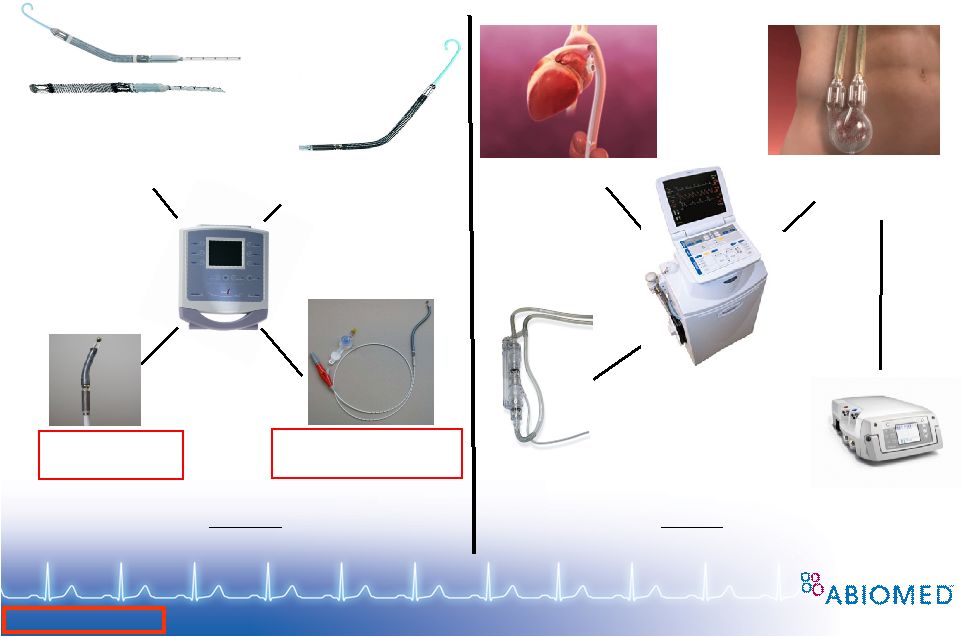
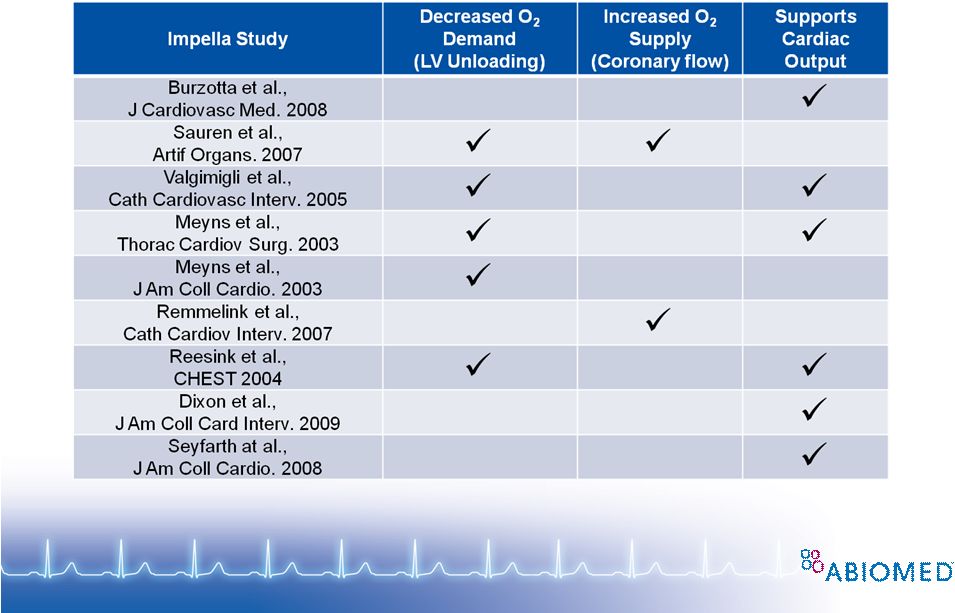
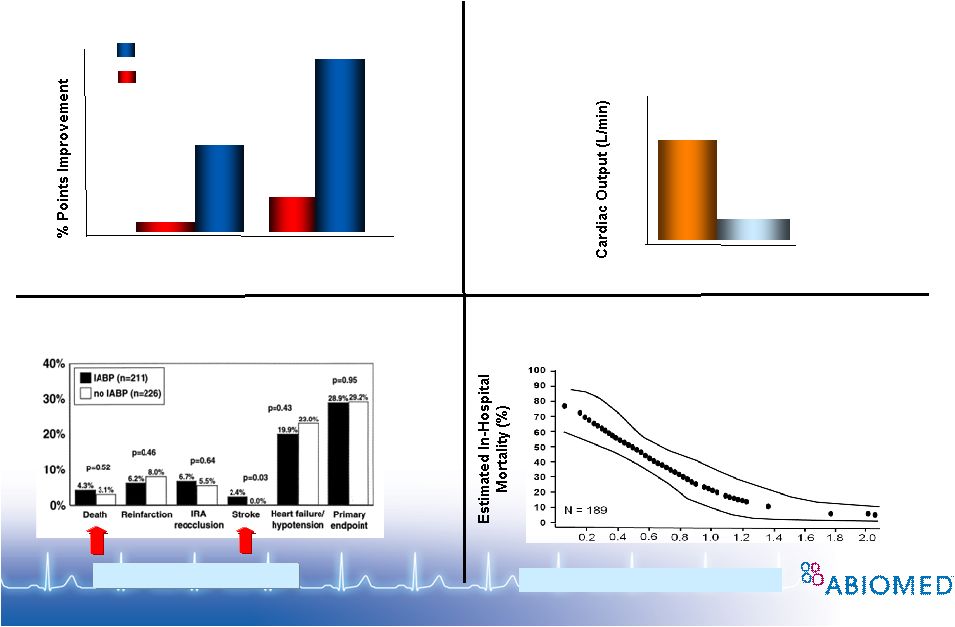
Free signup for more
- Track your favorite companies
- Receive email alerts for new filings
- Personalized dashboard of news and more
- Access all data and search results
Filing tables
Filing exhibits
Related financial report
ABMD similar filings
- 31 Jul 09 Other Events
- 22 Jul 09 Other Events
- 3 Jun 09 Departure of Directors or Certain Officers
- 18 May 09 Results of Operations and Financial Condition
- 12 May 09 Abiomed Announces Fiscal Fourth Quarter Revenue of $19.6 Million
- 23 Apr 09 Abiomed receives FDA 510(k) Clearance for Impella® 5.0 and Impella® LD
- 21 Apr 09 Departure of Directors or Certain Officers
Filing view
External links