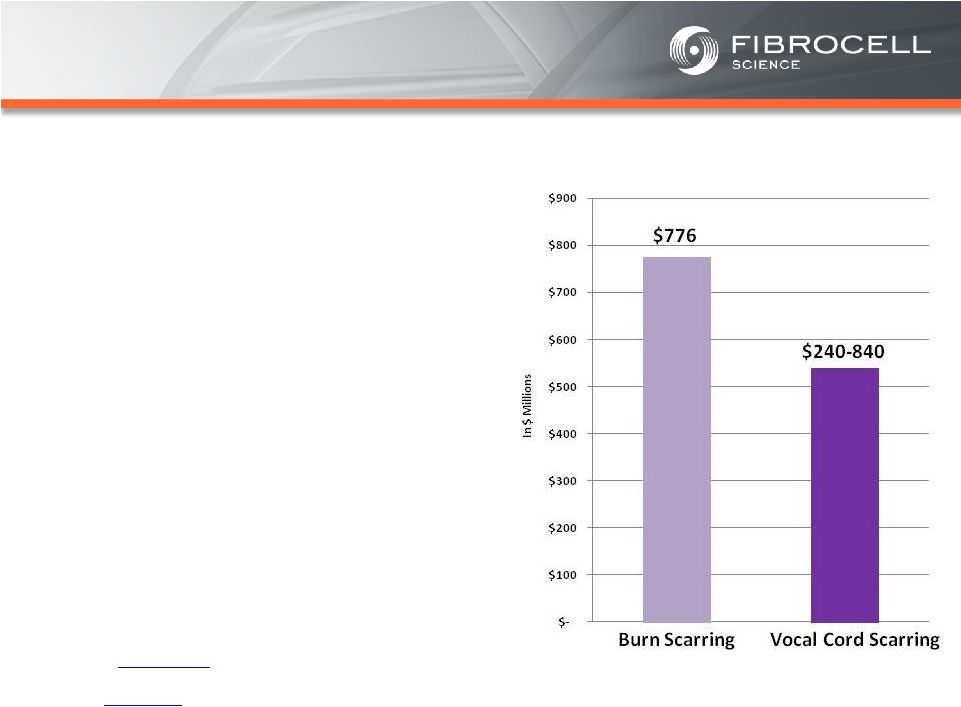
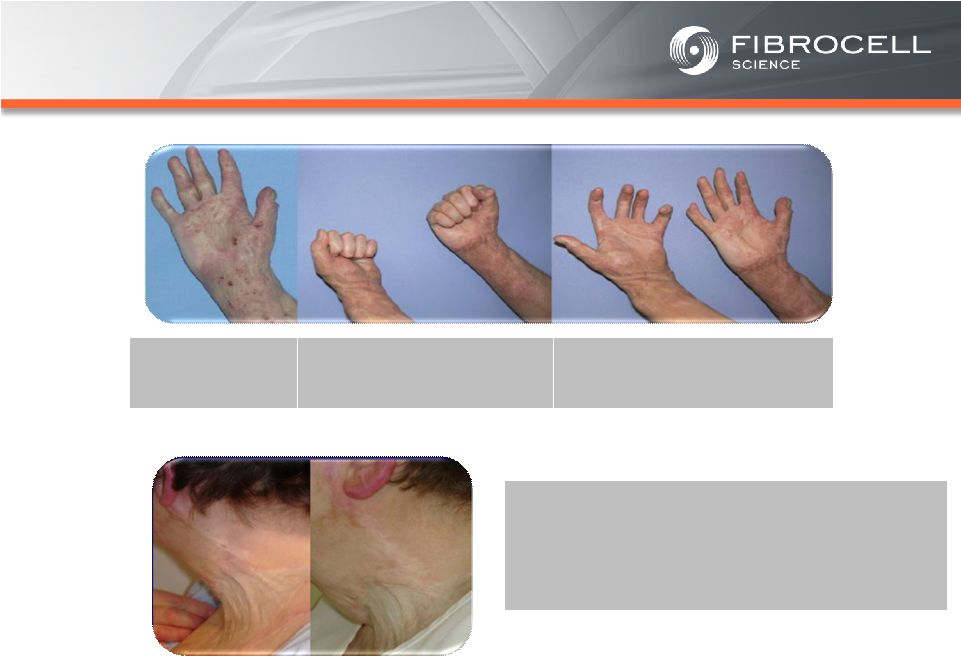
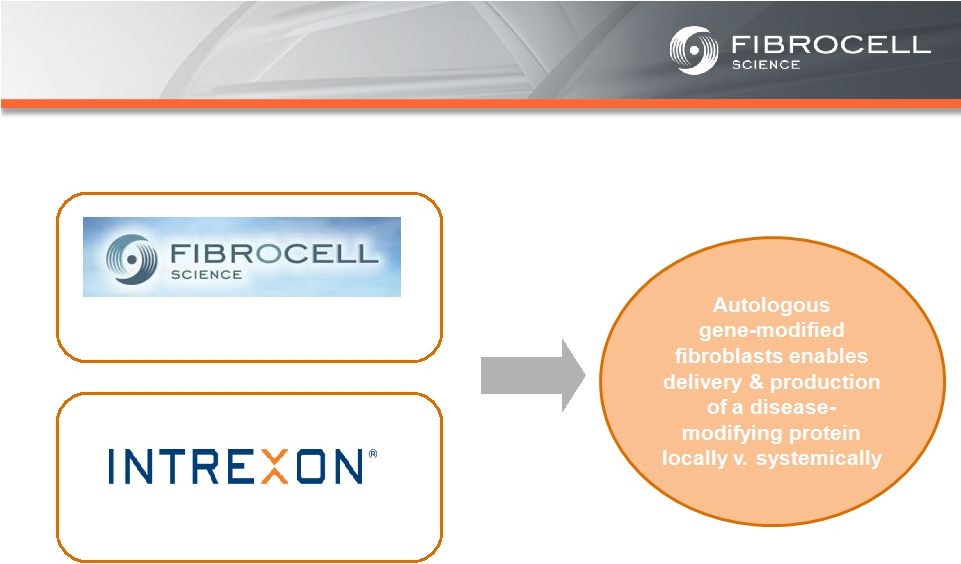
Appendix –References Slide 21 – Orphan Skin Diseases (1) Cutaneous eosinophilias represent a family of more than 30 different conditions ranging from eosinophilic cellulitis (Wells’ syndrome) to eosinophilic dermatosis and eosinophilic fasciitis. The prevalence varies based on which conditions are targeted for treatment. It could be as low as a few hundred patients (Wells’ syndrome and eosinophilic fasciitis) to thousands of patients (Duhring disease, which affects 15% to 25% of celiac patients). We chose 4,000 patients based on the Fibrocell press release dated July 1, 2013. (2) Beyer et al. “Recent Trends in Systemic Psoriasis Treatment Costs” Arch Dermatol 2010;146(1):46-54. We assume that the cutaneous eosinophilias indication will command a higher price than the psoriasis indication due to the need for new treatment options, the frequency of treatment, and the severity of the condition. (3) Kaplan et al. “Localized Fibrosing Disorders – Linear Scleroderma, Morphea, and Regional Fibrosis” eMedicine March 6, 2013. (4) Beyer et al. “Recent Trends in Systemic Psoriasis Treatment Costs” Arch Dermatol 2010;146(1):46-54. We assume that the morphea indication will command a higher price than the psoriasis indication due to the need for new treatment options, the frequency of treatment, and the severity of the condition. (5) Stanford School of Medicine, “Epidermolysis Bullosa Clinic Frequently Asked Questions” Available at: http://dermatology.stanford.edu/gsdc/eb_clinic/eb-faqs.html. Accessed July 11, 2013. (6) The Dystrophic Epidermolysis Bullosa Research Association of America (DEBRA), “About EB” http://www.debra.org/abouteb. Accessed July 11, 2013. (7) Herper, Matthew. “How A $440,000 Drug Is Turning Alexion Into Biotech‘s New Innovation Powerhouse.” Forbes. 5 September 2012 (8) The price range represents the price potential for a new therapy for a severe ultra rare disease based on currently marketed rare disease therapies such as Soliris® (eculizumab - ~$400,000/year), Elaprase® (idursulfase - ~$375,000/year), Naglazyme® (galsulfase - ~$365,000/year), Myozyme® (alglucosidasealfa - ~$300,000/year), and Fabrazyme® (agalsidase beta - ~$200,000/year). 31 |