UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 10-Q
(Mark One)
| | | | | |
| ☒ | QUARTERLY REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the quarterly period ended June 30, 2023
or
| | | | | |
| ☐ | TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the transition period from to
Commission File Number: 000-21088
FRESH TRACKS THERAPEUTICS, INC.
(Exact name of registrant as specified in its charter)
| | | | | | | | | | | | | | | | | | | | | | | |
| Delaware | | 93-0948554 | |
| (State or other jurisdiction of incorporation or organization) | | (I.R.S. Employer Identification No.) | |
| | | | | | | |
| 5777 Central Avenue, | Boulder, | CO | | | 80301 | |
| (Address of principal executive offices) | | (Zip Code) | |
(720) 505-4755
(Registrant’s telephone number, including area code)
Securities registered pursuant to Section 12(b) of the Act:
| | | | | | | | |
| Title of each class | Trading symbol(s) | Name of each exchange on which registered |
| Common stock, $0.01 par value per share | FRTX | The Nasdaq Stock Market LLC |
Indicate by check mark whether the registrant: (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. Yes ☒ No ☐
Indicate by check mark whether the registrant has submitted electronically every Interactive Data File required to be submitted pursuant to Rule 405 of Regulation S-T (§232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit such files). Yes ☒ No ☐
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, a smaller reporting company, or an emerging growth company. See the definitions of “large accelerated filer,” “accelerated filer,” “smaller reporting company,” and “emerging growth company” in Rule 12b-2 of the Exchange Act.
| | | | | | | | | | | |
| Large accelerated filer | ☐ | Accelerated filer | ☐ |
| Non-accelerated filer | ☒ | Smaller reporting company | ☒ |
| | Emerging growth company | ☐ |
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Exchange Act). Yes ☐ No ☒
As of August 3, 2023, there were 5,906,475 shares of the registrant’s common stock outstanding.
FORWARD-LOOKING STATEMENTS
This Quarterly Report on Form 10-Q (“Quarterly Report”) contains forward-looking statements that involve substantial risks and uncertainties for purposes of the safe harbor provided by the Private Securities Litigation Reform Act of 1995. All statements contained in this Quarterly Report other than statements of historical fact, including statements relating to future financial, business, conditions, plans, prospects, impacts, shifts, trends, progress, or strategies and other such matters, including without limitation, our strategy; future operations; future potential; future financial position; future liquidity; future revenue; territorial focus; projected expenses; results of operations; the anticipated timing, scope, design, results, and regulation of clinical trials; intellectual property rights, including the acquisition, validity, term, and enforceability of such; the expected timing and/or results of regulatory submissions and approvals; and prospects for treatment of patients and commercializing (and competing with) any product candidates for any disease by Fresh Tracks or third parties, or research and/or licensing collaborations with, or actions of, its partners, including in the United States (“U.S.”), Japan, South Korea, or any other country. The words “may,” “could,” “should,” “might,” “show,” “maximize,” “continue,” “additional,” “announce,” “anticipate,” “explore,” “reflect,” “believe,” “sufficient,” “transform,” “estimate,” “expect,” “intend,” “plan,” “predict,” “potential,” “will,” evaluate,” “aim,” “help,” “progress,” “meet,” “support,” “look forward,” “develop,” “strengthen,” “promise,” “successful,” “positive,” “provide,” “commit,” “first-in-class,” “opportunity,” “disrupt,” “reduce,” “restore,” “demonstrate,” “suggest,” “target,” “shift,” “inhibit,” and similar expressions and their variants, are intended to identify forward-looking statements. Such statements are based on management’s current expectations and involve risks and uncertainties. Actual results and performance could differ materially from those projected in the forward-looking statements as a result of many factors. Unless otherwise mentioned or unless the context requires otherwise, all references in this Quarterly Report to “Fresh Tracks,” “Brickell Subsidiary,” “Company,” “we,” “us,” and “our,” or similar references, refer to Fresh Tracks Therapeutics, Inc. and its consolidated subsidiaries.
We based these forward-looking statements largely on our current expectations and projections about future events and trends that we believe may affect our financial condition, results of operations, business strategy and business development activities, pipeline development status, clinical trials, short-term and long-term business operations and objectives, employees, and financial needs. These forward-looking statements are subject to a number of risks, uncertainties, and assumptions, including those described in Part I, Item 1A. “Risk Factors” in our Annual Report on Form 10-K for the year ended December 31, 2022, in Part II, Item 1A. “Risk Factors” in our Quarterly Report on Form 10-Q for the quarter ended March 31, 2023, in Part II, Item 1A, “Risk Factors” in this Quarterly Report, and under a similar heading in any other periodic or current report we may file with the U.S. Securities and Exchange Commission (the “SEC”) in the future. Moreover, we operate in a very competitive and rapidly changing environment. New risks emerge quickly and from time to time. It is not possible for our management to predict all risks, nor can we assess the impact of all factors on our business and operations or the extent to which any factor, or combination of factors, may cause actual results to differ materially from those contained in any forward-looking statements we may make. In light of these risks, uncertainties, and assumptions, the future events and trends discussed in this Quarterly Report may not occur and actual results could differ materially and adversely from those anticipated or implied in the forward-looking statements. We undertake no obligation to revise or publicly release the results of any revision to these forward-looking statements, except as required by law. Given these risks and uncertainties, readers are cautioned not to place undue reliance on such forward-looking statements. All forward-looking statements are qualified in their entirety by this cautionary statement.
You should read carefully the factors described in Part I, Item 1A. “Risk Factors” in our Annual Report on Form 10-K for the year ended December 31, 2022, in Part II, Item 1A, “Risk Factors” in this Quarterly Report, and under a similar heading in any other periodic or current report we may file with the SEC to better understand the risks and uncertainties inherent in our business and underlying any forward-looking statements. You are advised to consult any further disclosures we make on related subjects in our future public filings and on our website.
FRESH TRACKS THERAPEUTICS, INC.
FORM 10-Q
TABLE OF CONTENTS
PART I. FINANCIAL INFORMATION
ITEM 1. FINANCIAL STATEMENTS
FRESH TRACKS THERAPEUTICS, INC.
CONDENSED CONSOLIDATED BALANCE SHEETS
(in thousands, except share and per share data)
(unaudited)
| | | | | | | | | | | |
| |
| | | |
| June 30,
2023 | | December 31,
2022 |
| | | |
| Assets | | | |
| Current assets: | | | |
| Cash and cash equivalents | $ | 8,948 | | | $ | 8,680 | |
| | | |
| Prepaid expenses and other current assets | 761 | | | 1,403 | |
| Total current assets | 9,709 | | | 10,083 | |
| Property and equipment, net | 55 | | | 75 | |
| Contract asset, net of current portion | 61 | | | 64 | |
| Operating lease right-of-use asset | 17 | | | 49 | |
| | | |
| Total assets | $ | 9,842 | | | $ | 10,271 | |
Liabilities and stockholders’ equity | | | |
| Current liabilities: | | | |
| Accounts payable | $ | 256 | | | $ | 571 | |
| Accrued liabilities | 1,575 | | | 2,457 | |
| Lease liability | 19 | | | 49 | |
| | | |
| | | |
| | | |
| | | |
| | | |
| Total current liabilities | 1,850 | | | 3,077 | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| Commitments and contingencies (Note 5) | | | |
| | | |
| | | |
| Stockholders’ equity: | | | |
| Common stock, $0.01 par value, 300,000,000 shares authorized as of June 30, 2023 and December 31, 2022; 5,906,475 and 3,018,940 shares issued and outstanding as of June 30, 2023 and December 31, 2022, respectively | 59 | | | 30 | |
| Additional paid-in capital | 180,962 | | | 173,633 | |
| | | |
| Accumulated deficit | (173,029) | | | (166,469) | |
| Total stockholders’ equity | 7,992 | | | 7,194 | |
| Total liabilities and stockholders’ equity | $ | 9,842 | | | $ | 10,271 | |
| | | |
See accompanying notes to these condensed consolidated financial statements.
FRESH TRACKS THERAPEUTICS, INC.
CONDENSED CONSOLIDATED STATEMENTS OF OPERATIONS
(in thousands, except share and per share data)
(unaudited)
| | | | | | | | | | | | | | | | | | | | | | | |
| Three Months Ended
June 30, | | Six Months Ended
June 30, |
| 2023 | | 2022 | | 2023 | | 2022 |
| Revenue | | | | | | | |
| Contract revenue | $ | 53 | | | $ | 4,315 | | | $ | 62 | | | $ | 4,315 | |
| Royalty revenue | — | | | — | | | — | | | 92 | |
| Total revenue | 53 | | | 4,315 | | | 62 | | | 4,407 | |
| | | | | | | |
| Operating expenses: | | | | | | | |
| Research and development | 609 | | | 1,865 | | | 2,545 | | | 7,878 | |
| General and administrative | 1,808 | | | 3,908 | | | 4,222 | | | 7,394 | |
| Total operating expenses | 2,417 | | | 5,773 | | | 6,767 | | | 15,272 | |
| Loss from operations | (2,364) | | | (1,458) | | | (6,705) | | | (10,865) | |
| Other income | 82 | | | 313 | | | 150 | | | 314 | |
| | | | | | | |
| Interest expense | (2) | | | (2) | | | (5) | | | (6) | |
| | | | | | | |
| | | | | | | |
| | | | | | | |
| | | | | | | |
| Net loss attributable to common stockholders | $ | (2,284) | | | $ | (1,147) | | | $ | (6,560) | | | $ | (10,557) | |
| | | | | | | |
| | | | | | | |
| | | | | | | |
| | | | | | | |
| Net loss per common share attributable to common stockholders, basic and diluted | $ | (0.39) | | | $ | (0.43) | | | $ | (1.36) | | | $ | (3.98) | |
| | | | | | | |
| Weighted-average shares used to compute net loss per common share attributable to common stockholders, basic and diluted | 5,906,475 | | | 2,655,251 | | | 4,837,483 | | | 2,654,046 | |
See accompanying notes to these condensed consolidated financial statements.
FRESH TRACKS THERAPEUTICS, INC.
CONDENSED CONSOLIDATED STATEMENTS OF STOCKHOLDERS’ EQUITY
(in thousands, except share data)
(unaudited)
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| Series A Redeemable
Preferred Stock | | Common Stock | | Additional
Paid-In-Capital | | | | Accumulated
Deficit | | Total
Stockholders’
Equity |
| Shares | | Par Value | | Shares | | Par Value | | |
| Balance, December 31, 2021 | — | | | $ | — | | | 2,652,828 | | | $ | 27 | | | $ | 170,247 | | | | | $ | (145,367) | | | $ | 24,907 | |
| | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | |
| Stock-based compensation | — | | | — | | | — | | | — | | | 551 | | | | | — | | | 551 | |
| | | | | | | | | | | | | | | |
| Net loss | — | | | — | | | — | | | — | | | — | | | | | (9,410) | | | (9,410) | |
| Balance, March 31, 2022 | — | | | — | | | 2,652,828 | | | 27 | | | 170,798 | | | | | (154,777) | | | 16,048 | |
| Issuance of redeemable preferred stock | 1 | | | — | | | — | | | — | | | — | | | | | — | | | — | |
| | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | |
| Common stock issued, net of issuance costs of $46 | — | | | — | | | 31,557 | | | — | | | 131 | | | | | — | | | 131 | |
| | | | | | | | | | | | | | | |
| Issuance of common stock for cash under employee stock purchase plan | — | | | — | | | 5,975 | | | — | | | 29 | | | | | — | | | 29 | |
| Stock-based compensation | — | | | — | | | — | | | — | | | 576 | | | | | — | | | 576 | |
| | | | | | | | | | | | | | | |
| Net loss | — | | | — | | | — | | | — | | | — | | | | | (1,147) | | | (1,147) | |
| Balance, June 30, 2022 | 1 | | | $ | — | | | 2,690,360 | | | $ | 27 | | | $ | 171,534 | | | | | $ | (155,924) | | | $ | 15,637 | |
| | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| Series A Redeemable
Preferred Stock | | Common Stock | | Additional
Paid-In-Capital | | | | Accumulated
Deficit | | Total
Stockholders’
Equity |
| Shares | | Par Value | | Shares | | Par Value | | |
| Balance, December 31, 2022 | — | | | $ | — | | | 3,018,940 | | | $ | 30 | | | $ | 173,633 | | | | | $ | (166,469) | | | $ | 7,194 | |
| | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | |
| Common stock issued pursuant to ATM agreements, net of issuance costs of $202 | — | | | — | | | 2,887,535 | | | 29 | | | 6,540 | | | | | — | | | 6,569 | |
| | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | |
| Stock-based compensation | — | | | — | | | — | | | — | | | 379 | | | | | — | | | 379 | |
| | | | | | | | | | | | | | | |
| Net loss | — | | | — | | | — | | | — | | | — | | | | | (4,276) | | | (4,276) | |
| Balance, March 31, 2023 | — | | | — | | | 5,906,475 | | | 59 | | | 180,552 | | | | | (170,745) | | | 9,866 | |
| | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | |
| Stock-based compensation | — | | | — | | | — | | | — | | | 410 | | | | | — | | | 410 | |
| | | | | | | | | | | | | | | |
| Net loss | — | | | — | | | — | | | — | | | — | | | | | (2,284) | | | (2,284) | |
| Balance, June 30, 2023 | — | | | $ | — | | | 5,906,475 | | | $ | 59 | | | $ | 180,962 | | | | | $ | (173,029) | | | $ | 7,992 | |
See accompanying notes to these condensed consolidated financial statements.
FRESH TRACKS THERAPEUTICS, INC.
CONDENSED CONSOLIDATED STATEMENTS OF CASH FLOWS
(unaudited, in thousands)
| | | | | | | | | | | |
| Six Months Ended
June 30, |
| 2023 | | 2022 |
| CASH FLOWS FROM OPERATING ACTIVITIES: | | | |
| Net loss | $ | (6,560) | | | $ | (10,557) | |
| Adjustments to reconcile net loss to net cash used in operating activities: | | | |
| Stock-based compensation | 789 | | | 1,127 | |
| Non-cash operating lease expense | 43 | | | 29 | |
| Depreciation | 20 | | | 13 | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| Changes in operating assets and liabilities: | | | |
| Prepaid expenses and other current assets, including noncurrent portion of contract asset | 645 | | | (1,236) | |
| Accounts payable | (315) | | | (342) | |
| Accrued liabilities | (882) | | | (1,510) | |
| Operating lease liability | (41) | | | (33) | |
| | | |
| | | |
| | | |
| | | |
| Net cash used in operating activities | (6,301) | | | (12,509) | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| CASH FLOWS FROM FINANCING ACTIVITIES: | | | |
| | | |
| | | |
| Proceeds from the issuance of common stock pursuant to ATM agreements, net of issuance costs | 6,569 | | | 131 | |
| Payments of taxes related to net share settlement of equity awards | — | | | (55) | |
| Proceeds from the issuance of common stock under employee stock purchase plan | — | | | 29 | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| Net cash provided by financing activities | 6,569 | | | 105 | |
| | | |
| NET INCREASE (DECREASE) IN CASH AND CASH EQUIVALENTS | 268 | | | (12,404) | |
| CASH AND CASH EQUIVALENTS—BEGINNING | 8,680 | | | 26,884 | |
| CASH AND CASH EQUIVALENTS—ENDING | $ | 8,948 | | | $ | 14,480 | |
| | | |
| | | |
| | | |
| Supplemental Disclosure of Non-Cash Investing and Financing Activities: | | | |
| Acquisition of right-of-use asset through lease liability | $ | 11 | | | $ | — | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
See accompanying notes to these condensed consolidated financial statements.
FRESH TRACKS THERAPEUTICS, INC.
