
Merck to Acquire Pandion Therapeutics Media Coverage February 26, 2021 Newswires/National Page 3 Bloomberg Reuters Dow Jones/Wall Street Journal.com Forbes Business/Top Investor Page 7 Investors Business Daily Barron’s TheStreet.com Fox Business Biotech Page 13 Endpoints Fierce Biotech Scrip BioPharma Dive PharmaPhorum BioWorld Vantage BioCentury First Word Pharma BioSpace STAT News Investor Trade Page 33 Business Insider S&P Global MarketWatch RTT News 1

Seeking Alpha Benzinga Financial Buzz Yahoo Finance Econotimes Business News Insider Pulse 2.0 Industry Trade Page 49 The Science Advisory Board MedCity News Law 360 Bioprocess International Biopharmaceutical Technology Pharmaceutical Business Review Gene Online News Local Page 59 Boston Globe (adapted Bloomberg story) Boston Business Journal Social Media Posts of Interest Page 61 2

BLOOMBERG https://www.bloomberg.com/news/articles/2021-02-25/merck-to-buy-autoimmune-drug-developer-pandion-for-1-85-billion Merck to Buy Autoimmune Drug Developer Pandion for $1.85 Billion By Eric Pfanner February 25, 2021, 7:03 AM EST Updated on February 25, 2021, 7:43 AM EST $60 per share offer is double Wednesday’s closing stock price Drugmaker has been making acquisitions to expand pipeline Merck & Co. agreed to acquire Pandion Therapeutics Inc., a biotechnology company developing therapies for autoimmune diseases, for $1.85 billion. The agreement, worth $60 per share in cash, is at more than double the Cambridge, Massachusetts-based company’s closing price Wednesday. Pandion’s potential treatment for ulcerative colitis and other autoimmune diseases, known as PT101, has completed a Phase 1a clinical trial that achieved its primary objective of safety and tolerability, the companies said in a statement. The planned purchase comes about three weeks after Merck named Robert M. Davis to succeed Kenneth Frazier as chief executive officer, and about a month after it ended its Covid-19 vaccine program. Pandion shares rose to just below the agreed price in premarket trading, with Merck up 0.5%. Under the terms of the deal, Merck will initiate a tender offer for Pandion shares via a subsidiary. The deal is expected to close in the first half of the year. Merck also recently agreed to buy VelosBio Inc. for $2.75 billion, moving to bolster its lineup of cancer therapies, and privately held biopharma company OncoImmune for an upfront $425 million to gain a potential therapy for severe Covid. (Updates with shares, deal terms, other deals in last three paragraphs) 3
REUTERS https://www.reuters.com/article/pandion-m-a-merck-co/merck-to-buy-drug-developer-pandion-therapeutics-for-1-85-billion-idINKBN2AP1Q3 Merck to buy drug developer Pandion Therapeutics for $1.85 billion Merck & Co Inc said on Thursday it would buy drug developer Pandion Therapeutics Inc for about $1.85 billion in cash, expanding its portfolio of drugs that target autoimmune diseases. The $60-per-share deal represents about 134% premium to Pandion’s closing price on Wednesday. Shares of Pandion rose 131.6% to a record high of $59.37 before the bell, while Merck shares were up about 1%. Pandion’s lead drug candidate, PT101, met its main goals of safety and tolerability last month in an early-stage trial in patients with autoimmune diseases. In November, Merck agreed to buy privately-held VelosBio for $2.75 billion to strengthen its cancer drug portfolio as the drugmaker looks to reduce its reliance on its blockbuster cancer therapy Keytruda. The deal between Merck and Pandion is expected to close in the first half of 2021, the companies said in a joint statement. Merck was represented by Credit Suisse Securities, Covington & Burling, while Centerview Partners LLC and Skadden, Arps, Slate, Meagher & Flom LLP represented Pandion. 4

DOW JONES/WSJ.com https://www.morningstar.com/news/dow-jones/202102257257/merck-to-buy-pandion-therapeutics-for-185-billion Merck to Buy Pandion Therapeutics for $1.85 Billion Provided by Dow Jones Feb 25, 2021 7:13 AM EST By Colin Kellaher Merck & Co. on Thursday said it agreed to buy clinical-stage biotechnology company Pandion Therapeutics Inc. for $60 a share, or about $1.85 billion, in cash. The purchase price is more than double Wednesday’s closing price of $25.63 for Pandion, a Watertown, Mass., company that is developing novel therapeutics for autoimmune diseases. Merck said the acquisition adds a pipeline of candidates designed to rebalance the immune response, with potential applications across an array of autoimmune diseases. The Kenilworth, N.J., drugmaker said it expects to complete the acquisition in the first half of 2021. Write to Colin Kellaher at colin.kellaher@wsj.com 5

(END) Dow Jones Newswires FORBES https://www.forbes.com/sites/palashghosh/2021/02/25/merck-agrees-to-acquire-pandion-therapeutics-amid-expectations-of-rebound-in-pharma-ma-this-year/?sh=1eaf9a921564 Merck Agrees To Acquire Pandion Therapeutics Amid Expectations Of Rebound In Pharma M&A This Year Merck said Thursday it agreed to acquire Pandion Therapeutics, a clinical-stage biotech firm developing products for patients with autoimmune diseases, in a deal valued at $1.85 billion, amid hopes that pharma M&A activity rebounds this year. KEY FACTS Merck will buy Pandion for $60 per share in cash, representing a 134% premium based on Pandion shares’ closing price on Wednesday. The transaction, which is expected to close in the first half of 2021, comes only a few weeks after Merck named Robert M. Davis as its new CEO, succeeding Kenneth Frazier, and one month after Merck ended its Covid-19 vaccine program. Based in Cambridge, Mass., Pandion’s lead candidate product, PT101, a potential treatment of ulcerative colitis and other autoimmune diseases, 6

completed a phase 1-A clinical trial earlier this year, which, Merck said, “achieved its primary objective of safety and tolerability.” CRUCIAL QUOTE “We believe Merck is well positioned to bring our novel approach to the millions of those living with autoimmune diseases, and we look forward to seeing these molecules progress in the clinic,” said Dr. Rahul Kakkar, chief executive officer, Pandion Therapeutics. KEY BACKGROUND Although the pharma industry announced some big acquisitions in the last year – notably AstraZeneca’s $39 billion deal for Alexion – 2020 witnessed a decline in pharma M&A activity. According to PricewaterhouseCoopers (PwC), excluding the huge AstraZeneca merger, the number of pharma/life sciences transactions fell by 2.3% to 242 last year, while the aggregate value of those deals plunged by 61% to $141 billion. PwC projects that 2021 will see M&A deals in the sector totaling between $250 billion to $275 billion, including several megamergers each at least $50 billion in size. Big cap companies like Amgen, Biogen and Bristol Myers Squibb are expected to lead the way with major acquisitions. “Companies will be looking for acquisitions that help support their strategic agendas, but we also see an opportunity for at least one really transformational transaction,” Sky Milch, U.S. pharmaceutical and life sciences deals leader at PwC, told FiercePharma news website. Milch estimated that as of December 2020, the global biopharma industry had about $1.47 trillion in capital (down only 6% from 2019) that it could use to make M&A deals. Last spring and summer, the Covid-19 pandemic forced some companies to develop drugs and vaccines to treat the virus, rather than use resources for M&A, while government-ordered shutdowns disrupted the industry’s supply chains, FiercePharma noted. By the fall, the rebound in pharma M&A got a big push from Gilead Sciences’ $21 billion purchase of breast cancer drug maker Immunomedics for $21 billion. INVESTORS BUSINESS DAILY 7

https://www.investors.com/news/technology/ipo-stock-pandion-surges-record-merck-takeover/ Pandion Therapeutics Surges To Record High On Merck’s $1.85 Billion Buyout ALLISON GATLIN 09:35 AM ET 02/25/2021 Pandion Therapeutics (PAND) stock blasted to a record high Thursday on Merck’s (MRK) plan to buy the IPO stock for $1.85 billion. Biotech company Pandion only wrapped its initial public offering seven months ago. But Merck is planning to take over the fledgling company for $60 per share. That represents a roughly 130% premium to the IPO stock’s share price, SVB Leerink analyst Geoffrey Porges said in a report to clients. Porges called the deal “an outsized outcome for Pandion’s shareholders given the company’s early stage of development.” Pandion’s lead asset is in Phase 1 testing to treat ulcerative colitis and a form of lupus. “This transaction also validates the notion that investors in recently public and early stage biopharma companies can still be tempted by the possibility by an early huge return from a large transaction at a high premium,” he said. “This transaction seems likely to contribute to a sustained buoyancy in the appetite for emerging biopharma equity offerings in the public markets.” IPO Stock Surges On Buyout In morning trading on the stock market today, IPO stock Pandion soared 131.9% near 59.40. Merck stock dipped a fraction near 74.20. Under the terms of the deal, Merck will acquire IPO stock Pandion through a subsidiary. Dr. Dean Y. Li, Merck Research Laboratories president, noted 8
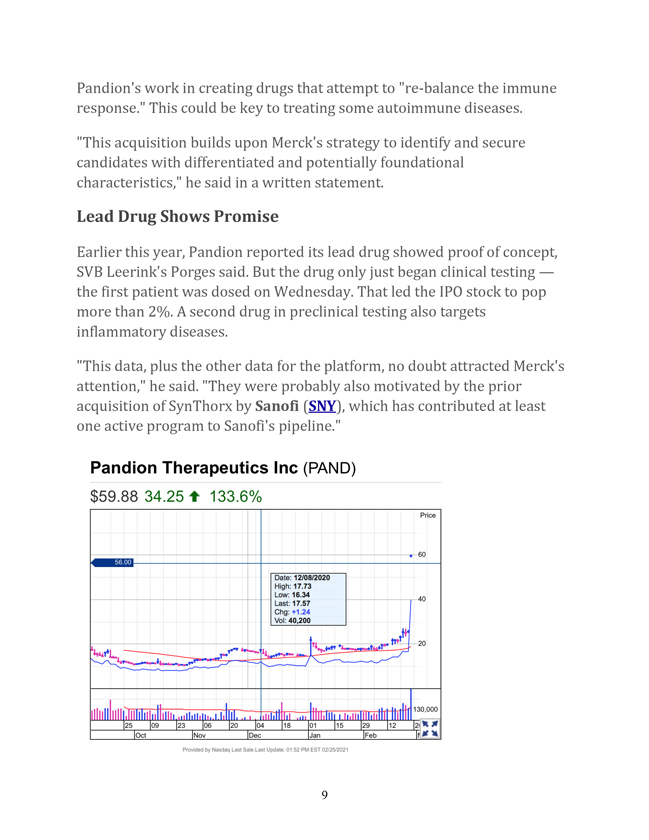
Pandion’s work in creating drugs that attempt to “re-balance the immune response.” This could be key to treating some autoimmune diseases. “This acquisition builds upon Merck’s strategy to identify and secure candidates with differentiated and potentially foundational characteristics,” he said in a written statement. Lead Drug Shows Promise Earlier this year, Pandion reported its lead drug showed proof of concept, SVB Leerink’s Porges said. But the drug only just began clinical testing — the first patient was dosed on Wednesday. That led the IPO stock to pop more than 2%. A second drug in preclinical testing also targets inflammatory diseases. “This data, plus the other data for the platform, no doubt attracted Merck’s attention,” he said. “They were probably also motivated by the prior acquisition of SynThorx by Sanofi (SNY), which has contributed at least one active program to Sanofi’s pipeline.” 9

IPO stock Pandion began trading in July at 18. Shares recently broke out of a cup base with a buy point at 23.56, according to MarketSmith.com. The news Thursday sent the IPO stock well above that entry. Follow Allison Gatlin on Twitter at @IBD_AGatlin. BARRONS https://www.barrons.com/articles/merck-pays-double-to-buy-pandion-therapeutics-51614259278 Merck Pays Double to Buy Pandion Therapeutics By Josh Nathan-Kazis Updated Feb. 25, 2021 10:50 am ET / Original Feb. 25, 2021 8:21 am ET Merck said early Thursday that it had cut a deal to buy a biotech firm called Pandion Therapeutics for $60 per share in cash, more than twice Pandion’s current share price. Shares of Pandion (ticker: PAND), which develops drugs to treat autoimmune diseases, closed at $25.63 on Wednesday. The stock has traded as low as $10.28, and no higher than $27.75, over the past 12 months. Shortly after the deal was announced, Pandion shares climbed to $59.32 in premarket trading, up 134.1%. The deal values Pandion at approximately $1.85 billion. The company is developing drugs called immune modulators to target autoimmune diseases. Its most advanced drug candidate, called PT101, is being tested as a treatment for ulcerative colitis and other autoimmune disease. It has successfully completed a Phase 1a safety and tolerability trial. 10

