Exhibit 99.1

APRI L 2023 In v e stor P re s e n ta tion Co gn o s t h e ra p e u t i cs, i n c. No c t urne ac q u i s i t i o n c o rp .

FORWARD LOOKING STATEMENTS This presentation (“ Presentation ”) is for informational purposes only to assist interested parties in making their own evaluation with respect to the proposed offering (the “ Offering ”) of Cognos Therapeutics, Inc., a Delaware corporation (“ Cognos ”), of common stock. The offering is being made in connection with, but is not subject to or conditioned upon, the consummation of a proposed business combination (the “ Business Combination ”) between Nocturne Acquisition Corporation (“ Nocturne ”) and the Company (the “ Purpose ”). By accepting this Presentation, the recipient acknowledges and agrees that all of the information contained herein is confidential, that the recipient will distribute, disclose, and use such information only for such Purpose and that the recipient shall not distribute, disclose or use such information in anyway detrimental to Cognos or Nocturne. The information contained herein does not purport to be all - inclusive and none of Nocturne, Cognos, and Maxim Group nor any of their respective affiliates nor any of its or their controlling persons, officers, directors, employees or representatives makes any representation or warranty, express or implied, as to the accuracy, completeness or reliability of the information contained in this Presentation. You should consult your own counsel and tax and financial advisors as to legal and related matters concerning the matters described herein, and, by accepting this Presentation, you confirm that you are not relying upon the information contained herein to make any decision. Any securities to be offered in any transaction contemplated hereby have not been registered under the Securities Act of 1933, as amended (the “ Securities Act ”), or any applicable state or foreign securities law. Any securities to be offered in any transaction contemplated hereby have not been approved or disapproved by the Securities and Exchange Commission (the “ SEC ”), any state securities commission or other United States or foreign regulatory authority and will be offered and sold solely in reliance on the exemption from the registration requirements provided by the Securities Act and rules and regulations promulgated thereunder (including Regulation D or Regulation S under the Securities Act). Neither the SEC or any U.S. state securities commission has approved or disapproved the Offering, passed upon the merits or fairness of the Offering, or passed any comment upon the adequacy, accuracy or completeness of any disclosure in relation to the Offering. Any representation to the contrary is a criminal offence in the United States. This Presentation does not constitute, or form a part of, an offer to sell or the solicitation of an offer to buy in any state or other jurisdiction to any person to whom it is unlawful to make such offer or solicitation in such state or jurisdiction. The financial information contained in this presentation is preliminary and unaudited . As such the Company’s independent auditors have not audited, studied, reviewed or performed any procedures with respect to such preliminary information, and accordingly, have not expressed an opinion or provided any other form of assurances thereto for the Purposes of this presentation . There can be no assurance that such preliminary results are indicative of the future performance of the Company and actual results may materially differ . Certain statements in this Presentation may be considered forward - looking statements. Forward - looking statements generally relate to future events or Cognos’ or Nocturne’s future financial or operating performance. For example, statements concerning the following include forward - looking statements: Cognos’ ability to identify, develop and commercialize product candidates; the initiation, cost, timing, progress and results of research and development activities, preclinical or clinical trials with respect to Cognos’ drug candidates; future revenue, expenses, capital requirements and needs for additional financing; and the potential effects of the Business Combination on Nocturne and Cognos and related capital raising activities. In some cases, you can identify forward - looking statements by terminology such as “may”, “should”, “expect”, “intend”, “will”, “estimate”, “anticipate”, “believe”, “predict”, “plan”, “potential” or “continue”, or the negatives of these terms or variations of them or similar terminology. Such forward - looking statements are subject to risks, uncertainties, and other factors which could cause actual results to differ materially from those expressed or implied by such forward - looking statements. These forward - looking statements are based upon estimates and assumptions that, while considered reasonable by Nocturne and its management, and Cognos and its management, as the case may be, are inherently uncertain. New risks and uncertainties may emerge from time to time, and it is not possible to predict all risks and uncertainties. Factors that may cause actual results to differ materially from current expectations include, but are not limited to, various factors beyond management’s control including general economic conditions (such as rising interest rates or bank failures) and other risks, uncertainties and factors set forth in the section entitled “Risk Factors” and “Cautionary Note Regarding Forward - Looking Statements” in Nocturne’s final prospectus relating to its initial public offering, dated February 22, 2021, and other filings with the SEC, as well as factors associated with companies, such as Cognos, that are engaged in preclinical studies and other research and development activities in the biopharma industry, including uncertainty in the timing or results of preclinical studies and clinical trials, product acceptance and/or receipt of regulatory approvals for product candidates, including any delays and other impacts from the Covid - 19 pandemic. Additionally, important factors that could cause actual results to differ materially from those presented or implied in the forward - looking statements include, among others: without limitation, (i) the ability of the Company to complete the development of its products in a timely manner, (ii) the demand for and timing of demand for such products, (iii) competition from other products and companies, (iv) the results of the Company’s safety and efficacy studies, (v) delay and challenges the results of the regulatory approval process, (vi) the Company’s sales and marketing capabilities, (vii) the Company’s ability to sell its products profitably, (viii) the ability of the Company’s third party suppliers to provide products and services in timely and a reliable manner ; (ix) availability of adequate debt and equity financing on favorable terms and (x) general business and economic conditions . There can be no assurance that the Company will be able to anticipate, respond to, or adapt to changes in any factors affecting the Company’s business and financial results . 2

FORWARD LOOKING STATEMENTS With the exception of the historical information contained in this presentation, the matters described herein contain forward - looking statements that involve risk and uncertainties that individually or jointly impact the matters herein described, including but not limited to financial projections, product demand and market acceptance, the effect of economic conditions, the impact of competitive products and pricing, governmental regulations, technological difficulties and/or other factors outside the control of the Company. These important factors and certain other factors that might affect the Company’s financial and business results are discussed in this presentation. Nothing in this Presentation should be regarded as a representation by any person that the forward - looking statements set forth herein will be achieved or that any of the contemplated results of such forward - looking statements will be achieved. You should not place any reliance on forward - looking statements in this Presentation, which speak only as of the date they are made and are qualified in their entirety by reference to the cautionary statements herein. Neither Nocturne nor Cognos undertakes any duty to update these forward - looking statements. Certain information contained in this Presentation relates to or is based on studies, publications, surveys and Cognos’ own internal estimates and research. In addition, all of the market data included in this Presentation involve a number of assumptions and limitations, and there can be no guarantee as to the accuracy or reliability of such assumptions. Finally, while Cognos believes its internal research is reliable, such research has not been verified by any independent source. Cognos has retained Maxim Group as placement agent (together with its affiliates, partners, directors, agents, employees, representatives, and controlling persons, the “ Placement Agent ”) with respect to capital raising activities in connection with the Business Combination. The Placement Agent is acting solely as placement agent (and, for the avoidance of doubt, not as underwriter, initial purchaser, dealer or any other principal capacity) for Cognos in connection with a potential transaction. The Placement Agent has not independently verified any of the information contained herein or any other information that has been or will be provided to you. Nothing contained herein or in any other oral or written information provided to you is, nor shall be relied upon as, a promise or representation of any kind by the Placement Agent, Nocturne or Cognos, whether as to the past or the future. Without limitation of the foregoing, none of the Placement Agents, Nocturne or Cognos shall be liable to you or any prospective investor or any other person for any information contained herein or that otherwise has been or will be provided to you, or any action heretofore or hereafter taken or omitted to be taken, in connection with this potential transaction. This Presentation is being distributed solely for the consideration of sophisticated prospective purchasers who are accredited investors, including institutional investors who are accredited investors, with sufficient knowledge and experience in investment, financial and business matters and the capability to conduct their own due diligence investigation and evaluation in connection with the Purpose. This Presentation does not purport to summarize all of the conditions, risks and other attributes of an investment in Nocturne or Cognos. Information contained herein will be superseded by, and is qualified in its entirety by reference to, any other information that is made available to you in connection with the Purpose, including your investigation of Nocturne and Cognos. Additional Information . In connection with the proposed Business Combination, Nocturne intends to file with the SEC a registration statement on Form S - 4 containing a preliminary proxy statement/prospectus of Nocturne, and after the registration statement is declared effective, Nocturne will mail a definitive proxy statement/prospectus relating to the proposed Business Combination to its shareholders. This Presentation does not contain all the information that should be considered concerning the proposed Business Combination and is not intended to form the basis of any investment decision or any other decision in respect of the Business Combination. Nocturne’s shareholders and other interested persons are advised to read, when available, the preliminary proxy statement/prospectus and the amendments thereto and the definitive proxy statement/prospectus and other documents filed in connection with the proposed Business Combination, as these materials will contain important information about Cognos, Nocturne and the Business Combination. When available, the definitive proxy statement/prospectus and other relevant materials for the proposed Business Combination will be mailed to shareholders of Nocturne as of a record date to be established for voting on the proposed Business Combination. Nocturne shareholders will also be able to obtain copies of the preliminary proxy statement/prospectus, the definitive proxy statement/prospectus and other documents filed with the SEC, without charge, once available, at the SEC’s website at www.sec.gov. Participants in the Solicitation. Nocturne and its directors and executive officers may be deemed participants in the solicitation of proxies from Nocturne’s shareholders with respect to the proposed Business Combination. A description of the interests of Nocturne’s directors and executive officers in Nocturne is contained in Nocturne’s final prospectus relating to its initial public offering, February 22, 2021, which was filed with the SEC and is available free of charge at the SEC’s website at www.sec.gov. Additional information regarding the interests of such participants will be contained in the proxy statement/prospectus for the proposed Business Combination when available. 3

