
STRATUM Phase 3 Seborrheic Dermatitis Topline Data Presentation June 2022

2©Copyright 2022. Arcutis Biotherapeutics, Inc. All rights reserved. Presentation designed for an investor audience. Legal Disclaimers This presentation and the accompanying oral presentation contain “forward-looking” statements that are based on our management’s beliefs and assumptions and on information currently available to management. Forward-looking statements include all statements other than statements of historical fact contained in this presentation, including information concerning our current and future financial performance, business plans and objectives, current and future clinical and preclinical development activities, current and future commercialization activities, timing and success of our ongoing and planned clinical trials and related data, the timing of announcements, updates and results of our clinical trials and related data, our ability to obtain and maintain regulatory approval, the potential therapeutic benefits and economic value of our product candidates, competitive position, industry environment, and potential market opportunities. Forward-looking statements are subject to known and unknown risks, uncertainties, assumptions and other factors including, but not limited to, those related to the success, cost and timing of our product candidate development activities and ongoing and planned clinical trials; our plans to develop and commercialize targeted therapeutics, including our lead product candidates roflumilast cream and roflumilast foam; the progress of patient enrollment and dosing in our clinical trials; the ability of our product candidates to achieve applicable endpoints in the clinical trials; the safety profile of our product candidates; the potential for data from our clinical trials to support a marketing