
Investor Presentation August 2016 Exhibit 99.1

Important Information and Where to Find It This communication does not constitute an offer to sell or the solicitation of an offer to buy any securities or a solicitation of any vote or approval. A definitive proxy statement and a proxy card will be filed with the SEC and will be mailed to StemCells stockholders seeking any required stockholder approvals in connection with the proposed transactions. BEFORE MAKING ANY VOTING OR INVESTMENT DECISION, INVESTORS AND STOCKHOLDERS ARE URGED TO READ THE PROXY STATEMENT (INCLUDING ANY AMENDMENTS OR SUPPLEMENTS THERETO) AND ANY OTHER RELEVANT DOCUMENTS THAT STEMCELLS MAY FILE WITH THE SEC WHEN THEY BECOME AVAILABLE BECAUSE THEY WILL CONTAIN IMPORTANT INFORMATION ABOUT THE PROPOSED TRANSACTIONS. Stockholders may obtain, free of charge, copies of the definitive proxy statement and any other documents filed by StemCells with the SEC in connection with the proposed transactions at the SEC’s website (http://www.sec.gov), at StemCells’ website, or by directing written request to: StemCells, Inc. 39899 Balentine Drive, Suite 200, Newark, CA 94560, Attention: Kenneth Stratton, Esq. StemCells and its directors and executive officers and Microbot and its directors and executive officers may be deemed to be participants in the solicitation of proxies from the stockholders of StemCells in connection with the proposed transaction. Information regarding the special interests of these directors and executive officers in the merger will be included in the proxy statement referred to above. Additional information regarding the directors and executive officers of StemCells is also included in the StemCells Definitive Proxy Statement on Schedule 14A relating to the 2016 Annual Meeting of Stockholders, which was filed with the SEC on April 8, 2016. This document is available free of charge at the SEC web site (www.sec.gov), at the StemCells website, or by directing a written request to StemCells as described above. This document contains summary information about Microbot, does not purport to be complete, and no representations or warranties about such information are made by Microbot or its representatives. This document does not constitute an offer to sell or a solicitation of an offer to purchase any securities of Microbot. This document contains forward-looking statements within the meaning of Section 27A of the Securities Act of 1933, as amended, and Section 21E of the Securities Exchange Act of 1934, as amended, relating to future events or the future financial performance and operations of Microbot. Forward-looking statements, which involve assumptions and describe Microbot’s intent, belief or current expectations about its business opportunities, prospects, performance and results, are generally identifiable by use of the words “may,” “could,” “should,” “will,” “would,” “expect,” “anticipate,” “plan,” “potential,” “estimate,” “believe,” “intend,” “project,” “forecast,” the negative of such words and other variations on such words or similar terminology. These forward-looking statements are not guarantees of future performance and by their nature involve known and unknown risks and uncertainties that may cause actual opportunities, prospects, performance and results to vary from those presented in this document, and those variances may be material. In evaluating such statements, prospective investors should carefully consider the various risks and uncertainties, such as market risk, liquidity risk, competitive risk, regulatory risk and other forms of risk. In light of these risks, uncertainties and assumptions, the forward-looking events discussed in this document might not occur. Microbot is not obligated to publicly update or revise any forward-looking statements, whether as a result of new information, future events or otherwise. Presentation Disclaimer

