Free signup for more
- Track your favorite companies
- Receive email alerts for new filings
- Personalized dashboard of news and more
- Access all data and search results
Filing tables
Filing exhibits
IONS similar filings
- 9 Aug 23 Ionis reports second quarter 2023 financial results
- 4 Aug 23 Termination of a Material Definitive Agreement
- 3 Aug 23 Entry into a Material Definitive Agreement
- 10 Jul 23 Eplontersen continued to show improvement in ATTRv-PN through 85 weeks
- 12 Jun 23 Ionis prices private placement of convertible senior notes
- 6 Jun 23 Ionis announces proposed private placement
- 5 Jun 23 Submission of Matters to a Vote of Security Holders
Filing view
External links
Exhibit 99.2

Eplontersen: NEURO-TTRansform Week 85 Topline Results July 10, 2023 Nasdaq: IONS

Forward-Looking Statements This presentation includes forward-looking statements regarding our business and the therapeutic and commercial potential of eplontersen, and Ionis' technologies. Any statement describing Ionis’ goals, expectations, financial or other projections, intentions or beliefs is a forward-looking statement and should be considered an at-risk statement. Such statements are subject to certain risks and uncertainties including but not limited to those related to our commercial products and the medicines in our pipeline, and particularly those inherent in the process of discovering, developing and commercializing medicines that are safe and effective for use as human therapeutics, and in the endeavor of building a business around such medicines. Ionis’ forward-looking statements also involve assumptions that, if they never materialize or prove correct, could cause its results to differ materially from those expressed or implied by such forward-looking statements. Although Ionis’ forward-looking statements reflect the good faith judgment of its management, these statements are based only on facts and factors currently known by Ionis. As a result, you are cautioned not to rely on these forward-looking statements. These and other risks concerning Ionis' programs are described in additional detail in Ionis' annual report on our Form 10-K for the year ended December 31, 2022, and our most recent Form 10-Q quarterly filing, which are on file with the SEC. Copies of these and other documents are available at www.ionispharma.com. In this presentation, unless the context requires otherwise, “Ionis,” “Company,” “we,” “our,” and “us” refers to Ionis Pharmaceuticals and its subsidiaries. Ionis Pharmaceuticals® is a registered trademark of Ionis Pharmaceuticals, Inc. 2
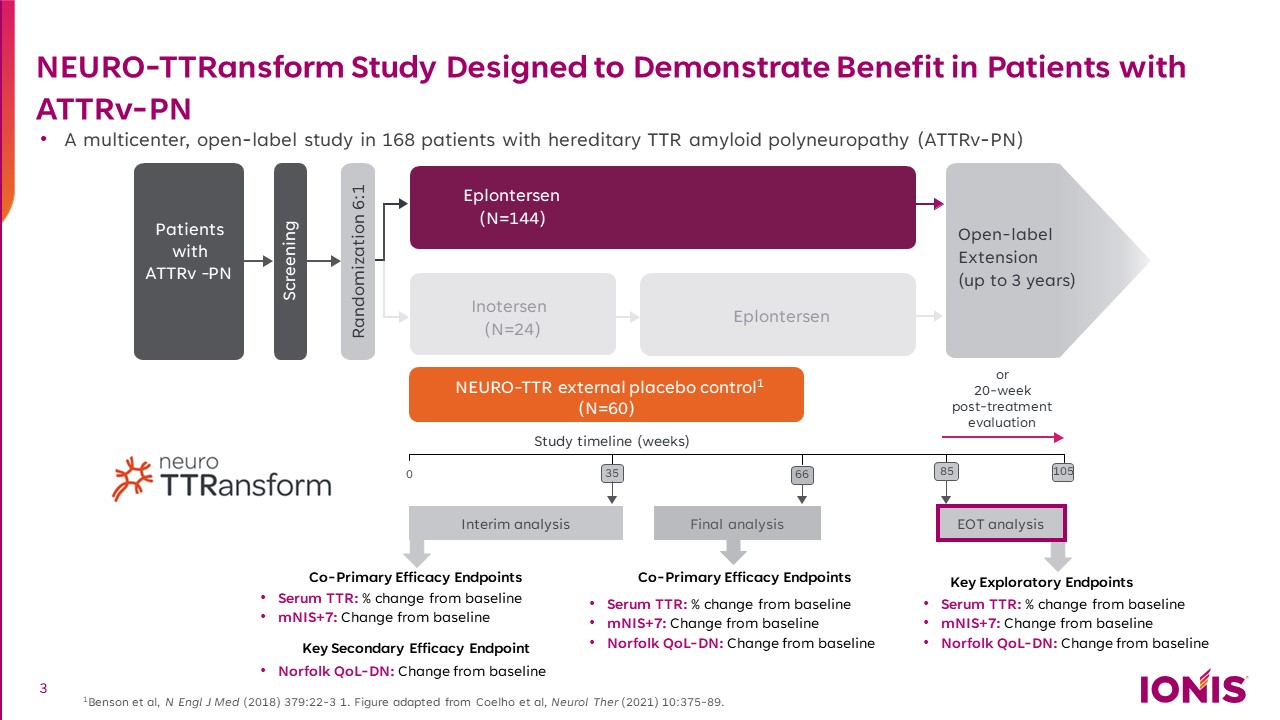
