Exhibit 99.2
AMAG Pharmaceuticals J.P. Morgan Healthcare Conference January 2016 AMAG Pharmaceuticals, Inc. 1100 Winter Street Waltham, MA 02451 617.498.3300 www.amagpharma.com A SPECIALTY PHARMACEUTICAL COMPANY DEDICATED TO BRINGING TO MARKET THERAPIES THAT IMPROVE PATIENTS’ LIVES Copyright © 2016 AMAG Pharmaceuticals, Inc. All Rights Reserved.

This presentation contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995 and other federal securities laws. Any statements contained herein which do not describe historical facts, including among others, statements regarding expected 2015 preliminary financial results and 2016 financial guidance, including revenues, adjusted EBITDA and cash earnings; future growth drivers for Makena, including estimated market size, the impact of the increased size of the sales force and patient support services and their effect on brand loyalty from physicians and patients and expectations for approval and commercialization of the single-dose (1 mL) preservative-free vial; expectations for the next generation development programs for Makena, including the development and advantages of a single-dose preservative free vial, a subcutaneous auto-injector and a longer-acting formulation of Makena (including the timing, cost-effectiveness, clinical trial efforts and targeted approval and commercialization related to such activities); future growth opportunities for CBR, including the impact of the increased size of the sales force, the benefits of a segmentation and pricing study that is underway and potential future medical applications in regenerative medicine; key drivers for the growth of Feraheme, including opportunities and plans to grow market share in the hospital and hematology/oncology segments, improved patient identification, and plans to realize the benefits of performance-based contracting strategies; growth opportunities for Feraheme in the CKD market through increased market share of the current U.S. indication and the potential approval of Feraheme for the broad IDA indication; plans and expectations (including timing and approval) for the head-to-head Phase 3 clinical trial for the broad IDA indication for Feraheme; and AMAG’s 2016 key priorities, including plans to achieve financial, product development and portfolio expansion objectives are forward-looking statements which involve risks and uncertainties that could cause actual results to differ materially from those discussed in such forward-looking statements. Such risks and uncertainties include, among others, the possibility that AMAG’s Phase 3 clinical trial for broad IDA indication for Feraheme will absorb significant resources, financial or otherwise over a considerable period of time and despite such efforts, might not be deemed adequate by the FDA, as well as those risks identified in AMAG’s filings with the U.S. Securities and Exchange Commission (SEC), including its Annual Report on Form 10-K for the year ended December 31, 2014, its Quarterly Reports on Form 10-Q for the quarters ended June 30, 2015 and September 30, 2015 and subsequent filings with the SEC. Any of the above risks and uncertainties could materially and adversely affect AMAG’s results of operations, its profitability and its cash flows, which would, in turn, have a significant and adverse impact on AMAG’s stock price. Use of the term “including” in this paragraph shall mean in each case “including, but not limited to.” AMAG cautions you not to place undue reliance on any forward-looking statements, which speak only as of the date they are made. AMAG disclaims any obligation to publicly update or revise any such statements to reflect any change in expectations or in events, conditions or circumstances on which any such statements may be based, or that may affect the likelihood that actual results will differ from those set forth in the forward-looking statements. AMAG Pharmaceuticals® and Feraheme® are registered trademark of AMAG Pharmaceuticals, Inc. MuGard® is a registered trademark of Abeona Therapeutics, Inc. (formerly known as PlasmaTech Biopharmaceuticals, Inc. and Access Pharmaceuticals, Inc.). Makena® is a registered trademark of Lumara Health Inc. Lumara HealthTM is a registered trademark of Lumara Health. Cord Blood Registry® and CBR® are registered trademarks of CBR Systems, Inc. Forward-Looking Statements Copyright © 2016 AMAG Pharmaceuticals, Inc. All Rights Reserved.