“The pain, discomfort, and disability, or other morbidity as a result of surgery is more frequently due to trauma involved in gaining access to the area to perform the intended procedure rather than from the procedure itself.” Medtech Insight on Minimal Invasive Surgery Soft tissue repair market (USA & EU), 2009 A well known problem
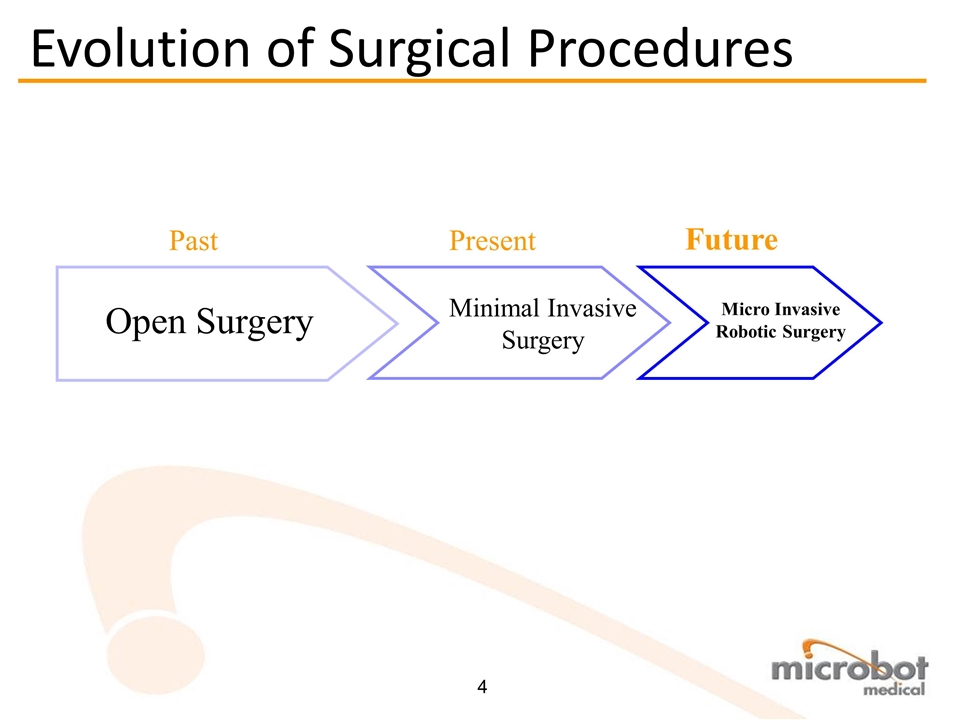
Business Confidential Open Surgery Past Minimal Invasive Surgery Present Micro Invasive Robotic Surgery Future Evolution of Surgical Procedures
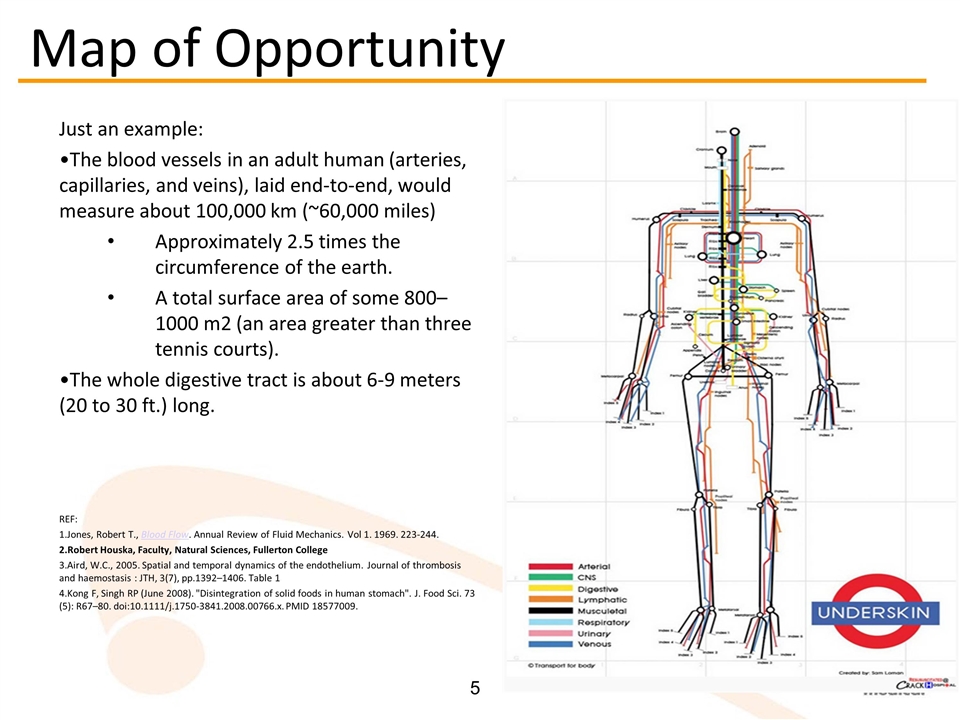
Just an example: The blood vessels in an adult human (arteries, capillaries, and veins), laid end-to-end, would measure about 100,000 km (~60,000 miles) Approximately 2.5 times the circumference of the earth. A total surface area of some 800–1000 m2 (an area greater than three tennis courts). The whole digestive tract is about 6-9 meters (20 to 30 ft.) long. REF: Jones, Robert T., Blood Flow. Annual Review of Fluid Mechanics. Vol 1. 1969. 223-244. Robert Houska, Faculty, Natural Sciences, Fullerton College Aird, W.C., 2005. Spatial and temporal dynamics of the endothelium. Journal of thrombosis and haemostasis : JTH, 3(7), pp.1392–1406. Table 1 Kong F, Singh RP (June 2008). "Disintegration of solid foods in human stomach". J. Food Sci. 73 (5): R67–80. doi:10.1111/j.1750-3841.2008.00766.x. PMID 18577009. Map of Opportunity
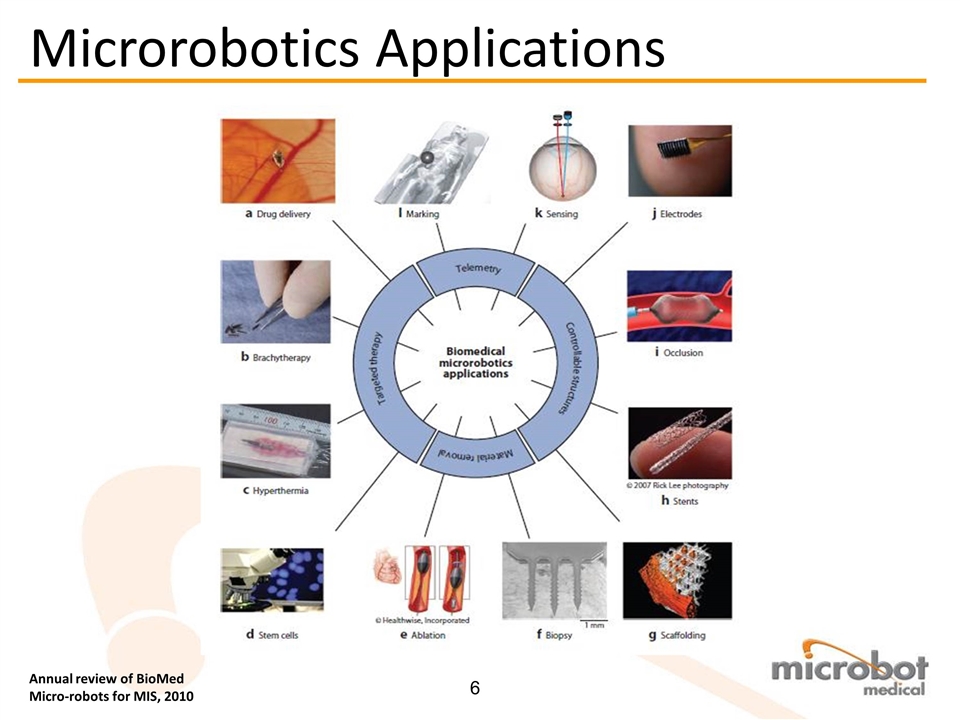
Annual review of BioMed Micro-robots for MIS, 2010 Microrobotics Applications

