 Global Strategies for Life-Enhancing Drugs Corporate Presentation February 9, 2009 Exhibit 99.2 |
 2 Safe Harbor Statement This presentation contains forward-looking statements regarding the business, products and financial results of SciClone Pharmaceuticals, Inc. You are urged to consider statements that include the words “may,” “will,” “would,” “could,” “should,” “might,” “believes,” “estimates,” “projects,” “potential,” “expects,” “plans,” “anticipates,” “intends,” “continues,” “forecast,” “designed,” “goal,” or the negative of those words or other comparable words to be uncertain and forward-looking. These statements are subject to risks and uncertainties that are difficult to predict and actual outcomes may differ materially. These risks and uncertainties include changes to our final fiscal year 2008 results that may result in the course of completion of our audit, developments with respect to our products as well as the outcome our audit and of the director nomination notice received by SciClone in December 2008 from a company controlled by Sigma-Tau Finanziaria S.p.A. Please also refer to other risks and uncertainties described in SciClone’s filings with the Securities and Exchange Commission. All forward-looking statements are based on information currently available to SciClone and SciClone assumes no obligation to update any such forward-looking statements. |
 3 Important Information/ Solicitation Participants Legend SciClone Pharmaceuticals, Inc. will file a proxy statement in connection with its 2009 annual meeting of stockholders and advises its stockholders to read that proxy statement when it becomes available because it will contain important information. Stockholders will be able to obtain a free copy of that proxy statement and other documents (when available) that SciClone files with the Securities and Exchange Commission at the Commission's website at www.sec.gov. That proxy statement and these other documents will also be available free of charge by directing a request to SciClone Pharmaceuticals, Inc., Attn: Investor Relations, 950 Tower Lane, Suite 