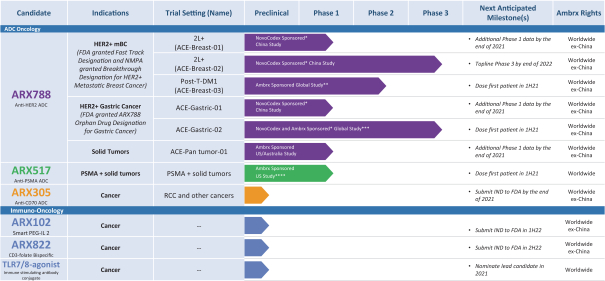
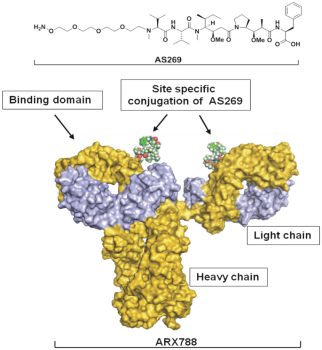
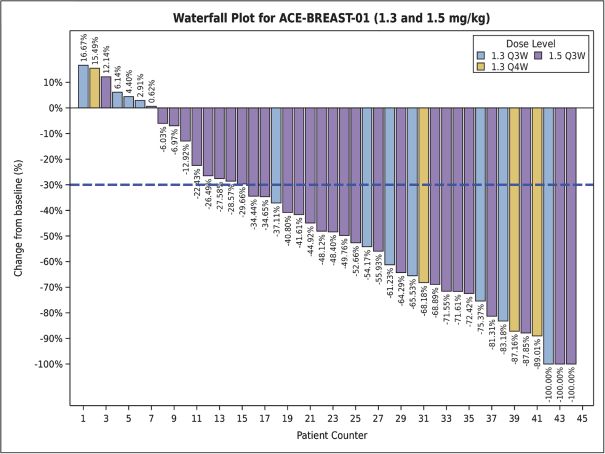
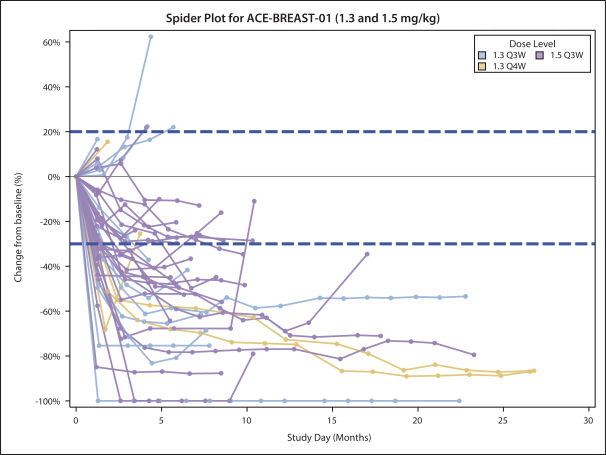
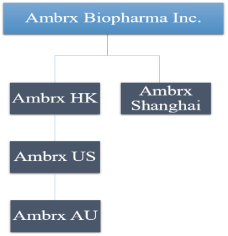
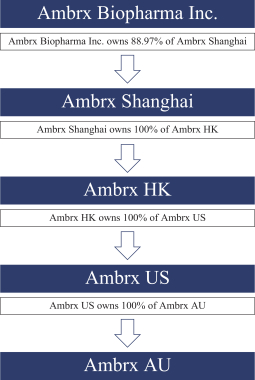
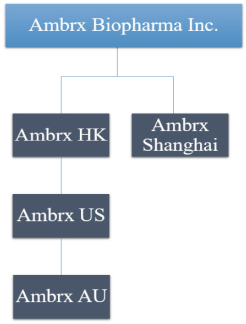
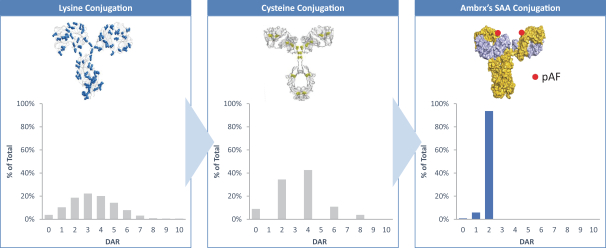
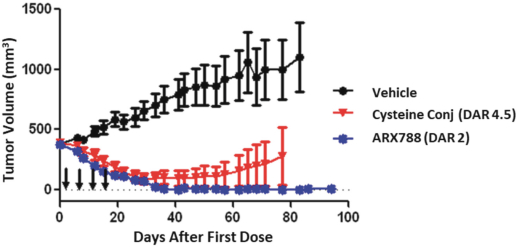
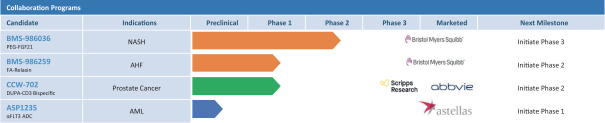
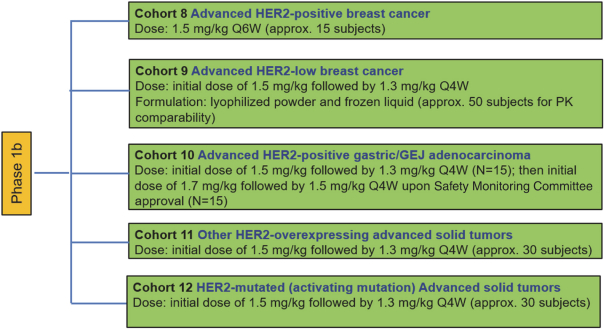
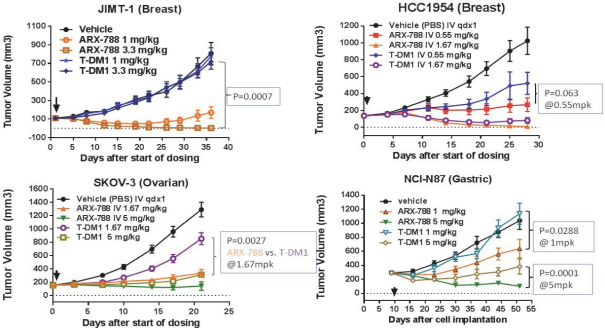
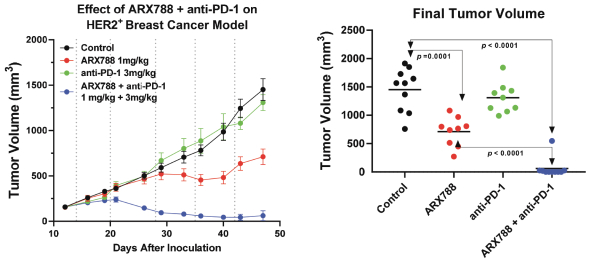
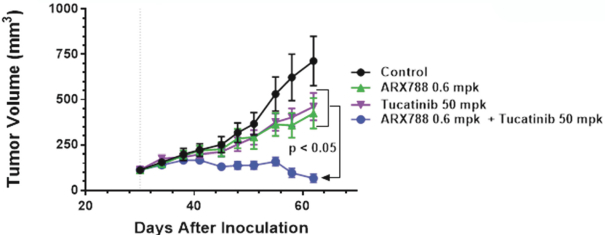
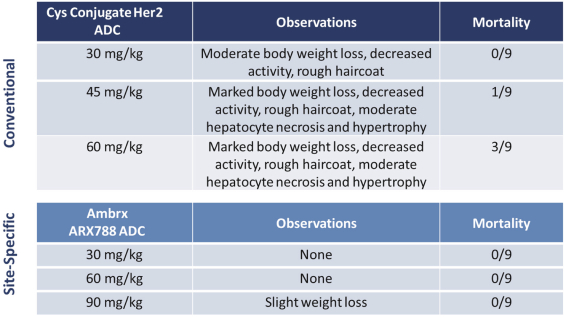
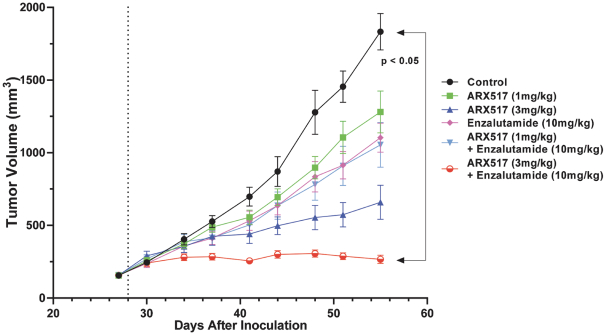
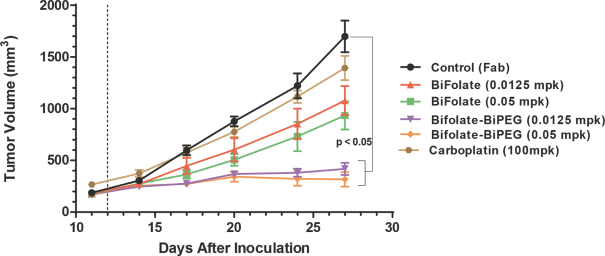
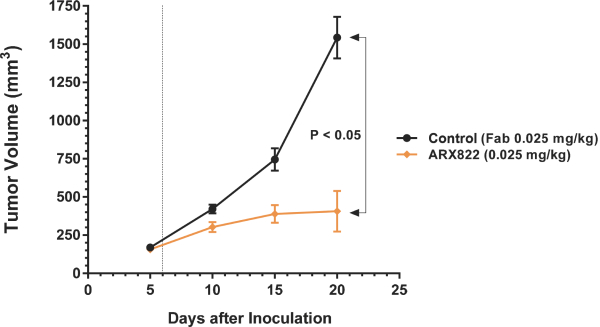
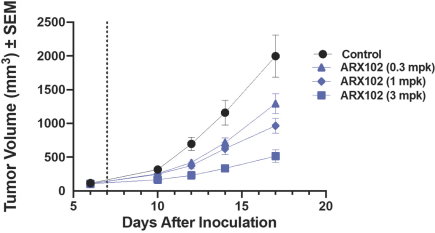
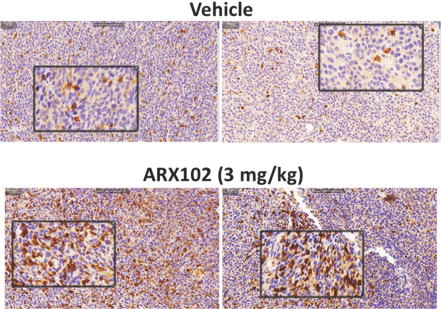
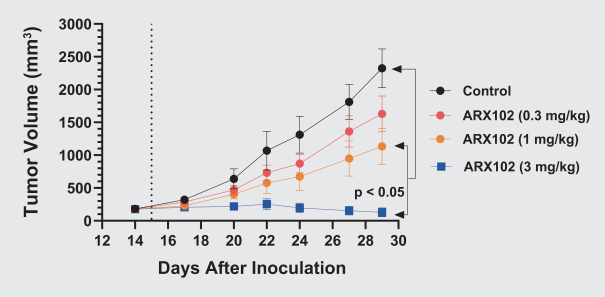
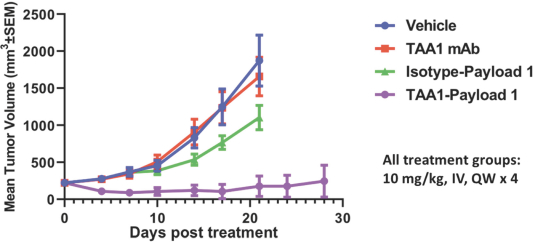
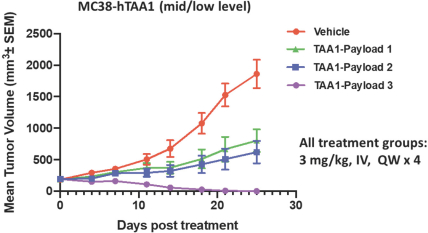
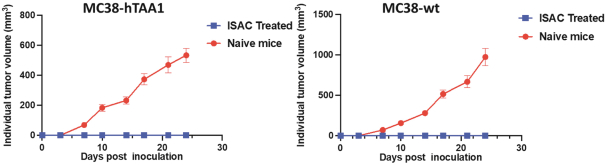
practicing our proprietary technology and commercializing our EuCODE and ReCODE technology platforms and product candidates. Our issued patents and those that may issue in the future may be challenged, narrowed, circumvented, or invalidated, which could limit our ability to stop competitors from marketing related platforms or product candidates or limit the length of the term of patent protection that