Exhibit 99.2
MANAGEMENT’S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF
OPERATIONS FOR THE THREE MONTHS ENDED MARCH 31, 2021 AND 2020.
This management discussion and analysis (“MD&A”) of the financial condition and results of operations of Cresco Labs Inc. (the “Company” or “Cresco Labs”) is for the three months ended March 31, 2021 and 2020. The Company’s interim financial statements are prepared in accordance with accounting principles generally accepted in the United States of America (“U.S. GAAP”). Prior period amounts included in the MD&A have been recast and adjusted to update for historical changes necessary to present the financial results in accordance with U.S. GAAP. It is supplemental to, and should be read in conjunction with, the Company’s audited Consolidated Financial Statements and accompanying notes as of and for the years ended December 31, 2020 and 2019 prepared in accordance with International Financial Reporting Standards (“IFRS”) previously filed on SEDAR. Financial information presented in this MD&A is presented in United States dollars (“$”), unless otherwise indicated. The three months ended data presented below is unaudited.
The Company has provided certain supplemental non-GAAP financial measures in this MD&A. Where the Company has provided such non-GAAP financial measures, we have also provided a reconciliation to the most comparable U.S. GAAP financial measure. These supplemental non-GAAP financial measures should not be considered superior to, as a substitute for or as an alternative to, and should only be considered in conjunction with, the U.S. GAAP financial measures presented herein. Please see the information under the header “Non-GAAP Financial Measures” for additional information on the Company’s use of non-GAAP financial measures and, the reasons therefore.
This MD&A contains certain “forward-looking statements” and certain “forward-looking information” as defined under applicable United States securities laws. Please refer to the discussion of forward-looking statements and information set out under the heading “Cautionary Note Regarding Forward Looking Information,” located at the beginning of the Company’s Annual Information Form for the year ended December 31, 2020, filed on SEDAR. As a result of many factors, the Company’s actual results may differ materially from those anticipated in these forward- looking statements and information. Please refer to the discussion of risks and uncertainties set out under the heading “Risk Factors,” located within the Company’s Annual Information Form for the year ended December 31, 2020, filed on SEDAR and other filings the Company makes on SEDAR.
OVERVIEW OF THE COMPANY
Cresco Labs Inc. (the “Company” or “Cresco Labs”) was incorporated in the Province of British Columbia and is licensed to cultivate, manufacture and sell cannabis and cannabis products. The Company operates in and/or has ownership interests in Illinois, Pennsylvania, Ohio, California, Nevada, Arizona, Maryland, Massachusetts, New York, Michigan, and Florida.
Cresco Labs is primarily engaged in the business of cultivating medical grade cannabis, manufacturing medical grade products derived from cannabis cultivation, and distributing such products to medical or adult-use consumers in legalized cannabis markets. Cresco Labs exists to provide high-quality and consistent cannabis-based products to consumers. Cresco Labs’ business focuses on regulatory compliance while working to develop condition-specific strains of cannabis and non-invasive delivery methods (alternatives to smoke inhalation) to provide controlled- dosage medicinal cannabis relief to qualified patients and consumers in legalized cannabis markets. The Company currently operates three (3) medical and adult-use cannabis cultivation and manufacturing centers and ten (10) dispensary locations in Illinois; one (1) medical cannabis cultivation and manufacturing center and three (3) dispensary locations in Pennsylvania; one (1) medical cannabis cultivation center and five (5) dispensary location in Ohio; three (3) cultivation centers, one (1) manufacturing facility and two (2) distribution facilities in California; one (1) cultivation center and one (1) cultivation and manufacturing center and dispensary location in Arizona; one (1) processing center in Maryland; one (1) medical cannabis manufacturing center and four (4) dispensary locations in New York; one (1) cultivation and manufacturing center and dispensary in Massachusetts; one (1) processing facility in Michigan; eight (8) dispensary locations and one (1) medical cannabis cultivation and manufacturing center in Florida. For additional information on wholly-owned or effectively controlled subsidiaries and affiliates of Cresco Labs, refer to Note 2 under the heading “Basis of Consolidation” of the Company’s Consolidated Financial Statements for the years ended December 31, 2020 and 2019.
1
During 2019, the Company announced a new dispensary brand, Sunnyside*, created to accelerate industry growth and shift consumer expectations and perceptions around shopping for cannabis from intimidation and doubt to curiosity and acceptance through a new trial and marketing approach. During 2020, five (5) Sunnyside* dispensaries opened in the Illinois market, four (4) dispensaries were rebranded as Sunnyside* in New York, one (1) dispensary was rebranded as Sunnyside* in Arizona, one (1) dispensary was rebranded as Sunnyside* in Massachusetts, and three (3) dispensaries were rebranded as Sunnyside* in Pennsylvania. Cresco Labs’ portfolio of owned cannabis consumer packaged goods includes CrescoTM, Cresco ReserveTM, High SupplyTM, Mindy’sTM, Good NewsTM, RemediTM, Wonder Wellness Co.TM and FloraCalR Farms. The Company distributes and markets these products primarily to third-party licensed retail cannabis stores across the United States as well as to Cresco Labs-owned retail stores. During 2021, the Company closed its acquisition of four (4) dispensaries in Ohio previously operated by Verdant Creations, LLC and its affiliates (collectively “Verdant”).
Cresco Labs is currently located at Suite 110, 400 W. Erie St, Chicago, IL 60654 and employs approximately 2,700 people, while being named as a “Top Diversity Employer” by Diversity Jobs in 2021.
Issuing IPO, Reverse Takeover & Corporate Structure
The Company (then Randsburg Gold Corporation) was incorporated in the Province of British Columbia under the Company Act (British Columbia) on July 6, 1990. On December 30, 1997, the Company changed its name from Randsburg Gold Corporation to Randsburg International Gold Corp. (“Randsburg”) and consolidated its common shares on a five (5) old for one (1) new basis. On November 30, 2018, in connection with the Reverse Takeover, the Company (i) consolidated its outstanding Randsburg common shares on an 812.63 old for one (1) new basis, and (ii) filed an alteration to its Notice of Articles with the British Columbia Registrar of Companies to change its name from Randsburg to Cresco Labs Inc. and to amend the rights and restrictions of its existing class of common shares, redesignate such class as the class of Subordinate Voting Shares (“SVS”) and create the Proportionate Voting Shares and the Super Voting Shares.
Pursuant to the Transaction, among the Company (then Randsburg) and Cresco Labs, LLC, a series of transactions were completed on November 30, 2018 resulting in a reorganization of Cresco Labs and Randsburg in which Randsburg became the indirect parent and sole voting unitholder of Cresco Labs. The transaction constituted a reverse takeover of Randsburg by Cresco Labs under applicable securities laws. Cresco Labs was formed as a limited liability company under the laws of the state of Illinois on October 8, 2013 and is governed by the Pre- Combination LLC Agreement. The Pre-Combination LLC Agreement was further amended and restated in connection with the completion of the Transaction. The Company’s registered office is located at Suite 2200, 1055 West Hastings Street, Vancouver, BC V6E 2E9.
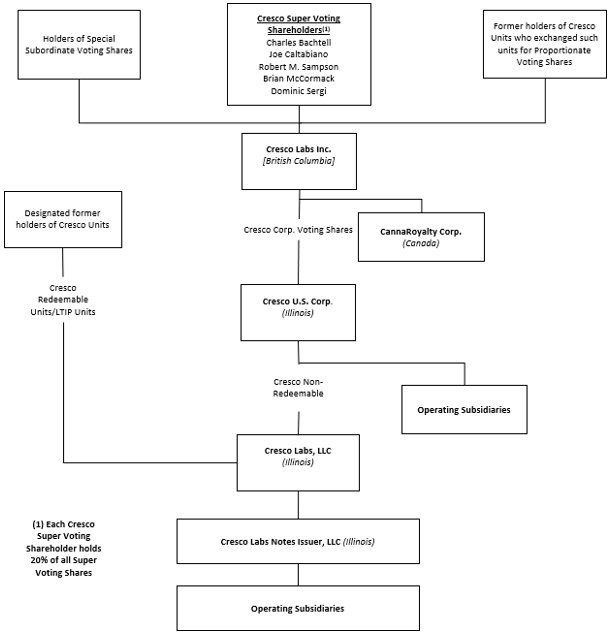
Set forth below is the organization chart of the Company.
2
Recent Developments
On February 2, 2020, the Company closed on a senior secured term loan agreement (the “Term Loan”) for an aggregate principal amount of $100.0 million, with the mutual option to increase the principal amount to $200.0 million. The Tranche A and Tranche B Commitments had a stated maturity of July 2021 and January 2022, respectively.
In conjunction with its January 8, 2020 acquisition of CannaRoyalty Corp. d/b/a Origin House (“Origin House”), the Company recorded a short-term liability with Opaskwayak Cree Nation (the “OCN Loan”) with an aggregate balance of $22.0 million in Canadian dollars as of the acquisition date, subject to a 10.0% interest rate and a stated maturity of June 2020. During the year ended December 31, 2020, the OCN Loan was amended to extend the maturity date to June 30, 2021.
3
On December 11, 2020, the Company entered into an amendment to extend the maturity of the Term Loan and exercised the option to increase the principal amount to $200.0 million (the “Amended Term Loan”). The Amended Term Loan includes principal amounts of $11.7 million, subject to a 12.7% interest rate and a stated maturity of July 2021, and $91.0 million, subject to a 12.0% interest rate and a stated maturity of January 2023, including the conversion of $15.0 million of the OCN loan to the long-term portion of the Amended Term Loan, with the remaining $5.4 million settled in conjunction with the closing of the Amended Term Loan.
On January 14, 2021, the Company announced the commencement of a best efforts overnight marketed offering (the “January 2021 Offering”) of SVS. On January 15, 2021, the Company closed the January 2021 Offering of 9.9 million SVS at a price of C$16.00 per share for total gross proceeds of approximately $125.0 million. The SVS were offered in each of the Provinces of Canada, other than Québec, pursuant to a prospectus supplement dated January 19, 2021 to the Company’s base shelf prospectus dated July 25, 2019 and in the United States on a private placement basis to “qualified institutional buyers.”
On January 13, 2021, the Company filed a Form 40-F with the United States (“U.S.”) Securities and Exchange Commission (“SEC”).
On January 14, 2021, the Company entered into a definitive agreement with Bluma Wellness Inc. (“Bluma”) (the “Bluma Agreement”), pursuant to which Cresco Labs will acquire all of the issued and outstanding shares of Bluma in an all-share transaction that values Bluma at an equity value of $213 million (the “Bluma Transaction”), or $1.12 per Bluma share. Under the terms of the Bluma Agreement, holders of common shares of Bluma will receive 0.0859 subordinate voting shares of Cresco Labs for each Bluma share. On March 15, 2021, Cresco Labs agreed to extend $7.5 million to One Plant Florida (“One Plant”), Bluma’s operating subsidiary. The proceeds of the loan will be used for the expansion of One Plant’s operations in Florida and to satisfy tax liabilities relating to the settlement of vested restricted share units. The acquisition closed on April 14, 2021.
On March 1, 2021, the Company filed and received a receipt for a preliminary short form base shelf prospectus (the “Shelf Prospectus”) with the securities commissions in each of the provinces of Canada, except Québec, and filed a corresponding shelf registration statement on Form F-10 (the “Registration Statement”) with the SEC under the U.S./Canada Multijurisdictional Disclosure System (“MJDS”). The Shelf Prospectus and Registration Statement, was made effective on April 23, 2021 and will allow the Company to offer up to $1.0 billion SVS, debt securities, subscription receipts, warrants, and units, or any combination thereof, from time to time during the 25-month period that the 2021 Shelf Prospectus is effective (subject to MJDS eligibility). The Company filed the 2021 Shelf Prospectus in order to maintain financial strength and flexibility.
On March 18, 2021, the Company entered into a definitive agreement to acquire all of the issued and outstanding equity interests in Cultivate Licensing LLC and BL Real Estate LLC (collectively, “Cultivate”), a vertically integrated Massachusetts operator, for maximum consideration of up to $158.0 million (the “Purchase Price”). A portion of the Purchase Price is payable upon closing of $15.0 million cash consideration and Cresco Labs shares having an equity value equal to $75.0 million. The remaining portion of the Purchase Price will be structured as an earnout of up to $68.0 million in which approximately 12.5% of earnout payments shall be paid in cash and the remaining portion satisfied through issuance of Cresco Labs shares (the “Cultivate Transaction”). The Cultivate Transaction is expected to close in the fourth quarter of 2021.
