to
UNDER
THE SECURITIES ACT OF 1933
| Delaware | 2834 | 52-1253406 | ||
| (State or other jurisdiction of incorporation or organization) | (Primary Standard Industrial Classification Code Number) | (I.R.S. Employer Identification Number) |
Rockville, MD 20850
(301) 208-9191
(Address, including zip code, and telephone number, including area code, of registrant’s principal executive offices)
President and Chief Executive Officer
15245 Shady Grove Road, Suite 470
Rockville, MD 20850
(301) 208-9191
(Name, address, including zip code, and telephone number, including area code, of agent for service)
Brian F. Leaf, Esq.
Cooley LLP
One Freedom Square, Reston Town Center
11951 Freedom Drive
Reston, VA 20190-5656
(703) 456-8000
| Large accelerated filero | Accelerated filero | Non-accelerated filero(Do not check if a smaller reporting company) | Smaller reporting companyþ |
The information in this prospectus is not complete and may be changed. We may not sell these securities until the registration statement filed with the Securities and Exchange Commission is effective. This prospectus is not an offer to sell these securities and we are not soliciting offers to buy these securities in any state where the offer or sale is not permitted.

| Page | ||||
| 1 | ||||
| 8 | ||||
| 22 | ||||
| 23 | ||||
| 24 | ||||
| 25 | ||||
| 26 | ||||
| 27 | ||||
| 28 | ||||
| 36 | ||||
| 45 | ||||
| 54 | ||||
| 62 | ||||
| 65 | ||||
| 67 | ||||
| 71 | ||||
| 74 | ||||
| 75 | ||||
| 75 | ||||
| 75 | ||||
| 75 | ||||
| F-1 | ||||
-i-
| • | RGN-352, an injectable product candidate to treat cardiovascular diseases, central nervous system diseases, and other medical indications that may be treated by systemic administration; | ||
| • | RGN-259, a topical eye drop for regeneration of corneal tissues damaged by injury, disease or other pathology; and | ||
| • | RGN-137, a topically applied gel for chronic dermal wounds and reduction of scar tissue. |
1
| • | signaling adult epicardial progenitor cells, or EPCs, to differentiate into coronary blood vessels; | ||
| • | forming cardiomyocytes that repair damaged heart tissue; | ||
| • | triggering the maturation of stem cells into cells that produce myelin, the outer covering of nerve cells in the central nervous system; | ||
| • | improving neurologic functional recovery; | ||
| • | regulating actin, which is critical to cell structure and mobility; | ||
| • | stimulating angiogenesis, or blood vessel development; | ||
| • | reducing inflammation, which is implicated in many medical indications; | ||
| • | stimulating the formation of collagen and up-regulation of laminin-5 to accelerate tissue repair; and | ||
| • | preventing apoptosis, or programmed cell death. |
2
3
4
| Common stock outstanding prior to the offering | 79,860,282 shares, including the 958,333 initial commitment shares already issued to Lincoln Park under the Purchase Agreement and the 1,851,852 shares already issued to Lincoln Park outside of the Purchase Agreement | |
| Common Stock offered by the selling stockholder | 15,000,000 shares, consisting of up to 958,333 shares to be issued to Lincoln Park as additional commitment shares and the remaining shares to be purchased from time to time under the Purchase Agreement | |
| Common stock to be outstanding after giving effect to the issuance of 15,000,000 shares to Lincoln Park under the Purchase Agreement | 94,860,282 shares | |
| Use of proceeds | We will not receive any proceeds from the sale of the shares of common stock by Lincoln Park. However, we may receive up to $11,000,000 from sales of shares under the Purchase Agreement. Any proceeds that we receive from sales to Lincoln Park under the Purchase Agreement will be used to further development of our drug candidates and for general corporate purposes. See “Use of Proceeds.” | |
| OTC Bulletin Board symbol | RGRX | |
| Risk factors | This investment involves a high degree of risk. See “Risk Factors” for a discussion of factors you should consider carefully before making an investment decision. |
| • | 5,348,863 shares of our common stock issuable upon the exercise of outstanding stock options, with a weighted average exercise price of $1.37 per share; | ||
| • | 4,327,500 shares of our common stock available for future issuance under our 2010 Equity Incentive Plan; | ||
| • | 16,136,900 shares of our common stock issuable upon the exercise of outstanding warrants, with a weighted-average exercise price of $0.89 per share; and | ||
| • | additional shares of our common stock that are potentially issuable to Lincoln Park under the Purchase Agreement beyond the 15,000,000 shares being offered by this prospectus. |
5
| • | 958,333 shares represent shares that we are required to issue proportionally in the future, as a commitment fee, if and when we sell additional shares to Lincoln Park under the Purchase Agreement; and | ||
| • | The remainder represent shares we may sell to Lincoln Park under the Purchase Agreement. |
6
| Year Ended | Nine Months Ended | |||||||||||||||
| December 31, | September 30, | |||||||||||||||
| 2009 | 2008 | 2010 | 2009 | |||||||||||||
Statement of Operations Data: | ||||||||||||||||
| Sponsored research revenue | $ | — | $ | 168,412 | $ | 53,819 | $ | — | ||||||||
| Operating expenses: | ||||||||||||||||
| Research and development | 3,724,514 | 7,149,808 | 1,819,036 | 3,064,248 | ||||||||||||
| General and administrative | 2,781,790 | 3,805,346 | 2,345,619 | 2,161,539 | ||||||||||||
| Total operating expenses | 6,506,304 | 10,955,154 | 4,164,655 | 5,225,787 | ||||||||||||
| Loss from operations | (6,506,304 | ) | (10,786,742 | ) | (4,110,836 | ) | (5,225,787 | ) | ||||||||
| Interest income | 12,444 | 149,777 | 6,840 | 10,304 | ||||||||||||
| Net loss | $ | (6,493,860 | ) | (10,636,965 | ) | (4,103,996 | ) | (5,215,483 | ) | |||||||
| Basic and diluted net loss per share | $ | (0.12 | ) | $ | (0.21 | ) | $ | (0.06 | ) | $ | (0.10 | ) | ||||
| Shares used to compute basic and diluted net loss per share | 55,680,525 | 50,967,617 | 66,729,519 | 54,216,430 | ||||||||||||
| As of September 30, 2010 | ||||
Balance Sheet Data: | ||||
| Cash and cash equivalents | $ | 4,975,947 | ||
| Working capital | 4,423,905 | |||
| Total assets | 5,615,670 | |||
| Common stock | 73,531 | |||
| Additional paid-in capital | 92,997,669 | |||
| Accumulated deficit | (88,605,400 | ) | ||
| Total stockholders’ equity | 4,465,800 | |||
7
8
| • | the scope of our clinical trials, which is significantly influenced by the quality of clinical data achieved as trials are completed and the requirements established by regulatory authorities; | ||
| • | the speed with which we complete our clinical trials, which depends on our ability to attract and enroll qualifying patients and the quality of the work performed by our clinical investigators; | ||
| • | the time required to prosecute, enforce and defend our intellectual property rights, which depends on evolving legal regimes and infringement claims that may arise between us and third parties; | ||
| • | the ability to manufacture at scales sufficient to supply commercial quantities of any of our product candidates that receive regulatory approval, which may require levels of effort not currently anticipated; and | ||
| • | the successful commercialization of our product candidates, which will depend on our ability to either create or partner with an effective commercialization organization and which could be delayed or prevented by the emergence of equal or more effective therapies. |
9
10
| • | be found ineffective or cause harmful side effects during preclinical studies or clinical trials; | ||
| • | fail to receive necessary regulatory approvals; | ||
| • | be precluded from commercialization by proprietary rights of third parties; | ||
| • | be difficult to manufacture on a large scale; or | ||
| • | be uneconomical or otherwise fail to achieve market acceptance. |
| • | the FDA or other health regulatory authorities, or institutional review boards, or IRBs, do not approve a clinical trial protocol or place a clinical trial on hold; | ||
| • | suitable patients do not enroll in a clinical trial in sufficient numbers or at the expected rate, for reasons such as the size of the patient population, the proximity of patients to clinical sites, the eligibility criteria for the trial, the perceptions of investigators and patients regarding safety, and the availability of other treatment options; | ||
| • | clinical trial data is adversely affected by trial conduct or patient withdrawal prior to completion of the trial; | ||
| • | there may be competition with ongoing clinical trials and scheduling conflicts with participating clinicians; | ||
| • | patients experience serious adverse events, including adverse side effects of our drug candidates, for a variety of reasons that may or may not be related to our product candidates, including the advanced stage of their disease and other medical problems; | ||
| • | patients in the placebo or untreated control group exhibit greater than expected improvements or fewer than expected adverse events; | ||
| • | third-party clinical investigators do not perform the clinical trials on the anticipated schedule or consistent with the clinical trial protocol and good clinical practices, or other third-party organizations do not perform data collection and analysis in a timely or accurate manner; | ||
| • | service providers, collaborators or co-sponsors do not adequately perform their obligations in relation to the clinical trial or cause the trial to be delayed or terminated; | ||
| • | we are unable to obtain a sufficient supply of manufactured clinical trial materials; | ||
| • | regulatory inspections of manufacturing facilities, which may, among other things, require us or a co-sponsor to undertake corrective action or suspend the clinical trials; | ||
| • | the interim results of the clinical trial are inconclusive or negative; |
11
| • | the clinical trial, although approved and completed, generates data that is not considered by the FDA or others to be sufficient to demonstrate safety and efficacy; and | ||
| • | changes in governmental regulations or administrative actions affect the conduct of the clinical trial or the interpretation of its results. |
| • | we do not achieve our objectives under our collaboration agreements; | ||
| • | we or our collaborators are unable to obtain patent protection for the products or proprietary technologies we develop in our collaborations; | ||
| • | we are unable to manage multiple simultaneous product development collaborations; | ||
| • | our collaborators become competitors of ours or enter into agreements with our competitors; |
12
