TABLE OF CONTENTS
| 1 |
| | |
| PART I | |
| | | |
| | 2 |
| | 2 |
| | 2 |
| | 24 |
| | 38 |
| | 38 |
| | 45 |
| | 58 |
| | 60 |
| | 60 |
| | 61 |
| | 68 |
| | 68 |
| | | |
| PART II | |
| | | |
| | 70 |
| | 70 |
| | 70 |
| | 70 |
| | 70 |
| | 71 |
| | 71 |
| | 71 |
| | 71 |
| | 71 |
| | 72 |
| | 72 |
| | 72 |
| | | |
| PART III | |
| | | |
| | 73 |
| | 73 |
| | 74 |
| | | |
| 75 |
INTRODUCTION
Certain Definitions
In this Annual Report on Form 20-F, unless the context otherwise requires:
| • | references to “MeaTech,” the “Company,” “us,” “we” and “our” refer to MeaTech MT Ltd. (formerly MeaTech Ltd.) from its inception until the consummation of the January 2020 merger described herein, and MeaTech 3D Ltd. (the “Registrant”), an Israeli company, thereafter, unless otherwise required by the context; |
| • | references to “ordinary shares,” “our shares” and similar expressions refer to the Registrant’s ordinary shares, no nominal (par) value per share; |
| • | references to “ADS” refer to the American Depositary Shares listed on the Nasdaq Capital Market (“Nasdaq”) under the symbol “MITC,” each representing ten ordinary shares of the Registrant; |
| • | references to “dollars,” “U.S. dollars” and “$” are to United States Dollars; |
| • | references to “NIS” are to New Israeli Shekels, the currency of the State of Israel; |
| • | references to the “Companies Law” are to Israel’s Companies Law, 5759-1999, as amended; and |
| • | references to the “SEC” are to the United States Securities and Exchange Commission. |
Forward-Looking Statements
This Annual Report on Form 20-F contains forward-looking statements concerning our business, operations and financial performance and condition as well as our plans, objectives and expectations for our business operations and financial performance and condition. Any statements that are not historical facts may be deemed to be forward-looking statements. These statements involve known and unknown risks, uncertainties and other factors that may cause our actual results, performance or achievements to be materially different from any future results, performance or achievements expressed or implied by the forward-looking statements. In some cases, you can identify forward-looking statements by terms including “anticipates,” “believes,” “could,” “estimates,” “expects,” “intends,” “may,” “plans,” “potential,” “predicts,” “projects,” “should,” “will,” “would,” and similar expressions intended to identify forward-looking statements, but these are not the only ways these statements are identified. Forward-looking statements reflect our current views with respect to future events and are based on assumptions and subject to risks and uncertainties. You should not put undue reliance on any forward-looking statements. Unless we are required to do so under U.S. federal securities laws or other applicable laws, we do not intend to update or revise any forward-looking statements. Readers are encouraged to consult the Company’s filings made on Form 6-K, which are periodically filed with or furnished to the SEC.
Forward-looking statements contained in this prospectus include, but are not limited to:
| • | our estimates regarding our expenses, future revenue, capital requirements and needs for additional financing; |
| • | our expectations regarding the success of our cultured meat manufacturing technologies we are developing, which will require significant additional work before we can potentially launch commercial sales; |
| • | our research and development activities associated with technologies for cultured meat manufacturing, including three-dimensional meat production, which involves a lengthy and complex process; |
| • | our expectations regarding the timing for the potential commercial launch of our cultured meat technologies; |
| • | our ability to successfully manage our planned growth, including with respect to our acquisition of Peace of Meat BV, or Peace of Meat, and any future acquisitions, joint ventures, collaborations or similar transactions; |
| • | the potential business or economic disruptions caused by the COVID-19 pandemic; |
| • | the competitiveness of the market for our cultured meat technologies; |
| • | our ability to enforce our intellectual property rights and to operate our business without infringing, misappropriating, or otherwise violating the intellectual property rights and proprietary technology of third parties; |
| • | our ability to predict and timely respond to preferences for alternative proteins and cultured meats and new trends; |
| • | our ability to attract, hire and retain qualified employees and key personnel; and |
| • | other risks and uncertainties, including those listed in “Item 3. —Key Information—Risk Factors.” |
PART I
| | ITEM 1. | IDENTITY OF DIRECTORS, SENIOR MANAGEMENT AND ADVISERS |
Not applicable.
| | ITEM 2. | OFFER STATISTICS AND EXPECTED TIMETABLE |
Not applicable.
A. Selected Financial Data
[Reserved]
B. Capitalization and Indebtedness
Not applicable.
C. Reasons for the Offer and Use of Proceeds
Not applicable.
D. Risk Factors
Our business is subject to various risks, including those described below. You should carefully consider the risks and uncertainties described below and in our future filings with the SEC. If any of the following risks are realized, our business, financial condition, results of operations and prospects could be materially and adversely affected. Additionally, risks and uncertainties not currently known to us or that we currently deem to be immaterial also may materially adversely affect our business, financial condition, results of operations and/or prospects.
SUMMARY OF RISKS ASSOCIATED WITH OUR BUSINESS
Our business is subject to a number of risks of which you should be aware before a decision to invest in the ADSs. You should carefully consider all the information set forth in this prospectus and, in particular, should evaluate the specific factors set forth in the sections titled “Risk Factors” before deciding whether to invest in the ADSs. Among these important risks are, but not limited to, the following:
| • | We have experienced net losses in every period since the inception of MeaTech and we expect to continue incurring significant losses for the foreseeable future and may never become profitable; |
| • | We have a limited operating history to date and our prospects will be dependent on our ability to meet a number of challenges; |
| • | Our business and market potential are unproven, and we have limited insight into trends that may emerge and affect our business; |
| • | We are wholly dependent on the success of our cultured meat manufacturing technologies, including our cultured steak technologies, and we have limited data on the performance of our technologies to date; |
| • | The research and development associated with technologies for cultured meat manufacturing, including three-dimensional meat production, is a lengthy and complex process; |
| • | Business or economic disruptions or global health concerns, including the COVID-19 pandemic, may have an adverse impact on our business and results of operations; |
| • | We may not be able to compete successfully in our highly competitive market; |
| • | We may suffer reputational harm due to real or perceived quality or health issues with products manufactured by our licensees using our technology; |
| • | Consumer preferences for alternative proteins in general, and more specifically cultured meats, are difficult to predict and may change, and, if we are unable to respond quickly to new trends, our business may be adversely affected; |
| • | We have no manufacturing experience or resources and we expect we will incur significant costs to develop this expertise or need to rely on third parties for manufacturing; |
| • | We expect that a small number of customers will account for a significant portion of our revenues, and the loss of one or more of these customers could adversely affect our financial condition and results of operations; |
| • | We expect that products utilizing our technologies will be subject to regulations that could adversely affect our business and results of operations; |
| • | Regulatory authorities may impose new regulations on manufacturers of alternative proteins; |
| • | Any changes in, or changes in the interpretation of, applicable laws, regulations or policies of the U.S. Department of Agriculture, state regulators or similar foreign regulatory authorities that relate to the use of the word “meat” or other similar words in connection with cultured meat products could adversely affect our business, prospects, results of operations or financial condition; |
| • | If we are unable to obtain and maintain effective intellectual property rights for our technologies, we may not be able to compete effectively in our markets; |
| • | If there are significant shifts in the political, economic and military conditions in Israel, it could have an adverse impact on our operations; and |
| • | If we encounter delays or challenges, such as operational challenges inherent in managing a foreign business, we may not fully realize the anticipated benefits of the acquisition of Peace of Meat. |
RISKS RELATED TO OUR FINANCIAL CONDITION AND LIQUIDITY REQUIREMENTS
We expect to continue incurring significant losses for the foreseeable future and may never become profitable.
We have experienced net losses in every period since the inception of MeaTech. We anticipate that we will continue to incur significant losses for the foreseeable future as our operating expenses and capital expenditures increase substantially due to our continued investment in our research and development activities and as we hire additional employees over the coming years. These activities may prove more expensive than we anticipate. We incur significant expenses in developing our technologies. Accordingly, we may not be able to achieve or sustain profitability, and we expect to incur significant losses for the foreseeable future.
Our predecessor entity, MeaTech Ltd., commenced cultured meat development operations in September 2019, and we continue to be in the early stages of development of our technologies. As a result, we have not generated any revenues since inception of our cultured meat operations, and we do not expect to generate any revenue from operations in the near term. We may not be able to develop the technology for manufacturing cultured meat at all, or meet the additional technological challenges to scaling such technology up to an industrial scale from our research and development efforts or successfully market and license our technologies, once approved. In addition, there is no certainty that there will be sufficient demand to justify the production and marketing of cultured meat products. The market for alternative proteins in general, and cultured meats specifically, may be small or may not develop.
If cultured meats produced using our industrial-scale cultured meat manufacturing processes do not gain wide market acceptance, we will not be able to achieve our anticipated growth, revenues or profitability and we may not be able to continue our business operations.
We will require substantial additional funds to complete our research and development activities and, if additional funds are not available on acceptable terms or at all, we may need to significantly scale back or cease our operations.
A significant portion of our research and development activities has been financed by the issuance of equity securities. We believe that we will continue to expend substantial resources for the foreseeable future as we work to develop our technologies. These expenditures are expected to include costs associated with research and development, and manufacturing and supply, as well as general operating expenses. In addition, other unanticipated costs may arise.
There is no certainty that we will be able to obtain funding for our research and development activities when we need it, on acceptable terms or at all. A lack of adequate funding may force us to reduce or cease all or part of our research and development activities and business operations. Our operating plan may change because of factors currently unknown to us, and we may need to seek additional funds sooner than planned, through public or private equity or debt financings or other sources, such as strategic collaborations. Such financing may result in dilution to shareholders, imposition of debt covenants and repayment obligations, or other restrictions that may adversely affect our business. In addition, we may seek additional capital due to favorable market conditions or strategic considerations even if we believe we have sufficient funds for our current or future operating plans.
Our future capital requirements depend on many factors, including:
| • | our progress with current research and development activities; |
| • | the number and characteristics of any products or manufacturing processes we develop or acquire; |
| • | the expenses associated with our marketing initiatives; |
| • | the timing, receipt and amount of milestone, royalty and other payments from future customers and collaborators, if any; |
| • | the scope, progress, results and costs of researching and developing future products or improvements to existing products or manufacturing processes; |
| • | any lawsuits related to our products or commenced against us; |
| • | the expenses needed to attract, hire and retain skilled personnel; |
| • | the costs associated with being a public company in the United States; and |
| • | the costs involved in preparing, filing, prosecuting, maintaining, defending and enforcing intellectual property claims, including litigation costs and the outcome of such litigation. |
If our estimates and predictions relating to any of these factors are incorrect, we may need to modify our operating plan. Additional funds may not be available to us when needed on acceptable terms, or at all. If adequate funds are not available to us on a timely basis, we may be required to delay, limit, reduce or terminate our manufacturing, research and development activities or other activities that may be necessary to generate revenue and achieve profitability.
There is substantial doubt as to whether we can continue as a going concern.
Our consolidated financial statements as of December 31, 2021 contain an explanatory paragraph that states that our recurring losses from operations raise substantial doubt about our ability to continue as a going concern. Our financial statements do not include any measurement or presentation adjustment for assets or liabilities that might result if we would be unable to continue as a going concern. We have incurred operating losses since inception, have not generated any revenues and have not achieved profitable operations. Our net loss, accumulated during the development stage through December 31, 2021, totaled approximately $37 million, and we expect to continue to incur substantial losses in future periods while we continue our research and development activities.
Raising additional capital may cause dilution to our existing shareholders, restrict our operations or require us to relinquish rights to our technologies.
Until such time, if ever, as we can generate substantial product revenues, we expect to finance our cash needs through equity offerings, debt financings, government contracts, government and/or other third-party grants or other third-party funding, marketing and distribution arrangements and other collaborations, strategic alliances and licensing arrangements. We will require substantial funding to fund our anticipated commercialization efforts and fund our operating expenses and other activities. To the extent that we raise additional capital through the sale of equity or convertible debt securities, your ownership interest will be diluted, and the terms of these securities may include liquidation or other preferences that adversely affect your rights as a shareholder. Debt financing, if available, may involve agreements that include covenants limiting or restricting our ability to take specific actions, such as incurring additional debt, making capital expenditures or declaring dividends. If we are unable to obtain funding on a timely basis, we may be required to significantly curtail our research or development program, or a part thereof, which would adversely impact our potential revenues, results of operations and financial condition.
RISKS RELATED TO OUR BUSINESS AND STRATEGY
We have a limited operating history to date and our prospects will be dependent on our ability to meet a number of challenges.
Our business prospects are difficult to predict due to a lack of operational history, and our success will be dependent on our ability to meet a number of challenges. Because we have a limited operating history and we are in the early stages of development, you may not be able to evaluate our future prospects accurately. Our prospects will be primarily dependent on our ability to successfully develop industrial-scale cultured meat manufacturing technologies and processes, and market these to our customers. If we are not able to successfully meet these challenges, our prospects, business, financial condition and results of operations could be adversely impacted.
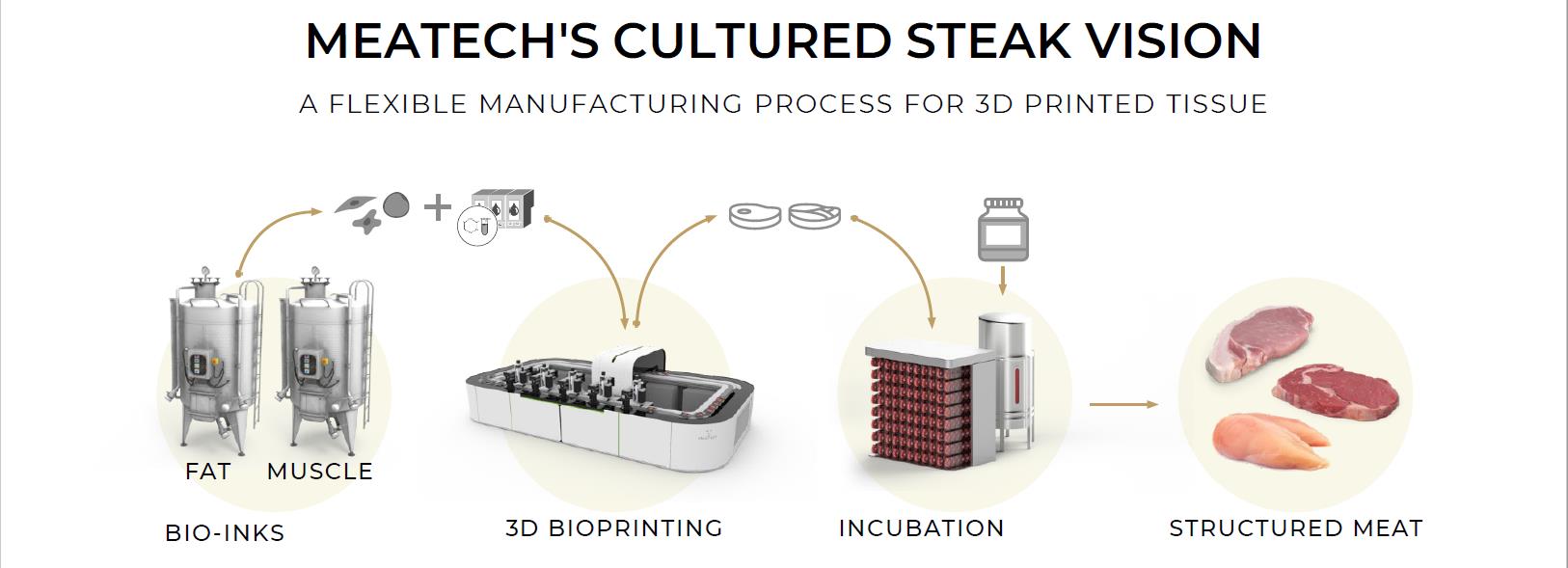
We are wholly dependent on the success of our cultured meat manufacturing technologies, including our cultured steak technologies, and we have limited data on the performance of our technologies to date.
We do not currently have any products or technologies approved for sale and we are still in the early stages of development. To date, we have limited data on the ability of our technologies to successfully manufacture cultured meat, towards which we have devoted substantial resources to date. We may not be successful in developing our technologies in a manner sufficient to support our expected scale-ups and future growth, or at all. We expect that a substantial portion of our efforts and expenditures over the next few years will be devoted to the development of technologies designed to enable us to market industrial-scale cultured meat manufacturing processes. We cannot guarantee that we will be successful in developing these technologies on the timeline we expect or at all. If we are able to successfully develop our cultured meat technologies, we cannot ensure that we will obtain regulatory approval or that, following approval, upon commercialization our technologies will achieve market acceptance. Any such delay or failure would materially and adversely affect our financial condition, results of operations and prospects.
The research and development associated with technologies for cultured meat manufacturing, including three-dimensional meat production, is a lengthy and complex process.
We are focused on developing commercial technologies that companies can license to manufacture alternative foods without the need for animal butchery, based on rapid growing cycles. To develop our cultured meat steak technology, we are developing cellular agriculture technology, such as cell lines and approaches to working with plant-based cell-growth media in a scalable process. We are currently planning to scale up the printing process to provide us with industrial-scale capabilities. If we are unable to successfully develop our cultured meat manufacturing technologies, we may not be able to achieve our anticipated growth, revenues or profitability and we may not be able to continue our business operations.
We intend to engage in future acquisitions, joint ventures or collaborations, similar to our acquisition of Peace of Meat, which may increase our capital requirements, dilute our shareholders, cause us to incur debt or assume contingent liabilities, and subject us to other risks. We may not realize the benefits of these acquisitions, joint ventures or collaborations.
We may evaluate various acquisitions and collaborations, including licensing or acquiring complementary technologies, intellectual property rights, or businesses. Any potential acquisition, joint venture or collaboration will entail numerous potential risks, including:
• increased operating expenses and cash requirements;
| • | the assumption of additional indebtedness or contingent liabilities; |
| • | assimilation of operations, intellectual property and products of an acquired company, including difficulties associated with integrating new personnel; |
| • | the diversion of our management’s attention from our existing programs and initiatives in pursuing such a strategic merger or acquisition; |
| • | retention of key employees, the loss of key personnel, and uncertainties in our ability to maintain key business relationships; |
| • | risks and uncertainties associated with the other party to such a transaction, including the prospects of that party and their existing technologies; and |
| • | our inability to generate revenue from acquired technologies or products sufficient to meet our objectives in undertaking the acquisition or even to offset the associated acquisition and maintenance costs. |
In addition, if we undertake acquisitions, we may utilize our cash, issue dilutive securities, assume or incur debt obligations, incur large one-time expenses and acquire intangible assets that could result in significant future amortization expense.
Moreover, we may not be able to locate suitable acquisition or collaboration opportunities and this inability could impair our ability to grow or obtain access to technologies that may be important to the development of our business.
Our existing potential collaborations, or any future collaboration arrangements that we may enter into, may not be successful, which could significantly limit the likelihood of receiving the potential economic benefits of the collaboration and adversely affect our ability to develop and commercialize our product candidates.
We have entered into a potential collaboration with Tiv Ta’am, which is reflected in a non-binding letter of intent. Tiv Ta’am has no obligation to collaborate with us and may withdraw from the proposed collaboration at any time without any liability.
Additionally, we may enter into future collaborations under which our collaborators have provided, and may in the future provide, funding and other resources for developing and commercializing our cultured meat manufacturing technologies. We expect to enter into additional collaborations to access additional funding, capabilities and expertise in the future. Our existing collaborations, and any future collaborations we enter into, may pose a number of risks, including the following:
| • | collaborators may not perform or prioritize their obligations as expected; |
| • | collaborators may not pursue development and commercialization of any of our cultured meat manufacturing technologies or may elect not to continue or renew development or commercialization, changes in the collaborators’ focus or available funding, or external factors, such as an acquisition, that divert resources or create competing priorities; |
| • | collaborators may provide insufficient funding for the successful development or commercialization of our cultured meat manufacturing technologies; |
| • | collaborators could independently develop, or develop with third parties, products or technologies that compete directly or indirectly with our products or cultured meat manufacturing technologies if the collaborators believe that competitive products are more likely to be successfully developed or can be commercialized under terms that are more economically attractive than ours; |
| • | cultured meat manufacturing technologies developed in collaborations with us may be viewed by our collaborators as competitive with their own products or technologies, which may cause collaborators to cease to devote resources to the development or commercialization of our products; |
| • | a collaborator with marketing and distribution rights to one or more of our products or technologies that achieve regulatory approval may not commit sufficient resources to the marketing and distribution of any such product; |
| • | disagreements with collaborators, including disagreements over proprietary rights, contract interpretation or the preferred course of development of cultured meat manufacturing technologies, may cause delays or termination of the research, development or commercialization of such technologies, may lead to additional responsibilities for us with respect to such technologies, or may result in litigation or arbitration, any of which would be time-consuming and expensive; |
| • | collaborators may not properly maintain, protect, defend or enforce our intellectual property rights or may use our proprietary information in such a way as to invite litigation that could jeopardize or invalidate our intellectual property or proprietary information or expose us to potential litigation; |
| • | disputes may arise with respect to the ownership of intellectual property developed pursuant to our collaborations; |
| • | collaborators may infringe, misappropriate or otherwise violate the intellectual property rights of third parties, which may expose us to litigation and potential liability; |
| • | collaborations may be terminated for the convenience of the collaborator and, if terminated, the development of our cultured meat manufacturing technologies may be delayed, and we could be required to raise additional capital to pursue further development or commercialization of the cultured meat manufacturing technologies; |
| • | future relationships may require us to incur non-recurring and other charges, increase our near- and long-term expenditures, issue securities that dilute our existing shareholders, or disrupt our management and business; and |
| • | we could face significant competition in seeking appropriate collaborators, and the negotiation process is time-consuming and complex. |
If our collaborations do not result in the successful development of our products, or if one of our collaborators terminates its agreement with us, we may not receive any future research funding or milestone, earn-out, royalty or other contingent payments under the collaborations. If we do not receive the funding we expect under these agreements, our development of our cultured meat manufacturing technologies could be delayed and we may need additional resources to develop our technologies. If one of our collaborators terminates its agreement with us, we may find it more difficult to attract new collaborators and the perception of us in the business and financial communities could be adversely affected. All of the risks relating to product development, regulatory approval and commercialization described in this report apply to the activities of our collaborators.
We may not be able to successfully manage our planned growth.
We expect to continue to make investments in our cultured meat manufacturing technologies. We expect that our annual operating expenses will continue to increase as we invest in further research and development activities and, ultimately, sales and marketing efforts and customer service and support resources for future customers. Our failure to expand operational and financial systems in a timely or efficient manner could result in operating inefficiencies, which could increase our costs and expenses more than we had planned and prevent us from successfully executing our business plan. We may not be able to offset the costs of operation expansion by leveraging the economies of scale from our growth in negotiations with our suppliers and contract manufacturers. Additionally, if we increase our operating expenses in anticipation of the growth of our business and this growth does not meet our expectations, our financial results will be negatively impacted.
If our business grows, we will have to manage additional product design projects, materials procurement processes, and sales efforts and marketing for an increasing number of products, as well as expand the number and scope of our relationships with suppliers, distributors and end customers. If we fail to manage these additional responsibilities and relationships successfully, we may incur significant costs, which may negatively impact our operating results. Additionally, in our efforts to be first to market with new products with innovative functionality and features, we may devote significant research and development resources to products and product features for which a market does not develop quickly, or at all. If we are not able to predict market trends accurately, we may not benefit from such research and development activities, and our results of operations may suffer.
As our future development and commercialization plans and strategies develop, we expect to need additional managerial, operational, sales, marketing, financial and legal personnel. Our management may need to divert a disproportionate amount of its attention away from our day-to-day activities and devote a substantial amount of time to managing these growth activities. We may not be able to effectively manage the expansion of our operations, which may result in weaknesses in our infrastructure, operational mistakes, loss of business opportunities, failure to deliver and timely deliver our products to customers, loss of employees and reduced productivity among remaining employees. Our expected growth could require significant capital expenditures and may divert financial resources from other projects, such as the development of additional new products. If our management is unable to manage our growth effectively, our expenses may increase more than expected, our ability to generate and/or grow revenue could be reduced, and we may not be able to implement our business strategy.
If the market does not grow as we expect, we may not achieve sustainable revenues.
The marketplace for alternative protein manufacturing plants, which we expect to be our primary market, is dominated by methods that do not involve three-dimensional printing technology. If the market does not broadly accept three-dimensional printing of cultured meats as an alternative for conventional meat harvesting, or if it adopts three-dimensional printing based on a technology other than our proprietary bio-ink technology, we may not be able to achieve a sustainable level of revenues, and our results of operations would be adversely affected as a result. Additionally, cultivated meat is significantly more expensive than conventional meat. If the price of cultivated meat remains high, this may limit the consumer demand for, and market acceptance of, products manufactured using our technologies, and we may never be able to compete successfully or generate sufficient revenue or sustained profitability.
Business or economic disruptions or global health concerns, including COVID-19, may have an adverse impact on our business and results of operations.
The COVID-19 pandemic has negatively impacted the global economy, disrupted consumer spending and global supply chains, and created significant volatility and disruption of financial markets. Many countries around the world, including in Israel, have significant governmental measures being implemented to control the spread of the virus, including temporary closure of businesses, severe restrictions on travel and the movement of people, and other material limitations on the conduct of business. To date, the impact of the pandemic on our operations has been mainly limited to a temporary closure of our facility earlier in the year, in the context of a government-mandated general lockdown, which temporary delayed certain of our development activities, while we implemented remote working and workplace protocols for our employees in accordance with government requirements. While government restrictions have recently been eased in Israel following a successful vaccination campaign, the extent of the impact of the COVID-19 pandemic on our business and financial performance, including our ability to execute our near-term and long-term business strategies and initiatives in the expected time frame, will depend on future developments, including the duration and severity of the pandemic and the impacts of reopening, including possible additional waves, which are uncertain and cannot be predicted.
As a result of the COVID-19 pandemic, including related governmental guidance or requirements, we may need to close our facilities, at least temporarily, or implement more restrictive policies to comply with social distancing rules and other requirements. As much of our research and development work requires on-site performance, such steps may negatively impact productivity and cause other disruptions to our business.
The full extent of the COVID-19 pandemic’s impact on our business and results of operations depends on future developments that are uncertain and unpredictable, including the duration and spread of the pandemic, its lasting impact on capital and financial markets, including any economic recession, and any new information that may emerge concerning the severity of the virus, its spread to other regions as well as the actions taken to contain it, among others. At this point in time, we cannot reasonably estimate the full extent of the COVID-19 pandemic’s impact on our business, financial condition, results of operations and cash flow.
RISKS RELATED TO OUR ACQUISITION OF PEACE OF MEAT
We may not fully realize the anticipated benefits of the acquisition or realize such benefits within the timing anticipated.
We acquired Peace of Meat because we believe that the acquisition will be beneficial to us and to our shareholders. However, we may not be able to achieve the anticipated long-term strategic benefits of the acquisition within the timing anticipated or at all. For example, the benefits from the acquisition will be partially offset by the significant costs incurred in completing the transaction. Any delays and challenges that may be encountered in the post-acquisition process of consolidation could have an adverse effect on our business and results of operations, and may affect the value of the ADSs and our ordinary shares after the completion of the acquisition.
We may have operational challenges in managing Peace of Meat’s business and staff following the acquisition.
Acquisitions inherently have risks including misjudging key elements of an acquisition or failing to integrate it in an efficient and timely manner that would disrupt operations. In addition, as Peace of Meat is located in a different country, which also brings inherent management challenges. Our agreement to acquire Peace of Meat provides that Peace of Meat will continue to be managed independently within our business for approximately two years from the time of purchase, adding to the operational complexity of the integration. We may further face operational challenges in managing Peace of Meat’s business following the acquisition, which could have an adverse effect on our business and results of our operations, and may affect the value of the ADSs and ordinary shares.
If intangible assets that we recorded in connection with the Peace of Meat acquisition become impaired, we may have to take significant charges against earnings.
In connection with the accounting for the Peace of Meat acquisition, we have recorded intangible assets. Under IFRS, we must assess, at least annually and potentially more frequently, whether the value of indefinite-lived intangible assets has been impaired. Amortizing intangible assets will be assessed for impairment in the event of an impairment indicator. Any reduction or impairment of the value of intangible assets will result in a charge against earnings, which could materially adversely affect our results of operations and shareholders’ equity in future periods.
RISKS RELATED TO COMPETITION AND COMMERCIALIZATION OF OUR TECHNOLOGIES
We are an early-stage company with an unproven business model, which makes it difficult to evaluate our current business and future prospects.
We have no established basis to assure investors that our business strategies will be successful. We are dependent on unproven technologies and we have no basis to predict acceptance of our technologies by potential licensees and their customers. The market for cultured meat is new and as yet untested. As a result, the revenue and income potential of our business and our market are unproven. Further, because of our limited operating history and early stage of development, and because the market for cultured meat is relatively new and rapidly evolving, we have limited insight into trends that may emerge and affect our business. We may make errors in predicting and reacting to relevant business trends, which could harm our business.
Before investing, you should consider an investment in the ADSs in light of the risks, uncertainties and difficulties frequently encountered by early-stage companies in new and rapidly evolving markets such as ours. We may not be able to successfully address any or all of these risks. Failure to adequately do so could cause our business, results of operations and financial condition to suffer.
We may not be able to compete successfully in our highly competitive market.
The alternative protein market is expected to be highly competitive, with numerous brands and products competing for limited retailer shelf space, foodservice and restaurant customers and consumers. For us to compete successfully, we expect that the cultured meats printed using our technologies will need to be competitive in taste, ingredients, texture, ease of integration into the consumer diet, nutritional claims, convenience, brand recognition and loyalty, product variety, product packaging and package design, shelf space, reputation, price, advertising and access to restaurant and foodservice customers.
Generally, the food industry is dominated by multinational corporations with substantially greater resources and operations than us. We cannot be certain that we will successfully compete with larger competitors that have greater financial, marketing, sales, manufacturing, distributing and technical resources than we do. Conventional food companies may acquire our competitors or launch their own competing products, and they may be able to use their resources and scale to respond to competitive pressures and changes in consumer preferences by introducing new products, reducing prices or increasing promotional activities, among other things. Competitive pressures or other factors could prevent us from acquiring market share or cause us to lose market share, which may require us to lower prices, or increase marketing and advertising expenditures, either of which would adversely affect our margins and could result in a decrease in our operating results and profitability. We cannot assure you that we will be able to maintain a competitive position or compete successfully against such sources of competition.
We may suffer reputational harm due to real or perceived quality or health issues with products manufactured by our licensees using our technology.
Any real or perceived quality or food safety concerns or failures to comply with applicable food regulations and requirements, whether or not ultimately based on fact and whether or not involving us, or even merely involving unrelated manufacturers, could cause negative publicity and reduced confidence in our company, or the industry as a whole, which could in turn harm our reputation and sales, and could adversely impact our business, financial condition and operating results. There can be no assurance that products manufactured by our licensees will always comply with regulatory standards. Although we expect that our licensees will strive to manufacture products free of pathogenic organisms, these may not be easily detected and cross-contamination can occur. We cannot assure you that this health risk will always be preempted by quality control processes.
We will have no control over the products manufactured by our licensees, especially once they are purchased by consumers, who may prepare these products in a manner that is inconsistent with directions or store them for excessive periods of time, which may adversely affect their quality and safety. If the products manufactured by our licensees are not perceived as safe or of high quality, then our business, results of operations and financial condition could be adversely affected.
The growing use of social and digital media increases the speed and extent that information or misinformation and opinions can be shared. Negative publicity about cultured meats produced using our technologies could seriously damage our reputation.
Failure to improve our technologies may adversely affect our ability to continue to grow.
In order to continue to grow, we expect we will need to continue to innovate by developing new technologies or improving existing ones, in ways that meet our standards for quality and will enable our eventual licensees to manufacture products that appeal to consumer preferences. Such innovation will depend on the technical capability of our staff in developing and testing product prototypes, including complying with applicable governmental regulations, and the success of our management and sales and marketing teams in introducing and marketing new technologies. Failure to develop and market new technologies may cause a negative impact on our business and results of operations.
Additionally, the development and introduction of new technologies requires substantial research, development and marketing expenditures, which we may be unable to recoup if the new technologies do not lead to products that gain widespread market acceptance. If we are unsuccessful in meeting our objectives with respect to new or improved technologies, our business could be harmed.
We may face difficulties if we expand our operations into new geographic regions, in which we have no prior operating experience.
We intend to license our technologies in numerous geographical markets. International operations involve a number of risks, including foreign regulatory compliance, tariffs, taxes and exchange controls, economic downturns, inflation, foreign currency fluctuations and political and social instability in the countries in which we will operate. Expansion may involve expanding into less developed countries, which may have less political, social or economic stability and less developed infrastructure and legal systems. It is costly to establish, develop and maintain international operations and develop and promote our brands in international markets. As we expand our business into other countries, we may encounter regulatory, legal, personnel, technological and other difficulties that increase our expenses and/or delay our ability to become profitable in such countries, which may have an adverse impact on our business and brand.
Consumer preferences for alternative proteins in general, and more specifically cultured meats, are difficult to predict and may change, and, if we are unable to respond quickly to new trends, our business may be adversely affected.
Our business is focused on the development and marketing of licensable cultured meat manufacturing technologies. Consumer demand for the cultured meats manufactured using these technologies could change based on a number of possible factors, including dietary habits and nutritional values, concerns regarding the health effects of ingredients and shifts in preference for various product attributes. If consumer demand for our products decreases, our business and financial condition would suffer. Consumer trends that we believe favor sales of products manufactured using our licensed technologies could change based on a number of possible factors, including a shift in preference from animal-based protein products, economic factors and social trends. A significant shift in consumer demand away from products manufactured using our technologies could reduce our sales or our market share and the prestige of our brand, which would harm our business and financial condition.
