Exhibit 99.1

MDRNA
The 9th Annual Needham
Healthcare Conference
9 June 2010

2 |
|
Forward Looking Statement
Statements made in this presentation may be forward-looking statements
within the meaning of Federal Securities laws that are subject to certain risks
and uncertainties and involve factors that may cause actual results to differ
materially from those projected or suggested. Factors that could cause actual
results to differ materially from those in forward-looking statements include,
but are not limited to: (i) the ability of MDRNA to obtain additional funding;
(ii) the ability of MDRNA to attract and/or maintain manufacturing, research,
development and commercialization partners; (iii) the ability of MDRNA
and/or a partner to successfully complete product research and development,
including preclinical and clinical studies and commercialization; (iv) the
ability of MDRNA and/or a partner to obtain required governmental approvals;
(v) |
| the ability of MDRNA and/or a partner to develop and commercialize |
products that can compete favorably with those of competitors; and (vi) the
failure of the stockholders of MDRNA to approve the merger with Cequent,
the failure of either party to meet any of the other conditions to closing the
merger, contractual restrictions on the conduct of our business included in
the merger agreement, and any impact on our relationships with third parties
as a result of the announcement of the proposed merger. Additional factors
that could cause actual results to differ materially from those projected or
suggested in any forward-looking statements are contained in MDRNA’s most
recent periodic reports on Form 10-K and Form 10-Q that are filed with the
Securities and Exchange Commission. MDRNA assumes no obligation to update
and supplement forward-looking statements because of subsequent events.

