Exhibit 99.1

Investor Presentation July 2022

2 PainReform Ltd. | www.painreform.com Post Operative Pain Management Forward - Looking Statements This presentation contains forward - looking statements about our expectations, beliefs and intentions . Forward - looking statements can be identified by the use of forward - looking words such as “believe”, “expect”, “intend”, “plan”, “may”, “should”, “could”, “might”, “seek”, “target”, “will”, “project”, “forecast”, “continue” or “anticipate” or their negatives or variations of these words or other comparable words or by the fact that these statements do not relate strictly to historical matters . These forward - looking statements are based on assumptions and assessments made in light of management’s experience and perception of historical trends, current conditions, expected future developments and other factors believed to be appropriate . Because forward - looking statements relate to matters that have not yet occurred, these statements are inherently subject to risks and uncertainties that could cause our actual results to differ materially from any future results expressed or implied by the forward - looking statements . Many factors could cause our actual activities or results to differ materially from the activities and results anticipated in forward - looking statements, including, but not limited to, the following : our history of significant losses, our need to raise additional capital and our ability to obtain additional capital on acceptable terms, or at all ; our dependence on the success of our initial product candidate, PRF - 110 ; the outcomes of preclinical studies, clinical trials and other research regarding PRF - 110 and future product candidates ; the impact of the COVID - 19 pandemic on our operations ; our limited experience managing clinical trials ; our ability to retain key personnel and recruit additional employees ; our reliance on third parties for the conduct of clinical trials, product manufacturing and development ; the impact of competition and new technologies ; our ability to comply with regulatory requirements relating to the development and marketing of our product candidates ; commercial success and market acceptance of our product candidates ; our ability to establish sales and marketing capabilities or enter into agreements with third parties and our reliance on third party distributors and resellers ; our ability to establish and maintain strategic partnerships and other corporate collaborations ; the implementation of our business model and strategic plans for our business and product candidates ; the scope of protection we are able to establish and maintain for intellectual property rights and our ability to operate our business without infringing the intellectual property rights of others ; the overall global economic environment ; our ability to develop an active trading market for our ordinary shares and whether the market price of our ordinary shares is volatile ; and statements as to the impact of the political and security situation in Israel on our business . More detailed information about the risks and uncertainties affecting us is contained under the heading “Risk Factors” included in the Company’s most recent Annual Report on Form 20 - F and in other filings that we have made and may make with the Securities and Exchange Commission in the future . These statements are only current predictions and are subject to known and unknown risks, uncertainties and other factors that may cause our or our industry’s actual results, levels of activity, performance or achievements to be materially different from those anticipated by the forward - looking statements . Given these uncertainties, you should not rely upon forward - looking statements as predictions of future events . All forward - looking statements attributable to us or persons acting on our behalf included in, but not limited to, this presentation speak only as of the date hereof and are expressly qualified in their entirety by the foregoing . We undertake no obligations to update or revise forward - looking statements to reflect events or circumstances that arise after the date made or to reflect the occurrence of unanticipated events . In evaluating forward - looking statements, you should consider these risks and uncertainties . This presentation shall not constitute an offer to sell or the solicitation of an offer to buy, nor shall there be any sale of these securities in any state or other jurisdiction in which such offer, solicitation or sale would be unlawful prior to registration or qualification under the securities laws of any such state or other jurisdiction . The presentation contains information about investigation - stage drug products under development, which have not yet been approved by the FDA for commercial distribution in the United States . All representations in this presentation are based upon investigations in certain clinical and other research, but which accordingly should not be construed as general claims for the safety or efficacy of the products when used by patients .

