 1 MPN Pacritinib Phase 3* MDS/AML Tosedostat Phase 3* NHL Pixuvri™ Marketed (EU) Cell Therapeutics, Inc. Company Overview * Phase 3 ready. Covering a Spectrum of Blood Related Cancers Exhibit 99.1 |
 2 Forward Looking Statement This presentation includes forward-looking statements that involve a number of risks and uncertainties, the outcome of which could materially and/or adversely affect actual future results and the market price of CTI's securities. Specifically, the risks and uncertainties that could affect the development of Pixuvri, OPAXIO, pacritinib, tosedostat and brostallicin include risks associated with preclinical and clinical developments in the biopharmaceutical industry in general and with Pixuvri, OPAXIO, pacritinib, tosedostat or brostallicin in particular including, without limitation, the potential failure of Pixuvri to prove safe and effective for the treatment of relapsed or refractory non-Hodgkin's lymphoma and/or other tumors as determined by the U.S. Food and Drug Administration (the “FDA”), that Pixuvri may not be available to patients in the E.U. beginning in September 2012, that CTI may not market and commercialize Pixuvri as planned, that CTI may not launch Pixuvri in the E.U. this year as planned or in any of the specific countries in the E.U. we are currently planning to launch, that CTI may not be able to complete the PIX306 clinical trial of Pixuvri-rituximab compared to gemcitabine-rituximab in patients who have relapsed after 1 to 3 prior regimens for aggressive B-cell NHL and who are not eligible for autologous stem cell transplant by June 2015 or at all as required by the European Medicines Agency (the “EMA”) or have the results of such trial available by June 2015 or at all, that CTI may not be able complete a post- marketing study aimed at confirming the clinical benefit observed in the PIX301 trial, that the conditional marketing authorization for Pixuvri may not be renewed, the potential failure of OPAXIO to prove safe and effective for treatment of non-small cell lung and ovarian cancers, that the interim survival results for the phase 3 clinical trial for OPAXIO may not be ready in 2012, the potential that phase III studies of pacritinib might not begin in the fourth quarter of 2012, the potential that pacritinib may fail to prove safe and effective for primary MF and MF secondary to other MPNs, the potential that phase III studies of tosedostat may not occur as planned, the potential failure of tosedostat to prove safe and effective for the treatment of elderly patients with