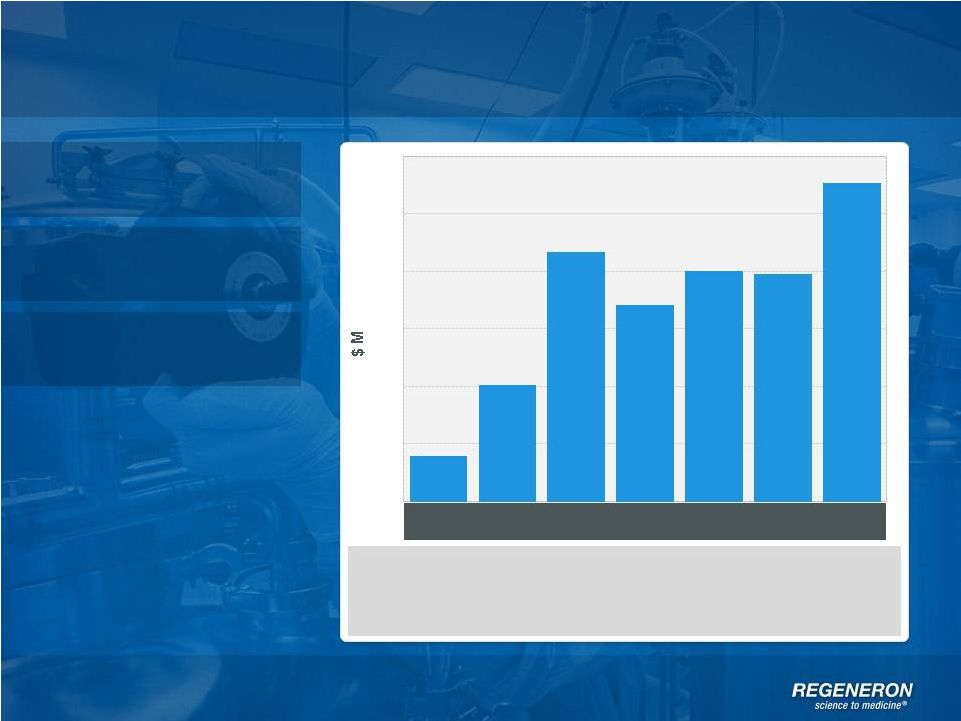
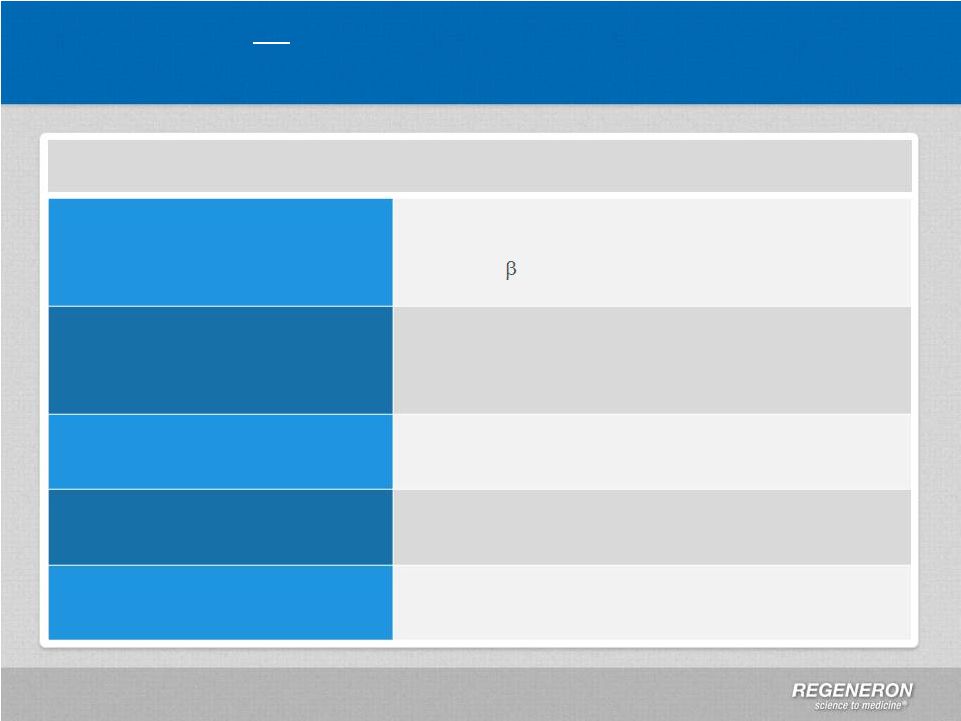
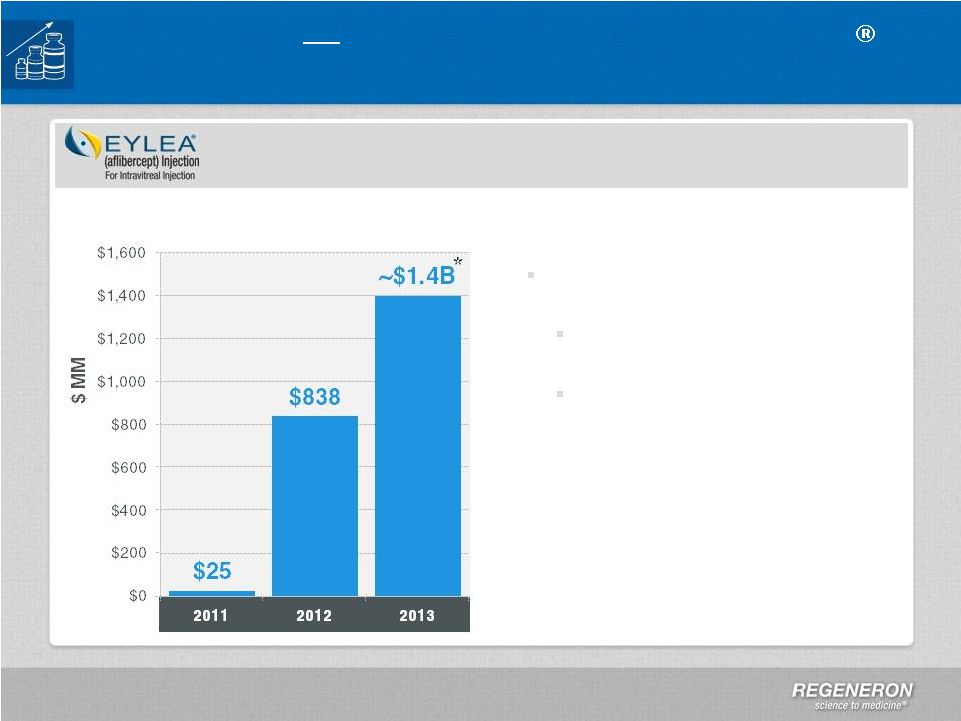
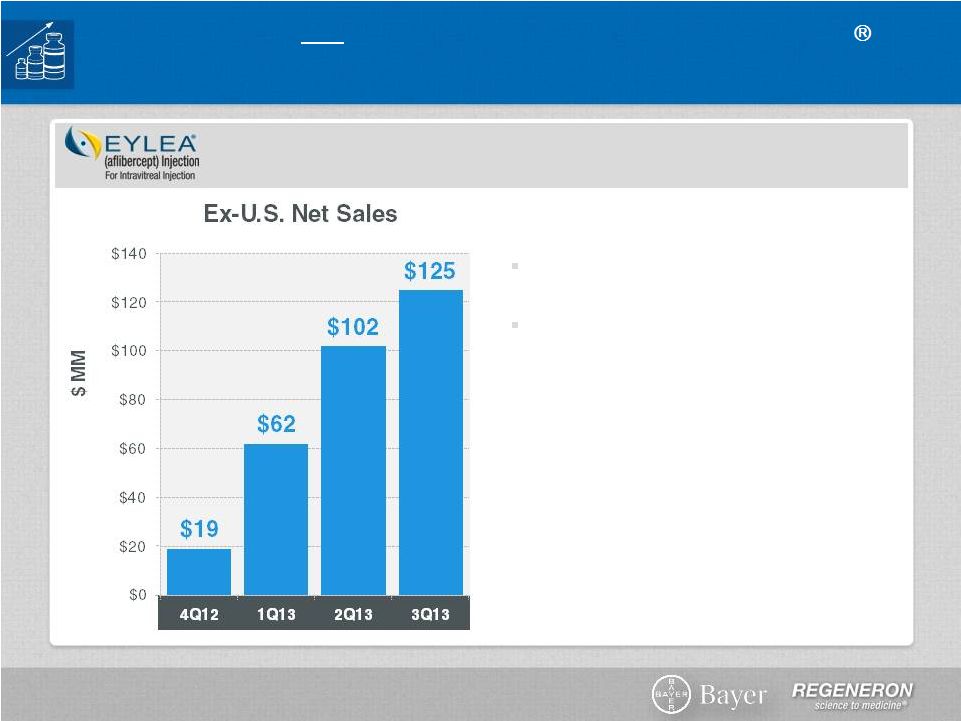
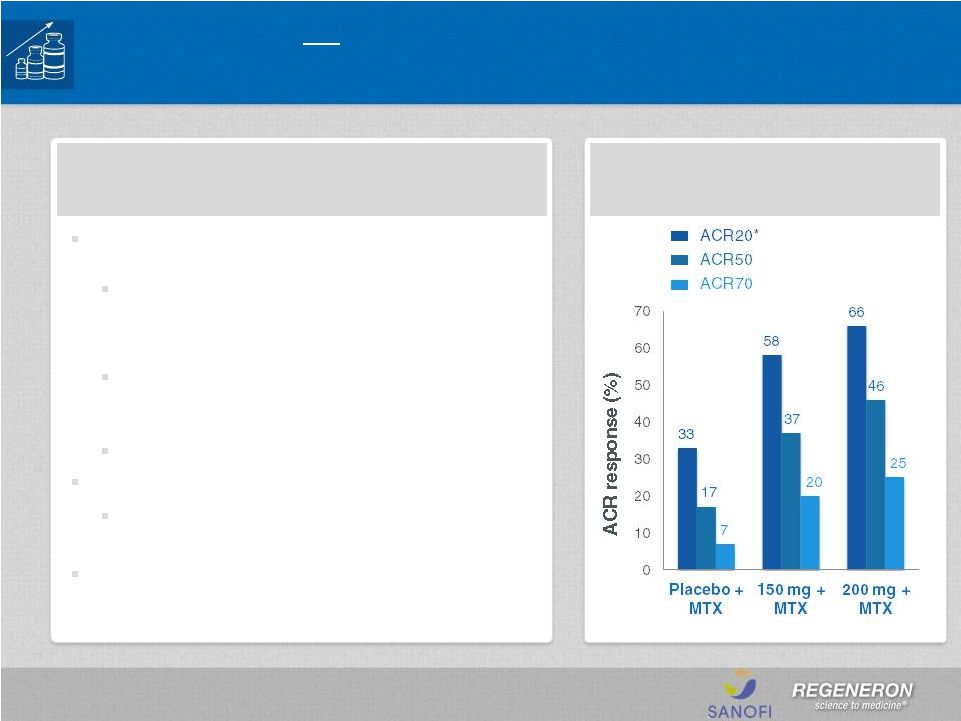
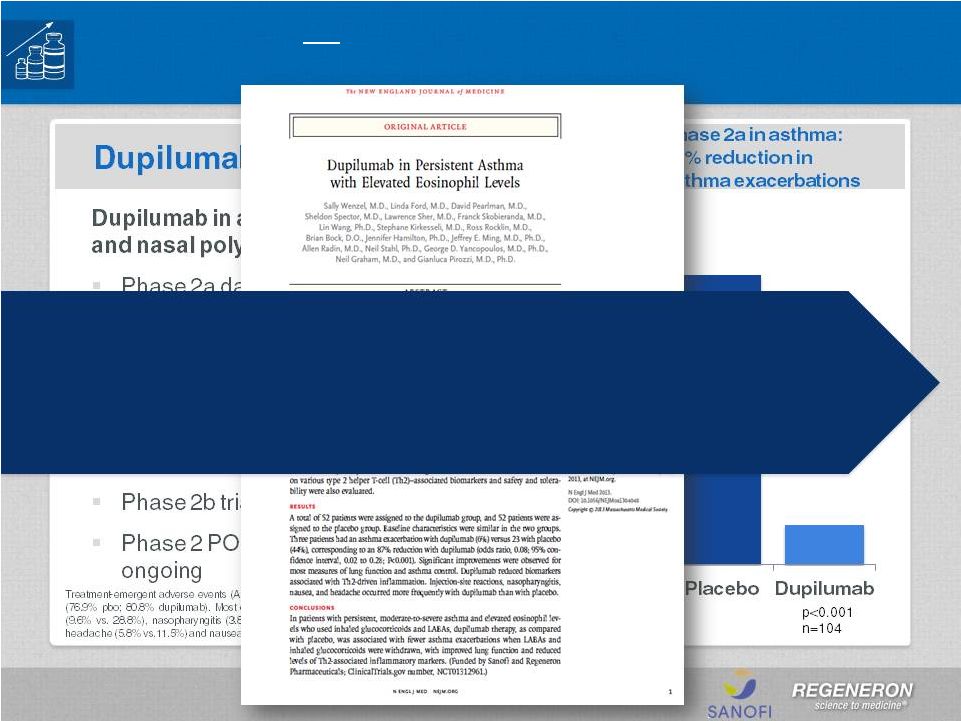
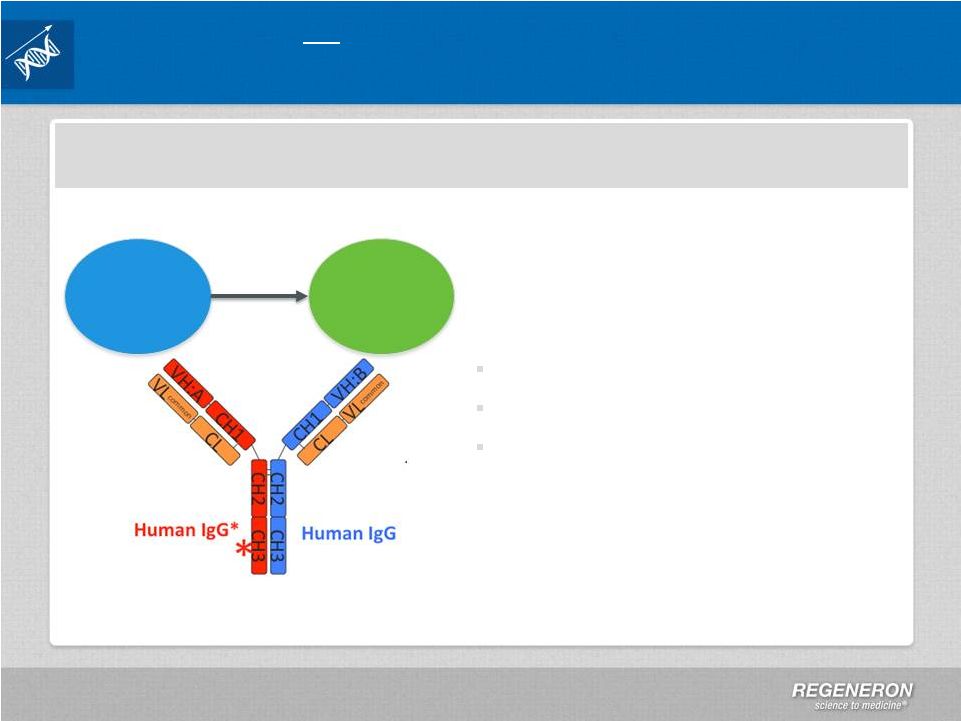
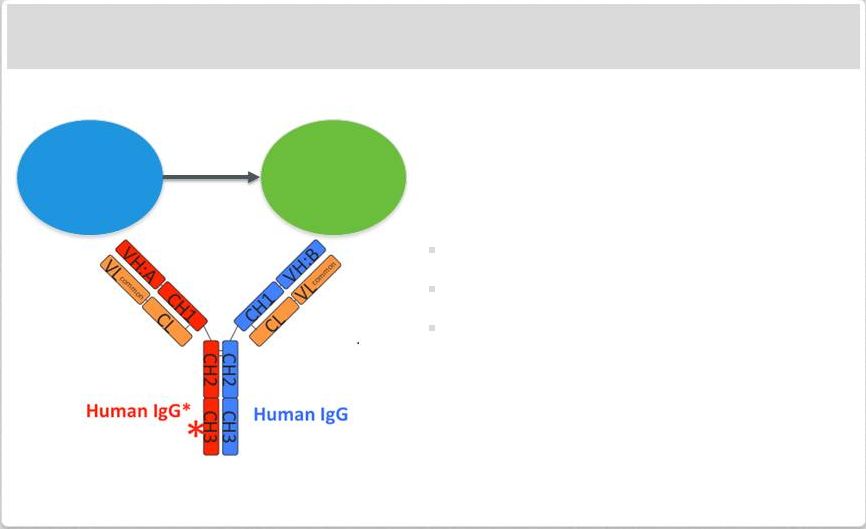
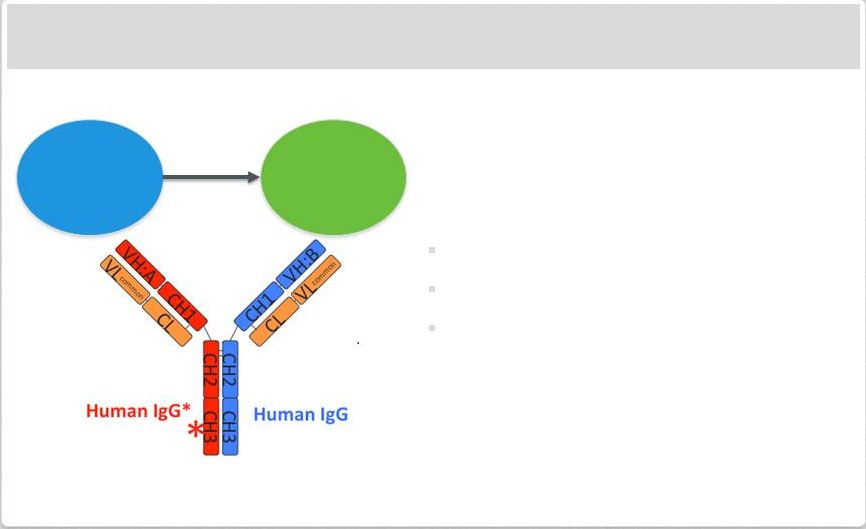
Page 2 | Copyright Regeneron 2014 Safe Harbor Statement This presentation includes forward-looking statements that involve risks and uncertainties relating to future events and the future performance of Regeneron Pharmaceuticals, Inc. (“Regeneron” or the “Company”), and actual events or results may differ materially from these forward-looking statements. Words such as “anticipate,” “expect,” “intend,” “plan,” “believe,” “seek,” “estimate,” variations of such words and similar expressions are intended to identify such forward-looking statements, although not all forward-looking statements contain these identifying words. These