On March 30, 2021, the Company completed the divestiture of all of its equity interest in the 180 Smoke business (“180 Smoke”) and related intercompany receivables to Spyder Cannabis Inc. and Plant-Based Investment Corp. for approximately $1.1 million, after certain adjustments. The sale resulted in a loss of $3.3 million, plus an additional loss of $0.3 million for accumulated foreign currency translation loss previously included in Other comprehensive loss.
On April 26, 2021, the Company announced a final base shelf prospectus, replacing the Company’s prior shelf prospectus, has been receipted with the securities commissions in each of the provinces of Canada. A corresponding registration statement on Form F-10 has been deemed effective by the U.S. Securities and Exchange Commission under the U.S./Canada MJDS. Additionally, the Company announced a new equity distribution agreement with Canaccord Genuity Corp. to replace the equity distribution agreement filed on December 2019 due to the expiration of the prior shelf prospectus. Pursuant to this agreement, the Company may, from time to time, sell up to $100 million of its subordinate voting shares in Canada.
4
Federal Regulatory Environment
Canadian-Securities Administrators Staff Notice 51-352 (Revised) – Issuers with U.S. Marijuana-Related Activities (“Staff Notice 51-352”) provides specific disclosure expectations for issuers that currently have, or are in the process of developing, cannabis-related activities in the United States as permitted within a particular state’s regulatory framework. All issuers with United States cannabis-related activities are expected to clearly and prominently disclose certain prescribed information in prospectus filings and other required disclosure documents.
In accordance with Staff Notice 51-352, Cresco Labs will evaluate, monitor and reassess the disclosure contained herein, and any related risks, on an ongoing basis and the same will be supplemented, amended and communicated to investors in public filings, including in the event of government policy changes or the introduction of new or amended guidance, laws or regulations regarding marijuana regulation. As a result of the Company’s operations, it is subject to Staff Notice 51-352 and accordingly provides the following disclosure:
Cresco Labs currently directly derives a substantial portion of its revenues from the cannabis industry in certain U.S. states, which industry is illegal under U.S. Federal Law. As of March 31, 2021, the Company is directly involved (through licensed subsidiaries) in both the adult-use and medical cannabis industry in the states of Illinois, Pennsylvania, Ohio, Arizona, Maryland, California, Michigan, New York and Massachusetts as permitted within such states under applicable state law which states have regulated such industries.
The cultivation, sale and use of cannabis is illegal under federal law pursuant to the U.S. Controlled Substance Act of 1970 (the “CSA”). Under the CSA, the policies and regulations of the U.S. Federal Government and its agencies are that cannabis has no medical benefit and a range of activities including cultivation and the personal use of cannabis is prohibited. The Supremacy Clause of the U.S. Constitution establishes that the U.S. Constitution and federal laws made pursuant to it are paramount and in case of conflict between federal and state law, the federal law shall apply.
On January 4, 2018, former U.S. Attorney General Jeff Sessions issued a memorandum to U.S. district attorneys which rescinded previous guidance from the U.S. Department of Justice specific to cannabis enforcement in the United States, including the Cole Memo. The Cole Memo previously provided guidance to prioritize a limited scope of federal enforcement including the prevention of the distribution of marijuana to minors, revenue from the sale of marijuana from going to criminal enterprises, diversion of marijuana from states where it is legal under state law in some form to other states, state-authorized marijuana activity from being used as a cover or pretext for the trafficking of other illegal drugs or other illegal activity, violence and the use of firearms in the cultivation and distribution of marijuana, drugged driving and the exacerbation of other adverse public health consequences associated with marijuana use, the growing of marijuana on public lands and marijuana possession or use on federal property. With the Cole Memo rescinded, U.S. federal prosecutors have been given discretion in determining whether to prosecute cannabis-related violations of U.S. Federal Law. If the Department of Justice policy was to aggressively pursue financiers or equity owners of cannabis-related business, and United States Attorneys followed such Department of Justice policies through pursuing prosecutions, then the Company could face (i) seizure of its cash and other assets used to support or derived from its cannabis subsidiaries and (ii) the arrest of its employees, directors, officers, managers and investors, who could face charges of ancillary criminal violations of the CSA for aiding and abetting and conspiring to violate the CSA by virtue of providing financial support to state-licensed or permitted cultivators, processors, distributors, and/or retailers of cannabis. Additionally, as has recently been affirmed by U.S. Customs and Border Protection, employees, directors, officers, managers and investors of the Company who are not U.S. citizens face the risk of being barred from entry into the United States for life. The Rohrabacher–Farr amendment (also known as the Rohrabacher–Blumenauer amendment) prohibits the Justice Department from spending funds to interfere with the implementation of state medical cannabis laws. It first passed the House in May 2014 becoming law in December 2014 as part of an omnibus spending bill. The passage of the amendment was the first time either chamber of Congress had voted to protect medical cannabis patients and is viewed as a historic victory for cannabis reform advocates at the federal level. The amendment does not change the
5
legal status of cannabis, however, and must be renewed each fiscal year in order to remain in effect. Since 2015, Congress has used a rider provision in the Consolidated Appropriations Acts (currently the Joyce Amendment, but previously called the Rohrabacher-Blumenauer Amendment, and before that the Rohrabacher-Farr Amendment) to prevent the federal government from using congressionally appropriated funds to enforce federal cannabis laws against state-compliant actors in jurisdictions that have legalized medical cannabis and cannabis-related activities.
Unless and until the United States Congress amends the CSA with respect to medical and/or adult-use cannabis (and as to the timing or scope of any such potential amendments there can be no assurance), there is a significant risk that federal authorities may enforce current U.S. Federal Law. If the U.S. Federal Government begins to enforce U.S. federal laws relating to cannabis in states where the sale and use of cannabis is currently legal, or if existing applicable state laws are repealed or curtailed, the Company’s business, results of operations, financial condition and prospects would be materially adversely affected.
Despite the current state of the federal law and the CSA, the states of Arizona, California, Nevada, Massachusetts, Maine, Michigan, New Mexico, New York, New Jersey, Illinois, Montana, Washington, Oregon, Colorado, Virginia, Vermont and Alaska, and the District of Columbia, have legalized recreational use of cannabis. Maine has not yet begun recreational cannabis commercial operations as of December 31, 2020. During the November 2020 election, voters in Arizona, New Jersey, South Dakota, and Montana passed an adult-use marijuana measure to allow for the sale of recreational marijuana in the state. South Dakota and Mississippi voters passed initiatives to allow medical marijuana. On February 8, 2021, South Dakota circuit court judge Christina Klinger rejected the measure approved by voters in the November election noting that it is a violation of the state’s requirement that constitutional amendments deal with one subject and would have broad changes to the state government. In April 2021, the South Dakota Supreme Court began hearing oral arguments on the constitutionality of the ballot initiative. Although the District of Columbia voters passed a ballot initiative in November 2014, no commercial recreational operations exist because of a prohibition on using funds for regulation within a federal appropriations amendment to local District spending powers. Early in 2021, the government moved to rectify the situation through local legislation. Two separate bills have been introduced: Mayor Muriel Bowser’s Safe Cannabis Sales Act of 2021, and Councilmember Phil Mendelson’s Comprehensive Cannabis Legalization and Regulation Act of 2021. On May 7, 2021, the Mississippi Supreme Court overturned the voter-approved initiate to legalize medical marijuana in Mississippi after legal challenges arguing the constitutional amendment violated procedural rules for placing measures on the ballot.
In addition, over half of the U.S. states have enacted legislation to legalize and regulate the sale and use of medical cannabis, provided that there are strict limits on the levels of THC. However, there is no guarantee that state laws legalizing and regulating the sale and use of cannabis will not be repealed or overturned, or that local governmental authorities will not limit the applicability of state laws within their respective jurisdictions.
The Company’s objective is to capitalize on the opportunities presented as a result of the changing regulatory environment governing the cannabis industry in the U.S. Accordingly, there are significant risks associated with the business of the Company. Unless and until the U.S. Congress amends the CSA with respect to medical and/or adult- use cannabis (and as to the timing or scope of any such potential amendments there can be no assurance), there is a significant risk that federal authorities may enforce current federal law, and the business of the Company may be deemed to be producing, cultivating, extracting, or dispensing cannabis or aiding or abetting or otherwise engaging in a conspiracy to commit such acts in violation of federal law in the U.S.
For these reasons, the Company’s investments in the U.S. cannabis market may subject the Company to heightened scrutiny by regulators, stock exchanges, clearing agencies and other Canadian authorities. There are risks associated with the business of the Company. See sections “Risk Factors,” “General Development of the Business” and “Description of the Business” in the Annual Information Form for the year ended December 31, 2020, filed on SEDAR.
On September 25, 2019, the Secure and Fair Enforcement Banking Act of 2019 (“SAFE Banking Act”) was passed by the U.S. House of Representatives in a 321 to 103 vote. The SAFE Banking Act would permanently protect state- chartered banks and credit unions that service state-legal cannabis companies from being penalized by federal regulators. On April 19, 2021 SAFE passed the US House of Representatives, 321 – 101, for the fourth time. Management believes, based on currently available information, that the likelihood of SAFE Banking’s passage is
6
high, however the particular timing and legislative vehicle is unknown. The U.S. Senate has declined to bring SAFE Banking up for a vote due to pending comprehensive federal reform legislation from Senators Chuck Schumer (D- NY), Cory Booker (D- N.J.), and Ron Wyden (D-Ore.).
On February 1, 2021 Senators Cory Booker, Ron Wyden and Chuck Schumer issued a joint statement announcing the imminent release of comprehensive cannabis reform legislation during the 117th Congress. The pending legislation is reported to focus on social justice reform. In the joint statement, the Senators stated; “we will release a unified discussion draft on comprehensive reform to ensure restorative justice, protect public health and implement responsible taxes and regulations.” To date, the legislation has not yet been released but is expected before the fall of 2021. On May 5, 2021 US Representatives David Joyce (R-OH) and Don Young (R-AK) introduced the Republican reform proposal called the Common Sense Cannabis Reform for Veterans, Small Businesses, and Medical Professionals Act.
On November 20, 2019, the House Judiciary Committee approved the Marijuana Opportunity Reinvestment and Expungement Act of 2019 (“MORE Act”) in a 24 to 10 vote. The MORE Act would decriminalize and remove Cannabis as a Schedule I substance. In April 2021, days before a floor vote in the US House of Representatives, the MORE Act was stalled due a late added amendment. While the main thrust of the bill remained intact, including a tax to fund programs to repair the harms of the drug war, a provision was added requiring a federal permit to operate a “cannabis enterprise” along with restrictions that could ban people with prior marijuana convictions from being eligible. Advocates viewed the amendment problematic as it allows for federal cannabis permit to be suspended or revoked if a person has a past or current legal proceeding related to a felony violation of any state or federal cannabis law.
The States in Which We Operate, Their Legal Framework and How it Affects Our Business
Illinois Operations
The Compassionate Use of Medical Cannabis Pilot Program Act, which allows individuals diagnosed with a debilitating medical condition access to medical marijuana, became effective January 1, 2014. There were over 41 qualifying conditions as part of the initial medical program.
The Opioid Alternative Pilot Program launched on January 31, 2019 and allows patients that receive or are qualified to receive opioid prescriptions access to medical marijuana as an alternative in situations where an opioid could generally be prescribed. Under this new program, patients with doctor approval can receive near-immediate access to cannabis products from an Illinois licensed dispensary. The Opioid Alternative Pilot Program eliminates the previously required fingerprinting and background checks that often delay patients’ access to medical cannabis by up to three months.
In January 2019, JB Pritzker was sworn into office as Governor of Illinois. Cresco Labs’ CEO and co-founder, Charles Bachtell, was appointed to the Cannabis Legalization Subcommittee of the governor’s transition team. Cannabis Legalization was one of four subcommittees under the Governor’s Restorative Justice and Safe Communities Transition Committee. The primary goals of the Cannabis Legalization Subcommittee were to evaluate and develop implementation recommendations for the Governor’s platform on legalizing cannabis.
