
Population Pharmacokinetics and Safety of sozinibercept (OPT-302), an anti-VEGF-C/-D ‘trap’ in Patients with Retinal Vascular Diseases Dante J. Pieramici, MD California Retina Consultants, Santa Barbara, CA ASRS, Seattle, 2023 Exhibit 99.1

Disclosures Presenter’s Financial Disclosures: - Adverum (C), Gemini, Genentech, Inc., Iveric Bio, NGM, Opthea (C), Regeneron, Regenxbio • This presentation will discuss IRB/IEC approved research of an investigational product. (C): Consultant; (S): Stock/shareholder; (R): Grants/Research Support
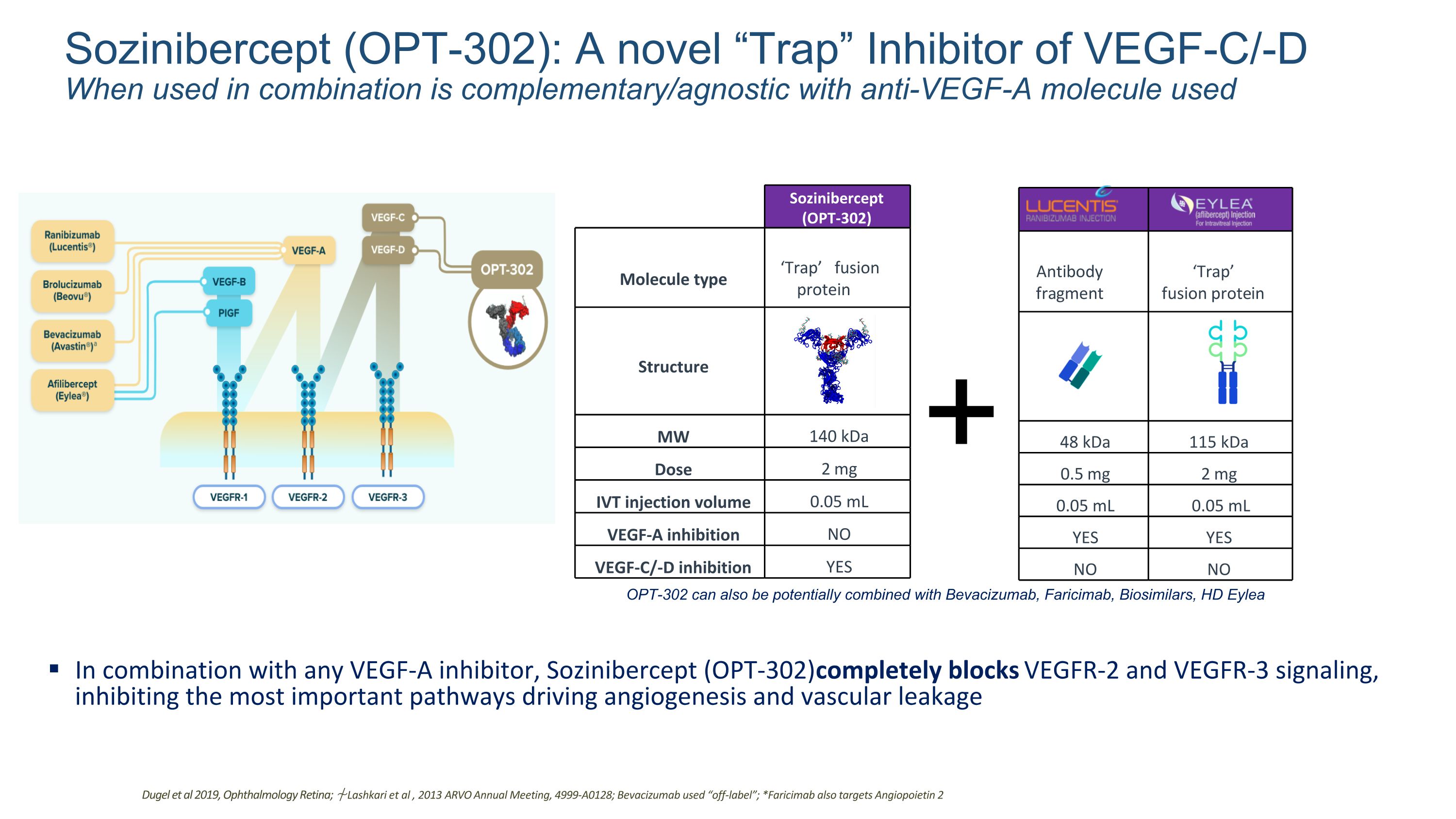
Sozinibercept (OPT-302): A novel “Trap” Inhibitor of VEGF-C/-D When used in combination is complementary/agnostic with anti-VEGF-A molecule used In combination with any VEGF-A inhibitor, Sozinibercept (OPT-302) completely blocks VEGFR-2 and VEGFR-3 signaling, inhibiting the most important pathways driving angiogenesis and vascular leakage VEGF-A inhibition elevates VEGF-C and VEGF-D which may contribute Dugel et al 2019, Ophthalmology Retina; ⍭ Lashkari et