Exhibit 99.3












Company Overview
We are an Israeli public corporation with shares dual listed for trading on the Tel Aviv Stock Exchange under the symbol “INCR” and on Toronto Stock Exchange under the symbol “INCR:U”. We are in the last stages of listing our ordinary shares on the NASDAQ exchange under the symbol “INCR”.
We have two main direct subsidiaries, Canndoc Ltd. (“Canndoc”) and Cannolam Ltd. (“Cannolam”). We currently own all of the issued and outstanding shares of Canndoc, 50.1% of the issued and outstanding shares of Cannolam and other holdings in additional pharmacies and trade houses. Unless otherwise specified, references in this section to “we”, “our” and “us” refer to the business of Intercure and its subsidiaries.
We are a pioneer in the production (including the breeding, cultivating, and processing), manufacturing and distribution of pharmaceutical-grade cannabis and cannabis-based products for medical use. For more than 13 years, we have been a leader in the licensed production and distribution of cannabis and cannabis-based products throughout Israel, one of the first countries with a governmentally sanctioned regime for the production, manufacturing, and distribution of cannabis for medical use. Our goal is to be a global leader in the production and distribution of high-quality pharmaceutical-grade cannabis and cannabis-based products to patients in all territories that permit and regulate the distribution of cannabis for medical use, including Israel, the European Union and Canada.
Since the beginning of 2020, we have focused on accelerating and growing our commercial activity in major markets around the world. As part of this strategy, we have entered into exclusive collaborations with the largest international cannabis companies in the world including Tilray, Organigram, Aphria, Fotmer and Charlotte’s Web. These strategic agreements serve to advance our capabilities and emphasize our focus on delivering premium quality and branding to Israel and other target markets. We have expanded cooperation agreements for the production, marketing and distribution of our products in countries with supportive regulations such as Germany, the United Kingdom, Canada and more, all of which are pending the permanent approval of commercial cannabis exports from Israel.
Through Cannolam, we operate the private chain Givol™ which is the first and leading chain of pharmacies focused on medical cannabis in Israel. The chain currently includes thirteen pharmacies across Israel. In addition, the chain operates a nationwide ordering and delivery system that serves the entire medical cannabis patient community in Israel. Eight of the pharmacies holds permits and licenses for the distribution of medical cannabis and we are in the process of obtaining those licenses.
Additionally, during the second quarter of 2021, we completed the purchase of a licensed leading operating trading house which will expand our sales channel, distribution, delivery, and storage capacity. The trading house is authorized to distribute GMP medical cannabis products to pharmacies.
Our production system, assuming that the facilities operate at their maximum capacity, and all regulatory approvals are received, allows for a maximum production capacity of over 100 tons of high-quality medical cannabis. This system enables us to be flexible and efficient, and to meet the standards required to execute commercial exports from Israel and to serve growing demand in Israel and around the world.
In 2019, we invested significant resources to upgrade and expand our production systems and establish a global network of advanced production facilities that meet the quality requirements and strict standards across target markets. In December 2020, we were granted a permit by the Israeli Ministry of Health (the “MOH”), as part of a cannabis-export pilot program,1 for the commercial export of its products to Tilray as part of a strategic partnership between the companies. The export permit was obtained after the Company secured an import permit from the Portuguese authorities, demonstrating its products complied with the requirements of European regulation in Portugal and the EU-GMP standard. The export request is a continuation of the developments that have taken place in Israel in recent months and the Company’s preparations for exporting its products.2
1 Note: during the fourth quarter of 2020, the Israeli government, as part of a pilot project to issue export permits for licensed producers, granted us a temporary export permit. The pilot program (as well as our temporary export permit) was set to expire on December 31, 2020, but was subsequently extended.
2 Note: We received an export license in the fourth quarter of 2020, which was subsequently extended.
The new Minister of Economy also enacted an ease on exporting GMP grade medical cannabis and CBD products from Israel. Combined with solid international demand for InterCure’s GMP branded products, the new regulations are expected to boost the Company’s global expansion. InterCure’s leadership and brand equity in international target markets, including Europe, position the Company for long term growth in these emerging markets.
We believe in the uncompromising quality of our products and we are leading the trend towards the pharmaceutical standard in the medical cannabis industry, both through a high quality, advanced production system and through extensive research and development with nine clinical studies approved by the MOH and one active phase 3 clinical trial. We have acquired a unique knowledge throughout our 13 years of experience operating in the cultivation, growth and genetics of cannabis strains. Combined with our analyses of patient use and experience data, we are uniquely positioned to enter into research collaboration agreements with leading organizations and companies. In addition, we have invested in a production system that adheres to the strictest regulatory and quality standards. In doing so, we achieve the highest standard of product quality for our patients and for commercial research collaborations. We believe this will enable us to enter into future partnerships and agreements with pharmaceutical companies.
On April 5, 2021, we signed a non-binding letter of intent to acquire 100% of the Israeli activity of Cann Pharmaceutical Ltd. (“Cann”) which holds the rights for cultivation and marketing medical cannabis products under the brand “Better”, one of our competitors and a pioneer in the Israeli medical cannabis market, with operations in Australia and several Asian countries. The transaction is subject to satisfaction of due diligence and signing of a definitive agreement. Pursuant to the letter of intent, we will allocate Cann shareholders a number of our ordinary shares with a value of $35 million. The allocated shares will be subject to a contractual lock-up for a period of three years, with one-third of the shares being released from lock-up every year (with a moderate monthly exercise mechanism).
We, through our wholly-owned subsidiary, Canndoc, and our 50.1% interest in Cannolam, operate primarily in the cannabis sector (“Cannabis Sector”). In addition, we, as a result of our operations prior to our acquisition of Canndoc, have financial assets in the biomed sector that were made for investments purposes and do not represent a material focus of our current business (“Biomed Sector”). Our only reporting segment as of the date of this MD&A is the medical cannabis sector which generates 100% of our revenue.
NASDAQ Listing
On July 14, 2021, we published our 20-F registration statement and F-1 registration statement and amendment for those statements on August 3rd 2021 after implementing the U.S. Securities Exchange Commission (the “SEC”) comments.