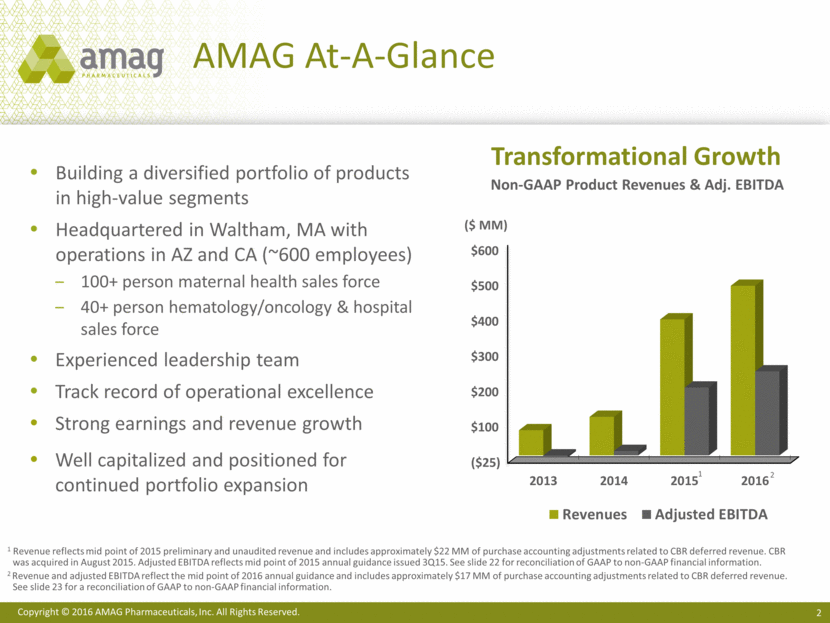
Building a diversified portfolio of products in high-value segments Headquartered in Waltham, MA with operations in AZ and CA (~600 employees) 100+ person maternal health sales force 40+ person hematology/oncology & hospital sales force Experienced leadership team Track record of operational excellence Strong earnings and revenue growth Well capitalized and positioned for continued portfolio expansion AMAG At-A-Glance Non-GAAP Product Revenues & Adj. EBITDA Transformational Growth 1 Revenue reflects mid point of 2015 preliminary and unaudited revenue and includes approximately $22 MM of purchase accounting adjustments related to CBR deferred revenue. CBR was acquired in August 2015. Adjusted EBITDA reflects mid point of 2015 annual guidance issued 3Q15. See slide 22 for reconciliation of GAAP to non-GAAP financial information. 2 Revenue and adjusted EBITDA reflect the mid point of 2016 annual guidance and includes approximately $17 MM of purchase accounting adjustments related to CBR deferred revenue. See slide 23 for a reconciliation of GAAP to non-GAAP financial information. $600 $500 $400 $300 $200 $100 ($25) ($ MM) 1 2 2013 2014 2015 2016 Revenues Adjusted EBITDA Copyright © 2016 AMAG Pharmaceuticals, Inc. All Rights Reserved.
Product Portfolio Feraheme Used for the treatment of iron deficiency anemia (IDA) in adult patients with chronic kidney disease (CKD) MuGard Prescriptive mucoadhesive for the management of oral mucositis, a common side effect of radiation or chemotherapy, affecting ~400,000 U.S. cancer patients with oral mucositis in U.S. annually1 Makena The only FDA-approved therapy to reduce recurrent preterm birth in certain at-risk women Maternal Health Business Hematology / Oncology & Hospital Business Velo Option At completion of a Phase 2b/3a clinical trial, AMAG has the option to acquire an orphan drug candidate for the treatment of severe preeclampsia Cord Blood Registry World’s largest umbilical cord stem cell collection and storage company 1 Sonis ST. Oral Oncol. 2009 Dex;45(12):1015-20. Copyright © 2016 AMAG Pharmaceuticals, Inc. All Rights Reserved.

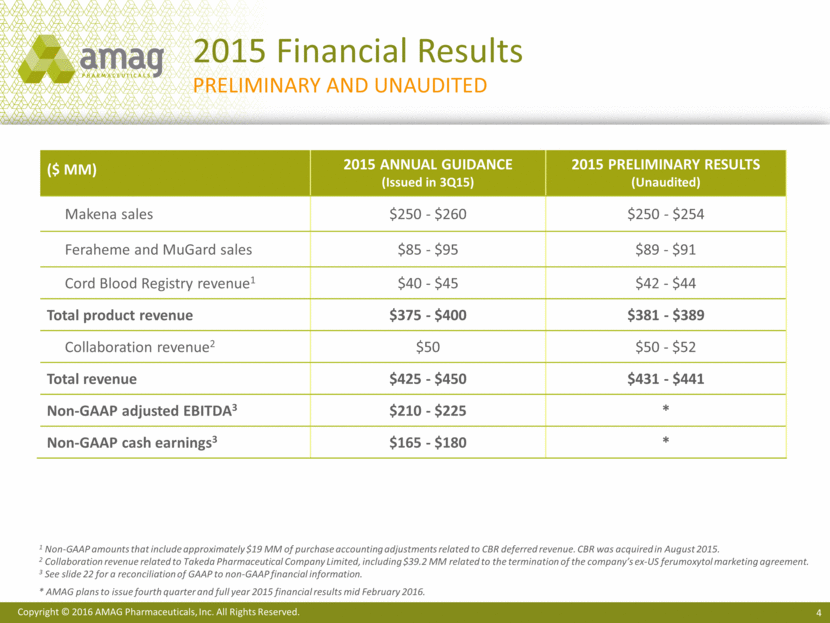
2015 Financial Results preliminary and Unaudited 1 Non-GAAP amounts that include approximately $19 MM of purchase accounting adjustments related to CBR deferred revenue. CBR was acquired in August 2015. 2 Collaboration revenue related to Takeda Pharmaceutical Company Limited, including $39.2 MM related to the termination of the company’s ex-US ferumoxytol marketing agreement. 3 See slide 22 for a reconciliation of GAAP to non-GAAP financial information. * AMAG plans to issue fourth quarter and full year 2015 financial results mid February 2016. ($ MM) 2015 Annual Guidance (Issued in 3Q15) 2015 Preliminary Results (Unaudited) Makena sales $250 - $260 $250 - $254 Feraheme and MuGard sales $85 - $95 $89 - $91 Cord Blood Registry revenue1 $40 - $45 $42 - $44 Total product revenue $375 - $400 $381 - $389 Collaboration revenue2 $50 $50 - $52 Total revenue $425 - $450 $431 - $441 Non-GAAP adjusted EBITDA3 $210 - $225 * Non-GAAP cash earnings3 $165 - $180 * Copyright © 2016 AMAG Pharmaceuticals, Inc. All Rights Reserved.