4 Nocturne’s team has extensive experience in disruptive technologies, business operations and investing. Henry Monzon Chairman & CEO Seasoned Executive and Entrepreneur with deep expertise in various technology domains such as wireless, semiconductors, blockchain and artificial intelligence. Former roles include: ■ CEO & Co - Founder of Katena Computing Technologies ■ Chief Financial Officer at Luminous Computing ■ Chief Revenue Officer at Bitfury Group ■ Sr. Director, Business Development at Qualcomm Direct access to strategic and financial capital pool in and out of Silicon Valley Ability to offer operator level expertise to targets Proven track record in tech operating, investing and deal - making Proprietary in - house target identification capability Ka Seng (Thomas) Ao Chief Financial Officer Extensive experience with M&A transactions and financial leasing. Current and former roles include: ■ Chief Executive Officer of MCL Financial Leasing, a provider of financial leasing services for bitcoin mining facilities, since January 2020 ■ Director of Mindfulness Capital ■ Previously the Vice President of HF Venture Overview of Nocturne Acquisition Corporation

AT A GLANCE FOUNDATION STRATEGIC COLLABORATORS CAPITAL RAISED $18.5 Million* 5 2006 2015 FOUNDED DELAWARE CORP AS PHARMACO - KINESIS COMPANY SPINOFF AS COGNOS THERAPEUTICS INTELLECTUAL PROPERTY ( US & International) Issued Patents 30 Filed Non - Provisional Patent Applications 52 *Includes $2.5 million from a revenue share agreement with Tako Ventures, LLC, a Larry Ellison controlled venture capital fund, that will convert into a 25% ownership interest in COGNOS immediately prior to closing of the proposed business combination with Nocturne. Developing propriety bonding technique Developing propriety piezoelectric micropump Center where Cognos conducted animal survival study in sheep Signed NDA and in discussion to do joint study for use of SINNAIS Œ to deliver drug to brain Conducted a small study to demonstrate local delivery of a combination of Avastin ® and Campto ® improves survival rate in mice HISTORY YEARS OF R&D EXPERTISE Cognos Therapeutics, Inc. 2015 to Present Pre - Spin Off - Pharmaco - Kinesis Corp. 2006 to 2015 COGNOS THERAPEUTICS (“ COGNOS ”) is a medical technology company focused on creating advanced implantable pump devices for neurological and oncological indications intended to adjust the course of therapeutics for the treatment and mitigation of disease models where current technology does not provide an effective solution . Our flagship product, the SINNAIS TM Implantable Smart Pump (ISP), is a fully implantable pump that is designed to metronomically administer therapeutics directly to the central nervous system (CNS), bypassing the blood - brain barrier (BBB) with potential to enable stable, continuous drug delivery for improved outcomes for brain cancers and other CNS diseases . COGNOS is also working on developing a Smart Drug Optical Sensor (SOS) which can collect important data from the disease target site and transmit that data to the cloud (big data, IA, and IoT) for further data analysis to customize drug dose (personalized medicine) and patient disease progression monitoring, which is intended to be part of the second generation SINNAIS TM pump – to bring virtual physician concept to reality . Our mission is to develop and commercialize medical products that combine diagnostic, therapeutic, and sensing technologies with state - of - the - art drug delivery to advance healthcare through improved patient outcomes