We have no manufacturing experience or resources and we expect we will incur significant costs to develop this expertise or need to rely on third parties for manufacturing.
We have no manufacturing experience. In order to develop and license our technologies, we will need to develop, contract for or otherwise arrange for the necessary manufacturing capabilities. We may experience difficulty in obtaining adequate and timely manufacturing capacity for our proprietary cultured meat printers and bio-inks. We do not own or lease facilities currently that could be used to manufacture any products that we might develop on an industrial scale, nor do we have the resources at this time to acquire or lease suitable facilities. If we are unable to build the necessary internal manufacturing capability or obtain this capability through third parties we will not be able to commercialize our technologies. Even if we develop or obtain the necessary manufacturing capacity, if we fail to comply with regulations, to obtain the necessary licenses and knowhow or to obtain the requisite financing in order to comply with all applicable regulations and to own or lease the required facilities in order to manufacture products, we could be forced to cease operations, which would cause you to lose all of your investment.
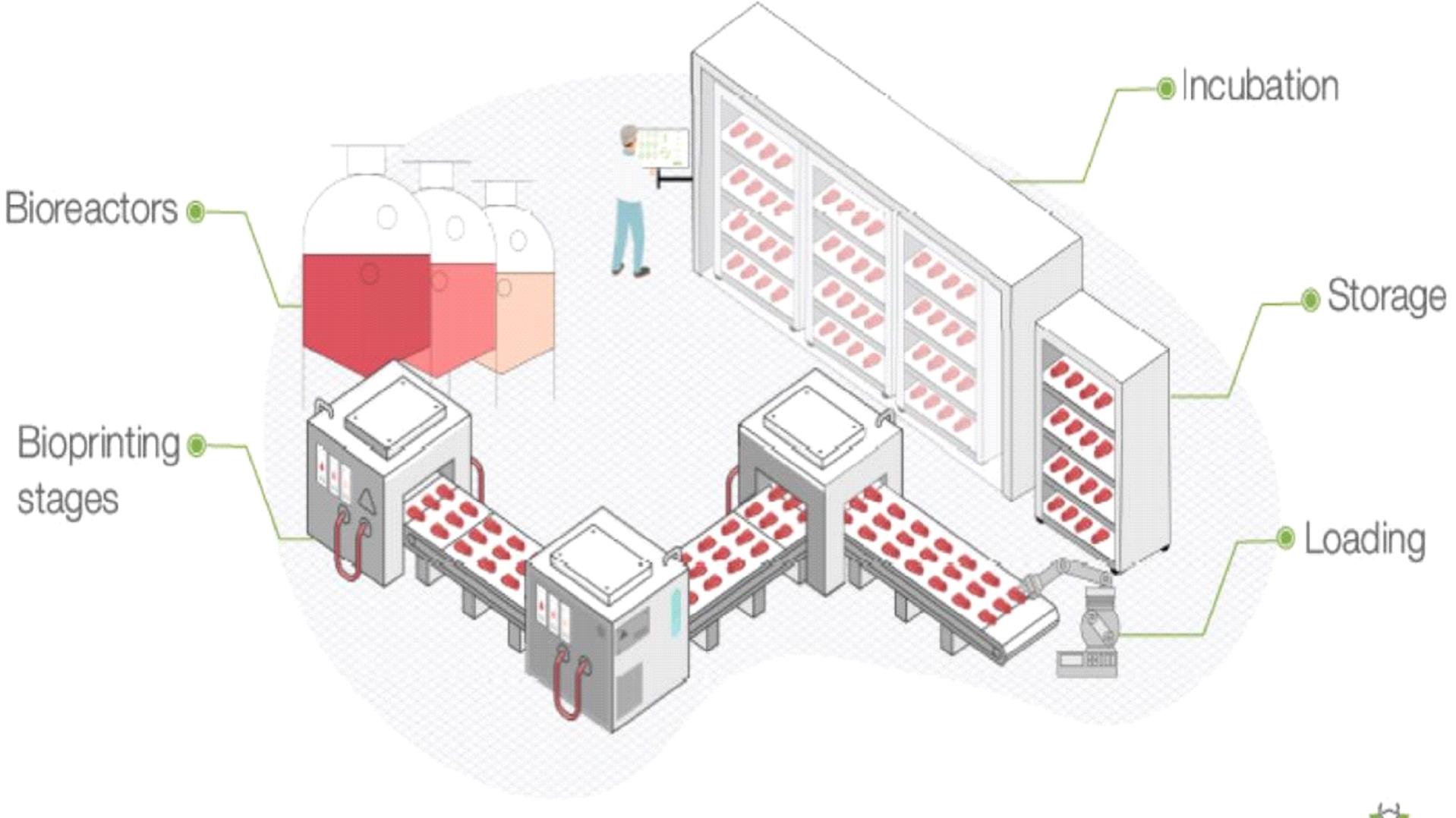
Our limited manufacturing capabilities are nascent, and if we fail to effectively develop manufacturing and production capabilities, our business and operating results and our brand reputation could be harmed.
We intend to begin manufacturing certain of our culture meat products in a pilot facility. If we are unable to develop manufacturing capacity to commence cultured chicken fat production, we may not be able to expedite our market entry or develop an industrial process for cultivating and producing real meat cuts. Additionally, there is risk in our ability to effectively scale production processes, if developed, to optimize manufacturing capacity for specific products and effectively manage our supply chain requirements, which involves accurately forecasting demand for each of our products and inventory needs in order to ensure we have adequate available manufacturing capacity for each such product and to ensure we are effectively managing our inventory. Our forecasts may be based on multiple assumptions that may cause our estimates to be inaccurate and affect our ability to obtain adequate manufacturing capacity (whether our own manufacturing capacity or co-manufacturing capacity) and adequate inventory supply in order to meet the demand for our products, which could prevent us from developing our industrial process and harm our business and prospects.
However, if we overestimate our demand and overbuild our capacity or inventory, we may have significantly underutilized assets, experience reduced margins, and have excess inventory which we may be required to write-down or write-off. If we do not accurately align our manufacturing capabilities and inventory supply with demand, if we experience disruptions or delays in our supply chain, or if we cannot obtain raw materials of sufficient quantity and quality at reasonable prices and in a timely manner, our business, financial condition and results of operations may be adversely affected.
If developed, our manufacturing and production operations will be subject to additional risks and uncertainties.
The interruption in, or the loss of operations at, our pilot facility, which may be caused by work stoppages, labor shortages, strikes or other labor unrest, production disruptions, product quality issues, local economic and political conditions, restrictive governmental actions, border closures, disease outbreaks or pandemics (such as COVID-19), the outbreak of hostilities, acts of war, terrorism, fire, earthquakes, severe weather, flooding or other natural disasters at one or more of these facilities, could delay, postpone or reduce production of some of our products, could impede our ability to develop an industrial process for cultivating and producing real meat cuts or could otherwise have an adverse effect on our business, results of operations and financial condition until such time as such interruption is resolved or an alternate source of production is secured.
Because we rely on a limited number of third-party suppliers, we may not be able to obtain raw materials on a timely basis or in sufficient quantities at competitive prices to produce our products or meet the demand for our products.
We rely on a limited number of vendors, a portion of which are located internationally, to supply us with raw materials. Our financial performance depends in large part on our ability to arrange for the purchase of raw materials in sufficient quantities at competitive prices. We are not assured a continued supply or pricing of raw materials. Any of our other suppliers could discontinue or seek to alter their relationship with us. We could experience similar delays in the future from any of our suppliers. Any disruption in the supply of embryonic stem cells or other raw materials would have a material adverse effect on our business if we cannot replace these suppliers in a timely manner, on commercially reasonable terms, or at all.
In addition, our suppliers manufacture their products at a limited number of facilities. A natural disaster, severe weather, fire, power interruption, work stoppage or other calamity affecting any of these facilities, or any interruption in their operations, could negatively impact our ability to obtain required quantities of raw materials in a timely manner, or at all, which could materially increase our cost and delay our timeline, and have a material effect on our business and financial condition.
Events that adversely affect our suppliers of raw materials could impair our ability to obtain raw material inventory in the quantities at competitive prices that we desire. Such events include problems with our suppliers’ businesses, finances, labor relations and/or shortages, strikes or other labor unrest, ability to import raw materials, product quality issues, costs, production, insurance and reputation, as well as local economic and political conditions, restrictive U.S. and foreign governmental actions, such as restrictions on transfers of funds and trade protection measures, including export/import duties and quotas and customs duties and tariffs, adverse fluctuations in foreign currency exchange rates, changes in legal or regulatory requirements, border closures, disease outbreaks or pandemics (such as COVID-19), acts of war, terrorism, natural disasters, fires, earthquakes, flooding, severe weather, agricultural diseases or other catastrophic occurrences. We continuously seek alternative sources of raw materials to use in our products, but we may not be successful in diversifying the raw materials we use in our products.
If we need to replace an existing supplier, there can be no assurance that supplies of raw materials will be available when required on acceptable terms, or at all, or that a new supplier would allocate sufficient capacity to us in order to meet our requirements, fill our orders in a timely manner or meet our strict quality standards.
Litigation or legal proceedings, government investigations or other regulatory enforcement actions could subject us to civil and criminal penalties or otherwise expose us to significant liabilities and have a negative impact on our reputation or business.
We operate in a constantly evolving legal and regulatory framework. Consequently, we are subject to heightened risk of legal claims, government investigations or other regulatory enforcement actions. Although we have implemented policies and procedures designed to ensure compliance with existing laws and regulations, we cannot assure you that our employees, temporary workers, contractors or agents will not violate our policies and procedures. Moreover, a failure to maintain effective control processes could lead to violations, unintentional or otherwise, of laws and regulations. Legal claims, government investigations or regulatory enforcement actions arising out of our failure or alleged failure to comply with applicable laws and regulations could subject us to civil and criminal penalties that could materially and adversely affect our product sales, reputation, financial condition and operating results. For example, in November 2020, the Israel Securities Authority, or ISA, initiated an administrative proceeding against us claiming negligent misstatement regarding certain immediate and periodic reports published by our predecessor (Ophectra) during the years 2017 and 2018, prior to the merger with MeaTech. These reports relate to Ophectra’s activities prior to establishment of the settlement fund in connection with the merger. In April 2021, following negotiations with the ISA, we agreed to settle the matter for NIS 0.7 million ($0.22 million). The settlement is subject to approval of the ISA’s Enforcement Committee.
RISKS RELATED TO OUR OPERATIONS
We expect that a small number of customers will account for a significant portion of our revenues, and the loss of one or more of these customers could adversely affect our financial condition and results of operations.
We do not expect to generate revenue in the short or medium term. If we are able to generate revenue, we believe that we will do so through three primary streams: (i) licensing our proprietary intellectual property to customers for the purpose of commercializing our technologies, including by way of setting up and operating cultured meat production factories; (ii) brokering the supply of materials needed in the manufacturing process; and (iii) providing consulting and implementation services to customers. These streams may arise in the context of product co-development. Under this model, we initially expect to derive a significant portion of our revenues from a few customers. Our financial condition and results of operations could be adversely impacted if any one of these customers interrupt or curtail their activities, fail to pay for the services that have been performed, terminate their cultured meat operations, or if we are unable to enter into agreements with new customers on favorable terms. The loss of customers could adversely affect our financial condition and results of operations.
We may be exposed to the credit risks of our customers, and nonpayment by these customers and other parties could adversely affect our financial position, results of operations and cash flows.
We may be subject to risks of loss resulting from nonpayment by our customers. Any material nonpayment by these entities could adversely affect our financial position, results of operations and cash flows. If customers default on their obligations to us, our financial results and condition could be adversely affected. Some of these customers may be highly leveraged and subject to their own operating and regulatory risks.
If we are unable to attract and retain qualified employees, our ability to implement our business plan may be adversely affected.
The loss of the service of employees, such as Mr. Arik Kaufman, our Chief Executive Officer, would likely delay our achievement of product development and other business objectives, as we may not be able to find suitable individuals to replace them on a timely basis, if at all. In addition, any such departure could be viewed in a negative light by investors and analysts, which may cause the price of our ordinary shares to decline. Although we have employment agreements with our key employees, these employees could terminate their employment with us at any time on relatively short notice. We do not carry key man life insurance on any of our executive officers.
Recruiting and retaining qualified scientific, manufacturing and sales and marketing personnel will also be critical to our success. We may not be able to attract and retain these personnel on acceptable terms given the competition among high technology and life sciences companies for similar personnel. We also experience competition from universities and research institutions in attracting and retaining scientific personnel. In addition, we rely on consultants and advisors, including scientific advisors, to assist us in formulating our research and development and commercialization strategy. Our consultants and advisors may be employed by employers other than us and may have commitments under consulting or advisory contracts with other entities that may limit their availability to us.
Under applicable employment laws, we may not be able to enforce covenants not to compete.
Our employment agreements generally include covenants not to compete. These agreements prohibit our employees, if they cease working for us, from competing directly with us or working for our competitors for a limited period. We may be unable to enforce these agreements under the laws of the jurisdictions in which our employees work. For example, Israeli courts have required employers seeking to enforce covenants not to compete to demonstrate that the competitive activities of a former employee will harm one of a limited number of material interests of the employer, such as the secrecy of a company’s confidential commercial information or the protection of its intellectual property. If we cannot demonstrate that such an interest will be harmed, we may be unable to prevent our competitors from benefiting from the expertise of our former employees or consultants and our competitiveness may be diminished.
We are exposed to a risk of substantial loss due to claims that may be filed against us in the future because our insurance policies may not fully cover the risk of loss to which we are exposed.
We are exposed to the risk of having claims seeking monetary damages being filed against us, for example with regard to securities-related claims. In the event that we are required to pay damages for any such claim, we may be forced to seek bankruptcy or to liquidate because our asset base and revenue base may be insufficient to satisfy the payment of damages and any insurance that we have obtained may not provide sufficient coverage against potential liabilities.
Our business and operations would suffer in the event of information technology system failures, including security breaches.
Despite the implementation of security measures, our internal computer systems and those of our contractors and consultants are vulnerable to damage from computer viruses, unauthorized access, natural disasters, terrorism, war and telecommunication and electrical failures. While we have not experienced any such system failure, accident or security breach to date, if such an event were to occur and cause interruptions in our operations, causing our business to suffer. To the extent that any disruption or security breach were to result in a loss of or damage to our data or applications, or inappropriate disclosure of confidential or proprietary information, we could incur liability and the further development of our product could be delayed.
A cybersecurity incident, other technology disruptions or failure to comply with laws and regulations relating to privacy and the protection of data relating to individuals could negatively impact our business, our reputation and our relationships with customers.
We use computers in substantially all aspects of our business operations. We also use mobile devices, social networking and other online activities to connect with our employees, suppliers and co-manufacturers. Such uses give rise to cybersecurity risks, including security breaches, espionage, system disruption, theft and inadvertent release of information. Our business involves the storage and transmission of numerous classes of sensitive and/or confidential information and intellectual property, including suppliers’ information, private information about employees and financial and strategic information about us and our business partners. Further, as we pursue new initiatives that improve our operations and cost structure, potentially including acquisitions, we may also be expand and improve our information technologies, resulting in a larger technological presence and corresponding exposure to cybersecurity risk. If we fail to assess and identify cybersecurity risks associated with new initiatives or acquisitions, we may become increasingly vulnerable to such risks. Additionally, while we have implemented measures to prevent security breaches and cyber incidents, our preventative measures and incident response efforts may not be entirely effective. The theft, destruction, loss, misappropriation, or release of sensitive and/or confidential information or intellectual property, or interference with our information technology systems or the technology systems of third parties on which we rely, could result in business disruption, negative publicity, brand damage, violation of privacy laws, loss of customers, potential liability and competitive disadvantage all of which could have a material adverse effect on our business, financial condition or results of operations.
In addition, we are subject to laws, rules and regulations in the United States, the European Union and other jurisdictions relating to the collection, use and security of personal information and data. Such data privacy laws, regulations and other obligations may require us to change our business practices and may negatively impact our ability to expand our business and pursue business opportunities. We may incur significant expenses to comply with the laws, regulations and other obligations that apply to us. Additionally, the privacy- and data protection-related laws, rules and regulations applicable to us are subject to significant change. Several jurisdictions have passed new laws and regulations in this area, and other jurisdictions are considering imposing additional restrictions. Privacy- and data protection-related laws and regulations also may be interpreted and enforced inconsistently over time and from jurisdiction to jurisdiction. Any actual or perceived inability to comply with applicable privacy or data protection laws, regulations, or other obligations could result in significant cost and liability, litigation or governmental investigations, damage our reputation, and adversely affect our business.
Disruptions in the worldwide economy may adversely affect our business, results of operations and financial condition.
The global economy can be negatively impacted by a variety of factors such as the spread or fear of spread of contagious diseases (such as the COVID-19 pandemic), man-made or natural disasters, actual or threatened war, terrorist activity, political unrest, civil strife and other geopolitical uncertainty. Such adverse and uncertain economic conditions may impact consumer demand for alternative proteins in general, and clean meats specifically, which may in turn impact manufacturer and retailer demand for our technologies. In addition, our ability to manage normal commercial relationships with suppliers may suffer. Consumers may shift purchases to lower-priced or other perceived value offerings during economic downturns as a result of various factors, including job losses, inflation, higher taxes, reduced access to credit, change in government economic policy and international trade disputes. In particular, consumers may reduce the amount of cultured meat that they purchase in favor of conventional meat or other alternative proteins, which may have lower retail prices, which could indirectly affect our results of operations. Manufacturer and retailers may become more conservative in response to these conditions and seek to delay commencing cultured market manufacturing operations or reduce existing operations. Our results of operations will depend upon, among other things, the financial condition of our business customers and our ability to supply them with the means to manufacture products that appeal to consumers at the right price. Decreases in demand for the products manufactured by our customers would put downward pressure on margins and would negatively impact our financial results. Prolonged unfavorable economic conditions or uncertainty may result in end consumers making long-lasting changes to their discretionary spending behavior on a more permanent basis, which may likewise have an indirect adverse effect on our sales and profitability.
RISKS RELATED TO GOVERNMENT REGULATION
We expect that products utilizing our technologies will be subject to regulations that could adversely affect our business and results of operations.
The manufacture and marketing of food products is highly regulated. We, our suppliers and licensees, may be subject to a variety of laws and regulations. These laws and regulations apply to many aspects of our business, including the manufacture, composition and ingredients, packaging, labeling, distribution, advertising, sale, quality and safety of food products, as well as the health and safety of our employees and the protection of the environment.
We are focused on developing a novel, proprietary three-dimensional bioprinter to deposit layers of cells (including stem cells and differentiated stem cells), scaffolding, and cell nutrients in a three-dimensional form of structured cultured meat. The cultured meat, in turn, will be produced by our customers. Peace of Meat intends to produce cultured avian fat that is anticipated to be used as an ingredient, inter alia, in the production of finished cultured poultry. Neither we nor Peace of Meat intend to manufacture, distribute and sell branded cultured-meat end products for consumer consumption.
Peace of Meat is a Business-To-Business, or B2B, ingredient producer and will be subject to regulation by the U.S. Food and Drug Administration, or FDA, to the extent its products are introduced to the United States for use by a manufacturer to produce cultured meat or other food in the United States, and analogous foreign regulatory bodies elsewhere. In the US, the FDA and the U.S. Department of Agriculture’s, or USDA's, Food Safety and Inspection Service, or FSIS share an ingredient approval process. FDA determines the safety of substances and prescribes safe conditions of use. USDA-FSIS determines the efficacy and suitability of food ingredients in meat, poultry, and egg products. Thus, the USDA’s efficacy and suitability requirements will also apply to the extent the ingredients are destined for use in USDA-regulated meat and poultry products.
For the reasons discussed below, we ourselves do not expect to be directly regulated by the FDA for United States compliance purposes but will apply FDA’s food contact substance standards or analogous foreign regulations when developing our three-dimensional bioprinter. Specifically, we intend to license our production technology, as well as provide associated products and services to food processing and food retail companies through a B2B model. From a regulatory perspective, in the United States, we expect companies manufacturing finished cultured meat products to be subject to regulation by various government agencies, including the FDA U.S. Department of Agriculture, U.S. Federal Trade Commission, or FTC, Occupational Safety and Health Administration and the Environmental Protection Agency, as well as the requirements of various state and local agencies, such as the California Safe Drinking Water and Toxic Enforcement Act of 1986. We likewise expect these products to be regulated by equivalent agencies outside the United States by various international regulatory bodies.
As the manufacturer of technology used to produce cultured meat, and consistent with the Federal Food, Drug and Cosmetic Act, Federal Meat Inspection Act, and Poultry Products Inspection Act, we believe we will not directly be regulated by the FDA or USDA. Rather, we believe the regulatory obligation falls on our customers — cultured meat producers — to ensure that all food produced using our technology is wholesome and not adulterated. Consistent with food industry norms, we expect that our customers will therefore request assurances from us that our products are suitable for their intended use from an FDA regulatory perspective. Therefore, we plan to apply FDA food safety standards when developing our three-dimensional bioprinter as a means of assuring our customers that our bioprinter is safe for its intended use and will not result in the production of adulterated food. In particular, we plan to apply applicable food contact substance requirements, such as those of the FDA, when developing its three-dimensional bioprinter as a means of assuring customers using the Company's technology that our bioprinter is safe for its intended use and will not result in the production of adulterated food. If we are unable to provide regulatory compliance assurance to our customers, we expect that our ability to license our production technology would be adversely impacted.
The manufacturing of cultured meat is expected to be subject to extensive regulations internationally, with products subject to numerous food safety and other laws and regulations relating to the sourcing, manufacturing, composition and ingredients, storing, labeling, marketing, advertising and distribution of these products. In addition, enforcement of existing laws and regulations, changes in legal requirements and/or evolving interpretations of existing regulatory requirements may result in increased compliance costs and create other obligations, financial or otherwise, that could adversely affect our business, financial condition or operating results. In addition, we could be adversely affected by violations of the U.S. Foreign Corrupt Practices Act, or FCPA, and similar worldwide anti-bribery laws, which generally prohibit companies and their intermediaries from making improper payments to officials or other third parties for the purpose of obtaining or retaining business. While our policies mandate compliance with anti-bribery laws, our internal control policies and procedures may not protect us from reckless or criminal acts committed by our employees, contractors or agents. Violations of these laws, or allegations of such violations, could disrupt our business and adversely impact our results of operations, cash flows and financial condition.
Any changes in, or changes in the interpretation of, applicable laws, regulations or policies of the USDA, state regulators or similar foreign regulatory authorities that relate to the use of the word “meat” or other similar words in connection with cultured meat products could adversely affect our business, prospects, results of operations or financial condition.
The USDA, state regulators or similar foreign regulatory authorities, such as Health Canada or the Canadian Food Inspection Agency, or CFIA, or authorities of the European Union (EU) or the EU member states (e.g., European Food Safety Authority, or EFSA), could take action to impact our ability to use the term “meat” or similar words, such as “beef”, to describe the product our bioprinters will produce. In addition, a food may be deemed misbranded if its labeling is false or misleading in any particular way, and the USDA, CFIA, EFSA or other regulators could interpret the use of the term “meat” or any similar phrase(s) to describe our cultured meat products as false or misleading or likely to create an erroneous impression regarding their composition. In the U.S., USDA will be developing new labeling requirements for foods under its jurisdiction produced through cell culture technology as noted in an Advance Notice of Proposed Rulemaking (ANPR) published in September 2021.
Failure by our raw materials suppliers to comply with food safety, environmental or other laws and regulations, or with the specifications and requirements of our products, may disrupt our supply of products and adversely affect our business.
If our suppliers fail to comply with food safety, environmental or other laws and regulations, or face allegations of non-compliance, their operations may be disrupted. In the event of actual or even alleged non-compliance, we might be forced to find an alternative supplier and we may be subject to lawsuits related to such non-compliance by our suppliers. As a result, our supply of raw materials could be disrupted or our costs could increase, which would adversely affect our business, results of operations and financial condition. The failure of any supplier to produce products that conform to our standards could adversely affect our reputation in the marketplace and result in economic loss. Additionally, actions we may take to mitigate the impact of any disruption or potential disruption in our supply of raw materials, including increasing inventory in anticipation of a potential supply or production interruption, may adversely affect our business, results of operations and financial condition.
RISKS RELATED TO OUR INTELLECTUAL PROPERTY AND POTENTIAL LITIGATION
If we are unable to obtain and maintain effective patent rights for our products, we may not be able to compete effectively in our markets. If we are unable to protect the confidentiality of our trade secrets or know-how, such proprietary information may be used by others to compete against us.
Since September 2019, we have sought patent protection for certain of our products, systems, processes, designs and applications. Our success depends in large part on our ability to obtain, maintain, monitor and enforce patent and other intellectual property protection in the United States and in other countries with respect to our proprietary technology and new products.
We seek to protect our proprietary position and sustain our competitive advantage by filing patent applications in the United States and in other countries. Patent prosecution in the United States and the rest of the world is uncertain, expensive and time consuming, and we may not be able to file and prosecute all necessary or desirable patent applications at a reasonable cost or in a timely manner in all the necessary locations. It is also possible that we will fail to identify Patentable aspects of our research and development output before it is too late to obtain patent protection.
We have a growing portfolio of 14 provisional and non-provisional pending patent applications, with a robust pipeline. These are filed with the U.S. Patent and Trademark Office, or USPTO, the World Intellectual Property Organization, or WIPO, and when the time comes, in various patent offices around the world, such as Israel, China, Japan, Europe, Canada, and South Korea. Three of the pending patent applications were filed through the Paris Convention Treaty, or PCT. We cannot offer any assurances about which, if any of the pending patent applications will issue, the scope of protection of any such patent or whether any issued patents will be found invalid and/or unenforceable or will be threatened by third parties. Any successful opposition to these patents or any other patents owned by or licensed to us after patent issuance could deprive us of rights necessary for the successful commercialization of any new products that we may develop.
Further, there is no assurance that all potentially relevant prior art relating to our patent applications has been found, which can invalidate a patent or prevent a patent from issuing from a pending patent application. Even if patents do successfully issue, and even if such patents cover our products, third parties may challenge their validity, enforceability, or scope, which may result in such patents being narrowed, found unenforceable or invalidated. Furthermore, even if they are unchallenged, our patent applications and any future patents may not adequately protect our intellectual property, provide exclusivity for our new products, or prevent others from designing around our claims. Any of these outcomes could impair our ability to prevent competition from third parties, which may have an adverse impact on our business.
If we cannot obtain and maintain effective patent rights for our products, we may not be able to compete effectively, and our business and results of operations would potentially be harmed.
If we are unable to maintain effective proprietary rights for our products, we may not be able to compete effectively in our markets.
In addition to the protection afforded by any patents currently owned and that may be granted, historically, we have relied on trade secret protection and confidentiality agreements to protect proprietary know-how that is not patentable or that we elect not to patent, processes and helpful devices that are not easily known, knowable or easily ascertainable, and for which patent infringement is difficult to monitor and enforce and any other elements of our product development processes, that involve proprietary know-how, as well as information or technology that is not covered by patents. However, trade secrets can be difficult to protect. We seek to protect our proprietary technology and processes, in part, by entering into confidentiality agreements with our employees, consultants, scientific advisors, and contractors. We also seek to preserve the integrity and confidentiality of our data, trade secrets and intellectual property by maintaining physical security of our premises and physical and electronic security of our information technology systems, as well as implementing various standard operating procedures designed to maintain that integrity. Agreements or security measures may be breached, and we may not have adequate remedies for any breach. In addition, our trade secrets and intellectual property may otherwise become known or be independently discovered by competitors.
We cannot provide any assurances that our trade secrets and other confidential proprietary information will not be disclosed in violation of our confidentiality agreements or that competitors will not otherwise gain access to our trade secrets or independently develop substantially equivalent information and techniques. Also, misappropriation or unauthorized and unavoidable disclosure of our trade secrets and intellectual property could impair our competitive position and may have a material adverse effect on our business. Additionally, if the steps taken to maintain our trade secrets and intellectual property are deemed inadequate, we may have insufficient recourse against third parties for misappropriating any trade secret.
Intellectual property rights of third parties could adversely affect our ability to successfully commercialize our products, and we might be required to litigate or obtain licenses from third parties in order to develop or market our product candidates. Such litigation or licenses could be costly or not available on commercially reasonable terms.
At this stage, and in the future it is inherently difficult to conclusively assess our freedom to operate without infringing on third party rights. Our competitive position may be adversely affected if existing patents or patents resulting from patent applications issued to third parties or other third-party intellectual property rights are held to cover our products, systems and processes or elements thereof, or our manufacturing or uses relevant to our development plans. In such cases, we may be limited, or not be in a position to develop or commercialize products or our product candidates unless we successfully pursue litigation to nullify or invalidate the third party intellectual property right concerned, or enter into a license agreement with the intellectual property rights’ holder, if available on commercially reasonable terms. There may also be pending patent applications that should they result in issued patents, could be alleged to be infringed by our new products. If such an infringement claim should be brought and be successful, we may be required to pay substantial damages, royalties, be forced to abandon our new products or seek a license from any patent holders. No assurances can be given that a license will be available on commercially reasonable terms, if at all.
It is also possible that we have failed to identify relevant third party patents or applications. For example, U.S. patent applications filed before July 8, 2019 and certain U.S. patent applications filed after that date that will not be filed outside the United States remain confidential until patents issue. Patent applications in the United States and elsewhere are published 18 months after the earliest filing for which priority is claimed, with such earliest filing date being commonly referred to as the priority date. Therefore, patent applications covering our new products or platform technology could have been filed by others without our knowledge. Additionally, pending patent applications which have been published can, subject to certain limitations, be later amended in a manner that could cover our platform technologies, our new products or the use of our new products. Third party intellectual property right holders may also actively bring infringement claims against us. We cannot guarantee that we will be able to successfully settle or otherwise resolve such infringement claims. If we are unable to successfully settle future claims on terms acceptable to us, we may be required to engage in or continue costly, unpredictable and time-consuming litigation and may be prevented from or experience substantial delays in pursuing the development of and/or marketing our new products. If we fail in any such dispute, in addition to being forced to pay damages, we may be temporarily or permanently prohibited from commercializing our new products that are held to be infringing. We might, if possible, also be forced to redesign our new products so that we no longer infringe the third party intellectual property rights. Any of these events, even if we were ultimately to prevail, could require us to divert substantial financial and management resources that we would otherwise be able to devote to our business.
Third-party claims of intellectual property infringement may prevent or delay our development and commercialization efforts.
Our commercial success depends in part on our avoiding infringement of the patents and proprietary rights of third parties. Numerous U.S. and foreign issued patents and pending patent applications, which are owned by third parties, exist in the fields in which we are developing new products. As our industries expand and more patents are issued, the risk increases that our products may be subject to claims of infringement of the patent rights of third parties.
Third parties may assert that we are employing their proprietary technology without authorization. There may be third-party patents or patent applications with claims to materials, designs or methods of manufacture related to the use or manufacture of our products. There may be currently pending patent applications that may later result in issued patents that our products may infringe. In addition, third parties may obtain patents in the future and claim that use of our technologies infringes upon these patents.
If any third-party patents were held by a court of competent jurisdiction to cover aspects of our formulations, processes for designs, or methods of use, the holders of any such patents may be able to block our ability to develop and commercialize the applicable product candidate unless we obtain a license or until such patent expires or is finally determined to be invalid or unenforceable. In either case, such a license may not be available on commercially reasonable terms or at all.
Parties making claims against us may obtain injunctive or other equitable relief, which could effectively block our ability to further develop and commercialize one or more of our products. Defense of these claims, regardless of their merit, would involve substantial litigation expense and would be a substantial diversion of employee resources from our business. In the event of a successful claim of infringement against us, we may have to pay substantial damages, including treble damages and attorneys’ fees for willful infringement, pay royalties, redesign our infringing products or obtain one or more licenses from third parties, which may be impossible or require substantial time and monetary expenditure.
Patent policy and rule changes could increase the uncertainties and costs surrounding the prosecution of our patent applications and the enforcement or defense of any issued patents.
Changes in either the patent laws or interpretation of the patent laws in the United States and other countries may diminish the value of any patents that may issue from our patent applications, or narrow the scope of our patent protection. The laws of foreign countries may not protect our rights to the same extent as the laws of the United States. Publications of discoveries in the scientific literature often lag behind the actual discoveries, and patent applications in the United States and other jurisdictions are typically not published until 18 months after filing, or in some cases not at all. We therefore cannot be certain that we were the first to file the invention claimed in our owned and licensed patent or pending applications, or that we or our licensor were the first to file for patent protection of such inventions. Assuming all other requirements for patentability are met, in the United States prior to March 15, 2013, the first to make the claimed invention without undue delay in filing, is entitled to the patent, while outside the United States, the first to file a patent application is entitled to the patent. After March 15, 2013, under the Leahy-Smith America Invents Act, or the Leahy-Smith Act, enacted on September 16, 2011, the United States has moved to a first to file system. The Leahy-Smith Act also includes a number of significant changes that affect the way patent applications will be prosecuted and may also affect patent litigation. In general, the Leahy-Smith Act and its implementation could increase the uncertainties and costs surrounding the prosecution of our patent applications and the enforcement or defense of any issued patents, all of which could have a material adverse effect on our business and financial condition.
We may be involved in lawsuits to protect or enforce our intellectual property, which could be expensive, time consuming, and unsuccessful.
Competitors may infringe our intellectual property. If we were to initiate legal proceedings against a third party to enforce a patent covering one of our new products, the defendant could counterclaim that the patent covering our product candidate is invalid and/or unenforceable. In patent litigation in the United States, defendant counterclaims alleging invalidity and/or unenforceability are commonplace. Grounds for a validity challenge could be an alleged failure to meet any of several statutory requirements, including lack of novelty, obviousness, or non-enablement. Grounds for an unenforceability assertion could be an allegation that someone connected with prosecution of the patent withheld relevant information from the USPTO, or made a misleading statement, during prosecution. Under the Leahy-Smith Act, the validity of U.S. patents may also be challenged in post-grant proceedings before the USPTO. The outcome following legal assertions of invalidity and unenforceability is unpredictable.