3 |
|
Business
Market
Products
Technology
Intellectual Property
Target to Clinic Capability for RNAi Drug Development
MDRNA’s business model:
? Platform technology for RNAi-based drug development
? Engage large pharma in multi-year therapeutic/target
collaborations
? Supported by acquisition of 2nd platform (in progress)
? Pipeline of therapeutic compounds
? Internal oncology development program
? Supported by acquisition of Phase 1 compound (in progress)
? In the future, we’ll be a company with:
? Multiple drug discovery platforms related to RNAi-based
therapeutics
? 2-3 compounds in clinical development
? R&D collaborations involving RNAi Drug Discovery Platforms
? Licensing transactions involving clinical programs
We lower risk and create stability through our platforms & pipeline business model
4 |
|
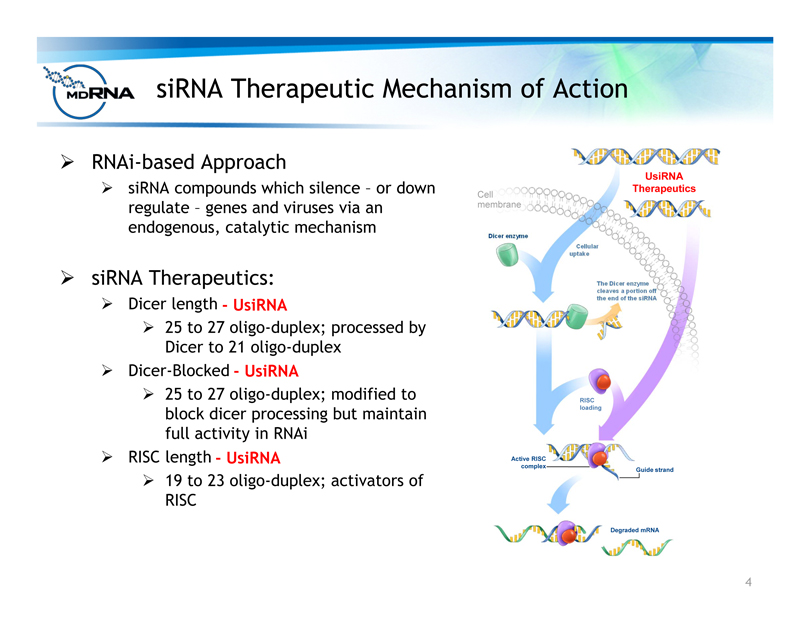
? RNAi-based Approach
? siRNA compounds which silence – or down
regulate – genes and viruses via an
endogenous, catalytic mechanism
? siRNA Therapeutics:
? Dicer length
? 25 to 27 oligo-duplex; processed by
Dicer to 21 oligo-duplex
? Dicer-Blocked
? 25 to 27 oligo-duplex; modified to
block dicer processing but maintain
full activity in RNAi
? RISC length
? 19 to 23 oligo-duplex; activators of
RISC
Cellular
uptake
Dicer enzyme
The Dicer enzyme
cleaves a portion off
the end of the siRNA
Degraded mRNA
RISC
loading
Active RISC
complex Guide strand
Cell
membrane
siRNA
Therapeutic
siRNA Therapeutic Mechanism of Action
- UsiRNA
- UsiRNA
- UsiRNA
UsiRNA
Therapeutics

5 |
|
Business
Market
Products
Technology
Intellectual Property
Next Generation Therapeutics based on RNAi
Current market niche:
? Diseases where:
? Small molecules or monoclonal antibodies (mAb) will
not work
? Targeted delivery is possible
? Focus is in oncology
Future market potential:
? Breakthrough RNAi-based therapeutics that compete with
small molecules and/or mAb
? Differentiators may include:
? Mode of administration
? Cost of goods
? Advances in therapeutic effect
Promising market opportunities with future upside

6 |
|
Business
Market
Products
Technology
Intellectual Property
? RNA interference technology is disruptive
? Naturally occurring process that uses the body’s own
biology to fix itself
? “Catalytic” i.e. one-to-many relationship between
compound and action
? Portfolio of capabilities in molecular biology, cellular
biology, peptide and alkylated amino acid chemistry,
pharmacology, and bioinformatics
? MDRNA is developing:
? Novel siRNA constructs: UsiRNA and Meroduplex
? Novel liposome (DiLA2) and peptide-based delivery
A Multi-faceted Approach to a Complex Problem
We leverage our novel siRNA constructs (UsiRNA) and delivery systems (DilA2)
7 |
|
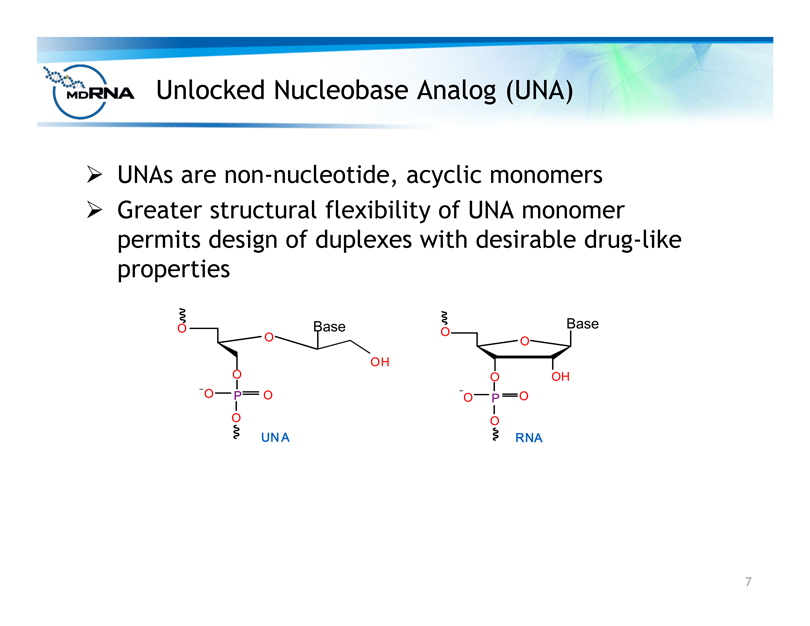
Unlocked Nucleobase Analog (UNA)
? UNAs are non-nucleotide, acyclic monomers
? Greater structural flexibility of UNA monomer
permits design of duplexes with desirable drug-like
properties
O
O Base
O
P O
O
O
OH
UN A
O
O
O
P O
O
O
OH
RNA
Base
8 |
|
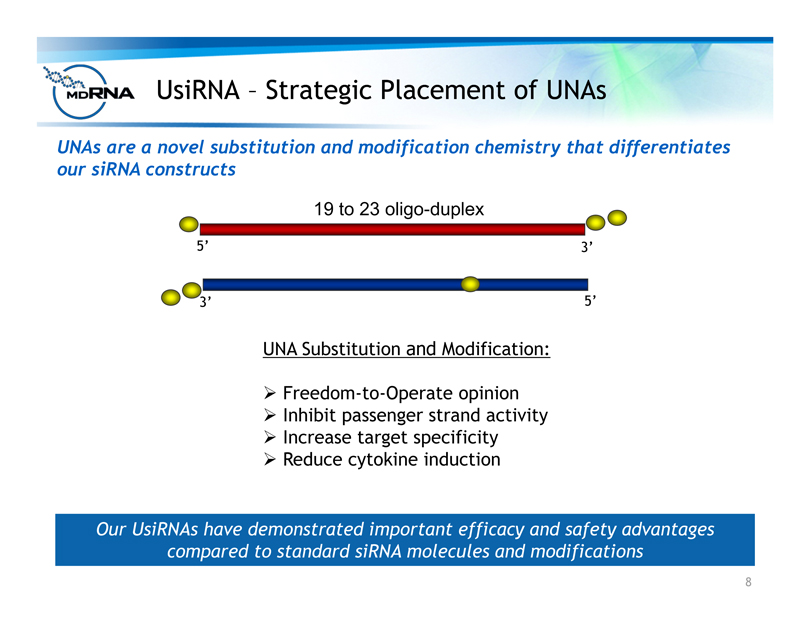
UsiRNA – Strategic Placement of UNAs
UNA Substitution and Modification:
? Freedom-to-Operate opinion
? Inhibit passenger strand activity
? Increase target specificity
? Reduce cytokine induction
5’ 3’
3’ 5’
19 to 23 oligo-duplex
Our UsiRNAs have demonstrated important efficacy and safety advantages
compared to standard siRNA molecules and modifications
UNAs are a novel substitution and modification chemistry that differentiates
our siRNA constructs
9
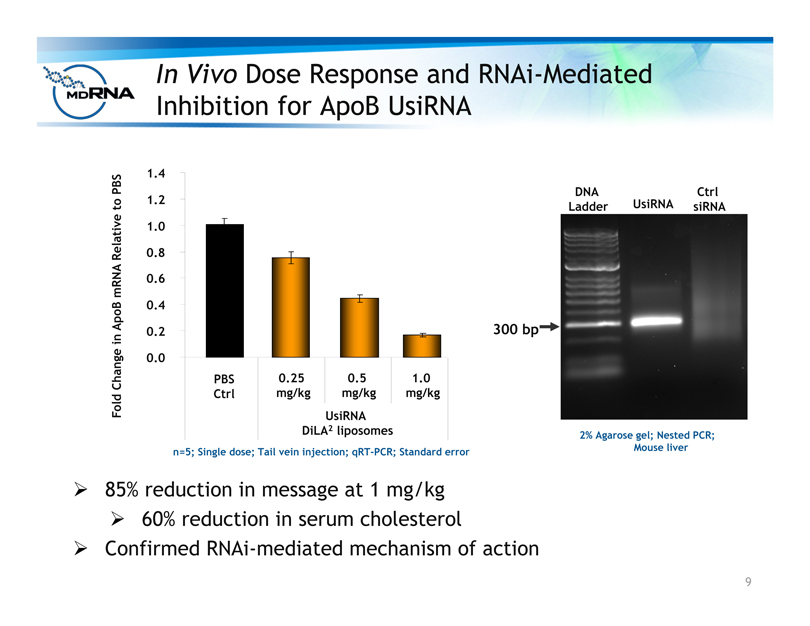
In Vivo Dose Response and RNAi-Mediated
Inhibition for ApoB UsiRNA
? 85% reduction in message at 1 mg/kg
? 60% reduction in serum cholesterol
