 CORPORATE PRESENTATION San Francisco – January 13, 2015 © Copyright 2015 CTI BioPharma Corp. All rights reserved. Exhibit 99.1 Conquering Cancer. It’s in our blood.™ |
 Forward Looking Statement 2 This presentation includes forward-looking statements within the meaning of the Safe Harbor provisions of the Private Securities Litigation Reform Act of 1995. Such statements are subject to a number of risks and uncertainties, the outcome of which could materially and/or adversely affect actual future results and the trading price of CTI BioPharma's securities. Such statements include, but are not limited to, statements regarding CTI BioPharma’s expectations with respect to the development of CTI BioPharma and its product and product candidate portfolio, and expectations concerning the benefits of our recently expanded rights to tosedostat, the anticipated reporting of top-line results from a Phase 2 trial of tosedostat, the development of a registrational strategy and development program for tosedostat, the anticipated completion of enrollment in PERSIST-2 in mid- 2015, potential release of top-line data from PERSIST-1 late in the first quarter of 2015, the potential submission of a rolling NDA to the FDA for pacritinib in late 2015, the advancement of a pacritinib development program in other hematologic malignancies in the second half of 2015, the initiation of a first-line study in elderly patients with AML in the second half of 2015, enrollment projections and expected outcome of the PIX306 post-marketing commitment study, liquidity, revenue and expense projections, the anticipated achievement of partner-related milestones and product and geographic sales, initiation of investigator-sponsored trials, the potential commencement of a pivotal program for tosedostat in early 2016, and the execution of CTI BioPharma’s strategy to commercialize globally and its strategy to continue the growth in aNHL in 2015. Risks that contribute to the uncertain nature of the forward-looking statements include, among others, risks associated with the biopharmaceutical industry in general and with CTI BioPharma and its product and product candidate portfolio in particular including, among others, risks associated with the following: that CTI BioPharma cannot predict or guarantee the pace or geography of enrollment of its clinical trials, that CTI BioPharma cannot predict or guarantee the outcome of preclinical and clinical studies, that CTI BioPharma may not obtain favorable determinations by other regulatory, patent and administrative governmental authorities, that CTI BioPharma may experience delays in the commencement of preclinical and clinical studies, risks related to the costs of developing pacritinib and CTI BioPharma’s