This acquisition builds upon Merck’s strategy to identify and secure candidates with differentiated and potentially foundational characteristics, “ said Dr. Dean Li, president of Merck Research Laboratories, in a statement. Merck shares are down 8.8% so far this year, and 6.7% over the past 12 months. The company’s longtime CEO, Kenneth Frazier, announced his long-expected retirement in early February. He will be succeeded by the company’s executive vice president, Robert Davis. Though the company is among the top vaccine makers in the world, and is generally renowned for its vaccine development capabilities, Merck’s efforts to develop a Covid-19 vaccine have so far failed. In late January, the company said it was giving up on its two experimental Covid-19 vaccines, after disappointing early trials. The company said that the Pandion acquisition is expected to close in the first half of this year. Pandion went public in July of 2020 in an initial public offering priced at $18 per share. It opened for trading at $27 per share on July 17. Merck announced a number of deal in late 2020, including the $2.75 billion acquisition of a private biotech company called VelosBio, and the $425 million acquisition of another privately held firm called OncoImmune. THE STREET.COM https://www.thestreet.com/investing/pandion-therapeutics-doubles-on-merck-1point85-billion-acquisition Pandion Therapeutics Doubles on Planned $1.85B Purchase by Merck Pandion Therapeutics shares more than double as it agrees to be acquired by Merck for $60 a share. ROB LENIHAN 11

Pandion Therapeutics (PAND)—Get Report more than doubled on Thursday after the biotech agreed to be acquired by Merck & Co. (MRK)—Get Report in a deal valued at about $1.85 billion. Shares of Pandion, Watertown, Mass., at last check were trading at $59.57, compared with Wednesday’s close at $25.63. Merck was little changed at $74.60. Under the terms, Merck, the Kenilworth, N.J., health-care giant, will pay $60 a share cash for Pandion. The deal is expected to close in the first half, subject to conditions including antitrust clearance and a vote of Pandion holders. Last month, Pandion said its leading drug candidate, PT101, met its main goals of safety and tolerability. The drug is being developed to treat ulcerative colitis, systemic lupus erythematosus, and other autoimmune diseases. SVB Leerink analyst Geoffrey Porges said in a note to clients that the deal “is an outsized outcome for Pandion’s shareholders given the company’s early stage of development,” according to Investors Business Daily. “This transaction also validates the notion that investors in recently public and early stage biopharma companies can still be tempted by the possibility by an early huge return from a large transaction at a high premium,” the analyst said. Earlier this month, Merck posted weaker-than-expected fourth-quarter earnings and said longtime Chief Executive Ken Frazier would retire later this year. In November, Merck said it would pay $425 million for OncoImmune, a privately held biotech that has developed a promising treatment for patients suffering a severe form of COVID-19. 12

“This acquisition builds upon Merck’s strategy to identify and secure candidates with differentiated and potentially foundational characteristics,” Dean Y. Li, president, Merck Research Laboratories, said in a statement. Fox Business https://www.foxbusiness.com/markets/merck-to-buy-drug-developer-pandion-therapeutics-for-1-85-billion Merck to buy drug developer Pandion Therapeutics for $1.85B The $60-per-share deal represents about 134% premium to Pandion’s closing price Merck & Co Inc said on Thursday it would buy drug developer Pandion Therapeutics Inc for about $1.85 billion in cash, to expand its portfolio of drugs that target autoimmune diseases. The $60-per-share deal represents about 134% premium to Pandion’s closing price on Wednesday. ENDPOINTS 13

https://endpts.com/merck-takes-a-swing-at-the-il-2-puzzlebox-with-a-1-85b-play-for-buzzy-pandion-and-its-autoimmune-hopefuls/ Merck takes a swing at the IL-2 puzzlebox with a $1.85B play for buzzy Pandion and its autoimmune hopefuls Kyle Blankenship Managing Editor When Roger Perlmutter bid farewell to Merck late last year, the drugmaker perhaps best known now for sales giant Keytruda signaled its intent to take a swing at early-stage novelty with the appointment of discovery head Dean Li. Now, Merck is signing a decent-sized check to bring an IL-2 moonshot into the fold. Merck will shell out roughly $1.85 billion for Pandion Pharmaceuticals, a biotech hoping to gin up regulatory T cells (Tregs) to treat a range of autoimmune disorders, the drugmaker said Thursday. The final price of the all-cash deal is equivalent to $60 per share, a roughly 134% premium on Pandion’s Wednesday closing price. The buyout is expected to close in the first half of this year. While Pandion plans to advance a larger range of therapies for autoimmune disorders, its lead candidate, IL-2 targeting PT101 for ulcerative colitis, scored a win in a Phase Ia trial earlier this year. In that 56-patient study, PT101 increased Treg levels by 260% and increased the level of high CD35-expressing Tregs by a whopping 6,250%, Pandion said. The drug also hits its marks on safety and tolerability. It’s a pretty remarkable ROI for Pandion investors after the biotech went public back in July for just $135 million. That big jump partly underscores the sort of crazy money floating around in biotech at the moment as well as the immense clinical need for a safe and effective drug targeting the IL-2 pathway. Meanwhile, Pandion, which launched four years ago with a star-studded pedigree, is also seeking new ways to regulate the immune system through its efforts to develop better bispecific antibodies. Astellas jumped on board the 14

biotech’s science in October 2019, agreeing to shell out up to $795 million for the company’s novel candidates for pancreatic autoimmune conditions. Merck’s interest in Pandion — and the steep price it paid — comes as it says goodbye to long-serving CEO Ken Frazier and has already bid farewell to Perlmutter, the man most responsible for shepherding checkpoint inhibitor Keytruda to the rarified air of pharma sales giants. Perlmutter handed the reins to Li, the former discovery lead at Merck, signaling the pharma giant’s intent to continue the search for the next big Keytruda follow-up. Perlmutter plans to stay on in a non-executive director role through mid-2021. Pandion’s bispecific work, though targeted on autoimmune conditions, could play well in Merck’s next-gen search for oncology blockbusters. Bispecific antibodies have become one of the big hot zones in cancer development, and Merck has good reason to chase novelty with Keytruda continuing to soak up sales and the rest of the portfolio sitting far behind. FIERCE BIOTECH https://www.fiercebiotech.com/biotech/merck-inks-1-9b-pandion-takeover-to-square-up-against-amgen-lilly-and-roche Merck inks $1.9B Pandion takeover to square up against Amgen, Lilly and Roche by Nick Paul Taylor | Feb 25, 2021 8:50am 15

Merck is paying a big premium to buy Pandion. (Merck) Merck has struck a $1.9 billion deal to buy Pandion. The takeover will give Merck control of a pipeline of immune modulators led by a rival to IL-2 drugs in development at Amgen, Eli Lilly and Roche. Pandion went public last year, pulling in $135 million to fund the development of a pipeline led by an engineered IL-2 mutein fused to a protein backbone. The clinical-phase drug, PT101, is designed to selectively activate and expand regulatory T cells (Tregs) and, in doing so, treat autoimmune diseases including ulcerative colitis. Merck has seen enough promise in the approach to offer to buy Pandion for $60 a share. The offer, which results in a total equity value of around $1.9 billion, is more than double the last closing price of Pandion. The stock traded below $20 apiece for almost all of Pandion’s time on public markets. Pandion commanded the big premium despite operating in a highly competitive space. Lilly is leading the IL-2 therapy field with its Nektar Therapeutics’ partnered LY3471851, which is in phase 2 trials in systemic lupus erythematosus and ulcerative colitis. Amgen’s efavaleukin alfa —formerly known as AMG592—and Roche’s RG7835 are in phase 1 development. Data on PT101 and its rivals suggest Pandion’s drug could be best in class. IL-2 activates Tregs at low doses, driving interest in its autoimmune applications, but activates proinflammatory natural killer (NK) cells at high doses, potentially exacerbating autoimmune diseases. The divergent effects of IL-2 at different doses make targeting the growth factor a balancing act. At high doses, Lilly’s PEGylated IL-2 has caused NK cell expansion. 16

Based on the available data, PT101 may be less prone than other IL-2 drugs to NK cell expansion. In preclinical, Pandion saw very little expansion of NK cells following administration of PT101. Pandion followed up at the start of this year with phase 1a data showing no evidence of NK cell expansion. Tregs increased by up to 3.6-fold over baseline, suggesting PT101 works about as well on that front as rivals from Amgen and Lilly. SCRIP https://scrip.pharmaintelligence.informa.com/authors/jessica-merrill Merck & Co. Buys Pandion, Building Out Capability In Autoimmune Disease 25 Feb 2021 Jessica Merrill@Jessicaemerrill Jessica.merrill@informa.com Executive Summary Merck & Co. reached a deal to acquire the developer of targeted immune modulators for $1.85bn. Pandion CEO Kakkar talked to Scrip about the acquisition. MERCK & CO. IS ACQUIRING AN AUTOIMMUNE DISEASE PLATFORM Source: Shutterstock Merck & Co., Inc. is building out its autoimmune disease capability with the $1.85bn acquisition of Pandion Therapeutics, Inc. announced on 25 February. The bulk of Merck & Co.’s recent string of deals has been in oncology, but this acquisition appears to balance the scale in a different therapeutic area. A newly public company focused on the development of precision immune modulators targeting key immune control nodes, Pandion has a discovery platform, several preclinical drug candidates and one clinical drug candidate in development for autoimmune diseases. The company’s lead 17

program is PT101, an engineered IL-2 mutein fused to a protein backbone to activate and expand regulatory T cells (T-regs) for the treatment of ulcerative colitis and other autoimmune diseases. “Pandion was started to try to usher in a fully different way of addressing autoimmune disease,” Pandion CEO Rahul Kakkar said in an interview. “Much of what is being developed these days or is in the clinic are really still in the realm of what I consider immunosuppression.” Current drug development efforts has hit up consistently against an efficacy ceiling, he said. “Pandion was really set up to move beyond that to what we like to consider true immunomodulation,” he said. Much of the foundation for the company comes from learnings and experience with immuno-oncology, which is why Merck & Co. was such an attractive buyer for Pandion. Merck & Co. is not considered a leader in autoimmune disease, but it is a leader in immuno-oncology, most notably as the developer of the immune checkpoint inhibitor Keytruda (pembrolizumab). “I like to think of Pandion as really just the flip side of IO being applied to autoimmune disease,” Kakkar said. Merck & Co.’s scientists, he said, “clearly understand the systems that we are talking about and the way these systems can be utilized for IO if you manipulate them in one direction but for autoimmunity if you manipulate them in another direction.” Pandion’s pipeline is in the very early stages of development, however. PT101 has just completed a Phase Ia clinical trial in healthy volunteers, demonstrating safety and tolerability, so there will be a lot of work ahead for Merck & Co. to realize the full potential of the deal. Behind PT101 is a PD-1 agonist in the investigational new drug (IND)-enabling phase of development. Pandion has been planning to move PT101 forward in development for ulcerative colitis first and then systemic lupus erythematosus, based on compelling early data from academia showing the potential of the IL-2 pathway in those indications. Kakkar, who joined Pandion as CEO in 2019 as the company moved toward clinical development, said the decision to sell the company came as Pandion was about to embark on a potentially expensive clinical drug development program that could span a wide array of autoimmune indications. “If you are targeting a mechanism which modulates the immune system, it is going to have potential applications in many autoimmune diseases,” he said. “We really had to make a choice: Do we continue to build this organization narrowly focusing on clinical build as our discovery engine continues to make more molecules or do we think about engaging with an entity that has the profound R&D resources that are going to be required to fully realize the potential of these molecules for patients.” A Steep Premium For Pandion, the deal represents a big return on investment and a quick turnaround. The Watertown, MA-based company went public through an initial public offering at $18 per share 18

