
HTX-011 Postoperative Pain Program Topline Results from Phase 2b Studies June 21, 2018 Exhibit 99.3

Forward-Looking Statements This presentation contains "forward-looking statements" as defined by the Private Securities Litigation Reform Act of 1995. We caution investors that forward-looking statements are based on management’s expectations and assumptions as of the date of this presentation and involve substantial risks and uncertainties that could cause our clinical development programs, future results, performance or achievements to differ significantly from those expressed or implied by the forward-looking statements. These risks and uncertainties include, but are not limited to, those associated with: the potential market opportunity for HTX-011; the timing of the NDA filing for HTX-011; the timing of completion and results of clinical studies for HTX-011; and other risks and uncertainties identified in the Company's filings with the Securities and Exchange Commission. Forward-looking statements reflect our analysis only on their stated date, and we take no obligation to update or revise these statements except as may be required by law.
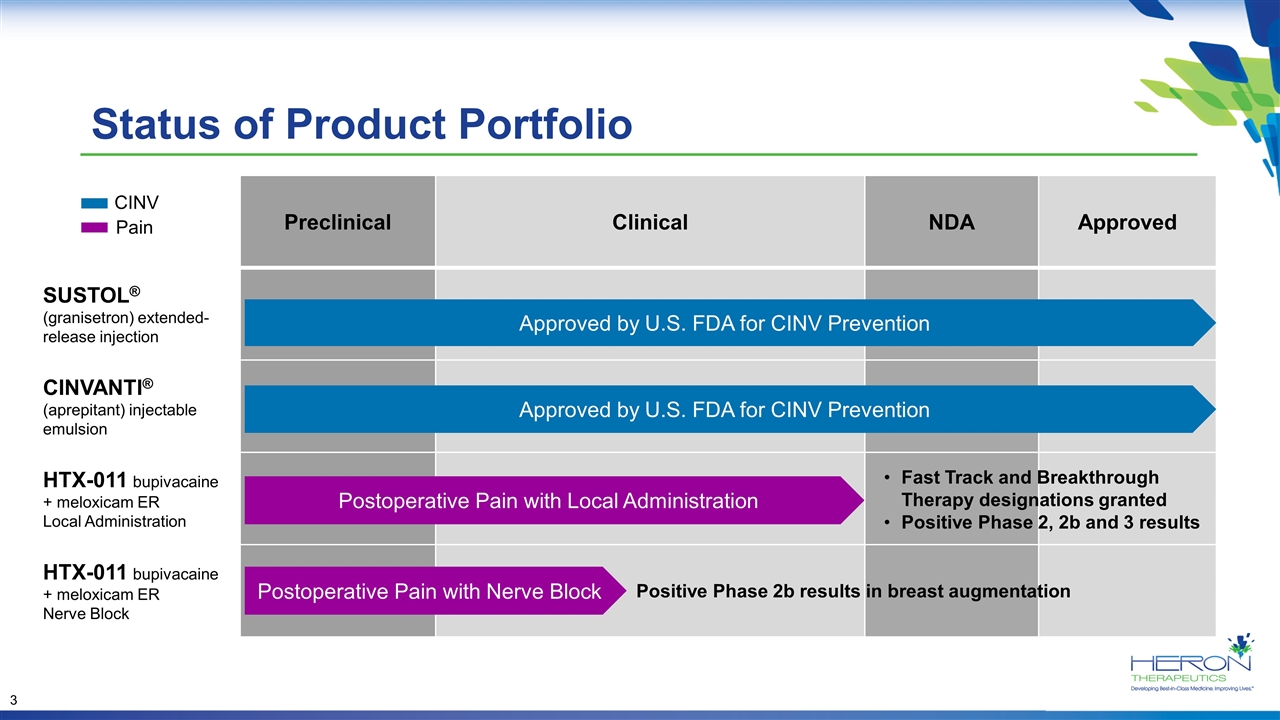
Preclinical Clinical NDA Approved SUSTOL® (granisetron) extended-release injection CINVANTI® (aprepitant) injectable emulsion HTX-011 bupivacaine + meloxicam ER Local Administration HTX-011 bupivacaine + meloxicam ER Nerve Block Approved by U.S. FDA for CINV Prevention Status of Product Portfolio Approved by U.S. FDA for CINV Prevention Postoperative Pain with Local Administration Fast Track and Breakthrough Therapy designations granted Positive Phase 2, 2b and 3 results CINV Pain Positive Phase 2b results in breast augmentation Postoperative Pain with Nerve Block