newly-diagnosed AML or high-risk myelodysplastic syndrome ("MDS") (including when administered in combination with cytarabine or decitabine) as determined by the FDA and/or the EMA, the potential failure of combination studies of tosedostat with hypomethylating agents in treating AML and/or MDS, that the studies of tosedostat may not achieve their primary and/or secondary objectives, that the development of tosedostat may be delayed by disputes with Chroma Therapeutics Ltd. (“Chroma”) under CTI’s co-development and license agreement with Chroma, that OPAXIO, pacritinib, tosedostat and/or brostallicin may not be approved by the FDA and/or the EMA, that CTI cannot predict or guarantee what actual sales of its product candidates will be, that CTI cannot predict or guarantee the pace or geography of enrollment of its clinical trials or the total number of patients enrolled, that CTI may not achieve a burn rate of an average of $4.5 million per month through the remainder of the year, that CTI's average net operating burn rate may increase, that CTI cannot guarantee that it will not exceed it estimated net operating burn rate for 2013, that CTI cannot guarantee that it will not incur any debt or maintain its current cash amount of cash available, that CTI cannot guarantee that its current amount of cash available will be sufficient to sustain CTI’s operations until January 2013, that CTI cannot predict or guarantee when CTI will achieve profitability, if ever, or if CTI will achieve cash flow break even in the fourth quarter of 2014, and that CTI may not be able to continue to raise capital as needed to fund its operations in general, and, including, without limitation, competitive factors, technological developments, costs of developing, producing, and selling its product candidates, and the risk factors listed or described from time to time in CTI's filings with the Securities and Exchange Commission including, without limitation, CTI's most recent filings on Forms 10-K, 8-K, and 10-Q. Except as may be required by law, CTI does not intend to update or alter its forward-looking statements whether as a result of new information, future events, or otherwise. |