The Company believes that the NASDAQ listing will enable the Company to broaden its global exposure through the world’s largest capital market, enhancing shareholder value. The Company believes that the listing will provide current investors with additional liquidity and prospective investors with better access to one of the leading and most profitable international cannabis companies outside of North America.
Qualified Transaction
On February 9, 2021, we entered into an amended and restated definitive agreement (hereinafter: the “Arrangement Agreement”) with Subversive Acquisition LP (formerly Subversive Acquisition REIT LP), a limited partnership established under the Limited Partnerships Act (Ontario) and a special purpose acquisition company (SPAC) (“Subversive LP”). As a SPAC, Subversive had limited operational activity.
Pursuant to the Arrangement Agreement, on April 23, 2021 our subsidiary acquired all of the outstanding limited partnership units of Subversive LP, in exchange for our ordinary shares by way of a plan of arrangement (the “SPAC Transaction”). At the closing of the SPAC Transaction, which occurred on April 23, 2021, the Company issued 15,650,280 ordinary shares to Subversive LP unit holders, including those that participated in the concurrent private placement. 5,243,616 of our ordinary shares were allocated as part of the SPAC Transaction and are subject to forfeiture unless the Company’s ordinary shares are listed on NASDAQ and obtain a target weighted average price per share of $13.00 (subject to appropriate adjustments) for any five (5) consecutive trading days during the thirty (30) trading days after the shares are traded on NASDAQ. Total net funds raised from the SPAC Transaction, after redemptions, and the private placement equaled USD $56 million (excluding transaction related expenses).
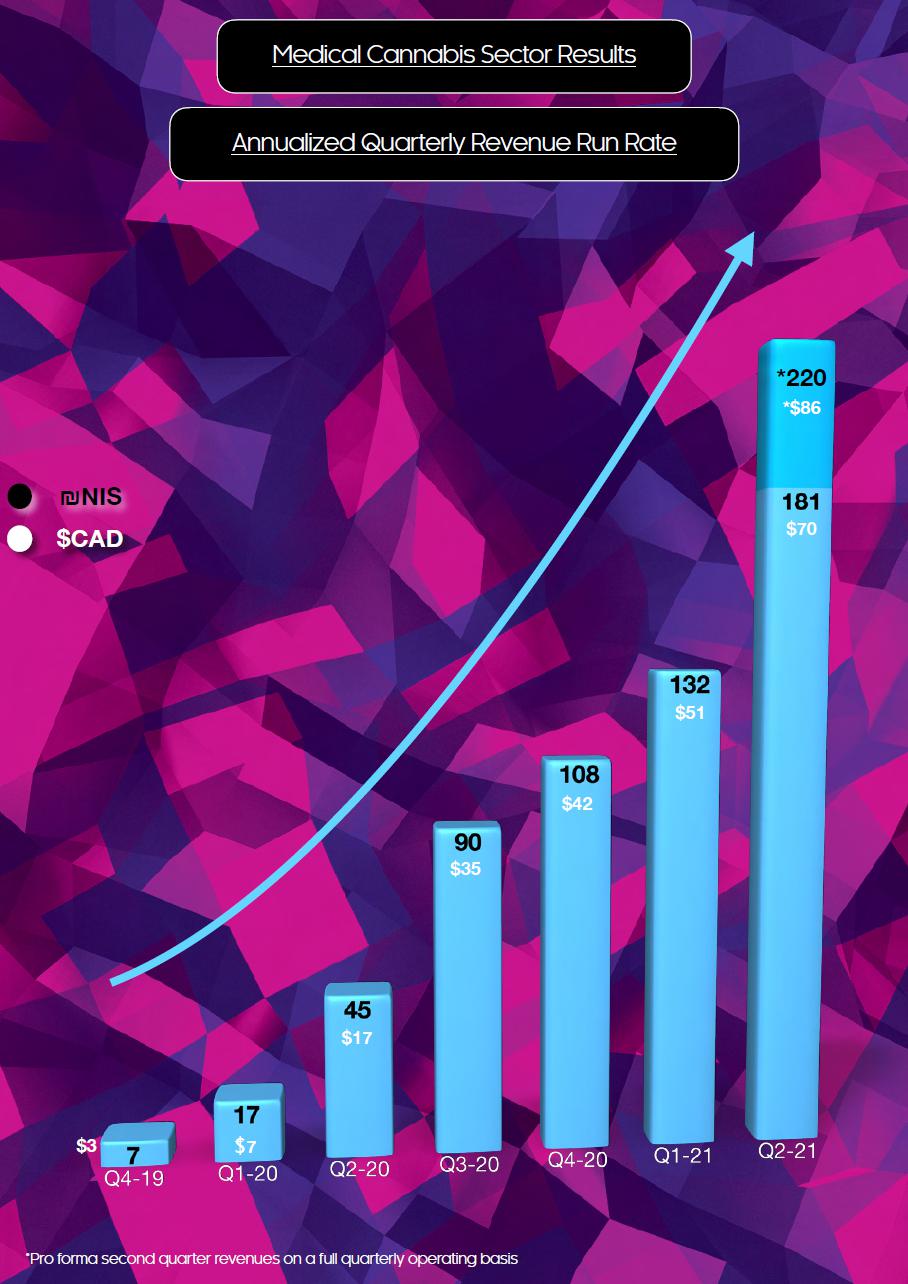
Key Q2 2021 Financial Highlights – Cannabis Sector
| Q2-20 | Q2-21 | Change (%) | ||||||||||
| Revenues | 11,185 | 45,230 | 304 | % | ||||||||
| Gross Profit (1) | 4,814 | 19,268 | 300 | % | ||||||||
| % Gross Profit | 43 | % | 43 | % | — | |||||||
| Operating Profit | (847 | ) | 11,127 | 1414 | % | |||||||
| Adjusted EBITDA (2) | 1,582 | 11,701 | 640 | % | ||||||||
| Net Cash from Operating Activities (consolidated) | (4,007 | ) | 2,005 | — | ||||||||
| (1) | Gross profit before effect of fair value. |
| (2) | EBITDA adjusted for changes in the fair value of inventory, share-based payment expense, impairment losses (and gains) on financial assets, non-controlling interest and other expenses (or income). This is a non-IFRS financial measure and does not have a standardized meaning prescribed by IFRS. See “Non-IFRS Measure”. |
| ● | Achieved record revenue of NIS 45 million (CAD$17 million), four times greater than the second quarter of 2020 and up 37% compared to the first quarter of 2021. | |
| ● | EBITDA for the second quarter of the Company’s cannabis sector was NIS 12 million (CAD$5 million), and NIS 11 million (CAD$4 million) on a consolidated basis. This represents an annual run rate of close to NIS 50 million (CAD$20 million), a significant increase year over year, driven by revenue growth, improvement in gross profit and operating profit. | |
| ● | Reported positive cash flow from operations for the fourth consecutive quarter. | |
| ● | Strong balance sheet with NIS 201 million (CAD$78 million) cash on June 30, 2021. |
Review of the Company’s Operations
| a. | Expansion of the Medical Cannabis Dispensing Operation |
We, through Cannolam, operate the private chain Givol™ which is the first and leading chain of pharmacies focused on medical cannabis in Israel. The chain currently includes thirteen pharmacies across Israel, eight of which are licensed to dispense medical cannabis. In addition, the chain operates a nationwide ordering and delivery system that serves the entire medical cannabis patient community in Israel.