HTX-011 for Postoperative Pain Management Has Received Breakthrough Therapy Designation Breakthrough Therapy designation designed to expedite development and review of drugs: Intended to treat serious conditions; and For which preliminary clinical evidence indicates substantial improvement over available therapies on clinically significant endpoint(s) Designation granted based on results of Phase 2 studies and two recently completed Phase 3 studies HTX-011 produced significant reductions in both pain intensity and need for opioids through 72 hours post-surgery compared to placebo and bupivacaine solution, the standard of care HTX-011 was also granted Fast Track designation in November 2017

Postoperative Opioids Are a Gateway to Addiction $ AS MANY AS 2.6 MILLION patients that take opioids to manage pain after surgery may become persistent opioid users each year . MORE THAN 40 MILLION patients undergoing surgical procedures are prescribed opioids for pain management in the United States each year UP TO 440,000 patients will become addicted to opioids each year In addition >1 BILLION OPIOID PILLS are taken home from the hospital after surgery each year 70% of all these opioid pills go unused 90% of these pills remain inside the home in unsecured locations >$15 BILLION of the annual healthcare costs associated with addiction can be attributed to postoperative pain management 32% of all opioid addicts report first opioid exposure through leftover pills

HTX-011 achieved statistically significant reductions in pain and the need for opioids Vs. Bupivacaine in every phase 2 study and both phase 3 studies
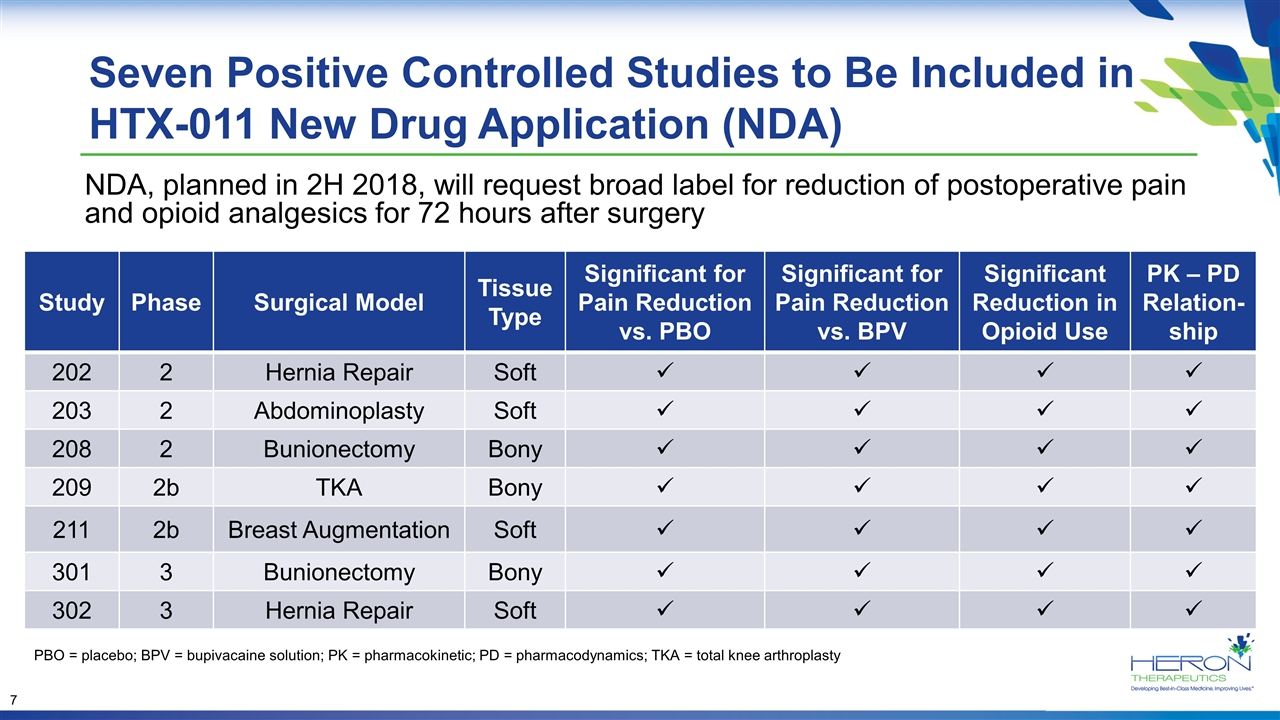
