Exhibit 99.2

Fourth Quarter 2020 ResultsMarch 18, 2021

Disclaimer This presentation has been prepared by Legend Biotech Corporation (“Legend Biotech” or the “Company”) solely for informational purposes and does not contain all relevant information relating to the Company. The safety and efficacy of the agents and/or uses under investigation discussed in this presentation have not been established. There is no guarantee that the agents will receive health authority approval or become commercially available in any country for the uses being investigated. Forward-Looking Statements This presentation contains “forward-looking statements” within the meaning of The Private Securities Litigation Reform Act of 1995. The words “anticipate,” “believe,” “continue,” “could,” “estimate,” “expect,” “intend,” “may,” “plan,” “potential,” “predict,” “project,” “should,” “target,” “will,” “would” and similar expressions are intended to identify forward-looking statements, although not all forward-looking statements contain these identifying words. These forward-looking statements include, but are not limited to, statements relating to the Company’s strategies and objectives; the anticipated timing of, and ability to progress, clinical trials, including the initiation of the phase 1 clinical trial of LB1901 in RRTCL; the ability to make, the timing of, and the ultimate success of regulatory submissions globally, including the rolling BLA for cilta-cel with the U.S. FDA, the MAA for cilta-cel with the EMA, and the submissions for cilta-cel to the CDE and JMHLW;the ability to generate, analyze and present data from clinical trials; patient enrollment; the potential benefits of our product candidates; and the status and