
2006 Annual
Stockholders Meeting
May 25, 2006

Financial Highlights
0.23
0.40
Diluted Earnings Per Share
1,821
3,318
Net Income
4,303
5,524
Income from Operations
23,834
27,120
Gross Profit
$56,736
$63,047
Revenues
2004
2005
in thousands, except for per share amounts

Financial Highlights
2005 Continued focus on:
Maximize operating income
Increase cash flows
Improve balance sheet

Maximize Operating Income
Revenue Growth
Improved Gross Margins
Reduced SG&A Expense as a
Percentage of Revenues
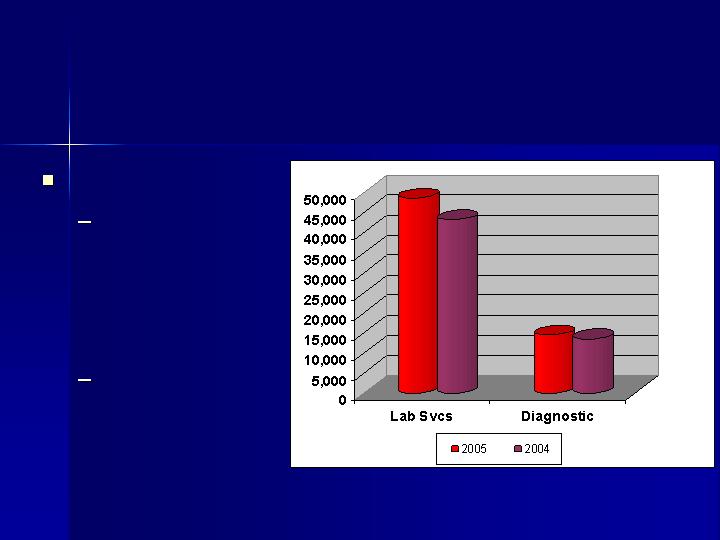
Maximize Operating Income
Revenue Growth
Laboratory
Services
Revenues
increased 12%
$48.6 million
Diagnostic
Revenues
increased 7%
$14.5 million

Improve Gross Margin
Consolidated Gross Margin increased to 43.0% of
revenues, compared to 42.0% in 2004
Laboratory Services Gross Margin increased to
38.0% compared to 36.1% in 2004
Diagnostic Gross Margins declined to 59.8%
from 60.9% in 2004
Maximize Operating Income

Reduced SG&A Expenses as a
Percentage of Revenues
SG&A Expenses were 30.6% of Revenues
in 2005, compared to 31.4% of Revenues
in 2004
Maximize Operating Income
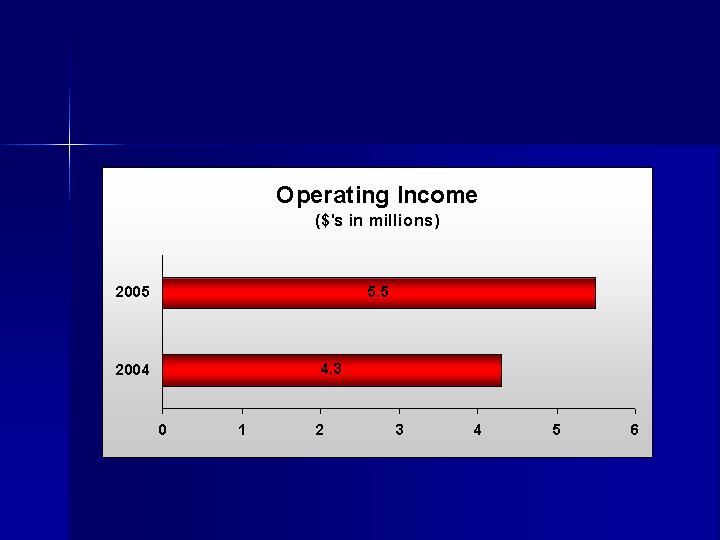
Maximize Operating Income
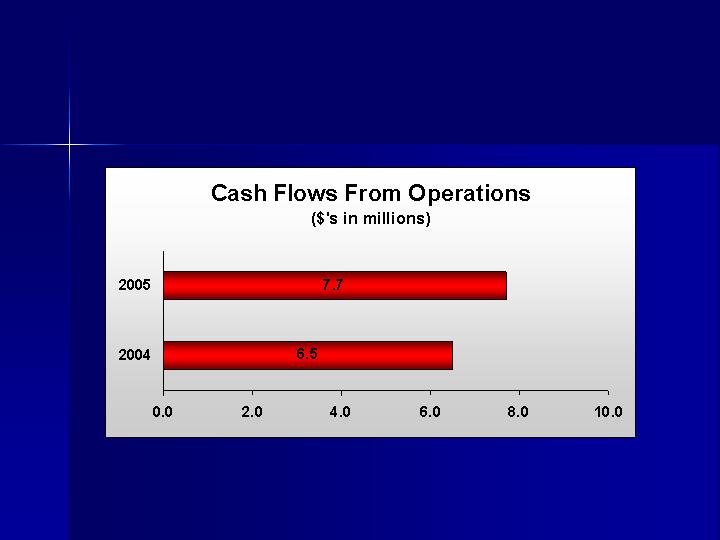
Increase Cash Flows

