Use these links to rapidly review the document
TABLE OF CONTENTS
TABLE OF CONTENTS
Table of Contents
NaPro BioTherapeutics, Inc. and Subsidiaries Financial Statements Index to Consolidated Financial Statements
NaPro BioTherapeutics, Inc. and Subsidiaries Table of Contents
SCHEDULE 14A
(Rule 14a-101)
INFORMATION REQUIRED IN PROXY STATEMENT
SCHEDULE 14A INFORMATION
Proxy Statement Pursuant to Section 14(a) of
the Securities Exchange Act of 1934 (Amendment No. )
| Filed by the Registrantý | ||
Filed by a Party other than the Registranto | ||
Check the appropriate box: | ||
ý | Preliminary Proxy Statement | |
o | Confidential, for Use of the Commission Only (as permitted by Rule 14a-6(e)(2)) | |
o | Definitive Proxy Statement | |
o | Definitive Additional Materials | |
o | Soliciting Material Pursuant to §240.14a-12 | |
NAPRO BIOTHERAPEUTICS, INC. | ||||
(Name of Registrant as Specified In Its Charter) | ||||
(Name of Person(s) Filing Proxy Statement, if other than the Registrant) | ||||
| Payment of Filing Fee (Check the appropriate box): | ||||
o | No fee required | |||
ý | Fee computed on table below per Exchange Act Rules 14a-6(i)(1) and 0-11 | |||
(1) | Title of each class of securities to which transaction applies: | |||
N/A | ||||
| (2) | Aggregate number of securities to which transaction applies: | |||
N/A | ||||
| (3) | Per unit price or other underlying value of transaction computed pursuant to Exchange Act Rule 0-11 (set forth the amount on which the filing fee is calculated and state how it was determined): | |||
The filing fee was determined based upon the estimated aggregate purchase price of $68,499,000 to be paid to the Company for its paclitaxel business, calculated by taking the cash payment of $71,700,000 minus an estimated reduction of $3,201,000 for an inventory adjustment to reflect the Company's actual inventory as of the closing. The filing fee is calculated at $80.90 per million of aggregate purchase price. | ||||
| (4) | Proposed maximum aggregate value of transaction: | |||
$68,499,000 | ||||
| (5) | Total fee paid: | |||
$5,542 | ||||
o | Fee paid previously with preliminary materials. | |||
o | Check box if any part of the fee is offset as provided by Exchange Act Rule 0-11(a)(2) and identify the filing for which the offsetting fee was paid previously. Identify the previous filing by registration statement number, or the Form or Schedule and the date of its filing. | |||
(1) | Amount Previously Paid: | |||
| (2) | Form, Schedule or Registration Statement No.: | |||
| (3) | Filing Party: | |||
| (4) | Date Filed: | |||
NAPRO BIOTHERAPEUTICS, INC.
4840 Pearl East Circle, Suite 300W
Boulder, Colorado 80301
September , 2003
To our stockholders:
You are cordially invited to attend a special meeting of stockholders of NaPro BioTherapeutics, Inc. (the "Company") on , , 2003, at .m. local time. The special meeting will be held at [location to be determined]. At this special meeting, you will be asked to consider and vote on a resolution to approve the proposed sale of our paclitaxel business to Faulding Pharmaceutical Co. for a purchase price of $71.7 million in cash minus an inventory adjustment to reflect our actual inventory as of the closing. Faulding will assume certain liabilities associated with the paclitaxel business. Approximately $21.7 million of the proceeds of the purchase price will be paid to Abbott Laboratories or its designee at closing to retire all outstanding debt and payables we owe to Abbott.
The proposed sale includes the transfer to Faulding of the assets and liabilities of our worldwide polyethoxylated castor oil formulated injectable paclitaxel business. Under the terms of the agreement, Faulding will acquire all of our paclitaxel manufacturing assets, yew plantations, domestic and international issued and pending paclitaxel patents, a worldwide registration dossier, worldwide development and supply agreements and inventories. We will retain all our intellectual property not directly used in connection with the business being sold.
The proposed sale may constitute the sale of substantially all of our assets under Delaware law and thus may require stockholder approval. The proposed sale would be consummated pursuant to the terms of an asset purchase agreement entered into between the Company and Faulding as of August 25, 2003, as described in more detail in the attached Proxy Statement (Proposal 1). Your board of directors has unanimously approved the proposed sale.
Your Board has determined that the proposed sale as contemplated by the asset purchase agreement is fair to, and in the best interests of, the Company and its stockholders. Accordingly, it recommends that you vote "FOR" the resolution approving the proposed sale and "FOR" the proposal to adjourn the special meeting in certain circumstances.