Derivation proceedings initiated by third parties or brought by us may be necessary to determine the priority of inventions and/or their scope with respect to our patent or patent applications or those of our licensors. An unfavorable outcome could require us to cease using the related technology or to attempt to license rights to it from the prevailing party. Our business could be harmed if the prevailing party does not offer us a license on commercially reasonable terms. Our defense of litigation or interference proceedings may fail and, even if successful, may result in substantial costs and distract our management and other employees. In addition, the uncertainties associated with litigation could have a material adverse effect on our ability to raise the funds necessary to continue our clinical trials, continue our research programs, license necessary technology from third parties, or enter into development partnerships that would help us bring our new products to market.
Furthermore, because of the substantial amount of discovery required in connection with intellectual property litigation, there is a risk that some of our confidential information could be compromised by disclosure during this type of litigation. There could also be public announcements of the results of hearings, motions, or other interim proceedings or developments. If securities analysts or investors perceive these results to be negative, it could have a material adverse effect on the price of our traded securities.
We may, in the future, be subject to claims that our employees, consultants, or independent contractors have wrongfully or unavoidably used or disclosed confidential information of third parties or that our employees have wrongfully used or disclosed alleged trade secrets of their former employers.
We continue to employ individuals who were previously employed at our competitors or potential competitors. We have established standard operating procedures to try and ensure that our employees, consultants, and independent contractors do not use the proprietary information or know-how of others in their work for us, but we may nevertheless be subject to claims that we or our employees, consultants, or independent contractors have inadvertently or otherwise used or disclosed intellectual property, including trade secrets or other proprietary information, of any of our employees’ former employers or other third parties. Litigation may result and be necessary to defend against these claims. If we fail in defending any such claims, in addition to paying monetary damages, we may lose valuable intellectual property rights or personnel, which could adversely impact our business. Even if we are successful in defending against such claims, litigation could result in substantial costs and be a distraction to management and other employees.
We may be subject to claims challenging the inventorship of our intellectual property.
We may be subject to claims that former employees, collaborators or other third parties have an interest in, or right to compensation, with respect to our current patent and patent applications, future patents or other intellectual property as an inventor or co-inventor. For example, we may have inventorship disputes arise from conflicting obligations of consultants or others who are involved in developing our products. Litigation may be necessary to defend against these and other claims challenging inventorship or claiming the right to compensation. If we fail in defending any such claims, in addition to paying monetary damages, we may lose valuable intellectual property rights, such as exclusive ownership of, or right to use, valuable intellectual property. Such an outcome could have a material adverse effect on our business. Even if we are successful in defending against such claims, litigation could result in substantial costs and be a distraction to management and other employees.
We may not be able to protect our intellectual property rights throughout the world.
Filing, prosecuting, and defending patents on products, as well as monitoring their infringement in all countries throughout the world would be prohibitively expensive, and our intellectual property rights in some countries can be less extensive than those in the United States. In addition, the laws of some foreign countries do not protect intellectual property rights to the same extent as federal and state laws in the United States.
Competitors may use our technologies in jurisdictions where we have not obtained patent protection to develop their own products and may also export otherwise infringing products to territories where we have patent protection, but enforcement is not as strong as that in the United States. These products may compete with our products. Future patents or other intellectual property rights may not be effective or sufficient to prevent them from competing.
Many companies have encountered significant problems in protecting and defending intellectual property rights in foreign jurisdictions. The legal systems of certain countries, particularly certain developing countries, do not favor the enforcement of patents, trade secrets, and other intellectual property protection, which could make it difficult for us to stop the marketing of competing products in violation of our proprietary rights generally. Proceedings to enforce our patent rights in foreign jurisdictions, whether or not successful, could result in substantial costs and divert our efforts and attention from other aspects of our business, could put our future patents at risk of being invalidated or interpreted narrowly and our patent applications at risk of not issuing and could provoke third parties to assert claims against us. We may not prevail in any lawsuits that we initiate and the damages or other remedies awarded, if any, may not be commercially meaningful. Accordingly, our efforts to monitor and enforce our intellectual property rights around the world may be inadequate to obtain a significant commercial advantage from the intellectual property that we develop or license.
RISKS RELATED TO OUR OPERATIONS IN ISRAEL
If there are significant shifts in the political, economic and military conditions in Israel, it could have an adverse impact on our operations.
Our corporate headquarters and research and development facilities are located in Israel. In addition, most of our employees, officers and directors are residents of Israel. Accordingly, political, economic and military conditions in Israel may directly affect our business. Since the establishment of the State of Israel in 1948, a number of armed conflicts have taken place between Israel and its neighboring countries. In recent years, these have included hostilities from Hezbollah in Lebanon and between Israel and Hamas in the Gaza Strip, which resulted in rockets being fired into Israel, causing casualties and disruption of economic activities. In addition, Israel faces threats from the civil war in Syria and from Iran. Our commercial insurance does not cover losses that may occur as a result of an event associated with the security situation in the Middle East. Although we expect that the Israeli government will cover the reinstatement value of direct damages that are caused by terrorist attacks or acts of war, we cannot assure you that such government coverage will be maintained, or if maintained, will be sufficient to compensate us fully for damages incurred. Several countries, principally in the Middle East, restrict doing business with Israel and Israeli companies, and additional countries may impose restrictions on doing business with Israel and Israeli companies, whether as a result of hostilities in the region or otherwise. Any such matters could adversely affect our operations and results of operations, and any losses or damages incurred by us as the result of such a conflict could have an adverse impact on our business.
Furthermore, our operations could be disrupted by the obligations of our personnel to perform military service. Some of our employees based in Israel may be called upon to perform military reserve duty and, in emergency circumstances, may be called to immediate and unlimited active duty. Our operations could be disrupted by the absence of a significant number of employees due to military service, which could adversely impact our business and results of operations.
Because a substantial portion of our revenues is expected to be generated in currencies other than our functional currency, we will be exposed to fluctuations in currency exchange rates, which could negatively affect our financial condition.
In the future, we expect that a substantial portion of our revenues will be generated in currencies other than our functional currency. Our functional currency, in which we currently maintain our financial records, is NIS, and our presentation currency, in which we report our financial results, is USD. The functional currency of Peace of Meat, in which it currently maintains its financial records, is Euros. As a result, our revenues for financial statement purposes might be negatively affected by fluctuations in the exchange rates of currencies in the countries in which our technologies may be licensed, and supplementary services provided and products sold.
Currency exchange controls may restrict our ability to utilize our cash flows.
We expect to receive proceeds from sales of any product we may develop, and also to pay a portion of our operational costs and expenses, in U.S. dollars, Euros and other foreign currencies. However, we may be subject to existing or future rules and regulations on currency conversion. In 1998, the Israeli currency control regulations were liberalized significantly, and there are currently no currency controls in place. Legislation remains in effect, however, pursuant to which such currency controls could be imposed in Israel by administrative action at any time. We cannot assure that such controls will not be reinstated, or if reinstated, that they would not have an adverse effect on our operations.
Enforcing a U.S. judgment against us and our executive officers and directors, or asserting U.S. securities law claims in Israel, may be difficult.
We are incorporated in Israel. Most of our executive officers and directors reside in Israel and most of our assets and the assets of these persons are located outside of the United States. Therefore, a judgment obtained against us or any of these persons in the United States, including one based on the civil liability provisions of the U.S. federal securities laws, may not be collectible in the United States and may not be enforced by an Israeli court. It may also be difficult to affect service of process on these persons in the United States or to assert U.S. securities laws claims in original actions instituted in Israel.
Even if an Israeli court agrees to hear such a claim, it may determine that Israeli, and not U.S., law is applicable to the claim. Under Israeli law, if U.S. law is found to be applicable to such a claim, the content of applicable U.S. law must be proved as a fact, which can be a time-consuming and costly process, and certain matters of procedure would be governed by Israeli law. There is little binding case law in Israel addressing these matters.
Subject to specified time limitations and legal procedures, under the rules of private international law currently prevailing in Israel, Israeli courts may enforce a final U.S. judgment in a civil matter, including judgments based upon the civil liability provisions of the U.S. securities laws and including a monetary or compensatory judgment in a non-civil matter, provided that:
| • | the judgment is enforceable in the state in which it was given; |
| • | the judgment was rendered by a court of competent jurisdiction under the rules of private international law prevailing in Israel; |
| • | the laws of the state in which the judgment was given provide for the enforcement of judgments of Israeli courts; |
| • | adequate service of process has been effected and the defendant has had a reasonable opportunity to be heard; |
| • | the judgment and the enforcement of the judgment are not contrary to the law, public policy, security or sovereignty of the State of Israel; |
| • | the judgment was not obtained by fraudulent means and does not conflict with any other valid judgment in the same matter between the same parties; and |
| • | an action between the same parties in the same matter is not pending in any Israeli court at the time the lawsuit is instituted in the foreign court. |
There is little binding case law in Israel that addresses the matters described above. As a result of the difficulty associated with enforcing a judgment against us in Israel, you may not be able to collect any damages awarded by either a U.S. or foreign court.
If a foreign judgment is enforced by an Israeli court, it generally will be payable in Israeli currency, which can be converted into non-Israeli currency and transferred out of Israel. The usual practice in an action before an Israeli court to recover an amount in a non-Israeli currency is for the Israeli court to issue a judgment for the equivalent amount in Israeli currency at the rate of exchange in force on the date of the judgment, but the judgment debtor may make payment in foreign currency. Pending collection, the amount of the judgment of an Israeli court stated in Israeli currency ordinarily will be linked to the Israeli CPI plus interest at the annual statutory rate set by Israeli regulations prevailing at the time. Judgment creditors must bear the risk of unfavorable exchange rate fluctuations.
Your rights and responsibilities as our shareholder will be governed by Israeli law, which may differ in some respects from the rights and responsibilities of shareholders of U.S. corporations.
Since we are incorporated under Israeli law, the rights and responsibilities of our shareholders are governed by our articles of association and Israeli law. These rights and responsibilities differ in some respects from the rights and responsibilities of shareholders of U.S. corporations. In particular, each of our shareholders has a duty to act in good faith and in a customary manner in exercising its rights and performing its obligations toward us and other shareholders and to refrain from abusing its power in, among other things, voting at shareholder meetings on certain matters, such as an amendment to our articles of association, an increase of our authorized share capital, a merger and approval of related party transactions that require shareholder approval. A shareholder also has a general duty to refrain from discriminating against other shareholders. In addition, a controlling shareholder or a shareholder who knows that it possesses the power to determine the outcome of a shareholders vote or to appoint or prevent the appointment of an office holder has a duty to act in fairness toward us. Israeli law does not clearly define the substance of these duties, but these provisions may be interpreted to impose additional obligations and liabilities on our shareholders that are not typically imposed on shareholders of U.S. corporations.
Provisions of Israeli corporate and tax law may deter acquisition transactions.
Israeli corporate law regulates mergers, requires tender offers for acquisitions of shares above specified thresholds, requires special approvals for transactions involving directors, officers or significant shareholders and regulates other matters that may be relevant to such types of transactions. For example, a merger may not be consummated unless at least 50 days have passed from the date on which a merger proposal is filed by each merging company with the Israel Registrar of Companies and at least 30 days have passed from the date on which the shareholders of both merging companies have approved the merger. In addition, a majority of each class of securities of the target company must approve a merger. Moreover, a tender offer for all of a company's issued and outstanding shares can only be completed if the acquirer receives positive responses from the holders of at least 95% of the issued share capital. Completion of the tender offer also requires approval of a majority of the offerees that do not have a personal interest in the tender offer, unless, following consummation of the tender offer, the acquirer would hold at least 98% of the company's outstanding shares. Furthermore, the shareholders, including those who indicated their acceptance of the tender offer, may, at any time within six months following the completion of the tender offer, claim that the consideration for the acquisition of the shares does not reflect their fair market value, and petition an Israeli court to alter the consideration for the acquisition accordingly, unless the acquirer stipulated in its tender offer that a shareholder that accepts the offer may not seek such appraisal rights, and the acquirer or the company published all required information with respect to the tender offer prior to the tender offer's response date.
Furthermore, Israeli tax considerations may make potential transactions unappealing to us or to our shareholders whose country of residence does not have a tax treaty with Israel exempting such shareholders from Israeli tax. For example, Israeli tax law does not recognize tax-free share exchanges to the same extent as U.S. tax law. With respect to mergers, Israeli tax law allows for tax deferral in certain circumstances but makes the deferral contingent on the fulfillment of a number of conditions, including, in some cases, a holding period of two years from the date of the transaction during which sales and dispositions of shares of the participating companies are subject to certain restrictions. Moreover, with respect to certain share swap transactions, the tax deferral is limited in time, and when such time expires, the tax becomes payable even if no disposition of the shares has occurred. These provisions could delay, prevent or impede an acquisition of us or our merger with another company, even if such an acquisition or merger would be beneficial to us or to our shareholders.
RISKS RELATED TO OWNERSHIP OF THE ADSs
The ADS price may be volatile, and you may lose all or part of your investment.
The market price of the ADSs could be highly volatile and may fluctuate substantially, including downward, as a result of many factors, including:
| • | changes in the prices of our raw materials or the products manufactured in factories using our technologies; |
| • | the trading volume of the ADSs; |
| • | the effects of the COVID-19 pandemic; |
| • | general economic, market and political conditions, including negative effects on consumer confidence and spending levels that could indirectly affect our results of operations; |
| • | actual or anticipated fluctuations in our financial condition and operating results, including fluctuations in our quarterly and annual results; |
| • | announcements by us or our competitors of innovations, other significant business developments, changes in distributor relationships, acquisitions or expansion plans; |
| • | announcement by competitors or new market entrants of their entry into or exit from the alternative protein market; |
| • | overall conditions in our industry and the markets in which we intend to operate; |
| • | market conditions or trends in the packaged food sales industry that could indirectly affect our results of operations; |
| • | addition or loss of significant customers or other developments with respect to significant customers; |
| • | adverse developments concerning our manufacturers and suppliers; |
| • | changes in laws or regulations applicable to our products or business; |
| • | our ability to effectively manage our growth and market expectations with respect to our growth, including relative to our competitors; |
| • | changes in the estimation of the future size and growth rate of our markets; |
| • | announcements by us or our competitors of significant acquisitions, strategic partnerships, joint ventures or capital commitments; |
| • | additions or departures of key personnel; |
| • | competition from existing products or new products that may emerge; |
| • | issuance of new or updated research or reports about us or our industry, or positive or negative recommendations or withdrawal of research coverage by securities analysts; |
| • | variance in our financial performance from the expectations of market analysts; |
| • | our failure to meet or exceed the estimates and projections of the investment community or that we may otherwise provide to the public; |
| • | fluctuations in the valuation of companies perceived by investors to be comparable to us; |
| • | disputes or other developments related to proprietary rights, including patents, and our ability to obtain intellectual property protection for our products; |
| • | litigation or regulatory matters; |
| • | announcement or expectation of additional financing efforts; |
| • | sales and short-selling of the ADSs; |
| • | our issuance of equity or debt; |
| • | changes in accounting practices; |
| • | ineffectiveness of our internal controls; |
| • | negative media or marketing campaigns undertaken by our competitors or lobbyists supporting the conventional meat industry; |
| • | the public’s response to publicity relating to the health aspects or nutritional value of products to be manufactured in factories using our technologies; and |
| • | other events or factors, many of which are beyond our control. |
In addition, the stock markets have experienced extreme price and volume fluctuations. Broad market and industry factors may materially harm the market price of the ADSs, regardless of our operating performance. These fluctuations often have been unrelated or disproportionate to the operating performance of those companies. These fluctuations, as well as general economic, political and market conditions such as recessions, interest rate changes, tariffs, international currency fluctuations, or the effects of disease outbreaks or pandemics (such as the COVID-19 pandemic), may negatively impact the market price of the ADSs. In the past, following periods of volatility in the market price of a company’s securities, securities class action litigation has often been instituted against that company. If we were involved in any similar litigation, we could incur substantial costs and our management’s attention and resources could be diverted.
We have never paid dividends on our share capital and we do not intend to pay dividends for the foreseeable future.
We have never declared or paid any dividends on our share capital and do not intend to pay any dividends in the foreseeable future. We anticipate that we will retain all of our future earnings for use in the development and growth of our business and for general corporate purposes. Accordingly, any gains from an investment in the ADSs will depend on price appreciation of the ADSs, which may never occur. In addition, Israeli law limits our ability to declare and pay dividends, and may subject our dividends to certain Israeli withholding taxes.
ADS holders may not receive the same distributions or dividends as those we make to the holders of our ordinary shares, and, in some limited circumstances, they may not receive dividends or other distributions on our ordinary shares and may not receive any value for them, if it is illegal or impractical to make them available.
The depositary for the ADSs has agreed to pay to ADS holders the cash dividends or other distributions it or the custodian receives on ordinary shares or other deposited securities underlying the ADSs, after deducting its fees and expenses. ADS holders will receive these distributions in proportion to the number of ordinary shares their ADSs represent. However, the depositary is not responsible if it decides that it is unlawful or impractical to make a distribution available to any holders of ADSs. For example, it would be unlawful to make a distribution to a holder of ADSs if it consists of securities that require registration under the Securities Act of 1933, as amended, or the Securities Act, but that are not properly registered or distributed under an applicable exemption from registration. In addition, conversion into U.S. dollars from foreign currency that was part of a dividend made in respect of deposited ordinary shares may require the approval or license of, or a filing with, any government or agency thereof, which may be unobtainable. In these cases, the depositary may determine not to distribute such property and hold it as “deposited securities” or may seek to affect a substitute dividend or distribution, including net cash proceeds from the sale of the dividends that the depositary deems an equitable and practicable substitute. We have no obligation to register under U.S. securities laws any ADSs, ordinary shares, rights or other securities received through such distributions. We also have no obligation to take any other action to permit the distribution of ADSs, ordinary shares, rights or anything else to holders of ADSs. In addition, the depositary may deduct from such dividends or distributions its fees and may withhold an amount on account of taxes or other governmental charges to the extent the depositary believes it is required to make such withholding. These restrictions may cause a material decline in the value of the ADSs.
ADS holders do not have the same rights as our shareholders.
ADS holders do not have the same rights as our shareholders. For example, ADS holders may not attend shareholders’ meetings or directly exercise the voting rights attaching to the ordinary shares underlying their ADSs. ADS holders may vote only by instructing the depositary to vote on their behalf. If we request the depositary to solicit voting instructions from ADS holders (which we are not required to do), the depositary will notify ADS holders of a shareholders’ meeting and send or make voting materials available to them. Those materials will describe the matters to be voted on and explain how ADS holders may instruct the depositary how to vote. For instructions to be valid, they must reach the depositary by a date set by the depositary. The depositary will try, as far as practical, subject to the laws of Israel and the provisions of our articles of association or similar documents, to vote or to have its agents vote the deposited ordinary shares as instructed by ADS holders. If we do not request the depositary to solicit voting instructions from ADS holders, they can still send voting instructions, and, in that case, the depositary may try to vote as they instruct, but it is not required to do so. Except by instructing the depositary as described above, ADS holders won’t be able to exercise voting rights unless they surrender their ADSs and withdraw the ordinary shares. However, they may not know about the meeting enough in advance to withdraw the ordinary shares. We cannot assure ADS holders that they will receive the voting materials in time to ensure that they can instruct the depositary to vote their ordinary shares. In addition, the depositary and its agents are not responsible for failing to carry out voting instructions or for the manner of carrying out voting instructions. This means that ADS holders may not be able to exercise voting rights and there may be nothing they can do if their ordinary shares are not voted as they requested. In addition, ADS holders have no right to call a shareholders’ meeting.
ADS holders may be subject to limitations on transfer of their ADSs.
ADSs will be transferable on the books of the depositary. However, the depositary may close its transfer books at any time or from time to time when it deems expedient in connection with the performance of its duties. In addition, the depositary may refuse to deliver, transfer or register transfers of ADSs generally when our books or the books of the depositary are closed, or at any time if we or the depositary deem it advisable to do so because of any requirement of law or of any government or governmental body, under any provision of the deposit agreement, or for any other reason in accordance with the terms of the deposit agreement.
As a foreign private issuer whose ADSs are listed on Nasdaq, we follow certain home country corporate governance practices instead of certain Nasdaq requirements, we are not subject to U.S. proxy rules and are exempt from certain Exchange Act reporting requirements. If we were to lose our foreign private issuer status, our costs to modify our practices and maintain compliance under U.S. securities laws and Nasdaq rules would be significantly higher.
We are a foreign private issuer and are not subject to the same requirements that are imposed upon U.S. domestic issuers by the SEC. We are permitted to follow certain home country corporate governance practices instead of certain requirements of the rules of Nasdaq. As permitted under the Israeli Companies Law 1999, or Companies Law, pursuant to our articles of association, the quorum for an ordinary meeting of shareholders shall be the presence of at least two shareholders present in person, by proxy or by a voting instrument, who hold at least 25% of the voting power of our shares (and in an adjourned meeting, with some exceptions, a minimum of one shareholder) instead of 33 1⁄3% of our issued share capital as otherwise required under the Nasdaq corporate governance rules. We may also adopt and approve material changes to equity incentive plans in accordance with the Companies Law, which does not impose a requirement of shareholder approval for such actions. In addition, we follow Israeli corporate governance practice instead of the Nasdaq requirements to obtain shareholder approval for certain dilutive events (such as issuances that will result in a change of control, certain transactions other than a public offering involving issuances of a 20% or greater interest in us and certain acquisitions of the stock or assets of another company). Additionally, we follow Israeli corporate governance practices instead of Nasdaq requirements with regard to, among other things, the composition of our board of directors. For example, our board of directors currently comprises four directors, three of whom we have determined are independent, however during 2021, the majority of our board of directors was not deemed to be independent, in compliance with our home-country requirements. Accordingly, our shareholders may be afforded less protection that what is provided under the Nasdaq corporate governance rules to investors in U.S. domestic issuers. See “Item 16G. —Corporate Governance—Nasdaq Listing Rules and Home Country Practices.”
Additionally, we are exempt from the rules and regulations under the Exchange Act related to the furnishing and content of proxy statements, and our officers, directors, and principal shareholders are exempt from the reporting and short-swing profit recovery provisions contained in Section 16 of the Exchange Act. Furthermore, although under regulations promulgated under the Companies Law, as an Israeli public company listed on Nasdaq, we are required to disclose the compensation of our five most highly compensated officers on an individual basis, this disclosure may not be as extensive as that required of U.S. domestic reporting companies. In addition, we are not required under the Exchange Act to file current reports and quarterly reports, including financial statements, with the SEC as frequently or as promptly as U.S. domestic reporting companies whose securities are registered under the Exchange Act. Moreover, we are not required to comply with Regulation FD, which restricts the selective disclosure of material information. These exemptions and leniencies reduce the frequency and scope of information and protections available to ADS holders in comparison to those applicable to U.S. domestic reporting companies.
If we cease to qualify as a foreign private issuer, we would be required to comply fully with the reporting requirements of the Exchange Act applicable to U.S. domestic issuers. We would lose our foreign private issuer status if a majority of our shares are owned by U.S. residents and a majority of our directors or executive officers are U.S. citizens or residents or we fail to meet additional requirements necessary to avoid loss of foreign private issuer status. If we are not a foreign private issuer, we will be required to file periodic reports and registration statements on U.S. domestic issuer forms with the SEC, which are more detailed and extensive than the forms available to a foreign private issuer. We may also be required to modify certain of our policies to comply with accepted governance practices associated with U.S. domestic issuers and we would lose our ability to rely upon exemptions from certain corporate governance requirements on U.S. stock exchanges that are available to foreign private issuers. Such modifications and subsequent compliance would cause us to incur significant legal, accounting and other expenses that we would not incur as a foreign private issuer.
If we are a “passive foreign investment company” for U.S. federal income tax purposes, there may be adverse U.S. federal income tax consequences to U.S. investors
Based on our income and assets, we believe that we should be treated as a PFIC for the preceding taxable year. However, the determination of our PFIC status is made annually based on the factual tests described below. Consequently, while we may be a PFIC in future years, we cannot estimate with certainty at this stage whether or not we are likely to be treated as a PFIC in the current taxable year or any future taxable years. Generally, if, for any taxable year, at least 75 percent of our gross income is “passive income” or at least 50 percent of our gross assets during the taxable year (based on the average of the fair market values of the assets determined at the end of each quarterly period) are assets that produce or are held for the production of passive income, we will be characterized as a PFIC for U.S. federal income tax purposes. Passive income for this purpose generally includes, among other things, dividends, interest, rents, royalties, gains from commodities and securities transactions, and gains from assets that produce passive income. However, rents and royalties received from unrelated parties in connection with the active conduct of a trade or business should not be considered passive income for purposes of the PFIC test. For example, if we were to be characterized as a PFIC for U.S. federal income tax purposes in any taxable year during which a U.S. Holder (as defined in “Item 10.—Additional Information—Taxation — Material United States federal income tax considerations”) holds ordinary shares or ADSs, such U.S. Holder could be subject to additional taxes and interest charges upon certain distributions by us and any gain recognized on a sale, exchange or other disposition of our shares, whether or not we continue to be characterized as a PFIC. Certain adverse consequences of PFIC status can be mitigated if a U.S. Holder makes a “mark to market” election or an election to treat us as a qualified electing fund, or QEF. Upon request, we expect to provide the information necessary for U.S. Holders to make “qualified electing fund elections” if we are classified as a PFIC. See “Item 10.—Additional Information—Taxation—Passive foreign investment company considerations.”
Whether we are a PFIC for any taxable year will depend on the composition of our income and the composition and value of our assets from time to time. Each U.S. Holder is strongly urged to consult its tax advisor regarding these issues and any available elections to mitigate such tax consequences.
If we are a controlled foreign corporation, there could be adverse U.S. federal income tax consequences to certain U.S. Holders.
Each “Ten Percent Shareholder” (as defined below) in a non-U.S. corporation that is classified as a “controlled foreign corporation,” or a CFC, for U.S. federal income tax purposes generally is required to include in income for U.S. federal tax purposes such Ten Percent Shareholder’s pro rata share of the CFC’s “Subpart F income,” “tested income” and investment of earnings in U.S. property, even if the CFC has made no distributions to its shareholders. Subpart F income generally includes dividends, interest, rents, royalties, gains from the sale of securities and income from certain transactions with related parties. In addition, a Ten Percent Shareholder that realizes gain from the sale or exchange of shares in a CFC may be required to classify a portion of such gain as dividend income rather than capital gain. A non-U.S. corporation generally will be classified as a CFC for U.S. federal income tax purposes if Ten Percent Shareholders own, directly or indirectly, more than 50% of either the total combined voting power of all classes of stock of such corporation entitled to vote or of the total value of the stock of such corporation. A “Ten Percent Shareholder” is a United States person (as defined by the Internal Revenue Code of 1986, as amended, or the Code) who owns or is considered to own 10% or more of the value or total combined voting power of all classes of stock entitled to vote of such corporation.
The determination of CFC status is complex and includes complex attribution rules. A non-corporate Ten Percent Shareholder with respect to a CFC generally will not be allowed certain tax deductions or foreign tax credits generally available to a corporate Ten Percent Shareholder. Failure to comply with CFC reporting obligations may subject a Ten Percent Shareholder to significant monetary penalties. We cannot provide any assurances that we will furnish to any Ten Percent Shareholder information that may be necessary to comply with the reporting and tax paying obligations applicable under the CFC rules of the Code. U.S. Holders should consult their own tax advisors with respect to the potential adverse U.S. tax consequences of becoming a Ten Percent Shareholder in a CFC.
| | ITEM 4. | INFORMATION ON THE COMPANY |
A. History and Development of the Company
We were incorporated in May 2018 in Israel as DocoMed Ltd., and originally provided digital health services. In July 2019, we changed our name to MeaTech Ltd., or MeaTech, and commenced our cultured meat technology development operations. In January 2020, MeaTech completed a merger with Ophectra Real Estate and Investment Ltd., or Ophectra, a company incorporated in Israel whose shares were traded on the TASE, whereupon the name of Ophectra was changed to Meat-Tech 3D Ltd., and later further changed to MeaTech 3D Ltd., or MeaTech 3D.
According to the terms of the merger, MeaTech 3D acquired all outstanding shares of MeaTech from MeaTech’s shareholders, in return for the issuance of ordinary shares to the shareholders of MeaTech, as well as non-tradable merger warrants to receive ordinary shares upon the achievement of pre-defined milestones, which were achieved in 2020 and 2021. MeaTech become MeaTech 3D’s wholly-owned subsidiary, and later changed its name to Chicken Meat-Tech Ltd. and then to MeaTech MT Ltd.
In connection with the merger, the Tel Aviv District Court for Economic Affairs approved an arrangement whereby all of Ophectra’s assets and liabilities, whether certain or contingent, at the time of the merger were irrevocably assigned to a settlement fund, or Settlement Fund, for the purpose of settling Ophectra’s pre-merger liabilities (except for Ophectra’s ownership of 14.74% of the outstanding shares of Therapin Ltd., as described below). This includes all future liabilities arising from Ophectra’s activities prior to the merger (including tax liabilities, if any), and any commitments made by Ophectra prior to the merger. We also provided the Settlement Fund approximately NIS 1.3 million (approximately $0.4 million), which we included in our public listing expenses, for the purpose of settling any of Ophectra’s debts, and bear no additional liabilities to the Settlement Fund. Anyone who believed they had a claim to Ophectra’s assets were invited to lodge their claims to the trustees. Due to the fact that two years have passed since the merger, and the fact that the Settlement Fund no longer contains any assets, its trustees are expected to instigate proceedings to wind up the Settlement Fund.
Upon completion of the merger, all directors and officers of MeaTech became directors and officers of MeaTech 3D, in addition to some of the independent directors of Ophectra.
Although MeaTech 3D was the legal acquirer of MeaTech’s shares as described above, because (i) the shareholders of MeaTech received the majority of the voting rights in MeaTech 3D and the ability to determine its financial and operational policy, (ii) the management of MeaTech continues to serve as the management of MeaTech 3D and (iii) at the time of completion of the merger, MeaTech 3D was a company without significant business operations, the merger is not considered a business acquisition as defined in IFRS 3. As a result, it was determined that MeaTech is the acquirer of the business for accounting purposes and the transaction was treated as a reverse acquisition that does not constitute a business combination.
Therefore, the consolidated financial statements and financial data included herein for all periods through and including December 31, 2019 were adjusted retroactively to reflect the financial statements of MeaTech (now called MeaTech MT Ltd.), other than the information concerning earnings per share, which is presented according to the equity information of MeaTech 3D (then called Ophectra Real Estate and Investments Ltd.), and our consolidated financial statements and financial data included herein from January 1, 2020 onward relate to MeaTech 3D.
We temporarily maintained ownership of 14.74% of the outstanding shares of Therapin Ltd., or Therapin, a company incorporated in Israel, while considering a possible collaboration, however, in May 2020, we returned these holdings to Therapin, and agreed to convert our investment of NIS 7.25 million in Therapin into an interest-free loan, to be repaid by the latter at a rate of NIS 0.48 million per annum for ten years (NIS 4.8 million in total) plus NIS 2.45 million to be paid upon an exit event, including a public offering, or repayment of 14.74% of any distributable surplus or dividend distributed by Therapin, up to the amount of the outstanding balance, as detailed in our separation agreement with Therapin. As part of the agreement, Therapin gave us an option to convert the cash payment to equity of Therapin.
Peace of Meat Acquisition
On February 10, 2021, we consummated an agreement with all of the shareholders of Peace of Meat, a private limited liability company incorporated, organized and existing under the laws of Belgium, or Peace of Meat, to acquire all of the outstanding share capital of Peace of Meat not yet owned by us, through our wholly-owned subsidiary, MeaTech Europe BV, for total consideration of up to €16.3 million ($19.9 million). The total consideration payable by us in the acquisition consists of €7.7 million ($8.6 million), comprised of €4.1 million ($5.1 million) in cash and 4,070,766 of our ordinary shares, with a fair value of €3.6 million ($4.4 million), paid on the closing date, and up to an additional €7.6 million ($8.3 million) payable in a combination of €4.0 million ($4.4 million) in cash and 4,070,766 of our ordinary shares in the amount of €3.6 million ($3.9 million), upon the achievement of four defined milestones related to Peace of Meat’s biomass and bioreactor size, density, capacity and production. The acquisition agreement specified that each milestone must be reached within a six-month period, over a total of two years, which can be extended by up to nine additional months under circumstances set forth in the acquisition agreement. The agreement also includes acceleration events, such as breach of the acquisition agreement by us; certain merger, consolidation or acquisition transactions involving us; our delisting; and the termination of employment of two or more of the founders of Peace of Meat during the milestone period under circumstances set forth in the acquisition agreement. As of the date hereof, Peace of Meat had fully achieved the first two such milestones. We do not provision liabilities for future milestones before their achievement.
Peace of Meat was established in Belgium in 2019 and is developing cultured avian fat directly from animal cells without the need to grow or kill animals. In 2020, Peace of Meat was awarded a subsidy of approximately $1.33 million from the Flemish government, of which $0.5 million has been received, and has received approximately $1 million in private investments. We bought Peace of Meat for approximately $20 million in a cash- and equity-based milestone deal. We believe that the innovative technology of Peace of Meat has the potential to support an industrial process for the production of cultured avian fat. Peace of Meat has entered into a number of scientific and commercial collaborations, is in the process of positioning itself as a future B2B provider with the potential to cover the entire value chain and to accelerate research and production processes in the industry, and has conducted taste tests for hybrid products that it has developed. It intends to open a pilot plant in Belgium in 2023.
