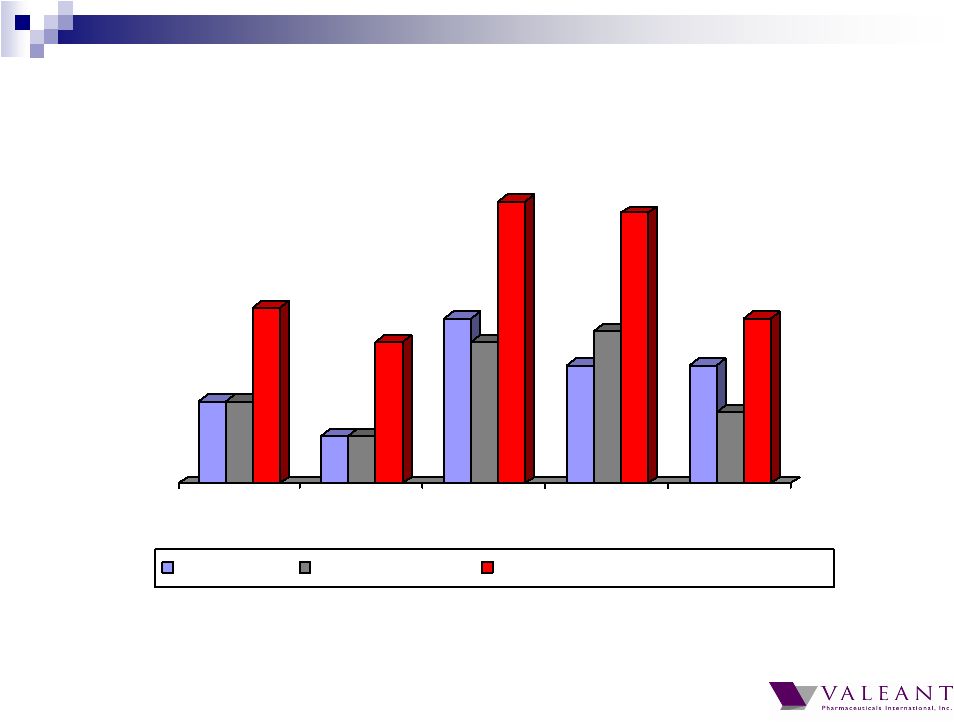
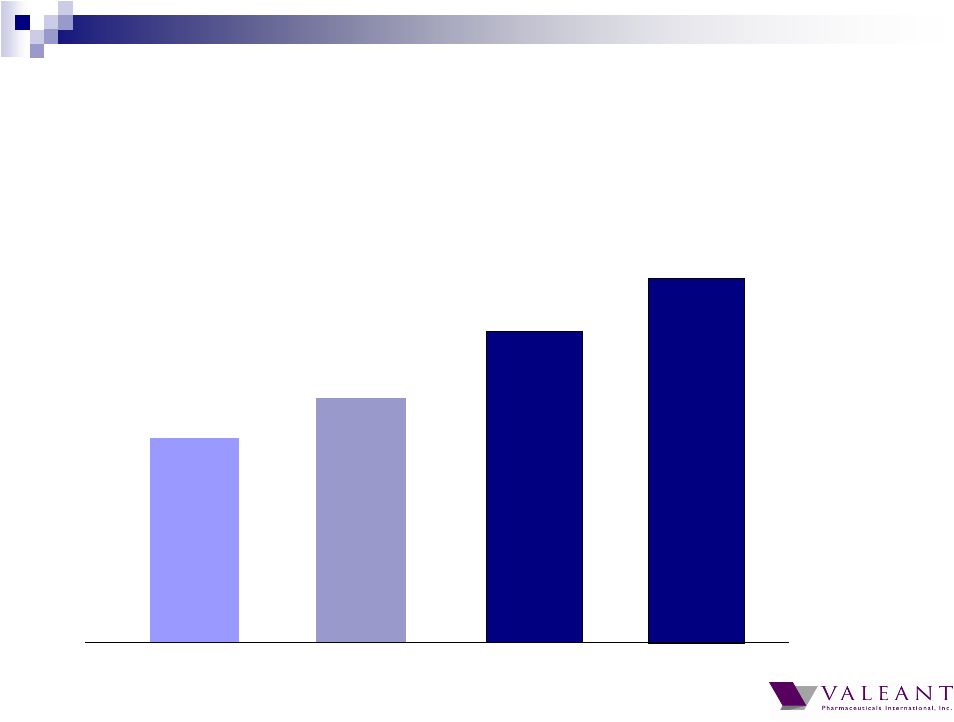
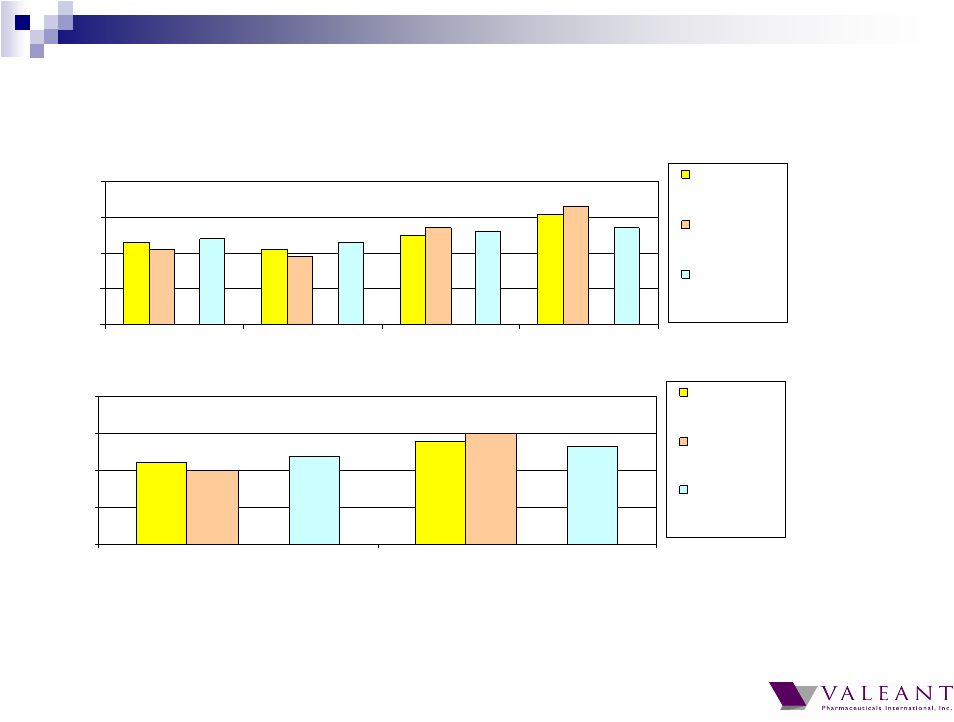
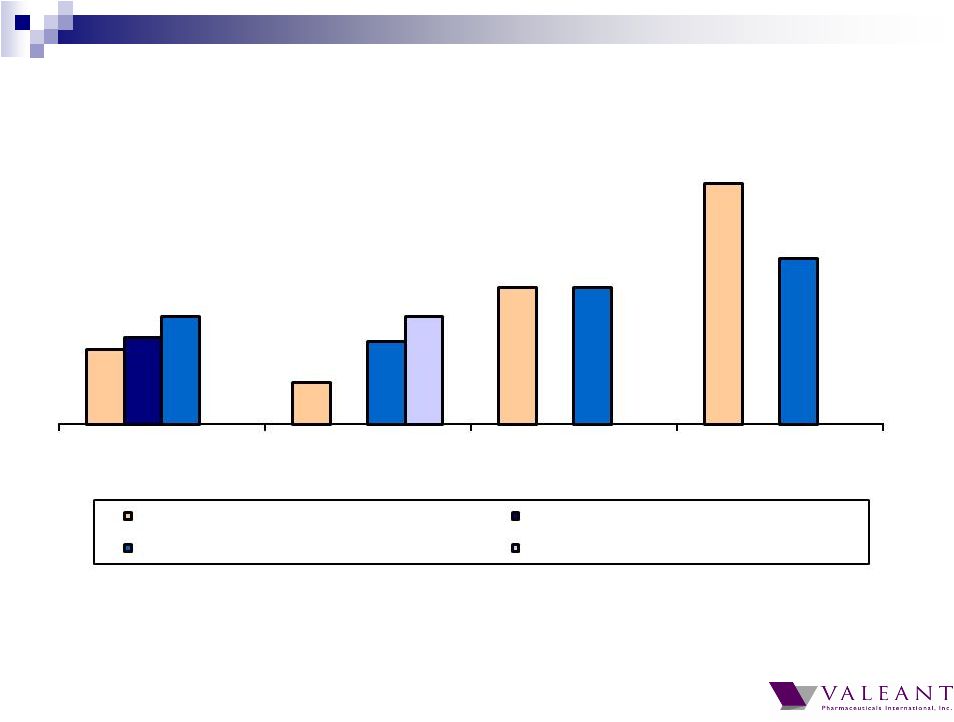
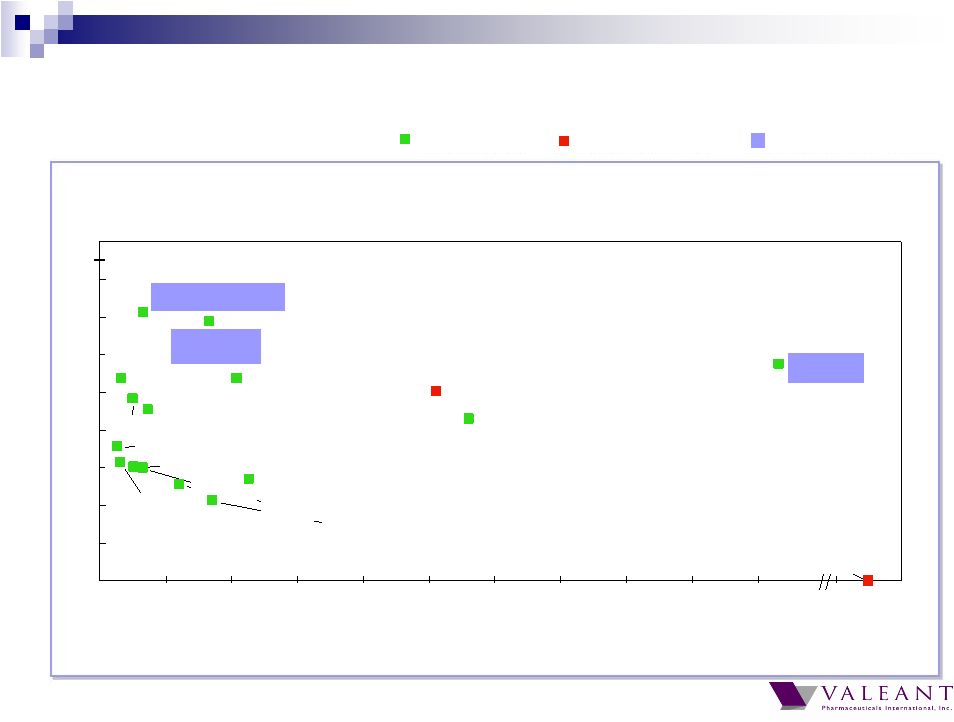
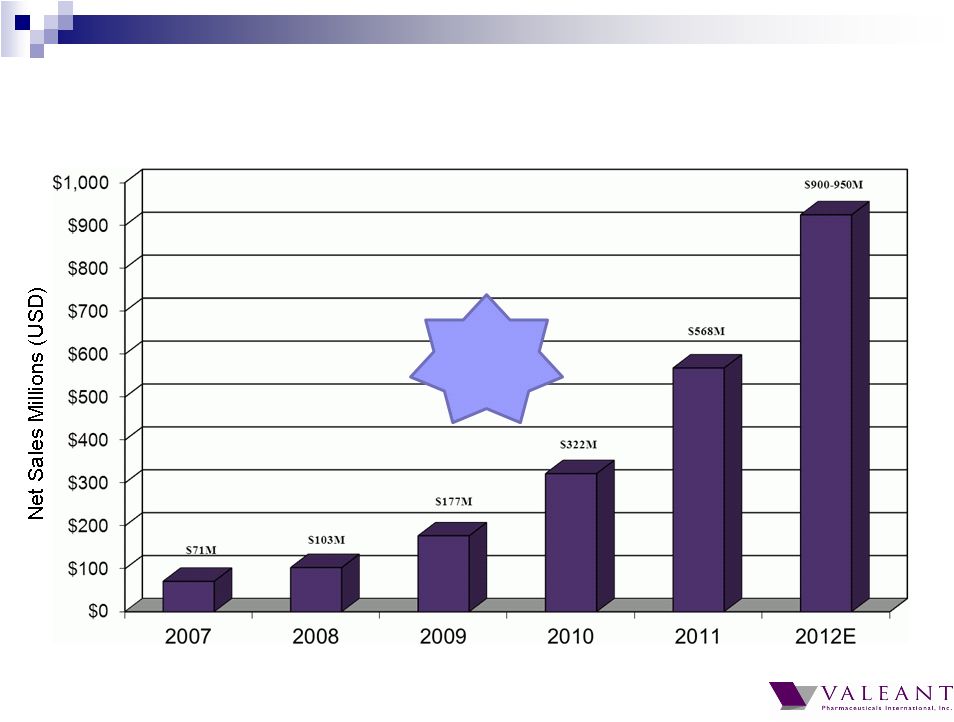
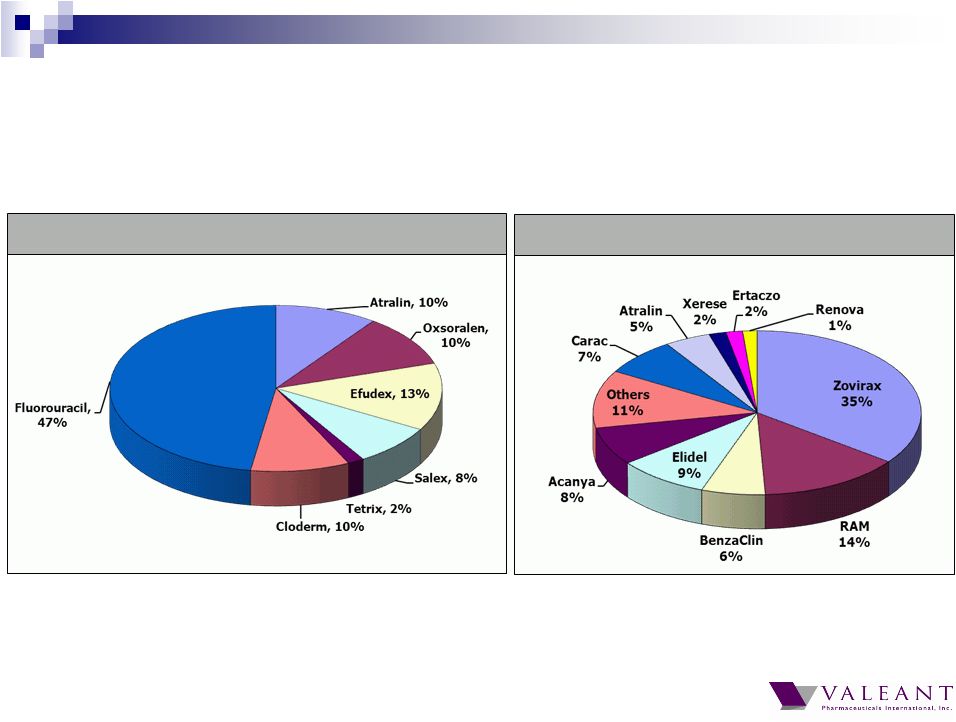
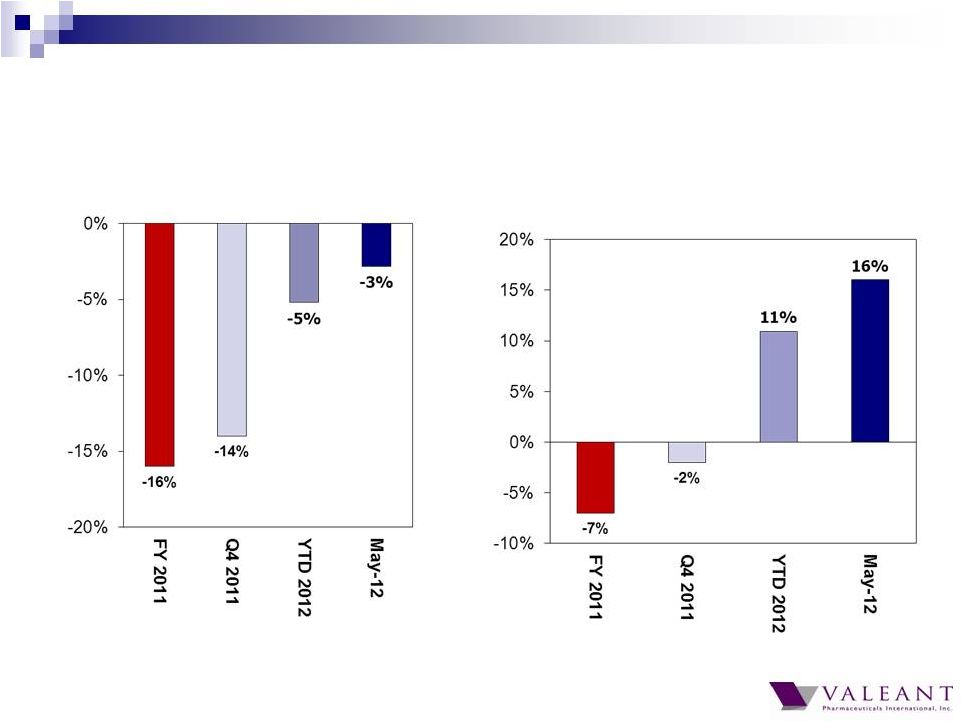
Important Information Forward-looking Statements This presentation may contain forward-looking statements, including, but not limited to, statements regarding our financial guidance (including quarterly trends) with respect to revenues, non-GAAP cash earnings per share and adjusted cash flow from operations, future growth, potential product launches, regulatory submissions and approvals, timing and costs of integration, synergies, expenses, business development