Filed pursuant to Rule 424(b)(3)
Registration No. 333-187466
PROSPECTUS SUPPLEMENT NO. 15
(to Prospectus dated July 21, 2013)

5,369 Shares of Series C 8% Convertible Preferred Stock
(and 2,753,348 Shares of Common Stock Underlying the Series C 8% Convertible Preferred Stock)
Warrants to Purchase up to 2,753,348 Shares of Common Stock
(and 2,753,348 Shares of Common Stock Issuable From Time to Time Upon Exercise of Warrants)
This prospectus supplement modifies and supplements the prospectus of Oxygen Biotherapeutics, Inc. (the “Company”) dated July 21, 2013 (as supplemented on July 25, 2013, July 31, 2013, August 13, 2013, August 23, 2013, August 26, 2013, September 17, 2013, October 25, 2013, November 8, 2013, November 19, 2013, December 9, 2013, December 17, 2013, December 20, 2013 and December 23, 2013) relating to 5,369 shares of Series C 8% Convertible Preferred Stock (and 2,753,348 shares of common stock issuable upon conversion of the Series C 8% Convertible Preferred Stock) and warrants exercisable for 2,753,348 shares of common stock at an exercise price of $2.60 per share (and 2,753,348 shares of common stock issuable upon exercise of the warrants).
This prospectus supplement should be read in conjunction with, and may not be delivered or utilized without, the prospectus. This prospectus supplement is qualified in its entirety by reference to the prospectus, except to the extent that the information in this prospectus supplement supersedes the information contained in the prospectus.
This prospectus supplement includes the attached Current Report on Form 8-K/A as filed with the Securities and Exchange Commission (the “SEC”) on January 22, 2014.
NEITHER THE SEC NOR ANY STATE SECURITIES COMMISSION HAS APPROVED OR DISAPPROVED THESE SECURITIES OR PASSED UPON THE ADEQUACY OR ACCURACY OF THIS PROSPECTUS SUPPLEMENT. ANY REPRESENTATION TO THE CONTRARY IS A CRIMINAL OFFENSE.
The date of this prospectus supplement is January 22, 2014.
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 8-K/A
(Amendment No. 1)
CURRENT REPORT
Pursuant to Section 13 OR 15(d) of The Securities Exchange Act of 1934
Date of Report (Date of earliest event reported): November 13, 2013
Oxygen Biotherapeutics, Inc.
(Exact name of registrant as specified in its charter)
| Delaware | 001-34600 | 26-2593535 | ||
| (State or other jurisdiction of incorporation) | (Commission File Number) | (IRS Employer Identification No.) |
ONE Copley Parkway, Suite 490
Morrisville, NC 27560
(Address of principal executive offices) (Zip Code)
919-855-2100
(Registrant’s telephone number, including area code)
N/A
(Former name or former address, if changed since last report)
Check the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of the registrant under any of the following provisions:
| o | Written communications pursuant to Rule 425 under the Securities Act (17 CFR 230.425) |
| o | Soliciting material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12) |
| o | Pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b)) |
| o | Pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c)) |
EXPLANATORY NOTE
Oxygen Biotherapeutics, Inc. (the “Company”) is filing this Amendment No. 1 to Form 8-K (this “Amendment”) to amend its Current Report on Form 8-K filed on November 19, 2013 (the “Original Form 8-K”) with the Securities and Exchange Commission (the “SEC”) disclosing, among other things, the Company’s acquisition of certain assets of Phyxius Pharma, Inc. (“Phyxius”). The purpose of this Amendment is to provide the financial information required under parts (a) and (b) of Item 9.01 of Form 8-K and amend and restate Item 9.01 of the Original Form 8-K.
No other changes have been made to the Original Form 8-K. This Amendment speaks as of the original filing date of the Original Form 8-K, does not reflect events that may have occurred subsequent to the original filing date, and does not modify or update in any way disclosures made in the Original Form 8-K.
Item 9.01 Financial Statements and Exhibits.
(a) Financial Statements of Businesses Acquired
The audited historical financial statements of Phyxius as of December 31, 2012 and 2011 and for the year ended December 31, 2012, the period from January 18, 2011 (inception) through December 31, 2011, and the period from January 18, 2011 (inception) through December 31, 2012 are filed as Exhibit 99.2 and incorporated herein by reference.
The unaudited financial statements of Phyxius as of September 30, 2013 and December 31, 2012 and for the nine months ended September 30, 2013 and 2012 and the period from January 18, 2011 (inception) through September 30, 2013 are filed as Exhibit 99.3 and are incorporated herein by reference.
(b) Pro Forma Financial Information
The unaudited pro forma condensed combined balance sheet and statement of operations of the Company as of and for the six months ended October 31, 2013 and the unaudited pro forma condensed combined statement of operations of the Company for the year ended April 30, 2013 are filed as Exhibit 99.4 and are incorporated herein by reference.
(d) Exhibits.
| Exhibit No. | Description | |
| 4.1* | Certificate of Designation of Series E Convertible Preferred Stock. | |
| 10.1* | Employment Agreement with John Kelley dated November 13, 2013. | |
| 10.2* | Second Amended and Restated Employment Agreement with Michael Jebsen dated November 13, 2013. | |
| 23.1 | Consent of WithumSmith+Brown, PC. | |
| 99.1* | Press Release dated November 14, 2013. | |
| 99.2 | Audited Financial Statements of Phyxius Pharma, Inc. as of December 31, 2012 and 2011 and for the year ended December 31, 2012, the period from January 18, 2011 (inception) through December 31, 2011, and the period from January 18, 2011 (inception) through December 31, 2012. | |
| 99.3 | Unaudited Financial Statements of Phyxius Pharma, Inc. as of September 30, 2013 and December 31, 2012 and for the nine months ended September 30, 2013 and 2012 and the period from January 18, 2011 (inception) through September 30, 2013. | |
| 99.4 | Unaudited Pro Forma Condensed Combined Financial Information of Oxygen Biotherapeutics, Inc. as of and for the six months ended October 31, 2013 and the unaudited pro forma condensed combined statement of operations of the Company for the year ended April 30, 2013. |
* Filed with the Original Form 8-K, which was filed with the SEC on November 19, 2013.