al , 2013 ARVO Annual Meeting, 4999-A0128; Bevacizumab used “off-label”; *Faricimab also targets Angiopoietin 2 Molecule type ‘Trap’ fusion protein Structure MW 140 kDa Dose 2 mg IVT injection volume 0.05 mL VEGF-A inhibition NO VEGF-C/-D inhibition YES + Antibody fragment ‘Trap’ fusion protein 48 kDa 115 kDa 0.5 mg 2 mg 0.05 mL 0.05 mL YES YES NO NO OPT-302 can also be potentially combined with Bevacizumab, Faricimab, Biosimilars, HD Eylea Sozinibercept (OPT-302)
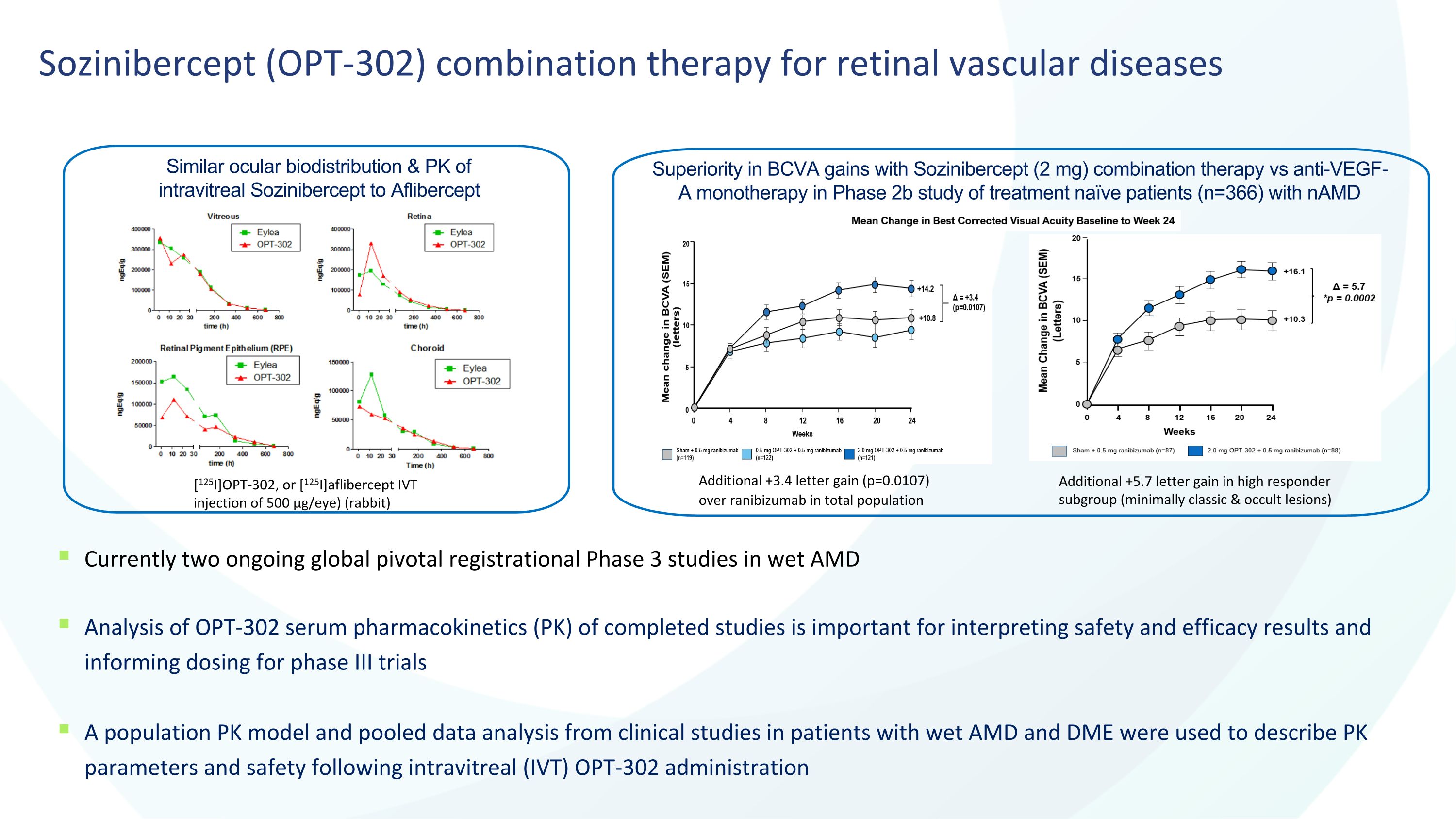
4 Sozinibercept (OPT-302) combination therapy for retinal vascular diseases Currently two ongoing global pivotal registrational Phase 3 studies in wet AMD Analysis of OPT-302 serum pharmacokinetics (PK) of completed studies is important for interpreting safety and efficacy results and informing dosing for phase III trials A population PK model and pooled data analysis from clinical studies in patients with wet AMD and DME were used to describe PK parameters and safety following intravitreal (IVT) OPT-302 administration