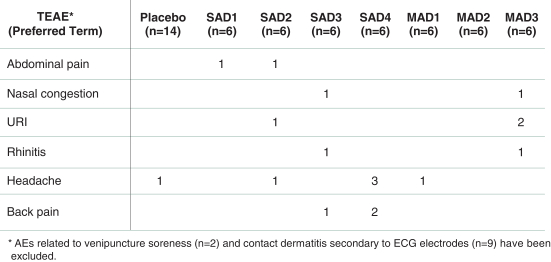
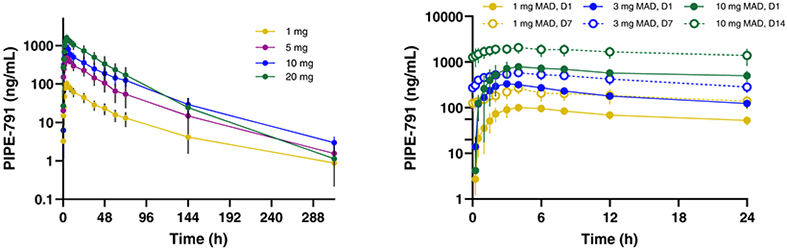
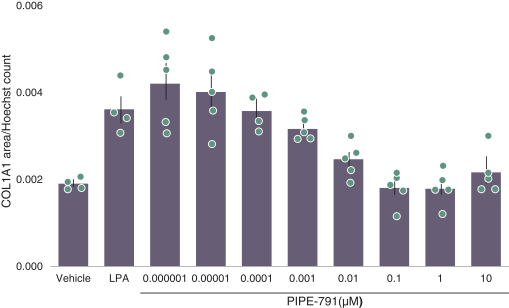
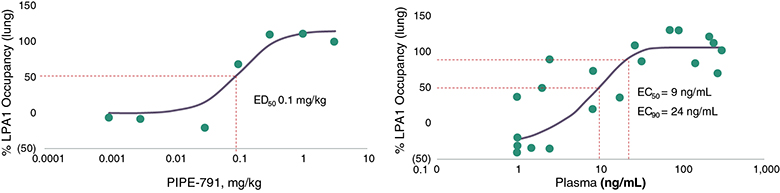
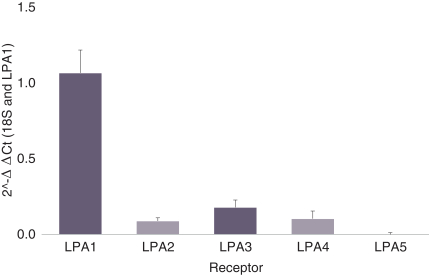
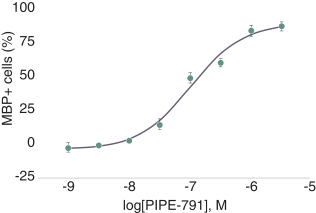
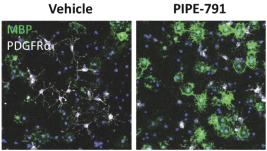
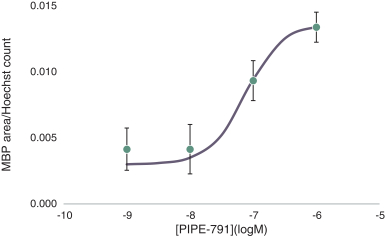
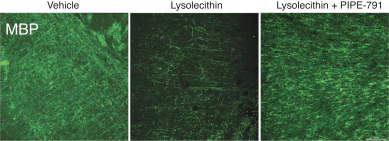
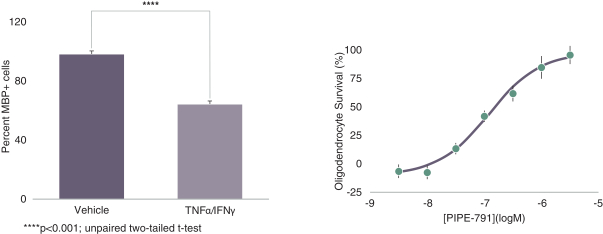
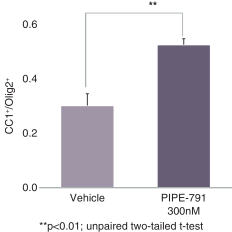
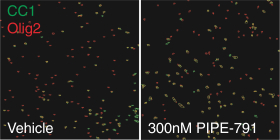
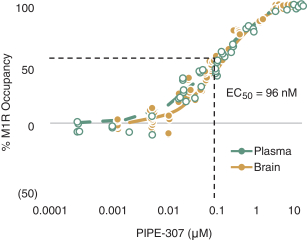
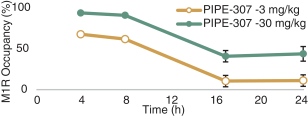
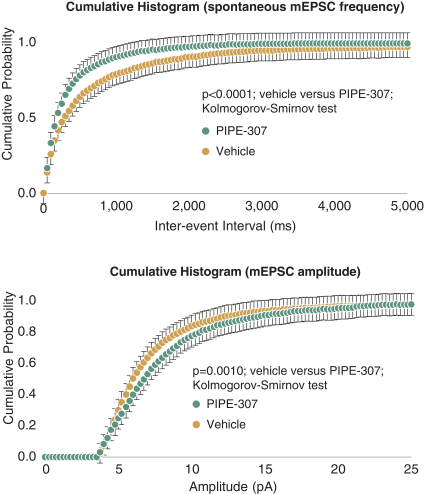
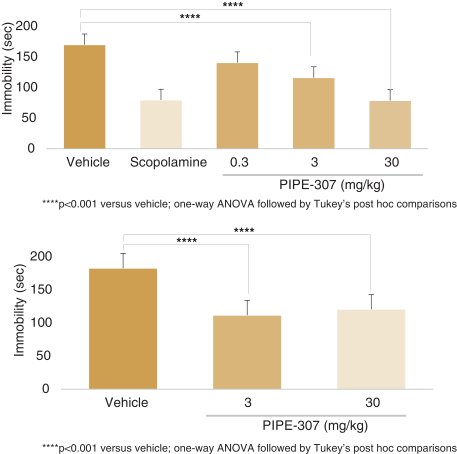
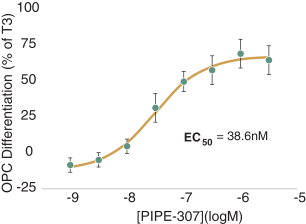
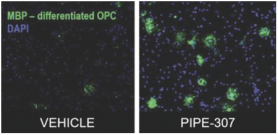
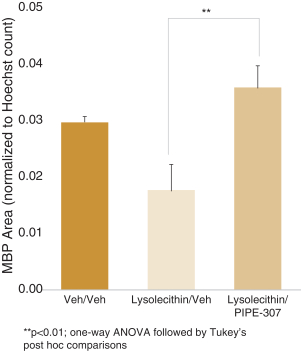
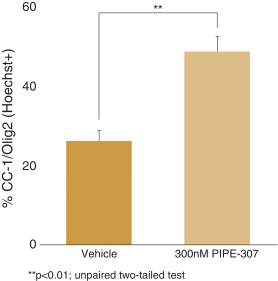
carcinogenic compounds, mutagenic compounds, and compounds that have a toxic effect on reproduction, laboratory procedures and exposure to blood-borne pathogens. If we fail to comply with such laws and regulations, we could be subject to fines or other sanctions.
As with other companies engaged in activities similar to ours, we face a risk of environmental liability inherent in our current and historical activities, including liability relating to releases of or exposure to hazardous or biological materials. Environmental, health and safety laws and regulations are becoming more stringent. We may be required to incur substantial expenses in connection with future environmental compliance or remediation activities, in which case, the production efforts of our third-party manufacturers or our development efforts may be interrupted or delayed.
We are subject to certain U.S. and foreign anti-corruption, anti-money laundering, export control, sanctions, and other trade laws and regulations. We can face serious consequences for violations.
U.S. and foreign anti-corruption, anti-money laundering, export control, sanctions, and other trade laws and regulations, which we collectively refer to as Trade Laws, prohibit, among other things, companies and their employees, agents, clinical research organizations, legal counsel, accountants, consultants, contractors, and other partners from authorizing, promising, offering, providing, soliciting, or receiving directly or indirectly, corrupt or improper payments or anything else of value to or from recipients in the public or private sector. Exports of our products are further subject to export controls and