 BIOLASE Technology, Inc. Federico Pignatelli Chairman, CEO & President NASDAQ: BLTI www.biolase.com Orlando | February 9 – 12, 2012 • Gaylord Palms Hotel & Convention Center Exhibit 99.1 |
 Safe Harbor Statement This presentation may contain forward-looking statements that are based on our current expectations, estimates and projections about our industry as well as management’s beliefs and assumptions. Words such as “anticipates,” “expects,” “intends,” “plans,” “believes,” “seeks,” “estimates,” “may,” “will,” and variations of these words or similar expressions are intended to identify forward-looking statements. These statements include projections about our future earnings and margins and speak only as of the date hereof. Such statements are based upon the information available to us now and are subject to change. We will not necessarily inform you of such changes. These statements are not guarantees of future performance and are subject to certain risks, uncertainties and assumptions that are difficult to predict. Therefore our actual results could differ materially and adversely from those expressed in any forward-looking statements as a result of various factors. The important factors which could cause actual results to differ materially from those in the forward-looking statements include, among others, a downturn or leveling off of demand for our products due to the availability and pricing of competing products and technologies, adverse international market or political conditions, a domestic economic recession, the volume and pricing of product sales, our ability to control costs, intellectual property disputes, the effects of natural disasters and other events beyond our control and other factors including those detailed in BIOLASE’s filings with the Securities and Exchange Commission including its prior filings on Form 10-K and 10-Q. PAGE 2 |
 BIOLASE is Revolutionizing Surgery in Medicine • The Er,Cr:YSGG (2780nm) laser crystal has the highest level of absorption in water in human tissue. • This absorption creates an expansion and vaporization of the water molecules causing a biological ablation of human tissue with very little trauma or bleeding. • Tooth enamel ~5% water, dentin and bone ~25% water, soft tissue ~80% water, and the eye ~90% water. How? Through the science of WaterLase! PAGE 3 Conventional Surgical Devices include the scalpel, high speed drill, electrosurge, electric bone saws, and heat generating lasers. …BIOLASE is NOT Just Another Medical Device Company! |