other product candidates, and other risks, including, without limitation, competitive factors, technological developments, that CTI BioPharma may not be able to sustain its current cost controls, further reduce its operating expenses or achieve its anticipated liquidity, revenue and expense projections, that CTI BioPharma may not achieve previously announced goals and objectives as or when projected, that CTI BioPharma’s average net operating burn rate may increase, that CTI BioPharma will continue to need to raise capital to fund its operating expenses, but may not be able to raise sufficient amounts to fund its continued operation, as well as other risks listed or described from time to time in CTI's most recent filings with the Securities and Exchange Commission on Forms 10-K, 10-Q and 8-K. Except as required by law, CTI does not intend to update any of the statements in this presentation upon further developments. |
 Accomplishments in 2014 3 Entered into a $133 million* collaboration for PIXUVRI to expand commercial potential of product Reported preclinical data highlighting pacritinib’s potential in other indications beyond myelofibrosis; ISTs in additional indications underway (AML, CMML, MDS) Secured worldwide rights to tosedostat and retired $209mm in potential payments and royalties to Chroma Grew revenue by 71% to approximately $60mm** reducing development and capital requirements risk Advanced pacritinib Phase 3 program in myelofibrosis; completed 1 st Phase 3 trial and received $20mm from Baxter Building Shareholder Value * Milestone and upfront payments excluding royalties. ** Unaudited 2014 financial results. |
 2015 Key Objectives Pacritinib 4 • Report top-line results from an investigator-sponsored Phase 2 AML trial • Based on clinical data, define registration directed strategy in AML Tosedostat • Generate sufficient E.U. sales to achieve a net positive contribution margin in 2015 • Complete enrollment of PIXUVRI post-marketing commitment study • Together with Servier, support the initiation of additional investigator- sponsored combination studies • Report top-line results from PERSIST-1 Phase 3 MF trial late 1Q-2015 • Complete enrollment of PERSIST-2 mid-2015 • Begin rolling submission of NDA late 2015 • Advance development program in other hematologic malignancies PIXUVRI |
