Exhibit 99.1
| Next-Generation TCR Bispecifics (TCER) Targeting Peptide-HLA Antigens for the Treatment of Patients with Solid Tumors
S. Bunk1, M. Hofmann1, G. Pszolla1, F. Schwoebel1, M. Hutt1, F. Unverdorben1, N. Aschmoneit1, M. Mølhøj1, C. Wagner1, M. Jaworski1, C. Schraeder1, H. Schuster1, S. Missel1, T. Weinschenk2, D. Maurer1, C. Reinhardt2
1 Immatics Biotechnologies GmbH, Tuebingen, Germany, 2 Immatics N.V., Tuebingen, Germany |  |


Abstract ID: 1319 |  |
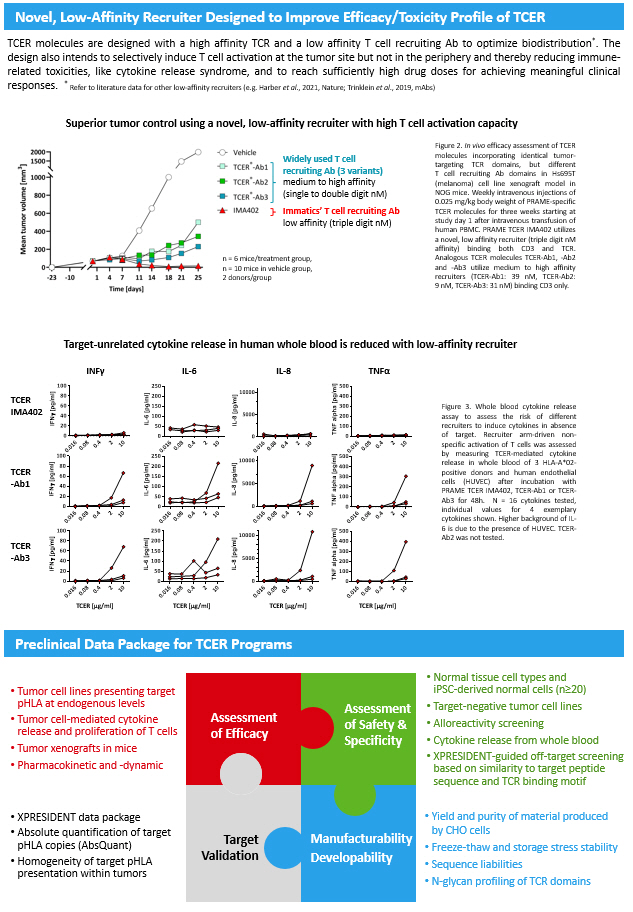
Next - Generation TCR Bispecifics (TCER) Targeting Peptide - HLA Antigens for the Treatment of Patients with Solid Tumors S. Bunk 1 , M. Hofmann 1 , G. Pszolla 1 , F. Schwoebel 1 , M. Hutt 1 , F. Unverdorben 1 , N. Aschmoneit 1 , M. Mølhøj 1 , C. Wagner 1 , M. Jaworski 1 , C. Schraeder 1 , H. Schuster 1 , S. Missel 1 , T. Weinschenk 2 , D. Maurer 1 , C. Reinhardt 2 T cell engaging receptor (TCER) – a Next - generation TCR Bispecific Format • Comparison of seven different TCR bispecific formats revealed highest anti - tumor potency for TCER format together with a 2+1 format that failed specificity requirements • High affinity TCR domains targeting peptide - HLA tumor antigens are generated by specificity - controlled affinity maturation • Novel, low affinity Ab domains developed for T cell recruitment aim to improve efficacy while minimizing toxicity of TCER molecules as demonstrated in tumor xenograft models in mice and by cytokine release in human whole blood • IgG Fc region incorporated into TCER format for the extension of serum half - life and improved manufacturability • Comprehensive preclinical data package including in vivo tumor models are generated for each TCER program PRAME TCER IMA402 with encouraging preclinical and CMC data • In vivo studies in mice demonstrate dose - dependent anti - tumor activity of IMA402 and that s ufficiently high drug doses are key to achieving sustained anti - tumor response over prolonged time period • IMA402 demonstrates a serum half - life of ≈ 8 days in mice suggesting a favorable dosing regimen and prolonged drug exposure at therapeutic levels when compared to TCR bispecifics lacking half - life extension • TCER IMA402 is manufactured by utilizing standard processes of mAb production resulting in high titer , protein quality and stability while unwanted side products are absent, a unique feature of our TCER format Conclusion Pharmacodynamic and Pharmacokinetic of PRAME TCER (IMA402) Background – Overcoming Challenges of T cell Engaging Bispecifics Manufacturing of PRAME TCER (IMA402) CMC data support antibody - like manufacturability and developability • Manufacturing in Chinese Hamster Ovary (CHO) cells able to process natural TCR glycosylation • High titer (>3.5 g/L) and good stability allowing liquid formulation • Manufacturing process development completed • Manufacturing advantages of TCER format: no formation of typical but unwanted Hole - Hole and Knob - Knob side products T cell engaging bispecifics have emerged as a promising therapeutic opportunity for patients with solid cancers . However, challenges related to target specificity and drug safety remain and many efforts are being made to generate optimized molecules with improved pharmacodynamics while reducing T cell engager - associated toxicities . We have developed a pipeline of bispecific T cell engaging receptor (TCER) molecules comprising a T cell receptor (TCR) for giving access to intracellular tumor antigens presented as peptide - HLA molecules . The next - generation design of TCER is established through a novel, low - affinity T cell recruiting antibody aiming at conferring a favorable drug safety profile while enabling a highly potent anti - tumor response . TCER molecules are further equipped with an effector function - silenced Fc region for prolongation of serum half - life . Target - unrelated cytokine release in human