| • | we or our collaborators encounter regulatory hurdles that prevent commercialization of our product candidates; or | ||
| • | we develop products and processes or enter into additional collaborations that conflict with the business objectives of our other collaborators. |
13
| • | the timing and receipt of marketing approvals; | ||
| • | the safety and efficacy of the products; | ||
| • | the emergence of equivalent or superior products; | ||
| • | the cost-effectiveness of the products; and | ||
| • | ineffective marketing. |
| • | changes and limits in import and export controls; | ||
| • | increases in custom duties and tariffs; | ||
| • | changes in currency exchange rates; | ||
| • | economic and political instability; | ||
| • | changes in government regulations and laws; | ||
| • | absence in some jurisdictions of effective laws to protect our intellectual property rights; and | ||
| • | currency transfer and other restrictions and regulations that may limit our ability to sell certain product candidates or repatriate profits to the United States. |
14
| • | their respective abilities to ensure quality and compliance with regulations relating to the manufacture of pharmaceuticals; | ||
| • | their manufacturing capacity may not be sufficient or available to produce the required quantities of our product candidates based on our planned clinical development schedule, if at all; | ||
| • | they may not have access to the capital necessary to expand their manufacturing facilities in response to our needs; | ||
| • | commissioning replacement suppliers would be difficult and time-consuming; | ||
| • | individual suppliers may have used substantial proprietary know-how relating to the manufacture of our product candidates and, in the event we must find a replacement or supplemental supplier, our ability to transfer this know-how to the new supplier could be an expensive and/or time-consuming process; | ||
| • | an individual supplier may experience events, such as a fire or natural disaster, that force it to stop or curtail production for an extended period; | ||
| • | an individual supplier could encounter significant increases in labor, capital or other costs that would make it difficult for them to produce our products cost-effectively; or | ||
| • | an individual supplier may not be able to obtain the raw materials or validated drug containers in sufficient quantities, at acceptable costs or in sufficient time to complete the manufacture, formulation and delivery of our product candidates. |
15
16
17
| • | the recent delisting of our common stock from the NYSE Amex exchange; | ||
| • | results of pre-clinical studies and clinical trials; | ||
| • | commercial success of approved products; | ||
| • | corporate partnerships; | ||
| • | technological innovations by us or competitors; |
18
| • | changes in laws and government regulations both in the U.S. and overseas; | ||
| • | changes in key personnel at our company; | ||
| • | developments concerning proprietary rights, including patents and litigation matters; | ||
| • | public perception relating to the commercial value or safety of any of our product candidates; | ||
| • | future sales of our common stock, including to Lincoln Park under the Purchase Agreement; | ||
| • | other issuances of our common stock causing dilution; | ||
| • | anticipated or unanticipated changes in our financial performance; | ||
| • | general trends related to the biopharmaceutical and biotechnological industries; and | ||
| • | general conditions in the stock market. |
19
20
21
| • | our anticipated cash needs and our estimates regarding our capital requirements and needs for additional financing; | ||
| • | the progress, outcome, timing or success of preclinical studies and clinical trials; | ||
| • | the expected timing of clinical trials and availability of data from those trials; | ||
| • | our ability to obtain and maintain regulatory approval for our product candidates from the FDA or foreign regulatory authorities; | ||
| • | future demand for our product candidates and our ability to sustain such demand; | ||
| • | the size of the potential market for our product candidates; | ||
| • | our plans to seek collaborative relationships and the success of those relationships; | ||
| • | the success of competing therapies that are or become available; | ||
| • | our compliance with federal, state and foreign regulatory requirements, and regulatory developments that impact those requirements; | ||
| • | our estimates and assumptions with respect to disease incidence; | ||
| • | our intellectual property and our strategies regarding filing additional patent applications to attempt to strengthen our intellectual property rights; | ||
| • | our ability to retain key management and scientific personnel; | ||
| • | estimates of our future financial performance; | ||
| • | our ability to implement financial controls and procedures on a timely basis; and | ||
| • | anticipated trends and challenges in our business. |
22
23
| Cash and cash equivalents | $ | 4,975,947 | ||
| Stockholders’ equity: | ||||
| Preferred stock, $0.001 per share, 1,000,000 shares authorized, no shares issued or outstanding | — | |||
| Common stock, $0.001 par value, 200,000,000 shares authorized, 73,531,578 shares issued and outstanding | 73,531 | |||
| Additional paid-in-capital | 92,997,669 | |||
| Accumulated deficit | (88,605,400 | ) | ||
| Total stockholders’ equity | 4,465,800 | |||
| Total capitalization | $ | 4.465,800 | ||
| • | 5,348,863 shares of our common stock issuable upon the exercise of outstanding stock options, with a weighted average exercise price of $1.37 per share; | |
| • | 4,327,500 shares of our common stock available for future issuance under our 2010 Equity Incentive Plan; | |
| • | 13,988,751 shares of our common stock issuable upon the exercise of outstanding warrants, with a weighted-average exercise price of $1.38 per share; and | |
| • | shares of our common stock issued to Lincoln Park and Sigma-Tau in January 2011. |
24
| 2009 | High | Low | ||||||
| First Quarter | $ | 1.75 | $ | 0.42 | ||||
| Second Quarter | $ | 0.85 | $ | 0.45 | ||||
| Third Quarter | $ | 1.12 | $ | 0.52 | ||||
| Fourth Quarter | $ | 0.83 | $ | 0.55 | ||||
| 2010 | High | Low | ||||||
| First Quarter | $ | 0.65 | $ | 0.53 | ||||
| Second Quarter | $ | 0.68 | $ | 0.26 | ||||
| Third Quarter | $ | 0.35 | $ | 0.24 | ||||
| Fourth Quarter | $ | 0.30 | $ | 0.21 | ||||
| 2011 | High | Low | ||||||
| First Quarter (through February 4, 2011) | $ | 0.27 | $ | 0.20 | ||||
25
| Assumed public offering price per share of common stock (minimum allowed price) | $ | 0.15 | ||||||
| As adjusted net tangible book value per share as of September 30, 2010 | $ | 0.07 | ||||||
| Increase in as adjusted net tangible book value per share attributable to this offering | 0.02 | |||||||
| Pro forma net tangible book value per share after this offering | 0.09 | |||||||
| Dilution per share to new investors | $ | 0.06 | ||||||
| • | 5,348,863 shares of our common stock issuable upon the exercise of outstanding stock options, with a weighted average exercise price of $1.37 per share; | ||
| • | 4,327,500 shares of our common stock available for future issuance under our 2010 Equity Incentive Plan; and | ||
| • | 13,988,751 shares of our common stock issuable upon the exercise of outstanding warrants, with a weighted-average exercise price of $1.38 per share. |
26
| Nine Months Ended | ||||||||||||||||||||||||||||
| Year Ended December 31, | September 30, | |||||||||||||||||||||||||||
| 2009 | 2008 | 2007 | 2006 | 2005 | 2010 | 2009 | ||||||||||||||||||||||
Statement of Operations Data: | ||||||||||||||||||||||||||||
| Sponsored research revenue | $ | — | $ | 168,412 | $ | 240,324 | $ | 272,491 | $ | — | $ | 53,819 | $ | — | ||||||||||||||
| Operating expenses: | ||||||||||||||||||||||||||||
| Research and development | 3,724,514 | 7,149,808 | 8,887,255 | 6,396,524 | 3,155,735 | 1,819,036 | 3,064,248 | |||||||||||||||||||||
| General and administrative | 2,781,790 | 3,805,346 | 3,197,685 | 2,665,652 | 2,513,792 | 2,345,619 | 2,161,539 | |||||||||||||||||||||
| Total operating expenses | 6,506,304 | 10,955,154 | 12,084,940 | 9,062,176 | 5,669,527 | 4,164,655 | 5,225,787 | |||||||||||||||||||||
| Loss from operations | (6,506,304 | ) | (10,786,742 | ) | (11,844,616 | ) | (8,789,685 | ) | (5,669,527 | ) | (4,110,836 | ) | (5,225,787 | ) | ||||||||||||||
| Interest income | 12,444 | 149,777 | 666,458 | 522,704 | 214,676 | 6,840 | 10,304 | |||||||||||||||||||||
| Net loss | $ | (6,493,860 | ) | $ | (10,636,965 | ) | $ | (11,178,158 | ) | $ | (8,266,981 | ) | $ | (5,454,851 | ) | $ | (4,103,996 | ) | (5,215,483 | ) | ||||||||
| Basic and diluted net loss per share | $ | (0.12 | ) | $ | (0.21 | ) | $ | (0.24 | ) | $ | (0.21 | ) | $ | (0.15 | ) | $ | (0.06 | ) | (0.10 | ) | ||||||||
| Shares used to compute basic and diluted net loss per share | 55,680,525 | 50,967,617 | 46,465,982 | 40,116,367 | 36,843,609 | 66,729,519 | 54,216,430 | |||||||||||||||||||||
| As of | ||||||||||||||||||||||||
| As of December 31, | September 30, | |||||||||||||||||||||||
| 2009 | 2008 | 2007 | 2006 | 2005 | 2010 | |||||||||||||||||||
Balance Sheet Data: | ||||||||||||||||||||||||
| Cash and cash equivalents | $ | 4,355,768 | $ | 5,655,367 | $ | 3,696,878 | $ | 13,052,308 | $ | 4,896,143 | $ | 4,975,947 | ||||||||||||
| Short-term investments | — | — | 4,579,592 | 4,000,000 | 2,679,693 | — | ||||||||||||||||||
| Working capital | 3,671,910 | 4,565,932 | 6,102,596 | 16,187,188 | 6,939,195 | 4,423,905 | ||||||||||||||||||
| Total assets | 4,583,754 | 5,922,576 | 8,621,793 | 17,501,625 | 7,724,634 | 5,615,670 | ||||||||||||||||||
| Total liabilities | 880,404 | 1,325,912 | 2,469,069 | 1,249,290 | 714,127 | 1,149,870 | ||||||||||||||||||
| Accumulated deficit | (84,501,404 | ) | (78,007,544 | ) | (67,405,579 | ) | (56,227,421 | ) | (47,960,440 | ) | (88,605,400 | ) | ||||||||||||
| Stockholders’ equity | 3,703,350 | 4,596,664 | 6,152,724 | 16,252,335 | 7,010,507 | 4,465,800 | ||||||||||||||||||
27
FINANCIAL CONDITION AND RESULTS OF OPERATIONS
| • | RGN-352, an injectable product candidate to treat cardiovascular diseases, central nervous system diseases, and other medical indications that may be treated by systemic administration; | ||
| • | RGN-259, a topical eye drop for regeneration of corneal tissues damaged by injury, disease or other pathology; and | ||
| • | RGN-137, a topically applied gel for dermal wounds and reduction of scar tissue. |
28
29
30
31
32
| • | the progress of our clinical trials; | ||
| • | the progress of our research activities; | ||
| • | the number and scope of our research programs; | ||
| • | the progress of our preclinical development activities; | ||