2
SIGNATURES
Pursuant to the requirements of the Securities and Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned hereunto duly authorized.
| OXYGEN BIOTHERAPEUTICS, INC. | ||
| Date: January 22, 2014 | By: | /s/ Michael B. Jebsen |
| Michael B. Jebsen | ||
| Chief Financial Officer | ||
3
EXHIBIT INDEX
| Exhibit No. | Description | |
| 4.1* | Certificate of Designation of Series E Convertible Preferred Stock. | |
| 10.1* | Employment Agreement with John Kelley dated November 13, 2013. | |
| 10.2* | Second Amended and Restated Employment Agreement with Michael Jebsen dated November 13, 2013. | |
| 23.1 | Consent of WithumSmith+Brown, PC. | |
| 99.1* | Press Release dated November 14, 2013. | |
| 99.2 | Audited Financial Statements of Phyxius Pharma, Inc. as of December 31, 2012 and 2011 and for the year ended December 31, 2012, the period from January 18, 2011 (inception) through December 31, 2011, and the period from January 18, 2011 (inception) through December 31, 2012. | |
| 99.3 | Unaudited Financial Statements of Phyxius Pharma, Inc. as of September 30, 2013 and December 31, 2012 and for the nine months ended September 30, 2013 and 2012 and the period from January 18, 2011 (inception) through September 30, 2013. | |
| 99.4 | Unaudited Pro Forma Condensed Combined Financial Information of Oxygen Biotherapeutics, Inc. as of and for the six months ended October 31, 2013 and the unaudited pro forma condensed combined statement of operations of the Company for the year ended April 30, 2013. |
* Filed with the Original Form 8-K, which was filed with the SEC on November 19, 2013.
4
Exhibit 23.1
Consent of Independent Registered Public Accounting Firm
We hereby consent to the inclusion and/or incorporation by reference in the Registration Statement of Oxygen Biotherapeutics, Inc. on Form S-8 (No. 333-167175) and related prospectuses as well as the Registration Statements of Oxygen Biotherapeutics, Inc. on Form S-3 (No. 333-165733, No. 333-187441, No. 333-187888 and No. 333-191579) and on Form S-1 (No. 333-187466) of our audit report dated November 13, 2013, with respect to the financial statements of Phyxius Pharma, Inc., which report appears in this Current Report on Form 8-K/A of Oxygen Biotherapeutics, Inc.
/s/ WithumSmith+Brown, PC
New York, New York
January 22, 2014
Exhibit 99.2

1

2

3

4

5
6
7

8

9

10

11
Exhibit 99.3

(A Development Stage Company)
Financial Statements
as of September 30, 2013
1
TABLE OF CONTENTS
| PAGE | ||
FINANCIAL STATEMENTS | ||
| Balance Sheets (Unaudited) as of September 30, 2013 and December 31, 2012 | 2 | |
| Statements of Operations (Unaudited) for the Nine Months Ended September 30, 2013 and 2012 | 3 | |
| Statement of Changes in Stockholders’ Deficit (Unaudited) for the cumulative period from January 18, 2011 (date of inception) to September 30, 2013 | 4 | |
| Statements of Cash Flows (Unaudited) for the Nine Months Ended September 30, 2013 and 2013 | 5 | |
| Notes to Financial Statements | 6 | |
2
PHYXIUS PHARMA, INC.
(A Development Stage Company)
BALANCE SHEETS
| September 30, | December 31, | |||||||
| 2013 | 2012 | |||||||
| (unaudited) | ||||||||
| ASSETS | ||||||||
| License rights | $ | 1,000,000 | $ | –– | ||||
| Total assets | $ | 1,000,000 | $ | –– | ||||
| LIABILITIES AND STOCKHOLDERS’ DEFICIT | ||||||||
| Current liabilities | ||||||||
| Accounts payable and accrued expenses | $ | 255,995 | $ | 37,804 | ||||
| Accrued license fees | 1,000,000 | –– | ||||||
| Total current liabilities | 1,255,995 | 37,804 | ||||||
| Commitments and Contingencies (Note 2) | ||||||||
| Stockholders’ deficit | ||||||||
| Common stock, $0.01 par value, 2,000 shares authorized, 100 shares issued and outstanding at September 30, 2013 and December 31, 2012 | 1 | 1 | ||||||
| Preferred stock, $0.01 par value, 1,000 shares authorized, no shares issued and outstanding at September 30, 2013 and December 31, 2012 | –– | –– | ||||||
| Additional paid-in capital | 9,999 | 9,999 | ||||||
| Deficit accumulated during the development stage | (265,995 | ) | (47,804 | ) | ||||
| Total stockholders’ deficit | (255,995 | ) | (37,804 | ) | ||||
| Total liabilities and stockholders’ deficit | $ | 1,000,000 | $ | –– | ||||
The accompanying notes are an integral part of these Financial Statements.
3
PHYXIUS PHARMA, INC.
(A Development Stage Company)
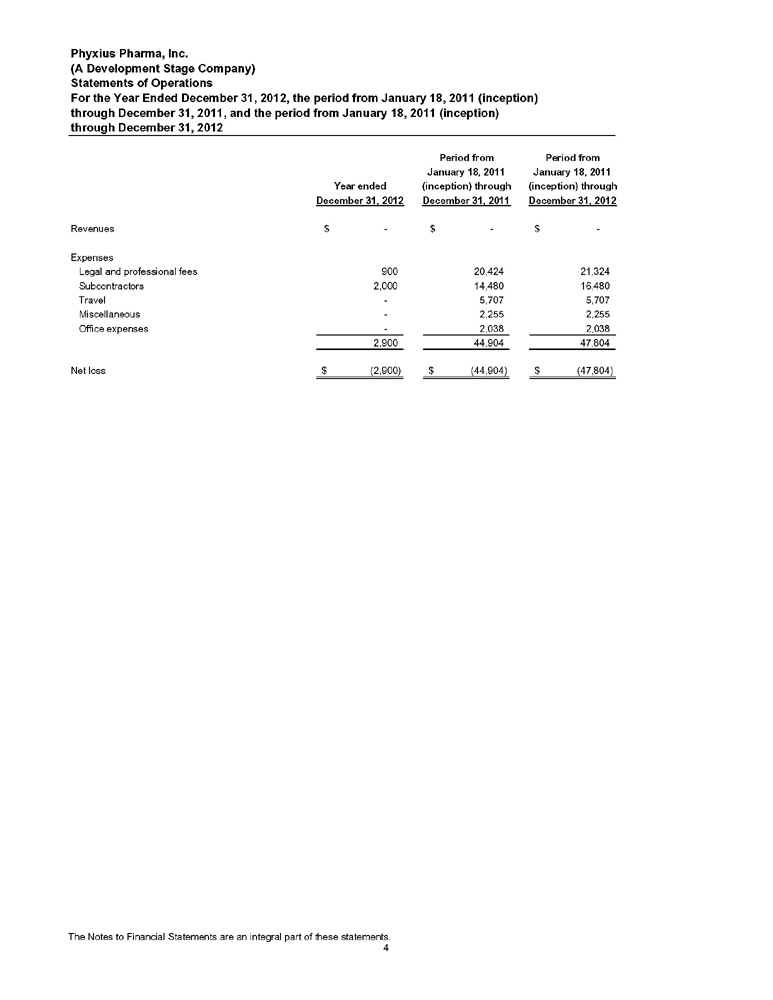
STATEMENTS OF OPERATIONS
Period from January 18, 2011 (Inception) through September 30, | Nine months ended September 30, | |||||||||||
| 2013 | 2013 | 2012 | ||||||||||
| (unaudited) | (unaudited) | (unaudited) | ||||||||||
| Revenue | $ | –– | $ | –– | $ | –– | ||||||
| Operating expenses | ||||||||||||
| Legal and professional fees | 39,515 | 18,191 | 900 | |||||||||
| Subcontractors | 216,480 | 200,000 | 2,000 | |||||||||
| Travel | 5,707 | –– | –– | |||||||||
| Miscellaneous | 2,255 | –– | –– | |||||||||
| Office expenses | 2,038 | –– | –– | |||||||||
| Total operating expenses | 265,995 | 218,191 | 2,900 | |||||||||
| Net loss | $ | 265,995 | $ | 218,191 | $ | 2,900 | ||||||
The accompanying notes are an integral part of these Financial Statements.