 3 Company Overview Hematology/Oncology-focused biotechnology company 1 commercial product • Pixuvri™ : for the treatment of non-Hodgkin's lymphoma (“NHL”) - Conditional Marketing Authorization May 2012- E.U. launch September 2012 - Attractive “niche” market ~16,000 patients (1) ; 10 yrs market exclusivity in E.U. 3 late-stage product candidates: • Pacritinib, : highly selective JAK2 inhibitor, Phase 3- expected Q4 2012 - Addresses unmet need in MF market post ruxolitinib approval • Tosedostat : novel aminopeptidase inhibitor anticipating Phase 3 in 2013 for MDS/AML • Opaxio : protein-paclitaxel conjugate completing Phase 3 trial in 1 st line maintenance therapy for Ovarian Cancer ~$24mm cash and no debt on the balance sheet (2) • Estimated cash burn of an average of ~$4.5mm per month through the remainder of 2012. (1) European Cancer Observatory, Publications on NHL subgroup and relapse rates includes 4th line. (2) Pro forma cash balance as of August 1,2012. |
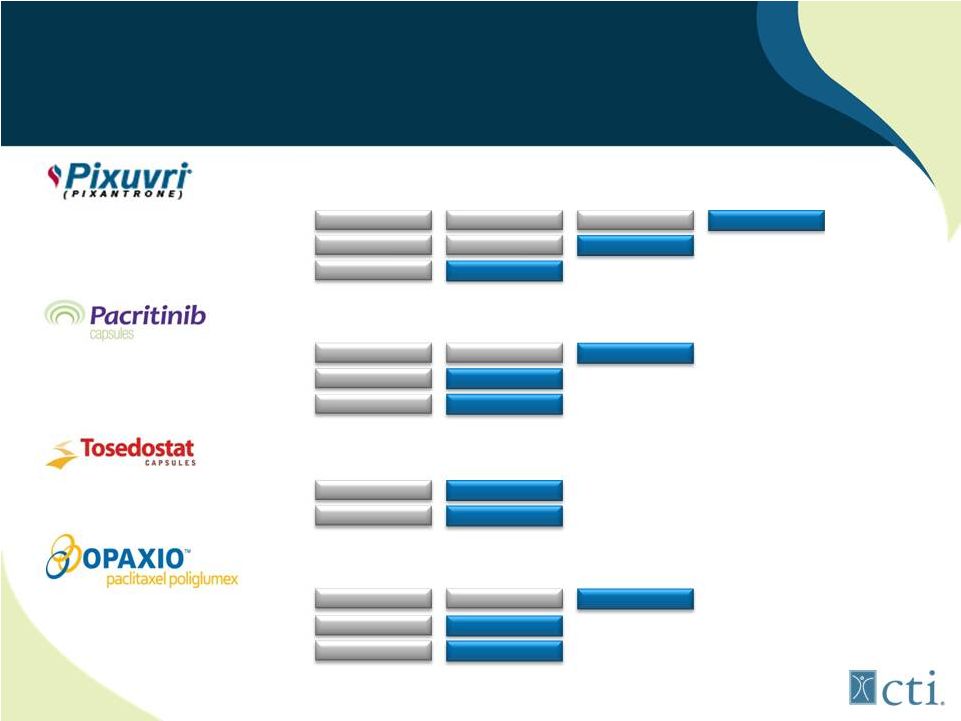 4 Current Pipeline Third-line r/r aggressive NHL (PIX301) Second-line r/r aggressive NHL (PIX306) First-line aggressive NHL (PIX203) Myelofibrosis (<100,000plts) (1) Primary Myelofibrosis Relapsed Lymphoma First-line or r/r Myelodysplastic Syndrome Elderly high-risk r/r AML (OPAL) Maintenance therapy for ovarian cancer First-line GBM +XRT vs standard of care Head and neck cancer w/ Erbitux+XRT Phase 1 Phase 2 Phase 3 Commercial (1) Phase 3 study is expected to initiate in 4Q 2012. |
 5 Investment Highlights Pixuvri Approved in E.U. Pacritinib is a Differentiated JAK2 Inhibitor Leading, Late-Stage Oncology Franchise Tosedostat has Shown Encouraging Data in AML/MDS Significant Upcoming Milestones • Late-stage portfolio focused on blood-related cancers • Deep pipeline with 1 product approved in the E.U., 1 product in Phase 3 trials, 2 products planning to initiate pivotal Phase 3 trials in 2012/2013 • Received E.U. conditional marketing approval in May 2012; launch roll-out September 2012 • Engaged Quintiles to hire Pixuvri dedicated sales force and MSL’s • $240mm potential peak sales in E.U., pricing –reimbursement studies support reasonable price • Pixuvri sales can help fund pipeline development and potentially lead to profitability by Q4-2014 • Highly selective JAK2 inhibitor (does not affect the JAK1 pathway, unlike competitors) • Positioned to be the first JAK2 inhibitor for MF patients with platelet counts below 100,000/uL, and potentially the second JAK2 inhibitor to enter the market after Incyte’s JAKAFI (peak worldwide sales of JAKAFI in MF are estimated at $1bn) - Only grade 3 side effect is diarrhea - Potential competitive advantage over JAKAFI in patients with thrombocytopenia (27% -33% of MF market) • Expected to enter pivotal Phase 3 trial in Q4 2012, target enrollment completion: 12 months • The only aminopeptidase inhibitor in advanced clinical development • Demonstrating results in Phase 2 trials • September 2012: Pixuvri launch in E.U. • Q4 2012: Expected to Initiate pacritinib Phase 3 trial in myelofibrosis • Q1-2013 Expected interim survival analysis on Opaxio (GOG212) phase 3 trial |
