| Inducing T Cells to Treat and Prevent Disease May 2023 Vaccitech Corporate Presentation |
| Vaccitech Overview This presentation includes express and implied “forward-looking statements,” including forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. Forward looking statements include all statements that are not historical facts, and in some cases, can be identified by terms such as “may,” “will,” “could,” “should,” “expect,” “intend,” “plan,” “anticipate,” “believe,” “estimate,” “potential,” “ongoing,” or the negative of these terms, or other comparable terminology intended to identify statements about the future. Forward-looking statements contained in this presentation include, but are not limited to, statements regarding: our product development activities and clinical trials, our regulatory filings and approvals, our estimated cash runway and cash burn, our ability to develop and advance our current and future product candidates and programs, our ability to establish and maintain collaborations or strategic relationships or obtain additional funding, the rate and degree of market acceptance and clinical utility of our product candidates, and the ability and willingness of our third-party collaborators to continue research and development activities relating to our product candidates. By their nature, these statements are subject to numerous risks and uncertainties, including factors beyond our control, that could cause actual results, performance or achievement to differ materially and adversely from those anticipated or implied in the statements. Such risks and uncertainties, include, without limitation, risks and uncertainties related to: preclinical and clinical studies, the success, cost and timing of our product development activities and planned and ongoing preclinical studies and clinical trials, our ability to execute on our strategy, regulatory developments, our ability to fund our operations, global economic uncertainty and the impact that the COVID-19 pandemic may have on our clinical trials, preclinical studies and access to capital, and other risks, uncertainties and other factors identified in our filings with the Securities and Exchange Commission (the “SEC”), including our Annual Report on Form 10-K for the year ended December 31, 2022, our Quarterly Report on Form 10-Q for the most recently ended fiscal quarter and subsequent filings with the SEC. You should not rely upon forward-looking statements as predictions of future events. Although our management believes that the expectations reflected in our statements are reasonable, we cannot guarantee that the future results, performance or events and circumstances described in the forward-looking statements will be achieved or occur and actual results may vary. Recipients are cautioned not to place undue reliance on these forward-looking statements, which speak only as of the date such statements are made and should not be construed as statements of fact. Except as required by law, we do not assume any intent to update any forward-looking statements after the date on which the statement is made, whether as a result of new information, future events or circumstances or otherwise. Certain information contained in this presentation and statements made orally during this presentation relate to or are based on studies, publications, surveys and other data obtained from third-party sources and our own internal estimates and research. While we believe these third-party studies, publications, surveys and other data to be reliable as of the date of this presentation, it has not independently verified, and makes no representations as to the adequacy, fairness, accuracy or completeness of, any information obtained from third-party sources. In addition, no independent source has evaluated the reasonableness or accuracy of our internal estimates or research and no reliance should be made on any information or statements made in this presentation relating to or based on such internal estimates and research. 2 |