outcome of the investigation being conducted by the Customs Anti-Smuggling Department of Zhenjiang in China and its impact on the Company’s operations. Actual results may differ materially from those indicated by such forward-looking statements as a result of various important factors. Legend Biotech’s expectations could be affected by, among other things, uncertainties involved in the development of new pharmaceutical products; unexpected clinical trial results, including as a result of additional analysis of existing clinical data or unexpected new clinical data; unexpected regulatory actions or delays, including requests for additional safety and/or efficacy data or analysis of data, or government regulation generally; unexpected delays as a result of actions undertaken, or failures to act, by our third party partners; uncertainties arising from challenges to Legend Biotech’s patent or other proprietary intellectual property protection, including the uncertainties involved in the US litigation process; competition in general; government, industry, and general public pricing and other political pressures; the duration and severity of the COVID-19 pandemic and governmental and regulatory measures implemented in response to the evolving situation; as well as the other factors discussed in the “Risk Factors” section of the Company’s prospectus filed with the Securities and Exchange Commission on June 8, 2020. Should one or more of these risks or uncertainties materialize, or should underlying assumptions prove incorrect, actual results may vary materially from those described in this presentation as anticipated, believed, estimated or expected. Legend Biotech specifically disclaims any obligation to update any forward-looking statement, whether as a result of new information, future events or otherwise. 2