CORPORATE HIGHLIGHTS 6 • Disruptive smart pump technology designed to offer metronomic drug delivery • A next - generation piezoelectric micropump technology for precise, metronomic dosing • The only pump under development to deliver drug intraventricularly, thereby bypassing the blood - brain barrier (BBB) • Development of biosensor capability for a second - generation pump to provide cloud - enabled physician monitoring and precision dosing • Technology may enable broad applicability across multiple indications and therapeutics • Potential for use with small molecule therapeutics and biologics (i.e.. large molecule, cell therapies, etc.) • Technology validated through multiple small and large animal model studies • Addressing large market opportunities in multiple neurological and other disease indications including cancer • Delivery of approved drugs could offer a regulatory path hoped to lower risk and greater opportunity for drug company participation • Multiple potential milestones over the next 24 months, including: • PMA submission to FDA for SINNAIS TM as a Class III pump indicated for infusion of Infumorph, potentially using FDA’s 6 - Year Rule which would allow PMA approval without conducting new human clinical trials • Investigational New Drug (IND) in effect for the initiation of Phase II human clinical trial of SINNAIS TM as a combination product for metronomic delivery of methotrexate (MTX) for the treatment of Leptomeningeal Carcinomatosis (LC) • Transition generic drugs to branded drugs utilizing FDA – combination product approval path 505(b)2 • Strong intellectual property portfolio covering the pump design, smart pump delivery, and method of use • Experienced management, board, and clinical advisors
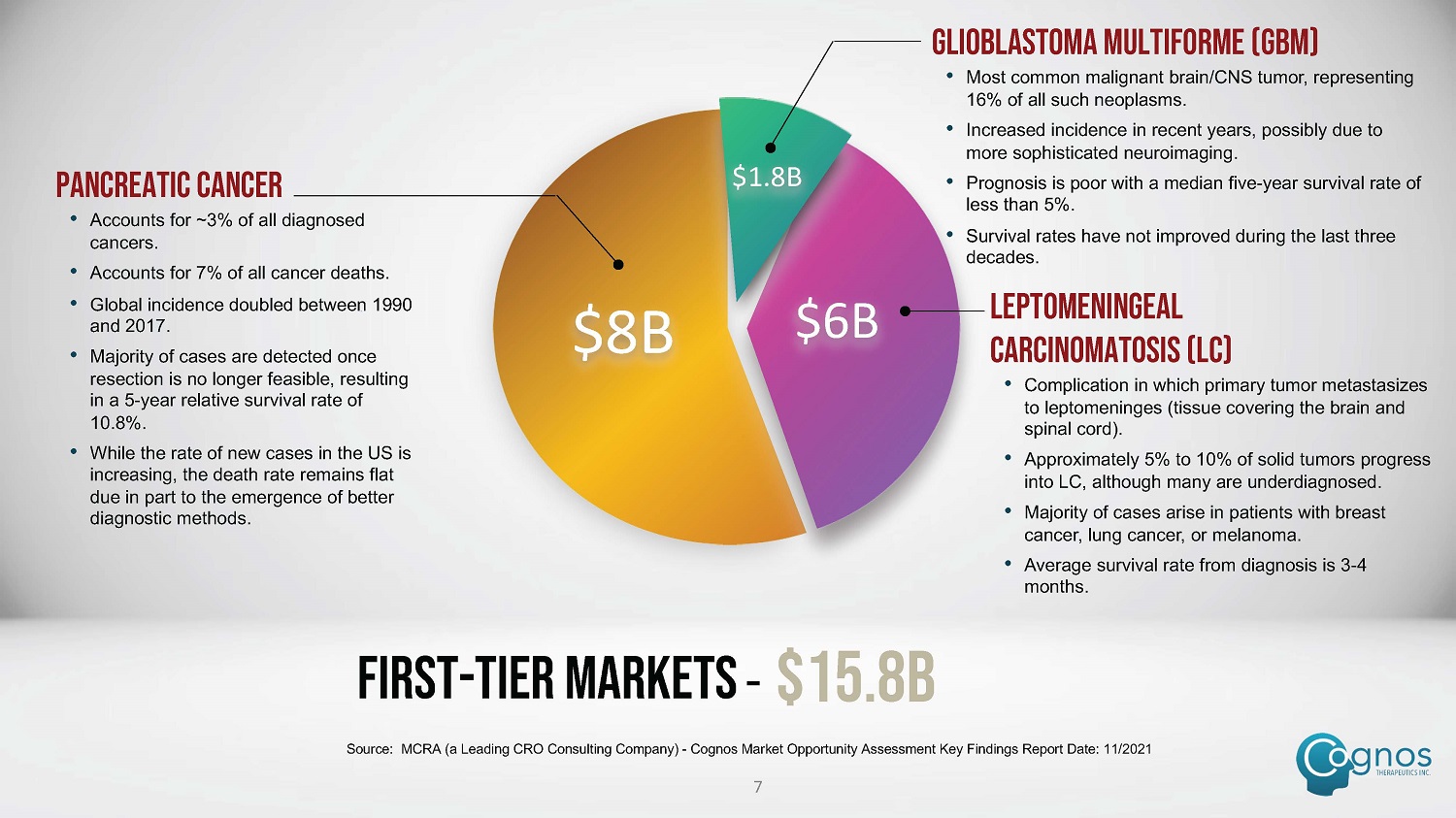
$8B $6B $1.8B LEPTOMENINGEAL CARCINOMATOSIS (LC) • Complication in which primary tumor metastasizes to leptomeninges (tissue covering the brain and spinal cord). • Approximately 5% to 10% of solid tumors progress into LC, although many are underdiagnosed. • Majority of cases arise in patients with breast cancer, lung cancer, or melanoma. • Average survival rate from diagnosis is 3 - 4 months. PANCREATIC CANCER • Accounts for ~3% of all diagnosed cancers. • Accounts for 7% of all cancer deaths. • Global incidence doubled between 1990 and 2017. • Majority of cases are detected once resection is no longer feasible, resulting in a 5 - year relative survival rate of 10.8%. • While the rate of new cases in the US is increasing, the death rate remains flat due in part to the emergence of better diagnostic methods. GLIOBLASTOMA MULTIFORME (GBM) • Most common malignant brain/CNS tumor, representing 16% of all such neoplasms. • Increased incidence in recent years, possibly due to more sophisticated neuroimaging. • Prognosis is poor with a median five - year survival rate of less than 5%. • Survival rates have not improved during the last three decades. FIRST - TIER MARKETS – $15.8B Source: MCRA (a Leading CRO Consulting Company) - Cognos Market Opportunity Assessment Key Findings Report Date: 11/2021 7
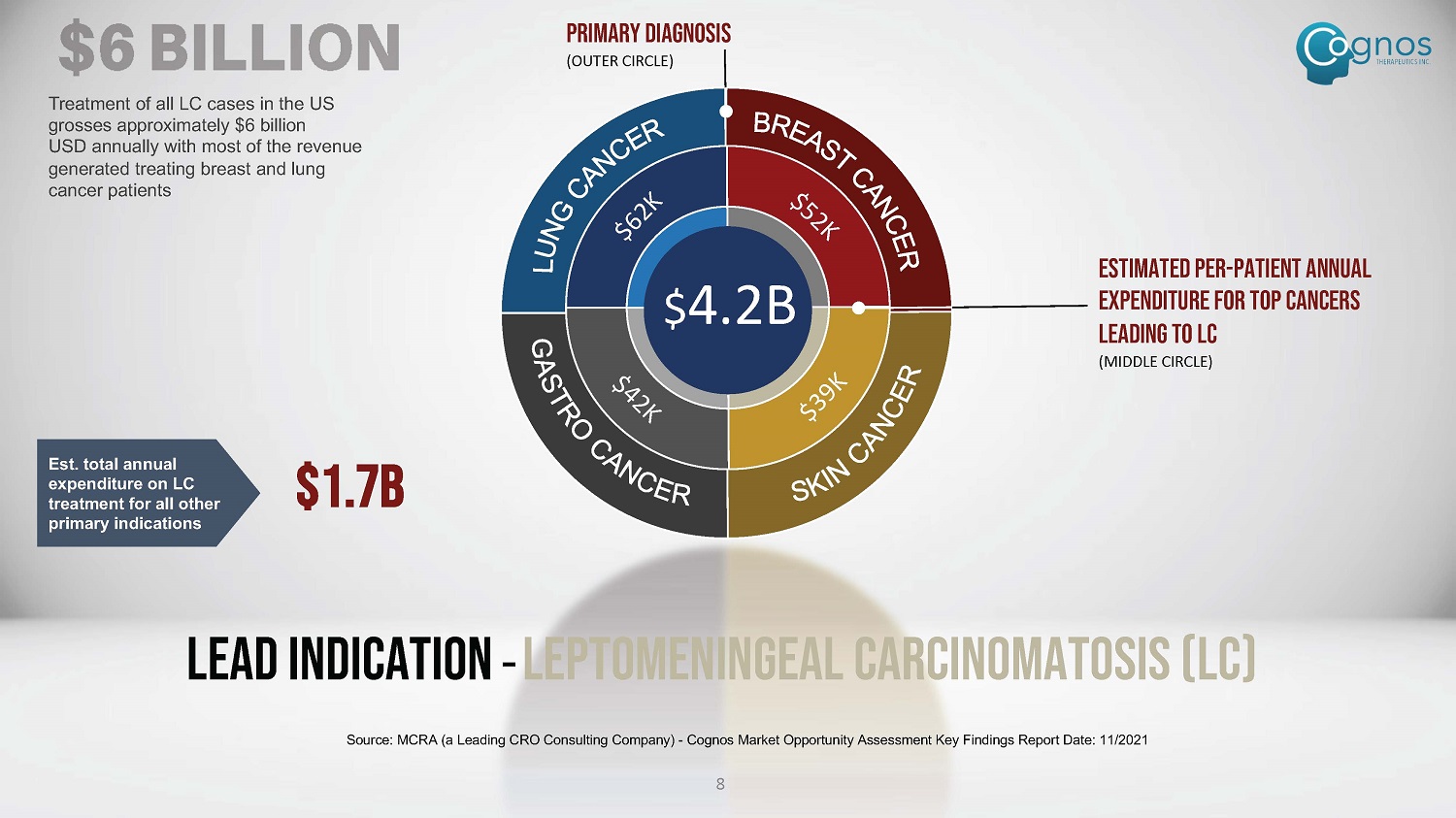
Estimated per - patient annual expenditure for top cancers leading to LC (MIDDLE CIRCLE) PRIMARY DIAGNOSIS (OUTER CIRCLE) Treatment of all LC cases in the US grosses approximately $6 billion USD annually with most of the revenue generated treating breast and lung cancer patients $ 4.2B Est. total annual expenditure on LC treatment for all other primary indications $1.7B 8 Lead Indication – LEPTOMENINGEAL CARCINOMATOSIS (LC) Source: MCRA (a Leading CRO Consulting Company) - Cognos Market Opportunity Assessment Key Findings Report Date: 11/2021

Challenges of CURRENT PUMP TECHNOLOGY All of the current commercially available pumps are based on technology and architecture that is more than three decades old and pose the following problems: • No intraventricular delivery – Drug cannot bypass blood - brain barrier (BBB). • Not customizable to each patient. • Do not automatically respond to situational changes in the patient’s condition • Have a limited ability to receive delivery commands. • Are unable to deliver precise and accurate dosing. • Can not provide a metronomic delivery of drugs, which provides programmable scheduled dosing within a therapeutic range vs bolus injection. • Are not able to provide real - time biofeedback and communication. • Have limited disease model application. • Do not integrate with other systems. • Are not MRI - compatible. • Cannot meet many of the new safety regulatory requirements set by the FDA to deliver therapeutics to the brain. • Due to crude dose size (1ml +) and not being able to provide information about level toxicity after drug administered in brain are not suitable to deliver drug to brain region. Source: MCRA (a Leading CRO Consulting Company) A Market Opportunity Assessment Key Findings, a report dated: 11/21/2022 9