in July, raising $135m. The company, formed in 2017, previously raised $58m in a series A round in early 2018 and $80m in a series B round in early 2020. (Also see “Finance Watch: Pandion’s $55m Series A Funds Localized Bispecifics For Autoimmune Diseases”—Scrip, 23 Jan, 2018.) and (Also see “Finance Watch: iTeos, Pandion And Aspen Show COVID-19 Hasn’t Slowed VC Deals Yet”—Scrip, 1 Apr, 2020.) Merck has agreed to pay $60 per share in cash for Pandion, a healthy premium over the company’s closing stock price of $25.63 on 25 February. Steep premiums are expected to persist within biotech given the optionality for young startups when it comes to private investment and the welcoming public markets. Big pharma leaders recently lamented the high premiums – particularly for early-stage assets – during the BIOCOM Global Life Science Partnering Conference. (Also see “High Deal Valuations & Volumes Will Persist, Industry Dealmakers Predict”—Scrip, 24 Feb, 2021.) SVB Leerink analyst Geoffrey Porges, in a same-day note, said the acquisition “validates the notion that investors in recently public and early-stage biopharma companies can still be tempted by the possibility of an early huge return from a large transaction at a high premium.” “This transaction seems likely to contribute to a sustained buoyancy in the appetite for emerging biopharma equity offerings in the public markets,” he added. Merck & Co. said the acquisition of Pandion builds on the company’s goal to develop drug candidates with “potentially foundational characteristics.” “Pandion has applied its TALON technology to develop a robust pipeline of candidates designed to re-balance the immune response with potential applications across a wide array of autoimmune diseases,” Merck Research Laboratories president Dean Li said in a statement. The acquisition comes during a period of transition for Merck & Co. within its top leadership ranks and within the portfolio as the company looks to build out beyond Keytruda (pembrolizumab). CEO Ken Frazier recently announced his planned retirement later in 2021, to be succeeded by chief financial officer Robert Davis, while Li took over as head of R&D at Merck & Co. from Roger Perlmutter in January. (Also see “Davis To Shepherd Merck & Co. Toward The Post-Keytruda Era, As Frazier Exits”—Scrip, 4 Feb, 2021.) Apart from this deal, Merck & Co.’s late-stage pipeline largely comprises candidates for cancer and infectious disease. The company has been on a deal-making streak since 2019 as part of an effort to bolster the portfolio and pipeline but Merck & Co. remains focused on licensing deals and bolt-on acquisitions over large-scale mergers. The bulk of the deals have been in oncology, including the $2.75bn acquisition of antibody drug conjugate and bispecific antibody developer VelosBio Inc. , announced in November. (Also see “Merck Pays $2.75bn To Access VelosBio’s ROR1-Targeting ADCs”—Scrip, 5 Nov, 2020.) 19

The company also partnered with Seagen Inc. in two cancer deals last year, to develop and commercialize ladiratuzumab vedotin, a Phase II antibody drug conjugate, and to commercialize Tukysa (tucatinib) outside the US, Canada and Europe, in exchange for $725m up front plus $1bn in equity. (Also see “Twice As Nice: Seattle Genetics, Merck & Co. Partner On Two Cancer Drugs”—Scrip, 14 Sep, 2020.) BioPharma Dive https://www.biopharmadive.com/news/merck-pandion-acquire-autoimmune-pd-1/595733/ Merck to buy autoimmune drugmaker for $2B Dive Brief: Merck & Co. has agreed to acquire Pandion Therapeutics, a Watertown, Massachusetts-based biotech developing drugs for autoimmune diseases, for $1.85 billion in cash. The deal values Pandion shares at $60 apiece, which is more than double their $25.03 closing price on Wednesday and nearly 3.5-times what the stock debuted at last July. Pandion has one drug in early-stage testing for ulcerative colitis and lupus, and preclinical research that could yield drugs for other autoimmune conditions. Typical treatments for autoimmune diseases broadly suppress the immune system, an approach that, while effective, can leave the body vulnerable to infections and other health problems. Pandion aims to develop safer drugs that 20

act more precisely, by dialing down an immune response associated with a specific site of inflammation. Dive Insight: The deal with Merck, which is expected to complete in the first half of this year, would bring an end to Pandion’s short run as an independent biotech. Formed in 2016 under the original name Immunotolerance, Pandion quickly attracted a substantial amount of investments on its mission to develop new, immune-regulating drugs. The biotech raised $58 million in 2018 through a Series A financing round co-led by its founding investor, Polaris Partners, as well as Versant Ventures and Roche’s corporate venture fund. By April 2020, Pandion had added another $80 million from a larger investor pool. And just a few months later, the biotech went public in an upsized offering that netted it another $135 million. The funding has been directed at developing a pipeline of precision medicines for autoimmune conditions, the most advanced of which is a drug called PT101. PT101 has two components: a protein backbone, and an engineered version of interleukin 2, a protein that helps regulate certain white blood cells. Pandion says it designed the drug to selectively activate and expand regulatory T cells, which keep the immune system from attacking the body. Early this year, Pandion reported positive results from a Phase 1 clinical trial, showing PT101 was well-tolerated and triggered its intended targets, without doing the same for other, pro-inflammatory T cells. With those results in hand, Pandion said a Phase 1/2 study of PT101 for ulcerative colitis should start in mid-2021, while a Phase 2 trial for systemic lupus erythematosus is expected to begin in the second half of this year. Behind PT101, Pandion is developing drugs meant to amplify another important regulatory protein known as PD-1. Notably, multiple blockbuster cancer immunotherapies, including Merck & Co.’s Keytruda, also target PD-1, though they work by inhibiting the protein instead. Pandion’s PD-1 drugs are preclinical testing for “numerous autoimmune diseases.” The biotech is also collaborating with Astellas Pharma to develop treatments for autoimmune diseases of the pancreas. For Merck, the Pandion deal adds to a string of recent and relatively small acquisitions for the pharmaceutical giant. In November alone, Merck agreed to drop $425 million to buy OncoImmune, a Maryland-based biotech with an experimental COVID-19 treatment, and then almost $3 billion more to snag VelosBio, a San Diego-based maker of antibody cancer drugs. 21

Last year also saw Merck acquire Themis, a privately held developer of vaccines and therapies for infectious diseases and cancer. The coronavirus vaccine acquired through that deal disappointed in testing, however. While Merck has favored these modest deals over the splashier ones made by many of its peers, the company is still under pressure to expand. Investors have expressed concerns that Merck is too reliant on Keytruda, a drug which last year accounted for about a third of its $43 billion in pharmaceutical sales. PharmaPhorum https://pharmaphorum.com/news/merck-takes-on-lilly-nektar-in-il-2-with-1-85bn-pandion-buy/ Merck takes on Lilly/Nektar in IL-2 with $1.85bn Pandion buy Merck & Co/MSD has agreed to buy Pandion for $1.85 billion, bolting on a pipeline of drugs for autoimmune and other immunological disorders headed by interleukin-2 (IL-2) based therapy PT-101. The $60 per share is a handsome, more than 100% premium on Pandion’s closing share price yesterday, and gives Merck a foothold in the hot area of treating disease by modifying the activity of regulatory T cells, or Tregs. Pandion’s lead candidate PT101 has completed a phase 1a clinical trial backing its safety and tolerability, and is due to start a phase 1a/2b study in ulcerative colitis and a phase 2 trial in systemic lupus erythematosus before the end of the year. The first trial showed that it could increase levels of Treg cells thought to be involved in dampening down immune responses. PT101 is an engineered IL-2 mutein fused to a protein backbone, and is vying to come to market along with other IL-2-based therapies, notably Eli Lilly and Nektar Therapeutics’ phase 2 candidate LY3471851 (also known as NKTR-358). LY3471851 – a long-acting, pegylated IL-2 formulation – is already in clinical trials for psoriasis, atopic dermatitis and SLE, with studies planned to get underway in ulcerative colitis shortly, according to the clinicaltrials.gov database. Data from a phase 1b study in patients with mild to moderate SLE reported last November showed that LY3471851 produced a dose-dependent increase in expression of Treg activation markers, backing up its proposed mode of action. 22

A little further behind in development are other IL-2 candidates including Amgen’s AMG-592, in phase 1/2 trials in SLE and graft versus host disease (GvHD), Roche’s RG 7835, which is in phase 1, and SAR444245 from Sanofi’s Synthorx unit, due to start clinical testing later this year. Nektar and Bristol-Myers Squibb also have a pegylated IL-2 drug heading for a phase 3 readout, but that candidate – bempegaldesleukin (NKTR-214) – is being developed as a cancer therapy. Nektar and Merck are also working on the combination of NKTR-214 with its Keytruda (pembrolizumab) cancer blockbuster. There are in fact dozens of companies trying to harness IL-2 to defeat cancer’s ability to avoid surveillance and attack from the immune system, but Pandion is one of a select few that are trying to flip that on its head and try to use the cytokine to protect organs from autoimmune reactions. Merck’s swoop on four-year-old Pandion comes just a few months after the company listed on the Nasdaq via an upsized IPO that raised $135 million, earmarked for the clinical development of PT101 as well as two PD-1 agonist candidates codenamed PT627 and PT001. Merck meanwhile has been steadily adding to its immunological disease pipeline in the last few years via a series of acquisitions. That includes deals to buy OncoImmune and its COVID-19 and GvHD candidate CD24Fc and COVID-19 vaccine developer Themis last year, and the $773 million takeover of Tilos Therapeutics in 2019. BIOWORLD—Preview https://www.bioworld.com/articles/504050-rapid-returns-for-shareholders-as-pandion-agrees-to-185b-merck-buyout Rapid returns for shareholders as Pandion agrees to $1.85B Merck buyout Feb. 25, 2021 Merck & Co. Inc. is paying $1.85 billion, or $60 per share, to acquire Pandion Therapeutics Inc. on the back of early stage data in human volunteers for its lead program, PT-101, an engineered interleukin-2 (IL-2) mutein fused to an Fc backbone, which is designed to stimulate targeted expansion of regulatory T cells (Tregs) for use in autoimmune disease indications. By any yardstick, the deal represents both a large and a rapid return on investment. Watertown, Mass.-based Pandion raised $291 million in equity financing since its formation in 2017, including a $58 million series A round in 2018 and an $80 million series B round in 2020. For investors in Pandion’s IPO, which it completed last July, the deal is a sweet outcome. The company priced its IPO at $18 23

per share. Its closing price (NASDAQ:PAND) on Feb. 24, immediately before the acquisition deal was disclosed, was $25.63. Overnight, the company’s valuation has jumped from just over $750 million to almost two and a half times that. BIOWORLD – Full Article Rapid returns for shareholders as Pandion agrees to $1.85B Merck buyout Feb. 25, 2021 By Cormac Sheridan Merck & Co. Inc. is paying $1.85 billion, or $60 per share, to acquire Pandion Therapeutics Inc. on the back of early stage data in human volunteers for its lead program, PT-101, an engineered interleukin-2 (IL-2) mutein fused to an Fc backbone, which is designed to stimulate targeted expansion of regulatory T cells (Tregs) for use in autoimmune disease indications. By any yardstick, the deal represents both a large and a rapid return on investment. Watertown, Mass.-based Pandion raised $291 million in equity financing since its formation in 2017, including a $58 million series A round in 2018 and an $80 million series B round in 2020. For investors in Pandion’s IPO, which it completed last July, the deal is a sweet outcome. The company priced its IPO at $18 per share. Its closing price on Feb. 24, immediately before the acquisition deal was disclosed, was $25.63. Overnight, the company’s valuation jumped from just over $750 million to almost two and a half times 24