In June 2019, the Illinois House of Representatives and Senate passed Senate Bill 2023 which added 11 additional debilitating illnesses such as chronic pain, migraines and irritable bowel syndrome to the list of qualifying medical conditions. This bill was signed into law in August 2019 by Governor JB Pritzker.
Additionally, in June 2019, Governor Pritzker signed the Cannabis Regulation and Taxation Act into law, making Illinois the 11th state to legalize recreational marijuana. Adult-use sales of marijuana started in Illinois on January 1, 2020.
Illinois’ retail market for 2020 was approximately $1 billion, representing a 312% year-over-year increase. Illinois’ retail market for the quarter ended March 31, 2021 was approximately $377 million and increased 12% quarter over quarter.
7
Cresco Labs currently owns and operates three (3) medical/adult-use cannabis cultivation and manufacturing centers in Illinois, five (5) medical/adult-use dispensary locations in Illinois, and five (5) adult-use dispensary locations in Illinois. Licenses were awarded based on merit in a highly competitive application process to applicants who demonstrated strong operational expertise and financial backing. Cresco Labs’ dispensary applications received the highest, second highest and third highest scores, respectively, of all applications reviewed by the State of Illinois.
Cresco Labs is licensed to operate in the state of Illinois as a medical and adult-use cultivator and medical and adult- use product manufacturer. Cresco Labs Phoenix Farms, LLC (“Phoenix”), PDI Medical III, LLC (“PDI”), FloraMedex, LLC (“FloraMedex”), MedMar Lakeview, LLC (“MedMar Lakeview”), and MedMar Rockford, LLC (“MedMar Rockford”) are licensed to operate retail dispensaries in the state of Illinois. These dispensaries were rebranded as Sunnyside* dispensaries in early 2020. Under applicable laws, the licenses permit Cresco Labs and its subsidiaries to collectively cultivate, manufacture, process, package, sell, and purchase marijuana pursuant to the terms of the licenses, which are issued by the Department of Agriculture and the Department of Financial and Professional Regulation under the provisions of the Illinois Revised Statutes 410 ILCS 130. All licenses are, as of the date hereof, active with the State of Illinois. There are two categories of licenses in Illinois: (i) cultivation/ processing and (ii) dispensary. The licenses are independently issued for each approved activity.
All cultivation/processing establishments must register with the Illinois Department of Agriculture (“IDOA”) and all dispensaries must register with the Illinois Department of Financial and Professional Regulation (“IDFPR”). If applications contain all required information and after vetting by officers, establishments are issued a medical marijuana establishment registration certificate. Registration certificates are valid for a period of one (1) year and are subject to annual renewals after required fees are paid and the business remains in good standing. Renewal requests are typically communicated through email from the IDOA or IDFPR and include a renewal form. While Cresco Labs compliance controls have been developed to mitigate the risk of any material violations of a license arising, there is no assurance that Illinois cannabis licenses will be renewed in the future in a timely manner. Any unexpected delays or costs associated with the licensing renewal process could impede the ongoing or planned operations of Illinois cannabis and could have a material adverse effect on the Company’s business, financial condition, results of operations or prospects.
The retail dispensary licenses held by Phoenix, PDI, FloraMedex, MedMar Lakeview, and MedMar Rockford permit the Company to purchase marijuana and marijuana products from cultivation/processing facilities and allows the sale of marijuana and marijuana products to registered patients and adult-use customers. As of March 31, 2021, the Company has opened ten (10) Sunnyside* dispensary locations in Illinois. Two (2) of the ten (10) are located within the City of Chicago.
The three (3) medical cultivation licenses held by Cresco Labs permit it to acquire, possess, cultivate, manufacture/ process into edible medical marijuana products and/or medical marijuana-infused products, deliver, transfer, have tested, transport, supply or sell marijuana and related supplies to medical marijuana dispensaries. In September 2019, the three (3) cultivation facilities were approved for growing adult-use cannabis by the Illinois Department of Agriculture, for a total cultivation capacity of 0.6 million square feet, the maximum allowed by law.
On September 27, 2019, the Company announced that it has signed a binding agreement to sell its Joliet and Kankakee, Illinois properties to Innovative Industrial Properties, Inc. (“IIP”) for approximately $46.3 million, which amount includes funding for additional tenant improvements at the Kankakee property. Concurrent with the closing of the sale, Cresco Labs entered into a long-term, triple-net lease agreement with IIP and will continue to operate each property as a licensed cannabis cultivation and processing facility. The Joliet transaction was accounted for as a financing transaction. The two properties represent approximately 100,000 square feet of industrial space in aggregate.
On December 12, 2019, the Company announced that it had completed the sale of to sell its Lincoln, Illinois cultivation facility to GreenAcreage Real Estate Corp. (“GreenAcreage”), for $50 million and accounted for as a financing transaction. Cresco Labs entered into a long-term, triple-net lease agreement with GreenAcreage and will continue to operate the property as a licensed medical & recreational cannabis cultivation and processing facility. The Company’s Lincoln property is approximately 215,000 square feet, making it the largest such facility in Illinois.
8
Pennsylvania Operations
The Pennsylvania medical marijuana program was signed into law on April 17, 2016 under Act 16 and provided access to state residents with one of 21 qualifying conditions. The state, which consists of over 12 million U.S. citizens and qualifies as the fifth largest population in the U.S., operates as a high-barrier market with very limited market participation. The state originally awarded only 12 licenses to cultivate/process and 27 licenses to operate retail dispensaries (which entitled holders up to three medical dispensary locations). Out of the hundreds of applicants in each license category, Cresco Yeltrah, LLC (“Yeltrah”) was awarded one (1) medical cannabis cultivation and processing center in Pennsylvania, and three (3) dispensary locations in Pennsylvania. Cresco Labs was awarded the second highest overall score during the application process.
Retail sales commenced in February 2018 to a limited number of retail locations across the state. On February 15, 2018, Yeltrah was the first cultivator/processor to release product into the Pennsylvania market (approximately six (6) weeks ahead of any other producer), and its dispensary was the first to sell product to patients in the state.
On March 22, 2018, it was announced that the final phase of the Pennsylvania medical marijuana program would initiate its rollout, which will include 13 additional cultivation/processing licenses and 23 additional dispensary licenses. The application period ran from April 2018 through May 17, 2018. Yeltrah submitted additional dispensary applications and in December 2018 an additional dispensary license was obtained to open three (3) additional dispensary locations, for a total of six (6) in the state of Pennsylvania. Of the six (6) approved dispensary locations, three (3) are not currently operational.
Under applicable laws, the licenses permit Yeltrah to cultivate, manufacture, process, package, sell, and purchase medical marijuana pursuant to the terms of the licenses, which are issued by the Pennsylvania Department of Health under the provisions of Medical Marijuana Act (35 P.S. §10231.101 — 10231.2110) and Chapters 1141, 1151 and 1161 of the Pennsylvania regulations. All licenses are, as of the date hereof, active with the Commonwealth of Pennsylvania. There are two categories of licenses in Pennsylvania: (i) cultivation/processing and (ii) dispensary. The licenses are independently issued for each approved activity for use at Yeltrah facilities in Pennsylvania.
All cultivation/processing establishments and all dispensaries must register with the Pennsylvania Department of Health. Registration certificates are valid for a period of one year and are subject to annual renewals after required fees are paid and the business remains in good standing. Specifically, licenses that Yeltrah currently holds have each undergone one or two renewals. While the Company’s compliance controls have been developed to mitigate the risk of any material violations of a license arising, there is no assurance that Pennsylvania cannabis licenses will be renewed in the future in a timely manner. Any unexpected delays or costs associated with the licensing renewal process could impede the ongoing or planned operations of Pennsylvania cannabis and could have a material adverse effect on the Company’s business, financial condition, results of operations or prospects.
The retail dispensary licenses permit Yeltrah to purchase marijuana and marijuana products from cultivation/ processing facilities and allows the sale of marijuana and marijuana products to registered patients.
The medical cultivation licenses permit Yeltrah to acquire, possess, cultivate, manufacture/process into edible medical marijuana products and/or medical marijuana-infused products, deliver, transfer, have tested, transport, supply or sell marijuana and related supplies to medical marijuana dispensaries. In May 2020, the Company announced the completion of its cultivation and manufacturing facility expansion which provides an additional 66,000 square feet of indoor and greenhouse cultivation area, bringing the total cultivation space in the facility to 88,000 square feet, subsequently updated to 85,000 square feet upon conversion of cultivation space to packaging space.
On September 25, 2019, Pennsylvania Governor Tom Wolf held a press conference to announce that a majority of Pennsylvania citizens were in favor of adult-use cannabis. He called on the General Assembly to consider the legalization of adult-use cannabis and provided additional actions to seek a path forward. On October 13, 2020, the Governor reaffirmed his support for adult-use cannabis and discussed the economic growth potential and restorative justice benefits of legalizing adult-use cannabis. On January 28, 2021, Governor Wolf further reiterated his support
9
for adult-use cannabis and called for the legalization in his 2021 agenda. On February 24, 2021, Senators Dan Laughlin, R-Erie County, joined by Senator Sharif Street, D-Philadelphia, introduced bipartisan legislation to legalize Adult Use Cannabis in the Commonwealth of Pennsylvania.
Ohio Operations
House Bill 523, effective on September 8, 2016, legalized medical marijuana in Ohio. The Ohio Medical Marijuana Control Program (“MMCP”) allows people with certain medical conditions, upon the recommendation of an Ohio- licensed physician certified by the State Medical Board, to purchase and use medical marijuana. House Bill 523 required that the framework for the MMCP become effective as of September 2018. This timeframe allowed for a deliberate process to ensure the safety of the public and to promote access to a safe product.
The three following state government agencies are responsible for the operation of MMCP: (1) the Ohio Department of Commerce is responsible for overseeing medical marijuana cultivators, processors and testing laboratories; (2) the State of Ohio Board of Pharmacy is responsible for overseeing medical marijuana retail dispensaries, the registration of medical marijuana patients and caregivers, the approval of new forms of medical marijuana and coordinating the Medical Marijuana Advisory Committee; and, (3) the State Medical Board of Ohio is responsible for certifying physicians to recommend medical marijuana and may add to the list of qualifying conditions for which medical marijuana can be recommended.
Several forms of medical marijuana are legal in Ohio, these include: inhalation of marijuana through a vaporizer (not direct smoking), oils, tinctures, plant material, edibles, patches and any other forms approved by the State of Ohio Board of Pharmacy.
On June 4, 2018, the State of Ohio Board of Pharmacy awarded 56 medical marijuana provisional dispensary licenses. The licenses were awarded after an extensive review of 376 submitted dispensary applications.
Provisional licensees are authorized to begin the process of establishing a dispensary in accordance with the representations in their applications and the rules adopted by the State of Ohio Board of Pharmacy. Per state of Ohio regulations, all provisional license holders have a maximum of six (6) months to demonstrate compliance with the dispensary operational requirements to obtain a certificate of operation. Compliance will be determined through an inspection by a Board of Medical Marijuana Compliance Agent. Once a dispensary is awarded a certificate of operation, it can begin selling medical marijuana to Ohio patients and caregivers in accordance with Ohio laws and rules.
By rule, the State of Ohio Board of Pharmacy is limited to issuing up to 60 dispensary licenses across the state but will have the authority to increase the number of licenses. To date, no announcement has been made if the number of licenses will be increased. Per the program rules, the Board will consider, on at least a biennial basis, whether enough medical marijuana dispensaries exist, considering the state population, the number of patients seeking to use medical marijuana, and the geographic distribution of dispensary sites.
Cresco Labs Ohio, LLC (“Cresco Labs Ohio”) was awarded one (1) dispensary license located in Wintersville, Ohio. The dispensary license permits Cresco Labs Ohio to purchase marijuana and marijuana products from cultivation/processing facilities and allows the sale of marijuana and marijuana products to registered patients.