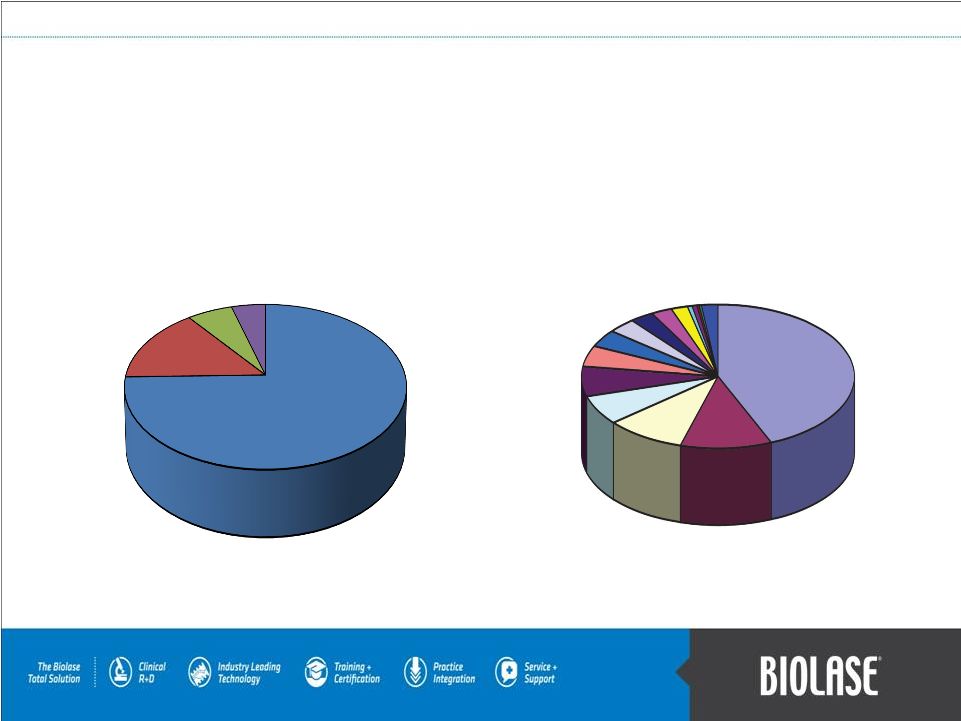 BIOLASE is the World Leader in Laser Dentistry… …and the Dental Community is the Largest Medical Community in the World! PAGE 4 BIOLASE 45% BIOLASE 45% Source: iData Research Inc. 2009 US Overall Dental Laser Market: Hard & Soft Tissue PAGE 4 BIOLASE 80% BIOLASE 80% Source: Biolase Technology Licensing Reports 2009 US Dental Laser Market: Hard Tissue |
 Medical: The Diolase 10 offers high intensity laser therapy in the palm of your hand for pain management. Digital Imaging: equipment, including cone beam 3-D dental imaging and CAD/CAM digital dental impressions. Diode soft-tissue dental lasers: Our soft tissue lasers include the ezLase total diode solution, which offers power and portability, and the revolutionary iLase, a portable diode laser with no foot pedal, power cord, or external controls. Current Line of BIOLASE Products PAGE 5 All-tissue dental lasers: revolutionary WaterLase iPlus, and the WaterLase MD Turbo. Our all-tissue lasers include our new flagship laser, the We offer a full line of digital imaging |
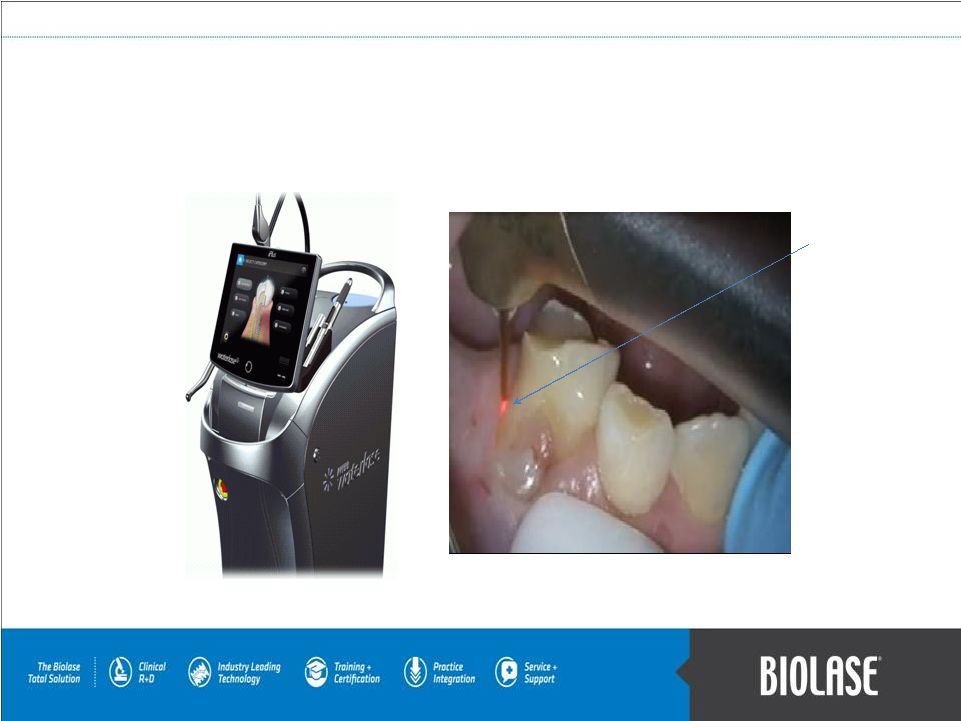 Alternative and Advanced Hi-Tech Dentistry Atraumatic Pediatric Tooth Extraction The red or green aiming beam on the WaterLase does not cut the tissue or generate any heat. The tissue is cut biologically, with little or no pain, by energizing water at the molecular level. Using BIOLASE WaterLase Systems, including our new WaterLase iPlus System: PAGE 6 The iPlus features an intuitive, touch-based computer screen, similar to a tablet personal computer. It is designed to allow for remote servicing and software upgrades. |
 WaterLase High Speed Cutting PAGE 7 |
 In 2011: Single Product Company 1. Moved from exclusive distribution with Schein to direct sales and multi-distributor model. 2. Launched iPlus which EQUALS the speed of a traditional dental drill. 3. Globalized distribution with approvals in Canada, Korea, Russia, Taiwan, & others. 4. Launched digital imaging products. 5. Growing revenues over 100%. 