opportunities and market position, and the impact of foreign exchange on our financial performance and projections with respect to market growth, size and spend. Forward-looking statements may be identified by the use of the words "anticipates," "expects," "intends," "plans," "should," "could," "would," "may," "will," "believes," "estimates," "potential," or "continue" and variations or similar expressions. These statements are based upon the current expectations and beliefs of management and are subject to certain risks and uncertainties that could cause actual results to differ materially from those described in the forward-looking statements. These risks and uncertainties include, but are not limited to, risks and uncertainties discussed in the company's most recent annual or quarterly report filed with the Securities and Exchange Commission (the “SEC”) and other risks and uncertainties detailed from time to time in the Company’s filings with the SEC and the Canadian Securities Administrators (“CSA”), which factors are incorporated herein by reference. Readers are cautioned not to place undue reliance on any of these forward-looking statements. Valeant undertakes no obligation to update any of these forward-looking statements to reflect events or circumstances after the date of this presentation or to reflect actual outcomes. Non-GAAP Information To supplement the financial measures prepared in accordance with generally accepted accounting principles (GAAP), the Company uses non-GAAP financial measures that exclude certain items. Management uses non-GAAP financial measures internally for strategic decision making, forecasting future results and evaluating current performance. By disclosing non-GAAP financial measures, management intends to provide investors with a meaningful, consistent comparison of the Company's core operating results and trends for the periods presented. Non-GAAP financial measures are not prepared in accordance with GAAP; therefore, the information is not necessarily comparable to other companies and should be considered as a supplement to, not a substitute for, or superior to, the corresponding measures calculated in accordance with GAAP. The Company has provided guidance with respect to cash earnings per share, adjusted cash flows from operations and organic growth rates, which are non-GAAP financial measures. The Company has not provided a reconciliation of these forward-looking non-GAAP financial measures due to the difficulty in forecasting and quantifying the amount of the items excluded from the non-GAAP financial measures that will be included in the comparable GAAP financial measures. Reconciliations for other non-GAAP financial measures can be found in the appendix to this presentation and in the Company’s earnings releases, which are available under the Investor Relations section of the Company’s website (www.valeant.com). Note on Financial Guidance The reaffirmation of the Company’s previous financial guidance issued on May 3, 2012 is effective only as of the date hereof, June 21, 2012, and will not be updated or confirmed until the Company publicly announces updated or affirmed guidance. |