3 PainReform Ltd. | www.painreform.com Post Operative Pain Management PRF - 110 Executive Summary ▪ Post - operative pain treatment is a growing market (~$12B) with a need for better therapeutics 1 o Local anesthetics provide pain relief for up to 6 hours and require augmentation with n on - steroidal anti - inflammatory drugs (NSAIDs) o NSAIDs or opioids for moderate to severe pain, leading to side effects and dependence o Opiate abuse and addiction cause 70,000 death in the US & an economic burden of $80B/yr 1 o Exparel (Pacira), a marketed long - acting liposomal generic local anesthetic has >$500M revenues: PCRX - market cap, at peak, over $4.0B – an important benchmark for PainReform 2 ▪ PainReform has developed PRF - 110, a novel formulation extended release ropivacaine o In a Phase 2 clinical study clinical study in 15 open hernia patients PRF - 110 demonstrated pain relief of up to 72 hours o Phase 3 study design and IND approved; FDA confirmed 505(b)(2) designation o Patent estate granted for PRF - 110 and formulation platform through 2033 prior to extensions - for US, Canada, EU, Israel, Australia, China, Japan, Russia and other countries o Green light from FDA to initiate two phase III trials (soft and hard tissue) of ~400 patients each for NDA submission ▪ Highly experienced board of directors and management team ▪ Inhouse clinical, manufacturing and QA know - how
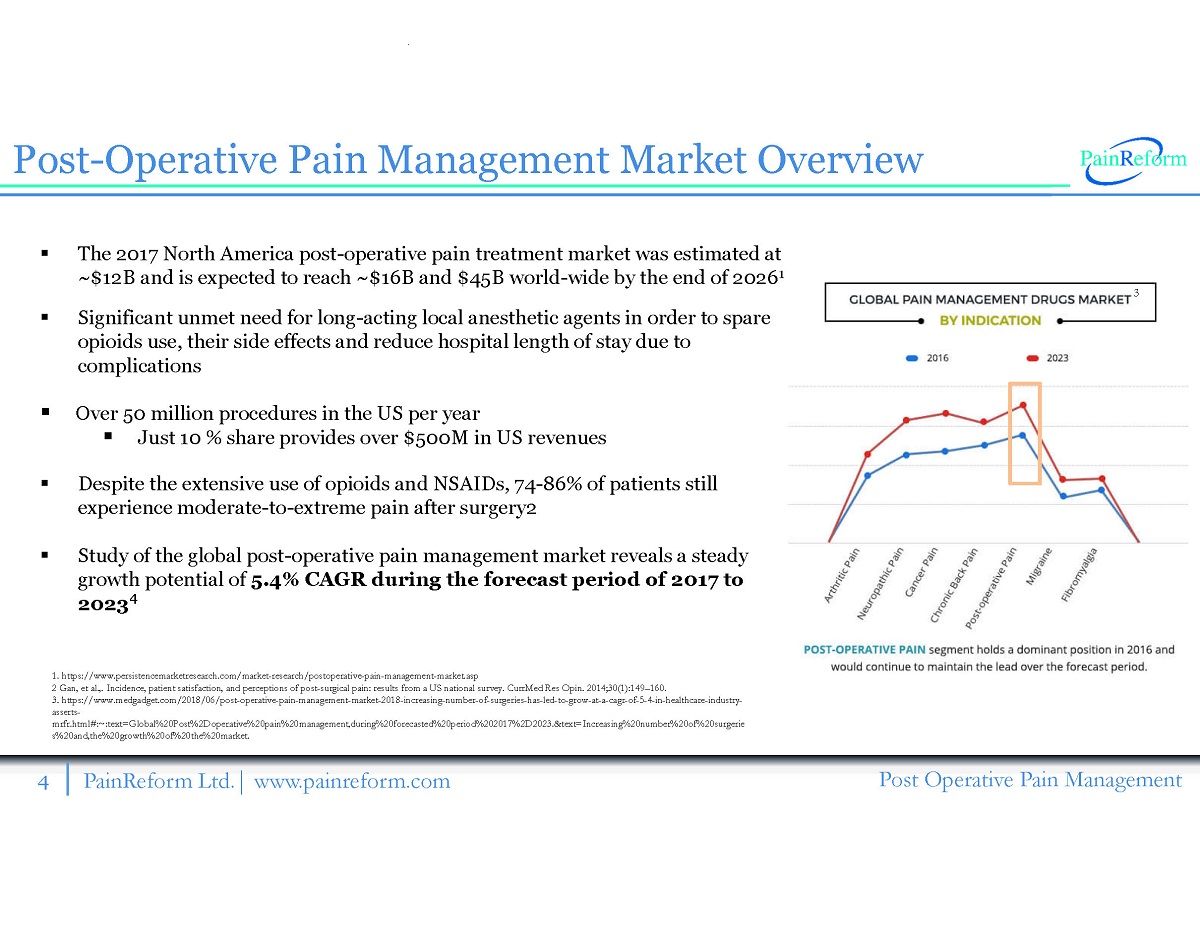
4 PainReform Ltd. | www.painreform.com Post Operative Pain Management Post - Operative Pain Management Market Overview ▪ The 2017 North America post - operative pain treatment market was estimated at ~$ 12 B and is expected to reach ~$ 16 B and $ 45 B world - wide by the end of 2026 1 ▪ Significant unmet need for long - acting local anesthetic agents in order to spare opioids use, their side effects and reduce hospital length of stay due to complications ▪ Over 50 million procedures in the US per year ▪ Just 10 % share provides over $ 500 M in US revenues ▪ Despite the extensive use of opioids and NSAIDs, 74 - 86 % of patients still experience moderate - to - extreme pain after surgery 2 ▪ Study of the global post - operative pain management market reveals a steady growth potential of 5.4 % CAGR during the forecast period of 2017 to 2023 3 1. https://www.persistencemarketresearch.com/market - research/postoperative - pain - management - market.asp 2 Gan, et al.,. Incidence, patient satisfaction, and perceptions of post - surgical pain: results from a US national survey. CurrM ed Res Opin. 2014;30(1):149 – 160. 