Similar ocular biodistribution & PK of intravitreal Sozinibercept to Aflibercept Superiority in BCVA gains with Sozinibercept (2 mg) combination therapy vs anti-VEGF-A monotherapy in Phase 2b study of treatment naïve patients (n=366) with nAMD Additional +3.4 letter gain (p=0.0107) over ranibizumab in total population [125I]OPT-302, or [125I]aflibercept IVT injection of 500 µg/eye) (rabbit) Additional +5.7 letter gain in high responder subgroup (minimally classic & occult lesions)
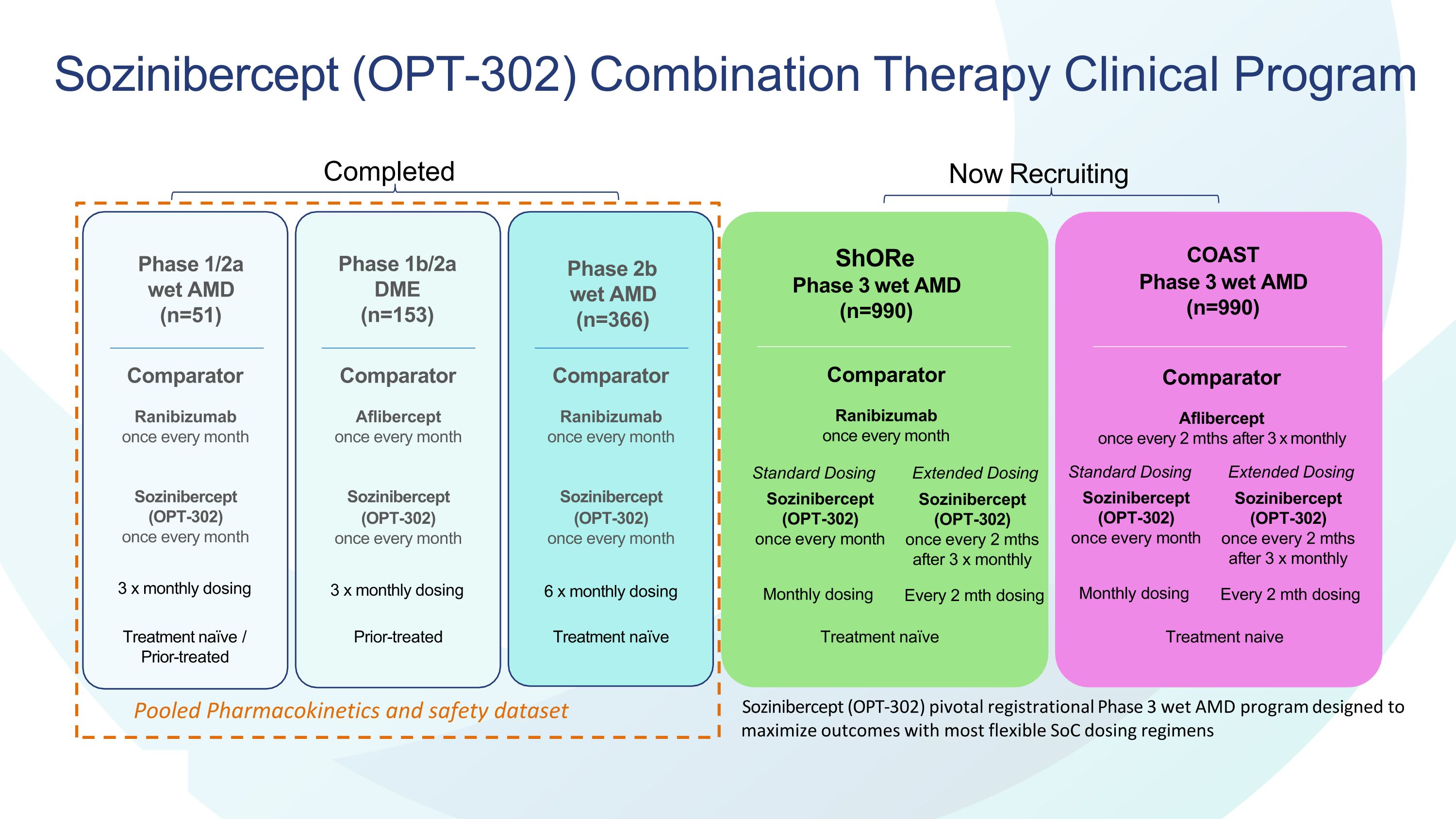
Sozinibercept (OPT-302) Combination Therapy Clinical Program Sozinibercept (OPT-302) pivotal registrational Phase 3 wet AMD program designed to maximize outcomes with most flexible SoC dosing regimens . ShORe Phase 3 wet AMD (n=990) Comparator Ranibizumab once every month COAST Phase 3 wet AMD (n=990) Standard Dosing Sozinibercept (OPT-302) once every month Monthly dosing Extended Dosing Comparator Aflibercept once every 2 mths after 3 x monthly Phase 1/2a wet AMD (n=51) Comparator Ranibizumab once every month