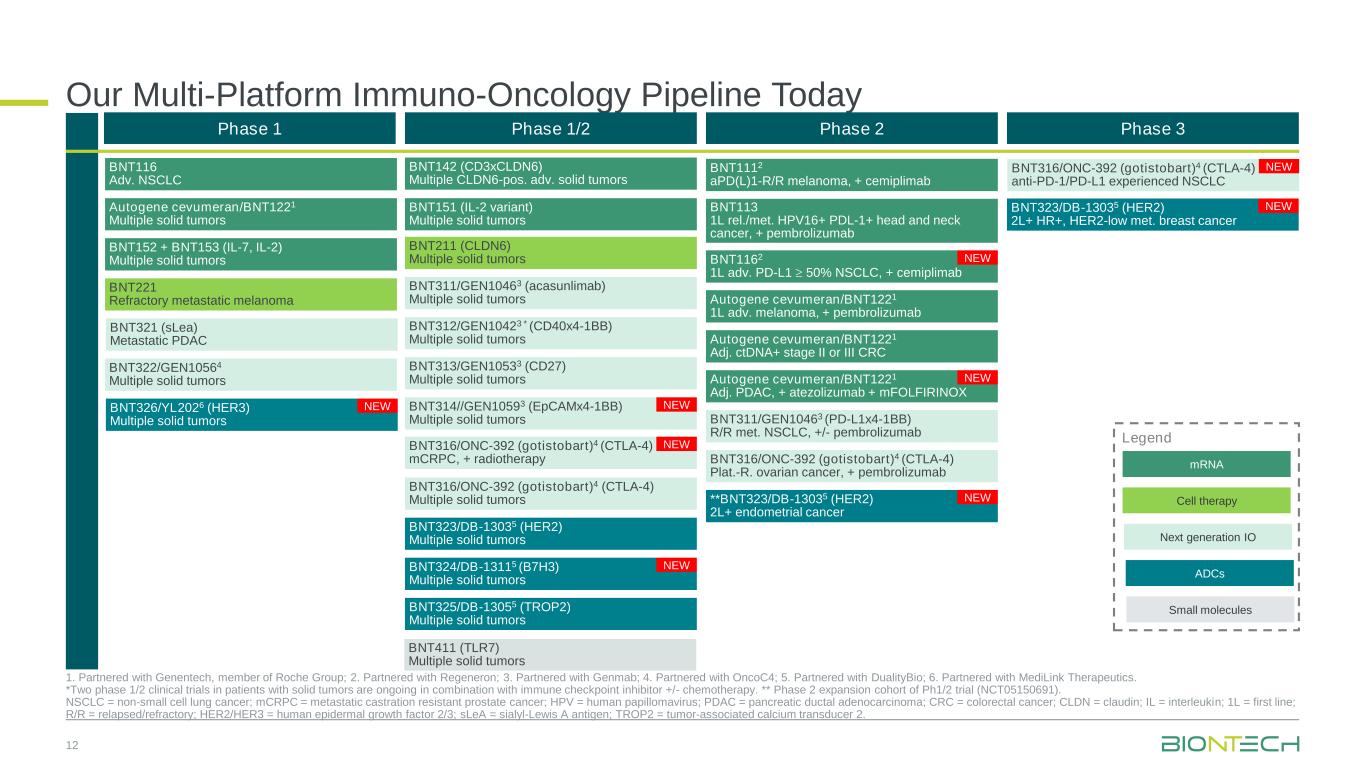
Our Multi-Platform Immuno-Oncology Pipeline Today 12 1. Partnered with Genentech, member of Roche Group; 2. Partnered with Regeneron; 3. Partnered with Genmab; 4. Partnered with OncoC4; 5. Partnered with DualityBio; 6. Partnered with MediLink Therapeutics. *Two phase 1/2 clinical trials in patients with solid tumors are ongoing in combination with immune checkpoint inhibitor +/- chemotherapy. ** Phase 2 expansion cohort of Ph1/2 trial (NCT05150691). NSCLC = non-small cell lung cancer; mCRPC = metastatic castration resistant prostate cancer; HPV = human papillomavirus; PDAC = pancreatic ductal adenocarcinoma; CRC = colorectal cancer; CLDN = claudin; IL = interleukin; 1L = first line; R/R = relapsed/refractory; HER2/HER3 = human epidermal growth factor 2/3; sLeA = sialyl-Lewis A antigen; TROP2 = tumor-associated calcium transducer 2. Phase 1 Phase 1/2 Phase 2 Phase 3 BNT211 (CLDN6) Multiple solid tumors BNT311/GEN10463 (acasunlimab) Multiple solid tumors BNT312/GEN10423 * (CD40x4-1BB) Multiple solid tumors BNT313/GEN10533 (CD27) Multiple solid tumors BNT316/ONC-392 (gotistobart)4 (CTLA-4) Multiple solid tumors BNT151 (IL-2 variant) Multiple solid tumors BNT142 (CD3xCLDN6) Multiple CLDN6-pos. adv. solid tumors BNT325/DB-13055 (TROP2) Multiple solid tumors BNT316/ONC-392 (gotistobart)4 (CTLA-4) anti-PD-1/PD-L1 experienced NSCLC BNT323/DB-13035 (HER2) Multiple solid tumors BNT324/DB-13115 (B7H3) Multiple solid tumors BNT323/DB-13035 (HER2) 2L+ HR+, HER2-low met. breast cancer BNT116 Adv. NSCLC BNT152 + BNT153 (IL-7, IL-2) Multiple solid tumors BNT221 Refractory metastatic melanoma BNT321 (sLea) Metastatic PDAC BNT322/GEN10564 Multiple solid tumors Autogene cevumeran/BNT1221 Multiple solid tumors BNT314//GEN10593 (EpCAMx4-1BB) Multiple solid tumors mRNA Next generation IO Cell therapy Legend Small molecules ADCs BNT326/YL2026 (HER3) Multiple solid tumors BNT311/GEN10463 (PD-L1x4-1BB) R/R met. NSCLC, +/- pembrolizumab BNT1112 aPD(L)1-R/R melanoma, + cemiplimab BNT113 1L rel./met. HPV16+ PDL-1+ head and neck cancer, + pembrolizumab Autogene cevumeran/BNT1221 1L adv. melanoma, + pembrolizumab Autogene cevumeran/BNT1221 Adj. ctDNA+ stage II or III CRC BNT1162 1L adv. PD-L1 50% NSCLC, + cemiplimab Autogene cevumeran/BNT1221 Adj. PDAC, + atezolizumab + mFOLFIRINOX BNT316/ONC-392 (gotistobart)4 (CTLA-4) Plat.-R. ovarian cancer, + pembrolizumab **BNT323/DB-13035 (HER2) 2L+ endometrial cancer BNT316/ONC-392 (gotistobart)4 (CTLA-4) mCRPC, + radiotherapy NEW NEW NEW NEW NEW NEW NEW NEW NEW BNT411 (TLR7) Multiple solid tumors