Business Confidential Minimally Invasive Surgery (MIS) is one of the largest trends in surgical healthcare Global market for MIS devices in 2012 was valued at USD 25.03 Billion and is expected to reach a market value of USD 50.60 Billion by 2019 The MIS market is expected to grow at a CAGR of 10.5% during the forecast period 2013 to 2019 Minimally invasive techniques have been applied to most of the surgical specialties (including cardiothoracic, orthopedic, urological, vascular and neurological procedures) Minimally invasive procedures are undergoing a transformation by becoming smaller, automated, more precise instruments Future of Medicine Source: Growing trend towards minimally invasive surgery – a potential market to exploit, Frost & Sullivan Market Insight, Nov 11, 2008 Source: ‘Global Minimally Invasive Surgery Market is Expected to Reach USD 50.60 Billion in 2019’, Transparency Market Research. http://www.transparencymarketresearch.com/minimally-invasive-surgery-market.html

Pioneer transformational micro robotic medical technologies, utilizing the natural and artificial lumens in the body to target the disease site with exquisite precision Our Vision

Prof. Moshe Shoham Scientific Director, Co-Founder Prof. Shoham is a worldwide acclaimed authority in the field of robotics. Prof. Shoham has been conducting research in the robotic field for over the past 20 years, with a special focus on kinematics and dynamics of robots, sensor integration, multi-finger hands and medical applications.. Founder of Mazor Surgical Technologies Ltd. (Nasdaq: MZOR) Foreign Member, US National Academy of Engineering Head of the robotics lab at Israel's Technion's Faculty of Mechanical Engineering. Formerly the director of the robotic laboratory of the Department of Mechanical Engineering, Columbia University, NY. Business Confidential Key Management Former Worldwide Group Marketing Director at Ethicon Inc., a multi-billion dollar division of Johnson & Johnson Company (NYSE: JNJ), responsible for global strategic marketing. Harel was with J&J for a decade between 2000-2010. Led business development activities and serves as a board member of ConTIPI Ltd., an early stage medical device company, which was acquired by Kimberly-Clark Corporation (NYSE: KMB), in 2012. Harel Gadot CEO & Chairman Mr. Sharon brings 23 years of R&D and general management in the medical devices space. Prior to joining Microbot Medical, Mr. Sharon managed the R&D at Icecure Medical, an early stage, public medical device company (TASE: ICCM) . Mr. Sharon was the General Manger of Anorad Israel, a subsidiary of Rockwell Automation which manufactures sub-micron precision motion systems. Mr. Sharon holds a B.Sc. from the Technion Institute of Technology, and an M.Sc in Mechanical engineering from MIT where he specialized in motion control and Robotics Ms. Stein, is a regulatory affairs, clinical and QA consultant with over 18 years of experience working directly with the FDA. Her regulatory experience includes all types of regulatory submissions for a wide variety of innovative medical devices. Ahava Stein Director, Regulatory Affairs Simon Sharon Chief Operating Officer