we may have for our ADC and IOC product candidates. In addition, the rights granted under any issued patents may not provide us with protection or competitive advantages against competitors with similar technology. Furthermore, our competitors may independently develop similar technologies. For these reasons, we may have competition for our ADC and IOC product candidates. Moreover, because of the extensive time required for development, testing and regulatory review of a potential product, it is possible that before any product candidate can be commercialized, any related patent may expire or remain in force for only a short period following commercialization, thereby reducing any advantage of the patent for this and other risks related to our proprietary technology, inventions, improvements, platforms and product candidates. Please see “Risk Factors—Risks Related to Our Intellectual Property” for additional information on the risks associated with our intellectual property strategy and portfolio.
Government Regulation
Government authorities in the United States at the federal, state and local level and in other countries and jurisdictions, including the People’s Republic of China (PRC), extensively regulate, among other things, the research, development, testing, manufacture, quality control, approval, safety, efficacy, labeling, packaging, storage, record-keeping, promotion, advertising, distribution, post-approval monitoring and reporting, marketing and export and import of drug and biological products. Generally, before a new drug or biologic can be marketed, considerable data demonstrating its quality, safety and efficacy must be obtained, organized into a format specific for each regulatory authority, submitted for review and approved by the regulatory authority.
Regulatory Approval of Biologics in the United States
In the United States, biological products used for the prevention, treatment or cure of a disease or condition of a human being, such as our product candidates, are subject to regulation under the Federal Food, Drug, and Cosmetic Act (the FDCA), and the Public Health Service Act (the PHSA), their implementing regulations, and other federal, state, and local regulations that govern, among other things, the research, development, testing, manufacture, quality control, approval, safety, efficacy, labeling, packaging, storage, record-keeping, promotion, advertising, distribution, import and export, post-approval monitoring and reporting, and marketing of biological products. The process of obtaining regulatory approvals and the subsequent compliance with appropriate federal, state, local and foreign statutes and regulations require the expenditure of substantial time and financial resources. Failure to comply with applicable U.S. requirements may subject a company to a variety of administrative or judicial sanctions, such as clinical hold, FDA refusal to approve pending BLAs, warning or untitled letters, product recalls, product seizures, total or partial suspension of production or distribution, injunctions, fines, civil penalties and criminal prosecution.
Further, even if we obtain the required regulatory approvals for our product candidates, pharmaceutical companies are subject to myriad federal, state, and foreign healthcare laws, rules, and regulations governing all aspects of our operations, including, but not limited to, our relationships with healthcare professionals, healthcare institutions, distributors of our products, and sales and marketing personnel; governmental and other third-party payor coverage and reimbursement of our products; and data privacy and security. Such laws, rules, and regulations are complex, continuously evolving, and, in many cases, have not been subject to extensive interpretation by applicable regulatory agencies or the courts. We are required to invest significant time and financial resources in policies, procedures, processes, and systems to ensure compliance with these laws, rules, and regulations, and our failure to do so may result in the imposition of substantial monetary or other penalties by federal or state regulatory agencies.
187