6. Continuing to expand global sales footprint. In 2012: Broaden Platform and Product Portfolio Becoming a Globally Diversified Platform Company 1. Diversify revenue base to expand addressable markets and reduce investor risk. 2. Launch 2 to 3 new dental products in 2Q12, including lasers and imaging. 3. Enter or begin clinical development & approval in: ophthalmology, orthopedics, pain management, podiatry, and aesthetics. 4. Re-launch revised iLase with new distribution partners. 5. Reset investor expectations… ,,,under promise, over deliver. PAGE 8 |
 Why BIOLASE WaterLase Technology in Dentistry? Traditional high-speed drills work by FRICTION which creates HEAT which causes PAIN. This PAIN necessitates injections of ANESTHESIA which results in PAIN and NUMB LIPS. In addition, the lack of feeling from the numbing injections can lead to THERMAL DAMAGE which can lead to PULPAL DEATH and result in a ROOT CANAL. The traditional high-speed drill also causes VIBRATION, which causes PAIN and leads to MICROFRACTURES. These MICROFRACTURES allow BACTERIA to penetrate the tooth which causes further DECAY and the FRACTURING of teeth. High speed drills also require the use of BURS which even after autoclaving have a 15% chance of carrying pathogenic micro-organisms. Needles and files can also carry bacteria. That means in traditional dentistry the patient has a 1 in 6 chance of cross contamination. WaterLase energy does not create heat or vibration so there is NO PAIN. Further, the WaterLase is bactericidal, antiviral and essentially eliminates the risk of cross contamination. PAGE 9 |
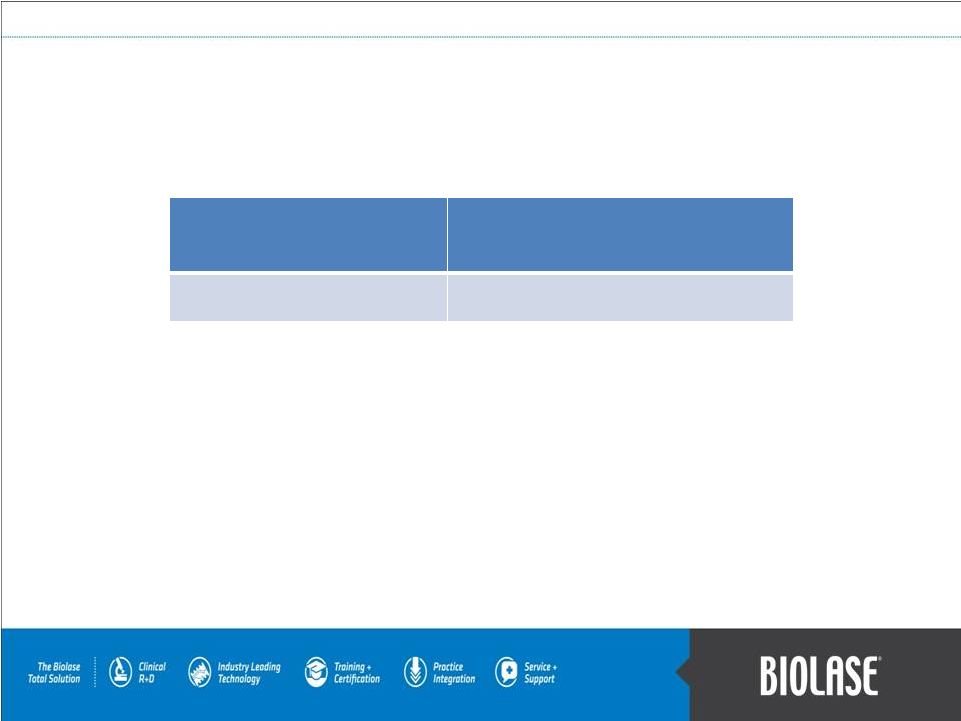 Why Dentists Should Buy an iPlus System? • WaterLase technology enables traditional dentists to perform procedures that they currently refer out to specialists or current WaterLase dentists, for example: • Gingivectomy = $160 • Perio Treatment = $375 • Hard-tissue Crown Lengthening = $520 • Herpetic or Aphthous Ulcer = $308 • Frenectomy = $355 • With no anesthesia, the WaterLase also increases efficiency and allows the dentist to work in multiple quadrants in a single visit. • This equates to $250-$750 per day in additional revenue. These procedures are easy to learn and training is included in the cost.. Monthly Lease Payment New Monthly Revenue Generated Approx. $1,000 $5,000 – $15,000 PAGE 10 A dentists’ return on investment can be between 500% and 1,500%! …WaterLase’s Tremendous Return on Investment! |