Solid execution of 2015 business plan and strong financial performance Increased Makena revenue by 52% (as compared to 2014 pro forma revenue) Increased Feraheme revenue by 5% vs. 2014 Reached 633,000+ cumulatively stored cord blood and tissue units at CBR Established and expanded maternal health business Fully integrated Lumara Health and Makena commercial team Acquired Cord Blood Registry; combined and expanded Makena and CBR sales team Acquired rights to Velo orphan drug candidate for the treatment of severe preeclampsia Expanded and strengthened executive teams 2015: hired new chief medical officer and regulatory & development personnel; hired new general counsel 2016: hired new chief commercial officer and strengthened maternal health commercial leadership 2015 Growth and Expansion Copyright © 2016 AMAG Pharmaceuticals, Inc. All Rights Reserved.

Maternal Health Copyright © 2016 AMAG Pharmaceuticals, Inc. All Rights Reserved.

2015 accomplishments: Improved access for Makena Quality of access, including: Expanded coverage to both pharmacy and medical benefit Removal of prior authorizations Implementation of treatment guidelines Breadth of access, including: Multiple new commercial plans Medicaid plans: preferred status in TX, GA, MS, OH Increased sales across all distribution channels, including within our network of compounding pharmacies which now distribute Makena in place of compounded product. Increased exit share to 35% in 2015, compared with 28% in 2014 Makena Sales Performance1 1 Preliminary and unaudited. Pro forma Makena sales for each period in 2014. Makena acquired in November 2014. $0 $40 $80 $240 $280 $200 $160 $120 2015 +52% +43% 2014 4Q15 $67.7 4Q14 ($ MM) Strong Makena Sales Growth $252.0 $165.8 $47.5 Copyright © 2016 AMAG Pharmaceuticals, Inc. All Rights Reserved.

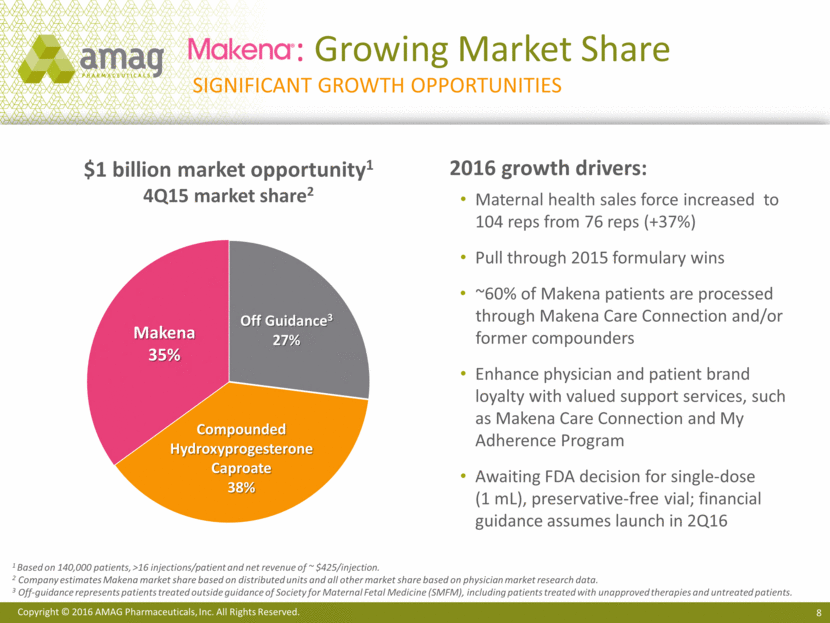
: Growing Market Share Significant Growth Opportunities $1 billion market opportunity1 4Q15 market share2 1 Based on 140,000 patients, >16 injections/patient and net revenue of ~ $425/injection. 2 Company estimates Makena market share based on distributed units and all other market share based on physician market research data. 3 Off-guidance represents patients treated outside guidance of Society for Maternal Fetal Medicine (SMFM), including patients treated with unapproved therapies and untreated patients. 2016 growth drivers: Maternal health sales force increased to 104 reps from 76 reps (+37%) Pull through 2015 formulary wins ~60% of Makena patients are processed through Makena Care Connection and/or former compounders Enhance physician and patient brand loyalty with valued support services, such as Makena Care Connection and My Adherence Program Awaiting FDA decision for single-dose (1 mL), preservative-free vial; financial guidance assumes launch in 2Q16 Compounded Hydroxyprogesterone Caproate 38% Makena 35% Off Guidance3 27% Copyright © 2016 AMAG Pharmaceuticals, Inc. All Rights Reserved.