900, Foster City, CA 94404, or from SciClone at www.sciclone.com. SciClone, its directors and named executive officers may be deemed to be participants in the solicitation of proxies from the SciClone stockholders in connection with the 2009 annual meeting. Stockholders may obtain information regarding the names, affiliations and interests of such individuals in SciClone's proxy statement filed on May 5, 2008 for the 2008 annual meeting of stockholders. To the extent that holdings of SciClone securities on the part of those individuals have changed since the date of that proxy statement, those changes have been reflected on Statements of Changes in Ownership on Forms 3 or 4 filed with the Securities and Exchange Commission. More current information regarding the interests of the directors and named executive officers of SciClone will be contained in the proxy statement referred to in the preceding paragraph. |
 4 Our Company Today Company Overview • Investment Opportunity • Pipeline • Strategy and Business Structure Product Overview • ZADAXIN ® • DC Bead • RP101 • SCV-07 Looking Forward • Milestones • Investment Opportunity TM |
 Company Overview |
 6 Investment Opportunity • Profitability-Driven Enterprise • Solid global sales growth drives sustainable profitability • 2008 revenues: $54 M (unaudited); 46% increase* • 2009 guidance: $60-$62 M revenues; $0.00 - $0.02 EPS • Revenue growth + tight expense management = positive cash flow and EPS • International Commercial Success • ZADAXIN marketed in more than 30 countries globally • Established business in China with >160-person sales organization • Strong international brand recognition • Consistent double-digit revenue growth • Rich Product Portfolio in Oncology and Infectious Disease • Invest in cost-efficient phase 1/2 trials; partner phase 3 trials • 1 marketed product; developing 3 molecules in 4 indications • Development programs with strong IP positions Note: All 2008 unaudited financial results included in this report are based on our current expectations and are preliminary unaudited results. |