Seven Positive Controlled Studies to Be Included in HTX-011 New Drug Application (NDA) Study Phase Surgical Model Tissue Type Significant for Pain Reduction vs. PBO Significant for Pain Reduction vs. BPV Significant Reduction in Opioid Use PK – PD Relation-ship 202 2 Hernia Repair Soft ü ü ü ü 203 2 Abdominoplasty Soft ü ü ü ü 208 2 Bunionectomy Bony ü ü ü ü 209 2b TKA Bony ü ü ü ü 211 2b Breast Augmentation Soft ü ü ü ü 301 3 Bunionectomy Bony ü ü ü ü 302 3 Hernia Repair Soft ü ü ü ü NDA, planned in 2H 2018, will request broad label for reduction of postoperative pain and opioid analgesics for 72 hours after surgery PBO = placebo; BPV = bupivacaine solution; PK = pharmacokinetic; PD = pharmacodynamics; TKA = total knee arthroplasty

Recently Completed Phase 2b Studies
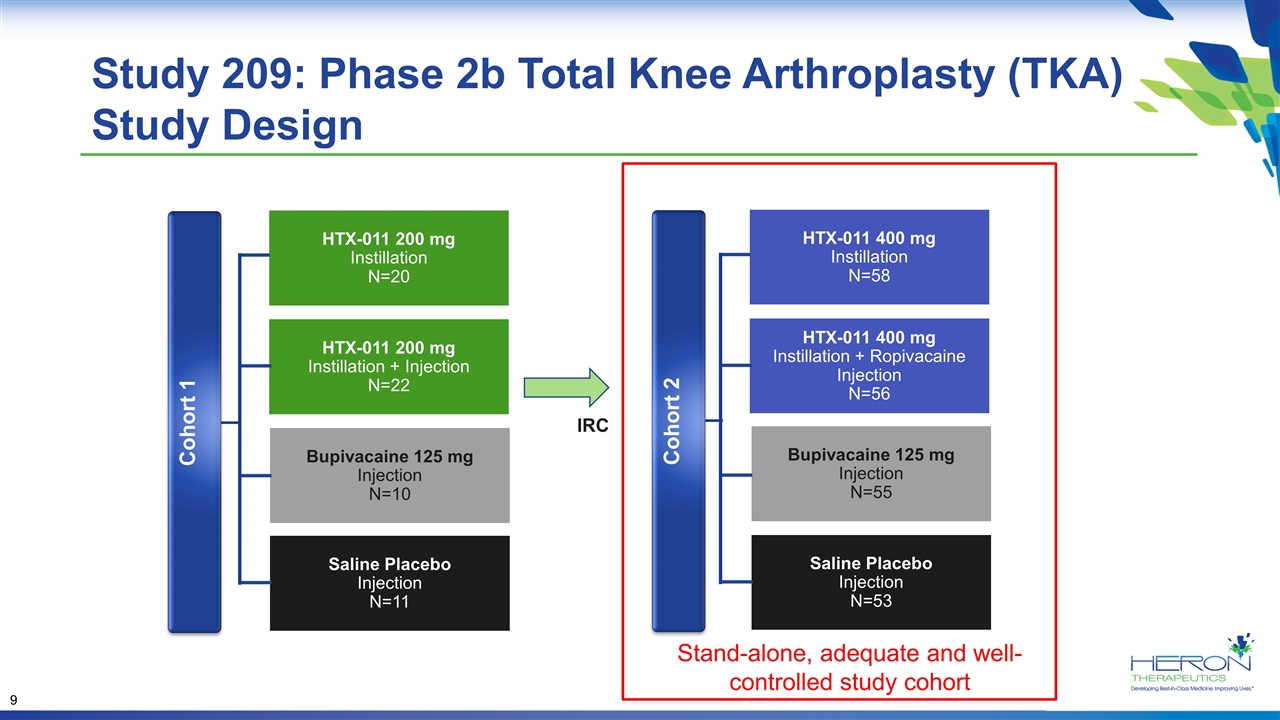
Study 209: Phase 2b Total Knee Arthroplasty (TKA) Study Design HTX-011 400 mg Instillation N=58 HTX-011 400 mg Instillation + Ropivacaine Injection N=56 Saline Placebo Injection N=53 Bupivacaine 125 mg Injection N=55 Cohort 1 HTX-011 200 mg Instillation N=20 HTX-011 200 mg Instillation + Injection N=22 Saline Placebo Injection N=11 Bupivacaine 125 mg Injection N=10 IRC Cohort 2 Stand-alone, adequate and well-controlled study cohort
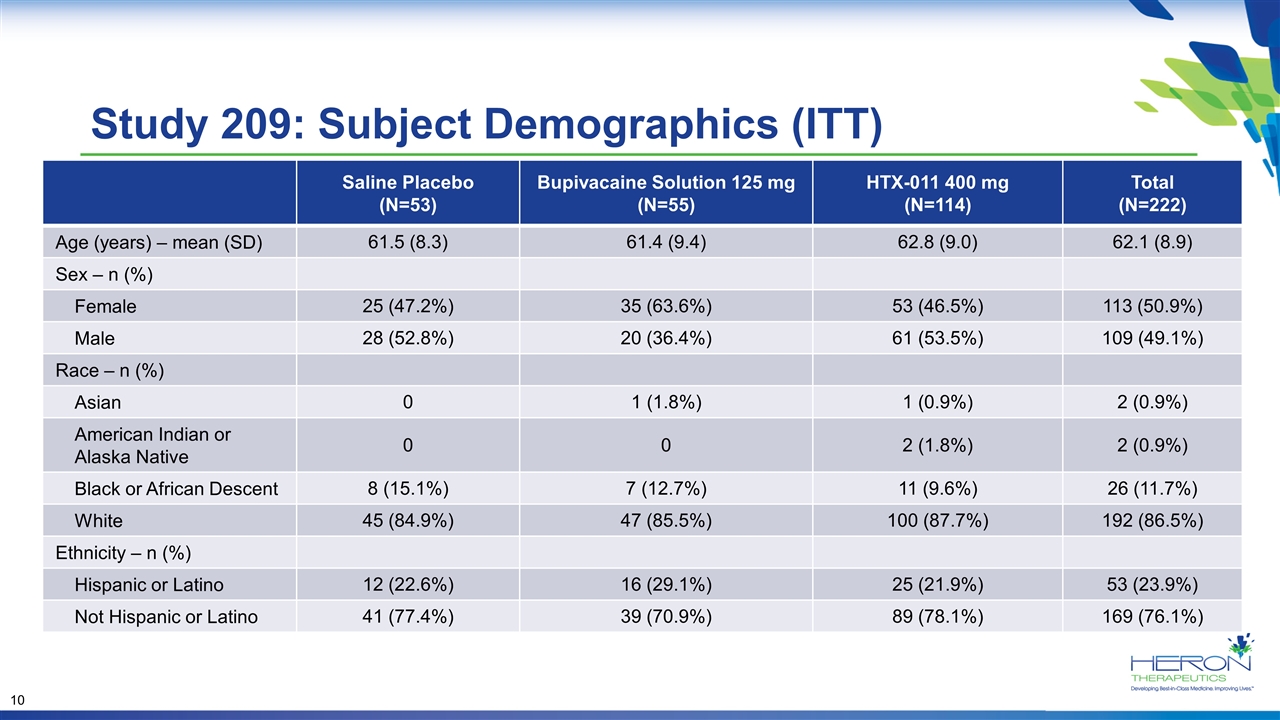
Study 209: Subject Demographics (ITT) Saline Placebo (N=53) Bupivacaine Solution 125 mg (N=55) HTX-011 400 mg (N=114) Total (N=222) Age (years) – mean (SD) 61.5 (8.3) 61.4 (9.4) 62.8 (9.0) 62.1 (8.9) Sex – n (%) Female 25 (47.2%) 35 (63.6%) 53 (46.5%) 113 (50.9%) Male 28 (52.8%) 20 (36.4%) 61 (53.5%) 109 (49.1%) Race – n (%) Asian 0 1 (1.8%) 1 (0.9%) 2 (0.9%) American Indian or Alaska Native 0 0 2 (1.8%) 2 (0.9%) Black or African Descent 8 (15.1%) 7 (12.7%) 11 (9.6%) 26 (11.7%) White 45 (84.9%) 47 (85.5%) 100 (87.7%) 192 (86.5%) Ethnicity – n (%) Hispanic or Latino 12 (22.6%) 16 (29.1%) 25 (21.9%) 53 (23.9%) Not Hispanic or Latino 41 (77.4%) 39 (70.9%) 89 (78.1%) 169 (76.1%)
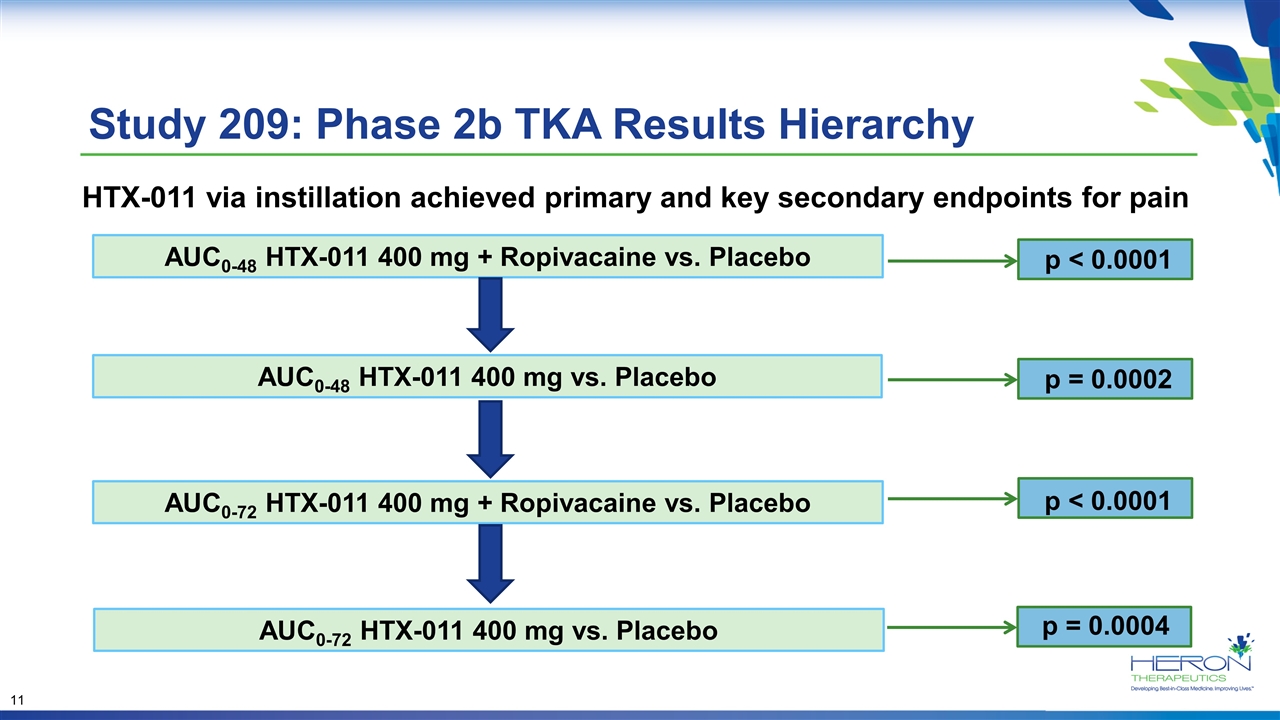