| • | the costs involved in preparing, filing, prosecuting, maintaining, enforcing and defending patent and other intellectual property claims; | ||
| • | the costs related to development and manufacture of preclinical, clinical and validation lots for regulatory purposes and commercialization of drug supply associated with our product candidates; | ||
| • | our ability to enter into corporate collaborations and the terms and success of these collaborations; | ||
| • | the costs and timing of regulatory approvals; and | ||
| • | the costs of establishing manufacturing, sales and distribution capabilities. |
| • | the number of patients that ultimately participate in the trial; | ||
| • | the duration of patient follow-up that seems appropriate in view of the results; | ||
| • | the number of clinical sites included in the trials; and | ||
| • | the length of time required to enroll suitable patient subjects. |
33
34
35
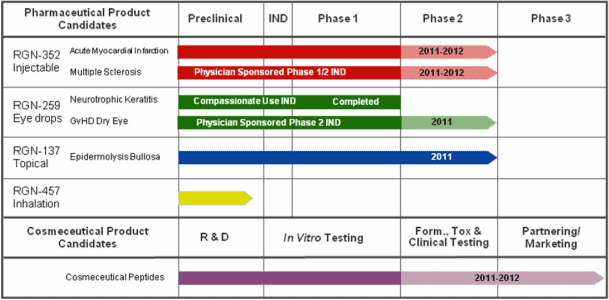
| • | RGN-352, an injectable product candidate to treat cardiovascular diseases, central nervous system diseases, and other medical indications that may be treated by systemic administration, for which we initiated a Phase 2 clinical trial in the second half of 2010; | ||
| • | RGN-259, a topical eye drop for ophthalmic indications for which we are supporting a physician-sponsored clinical trial in patients with dry eye secondary to graft versus host disease, or GvHD; and | ||
| • | RGN-137, a topically applied gel for chronic dermal wounds and reduction of scar tissue that is currently in a Phase 2 clinical trial for the treatment of the skin defect epidermolysis bullosa, or EB. |
| • | Progenitor (Stem) Cell Differentiation.Research published in the journalNaturein November 2006 featured the discovery that Tß4 is the key signaling molecule that triggers adult epicardial progenitor cells, or EPCs, to differentiate into coronary blood vessels. EPCs are partially differentiated stem cells that can further differentiate into specific cell types when needed. Confirmatory research published in 2009 in theJournal of Molecular and Cellular Cardiologyconcluded that Tß4 is responsible for the initiation of the embryonic coronary developmental program and EPC differentiation in adult mice. These publications confirm that Tß4’s interaction with EPCs is necessary for the maintenance of a healthy adult animal heart, as well as normal fetal animal heart development. |
36
| • | Actin Regulation.Tß4 regulates actin, which comprises up to 10% of the protein of non-muscle cells in the body and plays a central role in cell structure and in the movement of cells. Research studies have indicated that Tß4 stimulates the migration of human keratinocytes, or skin cells, human endothelial cells, and progenitor cells. Endothelial cells are the major cell type responsible for the formation of new blood vessels, a process known as angiogenesis. Certain of these studies conducted at the NIH were the first to suggest the role of Tß4 in wound healing. The data from these studies encouraged us to license the rights to Tß4 from the NIH in 2001 and to launch an initial clinical development program that targeted the use Tß4 for chronic dermal wounds. | ||
| • | Reduction of Inflammation.Uncontrolled inflammation is the underlying basis of many pathologies and injuries. Research has shown that Tß4 is a potent anti-inflammatory agent in skin cells and in corneal epithelial cells in the eye. Tß4 has also been shown to decrease the levels of inflammatory mediators and to significantly reduce the influx of inflammatory cells in the reperfused heart of animals. More recent preclinical research suggests that Tß4 blocks activation of the NFкB pathway, which is involved in DNA activation of inflammatory mediators, thereby modulating inflammation in the body. This anti-inflammatory activity may explain, in part, the mechanism by which Tß4 appeared to improve functional outcome in the mouse multiple sclerosis model described above, as well as promoting repair in the heart and skin. Identifying a factor such as Tß4 that blocks activation of NFкB suggests that Tß4 could have additional important therapeutic applications for inflammation-related diseases, such as cancer, osteoarthritis, rheumatic diseases, autoimmune diseases, inflammatory pulmonary disease and pancreatitis. | ||
| • | Collagen and Laminin-5 Stimulation.Tß4 has a number of additional biological activities shown to reduce inflammation, stimulate the formation of collagen, and up-regulate the expression of laminin-5, a subepithelial basement membrane protein. Both collagen and laminin-5 are central to healthy tissue and the prevention of disease. | ||
| • | Apoptosis.Tß4 has been shown to prevent apoptosis, or programmed cell death, in two animal models and in two tissue types. In the rodent model, corneal apoptosis, or loss of corneal epithelial cells leading to corneal epithelial thinning, was prevented through topical administration of Tß4, and in the heart muscle of ischemic animal models, such as in mice and pigs, cell death was prevented by the systemic administration of Tß4. |
37
38
39
40
| • | RGN-352.Currently, there are no approved pharmaceutical products for regenerating cardiac tissue following a heart attack, nor are there approved pharmaceutical products for the remyelination of axons for patients with multiple sclerosis. However, many pharmaceutical companies and research organizations are developing products and technologies that are intended to prevent cardiac damage, improve cardiac function, and regenerate cardiac muscle after a heart attack. There are also companies developing products that remyelinate neurons and provide functional improvement for multiple sclerosis patients. If we were to successfully develop RGN-352 for other cardiovascular indications, such as acute or chronic heart failure, such a product would have to compete with other drugs or therapies currently marketed by large pharmaceutical companies for similar indications, as would products for the treatment of multiple sclerosis. | ||
| • | RGN-259.Most specialty ophthalmic companies have a number of products on the market that could compete with RGN-259. There are numerous antibiotics to treat eye infections that cause corneal wounds and many eye lubrication products to help eye healing and function, many of which are sold without prescriptions. Companies also market steroids to treat certain severe conditions within our area of interest. Allergan, Inc. has marketed Restasis TM , a relatively new approved eye drop to treat dry eye. Dry eye is a condition related to a number of diseases and one that we believe could benefit from the use of RGN-259. | ||
| • | RGN-137.Johnson & Johnson has marketed RegranexTM for patients with diabetic foot ulcers. Companies such as Novartis are developing and marketing artificial skins, which would compete with RGN-137 in the treatment of dermal wound healing. There are other companies developing new pharmaceutical products for wound healing. Products and therapies such as antibiotics, honey-based ointments and low frequency cavitational ultrasound are also used to treat certain types of dermal wounds. Moreover, dermal wound healing is a large and highly fragmented marketplace that includes numerous therapeutic products and medical devices for treating acute and chronic dermal wounds. | ||
| • | RGN-457.CF is a genetic defect for which there is no cure. There are mucolytic agents and antibiotic drugs on the market, such as Genentech’s pulmozyme and Novartis’ TOBI® , an inhaled version of tobramycin, that relieve the symptoms posed by CF and could potentially compete with RGN-457. | ||
| • | Cosmeceuticals.The cosmetics industry is highly competitive and dependent on effective marketing and distribution. There are multiple products currently launched by major international cosmetic enterprises that claim the same or similar benefits that may be claimed with our product candidates. |
41
42
43
44
| Name | Age | Position | ||||
| J.J. Finkelstein | 58 | President, Chief Executive Officer and Director | ||||
| C. Neil Lyons | 53 | Chief Financial Officer | ||||
| David R. Crockford | 65 | Vice President, Clinical and Regulatory Affairs | ||||
| Allan L. Goldstein, Ph.D. | 73 | Director, Chairman of the Board and Chief Scientific Advisor | ||||
| R. Don Elsey | 57 | Director | ||||
| Joseph C. McNay | 77 | Director | ||||
| Mauro Bove | 56 | Director | ||||
| L. Thompson Bowles, M.D., Ph.D. | 79 | Director | ||||
45
46
47
| • | establishment of corporate and individual performance objectives relevant to the compensation of our chief executive officer, other executive officers and Board members; | ||
| • | evaluation of performance in light of these stated objectives; | ||
| • | review and approval of the compensation and other terms of employment or service, including severance and change-in-control arrangements, of our Chief Executive Officer and the other executive officers; and | ||
| • | administration of our equity compensation plans and other similar plan and programs. |
48
| Fees Earned | ||||||||||||||||
| or Paid | Option | All Other | ||||||||||||||
| in Cash | Awards | Compensation | Total | |||||||||||||
| Name | ($) | ($)(1) | ($) | ($) | ||||||||||||
| Allan Goldstein, Ph.D. | — | 15,284 | 187,460 | (2) | 202,744 | |||||||||||
| R. Don Elsey | 7,726 | 6,403 | — | 14,129 | ||||||||||||
| L. Thompson Bowles M.D., Ph.D. | 28,017 | 3,103 | — | 31,120 | ||||||||||||
| Joseph McNay | 25,028 | 3,103 | — | 28,131 | ||||||||||||
| Mauro Bove | 17,303 | 3,103 | — | 20,406 | ||||||||||||
| Richard Hindin (3) | 21,836 | — | — | 21,836 | ||||||||||||
| (1) | These amounts reflect the aggregate full grant date fair values (computed in accordance with FASB ASC Topic 718) of options granted to directors during 2010. Options held by each Board member as of December 31, 2010, are as follows: |
| Allan Goldstein, Ph.D. | 795,442 | |||
| R. Don Elsey | 40,000 | |||
| L. Thompson Bowles M.D., Ph.D. | 174,843 | |||
| Joseph McNay | 248,024 | |||
| Mauro Bove | 247,155 |
