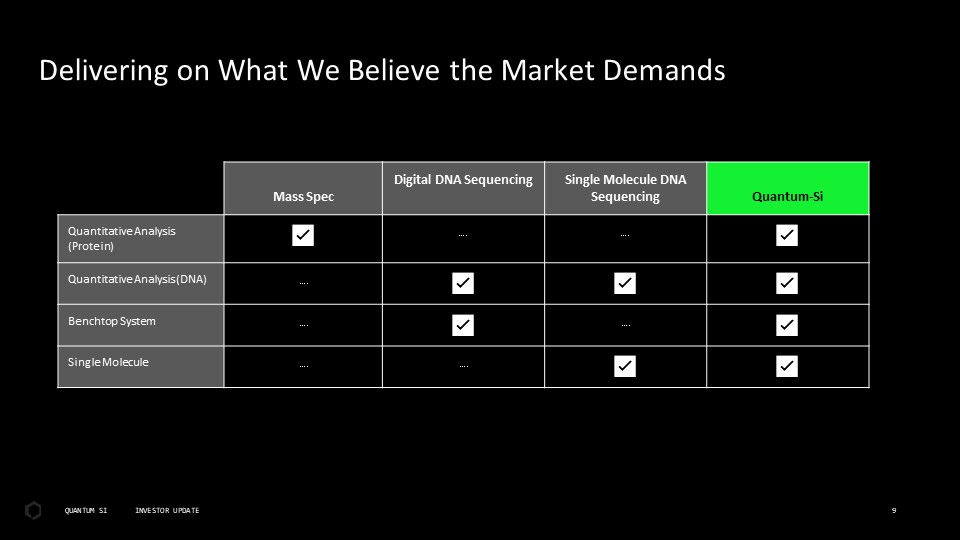
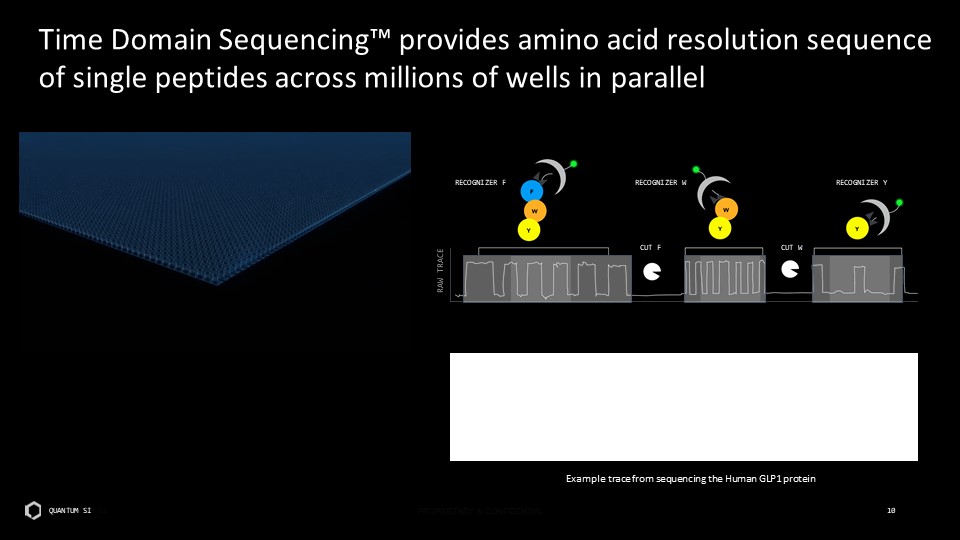
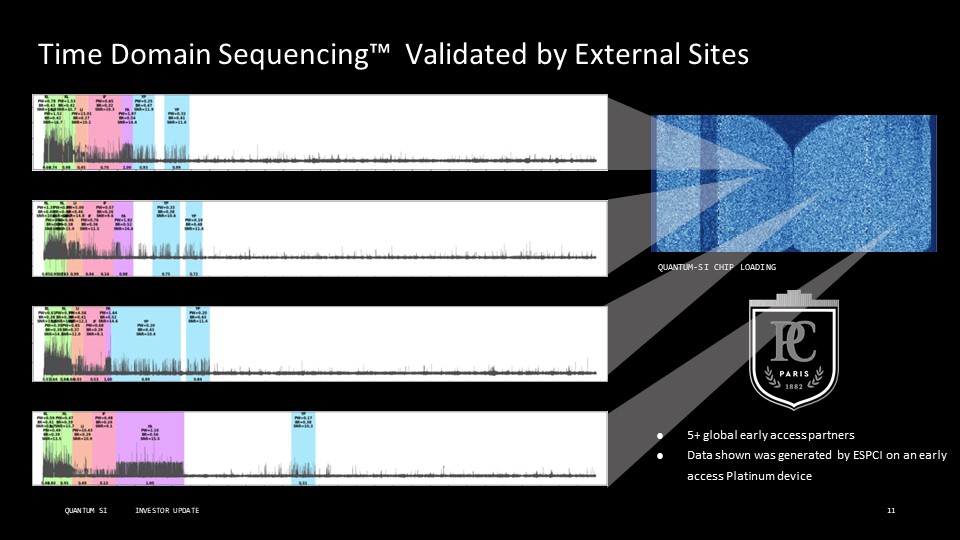
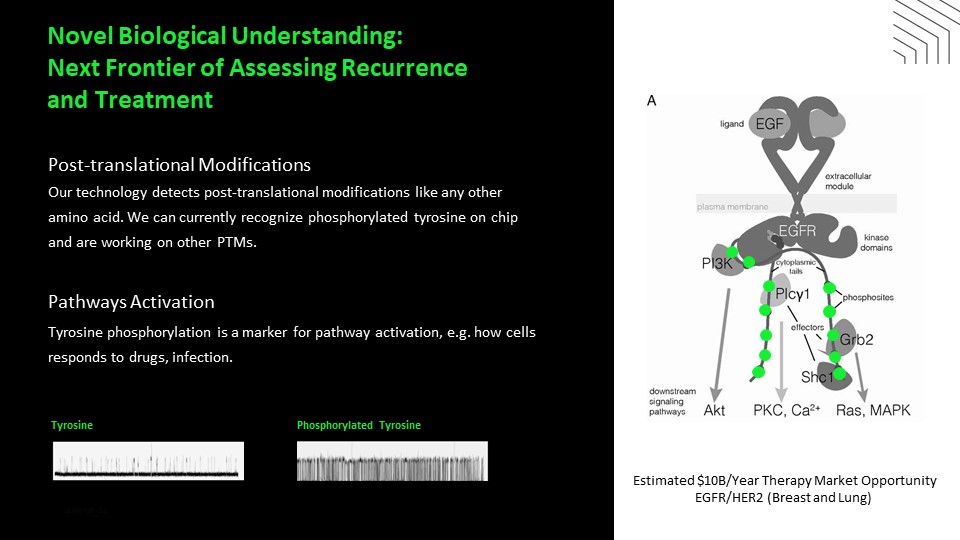
Disclaimer This presentation includes “forward-looking statements” within the meaning of the “safe harbor” provisions of the United States Private Securities Litigation Reform Act of 1995. Actual results of Quantum-Si Incorporated (the “Company”) may differ from its expectations, estimates, and projections and, consequently, you should not rely on these forward-looking statements as predictions of future events. Words such as “expect,” “estimate,” “project,” “budget,” “forecast,” “anticipate,” “intend,” “plan,” “may,” “will,” “could,” “should,” “believes,” “predicts,” “potential,” “continue,” and similar expressions (or the negative versions of such words or expressions) are intended to identify such forward-looking statements. These forward-looking statements include, without limitation, the Company’s expectations with respect to future performance, development of products and services, potential regulatory approvals, the size and potential growth of current or future markets for the Company’s future products and services, or the Company’s plans expectations or future operations, financial position, revenues, costs or expenses. These forward-looking statements involve significant risks and uncertainties that could cause the actual results to differ materially from those discussed in the forward-looking statements. Most of these factors are outside the Company’s control and are difficult to predict. Factors that may cause such differences include, but are not limited to: the impact of COVID-19 on the Company’s business; the inability to maintain the listing of the Company’s shares of Class A common stock on The Nasdaq Stock Market; the ability to recognize the anticipated benefits of the Company’s recently-completed business combination, which may be