   7 Three-Pillared Corporate Strategy Profitability-Driven Enterprise International Commercial Success Focused Development Approach SciClone |
     8 Three-Pillared Corporate Strategy • Created strong ZADAXIN brand in China during >10 years, reputation for quality • >$50 M revenues (unaudited) in China • Large, productive, educated China sales organization • Expected to launch DC Bead in 2009 • Emerging markets generate revenue; bolster long-term growth • Russia and Vietnam International Commercial Success SciClone |
     9 Three-Pillared Corporate Strategy Focused Product Development SciClone • Three phase 2/3 compounds in melanoma, pancreatic cancer, oral mucositis, and hepatitis C • All addressing serious unmet medical needs • Strong IP positions • Valuable partnering opportunities |
     10 Three-Pillared Corporate Strategy • Solid global sales growth drives sustainable profitability • $54 M 2008 revenue (unaudited) • 2009 guidance: $60-$62 M revenues; $0.00-$0.02 EPS • Sound cash position of >$27 M (unaudited) 12/31/08 • $6 M debt facility SciClone Profitability-Driven Enterprise |
   11 Three-Pillared Corporate Strategy Profitability-Driven Enterprise International Commercial Success Focused Development Approach SciClone |
 Business Update |
 13 Growth Opportunity: SciClone in China Our Position • Longstanding presence, ZADAXIN launched in 1996 • SPIL China 2008 sales >$50 million** (unaudited) • Sales: Offices in Beijing, Shanghai, Guangzhou, Hong Kong • >160-strong sales organization covers > 500 hospitals • Highly trained, highly productive, includes 40 MDs Our Opportunity • Markets: Large hepatitis B/C, cancer populations in China • SciClone and ZADAXIN well-regarded by physician community • EU manufacturing brings Western quality “seal of approval” • 1 class brand identity • Strong sales infrastructure will support new product introduction Our Growth Strategy • Expanding to tier 2/3 cities: opportunity to reach more patients • Launch DC Bead 2009 • Introducing additional products through licensing and M&A * McKinsey, 2007 ** In 2007, 80% of multinational pharmaceutical companies’ individual products reached <$10 M peak sales in China (McKinsey, 2007) China is expected to rank among the top five global pharmaceutical markets by 2012* st |