We cannot complete the proposed sale unless the holders of [15,377,693] of the outstanding shares of the Company's common stock vote to approve it. The obligations of the Company and Faulding under the sale agreement are also subject to the satisfaction or waiver of other conditions, which we expect will be satisfied. Whether or not you plan to attend the special meeting, please fill out, sign, date and return the enclosed proxy promptly in the envelope provided. Your shares will then be represented at the special meeting. If you attend the special meeting, at your discretion, you may withdraw the proxy and vote in person. If you hold your shares in "street name," you should instruct your broker how to vote in accordance with your voting instruction form.
Your vote is very important. If you fail to return the proxy card or fail to vote in person at the special meeting, it will have the same effect as a vote against the proposed sale.
The accompanying Notice of Special Meeting and Proxy Statement explain the proposed sale and provide specific information concerning the special meeting. Please read these materials carefully. We look forward to seeing you at the special meeting.
| Sincerely, | ||
Leonard P. Shaykin Chairman and Chief Executive Officer |
NAPRO BIOTHERAPEUTICS, INC.
4840 Pearl East Circle, Suite 300W
Boulder, Colorado 80301
Notice of Special Meeting Of Stockholders
To be held on , 2003
To our stockholders:
You are cordially invited to attend a special meeting of stockholders of NaPro BioTherapeutics, Inc., a Delaware corporation (the "Company") which will be held on , 2003, at .m. local time at [location to be determined] for the following purposes:
- 1.
- To adopt a resolution approving the Company's sale of its paclitaxel business to Faulding Pharmaceutical Co. under the terms set forth in the asset purchase agreement between Faulding and the Company dated as of August 25, 2003. The asset purchase agreement is attached as Annex A to the accompanying Proxy Statement;
- 2.
- To approve a proposal to adjourn the special meeting to a later date or dates, if necessary, to permit further solicitation of proxies in the event there are not sufficient votes to approve the asset sale at the time the special meeting is convened or to allow additional time for the satisfaction of conditions to the sale; and
- 3.
- To transact such other business as may properly come before the special meeting or any adjournment(s) or postponement(s) of the special meeting.
The board of directors has fixed the close of business on , 2003 as the record date for the determination of stockholders entitled to notice of and to vote at the special meeting and at any adjournment(s) or postponement(s) of the special meeting. Only holders of record of our common stock,$0.0075 par value per share, at the close of business on the record date are entitled to notice of and to vote at the special meeting. The Company's stock transfer books will not be closed following the record date. A complete list of stockholders entitled to vote at the special meeting will be available for inspection at the Company's offices at 4840 Pearl East Circle, Suite 300W, Boulder, Colorado 80301during normal business hours by any stockholder of the Company for the ten days prior to the special meeting. The list will also be available at the special meeting and may be inspected by any stockholder who is present.
Holders of our common stock will not have appraisal or dissenters' rights in connection with the proposed sale if it is approved.
This proxy solicitation is made on behalf of the board of directors of the Company. Whether or not you plan to attend the special meeting, please promptly complete, date, sign, and return the proxy card enclosed with the Proxy Statement. A postage prepaid envelope is enclosed for that purpose. Executed proxies with no instructions indicated thereon will be voted "FOR" the resolution approving the asset sale and "FOR" proposal number 2 to adjourn the special meeting in certain circumstances. Any stockholder attending the special meeting may vote in person even if that stockholder has returned a proxy. Please note, however, that if your shares are held of record by a broker, bank or other nominee and you wish to vote at the special meeting, you must obtain from the record holder a proxy issued in your name. If you fail to return your proxy or fail to vote in person at the special meeting, your shares will not be counted for purposes of determining whether a quorum is present at the special meeting, and will effectively be counted as a vote against the asset sale.
By Order of the Board of Directors
Patricia A. Pilia, Ph.D.
Secretary
Boulder, Colorado
September , 2003
i
ii
QUESTIONS AND ANSWERS ABOUT THE SPECIAL MEETING
- Q:
- Where and when is the special meeting?
- A:
- The special meeting will take place at [location to be determined] on , 2003, at .m., local time. In this Proxy Statement we refer to NaPro BioTherapeutics as "the Company" or "we."
- Q:
- What proposals will be voted on at the special meeting?
- A:
- The following two proposals will be voted on at the special meeting:
- •
- The first proposal (Proposal 1) to be voted on is whether to adopt a resolution approving the Company's sale of its worldwide polyethoxylated castor oil formulated injectable paclitaxel business (referred to in this Proxy Statement as the "paclitaxel business") to Faulding Pharmaceutical Co. ("Faulding") under the terms set forth in the asset purchase agreement between Faulding and the Company dated August 25, 2003. Pursuant to the asset purchase agreement, we will sell these assets to Faulding for a purchase price of $71.7 million in cash minus an inventory adjustment to reflect our actual inventory as of the closing, and Faulding will assume certain liabilities associated with the paclitaxel business. Approximately $21.7 million of the proceeds of the purchase price will be paid to one of our development and marketing partners, Abbott Laboratories, or to Abbott's designee, at closing to retire all outstanding debt and payables we owe to Abbott.
