
Corporate Presentation November 9, 2017 Exhibit 99.2

Forward-Looking Statements This presentation contains "forward-looking statements" as defined by the Private Securities Litigation Reform Act of 1995. We caution investors that forward-looking statements are based on management’s expectations and assumptions as of the date of this presentation, and involve substantial risks and uncertainties that could cause our clinical development programs, future results, performance or achievements to differ significantly from those expressed or implied by the forward-looking statements. These risks and uncertainties include, but are not limited to, those associated with: the potential market opportunity and net sales for SUSTOL® and CINVANTITM; the timing of completion and results of the Phase 2 and Phase 3 trials for HTX-011; the timing of the NDA filing for HTX-011; and other risks and uncertainties identified in the Company's filings with the Securities and Exchange Commission. Forward-looking statements reflect our analysis only on their stated date, and we take no obligation to update or revise these statements except as may be required by law.

CINVANTI™ Now Approved CINVANTI™ is the first and only polysorbate 80-free IV NK1 receptor antagonist approved for the prevention of both acute and delayed CINV CINVANTI™ is indicated in adults, in combination with other antiemetic agents, for the prevention of acute and delayed nausea and vomiting associated with initial and repeat courses of highly emetogenic cancer chemotherapy (HEC) including high-dose cisplatin and nausea and vomiting associated with initial and repeat courses of moderately emetogenic cancer chemotherapy (MEC). Please see Full Prescribing Information on www.CINVANTI.com
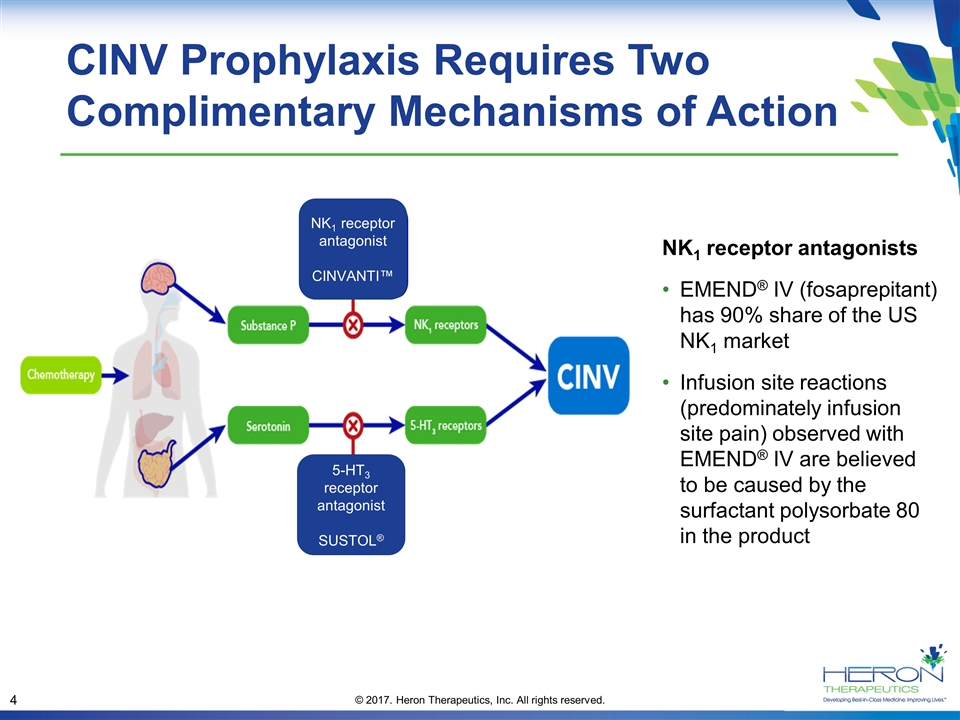
CINV Prophylaxis Requires Two Complimentary Mechanisms of Action NK1 receptor antagonists EMEND® IV (fosaprepitant) has 90% share of the US NK1 market Infusion site reactions (predominately infusion site pain) observed with EMEND® IV are believed to be caused by the surfactant polysorbate 80 in the product NK1 receptor antagonist CINVANTI™ 5-HT3 receptor antagonist SUSTOL®