 Current Status: BIOLASE has a tremendous IP portfolio with 284 issued and pending patents, 70% of which are related to our core WaterLase (Er,Cr:YSGG) technology and medical lasers. Filing costs and maintenance are closely monitored. Funding for redundant patents and patents with a low- probability for issuance are stopped. Patent Portfolio Issued Pending Total U.S. 84 53 137 International 76 71 147 Total 160 124 284 PAGE 11 |
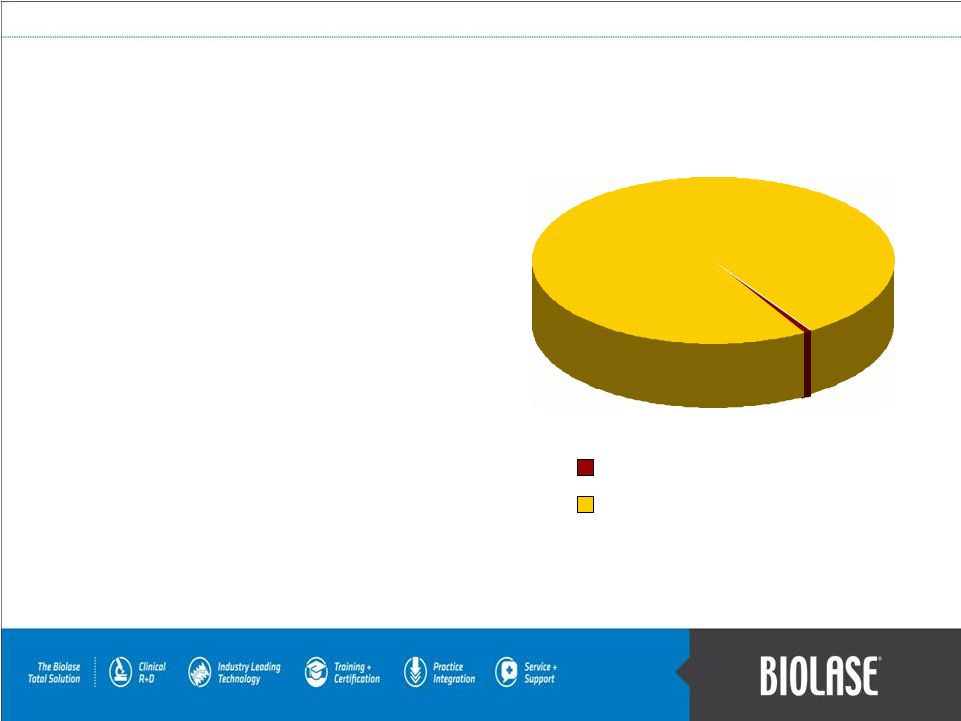 $50B to $100B WaterLase Market Opportunity The WaterLase’s global market opportunity is immense… because the dental community is the largest medical community in the world. • 176,000 dentists in the US and Canada. 1 • Over 1.2 million dentists in 134 countries and growing rapidly in emerging economies. 2 • 18,000 systems in over 11,000 practices. • Every 1% of further market penetration, just with the WaterLase, is equal to an opportunity of well over $600 million in revenues! 1. American Dental Association. 2. World Federation of Dentistry. 3. 1998-present over 17,300 BIOLASE systems sold. PAGE 12 Our current market penetration is approximately 3% in the US and slightly less than 1% internationally! Estimated total global market of 1,200,000, which is growing rapidly due to new markets such as China, India, and Indonesia. 18,000 3 systems sold worldwide. |
 1. Dental Health Magazine 2. Dental Burs and Endodontic Files: Are Routine Sterilization Procedures Effective?: Archie Morrison, DDS, MS, FRCD(C); Susan Conrod, DDS JCDA • www.cda-adc.ca/jcda • February 2009, Vol. 75, No. 1. 3. Contaminated dental instruments: Smith A, Dickson M, Aitken J, Bagg J.J Hosp Infect. 2002 Jul;51(3):233-5. 4. The antimicrobial efficacy of the erbium, chromium:yttrium-scandium-gallium-garnet laser with radial emitting tips on root canal dentin walls infected with Enterococcus faecalis: Wanda Gordon, DMD, Vahid A. Atabakhsh, DDS, Fernando Meza, DMD, Aaron Doms, DDS, Roni Nissan, DMD, Ioana Rizoiu, MS and Roy H. Stevens, DDS, MS JADA 2007; 138(7): 992-1002. 