 6 Recent Strategic Initiatives Reduced Projected Monthly Cash Burn from an average of ~$5.5mm to ~$4.5mm 1 • Do not plan to resubmit Pixuvri US NDA in the near-term • Discuss options for PIX306 trial with FDA/EMA for earlier data (PFS) • Delaying initiation of tosedostat Phase 3 trial until 1 st line phase 2 results • Reduced advisory, consulting, legal fees and occupancy costs Initiation of Pacritinib Phase 3 Trial to Q4 2012 4 Pixuvri Pre-launch Activities in Support of a September 2012 Launch 3 • Engaged Quintiles to hire CTI’s Pixuvri dedicated sales force and medical specialists • EU General Manager, Market Access Specialist, and VP of Medical Affairs in place New Hires with Significant Industry and Operational Expertise 2 • Recently hired new CMO and EVP, Corporate Development Implemented Reverse Stock Split 5 • Current shares outstanding ~56.6 million • Engaged CRO that conducted COMFORT II trial • Planned initiation Q4 2012 |
 7 Executive Management Team Officers Position Experience James Bianco, MD Principal Founder, President & Chief Executive Officer, and Director • Chief architect of CTI’s portfolio strategy and has secured more than $1.6bn in operating capital • Previously was an Assistant Member in the clinical research division of the Fred Hutchinson Cancer Research Center and an Assistant Professor of Medicine at the University of Washington and Director Bone Marrow Transplant Unit SVAMC • Received B.S. in Biology and Physics from New York University and M.D. from Mount Sinai School of Medicine Steven Benner, MD, MHS EVP, Chief Medical Officer • Joined CTI as EVP and CMO in June 2012 • Previously was SVP and CMO at OncoMed and CMO at Protein Design Labs • Industry experience Bristol –Myers Squibb • Completed his training in medical oncology at Johns Hopkins University, where he also earned an M.H.S. in Clinical Epidemiology from the University’s school of Public Health Matt Plunkett. PhD EVP, Corporate Development • Former CFO California Institute Regenerative Medicine, iPierian • Previously Managing Director, Head West Coast Biotechnology, CIBC • 70 transactions, $4.3bn equity linked transactions, $1bn M&A • Received PhD. from UC Berkeley |
 8 |