4
PHYXIUS PHARMA, INC.
(A Development Stage Company)
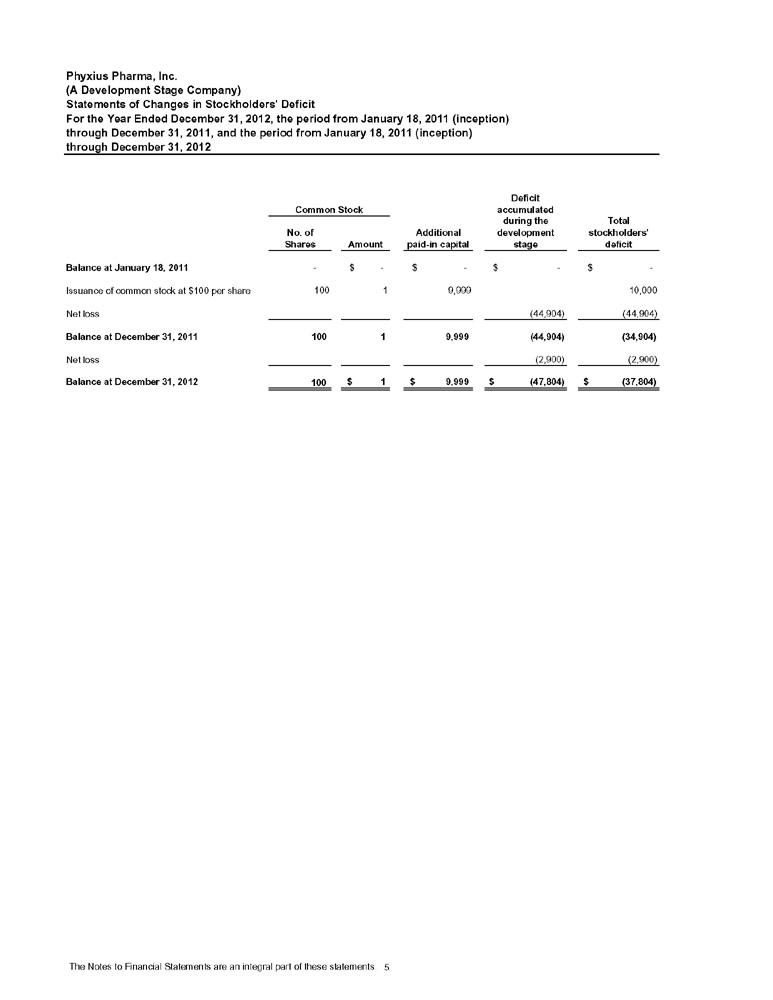
STATEMENTS OF CHANGES IN STOCKHOLDERS’ DEFICIT
For the cumulative period from January 18, 2011 (date of inception) to September 30, 2013
| Additional | Deficit accumulated | |||||||||||||||||||
| Common Stock | Paid-In | during the | ||||||||||||||||||
| Shares | Amount | Capital | development stage | Total | ||||||||||||||||
| Balances at January 18, 2011 | –– | $ | –– | $ | –– | $ | –– | $ | –– | |||||||||||
| Issuance of common stock | 100 | 1 | 9,999 | 10,000 | ||||||||||||||||
| Net Loss | (44,904 | ) | (44,904 | ) | ||||||||||||||||
| Balances at December 31, 2011 | 100 | $ | 1 | $ | 9,999 | $ | (44,904 | ) | $ | (34,904 | ) | |||||||||
| Net Loss | (2,900 | ) | (2,900 | ) | ||||||||||||||||
| Balances at December 31, 2012 | 100 | $ | 1 | $ | 9,999 | $ | (47,804 | ) | $ | (37,804 | ) | |||||||||
| Net Loss | (218,191 | ) | (218,191 | ) | ||||||||||||||||
| Balances at September 30, 2013 | 100 | $ | 1 | $ | 9,999 | $ | (265,995 | ) | $ | (255,995 | ) | |||||||||
The accompanying notes are an integral part of these Financial Statements.
5
PHYXIUS PHARMA, INC.
(A Development Stage Company)
STATEMENTS OF CASH FLOWS
Period from January 18, 2011 (Inception) through September 30, | Nine Months ended September 30, | |||||||||||
| 2013 | 2013 | 2012 | ||||||||||
| (unaudited) | (unaudited) | (unaudited) | ||||||||||
| Cash flows from operating activities | ||||||||||||
| Net loss | $ | (265,995 | ) | $ | (218,191 | ) | $ | (2,900 | ) | |||
| Adjustments to reconcile net loss to net cash provided by operating activities | ||||||||||||
| Changes in operating assets and liabilities: | ||||||||||||
| Accounts payable and accrued expenses | 255,995 | 218,191 | 2,900 | |||||||||
| Accrued license fees | 1,000,000 | 1,000,000 | –– | |||||||||
| Net cash provided by operating activities | 990,000 | 1,000,000 | –– | |||||||||
| Cash flows from investing activities | ||||||||||||
| Acquisition of license rights | (1,000,000 | ) | (1,000,000 | ) | –– | |||||||
| Net cash used in investing activities | (1,000,000 | ) | (1,000,000 | ) | –– | |||||||
| Cash flows from financing activities | ||||||||||||
| Proceeds from the issuance of common stock | 10,000 | –– | –– | |||||||||
| Net cash provided by financing activities | 10,000 | –– | –– | |||||||||
| Net decrease in cash and cash equivalents | –– | –– | –– | |||||||||
| Cash and cash equivalents at beginning of year | –– | –– | –– | |||||||||
| Cash and cash equivalents at end of year | $ | –– | $ | –– | $ | –– | ||||||
The accompanying notes are an integral part of these Financial Statements.