application, as well as the timing of these events; our ability to obtain funding for our operations, development and commercialization of our product candidates; the timing of and our ability to obtain and maintain regulatory approvals; the rate and degree of market acceptance and clinical utility of our product candidates; the size and growth potential of the markets for our product candidates, and our ability to serve those markets; our commercialization, marketing and manufacturing capabilities and strategy; future agreements with third parties in connection with the commercialization of our product candidates; our expectations regarding our ability to obtain and maintain intellectual property protection; our dependence on third party manufacturers; the success of competing therapies that are or may become available; our ability to attract and retain key scientific or management personnel; our ability to identify additional product candidates with significant commercial potential consistent with our commercial objectives; and our estimates regarding expenses, future revenue, capital requirements and needs for additional financing. Moreover, we operate in a very competitive and rapidly changing environment, and new risks may emerge from time to time. It is not possible for our management to predict all risks, nor can we assess the impact of all factors on our business or the extent to which any factor, or combination of factors, may cause actual results to differ materially from those contained in any forward-looking statements we may make. In light of these risks, uncertainties and assumptions, the forward- looking events and circumstances discussed herein may not occur and actual results could differ materially and adversely from those anticipated or implied in the forward-looking statements. Further information on these and other factors that could affect these forward-looking statements is contained in our our Form 10-K filed with U.S. Securities and Exchange Commission (SEC) on February 22, 2022, and other reports filed with the SEC from time to time. You should not rely upon forward-looking statements as predictions of future events. Although our management believes that the expectations reflected in our forward-looking statements are reasonable, we cannot guarantee that the future results, levels of activity, performance or events and circumstances described in the forward-looking statements will be achieved or occur. We undertake no obligation to publicly update any forward-looking statements, whether written or oral, that may be made from time to time, whether as a result of new information, future developments or otherwise. This presentation also contains estimates and other statistical data made by independent parties and by us relating to market size and growth and other data about our industry. This data involves a number of assumptions and limitations, and you are cautioned not to give undue weight to such estimates. Neither we nor any other person makes any representation as to the accuracy or completeness of such data or undertakes any obligation to update such data after the date of this presentation. In addition, projections, assumptions and estimates of our future performance and the future performance of the markets in which we operate are necessarily subject to a high degree of uncertainty and risk. All product and company names are trademarksTM or registered® trademarks of their respective holders.

3©Copyright 2022. Arcutis Biotherapeutics, Inc. All rights reserved. Presentation designed for an investor audience. Today’s Speakers Frank Watanabe President & CEO Patrick Burnett, MD, PhD, FAAD Chief Medical Officer Ken Lock Chief Commercial Officer Zoe Diana Draelos, MD, FAAD Principal Investigator & President, Dermatology Consulting Services, PLLC