THE SOLUTION BENEFITS TO STAKEHOLDERS The SINNAIS TM Implantable pump is expected to provide many key benefits across the healthcare spectrum to patients, healthcare providers and drug manufacturing companies FOR PATIENTS • Improved quality of life and mobility • Lower side effects • Shorter hospital stays • Lower health insurance premium • Lower hospitalization cost • Increases postoperative longevity FOR HEALTHCARE PROVIDERS • Reduced costs • Access to the patient’s “Big Data” using AI to apply triage and better diagnostics FOR PHARMACEUTICAL COMPANIES • Converts a generic drug into a proprietary BRANDED drug • Reduces cost of development for improved drug efficacy • Increases the life of the intellectual property • Ability to adapt the system to various disease models and drugs • SINNAIS TM is believed to be the world’s first implantable pump with SMART that delivers drugs locally and metronomically device under development • Intended to provide direct delivery of therapeutics into the brain ventricle, and tumor sites by passing BBB, or targeted organ • Potential to deliver neuroleptics locally to treat brain cancers, neurodegenerative diseases, and mental illnesses associated with the brain such as Alzheimer’s, Parkinson’s, and MS • Fully MRI and biocompatible implantable system • Can be implanted in the chest or abdomen • Precise programmable schedules • Micro - dose adjustment control based on a patient’s reaction and tolerance • Customizes and optimizes dosage to patients’ specific pharmacokinetic and pharmacodynamic needs – Personalized Medicine • Enables delivery of multiple drugs* • Secure high - level encrypted wireless communication through the cloud from anywhere in the world • Refilling Next Generation SINNAIS TM Additional Features: INTRODUCING SINNAIS TM Metronomic drug delivery directly into the brain, bypassing the Blood - Brain - Barrier (BBB). Some key features/attributes include: • Biofeedback capability in real - time • Uses AI for predictive disease outcome - - Virtual physician • SMART shunt and smart optical sensor (SOS) can monitor drug toxicity and concentration in real - time using a wireless connection to the cloud • Can be refilled and recharged transcutaneously *SINNAIS TM piezoelectric micropump is made of titanium and silicon therefore the chemistry of material in pump does not interact with drugs regardless of drug biological and chemistry composition. 10
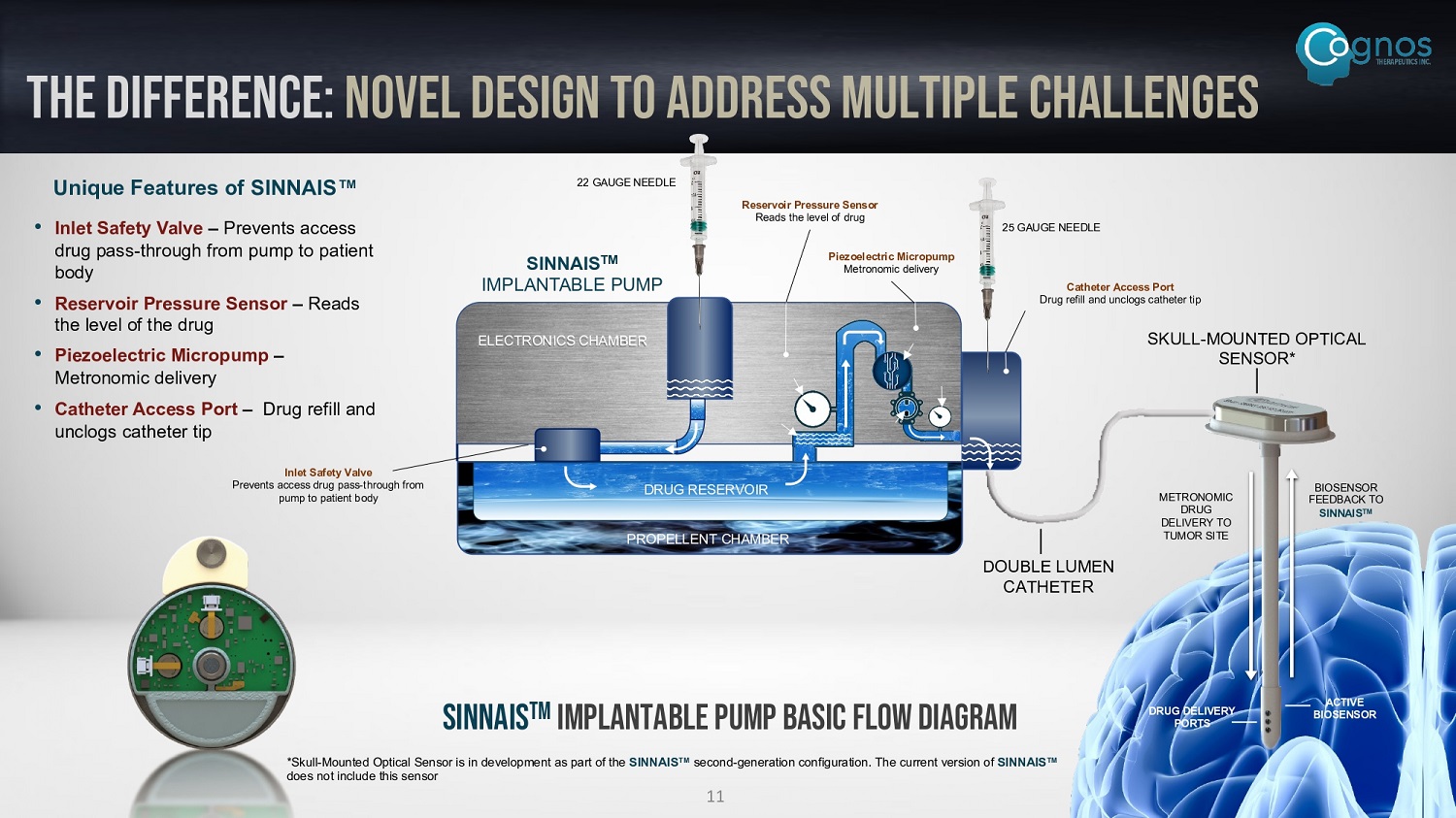
THE DIFFERENCE: novel design to address multiple challenges DRUG RESERVOIR PROPELLENT CHAMBER ELECTRONICS CHAMBER 22 GAUGE NEEDLE 25 GAUGE NEEDLE SKULL - MOUNTED OPTICAL SENSOR* SINNAIS TM IMPLANTABLE PUMP DOUBLE LUMEN CATHETER METRONOMIC DRUG DELIVERY TO TUMOR SITE BIOSENSOR FEEDBACK TO SINNAIS TM DRUG DELIVERY PORTS ACTIVE BIOSENSOR TM SINNAIS Implantable Pump Basic Flow Diagram Inlet Safety Valve Prevents access drug pass - through from pump to patient body Reservoir Pressure Sensor Reads the level of drug Piezoelectric Micropump Metronomic delivery Catheter Access Port Drug refill and unclogs catheter tip Unique Features of SINNAIS Œ • Inlet Safety Valve – Prevents access drug pass - through from pump to patient body • Reservoir Pressure Sensor – Reads the level of the drug • Piezoelectric Micropump – Metronomic delivery • Catheter Access Port – Drug refill and unclogs catheter tip *Skull - Mounted Optical Sensor is in development as part of the SINNAIS TM second - generation configuration. The current version of SINNAIS TM does not include this sensor 11
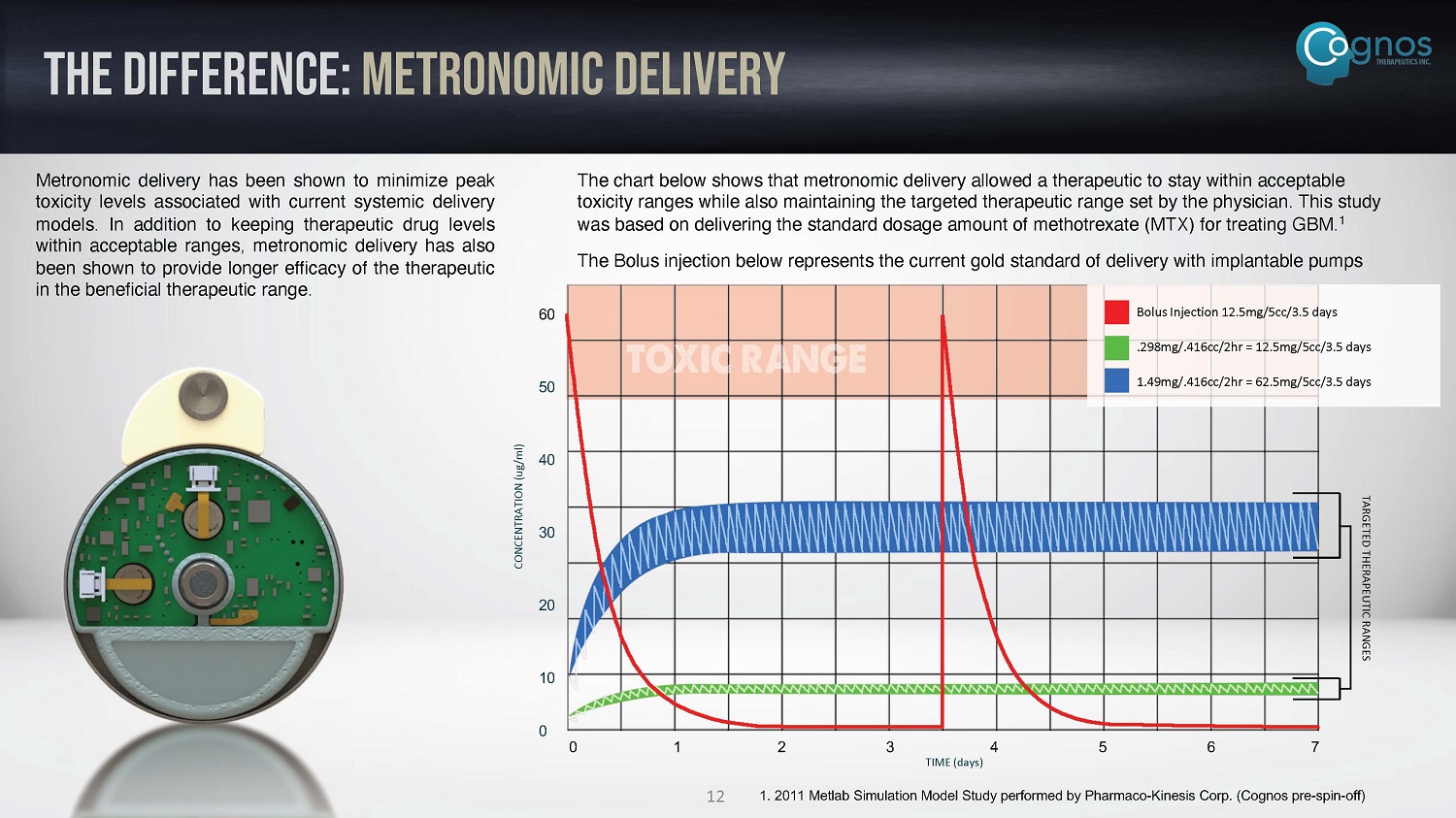
THE DIFFERENCE: metronomic delivery Metronomic delivery has been shown to minimize peak toxicity levels associated with current systemic delivery models . In addition to keeping therapeutic drug levels within acceptable ranges, metronomic delivery has also been shown to provide longer efficacy of the therapeutic in the beneficial therapeutic range . The chart below shows that metronomic delivery allowed a therapeutic to stay within acceptable toxicity ranges while also maintaining the targeted therapeutic range set by the physician. This study was based on delivering the standard dosage amount of methotrexate (MTX) for treating GBM. 1 The Bolus injection below represents the current gold standard of delivery with implantable pumps 0 1 2 3 4 5 6 7 TIME (days) 60 50 40 30 20 10 0 CONCENTRATION (ug/ml) Bolus Injection 12.5mg/5cc/3.5 days .298mg/.416cc/2hr = 12.5mg/5cc/3.5 days 1.49mg/.416cc/2hr = 62.5mg/5cc/3.5 days 1. 2011 Metlab Simulation Model Study performed by Pharmaco - Kinesis Corp. (Cognos pre - spin - off) TARGETED THERAPEUTIC RANGES 12
