
Investor Presentation January 2024 EXHIBIT 99.1

2 Disclaimer Forward-Looking Statements This presentation contains forward-looking statements within the meaning of Section 27A of the Securities Act of 1933, as amended, and Section 21E of the Securities Exchange Act of 1934, as amended, including statements regarding the future performance of 23andMe’s businesses in consumer genetics and therapeutics and the growth and potential of its proprietary research platform. All statements, other than statements of historical fact, included or incorporated in this presentation, including statements regarding 23andMe’s strategy, financial position, funding for continued operations, cash reserves, projected costs, plans, and objectives of management, are forward-looking statements. The words "believes," "anticipates," "estimates," "plans," "expects," "intends," "may," "could," "should," "potential," "likely," "projects," "continue," "will," “schedule,” and "would" or, in each case, their negative or other variations or comparable terminology, are intended to identify forward-looking statements, although not all forward-looking statements contain these identifying words. These forward-looking statements are predictions based on 23andMe’s current expectations and projections about future events and various assumptions. 23andMe cannot guarantee that it will actually achieve the plans, intentions, or expectations disclosed in its forward-looking statements and you should not place undue reliance on 23andMe’s forward-looking statements. The forward-looking statements contained herein are also subject generally to other risks and uncertainties that are described from time to time in the Company’s filings with the Securities and Exchange Commission, including under Item 1A, “Risk Factors” in the Company’s most recent Annual Report on Form 10-K, as filed with the Securities and Exchange Commission, and as revised and updated by our Quarterly Reports on Form 10-Q and Current Reports on Form 8-K. These forward-looking statements involve a number of risks, uncertainties (many of which are beyond the control of 23andMe), or other assumptions that may cause actual results or performance to be materially different from those expressed or implied by these forward-looking statements. Investors are cautioned not to place undue reliance on any such forward-looking statements, which speak only as of the date they are made. Except as required by law, 23andMe does not undertake any obligation to update or revise any forward-looking statements whether as a result of new information, future events, or otherwise. Use of Non-GAAP Financial Measures To supplement the 23andMe’s unaudited condensed consolidated statements of operations and unaudited condensed consolidated balance sheets, which are prepared in conformity with generally accepted accounting principles in the United States of America (“GAAP”), this presentation also includes references to Adjusted EBITDA, which is a non-GAAP financial measure that 23andMe defines as net income (loss) before net interest income (expense), net other income (expense), changes in fair value of warrant liabilities, income tax benefit, depreciation and amortization of fixed assets, amortization of internal use software, amortization of acquired intangible assets, goodwill and intangible assets impairment, non-cash stock-based compensation expense, acquisition-related costs, and expenses related to restructuring and other charges, if applicable, for the period. 23andMe has provided a reconciliation of net loss, the most directly comparable GAAP financial measure, to Adjusted EBITDA at the end of this presentation. Adjusted EBITDA is a key measure used by 23andMe’s management and the board of directors to understand and evaluate operating performance and trends, to prepare and approve 23andMe’s annual budget and to develop short- and long-term operating plans. 23andMe provides Adjusted EBITDA because 23andMe believes it is frequently used by analysts, investors and other interested parties to evaluate companies in its industry and it facilitates comparisons on a consistent basis across reporting periods. Further, 23andMe believes it is helpful in highlighting trends in its operating results because it excludes items that are not indicative of 23andMe’s core operating performance. In particular, 23andMe believes that the exclusion of the items eliminated in calculating Adjusted EBITDA provides useful measures for period-to-period comparisons of 23andMe’s business. Accordingly, 23andMe believes that Adjusted EBITDA provides useful information in understanding and evaluating operating results in the same manner as 23andMe’s management and board of directors. In evaluating Adjusted EBITDA, you should be aware that in the future 23andMe will incur expenses similar to the adjustments in this presentation. 23andMe’s presentation of Adjusted EBITDA should not be construed as an inference that future results will be unaffected by these expenses or any unusual or non-recurring items. Adjusted EBITDA should not be considered in isolation of, or as an alternative to, measures prepared in accordance with GAAP. Other companies, including companies in the same industry, may calculate similarly-titled non-GAAP financial measures differently or may use other measures to evaluate their performance, all of which could reduce the usefulness of Adjusted EBITDA as a tool for comparison. There are a number of limitations related to the use of these non-GAAP financial measures rather than net loss, which is the most directly comparable financial measure calculated in accordance with GAAP. Some of the limitations of Adjusted EBITDA include (i) Adjusted EBITDA does not properly reflect capital commitments to be paid in the future, and (ii) although depreciation and amortization are non-cash charges, the underlying assets may need to be replaced and Adjusted EBITDA does not reflect these capital expenditures. When evaluating 23andMe’s performance, you should consider Adjusted EBITDA alongside other financial performance measures, including net loss and other GAAP results. Intellectual Property All rights to the trademarks, copyrights, logos and other intellectual property listed herein belong to their respective owners 23andMe’s use thereof does not imply an affiliation with, or endorsement by the owners of such trademarks, copyrights, logos and other intellectual property. Solely for convenience, trademarks and trade names referred to in this Presentation may appear with the ® or ™ symbols, but such references are not intended to indicate, in any way, that such names and logos are trademarks or registered trademarks of 23andMe. Industry and Market Data This Presentation relies on and refers to certain information and statistics based on 23andMe’s management’s estimates, and/or obtained from third party sources which it believes to be reliable. 23andMe has not independently verified the accuracy or completeness of any such third party information. Copyright © 2024 23andMe, Inc.

To Help People Access, Understand, and Benefit from the Human Genome Mission

4Copyright © 2024 23andMe, Inc. Building Value with Three Distinct Business Verticals 1 / Consumer 2 / Research 3 / Therapeutics Personalized Health: genome, exome, lab (blood) work Telehealth & Telepharmacy Ancestry & DNA Relatives Recurring subscription revenue Worlds largest re-contactable genetic and phenotypic data engine Database licensing Target discovery Commercial and pharma services Genetics-informed targets, biologically validated Lead IO asset ‘610 enrolling phase 2A IND-ready IO asset with unique MOA Early-stage Immunology and Inflammation pipeline To achieve our three-part mission, we are executing across three different businesses
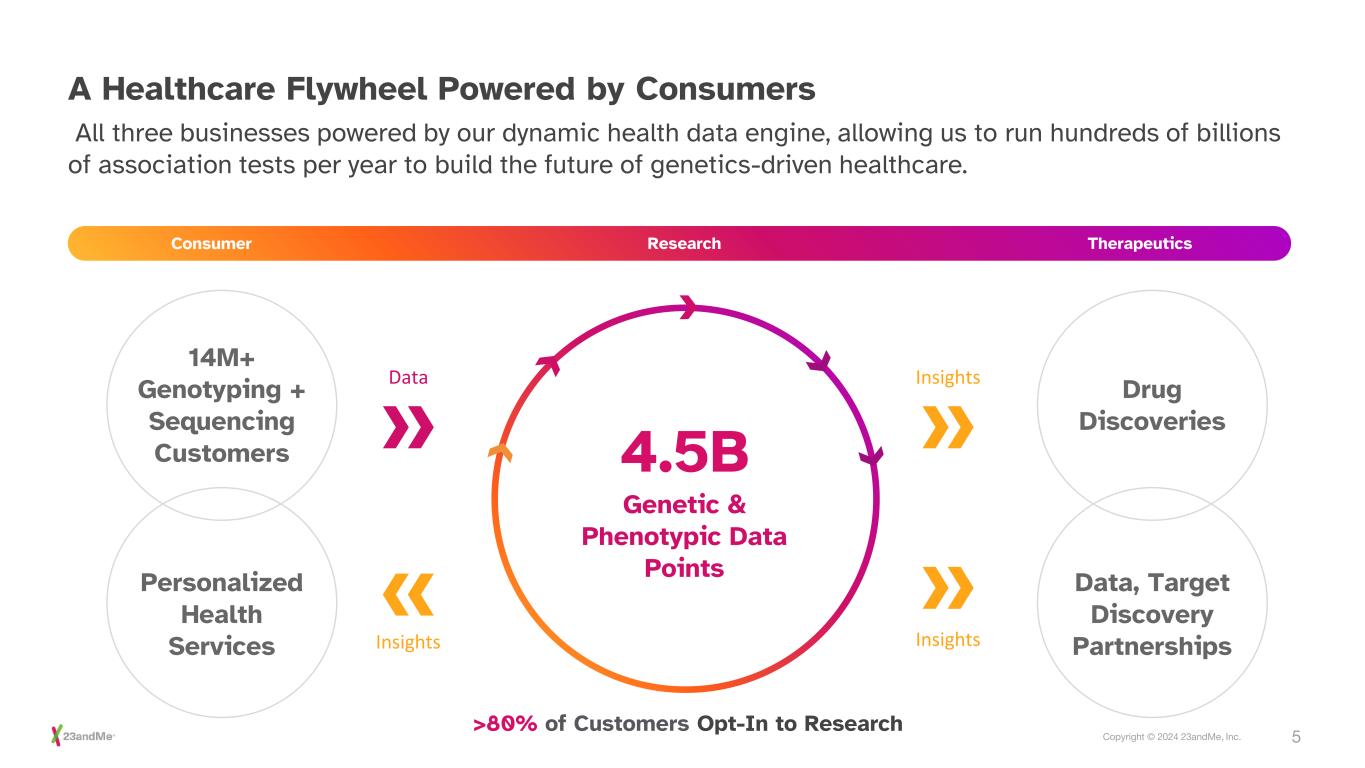
5Copyright © 2024 23andMe, Inc. A Healthcare Flywheel Powered by Consumers All three businesses powered by our dynamic health data engine, allowing us to run hundreds of billions of association tests per year to build the future of genetics-driven healthcare. Consumer Research Therapeutics Personalized Health Services Drug Discoveries Data, Target Discovery Partnerships 14M+ Genotyping + Sequencing Customers Data Insights Insights Insights 4.5B >80% of Customers Opt-In to Research Genetic & Phenotypic Data Points