5. The Bacterial Challenge: Time to React. A call to narrow the gap between multi-drug resistant bacteria in the EU and the development of new antibacterial agents., ECDC/EMEA Joint Working Group, Stockholm, Sweden, September, 2009. Warning: Patients Urged to Ask their Dentists About the Risk of Cross Contamination! Cross contamination is a huge threat: 1,2,3,4 • The CDC defines cross-contamination as the act of spreading bacteria and viruses from one surface to another. Blood-borne viruses have the ability to live on objects and surfaces for as long as a week. • Sterilization techniques used for dental burs used with high speed drill and endodontic files are not effective. This is a serious health threat! • Many bacteria are becoming more and more resistant to antibiotics. 5 PAGE 13 |
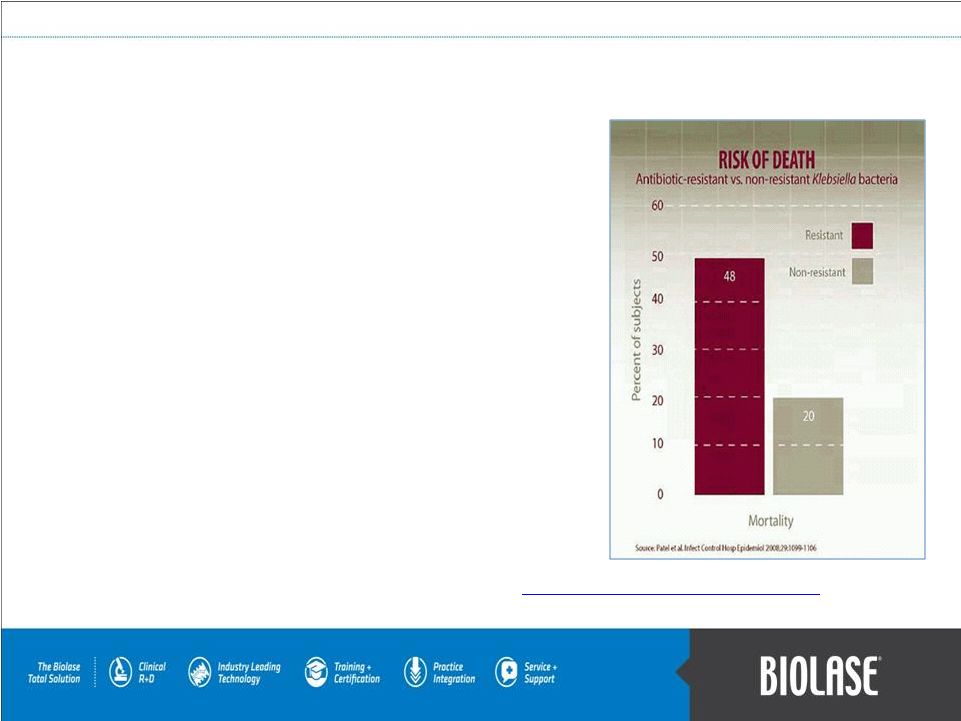 The Bacterial Challenge Antibiotic resistance adversely impacts the health of millions of patients every year. Of the patients receiving antibiotics, 50% receive unnecessary or redundant therapy, resulting in overuse of antibiotics. Resistance to antibiotics is high among bacteria that cause serious infections in humans and reaches 25% or more in several EU Member States. Each year, about 99,000 patients in the US die from hospital acquired infections and 25,000 patients in the EU die from an infection related to multidrug-resistant bacteria. Infections due to these multidrug-resistant bacteria result in extra healthcare costs and productivity losses of at least EUR 1.5 billion in the EU each year. Mortality rates of one resistant strain MRSA in the U.S. exceeded the combined death toll of AIDS, tuberculosis, and hepatitis B. 1. Centers for Disease Control and Prevention. Delivering safe care for patients: all healthcare providers play a role., November 15, 2011. Source: http://www.cdc.gov/getsmart/healthcare/learn-from-others/factsheets/hc_providers.html. 2. The Bacterial Challenge: Time to React. A call to narrow the gap between multi-drug resistant bacteria in the EU and the development of new antibacterial agents., ECDC/EMEA Joint Working Group, Stockholm, Sweden, September, 2009. 3. Klevens RM, Edwards JR, Richards CL, Horan TC. Estimating Health-Care Infections and Deaths in US Hospitals, 2002. Public Helth Reports 2007; 122: 160-166. 1 2 2 2 1 3 |
 WaterLase Tips: • Smooth tip surface does not harbor debris or bacteria like abrasive surface of burs or files. • YSGG laser energy is bacteriacidal. • Single-use, disposable tips that work without the need to contact tissue. Also eliminates accidental sticks with contaminated burs. WaterLase MD™ reduced E. faecalis 2.86 times more effectively than NaOCl³ Burs and Endo Files: • 15% of “sterilized” burs and up to 76% of “sterilized” endodontic files carry pathogenic micro-organisms. • Complex and rugged bur surface difficult to sterilize. • Autoclaving fails 15% of the time to decontaminate burs¹ •Only 32% of endodontic engine-driven files are replaced after every case and 36% after every other case. 4 BIOLASE is entering a new dental category utilizing WaterLase technology as it essentially eliminates the risk of cross contamination and contagion: PAGE 15 Eliminating the Risk of Cross Contamination 1, 2 1. Dental Burs and Endodontic Files: Are Routine Sterilization Procedures Effective?: Archie Morrison, DDS, MS, FRCD(C); Susan Conrod, DDS JCDA • www.cda-adc.ca/jcda • February 2009, Vol. 75, No. 1. 2. Contaminated dental instruments: Smith A, Dickson M, Aitken J, Bagg J.J Hosp Infect. 2002 Jul;51(3):233-5. 3. The antimicrobial efficacy of the erbium, chromium:yttrium-scandium-gallium-garnet laser with radial emitting tips on root canal dentin walls infected with Enterococcus faecalis: Wanda Gordon, DMD, Vahid A. Atabakhsh, DDS, Fernando Meza, DMD, Aaron Doms, DDS, Roni Nissan, DMD, Ioana Rizoiu, MS and Roy H. Stevens, DDS, MS JADA 2007; 138(7): 992-1002 4. Dentaltown Research: Endodontics; Survey, October 2011, dentaltown.com. 3 |
 Patented laser toothbrush for home use: • BIOLASE is currently developing a laser toothbrush for consumer home-use in connection with a license agreement with The Proctor and Gamble Company (“P&G”). • Research indicates that a laser toothbrush for home-use will whiten teeth, disinfect teeth and gums, bio- stimulate teeth and gums, relieve pain, and perform photodynamic therapy. New lasers: • BIOLASE expects to offer additional all-tissue WaterLase products in 2012 that complement the iPlus. • Planning to re-launch its handheld diode laser with several new options and applications. • Currently engineering more powerful diode soft-tissue lasers for launch in 2012. New Imaging products: • BIOLASE expects to offer additional imaging products in 2012 that complement its current products. Further Opportunities to Expand in Dentistry PAGE 16 |
