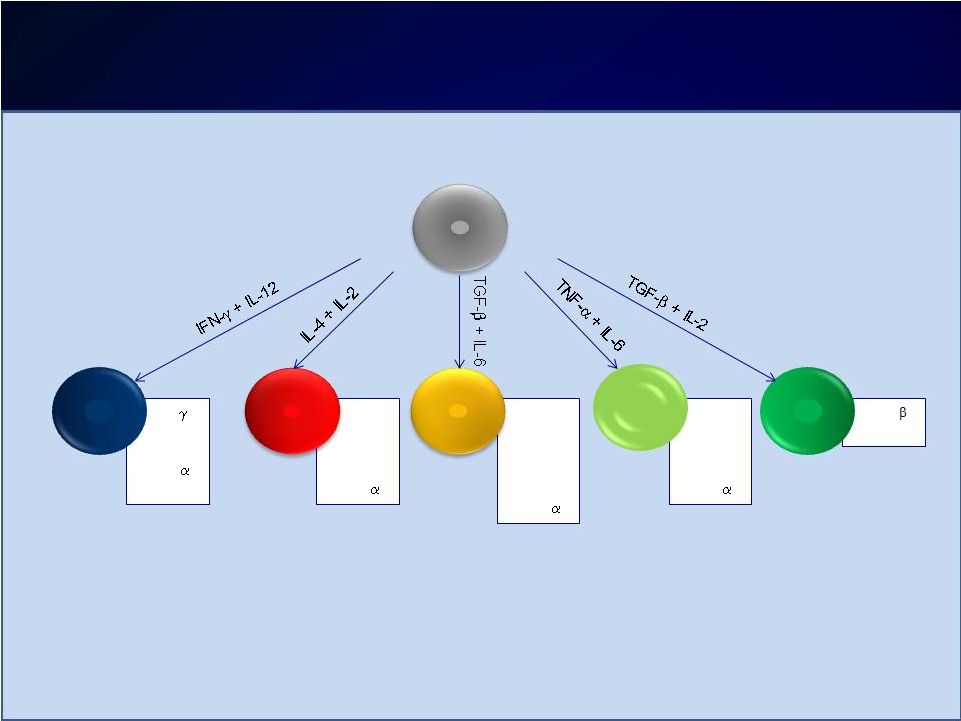
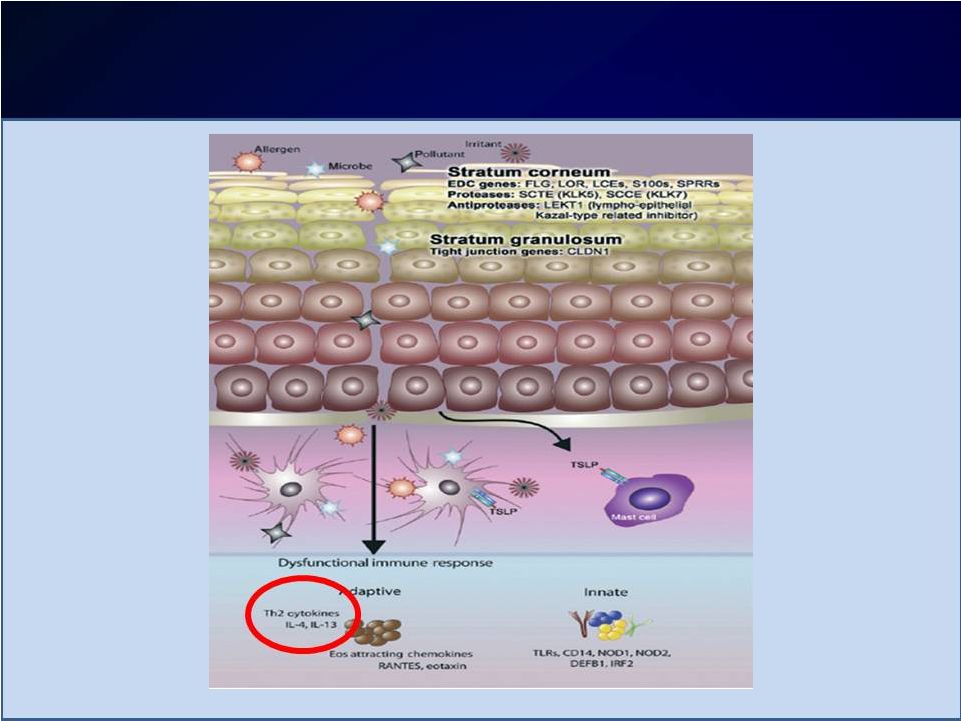
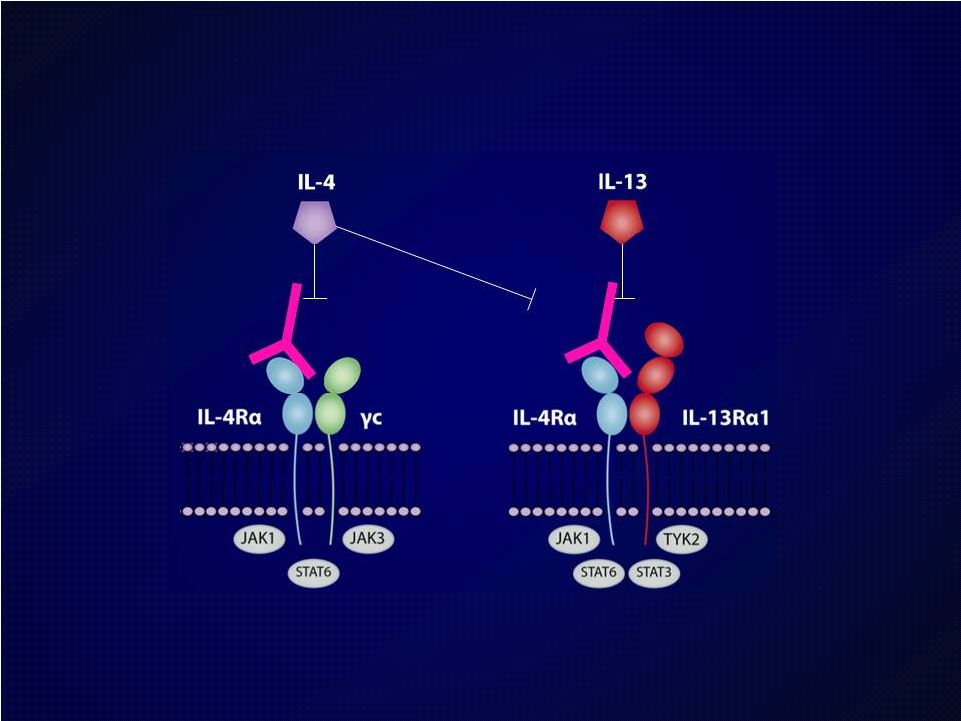
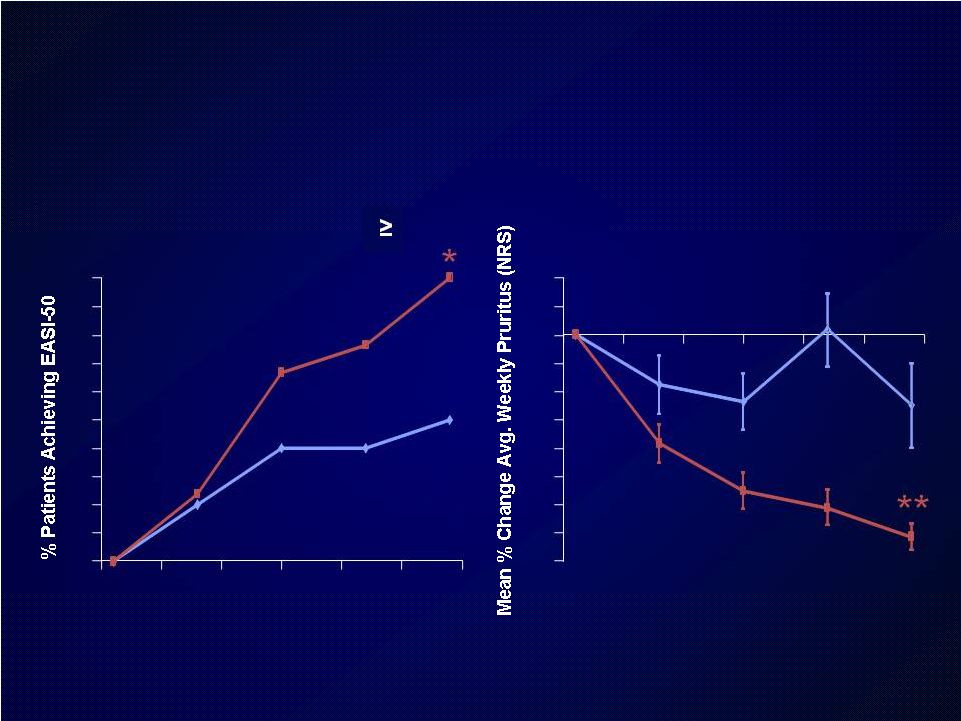
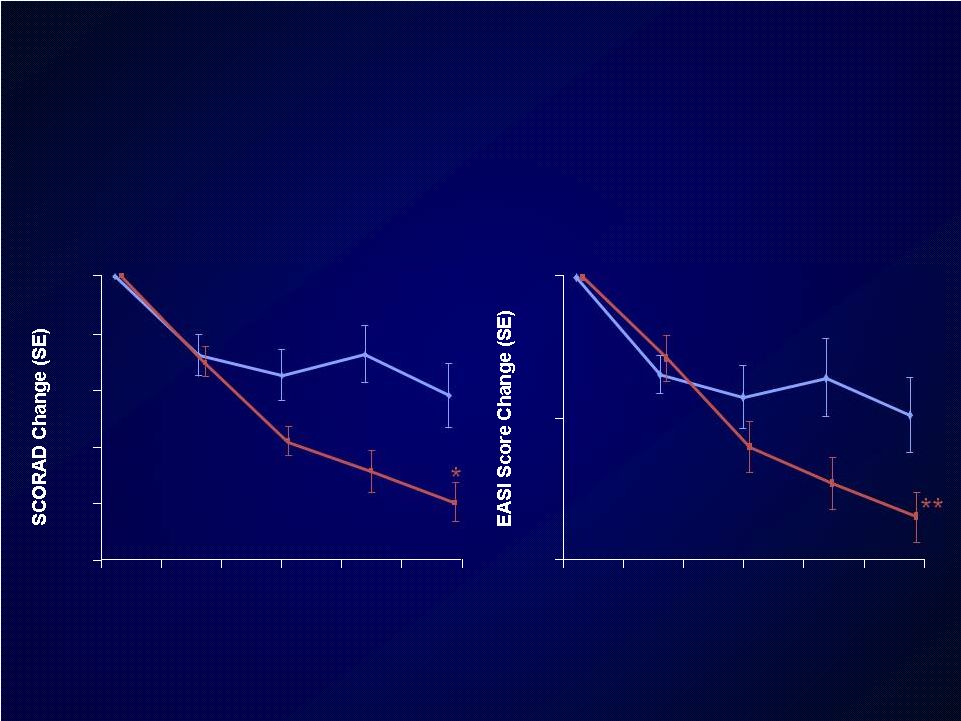
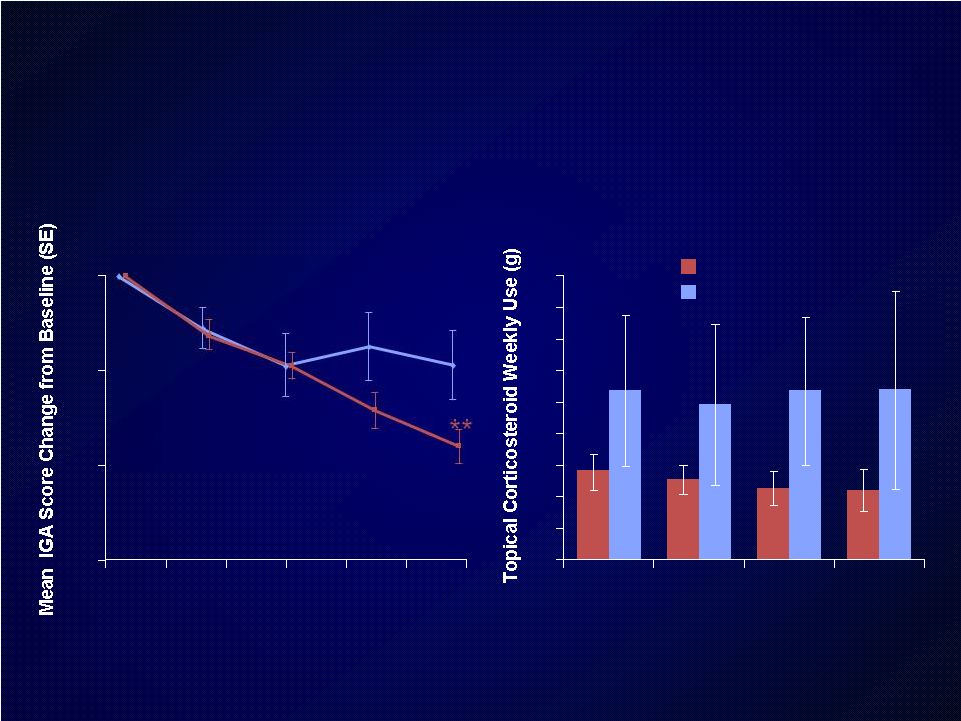
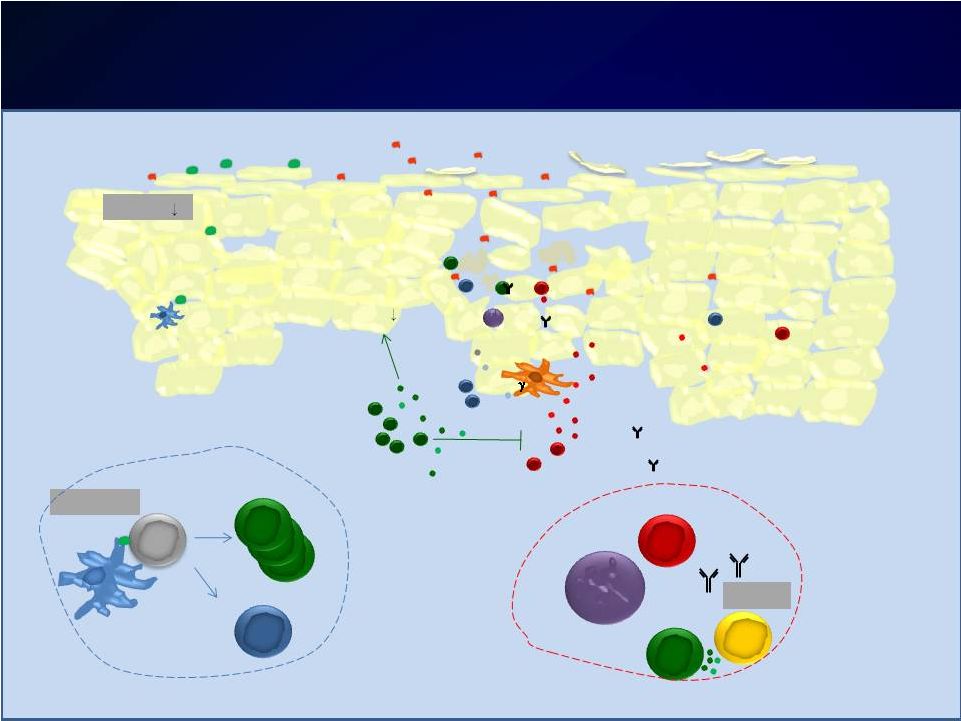
Introduction Introduction Moderate-to-severe atopic dermatitis (AD) is characterized by Moderate-to-severe atopic dermatitis (AD) is characterized by eczematous dermatitis with intractable pruritus associated with sleep eczematous dermatitis