6Copyright © 2024 23andMe, Inc. 14M+1 REGENERON ~2M+ OUR FUTURE HEALTH ~1M+ MILLION VETERAN PROGRAM 1M UK BIOBANK 500,000 DECODE GENETICS 500,000 FINNGEN 473,000+ ALL OF US 413,000+ The Scale of 23andMe Enables Impactful, Novel, Personalized Health With our growing database, we are uniquely positioned to understand human biology across areas of consumer health, research and therapeutics unlike any other genetics program globally. 1 Genotyped customers as of September 30, 2023.

Consumer Transforming Healthcare with Genetic Health Services at Scale 1

8Copyright © 2024 23andMe, Inc. Genome Sequencing Exome Sequencing Lab work (blood) Ancestry & DNA Relatives Recurring subscription revenue Telehealth Telepharmacy Medical Team & Online Pharmacy licensed in all 50 states Recurring subscription revenue Building Our Direct-to-Consumer Services In 2021, 23andMe acquired Lemonaid Health to build a new kind of care: access to Genetics-Informed Clinical Care. +

9Copyright © 2024 23andMe, Inc. Delivering Value with Our Direct-to-Consumer Product Line-up Dynamic data engine allows us to continually improve and expand product offerings. Product prices as of 12/31/23.

10Copyright © 2024 23andMe, Inc. U.S. Leading Causes of Death Genetics plays a role in 9 of the 10 leading causes of death in the US1 ● = Addressed by 23andMe genetic report • Heart disease • Cancer • COVID-19 • Accidents (unintentional injuries) • Stroke (cerebrovascular diseases) • Chronic lower respiratory diseases • Alzheimer’s disease • Diabetes • Chronic liver disease and cirrhosis • Nephritis, nephrotic syndrome, and nephrosis 1 Mortality in the US, 2021 - https://blogs.cdc.gov/genomics/2014/05/15/geography/
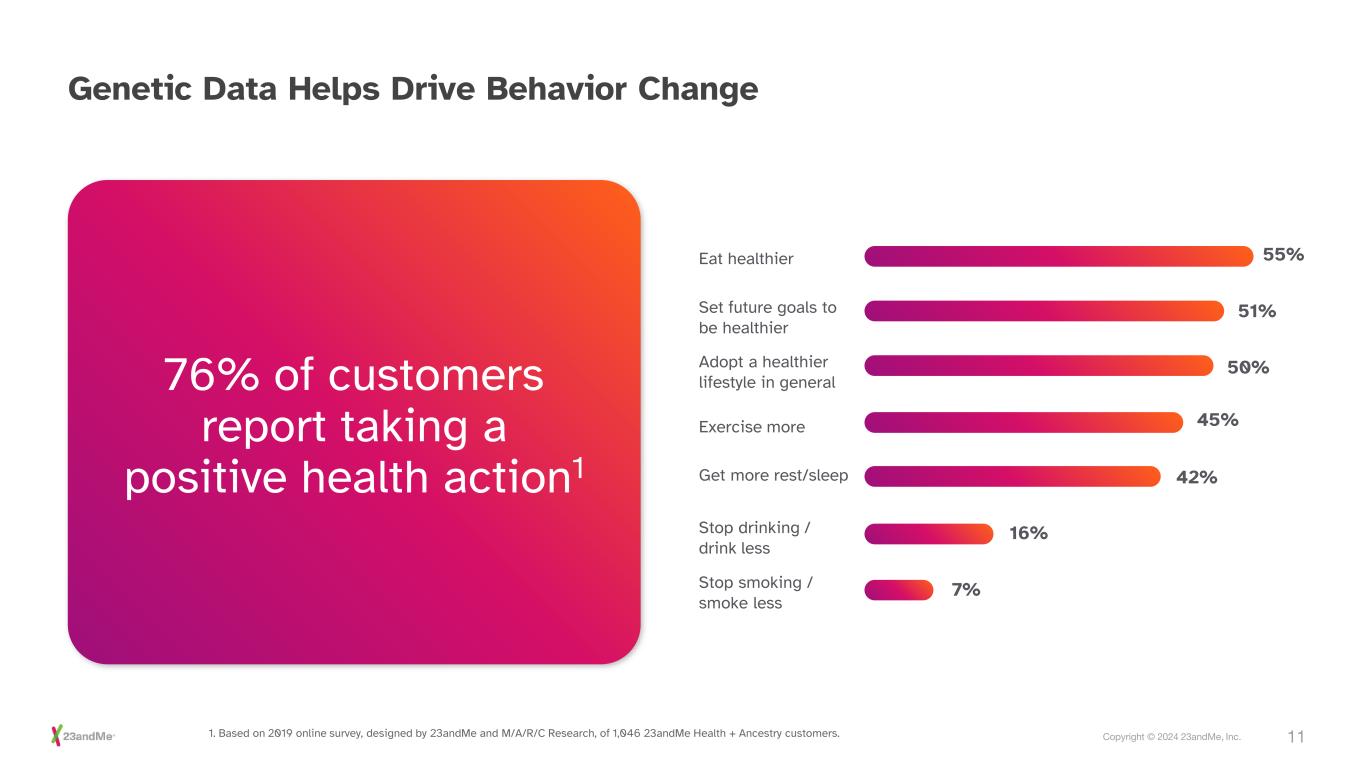
11Copyright © 2024 23andMe, Inc. Genetic Data Helps Drive Behavior Change Eat healthier Set future goals to be healthier Adopt a healthier lifestyle in general Exercise more Get more rest/sleep Stop drinking / drink less Stop smoking / smoke less 55% 51% 50% 45% 42% 16% 7% 1. Based on 2019 online survey, designed by 23andMe and M/A/R/C Research, of 1,046 23andMe Health + Ancestry customers. 76% of customers report taking a positive health action1