NOTES TO CONDENSED CONSOLIDATED FINANCIAL STATEMENTS
(unaudited)
NOTE 1. ORGANIZATION AND NATURE OF OPERATIONS
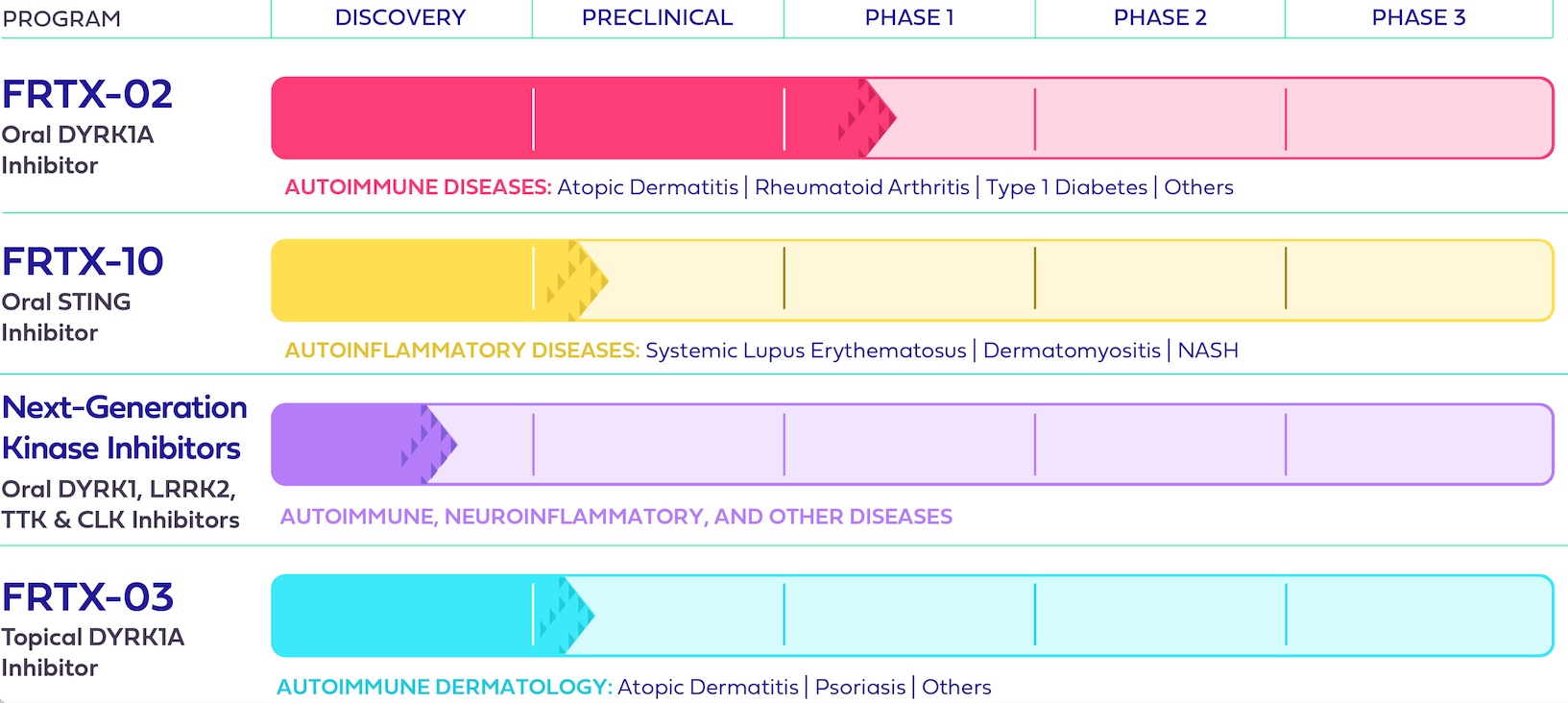
Fresh Tracks Therapeutics, Inc. (the “Company” or “Fresh Tracks”) is a clinical-stage pharmaceutical company striving to transform patient lives through the development of innovative and differentiated prescription therapeutics. The Company’s pipeline aims to disrupt existing treatment paradigms and features several new chemical entities that inhibit novel targets with first-in-class potential for autoimmune, inflammatory, and other debilitating diseases. This includes FRTX-02, a DYRK1A inhibitor for the treatment of certain autoimmune and inflammatory diseases; FRTX-10, a preclinical-stage Stimulator of Interferon Genes (“STING”) inhibitor candidate for the potential treatment of autoimmune, inflammatory, and rare genetic diseases; and a platform of next-generation kinase inhibitors that could produce treatments for autoimmune, inflammatory, and other debilitating diseases.
Reverse Stock Split
On July 5, 2022, the Company effected a 1-for-45 reverse stock split of outstanding shares of its common stock. All common stock share and per-share amounts, and other related balances and computations, reported as of and for all periods presented in the condensed consolidated financial statements and notes reflect the adjusted common stock share and per-share amounts, and other related balances and computations, that were effective on and after July 5, 2022.
Liquidity and Capital Resources
The Company has incurred significant operating losses and has an accumulated deficit as a result of ongoing efforts to in-license and develop product candidates, including conducting preclinical and clinical trials and providing general and administrative support for these operations. For the six months ended June 30, 2023, the Company had a net loss of $6.6 million and net cash used in operating activities of $6.3 million. As of June 30, 2023, the Company had cash and cash equivalents of $8.9 million and an accumulated deficit of $173.0 million. The Company believes that its cash and cash equivalents as of June 30, 2023, combined with $6.6 million in net proceeds received in July 2023 (as described further in Note 3. “Strategic Agreements”), will be sufficient to fund its operations for at least the next 12 months.
The Company expects to continue to incur additional losses for the foreseeable future. The Company’s board of directors (“Board”) and executive management team are conducting a comprehensive process to explore and evaluate strategic alternatives with the goal of maximizing shareholder value. Potential alternatives under evaluation include, but are not limited to, a financing, a merger or reverse merger, the sale of all or part of the Company, licensing of assets, a business combination, and/or other strategic transaction or series of related transactions involving the Company. In order to commence additional clinical trials for FRTX-02, or meaningfully progress the Company’s other pipeline assets, the Company would need to raise additional funds or enter into a strategic transaction with a partner who wishes to continue development of these assets.
NOTE 2. SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES
Basis of Presentation
The accompanying condensed consolidated financial statements include the accounts of the Company and its wholly-owned subsidiary, Brickell Subsidiary, Inc. (“Brickell Subsidiary”), and are presented in U.S. dollars and have been prepared in accordance with accounting principles generally accepted in the United States of America (“U.S. GAAP”) and applicable rules and regulations of the SEC for interim reporting. As permitted
under those rules and regulations, certain footnotes or other financial information normally included in financial statements prepared in accordance with U.S. GAAP have been condensed or omitted. These condensed consolidated financial statements have been prepared on the same basis as the annual financial statements and, in the opinion of management, reflect all adjustments, consisting only of normal recurring adjustments, which are necessary for a fair presentation of the Company’s financial information. The results of operations for the three and six months ended June 30, 2023 are not necessarily indicative of the results to be expected for the full year ending December 31, 2023, for any other interim period, or for any other future period. The condensed consolidated balance sheet as of December 31, 2022 has been derived from audited financial statements at that date but does not include all of the information required by U.S. GAAP for complete financial statements. All intercompany balances and transactions have been eliminated in consolidation. The Company operates in one operating segment and, accordingly, no segment disclosures have been presented herein.
Significant Accounting Policies
The Company’s significant accounting policies are described in Note 2. “Summary of Significant Accounting Policies” in the Company’s Annual Report on Form 10-K for the year ended December 31, 2022. During the six months ended June 30, 2023, the Company did not adopt any additional significant accounting policies.
Reclassifications
Certain comparative figures in the prior year condensed consolidated statement of cash flows within operating activities have been reclassified to conform to the current period presentation. These reclassifications did not impact total net cash used in operating activities.
Use of Estimates
The Company’s condensed consolidated financial statements are prepared in accordance with U.S. GAAP, which requires it to make estimates and assumptions that affect the reported amounts of assets and liabilities and disclosure of contingent assets and liabilities at the date of the financial statements and the reported amounts of revenue and expenses during the reporting period. Although these estimates are based on the Company’s knowledge of current events and actions it may take in the future, actual results may ultimately differ from these estimates and assumptions.
Risks and Uncertainties
The Company’s business is subject to significant risks common to early-stage companies in the pharmaceutical industry including, but not limited to, the ability to develop appropriate formulations, scale up and produce the compounds; dependence on collaborative parties; uncertainties associated with obtaining and enforcing patents and other intellectual property rights; nonclinical and clinical implementation and success; the lengthy and expensive regulatory approval process; compliance with regulatory and other legal requirements; competition from other products; significant competition; ability to manage third-party manufacturers, suppliers, contract research organizations, business partners and other alliances; and obtaining additional financing to fund the Company’s efforts.
The Company expects to incur additional losses for the foreseeable future and would need to obtain additional financing in order to develop its product candidates. There can be no assurance that such financing will be available or will be at terms acceptable to the Company.
Cash and Cash Equivalents
The Company considers all highly liquid investments with an original maturity of three months or less from date of purchase to be cash equivalents. Cash equivalents consist primarily of amounts held in short-term money market accounts with highly rated financial institutions.
Concentrations of Credit Risk
Financial instruments that potentially subject the Company to concentrations of credit risk consist principally of cash and cash equivalents, accounts receivable, and contract asset. The Company maintains cash and cash equivalents balances in several accounts with two financial institutions which, from time to time, are in excess of federally insured limits.
One third party individually accounted for all of the Company’s revenue for the three and six months ended June 30, 2023 and 2022, as well as associated accounts receivable and contract asset balances as of June 30, 2023 and December 31, 2022. Refer to Note 3. “Strategic Agreements” for a detailed discussion of agreements with Botanix SB Inc. and Botanix Pharmaceuticals Limited (“Botanix”).
Fair Value Measurements
Fair value is the price that the Company would receive to sell an asset or pay to transfer a liability in a timely transaction with an independent counterparty in the principal market, or in the absence of a principal market, the most advantageous market for the asset or liability. A three-tier hierarchy distinguishes between (1) inputs that reflect the assumptions market participants would use in pricing an asset or liability developed based on market data obtained from sources independent of the reporting entity (observable inputs) and (2) inputs that reflect the reporting entity’s own assumptions about the assumptions market participants would use in pricing an asset or liability developed based on the best information available in the circumstances (unobservable inputs). The hierarchy is summarized in the three broad levels listed below:
Level 1—quoted prices in active markets for identical assets and liabilities
Level 2—other significant observable inputs (including quoted prices for similar assets and liabilities, interest rates, credit risk, etc.)
Level 3—significant unobservable inputs (including the Company’s own assumptions in determining the fair value of assets and liabilities)
The following table sets forth the fair value of the Company’s financial assets measured at fair value on a recurring basis based on the three-tier fair value hierarchy (in thousands):
| | | | | | | | | | | | | | | |
| Level 1 | | | | |
| | | | | |
| | | | | | | |
| June 30,
2023 | | December 31,
2022 | |
| Assets: | | | | | | | |
| Money market funds | $ | 6,127 | | | $ | 7,680 | | | | | |
| | | | | | | |
| | | | | | | |
| | | | | | | |
| | | | | | | |
| | | | | | | |
| | | | | | | |
| | | | | | | |
| | | | | | | |
| | | | | | | |
Fair Value of Financial Instruments
The following methods and assumptions were used by the Company in estimating the fair values of each class of financial instrument disclosed herein:
Money Market Funds—The carrying amounts reported as cash and cash equivalents in the condensed consolidated balance sheets approximate their fair values due to their short-term nature and market rates of interest.
The carrying values of cash equivalents, accounts receivable, accounts payable, and accrued liabilities approximate fair value due to the short-term maturity of those items.
Revenue Recognition
The Company has historically recognized revenue primarily from upfront fees, research and development milestones, research reimbursements, and consulting services fees related to the development of previously owned or sublicensed assets associated with the proprietary compound sofpironium bromide, as well as sublicense income and royalty fees on sales of sofpironium bromide gel, 5% (ECCLOCK®) in Japan.
The Company recognizes revenue upon the transfer of promised goods or services to customers in an amount that reflects the consideration to which the Company expects to be entitled in exchange for those goods or services. In determining the appropriate amount of revenue to be recognized, the Company performs the following five steps: (i) identify the contract(s) with a customer; (ii) identify the performance obligations in the contract; (iii) determine the transaction price, including the constraint on variable consideration; (iv) allocate the transaction price to the performance obligations in the contract; and (v) recognize revenue when (or as) the Company satisfies the performance obligations. At contract inception, the Company assesses the goods or services promised within each contract and assesses whether each promised good or service is distinct and determines those that are performance obligations. The Company then recognizes as revenue the amount of the transaction price that is allocated to the respective performance obligation when (or as) the performance obligation is satisfied.
The Company utilizes judgment to assess the nature of the performance obligation to determine whether the performance obligation is satisfied over time or at a point in time and, if over time, the appropriate method of measuring progress. The Company evaluates the measure of progress each reporting period and, if necessary, adjusts the measure of performance and related revenue recognition.
Contract Revenue
The Company evaluates its contracts, including asset sale arrangements that involve the Company’s rights to intellectual property, to determine whether they are outputs of the Company’s ordinary activities and whether the counterparty meets the definition of a customer. If the arrangement is determined to be a contract with a customer and the goods or services sold are determined to be distinct from other performance obligations identified in the arrangement, the Company recognizes revenue primarily from non-refundable upfront fees, milestone payments, sales-based payments, and fees for consulting services allocated to the goods or services when (or as) control is transferred to the customer, and the customer can use and benefit from the goods or services.
Licenses of Intellectual Property
If a license for the Company’s intellectual property is determined to be distinct from the other performance obligations identified in the arrangement, the Company recognizes revenue when the functional license is transferred to the customer, and the customer can use and benefit from the license.
Milestones
At the inception of each arrangement that includes milestone payments (variable consideration), excluding sales-based milestone payments discussed below, the Company evaluates whether the milestones are considered probable of being reached and estimates the amount to be included in the transaction price using the most likely amount method. The most likely amount method is generally utilized when there are only two possible outcomes and represents the Company’s best estimate of the single most likely outcome to be achieved. If it is probable that a significant revenue reversal would not occur, the variable consideration for the associated milestone is included in the transaction price. Milestone payments contingent on regulatory approvals that are not within the Company or the Company’s collaboration partner’s control, as applicable, are generally not considered probable of being achieved until those approvals are received. At the end of each subsequent reporting period, the Company re-evaluates the probability of achievement of milestones and any related constraint, and if necessary, adjusts the Company’s estimate of the variable consideration. Any such adjustments are recorded on a cumulative catch-up basis, which would affect revenue in the period of adjustment.
Sales-Based Payments
For license arrangements that include sales-based payments such as royalties or milestone payments based on the level of sales, and for which the license is deemed to be the predominant item to which the sales-based payments relate, the Company recognizes revenue at the later of (i) when the related sales occur or (ii) when the performance obligation to which some or all of the sales-based payment has been allocated has been satisfied (or partially satisfied). Sales-based payments received under license arrangements are recorded as royalty revenue in the Company’s condensed consolidated statements of operations.
For non-license arrangements that include sales-based payments, including earnout payments and milestone payments based on the level of sales, the Company estimates the sales-based payments (variable consideration) to be achieved and recognizes revenue to the extent it is probable that a significant reversal in the amount of cumulative revenue recognized will not occur. The Company may use either the most likely amount, as described above, or the expected value method, in making such estimates based on the nature of the payment to be received and whether there is a wide range of outcomes or only two possible outcomes. The expected value method represents the sum of probability-weighted amounts in a range of possible consideration amounts. The Company bases its estimates using the applicable method described above on factors such as, but not limited to, required regulatory approvals, historical sales levels, market events and projections, and other factors as appropriate. The Company updates its estimates at each reporting period based on actual results and future expectations as necessary.