whole blood is reduced with low - affinity recruiter Abstract ID: 1319 1 Immatics Biotechnologies GmbH, Tuebingen, Germany, 2 Immatics N . V . , Tuebingen, Germany TCER IMA402 TCER - Ab1 TCER - Ab3 INF γ IL - 6 IL - 8 TNFα TCER [µg/ml] TCER [µg/ml] TCER [µg/ml] TCER [µg/ml] Figure 3 . Whole blood cytokine release assay to assess the risk of different recruiters to induce cytokines in absence of target . Recruiter arm - driven non - specific activation of T cells was assessed by measuring TCER - mediated cytokine release in whole blood of 3 HLA - A* 02 - positive donors and human endothelial cells (HUVEC) after incubation with PRAME TCER IMA 402 , TCER - Ab 1 or TCER - Ab 3 for 48 h . N = 16 cytokines tested, individual values for 4 exemplary cytokines shown . Higher background of IL - 6 is due to the presence of HUVEC . TCER - Ab 2 was not tested . Contact information: info@immatics.com Discovery and Affinity Maturation of TCRs Required for TCER Generation *high potency of 2+1 format D was caused by pronounced unspecific reactivity against T2 Killing of target - positive tumor cells by different TCR Bispecifics TCER Format Exhibits Highest Potency Combined with Specificity in Targeting pHLA Antigens TCER T cell recruiting antibody pHLA targeting TCR Fc region (silenced) with KiH technology High affinity (single digit nM ) TCR targeting tumor - specific peptide HLA molecules Low affinity (triple digit nM ) T cell recruiter against CD3/TCR Human IgG Fc region (silenced) with knob - in - hole technology for half - life extension, favorable stability and manufacturability Proprietary TCER format consisting of three distinct elements designed for optimal efficacy and minimal toxicity risk in patients Cytotoxic lytic granules T umor cell A ctivated T cell TCER binds to target, recruits and activates T cells and initiates tumor cell killing 2 1 3 The Next - generation of TCR Bispecifics – TCER Preclinical Data Package for TCER Programs Widely used T cell recruiting Ab (3 variants) medium to high affinity (single to double digit nM ) Immatics’ T cell recruiting Ab low affinity (triple digit nM ) TCER molecules are designed with a high affinity TCR and a low affinity T cell recruiting Ab to optimize biodistribution * . The design also intends to selectively induce T cell activation at the tumor site but not in the periphery and thereby reducing immune - related toxicities, like cytokine release syndrome, and to reach sufficiently high drug doses for achieving meaningful clinical responses . Figure 2 . In vivo efficacy assessment of TCER molecules incorporating identical tumor - targeting TCR domains, but different T cell recruiting Ab domains in Hs 695 T (melanoma) cell line xenograft model in NOG mice . Weekly intravenous injections of 0 . 025 mg/kg body weight of PRAME - specific TCER molecules for three weeks starting at study day 1 after intravenous transfusion of human PBMC . PRAME TCER IMA 402 utilizes a novel, low affinity recruiter ( triple digit nM affinity) binding both CD 3 and TCR . Analogous TCER molecules TCER - Ab 1 , - Ab 2 and - Ab 3 utilize medium to high affinity recruiters (TCER - Ab 1 : 39 nM , TCER - Ab 2 : 9 nM , TCER - Ab 3 : 31 nM ) binding CD 3 only . Novel, Low - Affinity Recruiter Designed to Improve Efficacy/Toxicity Profile of TCER n = 6 mice/treatment group, n = 10 mice in vehicle group, 2 donors/group Superior tumor control using a novel, low - affinity recruiter with high T cell activation capacity * R efer to literature data for other low - affinity recruiters (e.g. Harber et al ., 2021, Nature; Trinklein et al ., 2019, mAbs ) A B C D E F D* TCER A C B E F • TCR variable domains ( scTv format) are stabilized and optimized for facilitated production in mammalian cells • This enables combination with various bispecific platforms • Low binding affinity of natural TCRs is increased by at least 1,000 - fold while retaining