The assets Faulding would acquire include our paclitaxel manufacturing assets, yew plantations, domestic and international issued and pending paclitaxel patents, a worldwide registration dossier, worldwide development and supply agreements and inventories. In connection with the asset sale to Faulding, we have agreed that, for five years after the closing, we will not manufacture, use, or sell bulk active pharmaceutical ingredient paclitaxel for use in formulation of paclitaxel formulated in polyethoxylated castor oil, including, without limitation, paclitaxel formulated in polyethoxylated castor oil and ethanol. For three years after the closing, we have agreed not to manufacture or supply paclitaxel using certain proprietary processes for commercial purposes. We will retain all our intellectual property not used in connection with the business being sold. In this Proxy Statement, we refer to the sale of our paclitaxel business to Faulding pursuant to the asset purchase agreement as the "asset sale" in this Proxy Statement. See the section of this Proxy Statement entitled "Proposal 1—Sale of Our Paclitaxel Business to Faulding" for a more detailed description of the transaction with Faulding.
- •
- The second proposal (Proposal 2) to be voted on is whether to adjourn the special meeting to a later date or dates, if necessary, to permit further solicitation of proxies in the event there are not sufficient votes to approve the asset sale at the time the special meeting is convened or to allow additional time for the satisfaction of conditions to the asset sale. See the section of this Proxy Statement entitled "Proposal 2—Adjournment of the Special Meeting" for a more detailed description of Proposal 2.
- Q:
- What are the reasons for the asset sale?
- A:
- Over the past several months, our board of directors has reviewed our existing operations and its future growth opportunities. As a result, the board of directors believes that the Company should alter its existing generic pharmaceutical manufacturing strategy and focus on the development and commercialization of novel proprietary therapies that address specific clinical needs. These therapies include our oncology and gene editing platforms. The board of directors considered a number of strategic benefits that may result from the sale of its paclitaxel business to Faulding as well as a number of potentially negative factors. The board of directors does not believe that the negative factors were sufficient, individually or in the aggregate, to outweigh the potential strategic benefits of the asset sale. The potential strategic benefits and negative factors which the board examined are described in detail in the section of this Proxy Statement entitled "Proposal 1—Sale
of Our Paclitaxel Business to Faulding—Consideration of the Asset Sale by the Company's Board of Directors—The Company's Reasons for the Asset Sale."
- Q:
- What vote of our stockholders is required to approve the asset sale?
- A:
- For us to complete the asset sale, stockholders holding at least a majority of the shares of our common stock outstanding at the close of business on the record date must vote "FOR" the resolution approving the asset sale.
- Q:
- What will happen if the asset sale is approved by our stockholders? (see pages 52-56)
- A:
- If the asset sale is approved by our stockholders and the other conditions to closing of the asset sale are satisfied or waived, we will sell our paclitaxel business to Faulding. Upon consummation of this asset sale, we will exit the generic paclitaxel business, our development agreements with Abbott and Faulding will terminate, and we will transfer our other generic paclitaxel marketing agreements to Faulding. We intend to use the proceeds from the asset sale to fund the development and commercialization of products based on our proprietary gene editing and oncology platforms, to retire the debt and related payables to Abbott and for general corporate purposes. We do not intend to distribute the proceeds of the sale to stockholders. TL Ventures Funds, one of our principal institutional investors, has advised us that it believes that the completion of the asset sale will entitle it to have its $8 million of Company subordinated debentures redeemed. On September 8, 2003, TL Ventures Funds informed us that it intends to pursue legal remedies if we cannot resolve the issue promptly. We dispute TL Ventures Funds' position and will vigorously defend its legal position. See "Proposal 1—Sale of Our Paclitaxel Business to Faulding—Background of the Proposed Asset Sale."
Following the completion of the asset sale, we intend to focus our future efforts primarily on two distinct classes of therapeutics currently under development. First, we are developing an oncology program, consisting of both targeted as well as non-targeted chemical entities for the treatment of cancer. Our second class of therapeutics is based upon our "gene-editing" platform which provides a unique platform for the development of therapeutics which address hereditary diseases. Currently, we are engaged in developing therapeutic programs to treat Sickle Cell disease and Huntington disease.
The proceeds from the asset sale would provide resources to fund the introduction of what we determine to be the most promising of these therapies into the clinic. However, we cannot assure you that the proceeds from the asset sale, when realized, will alone be sufficient to fund our strategic plans to develop and commercialize any one of these therapies. If the proceeds from the asset sale are not sufficient, we may be required to seek additional financing that could be dilutive to our existing stockholders. In addition, upon completion of the asset sale, we will have no recurring sources of cash flow from revenue from operations. Our ability to generate revenues in the future will depend on developing and commercializing gene editing, oncology and other new products and technologies. All of these new products and technologies are in the early developmental stages and we cannot assure you that these efforts will be successful. See "Proposal 1—Sale of Our Paclitaxel Business to Faulding—Use of Proceeds; Operations After Asset Sale; Risks" for a more detailed description of our plans after the asset sale.
