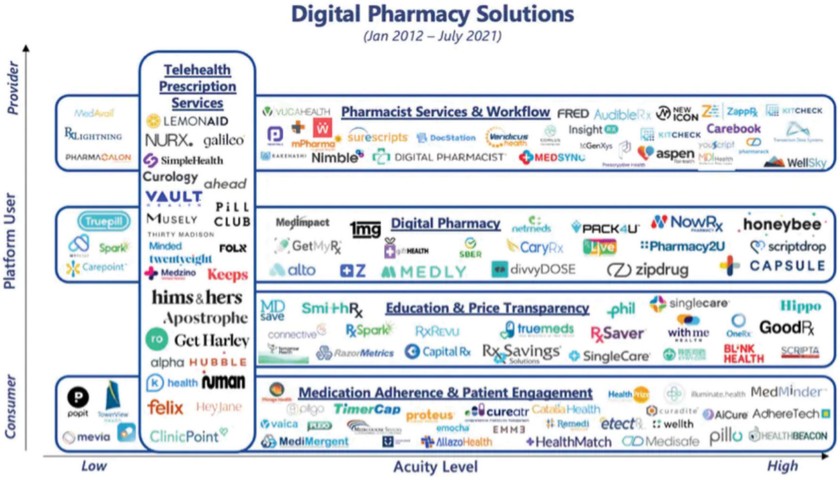
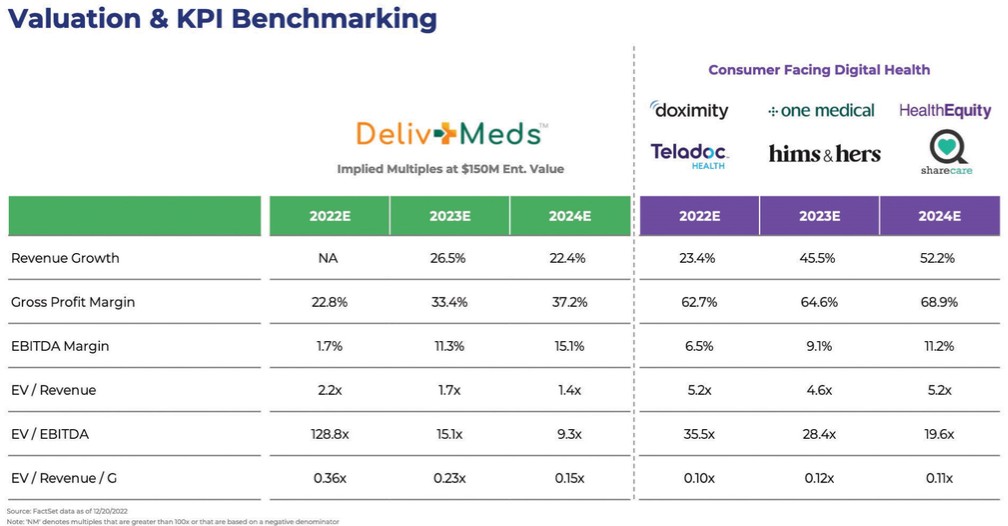
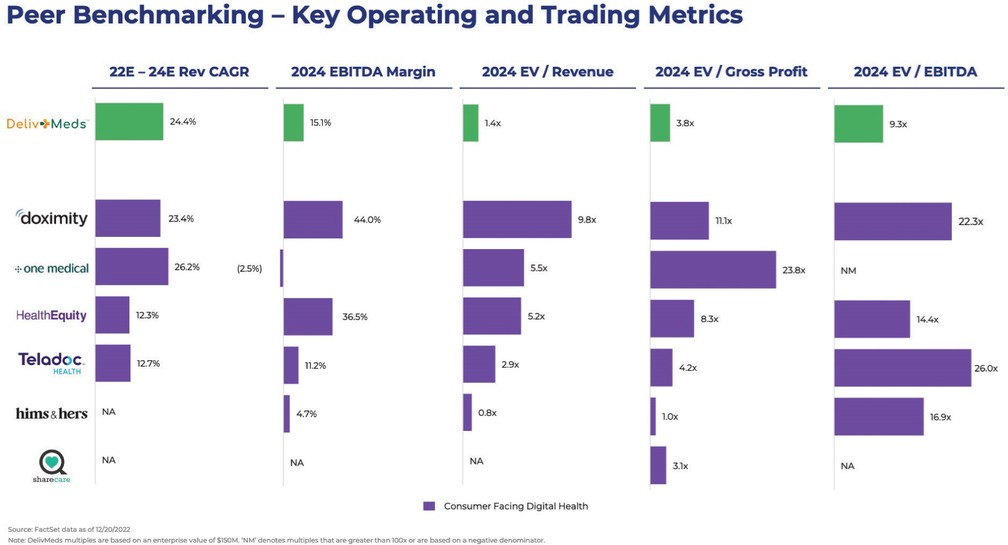
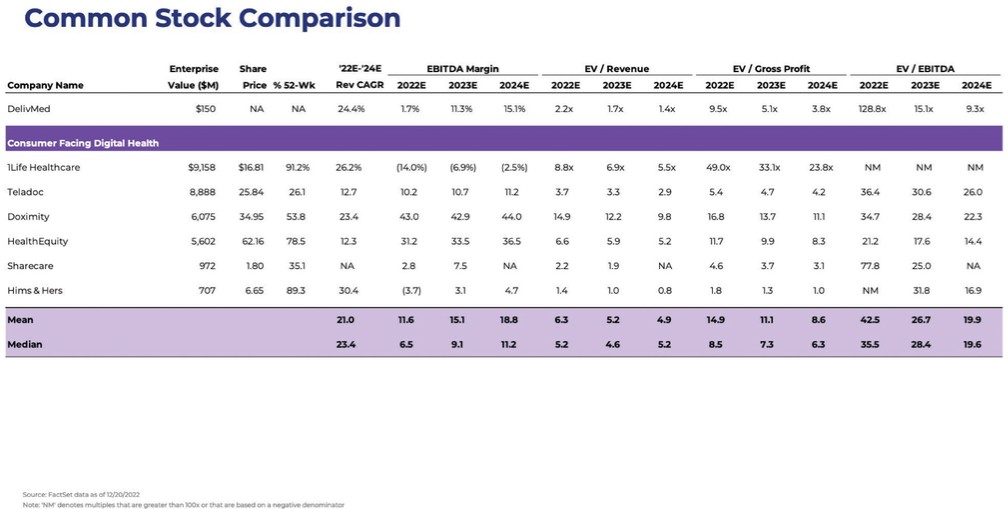
chemical” (or by any similar term) under any Environmental Law, or any other material regulated, under any Environmental Law, including petroleum and its by-products, asbestos, polychlorinated biphenyls, radon, mold, and urea formaldehyde insulation.