Contract Asset
For non-license arrangements involving the sale and transfer of the Company’s intellectual property rights, the Company recognizes estimated variable consideration as revenue as discussed above before the customer pays consideration or before payment is due. The estimated revenue recognized is presented as a contract asset on the Company’s condensed consolidated balance sheets. The current portion of the contract asset is presented in prepaid expenses and other current assets on the Company’s condensed consolidated balance sheets. Actual amounts paid or due by the customer are recorded as a reduction to the contract asset. Any revisions to the Company’s estimated revenue based on actual results and future expectations are recognized as an adjustment to the contract asset.
Research and Development
Research and development costs are charged to expense when incurred and consist of costs incurred for independent and collaboration research and development activities. The major components of research and
development costs include formulation development, nonclinical studies, clinical studies, clinical manufacturing costs, in-licensing fees for development-stage assets, salaries and employee benefits, and allocations of various overhead and occupancy costs. Research costs typically consist of applied research, preclinical, and toxicology work. Pharmaceutical manufacturing development costs consist of product formulation, chemical analysis, and the transfer and scale-up of manufacturing at contract manufacturers. Assets acquired (or in-licensed) that are utilized in research and development that have no alternative future use are expensed as incurred. Milestone payments related to the Company’s acquired (or in-licensed) assets are recorded as research and development expenses when probable and reasonably estimable.
Costs for certain research and development activities, such as clinical trial expenses, are recognized based on an evaluation of the progress to completion of specific tasks using data such as patient enrollment, clinical site activations, and information provided to the Company by its vendor on their actual costs incurred or level of effort expended. Payments for these activities are based on the terms of the individual arrangements, which may differ from the pattern of costs incurred, and are reflected on the condensed consolidated balance sheets as prepaid expenses and other current assets or accrued expenses.
When the Company enters into licensing or subscription arrangements to access and utilize certain technology, the Company evaluates if the license agreement results in the acquisition of an asset or a business. To date, none of the Company’s license agreements have been considered an acquisition of a business. For asset acquisitions, the upfront payments to acquire such licenses, as well as any future milestone payments made before product approval that do not meet the definition of a derivative, are immediately recognized as research and development expenses when they are paid or become payable, provided there is no alternative future use of the rights in other research and development projects.
Net Loss per Share
Basic and diluted net loss per share is computed by dividing net loss by the weighted-average number of common shares outstanding. When the effects are not anti-dilutive, diluted earnings per share is computed by dividing the Company’s net income by the weighted-average number of common shares outstanding and the impact of all potentially dilutive common shares. Diluted net loss per share is the same as basic net loss per share, as the effects of potentially dilutive securities are anti-dilutive for all periods presented.
The following table sets forth the potential common shares excluded from the calculation of diluted net loss per share because their inclusion would be anti-dilutive:
| | | | | | | | | | | | | | | |
| | | Three and Six Months Ended
June 30, |
| | | | | 2023 | | 2022 |
| Outstanding warrants | | | | | 621,063 | | | 621,063 | |
| Outstanding options | | | | | 193,777 | | | 232,084 | |
| Unvested restricted stock units | | | | | 141,250 | | | — | |
| | | | | | | |
| | | | | | | |
| Total | | | | | 956,090 | | | 853,147 | |
Leases
The Company determines if an arrangement is a lease at inception. Operating leases with a term greater than one year are recognized on the condensed consolidated balance sheets as right-of-use assets and lease liabilities. The Company does not currently hold any finance leases. The Company has elected the practical expedient not to recognize on the condensed consolidated balance sheets leases with terms of one year or less and not to separate lease components and non-lease components for real estate leases. Lease liabilities and their corresponding right-of-use assets are recorded based on the present value of lease payments over the expected lease term. The interest rate implicit in lease contracts is typically not readily determinable. As such, the
Company estimates the incremental borrowing rate in determining the present value of lease payments. Lease expense is recognized on a straight-line basis over the lease term.
New Accounting Pronouncements
From time to time, new accounting pronouncements are issued by the Financial Accounting Standards Board or other standard setting bodies that the Company adopts as of the specified effective date. The Company does not believe that the adoption of recently issued standards has had or will have a material impact on the Company's condensed consolidated financial statements or disclosures.
NOTE 3. STRATEGIC AGREEMENTS
License and Development Agreement with Voronoi
On August 27, 2021, the Company entered into a License and Development Agreement (the “Voronoi License Agreement”) with Voronoi Inc. (“Voronoi”), pursuant to which the Company acquired exclusive, worldwide rights to research, develop, and commercialize FRTX-02 and other next-generation kinase inhibitors.
With respect to FRTX-02, the Voronoi License Agreement provides that the Company will make payments to Voronoi of up to $211.0 million in the aggregate contingent upon achievement of specified development, regulatory, and commercial milestones. With respect to the compounds arising from the next-generation kinase inhibitor platform, the Company will make payments to Voronoi of up to $107.5 million in the aggregate contingent upon achievement of specified development, regulatory, and commercial milestones. Further, the Voronoi License Agreement provides that the Company will pay Voronoi tiered royalty payments ranging from low-single digits up to 10% of net sales of products arising from the DYRK1A inhibitor programs and next-generation kinase inhibitor platform. All of the contingent payments and royalties are payable in cash in U.S. Dollars, except for $1.0 million of the development and regulatory milestone payments, which amount is payable in equivalent shares of the Company’s common stock. Under the terms of the Voronoi License Agreement, the Company is responsible for, and bears the future costs of, all development and commercialization activities, including patenting, related to all the licensed compounds. As of June 30, 2023 and through the date of this Quarterly Report, the Company has not yet made any payments or recorded any liabilities related to the specified development, regulatory, and commercial milestones or royalties on net sales pursuant to the Voronoi License Agreement.
Exclusive License and Development Agreement with Carna
On February 2, 2022, the Company entered into an Exclusive License Agreement (the “Carna License Agreement”) with Carna Biosciences, Inc. (“Carna”), pursuant to which the Company acquired exclusive, worldwide rights to research, develop, and commercialize Carna’s portfolio of novel STING inhibitors. In accordance with the terms of the Carna License Agreement, in exchange for the licensed rights, the Company made a one-time cash payment of $2.0 million, which was recorded as research and development expenses in the condensed consolidated statements of operations during the six months ended June 30, 2022.
The Carna License Agreement provides that the Company will make success-based payments to Carna of up to $258.0 million in the aggregate contingent upon achievement of specified development, regulatory, and commercial milestones. Further, the Carna License Agreement provides that the Company will pay Carna tiered royalty payments ranging from mid-single digits up to 10% of net sales. All of the contingent payments and royalties are payable in cash in U.S. Dollars. Under the terms of the Carna License Agreement, the Company is responsible for, and bears the future costs of, all development and commercialization activities, including patenting, related to all the licensed compounds. As of June 30, 2023 and through the date of this Quarterly Report, the Company has not yet made any payments or recorded any liabilities related to the specified
development, regulatory, and commercial milestones or royalties on net sales pursuant to the Carna License Agreement.
Agreements with Botanix
Asset Purchase Agreement with Botanix
On May 3, 2022 (the “Effective Date”), the Company and Brickell Subsidiary entered into an asset purchase agreement with Botanix (the “Asset Purchase Agreement”), pursuant to which Botanix acquired and assumed control of all rights, title, and interests to assets primarily related to the proprietary compound sofpironium bromide that were owned and/or licensed by the Company or Brickell Subsidiary (the “Assets”). Prior to the sale of the Assets, the Company had previously entered into a License Agreement with Bodor Laboratories, Inc. (“Bodor”), dated December 15, 2012 (last amended in February 2020) that provided the Company with a worldwide exclusive license to develop, manufacture, market, sell, and sublicense products containing sofpironium bromide through which the Assets were developed (the “Amended and Restated License Agreement”). As a result of the Asset Purchase Agreement, Botanix is now responsible for all further research, development, and commercialization of sofpironium bromide globally and replaced the Company as the exclusive licensee under the Amended and Restated License Agreement.
In accordance with the sublicense rights provided to the Company under the Amended and Restated License Agreement, the Company also had previously entered into a License, Development, and Commercialization Agreement with Kaken Pharmaceutical Co., Ltd. (“Kaken”), dated as of March 31, 2015 (as amended in May 2018, the “Kaken Agreement”), under which the Company granted to Kaken an exclusive right to develop, manufacture, and commercialize the sofpironium bromide compound in Japan and certain other Asian countries (the “Territory”). In exchange for the sublicense, the Company was entitled to receive aggregate payments of up to $10.0 million upon the achievement of specified development milestones, which were earned and received in 2017 and 2018, and up to $19.0 million upon the achievement of sales-based milestones, as well as tiered royalties based on a percentage of net sales of licensed products in the Territory. In September 2020, Kaken received regulatory approval in Japan to manufacture and market ECCLOCK for the treatment of primary axillary hyperhidrosis, and as a result, the Company began recognizing royalty revenue earned on a percentage of net sales of ECCLOCK in Japan. Pursuant to the Asset Purchase Agreement, the Kaken Agreement was assigned to Botanix, which replaced the Company as the exclusive sub-licensor to Kaken. During the three and six months ended June 30, 2022, prior to entering into the Asset Purchase Agreement, the Company recognized royalty revenue under the Kaken Agreement of $0 and $0.1 million, respectively.
The Company determined that the development of and ultimate sale and assignment of rights to the Assets is an output of the Company’s ordinary activities and Botanix is a customer as it relates to the sale of the Assets and related activities.
In accordance with the terms of the Asset Purchase Agreement, in exchange for the Assets, the Company (i) received an upfront payment at closing in the amount of $3.0 million, (ii) was reimbursed for certain recent development expenditures in advancement of the Assets, (iii) received a milestone payment of $2.0 million upon the acceptance by the U.S. Food and Drug Administration (“FDA”) in December 2022 of the filing of a new drug application (“NDA”) for sofpironium bromide gel, 15%, and (iv) would have been eligible to receive a contingent milestone payment of $4.0 million if marketing approval in the U.S. for sofpironium bromide gel, 15%, is received on or before September 30, 2023, or $2.5 million if such marketing approval is received after September 30, 2023 but on or before February 17, 2024. Botanix submitted an NDA for sofpironium bromide gel, 15%, to the FDA in September 2022, which was accepted by the FDA in December 2022. Under the Asset Purchase Agreement, the Company also would have been eligible to receive additional success-based regulatory and sales milestone payments of up to $168.0 million. Further, the Company would have been eligible to receive tiered earnout payments ranging from high-single digits to mid-teen digits on net sales of sofpironium bromide gel (the “Earnout Payments”).
The Asset Purchase Agreement also provided that Botanix would pay to the Company a portion of the sales-based milestone payments and royalties that Botanix received from Kaken under the assigned Kaken Agreement (together, the “Sublicense Income”). Sublicense Income represented the Company’s estimate of payments that would be earned by the Company in the applicable period from sales-based milestone payments and royalties Botanix would receive from Kaken to the extent it was probable that a significant reversal in the amount of cumulative revenue recognized would not occur. Royalties vary based on net sales that are impacted by a wide variety of market and other factors and, as such, the Company utilized the expected value approach, which the Company believed would best predict the amount of consideration to which it would be entitled. In relation to the sales-based milestone payments that Botanix could receive from Kaken in the future, the Company utilized the most likely amount method and determined it was not yet probable that the Company would receive any payments from Botanix in relation to such milestone payments. Therefore, the Company determined that such milestone payments were fully constrained as of June 30, 2023, and, as such, had not yet been recognized such amounts as contract revenue. With respect to the recognition of contract revenue for the Sublicense Income based on future royalties that would be due to Botanix from Kaken, certain amounts were not yet due from Botanix. Therefore, the Company recorded a contract asset equal to the amount of revenue recognized related to the Sublicense Income, less the amount of payments received from or due by Botanix in relation to the Sublicense Income.
All other consideration due under the Asset Purchase Agreement was contingent upon certain regulatory approvals and future sales subsequent to such regulatory approvals, or was based upon future sales that the Company determined were not yet probable due to such revenues being highly susceptible to factors outside of the Company’s influence and uncertainty about the amount of such consideration that would not be resolved for an extended period of time. Therefore, the Company determined that such variable consideration amounts were fully constrained as of June 30, 2023, and as such, did not recognize such amounts as contract revenue.
Amendment No. 1 to the Asset Purchase Agreement
On July 21, 2023, the Company and Brickell Subsidiary entered into Amendment No. 1 to Asset Purchase Agreement (the “Asset Purchase Agreement Amendment”) with Botanix. The Asset Purchase Agreement Amendment provides that, in lieu of any remaining amounts potentially payable by Botanix to the Company pursuant to the Asset Purchase Agreement (collectively, the “Post-Closing Payment Obligations”), Botanix would pay $6.6 million to the Company and $1.65 million on behalf of the Company to Bodor, the payments for which were made on July 26, 2023. The Asset Purchase Agreement Amendment also provides that upon payment of the amounts by Botanix thereunder, all Post-Closing Payment Obligations under the Asset Purchase Agreement were terminated and of no further force or effect.
Transition Services Agreement with Botanix
In connection with the sale of the Assets, on the Effective Date, the Company and Botanix entered into a transition services agreement (the “TSA”) whereby the Company provides consulting services as an independent contractor to Botanix in support of and through filing and potential approval of the U.S. NDA for sofpironium bromide gel, 15%. In accordance with the terms of the TSA, in exchange for providing these services (i) prior to the acceptance of the filing by the FDA of such NDA in December 2022, the Company received from Botanix a fixed monthly amount of $71 thousand, and (ii) after the acceptance of the filing in December 2022, the Company receives from Botanix a variable amount based upon actual hours worked, in each case plus related fees and expenses of the Company’s advisors (plus a 5% administrative fee) and the Company’s out-of-pocket expenses. During the three and six months ended June 30, 2023, the Company recognized $0 and $9 thousand, respectively, of contract revenue associated with consulting services provided under the TSA. During the three and six months ended June 30, 2022, the Company recognized $0.4 million of contract revenue associated with consulting services provided under the TSA.
Contract Asset under the Botanix Agreements
The following table presents changes in the value of the Company’s contract asset related to Sublicense Income for the following periods (in thousands):
| | | | | |
| Contract asset as of January 1, 2023 | $ | 318 | |
| Sublicense Income recognized | 53 | |
| Amounts received or receivable | (65) | |
| Contract asset as of June 30, 2023 | $ | 306 | |
| Contract asset, included in prepaid expenses and other current assets | $ | 245 | |
| Contract asset, net of current portion | $ | 61 | |
| | | | | |
| Contract asset as of January 1, 2022 | $ | — | |
| Sublicense Income recognized | 333 | |
| |
| Contract asset as of June 30, 2022 | $ | 333 | |
| Contract asset, included in prepaid expenses and other current assets | $ | 152 | |
| Contract asset, net of current portion | $ | 181 | |
In July 2023, after entering into the Asset Purchase Agreement Amendment, the Company’s contract asset balances as of June 30, 2023 were fully settled.
Agreements with Bodor
In connection with the sale of the Assets, on the Effective Date, the Company, Brickell Subsidiary, and Bodor entered into an agreement (the “Rights Agreement”) to clarify that the Company and Brickell Subsidiary have the power and authority under the Amended and Restated License Agreement to enter into the Asset Purchase Agreement and the TSA, and that Botanix would assume the Amended and Restated License Agreement pursuant to the Asset Purchase Agreement. The Rights Agreement included a general release of claims and no admission of liability between the parties. Pursuant to such Rights Agreement, as subsequently amended on November 10, 2022, the Company agreed to pay Bodor (i) 20% of the amount of each payment due to the Company from Botanix for upfront and milestone payments, subject to deductions, credits, or offsets applied under the Asset Purchase Agreement, as well as (ii) certain tiered payments, set as a percentage ranging from mid-single digits to mid-teen digits, of the amount of each of the applicable Earnout Payments due to the Company from Botanix after deductions, credits or offsets applied under the Asset Purchase Agreement.