? Confirmed RNAi-mediated mechanism of action
Fold Change in ApoB mRNA Relative to PBS
0.0
0.2
0.4
0.6
0.8
1.0
1.2
1.4
1.0
mg/kg
0.5
mg/kg
0.25
mg/kg
PBS
Ctrl
n=5; Single dose; Tail vein injection; qRT-PCR; Standard error
UsiRNA
DiLA2 liposomes
300 bp
DNA
Ladder
Ctrl
UsiRNA siRNA
2% Agarose gel; Nested PCR;
Mouse liver
10
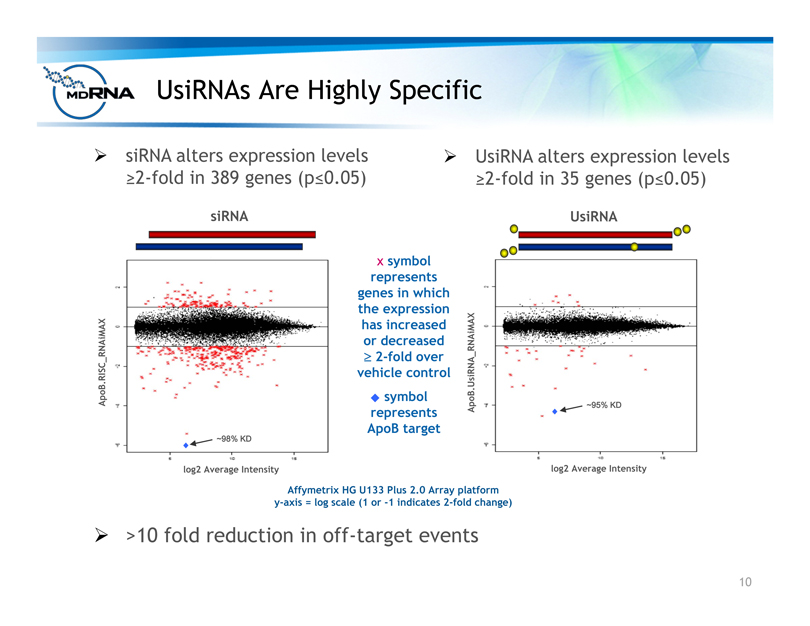
? >10 fold reduction in off-target events
Affymetrix HG U133 Plus 2.0 Array platform
y-axis = log scale (1 or -1 indicates 2-fold change)
log2 Average Intensity
ApoB.RISC_RNAiMAX
x symbol
represents
genes in which
the expression
has increased
or decreased
? 2-fold over
vehicle control
symbol
represents
ApoB target
siRNA
log2 Average Intensity ApoB.UsiRNA_RNAiMAX
UsiRNA
? siRNA alters expression levels
?2-fold in 389 genes (p?0.05)
? UsiRNA alters expression levels
?2-fold in 35 genes (p?0.05)
~98% KD
~95% KD
UsiRNAs Are Highly Specific
11
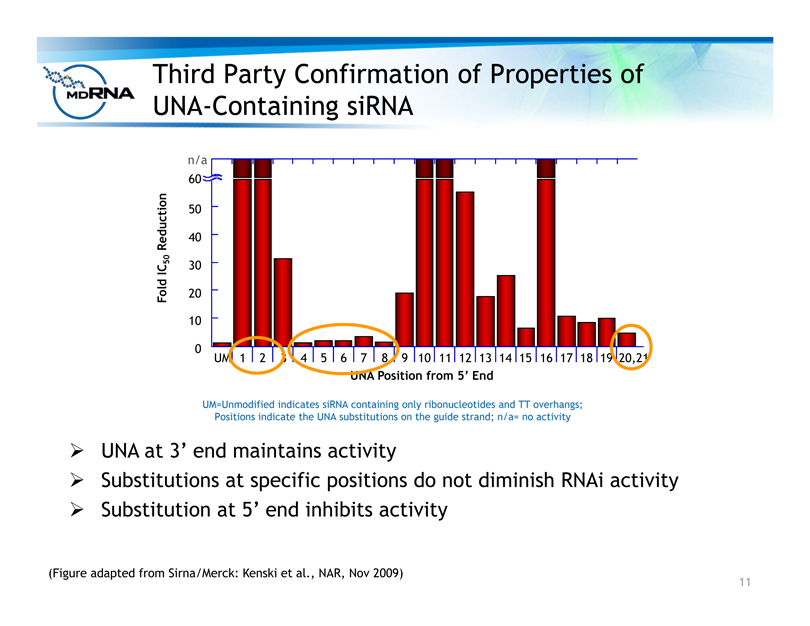
Third Party Confirmation of Properties of
UNA-Containing siRNA
0
10
20
30
40
50
60
n/a
UM 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20,21
UNA Position from 5’ End
Fold IC50 Reduction
(Figure adapted from Sirna/Merck: Kenski et al., NAR, Nov 2009)
UM=Unmodified indicates siRNA containing only ribonucleotides and TT overhangs;
Positions indicate the UNA substitutions on the guide strand; n/a= no activity
? UNA at 3’ end maintains activity
? Substitutions at specific positions do not diminish RNAi activity
? Substitution at 5’ end inhibits activity
12
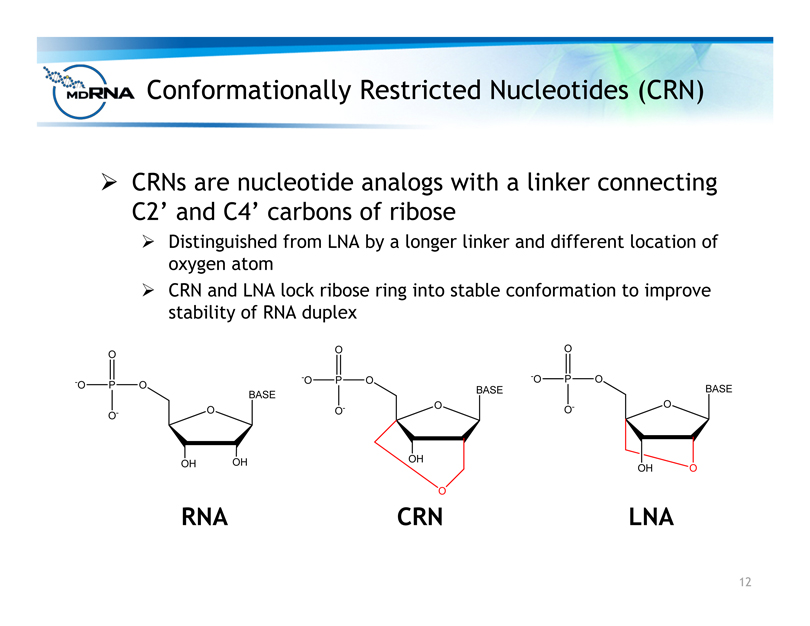
Conformationally Restricted Nucleotides (CRN)
? CRNs are nucleotide analogs with a linker connecting
C2’ and C4’ carbons of ribose
? Distinguished from LNA by a longer linker and different location of
oxygen atom
? CRN and LNA lock ribose ring into stable conformation to improve
stability of RNA duplex