During the reported period, we, through Cannolam, purchased three pharmacies (part of the 13 pharmacies mentioned above) located in major cities across Israel. We purchased 100% of two pharmacies and 51.1% of the third pharmacy. The operations of the three pharmacies were consolidated in our Q2 financials (based on the date of the purchase). After the date of the balance sheet (July 1st and up to date of this MD&A) we entered into transactions to purchase additional 3 pharmacies and active trading house, which those activities would be consolidated in our Q3 financials. The purchase price for each pharmacy was immaterial to the Company.
In addition, during the period and up to the date of publication of this MD&A, we purchased 100% of one of the leading operating trading houses in Israel, which is authorized to distribute GMP medical cannabis products to pharmacies. The purchase of the trading house will support our vertically integrated model and be an addition to our existing distribution channels. The operation of the trading house was partially consolidated in Q2 financials (based on the date of the purchase). The purchase price of the trading house was immaterial to the Company.
| b. | Exclusive Partnerships with Global Leaders |
We have entered into the following partnerships, all of which provides us with exclusive relationships to distribute the noted products within certain geographical areas:

Cookies is one of the most well-respected and top-selling cannabis brands in California and throughout the world. The company and its product are recognized globally and offer a collection of over 150 proprietary cannabis varieties and product lines.
Cannolam entered into an exclusive license agreement with Cookies in 2019 by which Cannolam will have the exclusive rights to use the Cookies brand in Israel. Cannolam opened a Cookies branded pharmacy in Jerusalem and is expected to open an additional branded pharmacy in Be’er Sheva during the second quarter of 2021
In April 2021, we expanded our partnership with Cookies by entering into a letter of intent to expand the Cookies brand into Europe. According to the letter of intent, we will establish joint ventures in European countries that will focus on cultivating, manufacturing, and distributing Cookies branded products. In addition, we will cultivate Cookies branded products at our southern facility in Israel which we also plan will supply Cookies products to Cookies stores throughout Europe.

Tilray Inc. (NASDAQ: TLRY) (“Tilray”) is a global pioneer in the research, cultivation, production, and distribution of cannabis and cannabinoids, currently serving patients and consumers in 16 countries spanning five continents.
In December 2019, we established a strategic collaboration with Tilray for the purpose of providing us with access to existing and potential markets in Tilray’s operating territories. The collaboration between us and Tilray consists of a set of agreements with Tilray Portugal Unipessoal Ltd., a wholly-owned subsidiary of Tilray, pursuant to which, Tilray will import GMP-quality medical cannabis products from us (the “Tilray Agreements”). Tilray’s facility in Portugal has an annual maximum production capacity of 25 metric tons of cannabis.3
As of the date of the MD&A, we agreed with Tilray that we are entitled to additional shipments of cannabis products, subject to both parties obtaining the required permits. In this regard, in July 2021 we successfully completed the import of additional shipment to Israel and the product sales for that shipment commenced in August 2021. Together with Tilray, we are exploring several more potential shipments.
3 Source: ttps://mjbizdaily.com/canadas-tilray-build-marijuana-production-arm-portugal-serve-eu/
The Tilray Agreements provide us with a seven-and-a-half year exclusivity period over all of the final Tilray-branded products sold in Israel.
Tilray’s Cantanhede site in Portugal


Organigram, Inc. (NASDAQ: OGI) (TSX: OGI) (“Organigram”), is a leading licensed producer of cannabis.
In June 2020, we entered into a contractual relationship with Organigram for the purpose of collaborating to develop, import and export medical cannabis products in the state of Israel and across Europe (the “Organigram Agreement”). Organigram’s facility located in New Brunswick has a potential annual capacity of 70 tons.
The Organigram Agreement specifies that, subject to obtaining the required permits, we will import from Organigram 3,000 kilograms of medical cannabis products from Organigram’s advanced indoor facility in Canada (“Indoor Products”) within a period of 18 months (the “Organigram Initial Period”). In accordance with the Organigram Agreement, we will produce and market the medical cannabis products imported from Organigram in pharmacies throughout Israel and Europe. We will be provided with the option to import from Organigram an additional 3,000 kilograms per year of medical cannabis products for a period of two years from the end of the Organigram Initial Period, under the same terms and conditions as those in place during the Organigram Initial Period. These products will be marketed under our “Canndoc Indoor” brand and we, and Organigram, will examine the possibility of selling these products under a joint brand, in compliance with and subject to the Israeli Medical Cannabis agency’s (“IMCA”) instructions. We will then manufacture and transform the imported product into Canndoc’s GMP-branded product. Final products will be distributed by Canndoc’s distribution channels to all pharmacies in Israel. In August 2020, we successfully imported our first shipment of the noted products from Organigram into Israel and successfully launched the “Canndoc Indoor” family of products.
The Organigram Agreement provides us with an aggregate of up to a seven-and-a-half year exclusivity period (in addition to certain other rights and subject to certain conditions) over all of the final Organigram-branded products sold in Israel.
Organigram’s Indoor site (Moncton Campus) in Canada


Aphria Inc. (NASDAQ: APHA) (TSX: APHA) (“Aphria”) is one of the largest leading worldwide cannabis production companies, with its “Diamond Facility” in Leamington, Ontario being one of the biggest and most advanced cannabis facilities in the world, and having an annual production capacity of 140 metric tons.