4©Copyright 2022. Arcutis Biotherapeutics, Inc. All rights reserved. Presentation designed for an investor audience. Arcutis Overview Speakers & Agenda

5©Copyright 2022. Arcutis Biotherapeutics, Inc. All rights reserved. Presentation designed for an investor audience. 2022: A Transformational Year for Arcutis Topical roflumilast offers a differentiated clinical profile, targeting three distinct disease areas each with >2 million topically treated patients today in U.S. Dermatology offices We are increasingly excited about the clinical profile and the underappreciated opportunity of roflumilast foam in seborrheic dermatitis We are progressing our commercial launch preparations in advance of our July PDUFA date for roflumilast cream in plaque psoriasis We remain confident in replicating our track record of Phase 3 success in subsequent pivotal readouts in atopic dermatitis and scalp and body psoriasis later this year
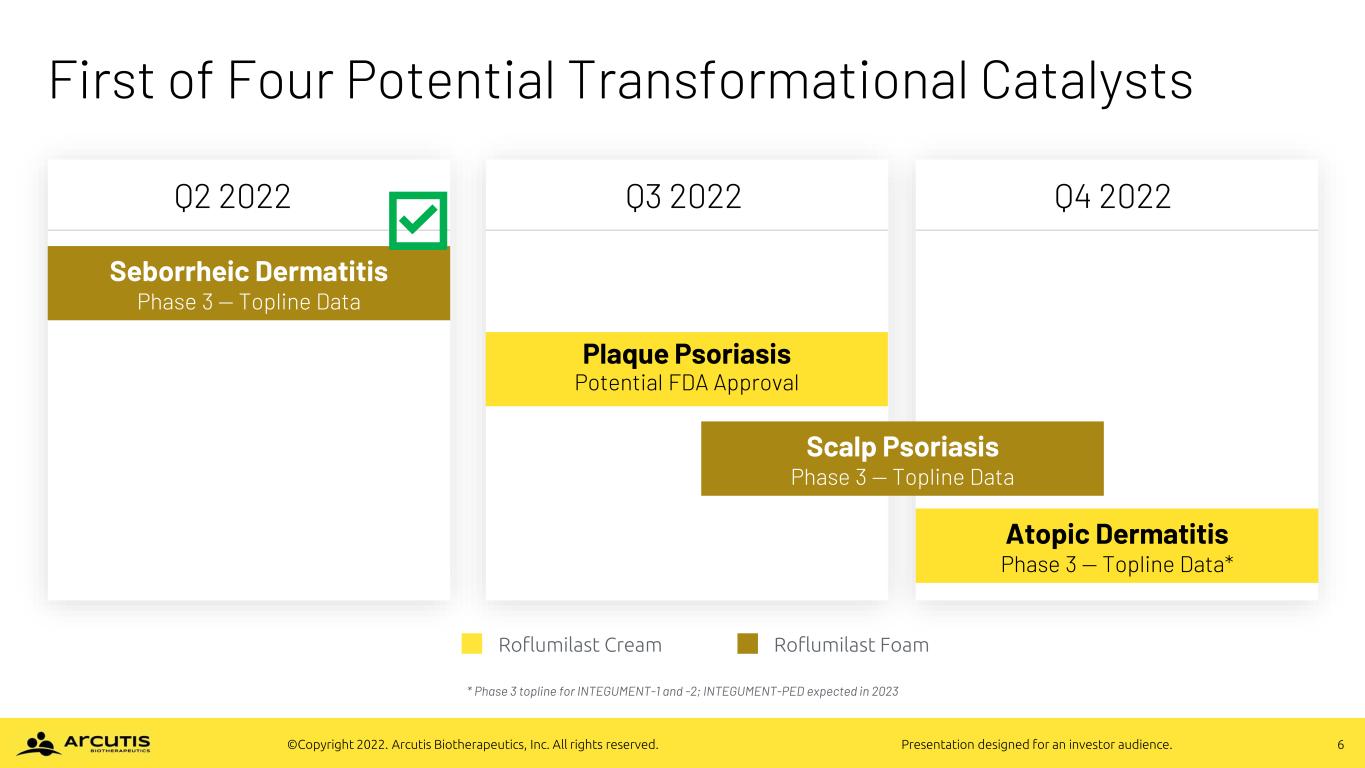
6©Copyright 2022. Arcutis Biotherapeutics, Inc. All rights reserved. Presentation designed for an investor audience. First of Four Potential Transformational Catalysts * Phase 3 topline for INTEGUMENT-1 and -2; INTEGUMENT-PED expected in 2023 Roflumilast Cream Roflumilast Foam Atopic Dermatitis Phase 3 — Topline Data* Seborrheic Dermatitis Phase 3 — Topline Data Scalp Psoriasis Phase 3 — Topline Data Plaque Psoriasis Potential FDA Approval Q4 2022Q3 2022Q2 2022
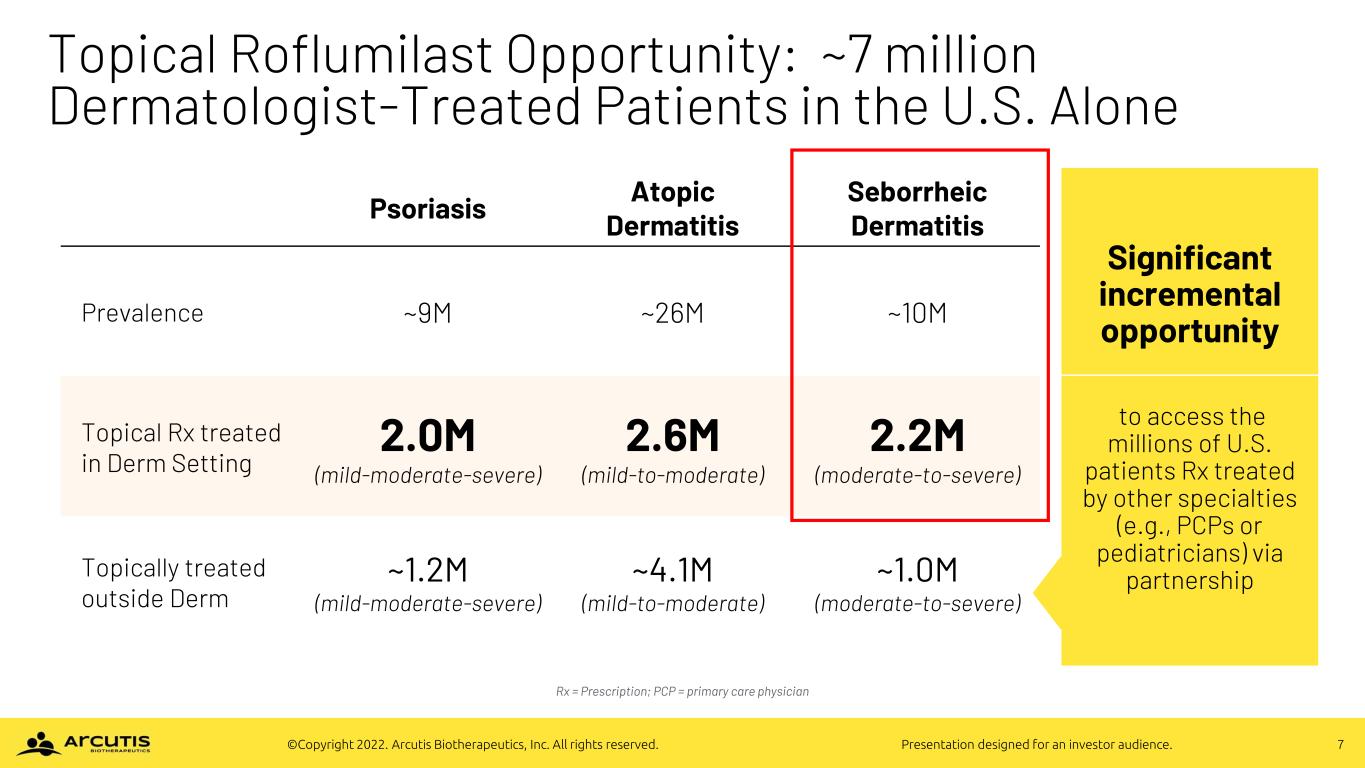