RNA CRN LNA
O
OH
BASE
OH
-O P O
OO

13
CRN Enhancement of UsiRNA Technology
? Provide greater opportunity to leverage UNA properties
? Improve strand bias for RISC loading
? CRN substitutions complementary to UNA
substitutions
? Position-by-position tailoring of specificity in conjunction
with UNA
? CRN or UNA for site-specific nuclease resistance
5’ 3’
3’ 5’
CRN
UNA
14
Additional Therapeutic Approaches Using CRN
? Increase activity and selectivity of molecule to target
mRNA and miRNA
? Antisense therapeutics
? CRN can substitute for RNA at ends of DNA-RNA hybrid
compounds
? Antagomir therapeutics
? CRN can be substituted throughout the sequence
? Location and extent of substitution will be sequence dependent
? Enhances resistance to nuclease
CRN

15
DiLA2 (Di-Alkylated Amino Acids) Delivery Platform
? Composed of unique combinations of head groups, linkers and alkyl
chains
? Self-assembly into liposomes with siRNA and other components
Linker region
Alkyl chain: R2
Alkyl chain: R1
Head group (AA and #)
? Size range from 60—130 nm; controlled by process
? Typical range 80 to 120 nm
? >80% siRNA encapsulation
? Narrow polydispersity index (PDI)
? One year stability has been demonstrated
? Chemical and physical parameters
? In vivo activity
A unique and proprietary approach to solving the most significant scientific obstacle to the
broad use of RNAi-based therapeutics to treat human diseases
16
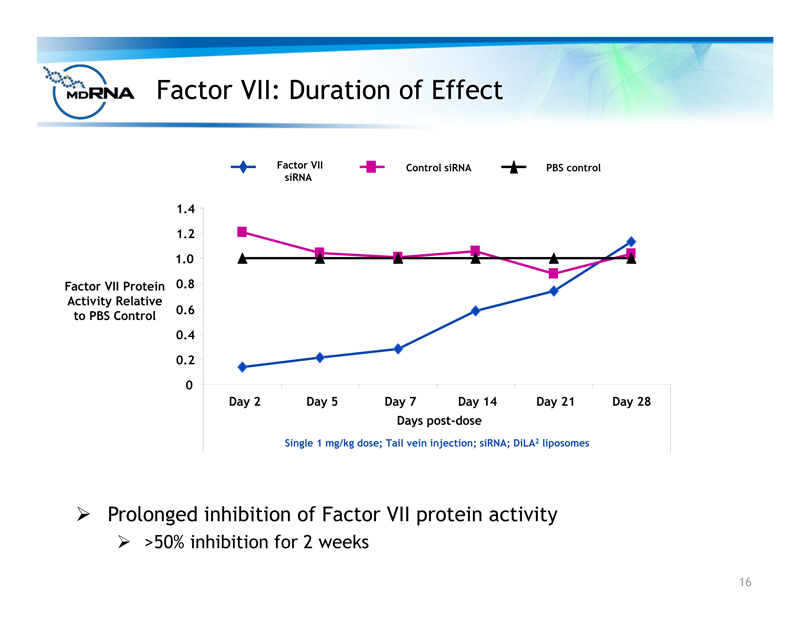
Factor VII: Duration of Effect
? Prolonged inhibition of Factor VII protein activity
? >50% inhibition for 2 weeks
0
0.2
0.4
0.6
0.8
1.0
1.2
1.4
Day 2 Day 5 Day 7 Day 14 Day 21 Day 28
Days post-dose
Factor VII Protein
Activity Relative
to PBS Control
Factor VII
siRNA
Control siRNA PBS control
Single 1 mg/kg dose; Tail vein injection; siRNA; DiLA2 liposomes
17
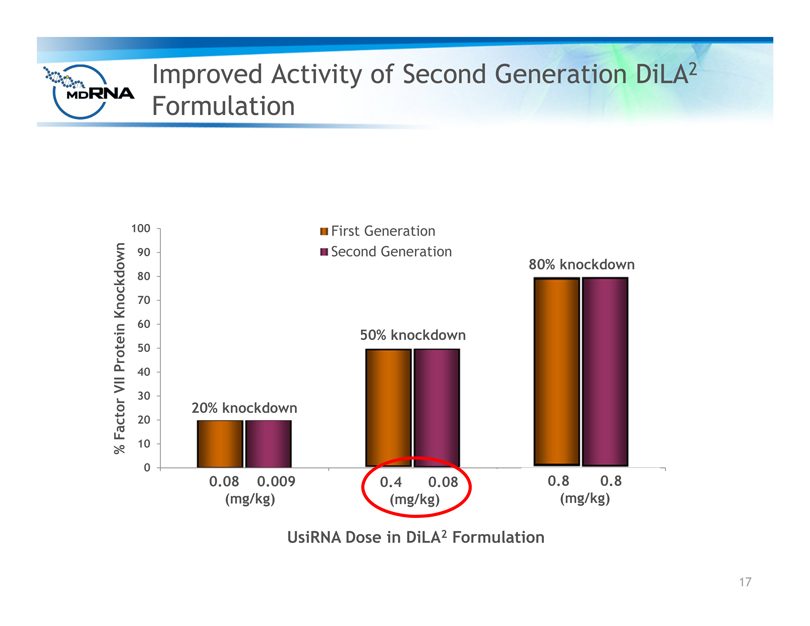
