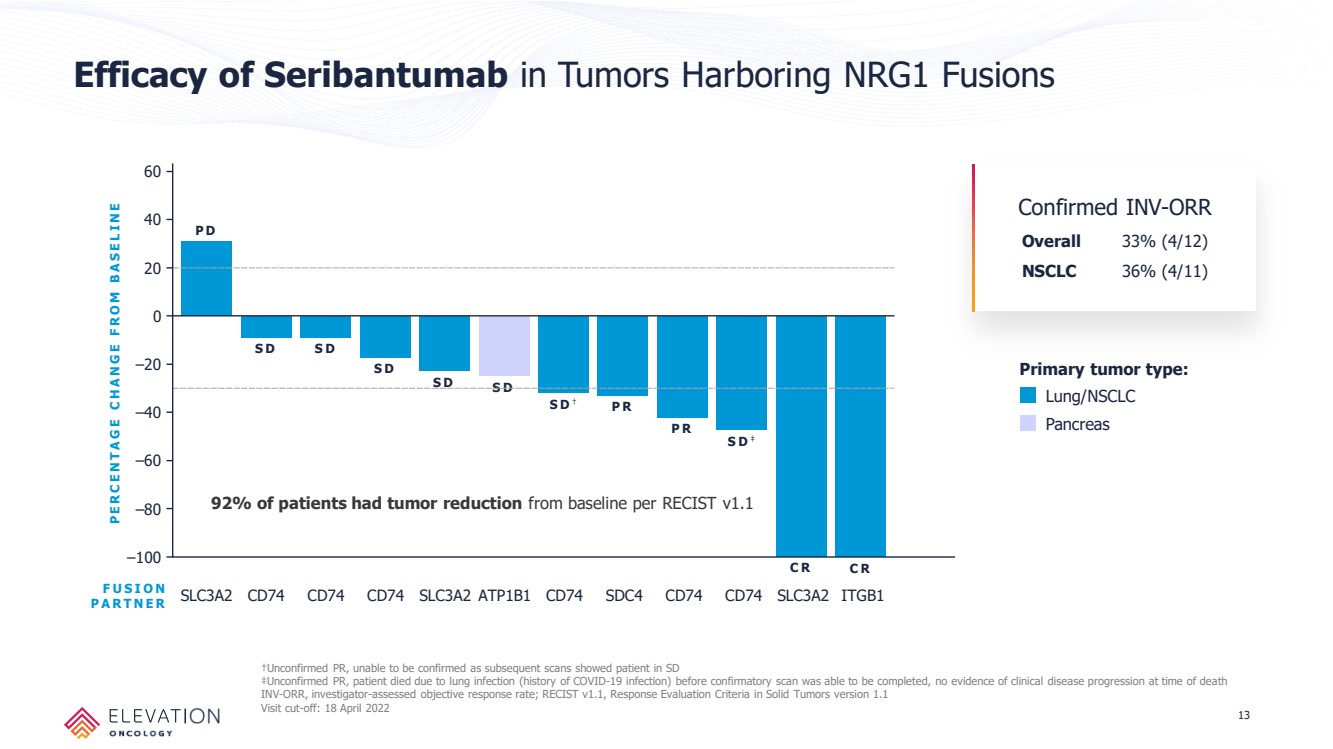
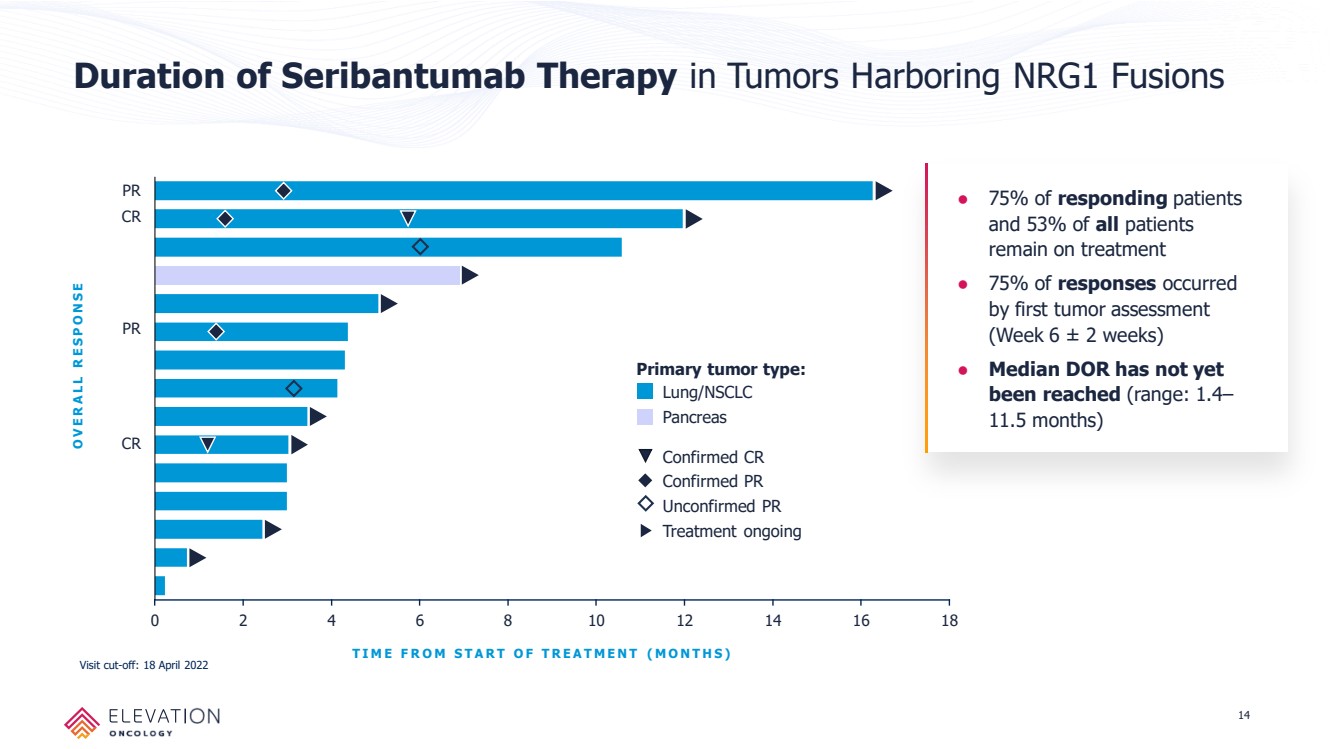
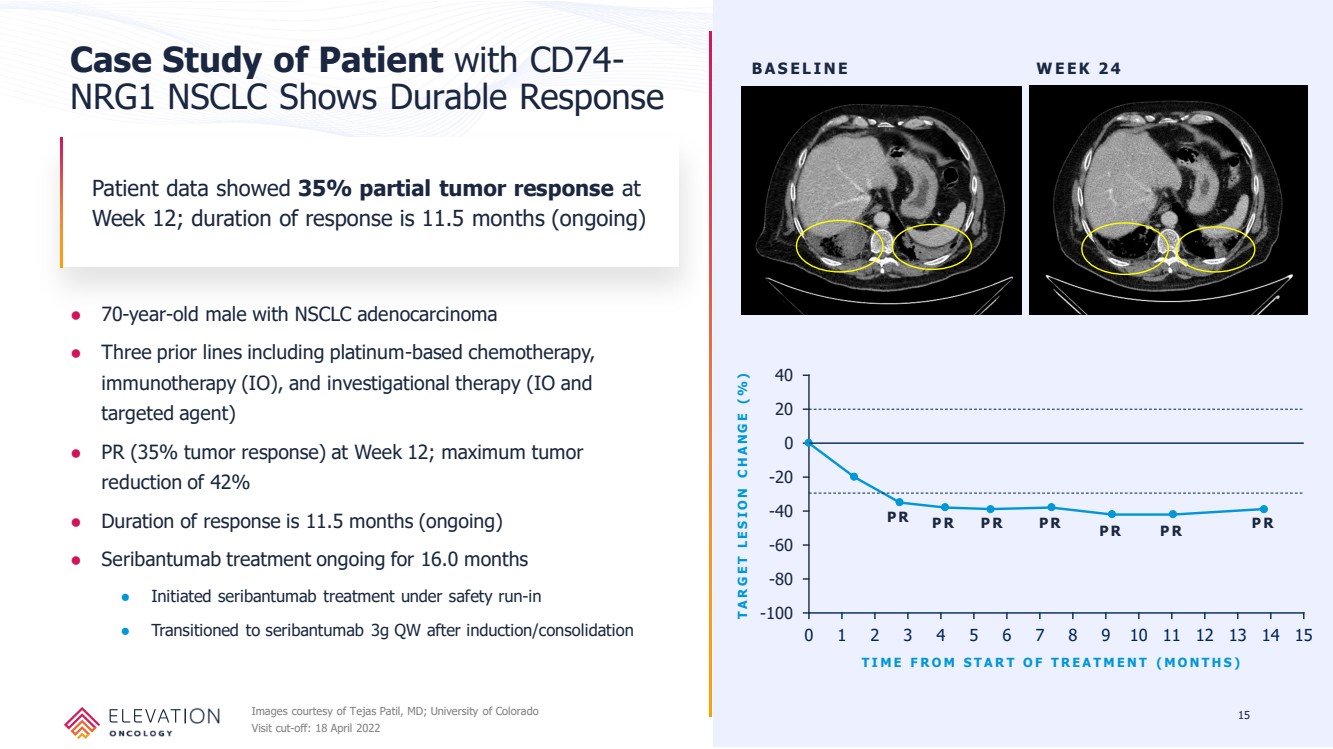
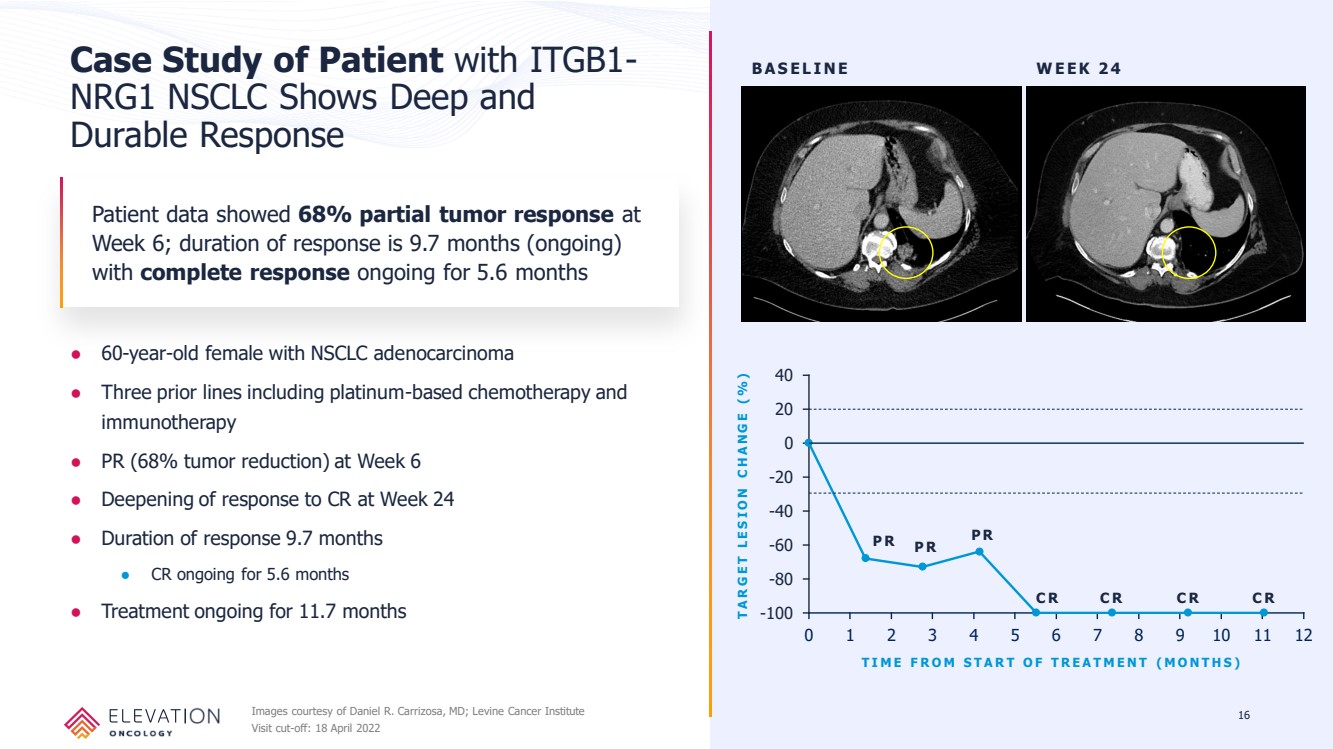
| Forward Looking Statements These slides contain forward - looking statements and information relating to Elevation Oncology, Inc. within the meaning of, and made pursuant to the safe harbor provisions of, the Private Securities Litigation Reform Act of 1995. You should not place undue reliance on forward - looki ng statements, as these statements are based upon our current expectations, forecasts, and assumptions and are subject to significant risks and uncer tai nties. Any statements contained herein or provided orally that are not statements of historical fact may be deemed to be forward - looking statements. I n some cases, you can identify forward - looking statements by terms such as “believe,” “may,” “will,” “potentially,” “estimate,” “continue,” “anticipate,” “inte nd,” “could,” “would,” “project,” “plan,” “expect,” “predict,” “potential” and similar expressions that convey uncertainty of future