B. Business Overview
We are an international deep-tech food company that initiated activities in 2019 and is listed on the Nasdaq Capital Market under the ticker “MITC”. We maintain facilities in Rehovot, Israel and Antwerp, Belgium and are in the process of expanding activities to California, USA. We believe that cultivated meat technologies hold significant potential to improve meat production, simplify the meat supply chain, and offer consumers a range of new product offerings.
We aim to provide an alternative to industrialized animal farming that reduces carbon footprint, minimizes water and land usage, and prevents the slaughtering of animals. By adopting a modular factory design, we expect to be able to offer a sustainable solution for producing a variety of beef, chicken and pork products, both as raw materials and whole cuts.
We are developing cultivated meat technologies, including three-dimensional printing technology, together with biotechnology processes and customizable manufacturing processes in order to manufacture cultivated meat that does not require animal slaughter. We are developing a novel, proprietary three-dimensional bioprinter to deposit layers of differentiated stem cells, scaffolding, and cell nutrients in a three-dimensional form of structured cultured meat. We believe the cultured meat production processes we are developing, which are designed to offer our eventual customers an alternative to industrial slaughter, have the potential to improve the quality of the environment, shorten global food supply chains, and reduce the likelihood of health hazards such as zoonotic diseases transferred from animals to humans (including viruses, such as virulent avian influenza and COVID-19, and drug-resistant bacterial pathogens, such as some strains of salmonella).
In December 2021, we announced that we had successfully three-dimensionally printed a 3.67 oz cultivated steak, primarily composed of cultivated real fat and muscle tissues. While cultivated meat companies have made some progress developing unstructured alternative meat products, such as minced meat and sausage, to the best of our knowledge, the industry has struggled in developing high-margin, high-value structured and cultured meat products such as steak. Unlike minced meat, a cultured meat steak product has to grow in fibers and contain connective tissues and fat. To be adopted by diners, we believe cultured steaks will need to be meticulously engineered to look and smell like conventional meat, both before and after cooking, and to taste and feel like meat to the diner. We believe that we are the first company to be developing both a proprietary bioprinter and the related processes for growing cultivated meat to focus on what we believe is a high value sector of the alternative protein market.
We are led by our Chief Executive Officer, Arik Kaufman, who has founded various Nasdaq- and TASE-traded foodtech companies, and currently serves as director of Wilk Technologies Ltd. He is also a founding partner of the BlueSoundWaves collective, led by Ashton Kutcher Guy Oseary and Effie Epstein, which recently partnered with MeaTech to assist in attempting to accelerate the Company’s growth. Mr. Kaufman holds extensive personal experience in the fields of food-tech and bio-tech law, and has led and managed numerous complex commercial negotiations, as part of local and international fundraising, M&A transactions and licensing agreements. We have carefully selected personnel for the rest of our executive management team who possess substantial industry experience and share our core values, from fields as diverse as tissue engineering, industrial stem cell growth, and printer and print materials development.
Cultivated Meat Industry and Market Opportunity
Protein is a necessary staple for healthy nutrition. The growth in recent years of both the human population and global wealth is driving a decades-long trend of accelerating demand for meat. The demand for protein products has consistently risen in recent decades and is expected to continue to do so. The rising growth of demand for farm animals for the food industry has created significant environmental, health, financial and ethical challenges.
According to Statista the value of the global meat sector was estimated at $838 billion in 2020, and was forecast to increase to $1,157 billion by 2025. According to market research firm Fortune Business Insights, the global meat substitute market was estimated at $5.4 billion in 2021 and is expected to grow to $10.8 billion by 2028. According to Facts and Factors Market Research, the cultivated meat category alone is expected to reach $248 million by 2026, with an annual growth rate of approximately 16%. With regard to the longer term, Barclays predicted in November 2021 that by 2040, 20% of the demand for meat globally will be provided by cultivated meat – a $450 billion market opportunity.
The meat industry is showing strong interest in the alternative protein space, both in plant-based and cell-based proteins. There are several drivers underlying the strong engagement with alternative proteins. We believe consumers are looking for less harmful protein sources, with approaches such as flexitarianism already an established middle path between vegetarian diets and those heavy in animal proteins, such as the paleo diet. Many meat processors have experienced the worst of the COVID-19 pandemic outbreaks and are seeking to minimize human involvement in the manufacturing process. To that end, retailers such as Costco and Walmart are increasingly opening their own meat processing facilities on which they can rely exclusively.
Limitations of Conventional Meat Production
In addition to questions about whether conventional meat production can adequately provide for the growing global population, conventional meat production raises serious environmental issues. According to the United Nations, 8% of the world's freshwater is used for raising livestock for meat and leather. At least 18% of the greenhouse gases entering the atmosphere are from the livestock industry. 26% of the planet's ice-free land is used for livestock grazing and 33% of croplands are used for animal feed. With regard to treatment of animals in conventional meat production, approximately More than 70 billion animals are slaughtered annually with steady increases to be expected in line with increased demand for meat.
Another common consumer concern with industrial-scale animal rearing is the reliance on intensive use of antibiotics. Antibiotics are used in livestock, especially pigs and poultry, to manage animal health, and to treat or prophylactically prevent diseases such as avian flu and swine flu. Their effects on human health have not been fully resolved, with concerns including the potential growth of antibiotic-resistant diseases in meat for human consumption.
Existing Alternative Proteins and their Limitations
Negative consumer sentiment towards the perceived ethical, health and environmental effects of the global meat industry help explain the strong focus that has developed on creating methods of protein production that are more sustainable, nutritious and conscious of animal welfare. Recent years have seen a combination of increasing consumer awareness and advanced technological development that has led to substantially increased demand for proteins that do not involve animal slaughter, beyond traditional plant-based proteins, such as soy, peas and chickpeas. Some of the alternative proteins being developed for human consumption for this purpose include:
Mycoproteins: Some of the most commercially successful novel alternative protein products are currently mycoproteins, derived from fungi. They are high in protein, high in fiber, low in saturated fat, and contain no cholesterol. However, they have been associated with allergic and gastrointestinal reactions. They are fermented to become a dough, which can develop a texture similar to that of meat.
Jackfruit: Jackfruit is a tropical fruit, native to India, which is high in protein. Its texture is somewhat similar to shredded meat, although its taste is similar to other fruits, such as apples and mangoes, so while it is a good source of protein, it is not generally viewed as an alternative to meat for consumers used to animal proteins.
Insects: Insects are an environmentally-friendly source of protein, requiring significantly less land and water, and emitting significantly less greenhouse gases than large mammals raised for slaughter. In addition, they can be fed food unsuitable for livestock that would otherwise be wasted. While crickets are the most common source of edible insects, research is taking place on new insect species of value for food production, as well as methods to produce them economically at scale. Insects can be consumed in their natural state; however many cultures consider insect consumption to be taboo and many people are disgusted by the idea. As a result, research is taking place into developing insect-based products in different forms not easily discernable as insect-based, including flour.
RECENT DEVELOPMENTS
Acquisition of Peace of Meat
In February 2021, we finalized our acquisition of Peace of Meat, a Belgian producer of cultured avian products, for up to $19.9 million in cash and equity, depending on milestone achievements. We intend to leverage Peace of Meat’s cultured avian technologies to diversify our own bovine-oriented technologies and expedite our entry into the market for plant-based meat alternatives and cultured products.
$28 million Nasdaq listing and voluntary delisting from the Tel Aviv Stock Exchange
In March 2021, we raised $28 million in an initial public offering of American Depository Shares (ADSs) on the Nasdaq Capital Market, making us the first cultured meat company to be publicly traded in the US. In August 2021, we completed the process of voluntarily delisting our ordinary shares from the Tel Aviv Stock Exchange (TASE), with our ADSs continuing to trade on the Nasdaq Capital Market. The decision was made in order to internationalize our investor and public relations efforts into the United States and globally.
Initiation of food technology development activities in Europe
In April 2021, we commenced food technology development activities through our European subsidiary, MeaTech Europe, with an initial focus on hybrid foods using Peace of Meat’s cultured fat. Hybrid foods are products composed of both plant and cultured meat ingredients that have the potential to offer a meatier experience than purely plant-based meat alternatives.
Letter of intent with Tiv Ta'am Holdings to develop and distribute cultured meat products
In July 2021, as part of our strategy, we signed a non-binding letter of intent with Tiv Ta'am, a leading food retailer and meat producer in Israel, to examine options to commercialize our products. We are unable to determine whether a binding cooperation agreement will result from negotiations, however such an agreement would be expected to include cooperation on research and the establishment of a production facility for cultivated meat products, as well as a grant of distribution and marketing rights to Tiv Ta’am (including possible exclusive rights on jointly developed products), in Israel and/or elsewhere in the world. We have also agreed to discuss expanding cooperation regarding the production and marketing of cultivated meat products in the future, with an emphasis on cultivated pork. We intend to publicize information regarding such an agreement, should it eventuate.
Manufacturing of 700g (25 oz) of cultivated fat biomass
In July 2021, Peace of Meat cultivated just over 700 grams of pure chicken fat biomass in a single production run. We believe that producing this quantity of pure cultured material in one run is a breakthrough toward potentially manufacturing cultivated chicken fat at an industrial scale.
Partnering with Ashton Kutcher and Guy Oseary of BlueSoundWaves
In October 2021, the BlueSoundWaves collective, led by Ashton Kutcher, Guy Oseary and Effie Epstein, partnered with us with the goal of accelerating our growth and development toward commercializing our proprietary cultured meat production technologies. BlueSoundWaves works closely with our management to advance our strategy, go-to-market activities and brand by leveraging the collective’s marketing and strategic expertise and network.
Cultivation of World’s Largest Bioprinted Steak
In December 2021, we announced that we had successfully three-dimensionally printed a 3.67 oz cultivated steak, primarily composed of cultivated real fat and muscle tissues, without using soy or pea protein. The cells used to make the steak were produced with an advanced proprietary process that starts by isolating bovine stem cells from tissue samples and multiplying them. Upon reaching sufficient cellular mass, stem cells were formulated into bio inks compatible with our proprietary 3D bioprinter. The bio-inks were printed from a digital design file of a steak structure. The printed product was placed in an incubator to mature, allowing the stem cells to differentiate into fat and muscle cells and develop into fat and muscle tissue to form our steak.
Promising results with muscle stem cell differentiation
In February 2022, we announced the successful development of a novel technology process in which muscle cells are fused into significant muscle fibers that better resemble those in whole cuts of meat. Bovine stem cells were isolated, proliferated in the lab, and differentiated into matured muscle cells with improved muscle fiber density, thickness and length. Based on these improvements, we have filed a provisional patent application with the USPTO.
Expansion of cultivated meat operation into the US to accelerate go-to-market strategy
In March 2022, we announced that we will be opening a US office. The new space will include activities in research and development, investor relations, and business development.
THE CULTIVATED MEAT SOLUTION
We believe cultivated meat grown through cellular agriculture, which aims to produce cultivated animal proteins without the need for large-scale slaughter, has the potential to satisfy consumer desire for meat while avoiding the negative impacts of conventional meat production. Cellular agriculture is an efficient, closely-controlled indoor agricultural process, utilizing advanced technologies with conceptual similarities to hydroponics, but used for growing meat cells, rather than fruit. Cultivated meat is grown in cell culture, rather than inside animals, applying tissue engineering practices for fat and muscle production for the purpose of human consumption. In place of animal slaughter, stem cells are removed from an animal, such as from an umbilical cord following birth, and then cultivated in vitro to form muscle fibers and fat cells. Also known as cultured meat, clean meat, in vitro meat, lab-grown meat, green meat, cell-based meat, and motherless meat, the term “cultivated meat” is the term believed to best appeal to consumers.
Cultivated meat production is an advanced technology operating as part of the wider field of cellular agriculture (growing animal cells in bioreactors), which is an emerging solution to the growing demand for alternative proteins. We are aware of a few dozen companies and institutions actively working to develop technologies and other products to meet this demand, some of whom are focused on producing red meats, while others are focused on fish and crustaceans. Some of these companies are working on culturing various types of cells, such as chicken, pork, kangaroo and foie gras. We believe this push on scaling-up cellular agriculture has the potential to offer a solution to the scale and environmental challenges confronting conventional meat production. Other alternative protein competitors are already selling plant-based meat substitutes, but to our knowledge, these companies are not focused on the production of real meat products produced with animal cells with no pea or soy ingredients.
We are engaged with experimentation to develop optimal and cost-effective cell culture media. In so doing, we are also exploring a range of types of and sources for growth factors suited to cell culture. These sources are expected to be sustainable and ethical, providing a route to enabling efficient and cost-effective processes. While many challenges remain, surveys are consistently showing consumer openness toward, and enthusiasm for, cultivated meat. According to ‘Consumer Acceptance of Cultured Meat: An Updated Review (2018–2020)’, published by researchers at the University of Bath, “the evidence suggests that, while most people see more societal benefits than personal benefits of eating cultured meat, there is a large potential market for cultured meat products in many countries around the world. Cultured meat is generally seen as more acceptable than other food technologies like GMOs, and more appealing than other alternative proteins like insects. Although it is not as broadly appealing as plant-based proteins, evidence suggests it may be more uniquely positioned to appeal to meat-lovers who are resistant to other alternative proteins, and it is more appealing to certain demographic groups".
We believe that cultivated meat could have several potential advantages over conventionally-harvested meat:
| • | Environmental: At least 18% of the greenhouse gases entering the atmosphere today are from the livestock industry. Research shows that the expected environmental footprint of cultivated meat includes approximately 78% to 96% fewer greenhouse gas emissions, 99% less land use, 82% to 96% less water use, and 7% to 45% less energy use than conventionally-produced beef, lamb, pork and poultry. This suggests that the environmental consequences of switching from large-scale, factory farming to lab-grown cultivated meat could have a long-term positive impact on the environment. |
| • | Cost: While the precise economic value of harvested cells has yet to be determined, the potential to harvest large numbers of cells from a small number of live donor animals gives rise to the possibility of considerably higher returns than traditional agriculture, with production cycles potentially measured in months, rather than years. By comparison, raising a cow for slaughter generally takes an average of 18 months, over which period 15,400 liters of water and 7 kilograms of feed will be consumed for every kilogram of beef produced. |
| • | Animal Suffering: More and more people are grappling with the ethical question of whether humanity should continue to slaughter animals for food. There is a growing trend of opposition to the way animals are raised for slaughter, often in small, confined spaces with unnatural feeding patterns. In many cases, such animals suffer terribly throughout their lives. This consideration is likely a factor in many consumers choosing to incorporate more flexitarian, vegetarian and vegan approaches to their diets in recent years. |
| • | Controlled Growing Environment: Another potential benefit of cultivated meat is that its growth environment is designed to be less susceptible to biological risk and disease, through standardized, tailored production methods consistent with good manufacturing practices, or GMP, that are controls to contribute to improved nutrition, health and wellbeing. |
| • | Alternate Use of Natural Resources: Eight percent of the world’s freshwater supply and one third of croplands are currently used to provide for livestock. The development of cultivated meat is expected to free up many of these natural resources, especially in developing economies where they are most needed. |
| • | Food Waste: The conventional meat industry’s largest waste management problem relates to the disposal of partially-used carcasses, which are usually buried, incinerated, rendered or composted, with attendant problems such as land, water or air pollution. Cultivated meat offers a potential solution for this problem, with only the desired cuts of meat being produced for consumption and only minimal waste product generated, with no leftover carcass. |
Our vision is to be a global leader in making real meat through advanced biotechnology and engineering solutions for a more sustainable world. We are committed to making the right choice of meat for end consumers simple by developing high-quality real meat that is slaughter-free, delicious, nutritious, and safer than farm-raised meat, by adopting a factory design intended to offer a sustainable solution for producing a variety of beef, chicken, and pork products, whether as raw materials or final consumer products.
Our technologies and processes have the potential to be sustainable. We are developing a meat production process that is designed to provide sustainability in an industry that is not otherwise expected to be able to meet the growing demand for protein caused by rising population numbers and global affluence, due to inefficiencies inherent in conventional meat farming. These include the large amount of land and water use needed for raising livestock, causing precious natural resources to be squandered.
We are designing our cellular agriculture and bioprinting processes to be modular so that customers can initiate their cultivated meat activities at scales suitable for their specific needs and to grow their activities as their needs evolve. Whether a customer wishes to manufacture cultivated fat or 3D-printed steak, each facility can be adapted to scale-out product capabilities and production volumes.
We are developing a fully automated, clean, proprietary process for cultivated meat manufacturing in a controlled, sterile environment, which is expected to significantly increase food safety. Our production facilities will not house a single animal and will contain robust integrated monitoring systems and minimal human interaction, which will greatly reduce the risk of pathogen contamination of the type claimed to have caused the COVID-19 pandemic and numerous other human health crises.
We have carefully selected personnel for our management team who possess substantial industry experience, from diverse fields including the food industry, bioprinting, tissue engineering, industrial stem cell growth, and bioprinter and print materials development. We believe this blend of talent and experience in managers who share our core values gives us the requisite insights and capabilities to execute our plan to develop technologies designed to meet demand in a scalable, profitable and sustainable way.
To achieve our mission, we intend to:
Perfect the development of our cultivated meat manufacturing technology and processes. We intend to continue developing and refining our processes, procedures and equipment until we are in a position to commercialize our technologies, whether by manufacturing final products for consumers (B2C and B2B2C models) or ingredients for industrial use, as well as in outlicensing (B2B models). We are continuing to tackle the technological challenges involved in scaling up both our biological and printing processes to industrial-scale levels.
Commercialize our technologies for use in consumer and business markets. We intend to provide ingredients to business customers for use in consumer products, in order to help meet the growing demand for sustainable, slaughter-free cultivated meat products. For example, manufacturers of meat alternatives, such as vegetarian sausages, may choose to include our cultivated fat biomass in their products, in order to deliver the signature meaty flavors, aromas, and textures of the meat otherwise provided by the conventional meat of species such as chicken, beef and pork. We believe that this combination has the potential to unlock a new level of meat experience.
In addition, we intend to license our production technology as well as provide associated products, such as cell lines, printheads, bioreactors and incubators, and services, such as technology implementation, training, and engineering support, whether directly or through contractors, to food processing and food retail companies. We intend to charge our customers a production license fee, based upon the amount of meat printed. We expect that each production facility will periodically require us to provide them with our proprietary materials, such as fresh sets of starter cells, for a fee. In addition, other materials used in the production process, such as cell-culture media and additives in our bio-inks may be sourced from third parties. Whether these materials are customized for the specifics of our production processes, or ‘white-labelled’ generic materials, or proprietary materials that we have developed, we may charge a fee for restocking such materials, however we have not yet reached the stage where it would be possible to estimate to what extent this would contribute to any future revenue stream. Finally, we intend to provide paid product implementation and guidance services to our customers looking to establish cultivated meat manufacturing facilities. We expect that each facility licensing our technologies will need to deal with novel challenges and, as a result, will require the assistance of our expert knowledge in order to set up and implement the licensed technologies.
In addition, we envisage demand for our ingredients in industrial applications other than human consumption, including cosmetics, which involve the extensive use of fat, and pet food. However, our current focus remains the development of cultivated meat and its ingredients for human consumption.
Develop additional alternative proteins, such as cultivated fish, to meet growing industry demand. There are substantial technological challenges inherent in expanding our offering beyond cultivated beef, pork and avian technologies to additional alternative proteins, such as cultivated fish. However, we believe that our experience, know-how and intellectual property portfolio form an excellent basis from which to surmount such challenges. Our first step in this direction was the 2021 acquisition of Peace of Meat, with the aim of developing avian fat for the alternative meat industry, applying proprietary technology to mimic the cellular composition of conventional poultry.
Acquire synergistic and complementary technologies and assets. We intend to optimize our processes and diversify our product range to expand the cultivated meat technologies upon which marketable products can be based, through a combination of internal development, acquisitions and collaborations, with a view to complementing our own processes and diversifying our product range along the cultivated meat production value chain in order to introduce cultivated products to the global market as quickly as possible. See also “- Additional Technologies” below.
The Commercialization Roadmap
The following table sets forth a road map for the expected commercialization of substitutes for conventionally-farmed meat, commencing with fully-plant-based meat-like offerings that are already commercially available but lack the organoleptic properties of meat (primarily flavor, aroma, texture and color), to be followed by hybrid meat products of the type that we are developing, combining real cultivated fat with plant-based protein to offer meatier products with enhanced organoleptic properties, prior to the commercialization of fully-cultivated unstructured meat products, such as hamburgers and minced meat, and lastly fully-cultivated structured meat products, such as 3D-printed steaks.
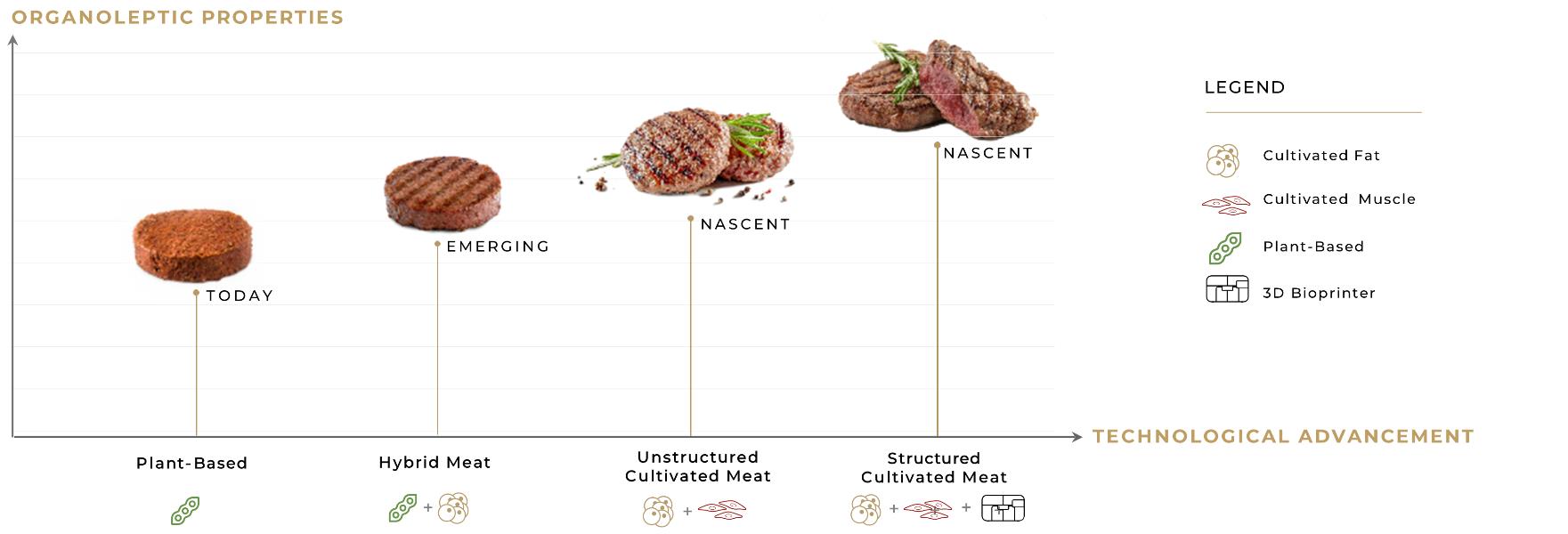
We are focusing on developing cultivated fat biomass, primarily for the purpose of commercializing hybrid meat products in the shorter term, and developing the technologies needed for both unstructured and 3D-printed, structured, cultivated meat products.
Meat Ingredients for Hybrid and Unstructured Cultivated Products
Both we and our Belgian subsidiary, Peace of Meat, continue to develop novel, proprietary, stem-cell-based technologies to produce fat, muscle and connective tissue biomass from multiple species, such as chicken, beef and pork, without harming any animals. We are leveraging this technology, including through novel hybrid food products, to expedite market entry while we develop an industrial process for cultivating and producing real meat, including using three-dimensional bioprinting technology. The first expected application of the technology is in hybrid food products, combining plant-based protein with cultivated animal fat biomass, designed to provide meat analogues with qualities of “meatiness” (taste and texture) closer to that of conventional meat products than currently available. To this end, we have conducted a number of taste tests, where we demonstrated the potential that our cultivated fat biomass has to enhance the taste of plant-based protein products. We believe that a product comprised of as little as 10-25% of our cultivated fat biomass combined with plant-based protein has the potential to enhance meatiness. Our cultivated fat biomass is designed to be free of antibiotics, can provide enhanced fatty acid profiles, and can be tailored to provide personalized nutritional profiles.
Our fat biomass production technology relies on the use of cells derived from proprietary cell lines. These cells grow naturally in suspension, in high densities. They also proliferate continuously, are relatively large and tend to easily accumulate lipid. This quality of the cells makes them an excellent candidate for producing cultured fat, so we have used them to build a robust cell line, free of genetic modifications, which we are now attempting to upscale towards industrial production volumes. Our most advanced cell line is being built with non-GMO pluripotent stem cells that can differentiate into muscle cells and fat cells, and form connective tissue, which need fewer high-cost media components, such as growth factors, for their development, which is why these cells may have higher growth potential with lower costs than alternative technologies. We have likewise developed the process for isolating, growing and differentiating bovine stem cells into muscle fibers, fat biomass and connective tissue.
Single production run of chicken fat biomass.
Some of the steps which we are taking in order to keep the growth media cost low include:
| • | Replacing expensive, animal-derived components in cell growth media with chemical replacements, including through in-house production; |
| • | Cell line optimizations, e.g. through high-throughput analyses of evolved isolates; |
| • | Bioprocess optimization and media recycling; |
| • | Upscaled growth factor production, e.g. through hollow fiber bioreactors; and |
| • | Long-term market optimization as a result of expected increased demand. |
Structured Fully-Cultivated Meat
In addition to meat ingredients for use in hybrid meats and unstructured, cultivated meat, we are developing the technology and processes to produce cultivated meat steak at an industrial scale. We are working to achieve this by creating an end-to-end technology that combines cellular agriculture with bioprinting to produce complex meat structures. We are developing cellular agriculture technology, such as cell lines, and approaches to working with growth media to support the growth of cells such as fat and muscle cells in a scalable process, and have demonstrated an ability to differentiate stem cells into fat and muscle cells. The media will be composed of food-grade ingredients and we expect their growth factors to be similar to those produced naturally in the bodies of cattle, albeit free of fetal bovine serum, traditionally a significant component of cellular growth media that is harvested from animals. We are engaged with experimentation to develop optimal and cost-effective cell culture media, and are exploring a range of types of, and sources for, growth factors suited to cell culture. These sources are expected to be sustainable and ethical, providing a route to enabling effective and cost-effective processes. The processes we are developing are designed to allow cells of interest, following humane tissue extraction from the umbilical cord or biopsy, to be isolated, replicated, grown and maintained in vitro under controlled, laboratory conditions.
We are developing proprietary bioprinting and tissue engineering technologies to enable the design and bioprinting of three-dimensional tissues. Our goal is for the meat produced using these technologies to have an authentic texture, flavor, appearance and aroma without being limited to the precise combinations of existing meat tissue (so that fat content of the meat, for example, can be adjusted to amounts other than those occurring naturally in animals, to meet varied consumer preferences for fattier or leaner cuts of meat). We believe the novel processes we are developing have the potential to eventually be competitive with conventional manufacturing technologies for premium products, as large-scale production of meat tissues will create new lines of meat without any unnecessary animal use.
In the course of developing our technologies, we intend to develop a large-scale technology demonstration model. We have set forth below an illustration of the process we are developing that we believe, upon completion, will allow us and our customers to develop and manufacture cultivated steaks at industrial scale.
We are working on slaughter-free meat development processes, including cell proliferation and differentiation, including experiments with stem cell growth media to grow high-density stem cells based solely on compounds produced in laboratory processes.
In these experiments, we have developed stem cells able to differentiate into fat or muscle cells, allowing the building of fat tissue and muscle fibers, following on from an isolation process of specific stem cells from sources such as bovine umbilical cords or muscle tissue. While preparing our 3.67 oz. steak, these cells were nourished with nutritional compounds that we developed as a growth medium to direct their differentiation into fat tissue or muscle tissue as needed.
Cell source for cultured meat products
The process of industrial scale meat printing necessitates the isolation and development of cells able to produce both animal muscle and fat tissues. Our proprietary cell lines are isolated from various sources harboring these properties, for example embryonic stem cells (stem cells isolated from embryos at a very early development stage), adult stem cells isolated from various adult tissues (e.g., fat and muscle tissues) and stem cells isolated from the umbilical cord immediately following birth. Each of these cells has advantages and disadvantages and their adaptation to our robust meat production process is currently being evaluated.
We are using software-controlled bioreactors to foster cell proliferation. The initial growth phase leverages exponential growth of stem cells to achieve sufficient cell volumes for food production. These stem cells undergo differentiation into multiple cell types, such as muscle and fat, as well as cell maturation, where cells continue to proliferate, spread and, for printed cells, produce the extra-cellular matrix which transforms the dispersed printed cells into a whole tissue.
We are in the process of developing cell-culturing processes and protocols for use in bioreactor systems. Such bioreactor systems will enable monitoring and control of growth parameters, as well as testing and development of efficient and economical cell-growth processes in industrial breeding containers. Separate from the bioreactor development process, we have commenced development of a cell-suspension growth process. This growth process is different from cell growth on laboratory plates. We expect that the newly-developed processes may allow cell growth on a scale needed for industrial-scale meat development. We have already developed a cell-suspension growth process using chicken and beef cells, in the course of developing both structured and unstructured products.
Structured, three-dimensional printed products require the use of bio-inks, which are printable biological materials produced from the biomass developed in our bioreactors, as well as scaffolding inks that may be non-biological in origin. Bioinks produced differ in their differentiation potential into muscle, fat and connective tissue. In this step, our bio-inks are printed in thin layers in the desired combination, providing creative control over the steak design, in a process that maintains the ongoing viability of the bio-ink cells. As the printed layers are composed of viable cells, they are then able to join together in an incubator with the help of bonding agents that serve as a scaffold, forming three-dimensional tissues. We are in the process of optimizing the characteristics of our proprietary bio-inks, including composition, motility, viscosity, temperature, structural stability, density and jettability, or the ability to be dispersed by a printer, as well as the factors helping the cells to connect in three-dimensional tissues.
To date, we have produced and manufactured bio-inks designed to create fat and muscle cells and tissues. We have also built and successfully tested a three-dimensional prototype digital bio-ink printer for printing cultivated meat cells, as well as supporting systems, with a view to developing industrial bio-print heads for the purpose of large-scale printing.
Bioprinting is a process of fashioning a specific type or types of native or manipulated cells configured to form the edible tissue analog, by depositing scaffolding material mixed with cells and other bio-inks using an inkjet-style printer with drop-on-demand capabilities, where inks are dispensed only where needed.
The image below depicts a potential laboratory model that we could use for the development and production of cultivated meat steaks.
Once the tissue is bioprinted, the culture is transferred to an incubator, where, in addition to providing nutrients and other chemical and biological agents, the systems provided may physically manipulate the tissues to increase differentiation (the process by which a cell changes from one type to another) and adjust the physical properties of the extracellular matrix, or ECM. The ECM is a three-dimensional network of very large molecules, such as collagen, that provide structural and biochemical support to surrounding cells. Collagen is the ECM of a scaffold that contains nutrients, adherents, and essential growth factors for the surrounding cells, supporting the development growth of complex muscle tissues in living animal bodies. Building muscle tissues in vitro requires the development of artificial scaffolds. Plants are an obvious candidate for artificial scaffolding, as plant fiber is similar in composition to collagen fiber.
To date, we have printed several cell types, which coalesced into fat and muscle tissue grown in our laboratory. To the best of our knowledge, this is the first digital bio-ink printing of food (using a bioprinter developed in-house for industrial use) to coalesce living tissue made up of several different cells that can be derived from a live cow. We observed that the digital printer arranged the cells in space as planned, with coalescence observed both between different cells and between cells and their environment, both of which are essential for tissue formation.
Cultivated Steak Scaffolding
Growing three-dimensional meat presents a unique challenge. Typically, animal cells must remain within 200 microns of a nutrient supply in order to survive. This is little more than the width of a human hair and is known as the diffusion limit. It is the reason that cells grow along the surface of a petri dish, rather than forming vertical piles.
In the next step of the process we are developing, we intend to build a scaffold to support the growth of three-dimensional meat. A “scaffold”, or “biocompatible scaffolding”, refers to an engineered platform having a predetermined three-dimensional structure, which mimics the environment of the natural extracellular material, or ECM, provides short-term mechanical support for the tissue culture, and provides an increased surface area for cell adhesion, proliferation, migration, and differentiation, eventually leading to accelerated tissue formation. We are developing technology to allow for the formation of a composite scaffold.
Modularity
We are focused on developing a process that will allow our food technology customers to operate a high-throughput manufacturing process for high-quality, healthy meat. Our cellular agriculture and bioprinting processes are being designed to be modular, in that they can work using different sizes of factory. We believe we could license our technology to customers with industrial plants close to urban areas seeking to provide ‘just in time’, logistically-efficient, local, and premium cellular agriculture. In addition, we believe a licensee of our technology could build a plant in a locality that does not have the resources needed for industrial animal husbandry, allowing places like the United Arab Emirates, Hong Kong or Singapore to potentially become more agriculturally independent, thus increasing food security. As costs continue to decrease, we believe licensees of our technology could also build production facilities in localities where there is high agricultural seasonality or desertification risk.
Illustration of a contemplated cultivated meat manufacturing plant.
Clean Energy
We are developing processes intended to achieve high-volume manufacturing capabilities in line with the needs of today’s value-added food processors and other meat and food industry players. To this end, we are working on processes to scale up production, starting with stem cells. We expect high-volume stem cell production to feed into differentiation bioreactors that are dedicated to producing fat and muscle cells. These cells are the key input for our downstream productization stages.
The processes we are developing are advanced biotechnological processes, intended to produce meat in a clean environment with minimal environmental impact. We envision factories utilizing our technologies that exist in greater harmony with their environment than typical current factories, supporting sustainability, utilizing renewable energy sources, and recycling or treating their own waste.