Making Progress Towards Submissions for Regulatory Approvals in Oncology 13 1. Partnered with OncoC4; 2. Partnered with DualityBio; 3. Partnered with Genentech, member of the Roche group; 4. Partnered with Genmab; 5. Partnered with Biotheus. PD-1 =programmed cell death protein 1; HER2 = human epidermal growth factor receptor 2; HR = hormone receptor; NSCLC = non-small cell lung cancer; PDAC = pancreatic ductal adenocarcinoma; CRC = colorectal cancer, HNSCC = head and neck squamous cell carcinoma; HPV = human papillomaviruses. Plans for at least six programs in 10+ potentially pivotal trials by end of 2024 Select ongoing mid- to late-stage trials Mid- to late-stage trials planned in 2024 & beyond BNT323/DB-13032 BNT211 BNT311/GEN1046 (acasunlimab)4 Autogene cevumeran/BNT1223 BNT327/PM80025 BNT316/ONC-392 (gotistobart)1 anti-PD-1/PD-L1-experienced NSCLC BNT323/DB-13032 HR+, HER2-low met. breast cancer Autogene cevumeran/BNT1223 Adj. PDAC, + atezolizumab + mFOLFIRINOX Autogene cevumeran/BNT1223 Adj. CRC BNT113 PDL-1+, HPV16+ HNSCC, + pembrolizumab Phase 3 Phase 3 Phase 2 Phase 2 Phase 2 Phase 3 Phase 3 Phase 3 Phase 2 Phase 2 Plan to have 10+ potentially registrational trials starting in 2024 and beyond
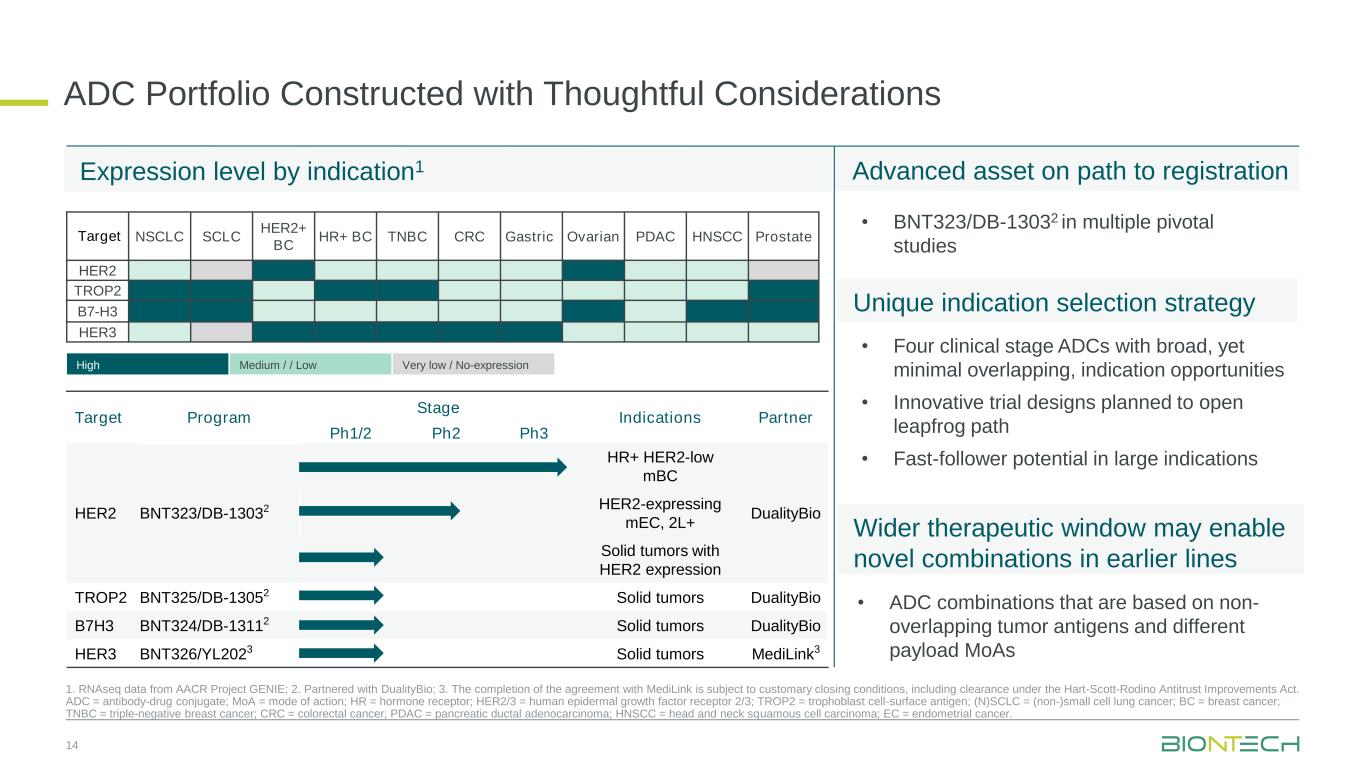
ADC Portfolio Constructed with Thoughtful Considerations 14 1. RNAseq data from AACR Project GENIE; 2. Partnered with DualityBio; 3. The completion of the agreement with MediLink is subject to customary closing conditions, including clearance under the Hart-Scott-Rodino Antitrust Improvements Act. ADC = antibody-drug conjugate; MoA = mode of action; HR = hormone receptor; HER2/3 = human epidermal growth factor receptor 2/3; TROP2 = trophoblast cell-surface antigen; (N)SCLC = (non-)small cell lung cancer; BC = breast cancer; TNBC = triple-negative breast cancer; CRC = colorectal cancer; PDAC = pancreatic ductal adenocarcinoma; HNSCC = head and neck squamous cell carcinoma; EC = endometrial cancer. Target Program Stage Indications Partner Ph1/2 Ph2 Ph3 HER2 BNT323/DB-13032 HR+ HER2-low mBC DualityBio HER2-expressing mEC, 2L+ Solid tumors with HER2 expression TROP2 BNT325/DB-13052 Solid tumors DualityBio B7H3 BNT324/DB-13112 Solid tumors DualityBio HER3 BNT326/YL2023 Solid tumors MediLink3 Target NSCLC SCLC HER2+ BC HR+ BC TNBC CRC Gastric Ovarian PDAC HNSCC Prostate HER2 TROP2 B7-H3 HER3 High Medium / / Low Very low / No-expression • ADC combinations that are based on non- overlapping tumor antigens and different payload MoAs • Four clinical stage ADCs with broad, yet minimal overlapping, indication opportunities • Innovative trial designs planned to open leapfrog path • Fast-follower potential in large indications • BNT323/DB-13032 in multiple pivotal studies Expression level by indication1 Unique indication selection strategy Wider therapeutic window may enable novel combinations in earlier lines Advanced asset on path to registration
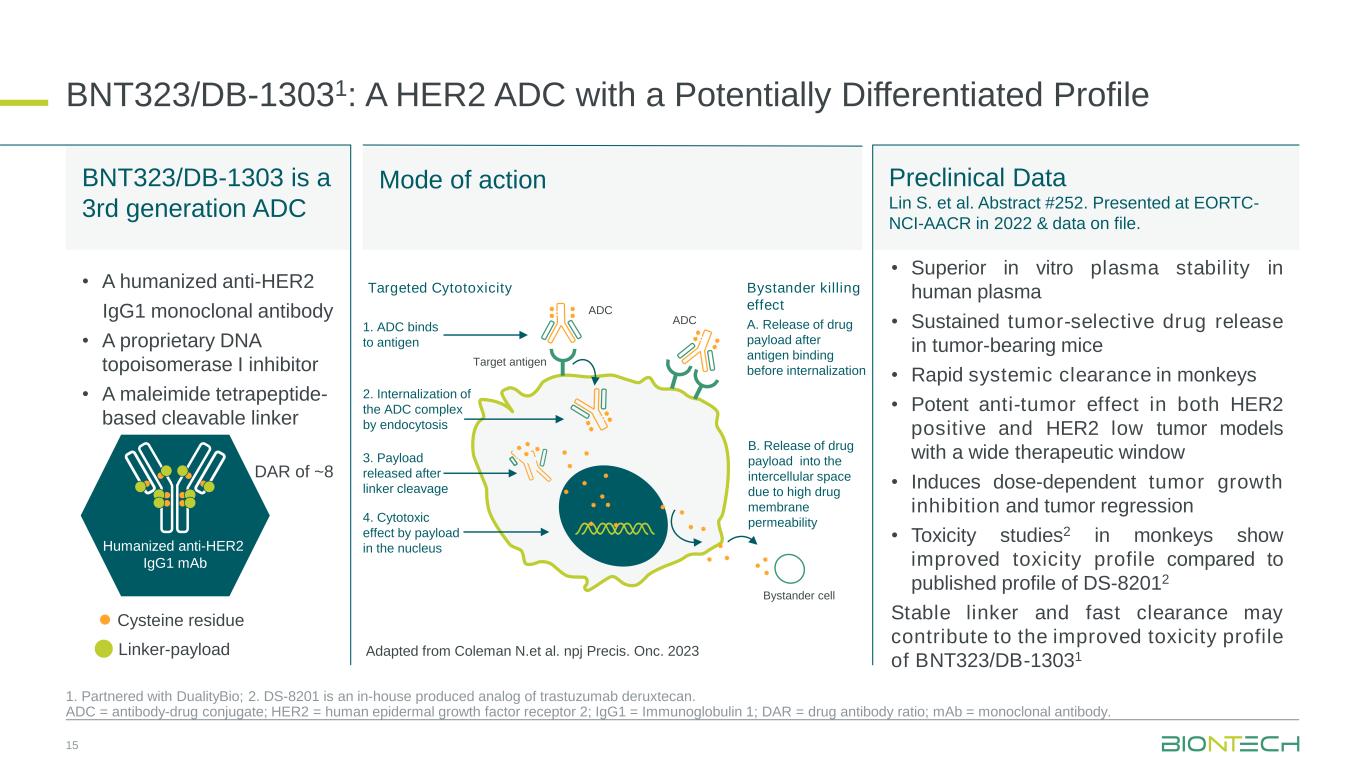
BNT323/DB-13031: A HER2 ADC with a Potentially Differentiated Profile 15 1. Partnered with DualityBio; 2. DS-8201 is an in-house produced analog of trastuzumab deruxtecan. ADC = antibody-drug conjugate; HER2 = human epidermal growth factor receptor 2; IgG1 = Immunoglobulin 1; DAR = drug antibody ratio; mAb = monoclonal antibody. Preclinical Data Lin S. et al. Abstract #252. Presented at EORTC- NCI-AACR in 2022 & data on file. BNT323/DB-1303 is a 3rd generation ADC Mode of action • Superior in vitro plasma stability in human plasma • Sustained tumor-selective drug release in tumor-bearing mice • Rapid systemic clearance in monkeys • Potent anti-tumor effect in both HER2 positive and HER2 low tumor models with a wide therapeutic window • Induces dose-dependent tumor growth inhibition and tumor regression • Toxicity studies2 in monkeys show improved toxicity profile compared to published profile of DS-82012 Stable linker and fast clearance may contribute to the improved toxicity profile of BNT323/DB-13031 Humanized anti-HER2 IgG1 mAb DAR of ~8 Cysteine residue Linker-payload • A humanized anti-HER2 IgG1 monoclonal antibody • A proprietary DNA topoisomerase I inhibitor • A maleimide tetrapeptide- based cleavable linker 1. ADC binds to antigen 2. Internalization of the ADC complex by endocytosis 3. Payload released after linker cleavage Target antigen ADC 4. Cytotoxic effect by payload in the nucleus Bystander cell Targeted Cytotoxicity Bystander killing effect A. Release of drug payload after antigen binding before internalization B. Release of drug payload into the intercellular space due to high drug membrane permeability ADC Adapted from Coleman N.et al. npj Precis. Onc. 2023