3. https://www.medgadget.com/2018/06/post - operative - pain - management - market - 2018 - increasing - number - of - surgeries - has - led - to - grow - at - a - cagr - of - 5 - 4 - in - healthcare - industry - asserts - mrfr.html#:~:text=Global%20Post%2Doperative%20pain%20management,during%20forecasted%20period%202017%2D2023.&text=Increasing%2 0nu mber%20of%20surgerie s%20and,the%20growth%20of%20the%20market.

5 PainReform Ltd. | www.painreform.com Post Operative Pain Management Current Approaches in Post - Operative Analgesia Are Lacking ▪ Poor efficacy in acute pain control ▪ Not intended for the treatment of post - operative pain Short - Acting Opioids ▪ Repeated dosing required ▪ Inconsistent pain control between doses ▪ Dependence risk increases with treatment duration Exparel ® ▪ Limited efficacy in acute pain control ▪ Liposomal bupivacaine ▪ Reduced postoperative opioid use ▪ Approved - revenues $500M ▪ Handling/delivery limitations Non - Steroidal Anti - inflammatory Drugs (NSAIDs) ▪ Moderate efficacy in acute pain control ▪ Repeated dosing required ▪ Inconsistent pain control between doses ▪ Significant safety issues, including bleeding, stroke, gastritis, renal toxicity Long - Acting Opioids Significant adverse effects including respiratory depression, sedation and postoperative nausea and vomiting Zynrelef ® ▪ Complex, high production - price ▪ Bupivacaine and Meloxicam leading to a black box in the label ▪ Approved, launched July, 2021

6 PainReform Ltd. | www.painreform.com Post Operative Pain Management Prescription Opioids ▪ Prescription opioids can be used to treat moderate - to - severe pain and are often prescribed following surgery or injury ▪ In recent years, there has been a dramatic increase in the acceptance and use of prescription Opioids ▪ Opioid's dependency can start within 3 days of initial use ▪ More than 191 million opioid prescriptions were dispensed to American patients in 2017 ▪ The most common drugs involved in prescription opioid overdose deaths include: ▪ Methadone ▪ Oxycodone (such as OxyContin®) ▪ Hydrocodone (such as Vicodin®) ▪ Anyone who takes prescription opioids runs the risk of becoming addicted to them. ▪ One in four patients receiving long - term opioid therapy in a primary care setting struggles with opioid addiction ▪ In 2016 and 2019 , approximately 11.5 million Americans reported misusing prescription opioids in the past year ▪ Taking too many prescription opioids can stop a person ’ s breathing — leading to death Source: CDC