- Q:
- What will happen if the asset sale is not approved or completed?
- A:
- If the asset sale is not approved by our stockholders or is not completed for any other reason, we would likely continue to operate our paclitaxel business for the foreseeable future. We also might explore other strategic alternatives, including a sale of our paclitaxel business or other assets to, or a business combination with, another party, or we may pursue other business opportunities and investments unrelated to our current paclitaxel business. We cannot assure you that any potential transaction will provide consideration equal to or greater than the price proposed to be paid by
2
Faulding in the asset sale, or that we will be able to complete any alternative transaction. At this time, our board of directors has not made any decision to pursue any one of these options.
In addition, if the asset sale is not completed, we may be required to pay Faulding liquidated damages of $2 million in certain circumstances (not including failure to receive the requisite stockholder approval). For a more complete discussion of the requirements relating to liquidated damages upon termination, see the section of this Proxy Statement entitled "Proposal 1—Sale of Our Paclitaxel Business to Faulding—The Asset Purchase Agreement—Payment of Liquidated Damages." The failure to sell the paclitaxel business may also prevent us from retiring the approximately $21.7 million of debt and payables we owe to Abbott, the majority of which is due in May 2004. Until we retire our debt to Abbott, the majority of our paclitaxel inventories and specific assets that relate to the manufacture of paclitaxel will remain pledged to Abbott as collateral for this debt.
- Q:
- How does our board of directors recommend that I vote?
- A:
- Our board of directors unanimously recommends that our stockholders vote "FOR" the resolution approving the asset sale. You should read "Proposal 1—Sale of Our Paclitaxel Business to Faulding—Board of Directors Recommendations to Stockholders and Reasons for the Asset Sale" for a discussion of the factors that our board of directors considered in deciding to recommend approval of the asset sale. Our board of directors also unanimously recommends that our stockholders vote "FOR" Proposal 2 to adjourn the special meeting in certain circumstances.
- Q:
- What do I need to do now? (see pages 13-15)
- A:
- After carefully reading and considering the information contained in this Proxy Statement, you should complete and sign your proxy and return it in the enclosed return, pre-paid envelope as soon as possible so that your shares may be represented at the special meeting. A majority of shares entitled to vote must be represented at the special meeting to enable us to conduct business at the special meeting. See the section of this Proxy Statement entitled "Information Concerning Voting and Solicitation of Proxies."
- Q:
- Can I change my vote after I have mailed my signed proxy? (see page 13)
- A:
- Yes. You can change your vote at any time before proxies are voted at the special meeting in one of three ways. First, you can send a written notice via mail to our secretary at our executive offices prior to the special meeting date, stating that you would like to revoke your proxy. Second, you can complete and submit a new proxy bearing a later date prior to the special meeting. Third, you can attend the special meeting and elect to vote in person. See the section of this Proxy Statement entitled "Information Concerning Voting and Solicitation of Proxies—Voting; Revocability of Proxy."
- Q:
- If my shares are held in "street name" by my broker, will the broker vote the shares on my behalf? (see pages 13-14)
- A:
- Shares held in "street name" by brokers or nominees who indicate on their proxies that they do not have discretionary authority to vote such shares as to a particular matter, referred to as "broker non-votes," will not be voted in favor of such matter. Proposal 1 requires the affirmative vote of a majority of our outstanding shares to be approved by our stockholders. Accordingly, broker non-votes will have the effect of a vote against Proposal 1. Broker non-votes will have no effect on Proposal 2. We anticipate that a broker will vote your shares only if you provide the broker with instructions on how to vote. We encourage all stockholders whose shares are held in street name to provide their brokers with instructions on how to vote.
3
- Q:
- What happens if I don't indicate how to vote my proxy? (see page 14)
- A:
- If you sign and send in your proxy, but do not include instructions on how to vote your properly signed proxy card, your shares will be voted "FOR" Proposal 1 to approve the asset sale and "FOR" Proposal 2 to adjourn the special meeting in certain circumstances.
- Q:
- What should I do if I receive more than one set of voting materials?
- A:
- You may receive more than one set of voting materials, including multiple copies of this Proxy Statement and multiple proxy cards or voting instruction cards. Please complete, sign, date and return each proxy card and voting instruction card that you receive.
- Q:
- When is the asset sale expected to be completed?
- A:
- We expect to complete the asset sale as soon as practicable after all of the conditions to completion of the asset sale contained in the asset purchase agreement have been satisfied or waived. The Company and Faulding currently plan to complete the asset sale in the fourth quarter of 2003 shortly following the special meeting of our stockholders, assuming our stockholders approve the asset sale and the other conditions to the asset purchase agreement are satisfied or waived. However, because the asset sale is subject to some conditions which are beyond our control, we cannot predict the exact timing of the completion of the asset sale. For a complete description of the conditions to completion of the asset sale, see the section of this Proxy Statement entitled "Proposal 1—Sale of Our Paclitaxel Business to Faulding—The Asset Purchase Agreement—Conditions to Completion of the Asset Sale."