7©Copyright 2022. Arcutis Biotherapeutics, Inc. All rights reserved. Presentation designed for an investor audience. Topical Roflumilast Opportunity: ~7 million Dermatologist-Treated Patients in the U.S. Alone Psoriasis Atopic Dermatitis Seborrheic Dermatitis Prevalence ~9M ~26M ~10M Topical Rx treated in Derm Setting 2.0M (mild-moderate-severe) 2.6M (mild-to-moderate) 2.2M (moderate-to-severe) Topically treated outside Derm ~1.2M (mild-moderate-severe) ~4.1M (mild-to-moderate) ~1.0M (moderate-to-severe) Significant incremental opportunity to access the millions of U.S. patients Rx treated by other specialties (e.g., PCPs or pediatricians) via partnership Rx = Prescription; PCP = primary care physician

8©Copyright 2022. Arcutis Biotherapeutics, Inc. All rights reserved. Presentation designed for an investor audience. Topical Roflumilast – A Differentiated & Transformational Clinical Profile Efficacy results on par with steroid / vitamin D combinations Non-steroidal with ability to use chronically, anywhere on the body No boxed warnings anticipated Favorable local tolerability Individuals evaluated with topical roflumilast across clinical programs ~3.5K

9©Copyright 2022. Arcutis Biotherapeutics, Inc. All rights reserved. Presentation designed for an investor audience. Seborrheic Dermatitis Disease State Education Speakers & Agenda Zoe Diana Draelos, MD, FAAD Principal Investigator & President, Dermatology Consulting Services, PLLC
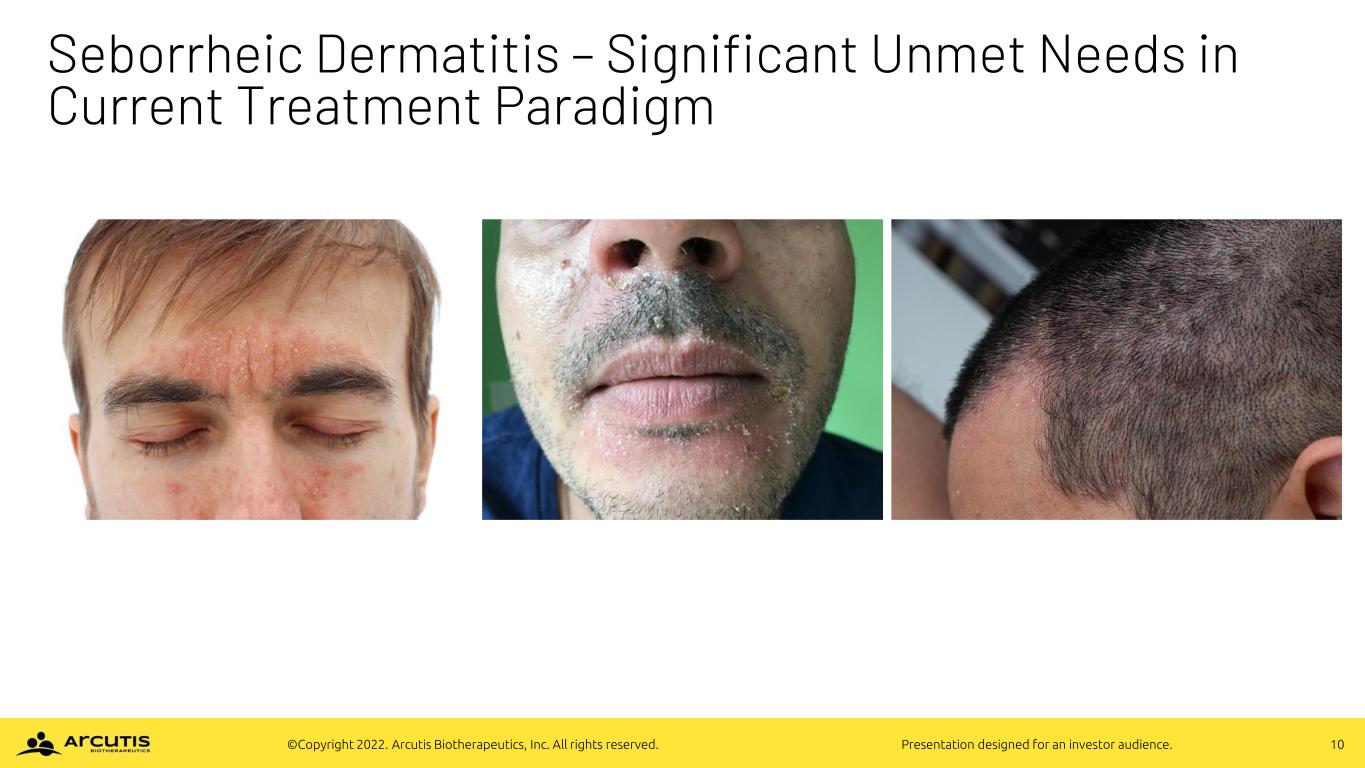
10©Copyright 2022. Arcutis Biotherapeutics, Inc. All rights reserved. Presentation designed for an investor audience. Seborrheic Dermatitis – Significant Unmet Needs in Current Treatment Paradigm

11©Copyright 2022. Arcutis Biotherapeutics, Inc. All rights reserved. Presentation designed for an investor audience. Speakers & Agenda Clinical Results Zoe Diana Draelos, MD, FAAD
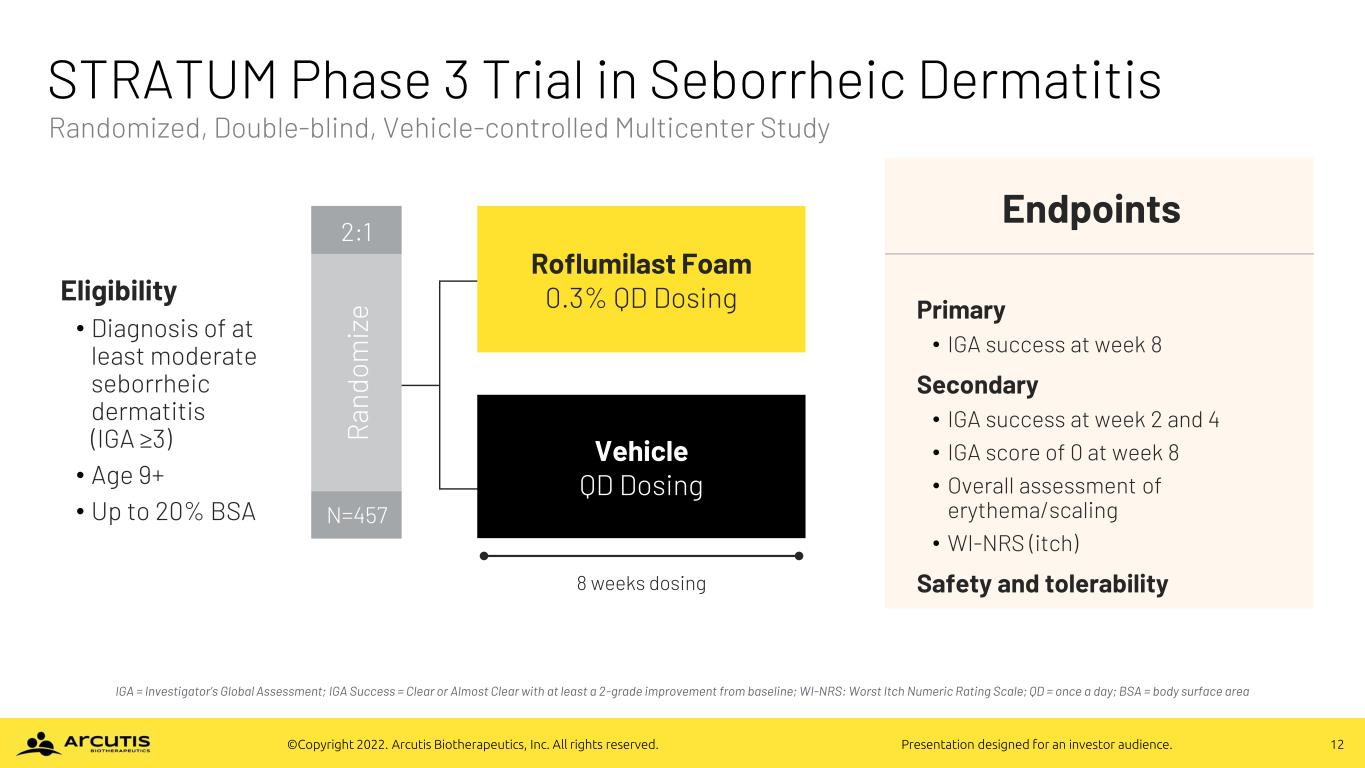
