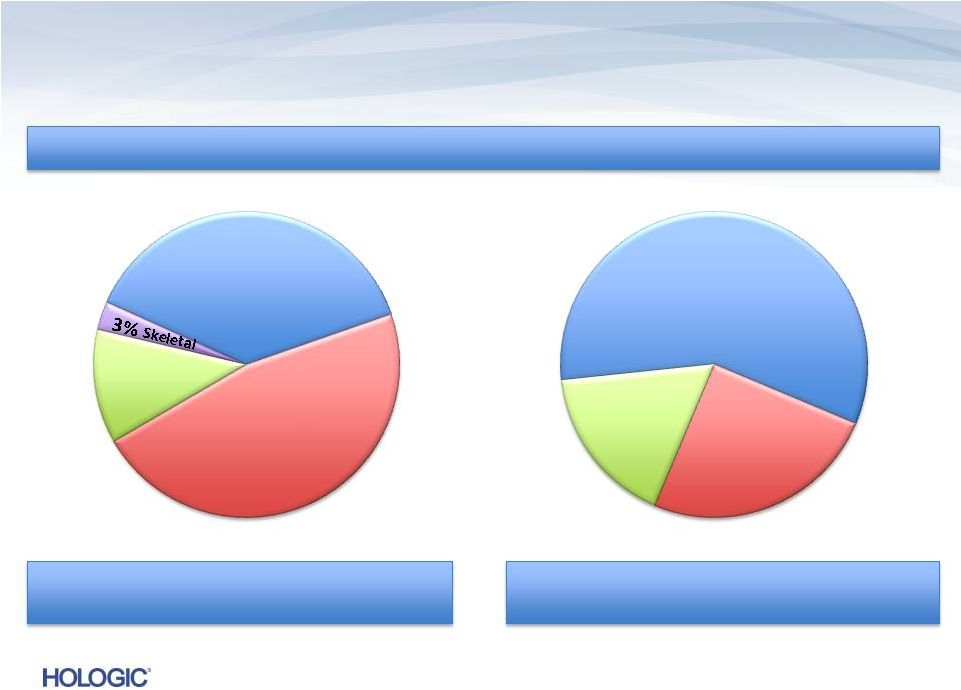
Safe Harbor Statement 2 This presentation contains forward-looking information that involves risks and uncertainties, including statements about the Company’s plans, objectives, expectations and intentions. Such statements include, without limitation: financial or other information included herein based upon or otherwise incorporating judgments or estimates relating to future performance, events or expectations; the Company’s strategies, positioning, resources, capabilities, and expectations for future performance; and the Company's outlook and financial and other guidance. These forward-looking statements are based upon assumptions made by the Company as of the date hereof and are subject to known and unknown risks and uncertainties that could cause actual results to differ materially from those anticipated. Risks and uncertainties that could adversely affect the Company’s business and prospects, and otherwise cause actual results to differ materially from those anticipated, include without limitation: the ability of the Company to successfully manage recent and ongoing leadership and organizational changes, including the ability of the Company to attract, motivate and retain key employees; U.S., European and general worldwide economic conditions and related uncertainties; the Company’s reliance on third-party reimbursement policies to support the sales and market acceptance of its products, including the possible adverse impact of government regulation and changes in the availability and amount of reimbursement and uncertainties for new products or product enhancements; uncertainties regarding the recently enacted or future healthcare reform legislation, including associated tax provisions, or budget reduction or other cost containment efforts; changes in guidelines, recommendations and studies published by various organizations that could affect the use of the Company’s products; uncertainties inherent in the development of new products and the enhancement of existing products, including FDA approval and/or clearance and other regulatory risks, technical risks, cost overruns and delays; the risk that products may contain undetected errors or defects or otherwise not perform as anticipated; risks associated with strategic alliances and the ability of the Company to realize anticipated benefits of those alliances; risks associated with acquisitions, including without limitation, the Company’s ability to successfully integrate acquired businesses, the risks that the acquired businesses may not operate as effectively and efficiently as expected even if otherwise successfully integrated, the risks that acquisitions may involve unexpected costs or unexpected liabilities including the risks and challenges associated with the Company’s recent acquisition of Gen-Probe and operations in China; the risks of conducting business internationally, including the effect of exchange rate fluctuations on those operations; manufacturing risks, including the Company’s reliance on a single or limited source of supply for key components, and the need to comply with especially high standards for the manufacture of many of its products; the Company’s ability to predict accurately the demand for its products, and products under development, and to develop strategies to address its markets successfully; the early stage of market development for certain of the Company’s products; the Company’s leverage risks, including the Company’s obligation to meet payment obligations and financial covenants associated with its debt; risks related to the use and protection of intellectual property; expenses, uncertainties and potential liabilities relating to litigation, including, without limitation, commercial, intellectual property, employment and product liability litigation; technical innovations that could render products marketed or under development by the Company obsolete; competition; and the Company’s ability to attract and retain qualified personnel. The risks included above are not exhaustive. Other factors that could adversely affect the company's business and prospects are described in the filings made by the Company with the SEC. The Company expressly disclaims any obligation or undertaking to release publicly any updates or revisions to any such statements presented herein to reflect any change in expectations or any change in events, conditions or circumstances on which any such statements are based. Hologic, Aptima, Cervista, Dimensions, Gen-Probe, MyoSure, NovaSure, Panther, ThinPrep, TMA and associated logos, as may be used throughout this presentation, are trademarks and/or registered trademarks of Hologic, Inc. and/or its subsidiaries in the United States and/or other countries. |