12Copyright © 2024 23andMe, Inc. Genetic Information Impacts Health and Clinical Outcomes Coronary Artery Disease APOL1 And CKD ● Increased initiation of lipid-lowering therapy in those with high vs. low CAD PRS (15% vs 6% statin initiation) ● Earlier initiation of lipid-lowering therapy in those with high vs. low CAD PRS (52 vs 65 years) Communication of CAD PRS through a digital app led to: Disclosure of APOL1 genetic results1 to African descent patients with hypertension (but no CKD) and to their primary care providers led to: ● Greater reduction in systolic blood pressure ● Increased guideline-appropriate kidney function testing ● Positive self-reported behavior changes Muse ED, et al. (2022). Impact of polygenic risk communication: an observational mobile application-based coronary artery disease study. NPJ Digit Med 5(1):30 Nadkarni GN, et al. (2022). Effects of Testing and Disclosing Ancestry-Specific Genetic Risk for Kidney Failure on Patients and Health Care Professionals: A Randomized Clinical Trial. JAMA Netw Open. 2022;5(3):e221048. EXAMPLE EXAMPLE
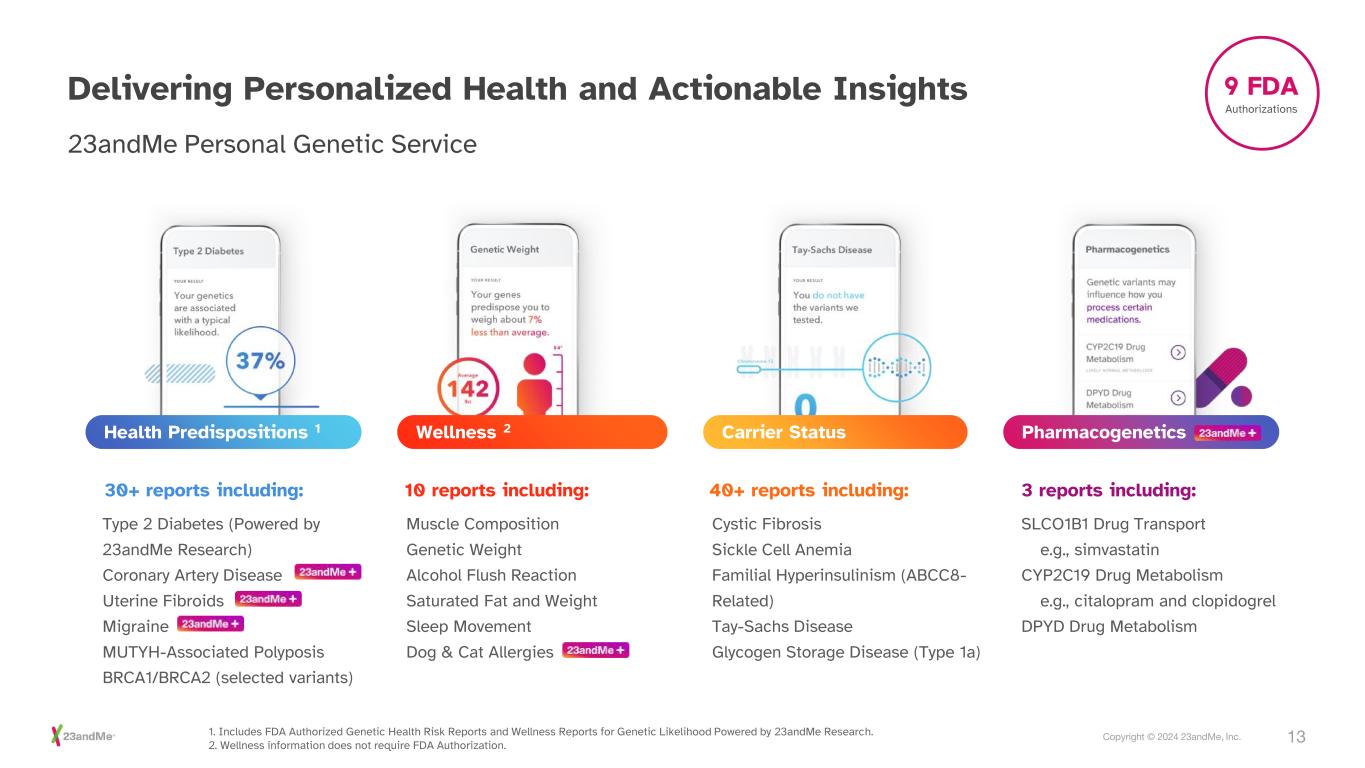
13Copyright © 2024 23andMe, Inc. Delivering Personalized Health and Actionable Insights 23andMe Personal Genetic Service Pharmacogenetics Carrier StatusWellness 2Health Predispositions 1 30+ reports including: 10 reports including: 40+ reports including: 3 reports including: Type 2 Diabetes (Powered by 23andMe Research) Coronary Artery Disease Uterine Fibroids Migraine MUTYH-Associated Polyposis BRCA1/BRCA2 (selected variants) Muscle Composition Genetic Weight Alcohol Flush Reaction Saturated Fat and Weight Sleep Movement Dog & Cat Allergies Cystic Fibrosis Sickle Cell Anemia Familial Hyperinsulinism (ABCC8- Related) Tay-Sachs Disease Glycogen Storage Disease (Type 1a) SLCO1B1 Drug Transport e.g., simvastatin CYP2C19 Drug Metabolism e.g., citalopram and clopidogrel DPYD Drug Metabolism 1. Includes FDA Authorized Genetic Health Risk Reports and Wellness Reports for Genetic Likelihood Powered by 23andMe Research. 2. Wellness information does not require FDA Authorization. 9 FDA Authorizations

14 Copyright © 2024 23andMe, Inc. New: 23andMe Total HealthTM Our new, premium subscription service: advanced, comprehensive sequencing for $1,188/year ($99/month). Next-Generation Sequencing Detects 200x more hereditary disease-causing variants than our personal genome service reports ‡. Screens for 55+ clinically actionable and under-diagnosed conditions. Clinical-grade genetic analysis. Access to clinicians with training in genetics-based care Annual virtual session with a clinician with ongoing conversations about reports, progress or questions. Bi-annual Blood Testing Track results, optimize and measure progress beyond routine labs. Access thyroid, kidney, heart health and more with biomarkers such as Lipoprotein(a) (Lp(a)) and Apolipoprotein B(ApoB). 23andMe+ Premium Service Includes an additional 190+ personalized genotyping reports with ongoing new reports and features delivered throughout the year. ‡ Our genotyping product detects 250 health-related variants in our Carrier Status and Genetic Health Risk reports. The Exome Sequencing reports detect 50,000+ hereditary disease-causing variants.
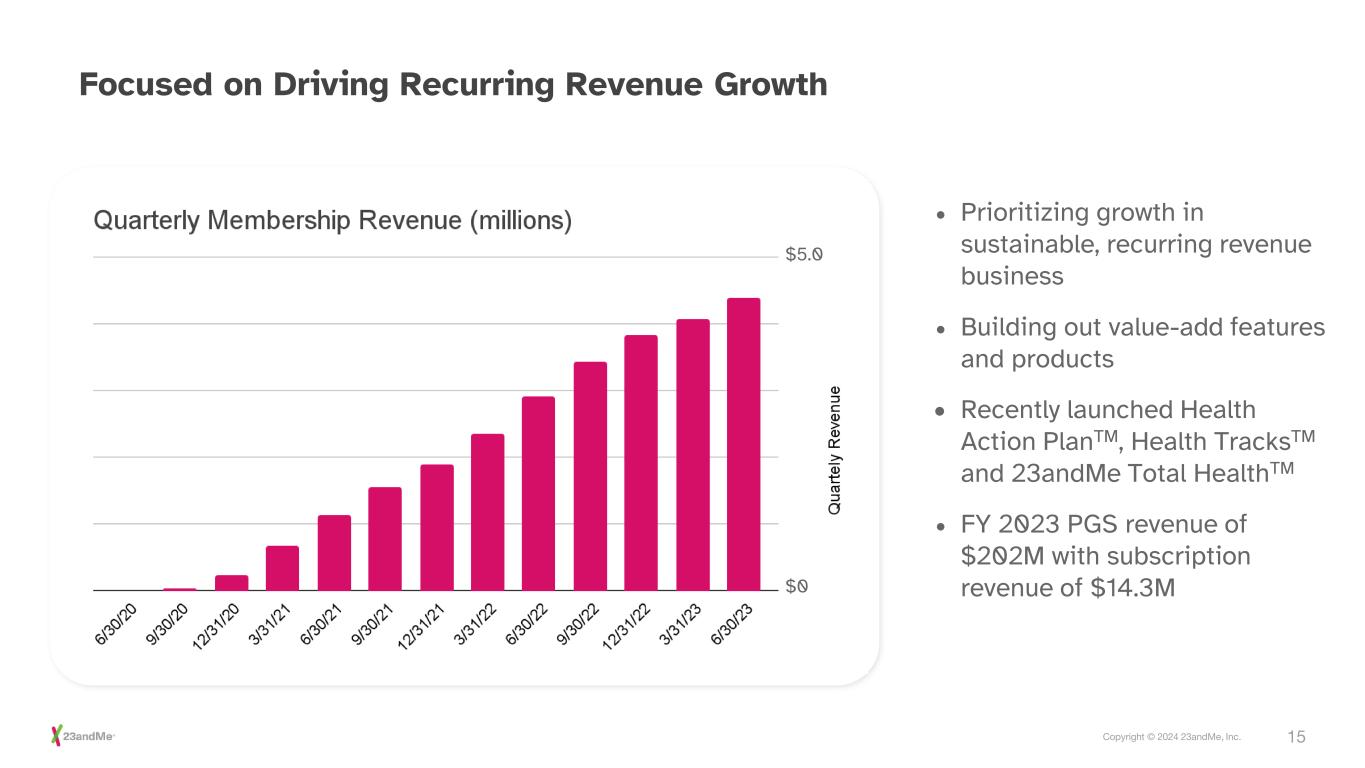
15Copyright © 2024 23andMe, Inc. • Prioritizing growth in sustainable, recurring revenue business • Building out value-add features and products • Recently launched Health Action PlanTM, Health TracksTM and 23andMe Total HealthTM • FY 2023 PGS revenue of $202M with subscription revenue of $14.3M Focused on Driving Recurring Revenue Growth $5.0 $0