| (2) | In addition to being Chairman of our Board of Directors, Dr. Goldstein also serves as our Chief Scientific Advisor. In this capacity, Dr. Goldstein received a base salary of $187,460 for 2010. Under Dr. Goldstein’s employment agreement, in the event that his employment is terminated by us without “cause,” as defined in his employment agreement, or if he voluntarily terminates his employment within 12 months following a “change in control,” as defined in his employment agreement, then in each case, subject to Dr. Goldstein’s entering into and not revoking a release of claims in a form acceptable to us, Dr. Goldstein will be entitled to receive a lump sum severance payment equal to his annual base salary then in effect, plus any earned bonus as of the date of termination, in each case less applicable taxes and withholdings. Dr. Goldstein is not entitled to receive any continuing health and welfare benefits as part of our severance obligation to him. If Dr. Goldstein’s employment had been terminated for any of the reasons described in this paragraph as of December 31, 2010, he would have been entitled to receive a lump sum payment of $187,460, less taxes and withholdings. Dr. Goldstein is eligible to receive options to purchase common stock under our equity incentive plans. The decision to grant any such options and the terms of such options are within the discretion of our board of directors or the compensation committee. In addition, if Dr. Goldstein’s employment is terminated without “cause,” or if there is a “change in control” event, in each case as defined in either the applicable benefit plan or in Dr. Goldstein’s employment agreement, then the unvested portion of Dr. Goldstein’s options would accelerate in full. All vested options are exercisable for a period of time following any termination of Dr. Goldstein’s employment as may be set forth in the applicable benefit plan or in any option agreement between Dr. Goldstein and us. | |
| (3) | Mr. Hindin’s term as a director ended in July 2010. |
49
| Non-Equity | ||||||||||||||||||||||||||||
| Incentive | ||||||||||||||||||||||||||||
| Option | Plan | All Other | ||||||||||||||||||||||||||
| Salary(1) | Bonus(2) | Awards(3) | Compensation(4) | Compensation(5) | Total | |||||||||||||||||||||||
| Name and Principal Position | Year | ($) | ($) | ($) | ($) | ($) | ($) | |||||||||||||||||||||
| J.J. Finkelstein, President and | 2010 | 299,520 | — | 19,396 | — | 18,425 | 337,341 | |||||||||||||||||||||
| Chief Executive Officer | 2009 | 244,608 | 18,720 | 116,198 | — | 13,005 | 392,531 | |||||||||||||||||||||
| C. Neil Lyons, | 2010 | 202,537 | 2,000 | 15,284 | — | 6,886 | 226,707 | |||||||||||||||||||||
| Chief Financial Officer | 2009 | 167,093 | 11,140 | 74,395 | — | 4,999 | 257,627 | |||||||||||||||||||||
| David R. Crockford, | 2010 | 210,223 | 2,000 | 15,284 | — | 10,321 | 237,828 | |||||||||||||||||||||
| Vice President, Clinical and Regulatory Affairs | 2009 | 210,223 | 5,781 | — | — | 6,818 | 222,822 | |||||||||||||||||||||
| (1) | Reflects base salary before pretax contributions and therefore includes compensation deferred under our 401(k) plan. | |
| (2) | Reflects the discretionary portion of our bonus plan. | |
| (3) | These amounts reflect the aggregate full grant date fair values (computed in accordance with FASB ASC Topic 718) of options granted to executives during the respective fiscal years. | |
| (4) | Reflects amounts earned under our bonus plan subject to the achievement of corporate performance goals. | |
| (5) | Primarily reflects our match of executive compensation deferrals into our 401(k) plan, along with supplemental life and disability insurance premiums. None of the individual items exceeded $10,000. |
50
51
| Number of | ||||||||||||||||||||
| Shares | Number of Shares | |||||||||||||||||||
| Underlying | Underlying | |||||||||||||||||||
| Unexercised | Unexercised | Option | ||||||||||||||||||
| Options (#) | Options (#) | Exercise Price | Option | |||||||||||||||||
| Name | Exercisable | Unexercisable | ($) | Expiration Date | Note | |||||||||||||||
| Mr. Finkelstein | 500,000 | — | 0.33 | 1/1/2012 | ||||||||||||||||
| 100,000 | — | 3.21 | 4/1/2015 | |||||||||||||||||
| 93,750 | 31,250 | 2.34 | 3/15/2014 | (1 | ) | |||||||||||||||
| 62,500 | 62,500 | 1.15 | 4/15/2015 | (1 | ) | |||||||||||||||
| 114,748 | — | 0.57 | 4/10/2019 | |||||||||||||||||
| 31,250 | 93,750 | 0.76 | 10/11/2016 | (1 | ) | |||||||||||||||
| — | 125,000 | 0.27 | 07/14/2017 | (1 | ) | |||||||||||||||
| Mr. Lyons | 166,667 | 33,333 | 3.10 | 4/7/2015 | (2 | ) | ||||||||||||||
| 56,250 | 18,750 | 2.34 | 3/15/2014 | (1 | ) | |||||||||||||||
| 37,500 | 37,500 | 1.50 | 6/15/2015 | (1 | ) | |||||||||||||||
| 77,728 | — | 0.57 | 4/10/2019 | |||||||||||||||||
| 18,750 | 56,250 | 0.76 | 10/11/2016 | (1 | ) | |||||||||||||||
| — | 98,500 | 0.27 | 07/14/2017 | (1 | ) | |||||||||||||||
| Mr. Crockford | 15,000 | — | 1.07 | 7/1/2013 | ||||||||||||||||
| 125,000 | — | 0.86 | 1/1/2014 | |||||||||||||||||
| 100,000 | — | 3.21 | 4/1/2015 | |||||||||||||||||
| 25,000 | — | 3.82 | 5/25/2015 | |||||||||||||||||
| 37,500 | 12,500 | 2.15 | 1/16/2014 | (1 | ) | |||||||||||||||
| 56,250 | 18,750 | 2.34 | 3/15/2014 | (1 | ) | |||||||||||||||
| 37,500 | 37,500 | 1.15 | 4/15/2015 | (1 | ) | |||||||||||||||
| — | 98,500 | 0.27 | 07/14/2017 | (1 | ) | |||||||||||||||
| (1) | This option vests in equal installments on the first four anniversaries of the grant date. In each case these options were granted seven years prior to the listed expiration dates. | |
| (2) | This option vests in equal installments on the first six anniversaries of the grant date which was April 7, 2005. |
52
| Number of Securities | ||||||||||||
| Remaining Available for | ||||||||||||
| Number of Securities to | Future Issuance Under | |||||||||||
| be Issued Upon Exercise | Weighted-Average Exercise | Equity Compensation Plans | ||||||||||
| of Outstanding Options, | Price of Outstanding Options, | (Excluding Securities | ||||||||||
| Warrants and Rights | Warrants and Rights | Reflected in Column (a)) | ||||||||||
| Plan Category | (a) | (b) | (c) | |||||||||
| Equity compensation plans approved by security holders | 5,348,863 | $ | 1.37 | 4,327,500 | ||||||||
| Equity compensation plans not approved by security holders | — | — | — | |||||||||
| Total | 5,348,863 | $ | 1.37 | 4,327,500 | ||||||||
53
54
| • | the option exercise price is at least 110% of the fair market value of the stock subject to the option on the date of grant; and | ||
| • | the term of the incentive stock option does not exceed five years from the date of grant. |
55
56
| • | The Board will arrange for assumption, continuation, or substitution of a stock award by a surviving or acquiring entity (or its parent company) and assign any reacquisition or repurchase rights held by us in respect of common stock issued pursuant to awards to our successor (or its parent). | ||
| • | With respect to stock awards that have not been assumed, continued or substituted, the vesting of such stock awards will be accelerated in full to a date prior to the effective date of the corporate transaction and any reacquisition or repurchase right held by us in respect of common stock issuable pursuant to such stock awards will lapse, and such stock awards will terminate if not exercised (if applicable) at or prior to the time of the corporate transaction. | ||
| • | With respect to stock awards that have not been assumed, continued or substituted, such outstanding stock awards will terminate if not exercised at or prior to the time of the corporate transaction, and the Board may, in its discretion, make a payment, in such form as the Board may determine, equal in value to the excess, if any, of (a) the value of the property the holder would have received upon the exercise of the stock award, over (b) any exercise price payable by such holder in connection with such exercise. |
57
58
59
60
61
62
| • | any breach of their duty of loyalty to us or our stockholders; | ||
| • | acts or omissions not in good faith or which involve intentional misconduct or a knowing violation of law; | ||
| • | voting or assenting to unlawful payments of dividends or other distributions; or | ||
| • | any transaction from which the director derived an improper personal benefit. |
63
64
| • | each person, or group of affiliated persons, who is known by us to beneficially own more than 5% of our common stock; | ||
| • | each of our named executive officers; | ||
| • | each of our directors; and | ||
| • | all of our executive officers and directors as a group. |
| Number of | Percentage of | |||||||
| Shares | Shares | |||||||
| Beneficially | Beneficially | |||||||
| Name of Beneficial Owner | Owned | Owned | ||||||
5% Stockholders: | ||||||||
| Entities affiliated with Sigma-Tau Finanziaria, S.p.A. Via Sudafrica, 20, Rome, Italy 00144 | 33,997,378 | (1) | 40.5 | % | ||||
Named Executive Officers and Directors: | ||||||||
| J.J. Finkelstein | 2,330,886 | (2) | 2.9 | % | ||||
| Allan L. Goldstein | 1,921,288 | (3) | 2.4 | % | ||||
| R. Don Elsey | — | * | ||||||
| Joseph C. McNay | 1,544,635 | (4) | 1.9 | % | ||||
| Mauro Bove | 204,655 | (5) | * | |||||
| L. Thompson Bowles | 132,343 | (6) | * | |||||
| C. Neil Lyons | 386,893 | (7) | * | |||||
| David R. Crockford | 408,750 | (8) | * | |||||
| All current directors and executive officers as a group (8 persons) | 6,929,450 | (9) | 8.4 | % | ||||
| * | Represents beneficial ownership of less than 1%. |
65
| (1) | Consists of 984,615 shares of common stock held of record held by Sigma-Tau Finanziaria, S.p.A. (“Sigma-Tau”); 12,937,111 shares of common stock held of record and 589,481 shares of common stock issuable upon exercise of warrants held by Defiante Farmaceutica S.A. (“Defiante”), a subsidiary of Sigma-Tau, that are exercisable within 60 days of January 7, 2011; 6,348,878 shares of common stock held of record and 1,228,486 shares of common stock issuable upon exercise of warrants held by Taufin International S.A. (“Taufin”), an entity wholly owned by Taufin SPA, which is owned directly by Claudio Cavazza, who directly and indirectly owns 57% of Sigma-Tau, that are exercisable within 60 days of January 7, 2011; and 9,711,407 shares of common stock held of record and 2,197,400 shares of common stock issuable upon exercise of warrants held by Sinaf S.A. (“Sinaf”), an indirect wholly-owned subsidiary of Aptafin S.p.A., which is owned by Paolo Cavazza and members of his family, that are exercisable within 60 days of January 7, 2011. Paolo Cavazza directly and indirectly owns 38% of Sigma-Tau. | |
| (2) | Consists of 1,377,638 shares of common stock held of record by Mr. Finkelstein and 51,000 shares of common stock held of record by Mr. Finkelstein’s daughter over which Mr. Finkelstein shares voting and dispositive power. Also includes 902,248 shares of common stock issuable upon exercise of options exercisable within 60 days of January 7, 2011. | |
| (3) | Consists of 1,336,846 shares of common stock held of record by Dr. Goldstein and 584,442 shares of common stock issuable upon exercise of options exercisable within 60 days of January 7, 2011. | |
| (4) | Consists of 1,339,111 shares of common stock held of record by Mr. McNay and 205,524 shares of common stock issuable upon exercise of options exercisable within 60 days of January 7, 2011. | |