 9 Pixuvri™: Addresses an Unmet Need Anthracyclines: most active class of chemotherapy • Cornerstone agents for NHL, AML, sarcoma, breast • Cumulative cardiac damage is dose limiting Pixuvri™ • Lacks structural motifs that lead to cardiotoxicity - Significant reduction in biochemical, echo-cardiographic and severe clinical symptoms (CHF) vs doxorubicin in randomized P2 clinical trial |
 10 Pixuvri™: Conditional Marketing Authorisation 3 rd /4 th line aggressive B-cell NHL |
 11 Lancet Oncology: May 2012 11 |
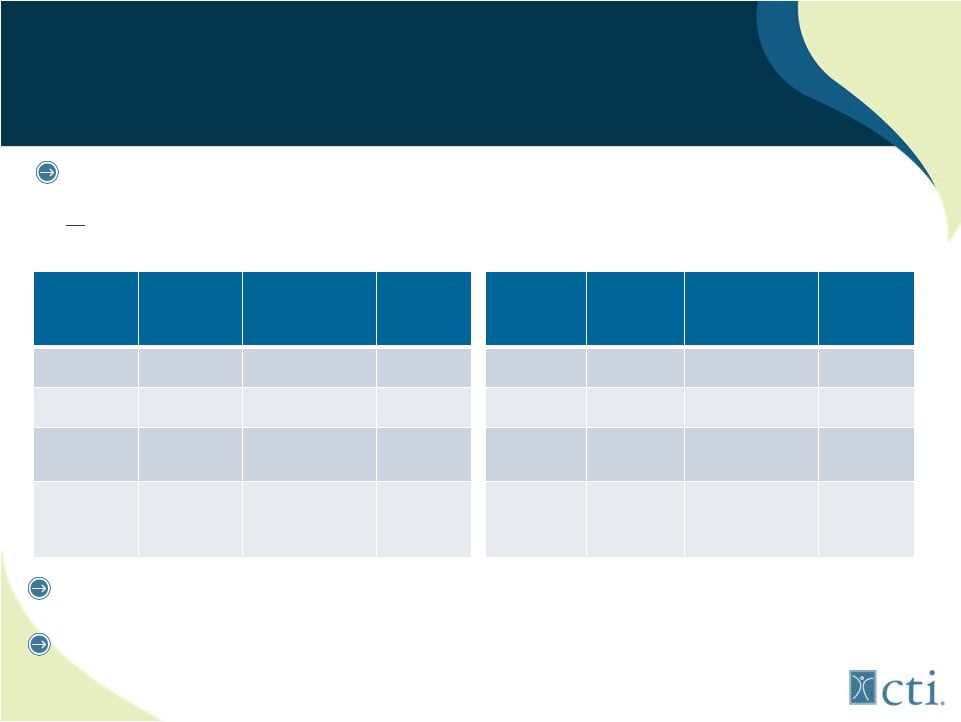 12 Pixuvri™: First to Market for r/r aggressive B-cell NHL Pixuvri (n=70) Comparator (n=70) P Value CR/CRu 20% 5.7% 0.021 ORR 40% 14.3% 0.001 PFS 5.3mos 2.6mos HR=0.60 0.005 OS 10.2mos 7.6mos HR=0.79 0.25 Pixuvri (n=50) Comparator (n=49) P value CR/CRu 28% 4.1% 0.002 ORR 48% 12.2% <0.001 PFS 5.7mos 2.8mos HR=0.50 0.002 OS 13.9mos 7.8mos HR=0.76 0.27 Basis: PIX301 trial, only randomized controlled trial in >3rd line aggressive NHL (n=140 patients) Manageable toxicities (neutropenia) Lower than expected incidence of cardiac events (7%) Intent to Treat Results On Label 3 rd /4 th line B-cell Results |
 Pixuvri™: EU Commercial Opportunity Market research supports potential peak penetration ~8,125 patients/year* Market access (pricing) strategy* • Quantitative benefit over physician choice • CR statistically correlative with OS • EU List Price corridor $29,500 - $35,000 - Allows for potential net ex-factory price $29,500 • Incremental cost/QALY (ICER) favorable Currently plan to launch in free market countries Q4 2012 • Nordic, Austria/Norway, Germany/UK/Netherlands • Balance of major market countries expected in 2013 13 *Market Access, Reimbursement and Market penetration projections provided by, UBC, and Kantar Research respectively |
 14 Pixuvri™: U.S. Regulatory Status FDA appeal: Office New Drugs (OND) provided path for resubmission - 2 Independent Radiographic Panel confirmed 1st panels results - Document Interim analysis was not conducted then PIX301 trial would be considered a positive study EU launch and pacritinib acquisition driving portfolio dynamics and priorities • NDA strategy under review • Considering potential to use PFS from PIX306 trial to provide data for accelerated approval while trial completes and survival data matures • Could potentially fulfill EMA-PMC and serve as 2 trial for US submission in 2014 • Plan to seek FDA and Scientific Advice on the above in 2013 nd nd 14 |
 A highly differentiated JAK2 inhibitor 15 |
 16 Pacritinib: Inhibits 2 important activating mutations (JAK2 + FLT3) |