First-in-Human Trial with BNT323/DB-13031 in Patients with Advanced HER2-Expressing Solid Tumors 1. Partnered with DualityBio. HER2 = human epidermal growth factor 2; IHC = immunohistochemistry; ISH = in-situ hybridization; NGS = next-generation sequencing; HR = hormone receptor; CRC = colorectal cancer; UC = uterine carcinosarcoma, USC = uterine serous carcinoma NSCLC = non-small cell lung cancer; BC = breast cancer, RP2D = recommended phase 2 dose; ECOG = Eastern Cooperative Oncology Group; MTD = maximum tolerated dose; . 16 Phase 1/2a trial design (NCT05150691), multicenter, non-randomized, open-label Moore K. et al. Presented at ASCO 2023. Abstract #3023. Part 1: Dose escalation (n=88 patients) Part 2a: Dose expansion cohorts (n=165 patients) (HER2 IHC 3+, IHC 2+, IHC 1+ or ISH +, or HER2 amplification and mutation by NGS) Status First patient in: Jan 2022 Trial ongoing Trastuzumab-treated HER2+ (IHC3+, IHC2+/ISH positive) gastric or gastroesophageal junction adenocarcinoma, esophageal carcinoma and CRC 10.0 mg/kg 12.0 mg/kg 8.0 mg/kg 7.0 mg/kg 6.0 mg/kg 4.4 mg/kg 2.2 mg/kg Both HER2 overexpression and HER2 low (IHC3+,2+,1+ or ISH positive) endometrial carcinoma, including UC and USC HR+/HER2 Low (IHC2+ /ISH negative, or IHC1+) breast cancer HER2+ (IHC3+, IHC2+/ISH positive) breast cancer NSCLC with activating HER2 mutation HER2+ or HR+/HER2-low breast cancer with treatment failure of trastuzumab deruxtecan (HER2+ BC; HR+/HER2-low BC) Key endpoints Safety, tolerability, pharmacokinetic, preliminary anti-tumor activity at the selected MTD/RP2D Inclusion criteria Pretreated advanced or metastatic solid tumors Histologically confirmed HER2-positive or HER2- expressing cancers Previous systemic therapies ECOG PS 0-1 Adequate organ function (RP2D=8 mg/kg) RP2D/ MTD