16Copyright © 2024 23andMe, Inc. • Focus on improving Gross Margin • Margin tailwinds from increasing subscription revenue and price optimization • Strong new product uptake would further positively impact consolidated GM over time Steadily Improving Consumer Gross Margin Profile

17Copyright © 2024 23andMe, Inc. Future of 23andMe Fully Integrated Genetics-Informed Clinical Care Genetic Health Evaluation Telehealth & Telepharmacy Services Lab Tests Precision Prescribing Using Pharmacogenetics Long-term Engagement All connected within a single technology platform.

2 Research Providing Unique Value and Insights for Research Partners

19Copyright © 2024 23andMe, Inc. The World’s Largest Recontactable Genetic Data Engine >80% consent to research >14M1 >4.5B1 customers datapoints • Participation is online • Fully opt-in, and opt-out at any time • IRB approved • Everyone can be included in multiple studies 1 as of September 30, 2023. 1

20Copyright © 2024 23andMe, Inc. Scale Enables Differentiated Research Across Multiple Disease Areas 1 23andMe multi-ancestry meta-analysis GWAS as of October 2023 Phenotype Number of Cases1 Asthma 1.1M Autoimmune Lupus Multiple Sclerosis Type 1 Diabetes 58k 31.5k 38.5k Solid Tumors > 1M Basal Cell Squamous Cell Melanoma Breast 388k 214k 125k 120k Hematologic Cancers NHL Leukemia 17k 14k Phenotype Number of Cases1 Retinal Diseases AMD Glaucoma 106k 186k Rare Diseases Scleroderma/SSc Sarcoidosis Idiopathic Pulmonary Fibrosis 12k 9.3k 5k Neurology + Psychiatry Depression Parkinson’s Essential Tremor 1.8M 33.5k 47k Numbers represent the number of research participants with the condition indicated
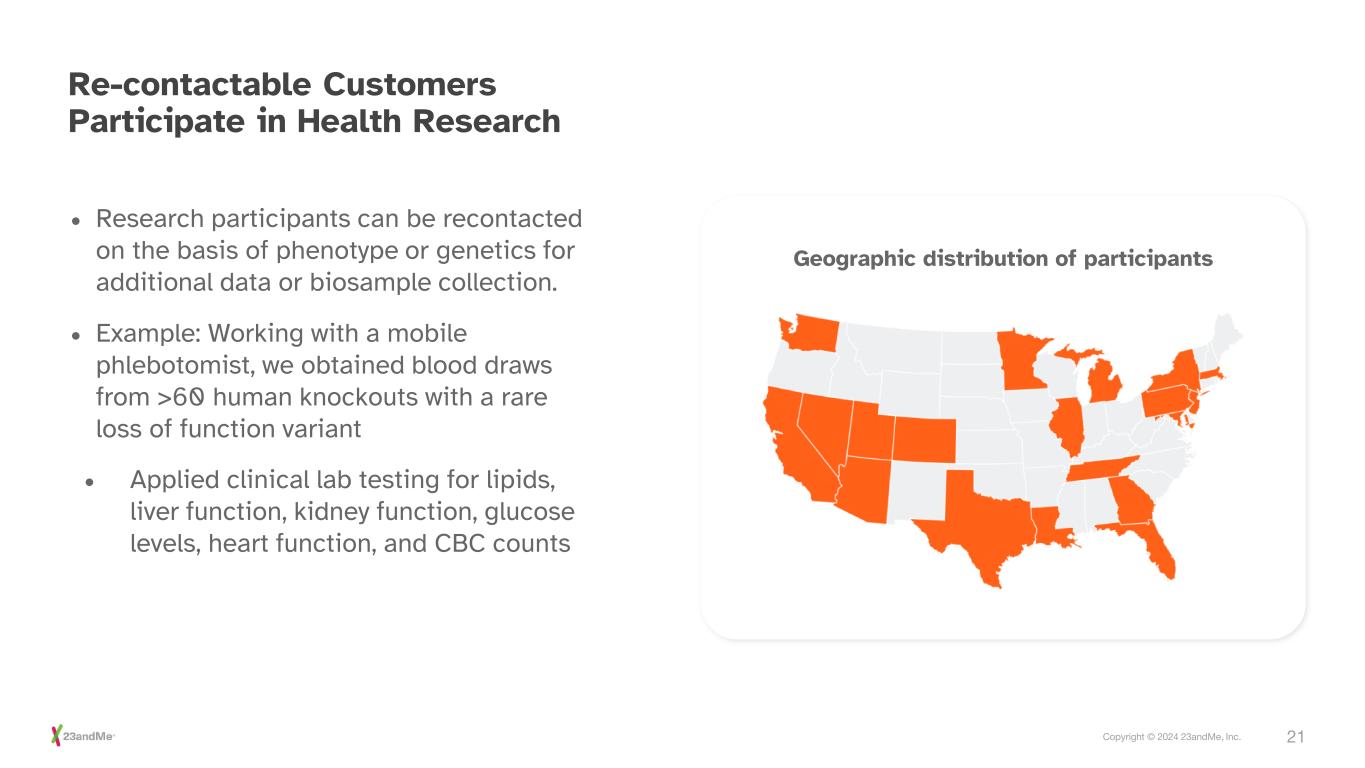