 17 Initial Target Market: MF patients with thrombocytopenia (platelets <100 x10 9 ) Table 2: Maximum Restarting Doses for Jakafi After Safety Interruption* Current Platelet Count Maximum Dose When Restarting Jakafi Treatment * Greater than or equal to 125 X 109/L 20 mg twice daily 100 to less than 125 X 109/L 15 mg twice daily 75 to less than 100 X 109/L 10 mg twice daily for at least 2 weeks; if stable, may increase to 15 mg twice daily 50 to less than 75 X 109/L 5 mg twice daily for at least 2 weeks; if stable, may increase to 10 mg twice daily Less than 50 X 109/L Continue hold *Maximum doses are displayed. When restarting, begin with a dose at least 5 mg twice daily below the dose at interruption. JAKAFI™ (ruxolitinib label for patients with low platelets) |
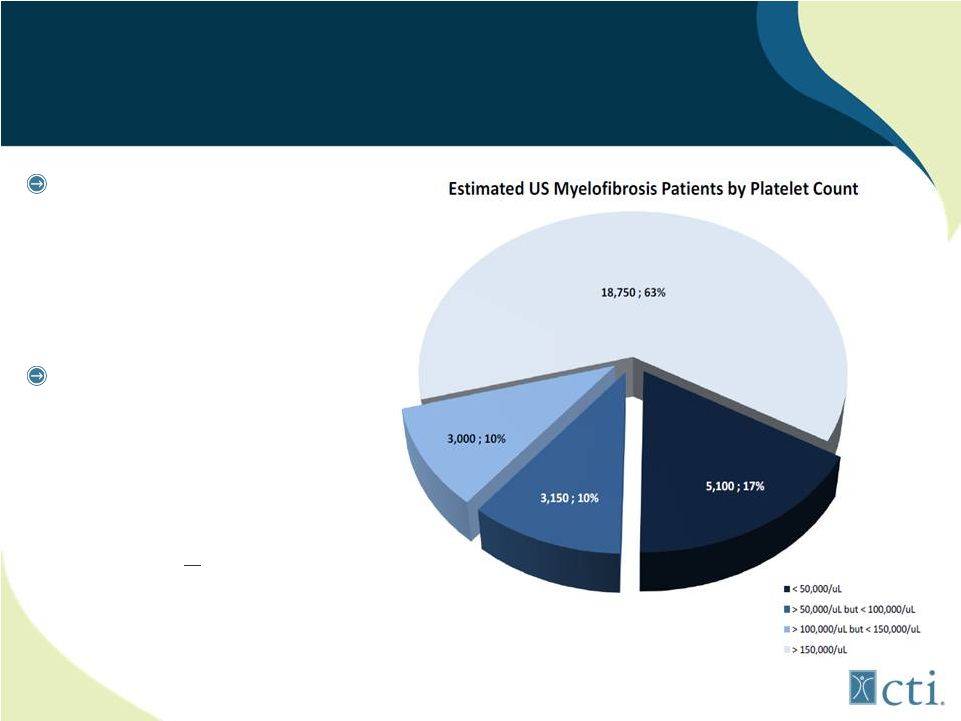 18 27% MF Patients Have Severe Thrombocytopenia JAK1/JAK2 inhibitors (ruxolitinib) associated with treatment emergent thrombocytopenia and anemia Pacritinib: selective JAK2 inhibitor lacks myelosupporession; potentially first to market for MF patients with low platelets (<100,000/uL) Source: MPN Research Foundation. |
 19 Pacritinib: Targets patient population unserved by ruxolitinib Ruxolitinib approval trials excluded patients <100,00/uL platelets - 93% COMFORT II had normal platelets, median COMFORT I, 244,000/uL - However, MF patients with low platelet counts are higher risk population Ruxolitinib study* (n=41) MF in patients with low platelets* • 75% tolerated up to 10mg bid by 24 weeks, but TET* still problematic *<100,000/uL > 50,000/uL ASCO June 2012 abstract #6630 preliminary data. TET: treatment emergent thrombocytopenia |
 20 Summary Clinical Trial Results JAK Inhibitors in MF Company Drug Stage SX Spleen >50% PE Spleen >35% MRI RBC JAK2 V617F* Platelet cutoff Side Effects Pacritinib JAK2-wt JAK2V617F FLT-3 Phase 3 (Phase 2 n=65)** >50% 42% 32% NR >30% NONE Nausea/diarrhea Ruxolitinib JAK1 JAK2-wt Marketed >50% 29-42% 14% NA 100,000 Anemia, thrombocytopenia, withdrawal syndrome CYT387 JAK1 JAK2-wt TYK2 Phase 3 (Phase 2 n=60) >50% 45% 50% NA 50,000 Thrombocytopenia, headaches, 1 st dose effect, neuropathy, lipase/amylasemia SAR302503 JAK2-wt JAK2V617F FLT-3 Phase 3 (Phase ½ n=59) >50% 39% 0% 21% 50,000 Nausea/diarrhea, anemia, thrombocytopenia, lipase/amylasemia Chart taken from Terferri, Supplement to Blood ASH 2012 *Median allele burden reduction at 24 cycles. ** Data combined from two Phase 2 trials. Pacritinib Demonstrates a Truly Differentiated Side-Effect Profile |
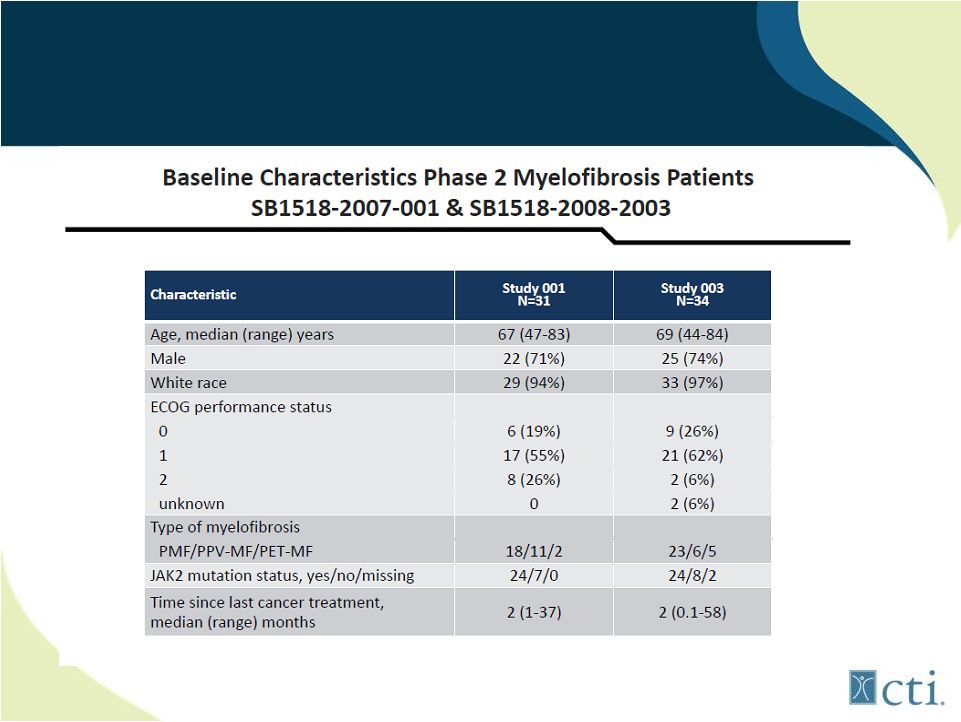 21 Pacritinib: Phase 2 Studies Source: ASH 2011. |
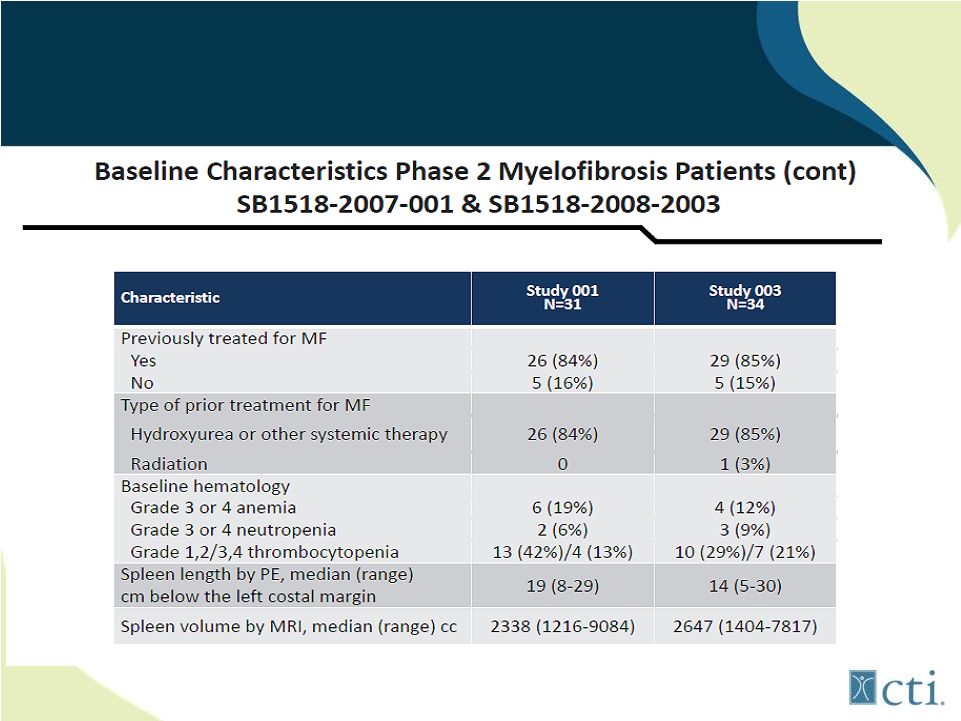 22 Pacritinib: Phase 2 Studies (combined) Source: ASH 2011. |