Sozinibercept (OPT-302) once every month 3 x monthly dosing Comparator Aflibercept once every month Sozinibercept (OPT-302) once every month 3 x monthly dosing Phase 1b/2a DME (n=153) Comparator Ranibizumab once every month Sozinibercept (OPT-302) once every month 6 x monthly dosing Treatment naïve / Prior-treated Treatment naïve Treatment naïve Treatment naive Prior-treated Phase 2b wet AMD (n=366) Now Recruiting Completed Pooled Pharmacokinetics and safety dataset Sozinibercept (OPT-302) once every 2 mths after 3 x monthly Every 2 mth dosing Standard Dosing Sozinibercept (OPT-302) once every month Monthly dosing Extended Dosing Sozinibercept (OPT-302) once every 2 mths after 3 x monthly Every 2 mth dosing
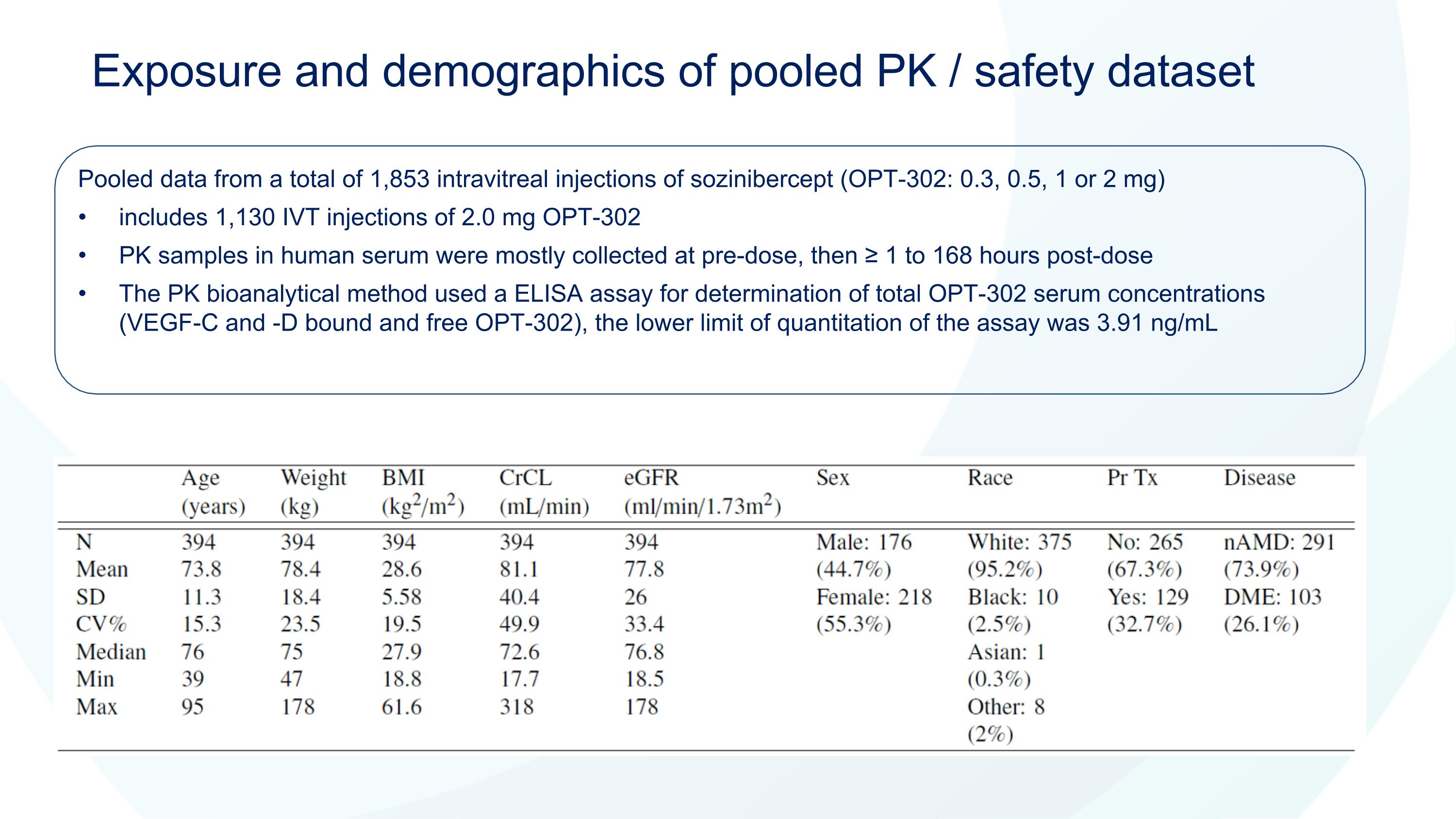
Exposure and demographics of pooled PK / safety dataset Pooled data from a total of 1,853 intravitreal injections of sozinibercept (OPT-302: 0.3, 0.5, 1 or 2 mg) includes 1,130 IVT injections of 2.0 mg OPT-302 PK samples in human serum were mostly collected at pre-dose, then ≥ 1 to 