| Vaccitech Overview 3 Our mission is to become a global leader in immunotherapies and vaccines that leverage T cell immune responses |
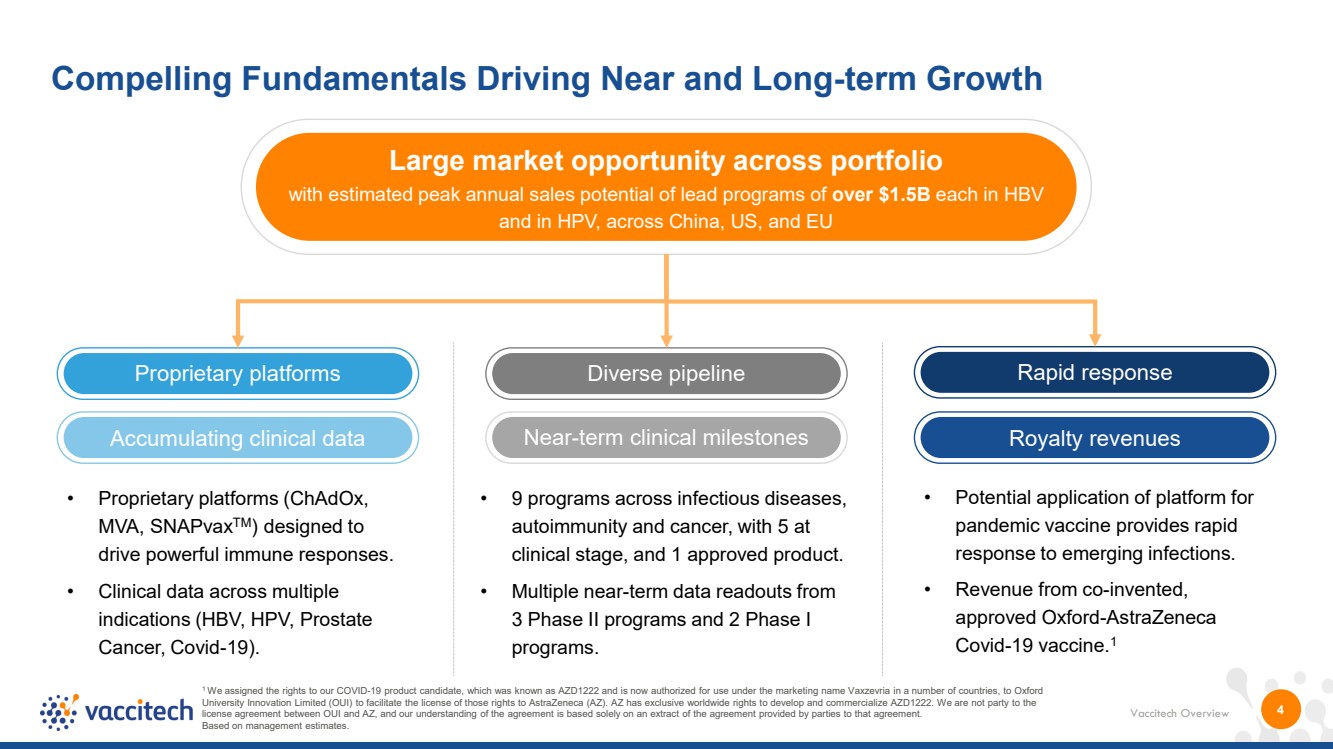
| Vaccitech Overview Large market opportunity across portfolio with estimated peak annual sales potential of lead programs of over $1.5B each in HBV and in HPV, across China, US, and EU Near-term clinical milestones Proprietary platforms Diverse pipeline 4 Compelling Fundamentals Driving Near and Long-term Growth 1 We assigned the rights to our COVID-19 product candidate, which was known as AZD1222 and is now authorized for use under the marketing name Vaxzevria in a number of countries, to Oxford University Innovation Limited (OUI) to facilitate the license of those rights to AstraZeneca (AZ). AZ has exclusive worldwide rights to develop and commercialize AZD1222. We are not party to the license agreement between OUI and AZ, and our understanding of the agreement is based solely on an extract of the agreement provided by parties to that agreement. Based on management estimates. Rapid response Accumulating clinical data Royalty revenues • Proprietary platforms (ChAdOx, MVA, SNAPvaxTM) designed to drive powerful immune responses. • Clinical data across multiple indications (HBV, HPV, Prostate Cancer, Covid-19). • 9 programs across infectious diseases, autoimmunity and cancer, with 5 at clinical stage, and 1 approved product. • Multiple near-term data readouts from 3 Phase II programs and 2 Phase I programs. • Potential application of platform for pandemic vaccine provides rapid response to emerging infections. • Revenue from co-invented, approved Oxford-AstraZeneca Covid-19 vaccine.1 |
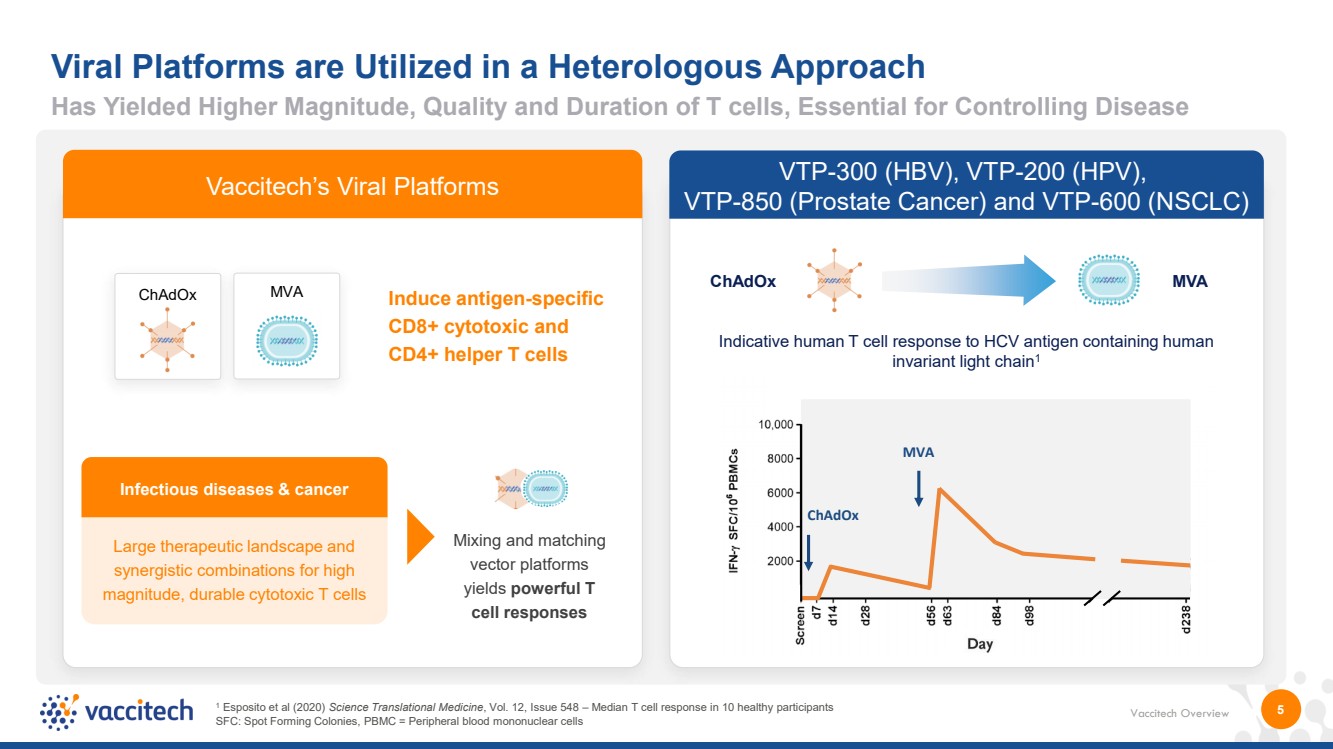
| Vaccitech Overview 5 Viral Platforms are Utilized in a Heterologous Approach Infectious diseases & cancer Large therapeutic landscape and synergistic combinations for high magnitude, durable cytotoxic T cells Induce antigen-specific CD8+ cytotoxic and CD4+ helper T cells Mixing and matching vector platforms yields powerful T cell responses ChAdOx MVA Key Platform Features: • Vectors encode the same transgene • Only boost target antigen responses • No anti-vector immunity (unlike most human adenoviruses) MVA ChAdOx VTP-300 (HBV), VTP-200 (HPV), VTP-850 (Prostate Cancer) and VTP-600 (NSCLC) ChAdOx MVA Indicative human T cell response to HCV antigen containing human invariant light chain1 1 Esposito et al (2020) Science Translational Medicine, Vol. 12, Issue 548 – Median T cell response in 10 healthy participants SFC: Spot Forming Colonies, PBMC = Peripheral blood mononuclear cells Vaccitech’s Viral Platforms Has Yielded Higher Magnitude, Quality and Duration of T cells, Essential for Controlling Disease |
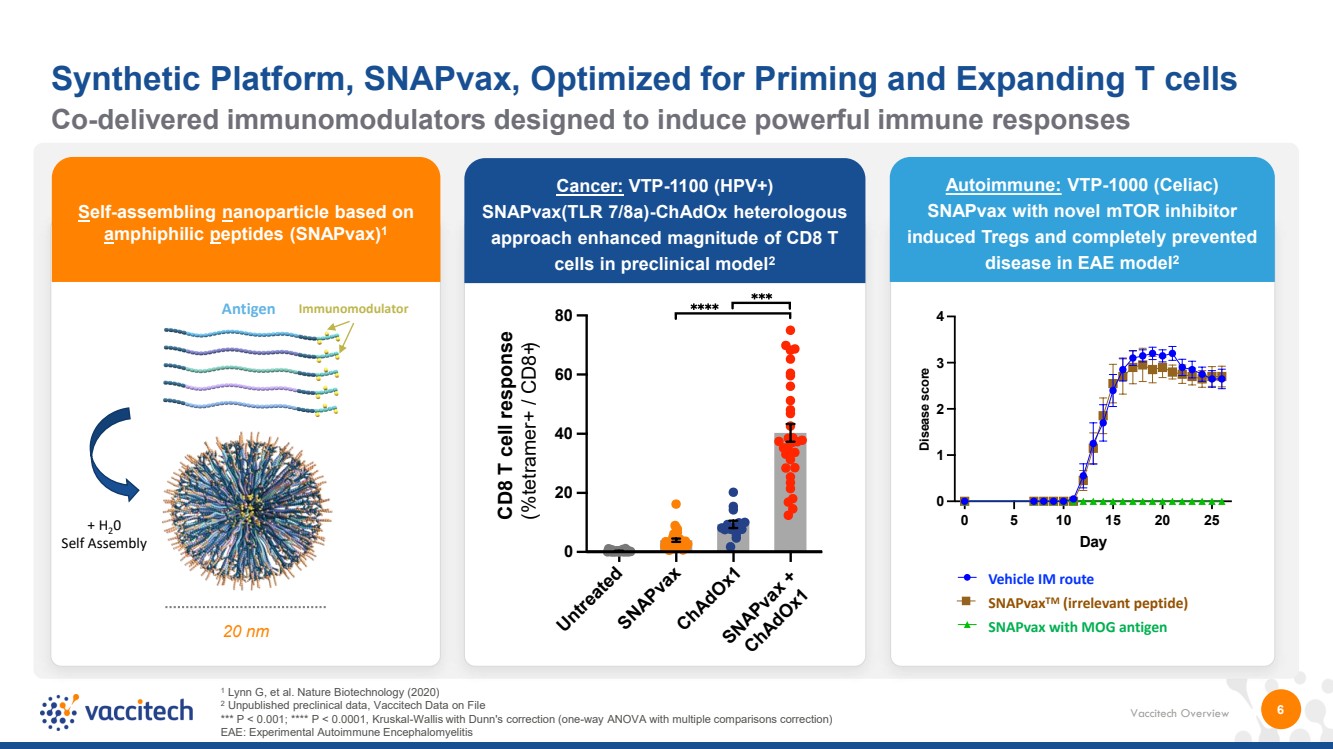
| Vaccitech Overview 6 Self-assembling nanoparticle based on amphiphilic peptides (SNAPvax)1 20 nm Co-delivered immunomodulators designed to induce powerful immune responses Synthetic Platform, SNAPvax, Optimized for Priming and Expanding T cells Cancer: VTP-1100 (HPV+) SNAPvax(TLR 7/8a)-ChAdOx heterologous approach enhanced magnitude of CD8 T cells in preclinical model2 Untreated SNAPvax ChAdOx1 SNAPvax + ChAdOx1 0 20 40 60 80 CD8 T cell response (%tetramer+ / CD8+) + H20 Self Assembly Antigen Immunomodulator 1 Lynn G, et al. Nature Biotechnology (2020) 2 Unpublished preclinical data, Vaccitech Data on File *** P < 0.001; **** P < 0.0001, Kruskal-Wallis with Dunn's correction (one-way ANOVA with multiple comparisons correction) EAE: Experimental Autoimmune Encephalomyelitis 0 5 10 15 20 25 0 1 2 3 4 Day Disease score Vehicle IM MOG + Novel Immun. IM Irr. peptide + Novel Immun. IM 0 5 10 15 20 25 0 1 2 3 4 Day Dis e a s e s c ore Vehicle IM MOG + Novel Immun. IM Irr. peptide + Novel Immun. IM Vehicle IM route SNAPvax TM (irrelevant peptide) SNAPvax with MOG antigen Autoimmune: VTP-1000 (Celiac) SNAPvax with novel mTOR inhibitor induced Tregs and completely prevented disease in EAE model2 |