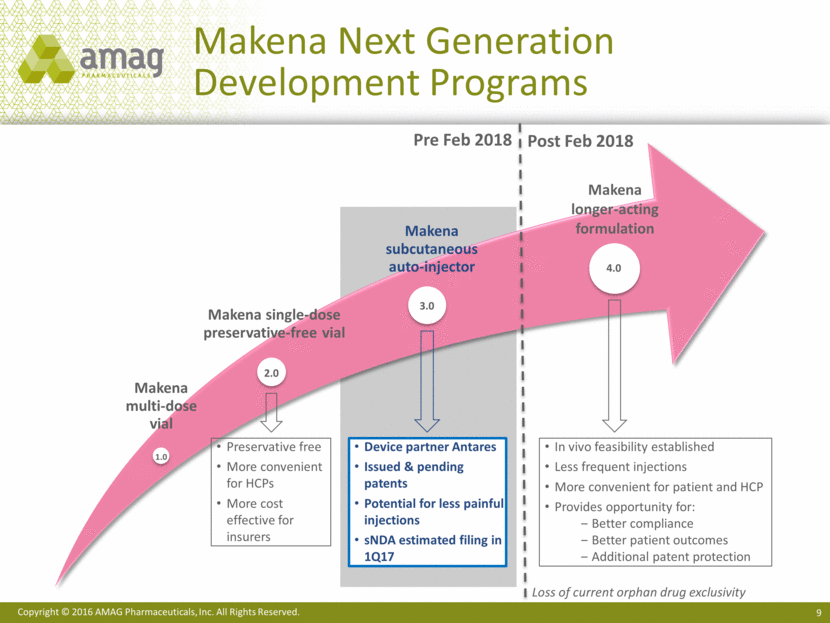
Makena Next Generation Development Programs Makena longer-acting formulation Pre Feb 2018 Post Feb 2018 Preservative free More convenient for HCPs More cost effective for insurers In vivo feasibility established Less frequent injections More convenient for patient and HCP Provides opportunity for: Better compliance Better patient outcomes Additional patent protection 1.0 2.0 3.0 4.0 Loss of current orphan drug exclusivity Device partner Antares Issued & pending patents Potential for less painful injections sNDA estimated filing in 1Q17 Makena multi-dose vial Makena single-dose preservative-free vial Makena subcutaneous auto-injector Copyright © 2016 AMAG Pharmaceuticals, Inc. All Rights Reserved.
Recurring revenue with upside potential: Customers pay: One-time, upfront collection and processing fee On-going annual storage fees (high-margin) 633,000+ units cumulatively stored Strong customer loyalty; <1% annual attrition rate Acquisition closed August 2015 4Q15 revenue decline vs. 4Q14 related to new customer discount program and sales force integration/disruption 2016 and beyond growth opportunities: More than doubled size of the sales force 47 104 sales representatives Expands reach to more obstetricians, more frequently Pricing Scaling back on 2015 discount strategy Segmentation & pricing study to refine market growth strategies Range of current clinical uses for stem cells, as well as additional potential future medical applications in regenerative medicine Pro Forma Revenue CBR Pro Forma Revenue (non-GAAP)1 $0 $75 $125 $150 $100 $50 $25 2015 2014 4Q15 4Q14 $118.6 ($ MM) 1 Preliminary and unaudited. Non-GAAP CBR revenue for each period in 2014 and 2015 includes purchase accounting adjustments related to CBR deferred revenue of $1 MM (4Q14), $12 MM (4Q15), $4 MM (2014) and $22 MM (2015). Revenues shown are pro forma for the periods 4Q14, 2014 and 2015. $126.8 $28.8 $31.4 Copyright © 2016 AMAG Pharmaceuticals, Inc. All Rights Reserved.