Study 209: Phase 2b TKA Results Hierarchy AUC0-48 HTX-011 400 mg vs. Placebo AUC0-72 HTX-011 400 mg + Ropivacaine vs. Placebo AUC0-48 HTX-011 400 mg + Ropivacaine vs. Placebo p < 0.0001 AUC0-72 HTX-011 400 mg vs. Placebo p = 0.0002 p < 0.0001 p = 0.0004 HTX-011 via instillation achieved primary and key secondary endpoints for pain
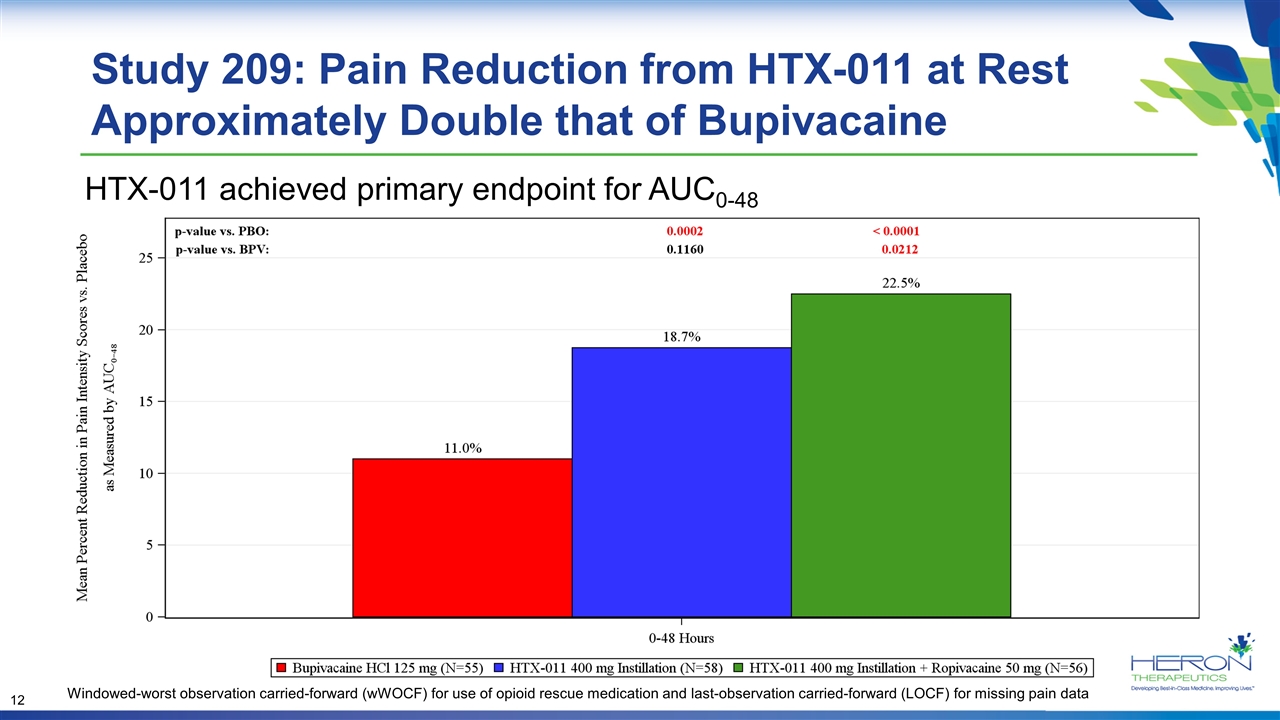
Study 209: Pain Reduction from HTX-011 at Rest Approximately Double that of Bupivacaine Windowed-worst observation carried-forward (wWOCF) for use of opioid rescue medication and last-observation carried-forward (LOCF) for missing pain data HTX-011 achieved primary endpoint for AUC0-48

Study 209: Both HTX-011 Arms Significantly Reduce Pain at Rest Compared to Placebo through 72 Hours wWOCF for use of opioid rescue medication and LOCF for missing pain data
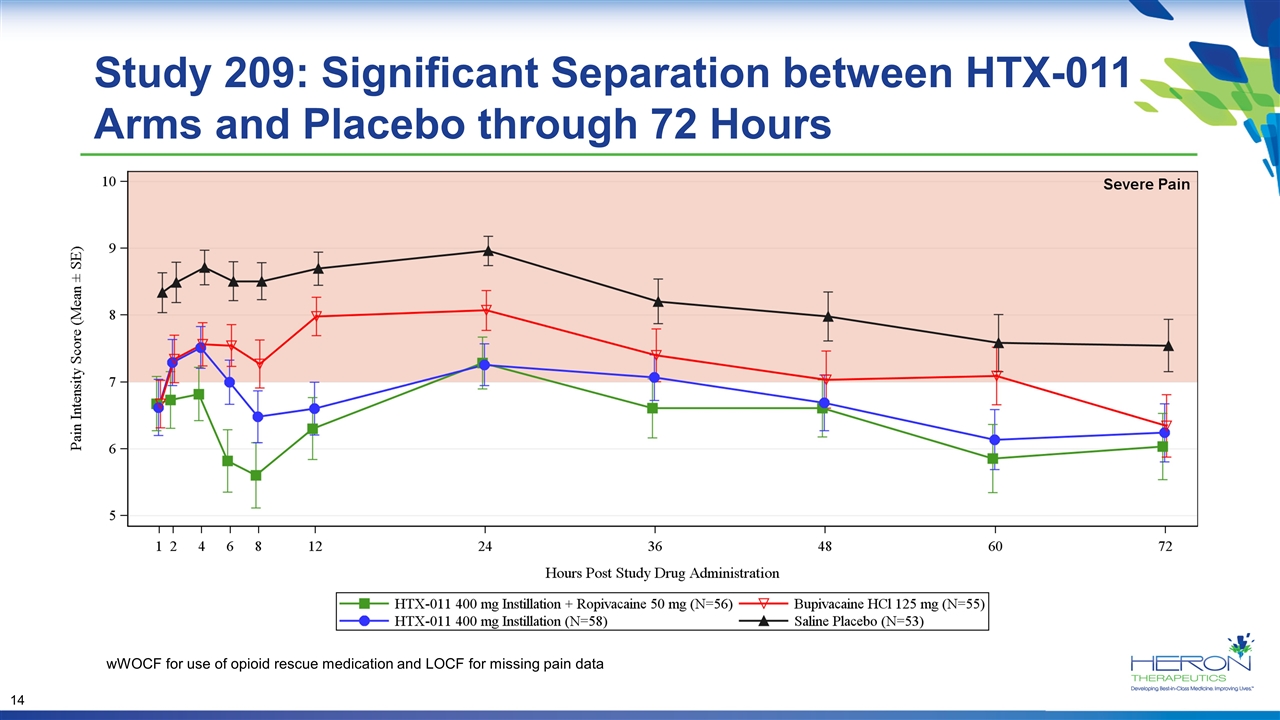
Study 209: Significant Separation between HTX-011 Arms and Placebo through 72 Hours wWOCF for use of opioid rescue medication and LOCF for missing pain data
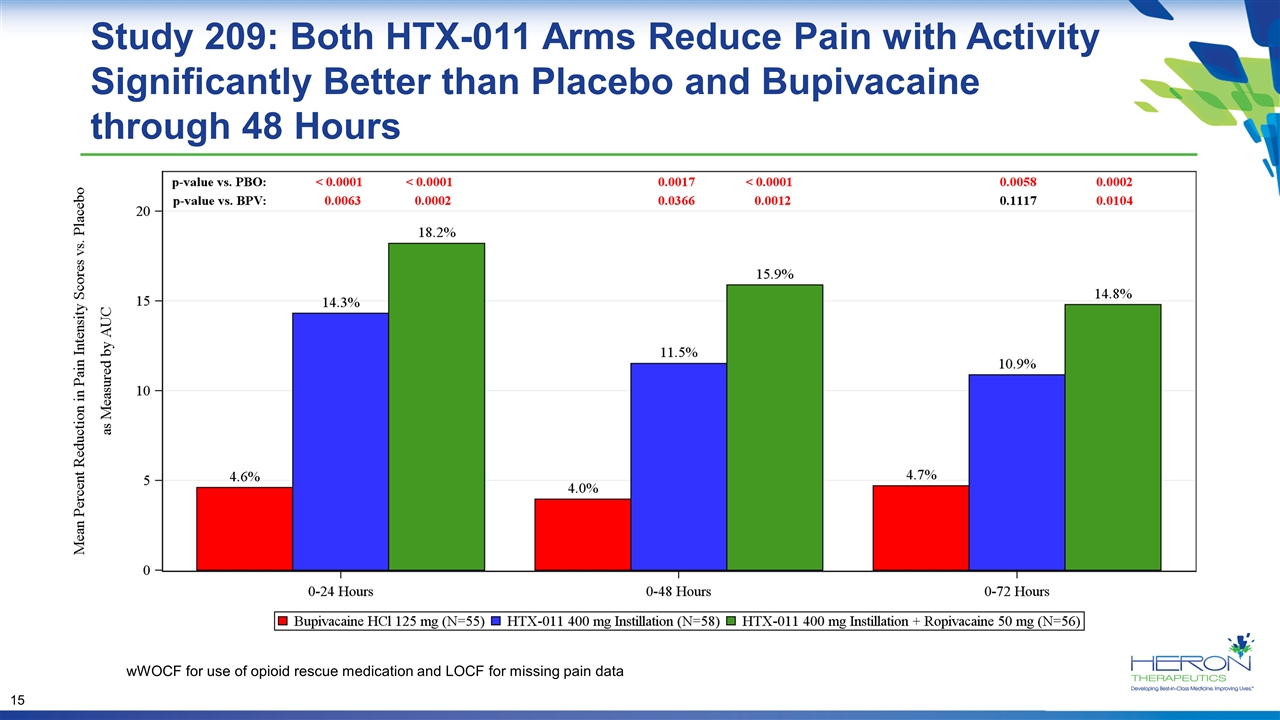
Study 209: Both HTX-011 Arms Reduce Pain with Activity Significantly Better than Placebo and Bupivacaine through 48 Hours wWOCF for use of opioid rescue medication and LOCF for missing pain data
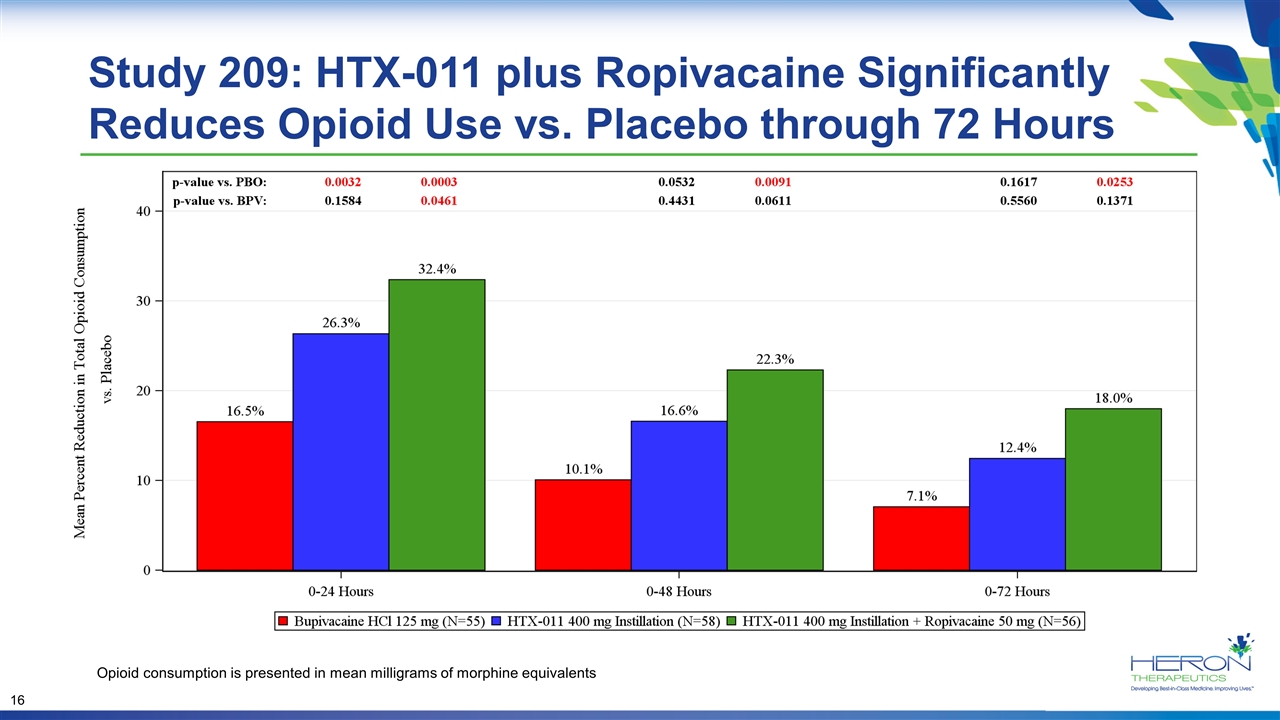
Study 209: HTX-011 plus Ropivacaine Significantly Reduces Opioid Use vs. Placebo through 72 Hours Opioid consumption is presented in mean milligrams of morphine equivalents
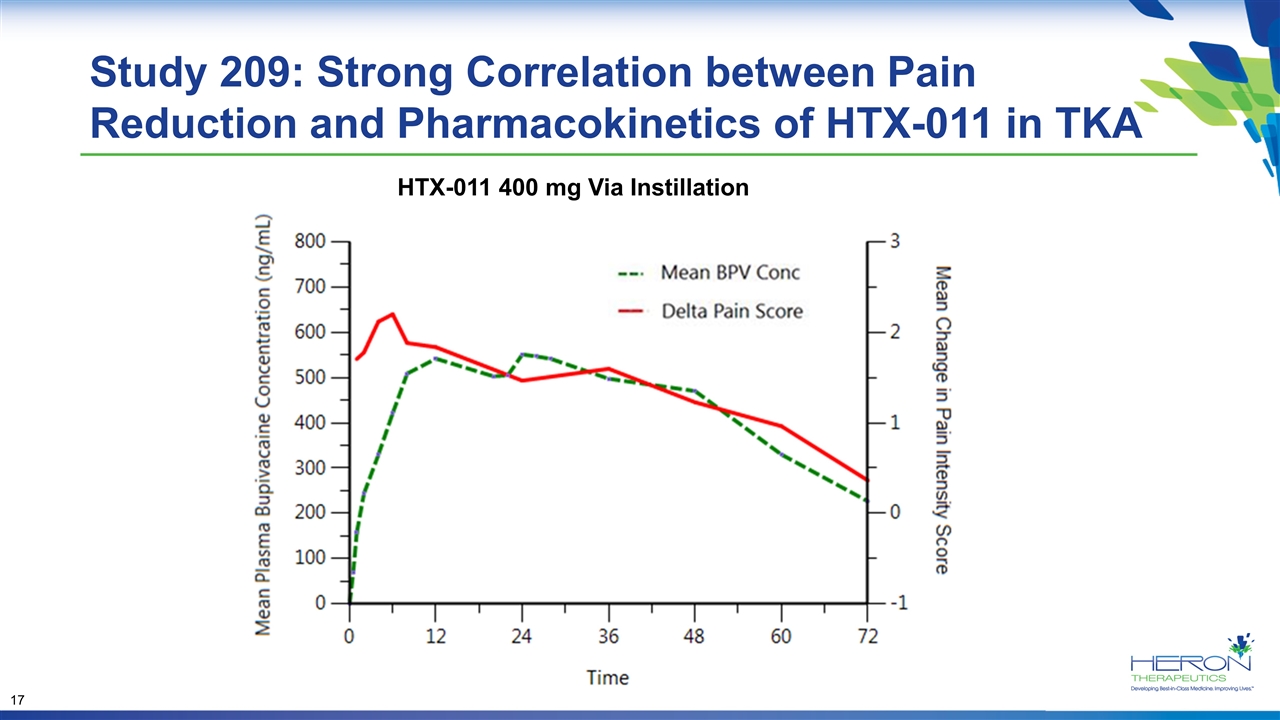
Study 209: Strong Correlation between Pain Reduction and Pharmacokinetics of HTX-011 in TKA HTX-011 400 mg Via Instillation

Study 209: HTX-011 Well Tolerated in TKA HTX-011 was well tolerated, with a safety profile comparable to placebo and bupivacaine solution: No clinically meaningful differences in overall adverse events No difference in the incidence of serious adverse events No difference in premature discontinuations