First Clinical Data for BNT323/DB-13031 Demonstrated Antitumor Activity in Heavily Pretreated HER2-Expressing Breast Cancer Patients 17 1. Partnered with DualityBio. HER2 = human epidermal growth factor receptor 2; HR = hormone receptor; ORR = objective response rate; DCR = disease control rate; FIH = first in human; IHC = immunohistochemistry; ISH = in-situ hybridization.. BNT323/DB-13031 was well-tolerated and all adverse events were manageable Results supported the initiation of a pivotal phase 3 study evaluating BNT323/DB-13031 in HR+/HER2 low br ast c ncer ORR, % DCR, % HER2+ breast cancer (n=26) 50 96.2 HER2 low breast cancer (n=13) 38.5 84.6 Anti-tumor activity in heavily pretreated HER2-expressing breast cancer patients Phase 1/2a FIH study (NCT05150691): Clinical activity and safety Adapted from Moore K. et al. Presented at ASCO 2023. Abstract #3023. B e s t c h a n g e f ro m b a s e li n e ( % ) IS H + IS H + IS H + IS H + IS H + IS H + 3+1+ 2+HER2 IHC Status: -100 -50 0 50 100 4.4 mg/kg 7 mg/kg6 mg/kg 8 mg/kg Best response across HER2 expression levels and dose levels
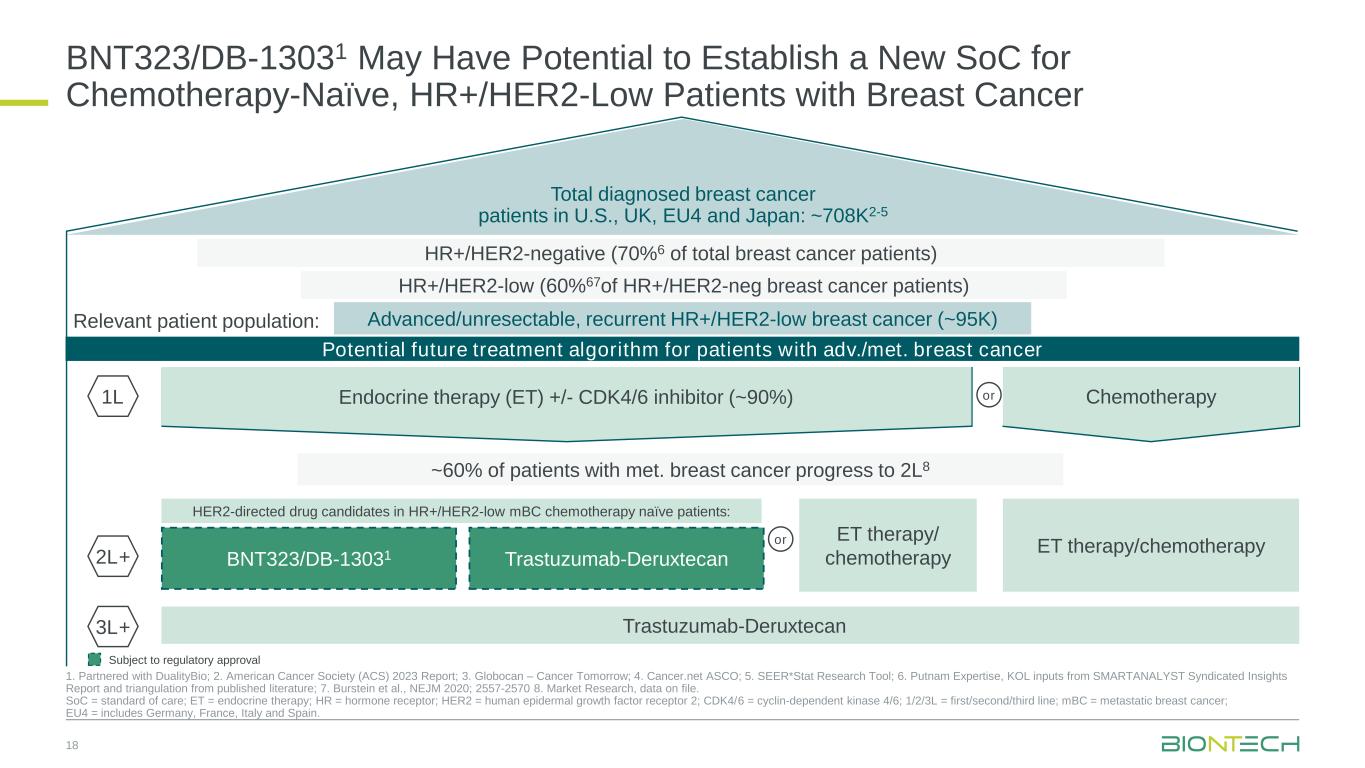
Total diagnosed breast cancer patients in U.S., UK, EU4 and Japan: ~708K2-5 Potential future treatment algorithm for patients with adv./met. breast cancer BNT323/DB-13031 May Have Potential to Establish a New SoC for Chemotherapy-Naïve, HR+/HER2-Low Patients with Breast Cancer 18 1. Partnered with DualityBio; 2. American Cancer Society (ACS) 2023 Report; 3. Globocan – Cancer Tomorrow; 4. Cancer.net ASCO; 5. SEER*Stat Research Tool; 6. Putnam Expertise, KOL inputs from SMARTANALYST Syndicated Insights Report and triangulation from published literature; 7. Burstein et al., NEJM 2020; 2557-2570 8. Market Research, data on file. SoC = standard of care; ET = endocrine therapy; HR = hormone receptor; HER2 = human epidermal growth factor receptor 2; CDK4/6 = cyclin-dependent kinase 4/6; 1/2/3L = first/second/third line; mBC = metastatic breast cancer; EU4 = includes Germany, France, Italy and Spain. ~60% of patients with met. breast cancer progress to 2L8 HER2-directed drug candidates in HR+/HER2-low mBC chemotherapy naïve patients: ET therapy/chemotherapy 1L 2L+ Advanced/unresectable, recurrent HR+/HER2-low breast cancer (~95K) Subject to regulatory approval Relevant patient population: HR+/HER2-negative (70%6 of total breast cancer patients) HR+/HER2-low (60%67of HR+/HER2-neg breast cancer patients) ET therapy/ chemotherapy or Trastuzumab-Deruxtecan3L+ Endocrine therapy (ET) +/- CDK4/6 inhibitor (~90%) Chemotherapyor BNT323/DB-13031 Trastuzumab-Deruxtecan
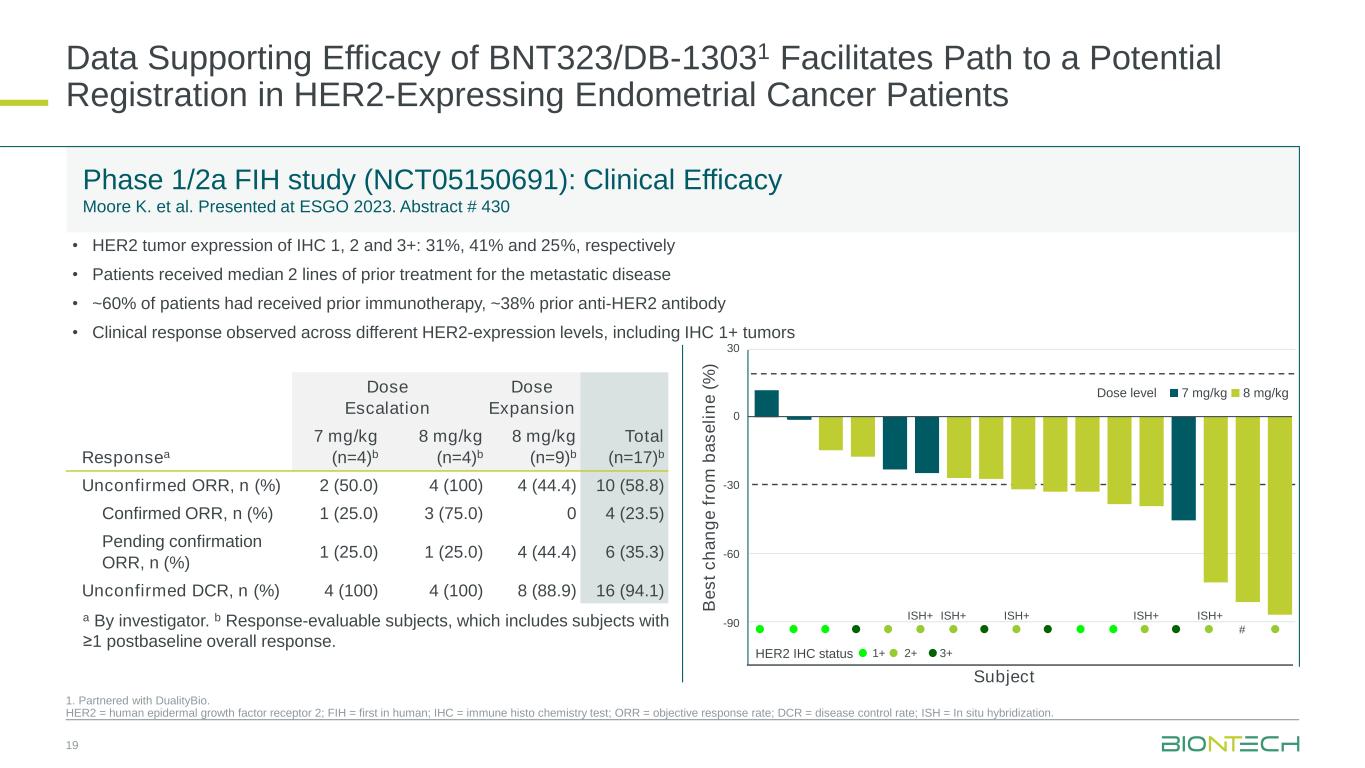
Data Supporting Efficacy of BNT323/DB-13031 Facilitates Path to a Potential Registration in HER2-Expressing Endometrial Cancer Patients 19 1. Partnered with DualityBio. HER2 = human epidermal growth factor receptor 2; FIH = first in human; IHC = immune histo chemistry test; ORR = objective response rate; DCR = disease control rate; ISH = In situ hybridization. Phase 1/2a FIH study (NCT05150691): Clinical Efficacy Moore K. et al. Presented at ESGO 2023. Abstract # 430 Responsea Dose Escalation Dose Expansion Total (n=17)b 7 mg/kg (n=4)b 8 mg/kg (n=4)b 8 mg/kg (n=9)b Unconfirmed ORR, n (%) 2 (50.0) 4 (100) 4 (44.4) 10 (58.8) Confirmed ORR, n (%) 1 (25.0) 3 (75.0) 0 4 (23.5) Pending confirmation ORR, n (%) 1 (25.0) 1 (25.0) 4 (44.4) 6 (35.3) Unconfirmed DCR, n (%) 4 (100) 4 (100) 8 (88.9) 16 (94.1) a By investigator. b Response-evaluable subjects, which includes subjects with ≥1 postbaseline overall response. B e s t c h a n g e f ro m b a s e li n e ( % ) ISH+ ISH+ ISH+ ISH+ ISH+ # Subject 30 0 -30 -60 -90 Dose level 7 mg/kg 8 mg/kg 1+ 2+ 3+HER2 IHC status • HER2 tumor expression of IHC 1, 2 and 3+: 31%, 41% and 25%, respectively • Patients received median 2 lines of prior treatment for the metastatic disease • ~60% of patients had received prior immunotherapy, ~38% prior anti-HER2 antibody • Clinical response observed across different HER2-expression levels, including IHC 1+ tumors