Business Confidential

Autonomous Advancing Micro-Robot (AAMR) ViRob demonstrates the ability to advance within cavities similar to the typical human body's lumens Including in liquid environment ViRob has the potential for: Minimally invasive medical procedures/treatments Targeted drug delivery Diagnosis & preventative capabilities Business Confidential ViRob Technology

Business Confidential ViRob Technology

Self Cleaning Shunt (SCS) A novel shunt technology for the continuous prevention of shunt blockage in hydrocephalus and NPH patients ViRob First Opportunity

Self Cleaning Shunt Markets (SCS) Hydrocephalus, also known as "water in the brain," is a medical condition, presented mostly in babies, in which there is an abnormal accumulation of cerebrospinal fluid (CSF) in the ventricles of the brain. The same condition, known as Normal Pressure Hydrocephalus (NPH), presented mostly in the elderly population, causing thinking and reasoning problems, difficulty walking, etc. Hydrocephalus occurs in about 1 in every 500 births in the U.S. alone1,2 Over 1,000,000 people in the United States currently live with hydrocephalus1 It is estimated that more than 700,000 Americans have NPH, but less than 20% receive an appropriate diagnosis1 The problem is often misdiagnosed as Dementia, Alzheimer's, or Parkinson's2 NPH can cause dementia, difficulty in walking and, urinary incontinence2 NIH, National Institute of Neurological Disorders and Stroke. http://www.ninds.nih.gov/disorders/hydrocephalus/detail_hydrocephalus.htm National Hydrocephalus Foundation. http://nhfonline.org/facts-about-hydrocephalus.htm
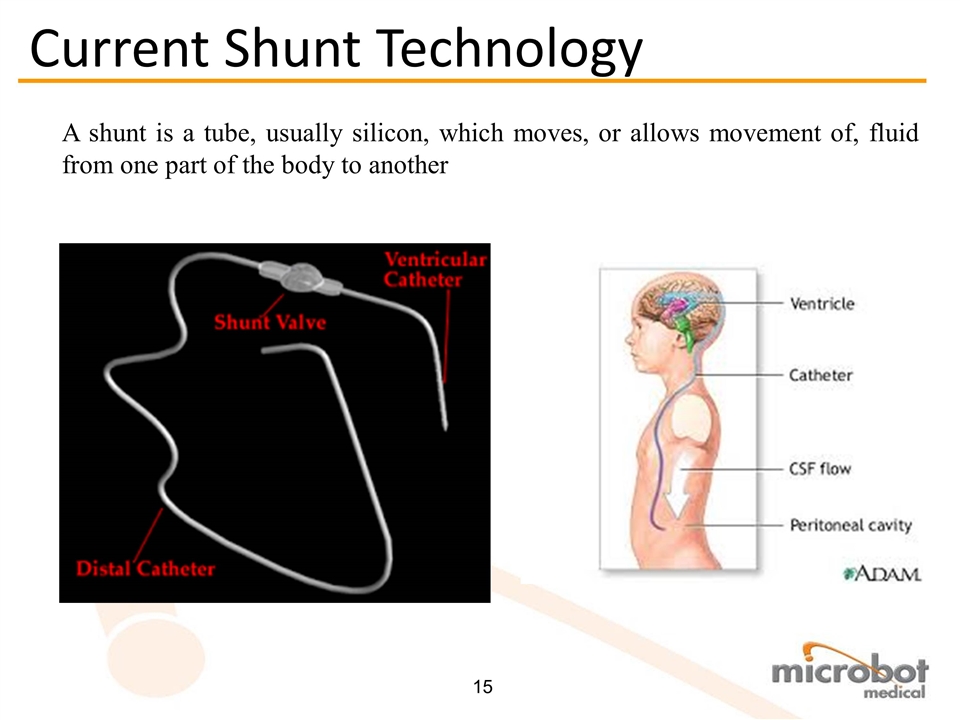
A shunt is a tube, usually silicon, which moves, or allows movement of, fluid from one part of the body to another Current Shunt Technology