- Q:
- What will I receive as a result of the asset sale?
- A:
- You will continue to own your shares of common stock. You will not receive any cash, stock or other property in connection with, or as a result of, the asset sale.
- Q:
- Am I entitled to appraisal or dissenters' rights in connection with the asset sale?
- A:
- No. Holders of our common stock will not have appraisal or dissenters' rights in connection with the sale of our paclitaxel business.
- Q:
- What are the tax consequences of the asset sale? (see page 47)
- A:
- The asset sale to Faulding is a taxable event to the Company. The Company will recognize gain or loss in an amount equal to the cash received, plus liabilities assumed in exchange for the assets and the amount paid to Abbott at closing to retire all outstanding debt and payables we owe to Abbott, less our adjusted tax basis in the purchased assets. Our gain will be offset to the extent of current year losses from operations plus available net operating loss carryforwards and general business carryforwards, subject to applicable limitations under the ownership changes rules under Internal Revenue Code section 382 and the alternative minimum tax rules. Any tax liabilities to us generated as a result of the asset sale are not expected to exceed $1million. We do not anticipate any direct tax consequence to you as a result of the asset sale. However, any tax consequence of the asset sale may vary depending on the particular circumstances of the stockholder. Accordingly, we recommend that each stockholder consult its own tax advisor regarding the federal income tax consequences of the asset sale, as well as the state, local and foreign consequences.
- Q:
- Who can help answer my questions about the proposals?
- A:
- If you have any questions about the proposals presented in this Proxy Statement, you should contact:
NaPro BioTherapeutics, Inc.
4840 Pearl East Circle, Suite 300W
Boulder, Colorado 80301
Attn: Kai Larson
Title: Vice President and General Counsel
Telephone: (303) 516-8500
In addition, our public filings can be accessed at the Securities and Exchange Commission's web site at www.sec.gov.
4
NAPRO BIOTHERAPEUTICS, INC.
4840 Pearl East Circle, Suite 300W
Boulder, Colorado 80301
PROXY STATEMENT
The following is a summary of the information contained in this Proxy Statement. This summary may not contain all of the information that is important to you. You should carefully read this entire Proxy Statement and the other documents referred to herein for a more complete understanding of the proposed asset sale to Faulding. In particular, you should read the annexes attached to this Proxy Statement, including the asset purchase agreement entered into between the Company and Faulding as of August 25, 2003, which is attached as Annex A. We have included page references in parentheses to direct you to a more complete description of the topics presented in this summary. This Proxy Statement and accompanying proxy were first mailed on or about , 2003 to all stockholders entitled to vote at the special meeting.
NaPro BioTherapeutics, Inc.
Today, NaPro BioTherapeutics, Inc. is a pharmaceutical company focused in three distinct areas:
- •
- the production and sale of paclitaxel, an approved cancer drug;
- •
- the development of novel, anti-cancer agents; and
- •
- the development of novel genomic technologies, primarily in the area of gene editing for applications in human therapeutics, diagnostics, agribiotechnology and pharmacogenomics.
Historically, the primary focus of our business has been the production and sale of paclitaxel, a naturally occurring chemotherapeutic anti-cancer agent found in certain species of yew, orTaxus, trees, and a majority of our resources has been devoted to this endeavor. In the field of oncology, we are developing several compounds that we believe have potential as anti-cancer agents to treat a variety of cancers including small cell lung, prostate, pancreatic and colorectal cancers and squamous cell head and neck carcinomas. In the field of genomics, we are engaged in the discovery, patenting, development and commercialization of a range of products using our proprietary gene editing technology. In the area of therapeutics, our efforts to date have been directed primarily to treatments for Sickle Cell disease and Huntington's disease. In addition, we are developing gene editing strategies for animals, plants, viruses and other microbes. In addition to gene editing and oncology research and development activities, we are also evaluating the in-licensing or purchase of potential new products and/or technologies.
We are a Delaware corporation, incorporated in 1991. Our principal executive offices are located at 4840 Pearl East Circle, Suite 300W, Boulder, Colorado 80301. Our telephone number is (303) 516-8500. Our web site is located at www.naprobio.com. We have not incorporated by reference into this prospectus the information on our website, and you should not consider it to be a part of this document. Our web site address is included in this document as an inactive textual reference only.
Faulding Pharmaceutical Co.
Faulding Pharmaceutical Co., a subsidiary of Mayne Group Limited, is a supplier of multi-source injectable pharmaceuticals focused on the hospital market in the United States. Faulding's products cover a number of therapeutic categories including oncology, anesthesia/pain and cardiac care.