due to adverse events No deaths No clinically meaningful differences in potential local anesthetic systemic toxicity (LAST) adverse events in this highly vascular model No increase in potential LAST when given with another local anesthetic, ropivacaine No difference in wound healing

Study 209: TKA Summary HTX-011 achieved primary and key secondary endpoints Both HTX-011 arms achieved significant reductions in pain at rest vs. placebo through 48 hours Also significantly reduced pain at rest through 72 hours vs. placebo Both HTX-011 arms achieved significant reductions in pain with activity (the most conservative assessment) vs. placebo and bupivacaine through 48 hours HTX-011 plus ropivacaine maintained superiority to both placebo and bupivacaine through 72 hours There was significant separation of the HTX-011 mean pain curves vs. placebo through 72 hours Strong correlation between PK and PD in TKA HTX-011 with or without ropivacaine was generally well tolerated in TKA
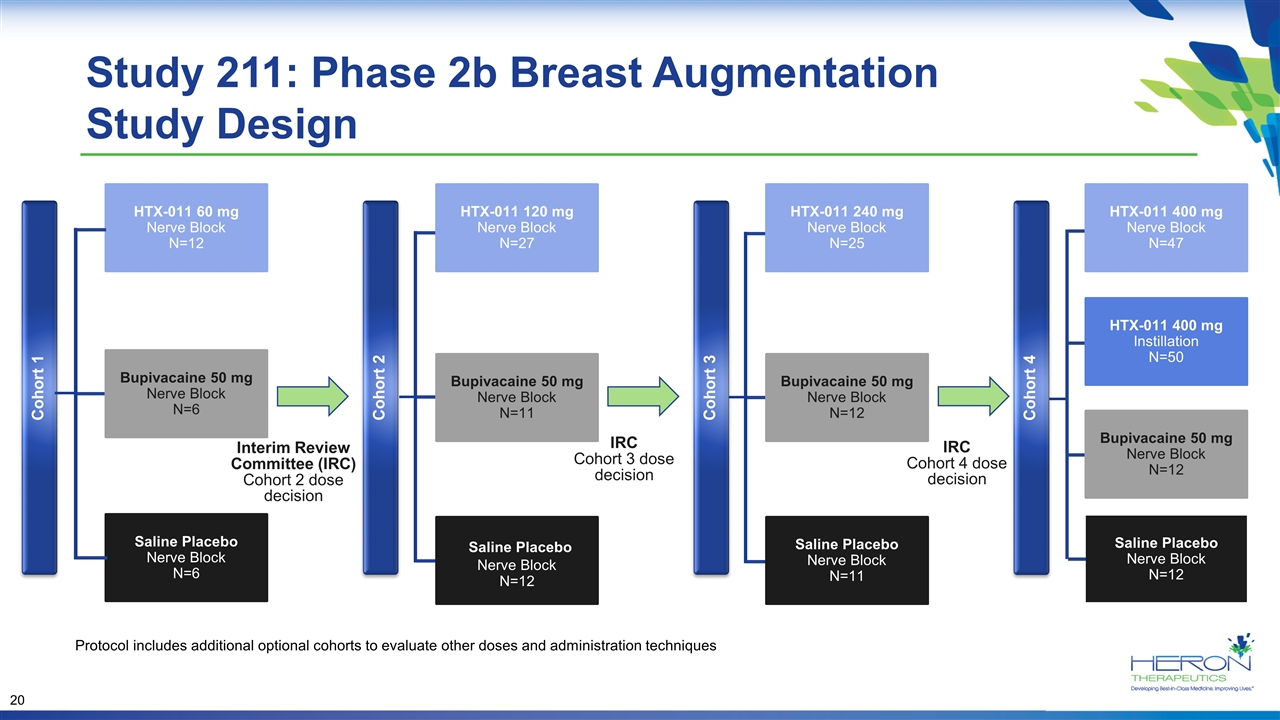
HTX-011 60 mg Nerve Block N=12 Saline Placebo Nerve Block N=6 Study 211: Phase 2b Breast Augmentation Study Design Cohort 1 Cohort 2 Interim Review Committee (IRC) Cohort 2 dose decision HTX-011 120 mg Nerve Block N=27 Saline Placebo Nerve Block N=12 Bupivacaine 50 mg Nerve Block N=11 HTX-011 240 mg Nerve Block N=25 Saline Placebo Nerve Block N=11 IRC Cohort 3 dose decision IRC Cohort 4 dose decision Bupivacaine 50 mg Nerve Block N=12 HTX-011 400 mg Nerve Block N=47 HTX-011 400 mg Instillation N=50 Saline Placebo Nerve Block N=12 Bupivacaine 50 mg Nerve Block N=12 Cohort 4 Cohort 3 Protocol includes additional optional cohorts to evaluate other doses and administration techniques Bupivacaine 50 mg Nerve Block N=6
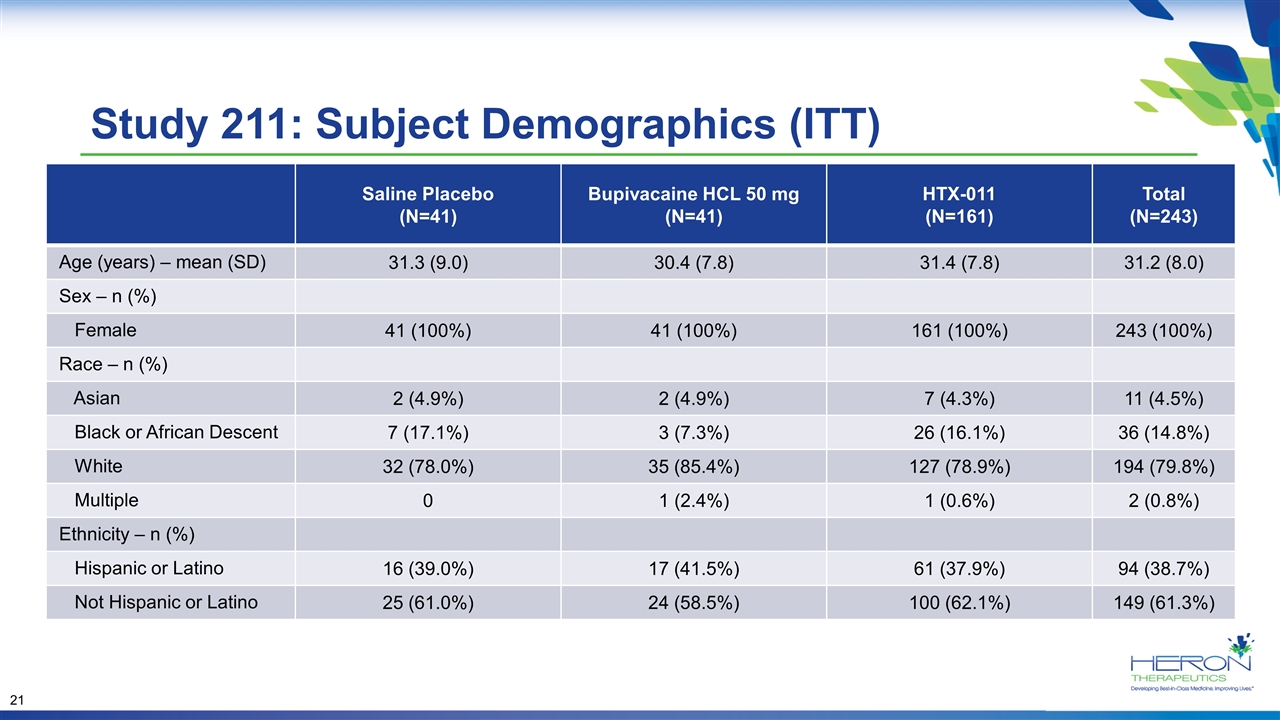