Pursuant to the terms of the Asset Purchase Agreement, the Company retained its obligation under the Amended and Restated License Agreement to issue $1.0 million in shares of its common stock to Bodor upon the FDA’s acceptance of an NDA filing for sofpironium bromide gel, 15%. On November 10, 2022, the Company entered into an Acknowledgment and Agreement Related to Asset Purchase Agreement and Amended and Restated License Agreement (the “Acknowledgment”) with Brickell Subsidiary, Botanix, and Bodor. Pursuant to the Acknowledgment, the Company paid $1.0 million in cash to Bodor in full satisfaction of the Company’s obligation to issue shares upon the FDA’s acceptance of the NDA.
In connection with the Asset Purchase Agreement Amendment, on July 21, 2023, the Company, Brickell Subsidiary, and Bodor entered into a Second Amendment to Rights Agreement (the “RA Amendment”). The RA Amendment provides that in exchange for the one-time payment of $1.65 million by Botanix on behalf of
the Company to Bodor, the Company shall have no further payment obligations to Bodor under or in connection with the Rights Agreement or the Amended and Restated License Agreement.
During the three and six months ended June 30, 2023, no expense was incurred or reported as general and administrative expenses in the condensed consolidated statements of operations associated with achieved milestones or payments due to Bodor related to sofpironium bromide gel, 15%. During the three and six months ended June 30, 2022, $0.5 million of general and administrative expense was associated with achieved milestones or payments due to Bodor related to sofpironium bromide gel, 15%. Prior to the execution of the Rights Agreement, the Company paid Bodor immaterial amounts with respect to the royalties the Company received from Kaken for sales of ECCLOCK in Japan during those periods.
NOTE 4. DETAILED ACCOUNT BALANCES
Prepaid expenses and other current assets consisted of the following (in thousands):
| | | | | | | | | | | |
| |
| | | |
| June 30,
2023 | | December 31,
2022 |
| Prepaid insurance | $ | 419 | | | $ | 521 | |
| Contract asset | 245 | | | 254 | |
| | | |
| Prepaid research and development expenses | 24 | | | 254 | |
| Accounts receivable | — | | | 250 | |
| | | |
| Other | 73 | | | 124 | |
| Total | $ | 761 | | | $ | 1,403 | |
Accrued liabilities consisted of the following (in thousands):
| | | | | | | | | | | |
| |
| | | |
| June 30,
2023 | | December 31,
2022 |
| Accrued compensation | $ | 920 | | | $ | 1,320 | |
| Accrued professional fees | 494 | | | 705 | |
| Accrued research and development expenses | 161 | | | 432 | |
| | | |
| | | |
| | | |
| | | |
| Total | $ | 1,575 | | | $ | 2,457 | |
NOTE 5. COMMITMENTS AND CONTINGENCIES
Operating Lease
In August 2016, the Company entered into a multi-year, noncancelable lease for its Colorado-based office space, which was amended on December 29, 2022 to, among other things, extend the lease term to December 31, 2025, eliminate options previously available to the Company to extend the lease, and provide that the Company may terminate the lease effective June 30, 2023 if notice is provided by April 30, 2023 (as amended, the “Boulder Lease”). Minimum base lease payments under the Boulder Lease are recognized on a straight-line basis over the term of the lease. In addition to base rental payments included in the contractual obligations table below, the Company is responsible for its pro rata share of the operating expenses for the building, which includes common area maintenance, utilities, property taxes, and insurance.
Upon modification of the Boulder Lease in December 2022, the Company reassessed classification of the lease and determined that the lease still met the criteria to be classified as an operating lease. Furthermore, the Company remeasured the lease liability as of the effective date by calculating the present value of the new lease payments, discounted at the Company’s incremental borrowing rate, over the lease term of six months. The
lease term includes periods covered by an option to terminate the lease that the Company was reasonably certain to exercise. The operating expenses are variable and are not included in the present value determination of the lease liability.
On May 4, 2023, the Company entered into an amendment to the Boulder Lease, which will terminate the Boulder Lease, effective August 31, 2023, and provided for the payment of a termination fee by the Company of approximately $5 thousand in May 2023.
Upon modification of the Boulder Lease in May 2023, the Company reassessed classification of the lease and determined that the lease still met the criteria to be classified as an operating lease. Furthermore, the Company remeasured the lease liability as of the effective date by calculating the present value of the new lease payments, discounted at the Company’s incremental borrowing rate, over the remaining lease term of four months. The operating expenses are variable and are not included in the present value determination of the lease liability.
The following is a summary of the contractual obligations related to operating lease commitments as of June 30, 2023 (in thousands):
| | | | | | | | |
| | |
| | |
| | |
| Total maturities, through December 31, 2023 | | $ | 19 | |
| | |
| Present value of lease liability | | $ | 19 | |
Licensing and Other Agreements
Refer to Note 3. “Strategic Agreements” for more information about the Company’s obligations under its licensing and other agreements.
NOTE 6. CAPITAL STOCK
Common Stock
Under the Company’s Restated Certificate of Incorporation, the Company’s Board has the authority to issue up to 300,000,000 shares of common stock with a par value of $0.01 per share. Each share of the Company’s common stock is entitled to one vote, and the holders of the Company’s common stock are entitled to receive dividends when and as declared or paid by its Board. The Company had reserved authorized shares of common stock for future issuance as of June 30, 2023 as follows:
| | | | | |
| June 30,
2023 |
| Common stock warrants | 621,063 | |
| Common stock options outstanding | 193,777 | |
| Unvested restricted stock units | 141,250 | |
| Shares available for grant under the Employee Stock Purchase Plan | 42,728 | |
| Shares available for grant under the 2020 Omnibus Long-Term Incentive Plan | 22,132 | |
| Total | 1,020,950 | |
The Company may be limited in its ability to sell a certain number of shares of its common stock under the Purchase Agreement or ATM Agreements described below, depending on the availability at any given time of authorized and available shares of common stock.
Public Offerings of Common Stock and Warrants
In October 2020, the Company completed a sale of 422,300 shares of its common stock, and, to certain investors, pre-funded warrants to purchase 40,663 shares of its common stock, and accompanying common stock warrants to purchase up to an aggregate of 462,979 shares of its common stock (the “October 2020 Offering”). Each share of common stock and pre-funded warrant to purchase one share of the Company’s common stock was sold together with a common warrant to purchase one share of the Company’s common stock. The shares of common stock and pre-funded warrants, and the accompanying common warrants, were issued separately and were immediately separable upon issuance. The common warrants are exercisable at a price of $32.40 per share of the Company’s common stock and will expire five years from the date of issuance. The pre-funded warrants were exercised in October 2020. No warrants associated with the October 2020 Offering were exercised during the three and six months ended June 30, 2023 or 2022.
In June 2020, the Company completed a sale of 328,669 shares of its common stock, and, to certain investors, pre-funded warrants to purchase 60,220 shares of its common stock, and accompanying common stock warrants to purchase up to an aggregate of 388,920 shares of its common stock (the “June 2020 Offering”). Each share of common stock and pre-funded warrant to purchase one share of common stock was sold together with a common warrant to purchase one share of common stock. The shares of common stock and pre-funded warrants, and the accompanying common warrants, were issued separately and were immediately separable upon issuance. The pre-funded warrants were exercised in the third quarter of 2020. The common warrants were immediately exercisable at a price of $56.25 per share of common stock and will expire five years from the date of issuance. No warrants associated with the June 2020 Offering were exercised during the three and six months ended June 30, 2023 or 2022.
At Market Issuance Sales Agreements
In March 2021, the Company entered into an At Market Issuance Sales Agreement (the “2021 ATM Agreement”) with Oppenheimer & Co. Inc. (“Oppenheimer”) and William Blair & Company, L.L.C. as the Company’s sales agents (the “Agents”). Pursuant to the terms of the 2021 ATM Agreement, the Company may sell from time to time through the Agents shares of its common stock having an aggregate offering price of up to $50.0 million. Such shares are issued pursuant to the Company’s shelf registration statement on Form S-3 (Registration No. 333-254037). Sales of the shares are made by means of ordinary brokers’ transactions on The Nasdaq Capital Market at market prices or as otherwise agreed by the Company and the Agents. Under the terms of the 2021 ATM Agreement, the Company may also sell the shares from time to time to an Agent as principal for its own account at a price to be agreed upon at the time of sale. Any sale of the shares to an Agent as principal would be pursuant to the terms of a separate placement notice between the Company and such Agent. During the six months ended June 30, 2023, the Company sold 2,887,535 shares of common stock under the 2021 ATM Agreement at a weighted-average price of $2.34 per share, for aggregate net proceeds of $6.6 million, after giving effect to a 3% commission to the Agents. During the three months ended June 30, 2023, no sales of common stock under the 2021 ATM Agreement occurred. During the three and six months ended June 30, 2022, the Company sold 31,557 shares of common stock under the 2021 ATM Agreement at a weighted-average price of $5.61 per share, for aggregate net proceeds of $0.2 million, after giving effect to a 3% commission to the Agents. As of June 30, 2023, approximately $38.0 million of shares of common stock were remaining, but had not yet been sold by the Company under the 2021 ATM Agreement.
In April 2020, the Company entered into an At Market Issuance Sales Agreement (the “2020 ATM Agreement” and, together with the 2021 ATM Agreement, the “ATM Agreements”) with Oppenheimer as the Company’s sales agent. Pursuant to the terms of the 2020 ATM Agreement, the Company may sell from time to time through Oppenheimer shares of its common stock having an aggregate offering price of up to $8.0 million. As of June 30, 2023, approximately $2.6 million of shares of common stock were remaining, but had not yet been sold by the Company under the 2020 ATM Agreement. However, the shares that may be sold pursuant to the 2020 ATM Agreement are not currently registered under the Securities Act of 1933, as amended. During the
three and six months ended June 30, 2023 and 2022, no sales of common stock under the 2020 ATM Agreement occurred.
The Company is subject to the SEC’s “baby shelf rules,” which prohibit companies with a public float of less than $75 million from issuing securities under a shelf registration statement in excess of one-third of such company’s public float in a 12-month period. These rules currently limit future issuances of shares by the Company under the ATM Agreements or other common stock offerings.
Private Placement Offerings
In February 2020, the Company and Lincoln Park Capital Fund, LLC (“Lincoln Park”) entered into (i) a securities purchase agreement (the “Securities Purchase Agreement”); (ii) a purchase agreement (the “Purchase Agreement”); and (iii) a registration rights agreement (the “Registration Rights Agreement”). Pursuant to the Securities Purchase Agreement, Lincoln Park purchased, and the Company sold, (i) an aggregate of 21,111 shares of common stock (the “Common Shares”); (ii) a warrant to initially purchase an aggregate of up to 13,476 shares of common stock at an exercise price of $0.45 per share (the “Series A Warrant”); and (iii) a warrant to initially purchase an aggregate of up to 34,588 shares of common stock at an exercise price of $52.20 per share (the “Series B Warrant” and, together with the Series A Warrant, the “Warrants”). No warrants associated with the Securities Purchase Agreement were exercised during the three and six months ended June 30, 2023 or 2022.
Under the terms and subject to the conditions of the Purchase Agreement, the Company has the right, but not the obligation, to sell to Lincoln Park, and Lincoln Park is obligated to purchase, up to $28.0 million in the aggregate of shares of common stock. In order to retain maximum flexibility to issue and sell up to the maximum of $28.0 million of the Company’s common stock under the Purchase Agreement, the Company sought and, at its annual meeting on April 19, 2021, received, stockholder approval for the sale and issuance of common stock in connection with the Purchase Agreement under Nasdaq Listing Rule 5635(d). Sales of common stock by the Company will be subject to certain limitations, and may occur from time to time, at the Company’s sole discretion, over the 36-month period commencing on August 14, 2020 (the “Commencement Date”).
Following the Commencement Date, under the Purchase Agreement, on any business day selected by the Company, the Company may direct Lincoln Park to purchase up to 2,222 shares of common stock on such business day (each, a “Regular Purchase”), provided, however, that (i) the Regular Purchase may be increased to up to 2,777 shares, provided that the closing sale price of the common stock is not below $3.00 on the purchase date; and (ii) the Regular Purchase may be increased to up to 3,333 shares, provided that the closing sale price of the common stock is not below $5.00 on the purchase date. In each case, Lincoln Park’s maximum commitment in any single Regular Purchase may not exceed $1,000,000. The purchase price per share for each such Regular Purchase will be based on prevailing market prices of common stock immediately preceding the time of sale. In addition to Regular Purchases, the Company may direct Lincoln Park to purchase other amounts as accelerated purchases or as additional accelerated purchases if the closing sale price of the common stock exceeds certain threshold prices as set forth in the Purchase Agreement. In all instances, the Company may not sell shares of its common stock to Lincoln Park under the Purchase Agreement if it would result in Lincoln Park beneficially owning more than 9.99% of the outstanding shares of common stock. During the three and six months ended June 30, 2023 and 2022, no sales of common stock under the Purchase Agreement occurred. As of June 30, 2023, approximately $26.9 million of shares of common stock were remaining, but had not yet been sold by the Company under the Purchase Agreement.
On September 9, 2022, a registration statement was declared effective covering the resale of up to 1,750,000 additional shares of the Company’s common stock that the Company has reserved for issuance and sale to Lincoln Park under the Purchase Agreement (Registration Statement No. 333-267254).
The Company has the right to terminate the Purchase Agreement at any time, at no cost or penalty.
The Securities Purchase Agreement, the Purchase Agreement, and the Registration Rights Agreement contain customary representations, warranties, agreements, and conditions to completing future sale transactions, indemnification rights, and obligations of the parties.
Preferred Stock
Under the Company’s Restated Certificate of Incorporation, the Company’s Board has the authority to issue up to 5,000,000 shares of preferred stock with a par value of $0.01 per share, at its discretion, in one or more classes or series and to fix the powers, preferences and rights, and the qualifications, limitations, or restrictions thereof, including dividend rights, conversion rights, voting rights, terms of redemption, and liquidation preferences, without further vote or action by the Company’s stockholders.
On May 25, 2022, the Company issued and sold one share of the Company’s preferred stock, which was designated as Series A Preferred Stock (the “Series A Preferred Stock”), for a nominal amount. During the time the Series A Preferred Stock was outstanding, it had 80,000,000 votes exclusively with respect to any proposal to amend the Company’s Restated Certificate of Incorporation to effect a reverse stock split of the Company’s common stock. The terms of the Series A Preferred Stock provided that it would be voted, without action by the holder, on any such proposal in the same proportion as shares of the Company’s common stock were voted. The Series A Preferred Stock otherwise had no voting rights except as otherwise required by the General Corporation Law of the State of Delaware. The Series A Preferred Stock was not convertible into, or exchangeable for, shares of any other class or series of stock or other securities of the Company and had no rights with respect to any distribution of assets of the Company, including upon a liquidation, bankruptcy, reorganization, merger, acquisition, sale, dissolution or winding up of the Company, whether voluntarily or involuntarily. The holder of the Series A Preferred Stock was not entitled to receive dividends of any kind. The Series A Preferred Stock was redeemed in whole on July 5, 2022 upon the effectiveness of the amendment to the Restated Certificate of Incorporation implementing the reverse stock split. As of June 30, 2023, there were no shares of preferred stock outstanding.
NOTE 7. STOCK-BASED COMPENSATION
Equity Incentive Plans
On April 20, 2020, the Company’s stockholders approved the 2020 Omnibus Long-Term Incentive Plan (the “Omnibus Plan”), which replaced, with respect to new award grants, the Company’s 2009 Equity Incentive Plan, as amended and restated (the “2009 Plan”), and the Vical Equity Incentive Plan (the “Vical Plan”) (collectively, the “Prior Plans”) that were previously in effect. Following the approval of the Omnibus Plan on April 20, 2020, no further awards were available to be issued under the Prior Plans but awards outstanding under those plans as of that date remain outstanding in accordance with their terms. As of June 30, 2023, 24,769 and 1,672 shares were subject to outstanding awards under the 2009 Plan and Vical Plan, respectively.