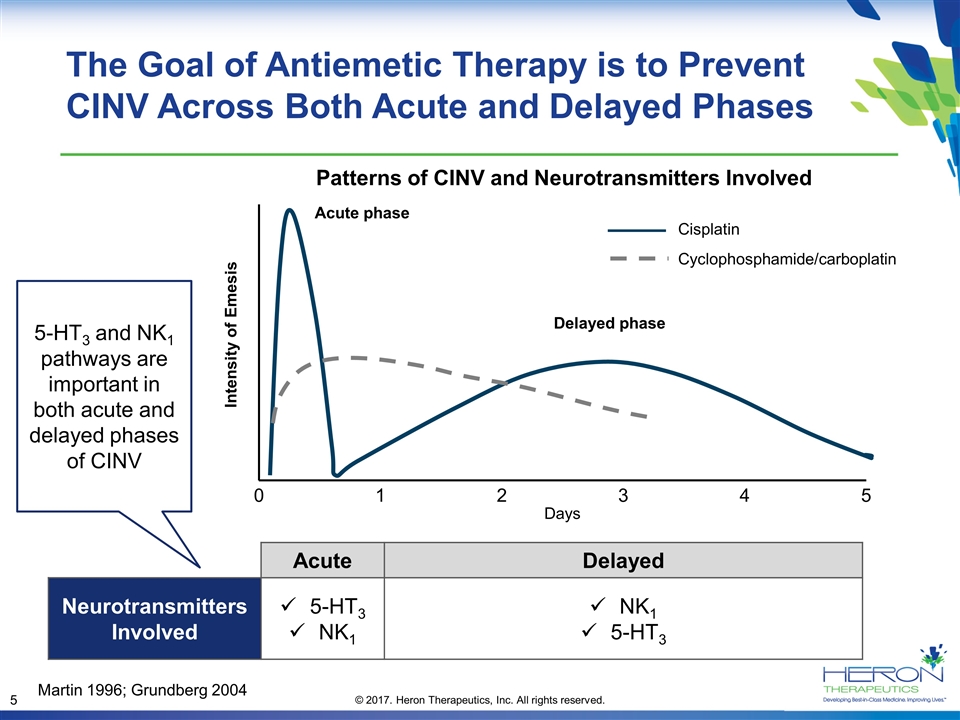
The Goal of Antiemetic Therapy is to Prevent CINV Across Both Acute and Delayed Phases Acute Delayed Neurotransmitters Involved 5-HT3 NK1 NK1 5-HT3 5-HT3 and NK1 pathways are important in both acute and delayed phases of CINV Martin 1996; Grundberg 2004 Patterns of CINV and Neurotransmitters Involved Cisplatin Cyclophosphamide/carboplatin Acute phase Delayed phase Intensity of Emesis 0 1 2 3 4 5 Days
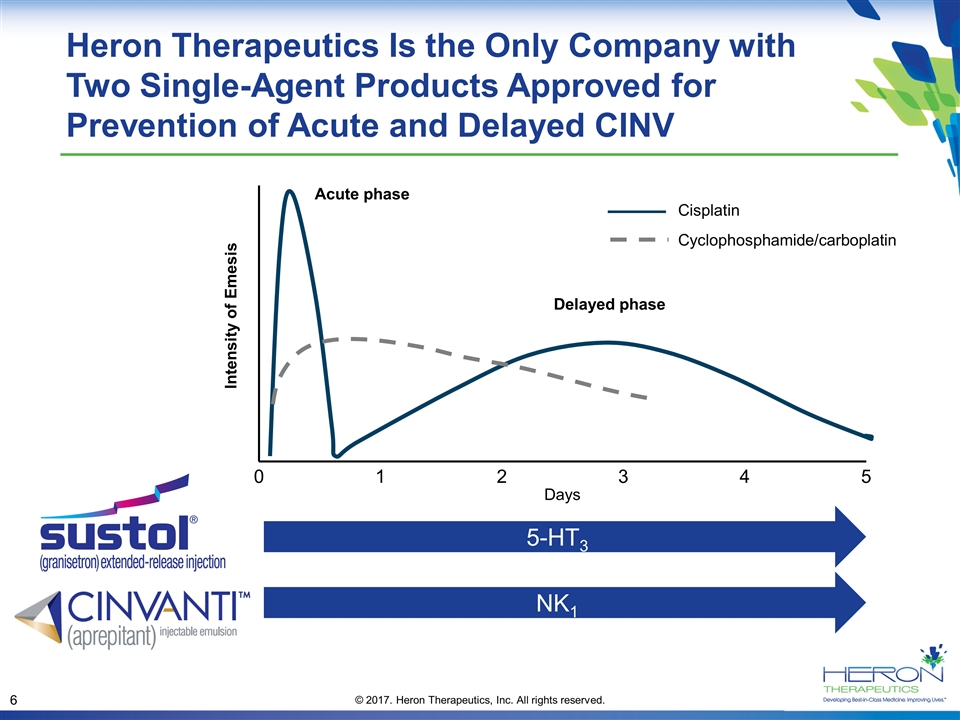
Cisplatin Cyclophosphamide/carboplatin Acute phase Delayed phase Intensity of Emesis 0 1 2 3 4 5 Days Heron Therapeutics Is the Only Company with Two Single-Agent Products Approved for Prevention of Acute and Delayed CINV NK1 5-HT3
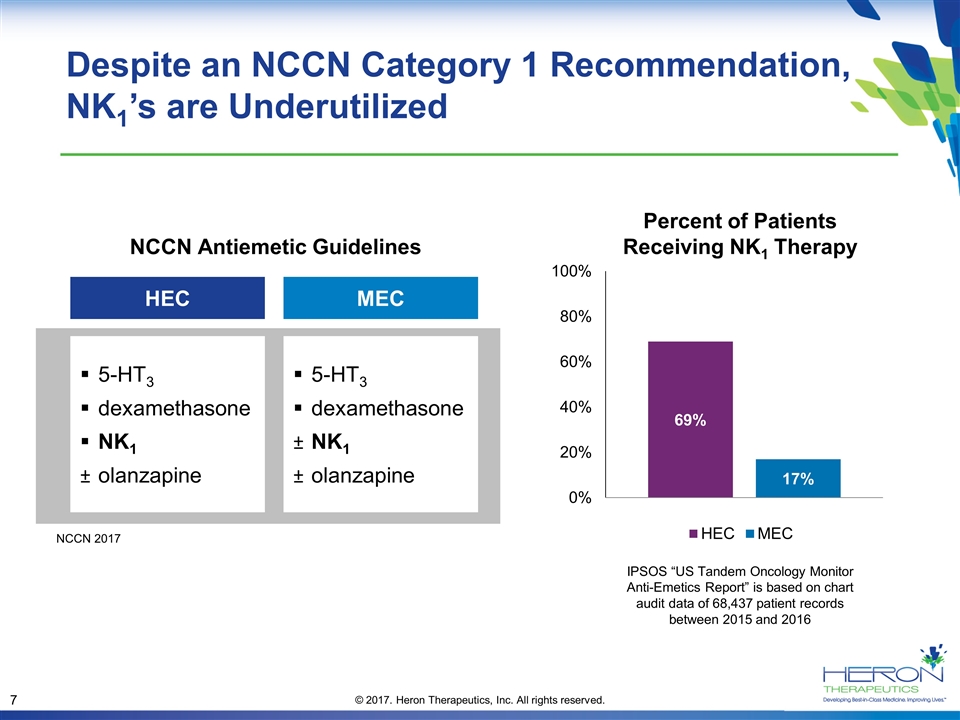
Despite an NCCN Category 1 Recommendation, NK1’s are Underutilized Percent of Patients Receiving NK1 Therapy HEC 5-HT3 dexamethasone NK1 olanzapine 5-HT3 dexamethasone NK1 olanzapine MEC NCCN Antiemetic Guidelines IPSOS “US Tandem Oncology Monitor Anti-Emetics Report” is based on chart audit data of 68,437 patient records between 2015 and 2016 NCCN 2017