| (5) | Consists of shares of common stock issuable upon exercise of options exercisable within 60 days of January 7, 2011. Mr. Bove is an officer of Sigma-Tau, but he has no beneficial ownership over the reported securities as he has no voting or dispositive power with respect to the securities held by Sigma-Tau and its affiliates described in Note 1 above. | |
| (6) | Consists of shares of common stock issuable upon exercise of options exercisable within 60 days of January 7, 2011. | |
| (7) | Consists of 30,000 shares of common stock held of record by Mr. Lyons and 356,893 shares of common stock issuable upon exercise of options exercisable within 60 days of January 7, 2011. | |
| (8) | Consists of shares of common stock issuable upon exercise of options exercisable within 60 days of January 7, 2011. | |
| (9) | Consists of 4,134,595 shares of common stock held of record and 2,794,855 shares of common stock issuable upon exercise of options exercisable within 60 days of January 7, 2011. |
66
67
| • | while any registration statement is required to be maintained effective pursuant to the terms of the registration rights agreement between us and Lincoln Park, the effectiveness of such registration statement lapses for any reason (including, without limitation, the issuance of a stop order) or is unavailable for sale of our shares of common stock in accordance with the terms of the registration rights agreement, and such lapse or unavailability continues for a period of twenty consecutive business days or for more than an aggregate of sixty business days in any 365-day period; | ||
| • | the suspension from trading or failure of our common stock to be listed on our principal market for a period of three consecutive business days; | ||
| • | the delisting of our common stock from our principal market, provided our common stock is not immediately thereafter trading on the OTC Bulletin Board, the NASDAQ Global Market, the NASDAQ Global Select Market, the New York Stock Exchange or the NASDAQ Capital Market; | ||
| • | our transfer agent’s failure to issue to Lincoln Park shares of our common stock which Lincoln Park is entitled to receive under the Purchase Agreement within five business days after an applicable purchase date; | ||
| • | any breach by us of the representations or warranties or covenants contained in the Purchase Agreement or any related agreements which could have a material adverse effect on us subject to a cure period of five business days; | ||
| • | if we become insolvent or are generally unable to pay our debts as they become due; or | ||
| • | any participation or threatened participation in insolvency or bankruptcy proceedings by or against us. |
68
| Additional Proceeds from the Sale | |||||||
| of | |||||||
| Percentage of Outstanding | Registered Shares | ||||||
| Assumed Average | Number of Registered | Shares After Giving Effect to | to Lincoln Park Under the | ||||
| Purchase Price | Shares to be Issued if | the | Purchase Agreement | ||||
| ($) | Full Purchase (1)(2) | Issuance to Lincoln Park(3) | ($) | ||||
| $0.15(4) | 15,000,000 | 15.8 | % | 2,220,976 | |||
| $0.22(5) | 15,000,000 | 15.8 | % | 3,237,940 | |||
| $0.50 | 15,000,000 | 15.8 | % | 7,186,933 | |||
| $1.00 | 11,958,333 | 13.0 | % | 11,000,000 | |||
| $1.50 | 8,291,666 | 9.4 | % | 11,000,000 |
| (1) | Although the Purchase Agreement provides that we may sell up to $11,000,000 of our common stock to Lincoln Park, we are only registering 15,000,000 shares to be purchased thereunder, which may or may not cover all such shares purchased by them under the Purchase Agreement, depending on the purchase price per share. As a result, we have included in this column only those shares which are registered in this offering. | |
| (2) | The number of registered shares to be issued includes a number of shares to be purchased at the applicable price plus the applicable number of pro rata additional commitment shares to be issued to Lincoln Park as a result of such purchase (but not the 958,333 initial commitment shares), although no additional proceeds will be attributable to such additional commitment shares. | |
| (3) | The denominator is based on 79,860,282 shares outstanding, and includes the 2,810,185 shares already owned by Lincoln Park not included in this offering, and the number of shares set forth in the adjacent column which includes the commitment fee issued pro rata up to the $11,000,000 of our stock if purchased by Lincoln Park. The numerator is based on the number of shares issuable under the Purchase Agreement at the corresponding assumed purchase price set forth in the adjacent column. | |
| (4) | Under the Purchase Agreement, we may not sell and Lincoln Park cannot purchase any shares in the event the price of our stock is below $0.15 per share. | |
| (5) | The closing price of our common stock on February 4, 2011. | |
69
| Shares to be | ||||||||||||||
| Sold | ||||||||||||||
| in the Offering | ||||||||||||||
| Assuming | ||||||||||||||
| Percentage of | RegeneRx Issues | Percentage of | ||||||||||||
| Shares Beneficially | Outstanding Shares | Maximum No. | Outstanding Shares | |||||||||||
| Owned Before | Beneficially Owned | of Shares | Beneficially Owned | |||||||||||
| Selling Stockholder | Offering | Before Offering | in the Offering | After Offering | ||||||||||
Lincoln Park Capital Fund, LLC(1) | 2,810,185 | 3.5 | % | 15,000,000 | (3) | 3.0 | % | |||||||
| (1) | Josh Scheinfeld and Jonathan Cope, the principals of Lincoln Park, are deemed to be beneficial owners of all of the shares of common stock owned by Lincoln Park. Messrs. Scheinfeld and Cope have shared voting and disposition power over the shares being offered under this prospectus. | |
| (2) | 2,810,185 shares of our common stock are already owned by Lincoln Park, which shares include (i) 958,333 shares of restricted common stock and (ii) 1,851,852 shares acquired by Lincoln Park in connection with the January 2011 registered direct offering of our securities. We may at our discretion elect to issue to Lincoln Park up to an additional 15,000,000 shares of our common stock under the Purchase Agreement, subject to the terms and provisions of such agreement, but Lincoln Park does not beneficially own any such shares that may be issued by us at our sole discretion and such shares are not included in determining the percentage of shares beneficially owned before the offering. | |
| (3) | Although the Purchase Agreement provides what we may sell up to $11,000,000 of our common stock to Lincoln Park, we are only registering 15,000,000 shares issuable under the Purchase Agreement on this registration statement. If we elect to issue more than the 15,000,000 shares offered by this prospectus, which we have the right but not the obligation to do, we must first register under the Securities Act any additional shares we may elect to sell to Lincoln Park before we can sell such additional shares. |
70
| • | options to purchase 5,348,863 shares of our common stock at a weighted average exercise price of $1.37 per share; and | ||
| • | warrants to purchase an aggregate of 16,136,900 shares of our common stock at a weighted average exercise price of $0.89 per share. |
71
| • | prior to the date of the transaction, the board of directors of the corporation approved either the business combination or the transaction that resulted in the stockholder becoming an interested stockholder; | ||
| • | the interested stockholder owned at least 85% of the voting stock of the corporation outstanding at the time the transaction commenced, excluding for this purpose shares owned by persons who are directors and also officers and shares owned by employee stock plans in which employee participants do not have the right to determine confidentially whether shares held subject to the plan will be tendered in a tender or exchange offer; or | ||
| • | on or subsequent to the date of the transaction, the business combination is approved by the board and authorized at an annual or special meeting of stockholders, and not by written consent, by the affirmative vote of at least 66 2 / 3 % of the outstanding voting stock that is not owned by the interested stockholder. |
| • | any merger or consolidation involving the corporation and the interested stockholder; | ||
| • | any sale, transfer, pledge or other disposition involving the interested stockholder of 10% or more of the assets of the corporation; | ||
| • | subject to exceptions, any transaction that results in the issuance or transfer by the corporation of any stock of the corporation to the interested stockholder; | ||
| • | any transaction involving the corporation that has the effect of increasing the proportionate share of the stock or any class or series of the corporation beneficially owned by the interested stockholder; and |
72
| • | the receipt by the interested stockholder of the benefit of any loans, advances, guarantees, pledges or other financial benefits provided by or through the corporation. |
| • | permit our board of directors to issue up to 1,000,000 shares of preferred stock, with any rights, preferences and privileges as they may designate, including the right to approve an acquisition or other change in our control; | ||
| • | provide that the authorized number of directors, which may not be less than three nor more than seven, may be changed only by resolution of the board of directors; | ||
| • | provide that stockholders seeking to nominate candidates for election as directors at a meeting of stockholders must provide notice in writing in a timely manner, and also specify requirements as to the form and content of a stockholder’s notice; | ||
| • | do not provide for cumulative voting rights, therefore allowing the holders of a majority of the shares of common stock entitled to vote in any election of directors to elect all of the directors standing for election, if they should so choose; and | ||
| • | provide that special meetings of our stockholders may be called only by the chairman of the board, our president or by the board of directors. |
73
| • | ordinary brokers’ transactions; | ||
| • | transactions involving cross or block trades; | ||
| • | through brokers, dealers, or underwriters who may act solely as agents; | ||
| • | “at the market” into an existing market for the common stock; | ||
| • | in other ways not involving market makers or established business markets, including direct sales to purchasers or sales effected through agents; | ||
| • | in privately negotiated transactions; or | ||
| • | any combination of the foregoing. |
74
SECURITIES ACT LIABILITY
75
| Page | ||||
| F-2 | ||||
| F-3 | ||||
| F-4 | ||||
| F-5 | ||||
| F-6 | ||||
| F-7 | ||||
| F-18 | ||||
| F-19 | ||||
| F-20 | ||||
| F-21 | ||||
F-1
RegeneRx Biopharmaceuticals, Inc.