7 PainReform Ltd. | www.painreform.com Post Operative Pain Management Problem: State of Opioid Crisis in America ▪ 99 % of surgical patients receive opioids to manage post - surgical pain 1 and released with opiate prescription ▪ Approximately 11.4 million people misused opioids in 2016 and 2019 . 886 K used heroin. 562 K misused both pain relievers and heroin ▪ 6 % to 10 % of surgical patients discharged with opiate prescription develop an opioid - dependency ▪ 75 % of patients who undergo surgery experience acute post - operative pain, which is often medium - high in severity ▪ A 2016 study which enrolled 799,449 patients, showed that reliance on opioid analgesics as the mainstay for perioperative pain management is still widespread ▪ 2.1 Million people have an opioid use disorder. 1.7 million people with a prescription pain reliever have a use disorder ▪ 62.6 % Of people listed pain as their main reason for opioid misuse where 36 % of people with an opioid problem received a prescription from a healthcare provider

8 PainReform Ltd. | www.painreform.com Post Operative Pain Management PainReform Solution: PRF - 110 Post Operative Pain Management ▪ PRF - 110 has the potential of reducing the consumption of opioids to manage post - surgical pain ▪ Studies have shown 48 hours of pain reduction in healthy volunteers and about 72 hours in a clinical setting ▪ PainReform has developed a platform formulation for extended release of drugs ▪ Avoiding multiple injection reduces delivery time and complexity, reduces the risk of hematoma ▪ Reliable PK and low Cmax ▪ Physical attributes provide ease of surgeon use ▪ No injections, thereby avoiding risk of inadvertent systemic administration ▪ Platform can be utilized for a wide - range of APIs to generate sustainable pipeline of new product applications ▪ Low variable costs allows for ease of manufacturing and production to meet high market demand ▪ Robust IP portfolio

9 PainReform Ltd. | www.painreform.com Post Operative Pain Management PRF - 110 Manufacturing ▪ Engaged Pharmaceutics International, Inc. (PII): ▪ US based contract manufacturing organization ( CMO ) ▪ Well experienced in sterile manufacturing ▪ PII is a premier, solutions - oriented, science driven CMO with over 25 years of proven success in providing high - quality dosage form development ▪ cGMP manufacturing services to the global biopharmaceutical industry ▪ Successfully developed a GMP manufacturing process for PRF - 110 ▪ Clinical trial material manufacturing scheduled for mid - August, 2022 ▪ Low COGS compared to current drug landscape