12©Copyright 2022. Arcutis Biotherapeutics, Inc. All rights reserved. Presentation designed for an investor audience. STRATUM Phase 3 Trial in Seborrheic Dermatitis IGA = Investigator’s Global Assessment; IGA Success = Clear or Almost Clear with at least a 2-grade improvement from baseline; WI-NRS: Worst Itch Numeric Rating Scale; QD = once a day; BSA = body surface area Endpoints Primary • IGA success at week 8 Secondary • IGA success at week 2 and 4 • IGA score of 0 at week 8 • Overall assessment of erythema/scaling • WI-NRS (itch) Safety and tolerability Eligibility • Diagnosis of at least moderate seborrheic dermatitis (IGA ≥3) • Age 9+ • Up to 20% BSA Roflumilast Foam 0.3% QD Dosing Vehicle QD Dosing 8 weeks dosing 2:1 R an do m iz e N=457 Randomized, Double-blind, Vehicle-controlled Multicenter Study
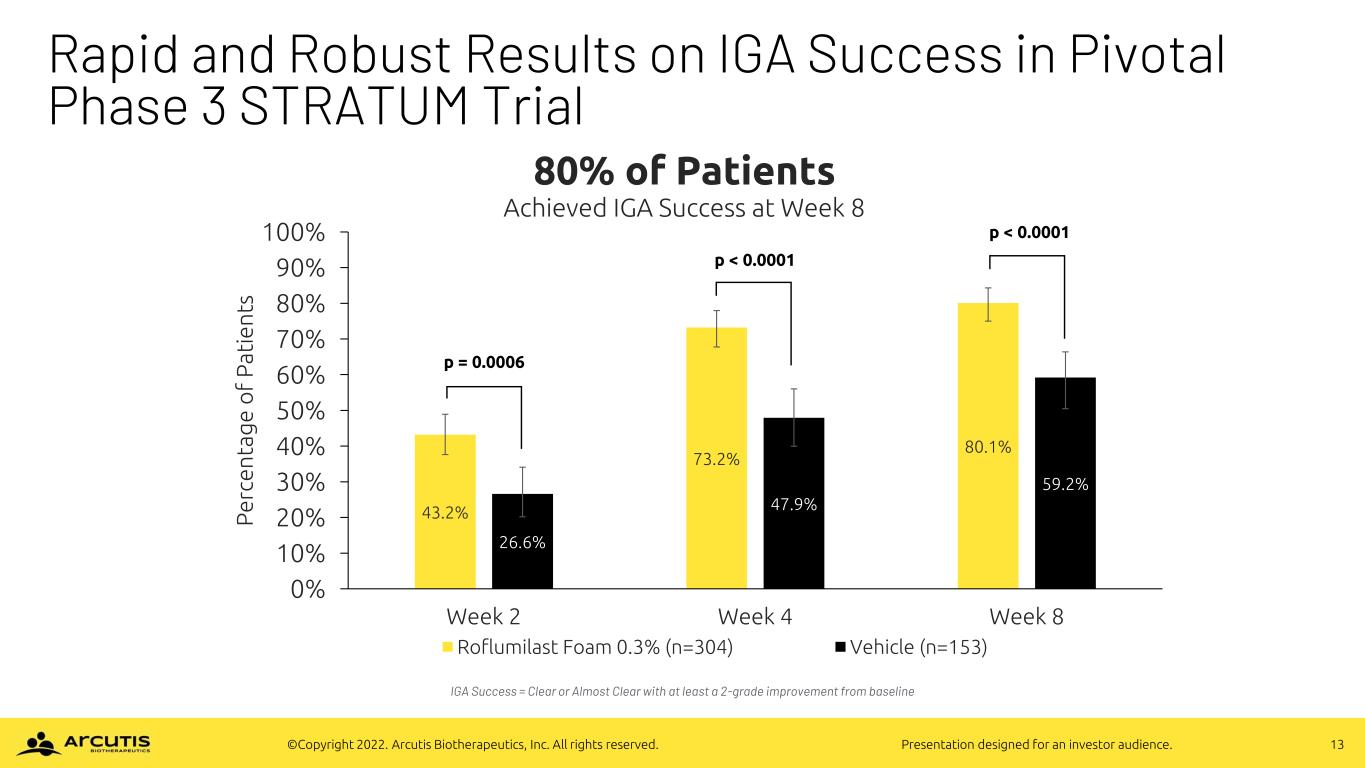
13©Copyright 2022. Arcutis Biotherapeutics, Inc. All rights reserved. Presentation designed for an investor audience. Rapid and Robust Results on IGA Success in Pivotal Phase 3 STRATUM Trial 43.2% 73.2% 80.1% 26.6% 47.9% 59.2% 0% 10% 20% 30% 40% 50% 60% 70% 80% 90% 100% Week 2 Week 4 Week 8 P e rc e n ta g e o f P a ti e n ts 80% of Patients Achieved IGA Success at Week 8 Roflumilast Foam 0.3% (n=304) Vehicle (n=153) IGA Success = Clear or Almost Clear with at least a 2-grade improvement from baseline p = 0.0006 p < 0.0001 p < 0.0001

14©Copyright 2022. Arcutis Biotherapeutics, Inc. All rights reserved. Presentation designed for an investor audience. Over 50% of Patients Achieved IGA of Clear at Week 8 IGA = Investigator’s Global Assessment 16.4% 35.6% 50.7% 6.5% 14.9% 28.2% 0% 10% 20% 30% 40% 50% 60% 70% 80% 90% 100% Week 2 Week 4 Week 8 P e rc e n ta g e o f P a ti e n ts >50% of Patients Achieved IGA of Clear (IGA = 0) at Week 8 Roflumilast Foam 0.3% (n=304) Vehicle (n=153) p = 0.0047 p < 0.0001 p < 0.0001
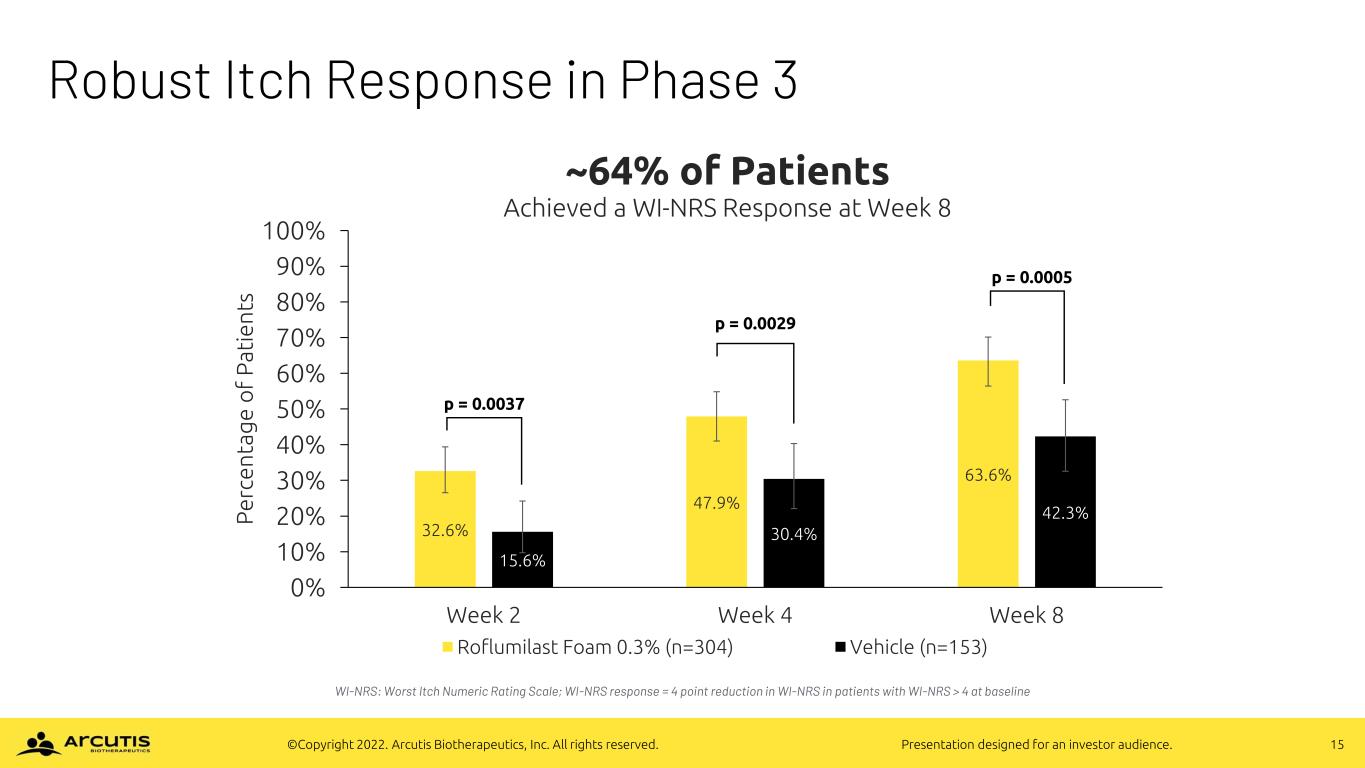