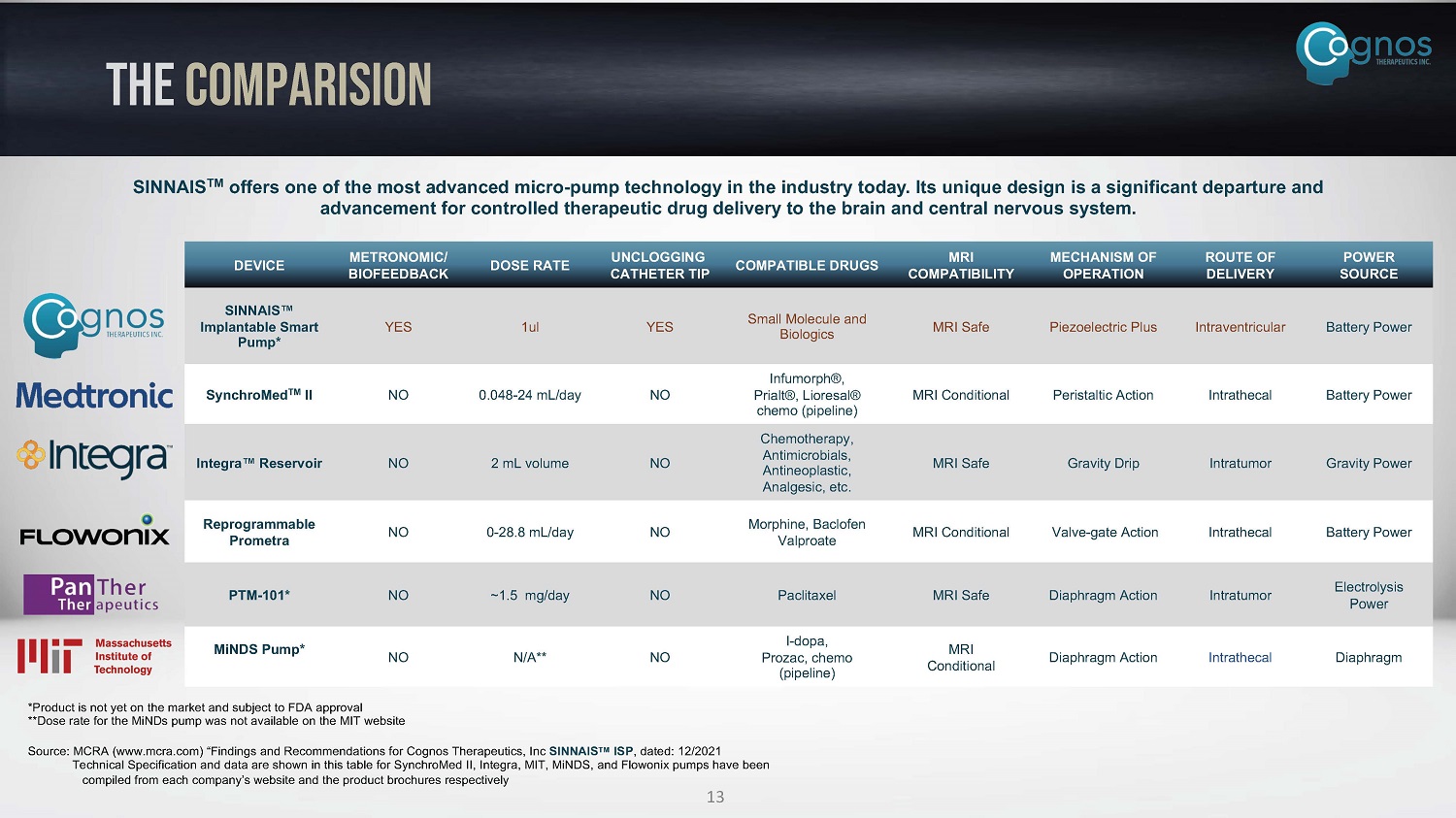
DEVICE METRONOMIC/ BIOFEEDBACK DOSE RATE UNCLOGGING CATHETER TIP COMPATIBLE DRUGS MRI COMPATIBILITY MECHANISM OF OPERATION ROUTE OF DELIVERY POWER SOURCE SINNAIS Œ Implantable Smart Pump* YES 1ul YES Small Molecule and Biologics MRI Safe Piezoelectric Plus Intraventricular Battery Power SynchroMed TM II NO 0.048 - 24 mL/day NO Infumorph®, Prialt®, Lioresal® chemo (pipeline) MRI Conditional Peristaltic Action Intrathecal Battery Power Chemotherapy, Integra Œ Reservoir NO 2 mL volume NO Antimicrobials, MRI Safe Gravity Drip Intratumor Gravity Power Antineoplastic, Analgesic, etc. Reprogrammable Prometra NO 0 - 28.8 mL/day NO Morphine, Baclofen Valproate MRI Conditional Valve - gate Action Intrathecal Battery Power PTM - 101* NO ~1.5 mg/day NO Paclitaxel MRI Safe Diaphragm Action Intratumor Electrolysis Power MiNDS Pump* NO N/A** NO I - dopa, Prozac, chemo (pipeline) MRI Conditional Diaphragm Action Intrathecal Diaphragm THE COMPARISION *Product is not yet on the market and subject to FDA approval **Dose rate for the MiNDs pump was not available on the MIT website Source: MCRA (www.mcra.com) “Findings and Recommendations for Cognos Therapeutics, Inc SINNAIS TM ISP , dated: 12/2021 Technical Specification and data are shown in this table for SynchroMed II, Integra, MIT, MiNDS, and Flowonix pumps have been compiled from each company’s website and the product brochures respectively 13 SINNAIS TM offers one of the most advanced micro - pump technology in the industry today. Its unique design is a significant departure and advancement for controlled therapeutic drug delivery to the brain and central nervous system.
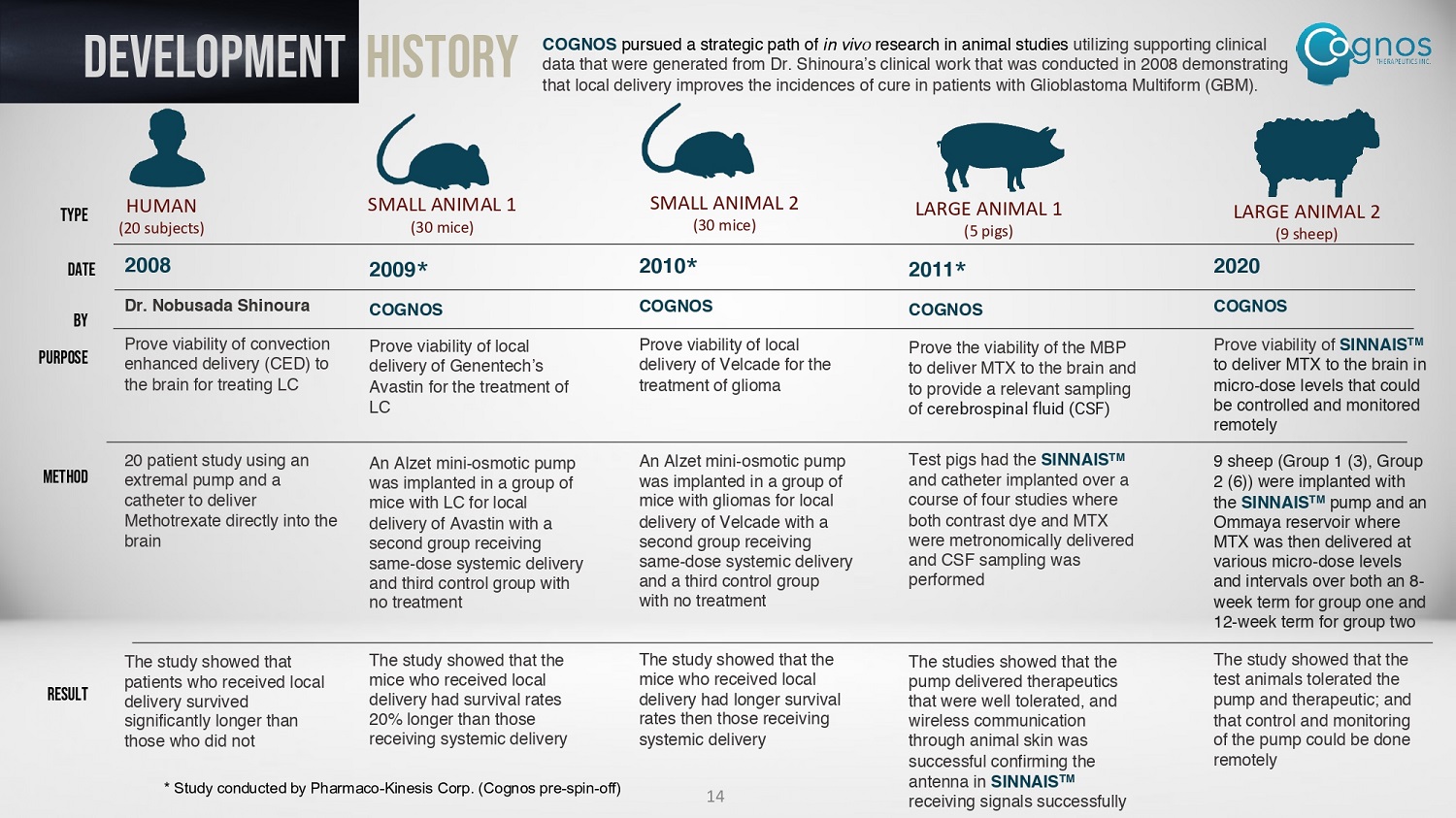
DEVELOPMENT History COGNOS pursued a strategic path of in vivo research in animal studies utilizing supporting clinical data that were generated from Dr. Shinoura’s clinical work that was conducted in 2008 demonstrating that local delivery improves the incidences of cure in patients with Glioblastoma Multiform (GBM). HUMAN (20 subjects) SMALL ANIMAL 1 (30 mice) SMALL ANIMAL 2 (30 mice) LARGE ANIMAL 1 (5 pigs) LARGE ANIMAL 2 (9 sheep) 2008 Dr. Nobusada Shinoura Prove viability of convection enhanced delivery (CED) to the brain for treating LC 20 patient study using an extremal pump and a catheter to deliver Methotrexate directly into the brain The study showed that patients who received local delivery survived significantly longer than those who did not 2010 * COGNOS Prove viability of local delivery of Velcade for the treatment of glioma An Alzet mini - osmotic pump was implanted in a group of mice with gliomas for local delivery of Velcade with a second group receiving same - dose systemic delivery and a third control group with no treatment The study showed that the mice who received local delivery had longer survival rates then those receiving systemic delivery 2009 * COGNOS Prove viability of local delivery of Genentech’s Avastin for the treatment of LC An Alzet mini - osmotic pump was implanted in a group of mice with LC for local delivery of Avastin with a second group receiving same - dose systemic delivery and third control group with no treatment The study showed that the mice who received local delivery had survival rates 20% longer than those receiving systemic delivery 2011 * COGNOS Prove the viability of the MBP to deliver MTX to the brain and to provide a relevant sampling of cerebrospinal fluid (CSF) Test pigs had the SINNAIS TM and catheter implanted over a course of four studies where both contrast dye and MTX were metronomically delivered and CSF sampling was performed The studies showed that the pump delivered therapeutics that were well tolerated, and wireless communication through animal skin was successful confirming the antenna in SINNAIS TM receiving signals successfully 2020 COGNOS Prove viability of SINNAIS TM to deliver MTX to the brain in micro - dose levels that could be controlled and monitored remotely 9 sheep (Group 1 (3), Group 2 (6)) were implanted with the SINNAIS TM pump and an Ommaya reservoir where MTX was then delivered at various micro - dose levels and intervals over both an 8 - week term for group one and 12 - week term for group two The study showed that the test animals tolerated the pump and therapeutic; and that control and monitoring of the pump could be done remotely TYPE DATE BY PURPOSE METHOD RESULT 14 * Study conducted by Pharmaco - Kinesis Corp. (Cognos pre - spin - off)