In August 2020, we entered into an agreement with Aphria (the “Aphria Agreement”) for the import of bulk cannabis products from Aphria’s facility in Canada into Israel. Pursuant to the Aphria Agreement, we will purchase from Aphria’s production facility in Canada, and import into Israel, up to 3,000 kilograms of “bulk” quality medical cannabis for a period of two years (“Aphria Initial Period”). We have the option to import up to 6,000 kilograms of additional product from Aphria for two additional periods of two years each. This option begins at the time on expiry of the Aphria Initial Period and under the same terms and conditions as during the Aphria Initial Period. We will then manufacture and transform the imported product from into Canndoc’s GMP-branded product. Final products will be distributed by Canndoc’s distribution channels to all pharmacies in Israel. In November 2020, we successfully imported our first shipment of the noted products from Aphria into Israel and successfully launched the “Canndoc Stars” family of products.
The Aphria Agreement provides us with an aggregate of up to a seven-and-a-half year exclusivity period (in addition to certain other rights and subject to certain conditions) over all of the final Aphria-branded products sold in Israel.
Aphria’s Diamond Site in Canada


Charlotte’s Web Inc. (TSX: CWEB) (OTCQX: CWBHF) (“Charlotte’s Web”) is the owner of one of the largest worldwide CBD brands.
In December 2020, we entered into a collaboration with Charlotte’s Web, under which we will be the sole partner of Charlotte’s Web in Israel, and through which its products will be marketed in Israel under a joint brand for the Israeli market, subject to certain conditions, including certain regulatory matters within central European countries and England (the “Charlotte’s Web Agreement”). The arrangement is subject to the receipt of the required regulatory agreements.
We will be responsible for obtaining the regulatory approvals required in order to register the purchased products and their importation and will take appropriate marketing and sales actions. Together with Charlotte’s Web, we will explore opportunities for clinical trials, product development and Israeli product manufacturing.
The Charlotte’s Web Agreement is for a period of five years (with a one year extension option) from the date that CBD is removed from the Israeli Dangerous Drug Ordinance [New Version], 5733-1973 (which has yet to occur).

Fotmer Corporation S.A. (“Fotmer”) is a corporation established in Uruguay that cultivates and produces medical cannabis at an internationally high level. In December 2020, we entered into an agreement with Fotmer, under which we will import from Fotmer approximately 3,000 kilograms of quality medical cannabis products, each year for a period of four years (the “Fotmer Agreement”).
During August 2021 we completed the first import shipment from Fotmer. We expect to commence sales of the products during Q3 2021. Together with Fotmer, we are working on several more potential shipments.
Subject to the terms set out therein, the Fotmer Agreement provides us with a seven-and-a-half year exclusivity period over all of the final Fotmer-branded products sold in Israel.
| c. | Global Production System |
As of the date of this MD&A, our production capacity, assuming that the facilities operate at their maximum capacity, and all regulatory approvals are received, produces over 100 tons of GMP-certified pharmaceutical-grade cannabis. In addition, through strategic partnerships with leading license producers, we may have access to additional high quality medical cannabis on demand.
Israeli Production Facilities
Through our partnership with Kibbutz Nir-Oz we operate one of the largest medical cannabis production sites in Israel and in the world, covering a total area of 1.7 million square feet, of which 300,000 square feet are operational and produce up to 10,000 kilograms of pharmaceutical-grade cannabis per year. Full operations in the Southern Kibbutz will allow us to produce 88 tons of pharmaceutical-grade cannabis per year. The development of the southern site is carried out in a modular manner in accordance with the regulatory developments concerning the export of medical cannabis from Israel.
Through our partnership with Beit HaEmek Kibbutz, we own and operate our primary production facility, located in northern Israel, utilizing climatized greenhouses. This site currently occupies approximately 55,000 square feet with the capacity to produce up to 3,000 kilograms of pharmaceutical-grade cannabis per year.
Canndoc – new genetics - CANNDOC Cali™ cultivated in Canndoc’s advanced southern facility

Denmark
In May 2020, we entered into an EU-GMP distribution agreement with a Danish partner for the production of up to 11.7 tonnes of cannabis per year for a period of 3 years. As part of this agreement, we will manufacture our products in a facility located in Denmark. This manufacturing facility is approved by the Good Manufacturing Practice of the European Union (“EU-GMP”) standard and has all the licenses and permits required for the cultivation, production, distribution and marketing of cannabis. The manufacturer will be responsible for the entire growth and production process of the products, as well as the logistical process of transporting and packaging the products in accordance with all applicable legal requirements. The partner will be entitled to a portion of the profits generated as a result of the sales made through our distribution channel. This facility is operational and we are currently in the process of obtaining approval for importing products from Denmark to Germany with this partner. As of the date of this MD&A, no sale of products has commenced and this partnership does not impact our financial statements in any way.
Canada
In May 2019, we entered into a partnership with a Canadian company that is in the advanced stages of building an indoor complex for the production and distribution of cannabis products for medical use in Canada. We established a joint venture with the Canadian partner, which pursuant to the joint venture agreement, will entitle us to 51% of the profits generated from the sale of our products. The production and distribution of the products will be done under the “CANNDOC” brand while the marketing of the products will be done by the partner. While this facility is operational for cultivation, it has not yet received all of the licenses and permits required for the sale of products. As of the date of this MD&A, no sale of products has commenced and this partnership does not impact our financial statements in any way.
| d. | Sales and Distribution |
Israel
Under current regulations, patients in Israel fill prescriptions directly from a registered pharmacy. Our products meet all of the IMCA standards and are permitted to be sold within all registered pharmacies across Israel that are otherwise permitted to dispense medical cannabis to patients. We sell our products through pharmaceutical distributors and licensed retail pharmacy locations where patients can fill their prescriptions on-site or have our products delivered directly to their residence. Under the old regulations, the IMCA instituted a fixed price for the monthly supply of cannabis products, regardless of the dosage or form of use. Under the current regulations, the price of cannabis products is not fixed and will be determined primarily by market demand.
We have developed wholesale supply relationships with government and academic research institutions and private businesses throughout Israel and these relationships require minimal selling, administrative and fulfillment costs. We believe there is potential for the wholesale of finished, packaged products to other licensed producers, and we intend to pursue this sales channel as a part of our growth strategy.
