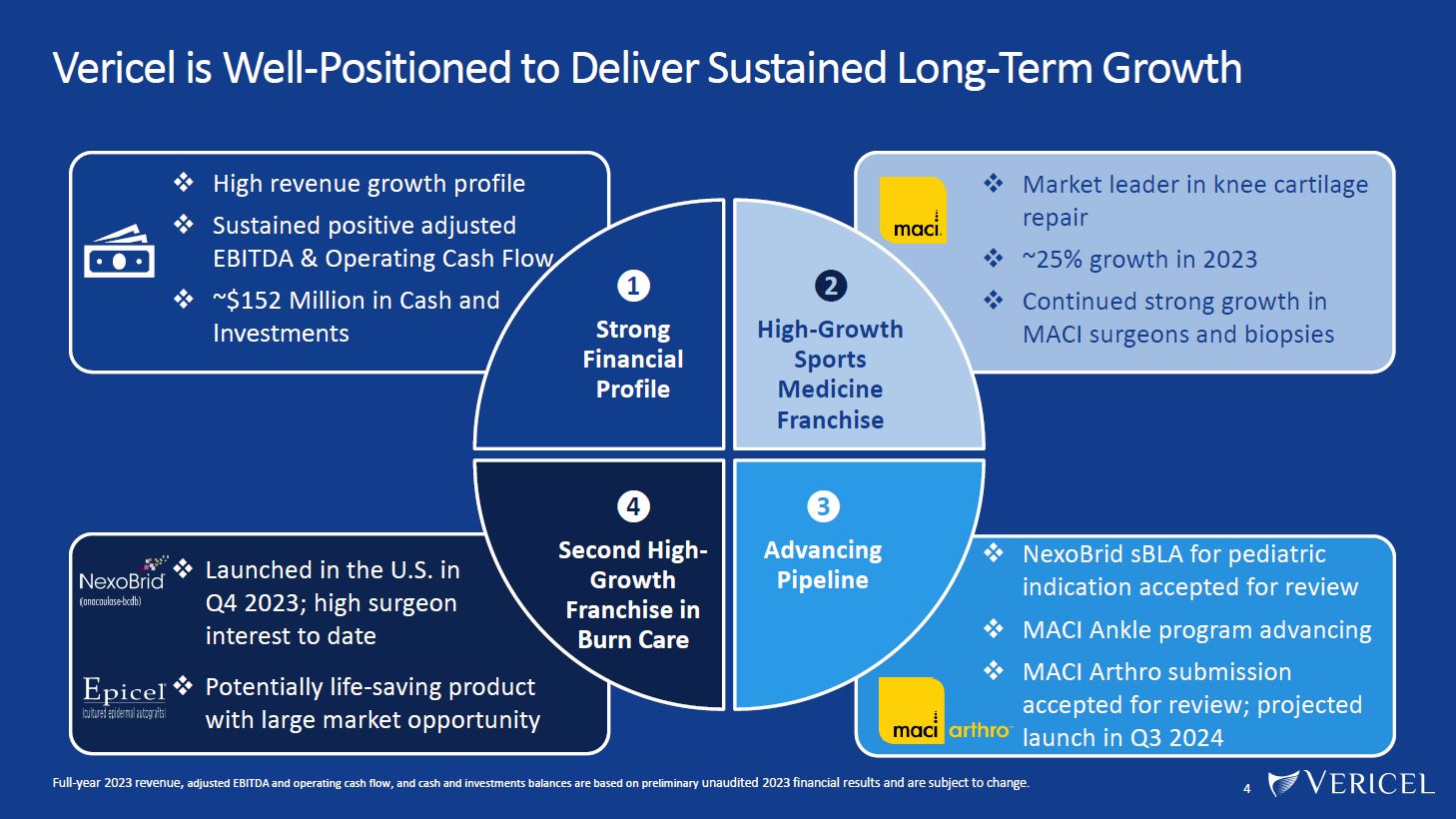
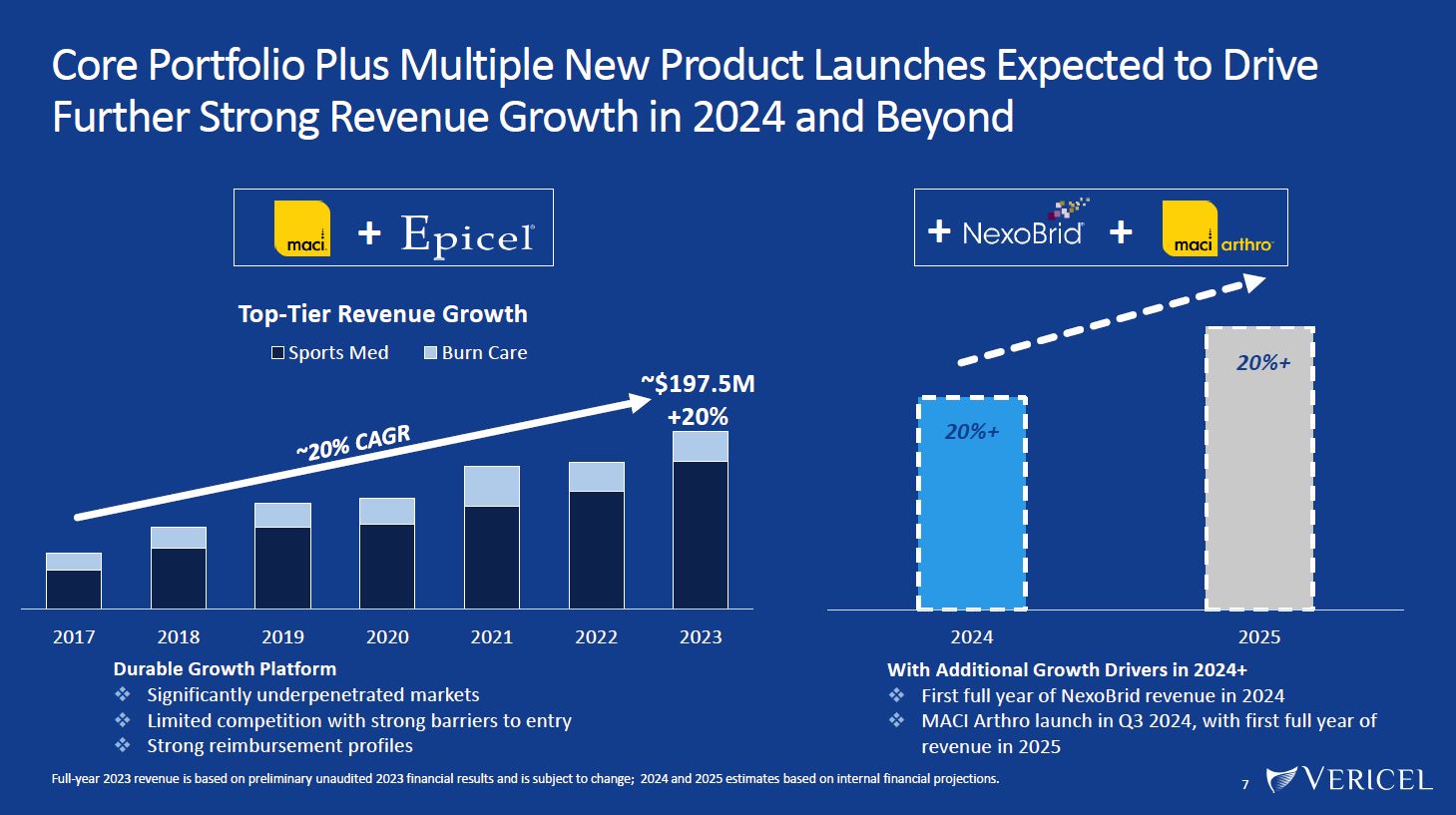
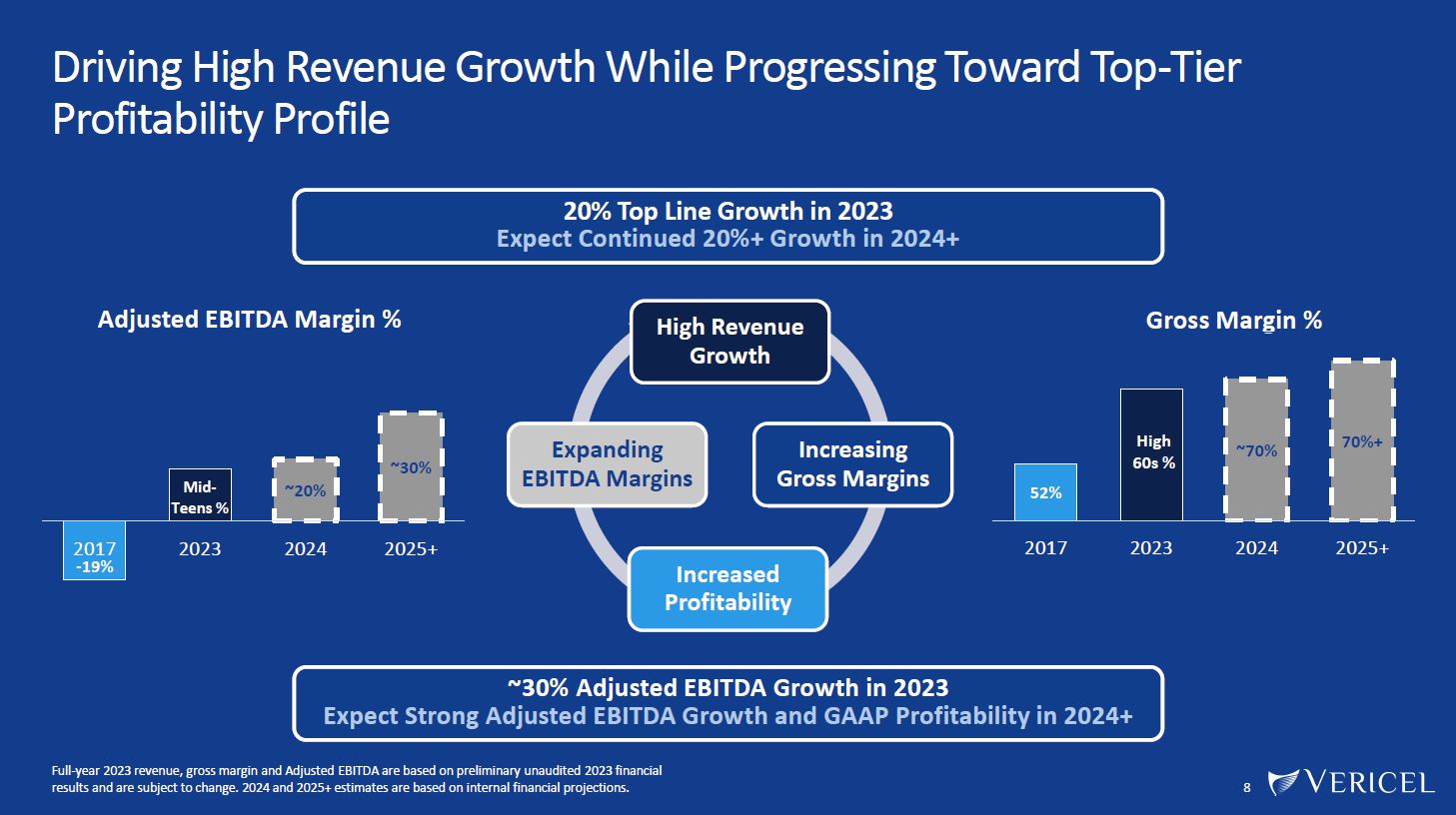
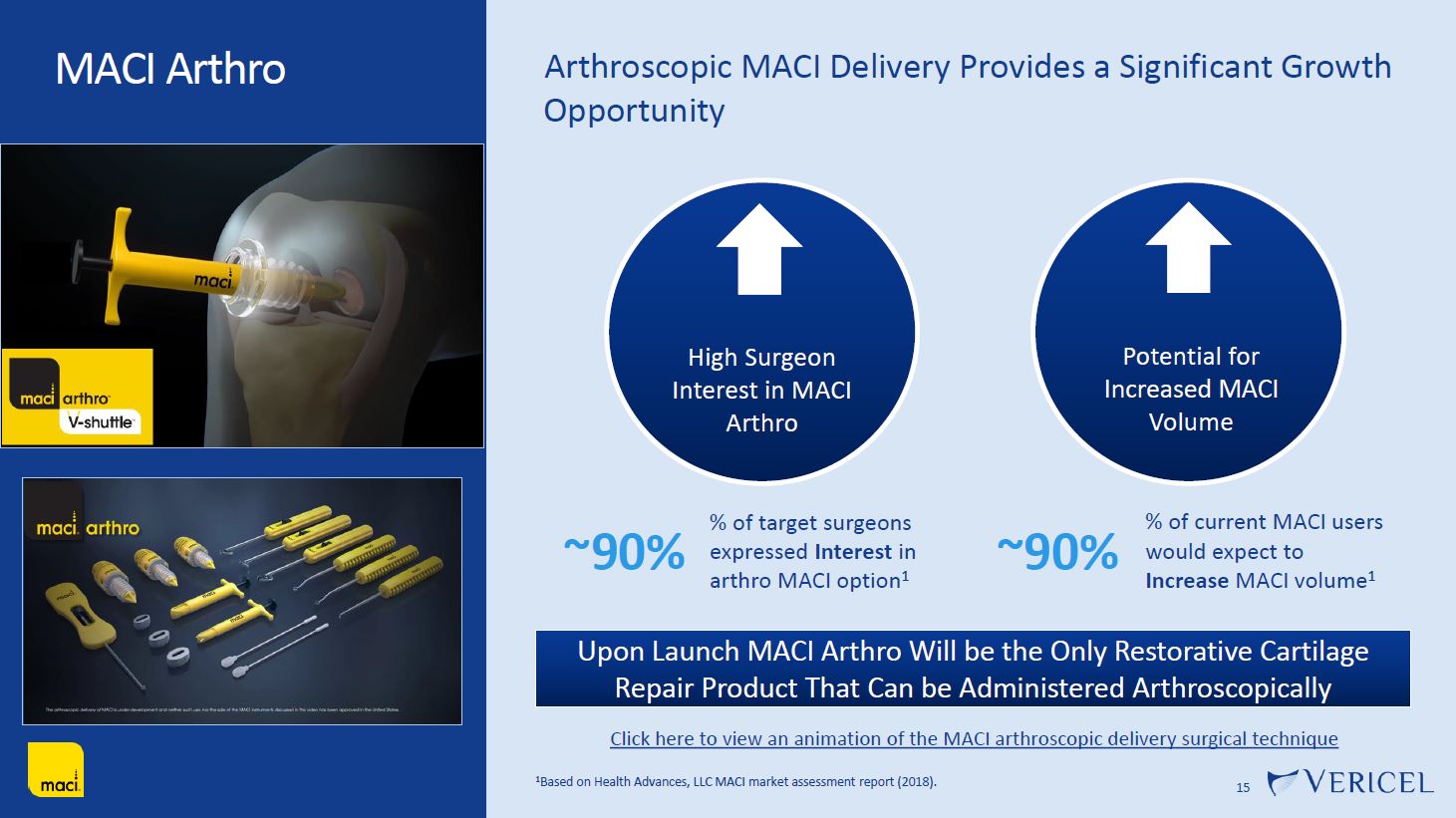
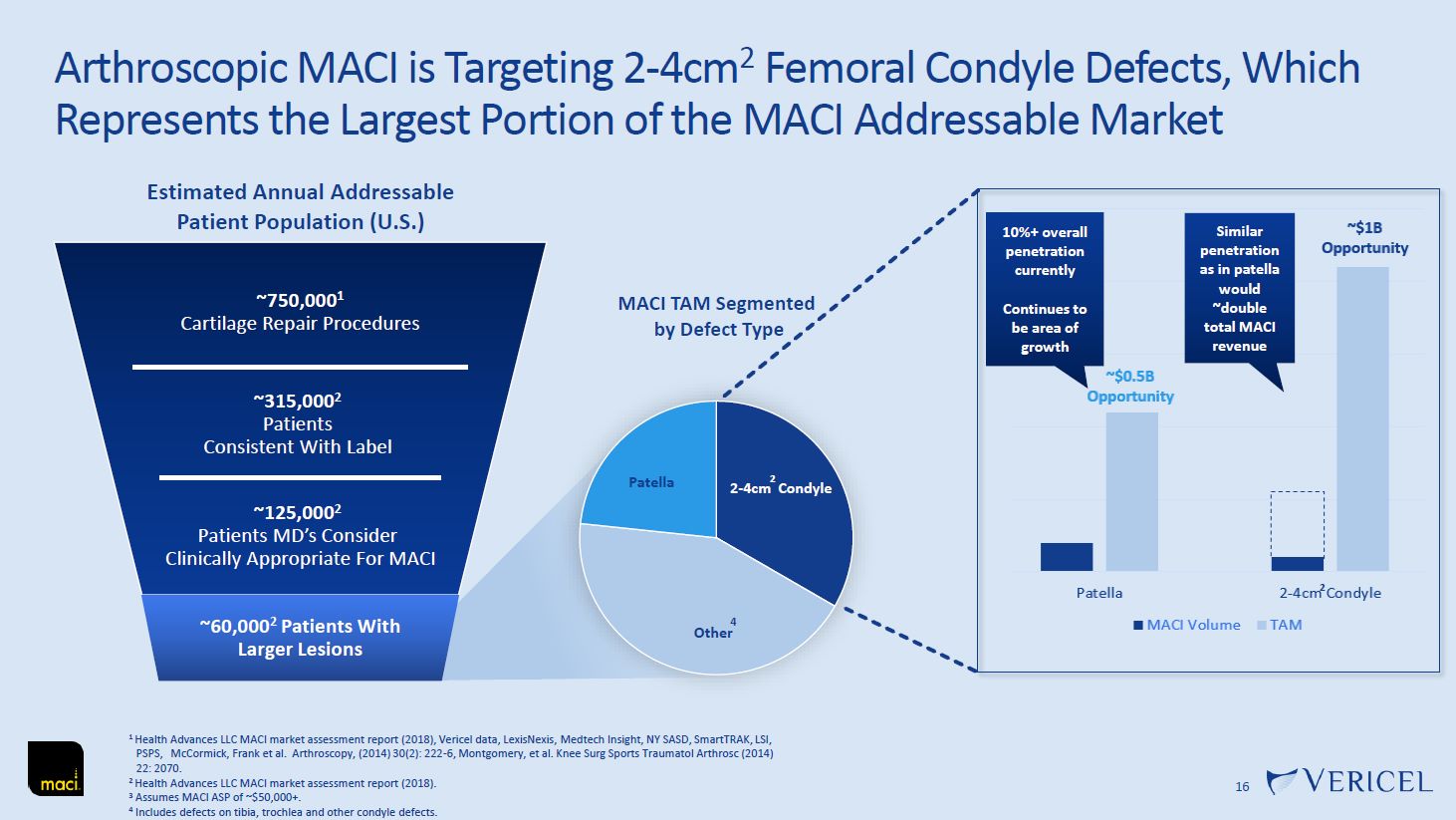
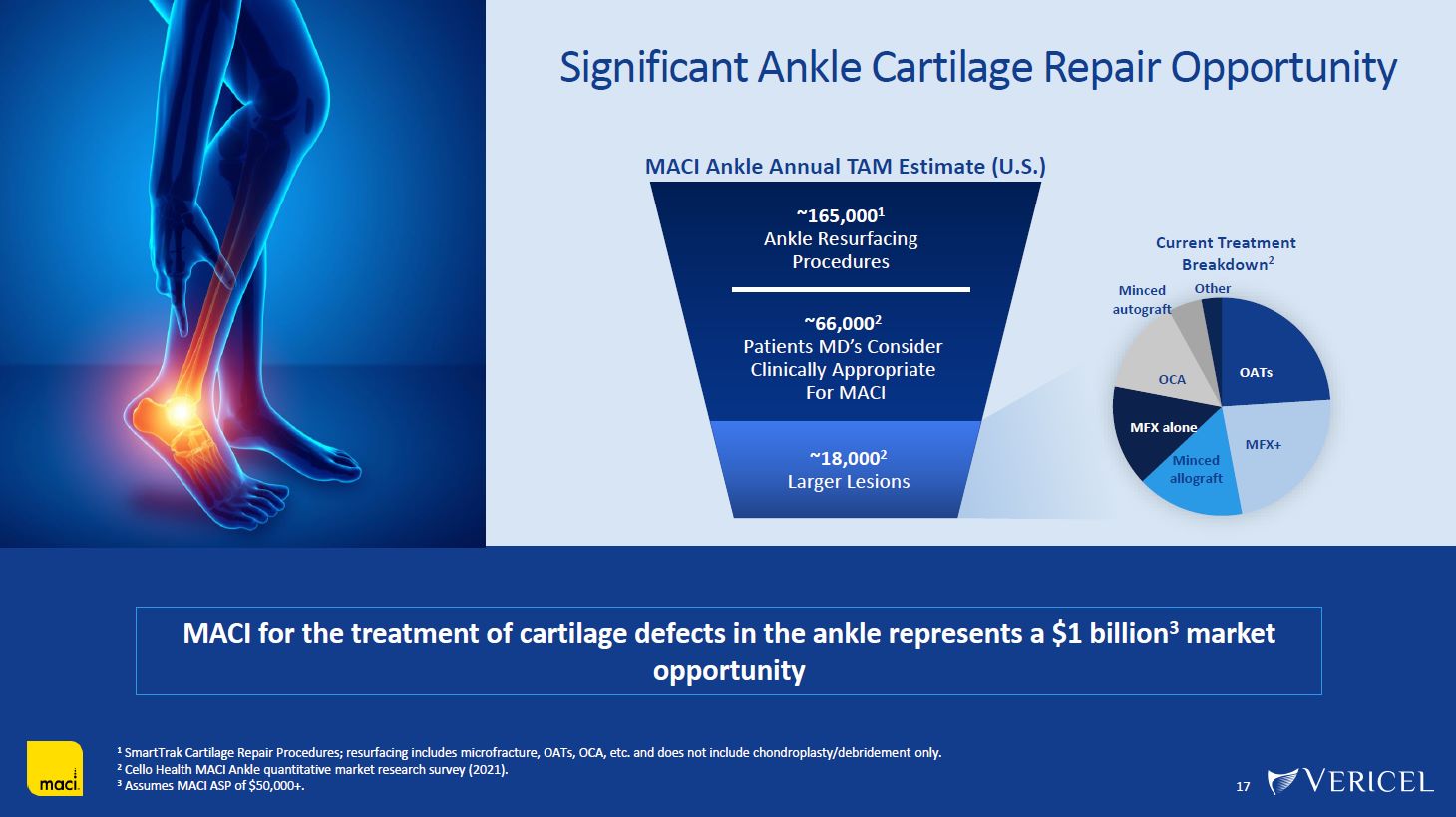
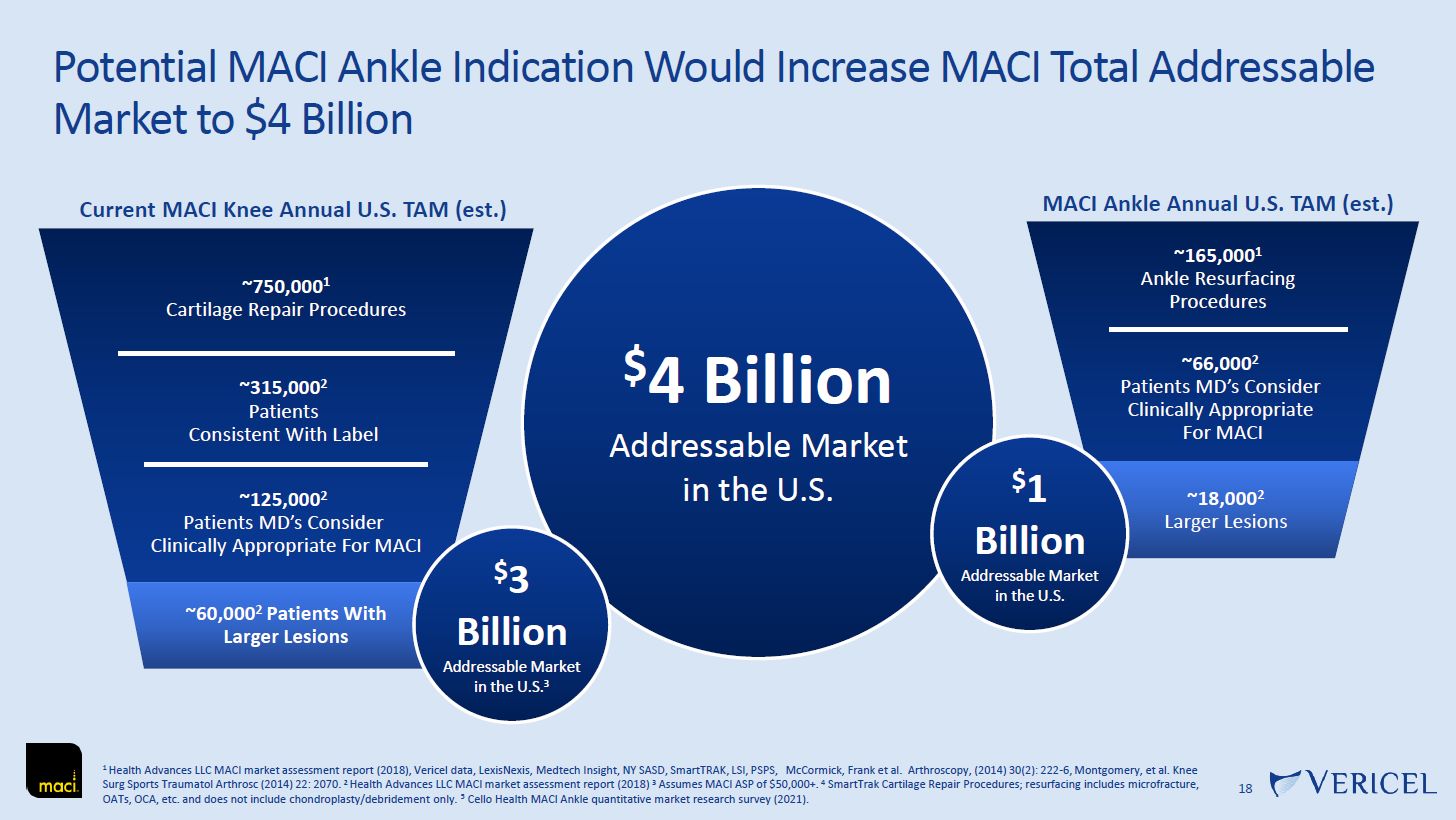
Forward-Looking Statements and Legal Disclosure 2 Forward-Looking Statements Vericel cautions you that all statements other than statements of historical fact included in this presentation that address activities, events or developments that we expect, believe or anticipate will or may occur in the future are forward-looking statements. Although we believe that we have a reasonable basis for the forward-looking