21Copyright © 2024 23andMe, Inc. Re-contactable Customers Participate in Health Research • Research participants can be recontacted on the basis of phenotype or genetics for additional data or biosample collection. • Example: Working with a mobile phlebotomist, we obtained blood draws from >60 human knockouts with a rare loss of function variant • Applied clinical lab testing for lipids, liver function, kidney function, glucose levels, heart function, and CBC counts Geographic distribution of participants
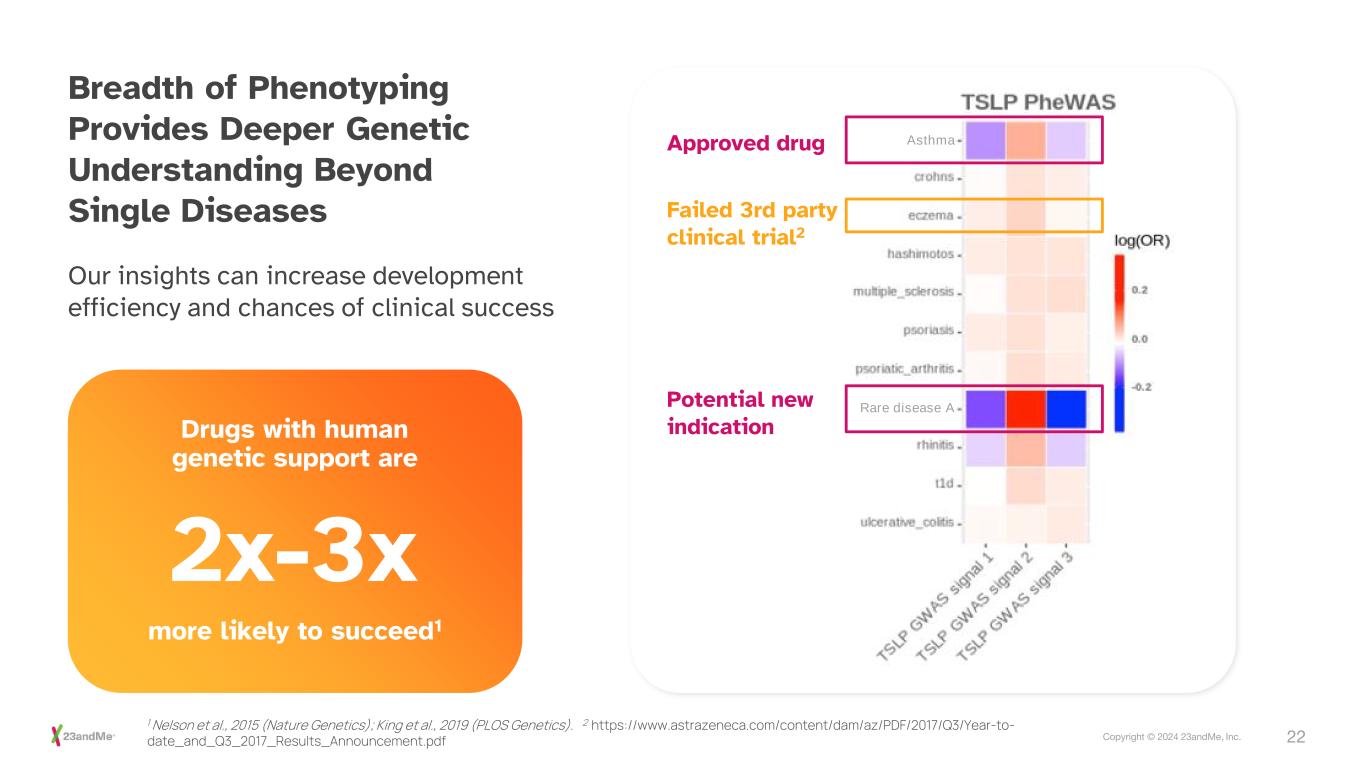
22Copyright © 2024 23andMe, Inc. Breadth of Phenotyping Provides Deeper Genetic Understanding Beyond Single Diseases Rare disease A AsthmaApproved drug Failed 3rd party clinical trial2 Potential new indication Our insights can increase development efficiency and chances of clinical success Drugs with human genetic support are more likely to succeed1 2x-3x 1 Nelson et al., 2015 (Nature Genetics); King et al., 2019 (PLOS Genetics). 2 https://www.astrazeneca.com/content/dam/az/PDF/2017/Q3/Year-to- date_and_Q3_2017_Results_Announcement.pdf
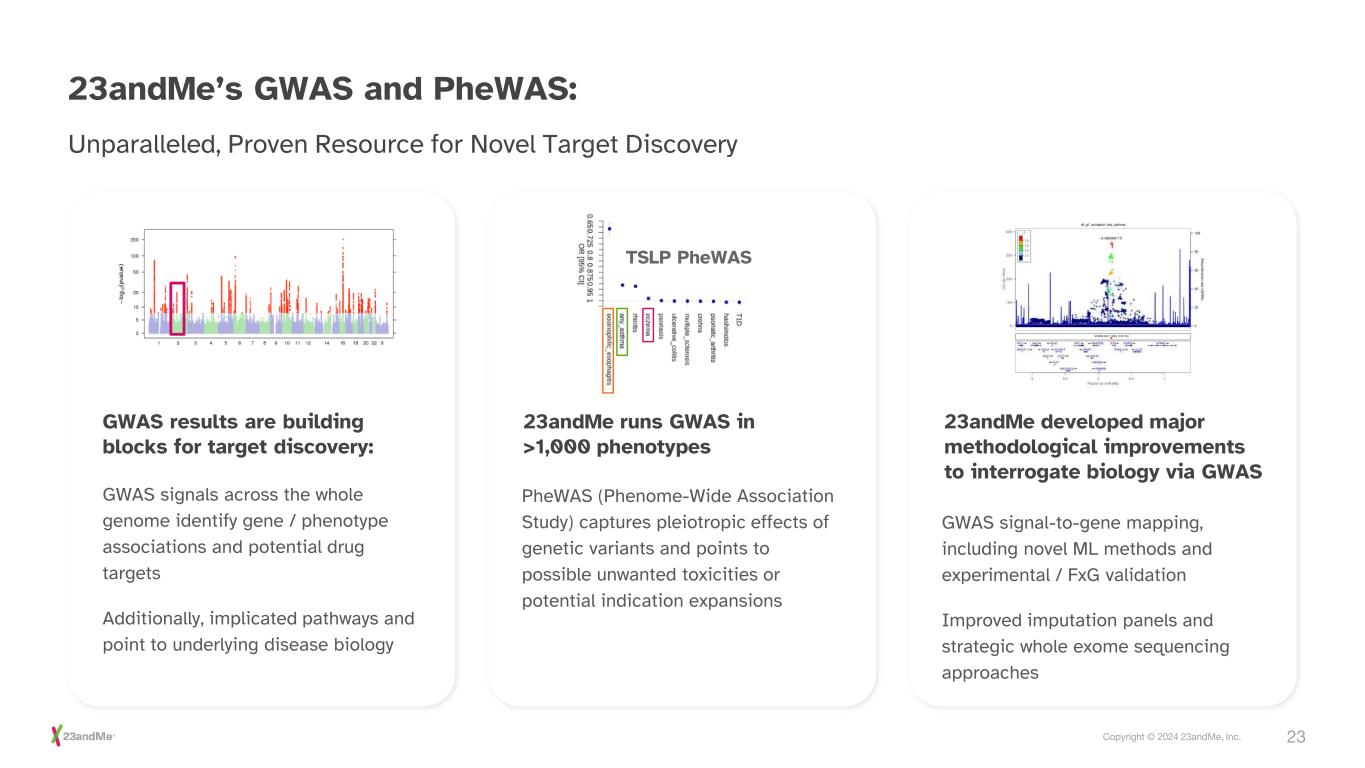
23Copyright © 2024 23andMe, Inc. 23andMe’s GWAS and PheWAS: GWAS results are building blocks for target discovery: 23andMe runs GWAS in >1,000 phenotypes 23andMe developed major methodological improvements to interrogate biology via GWAS GWAS signals across the whole genome identify gene / phenotype associations and potential drug targets Additionally, implicated pathways and point to underlying disease biology PheWAS (Phenome-Wide Association Study) captures pleiotropic effects of genetic variants and points to possible unwanted toxicities or potential indication expansions GWAS signal-to-gene mapping, including novel ML methods and experimental / FxG validation Improved imputation panels and strategic whole exome sequencing approaches Unparalleled, Proven Resource for Novel Target Discovery TSLP PheWAS

