 Fred Furry, COO & CFO January 7, 2013 Nasdaq: BIOL PAGE 1 EXHIBIT 99.1 |
 This presentation may contain forward-looking statements that are based on our current expectations, estimates and projections about our industry as well as management’s beliefs and assumptions. Words such as “anticipates,” “expects,” “intends,” “plans,” “believes,” “seeks,” “estimates,” “may,” “will,” and variations of these words or similar expressions are intended to identify forward-looking statements. These statements include projections about our future earnings and margins and speak only as of the date hereof. Such statements are based upon the information available to us now and are subject to change. We will not necessarily inform you of such changes. These statements are not guarantees of future performance and are subject to certain risks, uncertainties and assumptions that are difficult to predict. Therefore our actual results could differ materially and adversely from those expressed in any forward-looking statements as a result of various factors. The important factors which could cause actual results to differ materially from those in the forward-looking statements include, among others, a downturn or leveling off of demand for our products due to the availability and pricing of competing products and technologies, adverse international market or political conditions, a domestic economic recession, the volume and pricing of product sales, our ability to control costs, intellectual property disputes, the effects of natural disasters and other events beyond our control and other factors including those detailed in BIOLASE’s filings with the Securities and Exchange Commission including its prior filings on Form 10-K and 10-Q. PAGE 2 Safe Harbor Statement |
 • We are a biomedical company focused on developing, manufacturing, and selling dental lasers using our revolutionary, proprietary WaterLase technology. • We also market and distribute: – dental imaging equipment, – intra-oral scanners, and – other products designed for dental & medical procedures. • Net revenue of $49M in 2011; up 87% over 2010. • Projecting net revenue >$18M for Q4 and gross revenue >$57.4M for 2012. BIOLASE Company Overview PAGE 3 – up 34% over adjusted net revenues of $43M in 2011, which excludes prepaid purchase orders to Schein totaling $5.9M. 2012 gross revenue excludes $1.1M inventory repurchase in Q2. |
 Step Technology BIOLASE Product Offering Diagnose and establish treatment protocol 3D cone beam imaging • NewTom (QR/Cefla) • DaVinci Imaging (Cefla) Treatment: all tissue surgical device WaterLase • WaterLase iPlus • WaterLase MDX • WaterLase MD Turbo Treatment: soft tissue surgical device Diode laser • EPIC 10 diode laser • iLase portable, battery operated diode laser Impression taking for crowns Digital • TRIOS intra-oral scanner (3Shape) PAGE 4 BIOLASE’s Total Technology Solution |