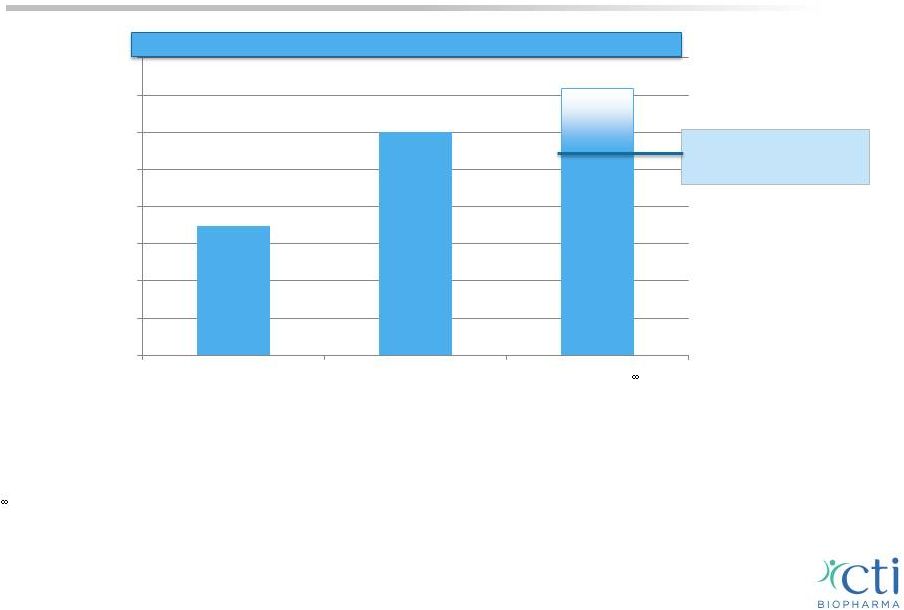 5 Preliminary 2015 Revenue Projections Projected range of $55mm - $72mm Preliminary Year-Over-Year Revenue Comparison* Actual $- $10 $20 $30 $40 $50 $60 $70 $80 2013 2014 E* 2015E * *This estimate is unaudited, preliminary and may change. CTI BioPharma and its auditors have not completed our normal annual closing and review procedures for the year ended December 31, 2014, and there can be no assurance that final results for the year ended December 31, 2014 will not differ materially from our estimates, including as a result of annual closing procedures or review adjustments and other developments that may arise between now and the time the financial results for this period are finalized and other factors described in “Forward Looking Statements” at the beginning of this presentation and in CTI BioPharma’s most recent filings with the SEC. Investors are cautioned not to rely on preliminary financial information. Revenue projections are subject to a number of risks and uncertainties and may change. Actual revenue for 2015 may vary based upon a number of factors, including the ability to complete and the timing of completion of events triggering milestone payments, the degree of market acceptance and other factors described in “Forward Looking Statements” at the beginning of this presentation and in CTI BioPharma's most recent filings with the SEC. Investors are cautioned not to rely on financial projections. |
 Strategic Approach to Commercialize Globally U.S. Europe ROW 3 core products focused on overlapping heme/onc patients, prescribers and payors *In collaboration with BAXTER in U.S. 6 Develop Full Commercial Capabilities For pacritinib*; PIXUVRI and tosedostat Core Markets [North Western Europe] Commercializing PIX and future products SERVIER for PIX in other markets BAXTER for pacritinib Collaboration For PIXUVRI, pacritinib and tosedostat • Build comprehensive field force, MSLs • Reimbursement specialists • Cost-effective commercial structure • CTILS Medical Affairs & Reimbursement specialists • SERVIER for PIXUVRI • BAXTER for pacritinib • Partner for tosedostat ROW |
 Building a Successful Oncology Business Therapeutic Focus: Novel Targeted Therapies 7 Lymphoma Leukemia MPNs NHL and CLL AML and MDS MF and PV Blood-Related Cancers |
 CTI BioPharma Opportunity 8 *Under the license and collaboration agreement with Servier, monetary amounts are generally denoted in euros, while the corresponding U.S. dollar amounts are based upon conversion on September 12, 2014. pacritinib tosedostat High (>50%) CR rates in elderly AML, MDS in combination with HMA or LDAC Acquired worldwide rights, extinguished potential $209 million obligations Targeting registration directed Phase 3 trial in early 2016 Fast Track designated product candidate Phase 3 top-line data expected late in 1Q’15; Second trial enrolling, FDA-SPA Data beyond myelofibrosis expected in 2015 Collaboration with BAXTER International of up to ~$360 million 1st approved therapy in the E.U. for relapsed aggressive B-cell NHL beyond 2 nd line; ongoing confirmatory Phase 3 trial Targeting cash flow positive operations in 2015 Collaboration with SERVIER of up to ~$133* million |
