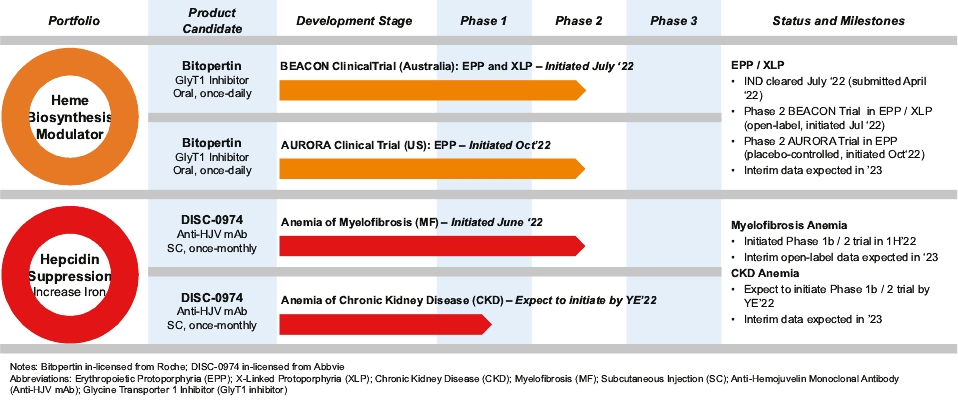
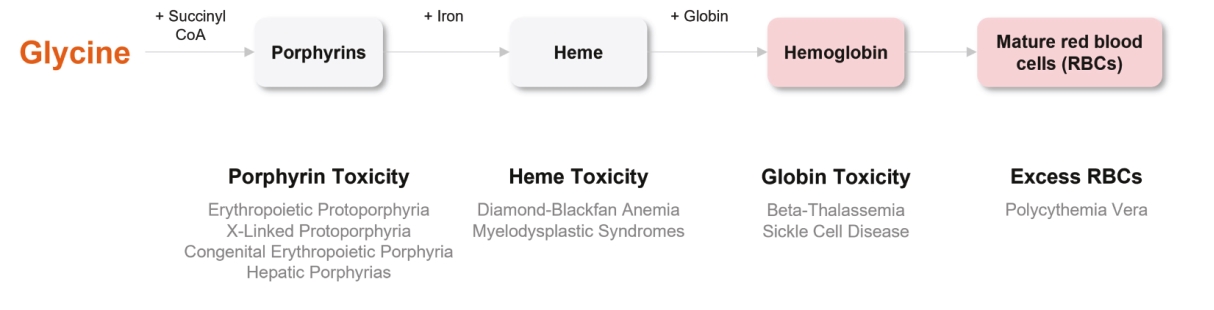
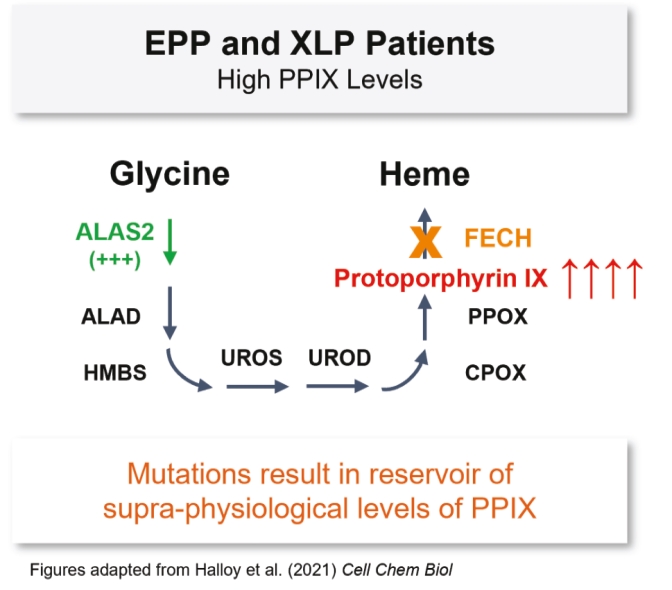
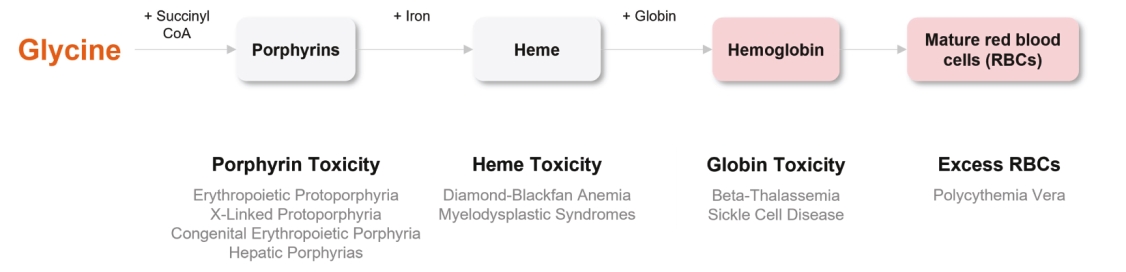
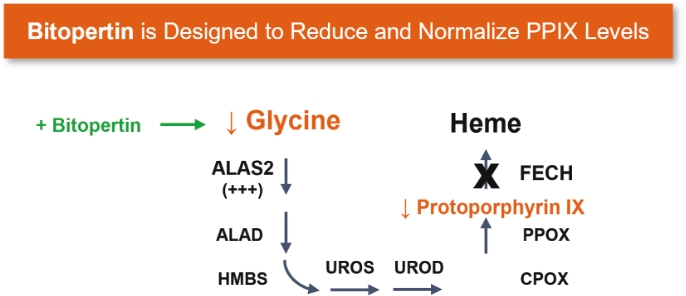