 • WaterLase is a biological method to cut tissue. • Water molecules within tissue absorb the laser energy from the WaterLase, expand, and vaporize, which causes a biological ablation of the tissue. – There is very little trauma or bleeding, – No heat or vibration are generated, and – Causes little or no pain. – Significantly reduces cross contamination. WaterLase All-Tissue Lasers PAGE 5 • Extensive patent protection. |
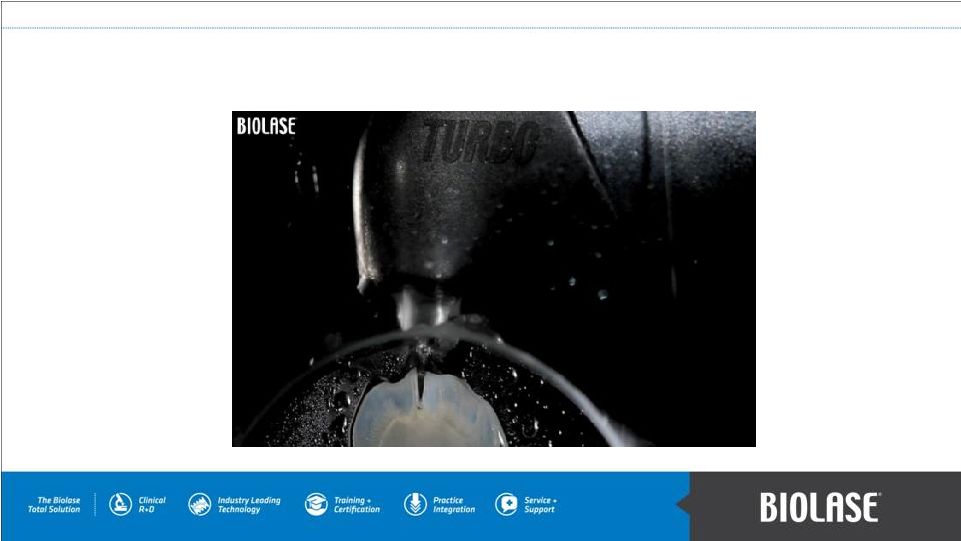 How It Works: WaterLase High Speed Cutting PAGE 6 |
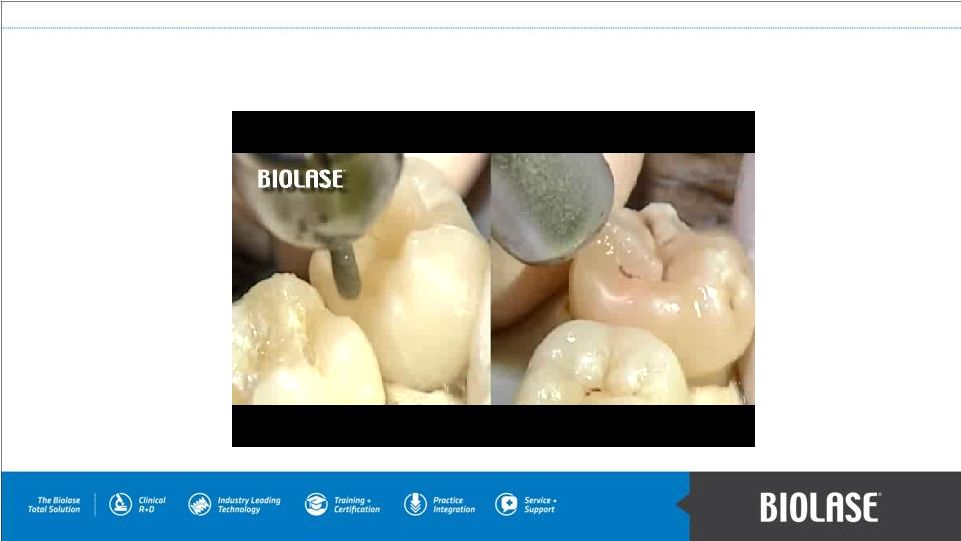 WaterLase Side by Side with Drill PAGE 7 |
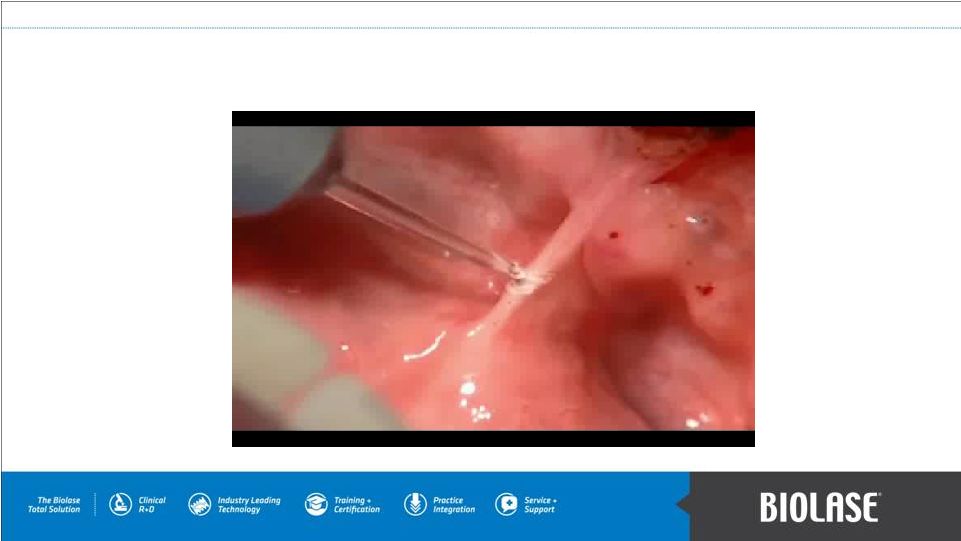 WaterLase Soft Tissue Lingual Frenectomy PAGE 8 |
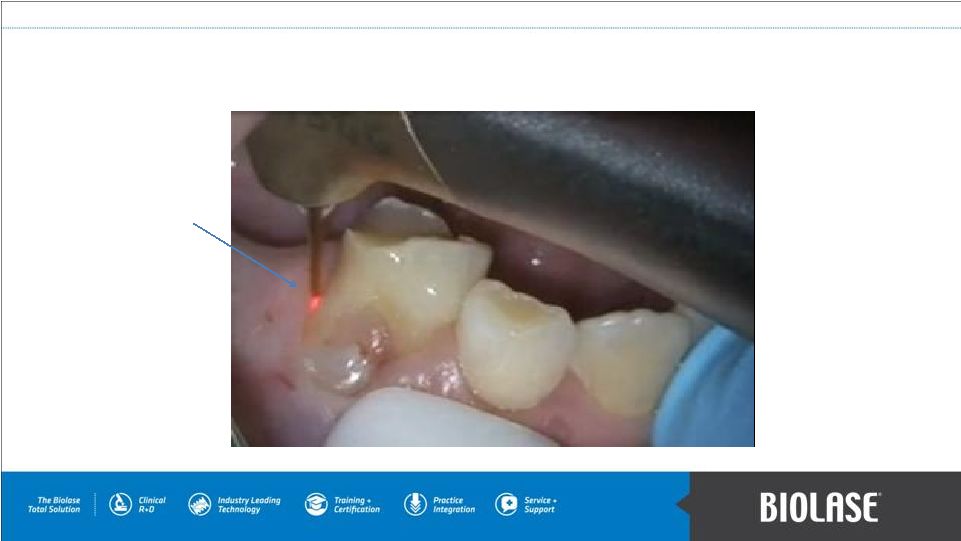 WaterLase Atraumatic Pediatric Tooth Extraction The WaterLase aiming beam does not cut the tissue or generate heat. The tissue is cut biologically, with little or no pain, when the laser energizes water at the molecular level. PAGE 9 |
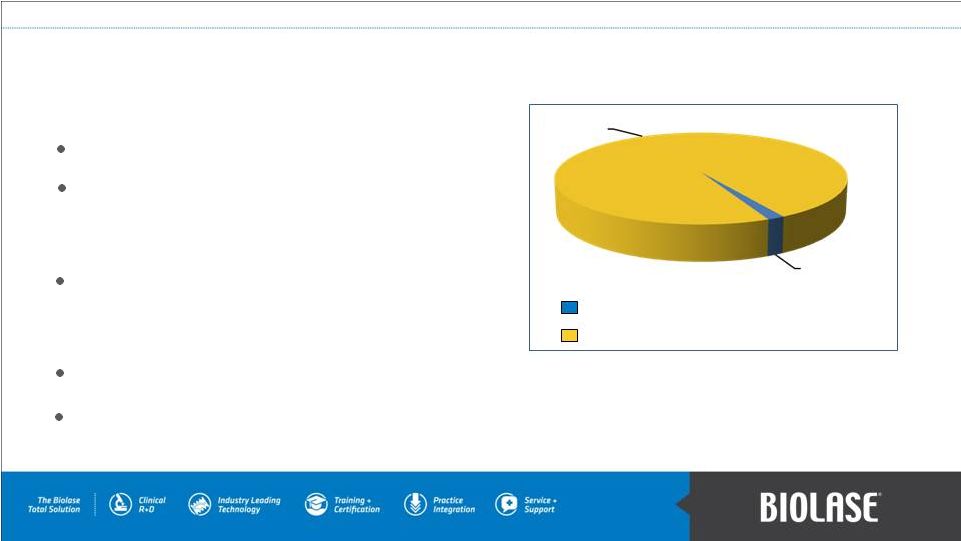 176,000 dentists in US & Canada. 1.2M+ dentists in 134 countries. – rapid growth in emerging economies e.g. China, India & Indonesia. Current market penetration. – approx. 5.0% of dental practices in US. – approx. 1.5% worldwide. Each incremental 1% market penetration equals over $600M revenue. Dental laser market opportunity is in excess of $50B. BIOLASE’s Market Opportunity 1 American Dental Association. 2 World Federation of Dentistry. PAGE 10 Est. total global market BIOLASE systems sold worldwide 1998-present 20,000+ 1,200,000 1 2 |