5
Mayne Group Limited is Australia's leading provider of private health care services, employing approximately 29,000 people in Australia. Mayne's business includes pharmaceuticals, consumer products, hospitals and health services. Mayne Pharma is a generic pharmaceutical company which develops and manufactures generic injectable pharmaceuticals for hospitals, and specializes in generic oncology products. Mayne Pharma's products cover therapeutic areas including oncology, anti-infectives, anaesthetics, and treatments relating to the cardiovascular and central nervous systems. Mayne Consumer Products manufactures and markets a wide range of health care-related consumer products, including vitamin and health supplements. Mayne's Health Services division encompasses pathology, diagnostic imaging, pharmacy services and medical centers.
We are party to an Amended and Restated Master Agreement, dated as of June 5, 2000, with F.H. Faulding & Co., Limited, an affiliate of Faulding Pharmaceutical Co., which is the entity that will purchase our paclitaxel business, and to an Agreement, dated as of March 2, 2001, with Central Laboratories, Limited, another affiliate of Faulding. Both of these agreements generally relate to development and supply relationships between us and the Faulding affiliates party to such agreements and are part of our paclitaxel business. These agreements will be terminated as part of the asset sale.
Faulding is a Delaware corporation which was incorporated in 1995. Faulding's principal executive offices are located at 650 From Road, Mack-Cali Centre II, Second Floor, Paramus, NJ 07652. Faulding's telephone number is (201) 225-5500 and its web site is located atwww.maynepharma.com/us. We have not incorporated by reference into this prospectus the information on Mayne's website, and you should not consider it to be a part of this document. Mayne's web site address is included in this document as an inactive textual reference only.
Voting Requirements for the Asset Sale (see page 14)
In order to complete the asset sale the holders of a majority of the outstanding shares of our common stock as of the record date must approve the proposed asset sale. The holders of our common stock will be entitled to cast one vote per share owned as of , 2003, the record date for the special meeting.
Share Ownership of Our Directors and Executive Officers (see pages 57-59)
As of the close of business on the record date for the special meeting of stockholders at which the asset sale will be presented and voted upon, our directors and executive officers collectively owned approximately 6.7% of the outstanding shares of our common stock entitled to vote at the special meeting. This does not include 2,471,958 shares of our common stock issuable upon the exercise of presently exercisable options which these directors and executive officers beneficially own. If all of these stock options had been exercised prior to the record date for the special meeting, the directors and executive officers of the Company would collectively beneficially own approximately 13.5% of the outstanding shares of our common stock entitled to vote at the special meeting. Two members of our board of directors, Leonard P. Shaykin, also our Chairman and Chief Executive Officer, and Sterling K. Ainsworth, also our Vice Chairman, have entered into agreements with Faulding to vote 622,554 shares and 913,273 shares of common stock, respectively, which together represent about 5.1% of the outstanding shares, in favor of the asset sale.
Board of Directors Recommendations to Stockholders and Reasons for the Asset Sale (see page 26)
Recommendation of the Company's Board of Directors (see page 26)
After careful consideration, the Company's board of directors unanimously determined that it is advisable and in the best interests of the Company and its stockholders to adopt a resolution to approve the proposed asset sale. Accordingly, the Company's board of directors unanimously approved the asset sale and recommends that the stockholders vote "FOR" Proposal 1.
6
The Company's Reasons for the Asset Sale (see pages 24-26)
In reaching its decision to approve the asset sale:
- •
- our board of directors considered several potential strategic benefits and material factors pertaining to the asset sale;
- •
- these included the board of directors' belief that the maximum long-term value of the Company is more likely to be realized in opportunities that are focused on the development and commercialization of novel proprietary therapies that address specific clinical needs;
- •
- our board of directors also considered potentially negative factors, including the risk that the potential strategic benefits of the asset sale may not be realized, in part or at all; and
- •
- our board of directors did not believe that the negative factors were sufficient, individually or in the aggregate, to outweigh the potential strategic benefits of the asset sale.
The board of directors examined a number of other potential strategic benefits and negative factors which are described in detail in the section of this Proxy Statement entitled "Proposal 1—Sale of Our Paclitaxel Business to Faulding—Consideration of the Asset Sale by the Company's Board of Directors—The Company's Reasons for the Asset Sale."
Opinion of the Company's Financial Advisor (see pages 26-32)
In connection with the asset sale, the Company's financial advisor, Wells Fargo Securities, LLC, delivered a written opinion dated as of August 25, 2003 to the Company's board of directors that, as of that date and based on the assumptions made, matters considered and the limitations on the review undertaken described in the written opinion, the consideration to be received by the Company in the transaction was fair, from a financial point of view, to the Company. The full text of the Wells Fargo Securities' written opinion, dated August 25, 2003, is attached to this Proxy Statement as Annex B. We encourage you to read this opinion and the section of this Proxy Statement entitled "Proposal 1—Sale of Our Paclitaxel Business to Faulding—Opinion of the Company's Financial Advisor" carefully in their entirety for a description of the procedures followed, assumptions made, matters considered and limitations on the review undertaken. Wells Fargo Securities' opinion was directed to the Company's board of directors and does not constitute a recommendation to any stockholder as to any matter relating to the asset sale.