24Copyright © 2024 23andMe, Inc. A New Paradigm for 23andMe Research: 2017 Mid-2023 Late-2023 Future Exclusive drug discovery and development collaboration with GlaxoSmithKline (GSK) ● $25-50M annual contract fee ● Co-development of targets ● Over 50 targets discovered ● Limited 23andMe control of costs ● Resource intensive ● Difficult to forecast due to cost sharing Non-exclusive research collaborations ● Database access, focused target discovery, portfolio optimization ● Full 23andMe control of costs ● Deal specific resource scaling ● Higher margin ● Easy to forecast ● Ex: GSK -$20M/yr database access
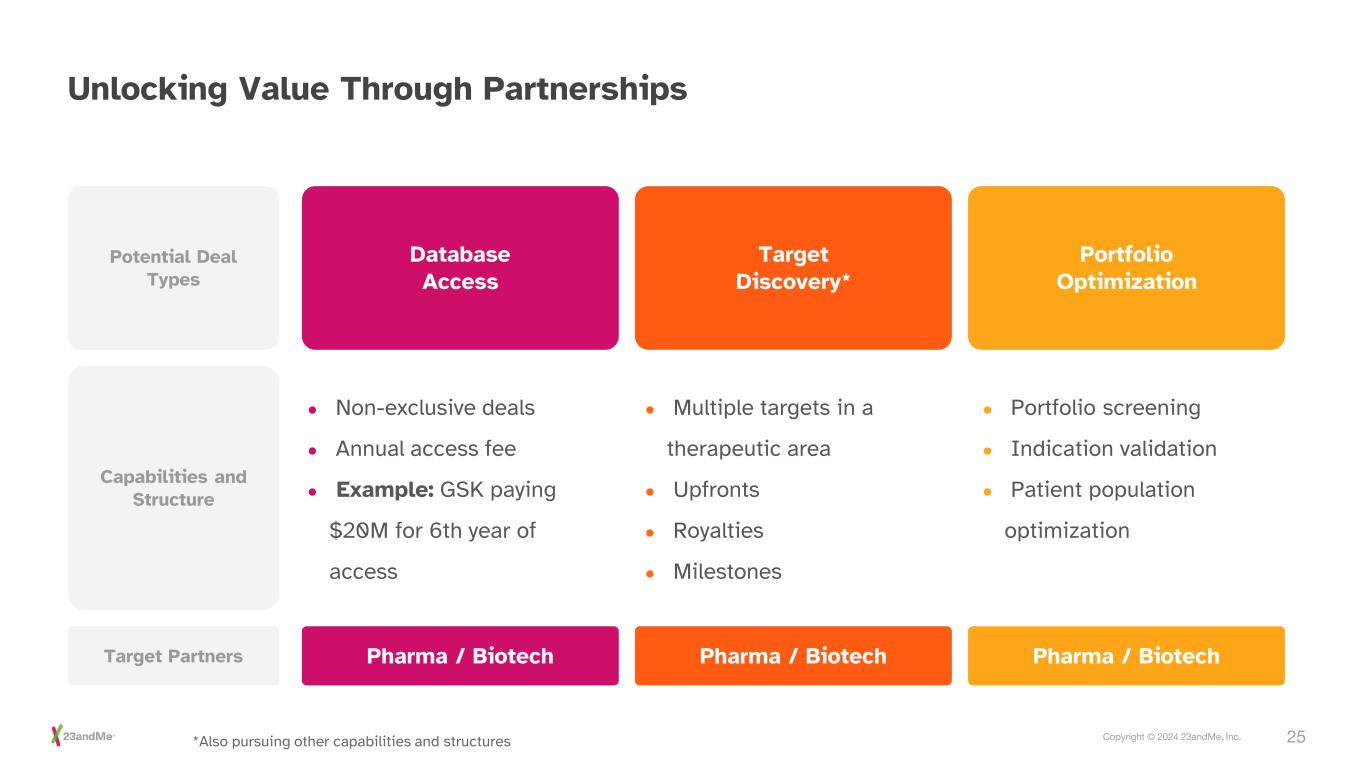
25Copyright © 2024 23andMe, Inc. Unlocking Value Through Partnerships Potential Deal Types Database Access Target Discovery* Portfolio Optimization Target Partners Pharma / Biotech Pharma / Biotech Pharma / Biotech ● Non-exclusive deals ● Annual access fee ● Example: GSK paying $20M for 6th year of access ● Multiple targets in a therapeutic area ● Upfronts ● Royalties ● Milestones ● Portfolio screening ● Indication validation ● Patient population optimization Capabilities and Structure *Also pursuing other capabilities and structures

3 Therapeutics Turning Data at Scale into New Treatments for Patients

27Copyright © 2024 23andMe, Inc. The Evolution of 23andMe Therapeutics Full-fledged Biotech Multiple programs identified to be brought forward independently Incredibly productive multi- modality drug discovery collaboration with GSK across many therapeutic areas Two novel, clinical stage Oncology antibody assets Discovery focus on Immunology and Inflammation In-silico target discovery, functional genomics, antibody design and wet-lab validation 2015 Today August 2023 - Today GSK Collaboration July 2018 - July 2023 23andMe Tx Began 2015 50+ programs

28Copyright © 2024 23andMe, Inc. Our Therapeutics Discovery Platform Capitalizing on 23andMe’s Capabilities & Genetic Advantage NEED Target areas with defined unmet medical need, creatively use our database Immunology & Inflammation SPEED Prioritizing speed to IND & Clinical PoC POWER Best Targets: Robust & unique GWAS / pleiotropy, world leading genetics capabilities
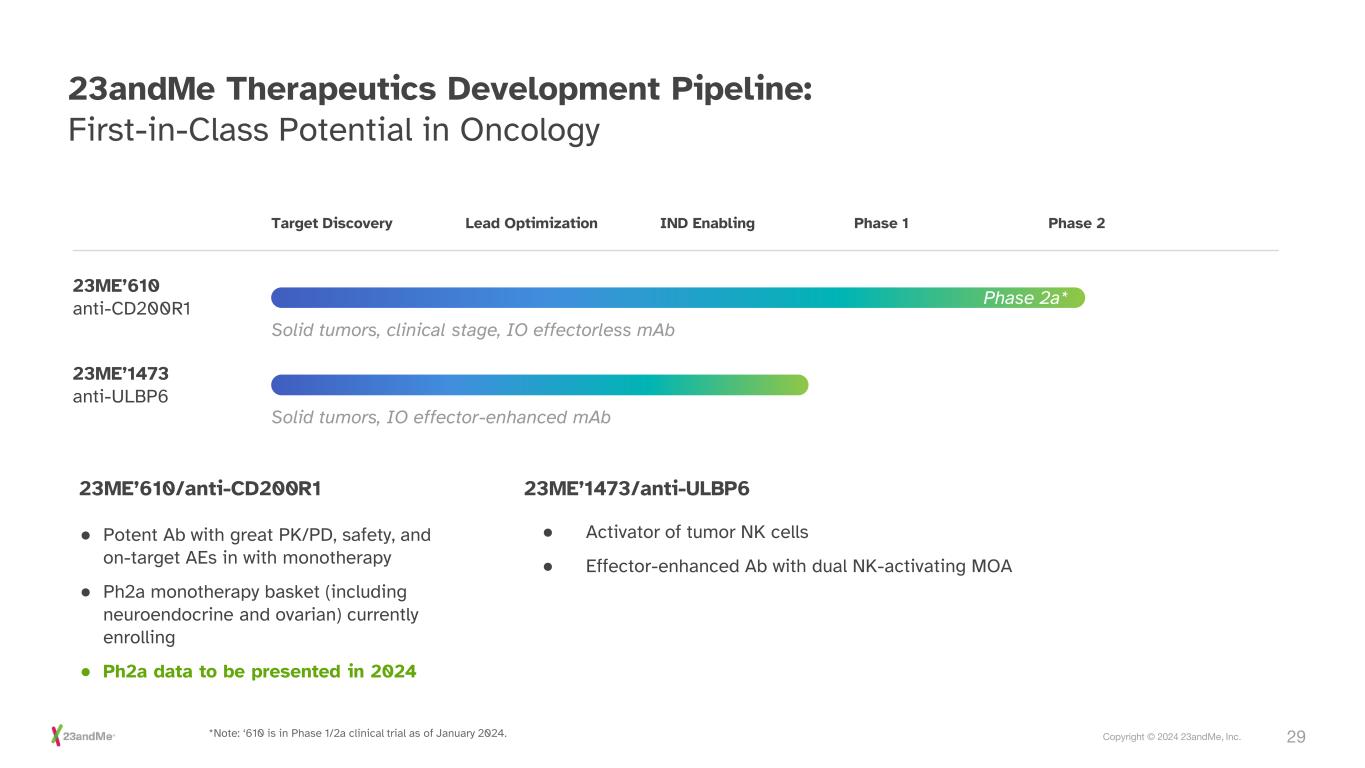
29Copyright © 2024 23andMe, Inc. 23andMe Therapeutics Development Pipeline: First-in-Class Potential in Oncology Target Discovery 23ME’610 anti-CD200R1 Solid tumors, clinical stage, IO effectorless mAb Solid tumors, IO effector-enhanced mAb 23ME’1473 anti-ULBP6 Lead Optimization IND Enabling Phase 1 Phase 2 Phase 2a* ● Potent Ab with great PK/PD, safety, and on-target AEs in with monotherapy ● Ph2a monotherapy basket (including neuroendocrine and ovarian) currently enrolling ● Ph2a data to be presented in 2024 23ME’610/anti-CD200R1 ● Activator of tumor NK cells ● Effector-enhanced Ab with dual NK-activating MOA 23ME’1473/anti-ULBP6 *Note: ‘610 is in Phase 1/2a clinical trial as of January 2024.