studies Laboratory Practices) using sheep • Eight - week survival study • Studied verified: • Wireless communication • Pump delivery capabilities • Ability to safely refill the implanted pump OBJECTIVES • Validate the pump’s ability to deliver metronomic dosing of a therapeutic to the brain in a large animal model • Test the ability of the pump’s wireless data and system control to allow for remote dose adjustment (metronomic delivery) and the flow delivery data to be received and monitored 1 st Pilot Study 2 nd Pilot Study • Three animal non - GLP (Good • Six animal non - GLP study using sheep • Twelve - week survival study • Study verified: • Metronomic pumping at micro - dose levels • Delivery of Methotrexate (MTX) to the brain • Durability and biocompatibility of the pump Dr. Wu checks communication protocols between the SINNAIS TM pump and several smartphones being used in the trial. SINNAIS TM pump is connected to catheter prior to implantation in a test animal. An Omamya reservoir connected to the SINNAIS TM pump is implanted in the test animal’s brain. The research team monitors the SINNAIS TM pump’s function after the implantation. 15 VALIDATION STUDIES – SHEEP MODEL RESULTS • Provided insight to continue to refine the pump’s accuracy and biocompatibility in providing adjustable, controlled micro - dose delivery of a therapeutic • Provided insight to develop the protocols for control of the system, CSF sampling, and data monitoring via wireless communication to a remote computer or an app - enabled smartphone
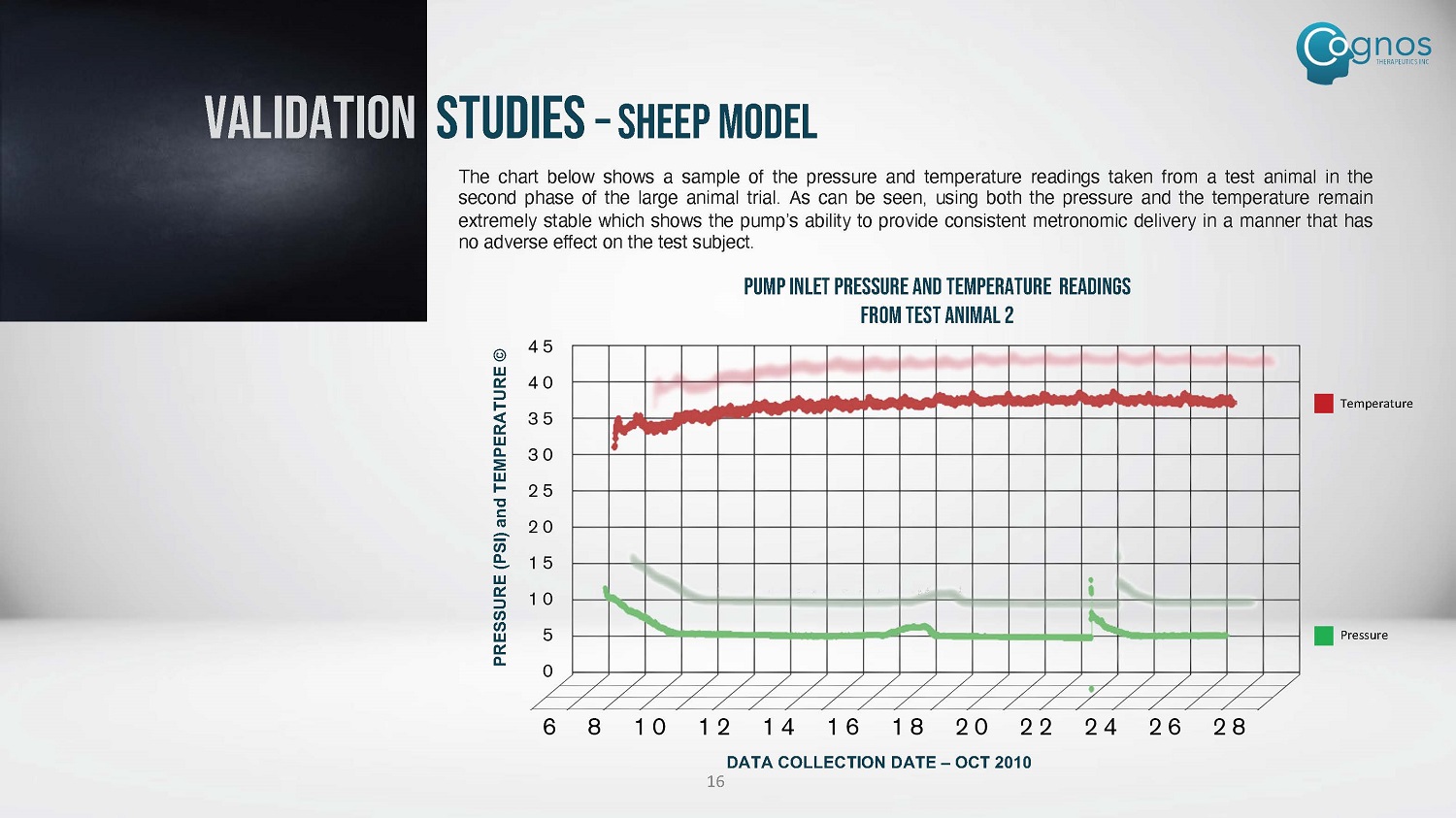
PRESSURE (PSI) and TEMPERATURE © The chart below shows a sample of the pressure and temperature readings taken from a test animal in the second phase of the large animal trial . As can be seen, using both the pressure and the temperature remain extremely stable which shows the pump’s ability to provide consistent metronomic delivery in a manner that has no adverse effect on the test subject . PUMP INLET PRESSURE and TEMPERATURE READINGS FROM TEST ANIMAL 2 Pressure Temperature VALIDATION STUDIES – SHEEP MODEL DATA COLLECTION DATE – OCT 2010 16
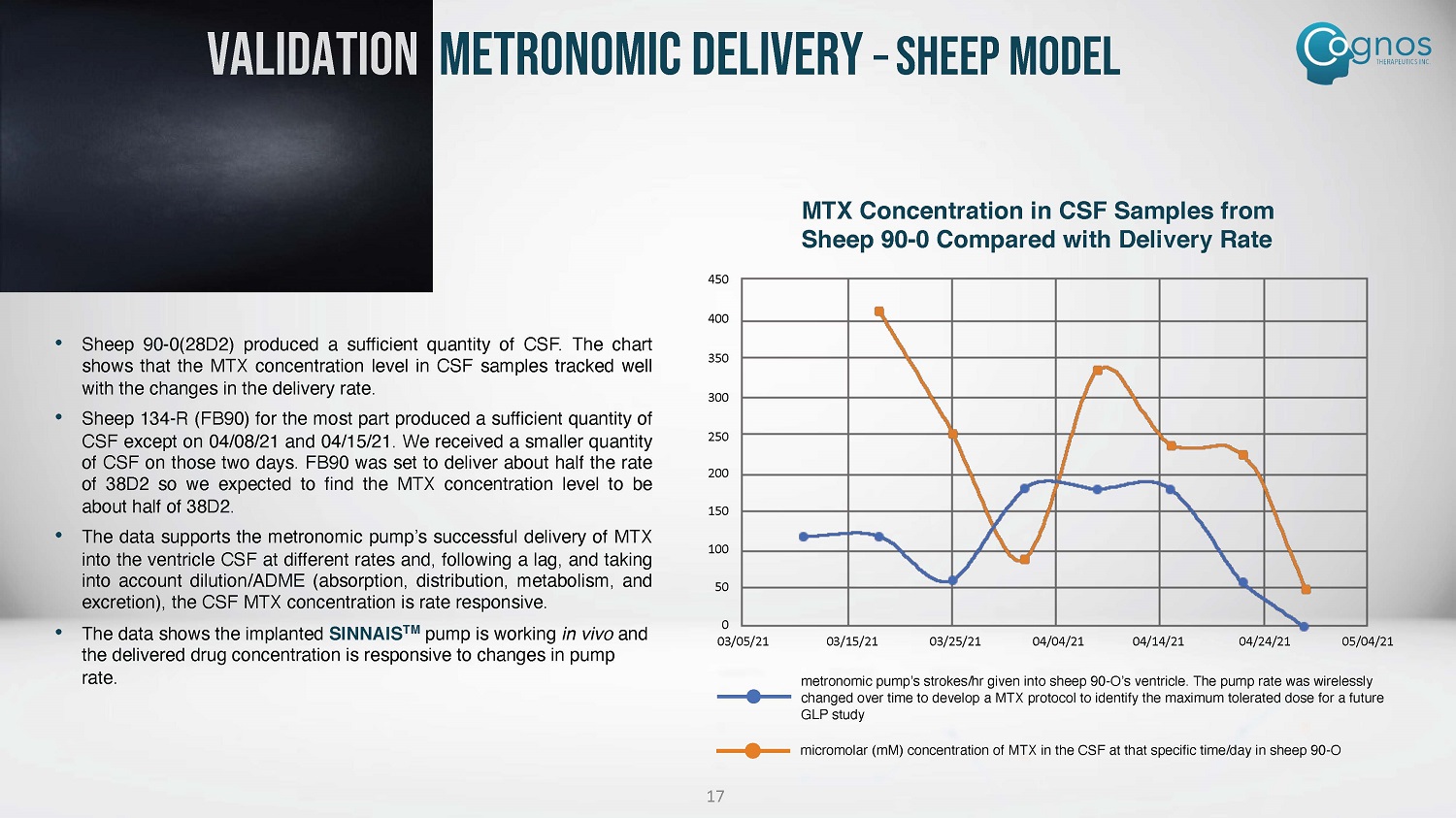
VALIDATION METRONOMIC DELIVERY – SHEEP MODEL • Sheep 90 - 0 ( 28 D 2 ) produced a sufficient quantity of CSF . The chart shows that the MTX concentration level in CSF samples tracked well with the changes in the delivery rate . • Sheep 134 - R (FB 90 ) for the most part produced a sufficient quantity of CSF except on 04 / 08 / 21 and 04 / 15 / 21 . We received a smaller quantity of CSF on those two days . FB 90 was set to deliver about half the rate of 38 D 2 so we expected to find the MTX concentration level to be about half of 38 D 2 . • The data supports the metronomic pump’s successful delivery of MTX into the ventricle CSF at different rates and, following a lag, and taking into account dilution/ADME (absorption, distribution, metabolism, and excretion), the CSF MTX concentration is rate responsive . • The data shows the implanted SINNAIS TM pump is working in vivo and the delivered drug concentration is responsive to changes in pump rate. MTX Concentration in CSF Samples from Sheep 90 - 0 Compared with Delivery Rate 450 400 350 300 250 200 150 100 50 0 03/05/21 03/15/21 03/25/21 04/04/21 04/14/21 04/24/21 05/04/21 metronomic pump’s strokes/hr given into sheep 90 - O’s ventricle. The pump rate was wirelessly changed over time to develop a MTX protocol to identify the maximum tolerated dose for a future GLP study micromolar (mM) concentration of MTX in the CSF at that specific time/day in sheep 90 - O 17