Potential future treatment algorithm for patients with adv./met. endometrial cancer3 BNT323/DB-13031 Offers the Potential to Establish a New SoC for Patients with HER2-Expressing Endometrial Cancer 20 1. Partnered with Duality Bio; 2. CancerMPact® Treatment Architecture Endometrial; U.S. and EU5 v1.1; 3. NCCN guidelines® Version 1.2024; 4. Makker V et al. NEJM. 2022; 5. Keytruda PI: https://www.merck.com/product/usa/pi_circulars/k/keytruda/keytruda_pi.pdf. SoC = standard of care;; HER2 = human epidermal growth factor receptor 2; 1L = first line; 2L+ = second line and beyond; EU4 = includes Germany, France, Italy and Spain; MSS/MSI = microsatellite in/stability; MMR = mismatch repair; PD-1 = programmed cell death protein 1; EC = endometrial cancer; * Dostarlimab approved in patients with MSI-H/dMMR tumors. NCCN guidelines recommend dostarlimab or pembrolizumab + chemotherapy irrespective of MMR status; ** pMMR tumors: pembrolizumab+lenvatinib, MSI-H/dMMR tumors pembroliumab or dostarlimab monotherapy; ***NCCN guidelines recommend Trastuzumab Deruxtecan for HER2-positive tumors (IHC 3+ or 2+). Chemotherapy combo (+/- bevacizumab) ~40 % progress and receive 2L therapy1 ICI + chemotherapy* Trastuzumab + chemotherapy (for HER2+ USC, carcinosarcoma) BNT323/ DB-1303 Anti-PD-1 +/- Lenvatinib** Chemotherapy (+/- bevacizumab) 1L 2L+ Advanced/unresectable, recurrent Stage IV (~50K) Subject to regulatory approval Relevant patient population: or or oror Total diagnosed endometrial cancer patients in US, UK, EU4 and Japan: ~130K2 MSS/MMR proficient MSI-H/MMR deficient HER2-positive ORR: 30% mPFS: 6.6 mo mOS: 17.4 mo4 ORR: 15%; mPFS: 3.8 mo; mOS: 12.0 mo5 Trastuzumab- Deruxtecan*** HER2-directed drug candidates in mEC:

Product candidate BNT323/ DB-13031 BNT316/ ONC-392 (gotistobart)2 BNT327/ PM80023 BNT211 BNT311/ GEN1046 (acasunlimab)4 Target HER2 CTLA-4 VEGF-A x PDL-1 VHH CLDN6 PD-L1 x 4-1BB First Wave of Potential Oncology Launches From 2026 Onwards Could Include: 21 1. Partnered with DualityBio; 2. Partnered with OncoC4; 3. Partnered with Biotheus; 4.. Partnered with Genmab MoA = mode of action; HER2 = human epidermal growth factor 2; CTLA-4 = cytotoxic T-lymphocyte-associated protein 4; PD1 = programmed cell death protein 1; CLDN6 = claudin 6. We believe we have multiple shots on goal, and that our in-licensed assets are starting to contribute to value creation and towards de-risking our pipeline Diverse MoAs Validated & new targets Mix of partnered and proprietary programs

3 Financial Results Jens Holstein, Chief Financial Officer

FY 2023 Key Financial Highlights1 23 1. Financial information is prepared and presented in Euros and numbers are rounded to millions and billions of Euros in accordance with standard commercial practice. 2. Consists of cash and cash equivalents of €11,663.7 million and security investments of €5,989.7 million, as of December 31, 2023. Profit before tax €3.83 €1.2bn €3.8 bn €17.7 bn Total revenues Diluted EPS Total cash plus security investments2