affected by, among other things, competition and the ability of the Company to grow and manage growth profitably and retain its key employees; changes in applicable laws or regulations; the Company’s ability to raise financing in the future; the success, cost and timing of the Company’s product development activities; the potential attributes and benefits of the Company’s products and services; the Company’s ability to obtain and maintain regulatory approval for its products, and any related restrictions and limitations of any approved product; the Company’s ability to identify, in-license or acquire additional technology; the Company’s ability to maintain its existing lease, license, manufacture and supply agreements; the Company’s ability to compete with other companies currently marketing or engaged in the development of products and services that the Company is developing; the size and growth potential of the markets for the Company’s future products and services, and its ability to serve those markets, either alone or in partnership with others; the pricing of the Company’s products and services following anticipated commercial launch; the Company’s estimates regarding future expenses, future revenue, capital requirements and needs for additional financing; the Company’s financial performance; and other risks and uncertainties indicated from time to time in the Company’s filings with the U.S. Securities and Exchange Commission. The Company cautions that the foregoing list of factors is not exclusive. The Company cautions readers not to place undue reliance upon any forward-looking statements, which speak only as of the date made. The Company does not undertake or accept any obligation or undertaking to release publicly any updates or revisions to any forward-looking statements to reflect any change in its expectations or any change in events, conditions, or circumstances on which any such statement is based.