On May 17, 2022, the Company’s stockholders approved an increase in the number of shares of common stock authorized for issuance under the Omnibus Plan by 119,377 shares. As of June 30, 2023, 323,364 shares were authorized, and 335,027 shares were subject to outstanding awards under the Omnibus Plan. As of June 30, 2023, 22,132 shares remained available for grant under the Omnibus Plan.
Employee Stock Purchase Plan
On April 19, 2021, the Company’s stockholders approved the Fresh Tracks Therapeutics, Inc. Employee Stock Purchase Plan (the “ESPP”), which had a first eligible purchase period commencing on July 1, 2021. The ESPP allows qualified employees to purchase shares of the Company’s common stock at a price per share equal to
85% of the lower of: (i) the closing price of the Company’s common stock on the first trading day of the applicable purchase period or (ii) the closing price of the Company’s common stock on the last trading day of the applicable purchase period. New six-month purchase periods begin each January 1 and July 1. As of June 30, 2023, the Company had 42,728 shares available for issuance and 15,049 cumulative shares had been issued under the ESPP.
Stock-Based Compensation Expense
Total stock-based compensation expense reported in the condensed consolidated statements of operations was allocated as follows (in thousands):
| | | | | | | | | | | | | | | | | | | | | | | |
| Three Months Ended
June 30, | | Six Months Ended
June 30, |
| 2023 | | 2022 | | 2023 | | 2022 |
| Research and development | $ | 103 | | | $ | 113 | | | $ | 195 | | | $ | 216 | |
| General and administrative | 307 | | | 463 | | | 594 | | | 911 | |
| Total stock-based compensation expense | $ | 410 | | | $ | 576 | | | $ | 789 | | | $ | 1,127 | |
ITEM 2. MANAGEMENT’S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS
Overview
We are a clinical-stage pharmaceutical company striving to transform patient lives through the development of innovative and differentiated prescription therapeutics. Our pipeline aims to disrupt existing treatment paradigms and features several new chemical entities that inhibit novel targets with first-in-class potential for autoimmune, inflammatory, and other debilitating diseases. Our executive management team and board of directors (our “Board”) have a proven track record of leadership across early-stage research, product development, and global commercialization. Our strategy has been to align this experience and clear vision to explore beyond the limitations of current therapies by identifying, pursuing, and developing next-generation therapeutics that can be groundbreaking in their ability to help millions of people struggling with autoimmune, inflammatory, and other debilitating diseases.
Exploration of Strategic Options
Our Board and executive management team are conducting a comprehensive process to explore and evaluate strategic alternatives with the goal of maximizing shareholder value. Potential alternatives under evaluation include, but are not limited to, a financing, a merger or reverse merger, the sale of all or part of the Company, licensing of assets, a business combination, and/or other strategic transaction or series of related transactions involving our Company. MTS Health Partners, LP has been retained as our exclusive financial advisor to assist in this review process.
In the second quarter of 2023, we paused substantially all of our research and development activities, including not proceeding with the previously planned Phase 1 Part 2 clinical trial for FRTX-02, in order to conserve capital resources during our ongoing evaluation of potential strategic options, to determine the appropriate next development steps based on the results of our first-in-human Phase 1 Part 1 clinical trial. The outcome of our strategic options initiative will dictate the Company’s future path including to what extent the current pipeline continues to be developed.
Research and Development Programs
The following image summarizes our current pipeline, corresponding development programs, and potential therapeutic areas for each program:
FRTX-02: A Potential First-in-Class Oral DYRK1A Inhibitor for the Treatment of Autoimmune and Inflammatory Diseases
FRTX-02 is a novel, potent, highly selective, and orally bioavailable potential first-in-class, small molecule DYRK1A inhibitor that aims to restore immune balance in patients whose immune systems have become dysregulated. We believe FRTX-02 has the potential to be a first-in-class therapy for the treatment of a wide array of debilitating autoimmune and inflammatory diseases.
FRTX-02 is our lead development-stage program and has demonstrated promising results in various preclinical models, including of atopic dermatitis (“AD”) and rheumatoid arthritis. In these models, FRTX-02 showed encouraging decreases in disease severity and reduction of pro-inflammatory cytokines compared to current standard-of-care agents, such as Janus kinase (JAK) inhibitors and anti-tumor necrosis factor (“TNF”) biologics. Notably, many current therapies for autoimmune disorders are broadly immunosuppressive, which may lead to severe side effects, such as increased infection risk. Preclinical data have shown FRTX-02 to drive regulatory T-cell differentiation while dampening pro-inflammatory TH17 cells and MyD88/IRAK4-related signaling pathways. Regulatory T-cells serve to maintain tolerance and keep the autoreactive, pro-inflammatory T-cells in check, thus inhibiting autoimmune disease and limiting chronic inflammation. The myeloid differentiation primary response 88 (“MyD88”) protein is normally spliced into a long form and a short form. The long form of MyD88 drives inflammation via pathways related to IRAK4, a protein kinase involved in signaling immune responses from toll-like receptors, while the short form of MyD88 limits IRAK4 phosphorylation and its respective downstream signaling pathway. DYRK1A inhibition shifts the balance to produce more MyD88 short form, which leads to deactivation of the downstream release of certain pro-inflammatory cytokines. Based on current understanding, this inhibition of the release of excess cytokines can be achieved by re-establishing the role of MyD88 short form as a negative regulator of this pathway. Unlike many existing therapies, as well as those currently being investigated, FRTX-02 may have the ability to target both the adaptive and innate immune imbalance simultaneously, potentially resulting in, or substantially achieving, restoration of immune homeostasis that, if proven, would represent a paradigm shift in the treatment of certain autoimmune and inflammatory diseases.
In May 2022, we initiated a first-in-human Phase 1 clinical trial for FRTX-02 (FRTX-02-101) in Canada, which marks the first time an oral DYRK1A inhibitor intended for patients with autoimmune diseases has been administered in humans. FRTX-02-101 was a randomized, double-blind, placebo-controlled study designed to evaluate the safety, tolerability, pharmacokinetics, and pharmacodynamics of FRTX-02 capsules in both healthy subjects and patients with AD. Parts 1A and 1B of the Phase 1 clinical trial were completed in the fourth quarter of 2022, and in March 2023, we reported positive topline results described in greater detail below. Part 2 of the study was originally designed to compare once-daily oral doses of FRTX-02 to placebo in subjects with moderate-to-severe AD and include an exploratory evaluation of efficacy.
In the second quarter of 2023, we paused substantially all of our research and development activities, including not proceeding with the previously planned Phase 1 Part 2 clinical trial for FRTX-02, in order to conserve capital resources during our ongoing evaluation of potential strategic options, to determine the appropriate next development steps based on the results of our first-in-human Phase 1 Part 1 clinical trial. The outcome of our strategic options initiative will dictate the Company’s future path including to what extent the current pipeline continues to be developed.
FRTX-02 is covered by a composition of matter patent issued in the U.S., Japan, China, and other key countries through at least 2038, subject to patent term extensions and adjustments that may be available depending on how this early-stage asset is developed, as well as a pending Patent Cooperation Treaty (“PCT”) application, and other foreign and U.S. applications for FRTX-02, as of the date of this Quarterly Report.
FRTX-10: A covalent Stimulator of Interferon Genes (STING) inhibitor for the Potential Treatment of Autoimmune, Inflammatory, and Rare Genetic Diseases
In February 2022, we acquired exclusive, worldwide rights to research, develop, and commercialize a portfolio of novel, preclinical-stage oral Stimulator of Interferon Genes (“STING”) inhibitors. STING is a well-known mediator of innate immune responses. Excessive signaling through STING is linked to numerous high unmet-need diseases, ranging from autoimmune disorders, such as systemic lupus erythematosus, to interferonopathies, which are a set of rare genetic conditions characterized by interferon overproduction and could have orphan drug potential.
STING is a key component of the cyclic GMP-AMP synthase (“cGAS”)-STING pathway, which plays an important role in the activation of innate immunity. cGAS acts as a DNA sensor, detecting DNA from sources such as invading bacteria, viruses, and cellular debris that can arise from aging and tissue damage. Upon DNA binding, cGAS produces the secondary messenger molecule cyclic GMP-AMP (“cGAMP”), which binds to STING. STING then undergoes the post-translational modification called palmitoylation, a step essential to the activation of STING. Activated STING then in turn activates the recruitment of kinases that phosphorylate IRF3 and IκBα. Phosphorylated IRF3 leads to activation of the type I interferon response, while phosphorylated IκBα activates NFκB and increases the secretion of pro-inflammatory cytokines such as IL-6 and TNFα, resulting in inflammation. While the innate immune response is an important defense mechanism, a dysregulated type I interferon response and overproduction of pro-inflammatory cytokines also represents a driving cause of multiple autoimmune and inflammatory diseases. As such, targeting the cGAS-STING pathway through STING inhibition may be a novel approach to treating these diseases.
FRTX-10, our lead early-stage STING inhibitor candidate, is a novel, potent, and orally bioavailable covalent STING inhibitor that specifically targets the palmitoylation site of STING. This allows it to inhibit both wild-type STING and gain-of-function mutants without competing with cGAMP binding, thus deactivating downstream signaling through IRF3 and IκBα and ultimately suppressing inflammation. FRTX-10 has exhibited strong proof-of mechanism and a promising profile in initial pharmacokinetics, toxicology, and safety pharmacology studies. In addition, in vitro studies show that FRTX-10 more potently blocks the STING pathway compared to other known STING palmitoylation inhibitors, and that mice treated with FRTX-10 in
vivo demonstrate significant decreases in production of key pro-inflammatory cytokines following stimulation of STING.
For FRTX-10, as of the date of this Quarterly Report, we currently have two pending PCT applications and pending applications in the U.S., Japan, Europe, and other key countries. We possess an exclusive license directed to a library of compounds targeting/inhibiting STING, pharmaceutical compositions containing the same, and methods of their use, which are being evaluated.
Next-Generation Kinase Inhibitors: A Cutting-Edge Platform with the Potential to Produce Treatments for Autoimmune, Inflammatory, and Other Debilitating Diseases
In August 2021, we acquired exclusive global rights to a cutting-edge platform of next-generation kinase inhibitors. This library of new chemical entities includes next-generation DYRK1 inhibitors, as well as other molecules that specifically inhibit Leucine-Rich Repeat Kinase 2 (“LRRK2”), CDC2-like kinase (“CLK”), and TTK protein kinase (“TTK”), also known as Monopolar spindle 1 (Mps1) kinases. A number of these drug candidates have the potential to penetrate the blood-brain barrier, presenting an opportunity to address neuroinflammatory conditions of high unmet need, such as Down Syndrome, Alzheimer’s Disease, and Parkinson’s Disease, while other peripherally acting novel LRRK2, TTK, and CLK kinase inhibitors could be developed in additional therapeutic areas within autoimmunity, inflammation, and oncology.
Compounds from the next-generation kinase inhibitor platform are covered by U.S. and foreign composition of matter patent applications, as well as other applications, that are currently pending in global prosecution.
Topline Results of FRTX-02 Phase 1 Clinical Trial
Study Design
The Phase 1 clinical trial of FRTX-02 is a two-part, randomized, double-blinded, placebo-controlled study designed to evaluate the safety, tolerability, pharmacokinetics, and pharmacodynamics of FRTX-02 capsules in both healthy subjects and patients with AD. Part 1A of the study was a single ascending dose (“SAD”) assessment, which enrolled a total of 56 healthy subjects across seven cohorts (single oral dose of 10 to 600 mg FRTX-02 or placebo). Part 1B of the study was a multiple ascending dose (“MAD”) assessment, which enrolled a total of 33 healthy subjects across three cohorts (75, 150, and 300 mg FRTX-02 or placebo, once-daily for 14 days). Part 2 of the study was previously designed to compare once-daily oral doses of FRTX-02 to placebo in subjects with moderate-to-severe AD and include an exploratory evaluation of efficacy. In the second quarter of 2023, we paused substantially all of our research and development activities, including the previously planned Phase 1 Part 2 clinical trial for FRTX-02, in order to conserve capital resources during our ongoing evaluation of potential strategic options to determine the appropriate next development steps for the program.
Safety
FRTX-02 was generally safe and well-tolerated in all seven SAD cohorts and in the 75 mg and 150 mg MAD cohorts, with no discontinuations due to Treatment-Emergent Adverse Events (“TEAEs”). No drug-related serious adverse events were reported. All but two TEAEs were classified as mild, with a single count of moderate back pain in the SAD cohort (assessed as unlikely related to treatment) and moderate headache in the MAD cohort (assessed as possibly related to treatment). No dose-dependent trend in the frequency or severity of TEAEs was observed. There were no electrocardiogram or lab findings of clinical relevance in any of the SAD cohorts and in the 75 mg and 150 mg MAD cohorts. In the 300 mg MAD cohort, QTc prolongation was observed in two subjects at Days 8 and 9, respectively. Both subjects were asymptomatic, and their QTc intervals returned to baseline levels and remained in the normal range after cessation of dosing. All subjects completed their scheduled study assessments.
Pharmacokinetics (“PK”)
A dose-proportional increase in exposure was observed through all SAD and MAD cohorts. PK data from the 75 mg and 150 mg MAD cohorts achieved maximum plasma concentrations (Cmax) and area under the concentration-time curve (AUC) values at or above the pharmacologically active exposure levels observed across multiple nonclinical autoimmune and inflammatory disease models. The PK data support once-daily oral dosing with FRTX-02. The time of maximum plasma FRTX-02 concentration (Tmax) occurred between 2.65 to 3.25 hours post-dose, and a plasma half-life of approximately 16.0 to 28.0 hours was observed at Day 14 in the 75 mg and 150 mg MAD cohorts, respectively. A minimal-to-moderate accumulation following once-daily oral administration of 75 mg and 150 mg FRTX-02 over 14 days was observed, and steady state plasma concentrations were attained before Day 14.
Pharmacodynamics (“PD”)
As part of an exploratory PD assessment, ex vivo lipopolysaccharide (LPS)-stimulated cytokine assays were conducted. FRTX-02 demonstrated a reduction in disease-relevant proinflammatory cytokines in whole blood, suggesting initial support for the FRTX-02 mechanism of action. Mean percent cytokine reduction from baseline after 14 days of once-daily 75 mg or 150 mg FRTX-02 treatment versus placebo were in the range of approximately 66% to 20% for IFNγ, IL-23, IL-10, IL-6, and TNFα. Additionally, maximum individual subject cytokine reductions from baseline were shown to be >90% for IFNγ, >50% for IL-23, IL-10, and TNFα, and approximately 40% for IL-6.
Strategic, Licensing, and Other Arrangements
License and Development Agreement with Voronoi
In August 2021, we entered into a License and Development Agreement (the “Voronoi License Agreement”) with Voronoi Inc. (“Voronoi”), pursuant to which we acquired exclusive, worldwide rights to research, develop, and commercialize FRTX-02 and other next-generation kinase inhibitors.
With respect to FRTX-02, the Voronoi License Agreement provides that we will make payments to Voronoi of up to $211.0 million in the aggregate contingent upon achievement of specified development, regulatory, and commercial milestones. With respect to the compounds arising from the next-generation kinase inhibitor platform, we will make payments to Voronoi of up to $107.5 million in the aggregate contingent upon achievement of specified development, regulatory, and commercial milestones. Further, the Voronoi License Agreement provides that we will pay Voronoi tiered royalty payments ranging from low-single digits up to 10% of net sales of products arising from the DYRK1A inhibitor programs and next-generation kinase inhibitor platform. All of the contingent payments and royalties are payable in cash in U.S. Dollars, except for $1.0 million of the development and regulatory milestone payments, which amount is payable in equivalent shares of our common stock. Under the terms of the Voronoi License Agreement, we are responsible for, and bear the future costs of, all development and commercialization activities, including patenting, related to all the licensed compounds. As of June 30, 2023 and through the date of this Quarterly Report, we have not yet made any payments or recorded any liabilities related to the specified development, regulatory, and commercial milestones or royalties on net sales pursuant to the Voronoi License Agreement.