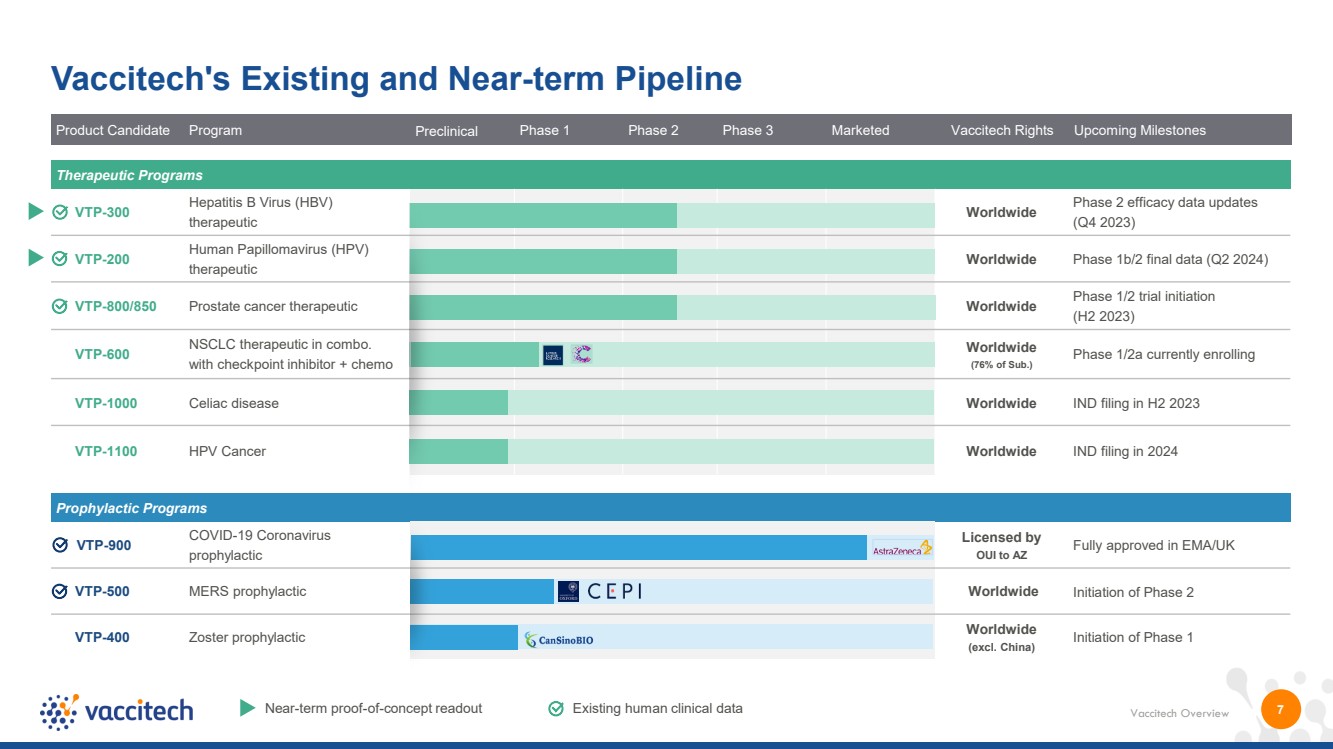
| Vaccitech Overview Product Candidate Program Preclinical Phase 1 Phase 2 Phase 3 Marketed Vaccitech Rights Upcoming Milestones 7 Vaccitech's Existing and Near-term Pipeline Near-term proof-of-concept readout Existing human clinical data Prophylactic Programs VTP-900 COVID-19 Coronavirus prophylactic Licensed by OUI to AZ Fully approved in EMA/UK VTP-500 MERS prophylactic Worldwide Initiation of Phase 2 VTP-400 Zoster prophylactic Worldwide (excl. China) Initiation of Phase 1 Therapeutic Programs VTP-300 Hepatitis B Virus (HBV) therapeutic Worldwide Phase 2 efficacy data updates (Q4 2023) VTP-200 Human Papillomavirus (HPV) therapeutic Worldwide Phase 1b/2 final data (Q2 2024) VTP-800/850 Prostate cancer therapeutic Worldwide Phase 1/2 trial initiation (H2 2023) VTP-600 NSCLC therapeutic in combo. with checkpoint inhibitor + chemo Worldwide (76% of Sub.) Phase 1/2a currently enrolling VTP-1000 Celiac disease Worldwide IND filing in H2 2023 VTP-1100 HPV Cancer Worldwide IND filing in 2024 |
| VTP-300 Hepatitis B Virus (HBV) Therapeutic |
| Vaccitech Overview 9 Potential for over $1.5B in Annual Sales across US, China and EU6 HBV: Global Long-term Pandemic in Need of a Functional Cure 1 WHO, Hepatitis B, 2022 2 Kosinska AD, et al, Curr Opin Virol. 2017 Apr;23:75-81 3 Boyd A, et al, Viruses. 2021 Jul 11;13(7):1341 4 Broquetas T and Carrion JA, Hepat Med. 2002 Jul;14:87-100 5 Van Zonneveld M, et al, Aliment Pharmacol Ther. 2005 May 1;21(9):1163-71 ~300 MILLION Patients are Chronically Infected with HBV1 Less than 10% get to Functional Cure with existing therapies3 HBV is the eighth most frequent cause of mortality globally2 ~1.4 million deaths per year from acute infection ~820,000 annual deaths due to liver cirrhosis and hepatocellular carcinoma (HCC) 1.5 MILLION new infections per year1 There is significant unmet medical need • Existing therapies typically require chronic treatment • NUCs are slow-acting with low cure efficacy4 • Pegylated Interferon has significant side effects5 Large market opportunity for VTP-300 ~300M Adults chronically infected with HBV1 ~52M 15-20% diagnosed6 ~30M ~10% Treated with current therapies6 There is an urgent need to develop effective therapeutic strategies to cure chronic HBV infection 6 Vaccitech, Data on file NUCs = Nucleos(t)ide analogs |
| Vaccitech Overview VTP-300 Could be Critical Component to a Functional Cure Regimen for HBV Three potential components to a functional cure • VTP-300 has been shown to induce sustained HBsAg reduction • It is likely that functional cure requires a combination of agents with complementary mechanisms of action 10 VTP-300 is designed to boost the host immune system The objective of ongoing clinical trials evaluating VTP-300 combinations in chronic HBV patients is to reach functional cure NUCs + VTP-300 + anti-PD-1 NUCs + siRNA + VTP-300 NUCs + VTP-300 NUCs + siRNA + VTP-300 + anti-PD-1 VTP-300 is an antigen-specific immunotherapy that could be a critical component to enhancing rates of functional cure 1 2 3 Directly lower viral antigen HBsAg burden Inhibit viral DNA cccDNA replication NUCs siRNA Oligo mAbs VTP-300 PD-1 Inhibitors Immunostimulants Stimulate host immune system response |
| Vaccitech Overview 11 HBV002 – Fully enrolled Ph 1b/2a Design Inclusion Criteria • HBV DNA <40 copies • HBsAg <4,000 IU/mL • On NUCs for 1 year Primary Endpoints • Safety and reactogenicity: incidence of AEs and SAEs Secondary Endpoints • Percentage of participants with reduction in HBsAg titre • Percentage of participants with HBsAg Loss • Percentage of participants with reduction of HBV DNA • Magnitude and avidity of HBV-specific CD4+ and magnitude of HBV-specific CD8+ T cells induced by each treatment regimen VTP-300 + Low-Dose Nivolumab (N=55) Objective: Evaluating safety, tolerability and immunogenicity Group 1 (N= 10) Group 2 (N=18) Group 3 (N=18) Group 4 (N=9) Randomization N=55 MVA-HBV ChAdOx1-HBV ChAdOx1-HBV ChAdOx1-HBV + LD Nivo* Day 1 Day 29 MVA-HBV MVA-HBV MVA-HBV + LD Nivo MVA-HBV + LD Nivo* Study reference: NCT04778904 *Nivolumab used with Day 1 and Day 29 doses in Group 4 |