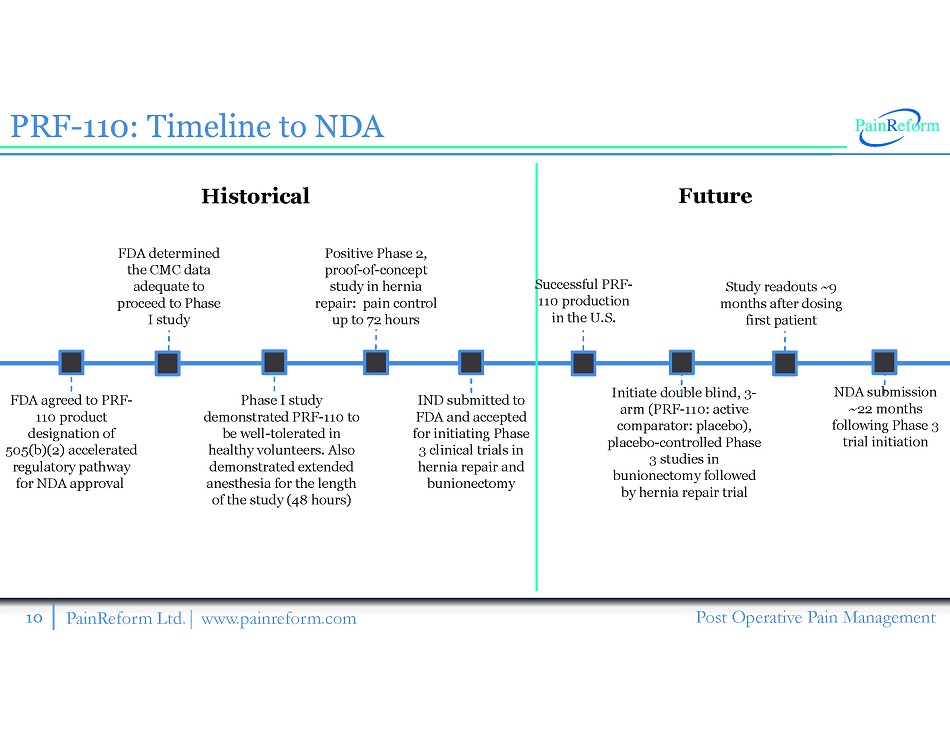
10 PainReform Ltd. | www.painreform.com Post Operative Pain Management PRF - 110 : Timeline to NDA FDA agreed to PRF - 110 product designation of 505 (b)( 2 ) accelerated regulatory pathway for NDA approval FDA determined the CMC data adequate to proceed to Phase I study Phase I study demonstrated PRF - 110 to be well - tolerated in healthy volunteers. Also demonstrated extended anesthesia for the length of the study ( 48 hours) IND submitted to FDA and accepted for initiating Phase 3 clinical trials in hernia repair and bunionectomy Study readouts ~9 months after dosing first patient Initiate double blind, 3 - arm (PRF - 110 : active comparator: placebo), placebo - controlled Phase 3 studies in bunionectomy followed by hernia repair trial Positive Phase 2, proof - of - concept study in hernia repair: pain control up to 72 hours NDA submission ~ 22 months following Phase 3 trial initiation Historical Future Successful PRF - 110 production in the U.S.
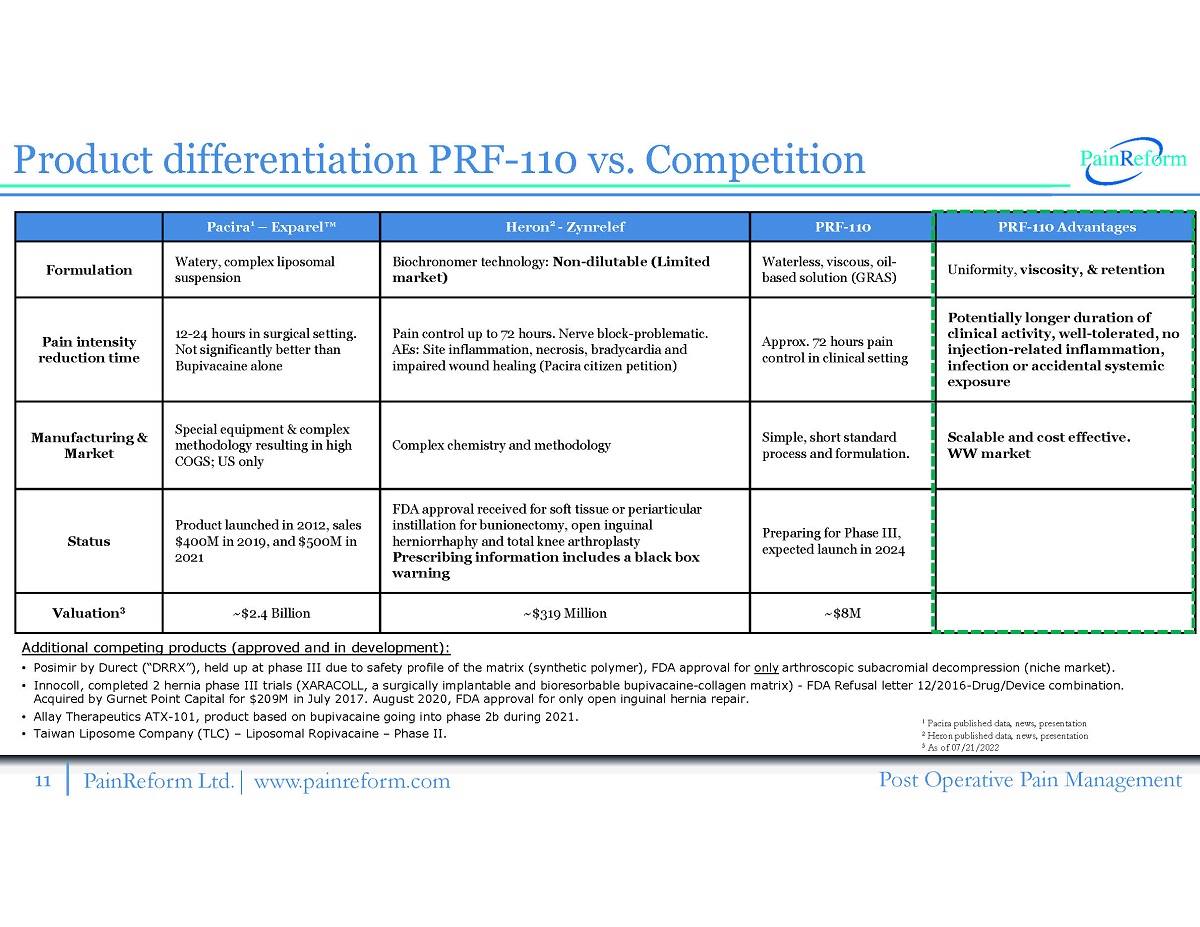
11 PainReform Ltd. | www.painreform.com Post Operative Pain Management Additional competing products (approved and in development): • Posimir by Durect ( “ DRRX ” ), held up at phase III due to safety profile of the matrix (synthetic polymer), FDA approval for only arthroscopic subacromial decompression (niche market). • Innocoll , completed 2 hernia phase III trials (XARACOLL, a surgically implantable and bioresorbable bupivacaine - collagen matrix) - FDA Refusal letter 12 / 2016 - Drug/Device combination. Acquired by Gurnet Point Capital for $ 209 M in July 2017 . August 2020 , FDA approval for only open inguinal hernia repair. • Allay Therapeutics ATX - 101 , product based on bupivacaine going into phase 2 b during 2021 . • Taiwan Liposome Company (TLC) – Liposomal Ropivacaine – Phase II. Pacira 1 – Exparel Œ Heron 2 - Zynrelef PRF - 110 PRF - 110 Advantages Formulation Watery, complex liposomal suspension Biochronomer technology: Non - dilutable (Limited market) Waterless, viscous, oil - based solution (GRAS) Uniform ity, viscosity , & retention Pain intensity reduction time 12 - 24 hours in surgical setting. Not significantly better than Bupivacaine alone Pain control up to 72 hours. Nerve block - problematic. AEs: Site inflammation, necrosis, bradycardia and impaired wound healing (Pacira citizen petition) Approx. 