Agenda 3 Near-Term Targets for Legend Biotech Q&A CEO Opening Remarks and 2020 Financial Results “2020 was a very successful year highlighted by solid financial performance as we expanded our pipeline, built a robust team to support our multiple platforms for fighting debilitating diseases, and we completed an initial public offering. Our year concluded with the initiation of rolling submission of BLA to US FDA for cilta-cel and our team worked tirelessly to obtain FDA clearance of the IND for LB1901. We believe these regulatory, partnering and clinical milestones position Legend Biotech for even stronger performance in 2021 and beyond.” – Ying Huang, CEO and CFO of Legend Biotech ASH 2020 Data Discussion

4 Phase 1b/2 Study Data of Cilta-cel (CARTITUDE-1) Data continued to show a very high overall response rate that deepened over time with 97% of patients achieving an overall response and 67% of patients achieving a stringent complete response (sCR) at a median follow-up of 12.4 monthsDemonstrated a manageable safety profile for cilta-cel at the recommended Phase 2 dose US FDA cleared Legend Biotech’s IND application to evaluate LB1901 for the treatment of adults with relapsed or refractory T-cell lymphoma Initiated a rolling submission of BLA to the US FDA for cilta-cel for treatment of adults with relapsed and/or refractory multiple myeloma (RRMM) FDA Clearance of the IND for LB1901 Initiation of Rolling Submission of Biologics License Application to US FDA 4th Quarter 2020 and Most Recent Company Highlights ASH 2020 Data Presentations Updated data from CARTITUDE-1 and LEGEND-2 studies presented at ASH Committee for Medicinal Products for Human Use of the European Medicines Agency has accepted a request for an accelerated assessment of the Marketing Authorisation Application for cilta-cel Accelerated Assessment in Europe for the Treatment of RRMM