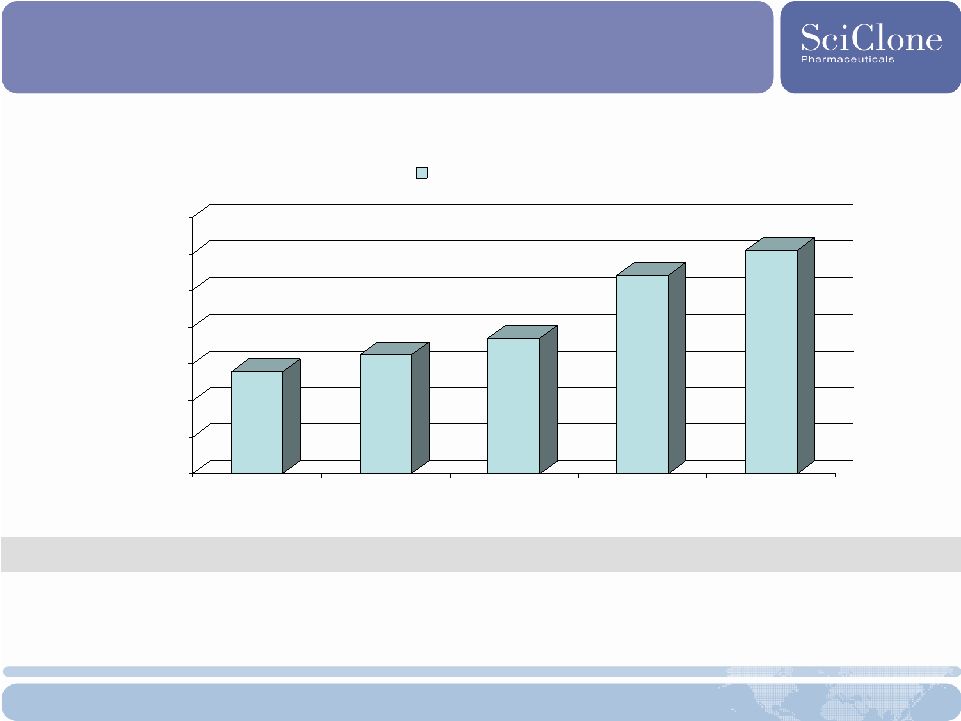 14 Revenues and EPS $.0 $10.0 $20.0 $30.0 $40.0 $50.0 $60.0 $70.0 2005 2006 2007 2008 2009 E Revenue ($M) SciClone Consolidated; 2008 revenues - unaudited * 2006 EPS do not include $8M settlement received that year 2005-2008 CAGR: 25% EPS: ($.17) ($.16)* ($.22) TBA $0.00 - $0.02 |
 Product Overview |
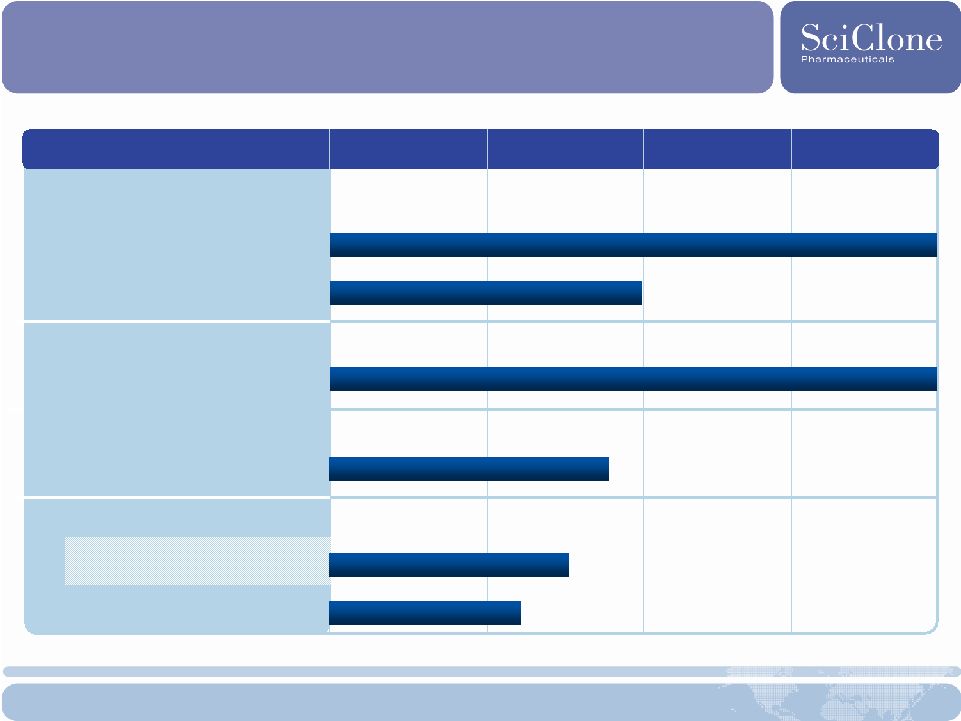          16 Product Portfolio Product Development Phase 1 Phase 2 Phase 3 Marketed ZADAXIN ® HBV/HCV & Other Indications China, Russia & >30 other countries Malignant Melanoma U.S./Europe RP101 Pancreatic Cancer U.S. & Canada DC Bead ™ China Marketing approval expected 2009 Oral Mucositis in Head & Neck Cancer SCV-07 HCV Global (Ex-Russia) Global (Ex-Russia) |
 17 Product Portfolio |
 18 ZADAXIN (thymalfasin): Immunomodulator for Malignant Melanoma Description • Synthetic thymosin alpha 1 (peptide produced naturally by thymus gland) • Stimulates multiple innate and adaptive immune and direct antiviral responses Commercial Opportunity • $200M worldwide market for Stage IV melanoma; Much larger market potential in adjuvant setting • U.S. Orphan Drug designation • Unique MOA suggests benefits as additive or combination therapy Development Strategy • Stage IV Melanoma phase 2 overall survival in normal LDH* subgroup ( 1.2*ULN**) extended 4.2 months • Stage IV Melanoma phase 2 progression free survival in normal LDH subgroup ( 1.2 *ULN**) increased from 1.9 months to 3.0 months (average of all thymalfasin arms) in a dose response manner • Stellar safety profile Development Status • SPA for phase 3 program approved by FDA • Actively exploring partnering opportunities * The level of LDH is a strong prognostic factor for survival; ** ULN: Upper Level of Normal |
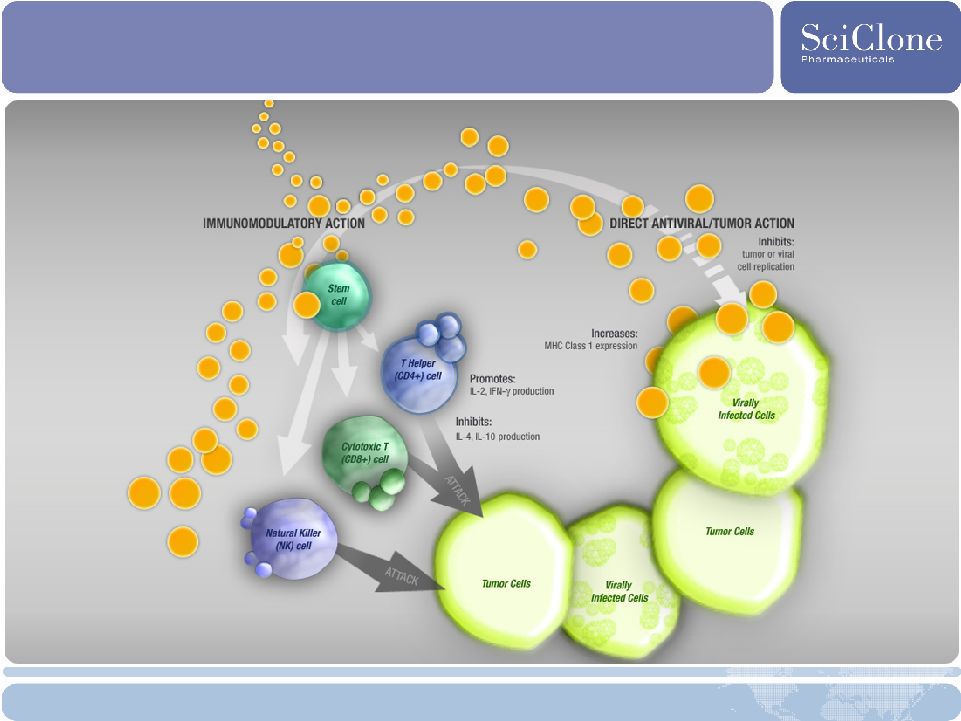 19 Thymalfasin’s Dual Mechanism of Action |