72 hours pain control in clinical setting Potentially longer duration of clinical activity, well - tolerated, no injection - related inflammation, infection or accidental systemic exposure Manufacturing & Market Special equipment & complex methodology resulting in high COGS; US only Complex chemistry and methodology Simple, short standard process and formulation. Scalable and cost effective. WW market Status Product launched in 2012, sales $400M in 2019, and $500M in 2021 FDA approval received for soft tissue or periarticular instillation for bunionectomy, open inguinal herniorrhaphy and total knee arthroplasty Prescribing information includes a black box warning Preparing for Phase III, expected launch in 2024 Valuation 3 ~$2.4 Billion ~$319 Million ~$8M Product differentiation PRF - 110 vs. Competition 1 Pacira published data, news, presentation 2 Heron published data, news, presentation 3 As of 07 / 21 / 2022

12 PainReform Ltd. | www.painreform.com Post Operative Pain Management PRF - 110 Safety ▪ PainReform carried out extensive FDA requested wound healing and related animal studies that showed: x PRF - 110 allows for normal wound healing of surgical incisions equal to both Naropin ® and saline without any untoward histological or radiologic ( microCT ) effects observed in soft or bony tissue x Tensile strength of healed surgical skin following exposure to PRF - 110 is equal to that of incisions exposed to either Naropin ® or saline x Integrity of surgical sutures and surgical meshes is not affected by PRF - 110 (compared to saline) x No systemic side effects observed in any models ▪ PRF - 110 safety in human trials showed no systemic, wound healing or scarring abnormalities . Wound healing in all patients was complete and similar to that expected in surgery without PRF - 110
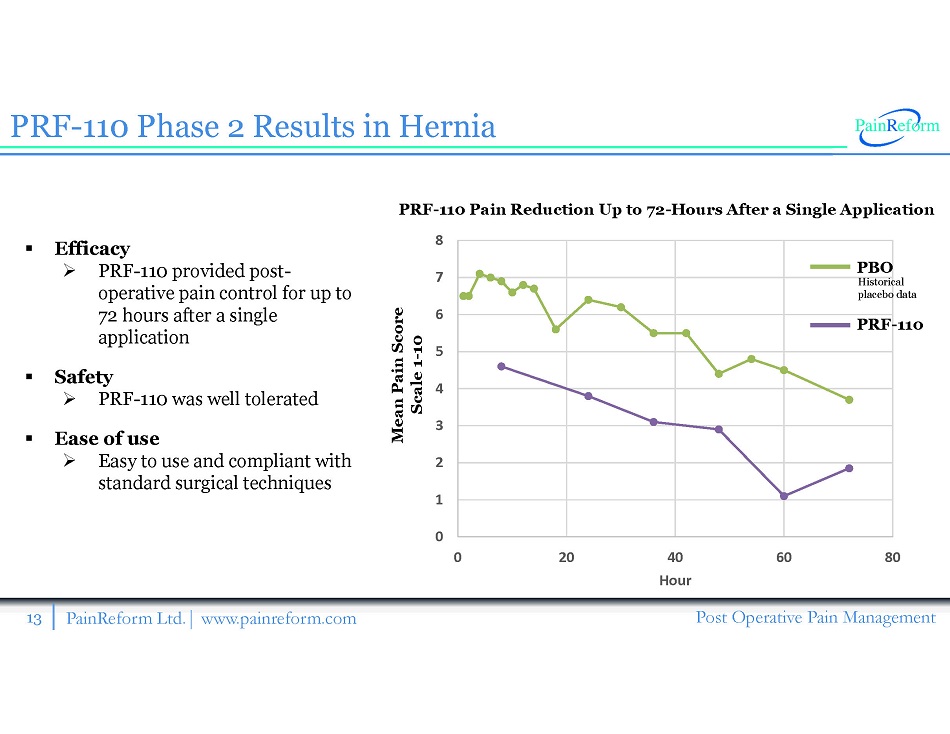
13 PainReform Ltd. | www.painreform.com Post Operative Pain Management PRF - 110 Phase 2 Results in Hernia ▪ Efficacy » PRF - 110 provided post - operative pain control for up to 72 hours after a single application ▪ Safety » PRF - 110 was well tolerated ▪ Ease of use » Easy to use and compliant with standard surgical techniques 0 1 2 3 4 5 6 7 8 0 20 40 60 80 Hour Mean Pain Score Scale 1 - 10 PRF - 110 Pain Reduction Up to 72 - Hours After a Single Application Historical placebo data PBO PRF - 110