high specificity • XPRESIDENT’s immunopeptidome database is essential for specificity - controlled affinity maturation • XCEPTOR for TCR Discovery, Engineering & Validation • Generation of multiple TCR candidates per target • Unique XPRESIDENT - guided on - and off - target toxicity screening Figure 4 . In vivo efficacy of IMA 402 in large ( ≈ 195 mm 3 average tumor volume ) melanoma cell line - derived tumors in MHC I/II knock - out NSG mice over a prolonged observation period of 71 days . Weekly intravenous injections of IMA 402 starting at study day 1 after intravenous transfusion of human PBMC . Treatment was discontinued when complete response was noted . Median values for n = 6 mice/group, 2 donors/group . Figure 5 . Pharmacokinetic analysis of IMA 402 in mice . NOG mice received a single intravenous injection of IMA 402 ( 2 mg/kg) . TCER plasma concentrations at different time points were determined by ELISA detecting binding of IMA 402 to the PRAME target via pHLA . The integrity of the molecule was confirmed via aV L or aFc detection . Terminal half - life (t 1 / 2 ) was calculated via linear regression of time points between 24 h and 360 h (n= 3 per timepoint, mean ц SD) . Figure 1 : Design and cytotoxic activity of TCER and six alternative TCR bispecific formats . Right panel : Based on an affinity - maturated TCR (TCR variable domains in red) specific for the HIV peptide SLYNTVATL presented on HLA - A* 02 , TCER and six alternative TCR bispecific formats were generated using the identical Fab region for T cell recruitment (variable Ab domains in blue) . Silenced IgG 1 Fc domains with knob - into - hole ( KiH ) were utilized to facilitate heterodimerization . Left panel : PBMC - mediated cytotoxicity of TCR bispecific formats against HLA - A* 02 – positive T 2 cells loaded with HIV peptide . Cytotoxicity was calculated based on LDH release during 24 h coculture of PBMC and T 2 . LDH content of T 2 cells alone was used as 100 % cytotoxicity reference leading to underestimated cytotoxicity values due to T 2 cell proliferation . Figure 6 . Theoretical product - related impurities such as hole - hole and knob - knob homodimers, are considered inactive due to absence of variable IgG or TCR domains . Total mass analysis by mass spectrometry ( MS) of transiently and CHO stably expressed IMA 402 has demonstrated lack of hole - hole and knob - knob homodimers in both Protein - L and Protein - A captured TCER fractions . IMA 402 was analyzed by MS after complete deglycosylation with PNGase F . With a 100 % correct chain pairing of the TCER knob and hole chains, IMA 402 can be captured with standard Protein - A chromatography, and n o specific downstream processing steps are needed for the removal of these product - related impurities . Detected by MS Not detected by MS • XPRESIDENT data package • Absolute quantification of target pHLA copies ( AbsQuant ) • Homogeneity of target pHLA presentation within tumors • Tumor cell lines presenting target pHLA at endogenous levels • Tumor cell - mediated cytokine release and proliferation of T cells • Tumor xenografts in mice • Pharmacokinetic and - dynamic • Normal tissue cell types and iPSC - derived normal cells (n≥20) • Target - negative tumor cell lines • Alloreactivity screening • Cytokine release from whole blood • XPRESIDENT - guided off - target screening based on similarity to target peptide sequence and TCR binding motif • Yield and purity of material produced by CHO cells • Freeze - thaw and storage stress stability • Sequence liabilities • N - glycan profiling of TCR domains Target Validation Assessment of Efficacy Assessment of Safety & Specificity Manufacturability Developability V α VL VH V β V α VL VH V β MW aver., SS , Δm = +0 Da Mass Intensity Hole - Hole Knob - Knob TCER: 100% correct chain pairing V α V α VH VH VL VL V β V β Hole chain Knob chain PNGase F digest Knob - Hole TCER