March 31, 2010
F-2
| December 31, | December 31, | |||||||
| 2009 | 2008 | |||||||
| ASSETS | ||||||||
| Current assets | ||||||||
| Cash and cash equivalents | $ | 4,355,768 | $ | 5,655,367 | ||||
| Prepaid expenses and other current assets | 196,546 | 236,477 | ||||||
| Total current assets | 4,552,314 | 5,891,844 | ||||||
| Property and equipment, net of accumulated depreciation of $98,171 and $81,623 | 8,492 | 25,039 | ||||||
| Other assets | 22,948 | 5,693 | ||||||
| Total assets | $ | 4,583,754 | $ | 5,922,576 | ||||
| LIABILITIES AND STOCKHOLDERS’ EQUITY | ||||||||
| Current liabilities | ||||||||
| Accounts payable | $ | 140,206 | $ | 70,554 | ||||
| Accrued expenses | 740,198 | 1,255,358 | ||||||
| Total current liabilities | 880,404 | 1,325,912 | ||||||
| Commitments | — | — | ||||||
| Stockholders’ equity | ||||||||
| Preferred stock, $.001 par value per share, 1,000,000 shares authorized; no shares issued | — | — | ||||||
| Common stock, $.001 par value per share, 100,000,000 shares authorized; 60,406,828 and 53,622,491 issued and outstanding | 60,407 | 53,623 | ||||||
| Additional paid-in capital | 88,144,347 | 82,550,585 | ||||||
| Accumulated deficit | (84,501,404 | ) | (78,007,544 | ) | ||||
| Total stockholders’ equity | 3,703,350 | 4,596,664 | ||||||
| Total liabilities and stockholders’ equity | $ | 4,583,754 | $ | 5,922,576 | ||||
F-3
| Years Ended December 31, | ||||||||
| 2009 | 2008 | |||||||
| Sponsored research revenue | $ | — | $ | 168,412 | ||||
| Operating expenses | ||||||||
| Research and development | 3,724,514 | 7,149,808 | ||||||
| General and administrative | 2,781,790 | 3,805,346 | ||||||
| Total operating expenses | 6,506,304 | 10,955,154 | ||||||
| Loss from operations | (6,506,304 | ) | (10,786,742 | ) | ||||
| Interest income | 12,444 | 149,777 | ||||||
| Net loss | $ | (6,493,860 | ) | $ | (10,636,965 | ) | ||
| Basic and diluted net loss per common share | $ | (0.12 | ) | $ | (0.21 | ) | ||
| Weighted average number of common shares outstanding | 55,680,525 | 50,967,617 | ||||||
F-4
| Accumulated | ||||||||||||||||||||||||
| Other | Total | |||||||||||||||||||||||
| Common Stock | Additional | Accumulated | Comprehensive | Stockholders’ | ||||||||||||||||||||
| Shares | Amount | Paid-in Capital | Deficit | Income/(loss) | Equity | |||||||||||||||||||
| Balance, December 31, 2007 | 46,553,527 | $ | 46,554 | $ | 73,513,292 | $ | (67,405,579 | ) | $ | (1,543 | ) | $ | 6,152,724 | |||||||||||
| Cumulative effect of a change in accounting principle — ASC Topic 730 | — | — | — | 35,000 | — | 35,000 | ||||||||||||||||||
| Issuance of common stock, net of offering costs of $52,240 | 7,068,964 | 7,069 | 7,940,691 | — | — | 7,947,760 | ||||||||||||||||||
| Share-based compensation expense | — | — | 1,096,602 | — | — | 1,096,602 | ||||||||||||||||||
| Net loss | — | — | — | (10,636,965 | ) | — | (10,636,965 | ) | ||||||||||||||||
| Unrealized gain on available for sale securities | — | — | — | — | 1,543 | 1,543 | ||||||||||||||||||
| Total comprehensive loss | (10,635,422 | ) | ||||||||||||||||||||||
| Balance, December 31, 2008 | 53,622,491 | 53,623 | 82,550,585 | (78,007,544 | ) | $ | — | $ | 4,596,664 | |||||||||||||||
| Issuance of common stock, net of offering costs of $447,933 | 6,784,337 | 6,784 | 4,845,282 | — | — | 4,852,066 | ||||||||||||||||||
| Share-based compensation expense | — | — | 748,480 | — | — | 748,480 | ||||||||||||||||||
| Net loss | — | — | — | (6,493,860 | ) | — | (6,493,860 | ) | ||||||||||||||||
| Balance, December 31, 2009 | 60,406,828 | $ | 60,407 | $ | 88,144,347 | $ | (84,501,404 | ) | $ | — | $ | 3,703,350 | ||||||||||||
F-5
| Years Ended December 31, | ||||||||
| 2009 | 2008 | |||||||
| Operating activities: | ||||||||
| Net loss | $ | (6,493,860 | ) | $ | (10,636,965 | ) | ||
| Adjustments to reconcile net loss to net cash used in operating activities: | ||||||||
| Depreciation and amortization | 16,547 | 19,396 | ||||||
| Non-cash share-based compensation | 748,480 | 1,096,602 | ||||||
| Gain on settlement of accrued expenses | (100,000 | ) | — | |||||
| Changes in operating assets and liabilities: | ||||||||
| Accounts receivable | — | 26,951 | ||||||
| Prepaid expenses and other current assets | 39,931 | 66,767 | ||||||
| Other assets | (17,255 | ) | — | |||||
| Accounts payable | 69,652 | (203,007 | ) | |||||
| Accrued expenses | (415,160 | ) | (940,150 | ) | ||||
| Net cash used in operating activities | (6,151,665 | ) | (10,570,406 | ) | ||||
| Investing activities: | ||||||||
| Sales/maturities of short-term investments | — | 4,581,135 | ||||||
| Net cash provided by investing activities | — | 4,581,135 | ||||||
| Financing activities: | ||||||||
| Net proceeds from issuance of common stock | 4,852,066 | 7,947,760 | ||||||
| Net cash provided by financing activities | 4,852,066 | 7,947,760 | ||||||
| Net (decrease) increase in cash and cash equivalents | (1,299,599 | ) | 1,958,489 | |||||
| Cash and cash equivalents at beginning of year | 5,655,367 | 3,696,878 | ||||||
| Cash and cash equivalents at end of year | $ | 4,355,768 | $ | 5,655,367 | ||||
F-6
F-7
F-8
F-9
| • | Level 1 — Quoted prices in active markets for identical assets and liabilities. | ||
| • | Level 2 — Observable inputs other than quoted prices in active markets for identical assets and liabilities. | ||
| • | Level 3 — Unobservable inputs. |
F-10
| December 31, | ||||||||
| 2009 | 2008 | |||||||
| Accrued clinical research | $ | 496,997 | $ | 944,283 | ||||
| Accrued professional fees | 122,590 | 155,000 | ||||||
| Accrued vacation | 35,300 | 61,714 | ||||||
| Accrued license fees | 30,000 | — | ||||||
| Accrued compensation | 28,995 | 84,361 | ||||||
| Other | 26,316 | 10,000 | ||||||
| $ | 740,198 | $ | 1,255,358 | |||||
F-11
F-12
| December 31, | ||||||||
| 2009 | 2008 | |||||||
| Research and development | $ | 369,814 | $ | 440,850 | ||||
| General and administrative | 378,666 | 655,752 | ||||||
| $ | 748,480 | $ | 1,096,602 | |||||
F-13
| Options Outstanding | ||||||||||||||||
| Weighted | ||||||||||||||||
| Shares | Average | |||||||||||||||
| Available for | Number of | Exercise Price | Exercise | |||||||||||||
| Grant | Shares | Range | Price | |||||||||||||
| December 31, 2007 | 620,000 | 3,545,000 | $ | 0.28 - $3.82 | $ | 1.80 | ||||||||||
| Grants | (572,500 | ) | 572,500 | 1.14 - 1.50 | 1.23 | |||||||||||
| Exercises | — | — | — | — | ||||||||||||
| Cancellations | — | — | — | — | ||||||||||||
| Newly authorized | 2,300,000 | — | — | — | ||||||||||||
| December 31, 2008 | 2,347,500 | 4,117,500 | 0.28 - 3.82 | 1.72 | ||||||||||||
| Grants | (1,192,939 | ) | 1,192,939 | 0.57 - 0.76 | 0.64 | |||||||||||
| Exercises | — | — | — | — | ||||||||||||
| Cancellations | 396,327 | (396,327 | ) | 0.57 - 2.59 | 0.82 | |||||||||||
| December 31, 2009 | 1,550,888 | 4,914,112 | $ | 0.28 - $3.82 | $ | 1.53 | ||||||||||
| Outstanding Options | Exercisable Options | |||||||||||||||||||||||
| Weighted- | Weighted- | |||||||||||||||||||||||
| Average | Weighted- | Average | Weighted- | |||||||||||||||||||||
| Number of | Remaining | Average | Number of | Remaining | Average | |||||||||||||||||||
| Shares | Contractual | Exercise | Shares | Contractual | Exercise | |||||||||||||||||||
| Range of Exercise Prices | Outstanding | Life (in Years) | Price | Exercisable | Life (in Years) | Price | ||||||||||||||||||
| $0.28-$0.86 | 2,151,612 | 4.5 | $ | 0.50 | 1,724,112 | 4.0 | $ | 0.43 | ||||||||||||||||
| $1.07-$1.93 | 827,500 | 5.0 | $ | 1.31 | 435,625 | 4.6 | $ | 1.40 | ||||||||||||||||
| $2.02-$2.68 | 860,000 | 4.3 | $ | 2.26 | 323,750 | 4.4 | $ | 2.31 | ||||||||||||||||
| $3.00-$3.82 | 1,075,000 | 5.4 | $ | 3.19 | 950,832 | 5.4 | $ | 3.19 | ||||||||||||||||
| 4,914,112 | 3,434,319 | |||||||||||||||||||||||
| Intrinsic value of in-the-money options, using the December 31, 2009 closing price of $0.55 | $ | 254,450 | $ | 254,450 | ||||||||||||||||||||
F-14
| 2009 | 2008 | |||||||
| Dividend yield | 0.0 | % | 0.0 | % | ||||
| Risk free rate of return | 1.9 - 2.3 | % | 0.8 - 3.7 | % | ||||
| Expected life in years | 4.75 - 5.38 | 1.00 - 4.75 | ||||||
| Volatility | 71 - 72 | % | 68 - 82 | % | ||||
| Forfeitures | 2.61 | % | — | |||||
| Warrants Outstanding | ||||||||||||
| Weighted | ||||||||||||
| Average | ||||||||||||
| Number of | Exercise Price | Exercise | ||||||||||
| Shares | Range | Price | ||||||||||
| December 31, 2007 | 3,522,544 | $ | 2.75 - $4.06 | $ | 3.26 | |||||||
| Grants | 1,745,104 | 1.60 - 1.74 | 1.66 | |||||||||
| Exercises | — | — | — | |||||||||
| Cancellations | (18,558 | ) | 4.05 - 4.06 | 4.05 | ||||||||