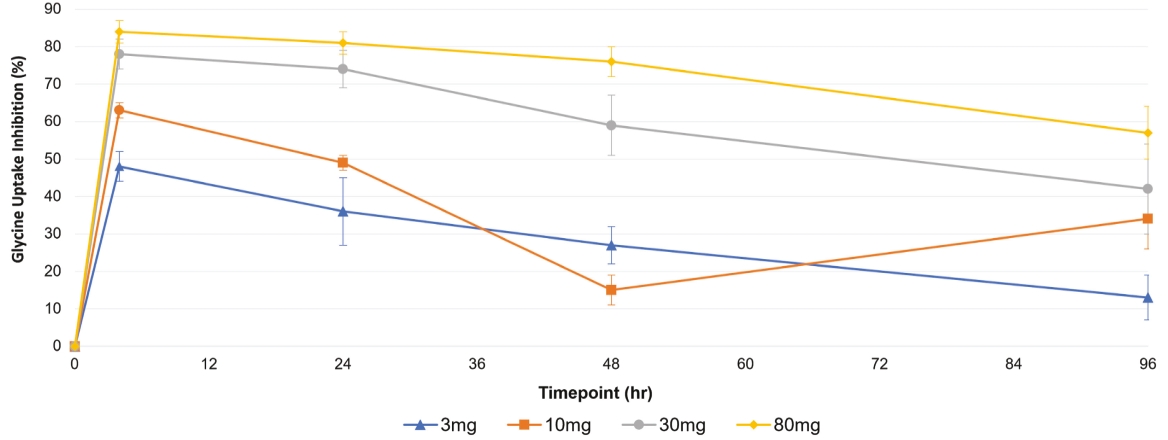
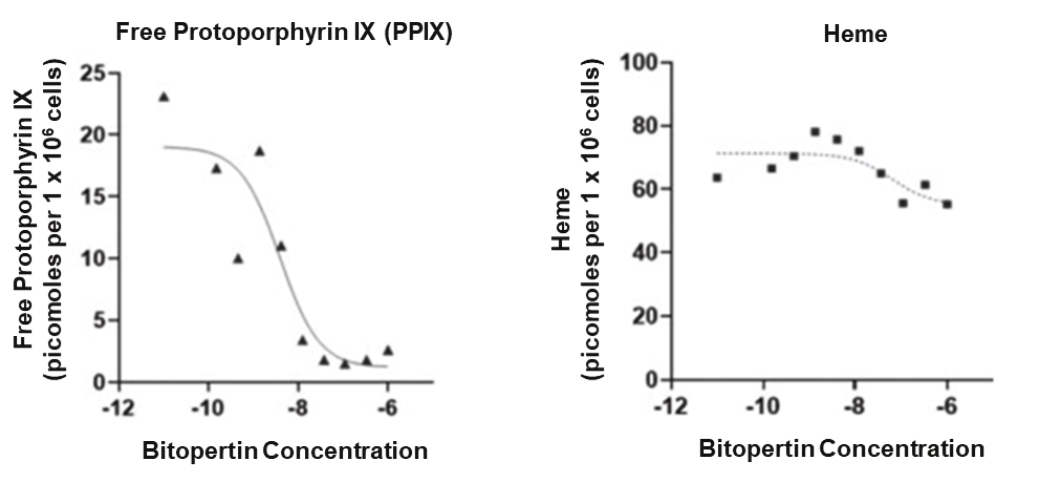
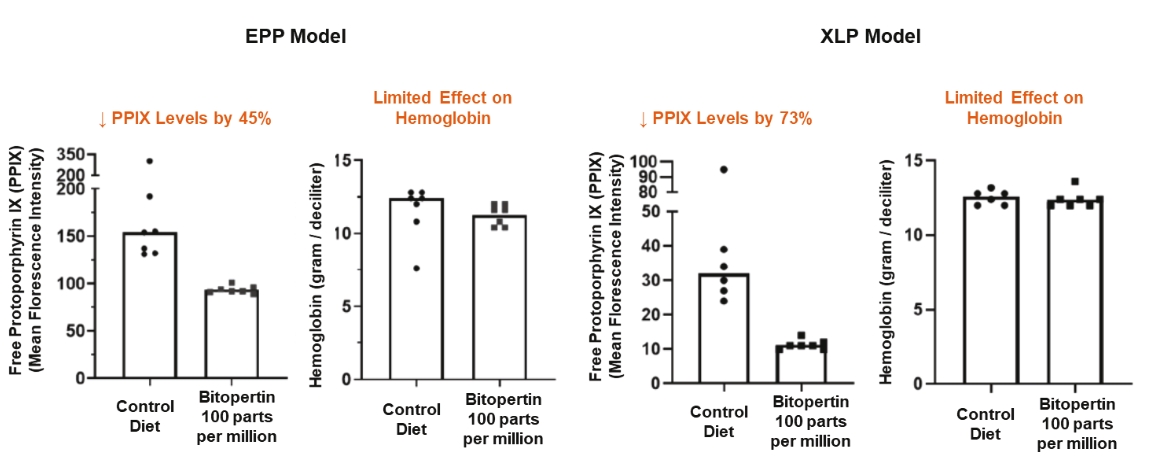
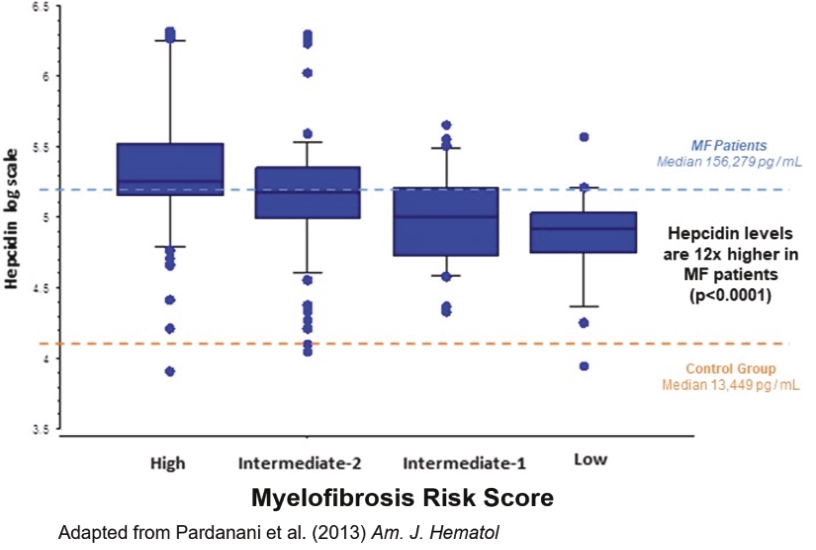