Cresco Labs Ohio applied for and, on November 30, 2017, received one (1) cultivation license. Cresco Labs Ohio’s cultivation facility is a hybrid greenhouse structure located in Yellow Springs, Ohio. The medical cultivation license authorizes Cresco Labs Ohio to grow, harvest, package and transport medical marijuana products.
On December 12, 2018, Cresco Labs Ohio was granted the first dispensary Certificate of Operation in the state, which was over a month in advance of any other dispensary operator. Retail sales commenced on January 16, 2019 with the first cannabis sale taking place at the Wintersville dispensary. This was the second state medical marijuana program in which the Company was first to market.
10
On June 8, 2020, Cresco Labs Ohio was granted a provisional processing license by the State of Ohio. This license allows Cresco Labs Ohio to extract oils and manufacture products from cannabis which will now provide the Company the ability to sell its entire brand portfolio in Ohio.
Ohio cultivation and processor licenses are renewable annually by the Ohio Department of Commerce. Renewal applications are due at least thirty days prior to the expiration date of the Certificate of Operation. The Department of Commerce shall grant a renewal if the renewal application was timely filed, the annual fee was timely paid, there are no reasons warranting denial of the renewal and the cultivator/processor passes inspection. Ohio dispensary licenses expire biennially on the date identified on the certificate. Renewal information, including a renewal fee, must be submitted at least 45 days prior to the date the existing certificate expires. If the dispensary is operated in compliance with Ohio dispensary regulations, and the renewal fee is paid, the state board of pharmacy shall renew the Certificate of Operation within 45 days after the renewal application is received. While the Company’s compliance controls have been developed to mitigate the risk of any material violations of a license arising, there is no assurance that Ohio cannabis licenses will be renewed in the future in a timely manner. Any unexpected delays or costs associated with the licensing renewal process could impede the ongoing or planned operations of Ohio cannabis and could have a material adverse effect on the Company’s business, financial condition, results of operations or prospects.
On January 28, 2020, the Company announced that it had completed the sale of its Yellow Springs, OH property to IIP. The previously announced sale was for consideration equal to approximately $10.5 million, which includes funding for additional tenant improvements. Concurrent with the closing of the sale, Cresco Labs entered into a long-term, triple-net lease agreement with IIP and will continue to operate the property as a licensed cannabis cultivation and processing facility. The Company accounted for the transaction as a financing transaction. The property represents approximately 50,000 square feet of industrial space in aggregate. This sale marked the Company’s fourth completed sale-and-leaseback, or financing, transaction, the third with IIP.
On February 16, 2021, the Company announced the closing of its acquisition of Verdant dispensaries in Cincinnati, Chillicothe, Newark and Marion, Ohio. These acquisitions give the Company four additional dispensaries, bringing the Company’s dispensary presence in Ohio to five (5), the maximum allowed by the state.
California Operations
In 1996, California was the first state to legalize medical marijuana through Proposition 215, the Compassionate Use Act of 1996 (“CUA”). This legalized the use, possession and cultivation of medical marijuana by patients with a physician recommendation.
In 2003, Senate Bill 420 was signed into law establishing an optional identification card system for medical marijuana patients.
In September 2015, the California legislature passed three bills collectively known as the “Medical Cannabis Regulation and Safety Act” (“MCRSA”). The MCRSA established a licensing and regulatory framework for medical marijuana businesses in California. The system created multiple license types for dispensaries, infused products manufacturers, cultivation facilities, testing laboratories, transportation companies and distributors. Edible infused product manufacturers would require either volatile solvent or non-volatile solvent manufacturing licenses depending on their specific extraction methodology. Multiple agencies would oversee different aspects of the program and businesses would require a state license and local approval to operate. However, in November 2016, voters in California overwhelmingly passed Proposition 64, the “Adult-Use of Marijuana Act” (“AUMA”) creating an adult-use marijuana program for adults 21 years of age or older. AUMA had some conflicting provisions with MCRSA, so in June 2017, the California State Legislature passed Senate Bill No. 94, known as Medicinal and Adult-Use Cannabis Regulation and Safety Act (“MAUCRSA”), which amalgamates MCRSA and AUMA to provide a set of regulations to govern medical and adult-use licensing regime for cannabis businesses in the state of California. MAUCRSA went into effect on January 1, 2018. The four agencies that regulate marijuana at the state level are the Bureau of Cannabis Control (“BCC”), the California Department of Food and Agriculture (“CDFA”), the California Department of Public Health (“CDPH”), and the California Department of Tax and Fee Administration (“CDTFA”).
11
In order to legally operate a medical or adult-use cannabis business in California, the operator must have both a local and state license. This requirement limits license holders to operate only in cities with marijuana licensing programs. Therefore, cities in California are allowed to determine if they will have a marijuana licensing program and determine the number of licenses they will issue to marijuana operators.
On June 7, 2018, Cresco Labs acquired a 60% ownership interest in SLO Cultivation, Inc. (“SLO”), a marijuana cultivation facility in operation in the cities of Carpinteria (Santa Barbara County) and Mendota (Fresno County), California. On September 27, 2018, Cresco Labs acquired a further 20% ownership interest to bring the total ownership to 80%.
SLO is licensed to cultivate, process, manufacture, and distribute medical and adult-use cannabis in the state of California pursuant to the terms of the California state licenses issued by the CDFA, CDPH, CDTFA, and BCC under the provision of MAUCRSA and California Assembly Bill No. 133. In California, licenses are independently issued by the respective agency.
California state and local licenses are renewed annually. Each year, licensees are required to submit a renewal application per guidelines published by BCC, CDFA, CDTFA, and CDPH. While renewals are annual, there is no ultimate expiry after which no renewals are permitted. Additionally, in respect of the renewal process, provided that the requisite renewal fees are paid, the renewal application is submitted in a timely manner, and there are no material violations noted against the applicable license, SLO would expect to receive the applicable renewed license in the ordinary course of business. While SLO’s compliance controls have been developed to mitigate the risk of any material violations of a license arising, there is no assurance that the licenses will be renewed in the future in a timely manner. Any unexpected delays or costs associated with the licensing renewal process could impede the ongoing or planned operations of the Company and have a material adverse effect on its business, financial condition, results of operations or prospects.
SLO applied for and was granted licenses permitting it to cultivate, manufacture and distribute medical (and in some instances, adult-use) cannabis and cannabis-related products:
Mendota (Fresno County)
| • | SLO has been issued one (1) provisional license for Type 7 (Manufacturing 2 – Volatile), Adult-Use & Medical (“A&M”). |
| • | SLO has been issued one (1) provisional license for Type 11 (Distribution), A&M. |
| • | SLO submitted annual applications for the two (2) listed license types to the state regulator and is awaiting approval for these annual applications. |
Carpinteria (Santa Barbara County)
| • | SLO has been issued: |
| • | Twenty-three (23) Cultivation: Small Mixed-Light Tier 1 licenses. |
| • | One (1) Nursery license: allowing for the planting and cultivation of medical cannabis from seeds, clones and immature plants. |
| • | One (1) Processor license: allowing for the harvesting, drying, curing, grading or tanning of cannabis as well as the packaging and labelling of certain non-manufactured cannabis. |
12
| • | SLO submitted annual for the three (3) listed license types to the state regulator and is awaiting approval of annual applications. |
Origin House
On January 8, 2020, Cresco Labs acquired Origin House, a leading distributor and provider of brand support services in California, pursuant to which Cresco Labs acquired all issued and outstanding shares of Origin House. Under the terms of the Origin House Agreement and subsequent amendments, holders of common shares of Origin House received 0.7031 SVS of Cresco Labs for each Origin House share (the “Transaction”). The Company acquired 100% of all equity interests of Origin House for 66.5 million Cresco Labs shares and 5.7 million replacement equity awards, valued at $428.2 million.
The Transaction represents a total consideration of $428.2 million on a fully-diluted basis, and as of this date, is among the largest of public company acquisitions in the history of the U.S. cannabis industry. The combined entity is one of the largest vertically integrated multi-state cannabis operators in the United States; a leading North American cannabis company, by footprint; and one of the largest cannabis brand distributors.
Origin House’s proven strategy has been to build relationships with established dispensaries, build partnerships with established market-leading brands, develop promising cannabis product companies and then leverage its full suite of support services to transform those products into strong California consumer brands. Since the closing of this acquisition, Cresco Labs has access to several additional licenses for cultivation, manufacturing and distribution of cannabis within the state of California.
West Sacramento (Yolo County)
| • | Origin House has been issued one (1) provisional license for Type 11 (Distribution), A&M. |
| • | Origin House submitted an annual application for the one (1) listed license type to the state regulator and is awaiting approval for the annual application. |
La Habra (Orange County)
| • | Origin House has been issued one (1) provisional license for Type 11 (Distribution), A&M. |
| • | Origin House submitted an annual application for the one (1) listed license type to the state regulator and is awaiting approval for the annual application. |
Unincorporated Sonoma (Sonoma County)
| • | Origin House has been issued one (1) provisional license for Cultivation, Medical Medium Indoor. |
| • | Origin House has been issued one (1) provisional license for Processor, Medical. |
| • | Origin House has been issued one (1) provisional license for Type 11 (Distribution), A&M. |
| • | Origin House has been issued one (1) provisional license for Cultivation: Medical Small Indoor. |
| • | Origin House submitted annual applications for the four (4) listed license types to the state regulator and is awaiting approval for these annual applications. |
In addition to the six (6) active licenses listed above, Origin House continues to pursue new state license opportunities and recently applied for a Type 11 (Distribution) license for the Unincorporated Sonoma (Sonoma County) location.
13
Arizona Operations
In 2010, Arizona passed Ballot Proposition 203, which amended Title 36 to the Arizona Revised Statutes. This amendment added Chapter 28.1, titled the Arizona Medical Marijuana Act. (the “AMMA”). The AMMA is codified in Arizona Revised Statutes §36-2801 et. seq. The AMMA also appointed the Arizona Department of Health Services (the “ADHS”) as the regulator for the program and authorized ADHS to promulgate, adopt and enforce regulations for the AMMA. These ADHS regulations are embodied in the Arizona Administrative Code Title 9 Chapter 17 (the “Rules”). In order to qualify to use medical marijuana under the AMMA, a patient is required to have a “debilitating medical condition.”
The ADHS has established the Arizona Department of Health Services Medical Marijuana Program (“MMJ Program”), which includes a vertically integrated license, meaning if allocated a Medical Marijuana Dispensary Registration Certificate (“Dispensary License”), entities are authorized to dispense and cultivate medical cannabis. Each Dispensary License allows the holding entity to operate one (1) on-site cultivation facility, and one (1) off-site cultivation facility which can be located anywhere within the state of Arizona. An entity holding a Dispensary License is required to file an application to renew with the ADHS on an annual basis, which must also include audited annual financial statements. While a Dispensary License may not be sold, transferred or otherwise conveyed, Dispensary License holders typically contract with third parties to provide various services related to the ongoing operation, maintenance and governance of its dispensary and/or cultivation facility so long as such contracts do not violate the requirements of the AMMA or the MMJ Program.
On December 6, 2012, Arizona’s first licensed medical marijuana dispensary opened in Glendale.
Once an applicant has been issued a Dispensary Registration Certificate (a “Certificate”), they are allowed to establish one (1) physical retail dispensary location, one (1) cultivation location which is co-located at the dispensary’s retail site (if allowed by local zoning) and one (1) additional off-site cultivation location. None of these sites can be operational, however, until the dispensary receives an approval to operate from ADHS for the applicable site.
On October 24, 2018, Cresco Labs obtained a 100% ownership interest in Arizona Facilities Supply, LLC which includes a vertically integrated cultivation, processing and dispensary operation in Arizona.
The licenses in Arizona are renewed annually. Before expiry, licensees are required to submit a renewal application. While renewals are granted annually, there is no ultimate expiry after which no renewals are permitted. Additionally, in respect of the renewal process, provided that the requisite renewal fees are paid, the renewal application is submitted in a timely manner, and there are no material violations noted against the applicable license, Cresco Labs would expect to receive the applicable renewed license in the ordinary course of business. While the Company’s compliance controls have been developed to mitigate the risk of any material violations of a license arising, there is no assurance that Arizona Cannabis licenses will be renewed in the future in a timely manner. Any unexpected delays or costs associated with the licensing renewal process could impede the ongoing or planned operations of Arizona Cannabis and could have a material adverse effect on the Company’s business, financial condition, results of operations or prospects.