6
Phyxius Pharma, Inc.
(A Development Stage Company) Notes to Financial Statements
December 31, 2012 and 2011
NOTES TO FINANCIAL STATEMENTS
(Unaudited)
1. Background, Liquidity and Summary of Significant Accounting Policies
Nature of Business
Phyxius Pharma, Inc. (the "Company") was incorporated in the State of Delaware on January 18, 2011. The Company is a privately held specialty pharmaceutical company focused on commercializing critical care pharmaceutical products in the United States. The Company intends to obtain or license the rights to develop and commercialize critical care pharmaceutical products from the holders of such rights and sell the products within North America. The Company also coordinates the obtainment of regulatory approval for its products.
Development Stage Company
The Company’s primary activities since formation have been to establish its operations, negotiate and obtain product rights, and conduct clinical trials. As of September 30, 2013, planned principal operations have not begun. The Company has no extended operating history and has not generated any revenues for the nine months ended September 30, 2013 or the period from January 18, 2011 (inception) through December 31, 2012. Accordingly, the Company is considered to be in the “development stage” and has prepared the accompanying financial statements in accordance with the provisions of Accounting Standards Codification (“ASC”) No. 915 Accounting for Development Stage Entities. The financial statements reflect the financial activities of the Company from its date of inception (January 18, 2011).
As discussed in Note 4 to the financial statements, the Company completed a sale of substantially all of its assets, including licensing rights obtained subsequent to September 30, 2013, to an unrelated entity in November 2013. In conjunction with this sale, all assets and liabilities of the Company were assumed by the purchasing entity.
Use of Estimates
The preparation of financial statements in conformity with accounting principles generally accepted in the United States of America requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities and disclosure of contingent assets and liabilities at the date of the financial statements and reported amounts of revenues and expenses during the reporting period. Actual results may differ from those estimates.
Revenue Recognition
Revenue is recognized when (i) persuasive evidence of an arrangement exists, (ii) delivery has occurred or services have been rendered, (iii) the fee is fixed or determinable, and (iv) collectability is reasonably assured. Revenue generated from products sold is recognized when the risks and rights of ownership have passed to the customer; this generally occurs upon shipment of the product. From January 18, 2011 (inception) through September 30, 2013 the Company did not generate any revenues as it is in the development stage.
Income Taxes
Income taxes are accounted for in accordance with the provisions of Accounting Standards Codification Topic 740, Accounting for Income Taxes. Deferred tax assets and liabilities are recognized for the future tax consequences attributable to differences between the financial statement carrying amounts of existing assets and liabilities and their tax basis. The differences are measured using enacted tax rates expected to be applied to taxable income in the years in which those temporary differences are expected to be recovered or settled. The effect on deferred tax assets and liabilities of a change in tax rates is recognized in income in the period that includes the enactment date. Valuation allowances are established, when necessary, to reduce deferred tax assets to the amounts expected to be realized.
7
Significant components of the Company’s deferred tax assets as of September 30, 2013 and December 31, 2012 are as follows:
| September 30, | December 31, | |||||||
| 2013 | 2012 | |||||||
| Federal | $ | 34,580 | $ | 6,215 | ||||
| State | 23,939 | 4,302 | ||||||
| Total deferred tax assets | 58,519 | 10,517 | ||||||
| Valuation allowance | (58,519 | ) | (10,517 | ) | ||||
| Net deferred tax assets | $ | –– | $ | –– | ||||
For the nine months ended September 30, 2013 and for the years ended December 31, 2012 and December 31, 2011, the company incurred net operating losses and, accordingly, no provision for income taxes has been recorded. In addition, no benefit for income taxes has been recorded due to the uncertainty of the realization of any tax assets. At September 30, 2013, the Company had approximately $266,000 of federal and state net operating losses. The net operating loss carryforwards, if not utilized, will begin to expire in 2032.
The pronouncement dealing with uncertain tax position clarifies the accounting for uncertainty in income taxes recognized in the Company's consolidated financial statements and prescribes a recognition threshold and measurement process for financial statement recognition and measurement of a tax position taken or expected to be taken in a tax return. The pronouncement also provides guidance on derecognition, classification, interest and penalties, accounting in interim periods, disclosure and transition. The Company had no uncertain tax positions at September 30, 2013 or December 31, 2012. Further, there were no tax related interest or penalties included in the consolidated financial statements. There are no tax years open to examination prior to 2011, which is the year of the Company’s inception.
2. Commitments and Contingencies
Agreement with Orion Pharma- In September 2013 the Company entered into a license agreement (“License”) with Orion Pharma, Orion Corporation, Espoo, Finland ("Licensor", "Orion") whereby it obtained an exclusive, sublicenseable right to develop and commercialize levosimendan (“Product”) in the United States and Canada (the “Territory”). Pursuant to the License, the Company must use Orion’s “Simdax®” trademark to commercialize the Product. The License also grants to the Company a right of first refusal to commercialize new developments of the Product, including developments as to the formulation, presentation, means of delivery, route of administration, dosage or indication. Orion’s ongoing role under the License includes sublicense approval, serving as the sole source of manufacture, holding a first right to enforce intellectual property rights in the Territory, and certain regulatory participation rights. Additionally, the Company must grant back to Orion a broad non-exclusive license to any patents or clinical trial data related to the Product developed by the Company under the License. The License has a fifteen (15) year term, provided, however, that the License will continue after the end of the ten year term in each country in the Territory until the expiration of Orion’s patent rights in the Product in such country (the “Term”). Orion may terminate the License if the human clinical trial using the Product and studying reduction in morbidity and mortality of cardiac surgery patients at risk of low cardiac output syndrome (“LCOS”) as described in the US Food and Drug Administration (the “FDA”) agreed upon clinical study protocol (the “Study”) is not started by July 31, 2014.