events or outcomes, although not a ll forward - looking statements contain these words. Forward - looking statements include all statements other than statements of historical fact contained in this presen tation, including information concerning our future financial performance, business plans and objectives, timing and success of our planned development act ivi ties, timing of data of our clinical trials, our ability to obtain regulatory approval, the potential therapeutic benefits and economic value of our prod uct candidates, potential growth opportunities, competitive position, industry environment and potential market opportunities, and the impact of the COVID - 19 pan demic on our business and operations. Forward - looking statements are subject to known and unknown risks, uncertainties, assumptions and other factors. It is not possi ble for our management to predict all risks, nor can we assess the impact of all factors on our business or the extent to which any factor, or combinat ion of factors, may cause actual results to differ materially from those contained in any forward - looking statements we may make. These factors, together with th ose that are described in under the heading “Risk Factors” contained in documents the Company files with the Securities and Exchange Commission (“SEC”) from tim e to time, may cause our actual results, performance or achievements to differ materially and adversely from those anticipated or implied by our forwa rd - looking statements. Statements, including forward - looking statements, speak only to the date they are provided (unless an earlier date is indicated). We undert ake no obligation to update publicly any forward - looking statements for any reason after the date of this prospectus to conform these statements to actual r esults or to changes in our expectations, except as required by law. Although we believe the expectations reflected in such forward - looking statements are r easonable, we can give no assurance that such expectations will prove to be correct. Accordingly, readers are cautioned not to place undue reliance on the se forward - looking statements. 2 |