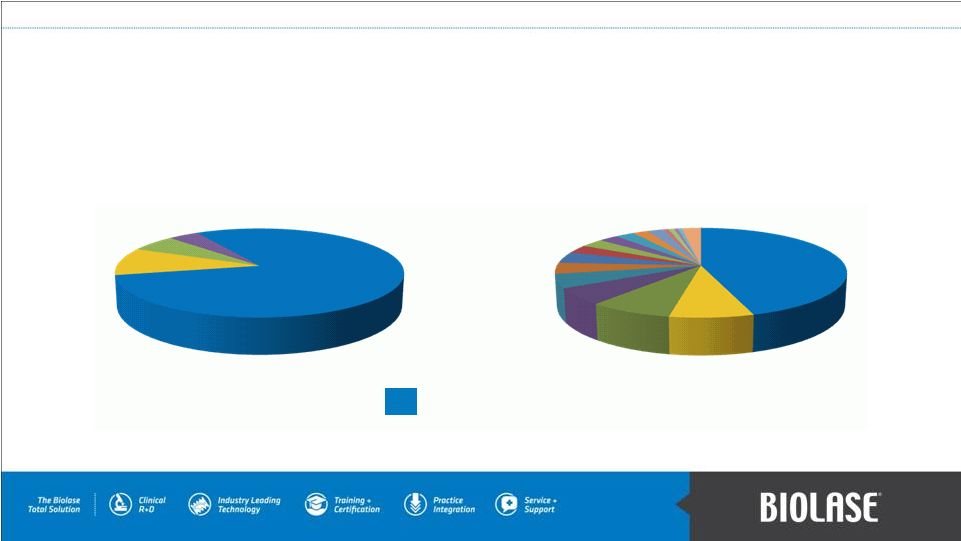 Dominant Market Position Hard-tissue Dental Laser Market Total Dental Laser Market 80% 45% BIOLASE Market Share PAGE 11 |
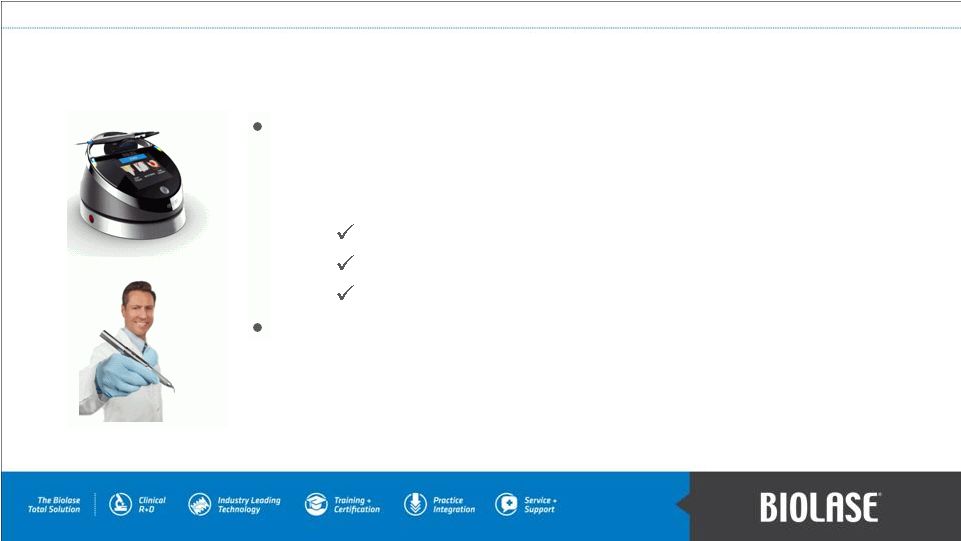 EPIC 10 total diode solution – regulatory clearance late Q3 (EU) & early Q4 (US) 2012. – cleared for 3 unique uses: Soft Tissue Surgery, Whitening, Pain Therapy. iLase – portable diode laser. – no foot pedal, power cord, or external controls. Diode Soft-Tissue Dental Lasers PAGE 12 |
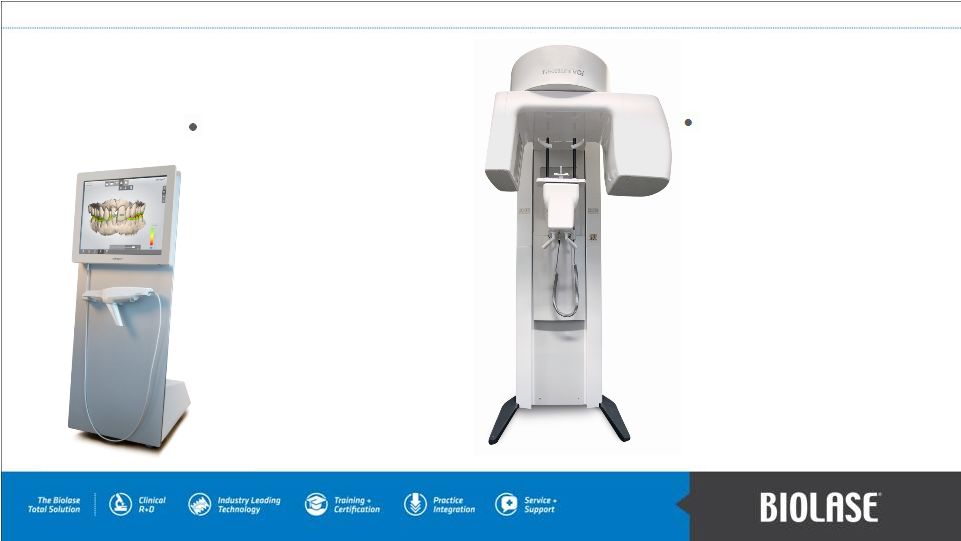 TRIOS intra-oral scanner and cart: – digital impression taking. – handheld scanner, operator's control cart, and software. – North American distribution agreement through Aug 2017. In-Licensed Products NewTom and Cefla 3D cone beam imaging products: – medical grade imaging technology. – less cost, less radiation exposure. – U.S. & Canada distribution agreement through Feb 2015. PAGE 13 |
 PAGE 14 • 291 patents issued & pending. • 70% are related to WaterLase technology & medical lasers. Extensive Patent Portfolio Issued & Active Pending Total U.S. 81 52 133 International 81 77 158 Total 162 129 291 |
 Returned to direct sales model in North America in August 2010. Currently consists of 47-person team: – 37 direct outside sales. – 10 inside sales representatives and lead generators. Targeted plans for 42-45 outside reps and 12-15 inside reps. Global sales offices: – Current: Irvine, CA (USA), Floss (Germany), Madrid (Spain),Shanghai (China), and Mumbai (India). – Planned: Dubai (UAE) and Rio de Janero (Brazil). Sales and Marketing PAGE 15 |