statements concern, and these risks and uncertainties include, among others, the nature, timing, and possible success and therapeutic applications of Regeneron's products, product candidates, and research and clinical programs now underway or planned; unforeseen safety issues resulting from the administration of products and product candidates in patients, including serious complications or side effects in connection with the use of Regeneron’s product candidates in clinical trials; the likelihood and timing of possible regulatory approval and commercial launch of Regeneron's late-stage product candidates and new indications for marketed products, including without limitation EYLEA ® , Alirocumab, Sarilumab, and Dupilumab; ongoing regulatory obligations and oversight and determinations by regulatory and administrative governmental authorities which may delay or restrict Regeneron's ability to continue to develop or commercialize Regeneron's products and product candidates; competing drugs and product candidates that may be superior to Regeneron's products and product candidates; uncertainty of market acceptance and commercial success of Regeneron's products and product candidates; the ability of Regeneron to manufacture and manage supply chains for multiple products and product candidates; coverage and reimbursement determinations by third-party payers, including Medicare and Medicaid; unanticipated expenses; the costs of developing, producing, and selling products; the ability of Regeneron to meet any of its sales or other financial projections or guidance and changes to the assumptions underlying those projections or guidance, including without limitation those relating to non-GAAP unreimbursed R&D, non-GAAP SG&A and capital expenditures, the potential for any license or collaboration agreement, including Regeneron's agreements with Sanofi and Bayer HealthCare, to be cancelled or terminated without any further product success; and risks associated with third party intellectual