Pursuant to the terms of the License, the Company must pay to Orion a non-refundable up-front payment in the amount of $1 million within thirty (30) days of the Company receiving funding for the Study, but in no event later than April 1, 2014. The License also includes the following development milestones for which the Company shall make non-refundable payments to Orion no later than twenty-eight (28) days after the occurrence of the applicable milestone event: (i) $2.0 million upon the grant of FDA approval, including all registrations, licenses, authorizations and necessary approvals, to develop and/or commercialize the Product in the United States; and (ii) $1.0 million upon the grant of regulatory approval for the Product in Canada. Once commercialized, the Company is obligated to make certain non-refundable commercialization milestone payments to Orion, aggregating up to $13.0 million, contingent upon achievement of certain cumulative net sales amounts in the Territory. The Company must also pay Orion tiered royalties based on net sales of the Product in the Territory made by the Company and its sublicensees. After the end of the Term, the Company must pay Orion a royalty based on net sales of the Product in the Territory for as long as the Company sells the Product in the Territory.
As of September 30, 2013, the Company has capitalized $1,000,000 to acquire the license rights and recorded an accrued liability arising from a contingency of $1,000,000 that has been recognized for expected license fee payments due under the acquired license. The Company expects that this expenditure will be payable in the first quarter of its fiscal year 2014.
Agreement with DCRI- In June 2011 the Company entered into an agreement with Duke Clinical Research Institute (“DCRI”) to perform research and development activities on behalf of the Company through June 30, 2012. The terms of the agreement call for total payments by the Company of $200,000, plus pass through expenses, payable on a mutually agreed-upon date to be based upon the Company’s ability to secure funding for a clinical trial. As of September 30, 2013, subsequent to entering into the license agreement with Orion, the Company recorded a liability for the payable and expects that this expenditure will be payable in the fourth quarter of its fiscal year 2013.
8
3. Stockholders’ Equity
The authorized capital of the Company consists of 3,000 shares, divided into 2,000 shares of Common Stock with a par value of $0.01 per share and 1,000 shares of Preferred Stock with a par value of $0.01 per share, of which only 100 shares of Common Stock are issued and outstanding as of September 30, 2013 and December 31, 2012.
Common Stock
On January 18, 2011, four individuals (“founding stockholders”) purchased and were issued 25 shares of Common Stock each at a price of $100 per share. In consideration for the common shares, the founding stockholders paid certain expenses on behalf of the Company in the amount of $10,000.
Preferred Stock
The Preferred Stock holders have the same rights as common stockholders, but are not entitled to voting rights. There were no preferred shares issued or outstanding as of September 30, 2013 or December 31, 2012.
4. Subsequent Events
On October 21, 2013, the Company entered into an agreement for the sale of substantially all of its assets to Oxygen Biotherapeutics, Inc., an unrelated entity, which sale was completed in November 2013. In connection with the sale, the Company assigned its licensing agreement as described above, the right to enforce any agreement, any and all intellectual property, all product registration materials, all books, records and other relevant business documents, and any goodwill that may exist, to the purchasing entity. In addition, certain liabilities and future commitments of the Company were assumed by the purchasing entity on the date of sale. In consideration for the purchased assets, the purchasing entity issued shares of its own common stock and convertible preferred stock to the four stockholders of Phyxius Pharma, Inc.
Management has evaluated subsequent events through the date the financial statements were issued. Based on this evaluation, the Company has determined that no other subsequent events have occurred which require disclosure through the date that these financial statements were issued.
9
EXHIBIT 99.4
UNAUDITED PRO FORMA CONDENSED COMBINED FINANCIAL INFORMATION
UNAUDITED PRO FORMA CONDENSED COMBINED
BALANCE SHEET
As of October 31, 2013
| Historical | |||||||||||||||||
Oxygen Biotherapeutics, Inc. | Phyxius Pharma, Inc. | Pro Forma Adjustments | Pro Forma Combined | ||||||||||||||
| ASSETS | |||||||||||||||||
| Current assets | |||||||||||||||||
| Cash and cash equivalents | $ | 2,542,942 | $ | - | $ | (125,000 | ) | (a) | $ | 2,417,942 | |||||||
| Accounts receivable | 64,815 | - | - | 64,815 | |||||||||||||
| Government grant receivable | 59,058 | - | - | 59,058 | |||||||||||||
| Inventory | 97,985 | - | - | 97,985 | |||||||||||||
| Prepaid expenses | 78,222 | - | - | 78,222 | |||||||||||||
| Other current assets | 251,055 | - | - | 251,055 | |||||||||||||
| Total current assets | 3,094,077 | - | (125,000 | ) | 2,969,077 | ||||||||||||
| Property and equipment, net | 160,212 | - | - | 160,212 | |||||||||||||
| Debt issuance costs, net | 85,735 | - | - | 85,735 | |||||||||||||
| Intangible assets, net | 963,810 | 1,000,000 | 21,000,000 | (b) | 22,963,810 | ||||||||||||
| Goodwill | - | - | 3,302,995 | (c) | 3,302,995 | ||||||||||||
| Other assets | 58,262 | - | - | 58,262 | |||||||||||||
| Total assets | $ | 4,362,096 | $ | 1,000,000 | $ | 24,177,995 | $ | 29,540,091 | |||||||||
| LIABILITIES AND STOCKHOLDERS’ EQUITY (DEFICIT) | |||||||||||||||||
| Current liabilities | |||||||||||||||||
| Accounts payable | $ | 920,844 | $ | 255,995 | $ | - | $ | 1,176,839 | |||||||||
| Accrued liabilities | 322,443 | 1,000,000 | - | 1,322,443 | |||||||||||||
| Current portion of notes payable, net | 233,324 | - | - | 233,324 | |||||||||||||
| Total current liabilities | 1,476,611 | 1,255,995 | - | 2,732,606 | |||||||||||||
| Other liabilities | 32,796 | - | - | 32,796 | |||||||||||||
| Total liabilities | 1,509,407 | 1,255,995 | - | 2,765,402 | |||||||||||||
| Commitments and contingencies | |||||||||||||||||
| Stockholders' equity (deficit) | |||||||||||||||||
| Preferred stock | 2 | - | 1 | (d) | 3 | ||||||||||||
| Common stock | 682 | 1 | 136 | (d) | 819 | ||||||||||||
| Additional paid-in capital | 125,672,848 | 9,999 | 24,036,863 | (d) | 149,719,710 | ||||||||||||
| Deficit accumulated during the development stage | (122,820,843 | ) | (265,995 | ) | 140,995 | (d) | (122,945,843 | ) | |||||||||
| Total stockholders’ equity (deficit) | 2,852,689 | (255,995 | ) | 24,177,995 | 26,774,689 | ||||||||||||
| Total liabilities and stockholders' equity (deficit) | $ | 4,362,096 | $ | 1,000,000 | $ | 24,177,995 | $ | 29,540,091 | |||||||||
See accompanying notes to the pro forma condensed combined financial statements.