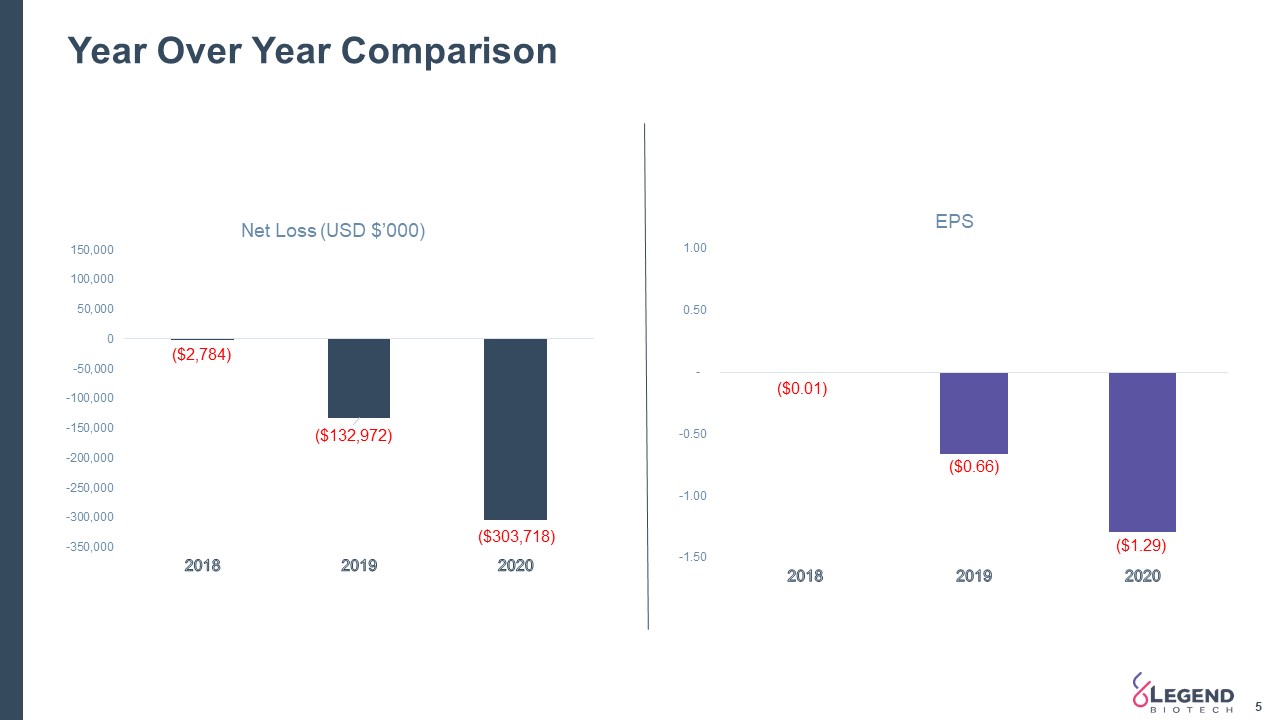
Year Over Year Comparison 5
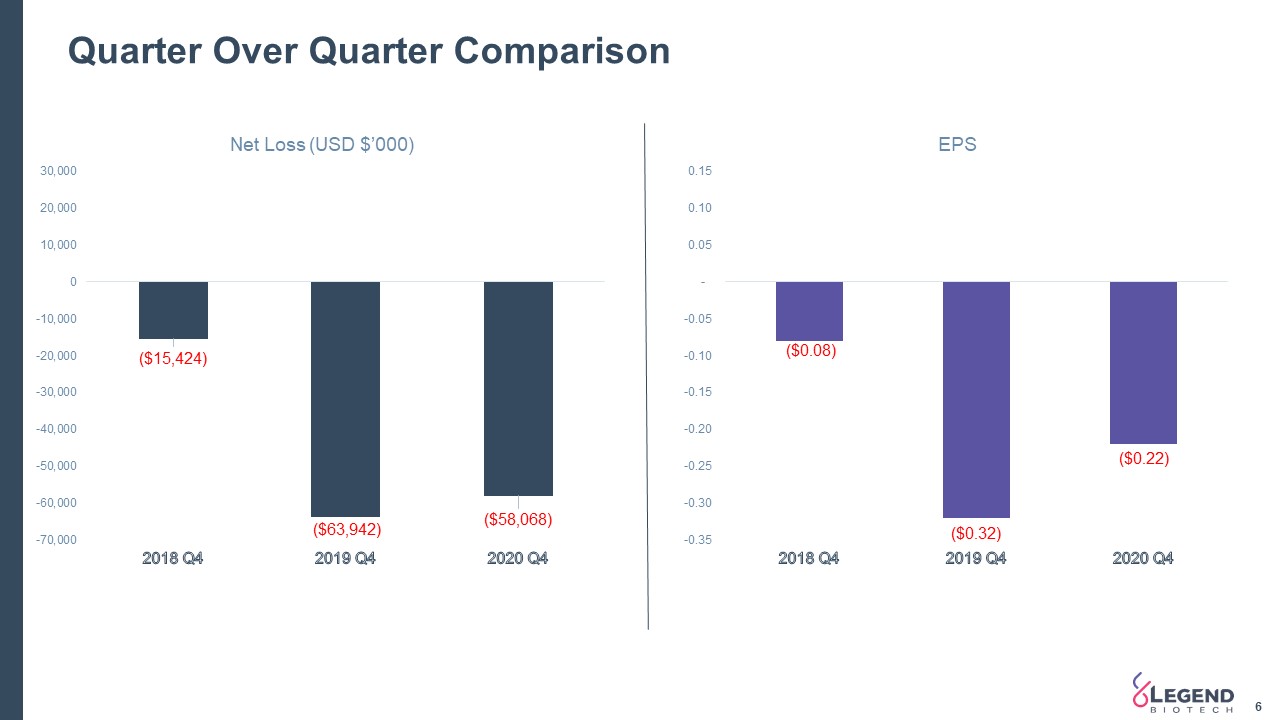
Quarter Over Quarter Comparison 6
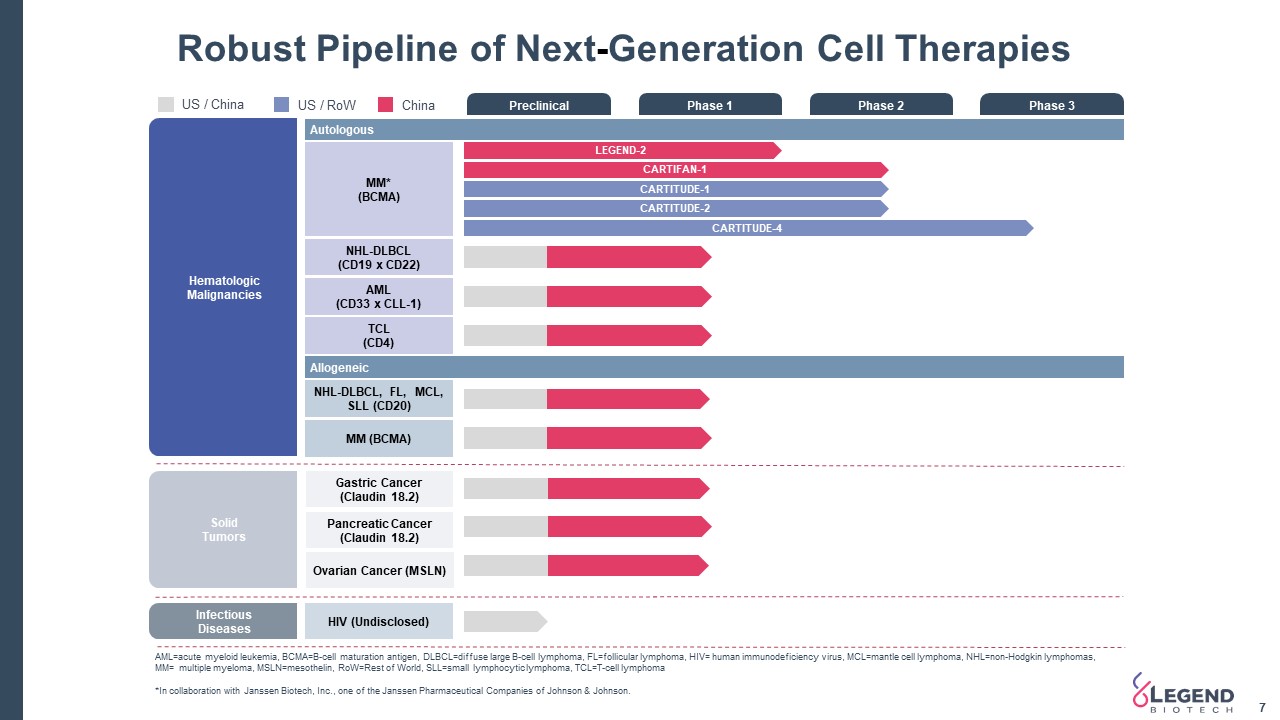
Robust Pipeline of Next-Generation Cell Therapies 7 InfectiousDiseases SolidTumors Preclinical Phase 1 Phase 2 Phase 3 Autologous TCL(CD4) NHL-DLBCL(CD19 x CD22) AML(CD33 x CLL-1) MM*(BCMA) Allogeneic NHL-DLBCL, FL, MCL, SLL (CD20) HIV (Undisclosed) LEGEND-2 CARTITUDE-4 Pancreatic Cancer(Claudin 18.2) Gastric Cancer(Claudin 18.2) Ovarian Cancer (MSLN) Hematologic Malignancies CARTITUDE-1 CARTIFAN-1 CARTITUDE-2 US / RoW China AML=acute myeloid leukemia, BCMA=B-cell maturation antigen, DLBCL=diffuse large B-cell lymphoma, FL=follicular lymphoma, HIV= human immunodeficiency virus, MCL=mantle cell lymphoma, NHL=non-Hodgkin lymphomas, MM= multiple myeloma, MSLN=mesothelin, RoW=Rest of World, SLL=small lymphocytic lymphoma, TCL=T-cell lymphoma*In collaboration with Janssen Biotech, Inc., one of the Janssen Pharmaceutical Companies of Johnson & Johnson. MM (BCMA) US / China
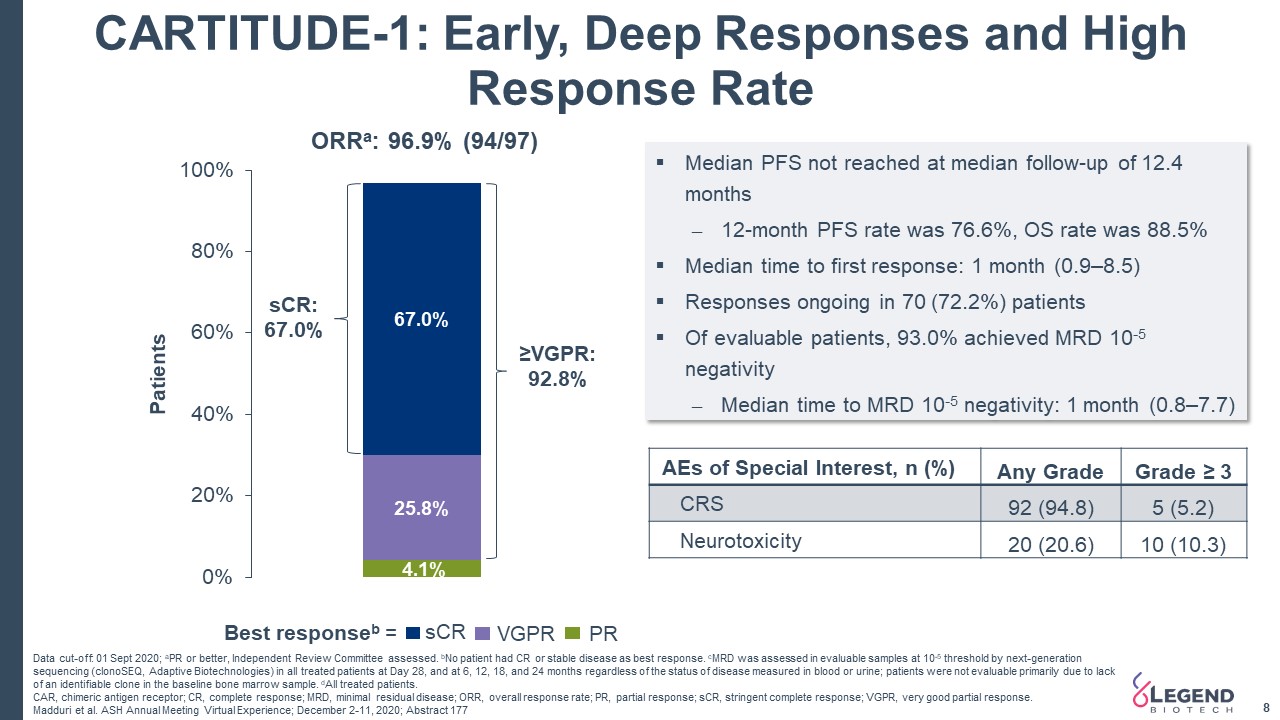
8 CARTITUDE-1: Early, Deep Responses and High Response Rate Data cut-off: 01 Sept 2020; aPR or better, Independent Review Committee assessed. bNo patient had CR or stable disease as best response. cMRD was assessed in evaluable samples at 10-5 threshold by next-generation sequencing (clonoSEQ, Adaptive Biotechnologies) in all treated patients at Day 28, and at 6, 12, 18, and 