that. Its share price (NASDAQ:PAND) moved accordingly before trading began on Feb. 25, quickly soaring to $59.75. Shares ended the day Feb. 25 at $59.81. The large premium is well above industry norms, but Merck, of Kenilworth, N.J., is betting that the early signs of biological activity reported from a recent first-in-human study in healthy volunteers will convert into an efficacy signal in ulcerative colitis and other autoimmune conditions. Rahul Kakkar, CEO, Pandion “The data speak for itself, frankly,” Pandion CEO Rahul Kakkar told BioWorld. In the study, PT-101 selectively expanded Tregs by 3.6-fold over baseline, while selectively expanding a Treg subset with high CD25 (called CD25 Bright Tregs) by 72.5-fold over baseline. The company observed no expansion of activated natural killer cells or pro-inflammatory T cells at any of the dose levels studied (1 mg – 10 mg). “PT-101 is on the cusp of entering patients,” Kakkar said. It is on track to start a phase Ia/IIb trial in ulcerative colitis around midyear, and Pandion is also prepping a phase II trial in systemic lupus erythematosus, which is due to get underway in the second half of the year. PT-101 is, unlike most IL-2 muteins, designed to dampen rather than activate immune responses. Proleukin (aldesleukin), the original recombinant version of IL-2, gained approval as a cancer drug several decades ago, but it was long hampered by a poor pharmacokinetic profile necessitating a grueling dose regimen, a lack of selectivity, which led to unwanted Treg expansion and a risk of causing capillary or vascular leak syndrome at high doses. The IL-2 receptor signaling is complex as it is mediated through a heterotrimeric complex of alpha (CD25), beta (CD122) and gamma (CD132) chains – differing configurations of some or all of those have different levels of affinity for IL-2. In recent years, multiple oncology drug developers have taken forward various IL-2 muteins that avoid those issues by selectively targeting the intermediate affinity IL-2 receptor, which comprises the beta and gamma chains. That approach strips out the Treg engagement activity, which is mediated by the alpha chain, and promotes immune 25

activation. “In autoimmune disease, we’re very much doing the flip side of that,” Kakkar said. “We’re trying to bias the molecule toward the CD25 receptor and away from the beta receptor.” The Fc antibody fragment to which the engineered IL-2 is fused is also modified, to eliminate Fc receptor binding – it serves purely as a half-life extension moiety. PT-101 is the lead example of Pandion’s modular approach to biologic drug design, which aims to achieve either systemic or localized Treg activation by linking active biologic drugs with protein backbones that determine the site of activity. It has several programs nearing or in IND-enabling studies, including PT-627, a systemic PD-1 agonist. PT-001 comprises a PD-1 agonist linked to Madcam (mucosal vascular addressin cell adhesion molecule), a lymphocyte homing receptor in the gut. It is designed to drive immunomodulation of the gastrointestinal tract. PT-002 is based on an IL-2 mutein fused to Madcam. Pandion has also developed tethers for the skin, kidneys and pancreas. The latter organ was the target of an early stage deal with Tokyo-based Astellas Pharma Inc. in October 2019, focused initially on type 1 diabetes. The deal is the second significant exit in less than a year in which Kakkar has had an involvement. Although he had left Corvidia Therapeutics Inc. by the time Ballerup, Denmark-based Novo Nordisk A/S acquired the Waltham, Mass.-based cardiometabolic drug developer for $725 million plus up to $1.375 billion in milestones, he played a significant role in its success, as founder, chief medical offer and chief strategy officer. The present transaction is due to close in the first half of this year. VANTAGE https://www.evaluate.com/vantage/articles/news/deals/merck-gets-treg-train-pandion-buy February 25, 2021 Merck gets on the Treg train with Pandion buy Madeleine Armstrong Merck & Co pays $1.9bn for an intriguing but early approach in autoimmune disease. 26
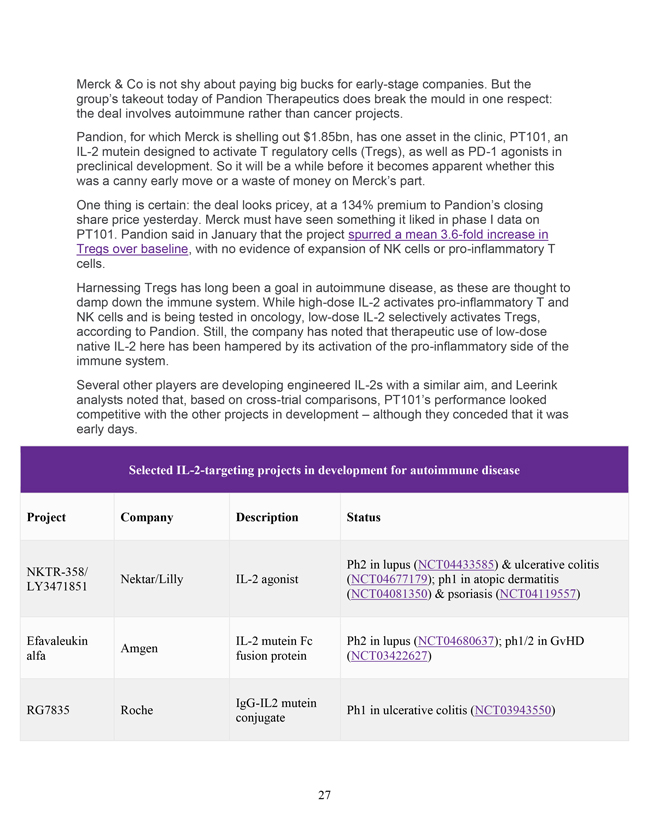
Merck & Co is not shy about paying big bucks for early-stage companies. But the group’s takeout today of Pandion Therapeutics does break the mould in one respect: the deal involves autoimmune rather than cancer projects. Pandion, for which Merck is shelling out $1.85bn, has one asset in the clinic, PT101, an IL-2 mutein designed to activate T regulatory cells (Tregs), as well as PD-1 agonists in preclinical development. So it will be a while before it becomes apparent whether this was a canny early move or a waste of money on Merck’s part. One thing is certain: the deal looks pricey, at a 134% premium to Pandion’s closing share price yesterday. Merck must have seen something it liked in phase I data on PT101. Pandion said in January that the project spurred a mean 3.6-fold increase in Tregs over baseline, with no evidence of expansion of NK cells or pro-inflammatory T cells. Harnessing Tregs has long been a goal in autoimmune disease, as these are thought to damp down the immune system. While high-dose IL-2 activates pro-inflammatory T and NK cells and is being tested in oncology, low-dose IL-2 selectively activates Tregs, according to Pandion. Still, the company has noted that therapeutic use of low-dose native IL-2 here has been hampered by its activation of the pro-inflammatory side of the immune system. Several other players are developing engineered IL-2s with a similar aim, and Leerink analysts noted that, based on cross-trial comparisons, PT101’s performance looked competitive with the other projects in development – although they conceded that it was early days. Selected IL-2-targeting projects in development for autoimmune disease Project Company Description Status Ph2 in lupus (NCT04433585) & ulcerative colitis NKTR-358/ Nektar/Lilly IL-2 agonist (NCT04677179); ph1 in atopic dermatitis LY3471851 (NCT04081350) & psoriasis (NCT04119557) Efavaleukin IL-2 mutein Fc Ph2 in lupus (NCT04680637); ph1/2 in GvHD Amgen alfa fusion protein (NCT03422627) IgG-IL2 mutein RG7835 Roche Ph1 in ulcerative colitis (NCT03943550) conjugate 27
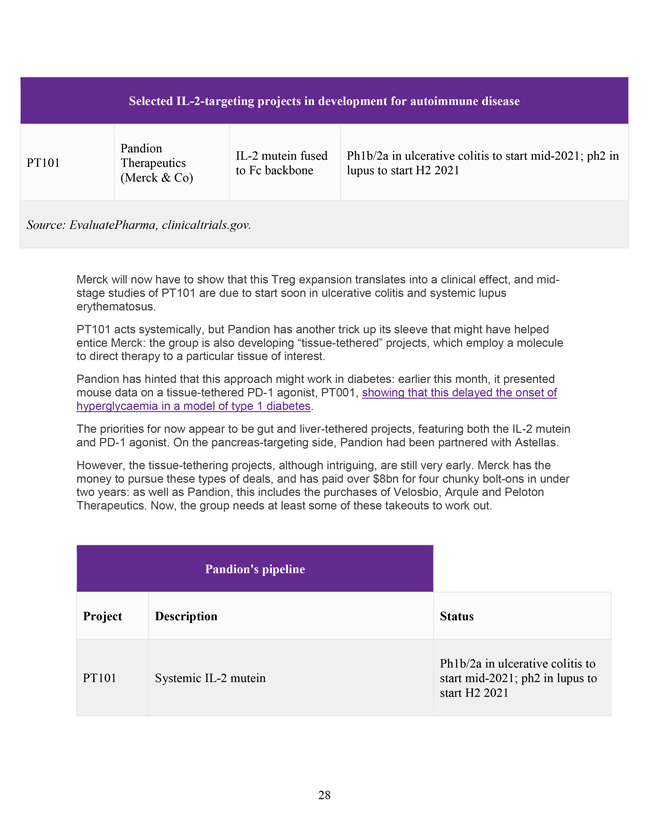
Selected IL-2-targeting projects in development for autoimmune disease Pandion IL-2 mutein fused Ph1b/2a in ulcerative colitis to start mid-2021; ph2 in PT101 Therapeutics to Fc backbone lupus to start H2 2021 (Merck & Co) Source: EvaluatePharma, clinicaltrials.gov. Merck will now have to show that this Treg expansion translates into a clinical effect, and mid-stage studies of PT101 are due to start soon in ulcerative colitis and systemic lupus erythematosus. PT101 acts systemically, but Pandion has another trick up its sleeve that might have helped entice Merck: the group is also developing “tissue-tethered” projects, which employ a molecule to direct therapy to a particular tissue of interest. Pandion has hinted that this approach might work in diabetes: earlier this month, it presented mouse data on a tissue-tethered PD-1 agonist, PT001, showing that this delayed the onset of hyperglycaemia in a model of type 1 diabetes. The priorities for now appear to be gut and liver-tethered projects, featuring both the IL-2 mutein and PD-1 agonist. On the pancreas-targeting side, Pandion had been partnered with Astellas. However, the tissue-tethering projects, although intriguing, are still very early. Merck has the money to pursue these types of deals, and has paid over $8bn for four chunky bolt-ons in under two years: as well as Pandion, this includes the purchases of Velosbio, Arqule and Peloton Therapeutics. Now, the group needs at least some of these takeouts to work out. Pandion’s pipeline Project Description Status Ph1b/2a in ulcerative colitis to PT101 Systemic IL-2 mutein start mid-2021; ph2 in lupus to start H2 2021 28