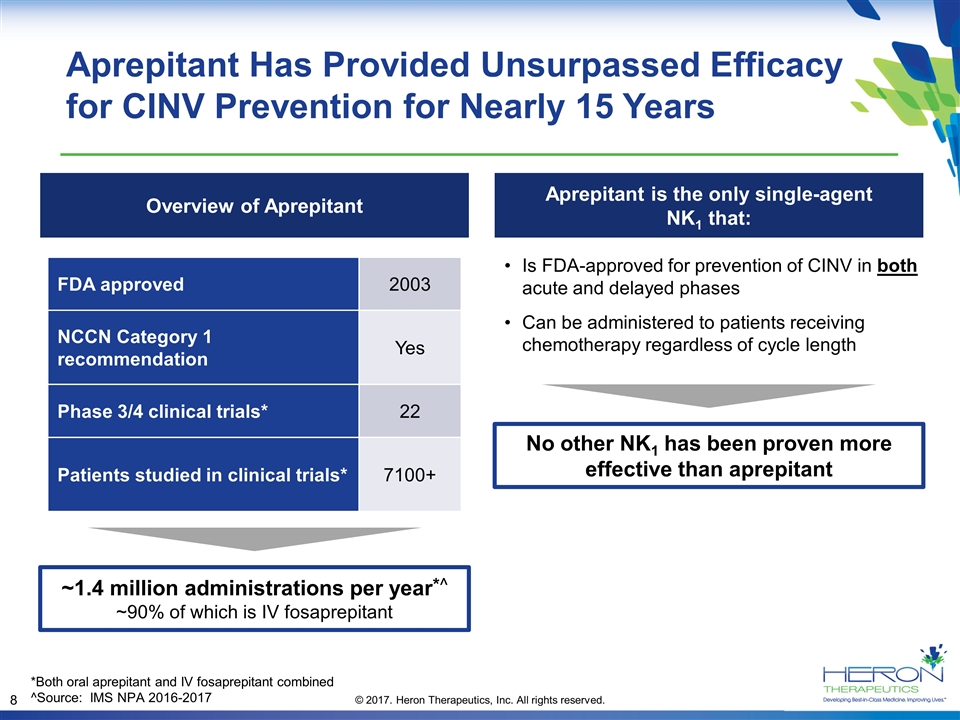
Aprepitant Has Provided Unsurpassed Efficacy for CINV Prevention for Nearly 15 Years FDA approved 2003 NCCN Category 1 recommendation Yes Phase 3/4 clinical trials* 22 Patients studied in clinical trials* 7100+ ~1.4 million administrations per year*^ ~90% of which is IV fosaprepitant Aprepitant is the only single-agent NK1 that: No other NK1 has been proven more effective than aprepitant Is FDA-approved for prevention of CINV in both acute and delayed phases Can be administered to patients receiving chemotherapy regardless of cycle length *Both oral aprepitant and IV fosaprepitant combined ^Source: IMS NPA 2016-2017 Overview of Aprepitant

Polysorbate 80 Is a Synthetic Surfactant Associated with Adverse Events Reactions related to PS-80 Injectable drugs containing PS-80 Chemotherapy Supportive Care Cabazitaxel Docetaxel Etoposide Darbepoetin alfa (Aranesp) Filgrastim (Neupogen) Fosaprepitant Polysorbate 80 (PS-80) is a synthetic surfactant used to