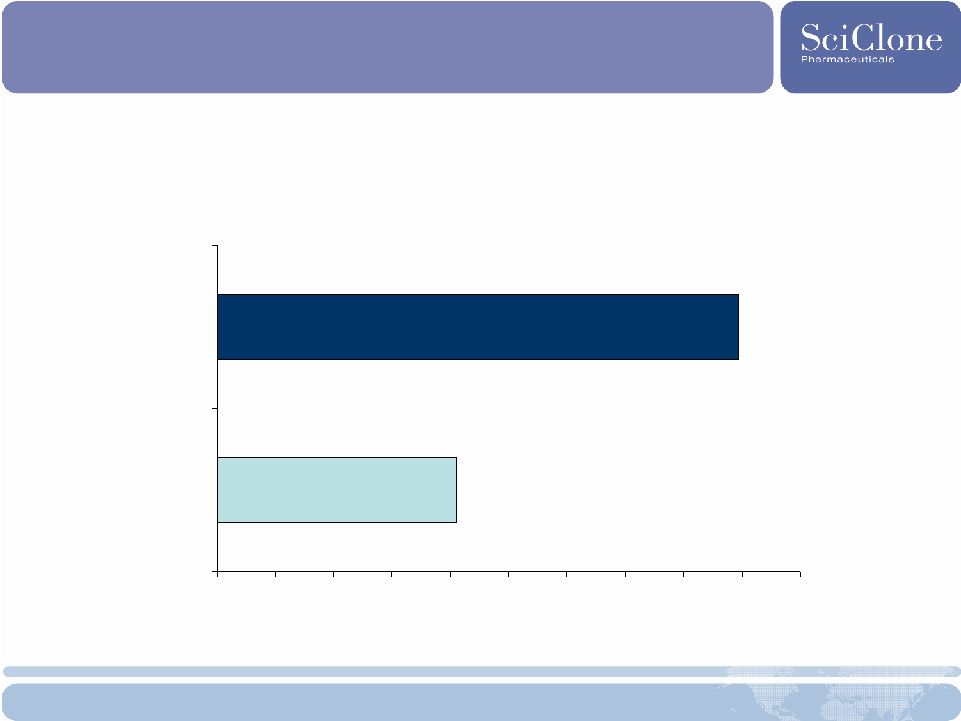 Positive Phase 2 Tumor Response Source: SciClone Pharmaceuticals and Sigma-Tau, ASCO, June 2007. Primary endpoint: Overall Tumor Response (Complete Response + Partial Response) 4.1% 9.0% 0% 1% 2% 3% 4% 5% 6% 7% 8% 9% 10% DTIC + IFN (Control) Total Thymalfasin |
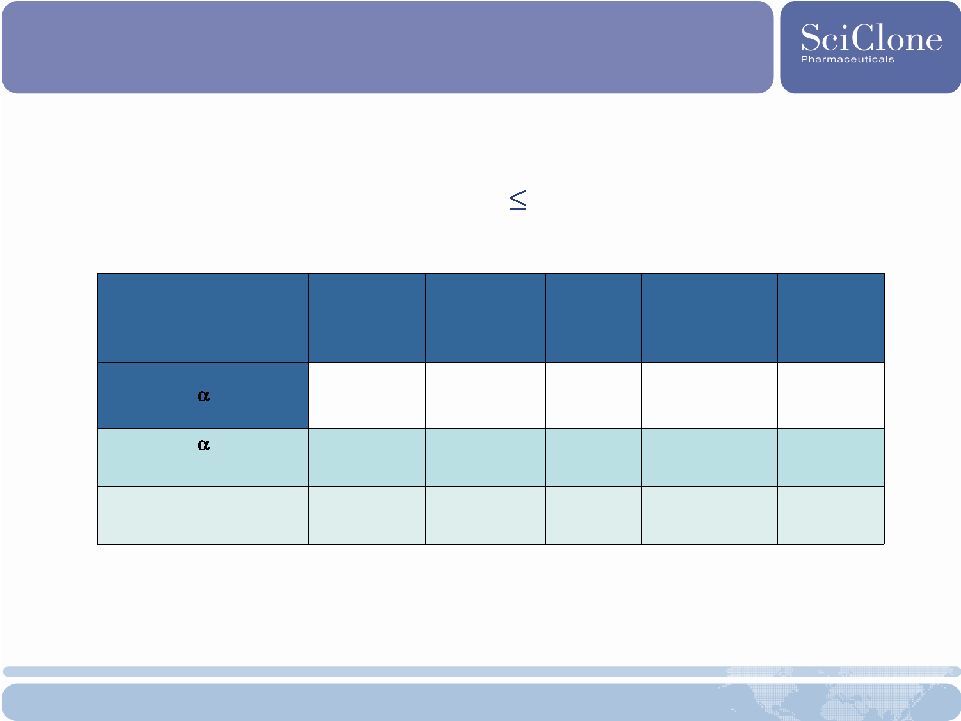 Thymalfasin Melanoma: Survival Data 21 Phase 2 Results Subset Analysis of LDH 1.2 ULN Patients n= Median Survival P= Median Progression Free Survival P= DTIC + IFN (control) 68 9.4 months 1.9 months DTIC + IFN thymalfasin 6.4mg 67 12.7 months 0.049 2.6 months 0.050 All thymalfasin arms 273 12.6 months 0.034 2.6 months 0.001 |
 22 DC Bead: Targeting Liver Cancer in China Description • Embolic agent, delivers chemotherapy drugs • Microscopic beads with dual anti-tumor effects • Deliver doxorubicin, potentially other chemotherapy agents directly to tumor Commercial Opportunity • Market-specific unmet need; Liver cancer one of most prevalent and deadly cancers in China • 271,300 diagnosed population; 64,530 drug-treated population • Leverage existing brand image, marketing and distribution channels, and strong in-country sales expertise Development Status • Exclusive Chinese marketing rights acquired in 2006 • Product marketed by other companies in multiple regions including EU, Canada, South Africa, India Future Plans • Expect SFDA marketing approval in 2009 • Launch sales and marketing efforts •Joint trial with drug Nexavar ® sponsored by Bayer |
 23 RP101: Phase 2 Chemotherapy Enhancer Description • Chemosensitizing agent: prevents tumor resistance to chemotherapy; orally delivered Commercial Opportunity • $350 M* market opportunity as an additive pancreatic cancer therapy • 37,680 new cases, 34,290 deaths in U.S. in 2008 • Orphan drug designation in the U.S. • >3 month survival benefit over standard of care in early studies • Tarceva ® approved with 11 day benefit • Biological activity shown in multiple solid tumor types Development Strategy • Deliver extended survival benefit to patients with Stage III and IV pancreatic cancer Development Status • Ongoing phase 2 trial in late stage patients (began in Q108) • Multi-center, double-blinded, randomized, placebo-controlled (n=153) Future Plans • Complete phase 2 trial, present data at major conferences • Obtain SPA for pivotal phase 3 trial *Source: Decision Resources |