| December 31, 2008 | 5,249,090 | 1.60 - 4.06 | 2.80 | |||||||||
| Grants | 3,129,011 | 0.91 - 1.12 | 1.10 | |||||||||
| Exercises | — | — | — | |||||||||
| Cancellations | (444,250 | ) | 4.06 | 4.06 | ||||||||
| December 31, 2009 | 7,933,851 | $ | 0.91 - $4.06 | $ | 2.01 | |||||||
F-15
| December 31, | ||||||||
| 2009 | 2008 | |||||||
| Deferred tax assets: | ||||||||
| Net operating loss carryforwards | $ | 16,988,000 | $ | 18,370,000 | ||||
| Research and development tax credit carryforward | 1,710,000 | 1,628,000 | ||||||
| Charitable contribution carryforward | 37,000 | 39,000 | ||||||
| Accrued vacation | 8,000 | 12,000 | ||||||
| Accrued expenses | 163,000 | 150,000 | ||||||
| Amortization | 5,000 | 6,000 | ||||||
| Depreciation | 1,000 | — | ||||||
| Stock option expense | 975,000 | 919,000 | ||||||
| 19,887,000 | 21,124,000 | |||||||
| Less — valuation allowance | (19,887,000 | ) | (21,123,000 | ) | ||||
| Net deferred tax asset | — | 1,000 | ||||||
| Deferred tax liabilities: | ||||||||
| Depreciation | — | (1,000 | ) | |||||
| Net deferred tax amounts | $ | — | $ | — | ||||
F-16
| December 31, | ||||||||
| 2009 | 2008 | |||||||
| Tax benefit at statutory rate | $ | (2,213,000 | ) | $ | (3,617,000 | ) | ||
| State taxes | (354,000 | ) | (579,000 | ) | ||||
| Permanent M-1s | 339,000 | 563,000 | ||||||
| Limited/expired net operating loss carryforwards | 3,546,000 | 6,150,000 | ||||||
| Limited/expired research and development tax credit carryforward | 120,000 | 284,000 | ||||||
| Research and development tax credit carryforward | (202,000 | ) | (504,000 | ) | ||||
| Change in effective tax rate | — | (455,000 | ) | |||||
| Change in valuation allowance | (1,236,000 | ) | (1,842,000 | |||||
| $ | — | $ | — | |||||
F-17
| September 30, | December 31, | |||||||
| 2010 | 2009 | |||||||
| (Unaudited) | ||||||||
| ASSETS | ||||||||
| Current assets | ||||||||
| Cash and cash equivalents | $ | 4,975,947 | $ | 4,355,768 | ||||
| Accounts receivable | 15,603 | — | ||||||
| Prepaid expenses and other current assets | 582,225 | 196,546 | ||||||
| Total current assets | 5,573,775 | 4,552,314 | ||||||
| Fixed assets, net of accumulated depreciation of $105,655 and $98,171 | 24,640 | 8,492 | ||||||
| Other non-current assets | 17,255 | 22,948 | ||||||
| Total assets | $ | 5,615,670 | $ | 4,583,754 | ||||
| LIABILITIES AND STOCKHOLDERS’ EQUITY | ||||||||
| Current liabilities | ||||||||
| Accounts payable | $ | 605,138 | $ | 140,206 | ||||
| Accrued expenses | 544,732 | 740,198 | ||||||
| Total current liabilities | 1,149,870 | 880,404 | ||||||
| Commitments | — | — | ||||||
| Stockholders’ equity | ||||||||
| Preferred stock, $.001 par value per share, 1,000,000 authorized; no shares issued | — | — | ||||||
| Common stock, par value $.001 per share, 200,000,000 shares authorized, 73,531,578 issued and outstanding as of September 30, 2010; 100,000,000 authorized, 60,406,828 issued and outstanding as of December 31, 2009 | 73,531 | 60,407 | ||||||
| Additional paid-in capital | 92,997,669 | 88,144,347 | ||||||
| Accumulated deficit | (88,605,400 | ) | (84,501,404 | ) | ||||
| Total stockholders’ equity | 4,465,800 | 3,703,350 | ||||||
| Total liabilities and stockholders’ equity | $ | 5,615,670 | $ | 4,583,754 | ||||
F-18
(Unaudited)
| Nine Months ended September 30, | ||||||||
| 2010 | 2009 | |||||||
| Sponsored research revenues | $ | 53,819 | $ | — | ||||
| Operating expenses: | ||||||||
| Research and development | 1,819,036 | 3,064,248 | ||||||
| General and administrative | 2,345,619 | 2,161,539 | ||||||
| Total operating expenses | 4,164,655 | 5,225,787 | ||||||
| Loss from operations | (4,110,836 | ) | (5,225,787 | ) | ||||
| Interest income | 6,840 | 10,304 | ||||||
| Net loss | $ | (4,103,996 | ) | $ | (5,215,483 | ) | ||
| Basic and diluted net loss per common share | $ | (0.06 | ) | $ | (0.10 | ) | ||
| Weighted average number of common shares outstanding | 66,729,519 | 54,216,430 | ||||||
F-19
(Unaudited)
| Nine Months ended September 30, | ||||||||
| 2010 | 2009 | |||||||
| Operating activities: | ||||||||
| Net loss | $ | (4,103,996 | ) | $ | (5,215,483 | ) | ||
| Adjustments to reconcile net loss to net cash used in operating activities: | ||||||||
| Depreciation and amortization | 7,484 | 12,578 | ||||||
| Non-cash share-based compensation | 361,195 | 607,440 | ||||||
| Gain on settlement of accrued liabilities | (141,016 | ) | — | |||||
| Changes in operating assets and liabilities: | ||||||||
| Accounts receivable | (15,603 | ) | — | |||||
| Prepaid expenses and other current assets | (385,679 | ) | (29,592 | ) | ||||
| Other non-current assets | 5,693 | — | ||||||
| Accounts payable | 464,932 | (13,804 | ) | |||||
| Accrued expenses | (54,450 | ) | (481,319 | ) | ||||
| Net cash used in operating activities | (3,861,440 | ) | (5,120,180 | ) | ||||
| Investing activities: | ||||||||
| Purchase of fixed assets | (23,632 | ) | — | |||||
| Net cash used in investing activities | (23,632 | ) | — | |||||
| Financing activities: | ||||||||
| Net proceeds from issuance of common stock | 4,505,251 | 573,683 | ||||||
| Net cash provided by financing activities | 4,505,251 | 573,683 | ||||||
| Net increase (decrease) in cash and cash equivalents | 620,179 | (4,546,497 | ) | |||||
| Cash and cash equivalents: | ||||||||
| Beginning of period | 4,355,768 | 5,655,367 | ||||||
| End of period | $ | 4,975,947 | $ | 1,108,870 | ||||
F-20
F-21
Notes to Financial Statements
(Unaudited)
F-22
Notes to Financial Statements
(Unaudited)
| 2010 | 2009 | |||||||
| Dividend yield | 0.0 | % | 0.0 | % | ||||
| Risk-free rate of return | 1.6 | % | 1.9 | % | ||||
| Expected life in years | 4.75 | 5.38 | ||||||
| Volatility | 70 | % | 72 | % | ||||
| Forfeiture rate | 2.6 | % | 2.6 | % | ||||
F-23
Notes to Financial Statements
(Unaudited)
| • | Level 1 — Quoted prices in active markets for identical assets and liabilities. | |
| • | Level 2 — Observable inputs other than quoted prices in active markets for identical assets and liabilities. | |
| • | Level 3 — Unobservable inputs. |
F-24
Notes to Financial Statements
(Unaudited)
F-25

| Amount to | ||||
| be Paid | ||||
| SEC registration fee | $ | 383 | ||
| Printing and engraving expenses | $ | 15,000 | ||
| Legal fees and expenses | �� | $ | 75,000 | |
| Accounting fees and expenses | $ | 20,000 | ||
| Transfer agent and registrar fees and expenses | $ | 5,000 | ||
| Miscellaneous expenses | $ | 10,000 | ||
| Total | $ | 125,383 | ||
| • | transaction from which the director derives an improper personal benefit; | ||
| • | act or omission not in good faith or that involves intentional misconduct or a knowing violation of law; | ||
| • | unlawful payment of dividends or redemption of shares; or | ||
| • | breach of a director’s duty of loyalty to the corporation or its stockholders. |
II-2
| Exhibit | ||
| Number | Description of Document | |
| 3.1(1) | Restated Certificate of Incorporation. | |
| 3.2(1) | Certificate of Amendment of Restated Certificate of Incorporation. | |
| 3.3(1) | Certificate of Amendment of Restated Certificate of Incorporation. | |
| 3.4(2) | Certificate of Amendment of Restated Certificate of Incorporation. | |
| 3.5(1) | Certificate of Designation of Series A Participating Cumulative Preferred Stock. | |
| 3.6(3) | Amended and Restated Bylaws adopted July 26, 2006. | |
| 3.7(4) | Amendment to Amended and Restated Bylaws. | |
| 4.1(1) | Specimen Common Stock Certificate. | |
| 4.2(1) | Specimen Rights Certificate. | |
| 4.3(1) | Rights Agreement, dated April 29, 1994, between the Company and American Stock Transfer & Trust Company, as Rights Agent. | |
| 4.4(1) | Amendment No. 1 to Rights Agreement, dated March 4, 2004, between the Company and American Stock Transfer & Trust Company, as Rights Agent. | |
| 4.5(5) | Form of Warrant Agreement. | |
| 4.6(6) | Form of Warrant Certificate. | |
| 4.7(5) | Form of Representative’s Warrant. | |
| 4.8(7) | Warrant issued to Lincoln Park. | |
| 4.9(7) | Form of Warrant issued to the Sigma-Tau Purchasers. | |
| 4.10(7) | Omnibus Warrant Amendment Agreement, dated as of January 5, 2011, by and among the Company and the Sigma-Tau Purchasers. | |
| 5.1# | Opinion of Cooley LLP. | |
| 10.1+(8) | Amended and Restated 2000 Stock Option Incentive Plan, as amended. | |
| 10.2*(9) | Patent License Agreement — Exclusive, dated January 24, 2001, between the Company and the U.S. Public Health Service. | |
| 10.3+(10) | Form of Stock Option Grant Notice and Stock Option Agreement under the 2010 Equity Incentive Plan. | |
| 10.4*(11) | Thymosin Beta 4 License and Supply Agreement, dated January 21, 2004, between the Company and Defiante Farmaceutica S.A. | |