24 months regardless of the status of disease measured in blood or urine; patients were not evaluable primarily due to lack of an identifiable clone in the baseline bone marrow sample. dAll treated patients. CAR, chimeric antigen receptor; CR, complete response; MRD, minimal residual disease; ORR, overall response rate; PR, partial response; sCR, stringent complete response; VGPR, very good partial response. Madduri et al. ASH Annual Meeting Virtual Experience; December 2-11, 2020; Abstract 177 Median PFS not reached at median follow-up of 12.4 months12-month PFS rate was 76.6%, OS rate was 88.5%Median time to first response: 1 month (0.9–8.5)Responses ongoing in 70 (72.2%) patientsOf evaluable patients, 93.0% achieved MRD 10-5 negativityMedian time to MRD 10-5 negativity: 1 month (0.8–7.7) ≥VGPR: 92.8% sCR: 67.0% sCR VGPR PR Best responseb = AEs of Special Interest, n (%) Any Grade Grade ≥ 3 CRS 92 (94.8) 5 (5.2) Neurotoxicity 20 (20.6) 10 (10.3)

Near-Term Targets for Legend Biotech 9 File BLA in China for cilta-cel Target FDA approval for cilta-cel in US 1H21 2H21 US: United States; BLA: Biologics License Application; FDA: Federal Drug Administration; MAA: Marketing Authorization Application; EMA: European Medicines Agency Initiate phase 1 study for LB1901 for T-cell Lymphoma in US 2021 File MAA to the EMA for cilta-cel 2H21

10 Target CDE approval for cilta-cel in China 2022 Target EMA approval for cilta-cel in EU 2022 Near-Term Targets for Legend Biotech File NDA to the Japan Ministry of Health, Labor and Welfare for cilta-cel 2H21

Data Update 11
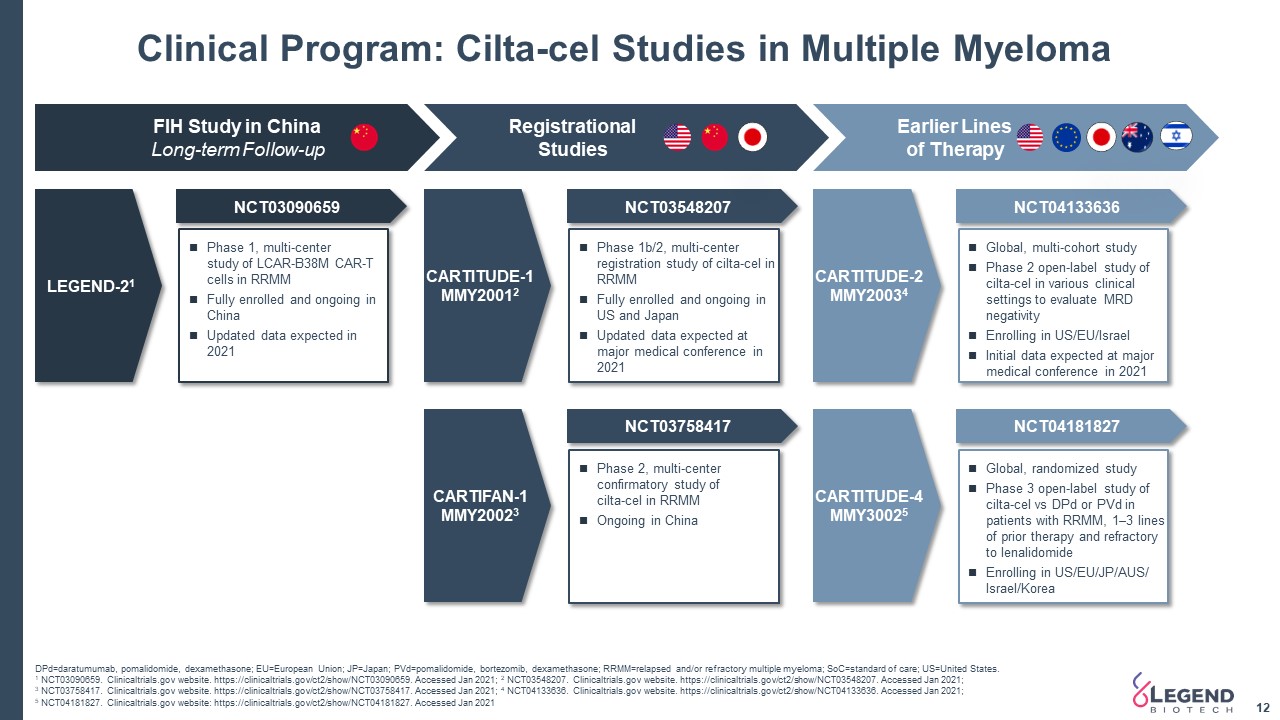