Improve Balance Sheet
Repaid $4.7 million on revolving credit
facility
Reduced long-term debt by $1.2
million
Stockholders Equity increased to $44.8
million from $37.8 million

MEDTOX Scientific, Inc.
Investing In Our Future

MEDTOX Scientific, Inc.
Update on 2005 initiatives:
eChain® System
MEDTOXscan™ Reader
SURE-SCREEN®

MEDTOX Scientific, Inc.
eChain ® System
(Corporate & Clinic markets)

MEDTOX Scientific, Inc.
eChain® System
Overview:
1st Web-based “thin client application”
chain-of-custody and data management
system
Easily deployed at clinics (PC & printer)
Used for lab-based and PROFILE® tests
Secure system architecture

MEDTOX Scientific, Inc.
eChain® System
Update:
Initial production transaction: 2/27/06
371 collection sites using eChain currently
264 clinics in initial rollout
107 subsequent clinics
Goal of 1,000 sites using eChain by
12/31/06
Corporate market launch in 2nd half of 2006

MEDTOX Scientific, Inc.
Reader
(Hospital Lab / ER market)
MEDTOX Scientific, Inc.

MEDTOX Scientific, Inc.
Overview:
Interprets drug strips electronically
Eliminates device interpretation ambiguity
Expedites throughput in hospital
laboratory
USB and Serial ports for connectivity

MEDTOX Scientific, Inc.
Update:
Completed beta testing at 3 hospitals
Positive response
Excellent feedback on enhancements
Internal QC being performed
Based on component availability,
objective is to have production models in
second half of 2006

MEDTOX Scientific, Inc.
SURE-SCREEN®
(Government & rehab markets)

MEDTOX Scientific, Inc.
SURE-SCREEN®

MEDTOX Scientific, Inc.
SURE-SCREEN®
Overview
First “zero-tolerance”- type device on
market
Substantially lower drug detection levels
User-friendly cup device:
Fast test reaction
All results hold for 15 minutes
Initially targeting larger states & counties
FDA-510(k)-clearence in October, 2005

MEDTOX Scientific, Inc.
SURE-SCREEN®
Update:
Product launch in Q4, 2005
225 established SURE-SCREEN® clients
Accounts for 28% of Government revenue
3 State Probation/Parole departments
have committed to using SURE-SCREEN ®
Contract cycle in Government = 3 years
Oxycodone FDA 510(k)-clearance 5/06

MEDTOX Scientific, Inc.
PROFILE ® -III Cup

MEDTOX Scientific, Inc.
PROFILE ® -III Cup
Same design & operation as SURE-SCREEN ®
Traditional detection levels
On-Board Adulterant strip
FDA-510(k)-cleared product
5 to 9 panel configurations
Target markets: corporations & clinics
Launched in Q1, 2006

2006 Annual
Stockholders Meeting
May 25, 2006