In November 2020, voters in Arizona passed an adult-use marijuana measure to allow for the sale of recreational marijuana in the state. During 2021, the Company received approval from the Arizona Department of Health Services to serve adult-use customers at its Sunnyside* dispensary in Phoenix, Arizona. Adult-use sales launched in February of 2021.
Nevada Operations
Medical marijuana use was legalized in Nevada by a ballot initiative in 2000. In November 2016, voters in Nevada passed an adult-use marijuana measure to allow for the sale of recreational marijuana in the state. The first dispensaries to sell adult-use marijuana began sales in July 2017. The Nevada Department of Taxation (“DOT”) is the regulatory agency overseeing the medical and adult-use cannabis programs. Similar to California, cities and counties in Nevada are allowed to determine the number of local marijuana licenses they will issue.
14
All marijuana establishments must register with DOT. If applications contain all required information and after vetting by officers, establishments are issued a medical marijuana establishment registration certificate. In a local governmental jurisdiction that issues business licenses, the issuance by DOT of a medical marijuana establishment registration certificate is considered provisional until the local government has issued a business license for operation and the establishment is in compliance with all applicable local governmental ordinances. Final registration certificates are valid for a period of one (1) year and are subject to annual renewals after required fees are paid and the business remains in good standing. The renewal periods serve as an update for DOT on the licensee’s status toward active licensure. It is important to note provisional licenses do not permit the operation of any commercial or medical cannabis activity. Only after a provisional licensee has gone through necessary state and local inspections, if applicable, and has received a final registration certificate from DOT may an entity engage in cannabis business operation.
On August 12, 2019, the Company settled its outstanding loan receivable with Lighthouse, which has cannabis operations in Nevada, through receipt of Lighthouse membership units approximating 1.2% ownership of the parent company. The remaining escrow balance was issued as a new secured convertible promissory note convertible, at the Company’s discretion, into additional membership units approximating 1% ownership of the parent company.
New York Operations
The state of New York’s medical cannabis program was introduced in July 2014 when Governor Andrew Cuomo signed the Compassionate Care Act, which legalized medical cannabis oils for patients with certain qualifying conditions. Under this program, five (5) registered organizations (“ROs”) were licensed to dispense cannabis oil to patients, with the first sale to a patient completed in January 2016. In December 2016, the New York State Department of Health (“NYSDOH”) added chronic pain as a qualifying condition and in the month-and-a-half following the addition of chronic pain, the number of registered patients increased by 18%. In August 2017, the NYSDOH granted licenses to five (5) additional registered organizations.
In July 2018, the NYSDOH added opioid replacement as a qualifying condition, meaning any condition for which an opioid could be prescribed is now a qualifying condition for medical cannabis. In August 2018, Governor Cuomo, prompted by a NYSDOH study which concluded the “positive effects” of cannabis legalization “outweigh the potential negative impacts,” appointed a group to draft a bill for regulating legal adult-use cannabis sales in New York. During Governor Cuomo’s January 2019 State of the State Address, he announced the proposal of the governmental agency, The Office of Cannabis Management. This agency would regulate and oversee the state’s medical marijuana program, adult-use program and hemp program. The executive director of this agency would have the authority to grant ROs currently registered and in good standing with the NYSDOH, the ability to be licensed to cultivate, process or sell adult-use cannabis and cannabis products.
Each RO license allows for the cultivation, processing, and dispensing of medical cannabis products. Each RO is permitted to open four (4) dispensaries in NYSDOH-designated regions throughout the state, and one (1) cultivation/ processing facility. Permitted products include oil-based formulations (i.e., vaporizer cartridges, tinctures and capsules), and ground-flower sold in tamper-proof vessels. Each RO is required to cultivate and process all medical cannabis products they dispense; however, wholesale transactions are permitted with approval from the state and home delivery is now permitted.
All cultivation/processing and dispensing establishments must register with the NYSDOH pursuant to Public Health Law §3365(9). Registrations issued by NYSDOH are valid for a two-year period. As embodied in New York Codes, Rules and Regulations §1004.7, an application to renew such registrations must be filed with the NYSDOH between four and six months prior to the expiration date, must include information prepared in the manner and detail as the commissioner may require, and should be accompanied by application fees and registration fees. Applications completed in accordance with §1004.7 would be expected to receive the applicable renewed license in a timely manner. While the Company’s compliance controls have been developed to mitigate the risk of any material
15
violations of a license arising, there is no assurance that New York cannabis licenses will be renewed in the future in a timely manner. Any unexpected delays or costs associated with the licensing renewal process could impede the ongoing or planned operations of New York cannabis and could have a material adverse effect on the Company’s business, financial condition, results of operations or prospects.
On October 8, 2019, the Company closed the acquisition of GSC, the parent entity of Valley Agriceuticals, LLC (“Valley Ag”), for consideration that consisted of cash, deferred consideration, equity and contingent consideration based upon the achievement or occurrence of certain milestones or events, all totaling $129.6 million. Valley Ag is one of the ten holders of a vertically integrated license from NYSDOH allowing for the cultivation and processing of medical cannabis as well as the establishment of four (4) medical cannabis dispensaries in the state of New York.
Through the aforementioned agreements and regulatory approval, Cresco Labs now has a license for a cultivation and manufacturing facility within the state of New York, as well as four (4) dispensary locations strategically located across the state. These four (4) locations are branded as Sunnyside* dispensaries. Valley Ag has successfully renewed their initial licenses and all licenses are, as of the date hereof, active with the State of New York.
On January 6, 2021, Governor Cuomo announced a proposal to legalize and create a comprehensive system to oversee and regulate adult-use cannabis in New York as part of the 2021 State of the State. Under the Governor’s proposal, a new Office of Cannabis Management would be created to oversee the new adult-use program, as well as the State’s existing medical and cannabinoid hemp programs. Additionally, an equitable structure for the adult-use market will be created by offering licensing opportunities and assistance to entrepreneurs in communities of color who have been disproportionately impacted by the war on drugs. Once fully implemented, legalization is expected to generate more than $300 million in tax revenue for the State of New York.
On February 16, 2021, Governor Cuomo announced 30-day amendments to the Governor’s proposal to establish a comprehensive adult-use cannabis program in New York. Specifically, these amendments will detail how the $100 million in social equity funding will be allocated, enable the use of delivery services and refine which criminal charges will be enforced as it relates to the improper sale of cannabis to further reduce the impact on communities.
Governor Andrew Cuomo signed Senate Bill 854/Assembly Bill 1248A on March 31, 2021 creating the Empire State’s adult-use cannabis program. The legislations expands our dispensary footprint to eight (8), with three (3) reserved to be co-located adult use, allows existing vertical ROs to wholesale branded products, and creates a strong social equity program with 50% of licenses dedicated to Social Equity Applicants. The Cannabis Control Board which will oversee the roll out of the program will be seated in summer 2021.
Massachusetts Operations
The Massachusetts medical cannabis market was established through “An Act for the Humanitarian Medical Use of Marijuana” in November 2012 when voters passed Ballot Question 3 “Massachusetts Medical Marijuana Initiative” with 63% of the vote. The first Massachusetts dispensary opened in June 2015 and by November 2016, Massachusetts voters legalized adult-use cannabis by passing ballot Question 4 – Legalize Marijuana with 54% of the vote. In July 2017, Governor Baker signed legislation that would lay the groundwork for the state’s adult-use market. The Cannabis Control Commission (the state’s regulatory body which creates regulations for both the medical and adult-use market) aimed to officially launch adult-use sales on July 1, 2018 but stumbling blocks such as a lack of licensed testing labs and disagreements between officials and businesses slowed the rollout, as sales for adult-use cannabis officially began in November 2018.
The Cannabis Control Commission oversees the medical and adult-use cannabis programs. Each medical licensee must be vertically-integrated and may have up to two (2) locations. Licensed medical dispensaries are given priority in adult-use licensing. Adult-use cultivators will be grouped into 11 tiers of production (ranging from up to 5,000 square feet to no larger than 100,000 square feet) and regulators will move a licensee down to a lower tier if that licensee has not shown an ability to sell at least 70% of what it produced. Medical dispensaries that wish to add the ability to sell cannabis products to nonpatients will be required to reserve 35% of their inventory or the six-month average of their medical cannabis sales for medical cannabis patients. In order to achieve an adult-use license, a
16
prospective licensee must first sign a “Host Community Agreement” with the town in which it wishes to locate. Roughly two-thirds of municipalities in the state have a ban or moratorium in place that prohibits cannabis businesses from operating within their jurisdiction. In both the medical and adult-use markets, extracted oils, edibles and flower products are permitted, as well as wholesaling.
On October 1, 2019, Cresco Labs acquired Cresco HHH, LLC (“HHH”) via certain agreements giving it operational control before cash consideration was settled. In August 2019, HHH entered into a host community agreement with the municipality of Fall River to allow for the siting of an adult-use cannabis dispensary. On February 7, 2020, the Company announced the legal close of the acquisition and cash funding of $27.5 million. The closing coincided with state approval allowing recreational cannabis sales at the Company’s Fall River dispensary.
Registration certificates are valid for a period of one year and are subject to annual renewals after required fees are paid and the business remains in good standing. Renewal requests are typically communicated through email from the Massachusetts Cannabis Commission and include a renewal form. While the Company’s compliance controls have been developed to mitigate the risk of any material violations of a license arising, there is no assurance that Massachusetts cannabis licenses will be renewed in the future in a timely manner. Any unexpected delays or costs associated with the licensing renewal process could impede the ongoing or planned operations of Massachusetts cannabis and could have a material adverse effect on the Company’s business, financial condition, results of operations or prospects.
On July 1, 2020, Cresco Labs announced that it had completed the sale of its Falls River, MA property to IIP. The sale was for consideration equal to approximately $29 million, which includes $21 million in funding for additional tenant improvements. Concurrent with the closing of the sale, the Company entered into a long-term, triple-net lease agreement with IIP and will continue to operate the property as a licensed cannabis cultivation, processing and dispensing facility upon completion of redevelopment. The Company accounted for the transaction as a financing transaction. The property represents approximately 50,000 square feet of industrial space in aggregate.
Michigan Operations
In November 2008, Michigan residents approved the Michigan Medical Marijuana Act (the “MMMA”) to provide a legal framework for a safe and effective medical marijuana program. In September 2016, the Michigan Senate passed the Medical Marihuana Facilities Licensing Act (the “MMFLA”) and the Marihuana Tracking Act (the “MTA” and together with the MMMA and the MMFLA, the “Michigan Cannabis Regulations”) to provide a comprehensive licensing and tracking scheme, respectively, for the medical marijuana program. Additionally, the Michigan Department of Licensing and Regulatory Affairs and its licensing board (“LARA”) has supplemented the Michigan Cannabis Regulations with “Emergency Rules” to further clarify the regulatory landscape surrounding the medical marijuana program. LARA is the main regulatory authority for the licensing of marijuana businesses.
Under the MMFLA, LARA administrates five (5) types of “state operating licenses” for medical marijuana businesses: (i) a “grower” license, (ii) a “processor” license, (iii) a “secure transporter” license, (iv) a “provisioning center” license and (e) a “safety compliance facility” license. There are no stated limits on the number of licenses that can be made available on a state level; however, LARA has discretion over the approval of applications and municipalities can pass additional restrictions.
On November 6, 2018, Michigan voters approved Proposal 1, to make marijuana legal under state and local law for adults 21 years of age or older and to control the commercial production and distribution of marijuana under a system that licenses, regulates and taxes the businesses involved. The act will be known as the Michigan Regulation and Taxation of Marihuana Act. In accordance with Proposal 1, LARA began accepting applications for retail (recreational) dispensaries on November 1, 2019.
On March 25, 2019, the Company announced Cresco Labs Michigan, LLC (“Cresco Michigan”) had completed the most comprehensive portion of Michigan’s application process, being pre-qualified for a cultivation and processing license by the Department of Licensing and Regulatory Affairs Medical Marihuana Licensing Board. The pre-qualification represents the authorization of the entity to move forward with the licensing process for its intended facilities.