Improved Activity of Second Generation DiLA2
Formulation
0
10
20
30
40
50
60
70
80
90
100
1 |
| 2 3 |
% Factor VII Protein Knockdown
First Generation
Second Generation
0.08 0.009
(mg/kg)
0.4 0.08
(mg/kg)
0.8 0.8
(mg/kg)
UsiRNA Dose in DiLA2 Formulation
50% knockdown
80% knockdown
20% knockdown

18
? Effective systemic delivery to hepatocytes demonstrated with
RNAi-mediated knockdown of:
?ApoB, PCSK9, DGAT2, and Factor VII in mouse
?FVII in NHP
? Effective systemic delivery to jejunum demonstrated with
RNAi-mediated knockdown of:
?ApoB
? Effective systemic delivery to solid tumor demonstrated with
RNAi-mediated knockdown of:
?Survivin and PLK1
? Effective local delivery to solid tumor demonstrated with
RNAi-mediated knockdown of:
?Survivin, PLK1, HRAS, & FGFR3
DiLA2 Delivery Accomplishments
19
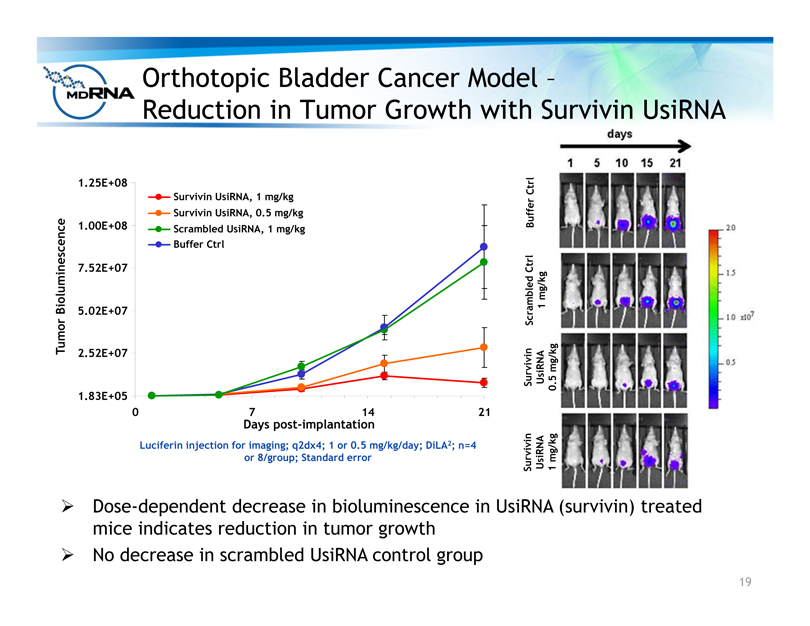
Orthotopic Bladder Cancer Model –
Reduction in Tumor Growth with Survivin UsiRNA
? Dose-dependent decrease in bioluminescence in UsiRNA (survivin) treated
mice indicates reduction in tumor growth
? No decrease in scrambled UsiRNA control group
Survivin
UsiRNA
1 |
| mg/kg |
Survivin
UsiRNA
0.5 mg/kg
Scrambled Ctrl
1 |
| mg/kg |
Buffer Ctrl
Luciferin injection for imaging; q2dx4; 1 or 0.5 mg/kg/day; DiLA2; n=4
or 8/group; Standard error
Days post-dose
1.83E+05
2.52E+07
5.02E+07
7.52E+07
1.00E+08
1.25E+08
0 7 14 21
Days post-implantation
Tumor Bioluminescence
Survivin UsiRNA, 1 mg/kg
Survivin UsiRNA, 0.5 mg/kg
Scrambled UsiRNA, 1 mg/kg
Buffer Ctrl
days
1 |
| 5 10 15 21 |
0.5
1.0 x107
1.5
2.0
20
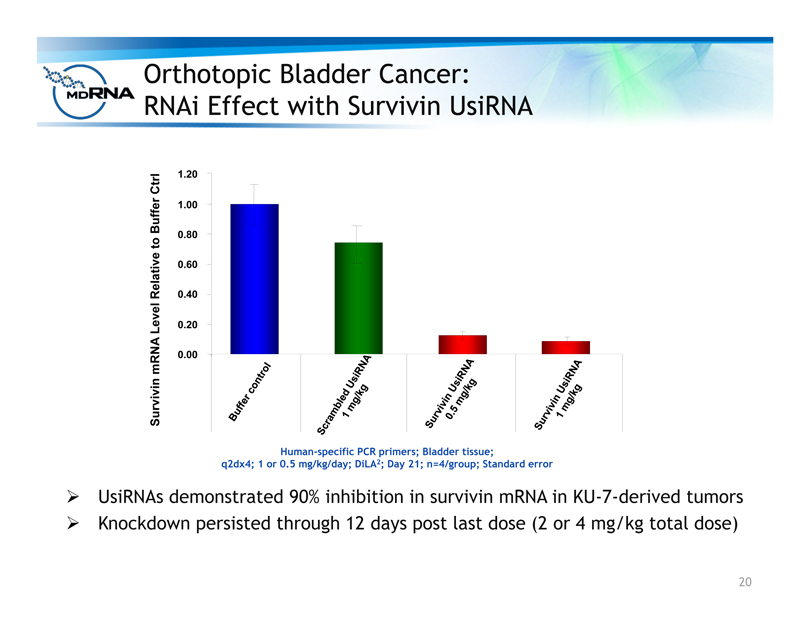
Orthotopic Bladder Cancer:
RNAi Effect with Survivin UsiRNA
? UsiRNAs demonstrated 90% inhibition in survivin mRNA in KU-7-derived tumors
? Knockdown persisted through 12 days post last dose (2 or 4 mg/kg total dose)
Human-specific PCR primers; Bladder tissue;