Additional Technologies
We may incorporate novel bioreactor technologies that benefit cellular agriculture and the development of low-cost cell culture media not based on fetal bovine serum.
We also plan to add cell line types to expand the development of cultivated meat to other types of animals, as well as achieving market penetration in the shortest timeframe possible, thus realizing the great potential in the market. We are developing cultivated meat, both unstructured hybrid products and structured, three-dimensional printed products, with an initial emphasis on bovine and porcine cells, while our subsidiary Peace of Meat is developing cultivated avian fat, initially for use in hybrid products. We estimate that the first hybrid products based on Peace of Meat technology may enter the market as early as 2025. Beyond hybrid products, cultivated fat is expected to be a component in other fat-based products (edible and otherwise), and an integrated component in MeaTech’s printing technology. We are working to create synergy and added value to the cultivated meat market, while sustaining animal welfare and meeting the growing global demand for meat.
Sales and Distribution
We do not yet have any sales, marketing or distribution infrastructure or capabilities. In the event that we complete development of our technologies and secure adequate funding, we intend to consider commercialization collaborations, where appropriate.
We have engaged in a consulting agreement with the Adom Group, or Adom, under which Adom serves as a consultant for the development of our operations in the cultivated meat production industry, and will assist us in penetrating the markets in which Adom operates in Europe and South America. Under the agreement with Adom, we granted Adom first refusal rights for establishing a production plant using the technology that we are developing, in any or all of Israel, Poland, Argentina and Brazil. According to the terms of the agreement, should Adom induce a leading producer in the local meat industry in the target country to invest in us at least $1 million and engage with the producer to establish a production plant franchise based on our technologies, we will grant Adom and the franchisee first refusal rights for production in that target country, subject to the completion of certain fundraising milestones, by or in conjunction with Adom.
Apart from end consumers in B2C and B2B2C models of branded products, we believe that our ideal business customers will be value-added food processors and retailers that wish to benefit from cultivated meat manufacturing capabilities. We intend to provide our corporate customers with a solution to these needs in the form of highly-automated, cleaner, ‘just-in-time’ manufacturing of cultivated meat products using a repeatable, consistent manufacturing process. Our goal is for our customers to be able to streamline their meat supply chain, introduce greater manufacturing flexibility, and locate their cultivated meat production facilities closer to the point of retail or consumption.
We intend to provide our business licensees with assistance in constructing facilities to employ our proprietary technology and processes. We expect that we will need to collaborate with third parties to obtain and make available to our customers the expertise necessary to provide this assistance. In addition, we intend to procure the equipment our licensees need to deploy our proprietary technology and processes from third-party providers. Some equipment, such as piping, clean rooms, and packing and freezing equipment, are standard industry equipment and can be sourced on open markets. Other equipment such as bioreactors and our proprietary bioprinters, will need to be produced by contract manufacturers.
Intellectual Property
We have sought and continue to seek patent protection as well as other intellectual property rights for our products, processes and technologies in the United States and internationally. Our policy is to pursue, maintain, expand, protect and defend our patent rights and trade secrets, which we believe enable us to deliver long-term protection for the proprietary technologies, inventions and improvements that are commercially important to the development of our business.
We have a growing portfolio of 14 provisional and non-provisional patent applications with the USPTO and WIPO filed through the PCT. A provisional patent application is a preliminary application, and establishes a priority date for the patenting process of inventions disclosed therein.
Our existing patent portfolio can currently be divided into two main areas:
Mechanical: covering printer components and peripherals used in the fabrication of the tissue cultures with two PCT applications filed:
The first is directed to print heads operable in a bioprinting systems for the fabrication of edible biostructures using drop-on-demand, the print heads specifically designed to accommodate bio fluids of suspended systems without causing demixing, while still delivering bio fluids with high accuracy and precision. The provisional application was filed in March 2020. Research, development and engineering of the technology is ongoing. A Go/No-go decision regarding entry to national stage is expected in September 2022.
The second is a PCT application directed to systems and methods of physically manipulating a resilient container (bladder) of bioprinted tissue culture having non-random three dimensional cell structure over 4 dimensions, namely elongation, compression, torsion and shear, to modulate the tissue and achieve the desired texture for each meat type. The PCT Application was filed in September 2020, with entry to national stage in March 2022. Research, development and engineering of the technology is ongoing.
Biological: covering initial materials used in the process and the methods for their use in the bioprinted tissues with two PCT applications and two provisional applications filed.
One PCT application is directed to methods for harvesting ICM from bovine blastocysts, the systems used to implement the methods with the disclosed compositions, are used to culture the harvested inner cell mass (ICM) for embryonic stem cells (ESC) for the formation of tissue cultivated to emulate tissues and/or organs for (non-vegan) food consumption. The PCT application was filed in January 2021 under the PCT, with publication due in July 2022.
Another pending provisional is directed to methods and compositions for the xeno-free propagation of bESC on bovine umbilical stem cells (bUCSC), derived from a bovine umbilical cord. The provisional application was filed in January 2021, and the PCT application in January 2022.
The second pending provisional application is directed to the use of plant-based lecithins and/or their components as a differentiation drivers for use in selectively promoting adipocytes differentiation by exposing MSCs post spontaneous immortalization, to the plant lecithins and/or their components for production of cultured fat ex-vivo. The Application was filed in August 2021. A Go/No Go decision concerning filing of the non-provisional application is expected in the second quarter of 2022.
Additional provisional applications are directed to spherified substrate layer, for bioprinting and myogenic differentiation enhancers.
In addition to patent applications, we maintain trade secrets covering know-how and proprietary information relating to our core technologies and make practicable efforts to protect our confidential trade secrets. To this end, we require our employees engaged in the development of intellectual property to enter into confidentiality agreements prohibiting the disclosure of confidential information and further, require disclosure and assignment of any inventions and associated intellectual property rights that are important to our business. Additionally, we require all entering employees to represent they are not bringing in, or are using any third party’s Trade Secrets. We have also registered our name and logo as registered trademarks in the United States, and maintain ongoing rights to our domain name.
While our policy is to obtain patents by application, license or otherwise, to maintain trade secrets and to seek to operate without infringing on the intellectual property rights of third parties, technologies related to our business have been rapidly developing in recent years. Additionally, patent applications that we may file or license from third parties may not result in the issuance of patents, and our current or future issued patents may be challenged, invalidated or circumvented. Therefore, we cannot predict the extent of claims that may be allowed or enforced against our patents, nor be certain of the priority of inventions covered by pending third-party patent applications. If third parties prepare and file patent applications that also claim technology or therapeutics to which we have rights, we may have to engage in proceedings to determine priority of invention, which could result in substantial costs to us, even if the eventual outcome is favorable. Moreover, because of the extensive time required for clinical development and regulatory review of products we may develop, it is possible that the patent or patents on which we rely to protect such products could expire or be close to expiration by the commencement of commercialization, thereby reducing the value of such patent. Loss or invalidation of certain of our patents, or a finding of unenforceability or limited scope of certain of our intellectual property, could also have a material adverse effect on us. See “Risk Factors — Risks Related to our Intellectual Property and Potential Litigation.”
Competition
We expect that demand for our cultivated meat manufacturing plants will be driven by consumer demand for alternative proteins and, more specifically, consumer acceptance of cultivated meat as the alternative protein of choice. We believe we will compete with other cultivated meat manufacturers, alternative protein manufacturers, and the conventional meat industry as a whole. We expect to directly compete with companies licensing know-how or otherwise enabling the establishment of cultivated meat manufacturing plants. We are aware of certain companies that have announced plans to provide their cultivated meat technology on a B2B basis, however we are not currently aware of a potential competitor focusing on complex, industrial-scale, bioprinted, high-value real meats, such as steak.
Companies such as Upside Foods and Mosa Meat are focused on producing red meats, BluNalu, Inc. on fish and Shiok Meats on crustaceans. There are different companies working on culturing varying cell types, such as chicken, pork, kangaroo and foie gras. This push on scaling-up cellular agriculture can serve as a solution to the scale and environmental challenges confronting traditional meat production. Other alternative protein competitors such as Beyond Meat and Impossible Foods, Inc. are already selling plant-based meat substitutes, but to the best of our knowledge, these companies are not focused on the production of real meat products produced with animal cells.
Companies Developing Vegetable and Insect Protein Alternatives
��
There are numerous companies focused on developing meat substitutes. In order for a product to achieve commercial acceptance as an alternative to meat, it must have an appearance, taste, smell and nutritional values that are similar enough to the type of meat that it seeks to replace or with which it seeks to compete. These meat substitute companies generally employ proprietary formulae for manufacturing, based wholly on ingredients of plant origin. In addition, we are aware of several companies developing insect-protein production capabilities, employing among other insects, flies, larvae and grasshoppers.
Companies Developing Cultivated Meat
The cellular agriculture meat sector is in early stages of development. The sector is currently primarily comprised of companies developing a full technology stack from developing cell lines to scaling up cellular cultivation, developing media, and researching the food technology aspects of the final product. Market dynamics have led to a large number of companies operating in this manner. We are aware of approximately 80 companies operating in the cell-based field, several of whom are developing cellular agriculture for ground-meat alternatives and appear to be progressing with their technological development. Some have indicated readiness to bring cell-based meat products to market as early as 2022 through 2023. We do not believe that any companies in this space have already developed the capability to produce industrial quantities at prices low enough to compete on a dollar-per-pound basis against conventionally-harvested meat.
A number of larger companies have begun engaging in this sector. For example, companies such as Merck & Co., Inc. and Lonza Group AG are currently investing in capabilities to accommodate the market’s desire for change in the cell culture media market. Additionally, a number of bioreactor companies are rumored to be interested in the cellular agriculture market opportunity. Over time, we expect that larger players will continue to increase their exposure to cellular meat production either by selling to, or collaborating with, the many start-ups in the space.
Currently cellular agriculture companies are for the most part paving their own path, with a goal of producing meat cells suitable as a replacement for ground meat. The ground meat type of cellular product may also be suitable as an ingredient in a hybrid plant-based food product. The cell-types relevant to this effort are primarily muscle and fat cells. What exactly these cell-based companies will offer is likely to be affected by consumer expectations and underlying cost structures. We believe these companies may have to mix their cellular meat product with plant-based ingredients in the interests of cost or appearance.
Companies Developing Structured Cultivated Meat Products
To our knowledge, there is currently no other company focused on the scaling up of three-dimensional bioprinting. However, there are companies attempting to produce steaks by means of other approaches, such as growing bovine cells including fat, muscle and connective tissue on a pre-prepared scaffold, in order to create a contiguous piece of meat, which has so far yielded steaks.
Government Regulation
Regulators around the world are in the process of developing a regulatory approval process for cultured meat. Cultured meat is not yet generally commercially available, but technologies like ours are anticipated to facilitate the imminent scaling up of cultured meat production. In general, cultured meat production is expected to be subject to extensive regulatory laws and regulations in the United States and in other jurisdictions such as Canada, Japan, the European Union and the United Kingdom. In the United States, existing food safety requirements are expected to apply. Additional details are being developed at the FDA and USDA pursuant to a Memorandum of Understanding, or MOU, published by the FDA and USDA on March 7, 2019 entitled “Formal Agreement to Regulate Cell-Cultured Food Products from Cell Lines of Livestock and Poultry.” For example, FDA anticipates publishing Draft Guidance on premarket safety oversight by December 31, 2022, and in September 2021 USDA published an Advance Notice of Proposed Rulemaking (ANPR), indicating that USDA will be developing new labeling requirements for foods under its jurisdiction produced through cell culture technology.
Under the MOU — which is expected to affect our customers producing cultured meat — the two agencies will operate under a joint regulatory framework wherein FDA will oversee cell collection, cell banks, and cell growth and differentiation. A transition from FDA to USDA oversight will then occur during the cell harvest stage, at which point the USDA will oversee the production and labeling of cultured meat. The USDA will be advancing new labeling requirements. To the best of our knowledge, the regulatory approval details under development, including the Draft Guidance on FDA premarket oversight, are not expected to apply to our business directly, but they are instructive as to the regulatory requirements our cultured meat production customers are expected to face and their expectations of us, in the form of customer assurances, regarding our product.
At this time, our business is limited to developing cultured meat production technology (i.e., bioprinters) that will be marketed to cultured meat producers, and that of Peace of Meat is limited to developing cultured meat ingredients (such as cultured avian fat). In the United States, and consistent with the Federal Food, Drug and Cosmetic Act, Federal Meat Inspection Act, and Poultry Products Inspection Act, food ingredient manufacturers (like Peace of Meat) must comply with the FDA’s food production requirements under the FDCA, as amended by the Food Safety Modernization Act, or the FSMA, to ensure that the food is safe, and the USDA's requirements that the ingredients, when used in USDA-regulated meat and poultry products, are effective and suitable for their intended use.
In addition, production equipment manufacturers must ensure that their products do not contribute to the production of adulterated food. The regulatory obligation falls on the food manufacturer to ensure that all food produced — including cultured meat — is wholesome and not adulterated. Therefore, when sourcing food processing equipment, such as the three-dimensional bioprinter we are developing, our customers will request assurances that the bioprinter is safe for its intended use and will not result in the production of adulterated food. We intend to monitor developments at the FDA and USDA in connection with the aforementioned FDA-USDA MOU to determine whether any specific requirements or recommendations are published with specific regard to cultured meat equipment manufacturers.
In the United States, we expect companies manufacturing cultured meat products to be subject to regulation by various government agencies, including the FDA, USDA, and the FTC. Equivalent foreign regulatory authorities include the Canadian Food Inspection Agency, the Japanese Food Safety Commission, the European Food Safety Authority and authorities of the EU member states, the State Food and Drug Administration of China and the Singapore Food Agency. These agencies, among other things, prescribe the requirements and establish the standards for food quality and safety, and regulate various food technologies, including alternative meat product composition, ingredients, manufacturing, labeling and other marketing and advertising to consumers.
We expect that federal, state and foreign regulators will have the authority to inspect our customers’ facilities to evaluate compliance with applicable food safety requirements. Federal, state, and foreign regulatory authorities also require that certain nutrition and product information appear on the product labels of our customers’ food products and, more generally, that such labels be truthful and non-misleading and that marketing and advertising be truthful, non-misleading and not deceptive to consumers.
As the cell-based agriculture industry is young and its regulatory framework is emerging and evolving, legislation and regulation may evolve to raise barriers to our go-to-market strategies.
In addition to federal regulatory requirements in the United States, certain states impose their own manufacturing and labeling requirements. For example, states typically require facility registration with the relevant state food safety agency, and those facilities are subject to state inspections as well as federal inspections. Further, states can impose state-specific labeling requirements. In the U.S., USDA will be developing new labeling requirements for foods under its jurisdiction produced through cell culture technology as noted in an Advance Notice of Proposed Rulemaking (ANPR) published in September 2021.
We are subject to labor and employment laws, laws governing advertising, privacy laws, safety regulations and other laws, including consumer protection regulations that regulate retailers or govern the promotion and sale of merchandise. Our operations are subject to various laws and regulations relating to environmental protection and worker health and safety matters. We monitor changes in these laws and believe that we are in material compliance with applicable laws.
Environmental, Health and Safety Matters
We, our agents and our service providers, including our manufacturers, may be subject to various environmental, health and safety laws and regulations, including those governing air emissions, water and wastewater discharges, noise emissions, the use, management and disposal of hazardous, radioactive and biological materials and wastes and the cleanup of contaminated sites. We believe that our business, operations and facilities, including, to our knowledge, those of our agents and service providers, are being operated in compliance in all material respects with applicable environmental and health and safety laws and regulations. Based on information currently available to us, we do not expect environmental costs and contingencies to have a material adverse effect on us. However, significant expenditures could be required in the future if we, our agents or our service providers are required to comply with new or more stringent environmental or health and safety laws, regulations or requirements.
Except as stated above, we are not aware of any environmental risks related to our operations, and therefore, we do not believe that environmental regulations will have a significant effect on us. However, in the future, we may be required to meet environmental protection standards or regulations which could have a material impact on our activities, activities, profitability and ability to remain competitive.
C. Organizational Structure
Our subsidiaries and the countries of their incorporation are as follows:
| Name | | Jurisdiction of
Incorporation | | | Parent | | % Ownership | |
| MeaTech U.S., Inc. | | | Delaware, U.S. | | | MeaTech 3D Ltd. | | | 100 | % |
| MeaTech MT Ltd. | | | Israel | | | MeaTech 3D Ltd. | | | 100 | % |
| MeaTech Europe BV | | | Belgium | | | MeaTech 3D Ltd. | | | 100 | % |
| Peace of Meat BV | | | Belgium | | | MeaTech Europe BV | | | 100 | % |
D. Property, Plant and Equipment
Our office and laboratory are located at 5 David Fikes St., Rehovot, Israel. The laboratory and office space total approximately 18,300 square feet. The lease for this facility will expire in January 2026, although we have an option to renew it for four years, and the annual rent (including parking fees) is approximately $0.7 million, linked to the Israeli CPI.
| | ITEM 4A. | UNRESOLVED STAFF COMMENTS |
None.
| | ITEM 5. | OPERATING AND FINANCIAL REVIEW AND PROSPECTS |
The following “Operating and Financial Review and Prospects” should be read together with the information in our financial statements and related notes included elsewhere in this Annual Report. The following discussion is based on our financial information prepared in accordance with the International Financial Reporting Standards, or IFRS, as issued by the International Accounting Standards Board, or IASB, which may differ in material respects from generally accepted accounting principles in other jurisdictions, including U.S. GAAP. The following discussion includes forward-looking statements that involve risks, uncertainties and assumptions. Our actual results may differ materially from those anticipated in these forward-looking statements as a result of many factors, including but not limited to those described in “Risk Factors” and elsewhere in this Annual Report. Please also see “Forward-Looking Statements.”
For a discussion of our results of operations for the year ended December 31, 2020, including a comparison between 2020 and 2019, refer to “Management’s Discussion and Analysis of Financial Condition and Results of Operations—Results of Operations—Year Ended December 31, 2020 Compared to Year Ended December 31, 2019” in our annual report on Form 20-F filed on April 21, 2021.
A. Operating Results
To date, we have not generated any revenue since we commenced our cultured meat operations. We do not expect to receive any revenue unless and until we complete development of and successfully commence out-licensing our technologies, or until we receive revenue from a collaboration or other partnership such as a co-development agreement, or the acquisition of a company that generates revenues. There can be no assurance that we will be successful in developing or ultimately commercializing our technologies, in establishing revenue-generating collaborations or acquiring revenue-generating companies.
Research and Development Expenses
Research and development activities are our primary focus. We do not believe that it is possible at this time to accurately project total expenses required for us to reach the point at which we will be ready to out-license our technologies. Development timelines, the probability of success and development costs can differ materially from expectations. In addition, we cannot forecast whether and when collaboration arrangements will be entered into, if at all, and to what degree such arrangements would affect our development plans and capital requirements. We expect our research and development expenses to increase over the next several years as our development program progresses. We would also expect to incur increased research and development expenses if we were to identify and develop additional technologies.
Research and development expenses include the following:
| • | employee-related expenses, such as salaries and share-based compensation; |
| • | expenses relating to outsourced and contracted services, such as external laboratories and consulting, research and advisory services; |
| • | supply and development costs; |
| • | expenses, such as materials, incurred in operating our laboratories and equipment; and |
| • | costs associated with regulatory compliance. |
We recognize research and development expenses as we incur them.
Marketing expenses consist primarily of professional services, personnel costs, including share-based compensation related to employees, and business development, public relations and investor relations services.
General and Administrative Expenses
General and administrative expenses consist primarily of personnel costs, including share-based compensation related to directors and employees, corporate costs (such as insurance), facility costs, patent application and maintenance expenses, and professional service costs, including legal, accounting, audit, finance and human resource services, and other consulting fees.
Based on the reverse acquisition method, the assets and liabilities of MeaTech (the acquirer for accounting purposes) were recognized in our financial statements at their book value at the date of closing of the merger in January, 2020. The acquisition consideration, in the amount of $11.4 million, was set based on the closing price of Ophectra's shares on the Tel Aviv Stock Exchange on the date of closing of the Merger, while any surplus proceeds of the acquisition over the fair value of Ophectra’s net assets (excluding its net assets that were transferred to a settlement fund as described in “- Merger” above) were recognized in profit or loss as public listing expenses in the amount of $10.2 million, that did not affect cash flow.
Finance Expenses (income), Net
Finance expenses (income), net, consisted primarily of a change in the fair value of financial instruments mandatorily measured at fair value through profit or loss, and exchange rate fluctuations.
We have yet to generate taxable income. As of December 31, 2021, our operating tax loss carryforwards were approximately $18.2 million.
Our results of operations have varied in the past and can be expected to vary in the future due to numerous factors. We believe that period-to-period comparisons of our operating results are not necessarily meaningful and should not be relied upon as indications of future performance.
Below is a summary of our results of operations for the periods indicated (in thousands):
| | | Year Ended December 31, | |
| | | 2021 | | | 2020 | |
| | | | | | | |
| Operating expenses: | | | | | | |
| Research and development expenses | | $ | 7,594 | | | $ | 2,491 | |
| Marketing expenses | | | 1,628 | | | | 506 | |
| General and administrative expenses | | | 8,010 | | | | 5,380 | |
| Public listing expenses | | | - | | | | 10,164 | |
| Loss from operations | | $ | 17,232 | | | $ | 18,541 | |
| Finance income | | | 509 | | | | 110 | |
| Finance expense | | | 1,299 | | | | 93 | |
| | | | | | | | | |
| Finance expense (income), net | | | 790 | | | | (17 | ) |
| Net loss | | $ | 18,022 | | | $ | 18,524 | |
Year Ended December 31, 2021 Compared to Year Ended December 31, 2020
Research and development expenses
Research and development expenses increased by approximately $5.1 million, or 205%, to approximately $7.6 million for the year ended December 31, 2021, compared to $2.5 million for year ended December 31, 2020. The increase resulted mainly from payroll expenses, materials and professional services expenditures related to our cultured meat research and development operations.
The increase reflects MeaTech’s growing investment in research and development as it achieves its milestones and expands its cultured meat technology capabilities.
Marketing expenses increased by approximately $1.1 million, or 222%, to approximately $1.6 million for the year ended December 31, 2021, compared to $0.5 million for year ended December 31, 2020. The increase resulted mainly from professional services, personnel costs, including share-based compensation related to employees, and business development, public relations and investor relations services.
General and administrative expenses
General and administrative expenses increased by approximately $2.7 million, or 49%, to approximately $8.0 million for the year ended December 31, 2021, compared to approximately $5.4 million for the year ended December 31, 2020. The increase resulted mainly from personnel costs, corporate expenses, professional services (such as legal and audit fees) and operating expenditures.
Net loss decreased by approximately $0.5 million to approximately $18.0 million for the year ended December 31, 2021, compared to $18.5 million for the year ended December 31, 2020. Net of the $10.2 million non-cash public listing expense that the Company recorded in 2020 in connection with its reverse merger into a TASE-traded shell company, the net loss increased by approximately $9.7 million, or 116%, driven mainly by increased research and development and general and administrative expenses.
Critical Accounting Policies
We describe our significant accounting policies and estimates in Note 3 to our annual financial statements contained elsewhere in this annual report. We believe that these accounting policies and estimates are critical in order to fully understand and evaluate our financial condition and results of operations.
We prepare our financial statements in accordance with IFRS as issued by the IASB.
In preparing these financial statements, management has made judgments, estimates and assumptions that affect the application of our accounting policies and the reported amounts recognized in the financial statements. On a periodic basis, we evaluate our estimates, including those related to share-based compensation and derivatives. We base our estimates on historical experience, authoritative pronouncements and various other assumptions that we believe to be reasonable under the circumstances. Actual results may differ from these estimates.
Recently-Issued Accounting Pronouncements
Certain recently-issued accounting pronouncements are discussed in Note 3, Summary of Significant Accounting Policies, to the consolidated financial statements included in elsewhere in this registration statement, regarding the impact of the IFRS standards as issued by the IASB that we will adopt in future periods in our consolidated financial statements.
Emerging Growth Company Status
We qualify as an “emerging growth company” as defined in the Jumpstart Our Business Startups Act of 2012, or the JOBS Act. An emerging growth company may take advantage of specified reduced reporting and other burdens that are otherwise applicable generally to public companies. These provisions include:
| • | to the extent that we no longer qualify as a foreign private issuer, (i) reduced disclosure obligations regarding executive compensation in our periodic reports and proxy statements and (ii) exemptions from the requirement to hold a non-binding advisory vote on executive compensation, including golden parachute compensation; |
| • | an exemption from the auditor attestation requirement in the assessment of our internal control over financial reporting pursuant to the Sarbanes-Oxley Act of 2002; and |
| • | an exemption from compliance with the Critical Audit Matters requirement that the Public Company Accounting Oversight Board has adopted regarding a supplement to the auditor’s report providing additional information about the audit and the financial statements. |
We may take advantage of these exemptions for up to five years or until such earlier time that we are no longer an emerging growth company. We would cease to be an emerging growth company upon the earliest to occur of: (i) the last day of the fiscal year in which we have total annual gross revenues of $1.07 billion or more; (ii) the date on which we have issued more than $1.0 billion in nonconvertible debt during the previous three years; (iii) the date on which we are deemed to be a large accelerated filer under the rules of the SEC; or (iv) the last day of the fiscal year following the fifth anniversary of our initial Nasdaq offering of March 2021. We may choose to take advantage of some but not all of these exemptions. Section 107 of the JOBS Act provides that an “emerging growth company” can take advantage of the extended transition period provided in Section 7(a)(2)(B) of the Securities Act for complying with new or revised accounting standards. This means that an “emerging growth company” can delay the adoption of certain accounting standards until those standards would otherwise apply to private companies. Given that we currently report and expect to continue to report our financial results under IFRS as issued by the IASB, we will not be able to avail ourselves of this extended transition period and, as a result, we will adopt new or revised accounting standards on the relevant dates on which adoption of such standards is required by the IASB.
B. Liquidity and Capital Resources
Since the commencement of our cultured meat operations, we have not generated any revenue and have incurred operating losses and negative cash flows from our operations. We have funded our operations primarily through the sale of equity securities. From the inception of MeaTech through December 31, 2021, we raised an aggregate of $42.3 million in four rounds of private placements of our securities and our initial public offering of securities on Nasdaq, and $6.0 million in proceeds from option exercises. As of December 31, 2021, we had $19.2 million in cash and cash equivalents.
The table below shows a summary of our cash flows for the periods indicated:
| | | Year Ended December 31, | |
| | | 2021 | | | 2020 | |
| | | | | | | |
| Net cash used in operating activities | | $ | (13,960 | ) | | $ | (3,832 | ) |
| Net cash used in investing activities | | | (9,340 | ) | | | (1,875 | ) |
| Net cash provided by financing activities | | | 29,023 | | | | 17,345 | |
| Net increase in cash and cash equivalents | | $ | 5,723 | | | $ | 11,638 | |
Year Ended December 31, 2021 Compared to Year Ended December 31, 2020
Net cash used in operating activities
Net cash used in operating activities increased by $10.1 million, or 264%, to approximately $14.0 million for the year ended December 31, 2021 compared to approximately $3.8 million for the year ended December 31, 2020. This increase was due to the increase in net loss.
Net cash used in investing activities
Net cash used in investing activities increased by $7.5 million, or 398%, to approximately $9.3 million for the year ended December 31, 2021 compared to $1.9 million for the year ended December 31, 2020. This increase was driven mainly by our investment in Peace of Meat and our acquisition of laboratory equipment and other fixed assets.
Net cash provided by financing activities
Net cash provided by financing activities increased by $11.7 million, or 67%, to approximately $29.0 million for the year ended December 31, 2021 compared to $17.3 million for the year ended December 31, 2020. This increase was driven mainly from the Company’s initial Nasdaq public offering and issuance of shares and warrants, and receipt of proceeds from the exercise of share options.
We have incurred losses and cash flow deficits from operations since the inception of MeaTech, resulting in an accumulated deficit as of December 31, 2021 of approximately $37 million. We anticipate that we will continue to incur net losses for the foreseeable future. We believe that our existing cash and cash equivalents will be sufficient to fund our projected cash needs through the fourth quarter of 2022. We do not currently have any specific commitments or plans for acquisitions; to the extent we do engage in acquisitions, we will do so after ensuring that we will have sufficient funds available to meet our capital requirements, and such acquisitions are likely to affect our projected cash needs. To meet future capital needs, we would need to raise additional capital through equity or debt financing or other strategic transactions. However, any such financing may not be on favorable terms or even available to us. Our failure to obtain sufficient funds on commercially acceptable terms when needed would have a material adverse effect on our business, results of operations and financial condition. Our forecast of the period of time through which our financial resources will be adequate to support our operations is a forward-looking statement that involves risks and uncertainties, and the actual amount of our expenses could vary materially and adversely as a result of a number of factors. We have based our estimates on assumptions that may prove to be wrong, and our expenses could prove to be significantly higher than we currently anticipate.
Our future capital requirements will depend on many factors, including, but not limited to:
| • | the progress and costs of our research and development activities; |
| • | the costs of development and expansion of our operational infrastructure; |
| • | the costs and timing of developing technologies sufficient to allow food production equipment manufacturers and food manufacturers to product products compliant with applicable regulations; |
| • | our ability, or that of our collaborators, to achieve development milestones and other events or developments under potential future licensing agreements; |
| • | the amount of revenues and contributions we receive under future licensing, collaboration, development and commercialization arrangements with respect to our technologies; |
| • | the costs of filing, prosecuting, enforcing and defending patent claims and other intellectual property rights; |
| • | the costs of contracting with third parties to provide sales and marketing capabilities for us or establishing such capabilities ourselves, once our technologies are developed and ready for commercialization; |
| • | the costs of acquiring or undertaking development and commercialization efforts for any future products or technology; |
| • | the magnitude of our general and administrative expenses; and |
| • | any additional costs that we may incur under future in- and out-licensing arrangements relating to our technologies and futures products. |
Until we can generate significant recurring revenues, we expect to satisfy our future cash needs through capital raising or by out-licensing and/or co-developing applications of one or more of our product candidates. We cannot be certain that additional funding will be available to us on acceptable terms, if at all. If funds are not available on favorable terms, or at all, we may be required to delay, reduce the scope of or eliminate research or development efforts or plans for commercialization with respect to our technologies and make necessary change to our operations to reduce the level of our expenditures in line with available resources.
We are a development-stage technology company and it is not possible for us to predict with any degree of accuracy the outcome of our research and development efforts. As such, it is not possible for us to predict with any degree of accuracy any significant trends, uncertainties, demands, commitments or events that are reasonably likely to have a material effect on our net loss, liquidity or capital resources, or that would cause financial information to not necessarily be indicative of future operating results or financial condition. However, to the extent possible, certain trends, uncertainties, demands, commitments and events are described herein.
Since inception, we have incurred significant losses and negative cash flows from operations and have an accumulated deficit of USD 37 million. We have financed our operations mainly through fundraising from various investors.
Our management expects that we will continue to generate losses and negative cash flows from operations for the foreseeable future, including as a result of material expenses such as leasing expenses (see “Item 4. —D. Property, Plant and Equipment”). Based on the projected cash flows and cash balances as of December 31, 2021, our management is of the opinion that our existing cash will be sufficient to fund operations until the fourth quarter of 2022. As a result, there is substantial doubt about our ability to continue as a going concern.
Management’s plans include continuing to secure sufficient financing through the sale of additional equity securities or capital inflows from strategic partnerships. Additional funds may not be available when we need them on terms that are acceptable to us, or at all. If we are unsuccessful in securing sufficient financing, we may need to cease operations.
Our financial statements include no adjustments for measurement or presentation of assets and liabilities, which may be required should we fail to operate as a going concern.
Quantitative and Qualitative Disclosures About Market Risk
Liquidity risk is the risk that we will encounter difficulty in meeting the obligations associated with our financial liabilities that are settled in cash. Cash flow forecasting is performed in our operating entities and aggregated at a consolidated level. We monitor forecasts of our liquidity requirements to ensure we have sufficient cash to meet operational needs. We may be reliant on our ability to raise additional investment capital from the issuance of both debt and equity securities to fund our business operating plans and future obligations.
Credit risk is the risk of financial loss to us if a debtor or counterparty to a financial instrument fails to meet its contractual obligations, and arises mainly from our receivables.
As part of an agreement with Therapin from May 2020, we agreed to convert an NIS 7.25 million investment in Therapin made by Ophectra and assumed by us at the Merger, into an interest-free loan, to be repaid by the latter at a rate of NIS 0.48 million per annum for ten years (NIS 4.8 million in total) plus NIS 2.45 million to be paid upon an exit event, including a public offering, or repayment of 14.74% of any distributable surplus or dividend distributed by Therapin, up to the amount of the outstanding balance, as detailed in our separation agreement with Therapin. As part of the agreement, Therapin gave us an option to convert the cash payment to equity of Therapin. Therapin has not provided any guarantees in connection with its repayment of our loan.
We restrict exposure to credit risk in the course of our operations by investing only in bank deposits.
As we have not invested in securities riskier than short-term bank deposits, we do not believe that changes in equity prices pose a material risk to our holdings. However, decreases in the market price of our ordinary shares or ADSs could make it more difficult for us to raise additional funds in the future or require us to raise funds at terms unfavorable to us.
Foreign Currency Exchange Risk
Currency fluctuations could affect us primarily through increased or decreased foreign currency-denominated expenses. Currency fluctuations had a material effect on our results of operations during the year ended December 31, 2021, although not in the year ended December 31, 2020.
C. Research and development, patents and licenses, etc.
For a description of the Company’s research and development policies for the last three years, see “Item 4.—Information on the Company—Business Overview—Intellectual Property.”
D. Trend Information
Not applicable.