Hematology / Oncology and Hospital Copyright © 2016 AMAG Pharmaceuticals, Inc. All Rights Reserved.

Key Growth Drivers 2015 Accomplishments: Returned to growth in 2H15 after label changes in 1H15 Maintained volume utilization in hematology/oncology segment Continued to grow volume utilization in hospital segment 2016 Growth Drivers: Grow market share in hospital segment Maximize opportunity in hematology/oncology segment Grow overall IV iron market through improved IDA-CKD patient identification Realize benefits of performance-based contracting strategies Feraheme Sales Performance $0 $100 $80 $60 $40 $20 4Q15 4Q14 +5% +4% 2015 2014 ($ MM) 1 Includes a favorable $1.8 MM release of product return reserves. 2 Preliminary and unaudited. 1 1 2 2 $88.5 $84.4 $23.3 $22.3 Copyright © 2016 AMAG Pharmaceuticals, Inc. All Rights Reserved.

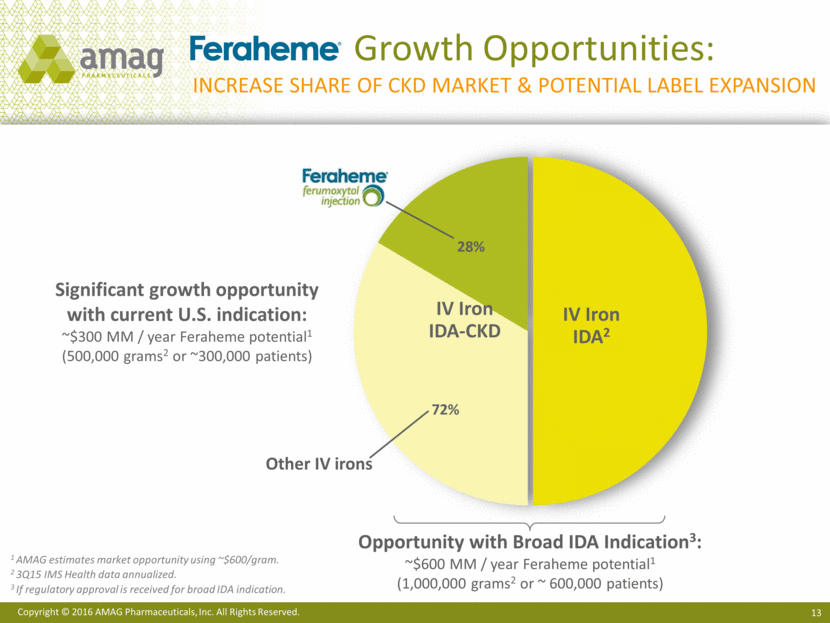
Growth Opportunities: Increase share of ckd market & potential label expansion 28% 1 AMAG estimates market opportunity using ~$600/gram. 2 3Q15 IMS Health data annualized. 3 If regulatory approval is received for broad IDA indication. Opportunity with Broad IDA Indication3: ~$600 MM / year Feraheme potential1 (1,000,000 grams2 or ~ 600,000 patients) IV Iron IDA2 72% IV Iron IDA-CKD Significant growth opportunity with current U.S. indication: ~$300 MM / year Feraheme potential1 (500,000 grams2 or ~300,000 patients) Other IV irons Copyright © 2016 AMAG Pharmaceuticals, Inc. All Rights Reserved.