30Copyright © 2024 23andMe, Inc. 23ME’610: Geno-Phenotypic Data Unveils Novel Immune Processes that Bear Out in the Clinic Rasco, D, et al., 2023, SITC Annual Meeting #619 Geno-Phenotypic Data Translates to Safety and Efficacy Signals in the Clinic Genetic data tracks AE profile observed in clinic (Ph 1/2a) with anti-CD200R1 Preliminary Monotx POC: 58% size reduction in longest dimension of paratracheal lesion IO-naive patient with pancreatic neuroendocrine cancer Disease-modifying potential as IO monotherapy across a broad spectrum of “cold” neoplasms (e.g., neuroendocrine, ovarian) Investigator-assessed immune-related adverse events seen in >5% of patients in cohort
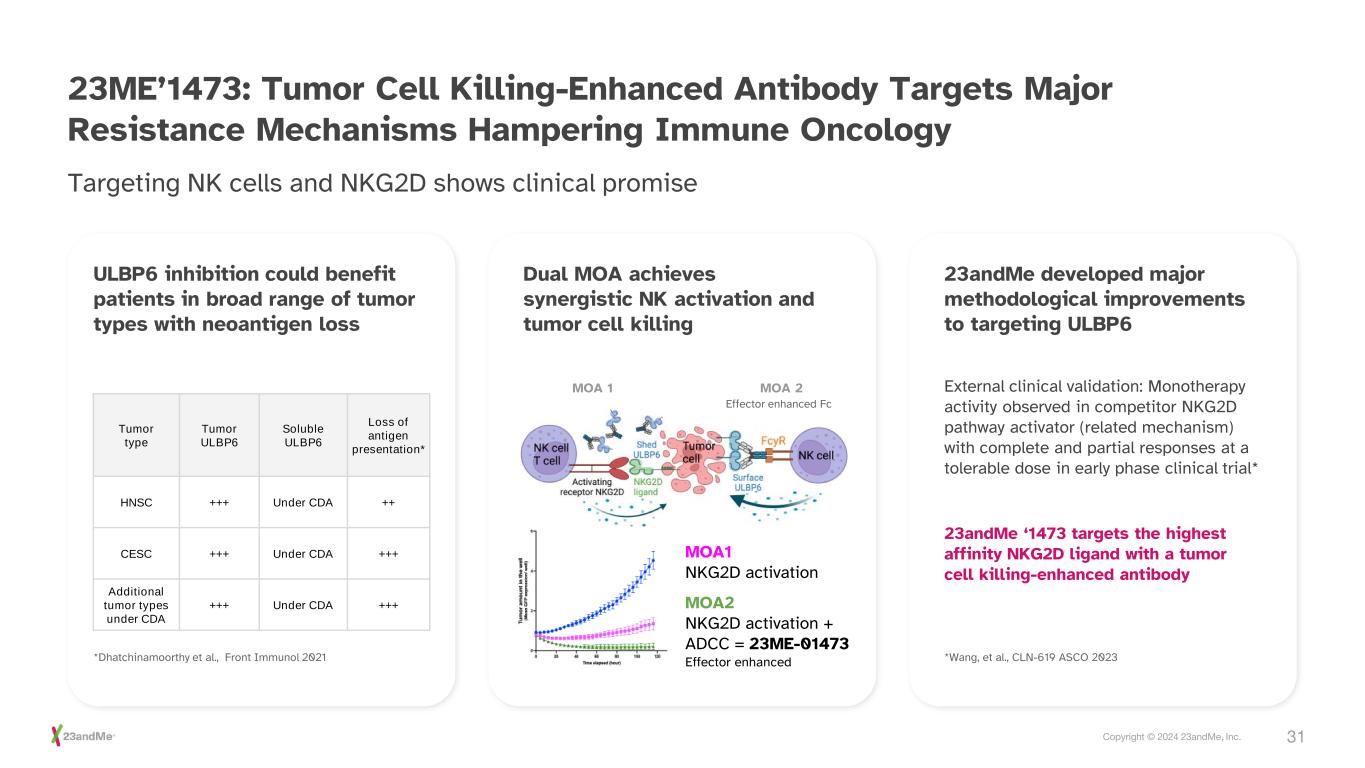
31Copyright © 2024 23andMe, Inc. 23ME’1473: Tumor Cell Killing-Enhanced Antibody Targets Major Resistance Mechanisms Hampering Immune Oncology Targeting NK cells and NKG2D shows clinical promise *Wang, et al., CLN-619 ASCO 2023 ULBP6 inhibition could benefit patients in broad range of tumor types with neoantigen loss Dual MOA achieves synergistic NK activation and tumor cell killing 23andMe developed major methodological improvements to targeting ULBP6 Tumor type Tumor ULBP6 Soluble ULBP6 Loss of antigen presentation* HNSC +++ Under CDA ++ CESC +++ Under CDA +++ Additional tumor types under CDA +++ Under CDA +++ *Dhatchinamoorthy et al., Front Immunol 2021 MOA1 NKG2D activation MOA2 NKG2D activation + ADCC = 23ME-01473 Effector enhanced MOA 1 MOA 2 Effector enhanced Fc External clinical validation: Monotherapy activity observed in competitor NKG2D pathway activator (related mechanism) with complete and partial responses at a tolerable dose in early phase clinical trial* 23andMe ‘1473 targets the highest affinity NKG2D ligand with a tumor cell killing-enhanced antibody

32Copyright © 2024 23andMe, Inc. For More Detailed Information on 23andMe Therapeutics: and visit our Investors page to view our full Therapeutics investor deck https://investors.23andme.com/news-events/events-presentations www.Therapeutics.23andMe.com