“Indebtedness” of any Person means, without duplication, (a) all indebtedness of such Person for borrowed money (including the outstanding principal and accrued but unpaid interest), (b) all obligations for the deferred purchase price of property or services (other than trade payables incurred in the ordinary course of business), (c) any other indebtedness of such Person that is evidenced by a note, bond, debenture, credit agreement or similar instrument, (d) all obligations of such Person under leases that should be classified as capital leases in accordance with GAAP, other than real estate leases, (e) all obligations of such Person for the reimbursement of any obligor on any line or letter of credit, banker’s acceptance, guarantee or similar credit transaction, in each case, that has been drawn or claimed against, (f) all obligations of such Person in respect of acceptances issued or created, (g) all interest rate and currency swaps, caps, collars and similar agreements or hedging devices under which payments are obligated to be made by such Person, whether periodically or upon the happening of a contingency, (h) all obligations secured by an Lien on any property of such Person, (i) any premiums, prepayment fees or other penalties, fees, costs or expenses associated with payment of any Indebtedness of such Person and (j) all obligation described in clauses (a) through (i) above of any other Person which is directly or indirectly guaranteed by such Person or which such Person has agreed (contingently or otherwise) to purchase or otherwise acquire or in respect of which it has otherwise assured a creditor against loss.
“Intellectual Property” means all of the following as they exist in any jurisdiction throughout the world: Patents, Trademarks, Copyrights, Trade Secrets, Internet Assets, Software and other intellectual property, and all licenses, sublicenses and other agreements or permissions related to the preceding property.
“Internet Assets” means any and all domain name registrations, web sites and web addresses and related rights, items and documentation related thereto, and applications for registration therefor.
“IPO” means the initial public offering of Purchaser Units pursuant to the IPO Prospectus.
“IPO Prospectus” means the final prospectus of the Purchaser, dated as of September 29, 2021, and filed with the SEC on October 1, 2021 (File No. 333-253092).
“IPO Underwriters” means Barclays and BMO Capital Markets.
“IRS” means the U.S. Internal Revenue Service (or any successor Governmental Authority).
“Knowledge” means, with respect to (i) the Company, the actual knowledge of the executive officers or directors of any Target Company, after reasonable inquiry or (ii) any other Party, (A) if an entity, the actual knowledge of its directors and executive officers, after reasonable inquiry, or (B) if a natural person, the actual knowledge of such Party after reasonable inquiry.
“Law” means any federal, state, local, municipal, foreign or other law, statute, legislation, principle of common law, ordinance, code, edict, decree, proclamation, treaty, convention, rule, regulation, directive, requirement, writ, injunction, settlement, Order or Consent that is or has been issued, enacted, adopted, passed, approved, promulgated, made, implemented or otherwise put into effect by or under the authority of any Governmental Authority.
“Liabilities” means any and all liabilities, Indebtedness, or obligations of any nature (whether absolute, accrued, contingent or otherwise, whether known or unknown, whether direct or indirect, whether matured or unmatured, whether due or to become due and whether or not required to be recorded or reflected on a balance sheet under GAAP or other applicable accounting standards), including Tax liabilities due or to become due.
“Lien” means any mortgage, pledge, security interest, attachment, right of first refusal, option, proxy, voting trust, encumbrance, lien or charge of any kind (including any conditional sale or other title retention agreement or lease in the nature thereof), restriction (whether on voting, sale, transfer, disposition or otherwise), any subordination arrangement in favor of another Person, or any filing or agreement to file a financing statement as debtor under the Uniform Commercial Code or any similar Law.
“Material Adverse Effect” means, with respect to any specified Person, any fact, event, occurrence, change or effect that has had, or would reasonably be expected to have, individually or in the aggregate, a material adverse effect upon (a) the business, assets, Liabilities, results of operations, prospects or condition (financial or otherwise) of such Person and its Subsidiaries, taken as a