14 PainReform Ltd. | www.painreform.com Post Operative Pain Management PRF - 110 : Phase III Clinical Trial Protocol ▪ Two, double blind, placebo control 72 - hour treatment period , studies planned (bunionectomy and hernia surgery). For each study: ▪ Three cohorts (n= ~ 400 ): PRF - 110 ; Naropin ® (ropivacaine); placebo; in a 2:2:1 ratio ▪ First patient in expected in Q 4 , 2022 Primary endpoint (Efficacy) • Compare the analgesic efficacy of PRF - 110 to placebo during the first 72 hours after completion of the surgery Secondary endpoints Objectives: • Efficacy: • C ompare the mean analgesic efficacy of PRF - 110 to that of plain Naropin ® during the first 72 hours after completion of bunionectomy or hernia surgery • Compare post - surgery opioid consumption through 72 hours for PRF - 110 to that of ropivacaine injection • Compare post - surgery opioid consumption (in morphine milligram equivalents) over 72 hours for PRF - 110 to that of placebo • Compare the proportion of subjects who were opioid free through 72 hours for PRF - 110 to that of plain ropivacaine • Safety Objective : • Evaluate the safety and tolerability of PRF - 110 in subjects undergoing bunionectomy or hernia surgery

15 PainReform Ltd. | www.painreform.com Post Operative Pain Management PRF - 110: Potential Best - In - Class Administration PRF - 110 viscosity and uniformity are highly suitable for standard surgical site administration. Efficacy Cross study comparison of Phase II data, 72 hours pain AUC Safety Met FDA required extensive pre - clinical studies to demonstrate no wound healing issues COGS Low cost of good sold allows a highly strategic pricing plan and considerations

16 PainReform Ltd. | www.painreform.com Post Operative Pain Management Highly experienced management Team Sigal Aviel, PhD, MBA, Chief Operating Officer ▪ Over 20 years of managerial experience in the Biotech industry. ▪ Former chief R&D officer at MediWound, a company specializing in deep burns and chronic wound care, where she was responsible fo r product development from early stages to final product approval by regulatory authorities. ▪ PhD in Immunology and Microbiology from Duke University Medical School as well as an executive MBA degree from the Kellogg schoo l of business at NW University Ehud Geller, PhD, MBA. Executive Chairman ▪ Former President & CEO of Interpharm Laboratories and EVP of Teva Group ▪ Former head of the Israeli Pharmaceutical Manufacturers Association and board member of the Tel Aviv Stock Exchange ▪ National Industry award for contribution to biotech industry and