FY 2023 Revenues and Cash plus Security Dynamics 24 13,9 17,72 YE 2022 YE 2023 +27% FY 2023 revenues reduced by significant write-downs Cash plus security investments grew in 20233 4,7 3,8 0,9 FY 2023 revenues without reflection of write-downs4 FY 2023 write-downs FY 2023 revenues (in billions €)1 1. Numbers have been rounded, numbers presented may not add up precisely to the totals and may have been adjusted in the table context. 2. Consists of cash and cash equivalents of €11,663.7 million and security investments of €5,989.7 million, as of December 31, 2023. 3. Contractual settlement of the gross profit share has a temporal offset of more than one calendar quarter and even has an additional time lag between the recognition of revenues and the payment receipt for gross profit of subsidiaries outside the United States. 4. Inventory write-downs and other charges identified on the collaboration partner Pfizer’s side, jointly referred to as write-downs, are reducing Pfizer’s gross profit, hence BioNTech’s revenues. (in billions €)1

Q4 and FY 2023 Financial Results 25 1. Numbers have been rounded, numbers presented may not add up precisely to the totals and may have been adjusted in the table context. Presentation of the consolidated statements of profit or loss has been condensed. 2. BioNTech’s profit share is estimated based on preliminary data shared between Pfizer and BioNTech as further described in the Annual Report on Form 20-F for the year ended December 31, 2023, filed on March 20, 2024. Any changes in the estimated share of the collaboration partner's gross profit will be recognized prospectively. 3. Adjustments to prior-year figures relate to costs for external legal advice in connection with certain legal litigations from general and administrative expenses to other operating expense to reflect changes in the internal reporting also in the external reporting. Three months ended December 31 Years ended December 31 (in millions €, except per share data)1 2023 2022 2023 2022 Commercial revenues2 1,478.9 4,271.3 3,815.5 17,194.6 Research & development revenues 0.1 7.0 3.5 116.0 Total revenues 1,479.0 4,278.3 3,819.0 17,310.6 Cost of sales (179,1) (183.5) (599.8) (2,995.0) Research and development expenses (577.8) (509.8) (1,783.1) (1,537.0) Sales and marketing expenses (18.0) (14.6) (62.7) (59.5) General and administrative expenses (124.3) (119.9) (495.0) (481.7) Other operating income less expenses3 (53.6) (157.6) (188.0) 405.3 Operating income 526.2 3,292.9 690.4 12,642.7 Finance income less expenses 137.0 (120.3) 495.7 311.4 Profit before tax 663.2 3,172.6 1,186.1 12,954.1 Income taxes (205.3) (893.9) (255.8) (3,519.7) Profit for the period 457.9 2,278.7 930.3 9,434.4 Earnings per share Basic profit for the period per share 1.91 9.38 3.87 38.78 Diluted profit for the period per share 1.90 9.26 3.83 37.77
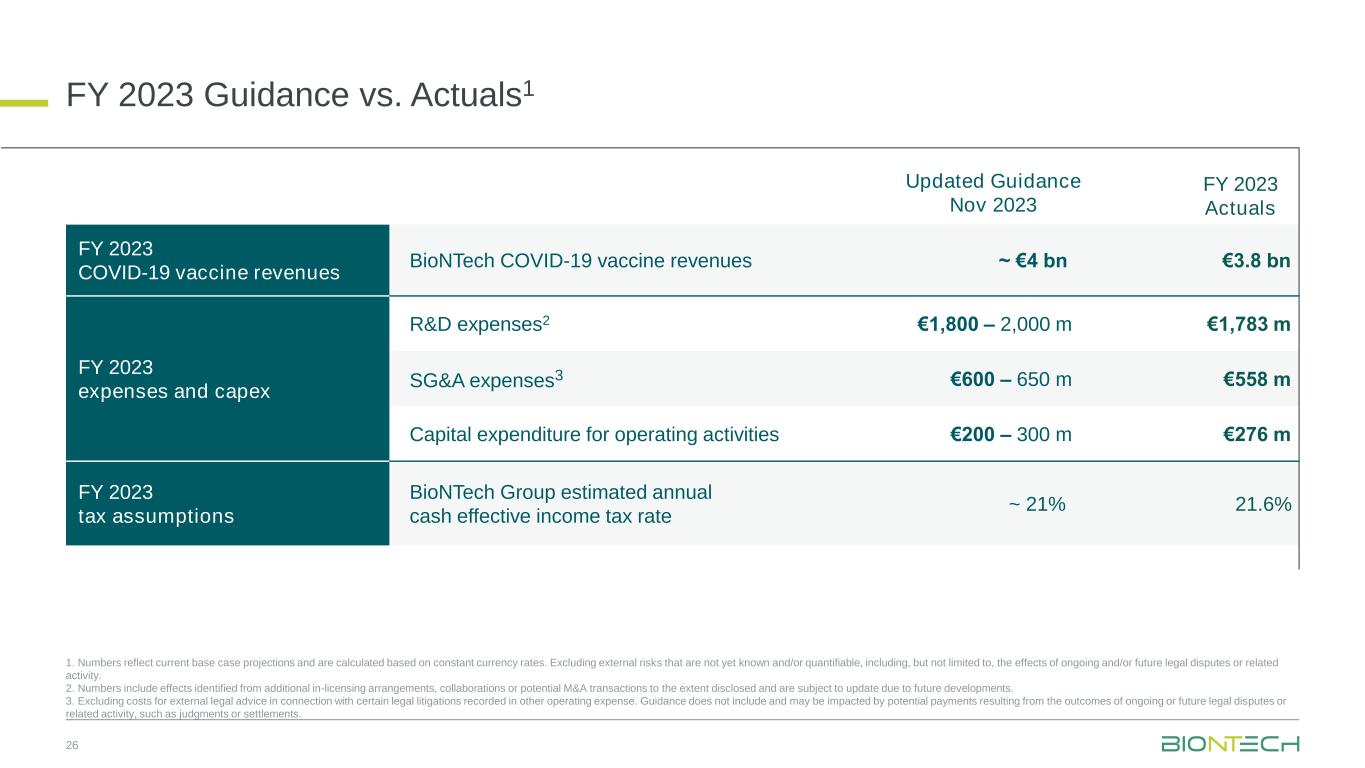