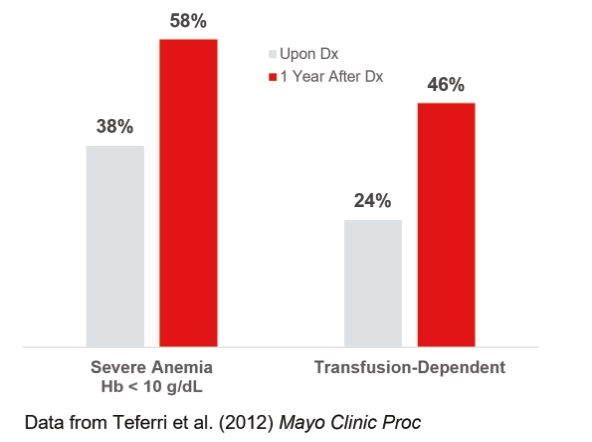
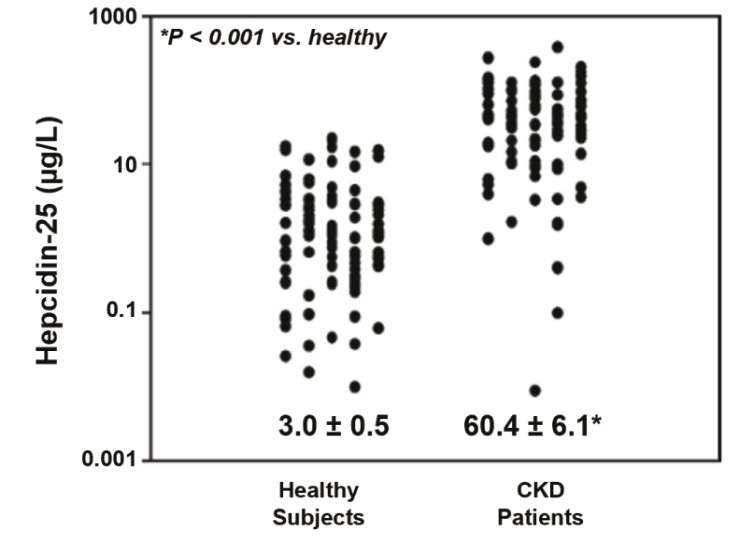
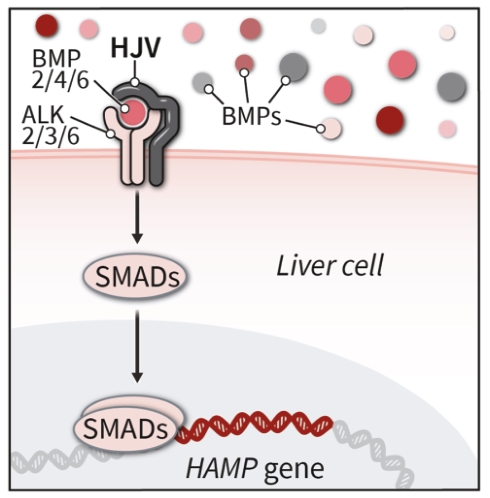
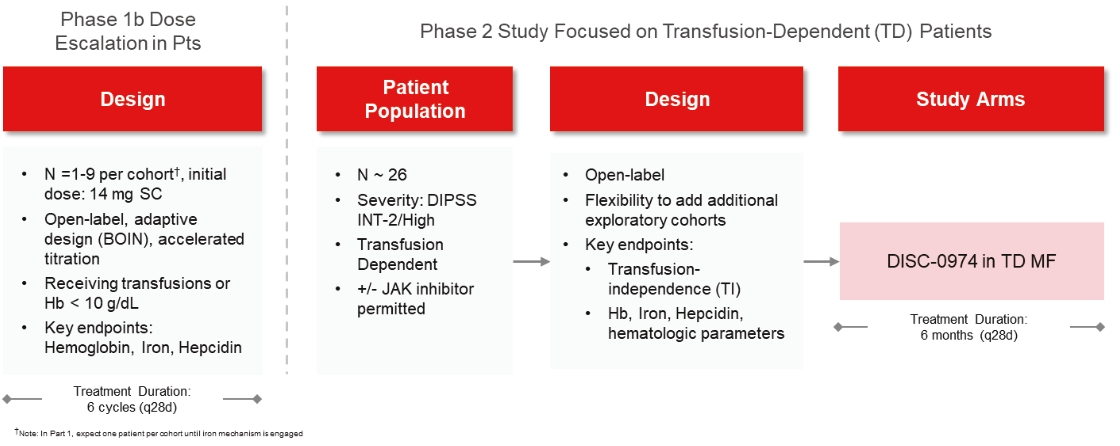
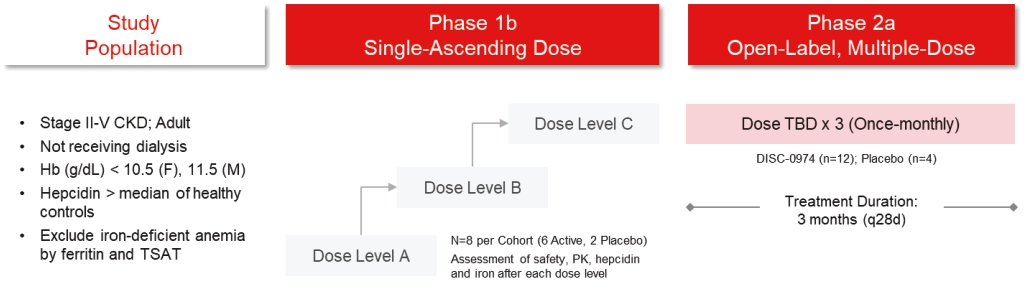