Interests of Our Directors and Executive Officers in the Asset Sale (see pages 44-46)
In considering the recommendation of our board of directors to vote for the proposal to adopt a resolution approving the asset sale, you should be aware that certain of our directors and executive officers have personal interests in the asset sale that are, or may be, different from, or in addition to, those of our stockholders generally:
- •
- pursuant to the terms of employment agreements with certain executive officers, the Company would owe these executives a severance payment and benefits upon a termination of their employment under certain circumstances;
- •
- the compensation committee of the board of directors recently approved a grant of stock options to purchase, in the aggregate, 1.25 million shares of our common stock and such grants were made in light of the facts that no options had been granted to senior management for over two years with limited exceptions and that the asset sale warranted special recognition; and
- •
- TL Ventures Funds, one of our principal institutional investors, has advised us that it believes that the completion of the asset sale will entitle it to have its $8 million of Company subordinated debentures redeemed. On September 8, 2003, TL Ventures Funds informed us that
7
it intends to pursue legal remedies if we cannot resolve the issue promptly. The Company disputes TL Ventures Funds' position and will vigorously defend its legal position. One of our directors, Marc Ostro, is also a partner in TL Ventures Funds.
Our board of directors was aware of these interests and approved the asset purchase agreement. The Company does not intend to take any action that would result in any payments being made by the Company under the employment agreements with certain executive officers discussed above,. For a detailed description of the interests of our directors and executive officers in the asset sale, see the section of this Proxy Statement entitled "Proposal 1—Sale of Our Paclitaxel Business to Faulding—Interests of Our Directors and Executive Officers in the Asset Sale."
Assets Being Sold (see page 33)
Our paclitaxel business may constitute substantially all of our assets under Delaware law and therefore the sale of that business may require approval of our stockholders. Under the terms of the asset purchase agreement, Faulding would acquire:
- •
- our paclitaxel manufacturing assets;
- •
- yew plantations;
- •
- domestic and international issued and pending paclitaxel patents;
- •
- a worldwide registration dossier;
- •
- worldwide development and supply agreements; and
- •
- inventories.
In connection with the asset sale to Faulding, we have agreed that, for five years after the closing, we will not manufacture, use, or sell bulk active pharmaceutical ingredient paclitaxel for use in formulation of paclitaxel formulated in polyethoxylated castor oil, including, without limitation, paclitaxel formulated in polyethoxylated castor oil and ethanol. For three years after the closing, we have agreed not to manufacture or supply paclitaxel using certain proprietary processes for commercial purposes. We will retain all our intellectual property not used in connection with the business being sold. We will also retain all other assets of our business including:
- •
- documentation related to our corporate existence and qualifications to do business;
- •
- cash on hand as of the closing;
- •
- accounts receivable that accrue in accordance with generally accepted accounting principles prior to the closing;
- •
- all assets, including patents and other intellectual property, not related to or used in the operation of our paclitaxel business;
- •
- our rights under the asset purchase agreement, including the consideration to be paid to us pursuant to the asset purchase agreement, and any other agreements between us and Faulding on or after the date of the asset purchase agreement;
- •
- our insurance policies and insurance contracts;
- •
- employee benefit plans, policies, and contracts; and
- •
- certain retained inventory.
8
Faulding has agreed to pay us $71.7 million in cash minus an inventory adjustment to reflect our actual inventory as of the closing. Faulding will assume certain liabilities associated with the paclitaxel business. Approximately $21.7 million of the proceeds of the purchase price will be paid to Abbott or its designee at closing to retire all outstanding debt and payables we owe to Abbott.
In connection with the asset sale, Faulding will assume certain of liabilities relating to obligations under the agreements related to our paclitaxel business we are selling, including transferred strategic alliances, partner agreements, cultivation agreements, certain consulting agreements, purchase orders and liabilities associated with certain regulatory proceedings and litigation. We will retain other liabilities relating to our business, including those relating to our employees and stockholders, our retained contracts, license agreements unrelated to the paclitaxel business, and certain leases and purchase orders, as well as certain other liabilities related to our manufacture and sale of paclitaxel which arose prior to the closing of the asset sale.
Indemnification Obligations (see page 40)
Under the terms of the asset purchase agreement, we have agreed to indemnify Faulding for the adverse consequences Faulding suffers as a result of:
- •
- breaches of our representations, warranties and covenants under the asset purchase agreement; and
- •
- liabilities not specifically assumed by Faulding.
Our liability for claims relating to breaches of certain of our representations and warranties is subject to certain minimum and maximum limitations that we negotiated with Faulding. However, none of these liability limitations apply to:
- •
- breaches of particular representations or warranties under the asset purchase agreement; or
- •
- liabilities not assumed by Faulding.