NEURO-TTRansform Study Designed to Demonstrate Benefit in Patients with ATTRv-PN A multicenter, open-label study in 168 patients with hereditary TTR amyloid polyneuropathy (ATTRv-PN) 1Benson et al, N Engl J Med (2018) 379:22-3 1. Figure adapted from Coelho et al, Neurol Ther (2021) 10:375-89. Randomization 6:1 0 Study timeline (weeks) 35 Interim analysis 85 EOT analysis Patients with ATTRv - PN Screening Eplontersen (N=144) NEURO-TTR external placebo control1 (N=60) Inotersen (N=24) Eplontersen Final analysis 105 Open-label Extension (up to 3 years) Co-Primary Efficacy Endpoints or 20-week post-treatment evaluation Serum TTR: % change from baseline mNIS+7: Change from baseline Norfolk QoL-DN: Change from baseline Co-Primary Efficacy Endpoints Serum TTR: % change from baseline mNIS+7: Change from baseline Key Secondary Efficacy Endpoint Norfolk QoL-DN: Change from baseline Key Exploratory Endpoints Serum TTR: % change from baseline mNIS+7: Change from baseline Norfolk QoL-DN: Change from baseline 66 66 3
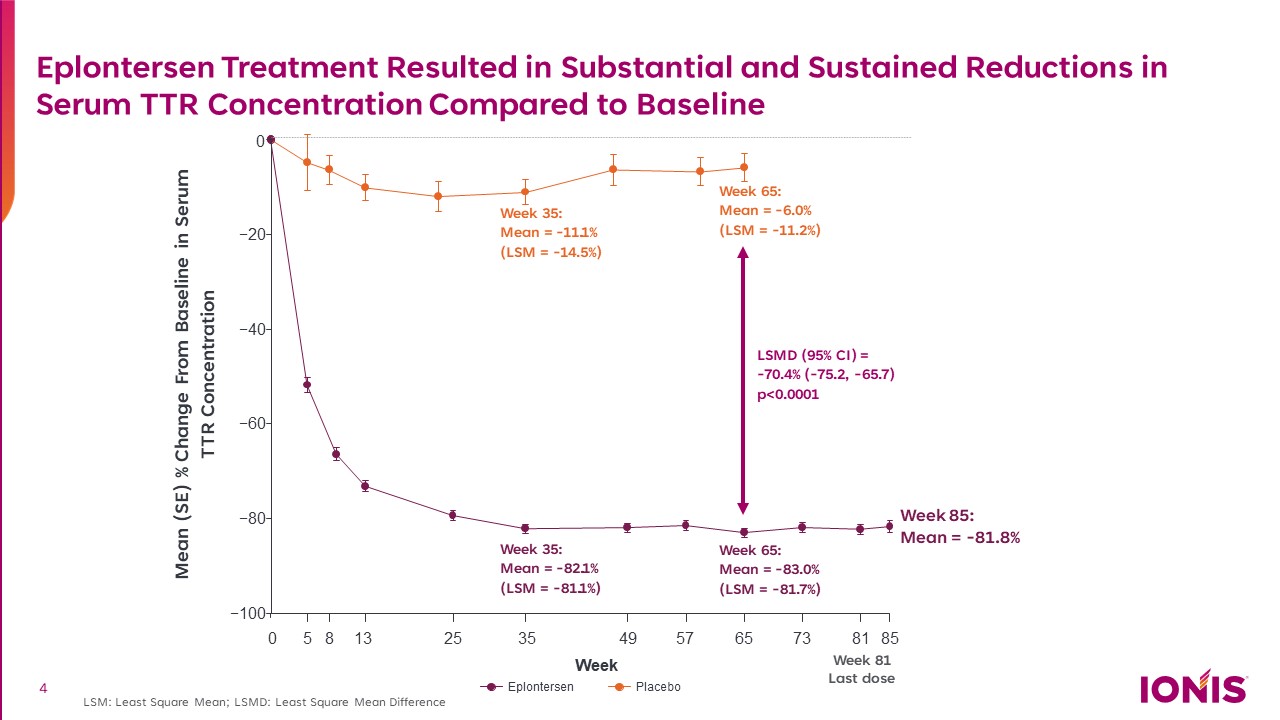
Eplontersen Treatment Resulted in Substantial and Sustained Reductions in Serum TTR Concentration Compared to Baseline LSM: Least Square Mean; LSMD: Least Square Mean Difference Week 35:Mean = -11.1% (LSM = -14.5%) Week 81Last dose Mean (SE) % Change From Baseline in Serum TTR Concentration Week 35:Mean = -82.1% (LSM = -81.1%) Week 65:Mean = -83.0% (LSM = -81.7%) Week 65:Mean = -6.0% (LSM = -11.2%) Week 85:Mean = -81.8% LSMD (95% CI) = -70.4% (-75.2, -65.7) p<0.0001 −100 −80 −60 −40 −20 0 0 5 8 13 25 35 49 57 65 73 81 85 W eek Eplontersen Placebo 4