Clinical Program: Cilta-cel Studies in Multiple Myeloma 12 DPd=daratumumab, pomalidomide, dexamethasone; EU=European Union; JP=Japan; PVd=pomalidomide, bortezomib, dexamethasone; RRMM=relapsed and/or refractory multiple myeloma; SoC=standard of care; US=United States. 1 NCT03090659. Clinicaltrials.gov website. https://clinicaltrials.gov/ct2/show/NCT03090659. Accessed Jan 2021; 2 NCT03548207. Clinicaltrials.gov website. https://clinicaltrials.gov/ct2/show/NCT03548207. Accessed Jan 2021;3 NCT03758417. Clinicaltrials.gov website. https://clinicaltrials.gov/ct2/show/NCT03758417. Accessed Jan 2021; 4 NCT04133636. Clinicaltrials.gov website. https://clinicaltrials.gov/ct2/show/NCT04133636. Accessed Jan 2021; 5 NCT04181827. Clinicaltrials.gov website: https://clinicaltrials.gov/ct2/show/NCT04181827. Accessed Jan 2021 FIH Study in ChinaLong-term Follow-up Registrational Studies Earlier Lines of Therapy LEGEND-21 NCT03090659 NCT03548207 NCT03758417 NCT04133636 NCT04181827 Phase 1, multi-centerstudy of LCAR-B38M CAR-T cells in RRMMFully enrolled and ongoing in ChinaUpdated data expected in 2021 Phase 2, multi-center confirmatory study ofcilta-cel in RRMMOngoing in China Phase 1b/2, multi-center registration study of cilta-cel in RRMMFully enrolled and ongoing in US and JapanUpdated data expected at major medical conference in 2021 Global, multi-cohort studyPhase 2 open-label study of cilta-cel in various clinical settings to evaluate MRD negativityEnrolling in US/EU/IsraelInitial data expected at major medical conference in 2021 Global, randomized studyPhase 3 open-label study of cilta-cel vs DPd or PVd in patients with RRMM, 1–3 lines of prior therapy and refractory to lenalidomideEnrolling in US/EU/JP/AUS/ Israel/Korea CARTITUDE-1MMY20012 CARTIFAN-1MMY20023 CARTITUDE-2MMY20034 CARTITUDE-4MMY30025

Q&A Session


Thank You !