 Pacritinib |
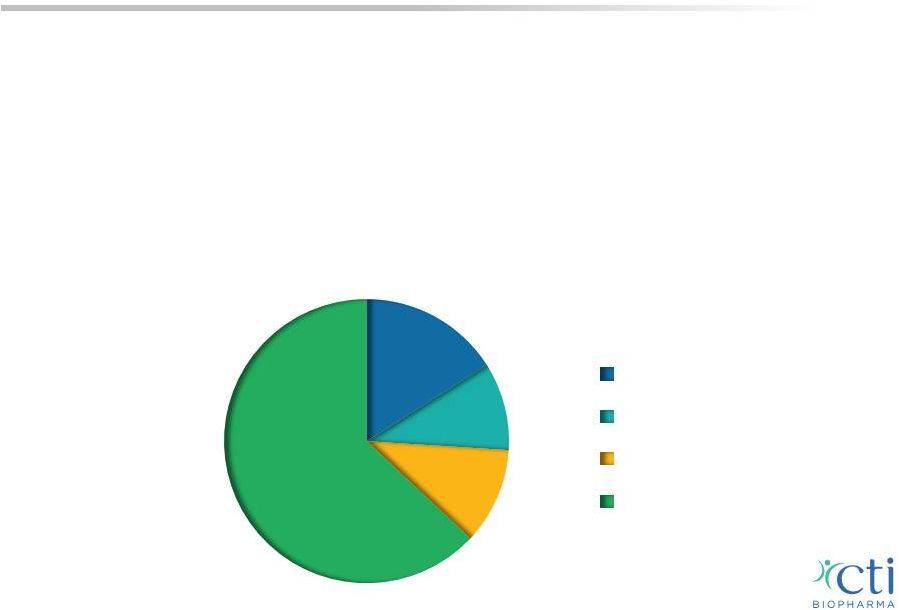 Myelofibrosis: A Chronic Disease • Malignant bone marrow disorder triggers an inflammatory response, scarring the bone marrow and limiting its ability to produce blood cells prompting the spleen and liver to compensate • Symptoms include: enlargement of the spleen, anemia, thrombocytopenia, extreme fatigue, pain, severe itching and GI side effects • Estimated prevalence ~18,000 patients in the U.S., 1 ~37% of whom are thrombocytopenic with platelet counts below 150,000/µL 2 1 Based on Mesa R, ASH 2012 poster 2 Visani et al. 1990 Br J Haematol; Caramazza et al. 2011 Leukemia; Tam et al. 2009 JCO <50,000/µL >50,000/µL but <100,000/µL >100,000/µL but <150,000/µL >150,000/µL 10% 11% 16% 63% 10 |
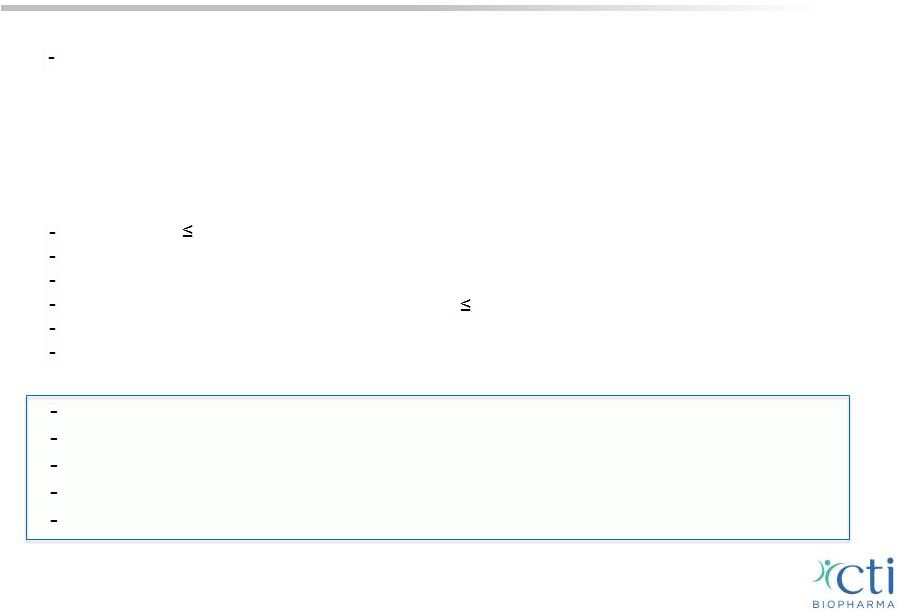 Pacritinib: Market Opportunity in MF • Approved drug Jakafi ® /Jakavi ® (ruxolitinib) Total 2014 U.S. net sales estimated at $350-360mm 1; $600mm - $1bn 2015 run-rate worldwide • Treatment emergent thrombocytopenia and anemia are limitations of most JAK1/JAK2 inhibitors, including ruxolitinib • Market research estimates 1/3 of patients do not respond adequately to ruxolitinib; dose adjustments used to “manage the drug” Low dose ( 10mg bid) provides minimal spleen reduction/symptom control 2 If ruxolitinib therapy is interrupted, symptoms return within 7 days 2 >50% of U.S. patients on low dose (5-10mg) Average time on drug 6-8 months at doses 20 mg/day 3 Minimal activity JAK/STAT pathway at lower doses Survival correlates with spleen response Ideal product profile for new MF market entrant would include the following: Efficacy not dose-limited by tolerability concerns Ability to treat entire spectrum of patients with MF Ability to maintain dose over a long period of time Demonstrated long-term symptom control and splenomegaly control No overlapping myelosuppression with other MF directed targeted agents 11 1 Incyte 3Q’14 financial results conference call 2 Verstovsek S, European Focus on MPNs & MDS 2014, Tips on using rux in everyday practice. 3 IMS data. |
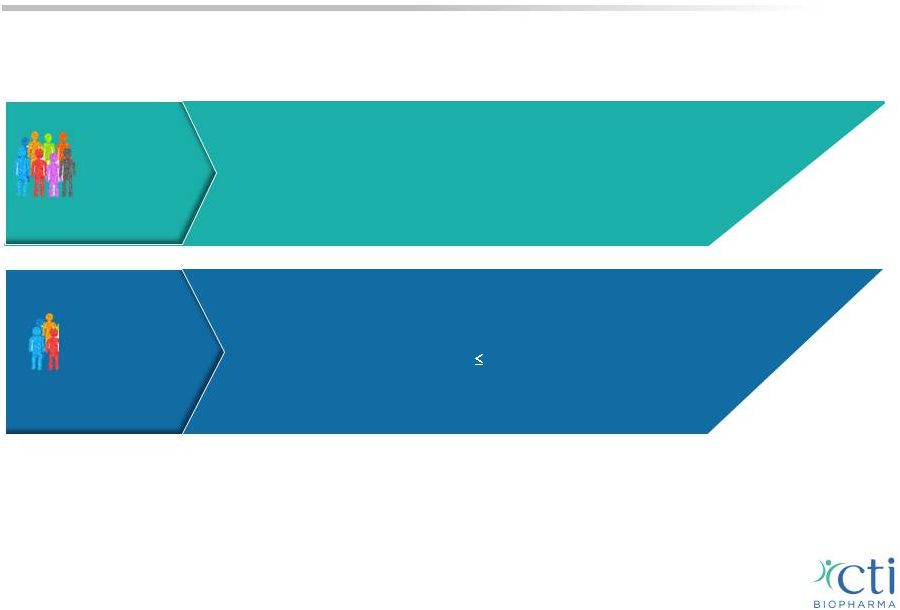 Evaluating Pacritinib in All MF Patients 12 Fast Track Designation; Global Registration Program • PERSIST-1 compares pacritinib to BAT, excludes ruxolitinib • Fully enrolled; Top-line results expected late in 1Q-2015 • Most inclusive randomized Phase 3 study All MF Patients • PERSIST-2 designed to demonstrate full dose pacritinib potential superior symptom and disease control over titrated dose ruxolitinib or other BAT in patients with disease or drug related thrombocytopenia ( 100,000/µL) • Conducted under Special Protocol Assessment (SPA) • Enrollment completion expected in mid-2015 MF Patients with Low Platelets Pacritinib Phase 3 trials incorporate FDA and EMA accepted endpoint • We believe the design is comparable to Incyte’s COMFORT II with the exception of inclusion INT-1, no exclusion on low platelet counts and primary endpoints at 24 weeks • INT-1, 2 High risk, no restrictions on platelet counts |