statements contained herein, they are based on current expectations about future events affecting us and are subject to risks, assumptions, uncertainties and factors relating to our operations and business environment, all of which are difficult to predict and many of which are beyond our control. Our actual results may differ materially from those expressed or implied by the forward-looking statements in this presentation. These statements are often, but are not always, made through the use of words or phrases such as “anticipates,” “intends,” “estimates,” “plans,” “expects,” “continues,” “believe,” “guidance,” “outlook,” “target,” “future,” “potential,” “goals” and similar words or phrases, or future or conditional verbs such as “will,” “would,” “should,” “could,” “may,” or similar expressions. Among the factors that could cause actual results to differ materially from those set forth in the forward-looking statements include, but are not limited to, the inherent uncertainties associated with our expectations concerning expected revenue results for the fourth quarter and full-year ended 2023, adjusted EBITDA, operating cash flow, and estimates of our cash, restricted cash and investments as of December 31, 2023. Vericel’s revenue expectations for the fourth quarter and full-year ended 2023, as well as its estimates concerning adjusted EBITDA, operating cash flow, and cash and investments are preliminary, unaudited and are subject to change during ongoing internal control, review, and audit procedures. Additional factors that could cause actual results to differ materially from those set forth in the forward- looking statements include, but are not limited to, uncertainties associated with our expectations regarding future revenue, growth in revenue, market penetration for MACI®, Epicel®, and NexoBrid®, growth in profit, gross margins and operating margins, the ability to continue to scale our manufacturing operations to meet the demand for our cell therapy products, including the timely completion of a new headquarters and manufacturing facility