Opportunity: Grow IV Iron Market 1 If regulatory approval is received for broad IDA indication. 2 Global Intravenous (I.V.) Iron Drugs Market Report: 2015 Edition. Initiating head-to-head Phase 3 clinical trial in 1Q16 to evaluate the safety of Feraheme compared to Injectafer®, with potential approval for broad IDA indication in 2018.1 Copyright © 2016 AMAG Pharmaceuticals, Inc. All Rights Reserved. 600,000 PATIENTS IV Iron IDA IV Iron IDA Oral Iron IDA-CKD patients Under the care of current AMAG call points; hematology/oncology & hospital infusion centers IV Iron IDA-CKD IV Iron IDA Oral Iron IDA patients Under the care of OB/GYNs Under the care of OB/GYNs (and other physician specialist) 4.5 MM patients diagnosed with IDA, many placed on oral iron therapy2
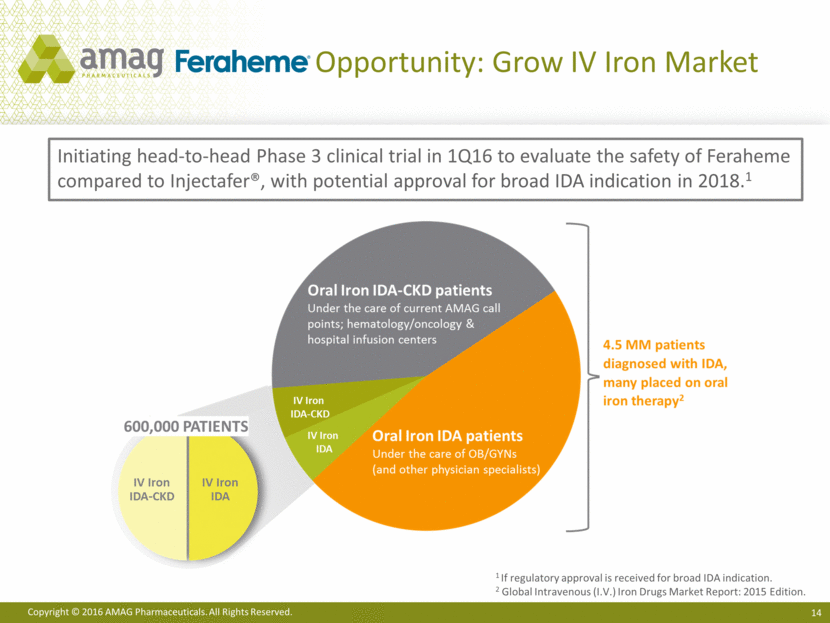
2016 Key Priorities Copyright © 2016 AMAG Pharmaceuticals, Inc. All Rights Reserved.

2016 Key Priorities Financial Grow product revenue +40% Increase non-GAAP adjusted EBITDA to >$255 MM Initiate stock repurchase program Product Development Conduct Makena PK study Initiate Feraheme IDA study Velo to complete preclinical work and initiate clinical program for severe preeclampsia option Portfolio Expansion Acquire marketed or late-stage development assets Copyright © 2016 AMAG Pharmaceuticals, Inc. All Rights Reserved.

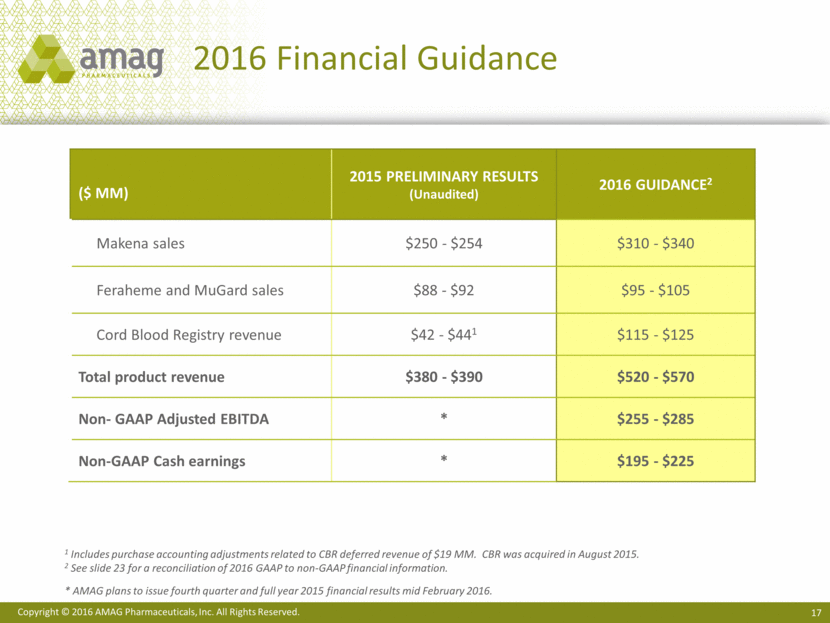
2016 Financial Guidance ($ MM) 2015 Preliminary Results (Unaudited) 2016 Guidance2 Makena sales $250 - $254 $310 - $340 Feraheme and MuGard sales $88 - $92 $95 - $105 Cord Blood Registry revenue $42 - $441 $115 - $125 Total product revenue $380 - $390 $520 - $570 Non- GAAP Adjusted EBITDA * $255 - $285 Non-GAAP Cash earnings * $195 - $225 1 Includes purchase accounting adjustments related to CBR deferred revenue of $19 MM. CBR was acquired in August 2015. 2 See slide 23 for a reconciliation of 2016 GAAP to non-GAAP financial information. * AMAG plans to issue fourth quarter and full year 2015 financial results mid February 2016. Copyright © 2016 AMAG Pharmaceuticals, Inc. All Rights Reserved.