Eplontersen Continued to Halt Neuropathy Progression at Week 85 Substantial number of patients showed improvement in neuropathy impairment through 19 months of treatment mNIS+7 Composite Score Worse Better Mean (SE) Change From Baseline in mNIS+7 Composite Score Week 35:Mean = 9.8 (LSM = 10.0) Week 35:Mean = -0.04 (LSM = 0.7) Week 66:Mean = 23.9 (LSM = 25.1) Week 66:Mean = -0.2 (LSM = 0.3) Week 85:Mean = -2.9 LSM: Least Square Mean; LSMD: Least Square Mean Difference LSMD (95% CI) = -24.8 (-31.0, -18.6) p<0.0001 5
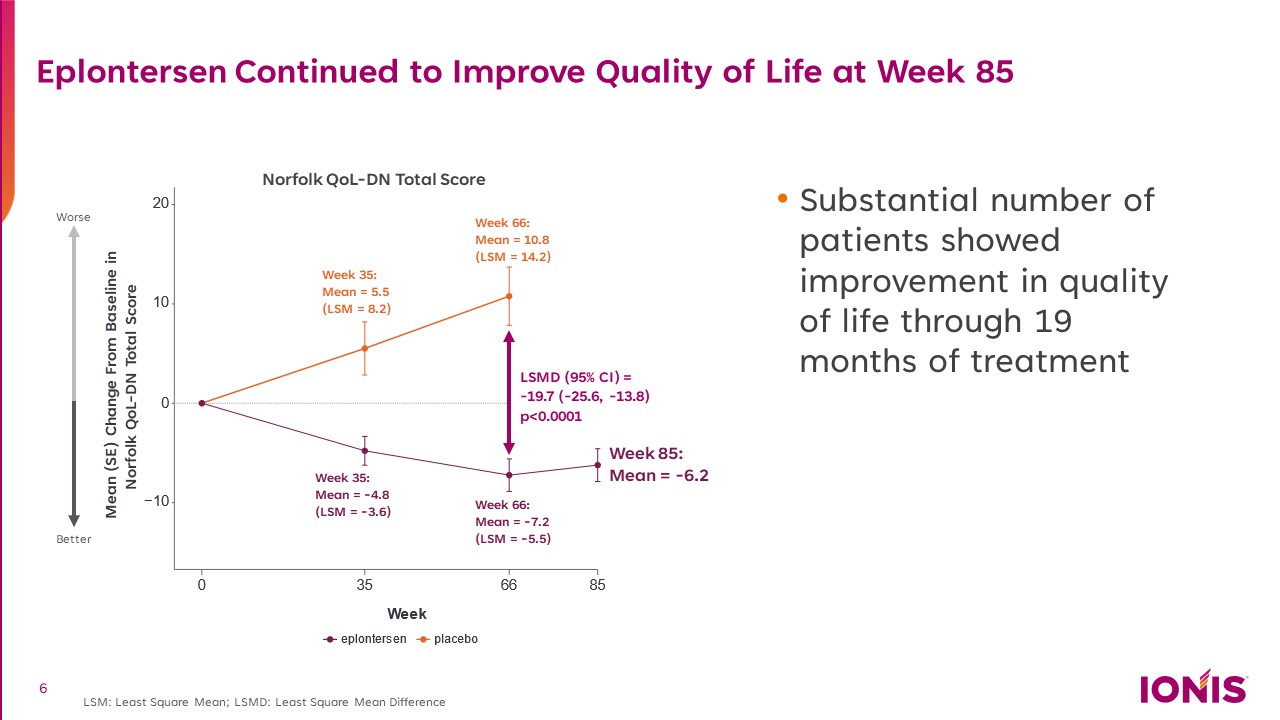
Eplontersen Continued to Improve Quality of Life at Week 85 Norfolk QoL-DN Total Score Worse Better Mean (SE) Change From Baseline in Norfolk QoL-DN Total Score Week 35:Mean = 5.5 (LSM = 8.2) Week 35:Mean = -4.8 (LSM = -3.6) Week 66:Mean = 10.8 (LSM = 14.2) Week 66:Mean = -7.2 (LSM = -5.5) Week 85:Mean = -6.2 Substantial number of patients showed improvement in quality of life through 19 months of treatment LSM: Least Square Mean; LSMD: Least Square Mean Difference LSMD (95% CI) = -19.7 (-25.6, -13.8) p<0.0001 6
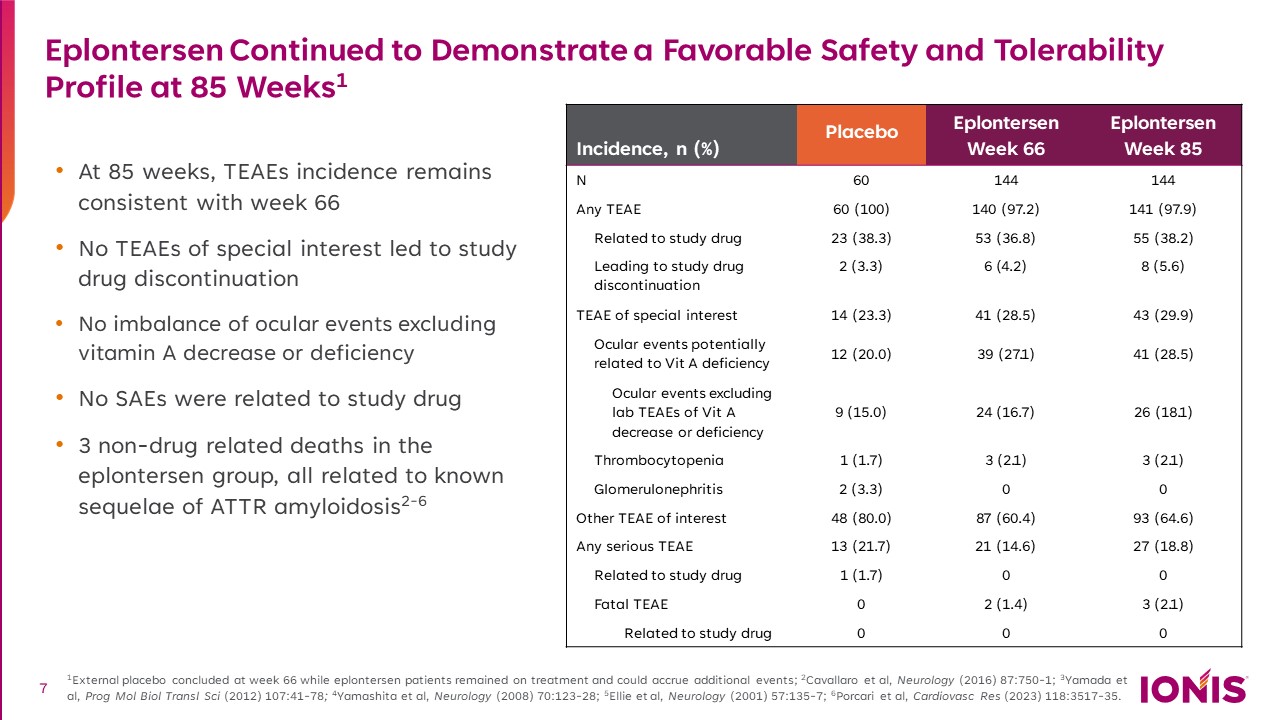