FY 2023 Guidance vs. Actuals1 26 FY 2023 COVID-19 vaccine revenues BioNTech COVID-19 vaccine revenues ~ €4 bn €3.8 bn FY 2023 expenses and capex R&D expenses2 €1,800 – 2,000 m €1,783 m SG&A expenses3 €600 – 650 m €558 m Capital expenditure for operating activities €200 – 300 m €276 m FY 2023 tax assumptions BioNTech Group estimated annual cash effective income tax rate ~ 21% 21.6% Updated Guidance Nov 2023 FY 2023 Actuals 1. Numbers reflect current base case projections and are calculated based on constant currency rates. Excluding external risks that are not yet known and/or quantifiable, including, but not limited to, the effects of ongoing and/or future legal disputes or related activity. 2. Numbers include effects identified from additional in-licensing arrangements, collaborations or potential M&A transactions to the extent disclosed and are subject to update due to future developments. 3. Excluding costs for external legal advice in connection with certain legal litigations recorded in other operating expense. Guidance does not include and may be impacted by potential payments resulting from the outcomes of ongoing or future legal disputes or related activity, such as judgments or settlements.
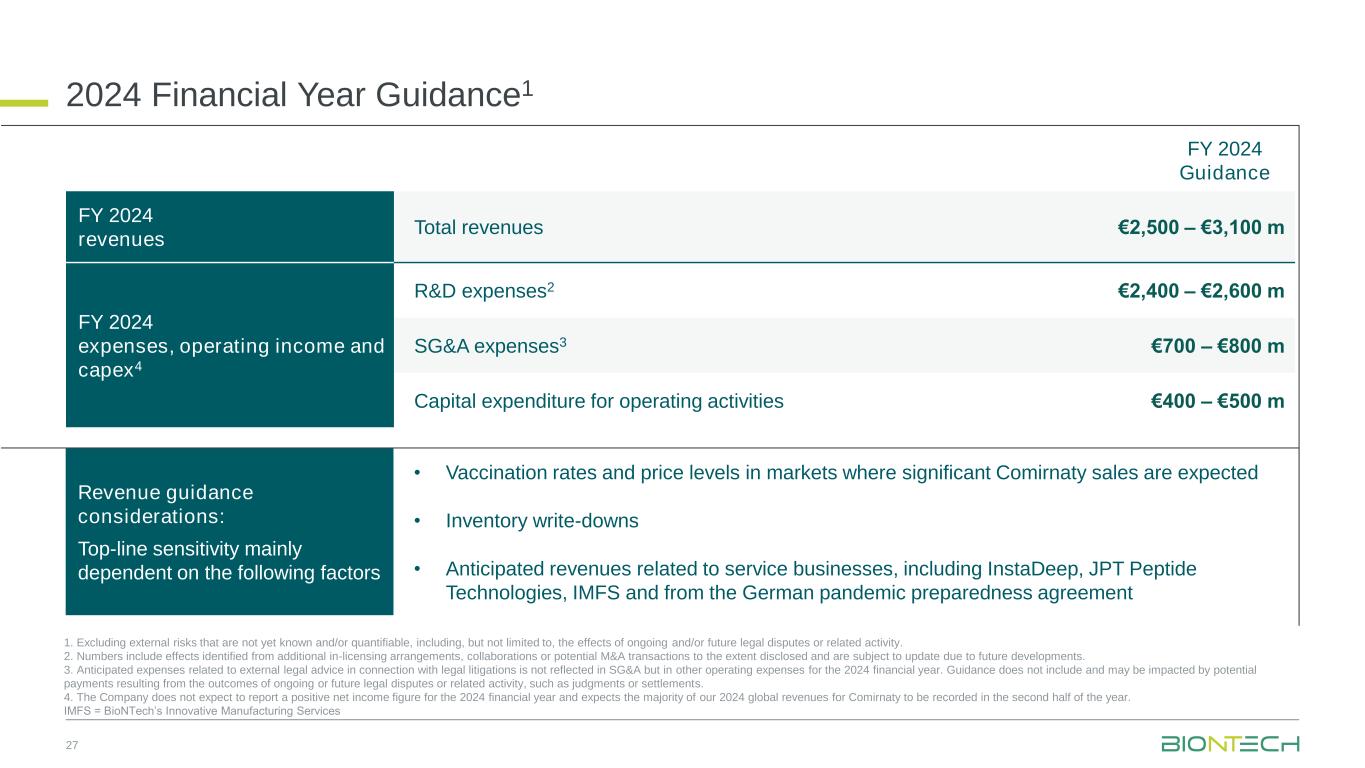
Revenue guidance considerations: Top-line sensitivity mainly dependent on the following factors • Vaccination rates and price levels in markets where significant Comirnaty sales are expected • Inventory write-downs • Anticipated revenues related to service businesses, including InstaDeep, JPT Peptide Technologies, IMFS and from the German pandemic preparedness agreement 2024 Financial Year Guidance1 27 1. Excluding external risks that are not yet known and/or quantifiable, including, but not limited to, the effects of ongoing and/or future legal disputes or related activity. 2. Numbers include effects identified from additional in-licensing arrangements, collaborations or potential M&A transactions to the extent disclosed and are subject to update due to future developments. 3. Anticipated expenses related to external legal advice in connection with legal litigations is not reflected in SG&A but in other operating expenses for the 2024 financial year. Guidance does not include and may be impacted by potential payments resulting from the outcomes of ongoing or future legal disputes or related activity, such as judgments or settlements. 4. The Company does not expect to report a positive net income figure for the 2024 financial year and expects the majority of our 2024 global revenues for Comirnaty to be recorded in the second half of the year. IMFS = BioNTech’s Innovative Manufacturing Services FY 2024 revenues Total revenues €2,500 – €3,100 m FY 2024 expenses, operating income and capex4 R&D expenses2 €2,400 – €2,600 m SG&A expenses3 €700 – €800 m Capital expenditure for operating activities €400 – €500 m FY 2024 Guidance