 • iPlus sales will continue grow rapidly as new products ramp enhancing revenue visibility. • Disposables and consumables will continue to represent ~20% of total sales. • New products will expand BIOLASE’s addressable markets 2 to 3 times. • BIOLASE will re-launch its handheld diode laser with new distributor partners in early 2012. 2011 (Projected) 2012 (Projected) WaterLase Systems ~60% ~50% Diode Systems ~19% ~15% Consumables & Other ~20% ~20% New ~1% ~15% BIOLASE will diversify in 2012 and expand product sales beyond dentistry. Diversification in 2012 PAGE 17 |
 With our patented WaterLase and diode technologies, we have created technological platforms that have the ability to extend far outside of dentistry. We expect to greatly expand our addressable markets in the coming years which, we believe, has the potential to substantially increase our revenues. Each of these potential markets represents a multi-billion dollar opportunity. Ophthalmology: We currently hold 14 issued and 19 pending U.S. and International patents in four patent families in the field of ophthalmology, giving us a wide range of applications and coverage. Our patented technology has the capability to restore the elasticity of the eye and allow it to return to normal function, eliminating presbyopia. Management has established a new subsidiary, OCULASE, which will own and develop BIOLASE’s ophthalmic assets and technologies. In 2012, we expect approval to market treatments for glaucoma and dry eyes. We expect approval to market WaterLase technology for presbyopia internationally in 2013 and in the United States in 2014. 2012: Expanding Beyond Dentistry PAGE 18 |
 Aesthetics: We have various FDA approvals for applications in dermatology, plastic surgery, and oculoplastics and are currently investigating options for entering these markets in 2012. Pain Management: We anticipate launching a new deep-tissue hand piece and upgraded laser for pain therapy in 2012 which will coincide with a new marketing campaign. Podiatry: We have found that our Diolase 10 technology is very effective in the treatment of nail fungus and we are completing the clinical and regulatory requirements necessary to enter the market in 2012. Orthopedics: We are working with several key manufacturers and universities to provide solutions that are not currently available. We are investigating opportunities for several orthopedic applications and anticipate filing several 510(k) applications over the next 12 months. PAGE 19 2012: Expanding Beyond Dentistry (Continued) |