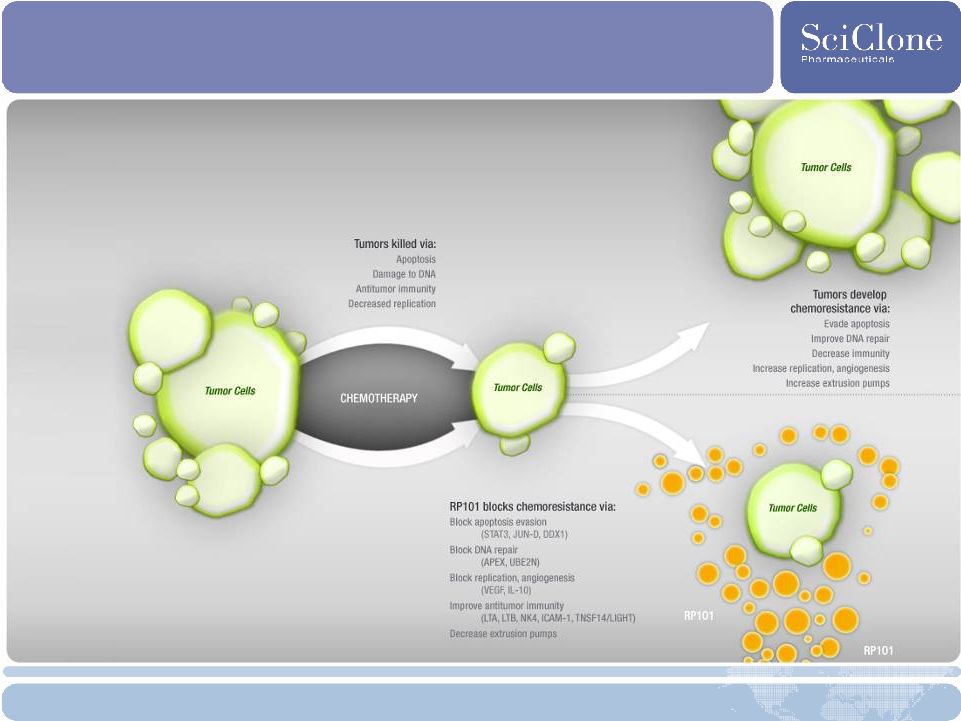 RP101: Blocks Resistance to Chemotherapy |
 25 SCV-07: Phase 2 Oral Mucositis and HCV Opportunities Description • Subcutaneously administered small molecule; also orally available • Stimulates immune system, increases T-helper cells (Th1 response specifically) Commercial Opportunity •Oral Mucositis: Vast unmet medical need with hundreds of thousands patients worldwide affected and largely untreated • Currently approved in Russia to treat depressed immune systems • SCLN holds exclusive worldwide rights ex-Russia • Demonstrated efficacy in multiple indications, including viral and bacterial infections, chemoradiation-induced mucositis, multiple cancers • HCV: Large unmet medical need; HCV market worth more than $2 B* in 2008 and expected to more than double with the launch of new therapies Development Strategy • Oral Mucositis: Protect normal cells following chemoradiotherapy for head and neck cancer •HCV: Safe, well-tolerated, no dose-limiting effects • Biological signal observed after 7 days of monotherapy Future Development • Oral Mucositis: Enrollment in phase 2 study to delay onset of severe oral mucositis and decrease its severity began in late 2008 • HCV: Phase 2b trial of SCV-07 with ribavirin in patients with genotype 1 chronic HCV who relapsed to standard of care * Source: Decision Resources, Data Monitor |
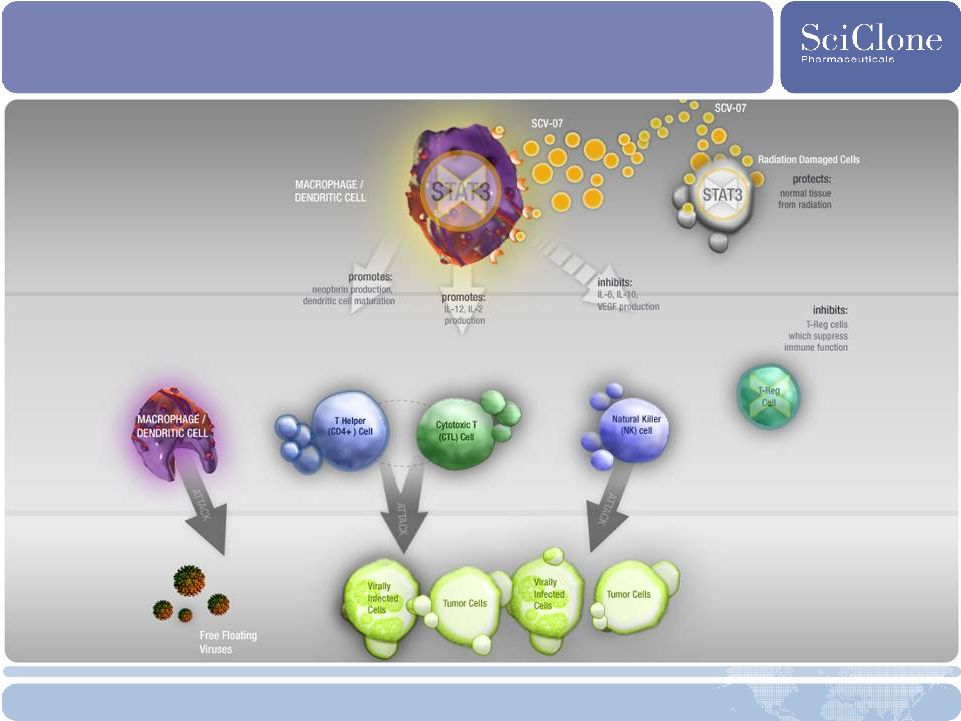 SCV-07: Multiple Immune Modulating Effects |
 27 Product Portfolio |
 What’s Ahead |
 29 Upcoming Milestones Ongoing Acquire additional product for Chinese market 1H10 RP101 Phase 2 unblinding 2008 guidance: $52-$54 revenues; YE cash >$20 M 2009 Launch DC Bead – China 2009 Phase 3 Thymalfasin Malignant Melanoma Trial; Ongoing partnering discussions 1H09 Complete enrollment for RP101 phase 2 pancreatic cancer trial YE 2009 Complete enrollement for SCV-07 in oral mucositis 2009 2009 guidance: $60-$62 M; $0.00-$0.02 EPS Milestone Timeline |