Pandion’s pipeline IL-2 mutein + MAdCAM tether for GI/liver PT002 Preclinical indications PT627 Systemic PD-1 agonist IND filing due in 2022 PD-1 agonist + MAdCAM tether for GI/liver IND-enabling studies to begin PT001 indications H1 2021 Source: Company presentation. BIOCENTURY Weeks after Pandion’s early clinical readout, Merck pays triple-digit premium in $1.9B takeout AT A 134% PREMIUM, PHARMA GAINS PLATFORM TO MODULATE THE IMMUNE SYSTEM BY PAUL BONANOS, ASSOCIATE EDITOR FEB 25, 2021 | 6:04 PM EST An early clinical proof-of-mechanism readout this year helped trigger Merck’s buyout of Pandion for nearly $1.9 billion, giving the pharma a platform with which to modulate the immune system as well as a lead program headed into mid-stage studies in two large indications. Merck & Co Inc. (NYSE:MRK) gains an immunomodulation platform to develop effector modules that can act systemically or be tethered to tissue-targeting constructs for increased localization and selectivity. CEO Rahul Kakkar told BioCentury that upon the company’s launch by Polaris Partners in 2017, the co-founders of Pandion Therapeutics Inc. (NASDAQ:PAND) set out to take lessons from the study of immuno-oncology to find ways to treat autoimmune diseases without 29

relying on immunosuppression — say, by stimulating immune control points, as opposed to inhibiting checkpoints as is done in cancer treatment. The deal comes about seven weeks after Watertown, Mass.-based Pandion revealed data from its Phase Ia trial of that lead program, PT101, in healthy volunteers. The fusion protein built on an engineered variant of IL-2 is designed to selectively expand regulatory T cells (Tregs) without stimulating pro-inflammatory conventional T cells or NK cells. The study showed that above a dosing threshold, PT101 led to at least twofold Treg expansion in more than 80% of subjects, with a mean maximum of 3.6-fold from baseline. Expansion of the CD25bright subset of Tregs, which the company said may be linked to enhanced immune regulation, was seen at up to 72.5-fold from baseline. Based on the results, Pandion planned to start a Phase Ib/IIa trial of PT101 around midyear in patients with ulcerative colitis, and a Phase II trial in systemic lupus erythematosus. Kakkar said Pandion’s decision was influenced by Merck’s ability to devote resources to its lead program and pipeline that would far outstrip what a smaller biotech could have accomplished. “We’re a platform company, and we don’t want to leave potential benefit behind,” he said. “We could have prosecuted UC and lupus on our own — we had money from our IPO — but they could bring a different order of resourcing to the molecule.” Merck will pay $60 per share to acquire Pandion, representing a 134% premium over Wednesday’s closing price of $25.63. Pandion had never traded higher than $27.75, its intraday peak on July 17, 2020, the day it went public. Since spiking briefly in its first trading day after pricing its IPO at $18, Pandion remained below $20 per share until last month’s data readout. Although Merck has been far stronger historically in cancer than in autoimmune diseases, with Keytruda pembrolizumab leading its immuno-oncology portfolio, Kakkar said the pharma has stressed inflammation and immunology as a strategic area of interest, and recognized the power of the biotech’s platform beyond its lead asset. Behind PT101, Pandion has two preclinical agonists PD-1 — one systemic, one tissue-targeted — and another preclinical IL-2 program that is drawn to the GI and liver. The biotech’s systemic therapy PT627 has begun IND-enabling work, and bifunctional molecule PT001 is to start an IND-enabling study this half. Both are PD-1 effector programs; the latter is tethered to MAdCAM to act in the GI. The GI and liver-targeting IL-2 program, PT002, is also tethered to MAdCAM. The company has not disclosed a clinical timeline. 30

Kakkar said preclinical evidence has shown that the tethered molecules have differentiated profiles from their systemic counterparts, although he acknowledged that the evidence is “still very conceptual” at the preclinical stage. The takeout is at least Merck’s eighth acquisition since the beginning of 2019, and fourth that was worth at least $1 billion up front. FIRST WORD PHARMA https://www.firstwordpharma.com/node/1803937?tsid=6 Merck & Co. to buy Pandion for $1.85 billion, gaining autoimmune drug pipeline (Ref: Business Wire, NASDAQ, Bloomberg, Morningstar, TheStreet) February 25th, 2021 By: Anna Bratulic Merck & Co. announced Thursday a definitive agreement to acquire Pandion Therapeutics for $60 per share in cash, representing an equity value of approximately $1.85 billion. The purchase will hand Merck a clutch of experimental drugs, including lead candidate PT101, that target a range of autoimmune diseases. Dean Li, who recently took over from Roger Perlmutter as head of Merck Research Laboratories, noted that “Pandion has applied its TALON technology to develop a robust pipeline of candidates designed to re-balance the immune response with potential applications across a wide array of autoimmune diseases.” According to Pandion, its TALON platform relies on immunomodulatory 31

effector modules that can be combined with tissue-selective tethers to create systemic and tissue-targeted new drugs. Selective expansion of Tregs PT101 is an engineered IL-2 mutein fused to a protein backbone designed to activate and expand regulatory T-cells (Tregs) as a potential treatment for ulcerative colitis and other autoimmune diseases. The drug completed a Phase Ia trial earlier this year, and besides achieving its primary objectives of safety and tolerability, it was able to induce “potent and selective” expansion of Tregs, including a subset with high CD25 expression, without triggering expansion of natural killer cells or pro-inflammatory conventional T-cells at any dose tested. “Multiple third-party clinical trials suggest that expansion of Tregs by low-dose IL-2 can benefit patients with autoimmune diseases,” Merck noted. Pandion has said a Phase Ia/II trial of PT101 in ulcerative colitis is expected to start in mid-2021, while a Phase II study in systemic lupus erythematosus is slated to get under way in the second half. Its pipeline also includes the IL-2 mutein PT002, as well as the PD-1 agonists PT627 and PT001, which are in preclinical development for numerous autoimmune diseases. Closing in H1 amid CEO transition The buyout, which represents a premium of around 134% to Pandion’s closing share price on February 24 and is expected to close in the first half of 2021, comes during a major transition period for Merck, as it prepares to hand over the reigns of the company to Robert Davis after current CEO Kenneth Frazier steps down on June 30. Late last year, it bolstered its oncology pipeline with ROR1-targeting drugs through the $2.75-billion purchase of VelosBio. Meanwhile, Pandion inked a licensing and collaboration deal with Astellas back in 2019 to develop locally acting immunomodulators for type 1 diabetes and other autoimmune diseases of the pancreas. BIOSPACE https://www.biospace.com/article/merck-acquires-autoimmune-focused-pandion-therapeutics-for-1-85-billion/ 32

Merck Snaps Up Autoimmune-Focused Pandion in Massive $1.85 Billion Deal Published: Feb 25, 2021 By Alex Keown Smith Collection/Gado/Getty Images Shares of Pandion Therapeutics have soared more than 131% in premarket trading after pharma giant Merck announced it is acquiring the company for up to $1.85 billion. Merck said the acquisition adds a pipeline of drug candidates that target a broad range of autoimmune diseases. This morning, Merck said it is acquiring Watertown, Mass.-based Pandion, which is developing novel therapeutics designed to address the unmet needs of patients living with autoimmune diseases, through a subsidiary for $60 per share in cash, a significant increase over Wednesday’s closing price of $25.63. This morning, Pandion’s stock price has skyrocketed to $59.36 after news of the deal broke. Pandion is advancing a pipeline of precision immune modulators targeting critical immune control nodes. The company’s lead candidate, PT101, is an engineered IL-2 mutein fused to a protein backbone designed to selectively activate and expand regulatory T cells (Tregs) for the potential treatment of ulcerative colitis and other autoimmune diseases. Tregs act as a control node within the immune system and can inhibit the activity of several different pro-inflammatory immune cell types. Defects in Tregs are associated with many autoimmune diseases. Research suggests that expansion of Tregs by low-dose IL-2 can benefit patients with autoimmune diseases. In January, Pandion announced PT101 hit the mark in a Phase Ia study. The company said PT101 was well-tolerated and selectively expanded total regulatory T cells. PT101 is in 33

development as a potential treatment for ulcerative colitis (UC), systemic lupus erythematosus (SLE), and other autoimmune diseases. The company’s pipeline also includes PD-1 agonists in development for numerous autoimmune diseases. Pandion’s PT001 is designed to activate, PD-1 receptors to reduce aberrant immune responses in tissues expressing mucosal addressing cell adhesion molecule (MAdCAM), such as the gastrointestinal tract, liver, and inflamed pancreas. Dean Y. Li, president of Merck Research Laboratories, said the acquisition of Pandion builds upon Merck’s strategy to identify and secure drug candidates that have differentiated and potentially foundational characteristics. “Pandion has applied its TALON (Therapeutic Autoimmune reguLatOry proteiN) technology to develop a robust pipeline of candidates designed to re-balance the immune response with potential applications across a wide array of autoimmune diseases,” Li said in a statement. Pandion Chief Executive Officer Rahul Kakkar said Merck is well-positioned to bring the company’s novel approach to treating autoimmune diseases to the patients that will benefit most from them. “We are proud that Merck has recognized our team’s innovation and drive in creating a pipeline of diverse candidates that activate natural immune regulatory mechanisms and thereby have the potential to achieve better clinical responses for patients,” Kakkar said in a statement. “Pandion grew out of our founders’ personal and scientific mission to change the way patients living with autoimmune diseases are treated. In just a few years, we have taken that mission from idea to clinical proof of mechanism with PT101, our lead IL-2 mutein.” The transaction is expected to close in the first half of 2021. STAT News By Ed Silverman Feb. 25, 2021 Merck (MRK) agreed to acquire Pandion Therapeutics (PAND), which is developing therapies for autoimmune diseases, for $1.85 billion, Bloomberg News informs us. Pandion is developing a treatment for ulcerative colitis, known as PT101, that has completed a Phase 1a clinical trial, which achieved its primary objective of safety and tolerability. Merck also recently agreed to buy VelosBio for $2.75 billion to bolster its cancer portfolio and OncoImmune for an upfront $425 million to gain a potential therapy for severe Covid. 34

BUSINESS INSIDER https://markets.businessinsider.com/news/stocks/pandion-therapeutics-stock-price-merck-merck-agrees-2-billion-buyout-2021-2-1030124405 Pandion Therapeutics soars 132% after Merck agrees to buy the biotech for $1.85 billion Matthew Fox Feb. 25, 2021, 03:18 PM Merck & Co. CEO Kenneth C. Frazier Stephanie Keith / Stringer / Getty Images Pandion Therapeutics soared 132% on Thursday after Merck agreed to acquire the biotech firm for $1.85 billion. Merck will initiate a tender offer through a subsidiary to acquire all outstanding shares of Pandion for $60 per share. Pandion develops therapeutics targeting patients living with autoimmune diseases. 35

Merck sparked a 132% surge in Pandion Therapeutics on Thursday after the pharmaceutical giant agreed to acquire the biotech firm for $1.85 billion. Pandion is a clinical-stage biotechnology company that is developing therapeutics for patients living with autoimmune diseases. Pandion’s lead candidate, PT101, completed a Phase 1a trial earlier this year and is a potential treatment for ulcerative colitis. Merck will initiate a tender offer through a subsidiary to acquire all outstanding shares of Pandion for $60 per share. Shares of Pandion closed at $25.63 on Wednesday. The completion of the acquisition will require at least a majority of Pandion shareholders tendering their shares to Merck. “Pandion has applied its TALON technology to develop a robust pipeline of candidates designed to re-balance the immune response with potential applications across a wide array of autoimmune diseases,” said Dr. Dean Y. Li, president of Merck Research Laboratories. The transaction is expected to close in the first half of 2021. S&P GLOBAL Market Intelligence https://www.spglobal.com/marketintelligence/en/news-insights/latest-news-headlines/merck-co-pays-134-premium-in-1-85b-purchase-of-pandion-therapeutics-62869463 25 Feb, 2021 Merck & Co. pays 134% premium in $1.85B purchase of Pandion Therapeutics AuthorMichael Gibney ThemeHealthcare & PharmaceuticalsBanking Merck & Co. Inc. has agreed to acquire autoimmune drugmaker Pandion Therapeutics Inc. for $1.85 billion, or $60 per share, amounting to a 134% premium on Pandion’s Feb. 24 closing price of $25.63 per share. 36