E. Critical Accounting Estimates
Critical accounting estimates are those estimates made in accordance with IFRS that involve a significant level of estimation uncertainty and have had or are reasonably likely to have a material impact on the financial condition or results of operations of the registrant. For further information, see Note 2E to our annual consolidated financial statements included in this Annual Report on Form 20-F.
ITEM 6. DIRECTORS, SENIOR MANAGEMENT AND EMPLOYEES
A. Directors and Senior Management
The following table sets forth the name, age and position of each of our executive officers and directors as of the date of this Annual Report on Form 20-F. Unless otherwise stated, the address of our executive officers and directors is MeaTech 3D Ltd., 5 David Fikes St., Rehovot 7638205, Israel.
| Name | | Age | | Position |
| | | | | |
| Executive Officers: | | | | |
| Arik Kaufman | | 41 | | Chief Executive Officer |
| Omri Schanin | | 32 | | Deputy Chief Executive Officer |
| Guy Hefer | | 40 | | Chief Financial Officer |
| Dan Kozlovski | | 37 | | Chief Technologies Officer |
| Non-Employee Directors: | | | | |
| Yaron Kaiser | | 44 | | Chairman of the Board of Directors |
| David Gerbi(1)(2)(3) | | 42 | | Director |
| Eli Arad(1)(2)(3) | | 49 | | Director |
| Sari Singer(1)(2)(3) | | 42 | | Director |
| (1) | Member of the Audit Committee |
| (2) | Member of the Compensation Committee |
| (3) | Independent director as defined under Nasdaq Marketplace Rule 5605(a)(2) and SEC Rule 10A-3(b)(1). |
Executive Officers
Arik Kaufman, Chief Executive Officer
Arik Kaufman has served as our Chief Executive Officer since January 2022. He has founded various Nasdaq- and TASE-traded foodtech companies, and currently serves as director of Wilk Technologies Ltd. He is also a founding partner of the BlueSoundWaves collective, led by Ashton Kutcher Guy Oseary and Effie Epstein, which recently partnered with MeaTech to assist in attempting to accelerate the Company’s growth. Mr. Kaufman holds extensive personal experience in the fields of food-tech and bio-tech law, and has led and managed numerous complex commercial negotiations, as part of local and international fundraising, M&A transactions and licensing agreements. He holds a B.A. degree in Law from Reichman University (formerly the Interdisciplinary Center Hertzliya).
Omri Schanin, Deputy Chief Executive Officer
Omri Schanin has served as our Chief Operating Officer and later Deputy Chief Executive Officer since October 2020, after co-founding and joining us in September 2019, and as a director between March 2021 and January 2022. Between 2018 and 2019, he was founder and CEO of Docomed, a digital health company offering better treatment through continuous pain monitoring and data collection, and co-founder and CEO of Cannova, a developer of sublingual alginate strips to supply active ingredients of the cannabis plant through the tongue into the bloodstream, between 2018 and 2019. He was previously a Merage Fellow at the University of California, Irvine. Between 2013 and 2016, he attained the rank of Major (Res.) while serving as a deputy commander of an Israeli Navy ship. He was included in the Forbes Israel 30 Under 30 list for 2021. Mr. Schanin holds B.Sc. degrees in Life Sciences and Biotechnology from the Hebrew University of Jerusalem, Israel, and Business Management and Political Science from the University of Haifa, and an MBA in Entrepreneurship and Innovation from the College of Management Academic Studies.
Guy Hefer, Chief Financial Officer
Guy Hefer has served as our Chief Financial Officer since October 2020. He has over ten years of experience in investment banking and corporate finance roles. Most recently he was the CFO of Prytek Holdings, a private holding group investing in technology companies globally. Prior to that, Mr. Hefer was an investment banker at Leumi Partners between 2018 and 2019 and GCA investment banking between 2017 and 2018 in Israel and at Barclays investment banking division between 2011 and 2016 in the UK and in Israel. Prior to that Guy worked at Grant Thornton Accounting firm in Israel between 2009 and 2011. Mr. Hefer holds a B.A. degree in Accounting and Economics from the Tel Aviv University, Israel.
Dan Kozlovski, Chief Technologies Officer
Dan Kozlovski has served as our Chief Technologies Officer since February 2022, having previously served as our Vice President of Research & Development from August 2020 after joining us in December 2019. He specializes in R&D and product development, with expertise in three-dimensional computer-aided design. Mr. Kozlovski has more than ten years of experience working in high-technology companies in the printing market. Previously, he served as Future Platform R&D Mechanical Engineer at HP Indigo Division from June 2018 to December 2019. Mr. Kozlovski has also worked as Mechanical Team Leader at Nano Dimension from August 2015 to June 2018. Mr. Kozlovski holds a B.Sc. degree in Mechanical Engineering from Ben Gurion University of the Negev and an Executive MBA in Technology, Innovation & Entrepreneurship Management from Tel Aviv University.
Non-Executive Directors
Yaron Kaiser, Chairman of the Board of Directors
Yaron Kaiser has founded various Nasdaq- or TASE-traded foodtech companies, and currently serves as Chairperson of Wilk Technologies Ltd. Mr. Kaiser is a founding partner of the BlueSoundWaves collective, and practices law in the fields of securities, commercial and corporate law, representing numerous public companies on fundraising, IPOs, M&A, Israel Securities Authority and corporate governance. He holds an LL.B. degree from the College of Management Academic Studies, Israel.
Eli Arad, Director
Eli Arad has served as a director since February 2018. Mr. Arad has been CEO of real-estate and life science investors Merchavia Holdings and Investments (TASE:MRHL) since 2010. Mr. Arad has served as a director of Cleveland Diagnostics, a clinical-stage biotechnology company developing technology to improve cancer diagnostics since 2016, as well as B.G.I. Investments (1961) (TASE:BGI), since 2016. He has had leadership roles in many biomedical startup companies, and has extensive experience in all areas of financial management. Mr. Arad is a certified practicing accountant who holds a B.A. in Accounting from Ramat Gan College and an M.B.A. from the Ruppin Academic Center.
David Gerbi, Director
David Gerbi has served as a director since August 2019. Mr. Gerbi is managing partner of accounting firm Gerbi & Co. and of consulting firm Do Finance Consulting, and serves as Chief Financial Officer of B.G.I. Investments (1961) (TASE:BGI). Mr. Gerbi holds a B.A. in Business Administration and Accounting from the Israeli College of Management Academic Studies and an M.B.A. in Finance from Tel Aviv University.
Sari Singer, Director
Sari Singer has served as a director since March 2021. Ms. Singer serves as General Counsel and Executive Vice President at Delek Drilling LP, the oil and gas arm of the Delek Group in Israel, and a partner in the Tamar and Leviathan offshore gas fields, as well as other petroleum assets offshore Israel and Cyprus, where she has led significant strategic processes, including restructurings and complex financing rounds totaling some $7 billion in various transactions in the international and domestic markets. Ms. Singer holds an LL.B. (cum laude) from Tel Aviv University and has been a member of the Israel Bar since 2007.
Family Relationships
There are no family relationships among any of our directors or officers.
B. Compensation
Aggregate Compensation of Office Holders
The aggregate compensation we paid to our executive officers and directors for the year ended December 31, 2021, was approximately $1.2 million. This amount includes approximately $0.2 million paid, set aside or accrued to provide pension, severance, retirement or similar benefits or expenses, but does not include share-based compensation expenses, or business travel, professional and business association dues and expenses reimbursed to office holders, and other benefits commonly reimbursed or paid by companies in our industry. As of the date of this annual report, options to purchase 2,639,200 ordinary shares granted to our officers and directors were outstanding under our share option plan at a weighted average exercise price of $0.71 per share, in addition to 287,600 restricted share units.
Individual Compensation of Office Holders
The table and summary below outlines the compensation granted to our Chief Executive Officer and Chief Technology Officer, the Chairman of our board of directors, our Deputy Chief Executive Officer, our Chief Financial Officer and our then-Vice President of Research and Development (now Chief Technologies Officer), with respect to the year ended December 31, 2021. For purposes of the table and the summary below, “compensation” includes base salary, bonuses, equity-based compensation, retirement or termination payments, benefits and perquisites such as car, phone and social benefits and any undertaking to provide such compensation.
| Name and Principal Position | | Salary(1) | | | Bonus(2) | | | Equity-Based
Compensation(3) | | | Other Compensation(4) | | | Total | |
| | | (USD in thousands) | |
| Mr. Steven H. Lavin | | | | | | | | | | | | | | | |
Chairman of the Board of Directors(5) | | $ | 180 | | | $ | - | | | $ | 281 | | | | - | | | $ | 461 | |
| Mr. Sharon Fima | | | | | | | | | | | | | | | | | | | | |
Chief Executive Officer & Chief Technology Officer(6) | | | 240 | | | | - | | | | 83 | | | | - | | | | 323 | |
| Mr. Omri Schanin | | | | | | | | | | | | | | | | | | | | |
| Deputy Chief Executive Officer | | | 190 | | | | 46 | | | | 121 | | | | - | | | | 357 | |
| Mr. Guy Hefer | | | | | | | | | | | | | | | | | | | | |
| Chief Financial Officer | | | 193 | | | | 39 | | | | 116 | | | | - | | | | 348 | |
| Mr. Dan Kozlovski | | | | | | | | | | | | | | | | | | | | |
| Vice-President, Research & Development (later Chief Technologies Officer) | | $ | 170 | | | $ | 48 | | | $ | 24 | | | | - | | | $ | 242 | |
| (1) | Salary includes the officer’s gross salary plus payment by us of social benefits on behalf of the officer. Such benefits may include payments, contributions and/or allocations for savings funds (e.g., Managers’ Life Insurance Policy), pension, severance, risk insurance (e.g., life, or work disability insurance), payments for social security and tax gross-up payments, vacation, medical insurance and benefits, convalescence or recreation pay and other benefits and perquisites consistent with our policies. |
| (2) | Represents annual bonuses paid with respect to 2021. |
| (3) | Represents the equity-based compensation expenses recorded in our consolidated financial statements for the year ended December 31, 2021, based on the options’ fair value on the grant date, calculated in accordance with applicable accounting guidance for equity-based compensation. For a discussion of the assumptions used in reaching this valuation, see Note 10(B) to our annual consolidated financial statements included in this Annual Report on Form 20-F. |
| (4) | Represents benefits and perquisites such as car, phone and social benefits. |
| (5) | Mr. Levin resigned his position as Chairman on January 24, 2022. |
| (6) | Mr. Fima resigned as Chief Executive Officer & Chief Technology Officer on January 24, 2022. |
Employment Agreements and Director Fees
We have entered into written employment agreements with each of our executive officers, which provide for notice periods of varying duration for termination of the agreement by us or by the relevant executive officer, during which time the executive officer will continue to receive base salary and benefits. These agreements also contain customary provisions regarding noncompetition, confidentiality of information and assignment of inventions. However, the enforceability of the noncompetition provisions may be limited under applicable law. See “Item 3. — Key Information — Risk Factors — Risks relating to our operations — Under applicable employment laws, we may not be able to enforce covenants not to compete” for a further description of the enforceability of non-competition clauses.
The material employment terms for Mr. Kaufman, our Chief Executive Officer, are as follows: (1) a gross annual salary of NIS 564,000 ($176,000); (2) reimbursement of annual travel expenses of up to NIS 60,000 ($19,000); (3) options to purchase 500,000 ordinary shares (currently equivalent to 50,000 ADSs), vesting over three years from the date of his appointment as Chief Executive Officer, pursuant to which 1/12 will vest every quarter until fully vested, expiring one year following Mr. Kaufman’s cessation of service in all then-applicable capacities, but in any case after four years, with an exercise price of $0.519 per ordinary share (currently equivalent to $5.19 per ADS) and subject to acceleration upon termination pursuant to our sale or change in control; (4) an annual performance bonus in the aggregate amount of NIS 282,000 ($87,000), subject to his meeting certain performance milestones as determined by our Board of Directors on an annual basis; (5) termination of the employment relationship upon provision of six months’ advance notice by either party; (6) severance pay equal to 25% of the gross annual salary upon termination of Mr. Kaufman’s employment by us, not for cause, following three to twelve months of service, or 50% following twelve or more months of service (or 50% of these amounts upon Mr. Kaufman’s resignation); and (7) social benefits that we pay on behalf of officers, such as payments, contributions and/or allocations for savings funds (e.g., Managers’ Life Insurance Policy), pension, severance, risk insurance (e.g., life, or work disability insurance), payments for social security and tax gross-up payments, vacation, medical insurance and benefits, convalescence or recreation pay and other benefits and perquisites consistent with our policies, such as inclusion in our directors’ and officers’ liability insurance policy, and provision of indemnification, exculpation and exemption undertakings to the fullest extent permitted by the Companies Law.
The material terms for Mr. Kaiser, the Chairman of our Board of Directors, are as follows: (1) an annual fee of $150,000, to be paid in four equal quarterly installments in USD or in NIS at the then-current exchange rate, which will automatically increase by an amount equal to seven percent at the end of each year of service; (2) reimbursement of annual travel expenses of up to $18,000; (3) options to purchase 350,000 ordinary shares (currently equivalent to 35,000 ADSs), vesting over three years from the date of his appointment as Chairman, pursuant to which 1/12 will vest every quarter until fully vested, expiring one year following Mr. Kaiser’s cessation of service in all then-applicable capacities, but in any case after four years, with an exercise price of $0.519 per ordinary share (currently equivalent to $5.19 per ADS) and subject to acceleration upon termination pursuant to our sale or change in control; (4) an annual bonus equal to 50% of the bonus awarded to the Chief Executive Officer in the applicable year; (5) severance pay equal to 12.5% of Mr. Kaiser’s annual fee upon the involuntary termination of his directorship, not for cause, following three to twelve months of service, or 25% following twelve or more months of service (or 50% of these amounts upon Mr. Kaiser’s resignation); and (6) other benefits and perquisites consistent with our policies, such as inclusion in our directors’ and officers’ liability insurance policy, and provision of indemnification, exculpation and exemption undertakings to the fullest extent permitted by the Companies Law.
In addition, we pay fees to our non-executive directors in return for their service on our board of directors, in accordance with our compensation policy.
Our other employees are employed under the terms prescribed in their respective employment contracts. The employees are entitled to the social benefits prescribed by law and as otherwise provided in their agreements. These agreements each contain provisions standard for a company in our industry regarding non-competition, confidentiality of information and assignment of inventions. Under currently applicable labor laws, we may not be able to enforce covenants not to compete and therefore may be unable to prevent our competitors from benefiting from the expertise of some of our former employees. See “Item 3.—Key Information—Risk Factors—Risks Related to Our Operations” for a further description of the enforceability of non-competition clauses.
Executive officers are also employed on the terms and conditions prescribed in employment agreements. These agreements provide for notice periods of varying duration for termination of the agreement by us or by the relevant executive officer, during which time the executive officer will continue to receive base salary and benefits. See “Item 3.—Key Information—Risk Factors—Risks Related to Our Operations—If we are unable to attract and retain qualified employees, our ability to implement our business plan may be adversely affected.”
Equity Incentive Plan
In June 2018, the board of directors of Ophectra adopted our Option and RSU Allocation Plan, as amended, or the share option plan, to issue options to purchase our ordinary shares and restricted stock units to our directors, officers, employees and consultants, and those of our affiliated companies (as such term is defined under share option plan), or the Grantees. The share option plan is administered by our Board or a committee that was designated by the Board for such purpose, or the Administrator.
Under the share option plan, we may grant options to purchase ordinary shares and/or RSUs, or options, under four tracks: (i) Approved 102 capital gains options through a trustee, which was approved by the Israeli Tax Authority in accordance with Section 102(a) of the Israeli Income Tax Ordinance (New Version), 1961, or ITO, and granted under the tax track set forth in Section 102(b)(2) of the ITO. The holding period under this tax track is 24 months from the date of issuance of options to the trustee or such period as may be determined in any amendment of Section 102 of the ITO, or any applicable tax ruling or guidelines; (ii) Approved 102 earned income options through a trustee, granted under the tax track set forth is Section 102(b)(1) of the ITO. The holding period under this tax track is 12 months from the date of issuance of options to the trustee or such period as may be determined in any amendment of Section 102 of the ITO; (iii) Unapproved 102 options (the options will not be issued through a trustee and will not be subject to a holding period); and (iv) 3(i) options (the options will not be subject to a holding period). These options shall be subject to taxation pursuant to Section 3(i) of the ITO, or Section 3(i).
Options pursuant to the first three tax tracks (under Section 102 of the ITO) can be granted to our employees and directors and the grant of options under Section 3(i) can be granted to our consultants and controlling shareholders (a controlling shareholder is defined under the Section 102 of the ITO is a person who holds, directly or indirectly, alone or together with a “relative,” (i) the right to at least 10% of the company’s issued capital or 10% of the voting power; (ii) the right to hold at least 10% of the company’s issued capital or 10% of the voting power, or the right to purchase such rights; (iii) the right to receive at least 10% of the company’s profits; or (iv) the right to appoint a company’s director). Grantees who are not Israeli residents may be granted options that are subject to the applicable tax laws in their respective jurisdictions.
We determine, in our sole discretion, under which of the first three tax tracks above the options are granted and we notify the Grantee in a grant letter, as to the elected tax track. As mentioned above, consultants and controlling shareholders can only be granted Section 3(i) options.
The number of ordinary shares authorized to be issued under the share option plan will be proportionately adjusted for any increase or decrease in the number of ordinary shares issued as a result of a distribution of bonus shares, change in our capitalization (split, combination, reclassification of the shares or other capital change), or issuance of rights to purchase ordinary shares or payment of a dividend. We will not issue fractions of ordinary shares and the number of ordinary shares shall be rounded up to the closest number of ordinary shares.
In the event of a (i) merger or consolidation in which we (in this context, specifically MeaTech 3D Ltd.) are not the surviving entity or pursuant to which the other company becomes our parent company or that pursuant to which we are the surviving company but another entity holds 50% or more of our voting rights, (ii) an acquisition of all or substantially all of our ordinary shares, (iii) the sale of all or substantially all of our assets, or (iv) any other event with a similar impact, we may exchange all of our outstanding options granted under the share option plan that remain unexercised prior to any such transaction for options to purchase shares of the successor corporation (or those of an affiliated company) following the consummation of such transaction.
The exercise price of an option granted under the share option plan will be specified in the grant letter every Grantee received from us in which the Grantee notifies of the decision to grant him/her options under the share option plan, and will be denominated in our functional currency at the time of grant or the currency in which the Grantee is paid, at our discretion.
The Administrator may, in its absolute discretion, accelerate the time at which options granted under the share option plan or any portion of which will vest.
Unless otherwise determined by the Administrator, in the event that the Grantee’s employment was terminated, not for Cause (as defined in the share option plan), the Grantee may exercise that portion of the options that had vested as of the date of such termination until the end of the specified term in the grant letter or the share option plan. The portion of the options that had not vested at such date, will be forfeited and can be re-granted to other Grantees, in accordance with the terms of the share option plan.
At the discretion of our Board of Directors, and subject to receipt of taxation authority approvals, we may allow Grantees to exercise their options on a cashless basis.
As of December 31, 2021, our Board of Directors had approved the issuance, under our incentive plans, of options and RSUs to purchase 9,444,145 ordinary shares at an average exercise price of $0.78 per share.
C. Board Practices
Board of Directors
Our board of directors consists of four directors, three of whom are deemed independent directors under the corporate governance standards of the Nasdaq Marketplace Rules and the independence requirements of Rule 10A-3 of the Exchange Act, as well as the standards of the Companies Law.
Under our articles of association, our board of directors must consist of no less than three and no more than 11 directors. Pursuant to our articles of association, the vote required to appoint a director is a simple majority vote of holders of our voting shares participating and voting at the relevant meeting.
In addition, our articles of association allow our board of directors to appoint new directors to fill vacancies which can occur for any reason or as additional directors, provided that the number of board members shall not exceed the maximum number of directors mentioned above. The appointment of a director by the board shall be in effect until the following annual general meeting of the shareholders or until the end of his tenure in accordance with our articles of association. Our board of directors may continue to operate for as long as the number of directors is no less than the minimum number of directors mentioned above.
In addition, under the Companies Law, our board of directors must determine the minimum number of directors who are required to have financial and accounting expertise. Under applicable regulations, a director with financial and accounting expertise is a director who, by reason of his or her education, professional experience and skill, has a high level of proficiency in and understanding of business accounting matters and financial statements. See “— External directors — Qualifications of external directors.” He or she must be able to thoroughly comprehend the financial statements of the company and initiate discussion regarding the manner in which financial information is presented. In determining the number of directors required to have such expertise, the board of directors must consider, among other things, the type and size of the company and the scope and complexity of its operations. Our board of directors has determined that we require at least one director with the requisite financial and accounting expertise and that Eli Arad and David Gerbi have such expertise.
Alternate Directors
Our articles of association provide, as allowed by the Companies Law, that any director may, by written notice to us, appoint another person who is qualified to serve as a director to serve as an alternate director. An alternate director has the same rights and responsibilities as a director, except for the right to appoint an alternate director. The appointment of an alternate director does not negate the responsibilities of the appointing director, who will continue to bear responsibility for the actions of the alternate, giving consideration to the circumstances of the appointment. The Companies Law specifies certain qualifications for alternate directors, and provides that one director may not serve as an alternate on the board of directors for another director, nor as an alternate on a committee of which he or she is already a member. As of the date of this Annual Report on Form 20-F, no director has appointed any other person as an alternate director.
External Directors
The Companies Law requires a public Israeli company to have at least two external directors who meet certain independence criteria to ensure that they are unaffiliated with the company and its controlling shareholder. An external director must have either financial and accounting expertise or professional qualifications, as defined in the regulations promulgated under the Companies Law, and at least one of the external directors is required to have financial and accounting expertise. An external director is entitled to reimbursement of expenses and compensation as provided in the regulations promulgated under the Companies Law, but is otherwise prohibited from receiving any other compensation from the company, directly or indirectly, during his or her term and for two years thereafter.
Pursuant to regulations promulgated under the Companies Law, as a company with shares traded on Nasdaq, we have elected no to comply with the requirements to appoint external directors and related rules concerning the composition of the audit committee and compensation committee of the board of directors. We are still subject to the gender diversity rule under the Companies Law, which requires that if, at the time a director is to be elected or appointed, all members of the board of directors are of the same gender, the director to be appointed must be of the other gender. The conditions to the exemptions from the Companies Law requirements are that: (i) the company does not have a “controlling shareholder,” as such term is defined under the Companies Law, (ii) its shares are traded on certain U.S. stock exchanges, including Nasdaq, and (iii) it comply with the director independence requirements and the audit committee and compensation committee composition requirements under U.S. laws, including the rules of the applicable exchange, that are applicable to U.S. domestic issuers.
Committees of the Board of Directors
Our board of directors has established the following committees. Each committee operates in accordance with a written charter that sets forth the committee’s structure, operations, membership requirements, responsibilities and authority to engage advisors.
Audit Committee
Under the Companies Law, the Exchange Act and Nasdaq Marketplace Rules, we are required to maintain an audit committee.
The responsibilities of an audit committee under the Companies Law include identifying and addressing flaws in the business management of the company, reviewing and approving related party transactions, establishing whistleblower procedures, overseeing the company’s internal audit system and the performance of its internal auditor, and assessing the scope of the work and recommending the fees of the company’s independent accounting firm. In addition, the audit committee is required to determine whether certain related party actions and transactions are “material” or “extraordinary” for the purpose of the requisite approval procedures under the Companies Law and to establish procedures for considering proposed transactions with a controlling shareholder.
In accordance with U.S. law and Nasdaq Marketplace Rules, our audit committee is also responsible for the appointment, compensation and oversight of the work of our independent auditors and for assisting our board of directors in monitoring our financial statements, the effectiveness of our internal controls and our compliance with legal and regulatory requirements.
Under the Companies Law, the audit committee must consist of at least three directors who meet certain independence criteria. Under the Nasdaq Marketplace Rules, we are required to maintain an audit committee consisting of at least three independent directors, all of whom are financially literate and one of whom has accounting or related financial management expertise. Each of the members of the audit committee is required to be “independent” as such term is defined in Rule 10A-3(b)(1) under the Exchange Act.
Our audit committee currently consists of Eli Arad, Sari Singer and David Gerbi. All members are independent directors as defined in the Companies Law, SEC rules and Nasdaq listing requirements. Our board of directors has determined that all members of our audit committee meet the requirements for financial literacy under the applicable rules and regulations of the SEC and Nasdaq Marketplace Rules. Our board of directors has determined that Eli Arad and David Gerbi are audit committee financial experts as defined by the SEC rules and have the requisite financial experience as defined by the Nasdaq Marketplace Rules.
Compensation Committee
Under both the Companies Law and Nasdaq Marketplace Rules, we are required to establish a compensation committee.
The responsibilities of a compensation committee under the Companies Law include recommending to the board of directors, for ultimate shareholder approval by a special majority, a policy governing the compensation of directors and officers based on specified criteria, reviewing modifications to and implementing such compensation policy from time to time, and approving the actual compensation terms of directors and officers prior to approval by the board of directors.
The Companies Law stipulates that the compensation committee must consist of at least three directors who meet certain independence criteria. Under Nasdaq Marketplace Rules, we are required to maintain a compensation committee consisting of at least two independent directors; each of the members of the compensation committee is required to be independent under Nasdaq Marketplace Rules relating to compensation committee members, which are different from the general test for independence of board and committee members.
Our compensation committee currently consists of Eli Arad, Sari Singer and David Gerbi. All members are independent directors as defined in the Companies Law, SEC rules and regulations, and Nasdaq Marketplace Rules.
Director Nominations
We do not have a standing nominating committee. In accordance with Rule 5605(e)(2) of the Nasdaq Rules, a majority of the independent directors may recommend a director nominee for selection by the board of directors. Our board of directors believes that the independent directors can satisfactorily carry out the responsibility of properly selecting or approving director nominees without the formation of a standing nominating committee. As we do not have a standing nominating committee, we will not have a nominating committee charter in place.
Our board of directors will consider candidates for nomination who have a high level of personal and professional integrity, strong ethics and values and the ability to make mature business judgments. In general, in identifying and evaluating nominees for director, our board of directors will also consider experience in corporate management such as serving as an officer or former officer of a publicly held company, experience as a board member of another publicly held company, professional and academic experience relevant to our business, leadership skills, experience in finance and accounting or executive compensation practices, whether candidate has the time required for preparation, participation and attendance at board meetings and committee meetings, if applicable, independence and the ability to represent the best interests of our stockholders.
Internal Auditor
Under the Companies Law, the board of directors is required to appoint an internal auditor recommended by the audit committee. The role of the internal auditor is to examine, among other things, whether the company’s actions comply with applicable law and proper business procedures. The internal auditor may not be an interested party, a director or an officer of the company, or a relative of any of the foregoing, nor may the internal auditor be our independent accountant or a representative thereof. Our current internal auditor is Mr. Daniel Spira, CPA, who is a member of the board of directors of the Institute of Internal Auditors in Israel and Chairman of its Auditing and Knesset Relations Committee.
Fiduciary Duties and Approval of Related Party Transactions
Fiduciary duties of directors and officers
Israeli law imposes a duty of care and a duty of loyalty on all directors and officers of a company. The duty of care requires a director or officer to act with the level of care with which a reasonable director or officer in the same position would have acted under the same circumstances. The duty of care includes, among other things, a duty to use reasonable means, under the circumstances, to obtain information on the advisability of a given action brought for his approval or performed by virtue of his position and other important information pertaining to such action. The duty of loyalty requires the director or officer to act in good faith and for the benefit of the company.
Disclosure of Personal Interests of an Office Holder and Approval of Certain Transactions
Under the Companies Law, a company may approve an act specified above which would otherwise constitute a breach of the office holder’s fiduciary duty, provided that the office holder acted in good faith, the act or its approval does not harm the company, and the office holder discloses to the company his or her personal interest in the transaction (including any significant fact or document) a reasonable time before the approval of such act. Any such approval is subject to the terms of the Companies Law, setting forth, among other things, the appropriate bodies of the company required to provide such approval, and the methods of obtaining such approval.
The Companies Law requires that an office holder promptly disclose to the company any direct or indirect personal interest that he or she may have and all related material information or documents known to him or her relating to any existing or proposed transaction by the company. An interested office holder’s disclosure must be made promptly and, in any event, no later than the first meeting of the board of directors at which the transaction is considered. An office holder is not obliged to disclose such information if the personal interest of the office holder derives solely from the personal interest of his or her relative in a transaction that is not considered an extraordinary transaction.
If the transaction is an extraordinary transaction, the office holder must also disclose any personal interest held by:
| • | the office holder’s relatives (spouse, siblings, parents, grandparents, descendants, spouse’s descendants and the spouses of any of these people); or |
| • | any company in which the office holder or his or her relatives holds 5% or more of the shares or voting rights, serves as a director or general manager or has the right to appoint at least one director or the general manager. |
Under the Companies Law, unless the articles of association of a company provide otherwise, a transaction with an office holder or with a third party in which the office holder has a personal interest, which is not an extraordinary transaction, requires approval by the board of directors or a committee authorized by the board of directors. If the transaction considered is an extraordinary transaction with an office holder or third party in which the office holder has a personal interest, then audit committee approval is required prior to approval by the board of directors. Under specific circumstances, shareholder approval may also be required. For the approval of compensation arrangements with directors and executive officers, see “Item 6.B. Compensation—Compensation of Directors and Executive Officers.”
Any persons who have a personal interest in the approval of a transaction that is brought before a meeting of the board of directors or the audit committee may not be present at the meeting or vote on the matter. However, if the chairman of the board of directors or the chairman of the audit committee, as applicable, has determined that the presence of an office holder with a personal interest is required, such office holder may be present at the meeting for the purpose of presenting the matter. Notwithstanding the foregoing, a director who has a personal interest may be present at the meeting of the board of directors or the audit committee (as applicable) and vote on the matter if a majority of the members of the board of directors or the audit committee (as applicable) have a personal interest in the approval of such transaction. If a majority of the directors at a board of directors meeting have a personal interest in the transaction, such transaction also generally requires approval of the shareholders of the company.
A “personal interest” is defined under the Companies Law as the personal interest of a person in an action or in a transaction of the company, including the personal interest of such person’s relative or the interest of any other corporate body in which the person and/or such person’s relative is a director or general manager, a 5% shareholder or holds 5% or more of the issued and outstanding share capital of the company or of its voting rights, or has the right to appoint at least one director or the general manager, but excluding a personal interest stemming solely from the fact of holding shares in the company. A personal interest also includes (i) a personal interest of a person who votes according to a proxy of another person, including in the event that the other person has no personal interest, and (ii) a personal interest of a person who gave a proxy to another person to vote on his or her behalf regardless of whether the discretion of how to vote lies with the person voting.
An “extraordinary transaction” is defined under the Companies Law as any of the following:
| • | a transaction other than in the ordinary course of business; |
| • | a transaction that is not on market terms; or |
| • | a transaction that may have a material impact on the company’s profitability, assets or liabilities. |
Disclosure of Personal Interests of a Controlling Shareholder and Approval of Transactions
Pursuant to the Companies Law, the disclosure requirements that apply to an office holder also apply to a controlling shareholder of a public company. Extraordinary transactions with a controlling shareholder or in which a controlling shareholder has a personal interest, including a private placement in which a controlling shareholder has a personal interest, and the terms of engagement of the company, directly or indirectly, with a controlling shareholder or a controlling shareholder’s relative (including through a corporation controlled by a controlling shareholder), regarding the company’s receipt of services from the controlling shareholder, and if such controlling shareholder is also an office holder or employee of the company, regarding his or her terms of employment, require the approval of each of (i) the audit committee (or the compensation committee with respect to the terms of the engagement as an office holder or employee, including insurance, indemnification and compensation), (ii) the board of directors and (iii) the shareholders, in that order. In addition, the shareholder approval must fulfill one of the following requirements:
| • | a majority of the shares held by shareholders who have no personal interest in the transaction and are voting at the meeting must be voted in favor of approving the transaction, excluding abstentions; or |
| • | the shares voted by shareholders who have no personal interest in the transaction who vote against the transaction represent no more than 2% of the voting rights in the company. |
Such majority determined in accordance with the majority requirement described above is hereinafter referred to as the Compensation Special Majority Requirement.
Any such transaction for which the term is more than three years must be approved in the same manner every three years, unless with respect to certain transactions as permitted by the Companies Law, the audit committee has determined that a longer term is reasonable under the circumstances. In addition, transactions with a controlling shareholder or a controlling shareholder’s relative who serves as an executive officer in a company, directly or indirectly (including through a corporation under his control), involving the receipt of services by a company or their compensation can have a term of five years from the company’s initial public offering under certain circumstances.
The Companies Law requires that every shareholder that participates, in person or by proxy, in a vote regarding a transaction with a controlling shareholder, must indicate in advance or in the ballot whether or not that shareholder has a personal interest in the vote in question. Failure to so indicate generally results in the invalidation of that shareholder’s vote.
Disclosure of Compensation of Executive Officers
For so long as we qualify as a foreign private issuer, we are not required to comply with the proxy rules applicable to U.S. domestic filers, including the requirement applicable to emerging growth companies to disclose the compensation of our chief executive officer and other two most highly compensated executive officers on an individual, rather than an aggregate, basis. Nevertheless, regulations promulgated under the Companies Law require us to disclose in the proxy statement for the annual general meeting of our shareholders (or to include a reference therein to other previously furnished public disclosure) the annual compensation of our five most highly compensated executive officers on an individual, rather than an aggregate, basis. This disclosure will not be as extensive as that required of a U.S. domestic issuer.
Compensation of Directors and Executive Officers
Directors. Under the Companies Law, the compensation of our directors requires the approval of our compensation committee, the subsequent approval of the board of directors and, unless exempted under regulations promulgated under the Companies Law, the approval of the shareholders at a general meeting. If the compensation of our directors is inconsistent with our compensation policy, then, provided that those provisions that must be included in the compensation policy according to the Companies Law have been considered by the compensation committee and board of directors, and provided that shareholder approval is obtained by the Compensation Special Majority Requirement.