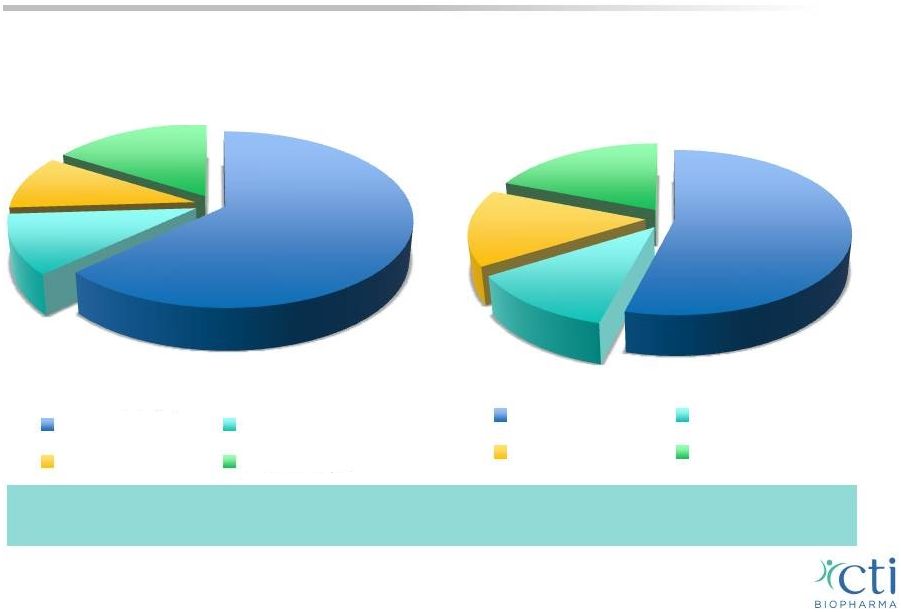 PERSIST-1 Representative of MF Population 13 1 Visani G et al. Br J Haematol 1990;75:4. 2 Caramazza D et al. Leukemia 2011;25:82. 3 Tam C et al. J Clin Oncol 2009;27:5587. 4 Based on blinded data. The duration of therapy is comparable across patient subsets with thrombocytopenia at baseline or patients with normal platelet counts at baseline. Distribution (%) Platelets in MF Patients Projected Distribution (%) Platelets in MF Patient in PERSIST-1 4 63 11 10 16 >150,000/µL 100 - 150,000/µL 50-100,000/µL <50,000/µL >150,000/µL 100 -150,000/µL 50-100,000/µL <50,000/µL 54 12 15 19 1-3 |
 14 March 2014: Open enrollment for PERSIST-2 March 2015: Expect to report PERSIST-1 top-line results PERSIST Program Phase 3 Trial - Timeline June 2012: Acquired pacritinib from S*BIO for $30mm January 2013: PERSIST-1 trial initiated October 2013: Reached agreement with FDA on SPA for PERSIST-2 November 2013: BAXTER collaboration 460mm upfront July 2014: PERSIST-1 enrollment complete; $20mm milestone |
 Pacritinib: Unique Kinome Profile 1 • Pacritinib is an oral multikinase inhibitor with specificity for kinases in several unrelated families • The inhibition of IRAK1 and c-fms (CSF1R) may contribute to its anti-inflammatory activity and early onset of symptomatic benefit in MF • Suggests its potential therapeutic utility in AML, MDS and, particularly CMML, CLL due to its potent inhibition of c-fms, IRAK1, JAK2 and FLT3 1 Singer J, et al., Abstract #1874 American Society of Hematology Annual Meeting 2014. |
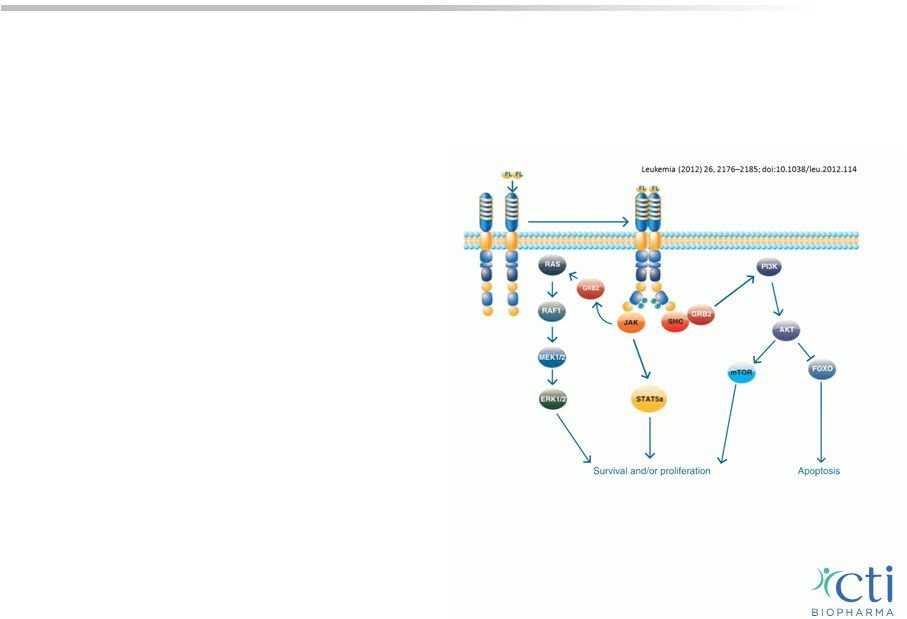 Pacritinib: AML Target Indication • Able to target multiple pathways, including suppression of microenvironmental tumor interactions that are key to overcoming longer-term drug resistance in this disease • FLT3 strong prognostic factor for AML; presents in 33% of patients 1 • Pacritinib also inhibits JAK2, which is independently associated with resistance to FLT3 inhibition and a poor prognosis in AML 2 • Phase 2 trial in patients with relapsed AML and FLT3 mutations is currently underway; a first-line study in elderly patients with AML is expected to be initiated in 2H-2015 16 1 Brunet S (2012) Impact of FLT3 Internal Tandem Duplication on the Outcome of Related and Unrelated Hematopoietic Transplantation for Adult Acute Myeloid Leukemia in First Remission: A Retrospective Analysis. J Clin Oncol 30(7):735-41. 2 Hart S et al., Pacritinib (SB1518), a JAK2/FLT3 inhibitor for the treatment of acute myeloid leukemia. Blood Cancer Journal (2011) 1, e44; doi:10.1038/bcj.2011.43. Signaling pathways initiated by FLT3 ligand activation of the FLT3 receptor |