Combined Regulatory FDA and EU Approval Strategy U.S. Q4 2021 – Filed pre - submission with FDA for PMA submission for SINNAIS TM Q1 2022 – Received FDA’s written pre - sub feedback, which informed PMA submission for SINNAIS TM ~18 Months* – Obtain PMA submission to FDA approval for for SINNAIS TM as a Class III pump indicated for infusion of Infumorph, potentially using FDA’s 6 - year Rule which would allow PMA approval without conducting new human clinical trials as a device alone (from Device Center (CDRH) and Drug Center (CDER) ~ 24 Months* – Complete Phase II human clinical trial of SINNAIS TM as a combination product for metronomic delivery of MTX for treatment of LC under IND from CDER E.U. ~24 Months* – Obtain CE - Mark submission for SINNAIS TM as device alone based upon data with the first drug: methotrexate. After SINNAIS TM receives CE - Mark in EU market, SINNAIS TM will be allowed to be used with any approved drug in the European market without requiring to go through additional regulatory approvals 18 * Expected development timeline of closing of the business combination – See slide 19 for further detail
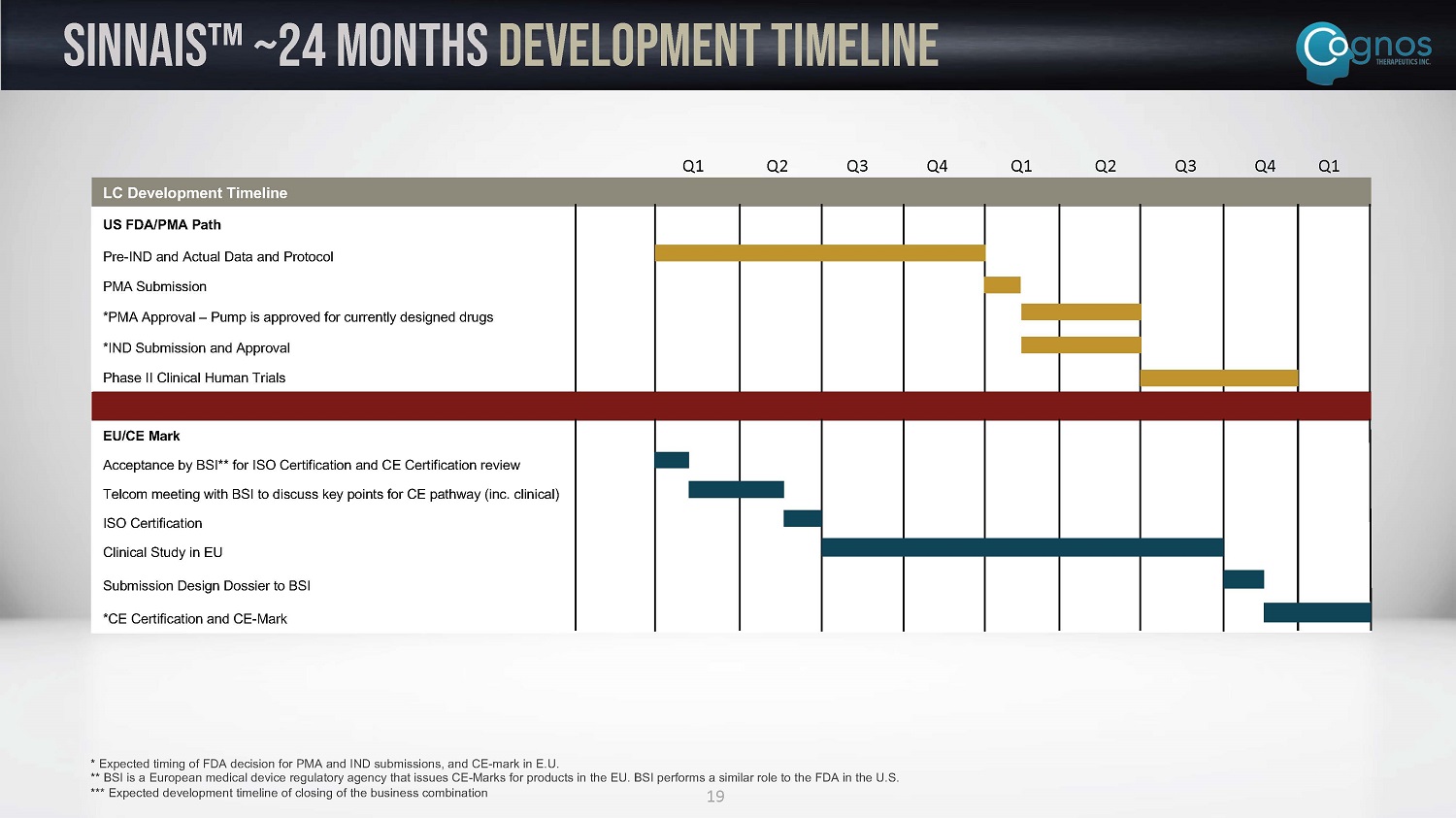
19 SINNAIS Ρ ~24 MONTHS development timeline * Expected timing of FDA decision for PMA and IND submissions, and CE - mark in E.U. ** BSI is a European medical device regulatory agency that issues CE - Marks for products in the EU. BSI performs a similar role to the FDA in the U.S. *** Expected development timeline of closing of the business combination LC Development Timeline US FDA/PMA Path Pre - IND and Actual Data and Protocol PMA Submission *PMA Approval – Pump is approved for currently designed drugs *IND Submission and Approval Phase II Clinical Human Trials EU/CE Mark Acceptance by BSI** for ISO Certification and CE Certification review Telcom meeting with BSI to discuss key points for CE pathway (inc. clinical) ISO Certification Clinical Study in EU Submission Design Dossier to BSI *CE Certification and CE - Mark Q1 Q2 Q3 Q4 Q1 Q2 Q3 Q4 Q1