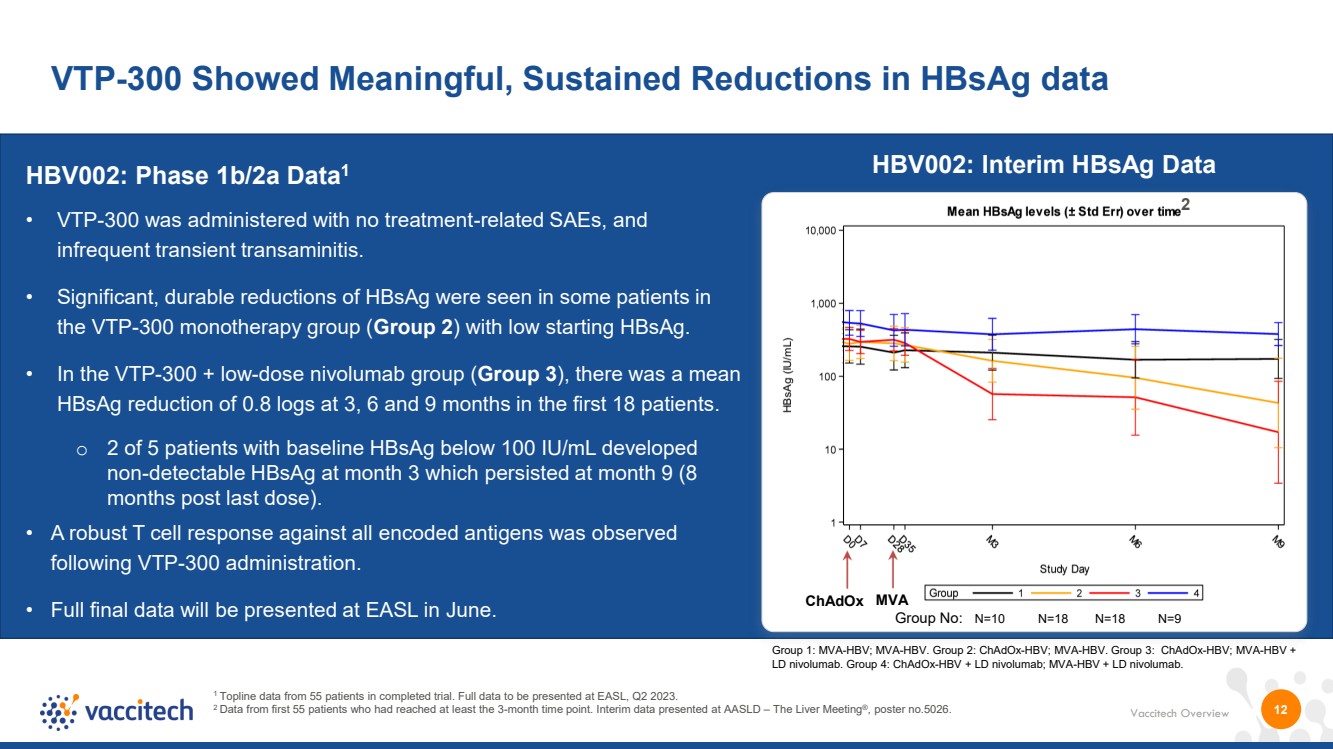
| Vaccitech Overview VTP-300 Showed Meaningful, Sustained Reductions in HBsAg data 1 Topline data from 55 patients in completed trial. Full data to be presented at EASL, Q2 2023. 2 Data from first 55 patients who had reached at least the 3-month time point. Interim data presented at AASLD – The Liver Meeting®, poster no.5026. HBV002: Phase 1b/2a Data1 • VTP-300 was administered with no treatment-related SAEs, and infrequent transient transaminitis. • Significant, durable reductions of HBsAg were seen in some patients in the VTP-300 monotherapy group (Group 2) with low starting HBsAg. • In the VTP-300 + low-dose nivolumab group (Group 3), there was a mean HBsAg reduction of 0.8 logs at 3, 6 and 9 months in the first 18 patients. o 2 of 5 patients with baseline HBsAg below 100 IU/mL developed non-detectable HBsAg at month 3 which persisted at month 9 (8 months post last dose). • A robust T cell response against all encoded antigens was observed following VTP-300 administration. • Full final data will be presented at EASL in June. 12 ChAdOx MVA Group No: N=10 N=18 N=18 N=9 Group 1: MVA-HBV; MVA-HBV. Group 2: ChAdOx-HBV; MVA-HBV. Group 3: ChAdOx-HBV; MVA-HBV + LD nivolumab. Group 4: ChAdOx-HBV + LD nivolumab; MVA-HBV + LD nivolumab. 2 HBV002: Interim HBsAg Data |
| Vaccitech Overview 13 AB-729-202 – Ph 2a Clinical Collaboration with Arbutus Study Design Primary Endpoints • Safety and reactogenicity: incidence of AEs and SAEs Secondary Endpoints • Change in HBsAg concentration from baseline • Proportion of participants with a change in HBsAg from baseline meeting response criteria (≥0.5, 1, 2, or 3 log10 reduction) • Change in HBV DNA, RNA, core-related antigen, HBsAg antibody, HBsAg e-antibody from baseline Inclusion Criteria • HBV DNA ≤20 IU/mL • HBsAg ≥100 to <5,000 IU/mL • On NUCs for 1 year NUC discontinuation (Evaluation at Week 48) • ALT <2 × ULN • HBV DNA < LLOQ • HBeAg negative • HBsAg <100 IU/mL 1 First patient dosed in expansion arm expected in H1 2023 * Additional MVA-HBV/Sham to be dosed at Week 38, if patients are HBsAg <100 IU/mL ** Additional MVA-HBV + Nivo to be dosed at Week 38, if patients are HBsAg <100 IU/mL LD = Low-dose AB-729 + VTP-300 + Low-dose Nivolumab (N=60) Trial expanded in Q4 2022 to include an arm with low-dose Nivolumab1 Week 1 AB-729 (N=60) Randomization Group 1 (N= 20) Group 2 (N=20) Week 24 Week 26 Week 48 (patient to discontinue NUCs if eligible) ChAdOx1-HBV Sham Group 3 (N=20) ChAdOx1-HBV Open Group Week 30 MVA-HBV + LD Nivo** MVA-HBV* Sham* |
| Vaccitech Overview 14 HBV003 – Ph 2b Study Design VTP-300 + Low-Dose Nivolumab (N=120) - Initiated in Q4 2022 Objective: Evaluating Additional Dosing and PD-1 Timing MVA-HBV + LD Nivo Day 1 Group 1 (N= 40) Group 2 (N=40) Group 3 (N=40) ChAdOx1-HBV Day 29 Day 36 Day 85 ChAdOx1-HBV ChAdOx1-HBV MVA-HBV + LD Nivo MVA-HBV MVA-HBV + LD Nivo LD Nivo MVA-HBV Primary Endpoint • Percentage of participants with a greater than 1 log HBsAg reduction at 6 months after initiation of therapy Secondary Endpoints • Safety and reactogenicity: incidence of AEs and SAEs • T cell response Study Reference: NCT05343481 Inclusion Criteria • HBV DNA ≤1,000 IU/mL • HBsAg ≥10 to <4,000 IU/mL • On NUCs for ≥6 months NUC discontinuation • Month 6 • HBsAg <100 IU/mL HBV003 is designed to obtain critical information on treatment dosing regimen • Group 1: Bridge/Mirrors Group 3 in HBV002 to further support response effect observed • Group 2: Assesses if additional dose of MVA-HBV with LD nivolumab at Day 85 further reduces HBsAg • Group 3: Assesses if delaying LD nivolumab until after MVA-HBV is more optimal (plus adds option of 2nd MVA-HBV dose) |
| VTP-200 Human Papillomavirus (HPV) Therapeutic |