4 Financials

34Copyright © 2024 23andMe, Inc. Solving for Fiscally Responsible Future Growth Investing in future growth potential ● Subscription Services ● New reports and insights ● Research partnerships ● Therapeutics Employing a conservative approach to planning ● Prioritizing the minimization of Adjusted EBITDA deficit rather than maximizing top-line growth in our Consumer business (PGS and telehealth). Investing in future growth potential ● Cash of $256 million1 supports 23andMe’s plans for targeted investment in high ROI growth initiatives. 1 2 3 1 As of September 30, 2023.
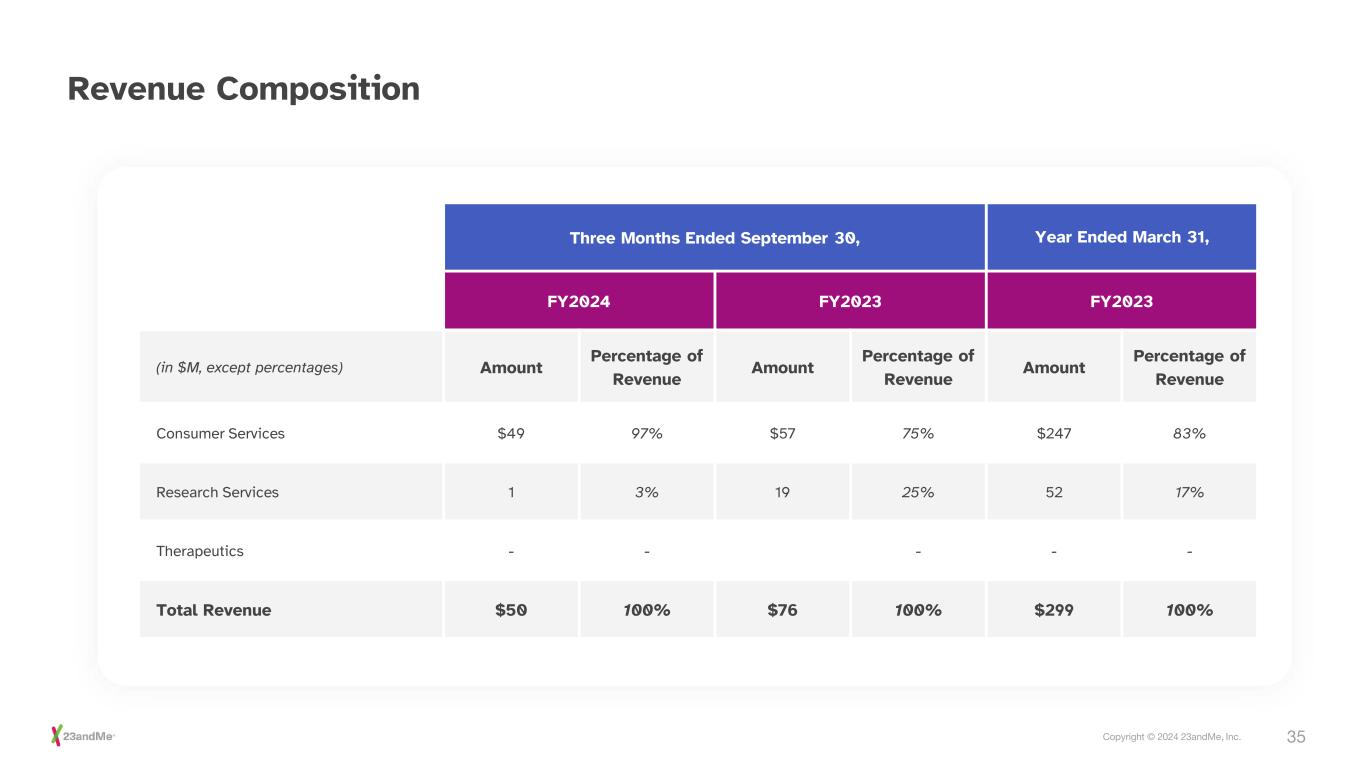
35Copyright © 2024 23andMe, Inc. Revenue Composition Three Months Ended September 30, Year Ended March 31, FY2024 FY2023 FY2023 (in $M, except percentages) Amount Percentage of Revenue Amount Percentage of Revenue Amount Percentage of Revenue Consumer Services $49 97% $57 75% $247 83% Research Services 1 3% 19 25% 52 17% Therapeutics - - - - - Total Revenue $50 100% $76 100% $299 100%

36Copyright © 2024 23andMe, Inc. Consumer Services Revenue Seasonality by Fiscal Quarter Q1 Q2 Q3 Q4 Full Year FY 2019 28% 19% 18% 35% 100% FY 2020 24% 24% 21% 31% 100% FY 2021 18% 21% 22% 39% 100% FY 2022 22% 20% 21% 38% 100% FY 2023 22% 25% 22% 31% 100% Note: Fiscal year ends March 31.
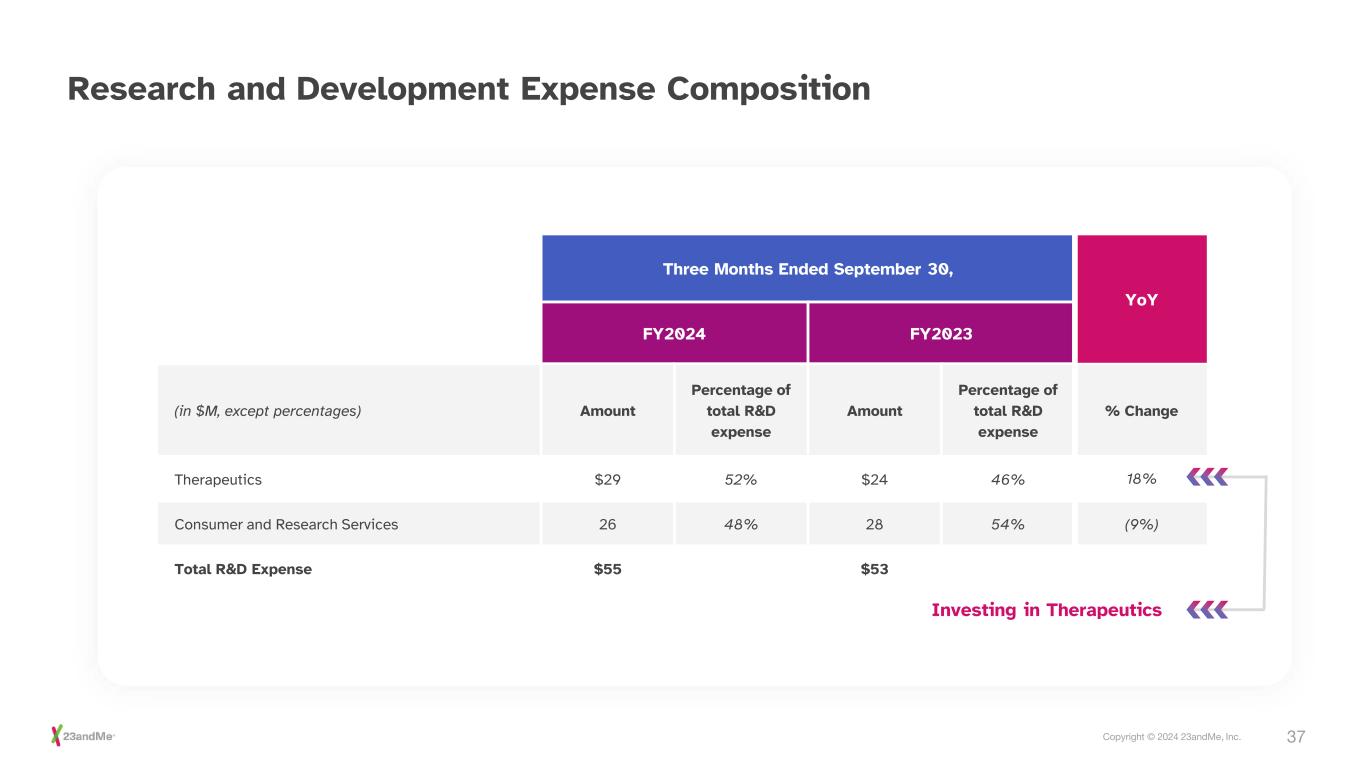
37Copyright © 2024 23andMe, Inc. Research and Development Expense Composition Three Months Ended September 30, YoY FY2024 FY2023 (in $M, except percentages) Amount Percentage of total R&D expense Amount Percentage of total R&D expense % Change Therapeutics $29 52% $24 46% 18% Consumer and Research Services 26 48% 28 54% (9%) Total R&D Expense $55 $53 Investing in Therapeutics

38Copyright © 2024 23andMe, Inc. Upcoming Value Drivers and Catalysts Consumer Research Therapeutics New product development, improved subscription value delivery, upgrades and cross-selling health services Continued customer LTV and margin improvement Progress toward adjusted EBITDA breakeven Research collaborations New GWAS Imputation innovations Initial ‘610 Phase 2A data PO14 IND Filing Potential collaborations

39Copyright © 2024 23andMe, Inc.