15©Copyright 2022. Arcutis Biotherapeutics, Inc. All rights reserved. Presentation designed for an investor audience. Robust Itch Response in Phase 3 WI-NRS: Worst Itch Numeric Rating Scale; WI-NRS response = 4 point reduction in WI-NRS in patients with WI-NRS > 4 at baseline 32.6% 47.9% 63.6% 15.6% 30.4% 42.3% 0% 10% 20% 30% 40% 50% 60% 70% 80% 90% 100% Week 2 Week 4 Week 8 P e rc e n ta g e o f P a ti e n ts ~64% of Patients Achieved a WI-NRS Response at Week 8 Roflumilast Foam 0.3% (n=304) Vehicle (n=153) p = 0.0037 p = 0.0029 p = 0.0005
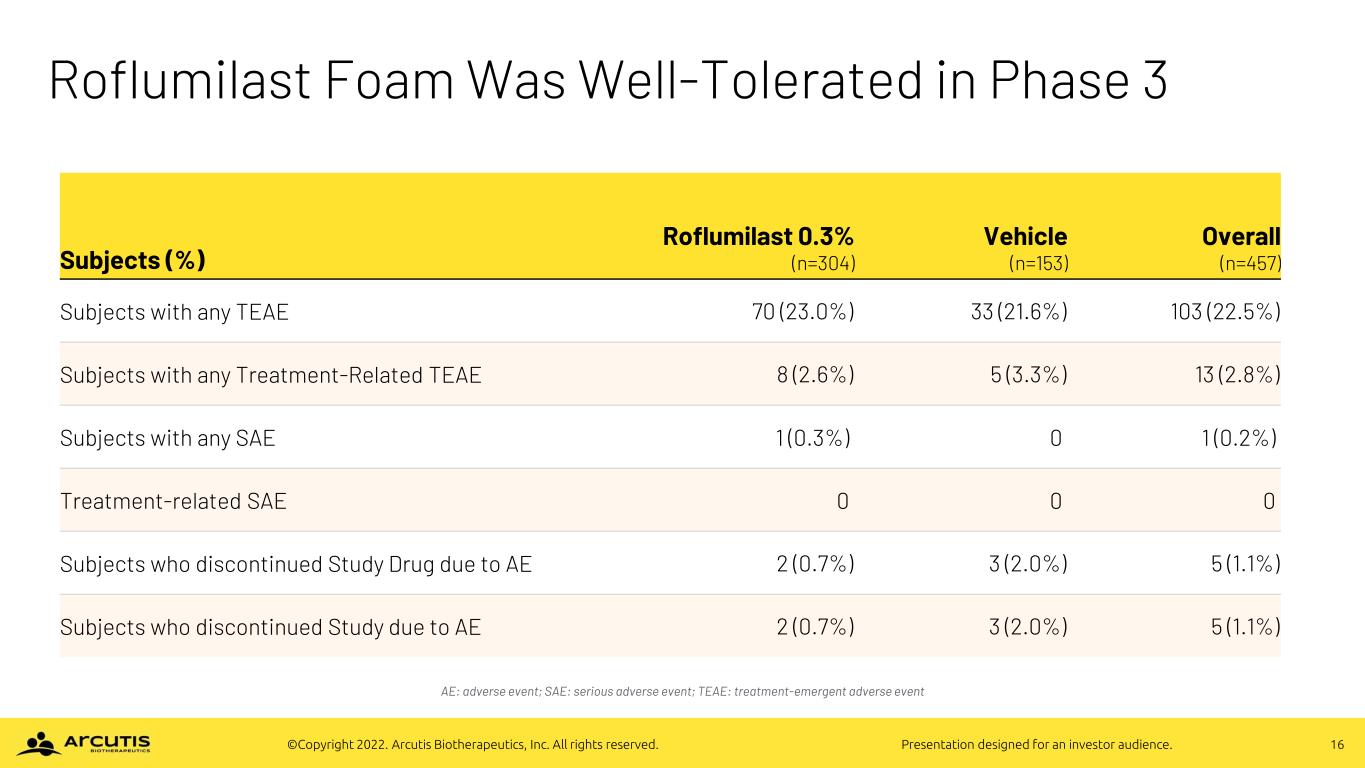
16©Copyright 2022. Arcutis Biotherapeutics, Inc. All rights reserved. Presentation designed for an investor audience. Roflumilast Foam Was Well-Tolerated in Phase 3 AE: adverse event; SAE: serious adverse event; TEAE: treatment-emergent adverse event Subjects (%) Roflumilast 0.3% (n=304) Vehicle (n=153) Overall (n=457) Subjects with any TEAE 70 (23.0%) 33 (21.6%) 103 (22.5%) Subjects with any Treatment-Related TEAE 8 (2.6%) 5 (3.3%) 13 (2.8%) Subjects with any SAE 1 (0.3%) 0 1 (0.2%) Treatment-related SAE 0 0 0 Subjects who discontinued Study Drug due to AE 2 (0.7%) 3 (2.0%) 5 (1.1%) Subjects who discontinued Study due to AE 2 (0.7%) 3 (2.0%) 5 (1.1%)
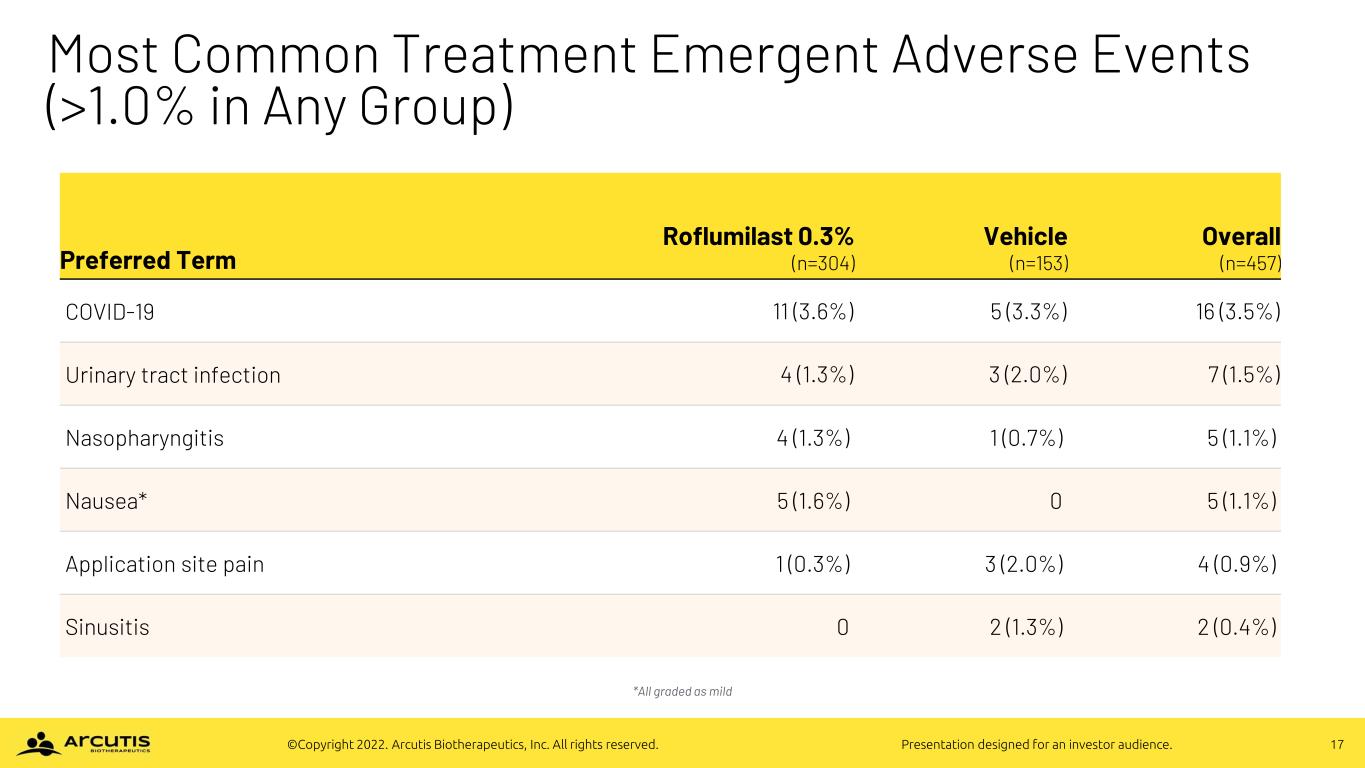
17©Copyright 2022. Arcutis Biotherapeutics, Inc. All rights reserved. Presentation designed for an investor audience. Most Common Treatment Emergent Adverse Events (>1.0% in Any Group) *All graded as mild Preferred Term Roflumilast 0.3% (n=304) Vehicle (n=153) Overall (n=457) COVID-19 11 (3.6%) 5 (3.3%) 16 (3.5%) Urinary tract infection 4 (1.3%) 3 (2.0%) 7 (1.5%) Nasopharyngitis 4 (1.3%) 1 (0.7%) 5 (1.1%) Nausea* 5 (1.6%) 0 5 (1.1%) Application site pain 1 (0.3%) 3 (2.0%) 4 (0.9%) Sinusitis 0 2 (1.3%) 2 (0.4%)

18©Copyright 2022. Arcutis Biotherapeutics, Inc. All rights reserved. Presentation designed for an investor audience. Speakers & Agenda Commercial Opportunity

