We cannot guarantee that future debt or equity financing will be available in sufficient amounts or on terms acceptable to us, if at all. If we are unable to raise additional capital when required or on acceptable terms, we may be required to take one or more of the following actions:
| | • | | significantly delay, scale back, or discontinue our product development and commercialization efforts; |
| | • | | seek collaborators for our hormone therapy drug products and candidates at an earlier stage than otherwise would be desirable or on terms that are less favorable than might otherwise be the case; or |
| | • | | license, potentially on unfavorable terms, our rights to our hormone therapy drug products and candidates that we otherwise would seek to develop or commercialize ourselves. |
The Credit Agreement does, and any agreements governing future debt financing, if available, may, include covenants limiting or restricting our ability to take specific actions, such as incurring additional debt, making capital expenditures, or declaring dividends. To the extent that we raise additional capital through the sale of equity or convertible debt securities, the ownership interest of our existing stockholders will be diluted, and the terms of these new securities may include liquidation or other preferences that adversely affect the rights of our existing stockholders. If we raise additional funds through collaborations, strategic alliances, or licensing arrangements with third parties, we may have to relinquish valuable rights to our technologies, future revenue streams, research programs, or proposed products or grant licenses on terms that may not be favorable to us.
If we are unable to raise additional capital in sufficient amounts or on terms acceptable to us, we will be prevented from pursuing discovery, development and commercialization efforts, and our ability to generate revenue and achieve or sustain profitability will be substantially harmed.
We are subject to extensive and costly government regulation.
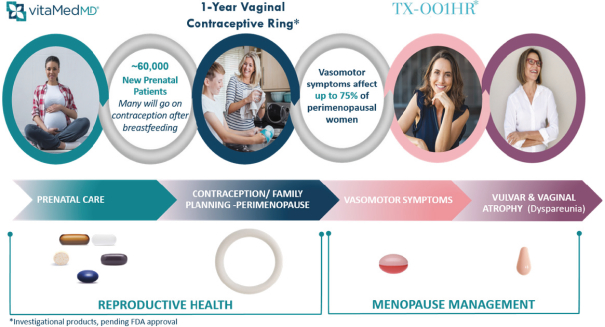
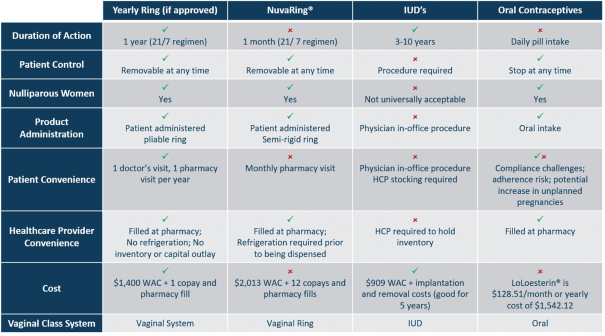
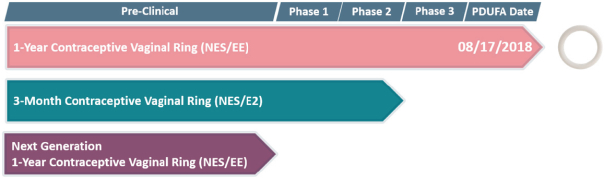
The products we currently market, including IMVEXXYTM and our prenatal vitamins, and the pharmaceutical products we are developing and planning to develop in the future, are subject to extensive and rigorous domestic government regulation, including regulation by the FDA, the Centers for Medicare & Medicaid Services, or CMS, other divisions of the U.S. Department of Health and Human Services, including its Office of Inspector General, the U.S. Department of Justice, the Departments of Defense and Veterans Affairs, to the extent our products are paid for directly or indirectly by those departments, state and local governments, and their respective foreign equivalents. The FDA regulates dietary supplements, cosmetics, and drugs under different regulatory schemes. For example, the FDA regulates the processing, formulation, safety, manufacturing, packaging, labeling, and distribution of dietary supplements and cosmetics under its dietary supplement and cosmetic authority, respectively. The FDA also regulates the research, development,pre-clinical and clinical testing, manufacture, safety, effectiveness, record keeping, reporting, labeling, storage, approval, advertising, promotion, sale, distribution, import, and export of pharmaceutical products under various regulatory provisions. If any drug products we develop are tested or marketed abroad, they will also be subject to extensive regulation by foreign governments, whether or not we have obtained FDA approval for a given product and its uses. Such foreign regulation may be equally or more demanding than corresponding U.S. regulation.
We are also subject to additional health care regulation and enforcement by the federal government and the states in which we conduct our business. Applicable federal and state health care laws and regulations include the following:
| | • | | The federal health care Anti-Kickback Statute, or AKS, prohibits, among other things, persons from knowingly and willfully soliciting, offering, receiving, or providing remuneration, directly or |
S-15