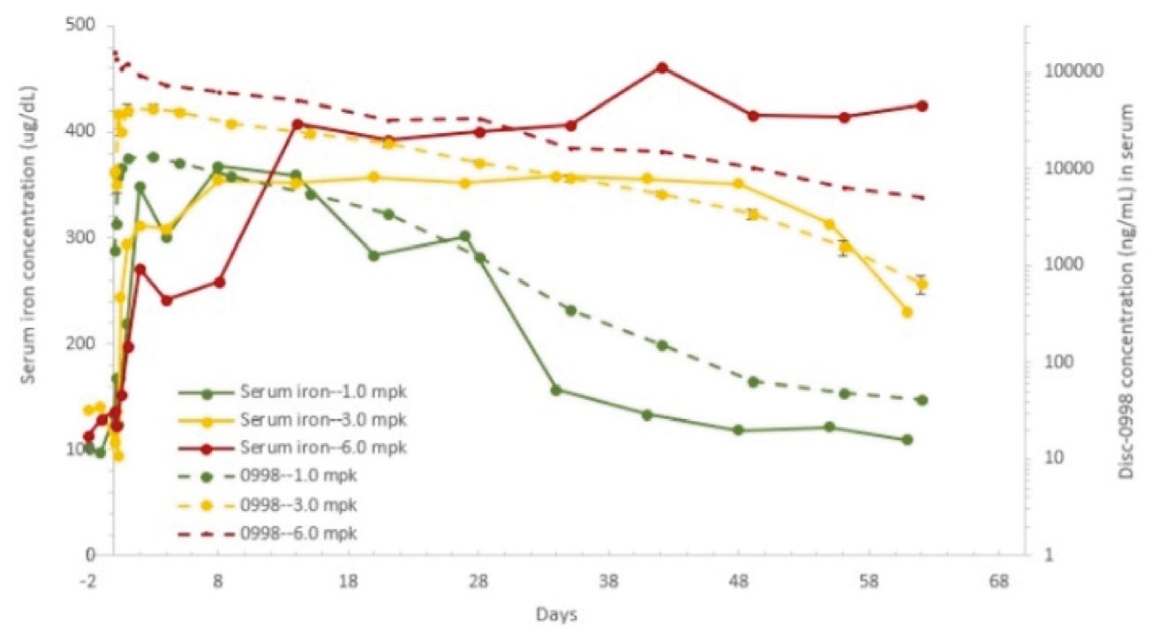
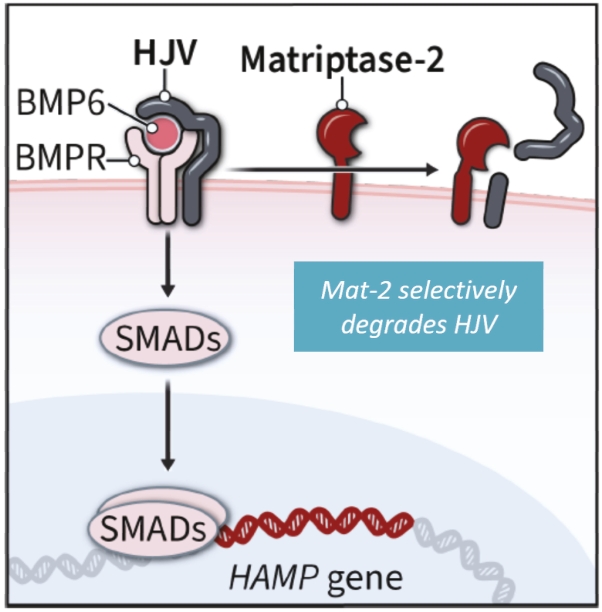
| | | owned | | | antagonists | | | | | | | | | |
3 | | | Disc Medicine owned | | | Claims to compositions of anti-HJV antibodies for treating anemia of chronic disease. | | | 2041 | | | PCT | | | n/a |
4 | | | Disc Medicine owned | | | Claims to compositions of anti-HJV antibodies for treating myelofibrosis | | | 2041 | | | PCT | | | n/a |
5 | | | Disc Medicine owned | | | Claims to methods of treating anemia of kidney disease with anti-HJV antagonists | | | 2042 | | | U.S.
