
Savient Redefined
Christopher Clement
President and Chief Executive Officer
NASDAQ: SVNT

Safe Harbor Statement
This presentation contains forward-looking statements within the meaning of Section 21E of the Securities
Exchange Act of 1934. All statements, other than statements of historical facts, included in this report regarding the
Company’s strategy, expected future financial position, discovery and development of products, strategic alliances,
competitive position, plans and objectives of management are forward-looking statements. Words such as
“anticipate,” “believe,” “estimate,” “expect,” “intend,” “plan,” “will” and other similar expressions help identify forward-
looking statements, although not all forward-looking statements contain these identifying words. In particular, the
statements as to the possible outcome of the Company’s request for continued listing on The Nasdaq Stock Market
are forward-looking statements. These forward-looking statements involve substantial risks and uncertainties and
are based on current expectations, assumptions, estimates and projections about the Company’s business and the
biopharmaceutical and specialty pharmaceutical industries in which the Company operates. Such risks and
uncertainties include, but are not limited to, delisting of the Company’s common stock from The Nasdaq Stock
Market, delay or failure in developing Prosaptide, Puricase and other product candidates; difficulties of expanding
the Company’s product portfolio through in-licensing; introduction of generic competition for Oxandrin; fluctuations
in buying patterns of wholesalers; potential future returns of Oxandrin or other products; our continuing to incur
substantial net losses for the foreseeable future; difficulties in obtaining financing; potential development of
alternative technologies or more effective products by competitors; reliance on third-parties to manufacture, market
and distribute many of the Company’s products; economic, political and other risks associated with foreign
operations; risks of maintaining protection for the Company’s intellectual property; risks of an adverse determination
in on-going or future intellectual property litigation; and risks associated with stringent government regulation of the
biopharmaceutical and specialty pharmaceutical industries. The Company may not actually achieve the plans,
intentions or expectations disclosed in its forward-looking statements, and you should not place undue reliance on
the Company’s forward-looking statements. Actual results or events could differ materially from the plans, intentions
and expectations disclosed in the forward-looking statements that the Company makes. The Company’s forward-
looking statements do not reflect the potential impact of any future acquisitions, mergers, dispositions, joint ventures
or investments that the Company may make. The Company does not assume any obligation to update any for
ward-looking statements.

Agenda
Savient Overview
Executing Our Strategy
Milestones
Summary

Long Heritage
Bio-Technology
General Corporation
1980 - 2002
2003
Name Change to

Time for Change
Need for clear strategic focus
Thorough internal review of existing
business uncovered:
– Potential generic introduction for
largest-selling brand – Oxandrin®
– Dwindling sources of funds
– Lack of other near-term commercial
opportunities
– Unsuccessful biologics research
– Languishing pipeline

Oxandrin®: Today
Anticipating generic competition
– Citizens petition filed February 2004
Maximizing sales and marketing efforts
until market dynamics change
– New leadership
– Terminated CSO
– Revitalized/refocused own field force

Transitioning to the New Savient
Focused
Strategic
Value Driven

Our Strategy
Savient Pharmaceuticals, Inc.
Unlocking Shareholder Value
EXPANDING
Our Portfolio
EXTENDING
Our Reach
ADVANCING
Our Pipeline
Rosemont
Oral Liquids
Puricase®
Business
Development

New Leadership
Zeb Horowitz, MD
SVP, CMO
25 Years pharmaceutical/medical research
13 Years industry experience – Procter & Gamble and Novartis
Assistant/Associate Professor of Medicine – NYU & University
of Cincinnati
Christopher Clement
President and CEO
25 Years global pharmaceutical management
Epicyte, Chairman and CEO
Senior Marketing Positions held at Ares-Serono, Searle,
Ciba-Geigy & Merck
David Fink
SVP, Commercial Operations
20 Years pharmaceutical sales and marketing experience
Specialized focus on business transitions – Med Pointe
Pharmaceuticals, Bristol-Myers Squibb
Lawrence Gyenes
SVP, CFO & Treasurer
30 Years experience
20 Years in pharmaceuticals – Searle, Lorex, DuPont & Reliant
CFO of public companies
CPA/MBA
Philip Yachmetz, Esq.
SVP, Corporate Strategy
& General Counsel
24 Years legal experience
16 Years pharmaceutical corporate legal and business
development – Hoffmann-La Roche, CytoTherapeutics and
Progenics
Admitted to Bar in NY and NJ
Transition to Specialty Pharma
Savient Pharmaceuticals, Inc.
Emerging
Rheumatology
Franchise
Rosemont
Oral Liquids
Biologics Mfg.
Business
X
$80 Million Cash
Executing Our Strategy
Savient Pharmaceuticals, Inc.
Unlocking Shareholder Value
EXPANDING
Our Portfolio
EXTENDING
Our Reach
ADVANCING
Our Pipeline
Rosemont
Oral Liquids
Puricase®
Business
Development