17
On November 13, 2019, the state’s Marijuana Regulatory Agency announced any existing medically licensed businesses would be allowed to sell recreational-use cannabis beginning December 1, 2019. On March 5, 2020, the Cresco Michigan was issued a medical processing license to begin manufacturing and processing flower into edible medical marijuana products and/or medical marijuana-infused products. Michigan has approximately 250,000 medical marijuana patients, which represents the fourth largest medical marijuana patient base in the United States.
On March 16, 2020, Cresco Michigan received pre-qualification to operate in the adult-use market and received an adult-use processor license on June 22, 2020. All Michigan marijuana licenses are renewed annually through the Marijuana Regulatory Agency after the required fees are paid and the business remains in good standing. In addition, a sworn statement is required that states that the business is in good standing and will uphold a continuing reporting duty. The renewal fees are to be determined by the amount of gross weight of marijuana product transferred during the past year. While the Company’s compliance controls have been developed to mitigate the risk of any material violations of a license arising, there is no assurance that Michigan cannabis licenses will be renewed in the future in a timely manner. Any unexpected delays or costs associated with the licensing renewal process could impede the ongoing or planned operations of Michigan cannabis and could have a material adverse effect on the Company’s business, financial condition, results of operations or prospects.
On April 22, 2020, Cresco Michigan and related parties of the Company executed an amended and restated operating agreement which increased the Company’s related parties’ ownership from 50% to 85% in exchange for a capital commitment of $25 million. Provisions contained in the operating agreement entitle related parties of the Company to a majority of profit and gives the Company control of Cresco Michigan and rights and exposure to variable returns. The Company has the right to direct all the relevant activities of and has the full decision-making power over Cresco Michigan.
On April 23, 2020, the Company announced that it had completed the sale of its Marshall, MI property to IIP. The previously announced sale was for consideration equal to approximately $16 million, which included $11 million in funding for tenant improvements. Concurrent with the closing of the sale, Cresco Labs entered into a long-term, triple-net lease agreement with IIP and will continue to operate the property as a licensed cannabis cultivation and processing facility upon completion of redevelopment. The property represents approximately 100,000 square feet of industrial space in aggregate. This sale marked Cresco Labs’ fifth completed sale-and-leaseback, or financing, transaction, the fourth with IIP.
Florida Operations
In 2014, the Florida Legislature passed the Compassionate Use Act (the “CUA”) which was a low-THC (CBD) law, allowing cannabis containing not more than 0.8% THC to be sold to patients diagnosed with severe seizures or muscle spasms and cancer. The CUA created a competitive licensing structure and originally allowed for one vertically integrated license to be awarded in each of five regions. The CUA set forth the criteria for applicants as well as the minimum qualifying criteria which included the requirement to hold a nursery certificate evidencing the capacity to cultivate a minimum of 400,000 plants, to be operated by a nurseryman and to be a registered nursery for at least 30 continuous years. The CUA also created a state registry to track dispensations. In 2016, the Florida Legislature passed the Right to Try Act (the “RTA”), which expanded the State’s medical cannabis program to allow for full potency THC products to be sold as “medical marijuana” to qualified patients.
In November of 2016, the Florida Medical Marijuana Legalization ballot initiative (the “Initiative”) to expand the medical cannabis program under the RTA was approved by 71.3% of voters, thereby amending the Florida constitution. The Initiative is now codified as Article X, Section 29 of the Florida Constitution.
The Initiative expanded the list of qualifying medical conditions include cancer, epilepsy, glaucoma, HIV and AIDS, ALS, Crohn’s disease, Parkinson’s disease, multiple sclerosis, or other debilitating medical conditions of the same kind or class or comparable to those other qualifying conditions and for which a physician believes the benefits outweigh the risks to the patient. The Initiative also provided for the implementation of state-issued medical cannabis identification cards. In 2017, the Florida Legislature passed legislation implementing the constitutional amendment and further codifying the changes set forth in the constitution into law. The 2017 law provides for the
18
issuance of 10 licenses to specific entities and another four licenses to be issued for every 100,000 active qualified patients added to the registry. The 2017 law also initially limited license holders to a maximum of 25 dispensary locations with the ability to purchase additional dispensary locations from one another, and for an additional five (5) locations to be allowed by the State for every 100,000 active qualified patients added to the registry. The 2017 legislation’s cap on dispensing facilities expired in April 2020.
On January 14, 2021, Cresco Labs and Bluma entered into a definitive agreement whereby Cresco Labs acquired all of the issued and outstanding shares of Bluma in an all-share transaction that valued Bluma at an equity value of $213.0 million. Under the terms of the transaction, shareholders of Bluma received 0.0859 of a subordinate voting share of Cresco Labs for each Bluma share held.
Bluma owns and operates a vertically integrated, licensed medical cannabis company in the State of Florida doing business as “One Plant Florida.” One Plant Florida cultivates, processes, dispenses and retails medical cannabis to qualified patients in the State of Florida through multiple retail dispensaries and an innovative next-day door-to-door e-commerce home delivery service, thereby offering convenient access for its customers and meeting the demands of an evolving retail landscape. Bluma, under One Plant Florida, has eight (8) strategically located dispensaries with seven (7) more locations under legal control and planned to open.
Components of Our Results of Operations
Revenue
We derive the majority of our revenue from wholesale of cannabis products to dispensary locations which, for the three months ended March 31, 2021, represents approximately 54% of our revenue. Revenue from company-owned retail dispensary locations, for the three months ended March 31, 2021 represents the remaining 46%. Retail revenue includes medical and adult-use cannabis sales in the United States, along with nicotine vape sales in Canada.
Gross Profit
Gross profit is calculated as revenue less cost of goods sold. Cost of goods sold include the direct costs attributable to the cultivation and production of the products sold and is comprised of the following:
| • | Direct labor costs: These expenses include all salaries, benefits, and taxes for all employees at the facility. |
| • | Direct supplies: The direct material cost for maintenance of the plants, the supplies and nutrients, and the production expenses and equipment used to process marijuana. |
| • | Facility expenses: The facility expense for the cultivation operations is the cost for the facility, utilities, property taxes, maintenance, and costs associated with monitoring the security systems. |
| • | Other operating expenses: These expenses include all costs associated with the facility itself including insurance, community outreach programs, professional services related to licenses and compliance, uniforms, employee training programs, tracking and inventory management systems, product testing, business development, information technology, license renewal fees and certain excise taxes. |
| • | Cultivation, manufacturing, and packaging costs: These expenses are comprised of cannabis plant costs and are initially capitalized into inventory in the period in which they are incurred. |
In addition to market fluctuations, cannabis costs are affected by various state regulations that limit the sourcing and procurement of cannabis products. The changes in regulatory environments may create fluctuations in gross profit over comparative periods.
19
Selling, General and Administrative Expenses (“SG&A”)
SG&A expenses consist mainly of salary and benefits costs of executive and back-office employees, consulting and professional fees, advertising and marketing, office and retail operation costs, share-based compensation, certain excise taxes, technology, insurance, security, travel and entertainment, rent expense and business expansion costs.
Selling costs generally correlate to revenue. As a percentage of sales, we expect SG&A costs to decrease as our business continues to grow. The decrease is expected to be driven primarily by efficiencies associated with scaling the business.
For the three months ended March 31, 2021 and March 31, 2020, SG&A was comprised of the following:
| Three months ended March 31, | ||||||||||||||||
| ($ in thousands) | 2021 | 2020 | $ Change | % Change | ||||||||||||
Payroll and employee costs | $ | 36,069 | $ | 19,676 | $ | 16,393 | 83 | % | ||||||||
Selling and marketing expenses | 7,250 | 3,984 | 3,266 | 82 | % | |||||||||||
Depreciation and amortization | 5,642 | 3,828 | 1,814 | 47 | % | |||||||||||
Excise taxes | 3,950 | 1,868 | 2,082 | 111 | % | |||||||||||
Facility expenses | 3,821 | 3,861 | (40 | ) | (1 | )% | ||||||||||
Consulting and professional fees | 2,806 | 8,346 | (5,540 | ) | (66 | )% | ||||||||||
Computer and software expense | 2,721 | 1,234 | 1,487 | 121 | % | |||||||||||
Business insurance | 2,130 | 993 | 1,137 | 115 | % | |||||||||||
Rental fees | 1,608 | 1,067 | 541 | 51 | % | |||||||||||
Accounting | 1,579 | 1,038 | 541 | 52 | % | |||||||||||
Legal | 1,537 | 1,989 | (452 | ) | (23 | )% | ||||||||||
Travel and employee expenses | 622 | 1,286 | (664 | ) | (52 | )% | ||||||||||
Intangible asset impairment | — | 1,194 | (1,194 | ) | (100 | )% | ||||||||||
Litigation accrual adjustment | (810 | ) | — | (810 | ) | 100 | % | |||||||||
Other expenses | 1,860 | 1,012 | 846 | 84 | % | |||||||||||
|
|
|
|
|
| |||||||||||
Total Selling, general & administrative expenses | $ | 70,785 | $ | 51,376 | $ | 19,409 | 38 | % | ||||||||
|
|
|
|
|
| |||||||||||
Income Taxes
The Company is classified for U.S. federal income tax purposes as a United States corporation under Section 7874 of the Internal Revenue Code (“IRC”). The Company is subject to income taxes in the jurisdictions in which it operates and, consequently, income tax expense is a function of the allocation of taxable income by jurisdiction and the various activities that impact the timing of taxable events. As the Company operates in the legal cannabis industry, the Company is subject to the limits of IRC Section 280E under which the Company is only allowed to deduct expenses directly related to sales of product. This results in permanent differences between ordinary and necessary business expenses deemed non-allowable under IRC Section 280E and a higher effective tax rate than most industries.
20
SELECTED FINANCIAL INFORMATION
The Company reports results of operations of its affiliates from the date that control commences, either through the purchase of the business, through a management agreement or other arrangements which grant such control. The following selected financial information includes only the results of operations after the Company established control of its affiliates. Accordingly, the information included below may not be representative of the results of operations if such affiliates had included their results of operations for the entire reporting period.
Three Months Ended March 31, 2021 Compared to Three Months Ended March 31, 2020
The following tables set forth selected consolidated financial information for the periods indicated that was derived from our unaudited condensed interim consolidated financial statements and the respective accompanying notes prepared in accordance with U.S. GAAP.
The selected consolidated financial information set out below may not be indicative of the Company’s future performance:
| Three months ended March 31, | ||||||||||||||||
($ in thousands) | 2021 | 2020 | $ Change | % Change | ||||||||||||
Revenue | $ | 178,437 | $ | 66,380 | $ | 112,057 | 169 | % | ||||||||
Cost of goods sold | (91,414 | ) | (42,792 | ) | (48,622 | ) | 114 | % | ||||||||
|
|
|
|
|
| |||||||||||
Gross profit | 87,023 | 23,588 | 63,435 | 269 | % | |||||||||||
Total operating expenses | 70,785 | 51,376 | 19,409 | 38 | % | |||||||||||
Total other (expense) income, net | (24,838 | ) | 3,515 | (28,353 | ) | nm | ||||||||||
Income tax expense | (15,524 | ) | (11,188 | ) | (4,336 | ) | 39 | % | ||||||||
Net loss1 | $ | (24,124 | ) | $ | (35,461 | ) | $ | 11,337 | (32 | )% | ||||||
|
|
|
|
|
| |||||||||||
| 1 | Net loss includes amounts attributable to non-controlling interests. |
Revenue
Revenue for the three months ended March 31, 2021 was $178.4 million, an increase of $112.1 million, or 169%, compared to Revenue of $66.4 million for the three months ended March 31, 2020. The increase in revenue was driven by continued growth in the states where the Company operates. Organic growth was primarily attributable to increased cultivation capacity and increased dispensary sales in states with recently adopted adult-use laws, such as Illinois, with the opening of additional dispensaries throughout 2020 contributing significantly to the increase in revenue for the three months ended March 31, 2021.
Cost of Goods Sold and Gross Profit
Cost of goods sold for the three months ended March 31, 2021 was $91.4 million, an increase of $48.6 million compared to cost of goods sold of $42.8 million for the three months ended March 31, 2020. The increase was primarily attributable to year-over-year revenue growth, increased cultivation capacity in Illinois and Pennsylvania, and other organic growth.