| Vaccitech Overview Potential peak annual sales for VTP-200 in persistent HPV of >$1.5B in US and EU56 HPV is the most common sexually transmitted viral infection in the world1 16 Vaccitech is targeting persistent HPV – which can lead to precancerous lesions and cervical cancer1 Persistent HPV Infection Remains a Significant Public Health Problem1 1 WHO, HPV vaccines: WHO position paper, 2022 2 WHO, Cervical Cancer 3 Center for Disease Control 4 Lancet Infect Dis. 2007 Jul;7(7):453-9. 10.1016/S1473-3099(07)70158-5 Approximately 291 million women worldwide are infected with HPV4 Cervical cancer was the 4 th most common cancer in women globally in 20202 .. >95% of cervical cancer is caused by HPV2 Cervical cancer worldwide2 : ~342,000 deaths per year ~604,000 cases per year Cervical cancer in the US3 : ~4,000 deaths per year even with screening & treatment ~12,000 cases per year There is a high unmet need for patients with persistent HPV infection • While HPV prophylactic vaccines are effective at preventing infection, these vaccines do not eliminate existing infections1 • Low vaccination rates in many regions of the world1 • >3.6M diagnosed persistent high-risk cervical HPV in US and across 5EU annually collectively6 • Standard of care is monitoring and excision once high-grade lesions develop1 • Currently no treatment before high-grade lesions develop1 • People with HPV infections report cancer-related fear, worry over lack of treatment and HPV being a ‘ticking time bomb’5 5 Psychooncology. 2021 Jan; 30(1): 84–92. doi: 10.1002/pon.5540 6 Vaccitech Data on File |
| Vaccitech Overview 17 APOLLO (HPV001) – Ph 1b/2 Study Design Main Phase1 : VTP-200 (N=99) – Enrolment complete 1 All groups open simultaneously Study Reference: NCT04607850 Inclusion Criteria • High risk HPV (hrHPV) positive for >6 months and low-grade cervical lesions Study Outputs • Efficacy Data: % clearance of high-risk HPV and cervical lesions evaluated at 12 months. • Interim data analysis: Interim data showed VTP-200 was generally well tolerated with no product-related serious adverse events and had encouraging initial immunogenicity results. The trial will continue as planned to the 12-month primary endpoint. • Final data analysis: Expected Q2 2024. Lead-in Phase: (N=9) Objective: Evaluating immunogenicity, safety data Objective: Evaluating safety data, efficacy data, immunogenicity, dose-response Group A (N=3) ChAdOx-HPV 2 x 108 vp MVA-HPV 1 x 107 pfu Group B (N=3) ChAdOx-HPV 2 x 109 vp MVA-HPV 1 x 10 7 pfu Group C (N=3) ChAdOx-HPV 2 x 1010 vp MVA-HPV 1 x 108 pfu Regions EU UK Group 1 (N=16) Group 2 (N=16) Group 3 (N=8) Group 4 (N=8) Group 5 (N=16) Group 6 (N=32) ChAdOx-HPV 2 x 109 vp MVA-HPV 1 x 107 pfu Day 1 ChAdOx-HPV 2 x 1010 vp ChAdOx-HPV 2 x 108 vp ChAdOx-HPV 2 x 109 vp ChAdOx-HPV 2 x 1010 vp Placebo MVA-HPV 1 x 107 pfu MVA-HPV 1 x 108 pfu MVA-HPV 1 x 108 pfu MVA-HPV 1 x 108 pfu Placebo Day 29 |
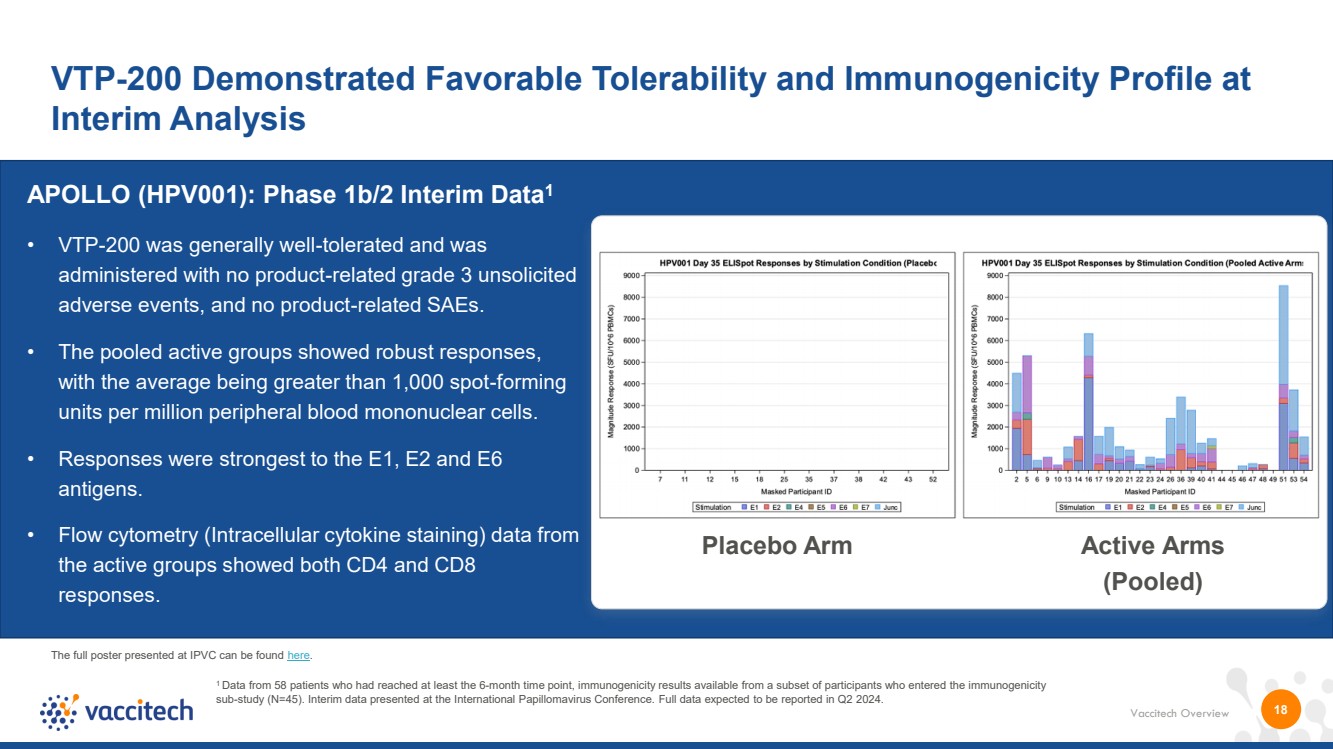
| Vaccitech Overview VTP-200 Demonstrated Favorable Tolerability and Immunogenicity Profile at Interim Analysis VTP-300 18 • VTP-200 was generally well-tolerated and was administered with no product-related grade 3 unsolicited adverse events, and no product-related SAEs. • The pooled active groups showed robust responses, with the average being greater than 1,000 spot-forming units per million peripheral blood mononuclear cells. • Responses were strongest to the E1, E2 and E6 antigens. • Flow cytometry (Intracellular cytokine staining) data from the active groups showed both CD4 and CD8 responses. Placebo Arm Active Arms (Pooled) APOLLO (HPV001): Phase 1b/2 Interim Data1 1 Data from 58 patients who had reached at least the 6-month time point, immunogenicity results available from a subset of participants who entered the immunogenicity sub-study (N=45). Interim data presented at the International Papillomavirus Conference. Full data expected to be reported in Q2 2024. The full poster presented at IPVC can be found here. |
| Cancer Programs |