SLE
In September 2019, we entered into a distribution agreement with SLE, a subsidiary of Teva Group Pharmaceutical Industries Ltd., a leading Israeli company in the health services field (the “SLE Agreement”).
Pursuant to the SLE Agreement, SLE will provide us with logistics, storage, collection and distribution services for our medical cannabis products throughout Israel for a term of three years, with two optional extensions of two years each. SLE holds an IMC-GDP distribution license and possesses an advanced logistics facility.
Novolog
In December 2020, we entered into a distribution agreement with Novolog, a leading Israeli company in the logistic health services field.
Pursuant to the noted agreement, Novolog will provide us with logistics, storage, collection and distribution services for our medical cannabis products throughout Israel for a term of three years, with two optional extensions of two years each. Novolog holds an IMC-GDP distribution license and possesses an advanced logistics facility.
Super-Pharm
In March 2020, we entered into a binding preliminary distribution agreement with Super-Pharm Ltd. (“Super Pharm”), the largest chain of pharmacies in Israel (which operates approximately 260 pharmacies) (the “Super Pharm Agreement”). Super Pharm currently operates 60 pharmacies that sell cannabis for medical purposes (the “Super Pharm Pharmacies”). Pursuant to the Super Pharm Agreement, Super Pharm agreed to purchase from us, and we agreed to sell to Super Pharm, 10,000 kilograms of our medical cannabis products for a period of 3 years. The Super Pharm Agreement requires our products to be in compliance with the Israel Medical Cannabis-Good Manufacturing Practice standards.
The parties to the Super Pharm Agreement have covenanted to negotiate in good faith and enter into a detailed agreement within 90 days from the date of the Super Pharm Agreement. The parties, by mutual agreement have agreed to extend the said period to September 30, 2021 and negotiations of the detailed agreement remain ongoing.
Pursuant to the Super Pharm Agreement, Super Pharm will be responsible for distributing the final products to each individual Super Pharm pharmacy, while we will provide professional training and clinical knowledge about our products to Super Pharm and Super Pharm Pharmacies over the term of the agreement.
International
Germany
In June 2019, we entered into a non-exclusive distribution agreement with a licensed distributor in Germany, for the purpose of distributing our pharmaceutical-grade products within Germany (the “German Distribution Agreement”). The German Distribution Agreement contains customary obligations, intellectual property, confidentiality and indemnification provisions. Each party to the German Distribution Agreement is entitled to terminate the German Distribution Agreement in the event of an uncured material breach of the agreement, the insolvency of the other party or a change of control event.
Austria
On April 4, 2021, we entered into a partnership with an Austrian entity to operate together in the developing cannabis markets in Austria and Luxembourg. Pursuant to the agreement, the partnership will replicate the successful model of our subsidiary Canndoc in Israel to establish and manage the distribution, marketing, and sales of the company’s products in selected countries in Europe. The partnership’s planned operations will be vertically integrated and will include both online and retail distribution for our branded products. The Austrian entity has committed to invest €10 million in an Austrian joint venture, which will be equally owned by the parties, with an option for the Austrian entity to increase its shares to 51% of all outstanding shares of the joint venture at any time.
| e. | Research and Development |
We believe that innovation is a key component of our competitiveness and growth in the medium and long-term and is driven by market research and analysis of potential new products and the development of new technologies. We engage in the research of agricultural techniques that utilize climatic advantages and our agrotech capabilities to improve the yield of cannabis plants in their production of various cannabinoids. Our research and development programs have also involved the development of high- quality protocols, elite genetics with improved disease and stress resistance, compound fractional distillation and separation and advanced formulation methods.
Since 2014, we have collaborated with various world-renowned research institutions, such as Technion – Israel Institute of Technology, Volcani Center (the research arm of the Israeli Ministry of Agriculture) and other universities and institutions accredited by the Israeli Council for Higher Education. As a result of these collaborations, we have enhanced our production capabilities, improved and optimized our genetics, and developed additional cannabinoid profiles. Our research and development operations also include collaborations with a renowned governmental institute as well as various research entities, researchers, start-up companies, mature companies and commercial entities holding licenses from the IMCA.
Clinical Trials
Based on our information and experience in providing medical cannabis to patients, we developed a broad and advanced clinical research program based on GMP - quality products approved by the IMCA.
During November 2019, we began clinical research with the Research and Development Foundation of the Shamir Medical Center (Assaf Harofeh) and with a principal investigator on his behalf to examine the effect of medical cannabis products on autism spectrum disorder in children. The study will be conducted at Assaf Harofeh Hospital, is expected to include about 100 participants and will last a period of 24 months.
We received the approval of the IMCA to conduct nine advanced clinical trials based on additional medical cannabis products in the IMC-GMP standard in strategic collaboration with leading medical centers in Israel. As part of the clinical trials, we will serve as the initiator of the clinical trials conducted by the research partners. The program includes clinical trials of the company’s products on a variety of medical indications (epilepsy, fibromyalgia, neuropathic pain, side effects of chemotherapy in cancer patients, Parkinson’s, rheumatoid arthritis, radicular pain, post-trauma) and radiculopathy (PTSD).
In addition, we submitted an application for approval of a clinical study to examine the effect of cannabis use on the dose and / or frequency of opioid use in collaboration with Sheba Hospital.
The studies are phase 2 studies and are performed randomly, double-blind and placebo-controlled (randomized, placebo-controlled, double-blind) as is customary in pharma studies according to FDA requirements, with dozens of subjects participating in each clinical study. It should be noted that due to the coronavirus Covid 19), a delay in studies is expected.