solubilize injectable chemotherapy and supportive care drugs PS-80 is a pharmacologically active compound and has been linked to adverse events in oncology patients Heron’s goal was to develop a new IV formulation of aprepitant that has the same efficacy as IV fosaprepitant without the potential risk of polysorbate 80-related AEs
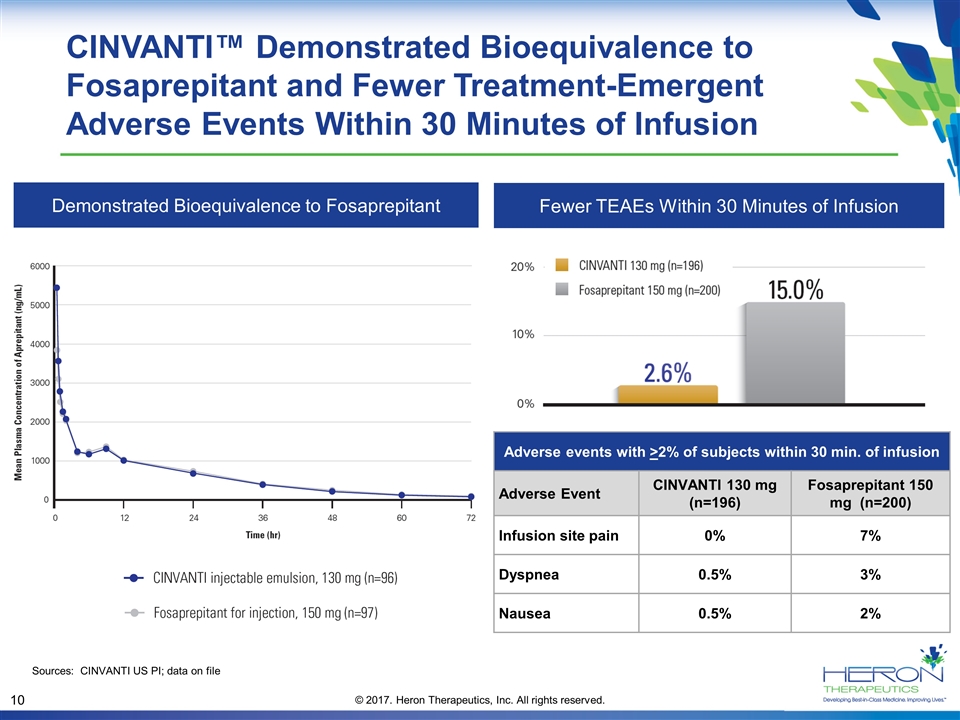
CINVANTI™ Demonstrated Bioequivalence to Fosaprepitant and Fewer Treatment-Emergent Adverse Events Within 30 Minutes of Infusion Demonstrated Bioequivalence to Fosaprepitant Fewer TEAEs Within 30 Minutes of Infusion Adverse events with >2% of subjects within 30 min. of infusion Adverse Event CINVANTI 130 mg (n=196) Fosaprepitant 150 mg (n=200) Infusion site pain 0% 7% Dyspnea 0.5% 3% Nausea 0.5% 2% Sources: CINVANTI US PI; data on file

CINVANTI™ Is the First and Only Polysorbate 80-Free IV NK1 Approved for the Prevention of Both Acute and Delayed CINV CINVANTI™ IV EMEND® IV Varubi® IV aprepitant emulsion fosaprepitant rolapitant Indicated for prevention of both acute and delayed CINV Yes Yes No Can