 23 Pacritinib: Phase 2 Efficacy (combined) 27 of 65 (42%) patients had response of 50% 14 of 65 (21%) had complete response with resolution of splenomegaly 8 of 26 (32%) treated with SB1518 had 35% reduction by MRI* Source: ASH 2011. *Among patients who had MRI measurements (8 patients did not have baseline MRI’s). |
 24 Pacritinib: Benefit Independent of Platelet Count Assessment Maximum decrease from baseline Patients with baseline platelet counts 150,000 N=36 All Evaluable Patients N=65 PE 50% 14 (39%) 27 (42%) Source: ASH 2011. * Reduction of splenomegaly was observed with similar frequency in patients with normal or low platelet counts |
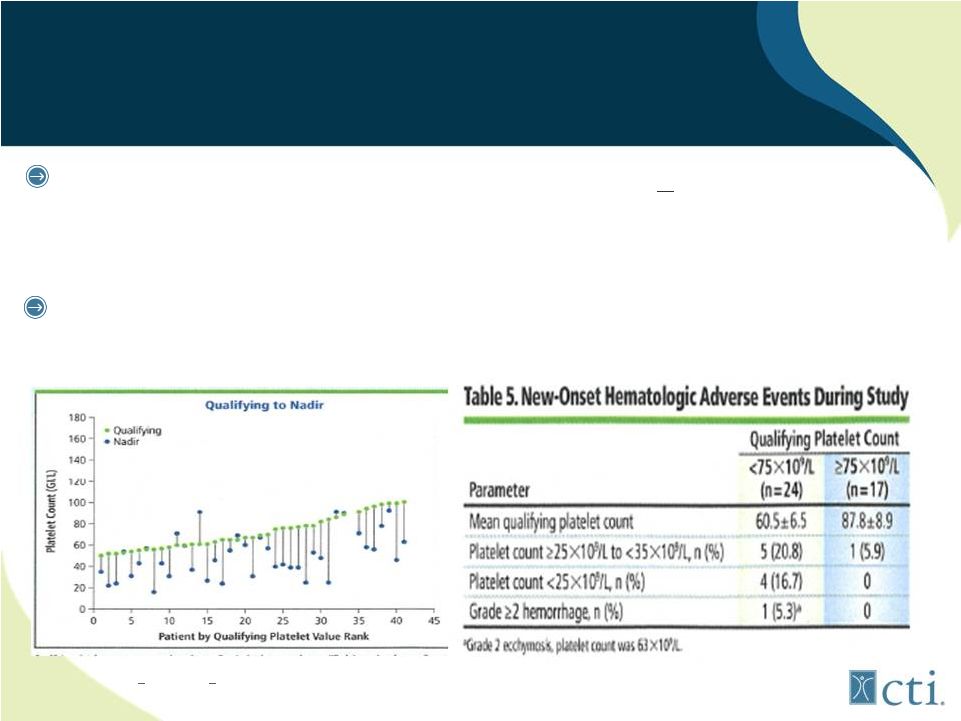
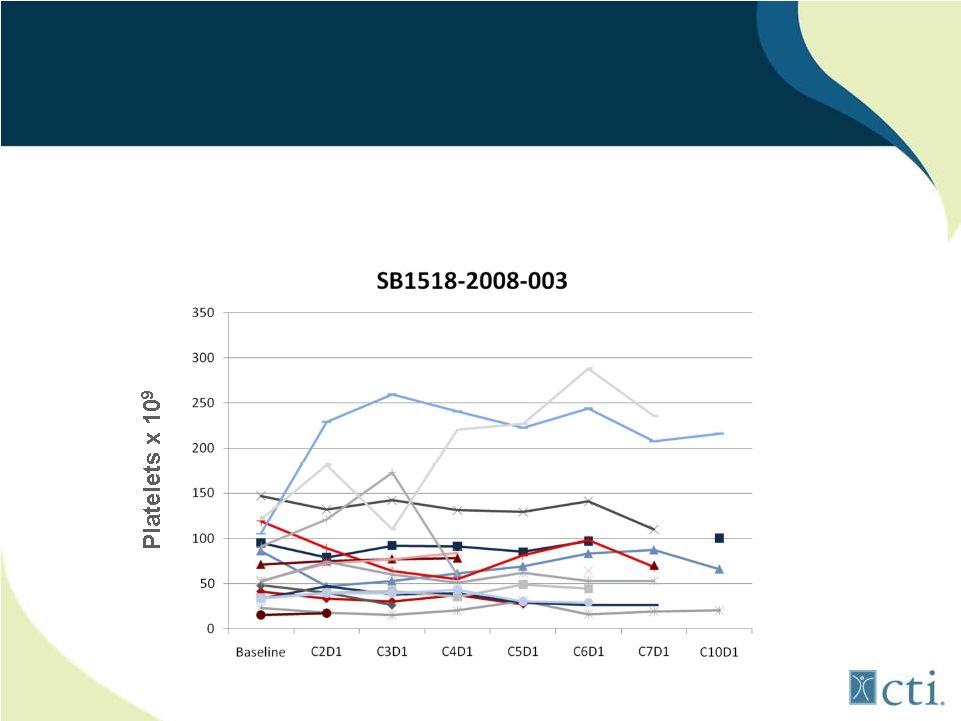 25 Lack of treatment emergent thrombocytopenia observed in patients with low starting platelet counts Pacritinib: Minimal Effect on Platelet Counts Source: Komrokji. R., et al |
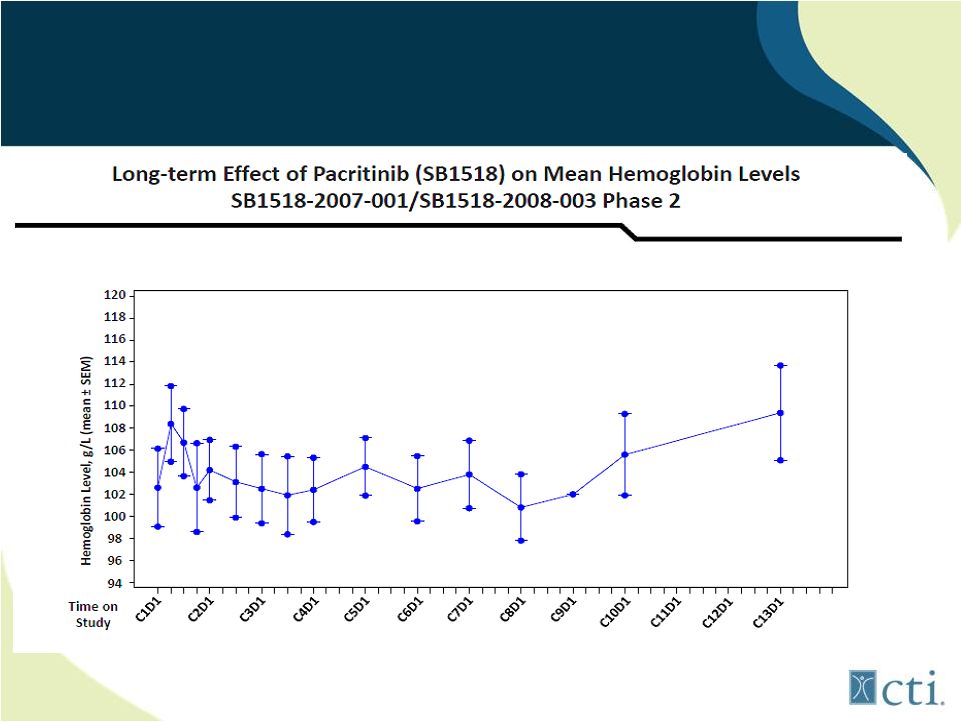 26 Pacritinib: Lack of treatment associated anemia Source: Komrokji. R., et al |
 27 Pacritinib: Durable Improvement in MF Related Symptoms Source: ASH 2011. |
 28 Contemplated Phase 3 Trial Designs Target Indication: • Patients with PMF, PPV-MF, PET-MF with splenomegaly and thrombocytopenia • Two phase 3 trials, each approximately 270 patients - Pacritinib Vs BAT - Pacritinib Vs BAT including patients on low dose ruxolitonib (5,10mg) with treatment emergent thrombocyopenia (<100,000/uL platelets) Target Initiation: Q4 2012 • Estimated enrollment time- 12 months + 6 months follow-up • Potential topline results mid 2014 Contemplated Phase 3 Trial Design(s) |
 29 Pacritinib: Certain Key Deal Terms Acquired molecule from S*Bio • S*BIO was established as a joint venture between The Singapore Economic Development Board Investments and Chiron Corporation as Singapore’s first fully- integrated, drug discovery company Upfront payment $30mm • $15 million cash • $15mm total in stock Up to $132.5mm in potential approval and sales milestones Sliding scale, low single digit royalty on sales |
 30 A new class of cancer targeted agents AADR inducers 30 |
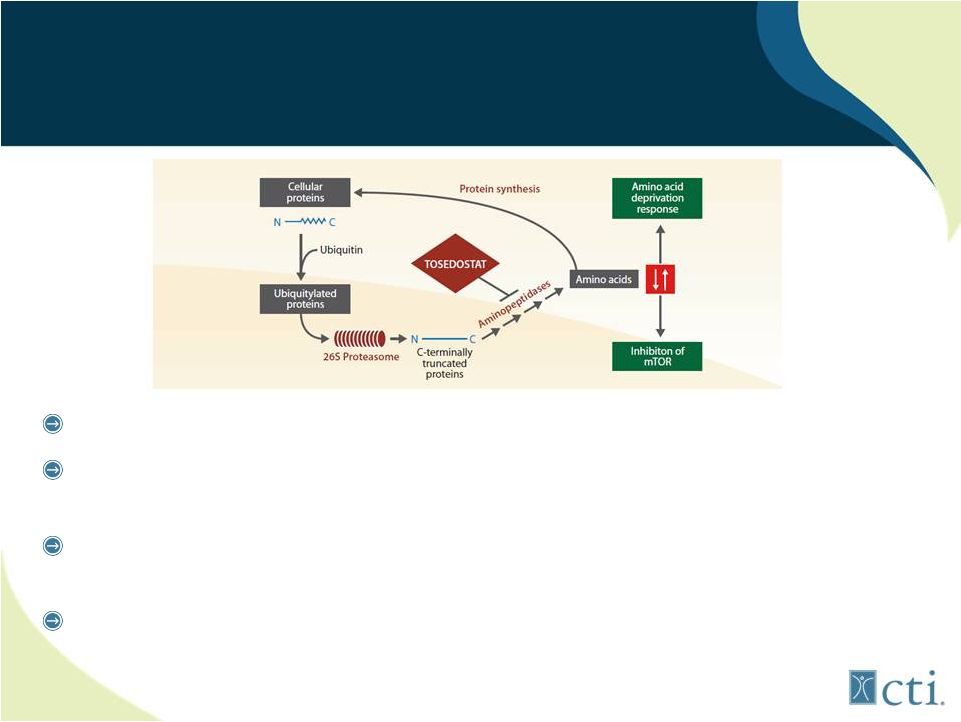 31 Tosedostat: Novel Mechanism of Action AADR Inducer Targets members of aminopeptidase family Induces genes characteristic of Amino Acid Deprivation Response (AADR) resulting in cell death AADR changes only seen in transformed (tumor) cells and not normal cells Demonstrated clinical activity across range of solid and liquid tumors |