Study 211: Subject Demographics (ITT) Saline Placebo (N=41) Bupivacaine HCL 50 mg (N=41) HTX-011 (N=161) Total (N=243) Age (years) – mean (SD) 31.3 (9.0) 30.4 (7.8) 31.4 (7.8) 31.2 (8.0) Sex – n (%) Female 41 (100%) 41 (100%) 161 (100%) 243 (100%) Race – n (%) Asian 2 (4.9%) 2 (4.9%) 7 (4.3%) 11 (4.5%) Black or African Descent 7 (17.1%) 3 (7.3%) 26 (16.1%) 36 (14.8%) White 32 (78.0%) 35 (85.4%) 127 (78.9%) 194 (79.8%) Multiple 0 1 (2.4%) 1 (0.6%) 2 (0.8%) Ethnicity – n (%) Hispanic or Latino 16 (39.0%) 17 (41.5%) 61 (37.9%) 94 (38.7%) Not Hispanic or Latino 25 (61.0%) 24 (58.5%) 100 (62.1%) 149 (61.3%)
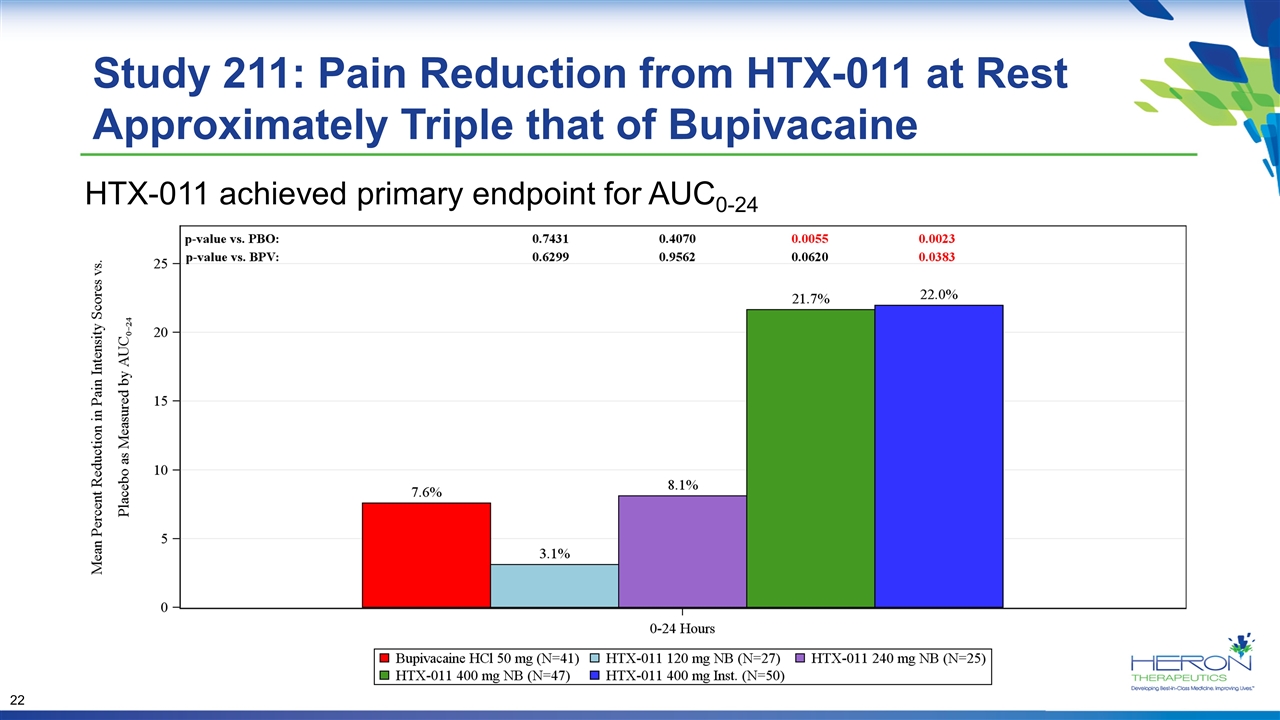
Study 211: Pain Reduction from HTX-011 at Rest Approximately Triple that of Bupivacaine HTX-011 achieved primary endpoint for AUC0-24
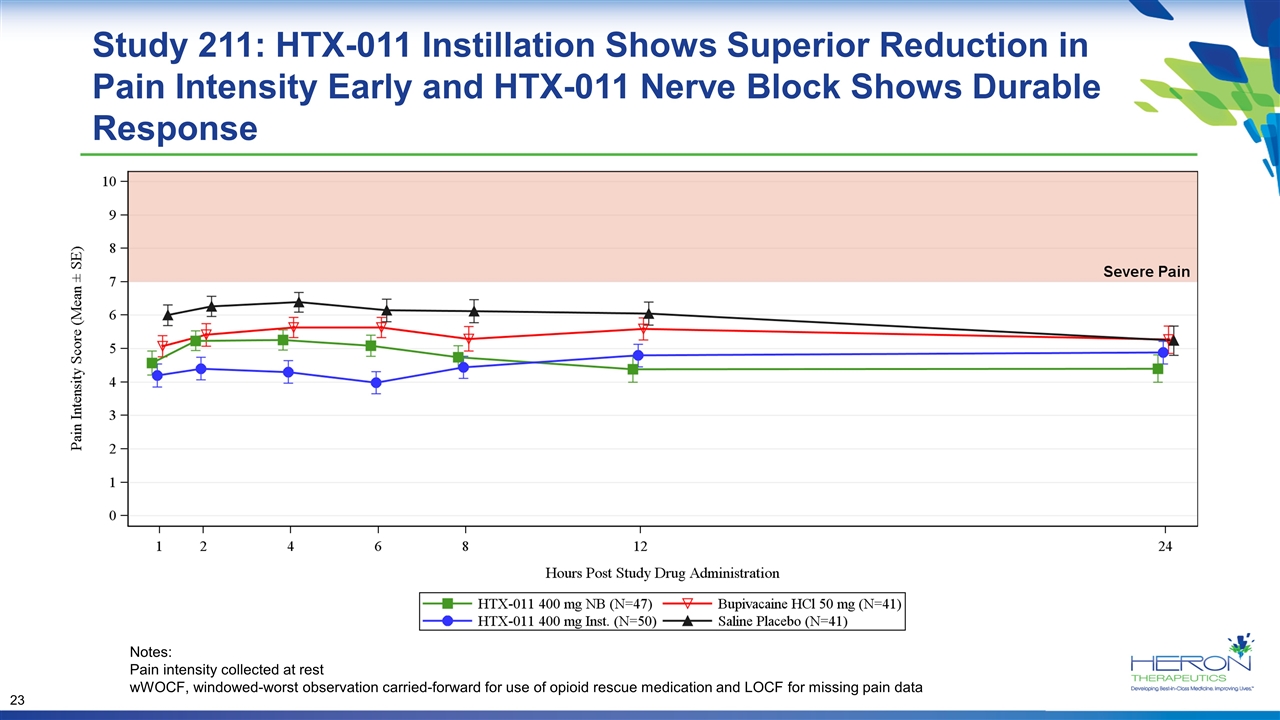
Study 211: HTX-011 Instillation Shows Superior Reduction in Pain Intensity Early and HTX-011 Nerve Block Shows Durable Response Notes: Pain intensity collected at rest wWOCF, windowed-worst observation carried-forward for use of opioid rescue medication and LOCF for missing pain data
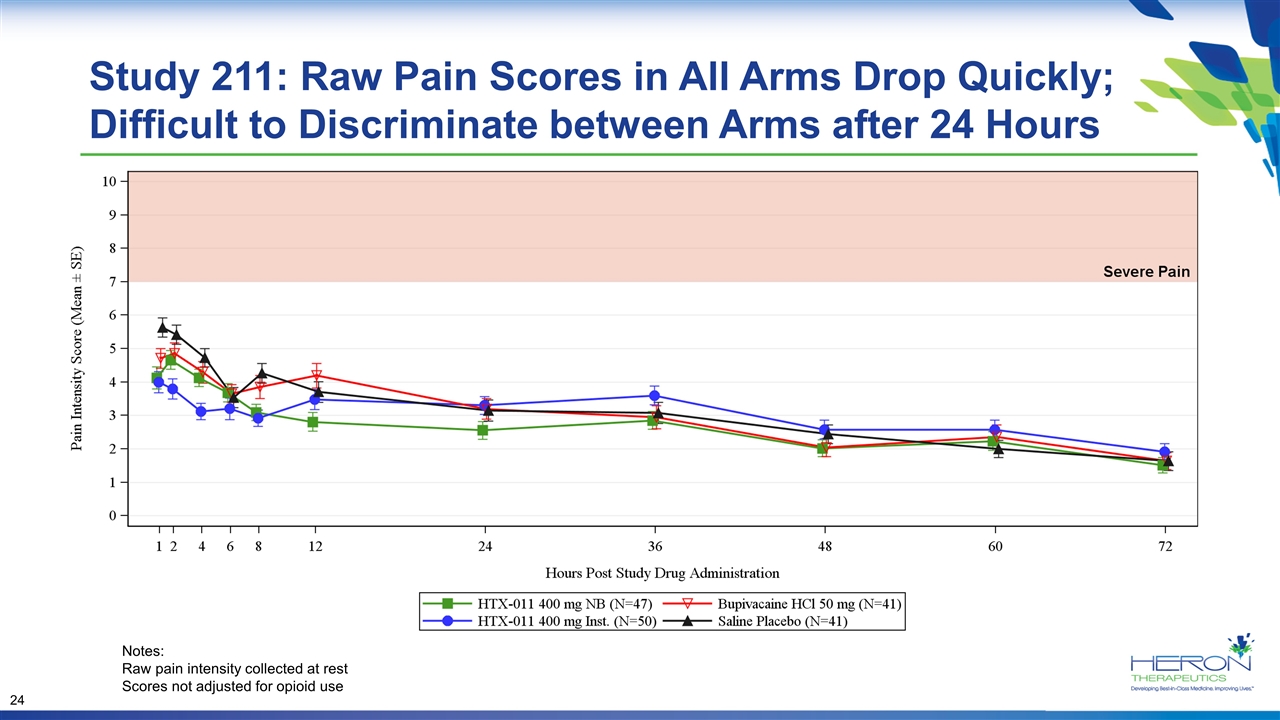
Study 211: Raw Pain Scores in All Arms Drop Quickly; Difficult to Discriminate between Arms after 24 Hours Notes: Raw pain intensity collected at rest Scores not adjusted for opioid use
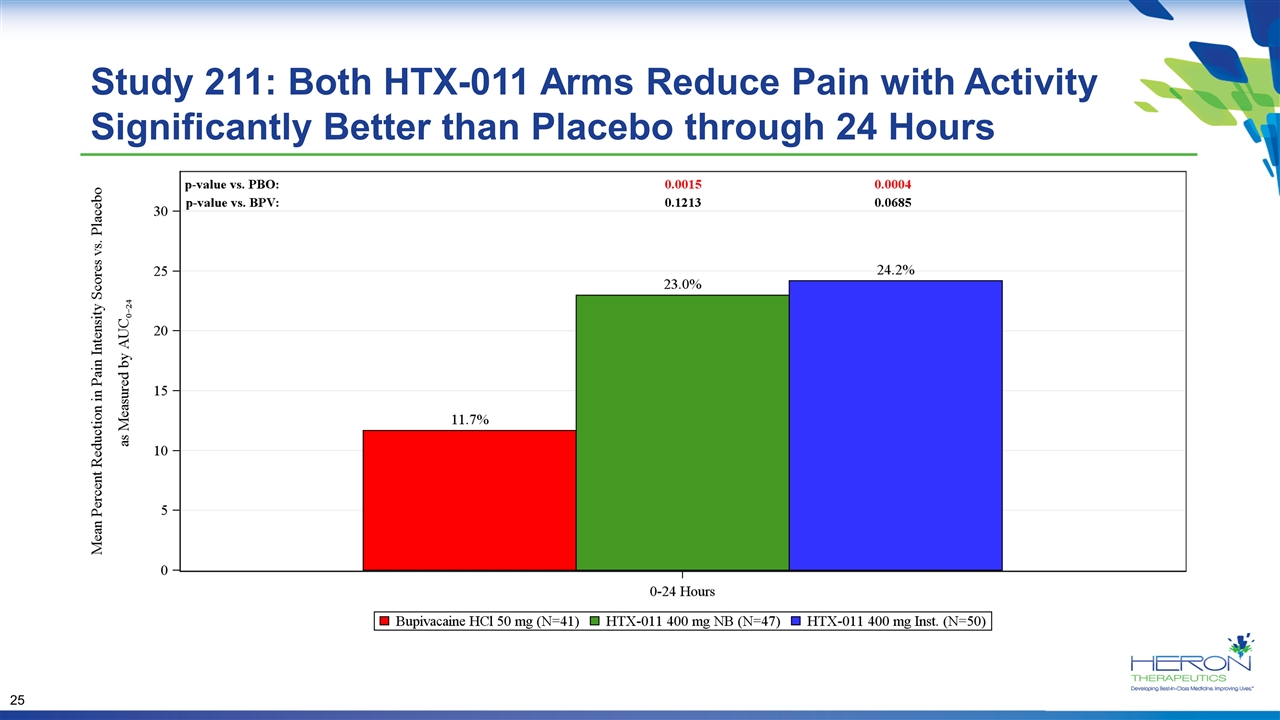
Study 211: Both HTX-011 Arms Reduce Pain with Activity Significantly Better than Placebo through 24 Hours
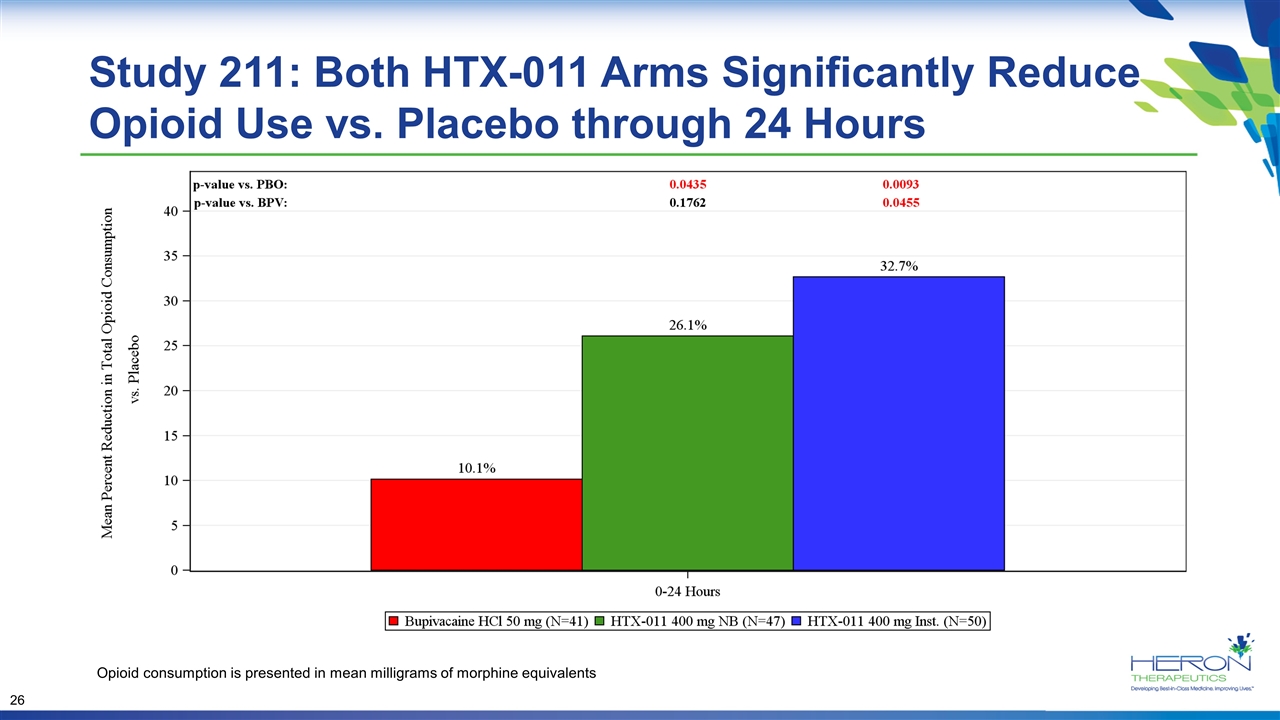
Study 211: Both HTX-011 Arms Significantly Reduce Opioid Use vs. Placebo through 24 Hours Opioid consumption is presented in mean milligrams of morphine equivalents
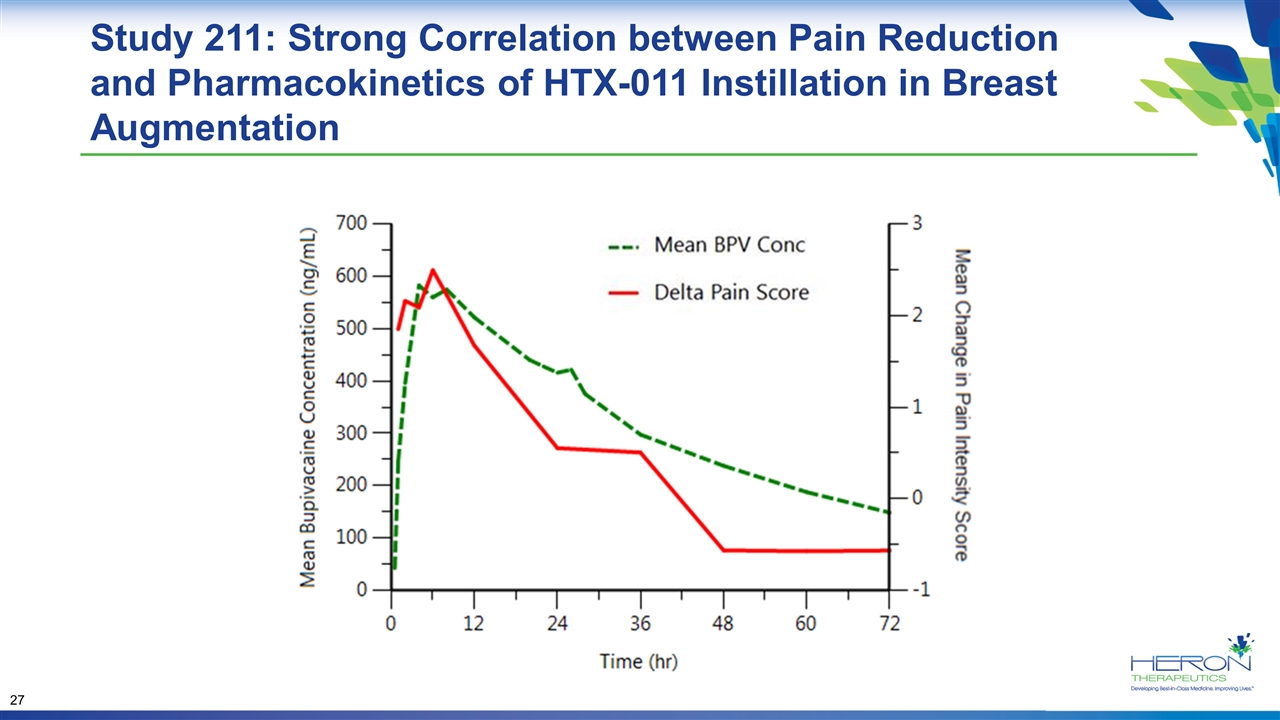
Study 211: Strong Correlation between Pain Reduction and Pharmacokinetics of HTX-011 Instillation in Breast Augmentation