168 hours post-dose The PK bioanalytical method used a ELISA assay for determination of total OPT-302 serum concentrations (VEGF-C and -D bound and free OPT-302), the lower limit of quantitation of the assay was 3.91 ng/mL
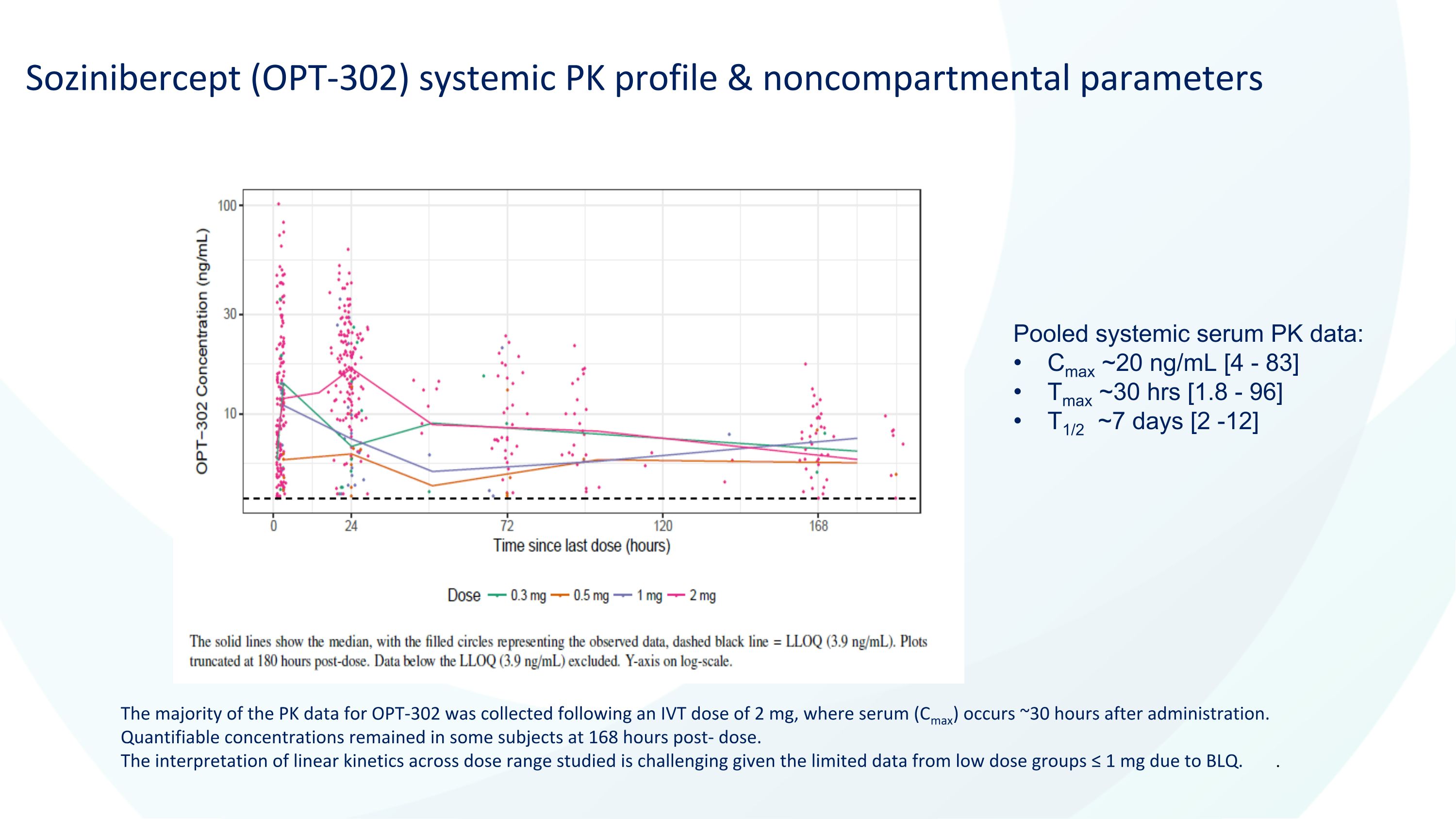
Sozinibercept (OPT-302) systemic PK profile & noncompartmental parameters The majority of the PK data for OPT-302 was collected following an IVT dose of 2 mg, where serum (Cmax) occurs ~30 hours after administration. Quantifiable concentrations remained in some subjects at 168 hours post- dose. The interpretation of linear kinetics across dose range studied is challenging given the limited data from low dose groups ≤ 1 mg due to BLQ. . Pooled systemic serum PK data: Cmax ~20 ng/mL [4 - 83] Tmax ~30 hrs [1.8 - 96] T1/2 ~7 days [2 -12]
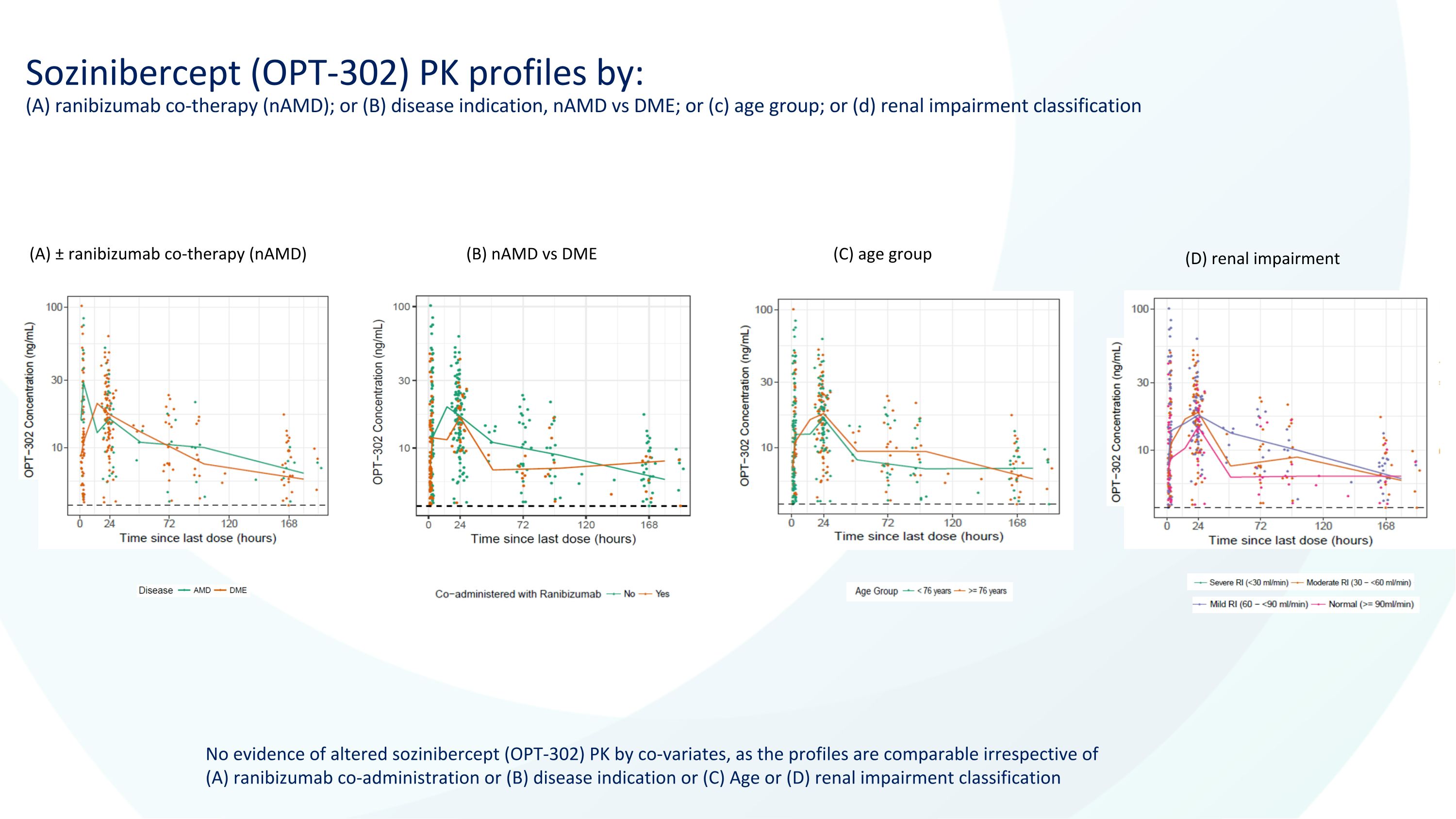
Sozinibercept (OPT-302) PK profiles by: (A) ranibizumab co-therapy (nAMD); or (B) disease indication, nAMD vs DME; or (c) age group; or (d) renal impairment classification No evidence of altered sozinibercept (OPT-302) PK by co-variates, as the profiles are comparable irrespective of (A) ranibizumab co-administration or (B) disease indication or (C) Age or (D) renal impairment classification (A) ± ranibizumab co-therapy (nAMD) (B) nAMD vs DME (C) age group (D) renal impairment

Vitreous Parameter Estimates from sozinibercept (OPT-302) Population PK Model The PK model retained the single distribution compartment and linear elimination. Absorption from the vitreous space was described by a first-order process. During selected dosing occasions, a small fraction, (≈7%) of the administered OPT-302 bypassed the vitreous compartment into the systemic circulation. This phenomena has previously been described for IVT ranibizumab using a population PK approach. The absorption of OPT-302 was the rate-limiting step, with the PK of OPT-302 via IVT administration described by ‘flip-flop’ kinetics. The model assumes no clearance of OPT-302 in the vitreous compartment. The M3 method was used during estimation given the BLQ data.