Exclusive License and Development Agreement with Carna
In February 2022, we entered into an Exclusive License Agreement (the “Carna License Agreement”) with Carna Biosciences, Inc. (“Carna”), pursuant to which we acquired exclusive, worldwide rights to research, develop, and commercialize Carna’s portfolio of novel STING inhibitors. In accordance with the terms of the Carna License Agreement, in exchange for the licensed rights, we made a one-time cash payment of $2.0
million, which was recorded as research and development expenses in the condensed consolidated statements of operations during the six months ended June 30, 2022.
The Carna License Agreement provides that we will make success-based payments to Carna of up to $258.0 million in the aggregate contingent upon achievement of specified development, regulatory, and commercial milestones. Further, the Carna License Agreement provides that we will pay Carna tiered royalty payments ranging from mid-single digits up to 10% of net sales. All of the contingent payments and royalties are payable in cash in U.S. Dollars. Under the terms of the Carna License Agreement, we are responsible for, and bear the future costs of, all development and commercialization activities, including patenting, related to all the licensed compounds. As of June 30, 2023 and through the date of this Quarterly Report, we have not yet made any payments or recorded any liabilities related to the specified development, regulatory, and commercial milestones or royalties on net sales pursuant to the Carna License Agreement.
Agreements with Botanix
Asset Purchase Agreement with Botanix
On May 3, 2022 (the “Effective Date”), we and Brickell Subsidiary entered into an asset purchase agreement with Botanix SB, Inc. and Botanix Pharmaceuticals Limited (“Botanix”) (the “Asset Purchase Agreement”), pursuant to which Botanix acquired and assumed control of all rights, title, and interests to assets primarily related to the proprietary compound sofpironium bromide that were owned and/or licensed by us or Brickell Subsidiary (the “Assets”). Prior to the sale of the Assets, we had previously entered into a License Agreement with Bodor Laboratories, Inc. (“Bodor”), dated December 15, 2012 (last amended in February 2020) that provided us with a worldwide exclusive license to develop, manufacture, market, sell, and sublicense products containing sofpironium bromide through which the Assets were developed (the “Amended and Restated License Agreement”). As a result of the Asset Purchase Agreement, Botanix is now responsible for all further research, development, and commercialization of sofpironium bromide globally and replaced us as the exclusive licensee under the Amended and Restated License Agreement.
In accordance with the sublicense rights provided to us under the Amended and Restated License Agreement, we also had previously entered into a License, Development, and Commercialization Agreement with Kaken Pharmaceutical Co., Ltd. (“Kaken”), dated as of March 31, 2015 (as amended in May 2018, the “Kaken Agreement”), under which we granted to Kaken an exclusive right to develop, manufacture, and commercialize the sofpironium bromide compound in Japan and certain other Asian countries (the “Territory”). In exchange for the sublicense, we were entitled to receive aggregate payments of up to $10.0 million upon the achievement of specified development milestones, which were earned and received in 2017 and 2018, and up to $19.0 million upon the achievement of sales-based milestones, as well as tiered royalties based on a percentage of net sales of licensed products in the Territory. In September 2020, Kaken received regulatory approval in Japan to manufacture and market sofpironium bromide gel, 5% (“ECCLOCK”) for the treatment of primary axillary hyperhidrosis, and as a result, we began recognizing royalty revenue earned on a percentage of net sales of ECCLOCK in Japan. Pursuant to the Asset Purchase Agreement, the Kaken Agreement was assigned to Botanix, which replaced us as the exclusive sub-licensor to Kaken. During the three and six months ended June 30, 2022, prior to entering into the Asset Purchase Agreement, we recognized royalty revenue under the Kaken Agreement of $0 and $0.1 million, respectively.
We determined that the development of and ultimate sale and assignment of rights to the Assets is an output of our ordinary activities and Botanix is a customer as it relates to the sale of the Assets and related activities.
In accordance with the terms of the Asset Purchase Agreement, in exchange for the Assets, we (i) received an upfront payment at closing in the amount of $3.0 million, (ii) were reimbursed for certain recent development expenditures in advancement of the Assets, (iii) received a milestone payment of $2.0 million upon the acceptance by the U.S. Food and Drug Administration (“FDA”) in December 2022 of the filing of a new drug
application (“NDA”) for sofpironium bromide gel, 15%, and (iv) would have been eligible to receive a contingent milestone payment of $4.0 million if marketing approval in the U.S. for sofpironium bromide gel, 15%, is received on or before September 30, 2023, or $2.5 million if such marketing approval is received after September 30, 2023 but on or before February 17, 2024. Botanix submitted an NDA for sofpironium bromide gel, 15%, to the FDA in September 2022, which was accepted by the FDA in December 2022. Under the Asset Purchase Agreement, we also would have been eligible to receive additional success-based regulatory and sales milestone payments of up to $168.0 million. Further, we would have been eligible to receive tiered earnout payments ranging from high-single digits to mid-teen digits on net sales of sofpironium bromide gel (the “Earnout Payments”).
The Asset Purchase Agreement also provided that Botanix would pay to us a portion of the sales-based milestone payments and royalties that Botanix received from Kaken under the assigned Kaken Agreement (together, the “Sublicense Income”). Sublicense Income represented our estimate of payments that would be earned by us in the applicable period from sales-based milestone payments and royalties Botanix would receive from Kaken to the extent it was probable that a significant reversal in the amount of cumulative revenue recognized would not occur. Royalties vary based on net sales that are impacted by a wide variety of market and other factors. We recorded a contract asset equal to the amount of revenue recognized related to the Sublicense Income, less the amount of payments received from or due by Botanix in relation to the Sublicense Income.
All other consideration due under the Asset Purchase Agreement was contingent upon certain regulatory approvals and future sales subsequent to such regulatory approvals, or was based upon future sales that we determined were not yet probable due to such revenues being highly susceptible to factors outside of our influence and uncertainty about the amount of such consideration that would not be resolved for an extended period of time. Therefore, we determined that such variable consideration amounts were fully constrained as of June 30, 2023, and, as such, did not recognize such amounts as contract revenue.
Amendment No. 1 to the Asset Purchase Agreement
On July 21, 2023, we and Brickell Subsidiary entered into Amendment No. 1 to Asset Purchase Agreement (the “Asset Purchase Agreement Amendment”) with Botanix. The Asset Purchase Agreement Amendment provides that, in lieu of any remaining amounts potentially payable by Botanix to us pursuant to the Asset Purchase Agreement (collectively, the “Post-Closing Payment Obligations”), Botanix would pay $6.6 million to us and $1.65 million on behalf of us to Bodor, the payments for which were made on July 26, 2023. The Asset Purchase Agreement Amendment also provides that upon payment of the amounts by Botanix thereunder, all Post-Closing Payment Obligations under the Asset Purchase Agreement were terminated and of no further force or effect. Except as explicitly amended by the Asset Purchase Agreement Amendment, the Asset Purchase Agreement remains in full force and effect.
Transition Services Agreement with Botanix
In connection with the sale of the Assets, on the Effective Date, we and Botanix entered into a transition services agreement (the “TSA”) whereby we provide consulting services as an independent contractor to Botanix in support of and through filing and potential approval of the U.S. NDA for sofpironium bromide gel, 15%. In accordance with the terms of the TSA, in exchange for providing these services (i) prior to the acceptance of the filing by the FDA of such NDA in December 2022, we are receiving from Botanix a fixed monthly amount of $71 thousand, and (ii) after the acceptance of the filing in December 2022, we receive from Botanix a variable amount based upon actual hours worked, in each case plus related fees and expenses of our advisors (plus a 5% administrative fee) and our out-of-pocket expenses. During the three and six months ended June 30, 2023, we recognized $0 and $9 thousand, respectively, of contract revenue associated with consulting services provided under the TSA. During the three and six months ended June 30, 2022, we recognized $0.4 million of contract revenue associated with consulting services provided under the TSA.
Agreements with Bodor
In connection with the sale of the Assets, on the Effective Date, we, Brickell Subsidiary, and Bodor entered into an agreement (the “Rights Agreement”) to clarify that we and Brickell Subsidiary have the power and authority under the Amended and Restated License Agreement to enter into the Asset Purchase Agreement and the TSA, and that Botanix would assume the Amended and Restated License Agreement pursuant to the Asset Purchase Agreement. The Rights Agreement included a general release of claims and no admission of liability between the parties. Pursuant to such Rights Agreement, as subsequently amended on November 10, 2022, we have agreed to pay Bodor (i) 20% of the amount of each payment due to us from Botanix for upfront and milestone payments, subject to deductions, credits, or offsets applied under the Asset Purchase Agreement, as well as (ii) certain tiered payments, set as a percentage ranging from mid-single digits to mid-teen digits, of the amount of each of the applicable Earnout Payments due to us from Botanix after deductions, credits, or offsets applied under the Asset Purchase Agreement.
Pursuant to the terms of the Asset Purchase Agreement, we retained our obligation under the Amended and Restated License Agreement to issue $1.0 million in shares of our common stock to Bodor upon the FDA’s acceptance of an NDA filing for sofpironium bromide gel, 15%. On November 10, 2022, we entered into an Acknowledgment and Agreement Related to Asset Purchase Agreement and Amended and Restated License Agreement (the “Acknowledgment”) with Brickell Subsidiary, Botanix, and Bodor. Pursuant to the Acknowledgment, we paid $1.0 million in cash to Bodor in full satisfaction of our obligation to issue shares upon the FDA’s acceptance of the NDA. We determined to prepay this obligation in cash in order to avoid the substantial dilution to our stockholders that would have resulted if we had issued the shares of our common stock originally provided for in the Amended and Restated License Agreement.
In connection with the Asset Purchase Agreement Amendment, on July 21, 2023, we, Brickell Subsidiary, and Bodor entered into a Second Amendment to Rights Agreement (the “RA Amendment”). The RA Amendment provides that in exchange for the one-time payment of $1.65 million by Botanix on behalf of us to Bodor, we shall have no further payment obligations to Bodor under or in connection with the Rights Agreement or the Amended and Restated License Agreement. Except as explicitly amended by the RA Amendment, the Rights Agreement remains in full force and effect.
During the three and six months ended June 30, 2023, no expense was incurred or reported as general and administrative expenses in the condensed consolidated statements of operations associated with achieved milestones related to sofpironium bromide gel, 15%. During the three and six months ended June 30, 2022, $0.5 million of general and administrative expense was associated with achieved milestones or payments due to Bodor related to sofpironium bromide gel, 15%. Prior to the execution of the Rights Agreement, we paid Bodor immaterial amounts with respect to the royalties we received from Kaken for sales of ECCLOCK in Japan during those periods.
Reverse Stock Split
On June 30, 2022, our stockholders approved a reverse stock split of our outstanding common stock, which was effected at a split ratio of 1-for-45 on July 5, 2022, at which date each forty-five (45) shares of common stock issued and outstanding immediately prior to the reverse stock split were automatically reclassified, combined and converted into one (1) validly issued, fully paid, and non-assessable share of our common stock, subject to the treatment of fractional share interests.
All common stock share and per-share amounts, and other related balances and computations, reported as of and for all periods presented in this Management’s Discussion and Analysis of Financial Condition and Results of Operations give effect to the 1-for-45 reverse stock split of our outstanding shares of common stock that occurred on July 5, 2022.
Significant Financing Arrangements
This section sets forth our recent and ongoing financing arrangements, all of which involve our common stock.
Public Offerings of Common Stock and Warrants
In October 2020, we completed the sale of 422,300 shares of our common stock, and, to certain investors, pre-funded warrants to purchase 40,663 shares of our common stock, and accompanying common stock warrants to purchase up to an aggregate of 462,979 shares of our common stock (the “October 2020 Offering”). The October 2020 Offering resulted in net proceeds of approximately $13.7 million to us after deducting underwriting commissions and discounts and other offering expenses payable by us of $1.3 million and excluding the proceeds from the exercise of the warrants. No warrants associated with the October 2020 Offering were exercised during the three and six months ended June 30, 2023 or 2022.
In June 2020, we completed the sale of 328,669 shares of our common stock, and, to certain investors, pre-funded warrants to purchase 60,220 shares of our common stock, and accompanying common stock warrants to purchase up to an aggregate of 388,920 shares of our common stock (the “June 2020 Offering”). The June 2020 Offering resulted in approximately $18.7 million of net proceeds after deducting underwriting commissions and discounts and other offering expenses payable by us of $1.4 million and excluding the proceeds from the exercise of the warrants. No warrants associated with the June 2020 Offering were exercised during the three and six months ended June 30, 2023 or 2022.
For additional information regarding the offerings described above, see Note 6. “Capital Stock” of the notes to our condensed consolidated financial statements included in this Quarterly Report.
At Market Issuance Sales Agreements
In March 2021, we entered into an At Market Issuance Sales Agreement (the “2021 ATM Agreement”) with Oppenheimer & Co. Inc. (“Oppenheimer”) and William Blair & Company, L.L.C. as our sales agents (the “Agents”). Pursuant to the terms of the 2021 ATM Agreement, we may sell from time to time through the Agents shares of our common stock having an aggregate offering price of up to $50.0 million. Such shares are issued pursuant to our shelf registration statement on Form S-3 (Registration No. 333-254037). Sales of shares are made by means of ordinary brokers’ transactions on The Nasdaq Capital Market at market prices or as otherwise agreed by us and the Agents. Under the terms of the 2021 ATM Agreement, we may also sell the shares from time to time to an Agent as principal for its own account at a price to be agreed upon at the time of sale. Any sale of the shares to an Agent as principal would be pursuant to the terms of a separate placement notice between us and such Agent. During the six months ended June 30, 2023, we sold 2,887,535 shares of common stock under the 2021 ATM Agreement at a weighted-average price of $2.34 per share, for aggregate net proceeds of $6.6 million, after giving effect to a 3% commission to the Agents. During the three months ended June 30, 2023, no sales of common stock under the 2021 ATM Agreement occurred. During the three and six months ended June 30, 2022, we sold 31,557 shares of common stock under the 2021 ATM Agreement at a weighted-average price of $5.61 per share, for aggregate net proceeds of $0.2 million, after giving effect to a 3% commission to the Agents. As of June 30, 2023, approximately $38.0 million of shares of common stock were remaining, but had not yet been sold under the 2021 ATM Agreement.
In April 2020, we entered into an At Market Issuance Sales Agreement (the “2020 ATM Agreement” and, together with the 2021 ATM Agreement, the “ATM Agreements”) with Oppenheimer as our sales agent. Pursuant to the terms of the 2020 ATM Agreement, we may sell from time to time through Oppenheimer shares of our common stock having an aggregate offering price of up to $8.0 million. As of June 30, 2023, approximately $2.6 million of shares of common stock were remaining, but had not yet been sold under the 2020 ATM Agreement, however, the shares that may be sold pursuant to the 2020 ATM Agreement are not
currently registered under the Securities Act of 1933, as amended. During the three and six months ended June 30, 2023 and 2022, no sales of common stock under the 2020 ATM Agreement occurred.
We are subject to the SEC’s “baby shelf rules,” which prohibit companies with a public float of less than $75 million from issuing securities under a shelf registration statement in excess of one-third of such company’s public float in a 12-month period. These rules currently limit future issuances of shares by us under the ATM Agreements or other common stock offerings.