 30 Selected Consolidated Financials FY Ended 12/31 (US$M) 2008 Guidance 2009 Cash & Equivalents >$27 (Unaudited) >$20 46% % Change $60-$62 Guidance 2009 Revenues FY Ended 12/31 (US$M) 2008 Revenues Guidance EPS 2009 $54 (Unaudited) $0.00 - $0.02 |
 31 Investment Opportunity • Profitability-Driven Enterprise • Solid global sales growth drives sustainable profitability • 2008 revenues: $54 M (unaudited); 46% increase • 2009 guidance: $60-$62 M revenues; $0.00 - $0.02 EPS • Revenue growth + tight expense management = positive cash flow and EPS • International Commercial Success • ZADAXIN marketed in more than 30 countries globally • Established business in China with >160-person sales organization • Strong international brand recognition • Consistent double-digit revenue growth • Rich Product Portfolio in Oncology and Infectious Disease • Invest in cost-efficient phase 1/2 trials; partner phase 3 trials • 1 marketed product; developing 3 molecules in 4 indications • Development programs with strong IP positions |
 Global Strategies to Enhance Health Corporate Presentation February 9, 2009 |
 33 Senior Management Team • Friedhelm Blobel, Ph.D., President and CEO • Gryphon Therapeutics, Hoechst Group, Boehringer Mannheim Group • Gary Titus, Chief Financial Officer • Kosan Biosciences, Nuvelo, Metabolex, IntraBiotics Pharmaceuticals, and J&J’s LifeScan Division • Hans P. Schmid, President and Managing Director of SciClone Pharmaceuticals International, Ltd. • Questcor Pharmaceuticals, Oread, Syntex, Roche • Israel Rios, M.D., Chief Medical Officer • Dendreon Corporation, Berlex Laboratories, Marion Merrell Dow, Inc., Bristol- Myers • Cynthia W. Tuthill, Ph.D., Chief Scientific Officer • Becton Dickinson Immunocytometry Systems, Scios, Inc. • Craig A. Halverson, Vice President, Regulatory Affairs and Quality Assurance • Zymogenetics, Introgen Therapeutics, CancerVax Therapeutics, Gen-Probe, Baxter Healthcare • Jeffery Lange, Vice President, Business Development • Novartis; variety of positions in business development and strategic planning |