comprehensive IP PORTFOLIO COGNOS has always pursued an aggressive strategy of identifying and protecting its Intellectual Property assets through global patent protection COGNOS IP Portfolio by the Numbers: ISSUED – 30 Patents issued in the U.S. and Internationally FILED – 52 Patent Applications filed in the U.S. and Internationally • Implantable pump for detection of spine issues • Implantable piezoelectric pump for delivery of therapeutics to the spine • A method for Cerebral Microdialysis to treat neurological disease • Implantable piezoelectric pump for delivery of biological response modifiers • Artificial Tooth Medicating device for local delivery of therapeutics • An implantable magnetic breather pump for local delivery of bevacizumab into a brain tumor • A method for creation and manufacture of a hermetic seal for use in an implantable metronomic drug pump • Creation of a MRI compatible drug pump with overpressure protection • Method for the intratumoral delivery of Bortezomib • Development of a Magnetic Breather Pump for tumor treatment • An implantable pump for the local delivery of intrathecal chemotherapy for Leptomeningpal Carcinomatosis • Development of a Multipurpose Cerebrospinal Fluid Sensor • Development of a Skull - Mounted Drug And Pressure Sensor 20 IP Portfolio Covers

Cognos Therapeutics Founders & Management TEAM JOSH SHACHAR* Co - Founder CHIEF IP & INNOVATION OFFICER THOMAS CHEN,* MD, PhD Co - Founder CHIEF NEUROSURGEON & ONCOLOGY OFFICER THOMAS LOBL, PhD* CHIEF OPERATING OFFICER SUSAN ALPERT*, MD, PhD* LEAD REGULATORY CONSULTANT WINSTON WU, PhD SR. VP. PRODUCT DEVELOPMENT AND CHIEF TECHNOLOGY OFFICER ROBERT ABRAMS* VICE PRESIDENT SOFTWARE ENGINEERING ELI GANG, MD, FACC, FACP* CHIEF MEDICAL OFFICER FRANK ADELL Co - Founder CHIEF EXECUTIVE OFFICER JAAP LAUFER, MD, PhD* LEAD REGULATORY FOR EU CE - MARK DARCI DIAGE* MANAGER, SPECIAL REGULATORY AND COMPLIANCE 21 * Consultant MEDI ABBIS* INTERIM CHIEF FINANCIAL OFFICER ROGER KORNBERG, PhD* STRATEGIC ADVISOR and CO - FOUNDER

Cognos Therapeutics ANTICIPATED BOARD OF DIRECTORS 22 JOSH SHACHAR DIRECTOR THOMAS CHEN, MD, PhD DIRECTOR FRANK ADELL DIRECTOR KEITH BRADLEY, Ph.D* DIRECTOR NOMINEE CHRISTOPHER SMITH* DIRECTOR NOMINEE CRAIG BURSON* DIRECTOR NOMINEE * Agreed to join the Board upon closing of the business combination RICK PANICUCCI, Ph.D* DIRECTOR NOMINEE

Summary transaction overview 23 Overview Ownership Financing Transaction Rationale Use of Proceeds • The combined company will be known as Cognos Therapeutics Holding Corp. • Will operate under the same management team as Cognos Therapeutics, Inc. • Anticipated closing in Q3 2023 • The transaction contemplates an enterprise value of $120 million for Cognos Therapeutics, Inc. • As part of the transaction, all shares owned by Cognos’ existing equity holders will be converted into Class A common stock • There is expected to be a minimum of $10 million in cash on the balance sheet at closing that may be sourced from a combination of the cash held in trust, net of redemptions, cash on the balance sheet, and through additional financing sources • Provides Cognos Therapeutics, Inc. with access to the capital markets, allowing it to accelerate the development and advancement of its technology and future applicability • Multiple near - term value inflection points • To advance the development of SINNAIS TM toward an Investigational New Drug (IND) for the initiation of Phase II human clinical trial of SINNAIS TM as a combination product for metronomic delivery of Methotrexate for the treatment of Leptomeningeal Carcinomatosis & obtaining a CE market in the E.U. • Working capital and general corporate purposes

detailed transaction overview 24 1. Assumes no further redemptions by Nocturne public shareholders and no cash from Cognos’ balance sheet 2. Estimated $10 million in gross proceeds to close concurrently with the business combination 3. Pro forma diluted basis at $10.30 per share, assumes no redemptions and excludes the to – be - established post - business combination company equity incentive plan 4. Includes Nocturne’s assumption of current Cognos warrants, options and restricted stock units 5. Does not factor in the shares underlying certain Cognos convertible notes 6. Includes the Nocturne Founder’s shares and Private Placement shares Stock Issuance to Cognos Nocturne Sponsor Shares Nocturne Cash in Trust Cash from PIPE $120.0 $34.9 $20.1 $10.0 Total Sources $185.0 Uses ($mm) Cognos Equity Rollover $120.0 Nocturne Sponsor Shares $34.9 Cash to Balance Sheet $17.9 Estimated Transaction Fees & Expenses $12.2 Total Uses $185.0 Pro Forma Valuation Illustrative Sources & Uses ($mm, except per share data) Sources ($mm) Pro Forma Shares Outstanding (x) Illustrative Share Price 18.8 $10.30 Pro Forma Equity Value $194.1 ( - ) Pro Forma Net Cash $17.9 Pro Forma Enterprise Value $176.2 Pro Forma Share Ownership 60% 16% 18% 6% Existing Cognos Shareholders Nocturne Public Shareholders Nocturne Sponsor Shares PIPE Investors