Gross profit increased by $63.4 million for the three months ended March 31, 2021 compared to the three months ended March 31, 2020 primarily due to the increase in revenue and greater scale in the Company’s established Illinois, Pennsylvania, and California markets, operating synergies realized through continued integration of Origin House, and continued efforts to increase cultivation yields.
Total Operating Expenses
Total expenses for the three months ended March 31, 2021 were $70.8 million, an increase of $19.4 million compared to total expenses of $51.4 million for the three months ended March 31, 2020. The increase in total
21
expenses was attributable to significant investments in our team, marketing, information technology, and operational infrastructure to drive strategic initiatives that better position the Company for future growth. Total expenses as a percentage of revenue have declined as the Company has gained efficiencies with the growth of the business.
Total Other (Expense) Income, Net
Total other expense for the three months ended March 31, 2021 was $24.8 million, an increase of $28.4 million compared to total other income of $3.5 million for the three months ended March 31, 2020. The increase in total other expense was primarily due to higher interest expense which resulted from the loan agreements the Company amended in the fourth quarter of 2020. The Company also realized higher mark-to-market losses on liability- classified warrants and contingent consideration liabilities primarily due to changes in the Company’s share price.
Provision for Income Taxes
Income tax expense for the three months ended March 31, 2021 was $15.5 million compared to $11.2 million for the three months ended March 31, 2020. The change was due to higher gross profit and increased amounts of permanently nondeductible expenses under Section 280E related to the expansion of the Company’s business operations through organic growth and acquisitions.
Net Income (Loss)
Net loss for the three months ended March 31, 2021 was $24.1 million, compared to net loss of $35.5 million for the three months ended March 31, 2020. Higher gross profit in the current period, driven by increased revenue and operational efficiencies, was partially offset by higher operating expenses, interest expense, and current period income tax expense.
22
Non-GAAP Financial Measures
EBITDA and Adjusted EBITDA are non-GAAP measures and do not have standardized definitions under U.S. GAAP. The Company has provided the non-GAAP financial measures, which are not calculated or presented in accordance with U.S. GAAP, as supplemental information and in addition to the financial measures that are calculated and presented in accordance with U.S. GAAP and may not be comparable to similar measures presented by other issuers. These supplemental non-GAAP financial measures are presented because management has evaluated the financial results both including and excluding the adjusted items and believe that the supplemental non-GAAP financial measures presented provide additional perspective and insights when analyzing the core operating performance of the business. These supplemental non-GAAP financial measures should not be considered superior to, as a substitute for or as an alternative to, and should only be considered in conjunction with, the U.S. GAAP financial measures presented herein. Accordingly, the Company has included below reconciliations of the supplemental non-GAAP financial measures to the most directly comparable financial measures calculated and presented in accordance with U.S. GAAP.
The following table provides a reconciliation of the Company’s Net loss to Adjusted EBITDA (non-GAAP):
| Three months ended March 31, | ||||||||||||||||
($ in thousands) | 2021 | 2020 | $ Change | % Change | ||||||||||||
Net loss1 | $ | (24,124 | ) | $ | (35,461 | ) | $ | 11,337 | (32 | )% | ||||||
Depreciation and amortization | 10,151 | 6,121 | 4,030 | 66 | % | |||||||||||
Interest expense, net | 11,302 | 6,796 | 4,506 | 66 | % | |||||||||||
Income tax expense | 15,524 | 11,188 | 4,336 | 39 | % | |||||||||||
|
|
|
|
|
| |||||||||||
Earnings before interest, taxes, depreciation, and amortization (EBITDA) (non-GAAP) | $ | 12,853 | $ | (11,356 | ) | $ | 24,209 | (213 | )% | |||||||
|
|
|
|
|
| |||||||||||
Other expense (income), net | 12,340 | (10,455 | ) | 22,795 | (218 | )% | ||||||||||
Loss from equity method investments | 1,196 | 144 | 1,052 | 731 | % | |||||||||||
Fair value mark-up for acquired inventory | 585 | 1,575 | (990 | ) | (63 | )% | ||||||||||
Adjustments for acquisition and other non-core costs | 1,738 | 9,369 | (7,631 | ) | (81 | )% | ||||||||||
Management incentive compensation (share-based) | 6,280 | 2,129 | 4,151 | 195 | % | |||||||||||
|
|
|
|
|
| |||||||||||
Adjusted EBITDA (non-GAAP) | $ | 34,992 | $ | (8,594 | ) | $ | 43,586 | nm | ||||||||
|
|
|
|
|
| |||||||||||
| 1 | Net loss includes amounts attributable to non-controlling interests. |
Adjusted EBITDA (non-GAAP)
Adjusted EBITDA, a non-GAAP measure which excludes depreciation and amortization, net interest expense, income taxes, other expense, share-based compensation, adjustments for acquisition and other non-core costs, loss on equity method investments and adjustments for fair value of mark-up of inventory was $35.0 million for the three months ended March 31, 2020 compared to a $8.6 million loss for the three months ended March 31, 2021. The increase in adjusted EBITDA of $43.6 million is due to higher gross profit partially offset by higher operating expense.
23
LIQUIDITY AND CAPITAL RESOURCES
Overview
As of March 31, 2021, the Company held $255.5 million in Cash and cash equivalents, $3.5 million in Restricted cash and $105.1 million of working capital compared to December 31, 2020, where the Company held $136.3 million in Cash and cash equivalents, $4.4 million in Restricted cash and $(3.4) million of working capital. The increase of $108.5 million in working capital was primarily due to an increase in cash and cash equivalents resulting from proceeds received for the Company’s offering of subordinate voting shares during the quarter.
The Company expects cash on hand and cash flows from operations, along with private and/or public financing, will be adequate to meet capital requirements and operational needs for the next 12 months.
Cash Flows
Operating Activities
Net cash provided by operating activities was $13.0 million for the three months ended March 31, 2021, an increase of cash of $51.1 million compared to $38.2 million of cash used during the three months ended March 31, 2020. The increase in net cash provided by operating activities was primarily due to an increase in gross profit through improved efficiencies and greater scale in the Company’s established markets.
Investing Activities
Net cash used in investing activities was $26.8 million for the year ended March 31, 2021 a decrease of cash used of
$25.4 million compared to $52.2 million used in the three months ended March 31, 2020. The decrease in net cash used in investing activities was primarily due to a decrease in capital expenditures, partially offset by fewer proceeds received from sale and leaseback transactions and an increase in cash extended to entities to complete acquisitions when compared to the prior period.
Financing Activities
Net cash provided by financing activities was $132.2 million for the three months ended March 31, 2021, an increase in cash provided of $24.8 million compared to $107.5 million for the three months ended March 31, 2020. The increase in net cash provided by financing activities was primarily due to higher proceeds received from the equity offering compared to the proceeds received from the term loan in the prior period.
24
CONTRACTUAL OBLIGATIONS
As of March 31, 2021, maturities of lease liabilities were as follows:
| Operating | Finance | |||||||||||
($ in thousands) | Total | Leases | Leases | |||||||||
2021 | $ | 14,596 | $ | 11,300 | $ | 3,296 | ||||||
2022 | 19,690 | 14,891 | 4,799 | |||||||||
2023 | 20,107 | 15,076 | 5,031 | |||||||||
2024 | 21,116 | 15,994 | 5,122 | |||||||||
2025 | 21,789 | 16,538 | 5,251 | |||||||||
Thereafter | 212,296 | 175,608 | 36,688 | |||||||||
|
|
|
|
|
| |||||||
Total lease payments | $ | 309,594 | $ | 249,407 | $ | 60,187 | ||||||
|
|
|
|
|
| |||||||
Less: imputed interest | (184,434 | ) | (153,048 | ) | (31,386 | ) | ||||||
Less: tenant improvement allowance | (33,103 | ) | (32,458 | ) | (645 | ) | ||||||
|
|
|
|
|
| |||||||
Present value of lease liabilities | 92,057 | 63,901 | 28,156 | |||||||||
|
|
|
|
|
| |||||||
Less: short-term lease liabilities | (15,419 | ) | (11,984 | ) | (3,435 | ) | ||||||
|
|
|
|
|
| |||||||
Present value of long-term lease liabilities | $ | 76,638 | $ | 51,917 | $ | 24,721 | ||||||
|
|
|
|
|
| |||||||
In addition to the future minimum lease payments disclosed above, the Company is responsible for real estate taxes and common operating expenses incurred by the building or facility in which it leases space. Additionally, Cresco Labs will continue to invest in its facilities through construction and other capital expenditures as it expands its footprint in existing and new markets.
OFF-BALANCE SHEET ARRANGEMENTS AND PROPOSED TRANSACTIONS
The Company has no material undisclosed off-balance sheet arrangements or proposed transactions that have, or are reasonably likely to have, a current or future effect on its results of operations, financial condition, revenues or expenses, liquidity, capital expenditures or capital resources that are material to investors.
RELATED PARTY TRANSACTIONS
The Company’s key management personnel, consisting of the executive management team and management directors, have the authority and responsibility for planning, directing, and controlling the activities of the Company. The material transactions with related parties and changes in related party balances for the three months ended March 31, 2021 and 2020, are discussed below.
As of March 31, 2021 and December 31, 2020, the Company had income tax related receivables of $nil and $0.2 million, respectively, with key management personnel.
Key management personnel hold 100.9 million redeemable units of Cresco Labs, LLC, which is equal to $78.6 million of Non-controlling interests as of March 31, 2021. During the three months ended March 31, 2021, 79.8% of required tax distribution payments to holders of Cresco Labs, LLC were made to key management personnel. During the three months ended March 31, 2020, no tax distributions were made to key management personnel.
As of March 31, 2021, the Company had borrowings with related parties of $16.6 million related to the Company’s Amended Term Loan. During the three months ended March 31, 2021 and 2020, the Company recorded interest expense related to borrowings with related parties of $0.5 million and $0.3 million, respectively. As of March 31, 2021 and December 31, 2020, the Company had interest payable related to borrowings with related parties of $0.5 million and $0.1 million, respectively.
25
Related party lenders include Charlie Bachtell, Chief Executive Officer and member of the Board of Directors (“the Board”); Robert Sampson, member of the Board; Global Green Debt, LLC which is owned by Randy Podolsky, member of the Board; Calti LLC which is owned by Joe Caltabiano, MVS shareholder; McCormack Capital which is owned by Brian McCormack, MVS shareholder; CL Debt which is owned by Dominic Sergi, member of the Board as of March 31, 2021; a holder of minority interest in MedMar; and Vero Management LLC which is owned by individuals owning 20.6% of the Company’s outstanding redeemable shares.
The Company has lease liabilities for real estate lease agreements in which the lessors have minority interest in SLO and MedMar. The lease liabilities were incurred in January 2019 and May 2020 and will expire in 2027 through 2036.
The Company has lease liabilities for real estate lease agreements in which the lessor is Clear Heights Properties where Dominic Sergi is Chief Executive Officer. The lease liabilities were incurred by entering into operating leases, sale and leaseback transactions and financing transactions during 2020 with lease terms that will expire in 2030. During the three months ended March 31, 2021, the Company received tenant improvement allowance reimbursements of $nil related to these lease agreements and expects to receive further reimbursements of $2.2 million as of March 31, 2021.