1
UNAUDITED PRO FORMA CONDENSED COMBINED
STATEMENT OF OPERATIONS
For the six months ended October 31, 2013
| Historical | |||||||||||||||||
Oxygen Biotherapeutics, Inc. | Phyxius Pharma, Inc. | Pro Forma Adjustments | Pro Forma Combined | ||||||||||||||
| Product revenue | $ | 60,079 | $ | - | $ | - | $ | 60,079 | |||||||||
| Cost of sales | 30,864 | - | - | 30,864 | |||||||||||||
| Net product revenue | 29,215 | - | - | 29,215 | |||||||||||||
| Government grant revenue | 192,297 | - | - | 192,297 | |||||||||||||
| Total net revenue | 221,512 | - | - | 221,512 | |||||||||||||
| Operating expenses | |||||||||||||||||
| Selling, general, and administrative | 2,403,926 | 18,191 | 1,148,333 | (e), (f) | 3,570,450 | ||||||||||||
| Research and development | 1,521,458 | 200,000 | - | 1,721,458 | |||||||||||||
| Total operating expenses | 3,925,384 | 218,191 | 1,148,333 | 5,291,908 | |||||||||||||
| Net operating loss | 3,703,872 | 218,191 | 1,148,333 | 5,070,396 | |||||||||||||
| Interest expense | 2,072,660 | - | - | 2,072,660 | |||||||||||||
| Other (income) expense | 227 | - | - | 227 | |||||||||||||
| Net loss | $ | 5,776,759 | $ | 218,191 | $ | 1,148,333 | $ | 7,143,283 | |||||||||
| Preferred stock dividend | 4,645,340 | - | - | 4,645,340 | |||||||||||||
| Net loss attributable to common stockholders | $ | 10,422,099 | $ | 218,191 | $ | 1,148,333 | $ | 11,788,623 | |||||||||
| Net loss per share, basic | $ | (2.94 | ) | $ | (2.40 | ) | |||||||||||
| Weighted average number of common shares outstanding, basic | 3,549,698 | 1,366,844 | (g) | 4,916,542 | |||||||||||||
| Net loss per share, diluted | $ | (2.96 | ) | $ | (2.39 | ) | |||||||||||
| Weighted average number of common shares outstanding, diluted | 3,556,357 | 1,366,844 | (g) | 4,923,201 | |||||||||||||
See accompanying notes to the pro forma condensed combined financial statements.
2
UNAUDITED PRO FORMA CONDENSED COMBINED
STATEMENT OF OPERATIONS
For the year ended April 30, 2013
| Historical | |||||||||||||||||
Oxygen Biotherapeutics, Inc. | Phyxius Pharma, Inc. | Pro Forma Adjustments | Pro Forma Combined | ||||||||||||||
| Product revenue | $ | 92,683 | $ | - | $ | - | $ | 92,683 | |||||||||
| Cost of sales | 43,111 | - | - | 43,111 | |||||||||||||
| Net product revenue | 49,572 | �� | - | - | 49,572 | ||||||||||||
| Government grant revenue | 1,141,356 | - | - | 1,141,356 | |||||||||||||
| Total net revenue | 1,190,928 | - | - | 1,190,928 | |||||||||||||
| Operating expenses | |||||||||||||||||
| Selling, general, and administrative | 3,676,145 | 595 | 2,296,667 | (e), (f) | 5,973,407 | ||||||||||||
| Research and development | 2,455,816 | 2,000 | - | 2,457,816 | |||||||||||||
| Restructuring expense | 220,715 | - | - | 220,715 | |||||||||||||
| Loss on impairment of long-lived assets | 27,279 | - | - | 27,279 | |||||||||||||
| Total operating expenses | 6,379,955 | 2,595 | 2,296,667 | 8,679,217 | |||||||||||||
| Net operating loss | 5,189,027 | 2,595 | 2,296,667 | 7,488,289 | |||||||||||||
| Interest expense | 4,238,456 | - | - | 4,238,456 | |||||||||||||
| Loss on extinguishment of debt | - | - | - | - | |||||||||||||
| Other (income) expense | (11,683 | ) | - | - | (11,683 | ) | |||||||||||
| Net loss | $ | 9,415,800 | $ | 2,595 | $ | 2,296,667 | $ | 11,715,062 | |||||||||
| Preferred stock dividend | 958,071 | - | - | 958,071 | |||||||||||||
| Net loss attributable to common stockholders | $ | 10,373,871 | $ | 2,595 | $ | 2,296,667 | $ | 12,673,133 | |||||||||
| Net loss per share, basic | $ | (6.29 | ) | $ | (4.20 | ) | |||||||||||
| Weighted average number of common shares outstanding, basic | 1,650,280 | 1,366,844 | (g) | 3,017,124 | |||||||||||||
| Net loss per share, diluted | $ | (6.68 | ) | $ | (4.05 | ) | |||||||||||
| Weighted average number of common shares outstanding, diluted | 1,759,025 | 1,366,844 | (g) | 3,125,869 | |||||||||||||
See accompanying notes to the pro forma condensed combined financial statements.
3
NOTES TO UNAUDITED PRO FORMA CONDENSED COMBINED
FINANCIAL STATEMENTS
1. Description of Transaction
On November 13, 2013, Oxygen Biotherapeutics, Inc. (the “Company”), through its wholly owned subsidiary, Life Newco, Inc., a Delaware corporation (“Life Newco”), consummated its previously announced acquisition of certain assets of Phyxius Pharma, Inc., a Delaware corporation (“Phyxius”) pursuant to an Asset Purchase Agreement, dated October 21, 2013 (the “Purchase Agreement”), by and among the Company, Life Newco, Phyxius and the stockholders of Phyxius (the “Phyxius Stockholders”).