 • Oral JAK2/FLT3 inhibitor that lacks treatment emergent myelosuppression associated with marketed JAK1/JAK2 agent - Potential to demonstrate superior disease and symptom control over titrated doses of ruxolitinib - May be used in combination regimens w/o overlapping myelosuppression - Potential for longer duration of therapy than observed with JAK1/JAK2 inhibitors • Durable improvement in splenomegaly (24+ months) and MF-related symptoms • Unique kinome profile with potential application in a spectrum of blood- related cancers Pacritinib: Key Takeaways 17 • Patent protection through 2026 / 2029 (+ possible patent term extension) • Orphan drug designation in the U.S. and E.U. |
 • CTI and BAXTER: Joint commercialization, profit split in U.S. • BAXTER: Exclusive rights to commercialize in all indications outside U.S., BAXTER generally responsible for 75% of global development • Potential total value: $362mm; potential milestones of up to $127mm through regulatory submission - $60mm upfront payment, includes $30mm equity investment in CTI - $67mm potential milestone progress payments expected through 2015 ($20mm received in conjunction with completion of PERSIST-1 enrollment) • CTI to receive tiered high single-digit to mid-teen percentage royalties based on net sales for myelofibrosis; higher double-digit royalties for all other indications Summary of Financial Terms Pacritinib: Collaboration with Baxter Announced November 2013 18 |
 PIXUVRI® |
 PIXUVRI: Fulfilling An Unmet Clinical Need 20 1 st LINE 2 nd LINE 3 rd LINE+ • Typically anthracycline- based treatment R-CHOP • Curative in 50% - 55% of patients 1 • Cardiac toxicity prevents re-use of anthracyclines in relapsed patients • Intensive (toxic) non-anthracycline based salvage therapy (R-DHAP), +/- ASCT • Also, ESHAP, ICE are commonly used 2 • 95% of patients will relapse after 2 nd -line therapy 3 • Palliative care (symptom control) or clinical trials 1 Adult Non-Hodgkin Lymphoma Treatment (PDQ®), Aggressive DLBCL, National Cancer Institute. Available at www.cancer.gov. Last accessed January 2015. 2 Papadatos-Pastos D, et al. Expert Rev. Hematol. 2013; 6(1), 25-33. 3 Hagemeister FB. Cancer Chemother Pharmacol 2002;49(suppl 1):S13-20. |