| Vaccitech Overview VTP-600: Non-Small Cell Lung Cancer (NSCLC) Novel Immunotherapy Candidate Targeting MAGE-A3 & NY-ESO-1 1World Cancer Research Fund International 2Johns Hopkins Medicine, Prostate Cancer Prognosis 3American Cancer Society, Global Cancer Facts and Figures 4th Edition 20 Opportunity: Prostate Cancer is the 2nd Most Common Cancer Diagnosis in Men: ~1.4M1 new cases diagnosed and ~375k deaths occurred in 2020 (worldwide). 20-30% of patients experience rising levels of PSA after local therapy (e.g., prostatectomy)2 , indicating that disease was not cured by local therapy. Systemic therapy generally required, resulting in toxicity and side effects. Our Solution: VTP-850 is a next generation ChAdOx1-MVA multi-antigen product candidate designed to induce a polyclonal T cell response to kill tumor cells and prevent advancement to metastatic disease. Status: Two completed studies with first generation product (VTP-800) containing single antigen (5T4). VTP-850 Phase 1/2 study expected to enrol first patient in H2 2023 (n=137)*. Opportunity: Lung cancer is the most common cause of cancer deaths worldwide (in 2018, ~2.1M cases of NSCLC & ~1.8M deaths)3 .. Better combination approaches are needed in first-line Standard-of-Care to improve long-term outcomes. Our Solution: VTP-600 has induced high levels of T cells that target MAGE-A3 and NY-ESO-1, leading to killing of tumor cells in pre-clinical trials. Status: Partnership with Ludwig Institute for Cancer Research by way of Vaccitech Oncology Limited (“VOLT”). Phase 1/2a in collaboration with Cancer Research UK (CRUK) underway with first patient dosed Q4 2021. Clinical-Stage Cancer Programs in Areas of High Unmet Need Heterologous approach paradigm utilizing ChAdOx-MVA PSA: Prostate Specific Antigen *Study Reference: NCT05617040 VTP-850: Prostate Cancer Novel Immunotherapy Candidate to Prevent Advanced Disease |
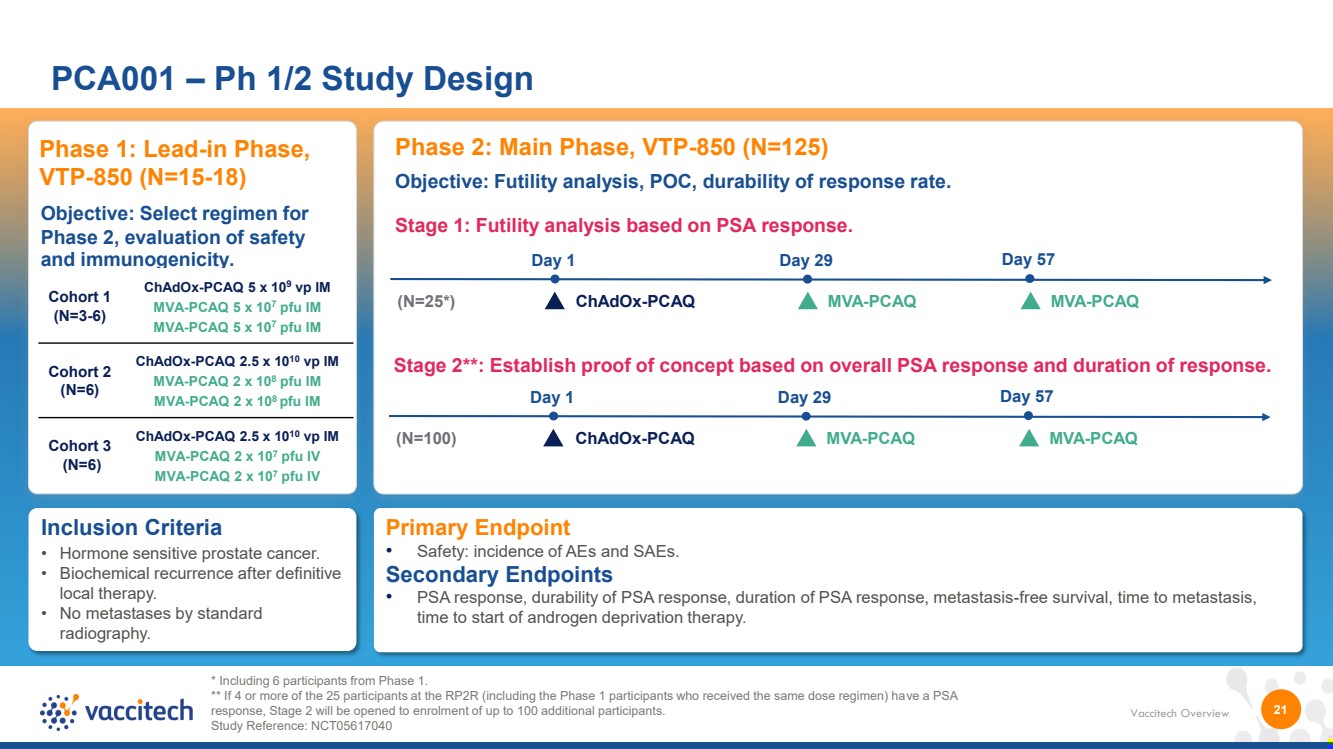
| Vaccitech Overview 21 PCA001 – Ph 1/2 Study Design * Including 6 participants from Phase 1. ** If 4 or more of the 25 participants at the RP2R (including the Phase 1 participants who received the same dose regimen) have a PSA response, Stage 2 will be opened to enrolment of up to 100 additional participants. Study Reference: NCT05617040 Inclusion Criteria • Hormone sensitive prostate cancer. • Biochemical recurrence after definitive local therapy. • No metastases by standard radiography. Phase 1: Lead-in Phase, VTP-850 (N=15-18) Objective: Select regimen for Phase 2, evaluation of safety and immunogenicity. Cohort 1 (N=3-6) ChAdOx-PCAQ 5 x 109 vp IM MVA-PCAQ 5 x 107 pfu IM MVA-PCAQ 5 x 107 pfu IM Cohort 2 (N=6) ChAdOx-PCAQ 2.5 x 1010 vp IM MVA-PCAQ 2 x 10 8 pfu IM MVA-PCAQ 2 x 10 8 pfu IM Cohort 3 (N=6) ChAdOx-PCAQ 2.5 x 1010 vp IM MVA-PCAQ 2 x 107 pfu IV MVA-PCAQ 2 x 10 7 pfu IV Phase 2: Main Phase, VTP-850 (N=125) Stage 1: Futility analysis based on PSA response. (N=25*) ChAdOx-PCAQ MVA-PCAQ Day 1 Day 29 Objective: Futility analysis, POC, durability of response rate. Day 57 MVA-PCAQ Stage 2**: Establish proof of concept based on overall PSA response and duration of response. (N=100) ChAdOx-PCAQ MVA-PCAQ Day 1 Day 29 Day 57 MVA-PCAQ Primary Endpoint • Safety: incidence of AEs and SAEs. Secondary Endpoints • PSA response, durability of PSA response, duration of PSA response, metastasis-free survival, time to metastasis, time to start of androgen deprivation therapy. |
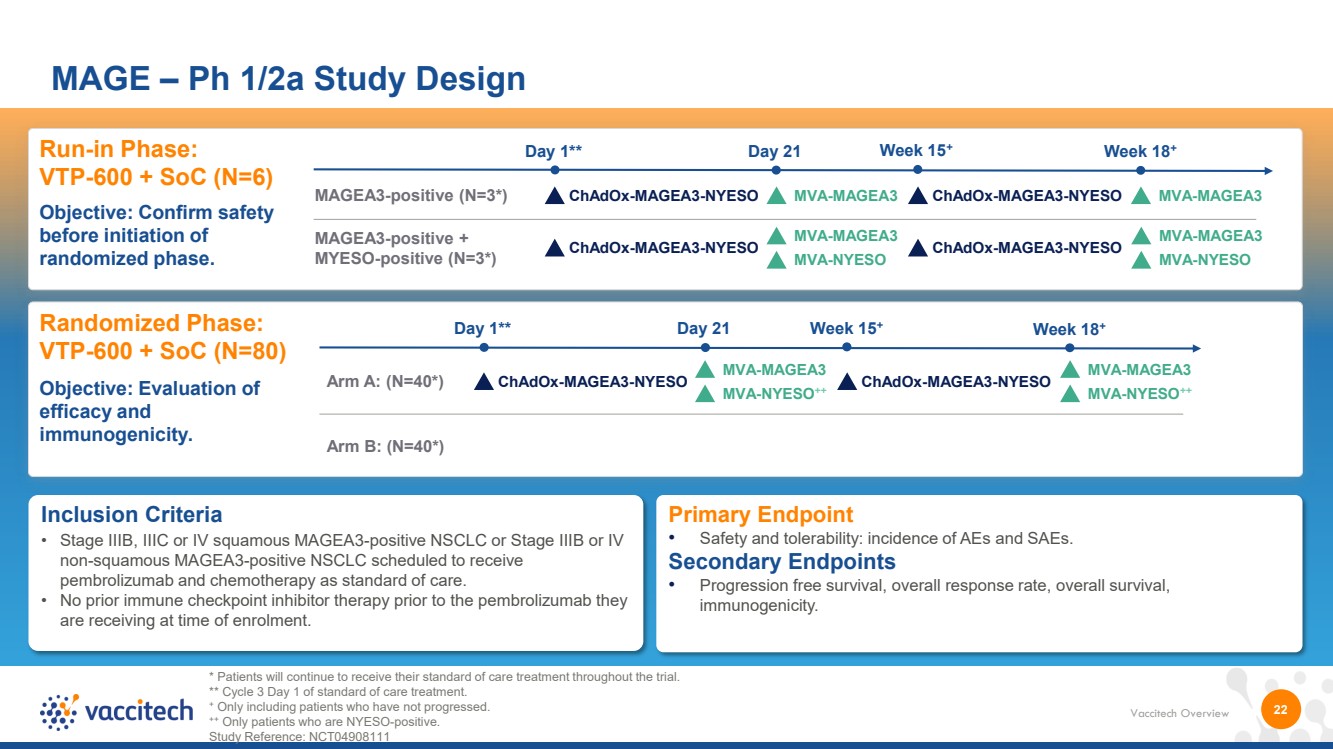
| Vaccitech Overview 22 MAGE – Ph 1/2a Study Design Inclusion Criteria • Stage IIIB, IIIC or IV squamous MAGEA3-positive NSCLC or Stage IIIB or IV non-squamous MAGEA3-positive NSCLC scheduled to receive pembrolizumab and chemotherapy as standard of care. • No prior immune checkpoint inhibitor therapy prior to the pembrolizumab they are receiving at time of enrolment. Run-in Phase: VTP-600 + SoC (N=6) Objective: Confirm safety before initiation of randomized phase. Randomized Phase: VTP-600 + SoC (N=80) Objective: Evaluation of efficacy and immunogenicity. Primary Endpoint • Safety and tolerability: incidence of AEs