Private Placement Offerings
In February 2020, we and Lincoln Park Capital Fund, LLC (“Lincoln Park”) entered into (i) a securities purchase agreement (the “Securities Purchase Agreement”); (ii) a purchase agreement (the “Purchase Agreement”); and (iii) a registration rights agreement (the “Registration Rights Agreement”). Pursuant to the Securities Purchase Agreement, Lincoln Park purchased, and we sold, (i) an aggregate of 21,111 shares of common stock (the “Common Shares”); (ii) a warrant to initially purchase an aggregate of up to 13,476 shares of common stock at an exercise price of $0.45 per share (the “Series A Warrant”); and (iii) a warrant to initially purchase an aggregate of up to 34,588 shares of common stock at an exercise price of $52.20 per share (the “Series B Warrant” and, together with the Series A Warrant, the “Warrants”). The aggregate gross purchase price for the Common Shares and the Warrants was $2.0 million. No warrants associated with the Securities Purchase Agreement were exercised during the three and six months ended June 30, 2023 or 2022.
Under the terms and subject to the conditions of the Purchase Agreement, we have the right, but not the obligation, to sell to Lincoln Park, and Lincoln Park is obligated to purchase, up to $28.0 million in the aggregate of shares of our common stock. In order to retain maximum flexibility to issue and sell up to the maximum of $28.0 million of our common stock under the Purchase Agreement, we sought and, at our annual meeting on April 19, 2021, received, stockholder approval for the sale and issuance of common stock in connection with the Purchase Agreement under Nasdaq Listing Rule 5635(d). Sales of common stock by us will be subject to certain limitations, and may occur from time to time, at our sole discretion, over the 36-month period commencing on August 14, 2020 (the “Commencement Date”).
Following the Commencement Date, under the Purchase Agreement, on any business day selected by us, we may direct Lincoln Park to purchase up to 2,222 shares of our common stock on such business day (each, a “Regular Purchase”), provided, however, that (i) the Regular Purchase may be increased to up to 2,777 shares, provided that the closing sale price of the common stock is not below $3.00 on the purchase date; and (ii) the Regular Purchase may be increased to up to 3,333 shares, provided that the closing sale price of the common stock is not below $5.00 on the purchase date. In each case, Lincoln Park’s maximum commitment in any single Regular Purchase may not exceed $1,000,000. The purchase price per share for each such Regular Purchase will be based on prevailing market prices of common stock immediately preceding the time of sale. In addition to Regular Purchases, we may direct Lincoln Park to purchase other amounts as accelerated purchases or as additional accelerated purchases if the closing sale price of the common stock exceeds certain threshold prices as set forth in the Purchase Agreement. In all instances, we may not sell shares of our common stock to Lincoln Park under the Purchase Agreement if it would result in Lincoln Park beneficially owning more than 9.99% of the outstanding shares of our common stock. During the three and six months ended June 30, 2023 and 2022, no sales of common stock under the Purchase Agreement occurred. As of June 30, 2023, approximately $26.9 million of shares of common stock were remaining, but had not yet been sold under the Purchase Agreement.
On September 9, 2022, a registration statement was declared effective covering the resale of up to 1,750,000 additional shares of our common stock that we have reserved for issuance and sale to Lincoln Park under the Purchase Agreement (Registration Statement No. 333-267254).
We have the right to terminate the Purchase Agreement at any time, at no cost or penalty.
Financial Overview
Our operations to date have been limited to business planning, raising capital, developing and entering into strategic partnerships for our pipeline assets, identifying and in-licensing product candidates, conducting clinical trials, and other research and development activities.
To date, we have financed operations primarily through funds received from the sale of common stock and warrants, convertible preferred stock, debt and convertible notes, and payments received under license, collaboration, and other agreements. Other than through arrangements as they relate to sales of ECCLOCK in Japan, none of our product candidates has been approved for sale and we have not generated any product sales. Since inception, we have incurred operating losses. We recorded a net loss of $6.6 million and $10.6 million for the six months ended June 30, 2023 and 2022, respectively. As of June 30, 2023, we had an accumulated deficit of $173.0 million. We expect to continue incurring expenses and operating losses for the foreseeable future. We would not be able to generate significant product revenue unless and until we successfully complete development of, obtain marketing approval for, and commercialize product candidates, either alone or in collaboration with third parties.
Our Board and executive management team are conducting a comprehensive process to explore and evaluate strategic alternatives with the goal of maximizing shareholder value. Potential alternatives under evaluation include, but are not limited to, a financing, a merger or reverse merger, the sale of all or part of the Company, licensing of assets, a business combination, and/or other strategic transaction or series of related transactions involving our Company.
In the second quarter of 2023, we paused substantially all of our research and development activities, including not proceeding with the previously planned Phase 1 Part 2 clinical trial for FRTX-02, in order to conserve capital resources during our ongoing evaluation of potential strategic options and to determine the appropriate next development steps based on the results of our first-in-human Phase 1 Part 1 clinical trial. The outcome of our strategic options initiative will dictate the Company’s future path including to what extent the current pipeline continues to be developed. In order to commence additional clinical trials for FRTX-02, or meaningfully progress our other pipeline assets, we would need to raise additional funds or enter into a strategic transaction with a partner who wishes to continue development of these assets.
Key Components of Operations
Revenue
Revenue generally consists of revenue recognized under our strategic agreements for the development and commercialization of our product candidates. Our strategic agreements generally outline overall development plans and include payments we receive at signing, payments for the achievement of certain milestones, sublicense income, earnout payments on net product sales, and royalties on net product sales. For these activities and payments, we utilize judgment to assess the nature of the performance obligations to determine whether the performance obligations are satisfied over time or at a point in time and, if over time, the appropriate method of measuring progress for purposes of recognizing revenue. Prior to entering into the Asset Purchase Agreement, we recognized royalty revenue earned on a percentage of net sales of ECCLOCK in Japan. Beginning in the second quarter of 2022, we began recognizing contract revenue pursuant to the terms of the Asset Purchase Agreement. After entering into the Asset Purchase Agreement Amendment in July 2023 and receiving $6.6 million from Botanix on July 27, 2023, we do not expect to generate any further significant revenue from any product candidates that we developed or develop unless and until we obtain regulatory approval and commercialize our products or enter into other collaboration agreements with third parties.
Research and Development Expenses
Research and development expenses principally consist of payments to third parties known as clinical research organizations (“CROs”) and upfront in-licensing fees of development-stage assets. CROs help plan, organize, and conduct clinical and nonclinical studies under our direction. Personnel costs, including wages, benefits, and share-based compensation, related to our research and development staff in support of product development activities are also included, as well as costs incurred for supplies, clinical and nonclinical studies, consultants, and facility and related overhead costs.
General and Administrative Expenses
General and administrative expenses consist primarily of personnel costs, including wages, benefits, and share-based compensation, related to our executive, sales, marketing, finance, and human resources personnel, as well as professional fees, including legal, accounting, and sublicensing fees.
Total Other Income, Net
Other income, net consists primarily of interest income, interest expense, and various income or expense items of a non-recurring nature. We have earned interest income from money market funds and interest-bearing accounts. Our interest income varies each reporting period depending on our average cash balances during the period and market interest rates. We expect interest income to fluctuate in the future with changes in average cash balances and market interest rates.
Critical Accounting Estimates
We have prepared the condensed consolidated financial statements in accordance with accounting principles generally accepted in the United States of America (“U.S. GAAP”). The preparation of these condensed consolidated financial statements requires us to make estimates, assumptions, and judgments that affect the reported amounts of assets, liabilities, and related disclosures at the date of the condensed consolidated financial statements, and the reported amounts of revenue and expenses during the reporting period. On an ongoing basis, management evaluates its critical estimates, including those related to revenue recognition and accrued research and development expenses. We base our estimates on our historical experience and on assumptions that we believe are reasonable; however, actual results may differ materially from these estimates under different assumptions or conditions.
There were no changes during the six months ended June 30, 2023 to our critical accounting estimates as disclosed in our Annual Report on Form 10-K for the year ended December 31, 2022. For information on our significant accounting policies, please refer to Note 2 of the notes to our condensed consolidated financial statements included elsewhere in this Quarterly Report.
Recent Accounting Pronouncements
We believe that the impact of recently issued guidance, whether adopted or to be adopted in the future, is not expected to have a material impact on our condensed consolidated financial statements upon adoption.
Results of Operations
Comparison of the Three Months Ended June 30, 2023 and 2022
| | | | | | | | | | | | | | | | | |
| Three Months Ended
June 30, | | |
| 2023 | | 2022 | | Change |
| (in thousands) | | |
| | | | | |
| | | | | |
| Revenue | 53 | | | 4,315 | | | (4,262) | |
| | | | | |
| | | | | |
| | | | | |
| Research and development expenses | (609) | | | (1,865) | | | (1,256) | |
| General and administrative expenses | (1,808) | | | (3,908) | | | (2,100) | |
| | | | | |
| | | | | |
| | | | | |
| | | | | |
| Total other income, net | 80 | | | 311 | | | (231) | |
| | | | | |
| Net loss | $ | (2,284) | | | $ | (1,147) | | | $ | (1,137) | |
Revenue
Revenue decreased by $4.3 million for the three months ended June 30, 2023 compared to the three months ended June 30, 2022. Revenue for the three months ended June 30, 2023 primarily consisted of Sublicense Income under the Asset Purchase Agreement with Botanix. Revenue for the three months ended June 30, 2022 primarily consisted of contract revenue recognized under the Asset Purchase Agreement and TSA with Botanix. Upon entering into the Asset Purchase Agreement on the Effective Date, whereby we sold all rights, title, and interests to assets primarily related to sofpironium bromide that were owned and/or licensed by us, and through June 30, 2022, we recognized contract revenue that was associated with the following: an upfront payment from Botanix of $3.0 million; reimbursed development expenditures from Botanix under the Asset Purchase Agreement of $0.6 million; fees for consulting services we provided under the TSA with Botanix of $0.4 million; and Sublicense Income under the Asset Purchase Agreement of $0.3 million.
Research and Development
Below is a summary of our research and development expenses by period related to our programs:
| | | | | | | | | | | | | | | | | | | | | | |
| Three Months Ended
June 30, | | | | | |
| 2023 | | 2022 | | | | | | | Change |
| | | | | | | | | | |
| (in thousands) | | | |
| | | | |
| | | | | | | | | | |
| | | | | | | | | | |
| | | | | | | | | | |
| Direct program expenses related to | | | | | | | | | | |
| | | | | | | | | | |
| DYRK1A inhibitor program (FRTX-02) | 153 | | | 958 | | | | | | | | (805) | |
| STING inhibitor program (FRTX-10) | 5 | | | 28 | | | | | | | | (23) | |
| | | | | | | | | | |
| | | | | | | | | | |
| | | | | | | | | | |
| | | | | | | | | | |
| | | | | | | | | | |
| | | | | | | | | | |
| Personnel and other unallocated expenses | 451 | | | 879 | | | | | | | | (428) | |
| Total research and development expenses | $ | 609 | | | $ | 1,865 | | | | | | | | $ | (1,256) | |
Research and development expenses decreased by $1.3 million for the three months ended June 30, 2023 compared to the three months ended June 30, 2022, driven primarily by decreased expenses of $0.8 million related to our DYRK1A inhibitor program and $0.4 million for personnel and other unallocated expenses. In May 2022, we initiated Part 1 of a two-part Phase 1 clinical trial in Canada for FRTX-02 that was completed in December 2022, with topline results reported in March 2023, as described above under the heading “Topline Results of FRTX-02 Phase 1 Clinical Trial.”
General and Administrative Expenses
General and administrative expenses decreased by $2.1 million for the three months ended June 30, 2023 compared to the three months ended June 30, 2022, which was primarily the result of $1.0 million in lower legal and compliance fees, $0.5 million for a payment to Bodor in June 2022 under the Rights Agreement that did not recur, $0.3 million in lower compensation-related expenses, and $0.3 million in lower other administrative fees.
Total Other Income, Net
Total other income, net decreased by $0.2 million for the three months ended June 30, 2023 compared to the three months ended June 30, 2022. Total other income, net in the 2023 period primarily consisted of interest income on our investment balances and in the 2022 period primarily consisted of a gain from the extinguishment of liabilities assumed by Botanix related to development costs prior to the Effective Date of the Asset Purchase Agreement.
Comparison of the Six Months Ended June 30, 2023 and 2022
| | | | | | | | | | | | | | | | | | | | | |
| | | Six Months Ended
June 30, | | |
| | | | | 2023 | | 2022 | | Change |
| | | | | (in thousands) | | |
| Revenue | | | | | $ | 62 | | | $ | 4,407 | | | $ | (4,345) | |
| | | | | | | | | |
| | | | | | | | | |
| Research and development expenses | | | | | (2,545) | | | (7,878) | | | 5,333 | |
| General and administrative expenses | | | | | (4,222) | | | (7,394) | | | 3,172 | |
| | | | | | | | | |
| | | | | | | | | |
| | | | | | | | | |
| | | | | | | | | |
| | | | | | | | | |
| | | | | | | | | |
| Other income, net | | | | | 145 | | | 308 | | | (163) | |
| Net loss attributable to common stockholders | | | | | $ | (6,560) | | | $ | (10,557) | | | $ | 3,997 | |
Revenue
Revenue decreased by approximately $4.3 million for the six months ended June 30, 2023 compared to the six months ended June 30, 2022. Revenue for the six months ended June 30, 2023 primarily consisted of Sublicense Income under the Asset Purchase Agreement with Botanix. Revenue for the six months ended June 30, 2022 primarily consisted of contract revenue recognized under the Asset Purchase Agreement and TSA with Botanix. Upon entering into the Asset Purchase Agreement on the Effective Date, whereby we sold all rights, title, and interests to assets primarily related to sofpironium bromide that were owned and/or licensed by us, and through June 30, 2022, we recognized contract revenue that was associated with the following: an upfront payment from Botanix of $3.0 million; reimbursed development expenditures from Botanix under the Asset Purchase Agreement of $0.6 million; fees for consulting services we provided under the TSA with Botanix of $0.4 million; and Sublicense Income under the Asset Purchase Agreement of $0.3 million.
Research and Development Expenses
Below is a summary of our research and development expenses by period related to our programs:
| | | | | | | | | | | | | | | | | | | | | | |
| | | Six Months Ended
June 30, | | | |
| | | | | 2023 | | 2022 | | | Change |
| | | | | | | | | | |
| | | | | (in thousands) | | | |
| | | | | | | | |
| | | | | | | | | | |
| | | | | | | | | | |
| | | | | | | | | | |
| Direct program expenses related to | | | | | | | | | | |
| Sofpironium bromide | | | | | $ | — | | | $ | 2,090 | | | | $ | (2,090) | |
| DYRK1A inhibitor program (FRTX-02) | | | | | 1,089 | | | 1,686 | | | | (597) | |
| STING inhibitor program (FRTX-10) | | | | | 107 | | | 2,038 | | | | (1,931) | |
| | | | | | | | | | |
| | | | | | | | | | |
| | | | | | | | | | |
| | | | | | | | | | |
| | | | | | | | | | |
| | | | | | | | | | |
| Personnel and other unallocated expenses | | | | | 1,349 | | | 2,064 | | | | (715) | |
| Total research and development expenses | | | | | $ | 2,545 | | | $ | 7,878 | | | | $ | (5,333) | |
Research and development expenses decreased by $5.3 million for the six months ended June 30, 2023 compared to the six months ended June 30, 2022, driven primarily by decreased costs of $2.1 million related to clinical development of sofpironium bromide, $1.9 million related to our STING inhibitor program, $0.7 million in lower personnel and other unallocated expenses, and $0.6 million related to our DYRK1A inhibitor program. Additional detail on our programs is as follows:
•Sofpironium bromide. In the fourth quarter of 2021, we completed our Phase 3 pivotal clinical program for sofpironium bromide gel, 15% and incurred $2.1 million in expenses during the six months ended June 30, 2022 associated with our Phase 3 pivotal clinical program. Subsequent to the Effective Date of the Asset Purchase Agreement, when we sold the assets primarily related to sofpironium bromide that we previously owned and/or licensed to Botanix, Botanix is responsible for all further research, development, and commercialization of sofpironium bromide.