HTX-011 Well Tolerated in Breast Augmentation HTX-011 was well tolerated, with a safety profile comparable to placebo and bupivacaine solution: No clinically meaningful differences in overall adverse events No difference in the incidence of serious adverse events No premature discontinuations due to adverse events No deaths No clinically meaningful differences in potential LAST adverse events No evidence of wound healing issues with local administration into the breast pocket

Study 211: Breast Augmentation Summary HTX-011 achieved the primary endpoint Both HTX-011 arms achieved significant reductions in pain at rest vs. placebo through 24 hours Both HTX-011 arms achieved significant reductions in pain with activity (the most conservative assessment) vs. placebo through 24 hours HTX-011 instillation (the most commercially relevant route for cosmetic surgery) produced the greatest reduction in pain and opioid use, beating both placebo and bupivacaine nerve block Strong correlation between PK and PD in breast augmentation with instillation HTX-011 administered via instillation or as a nerve block was generally well tolerated in breast augmentation with no wound healing issues

Consistent Pharmacokinetics
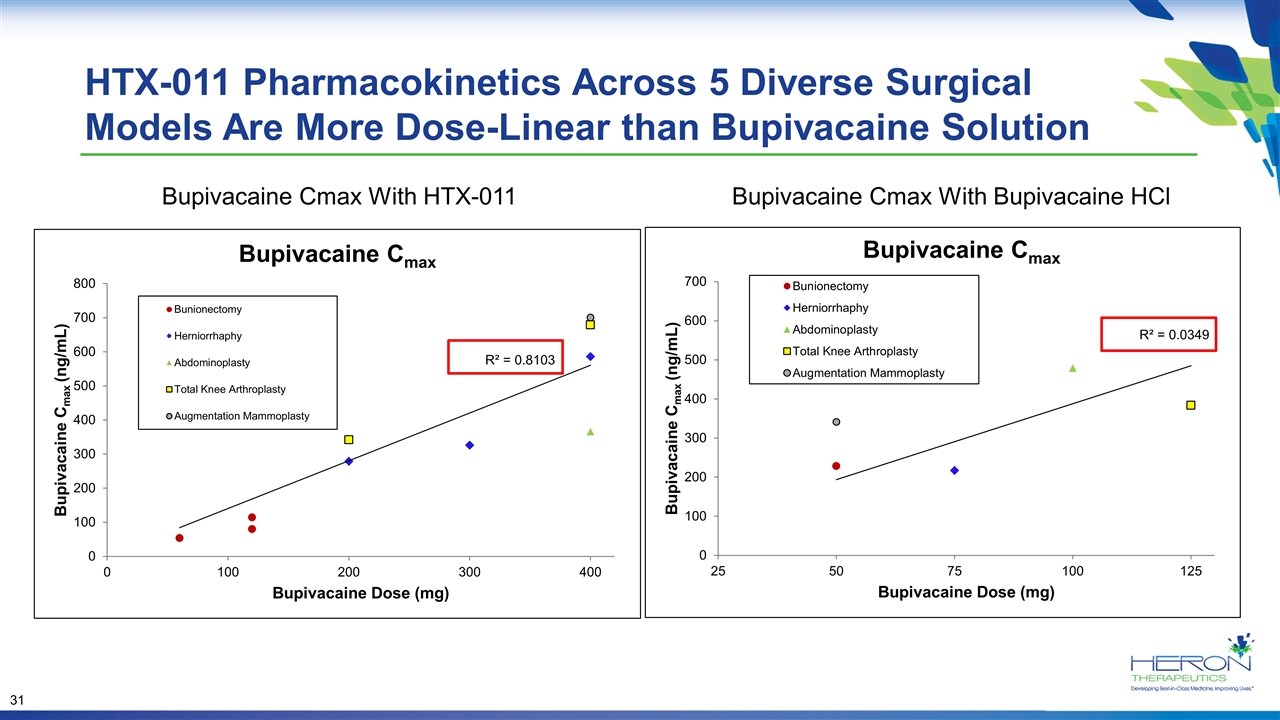
HTX-011 Pharmacokinetics Across 5 Diverse Surgical Models Are More Dose-Linear than Bupivacaine Solution Bupivacaine Cmax With HTX-011 Bupivacaine Cmax With Bupivacaine HCl

Phase 2b Conclusions HTX-011 plus ropivacaine was significantly superior to both placebo and bupivacaine in TKA HTX-011 demonstrated significant activity via both instillation and nerve block in breast augmentation Pharmacokinetics of HTX-011 remained consistent across 5 diverse surgical models with consistent correlation between PK and PD HTX-011 has been generally well tolerated up to 400 mg by instillation and as a nerve block Results from 7 positive Phase 2/3 studies across 5 surgical models are intended to support broad use of HTX-011 across a full range of surgical procedures