Approximately 50% of shunts in the pediatric population fail within two years of placement and repeated neurosurgical operations are often required1 Ventricular catheter blockage is by far the most frequent events2 Current Shunt Failure Hydrocephalus Association. http://www.hydroassoc.org/complications-of-shunt-systems/ World Federation of Neurological Societies. http://www.wfns.org/pages/read_the_reviews/97.php?rid=5
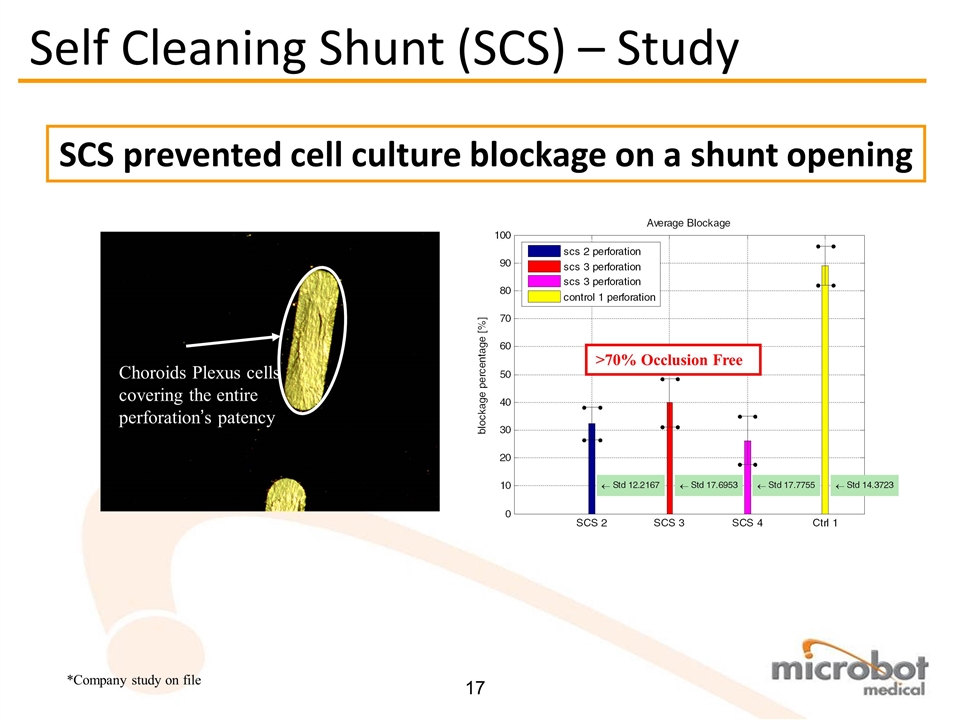
SCS prevented cell culture blockage on a shunt opening Choroids Plexus cells covering the entire perforation’s patency Self Cleaning Shunt (SCS) – Study >70% Occlusion Free *Company study on file

Conclusions: “Microbot Medical SCS presents low hydrodynamic resistance. The SCS behaves as a standard ventricular catheter and does not change the hydrodynamic performance of adjustable hydrocephalus valves.” Self Cleaning Shunt (SCS) – Study UK Shunt Testing Lab, Cambridge University, UK


TipCat is a disposable, flexible, self-propelled, see & treat endoscope/catheter Vs. push/pull, rigid or flexible endoscopes/catheters, applying high pressure on the tissue or presenting other limitations Business Confidential TipCat - Self Propelled Catheter Over guide-wire 0.018” (0.45mm) Catheter diameter 1.5mm