in Burlington, Massachusetts, the ability to achieve or sustain profitability, contributions to adjusted EBITDA, the expected target surgeon audience, potential fluctuations in sales and volumes and our results of operations over the course of the year, timing and conduct of clinical trial and product development activities, timing and likelihood of the FDA’s potential approval of the arthroscopic delivery of MACI to the knee or the use of MACI to treat cartilage defects in the ankle, the estimate of the commercial growth potential of our products and product candidates, competitive developments, changes in third-party coverage and reimbursement, physician and burn center adoption of NexoBrid, supply chain disruptions or other events or factors affecting MediWound’s ability to manufacture and supply sufficient quantities of NexoBrid to meet customer demand, including but not limited to the ongoing Israel-Hamas war, negative impacts on the global economy and capital markets resulting from the conflict in Ukraine and the Israel-Hamas war, adverse developments affecting financial institutions, companies in the financial services industry or the financial services industry generally, global geopolitical tensions or record inflation and potential future impacts on our business or the economy generally stemming from a resurgence of COVID-19 or another similar public health emergency. These and other significant factors are discussed in greater detail in Vericel’s Annual Report on Form 10-K for the year ended December 31, 2022, filed with the Securities and Exchange Commission (SEC) on February 23, 2023, Vericel’s Quarterly Report on Form 10-Q for the quarter ended September 30, 2023, filed with the SEC on November 8, 2023, and in other filings with the SEC. These forward-looking statements reflect our views as of the date hereof and Vericel does not assume and specifically disclaims any obligation to update any of these forward-looking statements to reflect a change in its views or events or circumstances that occur after the date of this release except as required by law. Discussion of Indications Currently Under Development Additionally, portions of this presentation discuss the potential clinical advantages of the arthroscopic delivery of MACI to treat cartilage defects in the knee joint and the use of MACI in the ankle joint, as well as the potential effect the approval of those additional indications could have on MACI’s total addressable market. The reader is reminded that the implantation of MACI in the knee is currently approved to be performed via an arthrotomy. The arthroscopic delivery of MACI to the knee joint and the use of MACI in the ankle joint are currently under development and such uses have not been approved in the U.S.