Under the terms and subject to the conditions of the Purchase Agreement, Life Newco acquired (the “Acquisition”) certain assets (the “Purchased Assets”), including that certain License Agreement (the “License”), dated September 20, 2013 by and between Phyxius and Orion Corporation, a global healthcare company incorporated under the laws of Finland (“Orion”), and that certain Side Letter (the “Side Letter”), dated October 15, 2013 by and between Phyxius and Orion. The License grants Life Newco an exclusive, sublicenseable right to develop and commercialize pharmaceutical products containing Levosimendan, 2.5 mg/ml concentrate for solution for infusion / 5ml vial (the “Product”) in the United States and Canada (the “Territory”). Pursuant to the License, Life Newco must use Orion’s “Simdax®” trademark to commercialize the Product. The License also grants to Life Newco a right of first refusal to commercialize new developments of the Product, including developments as to the formulation, presentation, means of delivery, route of administration, dosage or indication. Orion’s ongoing role under the License includes sublicense approval, serving as the sole source of manufacture, holding a first right to enforce intellectual property rights in the Territory, and certain regulatory participation rights. Additionally, Life Newco must grant back to Orion a broad non-exclusive license to any patents or clinical trial data related to the Product developed by Life Newco under the License. The License has a fifteen (15) year term, provided, however, that the License will continue after the end of the fifteen year term in each country in the Territory until the expiration of Orion’s patent rights in the Product in such country (the “Term”). Orion may terminate the License if the human clinical trial using the Product and studying reduction in morbidity and mortality of cardiac surgery patients at risk of low cardiac output syndrome (LCOS) as described in the US Food and Drug Administration (the “FDA”) agreed upon clinical study protocol (the “Study”) is not started by July 31, 2014.
Pursuant to the terms of the License, Life Newco must pay to Orion a non-refundable up-front payment in the amount of $1 million within thirty (30) days of Life Newco receiving funding for the Study, but in no event later than April 1, 2014. The License also includes the following development milestones for which Life Newco shall make non-refundable payments to Orion no later than twenty-eight (28) days after the occurrence of the applicable milestone event: (i) $2.0 million upon the grant of FDA approval, including all registrations, licenses, authorizations and necessary approvals, to develop and/or commercialize the Product in the United States; and (ii) $1.0 million upon the grant of regulatory approval for the Product in Canada. Once commercialized, Life Newco is obligated to make certain non-refundable commercialization milestone payments to Orion, aggregating up to $13.0 million, contingent upon achievement of certain cumulative net sales amounts in the Territory. Life Newco must also pay Orion tiered royalties based on net sales of the Product in the Territory made by Life Newco and its sublicensees. After the end of the Term, Life Newco must pay Orion a royalty based on net sales of the Product in the Territory for as long as Life Newco sells the Product in the Territory.
Pursuant to the Purchase Agreement, the Company issued 1,366,844 shares of its common stock (“Common Stock”) and 32,992 shares of its Series E convertible preferred stock, which are convertible into an aggregate of 3,299,200 shares of Common Stock (“Preferred Stock” and, together with the Common Stock, the “Consideration”) to the Phyxius Stockholders; however, 6,996 shares of the Preferred Stock, representing approximately 15% of the Consideration, are being held in escrow as security for post-closing indemnification obligations of Phyxius and the Phyxius Stockholders.
The rights, preferences and privileges of the Preferred Stock are set forth in the Certificate of Designation of Series E Convertible Preferred Stock (the “Certificate of Designation”) that the Company filed with the Secretary of State of the State of Delaware on November 13, 2013. Each share of Preferred Stock will automatically convert into 100 shares of Common Stock following receipt of stockholder approval for the transaction. Approximately 11% of the shares of converted Common Stock will vest immediately upon receipt of stockholder approval for the transaction, while the remainder will vest upon achievement of certain performance milestones related to the development and commercialization of the levosimendan product in North America. In addition, all unvested converted Common Stock will vest if certain change of control transactions or significant equity financings occur within 24 months of the closing of the Acquisition. The number of shares of Common Stock into which the Preferred Stock converts is subject to adjustment in the case of stock splits, stock dividends, combinations of shares and similar recapitalization transactions. The Preferred Stock does not carry dividends or a liquidation preference. The Preferred Stock carries voting rights aggregating 4.99% of the Company’s Common Stock voting power immediately prior to the closing of the Acquisition.
In connection with the closing of the Acquisition, Phyxius’ co-founder, Chief Executive Officer and stockholder, John Kelley, became the Company’s Chief Executive Officer and two other Phyxius employees and stockholders, Doug Randall and Douglas Hay, PhD became employees of the Company as Vice President, Business and Commercial Operations and Vice President, Regulatory Affairs, respectively. Michael Jebsen, the Company’s prior Interim Chief Executive Officer and current Chief Financial Officer, is continuing to serve as the Company’s Chief Financial Officer. In addition, Mr. Kelley was appointed to the Company’s Board of Directors on December 19, 2013, while another designee will be appointed to the Board of Directors following receipt of stockholder approval for the transaction.
4
2. Basis of Presentation
The accompanying pro forma balance sheet and pro forma statements of operations combine the historical financial information of the Company and Phyxius and are adjusted on a pro forma basis to give effect to the acquisition as described in the notes to the unaudited pro forma condensed combined financial statements. The pro forma balance sheet reflects the acquisition, which occurred on November 13, 2013, as if it had been consummated on October 31, 2013, and the pro forma statements of operations for the quarter ended October 31, 2013 and the year ended April 30, 2013 reflect the acquisition as if it had been consummated on May 1, 2012. The pro forma financial statements have been derived from and should be read in conjunction with the historical consolidated financial statements of each of the Company and Phyxius, which were (in the case of the Company’s financial statements) included in the Company’s Annual Report on Form 10-K filed with the SEC on June 26, 2013 and the Company’s Quarterly Report on Form 10-Q filed with the SEC on December 17, 2013 or are (in the case of Phyxius’ financial statements) included as Exhibits 99.2 and 99.3 to the Current Report on Form 8-K/A to which this exhibit is attached.
The pro forma financial statements are provided for illustrative purposes only and are not intended to represent, and are not necessarily indicative of, what the operating results or financial position of the Company would have been had acquisition been completed on the dates indicated, nor are they necessarily indicative of the Company’s future operating results or financial position. The pro forma financial statements do not reflect the impacts of any potential operational efficiencies, asset dispositions, cost savings or economies of scale that the Company may achieve with respect to the combined operations. Additionally, the pro forma statements of operations do not include non-recurring charges or credits which result directly from the transactions.