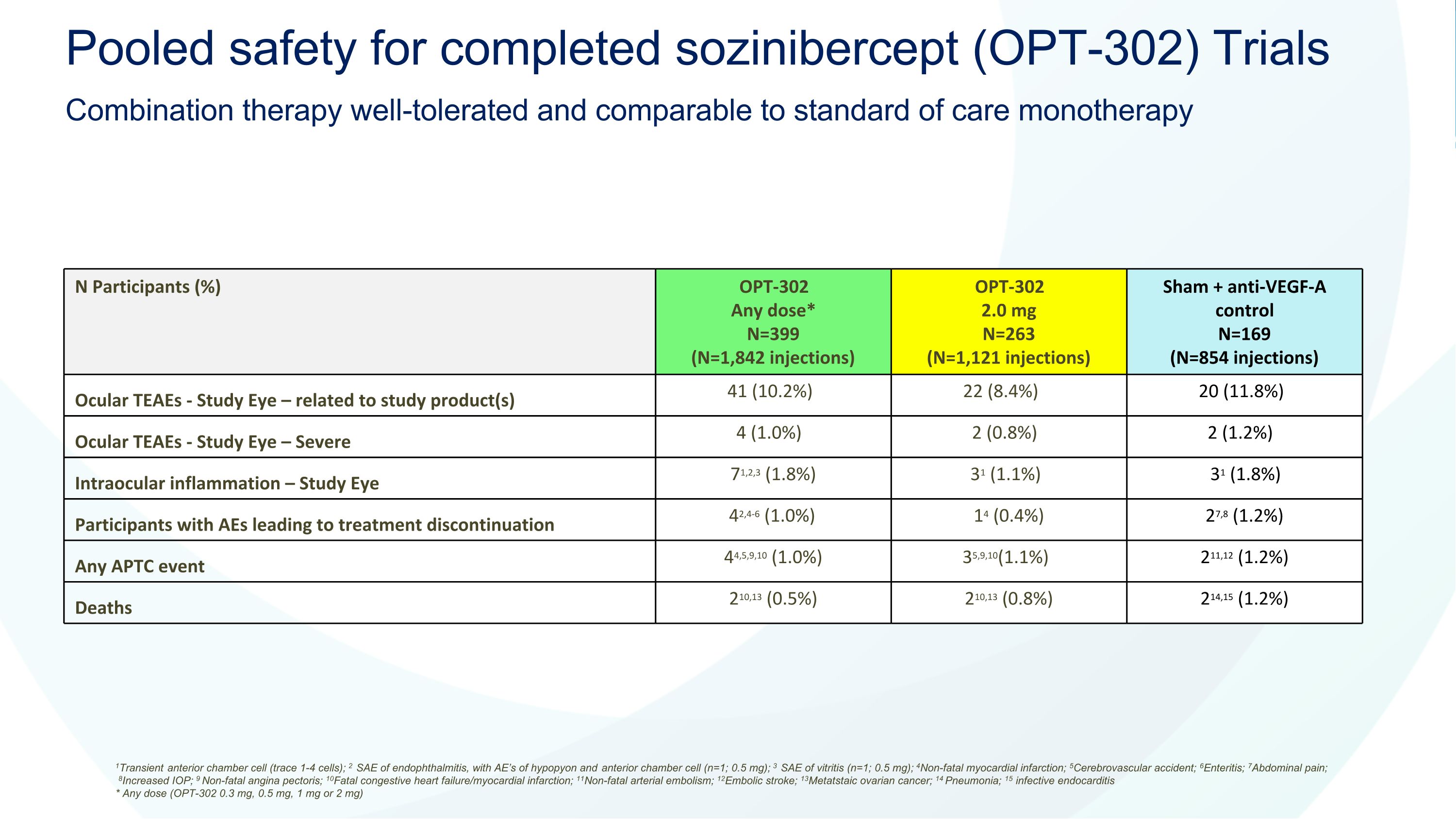
N Participants (%) OPT-302 Any dose* N=399 (N=1,842 injections) OPT-302 2.0 mg N=263 (N=1,121 injections) Sham + anti-VEGF-A control N=169 (N=854 injections) Ocular TEAEs - Study Eye – related to study product(s) 41 (10.2%) 22 (8.4%) 20 (11.8%) Ocular TEAEs - Study Eye – Severe 4 (1.0%) 2 (0.8%) 2 (1.2%) Intraocular inflammation – Study Eye 71,2,3 (1.8%) 31 (1.1%) 31 (1.8%) Participants with AEs leading to treatment discontinuation 42,4-6 (1.0%) 14 (0.4%) 27,8 (1.2%) Any APTC event 44,5,9,10 (1.0%) 35,9,10(1.1%) 211,12 (1.2%) Deaths 210,13 (0.5%) 210,13 (0.8%) 214,15 (1.2%) 1Transient anterior chamber cell (trace 1-4 cells); 2 SAE of endophthalmitis, with AE’s of hypopyon and anterior chamber cell (n=1; 0.5 mg); 3 SAE of vitritis (n=1; 0.5 mg); 4Non-fatal myocardial infarction; 5Cerebrovascular accident; 6Enteritis; 7Abdominal pain; 8Increased IOP; 9 Non-fatal angina pectoris; 10Fatal congestive heart failure/myocardial infarction; 11Non-fatal arterial embolism; 12Embolic stroke; 13Metatstaic ovarian cancer; 14 Pneumonia; 15 infective endocarditis * Any dose (OPT-302 0.3 mg, 0.5 mg, 1 mg or 2 mg) Pooled safety for completed sozinibercept (OPT-302) Trials Combination therapy well-tolerated and comparable to standard of care monotherapy
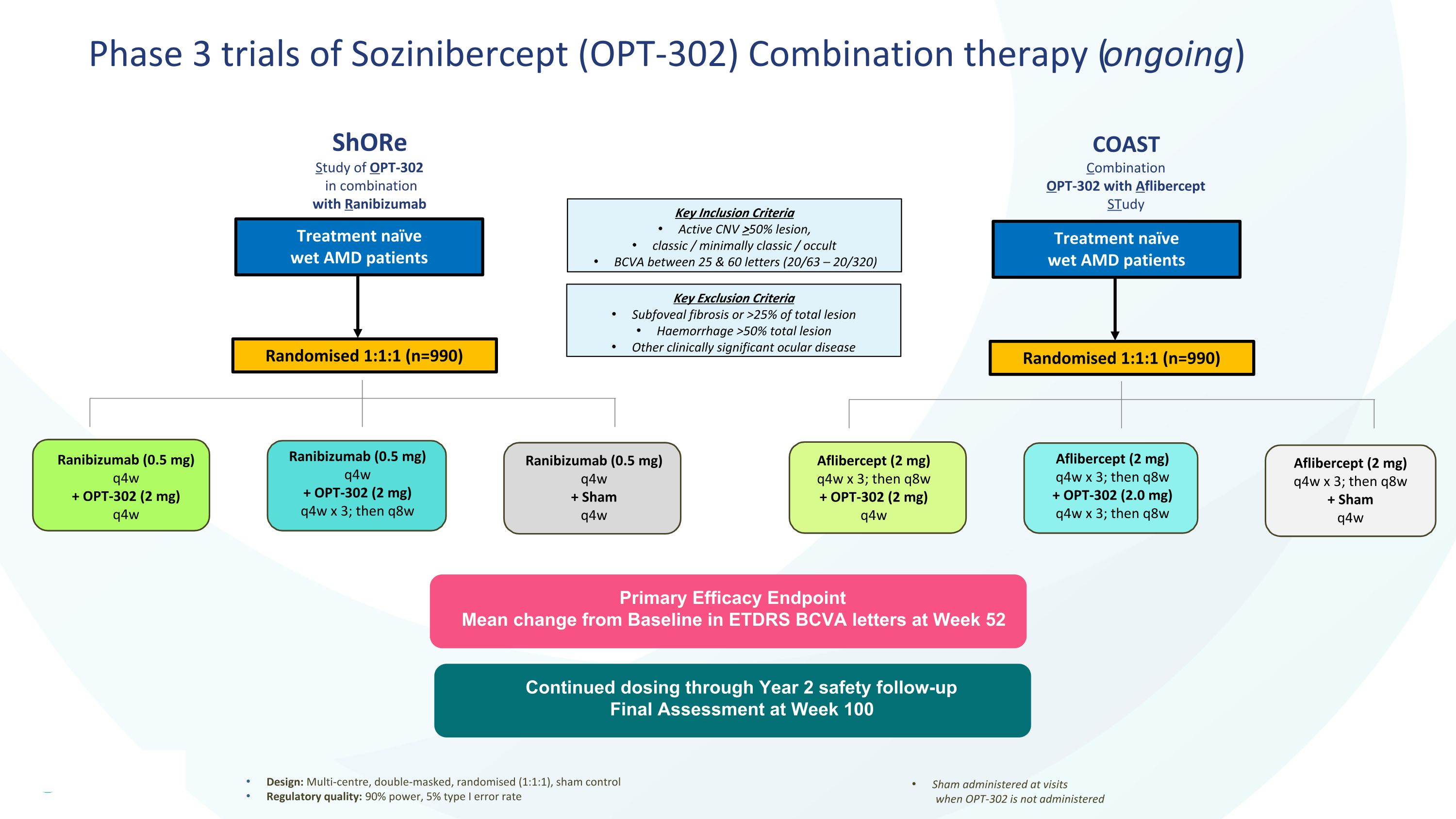
Phase 3 trials of Sozinibercept (OPT-302) Combination therapy (ongoing) Design: Multi-centre, double-masked, randomised (1:1:1), sham control Regulatory quality: 90% power, 5% type I error rate Sham administered at visits when OPT-302 is not administered Primary Efficacy Endpoint Mean change from Baseline in ETDRS BCVA letters at Week 52 Ranibizumab (0.5 mg) q4w + OPT-302 (2 mg) q4w