The TipCat colonoscope innovation is driven by two proprietary concepts: A SEMI-disposable design: Delivers high quality imaging No need for cleaning / reprocessing A simple self-propulsion mechanism Easy operation Allows a Physician Assistant (PA) to perform the insertion, while a GI specialist performs diagnosis and polyp removal Designed to reduce pain and discomfort Has the potential to increase patient acceptance Microbot TipCat – The Solution
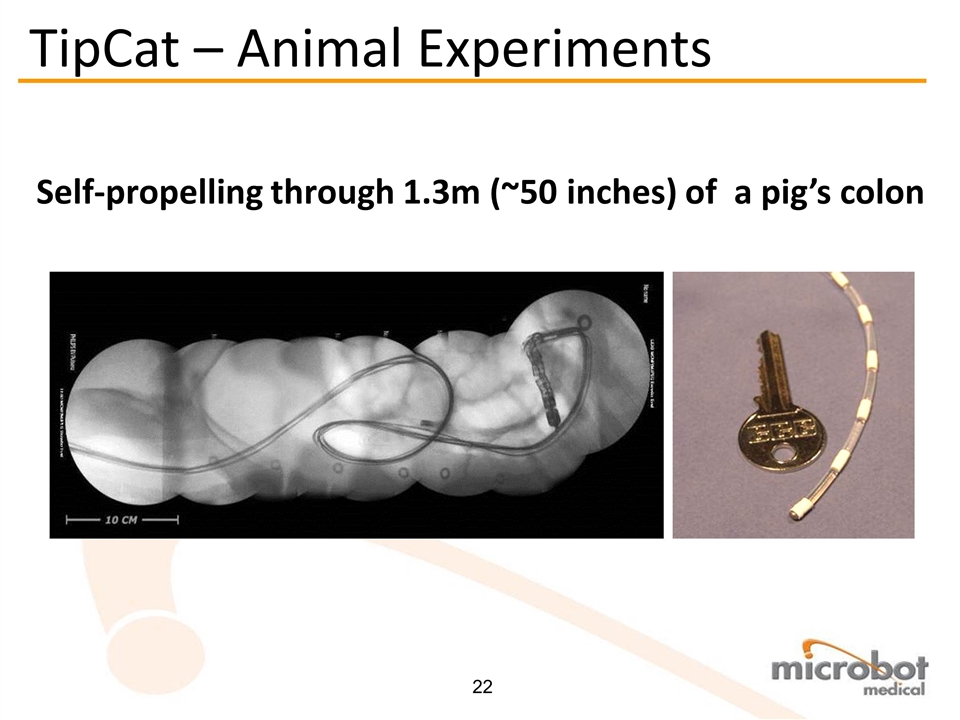
TipCat – Animal Experiments Self-propelling through 1.3m (~50 inches) of a pig’s colon
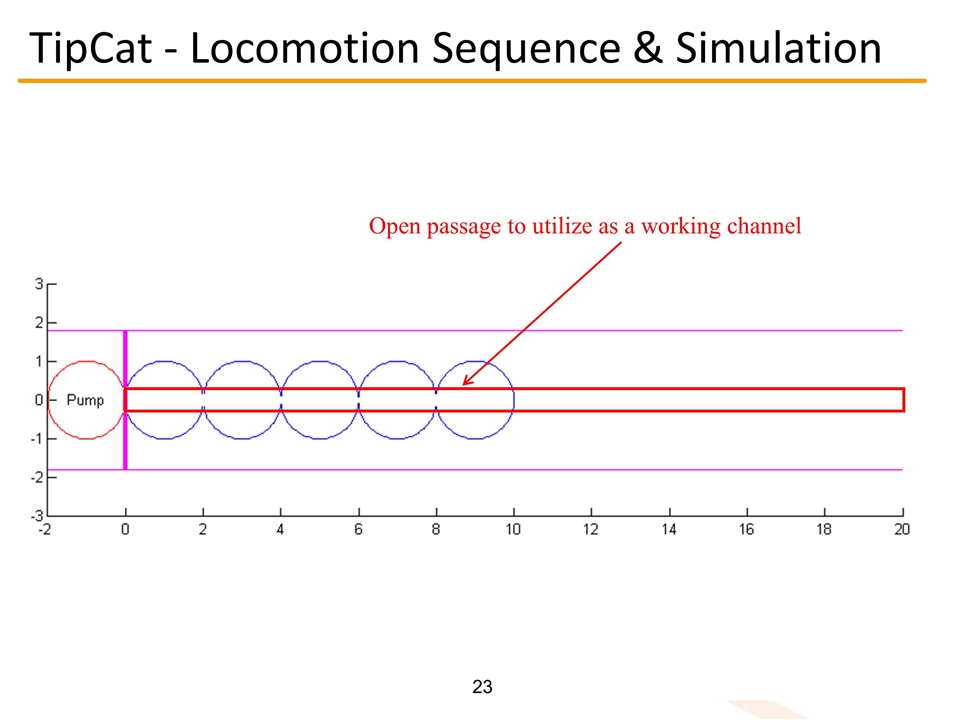
Open passage to utilize as a working channel TipCat - Locomotion Sequence & Simulation