COVID-19 Vaccine Business – major value contributor Innovative Oncology Pipeline – potential future value driver Profitable COVID-19 Vaccine Business supports Investment in Growth Drivers 28 : FY 2023 • Revenue of €3.8 bn • Gross Profit of €3.2 bn • COVID-19 associated R&D costs ~ €0.3 bn • S&M costs < €0.05 bn • COVID-19 vaccine business with lean cost structure expected to generate positive cash flows going forward • Aiming for 10+ potentially registrational trials ongoing by the end of 2024 • First potential oncology launch estimated for 2026 • Diversified clinical pipeline offers multiple potential growth opportunities for the years to come COVID-19 vaccine franchise and innovative oncology pipeline driving long-term value creation

4 Strategic Outlook Ryan Richardson, Chief Strategy Officer
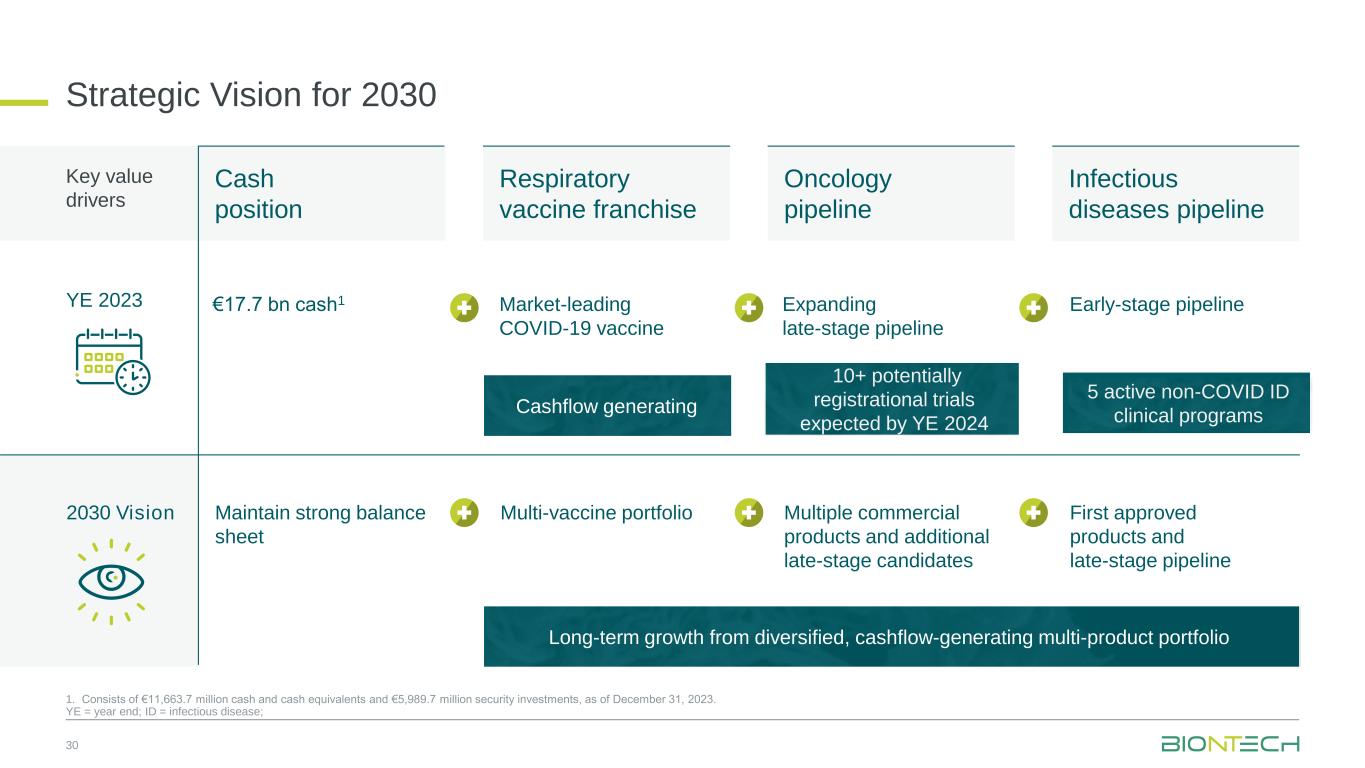
5 active non-COVID ID clinical programs 10+ potentially registrational trials expected by YE 2024 Cash position Respiratory vaccine franchise Oncology pipeline Infectious diseases pipeline Strategic Vision for 2030 30 1. Consists of €11,663.7 million cash and cash equivalents and €5,989.7 million security investments, as of December 31, 2023. YE = year end; ID = infectious disease; €17.7 bn cash1 Market-leading COVID-19 vaccine Expanding late-stage pipeline Early-stage pipeline Maintain strong balance sheet Multi-vaccine portfolio Multiple commercial products and additional late-stage candidates First approved products and late-stage pipeline YE 2023 2030 Vision Key value drivers Long-term growth from diversified, cashflow-generating multi-product portfolio Cashflow generating
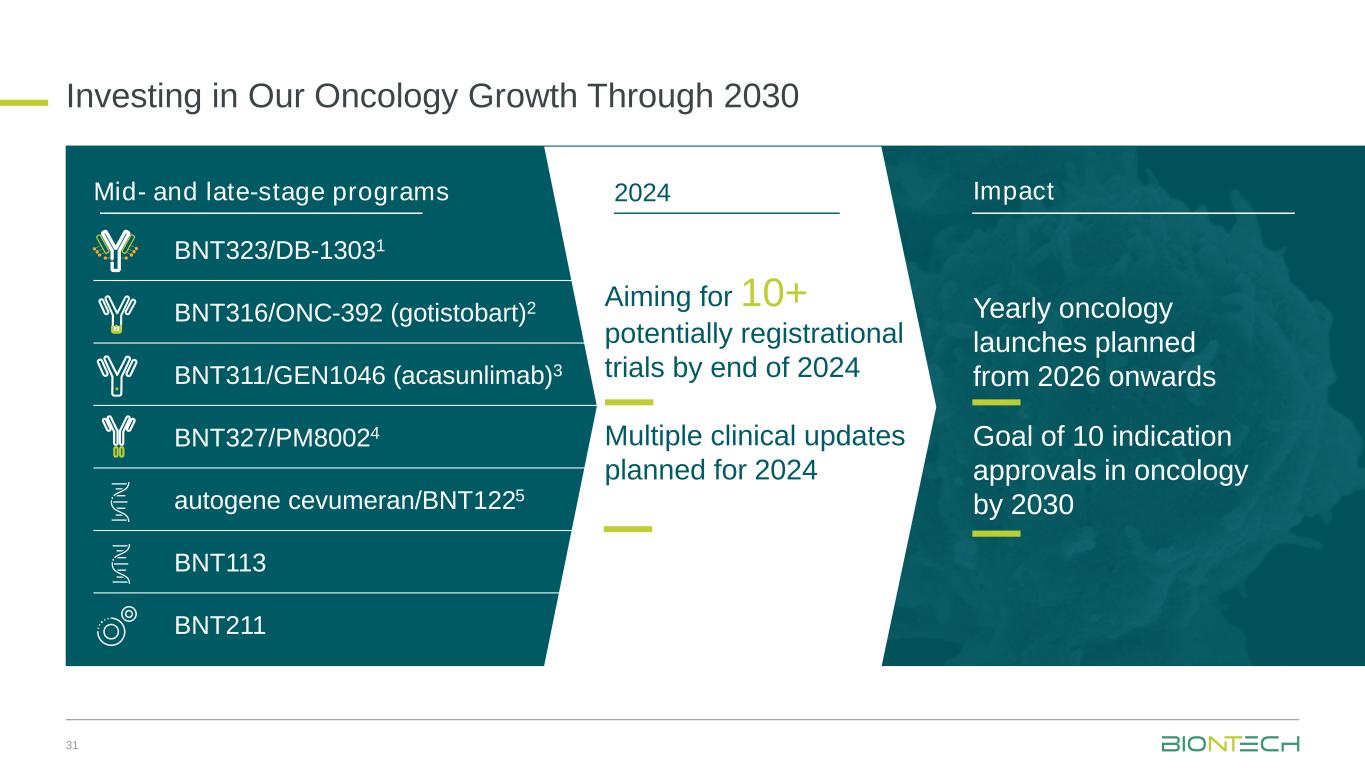
Investing in Our Oncology Growth Through 2030 31 Impact Yearly oncology launches planned from 2026 onwards Goal of 10 indication approvals in oncology by 2030 Aiming for 10+ potentially registrational trials by end of 2024 Multiple clinical updates planned for 2024 2024Mid- and late-stage programs BNT323/DB-13031 BNT316/ONC-392 (gotistobart)2 BNT311/GEN1046 (acasunlimab)3 BNT327/PM80024 autogene cevumeran/BNT1225 BNT113 BNT211

Save the date Annual General Meeting May 17, 2024 Innovation Series: Digital & AI October 1, 2024 Innovation Series November 14, 2024

Thank you

Appendix

Advancing our Pipeline: Select Data Milestones in 2024 35 1. Partnered with Genmab; 2. Partnered with OncoC4; 3. Partnered with DualityBio; 4. Partnered with Biotheus; 5. Partnered with Pfizer. NSCLC = non-small cell lung cancer, R/R = relapsed/refractors. Program Indication Targeted Milestone Oncology BNT311/GEN1046 (acasunlimab)1 R/R met. NSCLC, +/- pembrolizumab Phase 2 data BNT312/GEN10421 Multiple solid tumors Ph1/2 expansion cohort data BNT316/ONC-392 (gotistobart)2 Multiple solid tumors Ph1/2 expansion cohort data BNT323/DB-13033 Multiple solid tumors Ph1/2 expansion cohort data BNT325/DB-13053 Multiple solid tumors Ph1/2 data BNT327/PM80024 Multiple solid tumors Phase 2 data Infectious Disease BNT162b25 COVID-19, Omicron XBB.1.5 monovalent vaccine Phase 2/3 data BNT1675 Shingles Phase 1 trial update