19©Copyright 2022. Arcutis Biotherapeutics, Inc. All rights reserved. Presentation designed for an investor audience. Roflumilast Foam Could Become Standard of Care in Seborrheic Dermatitis Efficacy data showing meaningful symptomatic improvement The ability to use on all affected areas of the body Safety and tolerability data that supports potential for chronic use Simple, easy-to-use, 1x daily foam for scalp Dual Mechanism: Anti-fungal + Anti- inflammatory X
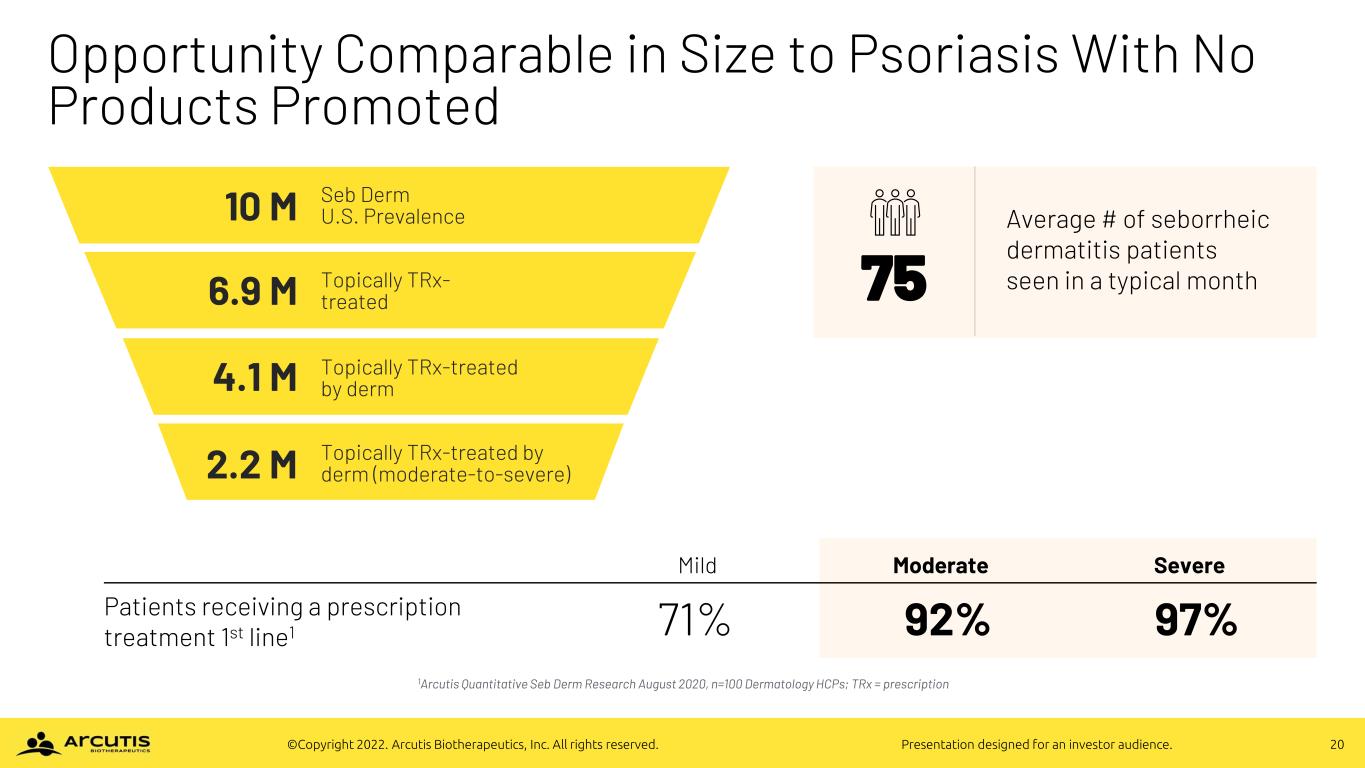
20©Copyright 2022. Arcutis Biotherapeutics, Inc. All rights reserved. Presentation designed for an investor audience. Opportunity Comparable in Size to Psoriasis With No Products Promoted 1Arcutis Quantitative Seb Derm Research August 2020, n=100 Dermatology HCPs; TRx = prescription Mild Moderate Severe Patients receiving a prescription treatment 1st line1 71% 92% 97% 75 Average # of seborrheic dermatitis patients seen in a typical month 10 M 6.9 M 4.1 M 2.2 M Seb Derm U.S. Prevalence Topically TRx- treated Topically TRx-treated by derm Topically TRx-treated by derm (moderate-to-severe)

21©Copyright 2022. Arcutis Biotherapeutics, Inc. All rights reserved. Presentation designed for an investor audience. Patients Are Dissatisfied with Complex and Onerous Treatment Regimens 1Harris Poll Seborrheic Dermatitis Survey (n>600 HCPs, n=300 patients), OTC = over the counter 1.4 2.1 2.4 Prescription treatments OTC treatments Alternative treatments TOTAL 5.9 Treatments Used Per Week1, Mean “I am interested in trying new treatment options.” 9 in 10 AGREE1 Patients ready for new options 9 in 10AGREE1 “I would be more likely to stick with a treatment plan if it meant using fewer treatments.”
22©Copyright 2022. Arcutis Biotherapeutics, Inc. All rights reserved. Presentation designed for an investor audience. Zoe Diana Draelos, MD, FAAD Thank You Q&A