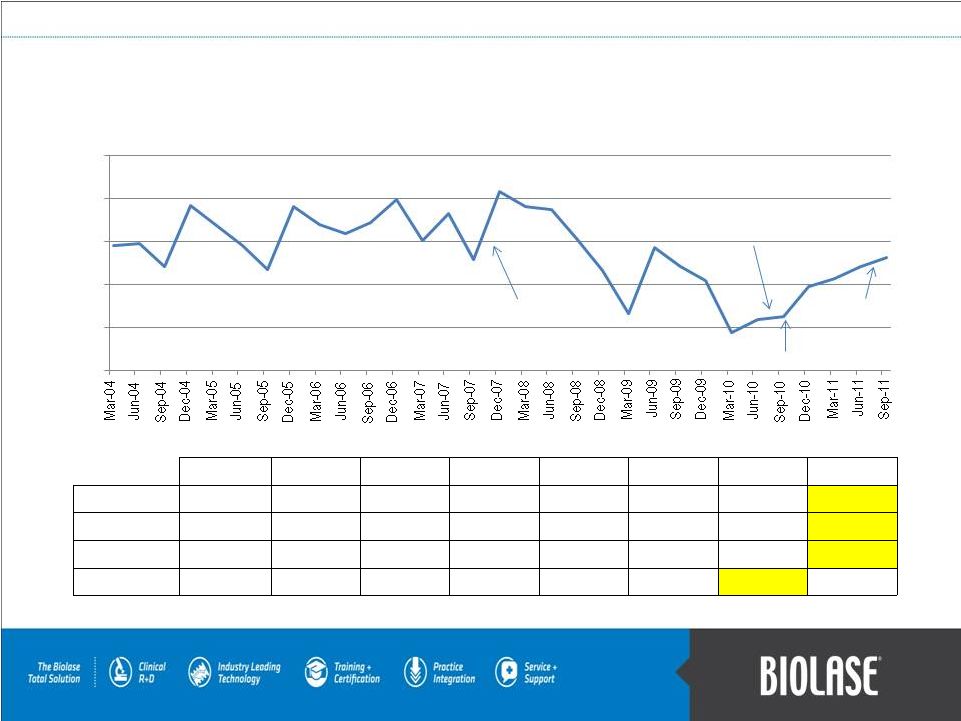 Quarterly Revenues (in Millions) PAGE 20 Federico Pignatelli restored as Chairman & CEO in Aug 2010 Record quarter with Federico Pignatelli interim CEO during Q407 2004 2005 2006 2007 2008 2009 2010 2011 Q1-Mar $ 14.5 $ 16.8 $ 16.9 $ 15.1 $ 19.0 $ 6.6 $ 4.4 $ 10.6 Q2-Jun $ 14.7 $ 14.5 $ 15.9 $ 18.2 $ 18.7 $ 14.3 $ 5.9 $ 12.1 Q3-Sep $ 12.0 $ 11.7 $ 17.1 $ 12.8 $ 15.3 $ 12.1 $ 6.2 $ 13.1 Q4-Dec $ 19.1 $ 19.0 $ 19.8 $ 20.8 $ 11.6 $ 10.4 $ 9.7 $- $5.0 $10.0 $15.0 $20.0 $25.0 Direct sales ramp begins in Sep 2010 Over 100% revenue growth to date in 2011 compared to 2010 |
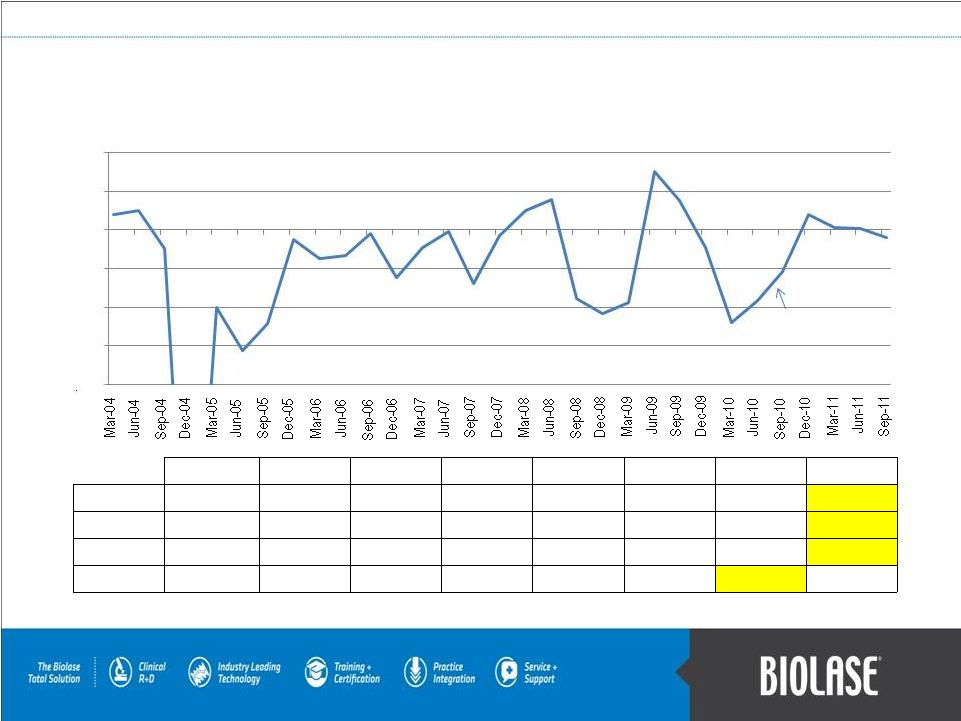 Quarterly Non-GAAP Net Income (in Millions) 2004 2005 2006 2007 2008 2009 2010 2011 Q1-Mar $ 0.783 $ (4.015) $ (1.462) $ (0.890) $ 0.985 $ (3.763) $ (4.797) $ 0.153 Q2-Jun $ 1.006 $ (6.222) $ (1.340) $ (0.089) $ 1.578 $ 3.020 $ (3.657) $ 0.069 Q3-Sep $ (0.962) $ (4.820) $ (0.167) $ (2.765) $ (3.551) $ 1.534 $ (2.173) $ (0.397) Q4-Dec $(23.257) $ (0.479) $ (2.466) $ (0.272) $ (4.338) $ (0.888) $ 0.787 Adjustments from net income (loss) include: depreciation and amortization, non-cash stock-based compensation expense, and interest expense. PAGE 21 $(8.0) $(6.0) $(4.0) $(2.0) $- $2.0 $4.0 Federico Pignatelli restored as Chairman & CEO in Aug 2010 |
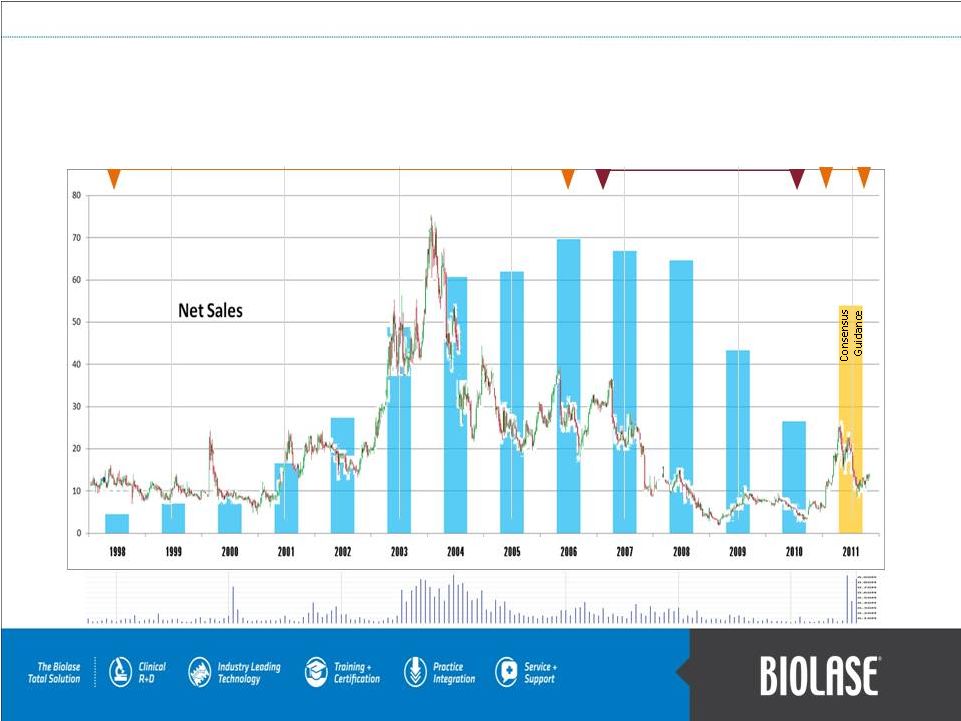 BLTI Revenues Chart & Financial History 1994-2006 Pignatelli Chairman of the Board Exclusive distribution partnership with Henry Schein Aug 2006–Aug 2010 Aug 2010-Present Pignatelli CEO & Board Chairman 1998 FDA Clearance WaterLase PAGE 22 Volume: Millions of Shares 20 15 10 5 BLTI Share Price |
 BIOLASE Technology, Inc. Today Company Headquarters in Irvine, California PAGE 23 • We have over 180 employees worldwide. • We also have manufacturing capabilities in Floss, Germany. • We have sales offices in the U.S., Floss (Germany), Madrid (Spain), Shanghai (China), and Mumbai (India), with expansion planned in Dubai (UAE), and Rio de Janero (Brazil). We also have training facilities in Charlotte, NC, and Irvine, CA. BIOLASE Europe in Floss, Germany • Our 57,000 sq. ft. corporate HQ in Irvine houses finance & administrative, sales, marketing, customer care, training, manufacturing, and R&D. This facility can accommodate growth to $250 million. • ISO 9001 certified and FDA GMP with clean room operations. |