Pandion’s treatment pipeline is still in early clinical stages, with its most advanced drug PT101 — in a class of therapies called IL-2 muteins — in the first of three stages of human testing for autoimmune diseases ulcerative colitis and systemic lupus erythematosus. The company also has autoimmune drugs in preclinical testing, including two in a class called PD-1 agonists. The deal is valued at $1.65 billion including equity, assumed current liabilities and net of current assets, according to data from S&P Global Market Intelligence. The announcement of the acquisition, which is expected to close in the first half of the year, follows the news that Merck’s Ken Frazier plans to step down as CEO at the end of June to be replaced by CFO Rob Davis. On a fourth-quarter earnings call, Frazier vowed to pass along the mantle of M&A that keeps new technology flowing into the U.S. pharmaceutical giant. Merck is best known as the maker of cancer blockbuster Keytruda, which brought in $14.38 billion for the company in 2020. The M&A announcement marks Kenilworth, N.J.-based Merck’s first acquisition of 2021; the company completed five such deals in 2020, according to S&P Global Market Intelligence data. Watertown, Mass.-based Pandion priced an IPO in July 2020 with expected proceeds of $135 million, or $18 per share. Beyond its therapies in the clinic, Pandion comes with a drug discovery platform called TALON, which uses the mechanisms in the natural immune system to design drugs to treat autoimmune diseases. “This acquisition builds upon Merck’s strategy to identify and secure candidates with differentiated and potentially foundational characteristics,” Merck Research Laboratories President Dean Li said. “Pandion has applied its TALON technology to develop a robust pipeline of candidates designed to rebalance the immune response with potential applications across a wide array of autoimmune diseases.” Credit Suisse Securities (USA) LLC acted as financial adviser to Merck, while Covington & Burling LLP acted as legal adviser. Centerview Partners LLC was financial adviser to Pandion, with Skadden Arps Slate Meagher & Flom LLP as its legal adviser. 37
38
MARKETWATCH https://www.marketwatch.com/story/merck-to-buy-pandion-therapeutics-for-134-premium-2021-02-25 Merck to buy Pandion Therapeutics for 134% premium Published: Feb. 25, 2021 at 6:57 a.m. ET By Tomi Kilgore Referenced Symbols MRK +0.07% PAND +133.36% SPX -2.45% Merck & Co. MRK, +0.07% announced an agreement Thursday to buy Pandion Therapeutics Inc. PAND, +133.36% in a deal valued at $1.85 billion. Under terms of the deal, Merck will pay $60 in cash for each Pandion share outstanding, which represents a 134.1% premium to Wednesday’s closing price. Pandion is a biotechnology company which uses its TALON technology to develop autoimmune treatments. The deal is expected to close in the first half of 2021. Merck shares rose 0.6% in premarket trading, while Pandion’s stock was still untraded. Over the past three months, Merck shares have slipped 6.9% through Wednesday, while Pandion’s stock has run up 41.7% and the S&P 500 SPX, -2.45% has gained 8.2%. RTT NEWS https://www.rttnews.com/3172838/merck-to-acquire-pandion-therapeutics-quick-facts.aspx Merck To Acquire Pandion Therapeutics—Quick Facts By RTTNews Staff Writer ✉ | Published: 2/25/2021 6:59 AM ET Merck (MRK) has agreed to acquire Pandion Therapeutics, Inc. (PAND), a clinical-stage biotechnology company developing therapeutics designed to address the unmet needs 39

of patients living with autoimmune diseases, for $60 per share in cash. This represents an approximate total equity value of $1.85 billion. Merck, through a unit, will initiate a tender offer to acquire all outstanding shares of Pandion. Upon the successful completion of the tender offer, Merck’s acquisition unit will be merged into Pandion. The deal is expected to close in the first half of 2021. PT101, the lead candidate of Pandion, is an engineered IL-2 mutein fused to a protein backbone designed to selectively activate and expand regulatory T cells (Tregs) for the potential treatment of ulcerative colitis and other autoimmune diseases. PT101 had completed a phase 1a clinical trial, which achieved its primary objective of safety and tolerability. For comments and feedback contact: editorial@rttnews.co SEEKING ALPHA https://seekingalpha.com/news/3666246-merck-to-acquire-pandion-in-a-deal-valued-at-2-billion Merck to acquire Pandion for $2 billion Feb. 25, 2021 7:09 AM ET Merck & Co., Inc. (MRK) By: Dulan Lokuwithana Merck (NYSE:MRK) has agreed to acquire Pandion Therapeutics (NASDAQ:PAND) for $60 per share in an all-cash deal valuing Pandion at $1.85B. Under the terms of the agreement, Merck, through a subsidiary, will initiate a tender offer to acquire all outstanding shares of Pandion, and subsequently Merck’s acquisition subsidiary will be merged into Pandion. 40

Pandion’s lead candidate PT101, targeting ulcerative colitis (UC), and systemic lupus erythematosus (SLE), has completed a Phase 1a clinical trial achieving its primary objective of safety and tolerability. BENZINGA https://www.benzinga.com/general/biotech/21/02/19853214/why-merck-is-buying-autoimmune-disease-drug-company-pandion-for-1-85b Why Merck Is Buying Autoimmune Disease Drug Company Pandion For $1.85B Shanthi Rexaline , Benzinga Staff Writer February 25, 2021 10:16am 2 min read Merck & Co., Inc. MRK 0.03% is making another big-ticket buy, this time to expand its presence in the autoimmune disease market. What Happened: Large-cap biopharma Merck announced Thursday it has signed a definitive agreement to buy Pandion Therapeutics Inc PAND 133.36% for $60 per share in cash, or a total equity value of $1.85 billion. 41

The per-share value represents a 134% premium over the $25.63 price at which Pandion shares closed Wednesday. Cambridge, Massachusetts-based Pandion is a clinical-stage biopharma that focuses on developing novel therapeutics to treat patients living with autoimmune disease. The transaction is expected to close in the first half of 2021. FINANCIAL BUZZ https://www.financialbuzz.com/merck-enters-definitive-agreement-to-acquire-pandion-therapeutics-for-usd-1-85-billion/ Merck Enters Definitive Agreement to Acquire Pandion Therapeutics for USD 1.85 Billion 02/25/2021 Merck (NYSE: MRK) and Pandion Therapeutics, Inc. (NASDAQ: PAND)have reported entry into a definitive agreement where Merck will acquire Pandion for USD 60 per share in cash or a total equity value of USD 1.85 Billion. “This acquisition builds upon Merck’s strategy to identify and secure candidates with differentiated and potentially foundational characteristics,” said Dr. Dean Y. Li, president, Merck Research Laboratories. “Pandion has applied its TALON technology to develop a robust pipeline of candidates designed to re-balance the immune response with potential applications across a wide array of autoimmune diseases.” 42

Pandion Therapeutics, Inc. $59.81 “Pandion grew out of our founders’ personal and scientific mission to change the way patients living with autoimmune diseases are treated. In just a few years, we have taken that mission from idea to clinical proof of mechanism with PT101, our lead IL-2 mutein. We are proud that Merck has recognized our team’s innovation and drive in creating a pipeline of diverse candidates that activate natural immune regulatory mechanisms and thereby have the potential to achieve better clinical responses for patients,” said Dr. Rahul Kakkar, chief executive officer, Pandion Therapeutics. “We believe Merck is well positioned to bring our novel approach to the millions of those living with autoimmune diseases, and we look forward to seeing these molecules progress in the clinic.” YAHOO FINANCE https://finance.yahoo.com/news/merck-inks-deal-buy-pandion-072318545.html Merck Inks Deal To Buy Pandion Therapeutics For $1.85B; Shares Skyrocket support@smarteranalyst.com (Ben Mahaney) Fri, February 26, 2021, 2:23 AM·2 min read PAND +133.36% MRK +0.07% 43

Merck & Company agreed to snap up Pandion Therapeutics, a clinical-stage biotechnology company, in a deal worth $1.85 billion. Pandion shares jumped almost 133.4% to close at $59.81 on Feb. 25. Per the terms of the deal, the pharmaceutical company will pay $60 per share in cash. Merck (MRK) will acquire all shares of Pandion through a subsidiary, which would initiate a tender offer. The tender offer will close subject to certain conditions. These include the conditions that at least a majority of the total number of Pandion’s fully-diluted common shares are tendered and the expiration of the waiting period is determined as per the Hart-Scott-Rodino Antitrust Improvements Act. Upon completion of the tender offer, the remaining shares of Pandion (PAND) will be canceled and converted into the right to receive at the price tag of $60 per share. The acquisition is likely to close in the first half of 2021. Pandion CEO Dr. Rahul Kakkar said, “Pandion grew out of our founders’ personal and scientific mission to change the way patients living with autoimmune diseases are treated.” “We are proud that Merck has recognized our team’s innovation and drive in creating a pipeline of diverse candidates that activate natural immune regulatory mechanisms and thereby have the potential to achieve better clinical responses for patients,” he added. (See Merck stock analysis on TipRanks) Following the deal, Mizuho Securities analyst Mara Goldstein maintained a Buy rating and a price target of $100. The analyst believes “MRK is augmenting its pipeline (with two clinical, three preclinical, and four discovery programs by YE21) ahead of the expected Organon & Co. Spin off (late 2Q21) with the acquisition of autoimmune-focused company Pandion.” The consensus rating among analysts is a Strong Buy based on 4 Buys versus 1 Hold. The average analyst price target stands at $94.40 and implies upside potential of 26.5% to current levels over the next 12 months. Additionally, Merck scores a 9 out of 10 from TipRanks’ Smart Score rating system, indicating that the stock has strong potential to outperform market expectations. 44

ECONOTIMES https://www.econotimes.com/Merck-to-acquire-Pandion-Therapeutics-for-185-billion-in-cash-1603161 Merck to acquire Pandion Therapeutics for $1.85 billion in cash Photo by: Merck/Facebook Friday, February 26, 2021 7:07 AM UTC Merck & Co Inc is buying Pandion Therapeutics, the biotechnology company developing bispecific antibody therapeutics headquartered in Massachusetts. It was said that the pharmaceutical firm is shelling out a total of $1.85 billion in cash to acquire it. 45

Merck’s target expansion The acquisition was revealed on Feb. 25 through a joint statement from Merck and Pandion Therapeutics. The deal is also equivalent to a $60 per share deal that amounts to a 134% premium to Pandion’s closing price on Wednesday. As per Fox Business, Merck & Co Inc. is purchasing the said drug developer to broaden its portfolio of drugs designed for autoimmune diseases. With the deal, the pharmaceutical company that is one of the largest in the world is also set to have Pandion’s major drug candidate called PT101, which is currently undergoing testing for public release. The drug already completed and passed the phase 1a clinical trial last month for safety and tolerability. It is now in the early stage of the trial involving volunteer patients with autoimmune diseases. “This acquisition builds upon Merck’s strategy to identify and secure candidates with differentiated and potentially foundational characteristics,” Dr. Dean Y. Li, Merck Research Laboratories president said in a press release. “Pandion has applied its TALON technology to develop a robust pipeline of candidates designed to re-balance the immune response with potential applications across a wide array of autoimmune diseases.” Merck and Pandion are expected to close the deal before the second half of this year begins. Both companies confirmed this during the announcement this week. Merck and Pandion’s stocks up after the buyout reveal The New Jersey-headquartered pharma company is paying more than double Pandion’s current stock price, and the news of this deal drove both Merck and Pandion’s share to shoot up. Shares of the drug developer closed at $25.63 after the confirmation of the acquisition, while Merck shares increased by 0.3% in premarket trading. 46

Meanwhile, this was Merck’s latest purchase as it had entered into a number of deals before 2020 closed. One of its major acquisitions was the biotech company VelosBio which was bought for $2.75 billion. It also acquired Oncolmmune, another private firm, for $425 million. BUSINESS NEWS INSIDER https://www.business-news-today.com/merck-to-acquire-us-biotech-company-pandion-therapeutics-for-1-8bn/ Merck to acquire US biotech company Pandion Therapeutics for $1.8bn Tagged with: Dr. Dean Y. Li | Dr. Rahul Kakkar | Massachusetts | Merck | Pharma acquisition news | PT101 | Ulcerative colitis | US biotech company | USA Merck has agreed to acquire Pandion Therapeutics, a Massachusetts-based clinical-stage biotech company, for about $1.85 billion in an all-cash deal. Pandion Therapeutics is engaged in developing therapeutics for addressing the unmet needs of patients suffering from autoimmune diseases. The US biotech company is being acquired by Merck for $60 per share. Commenting on Merck acquisition of Pandion Therapeutics, Dr. Dean Y. Li – president of Merck Research Laboratories, said: “This acquisition builds upon Merck’s strategy to identify and secure candidates with differentiated and potentially foundational characteristics. “Pandion has applied its TALON technology to develop a robust pipeline of candidates designed to re-balance the immune response with potential applications across a wide array of autoimmune diseases.” Pandion Therapeutics is focused on advancing a pipeline of precision immune modulators that target critical immune control nodes. 47

Its lead candidate is PT101, which is a modified IL-2 mutein fused to a protein backbone. PT101 is designed to selectively trigger and expand regulatory T cells (Tregs) for potentially treating ulcerative colitis and other autoimmune diseases. Earlier this year, Pandion Therapeutics wrapped up a phase 1a clinical trial for PT101, in which the primary objective of safety and tolerability was achieved. Also, part of its pipeline are PD-1 agonists, which are being developed for various autoimmune diseases. Merck acquisition of Pandion Therapeutics Dr. Rahul Kakkar – CEO of Pandion Therapeutics, commenting on Merck acquisition of Pandion Therapeutics, said: “Pandion grew out of our founders’ personal and scientific mission to change the way patients living with autoimmune diseases are treated. In just a few years, we have taken that mission from idea to clinical proof of mechanism with PT101, our lead IL-2 mutein. “We are proud that Merck has recognized our team’s innovation and drive in creating a pipeline of diverse candidates that activate natural immune regulatory mechanisms and thereby have the potential to achieve better clinical responses for patients. “We believe Merck is well positioned to bring our novel approach to the millions of those living with autoimmune diseases, and we look forward to seeing these molecules progress in the clinic.” 48