TipCat - Locomotion Sequence
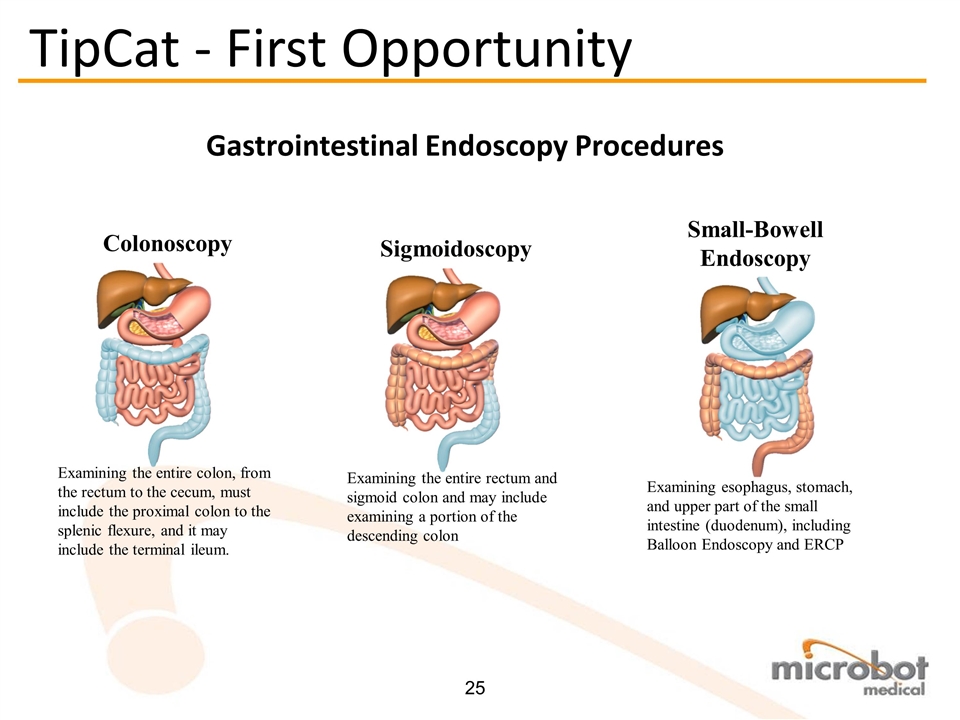
Examining esophagus, stomach, and upper part of the small intestine (duodenum), including Balloon Endoscopy and ERCP Small-Bowell Endoscopy Gastrointestinal Endoscopy Procedures Examining the entire colon, from the rectum to the cecum, must include the proximal colon to the splenic flexure, and it may include the terminal ileum. Colonoscopy Examining the entire rectum and sigmoid colon and may include examining a portion of the descending colon Sigmoidoscopy TipCat - First Opportunity

The North American flexible endoscopes market was valued at $1.27 billion in 2013, and is expected to reach $1.91 billion by 2018, at a CAGR of 8.5% during the forecast period, 2013 to 2018. 1 With a market value of about $600 million, the colonoscope and upper GI endoscope segments have been contributing 25.2% and 20.3% respectively to the total North American flexible endoscopes market.1 Annually, between 15 and 20 million endoscopy procedures are conducted with reusable endoscope devices to screen various components of a patient’s GI tract in the USA.2 North American Flexible Endoscopes Market Research Report. http://www.micromarketmonitor.com/market/north-america-flexible-endoscopes-1829607717.html American Journal of Infection Control ,V(6)41, June 2013, Pages S24, 40th Annual Conference Abstracts, APIC 2013, Ft Lauderdale, FL APIC 40th Annual Conference GI Endoscope Market

ADR: Approximately 30% of polyps are missed during Standard Colonoscopy (SC), e.g., due to polyps hidden behind colon folds and flexures 1. The ‘GI Gap’: Growing shortage of gastroenterologists and increasing patients volume affecting screening capacity for colorectal cancer2. Cross-Contamination: The reported incidence of bacteremia after colonoscopy, with or without biopsies and polypectomies, reaches up to 25%3. Whereas, 3 out of 20 flexible gastrointestinal (GI) endoscopes used for screening pose potential infection risk -- according to a study of endoscopes used at hospitals across the USA4. Presentation Abstract, 2013 UEGW (Abstract # OP286). http://www.smartmedsys.com/images/pdf/g-eye_tandem_abs_uegw2013.pdf http://www.olympusamerica.com/oai_pressDetails.asp?pressNo=655 Kovalevaa Jet al, Transmission of Infection by Flexible Gastrointestinal Endoscopy and Bronchoscopy, , Clin. Microbiol. Rev. April 2013 vol. 26 no. 2 231-254, http://cmr.asm.org/content/26/2/231.full. American Journal of Infection Control ,V(6)41, June 2013, Pages S24, 40th Annual Conference Abstracts, APIC 2013, Ft Lauderdale, FL APIC 40th Annual Conference The Unmet Need