Large US Market Opportunity Target Market Opportunity Initial Targets Higher volume procedures across 4 major specialties ~6.5M Orthopedic procedures ~4.3M General Surgery procedures ~3.3 M OB/GYN procedures ~1.1M Plastic Surgery procedures Secondary Targets Higher volume procedures in non-core specialties (e.g., ENT, urology, hand, others) Tertiary Targets Lower volume procedures and procedures where local anesthetics are not widely used today ~28M Annual US Surgical Procedures Requiring Postoperative Pain Management That Were Considered Potentially Suited for HTX-011 ~15M procedures ~6M procedures ~7M procedures Target Market Opportunity Size* $4.9B $1.9B $2.3B *Based on the current WAC of Exparel
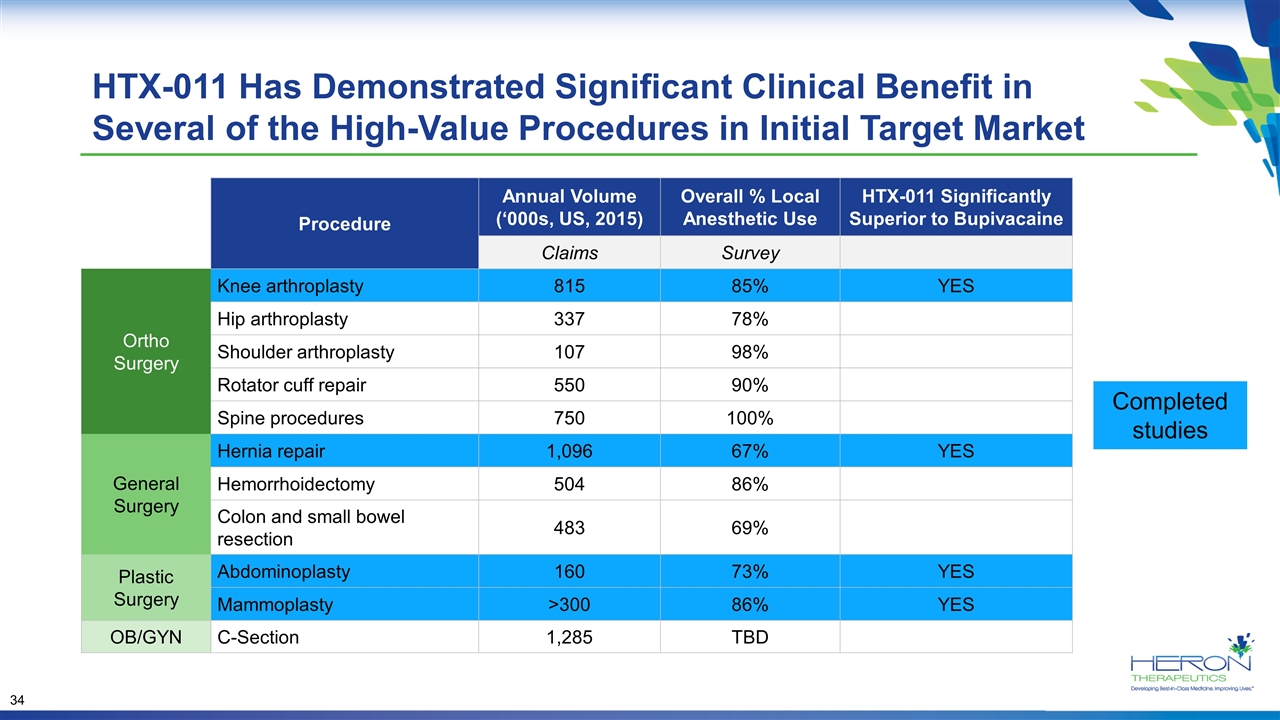
HTX-011 Has Demonstrated Significant Clinical Benefit in Several of the High-Value Procedures in Initial Target Market Completed studies Procedure Annual Volume (‘000s, US, 2015) Overall % Local Anesthetic Use HTX-011 Significantly Superior to Bupivacaine Claims Survey Ortho Surgery Knee arthroplasty 815 85% YES Hip arthroplasty 337 78% Shoulder arthroplasty 107 98% Rotator cuff repair 550 90% Spine procedures 750 100% General Surgery Hernia repair 1,096 67% YES Hemorrhoidectomy 504 86% Colon and small bowel resection 483 69% Plastic Surgery Abdominoplasty 160 73% YES Mammoplasty >300 86% YES OB/GYN C-Section 1,285 TBD
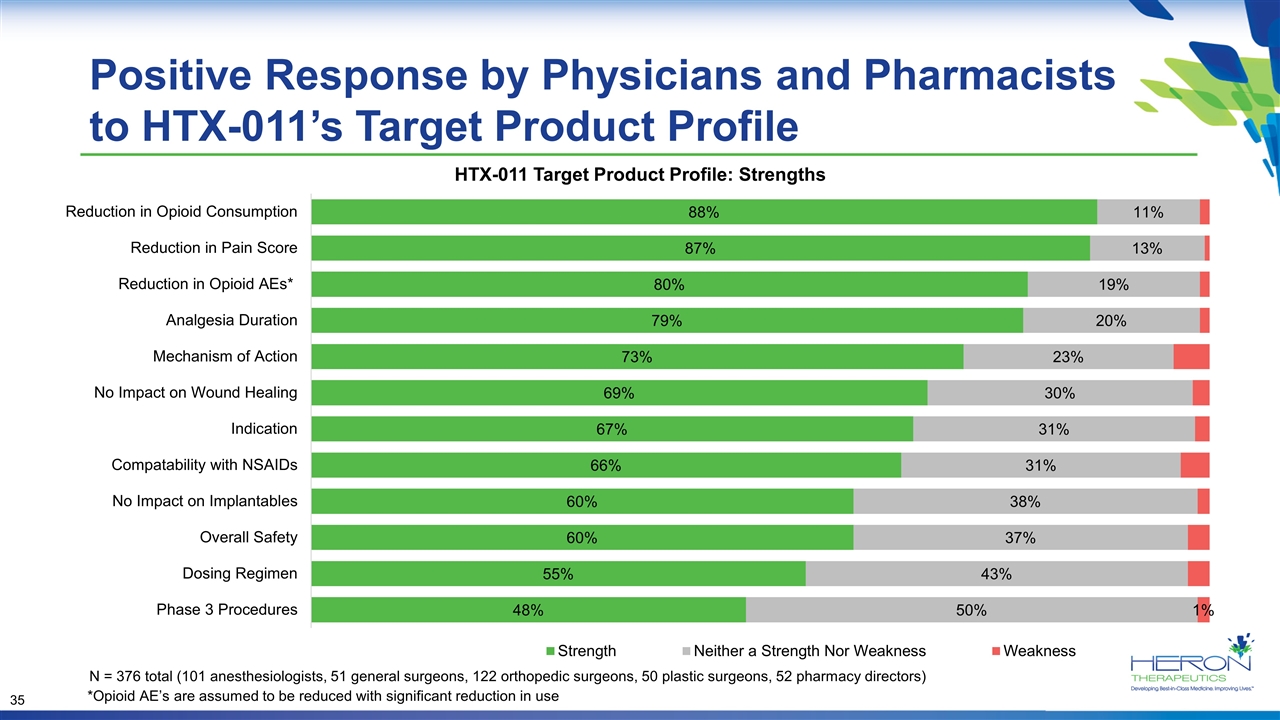
Positive Response by Physicians and Pharmacists to HTX-011’s Target Product Profile HTX-011 Target Product Profile: Strengths N = 376 total (101 anesthesiologists, 51 general surgeons, 122 orthopedic surgeons, 50 plastic surgeons, 52 pharmacy directors) *Opioid AE’s are assumed to be reduced with significant reduction in use