The deal, which is subject to customary conditions, is expected to be wrapped up in the first half of this year. PULSE 2.0 https://pulse2.com/merck-mrk-to-buy-pandion-therapeutics-pand-in-1-85-billion-deal/ Merck (MRK) To Buy Pandion Therapeutics (PAND) In $1.85 Billion Deal By Amit Chowdhry • February 25, 2021 Merck (NYSE: MRK) is buying Pandion Therapeutics, Inc. (Nasdaq: PAND) in a deal valued at $1.85 billion. These are the details. Merck (NYSE: MRK) — known as MSD outside the United States and Canada — and Pandion Therapeutics, Inc. (Nasdaq: PAND) announced today that the companies have entered into a definitive agreement, under which Merck through a subsidiary will buy Pandion, a clinical-stage biotechnology company developing novel therapeutics designed to address the unmet needs of patients living with autoimmune diseases for $60 per share in cash. And this represents an approximate total equity value of $1.85 billion. Pandion is advancing the pipeline of precision immune modulators targeting critical immune control nodes. And Pandion’s lead candidate PT101 is an engineered IL-2 mutein fused to a protein backbone designed to selectively activate and expand regulatory T cells (Tregs) for the potential treatment of ulcerative colitis and other autoimmune 49

diseases. And earlier this year, Pandion announced that PT101 had completed a Phase 1a clinical trial, which achieved its primary objective of safety and tolerability. The company’s pipeline also includes PD-1 agonists in development for numerous autoimmune diseases. Under the terms of the deal, Merck through a subsidiary will initiate a tender offer to acquire all outstanding shares of Pandion. And the closing of the tender offer will be subject to certain conditions, including the tender of shares representing at least a majority of the total number of Pandion’s shares of fully-diluted common stock, the expiration of the waiting period, and other customary conditions. Upon the successful completion of the tender offer, Merck’s acquisition subsidiary is going to be merged into Pandion. And any remaining shares of common stock of Pandion will be canceled and converted into the right to receive the same $60 per share price payable in the tender offer. The deal is expected to close in the first half of 2021. KEY QUOTES: “This acquisition builds upon Merck’s strategy to identify and secure candidates with differentiated and potentially foundational characteristics. Pandion has applied its TALON technology to develop a robust pipeline of candidates designed to re-balance the immune response with potential applications across a wide array of autoimmune diseases.” — Dr. Dean Y. Li, president, Merck Research Laboratories “Pandion grew out of our founders’ personal and scientific mission to change the way patients living with autoimmune diseases are treated. In just a few years, we have taken that mission from idea to clinical proof of mechanism with PT101, our lead IL-2 mutein. We are 50

proud that Merck has recognized our team’s innovation and drive in creating a pipeline of diverse candidates that activate natural immune regulatory mechanisms and thereby have the potential to achieve better clinical responses for patients. We believe Merck is well positioned to bring our novel approach to the millions of those living with autoimmune diseases, and we look forward to seeing these molecules progress in the clinic.” — Dr. Rahul Kakkar, chief executive officer, Pandion Therapeutics THE SCIENCE ADVISORY BOARD https://www.scienceboard.net/index.aspx?sec=ser&sub=def&pag=dis&ItemID=222 7 Merck to acquire Pandion for $1.85B By The Science Advisory Board staff writers February 25, 2021 — Merck has announced that it will acquire Pandion Therapeutics for $1.85 billion. The deal will boost Merck’s ability to further develop a range of drug candidates for autoimmune diseases, it said. It will make the purchase through one of its subsidiaries. Pandion is developing immune modulators that target immune system control nodes, according to Merck. The company’s lead candidate is PT101, an engineered interleukin-2 (IL-2) mutein (protein with an altered amino acid sequence) fused to a protein backbone that activates regulatory T cells for the treatment of autoimmune diseases; Pandion has completed a phase IA clinical trial for the drug. Pandion’s pipeline also includes programmed cell death 1 (PD-1) agonists in development for several autoimmune diseases. 51

The transaction is expected to close in the first half of this year. MEDCITY NEWS https://medcitynews.com/2021/02/merck-commits-1-85b-to-pandion-in-bid-to-boost-immunology-pipeline/ Merck commits $1.85B to Pandion in bid to boost immunology pipeline Merck’s top immunology drugs face biosimilar competition. The $1.85 billion acquisition of Pandion Therapeutics gives the pharma giant an opportunity to build up its immunology pipeline with a new approach to treating autoimmune disorders. By FRANK VINLUAN Post a comment / Feb 25, 2021 at 11:53 AM 52
Pandion Therapeutics is still in the earliest stage of testing its lead drug candidate in humans, but Merck sees enough potential for the biotech’s approach to autoimmune disease to plunk down $1.85 billion to buy the company outright. The Kenilworth, New Jersey-based pharmaceutical giant announced Thursday that it will pay $60 cash for each share of Pandion, which is a more than 138 percent premium to the Pandion’s $25.11 closing stock price on Wednesday. For Pandion, the buyout marks a quick exit for a company that emerged just three years ago, and seven months ago raised $135 million in an IPO priced at $18 per share. Based in Watertown, Massachusetts, Pandion develops drugs that target regulatory T cells, or Tregs. Whereas T cells activate an immune response, Tregs tamp it down by blocking immune cells that cause inflammation. Tregs play an important role in ensuring that the immune system doesn’t attack the body’s own cells. In some autoimmune diseases, defects in Tregs lead to inflammation in organs, contributing to autoimmune disorders. Using proprietary drug discovery and design technology that it calls TALON, Pandion is developing therapies intended to activate Tregs as a way of treating the overactive immune response. 53

Lead Pandion drug candidate, PT101, combines a signaling molecule called interleukin-2 (IL-2) with a mutated protein (“mutein”). It’s designed to selectively expand Tregs throughout the body without activating immune cells that spark inflammation. The lead disease target is moderate-to-severe ulcerative colitis, an inflammatory condition affecting the gut. In January, Pandion reported preliminary results from a Phase 1 study enrolling healthy volunteers. The data showed that the drug was well tolerated by patients with no safety problems observed. Pandion also reported that the drug induced Treg expansion at levels that exceeded levels associated with patient benefit in clinical studies of IL-2 that others have conducted in multiple autoimmune diseases. The company added that its drug was selective for Tregs and there was no evidence that T cells and natural killer cells, two types of pro-inflammatory immune cells, were activated by the experimental treatment at any dose. Based on those results, Pandion expects to start a Phase 1b/2a study in ulcerative colitis in the middle of this year. A Phase 2 study in lupus is on track to start in the second half of 2021. There’s more in the Pandion pipeline as the biotech demonstrated this week during the annual scientific meeting of the Network for Pancreatic Organ Donors with Diabetes. The biotech presented promising preclinical data for another pipeline candidate, PT-001, in type 1 diabetes. The drug is designed to hit PD-1, an inhibitory receptor on T cells that has the effect of attenuating an immune response when activated. In the mouse study, the Pandion drug delayed the onset of hyperglycemia. Other companies pursuing Treg-targeting drugs include Sonoma Biotherapeutics and GentiBio. Both startups, which launched last year, are still in preclinical development. For Merck, the Pandion deal represents an opportunity to shore up an immunology portfolio anchored by Remicade and Simponi, inflammatory disease drugs that came to the company via its 2009 acquisition of Schering Plough. Merck shares commercialization of those drugs with Johnson & Johnson. In the nine months ending Sept. 30, 2020, Merck reported $615 million in sales for Simponi and $242 million in Remicade sales. Revenue for both drugs has been slipping due to competition from biosimilars. “This acquisition builds upon Merck’s strategy to identify and secure candidates with differentiated and potentially foundational characteristics,” Dean Li, president of Merck Research Laboratories, said in a prepared statement. “Pandion has applied its TALON technology to develop a robust pipeline of candidates designed to rebalance the immune response with potential applications across a wide array of autoimmune diseases.” Merck and Pandion expect to close the transaction in the first half of this year. If another buyer emerges with a better offer and Pandion terminates Merck’s deal, the biotech must pay the pharma giant a $65 million termination fee, according to a securities filing. 54

LAW 360 https://www.law360.com/newjersey/articles/1358810/covington-skadden-rep-merck-s-1-9b-pandion-buy Covington, Skadden Rep Merck’s $1.9B Pandion Buy (February 25, 2021, 2:58 PM EST) Merck & Co. said Thursday it’s buying clinical-stage autoimmune and inflammatory disease biotechnology group Pandion Therapeutics Inc. in a $1.85 billion deal guided by Covington & Burling and Skadden. Watertown, Massachusetts-based Pandion said it’s agreed to be bought by the multinational pharmaceutical at $60 per share, a premium of 134% from its closing share price Wednesday of $25.63, and a more than 233% premium over its $18-per-share debut on the Nasdaq in July. We are proud that Merck recognized our team’s innovation and drive in creating a pipeline of diverse candidates that activate natural immune regulatory mechanisms and thereby have thereby have the potential to achieve better clinical responses for patients,” said Dr. Rahul Kakkar, chief executive officer, Pandion Therapeutics. BIOPROCESS INTERNATIONAL 55

https://bioprocessintl.com/bioprocess-insider/merck-buying-pandion-therapeutics-in-1-85bn-deal/ Merck buying Pandion Therapeutics in $1.85bn deal by Dan StantonThursday, February 25, 2021 7:18 am Merck & Co. will add a pipeline of precision immune modulators and a drug design and discovery platform through the proposed acquisition of Pandion . The deal, announced today, will see Merck & Co. pay $60 per share in cash for the Cambridge, Massachusetts-based firm, totalling roughly $1.85 billion. Pandion is developing therapies for the tissue-specific treatment of patients with autoimmune and inflammatory diseases and organ transplants. Its lead candidate, bispecific antibody PT101, is in early-phase clinical trial for ulcerative colitis (UC) and systemic lupus erythematosus. “Pandion grew out of our founders’ personal and scientific mission to change the way patients living with autoimmune diseases are treated. In just a few years, we have taken that mission from idea to clinical proof of mechanism with PT101, our lead IL-2 mutein,” Pandion’s CEO Rahul Kakkar said in a statement. Merck will also add Pandion’s owns Therapeutic Autoimmune reguLatOry proteiN (TALON) drug design platform. The firm’s TALON programs are based on immunomodulatory effector modules that act at known regulatory control nodes within the immune system. These have also been investigated in combination with an effector module with a tissue-targeted tether module in a bifunctional format to guide delivery of the effector to a targeted tissue, resulting in an accelerated pipeline of candidates for the treatment of autoimmune and inflammatory diseases. “Pandion has applied its TALON technology to develop a robust pipeline of candidates designed to re-balance the immune response with potential applications across a wide array of autoimmune diseases,” said Dean Li, president of Merck Research Laboratories. Pandion does not own its own manufacturing facilities but has inked deals with third-parties to support its candidates. In 2018, the firm – fresh from a $58 million Series A financing – contracted Just Biotherapeutics for the development and clinical manufacturing of PT101. PHARMACEUTICAL TECHNOLOGY 56

https://www.pharmaceutical-technology.com/news/merck-pandion-therapeutics-agreement/ Merck signs agreement to buy Pandion Therapeutics for $1.85bn 26 February 2021 (Last Updated February 26th, 2021 08:35) Merck has agreed to acquire biotechnology company Pandion Therapeutics for approximately $1.85bn to expand its portfolio of autoimmune disease-targeting candidates. Under the agreement, Merck will initiate a tender offer through its subsidiary to acquire all Pandion’s outstanding shares. Pandion is developing new therapeutics to address the unmet needs of patients living with autoimmune and inflammatory diseases. The company’s lead candidate PT101 completed a Phase Ia clinical trial last year. It is an engineered IL-2 mutein fused to a protein backbone designed to activate and expand regulatory T-cells selectively to treat autoimmune diseases. Pandion’s portfolio also includes PD-1 agonists that are in the development stage for numerous autoimmune diseases. Merck Research Laboratories president Dr Dean Li said: “This acquisition builds upon Merck’s strategy to identify and secure candidates with differentiated and potentially foundational characteristics. “Pandion has applied its TALON technology to develop a robust pipeline of candidates designed to re-balance the immune response with potential applications across a wide array of autoimmune diseases.” Once the tender offer concludes, the acquisition subsidiary of Merck will merge into Pandion. The acquisition should conclude in the first half of this year. 57