be administered regardless of chemo cycle length Yes Yes No Polysorbate 80-free formulation Yes No Yes Emulsion formulation requires no reconstitution Yes No Yes Can be stored at room temperature for 60 days Yes No Yes
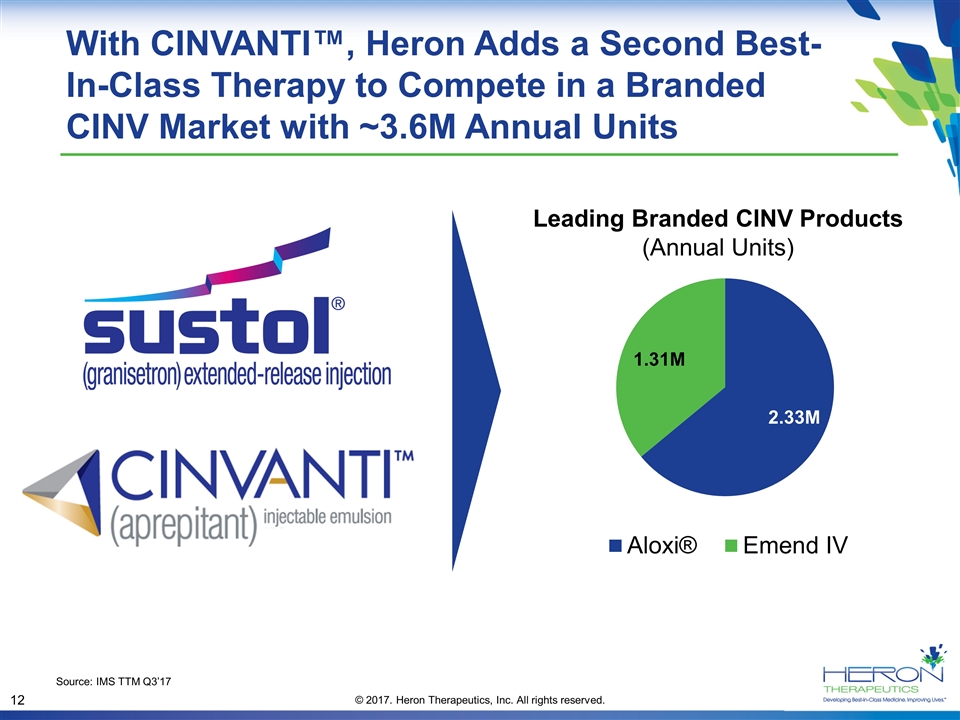
With CINVANTI™, Heron Adds a Second Best-In-Class Therapy to Compete in a Branded CINV Market with ~3.6M Annual Units Source: IMS TTM Q3’17 Leading Branded CINV Products (Annual Units)
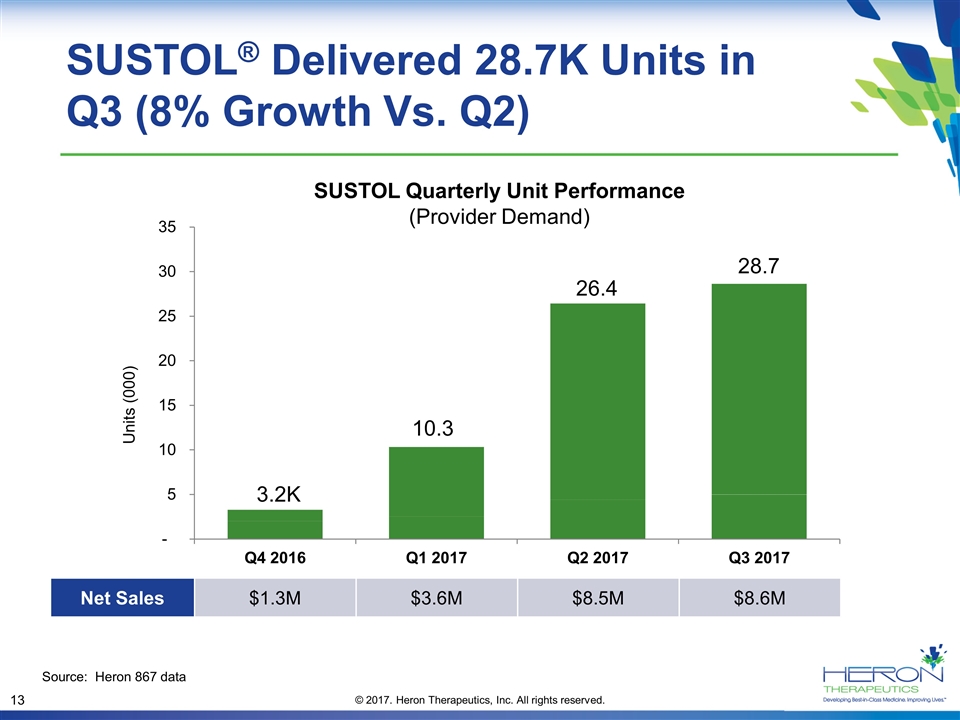
SUSTOL® Delivered 28.7K Units in Q3 (8% Growth Vs. Q2) SUSTOL Quarterly Unit Performance (Provider Demand) 3.2K 10.3 26.4 28.7 Source: Heron 867 data Net Sales $1.3M $3.6M $8.5M $8.6M
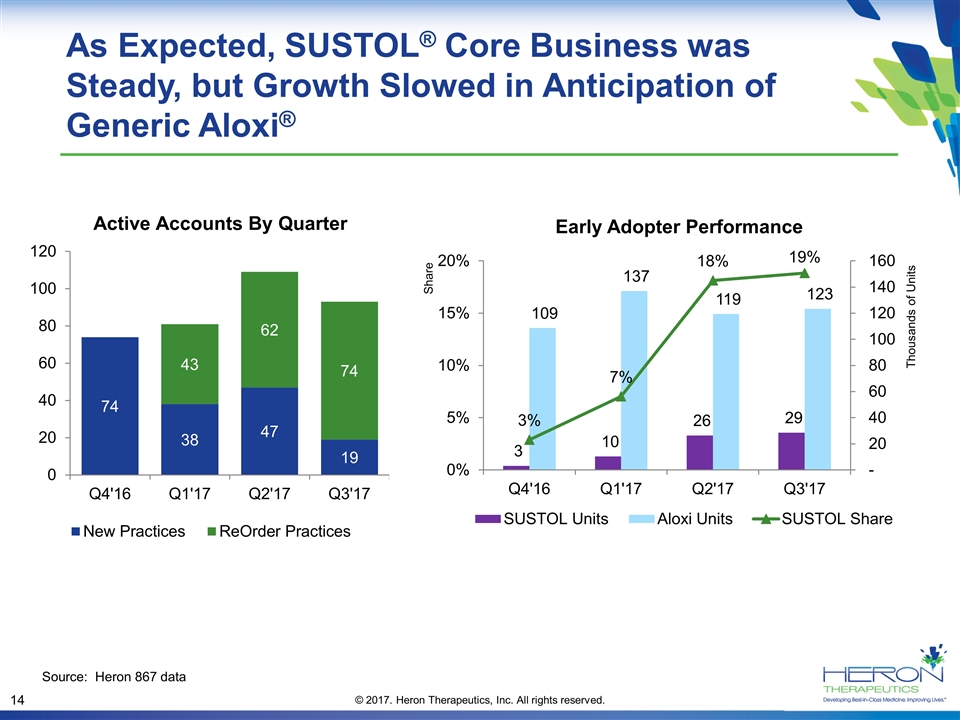
As Expected, SUSTOL® Core Business was Steady, but Growth Slowed in Anticipation of Generic Aloxi® Early Adopter Performance Share Source: Heron 867 data

Unit Sales Market Insights Suggest SUSTOL® May Decline Modestly Through the Arbitrage and Grow Thereafter – Consistent with Aloxi® Analogue Recent Market Insights Practices that are converting to SUSTOL are