and SAEs. Secondary Endpoints • Progression free survival, overall response rate, overall survival, immunogenicity. Arm A: (N=40*) ChAdOx-MAGEA3-NYESO Day 1** Day 21 Week 15+ Arm B: (N=40*) ChAdOx-MAGEA3-NYESO Week 18+ MAGEA3-positive (N=3*) ChAdOx-MAGEA3-NYESO MVA-MAGEA3 Day 1** Day 21 Week 15+ ChAdOx-MAGEA3-NYESO MVA-MAGEA3 Week 18+ * Patients will continue to receive their standard of care treatment throughout the trial. ** Cycle 3 Day 1 of standard of care treatment. + Only including patients who have not progressed. ++ Only patients who are NYESO-positive. Study Reference: NCT04908111 MAGEA3-positive + MYESO-positive (N=3*) ChAdOx-MAGEA3-NYESO ChAdOx-MAGEA3-NYESO MVA-MAGEA3 MVA-NYESO MVA-MAGEA3 MVA-NYESO MVA-MAGEA3 MVA-NYESO++ MVA-MAGEA3 MVA-NYESO++ |
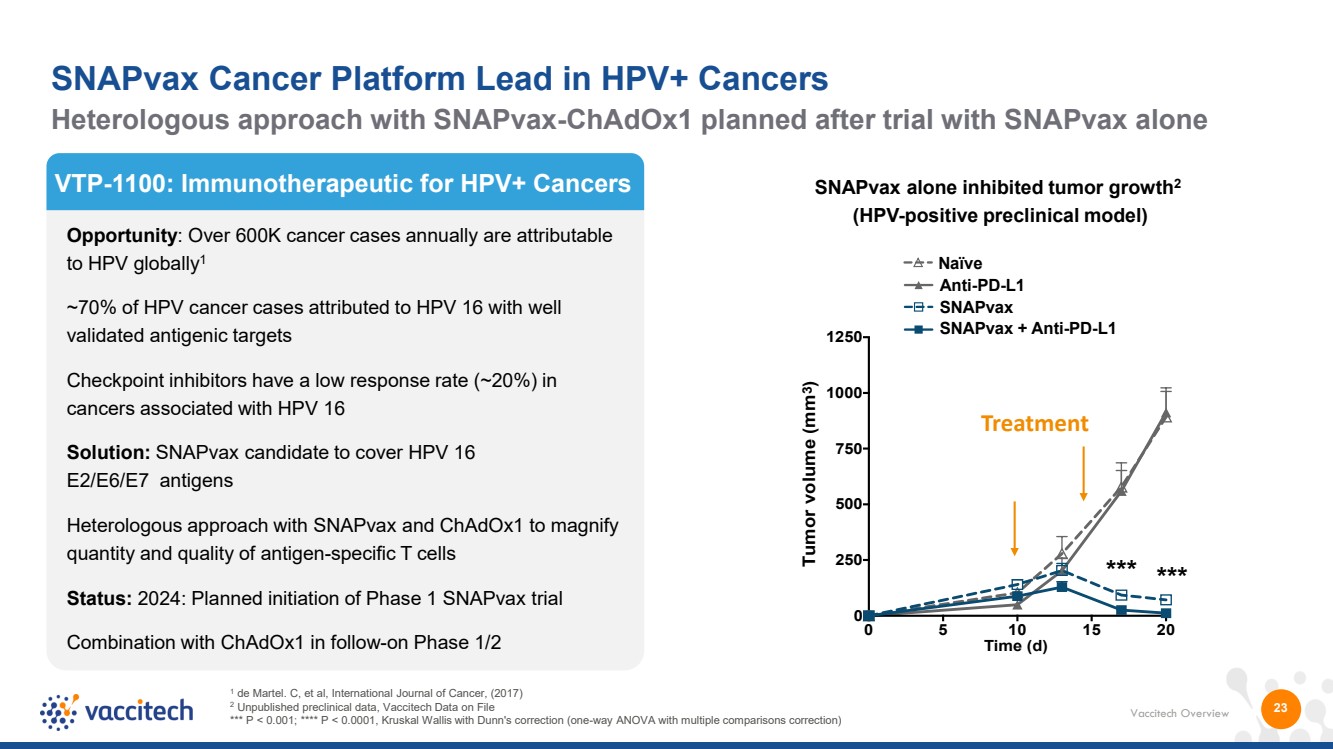
| Vaccitech Overview 23 VTP-1100: Immunotherapeutic for HPV+ Cancers Opportunity: Over 600K cancer cases annually are attributable to HPV globally1 ~70% of HPV cancer cases attributed to HPV 16 with well validated antigenic targets Checkpoint inhibitors have a low response rate (~20%) in cancers associated with HPV 16 Solution: SNAPvax candidate to cover HPV 16 E2/E6/E7 antigens Heterologous approach with SNAPvax and ChAdOx1 to magnify quantity and quality of antigen-specific T cells Status: 2024: Planned initiation of Phase 1 SNAPvax trial Combination with ChAdOx1 in follow-on Phase 1/2 1 de Martel. C, et al, International Journal of Cancer, (2017) 2 Unpublished preclinical data, Vaccitech Data on File *** P < 0.001; **** P < 0.0001, Kruskal Wallis with Dunn's correction (one-way ANOVA with multiple comparisons correction) SNAPvax alone inhibited tumor growth2 (HPV-positive preclinical model) 0 5 10 15 20 0 250 500 750 1000 1250 *** *** Time (d) Tumor volume (mm3) Naïve SNAPvax Anti-PD-L1 SNAPvax + Anti-PD-L1 Treatment Heterologous approach with SNAPvax-ChAdOx1 planned after trial with SNAPvax alone SNAPvax Cancer Platform Lead in HPV+ Cancers |
| Autoimmune Program |
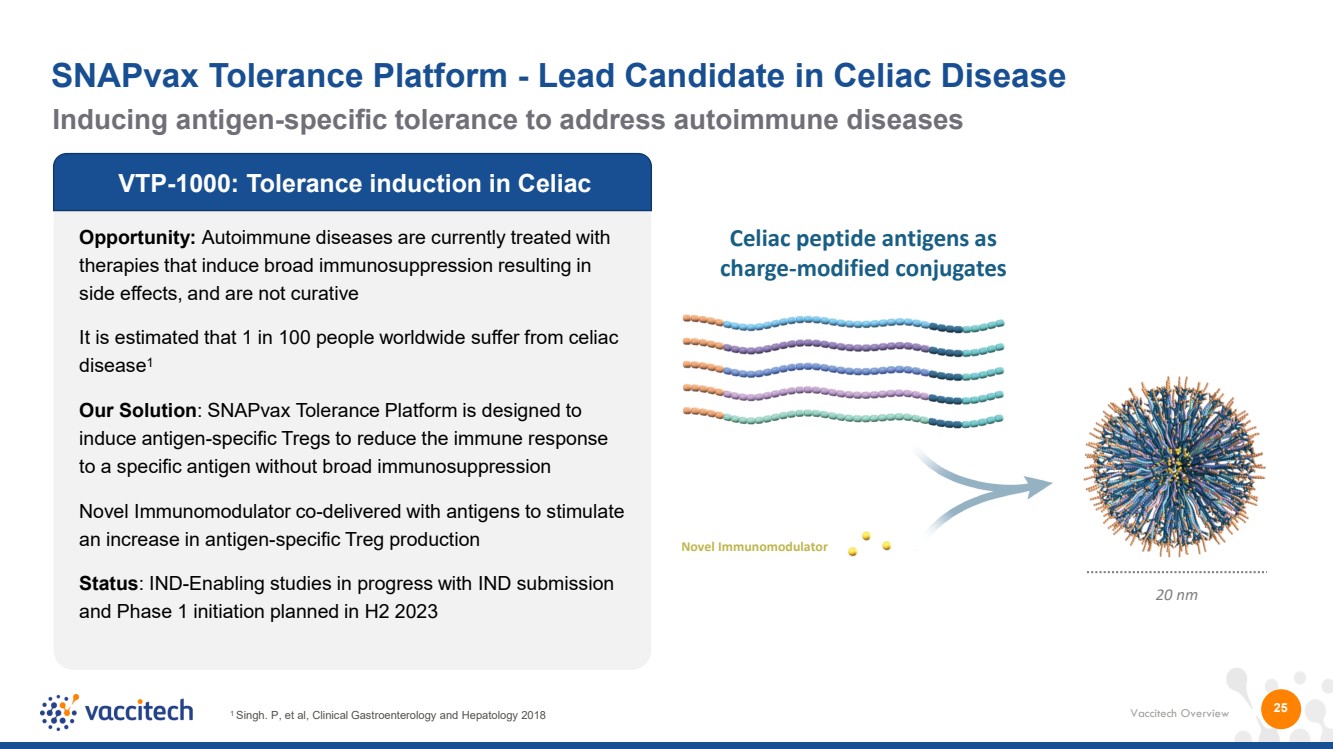
| Vaccitech Overview 25 VTP-1000: Tolerance induction in Celiac Opportunity: Autoimmune diseases are currently treated with therapies that induce broad immunosuppression resulting in side effects, and are not curative It is estimated that 1 in 100 people worldwide suffer from celiac disease1 Our Solution: SNAPvax Tolerance Platform is designed to induce antigen-specific Tregs to reduce the immune response to a specific antigen without broad immunosuppression Novel Immunomodulator co-delivered with antigens to stimulate an increase in antigen-specific Treg production Status: IND-Enabling studies in progress with IND submission and Phase 1 initiation planned in H2 2023 1 Singh. P, et al, Clinical Gastroenterology and Hepatology 2018 20 nm Celiac peptide antigens as charge-modified conjugates Novel Immunomodulator Inducing antigen-specific tolerance to address autoimmune diseases SNAPvax Tolerance Platform - Lead Candidate in Celiac Disease |
| Vaccitech Overview 26 Vaccitech & Oxford University Innovation (OUI) Co-Invented AstraZeneca (AZ) COVID-19 Vaccine ChAdOx Technology & Processes Validated in COVID-19 Sources: “BNT162b2 mRNA covid-19 vaccine in a nationwide mass vaccination setting”, by N. Dagan et al., 2021; studies run by Public Health England and Public Health Scotland; company press releases; The Economist 1) We are not party to the license agreement between OUI (Oxford University Innovation) and AZ (AstraZeneca) (the License Agreement), and our understanding of the License Agreement is based solely on an extract of the agreement provided by parties to that agreement. In addition, no party to the License Agreement has confirmed that there are no material terms in that agreement that are not included in the description above or that could adversely impact the economic and other terms of the License Agreement described above. Moreover, there can be no assurance that the License Agreement is an enforceable agreement, that the parties