Provisional | | | n/a |
6 | | | In-Licensed | | | Claims to compositions and methods for the diagnosis and treatment of iron-related disorders with anti-hemojuvelin (HJV) antagonists | | | 2032 | | | PCT | | | United Stated*, Australia, China, the United Kingdom, Germany, Mexico, and Japan |
Disc has also in-licensed multiple patent families from F. Hoffmann-La Roche Ltd and Hoffmann-La Roche Inc. comprising eight issued U.S. patents and additional granted patents in the following jurisdictions: Algeria, Australia, Austria, Belarus, Belgium, Brazil, Bulgaria, Canada, Chile, China, Colombia, Costa Rica, Croatia, Cyprus, Czech Republic, Denmark, Ecuador, Egypt, Estonia, Eurasian Patent Convention, European Patent Convention, Finland, France, Germany, Great Britain, Greece, Gulf Cooperation Council, Hong Kong, Hungary, India, Indonesia, Ireland, Israel, Italy, Japan, Kazakhstan, Kosovo, Latvia, Lithuania, Luxembourg, Malaysia, Malta, Mexico, Monaco, Montenegro, Morocco, Netherlands, New Zealand, Norway, Philippines, Poland, Portugal, Republic of Korea, Republic of Serbia, Romania, Russian Federation, Singapore, Slovak Republic, Slovenia, South Africa, Spain, Sweden, Switzerland, Taiwan, Turkey, Ukraine, and Vietnam. Patents and pending applications directed to bitopertin, synthetic intermediates, synthetic methods, synthetic processes of making bitopertin, treatment of hematologic disorders characterized by elevated cellular hemoglobin, and crystalline forms of bitopertin are expected to expire between 2024 and 2035, without accounting for any potential terminal disclaimers, available patent term adjustments or extensions. In particular, the first family is directed to composition of matter of bitopertin and processes of preparation, and this family has a twenty-year statutory expiration date of 2024. This family has issued patents in the U.S. and the following jurisdictions: Algeria, Australia, Austria, Belarus, Belgium, Brazil, Bulgaria, Canada, Chile, China, Colombia, Costa Rica, Croatia, Cyprus, Czech Republic, Denmark, Ecuador, Egypt, Estonia, Eurasian Patent Convention, European Patent Convention, Finland, France, Germany, Great Britain, Greece, Gulf Cooperation Council, Hong Kong, Hungary, India, Indonesia, Ireland, Israel, Italy, Japan, Kazakhstan, Kosovo, Latvia, Lithuania, Luxembourg, Malaysia, Mexico, Monaco, Montenegro, Morocco, Netherlands, New Zealand, Norway, Philippines, Poland, Portugal, Republic of Korea, Republic of Serbia, Romania, Russian Federation, Singapore, Slovak Republic, Slovenia, South Africa, Spain, Sweden, Switzerland, Taiwan, Turkey, Ukraine, and Vietnam. The second family is directed to processes of preparation of bitopertin, and this family has a twenty-year statutory expiration date of 2028. This family has issued patents in the U.S. and the following jurisdictions: Australia, Austria, Belgium, Brazil, Canada, China, European Patent Convention, Finland, France, Germany, Great Britain, Hungary, Ireland, Israel, Italy, Japan, Mexico, Netherlands, Republic of Korea, Spain, Sweden, and Switzerland. The third and fourth families are directed to synthetic processes for synthetic intermediates, and these families have twenty-year statutory expiration dates of 2026 and 2027, respectively. These families each have issued patents in the U.S. and the following jurisdictions: China, European Patent Convention, France, Germany, Great Britain, Japan, and Switzerland. The fifth family is directed to methods of treating hematological disorders characterized by elevated cellular hemoglobin levels with bitopertin, and this family has a twenty-year statutory expiration date of 2035. This family has issued patents in the U.S. and the following jurisdictions: Algeria, China, Croatia, Cyprus, European Patent Convention, France, Germany, Great Britain, Greece, Hong Kong, Indonesia, Italy, Japan, Malaysia, Morocco, Portugal, Republic of Korea, Republic of Serbia, Slovenia, South Africa, Spain, Switzerland, and Turkey. The sixth family is directed composition of matter of additional GlyT1 inhibitors, and this family has a twenty-year statutory expiration date of 2026. This family has issued patents in the U.S. and the following jurisdictions: China, European Patent Convention, France, Germany, Great Britain, Hong Kong, Japan, and Switzerland. The seventh family is directed to crystalline forms of bitopertin, and this family has a twenty-year statutory expiration date of 2027. This family has issued patents in the following jurisdictions: Australia, Austria, Belgium, Brazil, Bulgaria, Chile, Croatia, Cyprus, Czech Republic, Denmark, Estonia, European Patent Convention, Finland, France, Germany, Great Britain, Greece, Gulf Cooperation Council, Hungary, Indonesia, Ireland, Italy, Japan, Latvia, Lithuania, Luxembourg, Malaysia, Malta, Mexico,