likely to maintain use1 ~67% of current “dabblers” likely to stop or reduce use of SUSTOL during arbitrage2 ~20% of SUSTOL non-users would consider initiating SUSTOL during arbitrage2 “If generic Aloxi is available, it’s going to allow me to start using SUSTOL without having to worry about maintaining my Aloxi contract” – PM ~55% of HCPs said they would be interested in using SUSTOL post-arbitrage (equating to an addressable market of ~650K units)2 “When ASP [erodes], we would switch all patients from generic Aloxi to SUSTOL.” – PM “SUSTOL usage would increase. There’s no reason to keep people on generic Aloxi.” – PM 1 Customer discussions 2 Putnam Associates Qual Research Findings, June 2017
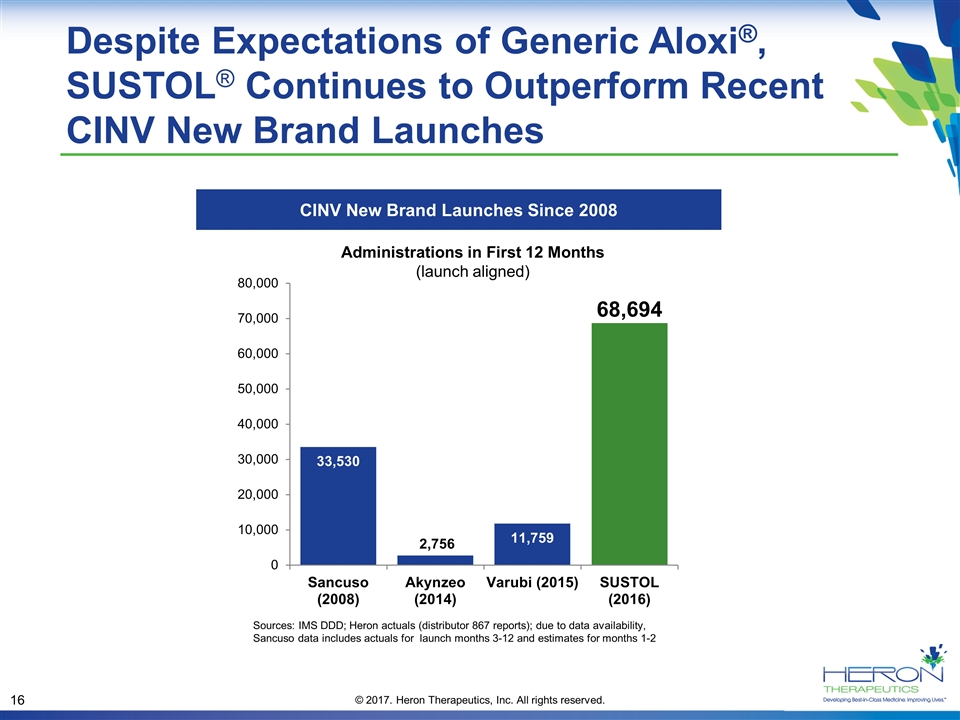
Despite Expectations of Generic Aloxi®, SUSTOL® Continues to Outperform Recent CINV New Brand Launches CINV New Brand Launches Since 2008 Sources: IMS DDD; Heron actuals (distributor 867 reports); due to data availability, Sancuso data includes actuals for launch months 3-12 and estimates for months 1-2 Administrations in First 12 Months (launch aligned) 68,694

2017 CINV Franchise Outlook SUSTOL®: We continue to expect core SUSTOL business to hold firm and with possibility of modest decline during arbitrage and growth thereafter Maintain guidance of $25M–$30M in SUSTOL net sales in 2017 Permanent J-code granted by CMS; effective January 1, 2018 CINVANTI™ Now Approved First and only polysorbate 80-free IV NK1 approved for the prevention of both acute and delayed CINV Product, pricing, and contracting available Jan 3, 2018 Offers strong strategic and operational fit with existing commercial organization Heron will build on the success of SUSTOL to win in a branded CINV market with ~3.6M annual units

Key Catalysts in Pain & CINV Franchises HTX-011 for Postoperative Pain CINVANTI™ for CINV SUSTOL® for CINV Top-line results abdominoplasty NDA submission 2017 net sales guidance: $25M - $30M Phase 2 program in nerve block initiated FDA approval Initiated TKA study (local administration) End-of-Phase 2 meeting Phase 3 program initiated Top-line Pivotal Phase 3 results 1H 2018 NDA filing 2018