| 10.5(12) | Lease by and between RegeneRx Biopharmaceuticals, Inc. and The Realty Associates Fund V, L.P., dated December 10, 2009. | |
| 10.6+(13) | Second Amended and Restated Employment Agreement, dated March 11, 2009, between the Company and Allan L. Goldstein, as amended. | |
| 10.6+(14) | Second Amended and Restated Employment Agreement, dated March 12, 2009, between the Company and J.J. Finkelstein, as amended. | |
| 10.7+(14) | Second Amended and Restated Employment Agreement, dated March 31, 2009, between the Company and C. Neil Lyons, as amended. | |
| 10.8+(14) | Second Amended and Restated Employment Agreement, dated March 31, 2009, between the Company and David Crockford. | |
| 10.9+ (10) | 2010 Equity Incentive Plan. |
II-3
| Exhibit | ||
| Number | Description of Document | |
| 10.10(15) | Stock Purchase Agreement, dated as of June 23, 2005. | |
| 10.11(16) | Form of Warrant to Purchase Common Stock, dated March 17, 2006. | |
| 10.12(17) | Form of Warrant to Purchase Common Stock, dated December 18, 2006. | |
| 10.13(17) | Registration Rights Agreement, dated as of December 15, 2006. | |
| 10.14(18) | Securities Purchase Agreement, dated as of February 27, 2008. | |
| 10.15(18) | Form of Warrant to Purchase Common Stock, dated February 29, 2008. | |
| 10.16(19) | Securities Purchase Agreement, dated as of December 10, 2008. | |
| 10.17(19) | Form of Warrant to Purchase Common Stock, dated December 10, 2008. | |
| 10.18(20) | Securities Purchase Agreement, dated as of April 13, 2009. | |
| 10.19(20) | Form of Warrant to Purchase Common Stock, dated April 30, 2009. | |
| 10.20(21) | Securities Purchase Agreement, dated as of September 30, 2009. | |
| 10.21(21) | Form of Warrant to Purchase Common Stock, dated October 5, 2009. | |
| 10.22(22) | Securities Purchase Agreement, dated as of September 30, 2009. | |
| 10.23(22) | Form of Warrant to Purchase Common Stock, dated October 15, 2009. | |
| 10.24(7) | Securities Purchase Agreement, dated as of January 5, 2011, by and between the Company and Lincoln Park | |
| 10.25(7) | Purchase Agreement, dated as of January 4, 2011, by and between the Company and Lincoln Park | |
| 10.26(7) | Registration Rights Agreement, dated as of January 4, 2011, by and between the Company and Lincoln Park | |
| 10.27(7) | Securities Purchase Agreement, dated as of January 5, 2011, by and between the Company and Defiante Farmaceutica S.A. | |
| 10.28(7) | Securities Purchase Agreement, dated as of January 5, 2011, by and between the Company and Taufin International S.A. | |
| 10.29(7) | Securities Purchase Agreement, dated as of January 5, 2011, by and between the Company and Sinaf S.A. | |
| 23.1 | Consent of Reznick Group, P.C., independent registered public accounting firm. | |
| 23.2# | Consent of Cooley LLP (included in Exhibit 5.1). | |
| 24.1# | Power of Attorney (Reference is made to page II-7 of the Registration Statement on Form S-1 (File No. 333-171982) filed with the Securities and Exchange Commission on January 31, 2011). | |
| (1) | Filed as an exhibit to the registrant’s Registration Statement on Form S-1 (File No. 333-166146) filed with the Securities and Exchange Commission on April 16, 2010 and incorporated herein by reference. | |
| (2) | Filed as an exhibit to the registrant’s Registration Statement on Form S-8 (File No. 333-168252) filed with the Securities and Exchange Commission on July 21, 2010 and incorporated herein by reference. | |
| (3) | Filed as an exhibit to the registrant’s Quarterly Report on Form 10-Q for the quarter ended June 30, 2006 filed with the Securities and Exchange Commission on August 14, 2006 and incorporated herein by reference. | |
| (4) | Filed as an exhibit to the registrant’s Registration Statement on Form S-8 (File No. 333-152250) filed with the Securities and Exchange Commission on July 10, 2008 and incorporated herein by reference. | |
| (5) | Filed as an exhibit to the registrant’s Current Report on Form 8-K filed with the Securities and Exchange Commission on May 21, 2010 and incorporated herein by reference. | |
| (6) | Filed as an exhibit to the registrant’s Amendment No. 1 to Registration Statement on Form S-1 (File No. 333-166146) filed with the Securities and Exchange Commission on May 17, 2010 and incorporated herein by reference. | |
| (7) | Filed as an exhibit to the registrant’s Current Report on Form 8-K filed with the Securities and Exchange Commission on January 7, 2011 and incorporated herein by reference. | |
| (8) | Filed as Annex A to the registrant’s definitive proxy statement on Schedule 14A filed with the Securities and Exchange Commission on May 9, 2008 and incorporated herein by reference. |
II-4
| (9) | Filed as an exhibit to the registrant’s Annual Report on Form 10-KSB for the year ended December 31, 2000 (File No. 1-15070) filed with the Securities and Exchange Commission on April 2, 2001 and incorporated herein by reference. | |
| (10) | Filed as an exhibit to the registrant’s Current Report on Form 8-K filed with the Securities and Exchange Commission on July 20, 2010 and incorporated herein by reference. | |
| (11) | Filed as an exhibit to the registrant’s Registration Statement on Form SB-2 (File No. 333-113417) filed with the Securities and Exchange Commission on March 9, 2004 and incorporated herein by reference. | |
| (12) | Filed as an exhibit to the registrant’s Annual Report on Form 10-K for the year ended December 31, 2009 filed with the Securities and Exchange Commission on March 31, 2010 and incorporated herein by reference. | |
| (13) | Filed as an exhibit to Amendment No. 1 the registrant’s Annual Report on Form 10-K/A for the year ended December 31, 2008 filed with the Securities and Exchange Commission on April 30, 2009 and incorporated herein by reference. | |
| (14) | Filed as an exhibit to the registrant’s Annual Report on Form 10-K for the year ended December 31, 2008 filed with the Securities and Exchange Commission on April 15, 2009 and incorporated herein by reference. | |
| (15) | Filed as an exhibit to the registrant’s Current Report on Form 8-K filed with the Securities and Exchange Commission on June 23, 2005 and incorporated herein by reference. | |
| (16) | Filed as an exhibit to the registrant’s Current Report on Form 8-K filed with the Securities and Exchange Commission on March 7, 2006 and incorporated herein by reference. | |
| (17) | Filed as an exhibit to the registrant’s Current Report on Form 8-K filed with the Securities and Exchange Commission on December 18, 2006 and incorporated herein by reference. | |
| (18) | Filed as an exhibit to the registrant’s Current Report on Form 8-K filed with the Securities and Exchange Commission on February 27, 2008 and incorporated herein by reference. | |
| (19) | Filed as an exhibit to the registrant’s Current Report on Form 8-K filed with the Securities and Exchange Commission on December 12, 2008 and incorporated herein by reference. | |
| (20) | Filed as an exhibit to the registrant’s Current Report on Form 8-K filed with the Securities and Exchange Commission on April 16, 2009 and incorporated herein by reference. | |
| (21) | Filed as an exhibit to the registrant’s Current Report on Form 8-K filed with the Securities and Exchange Commission on September 30, 2009 and incorporated herein by reference. | |
| (22) | Filed as an exhibit to the registrant’s Current Report on Form 8-K filed with the Securities and Exchange Commission on October 5, 2009 and incorporated herein by reference. | |
| + | Indicates management contract or compensatory plan. | |
| * | The registrant has been granted confidential treatment with respect to certain portions of this exhibit (indicated by asterisks), which have been filed separately with the Securities and Exchange Commission. | |
| # | Previously filed. | |
II-5
II-6
| REGENERX BIOPHARMACEUTICALS, INC. | ||||
| By: | /s/ J.J. Finkelstein | |||
| J.J. Finkelstein | ||||
| President and Chief Executive Officer | ||||
| Signature | Title | Date | ||
/s/ J.J. Finkelstein | President and Chief Executive Officer and Director(Principal Executive Officer) | February 7, 2011 | ||
/s/ C. Neil Lyons | Chief Financial Officer (Principal Accounting and Financial Officer) | February 7, 2011 | ||
* | Chairman of the Board of Directors | February 7, 2011 | ||
* | Director | February 7, 2011 | ||
* | Director | February 7, 2011 | ||
* | Director | February 7, 2011 | ||
* | Director | February 7, 2011 | ||
| * | By: | /s/ C. Neil Lyons | ||
| C. Neil Lyons | ||||
| Attorney-in-fact | ||||
II-7