Eplontersen Continued to Demonstrate a Favorable Safety and Tolerability Profile at 85 Weeks1 At 85 weeks, TEAEs incidence remains consistent with week 66 No TEAEs of special interest led to study drug discontinuation No imbalance of ocular events excluding vitamin A decrease or deficiency No SAEs were related to study drug 3 non-drug related deaths in the eplontersen group, all related to known sequelae of ATTR amyloidosis2-6 1External placebo concluded at week 66 while eplontersen patients remained on treatment and could accrue additional events; 2Cavallaro et al, Neurology (2016) 87:750-1; 3Yamada et al, Prog Mol Biol Transl Sci (2012) 107:41-78; 4Yamashita et al, Neurology (2008) 70:123-28; 5Ellie et al, Neurology (2001) 57:135-7; 6Porcari et al, Cardiovasc Res (2023) 118:3517-35. Incidence, n (%) Placebo Eplontersen Week 66 Eplontersen Week 85 N 60 144 144 Any TEAE 60 (100) 140 (97.2) 141 (97.9) Related to study drug 23 (38.3) 53 (36.8) 55 (38.2) Leading to study drug discontinuation 2 (3.3) 6 (4.2) 8 (5.6) TEAE of special interest 14 (23.3) 41 (28.5) 43 (29.9) Ocular events potentially related to Vit A deficiency 12 (20.0) 39 (27.1) 41 (28.5) Ocular events excluding lab TEAEs of Vit A decrease or deficiency 9 (15.0) 24 (16.7) 26 (18.1) Thrombocytopenia 1 (1.7) 3 (2.1) 3 (2.1) Glomerulonephritis 2 (3.3) 0 0 Other TEAE of interest 48 (80.0) 87 (60.4) 93 (64.6) Any serious TEAE 13 (21.7) 21 (14.6) 27 (18.8) Related to study drug 1 (1.7) 0 0 Fatal TEAE 0 2 (1.4) 3 (2.1) Related to study drug 0 0 0 7