Results of Operations
Financial data is expressed in thousands of NIS. The following table summarizes our historical consolidated statements of comprehensive income for the three and six months ended June 30, 2021 and 2020:
| For the 3-month period ended on June 30 | For the 6-month period ended on June 30 | |||||||||||||||
| 2021 | 2020(1) | 2021 | 2020(1) | |||||||||||||
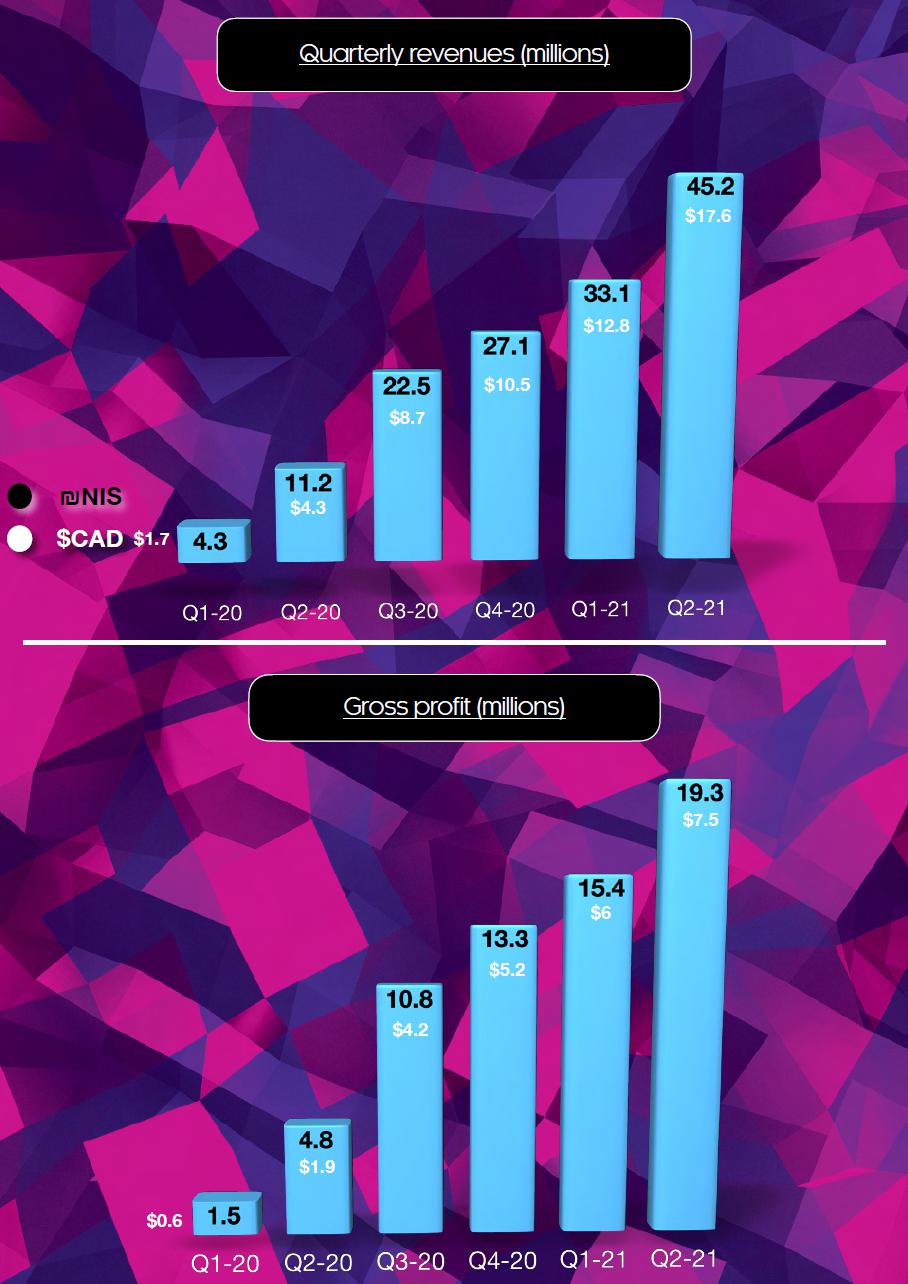
| Revenues | 45,230 | 11,185 | 78,281 | 15,444 | ||||||||||||
| Gross profit before effect of fair value | 19,268 | 4,814 | 34,694 | 6,330 | ||||||||||||
| Gross profit after effect of fair value | 19,948 | 5,495 | 34,709 | 7,104 | ||||||||||||
| Research and development expenses | (356 | ) | (412 | ) | (717 | ) | (786 | ) | ||||||||
| General and administrative expenses | (6,311 | ) | (4,211 | ) | (11,591 | ) | (9,234 | ) | ||||||||
| Marketing and selling expenses | (4,854 | ) | (1,765 | ) | (8,423 | ) | (2,884 | ) | ||||||||
| Impairment losses and (gains) on financial assets through profit or loss | 162 | 46 | 326 | (39,304 | ) | |||||||||||
| Other expenses (income), net | 290 | — | 290 | (3,127 | ) | |||||||||||
| Consolidated operating profit (loss) | 8,879 | (847 | ) | 14,594 | (48,231 | ) | ||||||||||
| Comprehensive income (loss) | 5,938 | (978 | ) | 9,793 | (48,187 | ) | ||||||||||
| Interest / Financing cost | 403 | 131 | 493 | (44 | ) | |||||||||||
| Tax expenses (income) | 2,538 | — | 4,309 | — | ||||||||||||
| Depreciation and amortization | 1,254 | 413 | 2,501 | 803 | ||||||||||||
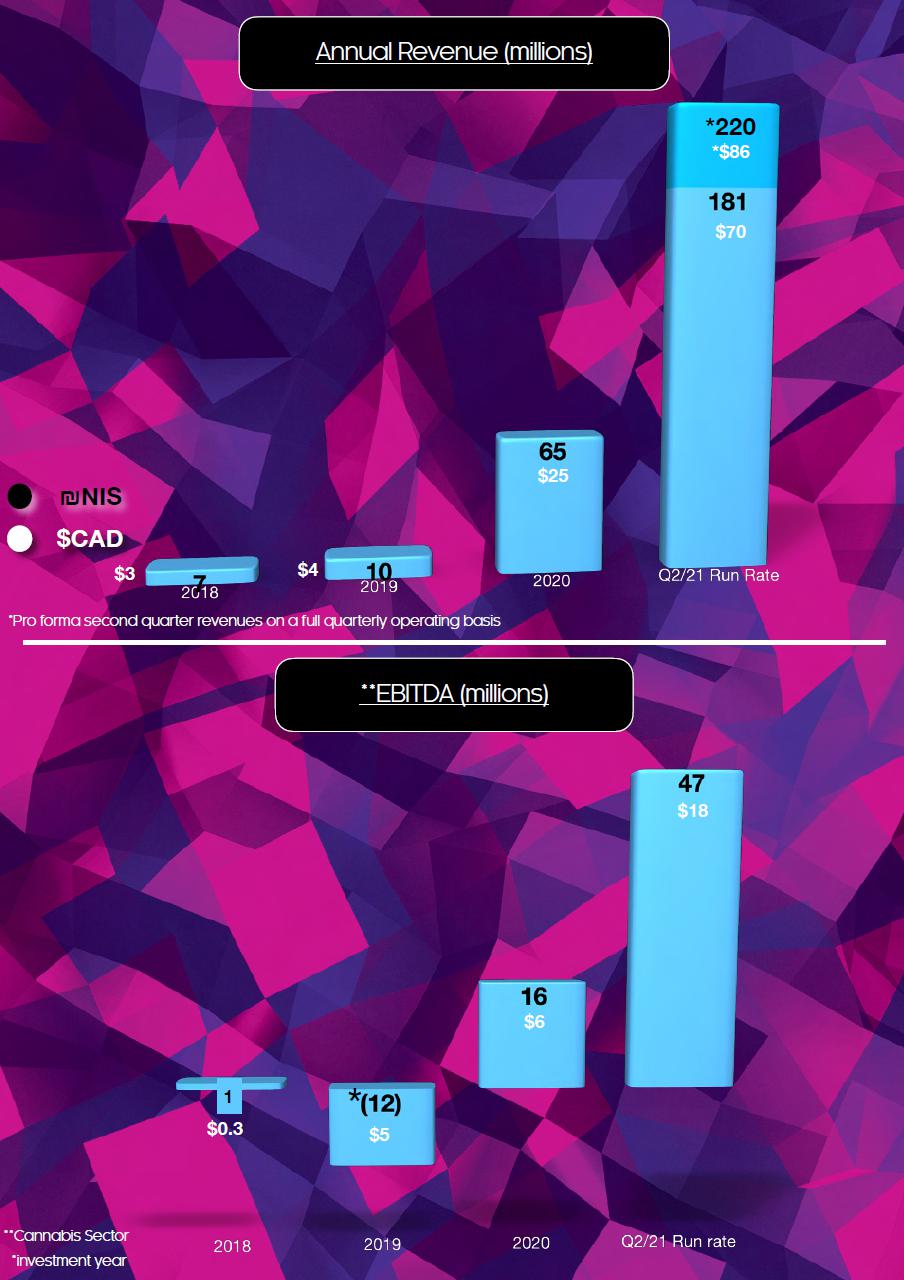
| EBITDA | 10,133 | (434 | ) | 17,095 | (47,428 | ) | ||||||||||
| Share-based payment expenses | 1,814 | 2,743 | 3,818 | 6,009 | ||||||||||||
| Other expenses (income), net | (290 | ) | — | (290 | ) | 3,127 | ||||||||||
| Impairment losses and (gains) on financial assets through profit and loss | (162 | ) | (46 | ) | (326 | ) | 39,304 | |||||||||
| Fair value adjustment to inventory | (680 | ) | (681 | ) | (15 | ) | (774 | ) | ||||||||
| Adjusted EBITDA | 10,815 | 1,582 | 20,282 | 238 | ||||||||||||
| Basic earnings (loss) per share | 0.29 | (0.02 | ) | 0.14 | (2.00 | ) | ||||||||||
| Diluted earnings per share | 0.24 | (0.02 | ) | 0.12 | (2.00 | ) | ||||||||||
| (1) | Cannolam operations consolidated for the first time on July 1st, 2020. |
