Nevada Lawsuit Against Genentech/Roche
• PDL filed a lawsuit against Genentech and Roche in Nevada state court
▪ Lawsuit states that fax constitutes a breach of 2003 Settlement Agreement because Genentech assisted
Roche in challenging PDL’s patents and SPCs
▪ Complaint seeks compensatory damages, including liquidated damages and other monetary remedies set
forth in the 2003 Settlement Agreement, punitive damages and attorney’s fees
• In November 2010, Genentech and Roche filed two motions to dismiss
▪ They contend that 2003 Settlement Agreement applies only to PDL’s U.S. patents
▪ They asserted that the Nevada court lacks personal jurisdiction over Roche
• On July 11, 2011, court denied Genentech and Roche's motion to dismiss four of PDL's
five claims for relief and denied Roche's separate motion to dismiss for lack of personal
jurisdiction.
▪ The court dismissed one of PDL's claims that Genentech committed a bad-faith breach of the covenant of
good faith and fair dealing
▪ Subsequent to the ruling, Roche has waived its defense that the Nevada court lacks personal jurisdiction for
the purposes of this lawsuit
• The court ruling allows PDL to continue to pursue its claims that:
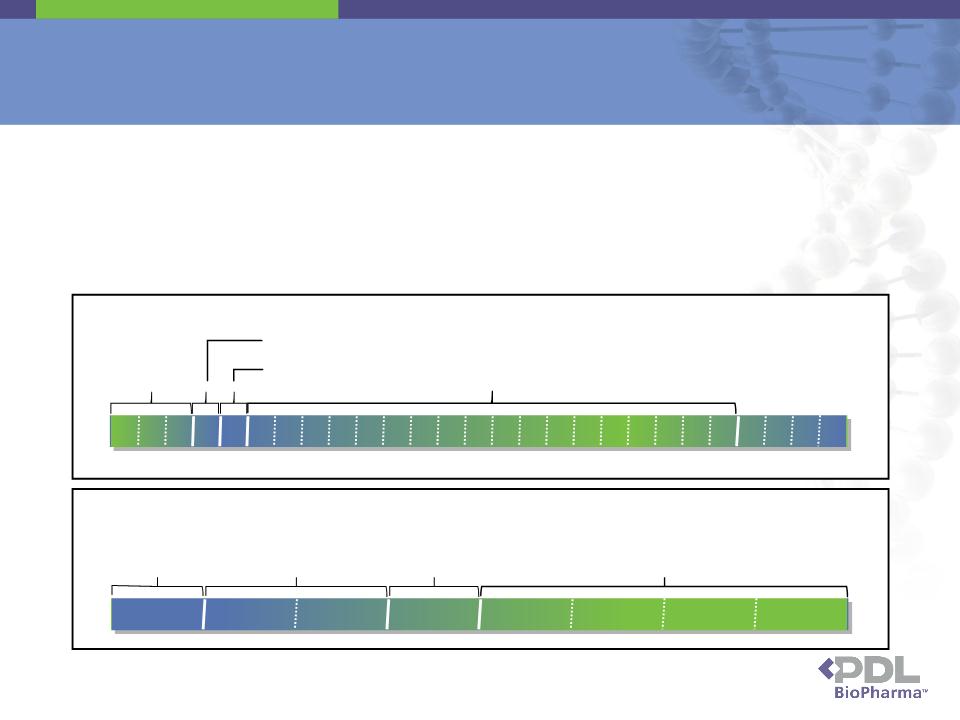
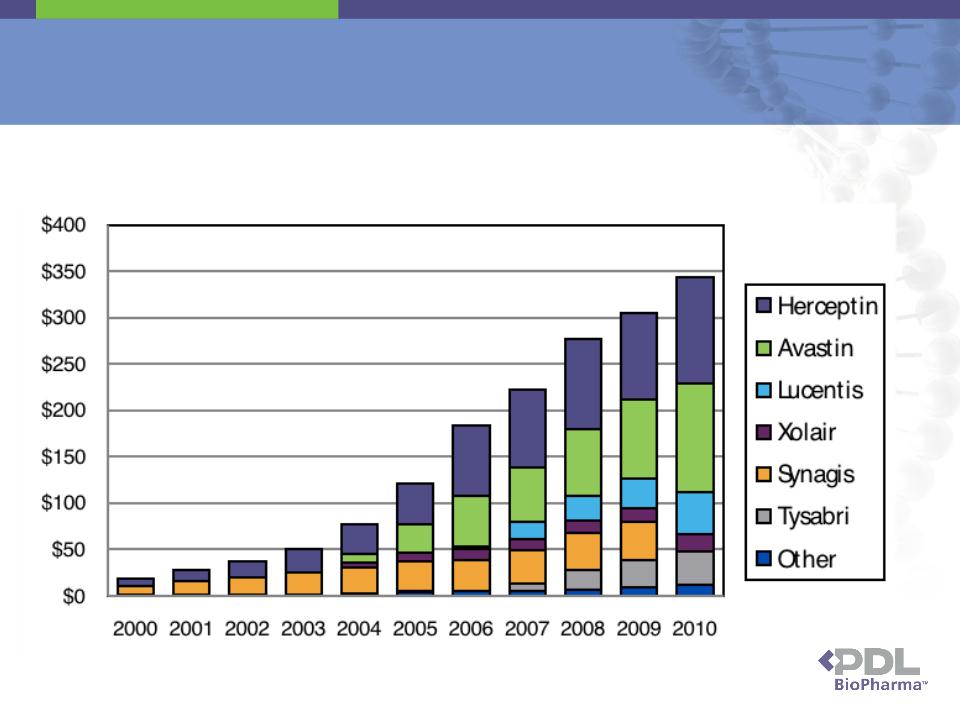
▪ Genentech is obligated to pay royalties to PDL on international sales of the Genentech Products
▪ Genentech, by challenging, at the behest of Roche and Novartis, whether PDL's SPCs cover the Genentech
Products breached its contractual obligations to PDL under the 2003 settlement agreement
▪ Genentech breached the implied covenant of good faith and fair dealing with respect to the 2003 settlement
agreement
▪ Roche intentionally and knowingly interfered with PDL's contractual relationship with Genentech in
conscious disregard of PDL's rights
▪ Parties are currently in discovery
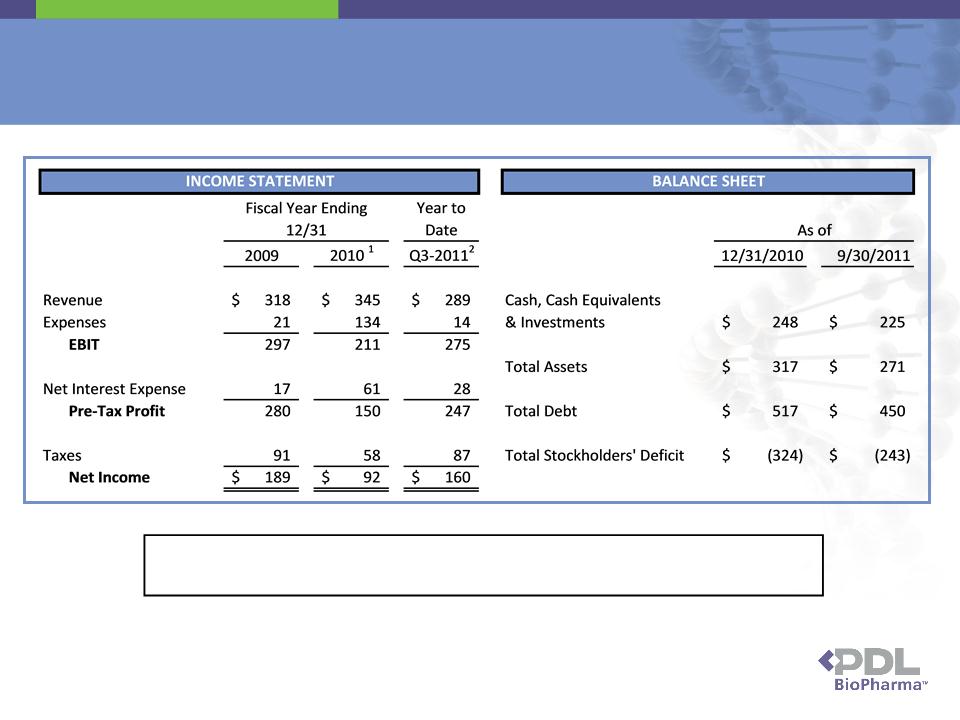
40