Rosemont Pharmaceuticals, Ltd.
Extending Our Reach

Rosemont Overview
Wholly-owned U.K. subsidiary
Acquired from Akzo Nobel in 2003
Liquid formulations of off-patent prescription
pharmaceuticals (solid dosage alternative)
Uniquely positioned, targeting the
elderly/geriatric population
High margin business
– Continued double-digit growth
Extending geographic reach into U.S.
– Filed first NDA for Soltamox – 12/04 / PDUFA
date 10/05
Rosemont Pharmaceuticals, Ltd.
Extending Our Reach
Numerous Opportunities For Growth
Rosemont Pharmaceuticals, Ltd.
U.S.
Soltamox
Potential for other ANDAs/NDAs
Obtain U.S. marketing and
distribution partner
U.K.
Pediatrics
Proprietary products
GP Market
Continental Europe
Other products/countries
Executing Our Strategy
Savient Pharmaceuticals, Inc.
Unlocking Shareholder Value
EXPANDING
Our Portfolio
EXTENDING
Our Reach
ADVANCING
Our Pipeline
Rosemont
Oral Liquids
Puricase®
Business
Development

Unmet Medical Need: Gout
Painful rheumatic disease
Symptomatic hyperuricemia
Results from over-production or under-excretion
of uric acid
Deposits crystals of uric acid in connective
tissues and/or joint spaces
– Gouty arthritis
– Gout tophi
– Gout flares
– Gout-related kidney disease
What is Gout?
Source: NIH Publication No. 02-5027 April 2002.

Refractory Gout
Painful / debilitating inflammation
Destructive arthritis
Development of tophi
Renal complications
Refractory Gout is a Serious Unmet Medical
Condition With Significant Clinical Consequences
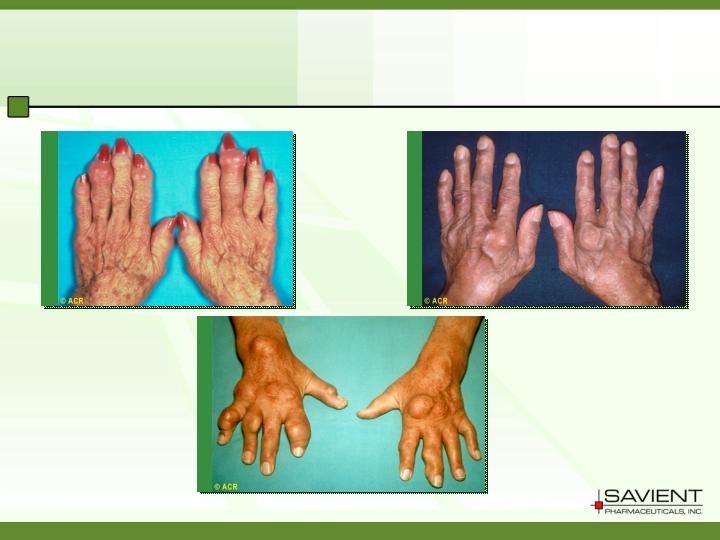
Source: ACR
Illustration of Gout Tophi

Puricase® Overview
PEG technology MPV and Duke
Exclusive license to trademark from MPV
Combined low double-digit royalties
Ownership:
Completed: Phase 2
Initiating Phase 3: 1Q06
Status:
Potential to be first effective therapy to
reverse course of the disease
Differentiation:
Estimates vary widely
25,000 to 75,000 patients in the U.S.
Equal numbers for Europe & Asia
Market Opportunity:
Treatment of severe gout in patients
unsuccessfully treated with conventional
therapy
Indication:
Polyethelene glycol (PEG)-modified
recombinant porcine uricase
Product/Category:

Puricase®
Randomized, double-blind, multi-center,
parallel groups
Two dose levels with two dose regimens:
– 4 mg and 8 mg every 2 weeks
– 8 mg and 12 mg every 4 weeks
Twelve week exposure
Sample size: ~10 per treatment group
9 Sites in U.S.
Phase II Study Design

Puricase®
Phase II Study Efficacy
Maximum % decrease in PUA from baseline within the first 24 hours
(p<.0001)
–93%
4 weeks
12 mg
(p<.0001)
–87%
4 weeks
8 mg
(p<.0001)
–94%
2 weeks
8 mg
(p=.0002)
–72%
2 weeks
4 mg
(p value)
Percent
Decrease
Arm
Study
% Decrease in PUA from baseline over the 12-week treatment period
(p<.0001)
–67%
4 weeks
12 mg
(p=.0003)
–58%
4 weeks
8 mg
(p<.0001)
–86%
2 weeks
8 mg
(p=.0002)
–38%
2 weeks
4 mg
(p value)
Percent
Decrease
Arm
Study

Puricase®
No increase in gout flare frequency with
normalization of plasma uric acid
Anecdotal reports from patients
and investigators
– Eradication of gout tophi
– Improvement in joint function
– Improvement in sense of well being
Phase II Study Efficacy (cont’d.)

Puricase®
General
– No medically important signal of
drug-related AEs or laboratory abnormalities
Gout flares
– No increase in frequency over time with
normalization of uric acid
Infusion reactions typical for biologicals
Allergy
– No clearly allergic manifestations
Phase II Study Safety

Puricase®
Working with FDA to finalize
Program designed to evaluate:
– Effectiveness in the control of uric acid
– Effectiveness in multiple
clinical outcomes
Elected to submit Special Protocol
Assessment (SPA)
Proposed Phase III Program

Puricase®
Development program to date strong basis for
full development and registration
Key efficacy endpoints
– Control of uric acid (determines approvability)
– Improvement in clinical outcomes
Correlations between suppression of PUA and
clinical outcomes will be evaluated in this study
Summary

Refining Our Therapeutic Focus
An Emerging Rheumatology Franchise

Rheumatology is an Area of Strategic
Long-Term Focus
Why Rheumatology?
Growing Market for Novel Technologies
A “Resource Manageable” Market
Puricase Will be Targeted to Rheumatologists
Unique Positioning for Savient

In-licensing
Co-promotion
Euflexxa™
Puricase®
Building our Rheumatology Franchise
Opportunities in Rheumatology
Time
2005
2008

Euflexxa™ Overview
Approximately $500 million
Synvisc: 61% market
2004 WW HA Market:
2004 Market Leader:
Only non-avian derived HA
Head-to-head data with Synvisc
Differentiation:
Approved
Scheduled launch 4Q 2005
Status:
July 2005 co-promotion agreement with
Ferring (product owner) to market to U.S.
Rheumatologists – 50% participation
in global revenues above agreed
upon thresholds
Deal/Term:
Treatment of pain in osteoarthritis
of the knee
Indication:
Hyaluronic Acid (HA) (three injections)
Product/Category:
Our Strategy
Savient Pharmaceuticals, Inc.
Unlocking Shareholder Value
EXPANDING
Our Portfolio
EXTENDING
Our Reach
ADVANCING
Our Pipeline
Rosemont
Oral Liquids
Puricase®
Business
Development

Expanding our Product Portfolio
Utilize experienced, focused business
development team
Maximize competitive advantages
– Fast and flexible management team
– Strong balance sheet
Expedite opportunity assessment process
– Proactive and disciplined
Seek savvy partnering agreements
Isolate and pursue strategic, attractive
in-licensing/co-promotion opportunities

Future Milestones
Delivering Results

2005 – 2006 Milestones
Approval of Soltamox®
Co-promotion with Ferring for Euflexxa ™ in U.S.
Announcement of strategic commercialization
partner for Soltamox ®
Agreement with FDA on SPA for Puricase ® Phase 3
Development Program
Contract with development and commercialization
partner for Puricase ® in Europe

The New Savient
Extending Our Reach
Executing New Strategy
Advancing Our Pipeline
Expanding Our Portfolio
Financially Stable
Delivering Results

Savient Redefined