Ranibizumab (0.5 mg) q4w + OPT-302 (2 mg) q4w x 3; then q8w Ranibizumab (0.5 mg) q4w + Sham q4w Treatment naïve wet AMD patients Key Exclusion Criteria Subfoveal fibrosis or >25% of total lesion Haemorrhage >50% total lesion Other clinically significant ocular disease Key Inclusion Criteria Active CNV >50% lesion, classic / minimally classic / occult BCVA between 25 & 60 letters (20/63 – 20/320) Randomised 1:1:1 (n=990) Aflibercept (2 mg) q4w x 3; then q8w + OPT-302 (2.0 mg) q4w x 3; then q8w Aflibercept (2 mg) q4w x 3; then q8w + Sham q4w Treatment naïve wet AMD patients Randomised 1:1:1 (n=990) Aflibercept (2 mg) q4w x 3; then q8w + OPT-302 (2 mg) q4w Continued dosing through Year 2 safety follow-up Final Assessment at Week 100 ShORe Study of OPT-302 in combination with Ranibizumab COAST Combination OPT-302 with Aflibercept STudy

Summary: Sozinibercept (OPT-302) pooled PK and safety The PK profiles of intravitreal sozinibercept (OPT-302) from completed studies in patients with nAMD and DME indicated: low systemic exposure within one week drug levels were mostly no longer detectable in serum estimated vitreous absorption half-life was ~4.6 days [95% CI 3.6-5.8] comparable to other IVT biologics the PK profile was unaltered by anti-VEGF-A co-therapy, disease indication (nAMD vs DME), age or renal function Pooled safety analysis shows soziniberept (OPT-302) combination therapy has a favourable safety and tolerability profile comparable to standard of care anti-VEGF-A monotherapy Promising treatment option for wet AMD currently in two pivotal registrational Phase 3 studies pooled PK and safety data support ShORe and COAST dosing regimens of 2 mg sozinibercept q4w or q8w following 3 loading doses