Cross-Contamination in Recent Mainstream Media

PORTFOLIO OF INNOVATIONS
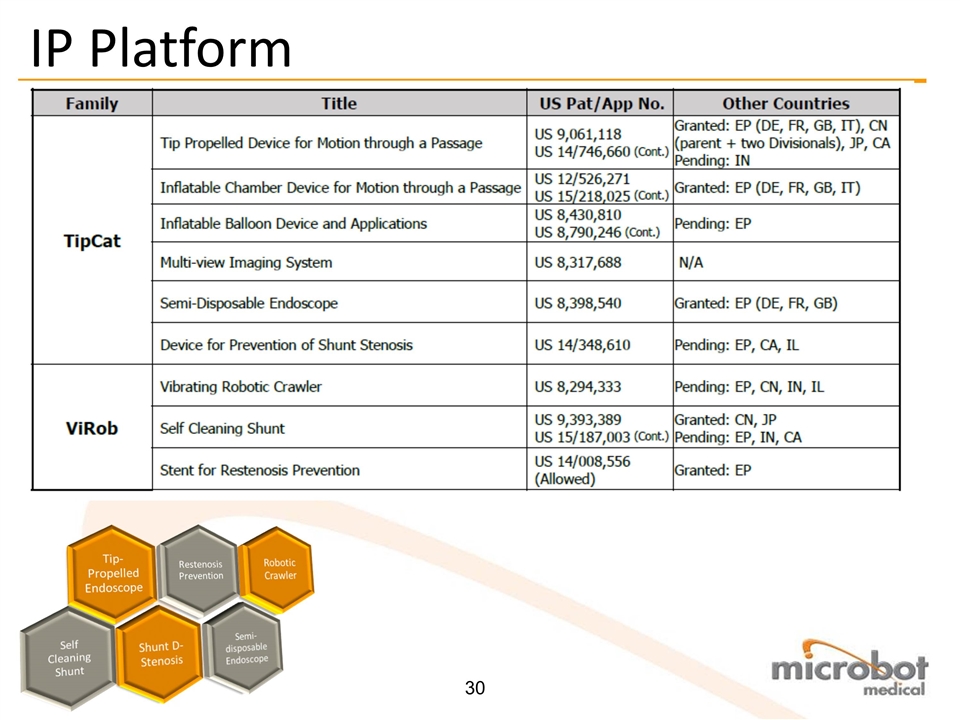
IP Platform
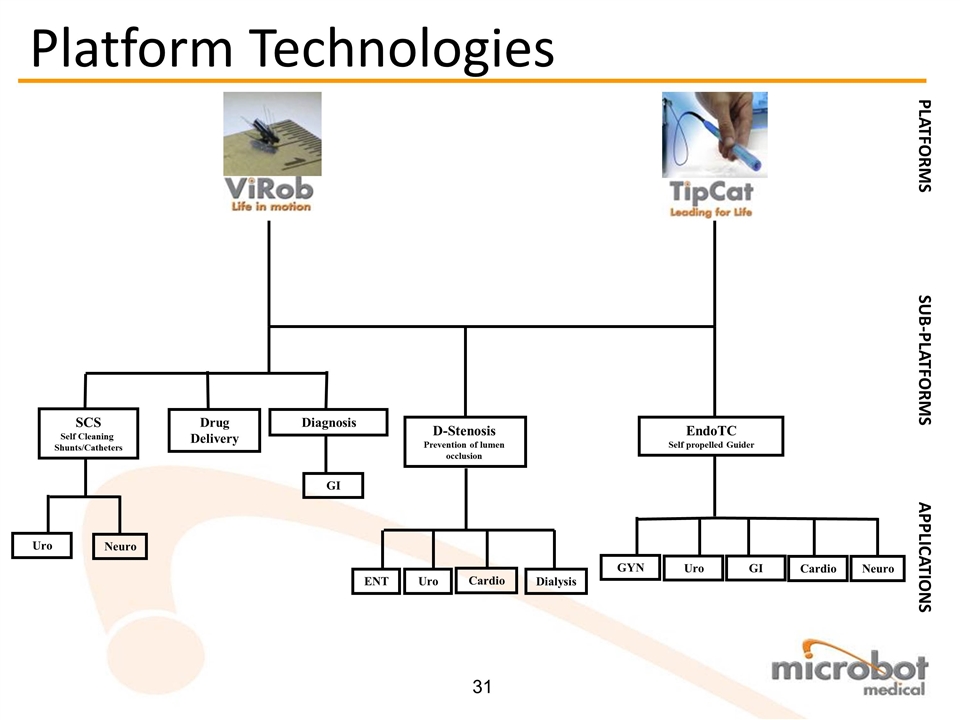
Business Confidential SCS Self Cleaning Shunts/Catheters PLATFORMS SUB-PLATFORMS D-Stenosis Prevention of lumen occlusion EndoTC Self propelled Guider APPLICATIONS Diagnosis Drug Delivery Cardio Dialysis Neuro Cardio GI Uro GYN Uro Neuro Uro GI ENT Platform Technologies

Summary Innovative and transformational technologies, capitalizing on the future trend of Micro-Invasive Robotic Surgery Microbot’s two platforms: ViRob - an Autonomous Advancing Micro-Robot (AAMR) Platform TipCat – a Self-Propelled Endoscope Platform Unique positioning for both platforms: Multi-disciplinary Platform technologies (multi-applications) Large market potential (Existing & future) Introducing transformational technology into the market Delivering prevention, diagnostic and treatment capabilities Extremely strong value proposition for all stakeholders: Patients, Physicians, Health care providers, Payers Leveraging growing trend of Minimal Invasive Surgery/Treatments Current status: Post intensive pre-clinical research and experiments Broad & specific IP Proven track record of the founders and management team Closed round A financing of >$5M to date Including multiple grants from the Office of the Chief Scientist in Israel