Executive Officers (other than the Chief Executive Officer). The Companies Law requires the approval of the compensation of a public company’s executive officers (other than the chief executive officer) in the following order: (i) the compensation committee, (ii) the company’s board of directors and (iii) if such compensation arrangement is inconsistent with the company’s compensation policy, the company’s shareholders (the Compensation Special Majority Requirement). However, if the shareholders of the company do not approve a compensation arrangement with an executive officer that is inconsistent with the company’s compensation policy, the compensation committee and board of directors may override the shareholders’ decision if each of the compensation committee and the board of directors provide detailed reasons for their decision.
Chief Executive Officer. The Companies Law requires the approval of the compensation of a public company’s chief executive officer in the following order: (i) the company’s compensation committee, (ii) the company’s board of directors and (iii) the company’s shareholders (the Compensation Special Majority Requirement). However, if the shareholders of the company do not approve the compensation arrangement with the chief executive officer, the compensation committee and board of directors may override the shareholders’ decision if each of the compensation committee and the board of directors provide a detailed report for their decision. The approval of each of the compensation committee and the board of directors should be in accordance with the company’s compensation policy; however, in special circumstances, they may approve compensation terms of a chief executive officer that are inconsistent with such policy provided that they have considered those provisions that must be included in the compensation policy according to the Companies Law and that shareholder approval was obtained (by a special majority vote as discussed above with respect to the approval of director compensation). In addition, the compensation committee may waive the shareholder approval requirement with regards to the approval of the engagement terms of a candidate for the chief executive officer position, if the compensation committee determines that the compensation arrangement is consistent with the company’s compensation policy, and that the chief executive officer did not have a prior business relationship with the company or a controlling shareholder of the company and that subjecting the approval of the engagement to a shareholder vote would impede the company’s ability to employ the chief executive officer candidate.
Compensation Policy
Under the Companies Law, we are required to approve, at least once every three years, a compensation policy with respect to our directors and officers. Following the recommendation of our compensation committee, the compensation policy must be approved by our board of directors and our shareholders. The shareholder approval must be by a simple majority of all votes cast, provided that (i) such majority includes a simple majority of the votes cast by non-controlling shareholders having no personal interest in the matter or (ii) the total number of votes of shareholders mentioned in clause (i) above who voted against such transaction does not exceed 2% of the total voting rights in the company.
Directors’ Service Contracts
There are no arrangements or understandings between us and any of our subsidiaries, on the one hand, and any of our directors, on the other hand, providing for benefits upon termination of their employment or service as directors of our company or any of our subsidiaries.
Under the Companies Law, a shareholder has a duty to refrain from abusing its power in the company and to act in good faith and in an acceptable manner in exercising its rights and performing its obligations to the company and other shareholders, including, among other things, when voting at meetings of shareholders on the following matters:
| • | an amendment to the articles of association; |
| • | an increase in the company’s authorized share capital; |
| • | the approval of related party transactions and acts of office holders that require shareholder approval. |
A shareholder also has a general duty to refrain from discriminating against other shareholders.
The remedies generally available upon a breach of contract also apply to a breach of the shareholder duties mentioned above, and in the event of discrimination against other shareholders, additional remedies may be available to the injured shareholder.
In addition, any controlling shareholder, any shareholder that knows that its vote can determine the outcome of a shareholder vote and any shareholder that, under a company’s articles of association, has the power to appoint or prevent the appointment of an office holder, or any other power with respect to a company, is under a duty to act with fairness towards the company. The Companies Law does not describe the substance of this duty except to state that the remedies generally available upon a breach of contract also apply in the event of a breach of the duty to act with fairness, taking the shareholder’s position in the company into account.
Exculpation, Insurance and Indemnification of Directors and Officers
Under the Companies Law, a company may not exculpate an office holder from liability for a breach of the duty of loyalty. An Israeli company may exculpate an office holder in advance from liability to the company, in whole or in part, for damages caused to the company as a result of a breach of duty of care but only if a provision authorizing such exculpation is included in its articles of association. Our articles of association include such a provision. The company may not exculpate in advance a director from liability arising from a breach of his or her duty of care in connection with a prohibited dividend or distribution to shareholders.
As permitted under the Companies Law, our articles of association provide that we may indemnify an office holder in respect of the following liabilities, payments and expenses incurred for acts performed by him or her as an office holder, either in advance of an event or following an event:
| • | financial liability that was imposed upon him in favor of another person pursuant to a judgment, including a compromise judgment or an arbitrator’s award approved by a court; |
| • | reasonable litigation expenses, including attorneys’ fees paid by an officeholder following an investigation or proceeding conducted against him by an authority authorized to conduct such investigation or proceeding, and which ended without the filing of an indictment against him and without any financial obligation being imposed on him as an alternative to a criminal proceeding, or which ended without the filing of an indictment against him but with the imposition of a financial obligation as an alternative to a criminal proceeding for an offense which does not require proof of mens rea or in connection with a financial sanction; |
| • | reasonable litigation expenses, including attorneys’ fees paid by the officeholder or which he was required to pay by a court, in a proceeding filed against him by the Company or on its behalf or by another person, or in criminal charges from which he was acquitted, or in criminal charges in which he was convicted of an offense which does not require proof of mens rea; |
| • | a financial obligation imposed on the officeholder for the benefit of all of the parties damaged by the violation of an administrative proceeding; |
| • | expenses incurred by an officeholder in connection with an Administrative Proceeding conducted in his regard, including reasonable litigation expenses, and including attorneys’ fees; |
| • | expenses incurred by an officeholder in connection with a proceeding under the Antitrust Law, 5748-1988 and/or in connection with it (a “Proceeding Under the Antitrust Law”), conducted regarding him, including reasonable litigation expenses, and attorneys' fees; and |
| • | any other liability or expense in respect of which it is permitted or shall be permitted by Law to indemnify an officeholder. |
As permitted under the Israeli Companies Law, our articles of association provide that we may insure an office holder against the following liabilities incurred for acts performed by him or her as an office holder:
| • | Breach of the duty of care to the Company or to any other person; |
| • | Breach of the fiduciary duty to the Company, provided that the officeholder acted in good faith and had reasonable grounds to assume that his act would not adversely affect the Company’s best interests; |
| • | financial liability imposed upon him in favor of another person; |
| • | financial liability imposed on the officeholder for the benefit of all of the parties damaged by the violation of an administrative proceeding; |
| • | expenses incurred or to be incurred by an officer in connection with an Administrative Proceeding, including reasonable litigation expenses, and including attorneys’ fees; |
| • | Expenses incurred or to be incurred in connection with a proceeding under the Antitrust Law, including reasonable litigation expenses, and including attorneys’ fees; and |
| • | any other event in respect of which it is permitted and/or shall be permitted by Law to insure the liability of an officeholder. |
Under the Companies Law, a company may not indemnify, exculpate or insure an office holder against any of the following:
| • | a breach of the duty of loyalty, except for indemnification and insurance for a breach of the duty of loyalty to the company to the extent that the office holder acted in good faith and had a reasonable basis to believe that the act would not prejudice the company; |
| • | a breach of duty of care committed intentionally or recklessly, excluding a breach arising out of the negligent conduct of the office holder; |
| • | an act or omission committed with intent to derive illegal personal benefit; or |
| • | a fine, monetary sanction or forfeit levied against the office holder. |
Under the Companies Law, exculpation, indemnification and insurance of office holders must be approved by the compensation committee and the board of directors and, with respect to directors or controlling shareholders, their relatives and third parties in which controlling shareholders have a personal interest, also by the shareholders.
Our articles of association permit us to exculpate, indemnify and insure our office holders to the fullest extent permitted or to be permitted by law. Our office holders are currently covered by a directors’ and officers’ liability insurance policy. As of the date of this annual report, no claims for directors’ and officers’ liability insurance have been filed under this policy and we are not aware of any pending or threatened litigation or proceeding involving any of our office holders, including our directors, in which indemnification is sought.
D. Employees
As of December 31, 2021, we had 38 employees based at our office and laboratory, then in Ness Ziona, Israel, while our Belgium-based subsidiaries had 14 employees.
Local labor laws govern the length of the workday and workweek, minimum wages for employees, procedures for hiring and dismissing employees, determination of severance pay, annual leave, sick days, advance notice of termination, Social Security payments or regional equivalents, and other conditions of employment and include equal opportunity and anti-discrimination laws. None of our employees is party to any collective bargaining agreements. We generally provide our employees with benefits and working conditions beyond the required minimums. We believe we have a good relationship with our employees, and have never experienced any employment-related work stoppages.
E. Beneficial Ownership of Executive Officers and Directors
The beneficial ownership of our ordinary shares (including ordinary shares represented by ADSs) is determined in accordance with the rules of the SEC. Under these rules, a person is deemed to be a beneficial owner of a security if that person has or shares voting power, which includes the power to vote or to direct the voting of the security, or investment power, which includes the power to dispose of or to direct the disposition of the security. For purposes of the table below, we deem ordinary shares issuable pursuant to options or warrants that are currently exercisable or exercisable within 60 days of the date of this Annual Report on Form 20-F, if any, to be outstanding and to be beneficially owned by the person holding the options or warrants for the purposes of computing the percentage ownership of that person, but we do not treat them as outstanding for the purpose of computing the percentage ownership of any other person.
Unless otherwise noted below, each shareholder’s address is c/o MeaTech 3D Ltd., 5 David Fikes St., Rehovot 7638205, Israel.
| | | Shares Beneficially Owned | |
| Name of Beneficial Owner | | Number | | | Percentage | |
| Directors and executive officers | | | | | | |
Arik Kaufman(1) | | | 129,170 | | | | * | |
Omri Schanin(2) | | | 3,626,900 | | | | 2.9 | % |
Guy Hefer(3) | | | 104,170 | | | | * | |
Dan Kozlovski(4) | | | 83,342 | | | | * | |
Yaron Kaiser(5) | | | 1,454,230 | | | | 1.2 | % |
David Gerbi(6) | | | 12,500 | | | | * | |
Eli Arad(7) | | | 12,500 | | | | * | |
| Sari Singer | | | — | | | | — | |
| | | | | | | | | |
| All directors and executive officers as a group (8 persons) | | | 5,422,812 | | | | 4.3 | % |
* Less than one percent (1%).
| (1) | Consists of 87,510 ordinary shares and options to purchase 41,660 ordinary shares exercisable within 60 days of the date of this annual report, with an exercise price of $0.519. These options expire on March 16, 2026. |
| (2) | Consists of 3,522,730 ordinary shares and options to purchase 104,170 ordinary shares exercisable within 60 days of the date of this annual report, with an exercise price of NIS 3.49 ($1.07). These options expire on March 24, 2025. |
| (3) | Consists of options to purchase 104,170 ordinary shares exercisable within 60 days of the date of this annual report, with an exercise price of NIS 3.49 ($1.07). These options expire on March 24, 2025. |
| (4) | Consists of options to purchase 83,342 ordinary shares exercisable within 60 days of the date of this annual report, with an exercise price of NIS 1.90 ($0.58). These options expire on August 5, 2024. |
| (5) | Consists of 1,425,070 ordinary shares and options to purchase 29,160 ordinary shares exercisable within 60 days of the date of this annual report, with an exercise price of $0.519. These options expire on March 16, 2026. |
| (6) | Consists of 10,000 ordinary shares and RSUs vesting into 2,500 ordinary shares within 60 days of the date of this annual report. |
| (7) | Consists of 10,000 ordinary shares and RSUs vesting into 2,500 ordinary shares within 60 days of the date of this annual report. |
ITEM 7. MAJOR SHAREHOLDERS AND RELATED PARTY TRANSACTIONS
A. Major Shareholders
The following table sets forth certain information regarding the beneficial ownership of our outstanding ordinary shares, including ordinary shares represented by ADSs, as of the date of this Annual Report on Form 20-F, by each person or entity who we know beneficially owns 5% or more of the outstanding ordinary shares. For purposes of the table below, we deem ordinary shares issuable pursuant to options or warrants that are currently exercisable or exercisable within 60 days of the date of this Annual Report on Form 20-F, if any, to be outstanding and to be beneficially owned by the person holding the options or warrants for the purposes of computing the percentage ownership of that person, but we do not treat them as outstanding for the purpose of computing the percentage ownership of any other person. The percentage of shares beneficially owned is based on 126,529,867 ordinary shares outstanding as of the date of this Annual Report on Form 20-F.
None of our shareholders have different voting rights from other shareholders. We are not aware of any arrangement that may, at a subsequent date, result in a change of control of our company.
As of the date of this Annual Report on Form 20-F, there are five shareholders of record of our ordinary shares, of whom two are in the United States. The number of record holders is not representative of the number of beneficial holders of our ordinary shares, as most of the shares we have issued, including those represented by ADSs are currently recorded in the name of our ADS registrar, The Bank of New York Mellon. Based upon a review of the information provided to us by The Bank of New York Mellon, as of March 1, 2022, there were 28 holders of record of the ADSs on record with the Depository Trust Company. These numbers are not representative of the number of beneficial holders of our ADSs nor is it representative of where such beneficial holders reside, since many of these ADSs were held of record by brokers or other nominees.
| | | Ordinary Shares Beneficially Owned | |
| Name of Beneficial Owner | | Number | | | Percentage | |
| 5% or greater shareholders | | | | | | |
| Shimon Cohen | | | 9,859,120 | (1) | | | 7.8 | % |
(1) Based on information provided to the Company on January 20, 2022 by Mr. Cohen regarding his holdings and those of companies through which he claims share ownership.
B. Related Party Transactions
The following is a description of the material transactions we entered into with related parties since the beginning of 2019. We believe that we have executed all of our transactions with related parties on terms no less favorable to us than those we could have obtained from unaffiliated third parties.
Our Board of Directors, acting through our Audit Committee, is responsible for the review, approval, or ratification of related party transactions between us and related persons. Under Israeli law, related party transactions are subject to special approval requirements, see “Management — Fiduciary duties and approval of specified related party transactions and compensation under Israeli law.”
Employment Agreements and Director Fees
We have entered into written employment agreements with each of our executive officers, which provide for notice periods of varying duration for termination of the agreement by us or by the relevant executive officer, during which time the executive officer will continue to receive base salary and benefits. These agreements also contain customary provisions regarding noncompetition, confidentiality of information and assignment of inventions. However, the enforceability of the noncompetition provisions may be limited under applicable law. See “Risk factors - Risks relating to our operations - Under applicable employment laws, we may not be able to enforce covenants not to compete” for a further description of the enforceability of non-competition clauses. For further information, see “Management - Employment and Consulting Agreements.”
Directors and Officers Insurance Policy and Indemnification and Exculpation Agreements
In accordance with our articles of association, we have obtained Directors and Officers insurance for our executive officers and directors, and provide indemnification, exculpation and exemption undertakings to each of our directors and officers to the fullest extent permitted by the Companies Law.
Private Issuances of Securities
In January 2020, following the closing of the merger between MeaTech and Ophectra, we issued former shareholders of MeaTech warrants to receive ordinary shares, including to the following related parties: (1) warrants to receive 1,036,098 ordinary shares each to Sharon Fima, then our Chief Executive Officer and Chief Technical Officer, and Omri Schanin, our Depuy Chief Executive Officer; and (2) warrants to receive 1,291,158 ordinary shares to Liran Damati, then a substantial shareholder. The warrants have no exercise price and vest upon the achievement of certain milestones (for further details, see “Item 4.—Information on the Company—History and Development of the Company”).
In May 2020, pursuant to approvals of our audit committee, board of directors and a general meeting of our shareholders: (1) we issued 1,043,846 ordinary shares and options to purchase 6,030,286 ordinary shares at an exercise price of NIS 3.36 (approximately $1.03) per share in return for a private investment of $750,000 by EL Capital Investments LLC, a company controlled by Mr. Steven Lavin, who was concurrently appointed to our board of directors as its chairman; and (2) we issued options to purchase 1,967,327 ordinary shares at an exercise price of NIS 2.49 (approximately $0.76) per share and options to purchase 1,967,328 ordinary shares at an exercise price of NIS 3.486 (approximately $1.07) to Silver Road Capital Ltd., the majority of whose shares were owned by directors at the time, Mr. Steven Lavin and Mr. Daniel Ayalon.
Engagement with BlueSoundWaves
On October 6, 2021, we entered into a services and collaboration agreement, or the Services and Collaboration Agreement, with BlueOcean Sustainability Fund, LLC, or BlueSoundWaves, pursuant to which BlueSoundWaves provides us with marketing and promotional services, strategic consulting advice, and partner and investor engagement services in the United States. As consideration for such services, BlueSoundWaves received (i) an option to purchase 6,215,770 ordinary shares, currently equal to 621,577 ADSs, and (ii) 1,243,150 of our ordinary restricted shares, currently equal to 124,315 ADSs.
BlueOcean Sustainability Management Fund LP, a Cayman partnership, and the managing partner of BlueSoundWaves holds all of the outstanding share capital of BlueOcean Kayomot Ltd., an Israeli company. Messrs. Kaufman and Kaiser are directors of BlueOcean Kayomot Ltd. and founding partners of BlueSoundWaves. Mr. Kaufman also serves as the chief executive officer of BlueOcean Kayomot Ltd. See Item 10.C for additional discussion of the Services and Collaboration Agreement.
C. Interests of Experts and Counsel
Not applicable.
| | ITEM 8. | FINANCIAL INFORMATION |
A. Consolidated Statements and other Financial Information
See “Item 18.—Financial Statements” in this Annual Report on Form 20-F.
Legal Proceedings
From time to time, we may be party to litigation or other legal proceedings that we consider to be a part of the ordinary course of our business. We are not currently involved in any legal proceedings that could reasonably be expected to have a material adverse effect on our business, prospects, financial condition or results of operations.
In November 2020, the ISA initiated an administrative proceeding claiming negligent misstatement regarding certain immediate and periodic reports published by our predecessor (Ophectra) during the years 2017 and 2018, prior to the merger with MeaTech. These reports relate to Ophectra’s activities prior to establishment of the settlement fund in connection with the merger. In February 2021, the trustee of the settlement fund informed us that the ISA views us as a party to this proceeding, notwithstanding the settlement and establishment of the settlement fund. This proceeding is of an administrative nature and carries a potential penalty in the form of a monetary fine which, under applicable Israeli law, could be as high as NIS 5 million. We were advised that the maximum fine likely to be imposed in this case, if any, is $0.26 million (NIS 0.85 million). In April 2021, following negotiations with the ISA, we agreed to settle the matter for $0.21 million (NIS 0.7 million). The settlement is subject to approval of the ISA’s Enforcement Committee.
In February 2021, a civil claim was lodged for which the settlement fund is a respondent, relating to Ophectra's activities prior to establishment of the settlement fund, in an amount of USD 0.8 million (NIS 2.5 million). We believe that there is a low probability of a final judgment against the settlement fund, relating to Ophectra's activities prior to establishment of the settlement fund, in an amount of $0.75 million (NIS 2.5 million). As described in “Item 4.—Information on the Company—History and Development of the Company” above, we do not bear any liability for such claims beyond the exercise value of the Therapin asset, which was the sole asset we retained from Ophectra in the merger.
Dividend Distributions
We have never declared or paid cash dividends to our shareholders. Currently we do not intend to pay cash dividends. We currently intend to reinvest any future earnings in developing and expanding our business. Any future determination relating to our dividend policy will be at the discretion of our Board of Directors and will depend on a number of factors, including future earnings, our financial condition, operating results, contractual restrictions, capital requirements, business prospects, applicable Israeli law and other factors our Board of Directors may deem relevant.
B. Significant Changes
Since December 31, 2021, the following significant changes have occurred:
Changes in Executive Roles
In January 2022, Mr. Sharon Fima stepped down from the positions of Chief Executive Officer, Chief Technology Officer and later Director, citing the Company’s current stage of development. Messrs. Steven H. Lavin (Chairman) and Danny Ayalon also stepped down from the Board of Directors, citing the Company’s current stage of development and to pursue other ventures, and Mr. Omri Schanin stepped down from the Board of Directors and continues to serve as MeaTech’s Deputy CEO.
The Company’s Board of Directors appointed Mr. Arik Kaufman to the position of Chief Executive Officer and Mr. Yaron Kaiser to the position of Chairman of the Board of Directors.
| | ITEM 9. | THE OFFER AND LISTING |
A. Offer and Listing Details
ADSs
The ADSs, representing our ordinary shares, have been trading on Nasdaq under the symbol “MITC” since March 12, 2021. Prior to that date, there was no public trading market for the ADSs.
Ordinary Shares
Our ordinary shares were traded on the TASE between January 26, 2020 and August 3, 2021, when we voluntarily de-listed them from trade on the TASE. Our ordinary shares were traded under the symbol “MEAT” until March 2021, and thereafter under the symbol “MITC.” Some of our ordinary shares are traded over the counter under the symbol MTTCF.
B. Plan of Distribution
Not applicable.
C. Markets
For a description of our publicly-traded ADSs, see “Item 9.— Offer and Listing Details —ADSs.” For a description of our ordinary shares, see “Item 9.— Offer and Listing Details —Ordinary Shares.”
D. Selling Shareholders
Not applicable.
E. Dilution
Not applicable.
F. Expenses of the Issue
Not applicable.
| | ITEM 10. | ADDITIONAL INFORMATION |
A. Share Capital
Not applicable.
B. Articles of Association
The information set forth in our prospectus dated March 12, 2021, filed with the SEC pursuant to Rule 424(b), under the headings “Description of Share Capital” is incorporated herein by reference.
C. Material Contracts
On October 6, 2021, we entered into the Services and Collaboration Agreement with BlueSoundWaves, a sustainability focused fund led by led by Ashton Kutcher, Guy Oseary and Effie Epstein. Pursuant to the Services and Collaboration Agreement, BlueSoundWaves provides us marketing and promotional services, strategic consulting advice, and partner and investor engagement services in the United States.
As consideration for such services, BlueSoundWaves received (i) an option to purchase 6,215,770 ordinary shares, currently equal to 621,577 ADSs, and (ii) 1,243,150 of our ordinary restricted shares, currently equal to 124,315 ADSs. The exercise price per ordinary share of the ordinary share option and restricted ordinary share option is the greater of (a) the closing price per ADS on October 5, 2021 ($6.65) divided by the number of ordinary shares represented by the ADS and (b) the closing price per ADS on the day prior to the exercise of the options less a discount ranging from 25% to 75% depending on how much higher the exercise price is compared to the price determined under subsection (a) of this paragraph. The options granted pursuant to this agreement will vest over a three-year period, with one-third vesting on the first anniversary of the Services and Collaboration Agreement with the remaining amount vesting in equal quarterly installments for the remaining period. If either party provides notice to terminate the agreement, the quarterly vesting will be cancelled. A percentage of the options described above will immediately vest and be exercisable upon the occurrence of certain trading milestones, investment milestones or change of control event. We have also agreed to reimburse BlueSoundWaves its reasonable out of pocket expenses which have been previously approved.
The Services and Collaboration Agreement will remain in effect until terminated in accordance with its terms. Each party has the right to terminate upon 60 days prior written notice after the 12-month anniversary of the effective date. Either party may also terminate the Services and Collaboration Agreement upon the occurrence of a material breach which remains uncured 30 days after the breaching party has received notice of the breach. Any portion of the vested options to purchase ordinary shares or restricted ordinary shares will expire on the second anniversary of the termination of the Services and Collaboration Agreement, or otherwise expire on the 10th anniversary.
For other agreements with related parties, see “Item 7.—Major Shareholders and Related Party Transactions—Related Party Transactions.”
D. Exchange Controls
Non-residents of Israel who purchase our ordinary shares outside of Israel with U.S. dollars or other foreign currency will be able to convert dividends (if any) thereon, and any amounts payable upon the dissolution, liquidation or winding up of the affairs of the Company, as well as the proceeds of any sale in Israel of the ordinary shares to an Israeli resident, into freely repatriable dollars, at a rate of exchange prevailing at the time of conversion, pursuant to regulations issued under the Currency Control Law, 1978, provided that Israeli income tax has been withheld by the Company with respect to such amounts.
E. Taxation
The following description is not intended to constitute a complete analysis of all tax consequences relating to the acquisition, ownership and disposition of our ordinary shares or ADSs. You should consult your own tax advisor concerning the tax consequences of your particular situation, as well as any tax consequences that may arise under the laws of any state, local, foreign or other taxing jurisdiction.
Israeli tax considerations and government programs
The following is a summary of the current tax regime in the State of Israel, which applies to us and to persons who hold our ordinary shares or ADSs.
This summary does not discuss all the aspects of Israeli tax law that may be relevant to a particular investor in light of his or her personal investment circumstances or to some types of investors subject to special treatment under Israeli law. Examples of this kind of investor include traders in securities or persons who do not hold our ordinary shares or ADSs as a capital asset. Some parts of this discussion are based on a new tax legislation which has not been subject to judicial or administrative interpretation. The discussion should not be construed as legal or professional tax advice and does not cover all possible tax considerations.
HOLDERS AND POTENTIAL INVESTORS ARE URGED TO CONSULT THEIR OWN TAX ADVISORS AS TO THE ISRAELI OR OTHER TAX CONSEQUENCES OF THE PURCHASE, OWNERSHIP AND DISPOSITION OF OUR ORDINARY SHARES OR ADSs, INCLUDING, IN PARTICULAR, THE EFFECT OF ANY FOREIGN, STATE OR LOCAL TAXES.
General corporate tax structure in Israel
Israeli resident companies are generally subject to corporate tax on both ordinary income and capital gains, currently at the rate of 23% of a company’s taxable income. Capital gains derived by an Israeli resident company are subject to tax at the prevailing corporate tax rate.
Taxation of our shareholders
Capital gains
Capital gains tax is generally imposed on the disposal of capital assets by an Israeli resident, and on the disposal of capital assets by a non-resident of Israel if those assets (i) are located in Israel, (ii) are shares or a right to shares in an Israeli resident corporation, (iii) represent, directly or indirectly, rights to assets located in Israel, or (iv) a right in a foreign resident corporation, which in its essence is the owner of a direct or indirect right to property located in Israel (with respect to the portion of the gain attributed to the property located in Israel), unless a specific exemption is available or unless a tax treaty between Israel and the shareholder’s country of residence provides otherwise. The ITO distinguishes between “Real Capital Gain” and “Inflationary Surplus.” Real Capital Gain is the excess of the total capital gain over Inflationary Surplus. The Inflationary Surplus is a portion of the total capital gain which is equivalent to the increase of the relevant asset’s price that is attributable to the increase in the Israeli consumer price index or, in certain circumstances, a foreign currency exchange rate, between the date of purchase and the date of sale. Inflationary Surplus is not currently subject to tax in Israel.
Real Capital Gain accrued by individuals on the sale of our ordinary shares or ADSs will be taxed at the rate of 25%. However, if the individual shareholder is a “Substantial Shareholder” (i.e., a person who holds, directly or indirectly, alone or together with another, 10% or more of one of the Israeli resident company’s means of control) at the time of sale or at any time during the preceding 12-month period, such gain will be taxed at the rate of 30%. In addition, capital gains generated by an individual claiming deduction of financing expenses in respect of such gain will be taxed at the rate of 30%.
Individual and corporate shareholders dealing in securities in Israel are taxed at the tax rates applicable to business income in 2021, a tax rate of 23% for corporations and a marginal tax rate of up to 47% for individuals, unless contrary provisions in a relevant tax treaty applies. In addition, a 3% excess tax (as discussed below) is levied on individuals whose total taxable income in Israel in 2021 exceeded NIS 647,640.
Notwithstanding the foregoing, generally, capital gain derived from the sale of our ordinary shares or ADSs by a shareholder who is a non-Israeli resident (whether an individual or a corporation) should be exempt from Israeli capital gain tax, provided, among others, that:(i) the ordinary shares or ADSs were purchased upon or after the listing of the securities on the stock exchange, and (ii) the seller does not have a permanent establishment in Israel to which the derived capital gain is attributable. However, non-Israeli entities (including corporations) will not be entitled to the foregoing exemption if Israeli residents, whether directly or indirectly: (i) have a controlling interest of more than 25% in such non-Israeli entity or (ii) are the beneficiaries of, or are entitled to, 25% or more of the revenues or profits of such non-Israeli entity. In addition, such exemption is not applicable to a person whose gains from selling or otherwise disposing of the shares or ADSs are deemed to be business income. In addition, the sale of ordinary shares or ADSs may be exempt from Israeli capital gains tax under the provisions of an applicable tax treaty. For example, the U.S.-Israel Tax Treaty, or the Treaty, generally exempts U.S. residents (for purposes of the U.S.-Israel Treaty) holding the shares as a capital asset from Israeli capital gains tax in connection with such sale, exchange or disposition unless either (i) the U.S. treaty resident own, directly or indirectly, 10% or more of the Israeli resident company’s voting rights at any time within the 12-month period preceding such sale, exchange or disposition; (ii) the seller, if an individual, has been present in Israel for a period or periods aggregating to 183 days or more during the relevant taxable year; (iii) the capital gain from the sale, exchange or disposition was derived through a permanent establishment of the U.S. resident in Israel; (iv) the capital gain arising from such sale, exchange or disposition is attributed to real estate located in Israel, or (v) the capital gains arising from such sale, exchange or disposition is attributed to royalties. In any such case, the sale, exchange or disposition of such shares or ADSs would be subject to Israeli tax, to the extent applicable; however, under the U.S.-Israel Treaty, a U.S. resident would be permitted to claim a credit for the Israeli tax against the U.S. federal income tax imposed with respect to the sale, exchange or disposition, subject to the limitations in U.S. laws applicable to foreign tax credits. The U.S.-Israel Treaty does not provide such credit against any U.S. state or local taxes.
In some instances where our shareholders may be liable for Israeli tax on the sale of their shares or ADSs, the payment of the consideration may be subject to withholding tax in Israel. Shareholders may be required to demonstrate that they are exempt from tax on their capital gains in order to avoid withholding tax at the time of sale. Specifically, in transactions involving a sale of all of the shares of an Israeli resident company, in the form of a merger or otherwise, the Israel Tax Authority may require from shareholders who are not liable for Israeli tax to sign declarations in forms specified by this authority or obtain a specific exemption from the Israel Tax Authority to confirm their status as non-Israeli resident, and, in the absence of such declarations or exemptions, may require the purchaser of the shares to withhold tax.
Upon the sale of securities traded on a stock exchange, a detailed return, including a computation of the tax due, must be filed and an advance payment must be made on January 31 and July 31 of every tax year, in respect of sales of securities made within the previous six months by Israeli residents for whom tax has not already been deducted. However, if all tax due was withheld at source according to applicable provisions of the ITO and the regulations promulgated thereunder, there is no need to file a return and no advance payment must be paid. Capital gains are also reportable on the annual income tax return.
Dividends
Israeli residents who are individuals are generally subject to Israeli income tax for dividends paid (other than bonus shares or share dividends) at 25%, or 30% if the recipient of such dividend is a Substantial Shareholder, as defined above, at the time of distribution or at any time during the preceding 12-month period. Israeli resident corporations are generally exempt from Israeli corporate tax on the receipt of dividends paid on shares of Israeli resident corporations.
Non-Israeli residents (whether individuals or corporations) are generally subject to Israeli income tax on the receipt of dividends paid on ordinary shares at the rate of 25% or 30% (if the dividend recipient is a Substantial Shareholder at the time of distribution or at any time during the preceding 12-month period). Such dividends are generally subject to Israeli withholding tax at a rate of 25% so long as the shares are registered with a nominee company (whether the recipient is a Substantial Shareholder or not), unless a reduced rate is provided under an applicable tax treaty (subject to the receipt in advance of a valid certificate from the Israel Tax Authority allowing for a reduced tax rate). Under the U.S.- Israel Treaty and subject to the eligibility to the benefits under such treaty, the maximum rate of tax withheld at source in Israel on dividends paid to a holder of our ordinary shares who is a U.S. resident (for purposes of the U.S.-Israel Treaty) is 25%. However, for dividends not generated by an Approved Enterprise, Benefited Enterprise or Preferred Enterprises (which are benefitted tax regimes under the Law for Encouragement of Capital Investments-1959) and paid to a U.S. corporation holding 10% or more of the outstanding voting capital throughout the tax year in which the dividend is distributed as well as during the previous tax year, the maximum rate of withholding tax is generally 12.5%, provided that not more than 25% of the gross income of the Israeli resident paying corporation for such preceding year consists of certain types of dividends and interest. Notwithstanding the foregoing, dividends distributed from income attributed to an Approved Enterprise, Benefited Enterprise or Preferred Enterprise are not entitled to such reduction under such tax treaty but are subject to withholding tax at the rate of 15% or 20% for such a United States corporate shareholder, provided that the conditions related to the holding of 10% of our voting capital and to our gross income for the previous year (as set forth in the previous sentence) are met. The aforementioned rates under the U.S.-Israel Treaty would not apply if the dividend income is derived through a permanent establishment of the U.S. resident in Israel.
If the dividend is attributable partly to income derived from an Approved Enterprise, Benefited Enterprise or Preferred Enterprise, and partly to other sources of income, the withholding rate will be a blended rate reflecting the relative portions of the two types of income. U.S. residents (for purposes of the U.S.-Israel Treaty) who are subject to Israeli withholding tax on a dividend may be entitled to a credit or deduction for United States federal income tax purposes up to the amount of the taxes withheld, subject to detailed rules contained in U.S. tax law.
A non-Israeli resident who receives dividends from which tax was withheld is generally exempt from the obligation to file tax returns in Israel in respect of such income, provided, inter alia, that (i) such income was not derived from a business conducted in Israel by the taxpayer, (ii) the taxpayer has no other taxable sources of income in Israel with respect to which a tax return is required to be filed and (iii) the taxpayer is not obliged to pay excess tax (as further explained below).