sanctions laws and regulations imposed by the U.S. government and administered by the U.S. Departments of State, Commerce, and Treasury. U.S. export control laws may require a license or other authorization to export products to certain destinations and end users. In addition, U.S. economic sanctions laws include restrictions or prohibitions on engaging in any transactions or dealings, including receiving investment or financing from, or engaging in the sale or supply of products and services to, U.S. sanctioned countries, governments, persons and entities.
Violations of Trade Laws can result in substantial criminal fines and civil penalties, imprisonment, the loss of trade privileges, debarment, tax reassessments, breach of contract and fraud litigation, reputational harm, and other consequences. We have direct or indirect interactions with officials and employees of government agencies or government-affiliated hospitals, universities, and other organizations. We also expect our non-U.S. activities to increase over time. We expect to rely on third parties for research, preclinical studies, and clinical trials and/or to obtain necessary permits, licenses, patent registrations, and other marketing approvals. We can be held liable for the corrupt or other illegal activities of our personnel, agents, or partners, even if we do not explicitly authorize or have prior knowledge of such activities. Any changes in Trade Laws could result in a decreased ability to export or sell our solutions to, existing or potential customers with international operations. Future changes in Trade Laws and enforcement could also result in increased compliance requirements and related costs which could materially adversely affect our business, results of operations, financial condition and/or cash flows.
Risks Related to our Employees, Managing our Growth and our Operations
Our future success depends on our ability to retain our key personnel and to attract, retain and motivate qualified personnel.
We are highly dependent on Carmine Stengone, our President and Chief Executive Officer, Daniel Lorrain, Ph.D., our Chief Scientific Officer, Stephen Huhn, M.D., our Chief Medical Officer and Senior Vice President of Clinical Development, Peter Slover, our Chief Financial Officer, as well as the other principal members of our management, scientific, and clinical teams. Although we have employment agreements, offer letters or consulting agreements with our executive officers, these
60