property and pending or future litigation relating thereto. A more complete description of these and other material risks can be found in Regeneron's filings with the U.S. Securities and Exchange Commission, including its Form 10-K for the fiscal year ended December 31, 2012 and its Form 10-Q for the quarterly period ended September 30, 2013, in each case including in the sections thereof captioned “Item 1A. Risk Factors.” Any forward-looking statements are made based on management's current beliefs and judgment, and the reader is cautioned not to rely on any forward-looking statements made by Regeneron. Regeneron does not undertake any obligation to update publicly any forward- looking statement, including without limitation any financial projection or guidance, whether as a result of new information, future events, or otherwise. This presentation uses non-GAAP net income, non-GAAP unreimbursed R&D, and non-GAAP SG&A, which are financial measures that are not calculated in accordance with the U.S. Generally Accepted Accounting Principles (“GAAP”). Regeneron believes that the presentation of these non-GAAP measures is useful to investors because they exclude, as applicable, (i) non-cash share-based compensation expense which fluctuates from period to period based on factors that are not within the Company's control, such as the Company's stock price on the dates share-based grants are issued, (ii) non-cash interest expense related to the Company's convertible senior notes since this is not deemed useful in evaluating the Company's operating performance, (iii) non-cash income tax expense, since the Company does not currently pay, or expect to pay in the near future, significant cash income taxes due primarily to the utilization of net operating loss and tax credit carry-forwards; therefore, non-cash income tax expense is not deemed useful in evaluating the Company's operating performance, and (iv) a non- cash tax benefit as a result of releasing substantially all of the valuation allowance associated with the Company’s deferred tax assets. Non-GAAP unreimbursed R&D represents non-GAAP R&D expenses reduced by R&D expense reimbursements from the Company's collaboration partners. Management uses these non- GAAP measures for planning, budgeting, forecasting, assessing historical performance, and making financial and operational decisions, and also provides forecasts to investors on this basis. However, there are limitations in the use of these and other non-GAAP financial measures as they exclude certain expenses that are recurring in nature. Furthermore, the Company's non-GAAP financial measures may not be comparable with non-GAAP information provided by other companies. Any non-GAAP financial measure presented by Regeneron should be considered supplemental to, and not a substitute for, measures of financial performance prepared in accordance with GAAP. A reconciliation of the Company's GAAP to non-GAAP results is included at the end of this presentation. |