Below is a summary of the expense resulting from the related party lease liabilities for the three months ended March 31, 2021 and 2020:
| Operating leases | ||||||||
| Three months ended March 31, | ||||||||
| 2021 | 2020 | |||||||
($ in thousands) | Rent expense | Rent expense | ||||||
Operating lease liability; lessor has minority | $ | 400 | $ | 606 | ||||
Operating lease liability; lessor has minority interest in MedMar | 57 | — | ||||||
Operating lease liability; lessor is a member of key management personnel | 277 | — | ||||||
| Finance leases | ||||||||||||||||
| Three months ended March 31, | ||||||||||||||||
| 2021 | 2020 | |||||||||||||||
| Depreciation | Interest | Depreciation | Interest | |||||||||||||
($ in thousands) | expense | expense | expense | expense | ||||||||||||
Finance lease liability; lessor has minority interest in MedMar | $ | 66 | $ | 71 | $ | 38 | $ | 44 | ||||||||
Finance lease liability; lessor is a member of key management personnel | 18 | 21 | 11 | 15 | ||||||||||||
26
Additionally, below is a summary of the ROU assets and lease liabilities attributable to related party lease liabilities.
| Operating leases | ||||||||||||||||
| As of March 31, | As of December 31, | |||||||||||||||
| 2021 | 2020 | |||||||||||||||
| ROU | Lease | ROU | Lease | |||||||||||||
($ in thousands) | asset | liability | asset | liability | ||||||||||||
Operating lease liability; lessor has minority | $ | 5,695 | $ | 9,665 | $ | 4,926 | $ | 8,560 | ||||||||
Operating lease liability; lessor has minority interest in MedMar | 1,131 | 1,143 | 1,146 | 1,187 | ||||||||||||
Operating lease liability; lessor is a member of | 6,283 | 4,760 | 6,334 | 4,783 | ||||||||||||
| Finance leases | ||||||||||||||||
| As of March 31, | As of December 31, | |||||||||||||||
| 2021 | 2020 | |||||||||||||||
| ROU | Lease | ROU | Lease | |||||||||||||
($ in thousands) | asset | liability | asset | liability | ||||||||||||
Finance lease liability; lessor has minority interest in MedMar | $ | 2,347 | $ | 2,572 | $ | 1,201 | $ | 1,365 | ||||||||
Finance lease liability; lessor is a member of key management personnel | 653 | 676 | 648 | 678 | ||||||||||||
Below is a summary of interest expense and the associated finance liability attributable to related party finance liabilities.
| Finance liabilities | ||||||||||||||||
| Three months ended March 31, 2021 | As of March 31, 2021 | Three months ended March 31, 2020 | As of December 31, 2020 | |||||||||||||
($ in thousands) | Interest expense | Finance liability | Interest expense | Finance liability | ||||||||||||
Finance lease liability; lessor is a member of key | $ | 63 | $ | 1,460 | $ | 42 | $ | 1,459 | ||||||||
FINANCIAL INSTRUMENTS AND FINANCIAL RISK MANAGEMENT
The Company’s financial instruments are held at amortized cost (adjusted for impairments or expected credit losses, as applicable) or fair value. The carrying values of financial instruments held at amortized cost approximate their fair values as of March 31, 2021 and 2020 due to their nature and relatively short maturity date. Financial assets and liabilities with embedded derivative features are carried at fair value.
Financial instruments recorded at fair value are classified using a fair value hierarchy that reflects the significance of the inputs to fair value measurements. The three levels of hierarchy are:
| • | Level 1 – Unadjusted quoted prices in active markets for identical assets or liabilities; |
27
| • | Level 2 – Inputs other than quoted prices that are observable for the asset or liability, either directly or indirectly; and |
| • | Level 3 – Inputs for the asset or liability that are not based on observable market data. |
There have been no transfers between fair value levels valuing these assets during the year.
The following tables summarize the Company’s financial instruments as of March 31, 2021 and December 31, 2020:
| March 31, 2021 | ||||||||||||||||||||
($ in thousands) | Amortized Cost | Level 1 | Level 2 | Level 3 | Total | |||||||||||||||
Financial Assets: | ||||||||||||||||||||
Cash and cash equivalents | $ | 255,503 | $ | — | $ | — | $ | — | $ | 255,503 | ||||||||||
Restricted cash1 | 3,468 | — | — | — | 3,468 | |||||||||||||||
Accounts receivable, net | 36,268 | — | — | — | 36,268 | |||||||||||||||
Loans receivable, short-term | 511 | — | — | 1,789 | 2,300 | |||||||||||||||
Loans receivable, long-term | 9,693 | — | — | — | 9,693 | |||||||||||||||
Investments | — | 9,969 | 1,049 | 119 | 11,137 | |||||||||||||||
Security deposits | 3,484 | — | — | — | 3,484 | |||||||||||||||
Financial Liabilities: | ||||||||||||||||||||
Accounts payable | $ | 36,999 | $ | — | $ | — | $ | — | $ | 36,999 | ||||||||||
Accrued liabilities | 110,395 | — | — | — | 110,395 | |||||||||||||||
Short-term borrowings | 28,014 | — | — | — | 28,014 | |||||||||||||||
Current portion of lease liabilities | 15,419 | — | — | — | 15,419 | |||||||||||||||
Deferred consideration, contingent consideration and other payables | — | 442 | — | 30,123 | 30,565 | |||||||||||||||
Derivative liabilities, long-term | — | — | — | 25,464 | 25,464 | |||||||||||||||
Lease liabilities | 76,637 | — | — | — | 76,637 | |||||||||||||||
Deferred consideration and contingent consideration | — | — | — | 7,568 | 7,568 | |||||||||||||||
Long-term notes payable and loans payable | 258,370 | — | — | — | 258,370 | |||||||||||||||
| 1 | Restricted cash balances include various escrow accounts related to investments, acquisitions, facility requirements and building improvements. |
28
| December 31, 2020 | ||||||||||||||||||||
| Amortized | ||||||||||||||||||||
($ in thousands) | Cost | Level 1 | Level 2 | Level 3 | Total | |||||||||||||||
Financial Assets: | ||||||||||||||||||||
Cash and cash equivalents | $ | 136,339 | $ | — | $ | — | $ | — | $ | 136,339 | ||||||||||
Restricted cash1 | 4,435 | — | — | — | 4,435 | |||||||||||||||
Accounts receivable, net | 29,943 | — | — | — | 29,943 | |||||||||||||||
Loans receivable, short-term | 921 | — | — | 1,517 | 2,438 | |||||||||||||||
Loans receivable, long-term | 1,204 | — | — | 20,019 | 21,223 | |||||||||||||||
Investments2 | 3,192 | — | 1,049 | 119 | 4,360 | |||||||||||||||
Security deposits | 3,558 | — | — | — | 3,558 | |||||||||||||||
Financial Liabilities: | ||||||||||||||||||||
Accounts payable | $ | 23,230 | $ | — | $ | — | $ | — | $ | 23,230 | ||||||||||
Accrued liabilities | 130,469 | — | — | — | 130,469 | |||||||||||||||
Short-term borrowings | 25,924 | — | — | — | 25,924 | |||||||||||||||
Current portion of lease liabilities | 18,040 | — | — | — | 18,040 | |||||||||||||||
Deferred consideration, contingent consideration and other payables | — | 22 | — | 19,093 | 19,115 | |||||||||||||||
Derivative liabilities, long-term | — | — | — | 17,505 | 17,505 | |||||||||||||||
Lease liabilities | 74,468 | — | — | — | 74,468 | |||||||||||||||
Deferred consideration and contingent consideration | — | — | — | 7,247 | 7,247 | |||||||||||||||
Long-term notes payable and loans payable | 255,439 | — | — | — | 255,439 | |||||||||||||||
| 1 | Restricted cash balances include various escrow accounts related to investments, acquisitions and facility licensing requirements. |
| 2 | Investment balances in the amortized cost column represent equity method investments. |
Financial Risk Management
The Company is exposed in varying degrees to a variety of financial instrument related risks. The Board and Company management mitigate these risks by assessing, monitoring and approving the Company’s risk management processes:
| (a) | Credit and Banking Risk |
Credit risk is the risk of a potential loss to the Company if a customer or a third party to a financial instrument fails to meet its contractual obligations. The maximum credit exposure at March 31, 2021 and December 31, 2020 is the carrying amount of cash, accounts receivable, and loans receivable. The Company does not have significant credit risk with respect to its customers or loan counterparties, based on cannabis industry growth in its key markets and the low interest rate environment. Although all deposited cash is placed with U.S. financial institutions in good standing with regulatory authorities, changes in U.S. federal banking laws related to the deposit and holding of funds derived from activities related to the cannabis industry have passed the House of Representatives but have not yet been voted on within the Senate. Given that current U.S. federal law provides that the production and possession of cannabis is illegal, there is a strong argument that banks cannot accept for deposit funds from businesses involved with the cannabis industry.
The novel coronavirus (“COVID-19”) was declared a pandemic by the World Health Organization on March 12, 2020. During the fourth quarter of 2020, the first vaccine utilized to prevent coronavirus infection was approved by the U.S. Food and Drug Administration. As of March 31, 2021 the vaccine is
29
becoming more widely available, however, there remains significant economic uncertainty and consequently it is difficult to reliably measure the potential impact of this uncertainty on the Company’s future financial results.
| (b) | Asset Forfeiture Risk |
Because the cannabis industry remains illegal under U.S. federal law, any property owned by participants in the cannabis industry which are either used in the course of conducting such business, or are the proceeds of such business, could be subject to seizure by law enforcement and subsequent civil asset forfeiture. Even if the owner of the property was never charged with a crime, the property in question could still be seized and subject to an administrative proceeding by which, with minimal due process, it could be subject to forfeiture.
| (c) | Liquidity Risk |
Liquidity risk is the risk that the Company will not be able to meet its financial obligations associated with financial liabilities. The Company primarily manages liquidity risk through the management of its capital structure by ensuring that it will have sufficient liquidity to settle obligations and liabilities when due. As of March 31, 2021, the Company had working capital (defined as current assets less current liabilities) of $105 million, which reflects the equity raise.
| (d) | Market Risk |
| (i) | Currency Risk |
The operating results and balance sheet of the Company are reported in U.S. dollars. As of March 31, 2021 and December 31, 2020, the Company’s financial assets and liabilities are denominated primarily in U.S. dollars. However, from time to time some of the Company’s financial transactions are denominated in currencies other than the U.S. dollar. The results of the Company’s operations are subject to currency transaction and translation risks. The Company recorded a $0.7 million foreign exchange loss and a $0.3 million foreign exchange gain during the three months ended March 31, 2021 and 2020, respectively.
As of March 31, 2021 and December 31, 2020, the Company had no hedging agreements in place with respect to foreign exchange rates. The Company has not entered into any agreements or purchased any instruments to hedge possible currency risks at this time.
| (ii) | Interest Rate Risk |
Interest rate risk is the risk that the fair value or future cash flows of a financial instrument will fluctuate because of changes in market interest rates. An increase or decrease in the Company’s incremental borrowing rate by 10% would result in an associated increase or decrease in Deferred consideration, contingent consideration and other payables and Interest expense, net of $0.2 million. The Company’s effective interest rates for its Term Loan range from 15.8% to 17.7% and the stated interest rate varies from 12.0% to 12.7% based on the term elected by the lender.
| (iii) | Price Risk |
Price risk is the risk of variability in fair value due to movements in equity or market prices. The Company is subject to price risk related to derivative liabilities and contingent consideration that are valued based on the Company’s own stock price. An increase or decrease in stock price by 10% would result in an associated increase or decrease to Deferred consideration, contingent consideration and other payables, Derivative liabilities, long-term and Deferred consideration and contingent consideration with a corresponding change to Other (expense) income net. As of March 31, 2021 an increase or decrease in stock price by 10% would result in an impact of $7.2 million or $7.0 million, respectively.
30
| (iv) | Tax Risk |
Tax risk is the risk of changes in the tax environment that would have a material adverse effect on the Company’s business, results of operations, and financial condition. Currently, state licensed marijuana businesses are assessed a comparatively high effective federal tax rate due to section 280E, which bars businesses from deducting all expenses except their cost of goods sold when calculating federal tax liability. Any increase in tax levies resulting from additional tax measures may have a further adverse effect on the operations of the Company, while any decrease in such tax levies will be beneficial to future operations.
| (v) | Regulatory Risk |
Regulatory risk pertains to the risk that the Company’s business objectives are contingent, in part, upon the compliance of regulatory requirements. Due to the nature of the industry, the Company recognizes that regulatory requirements are more stringent and punitive in nature. Any delays in obtaining, or failure to obtain regulatory approvals can significantly delay operational and product development and can have a material adverse effect on the Company’s business, results of operation, and financial condition. The Company is cognizant of the advent of regulatory changes occurring in the cannabis industry on the city, state, and national levels. Although regulatory outlook on the cannabis industry has been moving in a positive trend, the Company is aware of the effect that unforeseen regulatory changes could have on the goals and operations of the business as a whole.
31