 32 Tosedostat: OPAL Study Results Encouraging Phase 2 data - r/r elderly AML (n=73) • Oral once-a-day dosing for 6 months • 16 of 50 (32%) evaluable patients had bone marrow response - 9 (12%) of which were complete bone marrow responses • 9 of 25 (36%) patients who previously failed therapy with HMA had a bone marrow response Well-tolerated • Most common grade > 3 febrile neutropenia (29%), fatigue (21%) |
 33 Tosedostat: Phase 3 Study Data support pivotal trial: high risk MDS/secondary AML Phase 3 trial options: • Relapsed/refractory setting combination with LDAC - High risk heterogenous patient population - Several drugs currently targeting this population • 1 st line setting combination with HMA - Substantial investigator interest - Higher probability of success and shorter enrollment time Ongoing phase 2 data (1 st line HMA + tosedostat in MDS) will drive decision in 2013 |
 34 Partnership with Chroma Therapeutics Exclusive co-development and marketing rights in the Americas • Chroma Therapeutics has ROW rights Cost-sharing • CTI bears 75% of agreed upon direct development expenses • Registration trials aimed at US/EU regulatory approval Initial Payments • $5mm upon execution of license • $5mm upon initiation of first pivotal trial Development and sales based milestones Royalty rate based on net sales volume |
 35 Financial Overview |
 36 Financial Overview Exchanges / Tickers • NASDAQ: CTIC and MTA: CTIC.MI Market Capitalization • ~$125mm Shares Outstanding • ~56.6mm Cash • $24mm (1) Debt • None Projected Cash Burn • ~$4.5mm / month Cash Remaining • January 2013 Estimated 2013 Cash Burn (2) • $58mm net of Pixuvri product margin contribution Targeted Cash Flow Break Even (2) • Q4 2014 Financing Need • ~$75mm of additional financing is needed to reach potential CFBE and maintain a level of cash reserves Capital Structure and Financial Statistics (1) Estimated cash balance as of August 1, 2012. (2) Assumes Pixuvri price reimbursement and market penetration within corridors and percentages noted in Kantar Market Research and this presentation and also assumes burn rate stays in line with expectations |
 37 Expected News Flow Upcoming Planned Events • Pixuvri launch in the EU • Pacritinib phase 3 trial initiation in Myelofibrosis • Brostallicin- SABC triple negative breast cancer • OPAXIO interim survival analysis Phase 3 ovarian cancer trial • IQWiG innovation score assignment Upcoming publications • Journal Clinical Oncology Pacritinib in lymphoma • Blood Pacritinib in AML • Journal Clinical Oncology Pacritinib MF phase 2 • Journal Immunology SB1578 in RA |