with intractable pruritus associated with sleep disturbance and lower quality-of-life disturbance and lower quality-of-life For many patients, current therapies are inadequate and can be For many patients, current therapies are inadequate and can be associated with unwanted side effects associated with unwanted side effects IL-4 and IL-13 are thought to be central to T-helper 2 (Th2) IL-4 and IL-13 are thought to be central to T-helper 2 (Th2) inflammation, which mediates many features of AD inflammation, which mediates many features of AD Dupilumab is a fully human monoclonal antibody targeting the IL-4 Dupilumab is a fully human monoclonal antibody targeting the IL-4 receptor alpha subunit (IL-4R ), thus blocking the intracellular signaling receptor alpha subunit (IL-4R ), thus blocking the intracellular signaling of both IL-4 and IL-13 of both IL-4 and IL-13 Earlier clinical trials indicated that dupilumab monotherapy had Earlier clinical trials indicated that dupilumab monotherapy had an an acceptable safety profile and was efficacious in patients with moderate acceptable safety profile and was efficacious in patients with moderate to severe AD who cannot be adequately controlled with topical to severe AD who cannot be adequately controlled with topical medications medications Since topical corticosteroids (TCS) are commonly used in AD, we Since topical corticosteroids (TCS) are commonly used in AD, we assessed the safety and efficacy of dupilumab co-administered with assessed the safety and efficacy of dupilumab co-administered with TCS TCS |