Pandion Therapeutics CEO Dr Rahul Kakkar said: “We believe Merck is well-positioned to bring our novel approach to the millions of those living with autoimmune diseases, and we look forward to seeing these molecules progress in the clinic.” In October 2019, Pandion and Astellas Pharma signed a $795m agreement for the research, development and commercialisation of locally acting immunomodulators to treat Type 1 diabetes, as well as other autoimmune diseases of the pancreas. PHARMACEUTICAL BUSINESS REVIEW https://www.pharmaceutical-business-review.com/news/merck-signs-agreement-to-purchase-pandion-therapeutics/ Merck signs agreement to purchase Pandion Therapeutics By PBR Staff Writer 26 Feb 2021 World’s leading biopharmaceutical firm Merck has announced that it has entered into a definitive agreement to purchase Pandion Therapeutics through a subsidiary, for a total equity value of about $1.85m. 58

The office of Merck, located in Upper Gwynedd Township, Montgomery County, Pennsylvania. Credit: Montgomery County Planning Commission Biopharmaceutical firm Merck has announced that it has entered into a definitive agreement to purchase Pandion Therapeutics through a subsidiary, for a total equity value of about $1.85m. Through its subsidiary, Merck will initiate a tender offer to acquire all outstanding Pandion shares, under the terms of the acquisition agreement. The tender offer closes subject to certain conditions. Merck’s subsidiary will be merged into Pandion once the tender offer is completed successfully. Close of transaction is expected to occur in the first half of 2021. Clinical-stage biotechnology company Pandion develops novel therapeutics that are designed to address the unmet needs of patients suffering from autoimmune diseases. Merck Research Laboratories president Dean Y. Li said: “This acquisition builds upon Merck’s strategy to identify and secure candidates with differentiated and potentially foundational characteristics. Pandion has applied its TALON technology to develop a robust pipeline of candidates designed to re-balance the immune response with potential applications across a wide array of autoimmune diseases.” Pandion is developing precision immune modulators that target critical immune control nodes. Heading this strike force is Pandion’s lead candidate PT101, an engineered IL-2 mutein fused to a protein backbone. 59

PT101 selectively activates and expands regulatory T cells (Tregs) and can be utilised for the potential treatment of ulcerative colitis and other autoimmune diseases. Pandion announced earlier this year that PT101 had completed a Phase 1a clinical trial, achieving its primary objective of safety and tolerability. Pandion Therapeutics CEO Rahul Kakkar said: “Pandion grew out of our founders’ personal and scientific mission to change the way patients living with autoimmune diseases are treated. In just a few years, we have taken that mission from idea to clinical proof of mechanism with PT101, our lead IL-2 mutein. We are proud that Merck has recognised our team’s innovation and drive in creating a pipeline of diverse candidates that activate natural immune regulatory mechanisms and thereby have the potential to achieve better clinical responses for patients. “We believe Merck is well positioned to bring our novel approach to the millions of those living with autoimmune diseases, and we look forward to seeing these molecules progress in the clinic.” Merck’s financial advisor is Credit Suisse Securities (USA) LLC, while its legal advisor is Covington & Burling LLP. Pandion’s financial advisor is Centerview Partners LLC, while Skadden, Arps, Slate, Meagher & Flom LLP acted as legal advisor. GENE ONLINE NEWS https://geneonline.news/en/merck-acts-on-its-post-keytruda-plan-spends-1-85b-to-acquire-pandion-for-autoimmune-therapies/ Merck Acts on its Post-Keytruda Plan, Spends $1.85B to Acquire Pandion for Autoimmune Therapies 2021-02-25—by Ruchi Jhonsa 60

Pandion’s short run as independent biotech is coming to an end. Almost two years after coming out of stealth mode, Merck and Co. is acquiring the company for its impressive pipeline of autoimmune disease therapies. The $1.85 billion cash deal will buy Merck a promising drug in early-stage testing for ulcerative colitis and lupus and a set of preclinical drugs for other autoimmune diseases. Typically, treatment for diseases where the immune system starts killing its own cells involves suppressing the entire immune system. While this is effective at dialing down the negative effects of the immune system, it can make the body more susceptible to infections and inflammation. Pandion is developing a novel class of drugs that work by increasing the population of T-regulatory cells (T-regs) or by precisely activating PD-1 checkpoint protein. The novelty of the drugs is that they act at the local site of disease, avoiding a systemic immunosuppression characteristic of current treatments. Pandion’s quick progress with the drugs fetched the company $58 million in series A financing in 2018, $80 million financing in April last year, and $135 million in an IPO last July. Pandion’s Autoimmune Pipeline Regulatory T cells, or Tregs, play a key role in modulating other cells in the immune system, so they don’t attack the body’s own tissues. The failure of Tregs can lead to autoimmune diseases. Pandion’s lead drug candidate, PT101, is designed to selectively activate and expand Tregs. The drug comprises two components: a protein backbone and an engineered version of interleukin 2 protein that activates Tregs at low doses and proinflammatory natural killer (NK) cells at higher doses. An ideal autoimmune drug would be the one that activates only Tregs and suppresses NK cells. 61

In Phase 1 study, Pandion’s drug did exactly that. It prevented the expansion of NK cells and triggered activation and expansion of Tregs. A 3.6-fold increase in Tregs over baseline and an expansion of a subset of activated Tregs, called CD25 bright Tregs, was observed following drug treatment. With this data, Pandion is moving on to Phase1b/2a study for ulcerative colitis and Phase 2 trial for Lupus in the mid and second half of this year, respectively. Besides PT101, the company is also developing drugs to activate PD-1 receptors, a protein that is dear to many cancer immunotherapies. However, unlike checkpoint inhibitors, Pandion’s drugs will activate the protein, making the immune system unresponsive to its own cells. Pandion’s PT001 is designed to activate PD-1 receptors in gut tissues, which is directed at treating gut-restricted autoimmune diseases. What is in it for Merck? At the 39th Annual JPM conference last month, Merck discussed its growth plans after losing market exclusivity for Keytruda. The company was clear that it needs to move away from Keytruda’s shadow and start diversifying its portfolio. In that wake, Merck signed deals with VelosBio and Seattle Genetics for antibody-drug conjugates last year. The deal with Pandion is also part of the same plan and is expected to reap Merck a sizable profit in the post-Keytruda era. As per the report published by Fior Markets, the global autoimmune treatment market is expected to grow from $3.28 billion in 2017 to $7.95 billion in 2025. If Pandion’s drug stands the test of safety and efficacy, it might as well become the next Keytruda. BOSTON GLOBE [Published Bloomberg article with “Cambridge” in the headline} https://www.bostonglobe.com/2021/02/25/business/merck-buy-cambridge-autoimmune-drug-developer-pandion-185-billion/ Merck to buy Cambridge autoimmune drug developer Pandion for $1.85 billion By Eric Pfanner Bloomberg,Updated February 25, 2021 62

The Merck corporate headquarters in New Jersey.SETH WENIG/ASSOCIATED PRESS (Bloomberg) — Merck & Co. agreed to acquire Pandion Therapeutics Inc., a biotechnology company developing therapies for autoimmune diseases, for $1.85 billion. The agreement, worth $60 per share in cash, is at more than double the Cambridge, Massachusetts-based company’s closing price Wednesday. Pandion’s potential treatment for ulcerative colitis and other autoimmune diseases, known as PT101, has completed a Phase 1a clinical trial that achieved its primary objective of safety and tolerability, the companies said in a statement. The planned purchase comes about three weeks after Merck named Robert M. Davis to succeed Kenneth Frazier as chief executive officer, and about a month after it ended its Covid-19 vaccine program. BOSTON BUSINESS JOURNAL https://www.bizjournals.com/boston/news/2021/02/25/merck-acquires-newly-public-biotech-pandion-tx.html 63

Merck acquires newly public Watertown biotech for $1.85B By Rowan Walrath – Inno Associate Editor, Boston Business Journal Feb 25, 2021 Life sciences giant Merck is buying Pandion Therapeutics, the autoimmune disorder-focused biotech that went public last July in one of the largest IPOs of 2020, for $1.85 billion in stock. Pandion’s stock price nearly doubled upon the news of the Thursday morning announcement. Launched in 2018 by former executives at Pfizer and Biogen, Pandion aims to treat autoimmune disorders by targeting critical immune control nodes with immune system-modulating molecules. Its lead candidate, a drug that targets T-cells to treat ulcerative colitis and other autoimmune diseases, completed a Phase 1a clinical trial earlier this year. “We are proud that Merck has recognized our team’s innovation and drive in creating a pipeline of diverse candidates that activate natural immune regulatory mechanisms and thereby have the potential to achieve better clinical responses for patients,” Pandion CEO Rahul Kakkar said in a statement. “We believe Merck is well positioned to bring our novel approach to the millions of those living with autoimmune diseases, and we look forward to seeing these molecules progress in the clinic.” The acquisition is expected to be completed by early summer. Pandion employed 43 people full time as of last May, according to a securities filing. The company, which was incubated at LabCentral in Cambridge, moved to a 21,225-square-foot facility in Watertown last May. Pandion was the 19th-largest IPO in 2020, making its stock market debut at $135 million. Social Media Posts of Interest 64
23.2 million Twitter followers 3.9 million Twitter followers 9.62 million Facebook followers 764k Twitter followers 7.08 million Twitter followers 185k Twitter followers 65

182k Twitter followers 85.7k Twitter followers 148k Twitter followers 77.1k Twitter followers 88.7k Twitter followers 68.7k Twitter followers 66
67.6k Twitter followers 57.8k Twitter followers 64.5 Twitter followers 54.9k Twitter followers 61.5k Facebook followers 50.8k Twitter followers 67
30.7k Twitter followers 38.8k Twitter followers 35.3k Twitter followers 26.1k Twitter followers 33.9k Twitter followers 21.1k Twitter followers 68
17.7k Twitter followers 20.6k Twitter followers 17.1k Twitter followers 19.3k Twitter followers 18.3k Twitter followers 16.1k Twitter followers 69
13k Twitter followers 14.2k Twitter followers 13k Twitter followers 14.1k Twitter followers 13.5k Twitter followers 12k Twitter followers 70
10.4k Twitter followers 10.2k Twitter followers 11.4k Twitter followers 11.1k Twitter followers 11.1k Twitter followers 10.1k Twitter followers 11.1k Twitter followers 10.6k Twitter followers 8.39k Twitter followers 71
6.93k Twitter followers 8.2k Twitter followers 6.26k Twitter followers 7.56k Twitter followers 7.26k Twitter followers 5.97k Twitter followers 6.95k Twitter followers 5.8k Twitter followers 72

5.64k Twitter followers 4.43k Facebook followers 4.43k Facebook followers 5.59k Twitter followers 4.94k Twitter followers 4.332k Twitter followers 4.79k Twitter followers 3.93k Twitter followers 73
3.93k Twitter followers 3.44k Twitter followers 3.85k Twitter followers 3.41k Twitter followers 3.18k Twitter followers 3.73k Twitter followers 74
2.82k Twitter followers 3.18k Twitter followers 2.82k Twitter 2.98k Twitter followers 2.77k Twitter followers 2.82kTwitter 75
1.93k Twitter followers 2.48k Twitter followers 1.9k Twitter followers 1.85k Twitter followers 2.4k Twitter followers 1.35k Twitter followers 1.95k Twitter followers 1.26k Twitter followers 76
1.04k Twitter followers 1.04k Twitter followers 1.04k Twitter followers 77