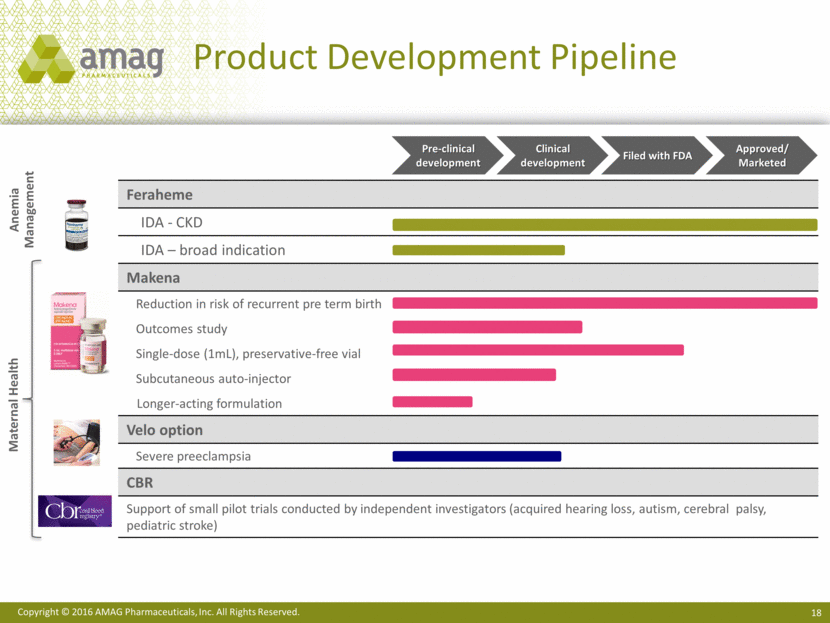
Feraheme IDA - CKD IDA – broad indication Makena Reduction in risk of recurrent pre term birth Outcomes study Single-dose (1mL), preservative-free vial Subcutaneous auto-injector Longer-acting formulation Velo option Severe preeclampsia CBR Support of small pilot trials conducted by independent investigators (acquired hearing loss, autism, cerebral palsy, pediatric stroke) Product Development Pipeline Maternal Health Anemia Management Pre-clinical development Clinical development Filed with FDA Approved/Marketed Copyright © 2016 AMAG Pharmaceuticals, Inc. All Rights Reserved.
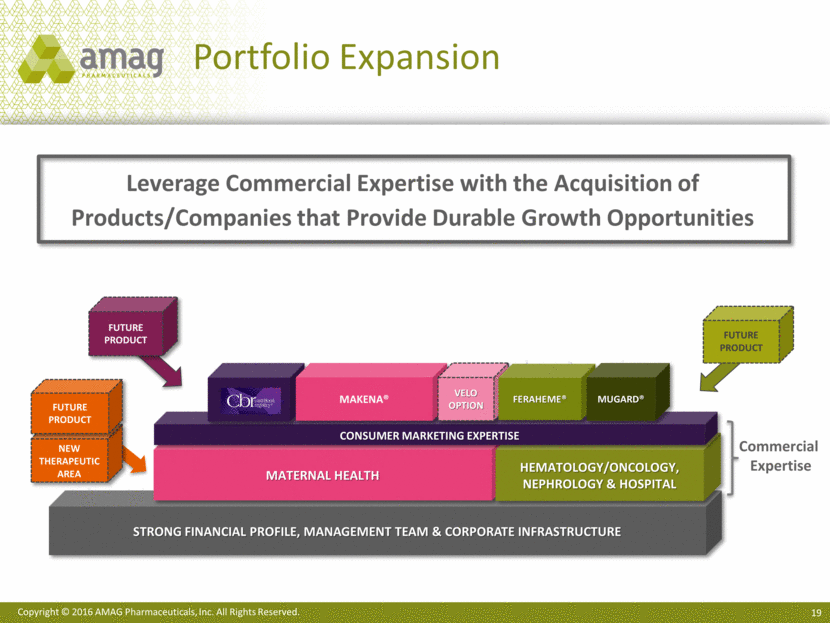
Leverage Commercial Expertise with the Acquisition of Products/Companies that Provide Durable Growth Opportunities Strong Financial profile, management team & corporate infrastructure FUTURE PRODUCT Maternal Health Hematology/Oncology, Nephrology & Hospital Consumer marketing expertise FUTURE PRODUCT CBR Commercial Expertise Makena® Velo Option Feraheme® MuGard® FUTURE PRODUCT New Therapeutic Area Portfolio Expansion Copyright © 2016 AMAG Pharmaceuticals, Inc. All Rights Reserved.