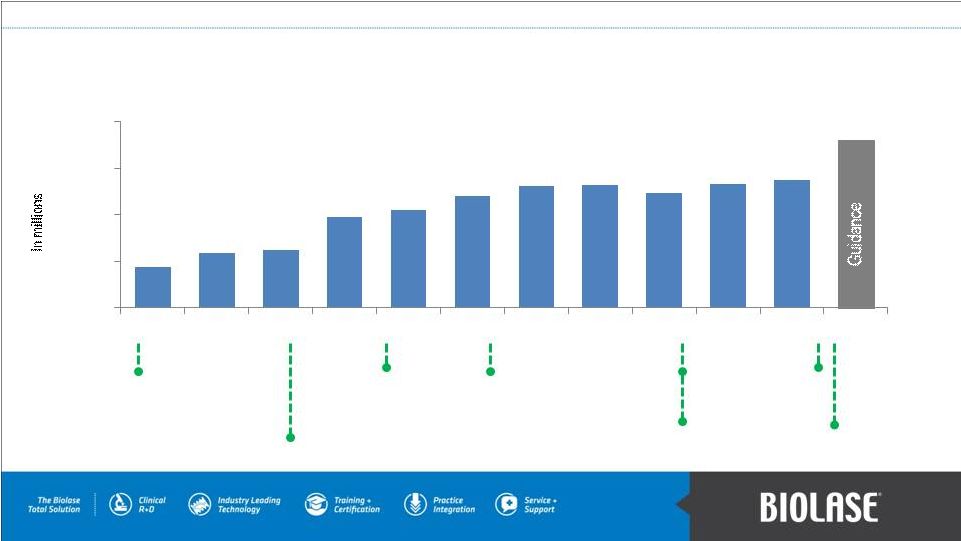 Gross Revenue and Influencers Launched Cefla’s NewTom Cone Beam Imaging products Introduced iLase™ hand-held soft- tissue diode laser Introduced DaVinci Imaging™ products Launched WaterLase ® iPlus™ all-tissue laser Launched 3Shape’s TRIOS™ intra-oral scanner CE Mark & FDA clearance for EPIC 10 F. Pignatelli becomes Chairman and CEO; Ended exclusive global distribution agreement with Henry Schein, Inc. Introduced WaterLase ® MDX™ all- tissue lasers PAGE 16 $4.4 $5.9 $6.2 $9.7 $10.6 $12.1 $13.1 $13.2 $12.3 $13.3 $13.8 >$18M $0.0 $5.0 $10.0 $15.0 $20.0 Q1 '10 Q3 '10 Q4 '10 Q1 '11 Q2 '11 Q3 '11 Q4 '11 Q1 '12 Q2 '12 Q3 '12 Q4 '12 Q2 '10 |
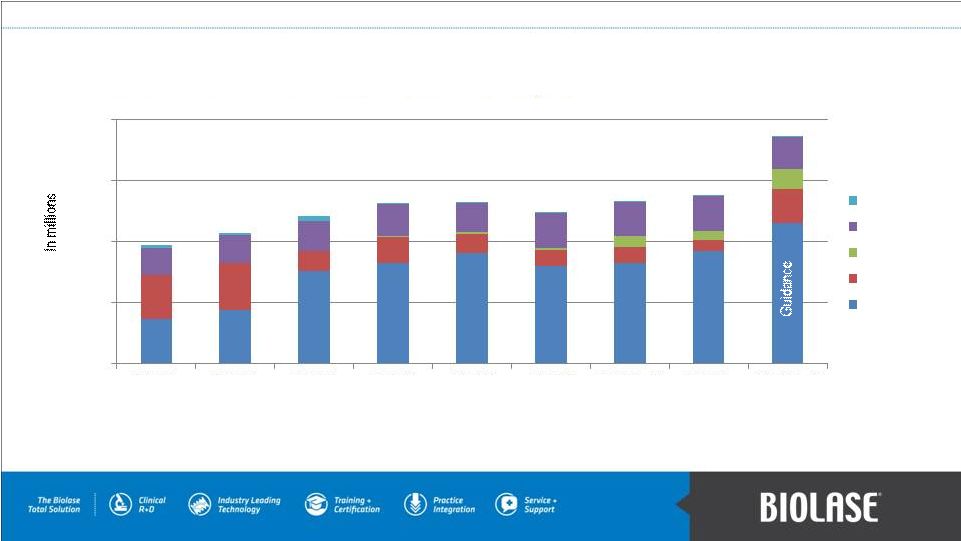 Gross Revenue Trend Analysis (1) Represents gross revenue for Q2 2012, which does not reflect $1.1 million inventory repurchase from Henry Schein, Inc. (2) Estimated revenue guidance of >$18M for Q4 2012, last revised January 7, 2013. PAGE 17 $- $5 $10 $15 $20 Q4 2010 Q1 2011 Q2 2011 Q3 2011 Q4 2011 Q1 2012 Q2 2012 (1) Q3 2012 Q4 2012 (2) Royalties Other Imaging Diodes WaterLase |
 • Continued adoption of core laser products. – The WaterLase product line, particularly the iPlus, has demonstrated a solid growth trend over the past 8 quarters. – Significant demad for our EPIC 10 diode soft tissue laser. • Growth of Imaging Division (cone beam and cad-cam). • Additional new product introductions based on our core technologies. – Two 510(k) submissions currently under FDA review. – Three additional submissions planned to be filed in near term. – More submissions planned for later in 2013. • Expand into new markets beyond dentistry. Growth Strategy PAGE 18 |