Revenues – Revenue for the second quarter of 2021 was approximately four times greater compared to the corresponding period last year, and increased in 37% compared to the fourth quarter of 2020. The growth was primarily derived from high demand for the company’s quality product lines, market growth, increase in the company’s market share, implementation of commercial agreements with pharmacies, international collaborations and the consolidation and continues grow of Cannolam’s pharmacy chain and the purchase and partially consolidation of the trading house.
During the period, Canndoc continued its successful launching of premium products under the brand CANNDOC Cali™. The series of GMP products were cultivated and manufactured in Canndoc’s advanced southern facility.
Gross profit after effect of fair value – Gross profit for the second quarter of 2021 increased of 263% to NIS 19.9 million compared to NIS 5.4 million in the corresponding quarter, mainly in light of the ramp up in Canndoc’s advanced southern facility.
Adjusted EBITDA – Significant improvement in comparison to the negative Adjusted EBITDA in the corresponding period and in comparison to the previous quarter. The improvement is mainly due to implementing our vertically integrated strategy.
Total Assets and Liabilities
| As of June 30th | As of March 31st | |||||||||||||||
| 2021 | 2020 | 2021 | 2020 | |||||||||||||
| Total current assets | 286,664 | 45,967 | 82,907 | 33,708 | ||||||||||||
| Total non-current assets | 301,822 | 211,889 | 259,970 | 206,928 | ||||||||||||
| Current Liabilities | 90,665 | 39,894 | 34,936 | 25,087 | ||||||||||||
| Non-current Liabilities | 53,302 | 2,578 | 9,020 | 2,763 | ||||||||||||
Total Current Assets
The increase was primarily due to capital raising of the SPAC Transaction and continuous increase in Intercure’s activity (trade receivables, inventories, and biologic assets).
Total Non-Current assets
The increase was primarily due to the consolidation of our subsidiaries - CannOlam’s pharmacy expansion and the purchase and first time consolidation of the trading house operation. The consolidation of those subsidiaries operations led to increase in the non-current assets and goodwill.
Current Liabilities
The change in compare to Q2 2020 was mainly due to: (a) the consolidation of the activities purchased during the period (additional pharmacies and trading house activities); (b) increase in the Company activity which led to increase in the trade payable, and other payables.
Non-Current Liabilities
The increase was primarily due to bank loans taken by the Company and its subsidiaries during the period in order to fund its capital investment to expend its operations.