AMAG Pharmaceuticals J.P. Morgan Healthcare Conference January 2016 Copyright © 2016 AMAG Pharmaceuticals, Inc. All Rights Reserved. AMAG Pharmaceuticals, Inc. 1100 Winter Street Waltham, MA 02451 617.498.3300 www.amagpharma.com A SPECIALTY PHARMACEUTICAL COMPANY DEDICATED TO BRINGING TO MARKET THERAPIES THAT IMPROVE PATIENTS’ LIVES

AMAG Pharmaceuticals Appendix J.P. Morgan Healthcare Conference January 2016 Copyright © 2016 AMAG Pharmaceuticals, Inc. All Rights Reserved. AMAG Pharmaceuticals, Inc. 1100 Winter Street Waltham, MA 02451 617.498.3300 www.amagpharma.com A SPECIALTY PHARMACEUTICAL COMPANY DEDICATED TO BRINGING TO MARKET THERAPIES THAT IMPROVE PATIENTS’ LIVES

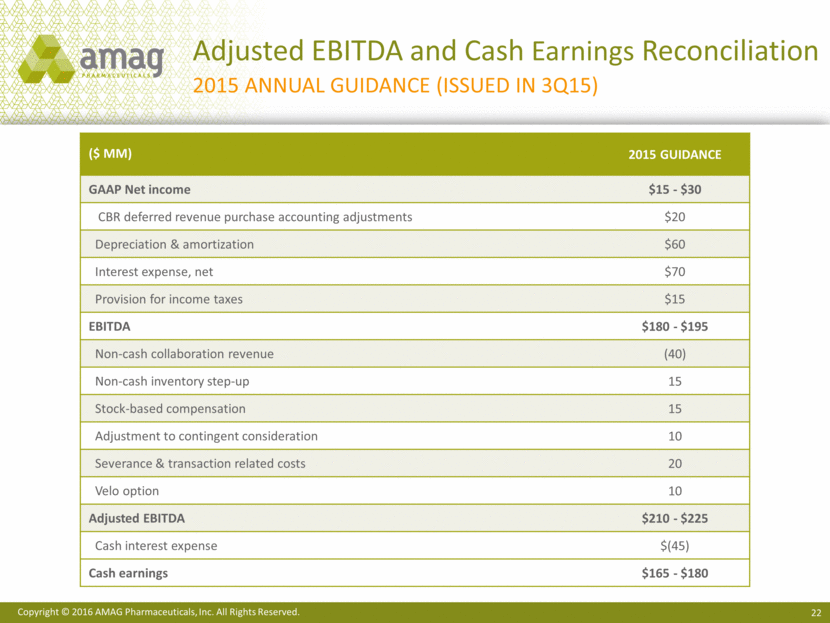
Adjusted EBITDA and Cash Earnings Reconciliation 2015 ANNUAL Guidance (Issued in 3Q15) ($ MM) 2015 Guidance GAAP Net income $15 - $30 CBR deferred revenue purchase accounting adjustments $20 Depreciation & amortization $60 Interest expense, net $70 Provision for income taxes $15 EBITDA $180 - $195 Non-cash collaboration revenue (40) Non-cash inventory step-up 15 Stock-based compensation 15 Adjustment to contingent consideration 10 Severance & transaction related costs 20 Velo option 10 Adjusted EBITDA $210 - $225 Cash interest expense $(45) Cash earnings $165 - $180 Copyright © 2016 AMAG Pharmaceuticals, Inc. All Rights Reserved.
2016 Financial Guidance Reconciliation ($ MM) 2016 Guidance GAAP Net income $11 - $41 CBR deferred revenue purchase accounting adjustments $17 Depreciation & amortization $90 Interest expense, net $72 Provision for income taxes $20 EBITDA $210 - $240 Non-cash inventory step-up $5 Stock-based compensation $27 Adjustment to contingent consideration $12 Restructuring costs $1 Adjusted EBITDA $255 - $285 Cash interest expense $(60) Cash earnings $195 - $225 Copyright © 2016 AMAG Pharmaceuticals, Inc. All Rights Reserved.

AMAG Pharmaceuticals J.P. Morgan Healthcare Conference January 2016 Copyright © 2016 AMAG Pharmaceuticals, Inc. All Rights Reserved. AMAG Pharmaceuticals, Inc. 1100 Winter Street Waltham, MA 02451 617.498.3300 www.amagpharma.com A SPECIALTY PHARMACEUTICAL COMPANY DEDICATED TO BRINGING TO MARKET THERAPIES THAT IMPROVE PATIENTS’ LIVES