•DYRK1A inhibitor program. In May 2022, we initiated Part 1 of a two-part Phase 1 clinical trial in Canada for FRTX-02 that was completed in December 2022, and we reported topline results in March 2023, as described above under the heading “Topline Results of FRTX-02 Phase 1 Clinical Trial.”
•STING inhibitor program. In February 2022, we acquired a portfolio of novel, potent, and orally available STING inhibitors that has broad potential in autoimmune, inflammatory, and rare genetic diseases, of which our primary product candidate is FRTX-10. To date, the expenses associated with our STING inhibitor program primarily relate to upfront in-licensing fees of $2.0 million incurred during the six months ended June 30, 2022.
•Personnel and other unallocated expenses. Personnel and other expenses include operating expenses related to research and development activities not specifically attributable to a specific program. Other expenses include travel, office supplies, license fees, and other miscellaneous expenses. These expenses vary over time depending on the development phase of the assets, the timing of acquisition or disposition of the assets, and other variables inherent in carrying out preclinical and clinical studies.
General and Administrative Expenses
General and administrative expenses decreased by $3.2 million for the six months ended June 30, 2023 compared to the six months ended June 30, 2022. The decrease of $3.2 million was primarily related to $1.6 million in lower legal and compliance fees, $0.5 million for a payment in June 2022 to Bodor under the Rights Agreement that did not recur, $0.5 million in lower compensation-related expenses, and $0.5 million in lower other expenses.
Total Other Income, Net
Total other income, net increased by $0.2 million for the six months ended June 30, 2023 compared to the six months ended June 30, 2022. Total other income, net in the 2023 period primarily consisted of interest income on our investment balances and in the 2022 period primarily consisted of a gain from the extinguishment of liabilities assumed by Botanix related to development costs during the three months ended June 30, 2022 prior to the Effective Date of the Asset Purchase Agreement.
Liquidity and Capital Resources
We have incurred significant operating losses and have an accumulated deficit as a result of ongoing efforts to in-license and develop our product candidates, including conducting preclinical and clinical trials and providing general and administrative support for these operations. For the six months ended June 30, 2023 and 2022, we had a net loss of $6.6 million and $10.6 million, respectively. As of June 30, 2023, we had an accumulated deficit of $173.0 million. As of June 30, 2023, we had cash and cash equivalents of $8.9 million. Since inception, we have financed our operations primarily through funds received from the sale of common stock and warrants, convertible preferred stock, debt and convertible notes, and payments received under license and other strategic agreements.
We believe that our cash and cash equivalents as of June 30, 2023, combined with $6.6 million in net proceeds received in July 2023 (as described further in Note 3. “Strategic Agreements”), will be sufficient to fund our operations for at least the next 12 months. However, it is difficult to predict our spending for our product candidates or other business opportunities. Moreover, changing circumstances may cause us to expend cash significantly faster than we currently anticipate, and we may need to spend more cash than currently expected because of circumstances beyond our control. We expect to continue to incur additional losses for the foreseeable future. Our Board and executive management team are conducting a comprehensive process to explore and evaluate strategic alternatives with the goal of maximizing shareholder value. Potential alternatives under evaluation include, but are not limited to, a financing, a merger or reverse merger, the sale of all or part of the Company, licensing of assets, a business combination, and/or other strategic transaction or series of related transactions involving our Company. In order to commence additional clinical trials for FRTX-02, or meaningfully progress our other pipeline assets, we would need to raise additional funds or enter into a strategic transaction with a partner who wishes to continue development of these assets. To the extent that additional funds are raised through the sale of equity, the issuance of securities will result in dilution to our stockholders.
Additionally, we are subject to the SEC’s “baby shelf rules,” which prohibit companies with a public float of less than $75 million from issuing securities under a shelf registration statement in excess of one-third of such company’s public float in a 12-month period. These rules currently limit our future issuances of shares under the ATM Agreements or other common stock offerings.
Cash Flows
Since inception, we have primarily used our available cash to fund expenditures related to efforts to in-license and develop our product candidates, including conducting preclinical and clinical trials and providing general
and administrative support for these operations. The following table sets forth a summary of cash flows for the periods presented:
| | | | | | | | | | | |
| | | |
| Six Months Ended
June 30, |
| 2023 | | 2022 |
| | | |
| | | |
| (in thousands) |
| Net cash provided by (used in): | | | |
| Operating activities | $ | (6,301) | | | $ | (12,509) | |
| | | |
| Financing activities | 6,569 | | | 105 | |
| Total | $ | 268 | | | $ | (12,404) | |
Operating Activities
Net cash used in operating activities of $6.3 million during the six months ended June 30, 2023 decreased compared to $12.5 million during the six months ended June 30, 2022, primarily as a result of a decrease in cash used to support our operating activities, including but not limited to our clinical trials, research and development activities, and general working capital requirements. The $6.2 million decrease was impacted by the net effect of a decrease in net loss of $4.0 million, reduced for non-cash operating expenses of approximately $0.3 million, and the net effect of changes in working capital of $2.5 million.
Financing Activities
Net cash provided by financing activities during the six months ended June 30, 2023 increased by $6.5 million compared to the six months ended June 30, 2022, primarily resulting from increased net proceeds received during the six months ended June 30, 2023 of $6.4 million from sales of our common stock under the 2021 ATM Agreement.
ITEM 3. QUANTITATIVE AND QUALITATIVE DISCLOSURES ABOUT MARKET RISK
We are a smaller reporting company as defined by Rule 12b-2 of the Securities Exchange Act of 1934, as amended (the “Exchange Act”), and are not required to provide the information under this item.
ITEM 4. CONTROLS AND PROCEDURES
Evaluation of Disclosure Controls and Procedures
We maintain disclosure controls and procedures that are designed to ensure that information required to be disclosed in the reports that we file under the Exchange Act is recorded, processed, summarized, and reported within the time periods specified in the rules and forms of the SEC, and that such information is accumulated and communicated to our management, including our principal executive officer and principal financial officer, as appropriate, to allow timely decisions regarding required disclosures. In designing and evaluating the disclosure controls and procedures, management recognizes that any controls and procedures, no matter how well designed and operated, can provide only reasonable assurance of achieving the desired control objectives, and management is required to apply its judgment in evaluating the cost-benefit relationship of possible controls and procedures.
Conclusion Regarding the Effectiveness of Disclosure Controls and Procedures
Under the supervision and with the participation of our management, including our principal executive officer and principal financial officer, we conducted an evaluation of the design and operation of our disclosure controls and procedures, as such term is defined in Rule 13a-15(e) and 15d-15(e) promulgated under the Exchange Act, as of the end of the period covered by this Quarterly Report. Based on this evaluation, our principal executive officer and principal financial officer concluded that our disclosure controls and procedures were effective and were operating at a reasonable assurance level as of June 30, 2023.
Changes in Internal Control over Financial Reporting
Management has determined that there were no changes in our internal control over financial reporting that occurred during the three months ended June 30, 2023 that have materially affected, or are reasonably likely to materially affect, our internal control over financial reporting.
PART II. OTHER INFORMATION
ITEM 1. LEGAL PROCEEDINGS
From time to time, we may become involved in legal proceedings arising in the ordinary course of our business. We are not presently a party to any legal proceedings that, if determined adversely to us, would individually or taken together have a material adverse effect on our Company, nor is any such litigation threatened as of the date of this filing.
ITEM 1A. RISK FACTORS
Our business, financial condition, and operating results may be affected by a number of factors, whether currently known or unknown, including but not limited to those described in Part I, Item 1A of our Annual Report on Form 10-K for the year ended December 31, 2022 under the heading “Risk Factors.” Any one or more of such factors could directly or indirectly cause our actual results of operations and financial condition to vary materially from past or anticipated future results of operations and financial condition. Any of these factors, in whole or in part, alone or combined with any of the other factors, could materially and adversely affect our business, financial condition, results of operations, and stock price. There have been no changes to our risk factors since our Annual Report on Form 10-K for the year ended December 31, 2022, except as set forth below.
The risk factor in our Annual Report on Form 10-K for the year ended December 31, 2022 entitled “Our inability to regain and maintain compliance with Nasdaq continued listing requirements could result in the delisting of our common stock” is replaced in its entirety by the following:
Our inability to regain and maintain compliance with Nasdaq continued listing requirements could result in the delisting of our common stock.
Our common stock is currently listed on The Nasdaq Capital Market. In order to maintain this listing, we must satisfy minimum financial, governance, and other requirements. On August 19, 2022, we received a notice (the “2022 Notice”) from the Listing Qualifications Department of the Nasdaq Stock Market (“Nasdaq”) stating that the departure of Dennison T. Veru from the Board in July 2022 resulted in noncompliance with the independent director and audit committee requirements set forth in Nasdaq Listing Rule 5605. More specifically, the Board was not comprised of a majority of “independent directors” within the meaning of Nasdaq Listing Rule 5605(a)(2), and the Board’s Audit Committee did not have at least three members, each of whom was independent and met the criteria for independence set forth in Rule 10A-3(b)(1) under the Exchange Act, as required by Nasdaq Listing Rule 5605(c)(2)(A). At the time of the 2022 Notice, the Board had two independent members and two non-independent members, and the Audit Committee consisted of two independent members.
On July 24, 2023, Robert B. Brown resigned from our Board, effective immediately, and the Board appointed Reginald L. Hardy to the Audit Committee of the Board under the exceptional and limited circumstances provision of Nasdaq Listing Rule 5605(d)(2)(B). On July 25, 2023, we received formal notice from Nasdaq stating that we have regained compliance with the independent director and audit committee requirements set forth in Nasdaq Listing Rule 5605. The Board is now comprised of a majority of “independent directors” within the meaning of Nasdaq Listing Rule 5605(a)(2), and the Board’s Audit Committee now has three members, each of whom meets the requirements of Nasdaq Listing Rule 5605(c)(2).
However, Mr. Lyons has informed us that he will resign from the Board on the date of our 2023 annual meeting of stockholders due to overboarding concerns under proxy advisor and other institutional investor policies. The date of our 2023 annual meeting of stockholders has not yet been determined. Upon Mr. Lyons’ resignation, we will no longer comply with Nasdaq Listing Rule 5605 unless a director is added to the Board who (i) qualifies as an “independent director” within the meaning of Nasdaq Listing Rule 5605(a)(2) and (ii) meets the enhanced
independence requirements applicable to audit committee members under Nasdaq Listing Rule 5605(c)(2). We will take appropriate future actions to address Mr. Lyons’ pending departure.
In addition, on April 24, 2023, we received a notice from the Listing Qualifications Department of Nasdaq informing us that because the closing bid price for our common stock listed on Nasdaq was below $1.00 per share for 30 consecutive business days, we do not comply with the minimum closing bid price requirement for continued listing on The Nasdaq Capital Market under Nasdaq Marketplace Rule 5550(a)(2). In accordance with Nasdaq’s Listing Rules, we have a period of 180 calendar days, or until October 23, 2023, to regain compliance with such rule.
However, there can be no assurance that we will be able to regain compliance with Nasdaq’s listing standards. If our common stock is delisted from Nasdaq and we are unable to list our common stock on another national securities exchange, we expect our common stock would be quoted on an over-the-counter market. If this were to occur, we and our stockholders could face significant material adverse consequences, including limited availability of market quotations for our common stock; substantially decreased trading in our common stock; decreased market liquidity of our common stock as a result of the loss of market efficiencies associated with Nasdaq and the loss of federal preemption of state securities laws; an adverse effect on our ability to issue additional securities or obtain additional financing in the future on acceptable terms, if at all; potential loss of confidence by investors, suppliers, partners, and employees and fewer business development opportunities; and limited news and analyst coverage. Additionally, the market price of our common stock may decline further, and stockholders may lose some or all of their investment.
Even if we are not delisted, the perception among investors that we are at a heightened risk of delisting could negatively affect the market price and trading volume of our common stock, or our ability to raise capital.
The risk factor in our Annual Report on Form 10-K for the year ended December 31, 2022 entitled “Our receipt of future payments from Botanix is contingent on various factors outside of our control, including the successful development, regulatory approval, and commercialization of sofpironium bromide gel, 15%, by Botanix outside of Japan, the successful continued commercialization of ECCLOCK by Kaken in Japan, and the sufficiency of funds by both entities to pay us and Bodor, the licensor of this product” is no longer applicable and is hereby deleted in its entirety.
ITEM 2. UNREGISTERED SALES OF EQUITY SECURITIES AND USE OF PROCEEDS
None.
ITEM 3. DEFAULTS UPON SENIOR SECURITIES
Not applicable.
ITEM 4. MINE SAFETY DISCLOSURES
Not applicable.
ITEM 5. OTHER INFORMATION
During the three months ended June 30, 2023, none of our directors or officers (as defined in Rule 16a-1(f) of the Exchange Act) adopted or terminated any contract, instruction, or written plan for the purchase or sale of our securities that was intended to satisfy the affirmative defense conditions of Rule 10b5-1(c) of the Exchange Act or any non-Rule 10b5-1 trading arrangement (as defined in the SEC’s rules).
ITEM 6. EXHIBITS
| | | | | | | | | | | | | | | | | | | | | | | | | | | |
Exhibit
Number | | Description of Exhibit | Form | Date of Filing | Exhibit Number | Filed Herewith | | | | | | | |
| | | | | | | | | | | | | |
| 3.1 | | | 8-K | 9/8/2022 | 3.2 | | | | | | | | |
| 3.2 | | | | | | × | | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | | | | | | | |
| | | | | | | | | | | | | |
| 10.1 | | | 10-Q | 5/10/2023 | 10.9 | | | | | | | | |
| 10.2 | | | 8-K | 7/21/2023 | 10.1 | | | | | | | | |
| 10.3 | | | 8-K | 7/21/2023 | 10.2 | | | | | | | | |
| 31.1 | | | | | | × | | | | | | | |
| 31.2 | | | | | | × | | | | | | | |
| 32.1* | | | | | | × | | | | | | | |
| 101.INS | | Inline XBRL Instance Document | | | | × | | | | | | | |
| 101.SCH | | Inline XBRL Taxonomy Extension Schema Document | | | | × | | | | | | | |
| 101.CAL | | Inline XBRL Taxonomy Extension Calculation Linkbase Document | | | | × | | | | | | | |
| 101.DEF | | Inline XBRL Taxonomy Extension Definition Linkbase Document | | | | × | | | | | | | |
| 101.LAB | | Inline XBRL Taxonomy Extension Label Linkbase Document | | | | × | | | | | | | |
| 101.PRE | | Inline XBRL Taxonomy Extension Presentation Linkbase Document | | | | × | | | | | | | |
| 104 | | Cover Page Interactive Data File (formatted as Inline XBRL and contained in Exhibit 101) | | | | × | | | | | | | |
__________________
| | | | | |
| |
| |
| |
| * | This certification is being furnished pursuant to 18 U.S.C. Section 1350 and is not being filed for purposes of Section 18 of the Exchange Act and is not to be incorporated by reference into any filing of the Registrant, whether made before or after the date hereof. |
| |
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this Quarterly Report to be signed on its behalf by the undersigned thereunto duly authorized.
| | | | | | | | | | | | | | |
| | Fresh Tracks Therapeutics, Inc. |
| | | | |
| Date: August 11, 2023 | | By: | | /s/ Andrew D. Sklawer |
| | | | Andrew D. Sklawer Chief Executive Officer (Principal Executive Officer) |
| | | | |
| | By: | | /s/ Aaron Fox-Collis |
| | | | Aaron Fox-Collis VP of Finance and Chief Accounting Officer (Principal Accounting Officer)
|