will comply with their obligations under that agreement (including any obligations of AZ to make milestone or royalty payments to OUI), that the License Agreement will not be terminated pursuant to its terms or otherwise, or that the terms of that agreement (including royalty rates and other economic terms) will not be modified by the parties in the future. Accordingly, these and other factors could cause amounts received by OUI pursuant to the License Agreement, and accordingly any share of the revenue under that agreement that we may receive, to differ from those described above, and any such differences could be material. Estimates of hospitalizations avoided make the simplifying assumption (that vaccines prevent death and hospitalizations by preventing infections https://jamanetwork.com/journals/jamanetworkopen/fullarticle/2793913) and that the death rate from hospitalization is the same in regions where AZ vaccine was administered and the US. The CDC estimates that the death rate from hospitalization in the US during late omicron was 4.9% and delta was 15.1%. Estimated range = 6 million deaths * [1/.049 hospitalization/death to 1/.151 hospitalizations/death]. Vaccitech co-invented the AZ COVID-19 vaccine, going from construct design to GMP, in 4 months in 2020 • More than 3 billion doses delivered to 180 countries by end of 2022 • Over 6 million lives saved worldwide (estimated) • Between 37.7 and 122.4 million hospitalizations prevented (estimated) • OUI entered into research collaboration and worldwide license agreement with AZ • Post-pandemic license structure includes milestones and royalties, approximately 1.4% of net sales • Royalty revenue payments commenced in 2022 • Revenue recognized in FY22 amounting to $43.7 million OUI / AZ Deal Summary Vaxzevria COVID-19 vaccine |
| Vaccitech Overview 27 Financial Overview & Catalysts Current cash position $191.3 million as of March 31, 2023 Estimated cash runway into the first quarter of 2025 Estimated cash burn (gross) of $23-$25 million per quarter in 2023 No debt or outstanding warrants Expected near-term catalysts VTP-850: Phase 1/2 PCA001 clinical trial FPFV expected VTP-300: Phase 1/2a HBV002 final results expected at EASL VTP-1000: IND filing expected VTP-300: AB-729-202 interim efficacy data expected VTP-300: Phase 2b HBV003 interim efficacy data expected Q2 2023 H2 2023 Based on management estimates. |
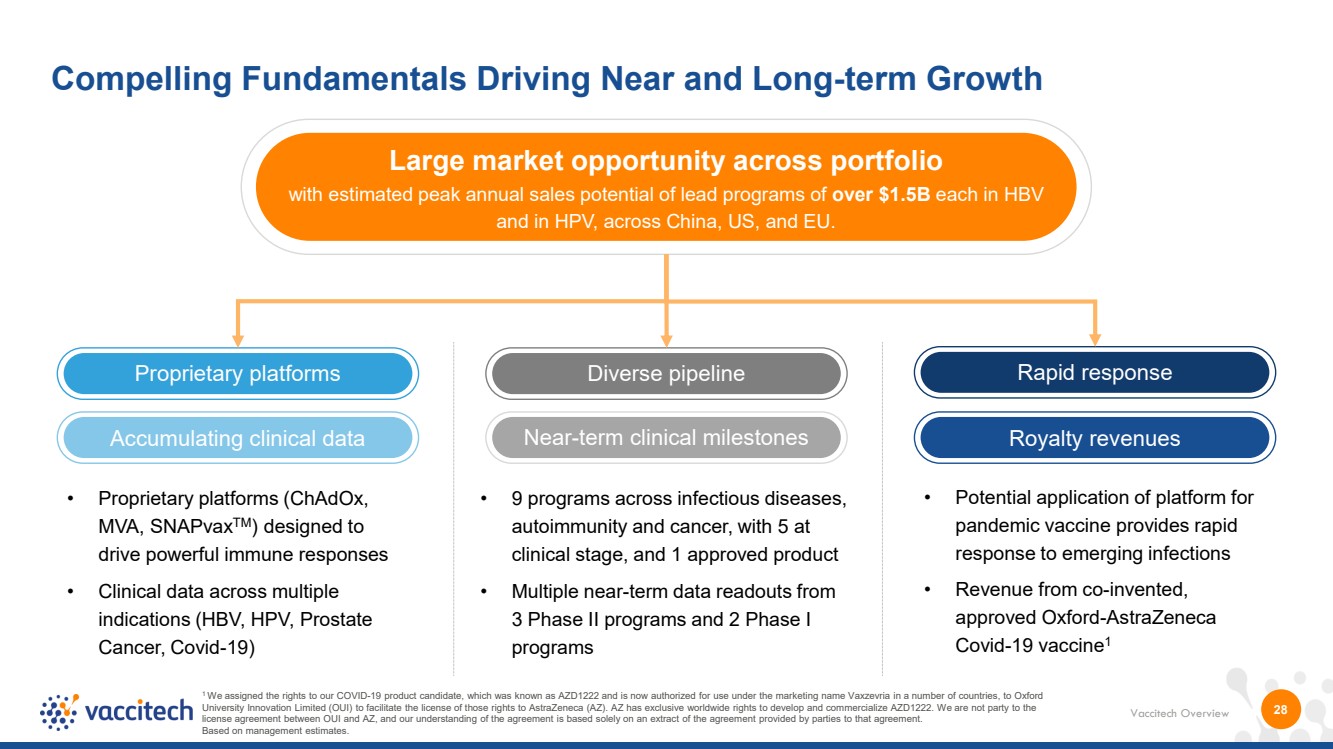
| Vaccitech Overview Large market opportunity across portfolio with estimated peak annual sales potential of lead programs of over $1.5B each in HBV and in HPV, across China, US, and EU. Near-term clinical milestones Proprietary platforms Diverse pipeline 28 Compelling Fundamentals Driving Near and Long-term Growth 1 We assigned the rights to our COVID-19 product candidate, which was known as AZD1222 and is now authorized for use under the marketing name Vaxzevria in a number of countries, to Oxford University Innovation Limited (OUI) to facilitate the license of those rights to AstraZeneca (AZ). AZ has exclusive worldwide rights to develop and commercialize AZD1222. We are not party to the license agreement between OUI and AZ, and our understanding of the agreement is based solely on an extract of the agreement provided by parties to that agreement. Based on management estimates. Rapid response Accumulating clinical data Royalty revenues • Proprietary platforms (ChAdOx, MVA, SNAPvaxTM) designed to drive powerful immune responses • Clinical data across multiple indications (HBV, HPV, Prostate Cancer, Covid-19) • 9 programs across infectious diseases, autoimmunity and cancer, with 5 at clinical stage, and 1 approved product • Multiple near-term data readouts from 3 Phase II programs and 2 Phase I programs • Potential application of platform for pandemic vaccine provides rapid response to emerging infections • Revenue from co-invented, approved Oxford-AstraZeneca Covid-19 vaccine1 |
| Inducing T Cells to Treat and Prevent Disease Thank you |