Excess Tax
Individuals who are subject to tax in Israel (whether any such individual is an Israeli resident or non-Israeli resident) are also subject to an additional tax at a rate of 3% on annual income exceeding NIS 647,640 for 2021 (which amount is linked to the annual change in the Israeli consumer price index), including, but not limited to, dividends, interest and capital gain.
Estate and gift tax
Israeli law presently does not impose estate or gift taxes.
Material United States federal income tax considerations
The following discussion describes material United States federal income tax considerations relating to the acquisition, ownership, and disposition of shares or ADSs by a U.S. Holder (as defined below) that acquires our shares or ADSs and holds them as a capital asset. This discussion is based on the tax laws of the United States, including the Code, Treasury regulations promulgated or proposed thereunder, and administrative and judicial interpretations thereof, all as in effect on the date hereof. These tax laws are subject to change, possibly with retroactive effect, and subject to differing interpretations that could affect the tax consequences described herein. In addition, this section is based in part upon the representations of the depositary and the assumption that each obligation in the deposit agreement and any related agreements will be performed in accordance with its terms. This discussion does not address the tax consequences to a U.S. Holder under the laws of any state, local or foreign taxing jurisdiction.
For purposes of this discussion, a “U.S. Holder” is a beneficial owner of our shares or ADSs that, for United States federal income tax purposes, is:
| • | an individual who is a citizen or resident of the United States, |
| • | a domestic corporation (or other entity taxable as a corporation); |
| • | an estate the income of which is subject to United States federal income taxation regardless of its source; or |
| • | a trust if (1) a court within the United States is able to exercise primary supervision over the trust’s administration and one or more United States persons have the authority to control all substantial decisions of the trust or (2) a valid election under the Treasury regulations is in effect for the trust to be treated as a United States person. |
A “Non-U.S. Holder” is a beneficial owner of our ordinary shares that is neither a U.S. Holder nor a partnership (or other entity treated as a partnership for United States federal income tax purposes).
This discussion does not address all aspects of United States federal income taxation that may be applicable to U.S. Holders in light of their particular circumstances or status (including, for example, banks and other financial institutions, insurance companies, broker and dealers in securities or currencies, traders that have elected to mark securities to market, regulated investment companies, real estate investment trusts, partnerships or other pass-through entities, corporations that accumulate earnings to avoid U.S. federal income tax, tax-exempt organizations, pension plans, persons that hold our shares as part of a straddle, hedge or other integrated investment, persons subject to alternative minimum tax or whose “functional currency” is not the U.S. dollar).
If a partnership (including any entity or arrangement treated as a partnership for United States federal income tax purposes) holds our shares or ADSs, the tax treatment of a person treated as a partner in the partnership for United States federal income tax purposes generally will depend on the status of the partner and the activities of the partnership. Partnerships (and other entities or arrangements so treated for United States federal income tax purposes) and their partners should consult their own tax advisors.
In general, and taking into account the earlier assumptions, for United States federal income and Israeli tax purposes, a holder that holds ADRs evidencing ADSs will be treated as the owner of the shares represented by those ADRs. Exchanges of shares for ADRs, and ADRs for shares, generally will not be subject to United States federal income or Israeli tax.
This discussion addresses only U.S. Holders and does not discuss any tax considerations other than United States federal income tax considerations. Prospective investors are urged to consult their own tax advisors regarding the United States federal, state, and local, and non-United States tax consequences of the purchase, ownership, and disposition of our shares or ADSs.
We do not expect to make any distribution with respect to our shares or ADSs. However, if we make any such distribution, under the United States federal income tax laws, and subject to the passive foreign investment company, or PFIC, rules discussed below, the gross amount of any dividend we pay out of our current or accumulated earnings and profits (as determined for United States federal income tax purposes) will be includible in income for a U.S. Holder and subject to United States federal income taxation. Dividends paid to a noncorporate U.S. Holder that constitute qualified dividend income will be taxable at a preferential tax rate applicable to long-term capital gains of, currently, 20 percent, provided that the U.S. Holder holds the shares or ADSs for more than 60 days during the 121-day period beginning 60 days before the ex-dividend date and meets other holding period requirements. If we are treated as a PFIC, dividends paid to a U.S. Holder will not be treated as qualified dividend income. If we are not treated as a PFIC, dividends we pay with respect to the shares or ADSs generally will be qualified dividend income, provided that the holding period requirements are satisfied by the U.S. Holder.
A U.S. Holder must include any Israeli tax withheld from the dividend payment in the gross amount of the dividend even though the holder does not in fact receive it. The dividend is taxable to the holder when the holder, in the case of shares, or the Depositary, in the case of ADSs, receives the dividend, actually or constructively. Because we are not a United States corporation, the dividend will not be eligible for the dividends-received deduction generally allowed to United States corporations in respect of dividends received from other United States corporations. The amount of the dividend distribution includible in a U.S. Holder’s income will be the U.S. dollar value of the NIS payments made, determined at the spot NIS/U.S. dollar rate on the date the dividend distribution is includible in income, regardless of whether the payment is in fact converted into U.S. dollars. Generally, any gain or loss resulting from currency exchange fluctuations during the period from the date the dividend payment is included in income to the date the payment is converted into U.S. dollars will be treated as ordinary income or loss and will not be eligible for the special tax rate applicable to qualified dividend income. The gain or loss generally will be income or loss from sources within the United States for foreign tax credit limitation purposes.
Dividends paid with respect to our ordinary shares or ADSs will be treated as foreign source income, which may be relevant in calculating the holder’s foreign tax credit limitation. The limitation on foreign taxes eligible for credit is calculated separately with respect to specific classes of income. For this purpose, dividends that we distribute generally should constitute “passive category income,” or, in the case of certain U.S. Holders, “general category income.” A foreign tax credit for foreign taxes imposed on distributions may be denied if holders do not satisfy certain minimum holding period requirements. The rules relating to the determination of the foreign tax credit are complex, and you should consult your tax advisor to determine whether and to what extent you will be entitled to this credit.
To the extent a distribution with respect to our shares or ADSs exceeds our current or accumulated earnings and profits, as determined under United States federal income tax principles, the distribution will be treated, first, as a tax-free return of the U.S. Holder’s investment, up to the holder’s adjusted tax basis in its shares or ADSs, and, thereafter, as capital gain, which is subject to the tax treatment described below in “—Gain on Sale, Exchange or Other Taxable Disposition.”
Subject to certain limitations, the Israeli tax withheld in accordance with the Treaty and paid over to Israel will be creditable or deductible against a U.S. Holder’s United States federal income tax liability.
Subject to the discussion below under “Information reporting and backup withholding,” if you are a Non-U.S. Holder, you generally will not be subject to United States federal income (or withholding) tax on dividends received by you on your ordinary shares, unless you conduct a trade or business in the United States and such income is effectively connected with that trade or business (or, if required by an applicable income tax treaty, the dividends are attributable to a permanent establishment or fixed base that such holder maintains in the United States).
Gain on sale, exchange or other taxable disposition
Subject to the PFIC rules described below under “—Passive Foreign Investment Company Considerations,” a U.S. Holder that sells, exchanges or otherwise disposes of shares or ADSs in a taxable disposition generally will recognize capital gain or loss for United States federal income tax purposes equal to the difference between the U.S. Dollar value of the amount realized and the holder’s tax basis, determined in U.S. Dollars, in the shares or ADSs. Gain or loss recognized on such a sale, exchange or other disposition of shares or ADSs generally will be long-term capital gain if the U.S. Holder’s holding period in the shares or ADSs exceeds one year. Long-term capital gains of non-corporate U.S. Holders are generally taxed at preferential rates. The gain or loss generally will be income or loss from sources within the United States for foreign tax credit limitation purposes. A U.S. Holder’s ability to deduct capital losses is subject to limitations.
Subject to the discussion below under “Information reporting and backup withholding,” if you are a Non-U.S. Holder, you generally will not be subject to United States federal income or withholding tax on any gain realized on the sale or exchange of such ordinary shares unless:
| • | such gain is effectively connected with your conduct of a trade or business in the United States (or, if required by an applicable income tax treaty, the gain is attributable to a permanent establishment or fixed base that such holder maintains in the United States); or |
| • | you are an individual and have been present in the United States for 183 days or more in the taxable year of such sale or exchange and certain other conditions are met. |
For a cash basis taxpayer, units of foreign currency paid or received are translated into U.S. dollars at the spot rate on the settlement date of the purchase or sale. In that case, no foreign currency exchange gain or loss will result from currency fluctuations between the trade date and the settlement date of such a purchase or sale. An accrual basis taxpayer, however, may elect the same treatment required of cash basis taxpayers with respect to purchases and sales of our ordinary shares that are traded on an established securities market, provided the election is applied consistently from year to year. Such election may not be changed without the consent of the IRS. An accrual basis taxpayer who does not make such election may recognize exchange gain or loss based on currency fluctuations between the trade date and the settlement date. Any foreign currency gain or loss a U.S. Holder realizes will be U.S. source ordinary income or loss.
The determination of whether the ADSs or ordinary shares are traded on an established securities market is not entirely clear under current U.S. federal income tax law. Please consult your tax advisor regarding the proper treatment of foreign currency gains or losses with respect to a sale or other disposition of our ordinary shares.
Passive foreign investment company considerations
Based on our income and assets, we believe that we should be treated as a PFIC for the preceding taxable year. However, the determination of our PFIC status is made annually based on the factual tests described below. Consequently, while we may be treated as a PFIC in future years, we cannot estimate with certainty at this stage whether or not we are likely to be treated as a PFIC for the current or future taxable years. If we were classified as a PFIC in any taxable year, a U.S. Holder would be subject to special rules with respect to distributions on and sales, exchanges and other dispositions of the shares or ADSs. We will be treated as a PFIC for any taxable year in which at least 75 percent of our gross income is “passive income” or at least 50 percent of our gross assets during the taxable year, assuming we were not a controlled foreign corporation, or CFC, for the year being tested, based on the average of the fair market values of the assets determined at the end of each quarterly period, are assets that produce or are held for the production of passive income. Passive income for this purpose generally includes, among other things, dividends, interest, rents, royalties, gains from commodities and securities transactions, and gains from assets that produce passive income. However, rents and royalties received from unrelated parties in connection with the active conduct of a trade or business are not considered passive income for purposes of the PFIC test. In determining whether we are a PFIC, a pro rata portion of the income and assets of each corporation in which we own, directly or indirectly, at least a 25% interest (by value) is taken into account.
Excess distribution rules
If we were a PFIC with respect to a U.S. Holder, then unless the holder makes one of the elections described below, a special tax regime would apply to the U.S. Holder with respect to (a) any “excess distribution” (generally, aggregate distributions in any year that are greater than 125% of the average annual distribution received by the holder in the shorter of the three preceding years or the holder’s holding period for the shares or ADSs) and (b) any gain realized on the sale or other disposition of the shares or ADSs. Under this regime, any excess distribution and realized gain will be treated as ordinary income and will be subject to tax as if (a) the excess distribution or gain had been realized ratably over the U.S. Holder’s holding period, (b) the amount deemed realized in each year had been subject to tax in each year of that holding period at the highest marginal rate for such year (other than income allocated to the current period or any taxable period before we became a PFIC, which would be subject to tax at the U.S. Holder’s regular ordinary income rate for the current year and would not be subject to the interest charge discussed below), and (c) the interest charge generally applicable to underpayments of tax had been imposed on the taxes deemed to have been payable in those years. If we were determined to be a PFIC, this tax treatment for U.S. Holders would apply also to indirect distributions and gains deemed realized by U.S. Holders in respect of stock of any of our subsidiaries determined to be PFICs. In addition, dividend distributions would not qualify for the lower rates of taxation applicable to long-term capital gains discussed above under “Dividends”.
A U.S. Holder that holds the shares or ADSs at any time during a taxable year in which we are classified as a PFIC generally will continue to treat such shares or ADSs as shares or ADSs in a PFIC, even if we no longer satisfy the income and asset tests described above, unless the U.S. Holder elects to recognize gain, which will be taxed under the excess distribution rules as if such shares or ADSs had been sold on the last day of the last taxable year for which we were a PFIC.
Certain elections by a U.S. Holder would alleviate some of the adverse consequences of PFIC status and would result in an alternative treatment of the shares or ADSs, as described below. However, we do not currently intend to provide the information necessary for U.S. Holders to make “QEF elections,” as described below, and the availability of a “mark-to-market election” with respect to the shares or ADSs is a factual determination that will depend on the manner and quantity of trading of our shares or ADSs, as described below. A mark-to-market election cannot be made with respect to the stock of any of our subsidiaries.
QEF election
If we were a PFIC, the rules above would not apply to a U.S. Holder that makes an election to treat our shares or ADSs as stock of a “qualified electing fund” or QEF. A U.S. Holder that makes a QEF election is required to include in income its pro rata share of our ordinary earnings and net capital gain as ordinary income and long-term capital gain, respectively, subject to a separate election to defer payment of taxes, which deferral is subject to an interest charge. A U.S. Holder makes a QEF election generally by attaching a completed IRS Form 8621 to a timely filed United States federal income tax return for the year beginning with which the QEF election is to be effective (taking into account any extensions). A QEF election can be revoked only with the consent of the IRS. In order for a U.S. Holder to make a valid QEF election, we must annually provide or make available to the holder certain information. While we intend to provide to U.S. Holders the information required to make a valid QEF election, we cannot provide any assurances that we will in fact provide such information.
Mark-to-market election
If we were a PFIC, the rules above also would not apply to a U.S. Holder that makes a “mark-to-market” election with respect to the shares or ADSs, but this election will be available with respect to the shares or ADSs only if they meet certain minimum trading requirements to be considered “marketable stock” for purposes of the PFIC rules. In addition, a mark-to-market election generally could not be made with respect to the stock of any of our subsidiaries unless that stock were itself marketable stock, and the election may therefore be of limited benefit to a U.S. Holder that wants to avoid the excess distribution rules described above. Shares or ADSs will be marketable stock if they are regularly traded on a national securities exchange that is registered with the Securities and Exchange Commission or on a non-U.S. exchange or market that meets certain requirements under the Treasury regulations. Shares or ADSs generally will be considered regularly traded during any calendar year during which they are traded, other than in de minimis quantities, on at least 15 days during each calendar quarter. Any trades that have as their principal purpose meeting this requirement will be disregarded.
A U.S. Holder that makes a valid mark-to-market election for the first tax year in which the holder holds (or is deemed to hold) our shares or ADSs and for which we are a PFIC will be required to include each year an amount equal to the excess, if any, of the fair market value of such shares or ADSs the holder owns as of the close of the taxable year over the holder’s adjusted tax basis in such shares or ADSs. The U.S. Holder will be entitled to a deduction for the excess, if any, of the holder’s adjusted tax basis in the shares or ADSs over the fair market value of such shares or ADSs as of the close of the taxable year, but only to the extent of any net mark-to-market gains with respect to such shares or ADSs included by the U.S. Holder under the election for prior taxable years. The U.S. Holder’s basis in such shares or ADSs will be adjusted to reflect the amounts included or deducted pursuant to the election. Amounts included in income pursuant to a mark-to-market election, as well as gain on the sale, exchange or other taxable disposition of such shares or ADSs, will be treated as ordinary income. The deductible portion of any mark-to-market loss, as well as loss on a sale, exchange or other disposition of our shares or ADSs to the extent that the amount of such loss does not exceed net mark-to-market gains previously included in income, will be treated as ordinary loss.
The mark-to-market election applies to the taxable year for which the election is made and all subsequent taxable years, unless the shares cease to be treated as marketable stock for purposes of the PFIC rules or the IRS consents to its revocation. The excess distribution rules described above generally will not apply to a U.S. Holder for tax years for which a mark-to-market election is in effect. However, if we were a PFIC for any year in which the U.S. Holder owns the shares or ADSs but before a mark-to-market election is made, the interest charge rules described above would apply to any mark-to-market gain recognized in the year the election is made.
PFIC reporting obligations
A U.S. Holder of PFIC shares must generally file an annual information return on IRS Form 8621 (Information Return by a Shareholder of a Passive Foreign Investment Company or Qualified Electing Fund) containing such information as the U.S. Treasury Department may require. The failure to file IRS Form 8621 could result in the imposition of penalties and the extension of the statute of limitations with respect to U.S. federal income tax.
U.S. Holders are urged to consult their tax advisors as to our status as a PFIC, and the tax consequences to them if we were a PFIC, including the reporting requirements and the desirability of making, and the availability of, a QEF election or a mark-to-market election with respect to the shares or ADSs.
Non-corporate U.S. Holders that are individuals, estates or trusts and whose income exceeds certain thresholds generally are subject to a 3.8% tax on all or a portion of their net investment income, which may include their gross dividend income and net gains from the disposition of shares or ADSs. A United States person that is an individual, estate or trust is encouraged to consult its tax advisors regarding the applicability of this Medicare tax to its income and gains in respect of any investment in our shares or ADSs.
Information reporting with respect to foreign financial assets
Individual U.S. Holders may be subject to certain reporting obligations on IRS Form 8938 (Statement of Specified Foreign Financial Asset) with respect to the shares or ADSs for any taxable year during which the U.S. Holder’s aggregate value of these and certain other “specified foreign financial assets” exceed a threshold amount that varies with the filing status of the individual. This reporting obligation also applies to domestic entities formed or availed of to hold, directly or indirectly, specified foreign financial assets, including the shares or ADSs. Significant penalties can apply if U.S. Holders are required to make this disclosure and fail to do so.
Information reporting and backup withholding
In general, information reporting, on IRS Form 1099, will apply to dividends in respect of shares or ADSs and the proceeds from the sale, exchange or redemption of shares of ADSs that are paid to a holder of shares or ADSs within the United States (and in certain cases, outside the United States), unless such holder is an exempt recipient such as a corporation. Backup withholding (currently at a 24% rate) may apply to such payments if a holder of shares or ADSs fails to provide a taxpayer identification number (generally on an IRS Form W-9) or certification of other exempt status or fails to report in full dividend and interest income.
Backup withholding is not an additional tax. A U.S. Holder generally may obtain a refund of any amounts withheld under the backup withholding rules that exceed the U.S. Holder’s income tax liability by filing a refund claim with the Internal Revenue Service.
F. Dividends and Paying Agents
Not applicable
G. Statement by Experts
Not applicable.
H. Documents on Display
We are subject to the information reporting requirements of the Exchange Act applicable to foreign private issuers and under those requirements will file reports with the SEC. Those reports may be inspected without charge at the locations described below. As a foreign private issuer, we are exempt from the rules under the Exchange Act related to the furnishing and content of proxy statements, and our officers, directors and principal shareholders are exempt from the reporting and short swing profit recovery provisions contained in Section 16 of the Exchange Act. In addition, we are not required under the Exchange Act to file periodic reports and financial statements with the SEC as frequently or as promptly as United States companies whose securities are registered under the Exchange Act.
We maintain a corporate website at www.meatech3d.com. We intend to post our Annual Report on Form 20-F on our website promptly following it being filed with the SEC. Information contained on, or that can be accessed through, our website does not constitute a part of this Annual Report on Form 20-F. We have included our website address in this Annual Report on Form 20-F solely as an inactive textual reference.
Our SEC filings are available to you on the SEC’s website at http://www.sec.gov. This site contains reports, proxy and information statements and other information regarding issuers that file electronically with the SEC. The information on that website is not part of this Annual Report on Form 20-F.
With respect to references made in this Annual Report on Form 20-F to any contract or other document of MeaTech, such references are not necessarily complete and you should refer to the exhibits attached or incorporated by reference to this Annual Report on Form 20-F for copies of the actual contract or document.
I. Subsidiary Information
Not applicable.
| | ITEM 11. | QUANTITATIVE AND QUALITATIVE DISCLOSURE ON MARKET RISK |
For quantitative and qualitative information regarding our market risk, see “Item 5 — Operating and Financial Review and Prospects — Liquidity and Capital Resources — Quantitative and Qualitative Disclosures About Market Risk” and Note 22 to our financial statements.
| | ITEM 12. | DESCRIPTION OF SECURITIES OTHER THAN EQUITY SECURITIES |
A. Debt Securities
Not applicable.
B. Warrants and Rights
Not applicable.
C. Other Securities
Not applicable.
D. American Depositary Shares
The Bank of New York Mellon is the depositary of the ADSs. Each ADS represents ten ordinary shares (or a right to receive ten ordinary shares). Each ADS will also represent any other securities, cash or other property which may be held by the depositary. The depositary’s office at which the ADSs are administered and its principal executive office are located at 240 Greenwich Street, New York, New York 10286.
A deposit agreement among us, the depositary, ADS holders and all other persons indirectly or beneficially holding ADSs sets out ADS holder rights as well as the rights and obligations of the depositary. New York law governs the deposit agreement and the ADSs. A copy of the deposit agreement was filed as Exhibit 4.1 to our amended registration statement on Form F-1 filed with the SEC on March 5, 2021 and is incorporated by reference herein.
Fees and Expenses
Pursuant to the terms of the deposit agreement, the holders of the ADSs will be required to pay the following fees:
Persons depositing or withdrawing ordinary
shares or ADS holders must pay | | For: |
| $5.00 (or less) per 100 ADSs (or portion of 100 ADSs) | | Issuance of ADSs, including issuances resulting from a distribution of ordinary shares or rights or other property Cancellation of ADSs for the purpose of withdrawal, including if the deposit agreement terminates |
| | | |
| $.05 (or less) per ADS | | Any cash distribution to ADS holders |
| | | |
| A fee equivalent to the fee that would be payable if securities distributed to you had been ordinary shares and the ordinary shares had been deposited for issuance of ADSs | | Distribution of securities distributed to holders of deposited securities (including rights) that are distributed by the depositary to ADS holders |
| | | |
| $.05 (or less) per ADS per calendar year | | Depositary services |
| | | |
| Registration or transfer fees | | Transfer and registration of ordinary shares on our share register to or from the name of the depositary or its agent when you deposit or withdraw ordinary shares |
| | | |
| Expenses of the depositary | | Cable, telex and facsimile transmissions (when expressly provided in the deposit agreement)
Converting foreign currency to U.S. dollars |
| | | |
| Taxes and other governmental charges the depositary or the custodian have to pay on any ADSs or ordinary shares underlying ADSs, such as stock transfer taxes, stamp duty or withholding taxes | | As necessary |
| | | |
| Any charges incurred by the depositary or its agents for servicing the deposited securities | | As necessary |
The depositary collects its fees for delivery and surrender of ADSs directly from investors depositing ordinary shares or surrendering ADSs for the purpose of withdrawal or from intermediaries acting for them. The depositary collects fees for making distributions to investors by deducting those fees from the amounts distributed or by selling a portion of distributable property to pay the fees. The depositary may collect its annual fee for depositary services by deduction from cash distributions or by directly billing investors or by charging the book-entry system accounts of participants acting for them. The depositary may collect any of its fees by deduction from any cash distribution payable (or by selling a portion of securities or other property distributable) to ADS holders that are obligated to pay those fees. The depositary may generally refuse to provide fee-attracting services until its fees for those services are paid.
From time to time, the depositary may make payments to us to reimburse us for costs and expenses generally arising out of establishment and maintenance of the ADS program, waive fees and expenses for services provided to us by the depositary or share revenue from the fees collected from ADS holders. In performing its duties under the deposit agreement, the depositary may use brokers, dealers, foreign currency or other service providers that are owned by or affiliated with the depositary and that may earn or share fees, spreads or commissions.
The depositary may convert foreign currency itself or through any of its affiliates and, in those cases, acts as principal for its own account and not as an agent, fiduciary or broker on behalf of any other person and earns revenue, including, without limitation, fees and spreads that it will retain for its own account. The revenue is based on, among other things, the difference between the exchange rate assigned to the currency conversion made under the deposit agreement and the rate that the depositary or its affiliate receives when buying or selling foreign currency for its own account. The depositary makes no representation that the exchange rate used or obtained in any currency conversion under the deposit agreement will be the most favorable rate that could be obtained at the time or that the method by which that rate will be determined will be most favorable to ADS holders, subject to its obligations under the deposit agreement. The methodology used to determine exchange rates used in currency conversions is available upon request.
PART II
| | ITEM 13. | DEFAULTS, DIVIDEND ARREARAGES AND DELINQUENCIES |
Not applicable.
| | ITEM 14. | MATERIAL MODIFICATIONS TO THE RIGHTS OF SECURITY HOLDERS AND USE OF PROCEEDS |
Initial Public Offering
In March 2021, we sold 2,721,271 ADSs, each representing ten ordinary shares, no par value, in our U.S. initial public offering at a public offering price of $10.30 per ADS. Gross proceeds, including proceeds generated from the partial exercise of the underwriter’s option to purchase additional ADSs, were $28 million. Net proceeds to us, after deducting underwriting discounts and commissions and estimated offering expenses payable by us were approximately $24.7 million. The effective date of the registration statement on Form F-1 (File No. 333-253257) for our U.S. initial public offering of ADSs was March 11, 2021. The offering closed on March 17, 2021. H.C. Wainwright, Inc. acted as the sole underwriter in the offering.
The net proceeds from our initial public offering are held in cash and cash equivalents and we expect they will meet our capital requirements until the fourth quarter of 2022. We do not currently have any specific commitments or plans for acquisitions; to the extent we do engage in acquisitions, we will do so after ensuring that we will have sufficient funds available to meet our capital requirements, and such acquisitions are likely to affect our projected cash needs. None of the net proceeds of our initial public offering were paid directly or indirectly to any director, officer, general partner of ours or to their associates, persons owning ten percent or more of any class of our equity securities, or to any of our affiliates.
| | ITEM 15. | CONTROLS AND PROCEDURES |
A. Disclosure Controls and Procedures
We have performed an evaluation of the effectiveness of our disclosure controls and procedures that are designed to ensure that the material financial and non-financial information required to be disclosed to the SEC is recorded, processed, summarized and reported timely. Based on our evaluation, our management, including our chief executive officer and chief financial officer, has concluded that our disclosure controls and procedures (as defined in Rules 13a-15(e) and 15d-15(e) of the Exchange Act) as of the end of the period covered by this report are effective. Notwithstanding the foregoing, there can be no assurance that our disclosure controls and procedures will detect or uncover all failures of persons within the Company to disclose material information otherwise required to be set forth in our reports.
B. Management’s Annual Report on Internal Control over Financial Reporting
Our management is responsible for establishing and maintaining adequate internal control over financial reporting, as such term is defined in Rule 13a-15(f) promulgated under the Exchange Act. Our internal control system was designed to provide reasonable assurance to our management and board of directors regarding the reliability of financial reporting and the preparation and fair presentation of published financial statements for external purposes in accordance with generally accepted accounting principles. All internal control systems, no matter how well designed, have inherent limitations. Therefore, even those systems determined to be effective can provide only reasonable assurance with respect to financial statement preparation and presentation and may not prevent or detect misstatements. Also, projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with policies or procedures may deteriorate.
Our management assessed our internal control over financial reporting as of December 31, 2020, the end of our fiscal year. Management based its assessment on criteria established in Internal Control—Integrated Framework (2013) issued by the Committee of Sponsoring Organizations of the Treadway Commission (COSO). The assessment included evaluation of elements such as the design and operating effectiveness of key financial reporting controls, process documentation, accounting policies and our overall control environment.
Based on its assessment, our management has concluded that our internal control over financial reporting was effective as of the end of the fiscal year to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external reporting purposes in accordance with International Financial Reporting Standards. We reviewed the results of our management’s assessment with the Audit Committee of our Board of Directors.
C. Attestation Report of the Registered Public Accounting Firm
This Annual Report on Form 20-F does not include an attestation report of the company’s registered public accounting firm as the company is considered an emerging growth company.
D. Changes in Internal Control over Financing Reporting
There were no changes in our internal control over financial reporting that occurred during the period covered by this Annual Report on Form 20-F that have materially affected, or that are reasonably likely to materially affect, our internal control over financial reporting.
| | ITEM 16A. | AUDIT COMMITTEE FINANCIAL EXPERT |
Our board of directors has determined that all members of our audit committee are financially literate as determined in accordance with Nasdaq rules and that Messrs. Eli Arad and David Gerbi are qualified to serve as “audit committee financial experts” as defined by SEC rules. The audit committee financial experts are independent directors.
We have adopted a Code of Business Conduct and Ethics (the “Code of Ethics”) that is applicable to all of our directors, executives and other employees, and those of our affiliates. A copy of the Code of Conduct is available on our website at www.meatech3d.com. Any waivers of the Code of Ethics for directors and officers require the approval of the audit committee of our board of directors. We expect that any amendments to the Code of Conduct will be disclosed on our website.
On April 18, 2021, in conjunction with the adoption of the Code of Ethics, we granted a waiver to Mr. Steven H. Lavin, at the time the Chairman of our Board of Directors, from provisions of the Code of Ethics where and to the extent that these conflict with the provisions of agreements to which Mr. Lavin and we are parties.
| | ITEM 16C. | PRINCIPAL ACCOUNTANT FEES AND SERVICES |
Fees Paid to Independent Registered Public Accounting Firm
Somekh Chaikin, Tel Aviv, Israel (PCAOB ID 1057), a member firm of KPMG International, has served as our independent registered public accounting firm for 2021 and 2020. Following are Somekh Chaikin fees for professional services in each of the respective fiscal years:
| | | Year ended December 31, | |
| | | 2021 | | | 2020 | |
| | | USD, in thousands | |
| | | | |
Audit fees(1) | | | 176 | | | | 145 | |
Tax fees(2) | | | 3 | | | | 10 | |
| Total | | | 179 | | | | 155 | |
| (1) | Audit fees consist of fees billed or expected to be billed for the annual audit services engagement and other audit services, which are those services that only the external auditor can reasonably provide, and include the Company audit; statutory audits; comfort letters and consents; attest services; and assistance with and review of documents filed with the TASE and SEC. |
| | |
| (2) | Tax fees include fees billed for tax compliance services that were rendered during the most recent fiscal year, including the preparation of original and amended tax returns and claims for refund; tax consultations, such as assistance and representation in connection with tax audits and appeals, tax advice related to mergers and acquisitions, transfer pricing, and requests for rulings or technical advice from taxing authority; tax planning services; and expatriate tax planning and services. |
Policy on Audit Committee Pre-Approval of Audit and Permissible Non-Audit Services of Independent Auditors
Our Audit Committee has the sole authority to approve the scope of the audit and any audit-related services, as well as all audit fees and terms. The Audit Committee must pre-approve any audit and non-audit services provided by our independent registered public accounting firm. The Audit Committee will not approve the engagement of the independent registered public accounting firm to perform any services that the independent registered public accounting firm would be prohibited from providing under applicable laws, rules and regulations, including those of self-regulating organizations. The Audit Committee reviews and pre-approves the statutory audit fees that can be provided by the independent registered public accounting firm on an annual basis.
ITEM 16D. EXEMPTIONS FROM THE LISTING STANDARDS FOR AUDIT COMMITTEES
Not applicable.
ITEM 16E. PURCHASES OF EQUITY SECURITIES BY THE ISSUER AND AFFILIATED PURCHASERS
Not applicable.
ITEM 16F. CHANGE IN REGISTRANT’S CERTIFYING ACCOUNTANT
Not applicable.
ITEM 16G. CORPORATE GOVERNANCE
Nasdaq Listing Rules and Home Country Practices
The Sarbanes-Oxley Act, as well as related rules subsequently implemented by the SEC, requires foreign private issuers, such as us, to comply with various corporate governance practices. As a foreign private issuer, we are permitted to follow certain Israeli corporate governance practices instead of Nasdaq rules, provided that we disclose which requirements we are not following and the equivalent Israeli requirements. Below is a concise summary of the significant ways in which our corporate governance practices differ from the corporate governance requirements of Nasdaq applicable to domestic U.S. listed companies:
| • | Quorum. As permitted under the Companies Law, pursuant to our articles of association, the quorum required for an ordinary meeting of shareholders consists of at least two shareholders present in person or by proxy who hold or represent between them at least 25% of the voting power of our shares (and, with respect to an adjourned meeting, generally one or more shareholders who hold or represent any number of shares), instead of 33 1/3% of the issued share capital provided under Nasdaq Listing Rule 5260(c). |
| • | Shareholder Approval. Although the Nasdaq Listing Rules generally require shareholder approval of equity compensation plans and material amendments thereto, we follow Israeli practice, which is to have such plans and amendments approved only by the board of directors, unless such arrangements are for the compensation of chief executive officer or directors, in which case they also require the approval of the compensation committee and the shareholders. In addition, rather than follow the Nasdaq Listing Rules requiring shareholder approval for the issuance of securities in certain circumstances, we follow Israeli law, under which a private placement of securities requires approval by our board of directors and shareholders if it will cause a person to become a controlling shareholder (generally presumed at 25% ownership) or if: (a) the securities issued amount to 20% or more of our outstanding voting rights before the issuance; (b) some or all of the consideration is other than cash or listed securities or the transaction is not on market terms; and (c) transaction will increase the relative holdings of a shareholder that holds 5% or more of our outstanding share capital or voting rights or will cause any person to become, as a result of the issuance, a holder of more than 5% of our outstanding share capital or voting rights. |
| • | Executive Sessions. While the Nasdaq Listing Rules require that “independent directors,” as defined in the Nasdaq Listing Rules, must have regularly scheduled meetings at which only “independent directors” are present. Israeli law does not require, nor do our independent directors necessarily conduct, regularly scheduled meetings at which only they are present. |
ITEM 16H. MINE SAFETY DISCLOSURE
Not applicable.
ITEM 16I. DISCLOSURE REGARDING FOREIGN JURISDICTIONS THAT PREVENT INSPECTIONS
Not applicable.
PART III
ITEM 17. FINANCIAL STATEMENTS
The Registrant has responded to Item 18 in lieu of responding to this Item.
ITEM 18. FINANCIAL STATEMENTS
See the financial statements beginning on page F-1. The following financial statements and financial statement schedules are filed as part of this Annual Report on Form 20-F together with the report of the independent registered public accounting firm.