management leadership, Samuel Johnson Medal – Columbia ▪ Columbia University, Drexel Institute – Chemical Engineering (bio - chemical technology), MBA, PhD Eli Hazum, PhD, MBA, Chief Technical Officer ▪ Spent 5 years at Glaxo Inc. as Head of Department of Receptor Research and Metabolic Diseases ▪ Over last 25 years Eli has taken leadership roles within Medica portfolio companies including interim CEO for Collgard Biopharmaceuticals and Ester Neurosciences, where he was responsible for executing Ester’s acquisition by Amarin Pharmaceuticals. ▪ Received Ph.D. from the Weizmann Institute of Science in the field of hormone biochemistry, and has an executive MBA from Hum ber side University in the UK Ilan Hadar, MBA, Chief Executive Officer ▪ Former Country Manger and CFO of Foamix Pharmaceuticals Ltd. (now Nasdaq: VYNE) ▪ Over 20 years of multinational managerial and corporate experience with pharmaceutical and high - tech companies ▪ Has been instrumental in building companies from start - ups to hundreds of millions of dollars in operations ▪ Successfully took part in the development, approval, and launch of new pharmaceutical products in the U.S. and Israel ▪ Received his MBA in Finance and Business Entrepreneurship and B.A. degree at The Hebrew University in Jerusalem Rita Keynan, V.P. Pharma Operations ▪ Over 25 years of managerial experience in the pharmaceutical industry . ▪ Former e xecutive director of drug development at VYNE Therapeutics Ltd., formerly Foamix Pharmaceuticals, where she managed the drug developme nt department ▪ Mrs. Keynan is the co - inventor of over two dozen patents ▪ Mrs. Keynan holds a B.Sc. in Chemistry and a M.Sc. in Pharm from the Hebrew University in Jerusalem.
17 PainReform Ltd. | www.painreform.com Post Operative Pain Management Summary Large Unmet Need in Non - Opioid Postoperative Pain Management 1 1 Market Watch Post - Operative Pain Management Market Size Analysis 2019 Report Innovative Formulation / Delivery System Opioid Epidemic Presents Significant Commercial Opportunity Seasoned Management Team with Decades of Experience Robust US and International IP Portfolio 4 Core platform can be utilized for wide - range of APIs to generate sustainable pipeline 5 1 2 3

18 PainReform Ltd. | www.painreform.com Post Operative Pain Management Key Statistics NASDAQ: PRFX Current Price (July 15, 22): $0.8 Shares Outstanding (4/11/22): 10.5 M Market Cap: $8.4 M Fiscal Year End: December 31 Inside Beneficial Ownership: 34.3%

Thank You