The pro forma financial statements have been prepared using the acquisition method of accounting in accordance with Accounting Standards Codification (“ASC”) 805, “Business Combinations.” ASC 805 requires, among other things, that assets acquired and liabilities assumed be recognized at their fair values, as determined in accordance with ASC 820, “Fair Value Measurements,” as of the acquisition date. For certain assets and liabilities, book value approximates fair value. In addition, ASC 805 establishes that the consideration transferred be measured at the closing date of the asset acquisition at the then-current market price, which may be different than the amount of consideration assumed in the pro forma financial statements. Under ASC 805, acquisition-related transaction costs (i.e., advisory, legal, valuation, other professional fees) and certain acquisition-related restructuring charges impacting the target company are expensed in the period in which the costs are incurred.
As of the filing date of the Current Report on Form 8-K/A to which this exhibit is attached, Phyxius’ assets and liabilities are presented at their preliminary estimated fair values, with the excess of the purchase price over the sum of these fair values presented as goodwill. The Company has not completed the detailed valuation studies necessary to arrive at the required estimates of fair market value of the Phyxuis’ assets acquired and liabilities assumed and the related allocations of purchase price. The valuations of acquired assets and liabilities are in process and are not expected to be finalized until later in 2014, as information may become available within the measurement period which indicates a potential change to these valuations. Accordingly, the final allocations of and the effects on the results of operations may differ materially from the preliminary allocations and unaudited pro forma combined amounts included herein.
Under the acquisition method of accounting, the Phyxius assets acquired and liabilities assumed will be recorded on the Company’s consolidated financial statements as of the consummation of the acquisition, primarily at their respective fair values. In addition, Phyxius’ results of operations will be included with the Company’s consolidated results of operations beginning on the closing date, and the Company’s consolidated results of operations prior to the closing date will not be retroactively restated to reflect Phyxius’ results of operations.
3. Accounting Policies
The Company is currently performing a detailed review of Phyxius’ accounting policies. As a result of this review, it may become necessary to conform Phyxius’ accounting policies to be consistent with the accounting policies of the Company. To date, the Company has not identified any significant differences in accounting policies.
5
4. Purchase Consideration
The following table reflects the acquisition consideration (in thousands, except per share amounts):
| Number of shares of Common Stock issued at closing (1) | 1,367 | |||
| Fair value per share of Common Stock (2) | $ | 6.40 | ||
| Fair value of shares of Common Stock issued (3) | $ | 8,748 | ||
| Fair value of Series E Convertible Preferred Stock issued at closing (4) | 15,299 | |||
| Total fair value of consideration transferred | $ | 24,047 |
| (1) | Represents the number of shares issued at the close of the acquisition on November 13, 2013 as set forth in the Purchase Agreement. |
| (2) | Represents the closing price of the Company’s common stock on The NASDAQ Stock Market LLC on November 13, 2013. |
| (3) | Represents the number of shares issued at closing multiplied by the fair value per share. |
| (4) | Represents the fair value of 32,992 shares of the Company’s Series E Convertible Preferred Stock issued at the close of the acquisition on November 13, 2013. The Preferred Stock is convertible into an aggregate of 3,299,200 shares of the Company’s Common Stock as set forth in the Purchase Agreement. The rights, preferences and privileges of the Preferred Stock are set forth in the Certificate of Designation of Series E Convertible Preferred Stock that the Company filed with the Secretary of State of the State of Delaware on November 13, 2013. Each share of Preferred Stock will automatically convert into 100 shares of Common Stock following receipt of stockholder approval for the transaction. Approximately 11% of the shares of converted Common Stock will vest immediately upon receipt of stockholder approval for the transaction, while the remainder will vest upon achievement of certain performance milestones related to the development and commercialization of the levosimendan product in North America. |
6
5. Preliminary Purchase Price Allocation
The following table summarizes the preliminary purchase price allocation based on estimated fair values as if the acquisition had been consummated on October 31, 2013 (in thousands):
| Intangible assets | $ | 22,000 | ||
| Accounts payable | (256 | ) | ||
| Accrued expenses | (1,000 | ) | ||
| Total identifiable net assets | 20,744 | |||
| Goodwill | 3,303 | |||
| Total fair value of consideration | $ | 24,047 | ||
6. Pro Forma Adjustments
Pro forma adjustments reflect those matters that are a direct result of the Purchase Agreement, which are factually supportable and, for pro forma adjustments to the pro forma statements of operations, are expected to have continuing impact. The pro forma adjustments are based on preliminary estimates that may change as additional information is obtained. Given the historical net loss positions of both companies and the full valuation allowances applied to the deferred tax assets at April 30, 2013, there is no expected tax impact of these adjustments on the pro forma balance sheet or statements of operations.
Adjustments to the pro forma balance sheet:
| (a) | Represents $125,000 of estimated transaction costs related to the acquisition that were not previously reflected in the historical financial statements |
| (b) | Represents the preliminary allocation of the fair value of the consideration transferred to the acquired intangible assets of Phyxius based on their estimated fair values. Intangible assets that have been identified in this preliminary allocation are in-process research and development (“IPR&D”). |
| (c) | Represents the difference between the estimated purchase price and the estimated fair values of the identifiable assets acquired and liabilities assumed. |
| (d) | The following pro forma adjustments represent the effects of eliminating Phyxius’ equity accounts and issuing the Company’s shares pursuant to the Purchase Agreement, and consist of: |
| (i) | the elimination of the par value of Phyxius’ common stock | (1 | ) | ||
| the par value of the Company’s shares issued at closing | 137 | ||||
| net change in common stock | 136 | ||||
| (ii) | the par value of the Series E preferred stock issued at closing | 1 | |||
| (iii) | the elimination of Phyxius’ paid-in capital | (9,999 | ) | ||
| paid-in capital for the Company’s shares issued at closing | 24,046,862 | ||||
| net change in paid-in capital | 24,036,863 | ||||
| (iv) | the elimination of Phyxius’ accumulated deficit | 265,995 | |||
| estimated acquisition transaction fees to be expensed | (125,000 | ) | |||
| net change in accumulated deficit | 140,995 |
| Adjustments to the pro forma statements of operations: |
| (e) | Represents the amortization of intangible assets acquired in the acquisition based on their estimated fair values and useful life of 15 years of $733,333 and $1,466,667 for the six months ended October 31, 2013 and the twelve months ended April 30, 2013, respectively. Amortization expense has been calculated on a straight-line basis. |
| (f) | Represents the salaries for the Phyxius employees hired by the Company in connection with the closing of the acquisition of $415,000 and $830,000 for the six months ended October 31, 2013 and the twelve months ended April 30, 2013, respectively. |
| (g) | Represents the impact of 1,366,844 shares of Common Stock issued in connection with the closing of the acquisition. |
7