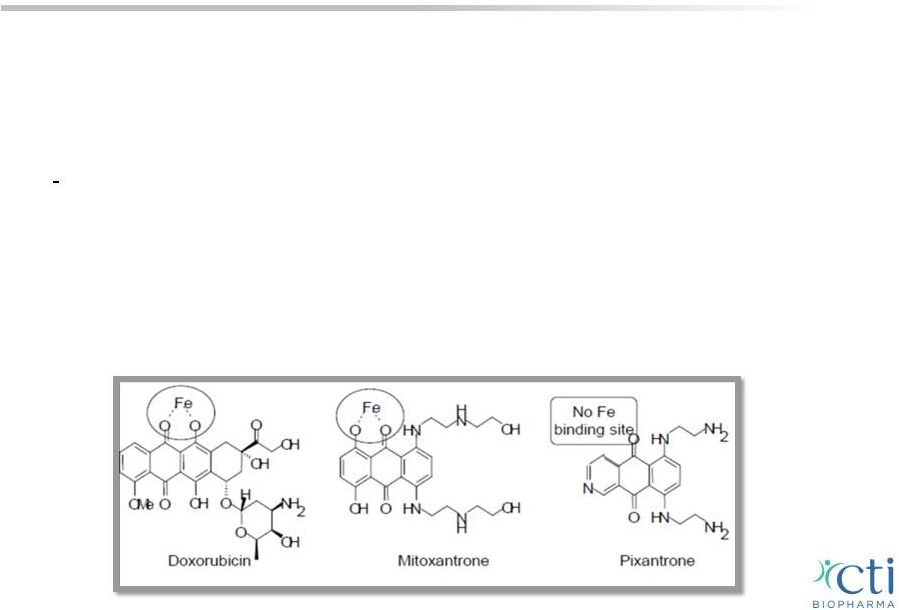 • Novel MOA: DNA alkylation/mitotic instability • Works in anthracycline-resistant aggressive B-cell NHL • No label warning for cardiotoxicity or cumulative dose restrictions PIXUVRI: Overview One of the most active agents in R/R aggressive B-cell NHL where there was no standard therapy 21 1 Beeharry N, et al., AACR 2013. PIXUVRI appears to be impairing chromosomal segregation during mitosis, thereby generating loss of genetic material in daughter cells, an abnormality, which is ultimately lethal, and that PIXUVRI would likely be effective in cells resistant to other cytotoxic agents such as doxorubicin. 1 |
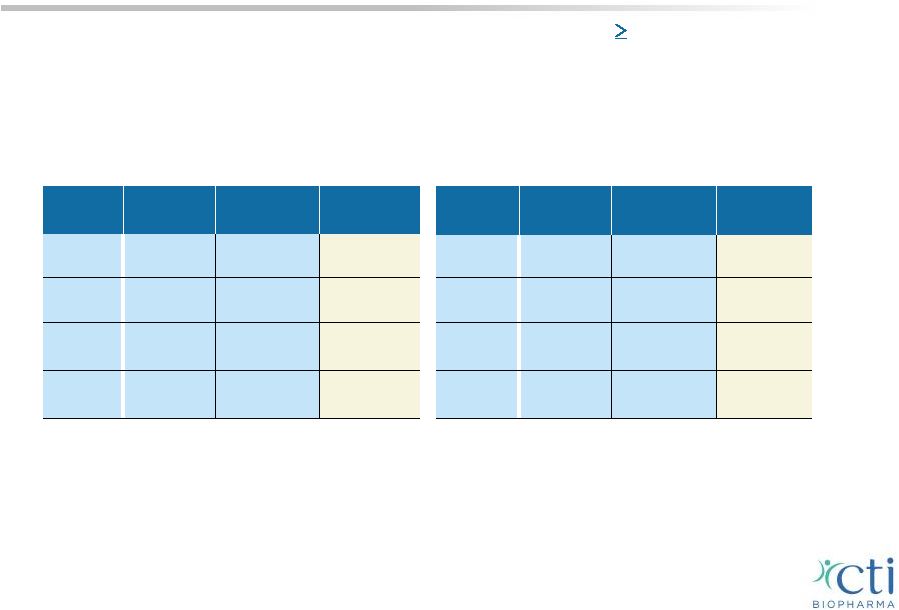 PIXUVRI: Demonstrated Benefit in Phase 3 Patient Population • Heavily pre-treated patients; median of 3 prior treatments • Median age was 60 years Safety profile • Predictable and manageable side effects; uncomplicated neutropenia - No severe nausea, vomiting or hair loss Administration • Pixantrone (50 mg/m 2 iv.) weekly infusion on days 1, 8 and 15 of a 28-day cycle, for maximum of 6 cycles on an outpatient basis 22 PIXUVRI (n=70) Comparator (n=70) p-value CR/CRu 24% 7.1% 0.009 ORR 40% 14.3% 0.001 PFS 5.3 mos. 2.6 mos. 0.005 (HR=0.60) OS 10.2 mos. 7.6 mos. 0.251 (HR=0.79) ITT Population PIXUVRI (n=50) Comparator (n=49) p-value CR/CRu 28% 4.0% 0.002 ORR 48% 12.2% <0.001 PFS 5.8 mos. 2.8 mos. 0.002 (HR=0.50) OS 13.9 mos. 7.8 mos. 0.275 (HR=0.76) 3 rd /4 th Line B-cell Results PIX301* - Only randomized controlled Phase 3 trial in 3 rd -line aggressive B-cell NHL (n=140 patients) * PIX301 (EXTEND Study) published in Lancet Oncology, May 2012. |
 PIXUVRI: Approved as Monotherapy by EMA • 1 st and only approved monotherapy in the E.U. for aggressive B-cell NHL 3 rd /4 th line • 2 nd salvage (3 rd line) = 2 nd chance at potentially curative ASCT* • Durable remissions and disease control (3 rd /4 th line) in non-SCT eligible • Label expansion study underway with potential as basis for filing U.S. NDA • Prospective investigator-sponsored studies underway evaluating potential in combination-salvage regimens for various hematological malignancies 23 *Autologous Stem Cell Transplant |
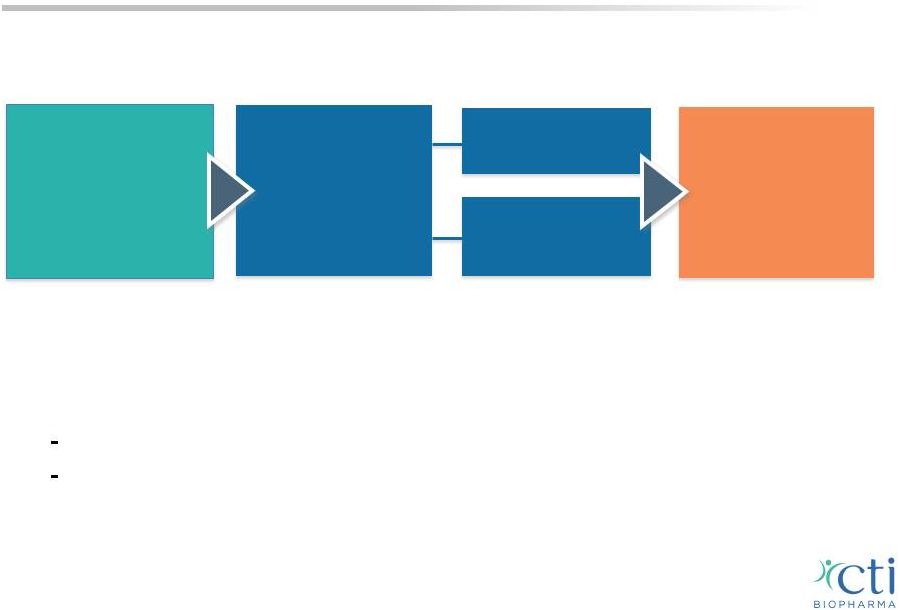 PIXUVRI: PIX306 Study Eligibility Criteria Relapse after CHOP-R therapy or an equivalent regimen and are ineligible for stem cell transplant 1:1 Randomization n = ~220 Primary Endpoint PFS Secondary Endpoints CR ORR OS Gemcitabine* Rituximab Pixantrone* Rituximab Post-marketing Commitment Phase 3 Study • Sites currently open in U.S. and E.U. • Target enrollment completion 4Q-2015; data in 2016 • If positive, could extend label to 2 nd line combination therapy Expected to increase E.U. addressable market by 12,000* patients Potential to provide basis for U.S. NDA 24 *Based on Kantar forecast model. |
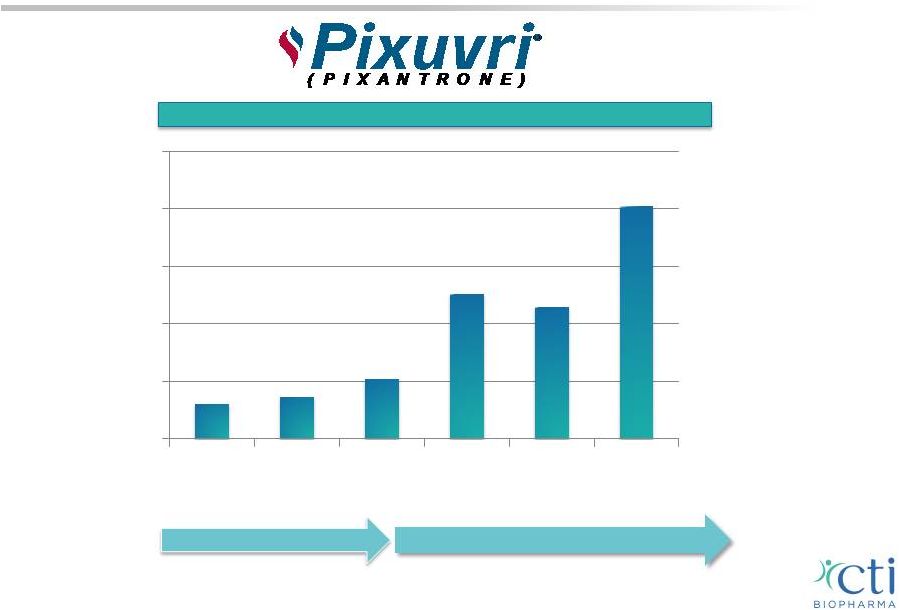 PIXUVRI EU Sales Since Launch 25 Net PIXUVRI Sales by Quarter ($ in millions) 2014 2013 Germany UK Netherlands Focus on Reimbursement 0 0.5 1 1.5 2 2.5 Q2 Q3 Q4 Q1 Q2 Q3 |