q2dx4; 1 or 0.5 mg/kg/day; DiLA2; Day 21; n=4/group; Standard error
0.00
0.20
0.40
0.60
0.80
1.00
1.20
Survivin mRNA Level Relative to Buffer Ctrl
21
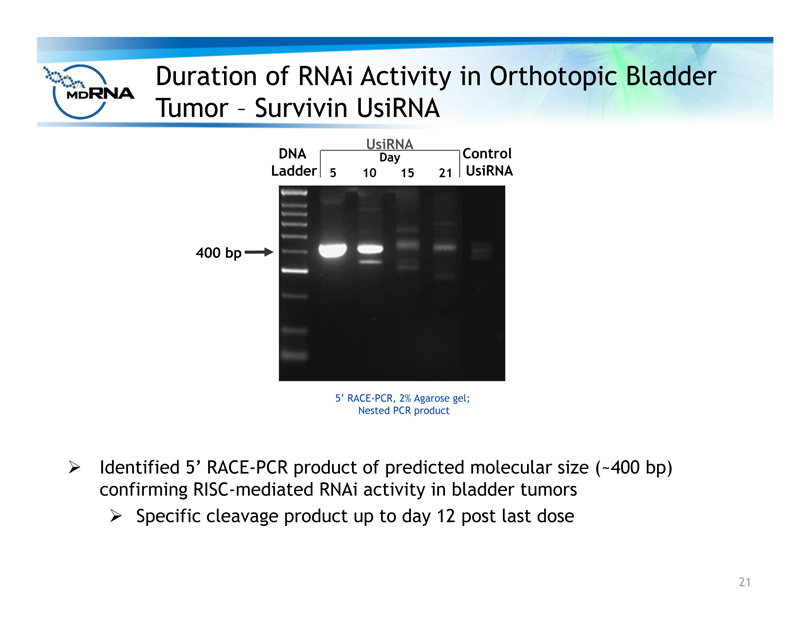
? Identified 5’ RACE-PCR product of predicted molecular size (~400 bp)
confirming RISC-mediated RNAi activity in bladder tumors
? Specific cleavage product up to day 12 post last dose
Duration of RNAi Activity in Orthotopic Bladder
Tumor – Survivin UsiRNA
5’ RACE-PCR, 2% Agarose gel;
Nested PCR product
400 bp
Control
UsiRNA
UsiRNA
DNA
Ladder 5 10 15 21
Day
22
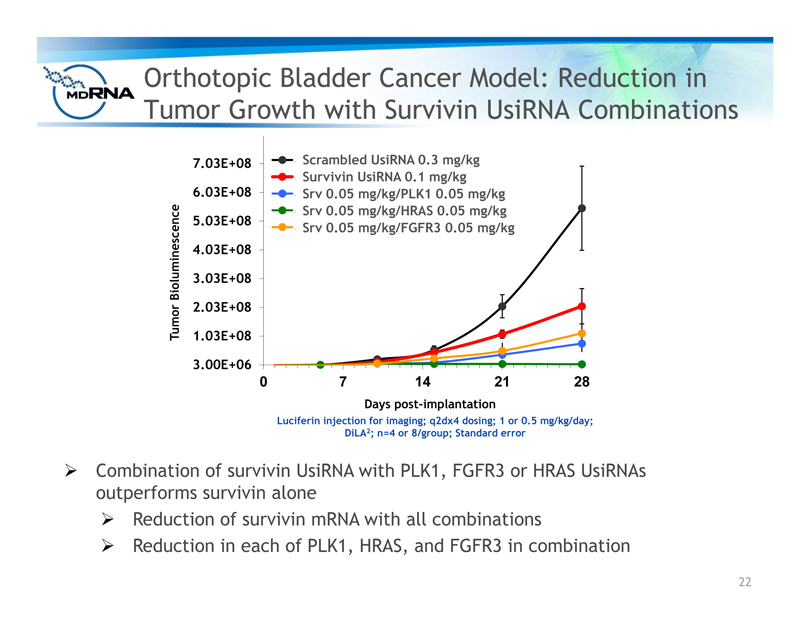
Orthotopic Bladder Cancer Model: Reduction in
Tumor Growth with Survivin UsiRNA Combinations
? Combination of survivin UsiRNA with PLK1, FGFR3 or HRAS UsiRNAs
outperforms survivin alone
? Reduction of survivin mRNA with all combinations
? Reduction in each of PLK1, HRAS, and FGFR3 in combination
3.00E+06
1.03E+08
2.03E+08
3.03E+08
4.03E+08
5.03E+08
6.03E+08
7.03E+08
0 7 14 21 28
SCR
SRV 0.1
SRV/PLK 0.1
SRV/hRAS 0.1
SRV/FGFR3 0.1
Luciferin injection for imaging; q2dx4 dosing; 1 or 0.5 mg/kg/day;
DiLA2; n=4 or 8/group; Standard error
Days post-implantation
Tumor Bioluminescence
Scrambled UsiRNA 0.3 mg/kg
Survivin UsiRNA 0.1 mg/kg
Srv 0.05 mg/kg/PLK1 0.05 mg/kg
Srv 0.05 mg/kg/HRAS 0.05 mg/kg
Srv 0.05 mg/kg/FGFR3 0.05 mg/kg

23
? Second validated RNAi Drug Discovery Platform:
? TransKingdom RNAi technology for oral delivery
? Expanded oncology and inflammatory disease pipeline with clinical program:
? FAP Phase 1 Clinical Trial expected in 3Q2010
? Two Inflammatory Bowel Disease INDs projected in 2011
? One pre-IND program in IBD under option with Novartis
? Expanded intellectual property estate1 covering: novel chemistries; RNAi constructs;
delivery platforms for systemic, local and oral delivery; and gene targets.