 • Ongoing growth in core WaterLase sales and trend of adoption. • High demand for our new EPIC 10 soft tissue diode laser. • Recent & planned new product introductions. – EPIC 10 & TRIOS introduced second half of 2012. – Additional product launches based on core technologies planned for 2013 and beyond. • Strong IP protection with additional patents pending. • Addressing $50B dental laser market opportunity. – Large market with little competition for all-tissue lasers. – Significant opportunities for WaterLase in other medical markets. • Improved financial position. Investment Considerations PAGE 19 |
 PAGE 20 • Over 180 employees worldwide. • Sales offices in Floss (Germany), Madrid (Spain), Shanghai (China), and Mumbai (India); expansion planned in Dubai (UAE), and Rio de Janero (Brazil). • Floss also has service and manufacturing capabilities. BIOLASE Europe in Floss, Germany • Corporate HQ: Irvine, California. • 57,000 sq. ft. facility houses finance & administrative, sales, marketing, customer care, training, manufacturing, and R&D. • Can accommodate growth to $250 million. BIOLASE Today BIOLASE Corporate Headquarters in Irvine, California |
 Fred Furry COO & CFO, BIOLASE, Inc. 949.226.8124 ffurry@biolase.net PAGE 21 Lisa Wilson President, In-Site Communications, Inc. 212.759.3929 lwilson@insitecony.com |