 PIXUVRI: Maximizing the European Commercial Opportunity • Exclusive license and collaboration agreement with SERVIER to develop and expand commercial reach • Reimbursed in Austria, England/Wales, Germany, Netherlands • Standard of care per treatment guidelines in certain E.U. countries • Weighted average pricing: €16,500 or $20,000/patient/year 26 10+ Countries ~5,000 Current eligible patients |
 PIXUVRI Collaboration with SERVIER Announced September 2014 • Exclusive license and collaboration agreement to develop and expand commercial reach of PIXUVRI • CTI retains 100% U.S. rights, continues its commercial presence in UK, Germany/Austria, Nordics, and via distribution partners in Turkey, Israel • SERVIER gains rights in ROW 27 • Potential deal value: ~$132mm • ~$18mm upfront • ~$63mm in potential clinical/regulatory milestone payments - ~$12mm tied to PIX306 Phase 3 trial enrollment • ~$52mm in potential sales milestone payments • CTI receives tiered royalty payments: low double-digit to mid-twenties on net sales in SERVIER’s territories Summary of Financial Terms |
 Continued Growth Expected in aNHL in 2015 • Continue to build awareness and educate HCPs • Expand field force; medical science liaisons key • Growing body of evidence on potential for combination studies • Limited near-term competition expected 28 |
 Tosedostat |
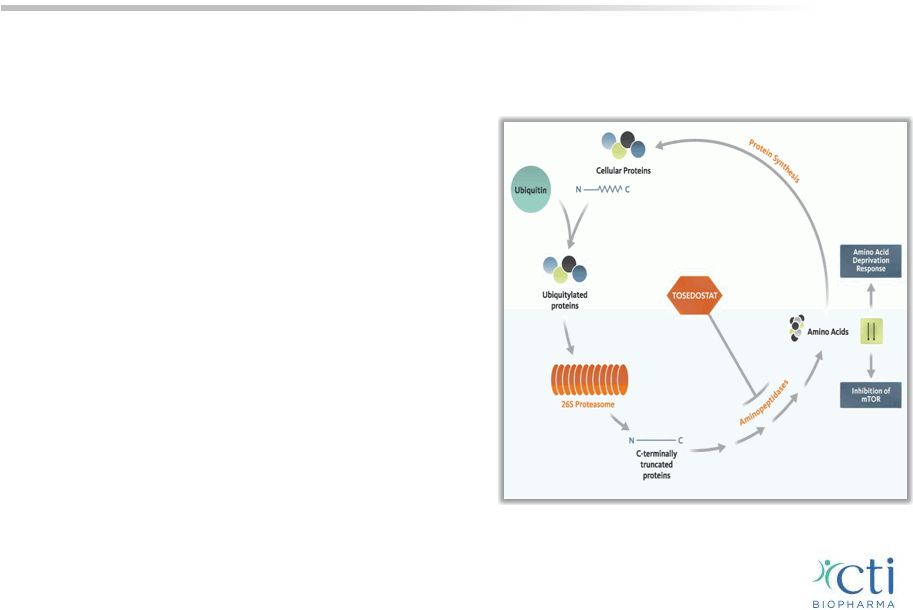 Tosedostat Novel Treatment for Blood-Related Cancers: MDS & AML • Oral, once-daily, aminopeptidase inhibitor interferes with protein recycling, prevents breakdown of peptides into amino acids necessary for tumor cell survival • Synergy with targeted agents (HMA, proteosome inhibitors) or chemotherapy • Encouraging CR rates (54%) and median OS (12 months) in combination with HMA or cytarabine in 1 st line elderly high risk AML/MDS • Several Phase 2 ISTs in AML or MDS underway 30 |
 Financial Overview Capital Structure and Financial Statistics Exchanges NASDAQ and MTA: CTIC Market Capitalization* ~$417mm Shares Outstanding** ~177mm Cash as of November 30, 2014 $82mm Debt $20mm *As of January 9, 2015 stock price of $2.36 per share. ** As of December 15, 2014. 31 |
 Why Invest in CTI BioPharma? 32 Biopharmaceutical company with a marketed oncology drug in the E.U. Strategic partner provides validation, financial resources and commercial support for high-potential JAK2/FLT3 inhibitor Phase 3 program Building value through development of late-stage pipeline, with emphasis in areas that address unmet medical needs of patients with blood-related cancers Commitment to manage expenses within guidance Stock with good liquidity and high potential near-term catalysts |
 NASDAQ & MTA: CTIC THANK YOU! |