? 19 issued or allowed patents;
? 45 pending U.S. patent applications;
? 139 pending foreign patent applications; and
? 7 PCT applications.
? Industry leading research team with proven success in demonstrating safety and
tolerability in non-human primates.
? Industry leading development team with success in bringing a RNAi-based therapeutic
into the clinic.
? World-class scientific advisors at Board of Director and Scientific Advisory Board
levels.
Increased Capabilities through Cequent Acquisition
1Numbers represent IP portfolio of combined company

24
Business
Market
Products
Technology
Intellectual Property
Strong IP Pipeline to Support Our Business
? Our portfolio consists of internally developed IP in combination
with licensed/acquired IP which covers siRNAs, chemistry,
delivery and gene targets:
? 17 issued/allowed US patents
? 41 U.S. patent applications
? 134 ex-U.S. patent applications
? Growing new patent filings at nearly 30% CAGR
? World-class team leading innovation
? Over 30 scientists with expertise in nucleic acid chemistry,
RNAi, and liposomal and peptide based delivery of nucleic
acids
? Average patent age = 5 years
? Younger than many others in the industry
? Positioning MDRNA for longer-term protection
Robust IP Portfolio developed by industry leading experts

25
? Five on-going early collaborative efforts
? 4 large international pharma companies (i.e. Pfizer, AstraZeneca)
? 1 biotechnology therapeutics company
? Focused on interactions with our world-class research team
and the capabilities of our RNAi drug discovery platform.
? Research Effort:
? Efficient and effective gene silencing
? Multiple targets in multiple tissue systems
? Systemic and local administration
? Pre-clinical program (hepatocellular carcinoma)
? Established as collaborations:
? Alliance Managers
? Project Managers
? Scientific Leads
? Goal is to convert two early collaborative efforts into multiyear,
multi-target R&D collaborations in 2010
Early Collaborative Efforts

26
? UsiRNA/DiLA2 compounds are highly potent and well tolerated in vivo
? Knockdown of ApoB, PCSK9, DGAT2, FVII, PLK1 and survivin via systemic delivery
? Knockdown of survivin, PLK1, FGFR3, and HRAS via local delivery
? Oncology Pipeline
? Bladder Cancer – IND expected in 1Q2011
? Hepatocellular Carcinoma – IND expected by mid-2011
? Validated drug discovery platform
? Roche and Novartis licensed MDRNA RNAi technology
? 5 early collaborative efforts (e.g. Pfizer, AstraZeneca)
? Integrated personalized medicine (therapeutic/diagnostic) platform
? Broad and comprehensive IP estate
? 17 Issued/Allowed Patents; 41 U.S. Applications; 6 PCT Applications
? Experienced management team
? CEO – Michael French (Former Sirna SVP, Corporate Development)
? CSO – Barry Polisky (Former Sirna CSO; Research VP at Merck)
? CFO – Pete Garcia (Former Nuvelo CFO; Dendreon CFO)
MDRNA - A Leader in RNAi Therapeutics

27
Additional Information about the Merger will
be filed with the SEC
This presentation may be deemed to be solicitation material regarding
the proposed merger of MDRNA and Cequent. In connection with the
proposed merger, MDRNA has filed relevant materials and documents
with the Securities and Exchange Commission (SEC), including a proxy
statement, which will be mailed to the stockholders of MDRNA.
Investors and the public are urged to read these materials carefully and
in their entirety when they become available because they will contain
important information about the companies, the proposed merger and
the expectations for the combined company. The proxy statement and
other relevant materials (when they become available), and any and all
documents filed with the SEC, may be obtained free of charge at the
SEC’s web site at www.sec.gov. In addition, investors and the public
may obtain free copies of the documents filed with the SEC by MDRNA
by directing a written request to MDRNA, Inc., 3830 Monte Villa
Parkway, Bothell, Washington 98021, Attention: Investor Relations. The
directors, executive officers and employees of MDRNA may be deemed
to be participants in the solicitation of proxies in connection with the
proposed transaction. Information regarding the special interests of
these directors, executive officers and employees in the proposed
transaction, if any, will be included in the proxy statement referred to
above.

MDRNA