Cash Flow
Intercure’s approach to liquidity is to always have sufficient liquidity to meet its liabilities as they come due. This is achieved by continuously monitoring cash flows and reviewing actual operating expenditures and revenue against budget.
| Cash Flow | For six months ended on June 30, 2021(1) | For Six months ended on June 30, 2020(1) | For three months ended on June, 2021(1) | For three months ended on June, 2020(1) | ||||||||||||
| Net cash provided by (used in) operating activities | 9,708 | (7,904 | ) | 2,005 | (4,007 | ) | ||||||||||
| Cash Flow | For six months ended on June 30, 2021(1) | For Six months ended on June 30, 2020(1) | For three months ended on June, 2021(1) | For three months ended on June, 2020(1) | ||||||||||||
| Net cash provided by financing activities | 171,191 | 10,990 | 170,288 | 11,156 | ||||||||||||
| Net cash provided by (used in) investing activities | (18,911 | ) | (22,005 | ) | (13,093 | ) | (15,873 | ) | ||||||||
| Change in cash during the period | 161,988 | (18,919 | ) | 159,200 | (8,724 | ) | ||||||||||
| Exchange differences in respect of cash and cash equivalent balances | 393 | 184 | 354 | 3 | ||||||||||||
| Cash and cash equivalents, beginning of year | 37,888 | 27,338 | 40,715 | 17,324 | ||||||||||||
| Cash and cash equivalents, end of year | 200,269 | 8,603 | 200,269 | 8,603 | ||||||||||||
(1) Cannolam operations consolidated for the first time on July 1st, 2020.
Net cash flow used in operating activities – The increase was primarily due to operating profitability and in continuous improvement and positive cash flow from operating activities.
Net cash provided by financing activities – the significant increase during the period was mainly due to the SPAC Transaction.
Net cash used in investing activities - The main investment for six months ended on June 30, 2021 were mainly were mainly continues investment in the Southern Kibbutz and the additional purchases of pharmacies and trading house.
Use of Proceeds
The Company’s intended use of proceeds from the SPAC Transaction has not changed from the disclosure set forth in the “Capitalization and Use of Proceeds” section of the Prospectus to the date of this MD&A.
Summary of Quarterly Results
The following table below sets out certain consolidated financial data for the Company:
| Q2 2021 | Q1 2021 | Q4 2020 | Q3 2020 | Q2 2020 | Q1 2020 | Q4 2019 | Q3 2019 | |||||||||||||||||||||||||
| Revenue | 45,230 | 33,051 | 27,094 | 22,497 | 11,185 | 4,259 | 1,828 | 2,598 | ||||||||||||||||||||||||
| Gross Profit (Loss) | 19,268 | 15,427 | 13,302 | 10,755 | 4,814 | 1,516 | 442 | (1,063 | ) | |||||||||||||||||||||||
| Adjusted EBITDA | 10,815 | 9,468 | 8,165 | 6,627 | 1,582 | (1,344 | ) | (3,914 | ) | (6,083 | ) | |||||||||||||||||||||
Liquidity and Capital Resources
Intercure has been generating profits and has experienced positive cash flows, which are expected to be the primary sources to fund its future operations. In addition, Intercure has cash reserves as a result of the completion of the noted private placements. Lastly, as a public company, Intercure may access the public and/or private markets to finance any additional needs it may have, including through the issuance of debt or equity securities.
Intercure does not expect to require any additional funding in the future as it projects a positive cash flow from operations.
Summary of Contractual Obligations
As of the date of this report, the company don’t have any material contractual obligations.
Critical Accounting Estimates
The Company’s critical accounting estimates are summarized in note 3 of the Annual Financial Statements and have not changed during the following interim period.
Outstanding Share Data
As of the date of this report (August 16, 2021) Intercure’s current outstanding shares capital can be summarized as follows:
| Type | Shares | Options / Warrants | ||||||
| Intercure Shares | 42,735,052 | * | ||||||
| Options (A) | 2,150,919 | |||||||
| Options (B) | 1,729,612 | |||||||
| ESOP (A) | 1,199,791 | |||||||
| ESOP (B) | 967,208 | |||||||
| Total | 42,735,052 | 6,047,530 | ||||||
(*) includes 5,243,616 shares allocated as part of the SPAC Transaction which are subject to (a) the Company shares listed on NASDAQ and (b) the Company’s shares listed on NASDAQ reaching the Target Price; and are subject to forfeiture as defined and described in the Prospectus.
| (1) | Options (A) with an expiration period of three year for the date of issuance (September 2018) with an exercise price of NIS 1.69 per Intercure Share. |
| (2) | Options (B) were issued to certain investors in July, 2020 and expire in August 2023 with an exercise price of NIS 19.57 per Intercure Share. |
| (3) | ESOP (A) were issued to our directors between September 2018 to January 2020 and expire in ten years from the date of issuance with an exercise price of NIS 15.57 per Intercure Share. |
| (5) | ESOP (B) were issued to certain employees in February 2021 and expire in four years from the date of issuance with an exercise price of NIS 18.37 per Intercure Share. |
Off-Balance Sheet Transactions
The Company has no off-balance sheet arrangements.
Financial Instruments and Other Instruments
We do not have any financial instruments other than normal course accounts receivable and payables associated with our business activities.
Risk and Uncertainties
We are subject to foreign exchange and liquidity risks.
Foreign Exchange Risk. Our reporting and functional currency is the NIS, but some portion of our operational expenses are in U.S. dollars, Canadian dollars and Euros. As a result, we are exposed to some currency fluctuation risks. We may, in the future, decide to enter into currency hedging transactions to decrease the risk of financial exposure from fluctuations in the exchange rate of the currencies mentioned above in relation to the NIS. These measures, however, may not adequately protect us and our operations could be adversely affected if we are unable to effectively hedge against currency fluctuations in the future.
Liquidity risk. We monitor forecasts of our liquidity reserve (comprising cash and cash equivalents available-for-sale financial assets and short-term deposits). We generally carry this out based on our expected cash flows in accordance with practice and limits set by our management. We are in the process of expanding our operations and the expenses associated therewith and we are therefore exposed to liquidity risk.
Subsequent Events
| 1. | On July 6th, 2021, we entered a transaction to purchase (100%) of a pharmacy and trading house located in northern Israel. The purchase was immaterial to the Company. | |
| 2. | On August 5th, 2021, we entered a transaction to purchase (100%) of a pharmacy located in northern Israel. The purchase was immaterial to the Company. | |
| 3. | On August 8, 2021, we entered a binding LOI to purchase (51%) of a pharmacy located in northern Israel. The purchase was immaterial to the Company. |
Additional Information
Additional information relating to the Company is available on SEDAR at www.sedar.com.