Completion of the Asset Sale (see page 35)
The Company and Faulding expect to complete the asset sale as soon as practicable. You should be aware that:
- •
- the asset sale cannot be completed until after all of the conditions to closing in the asset purchase agreement have been satisfied or waived;
- •
- the Company and Faulding are working toward satisfying these conditions to closing;
- •
- the Company anticipates that the sale will close in the fourth quarter of 2003, shortly following the special meeting of our stockholders, assuming that our stockholders approve the asset sale; and
- •
- some of the conditions to closing the asset sale are beyond our control and therefore the exact timing of the completion of the asset sale cannot be predicted.
The Company is Prohibited from Soliciting Other Offers (see page 37)
We have agreed that neither the Company nor any of our affiliates will directly or indirectly initiate or solicit any inquiries with respect to; have any discussion with or provide any confidential information to any person relating to; knowingly facilitate any effort to make; approve, recommend,
9
propose publicly to approve or recommend; or enter into any agreement to do any of the foregoing related to any merger, reorganization, share exchange, consolidation, business combination, recapitalization, liquidation, dissolution or similar transaction involving the Company or its affiliates; any purchase or sale of 33% or more of the assets of the Company and its affiliates; or any purchase, sale, tender offer or exchange offer for our equity securities representing more than 33% of our outstanding voting power for the election of a majority of directors. We may, however, engage in discussions or negotiations with a person who seeks to initiate discussions or negotiations and may provide information to such person if the board of directors concludes that such action is in the best interests of our stockholders. In addition, our board of directors may withdraw, modify or make no recommendation in favor of the asset sale if it concludes in good faith that such action is necessary in order to act in a manner consistent with its fiduciary obligations. See also "—Termination of the Asset Purchase Agreement and Payment of Liquidated Damages."
Conditions to Completion of the Asset Sale (see pages 38-40)
The Company's and Faulding's obligations to complete the asset sale are subject to specified conditions, including, among others:
- •
- the adoption of the asset sale by the affirmative vote of the holders of a majority of all outstanding shares of common stock;
- •
- the continued accuracy of each party's representations and warranties;
- •
- there being no order or decree preventing consummation of the asset sale;
- •
- the performance by each party of its obligations under the asset purchase agreement;
- •
- the fairness opinion of Wells Fargo Securities shall not have been withdrawn or modified in any material respect not acceptable to our board of directors; and
- •
- the expiration or termination of any applicable waiting period under the Hart-Scott-Rodino Antitrust Improvements Act of 1976 (the "HSR Act") and the receipt of all material consents from governmental authorities.
The conditions to completion of the asset sale are described in more detail in the section of this Proxy Statement entitled "Proposal 1—Sale of Our Paclitaxel Business to Faulding—The Asset Purchase Agreement—Conditions to Completion of the Asset Sale."
Termination of the Asset Purchase Agreement and Payment of Liquidated Damages (see pages 40-41)
The Company and Faulding may terminate the asset purchase agreement by mutual agreement and under other circumstances specified in the asset purchase agreement. If the asset purchase agreement is terminated under certain circumstances described under the section of this Proxy Statement entitled "Proposal 1—Sale of Our Paclitaxel Business to Faulding—The Asset Purchase Agreement—Payment of Liquidated Damages," the asset purchase agreement requires us to pay Faulding liquidated damages equal to $2 million.
Material U.S. Federal Income Tax Consequences to the Company (see page 47)
The asset sale to Faulding is a taxable event to the Company. As a result:
- •
- we will recognize gain or loss in an amount equal to the cash received, plus liabilities assumed in exchange for the assets and the amount paid to Abbott at closing to retire all outstanding debt and payables we owe to Abbott, less our adjusted tax basis in the assets;
- •
- our gain will be offset to the extent of current year losses from operations plus available net operating loss carryforwards and general business carryforwards;
10
- •
- the use of our loss carryforwards to offset our gain is subject to applicable limitations under the ownership changes rules under Internal Revenue Code section 382 and the Alternative Minimum Tax rules;
- •
- any tax liabilities to us as a result of the asset sale are not expected to exceed $1 million; and
- •
- we do not anticipate any direct tax consequence to our stockholders as a result of the asset sale.
However, any tax consequence of the asset sale may vary depending on the particular circumstances of the stockholder. ACCORDINGLY, WE RECOMMEND THAT EACH STOCKHOLDER CONSULT ITS OWN TAX ADVISOR REGARDING THE FEDERAL INCOME TAX CONSEQUENCES OF THE ASSET SALE, AS WELL AS THE STATE, LOCAL AND FOREIGN CONSEQUENCES.
Accounting Treatment of the Asset Sale (see page 47)
If the asset sale is approved by the Company's stockholders as described in this Proxy Statement:
- •
- the Company will record the asset sale in accordance with accounting principles generally accepted in the United States;
- •
- upon the completion of the asset sale, the Company will recognize a financial reporting gain, if any, equal to:
- •
- the net proceeds (the sum of the purchase price received less the estimated $1,258,000 in expenses relating to the asset sale) less
- •
- the net book value of the assets purchased and the fair value of the indemnification liability retained.
Regulatory Approvals Required to Complete the Asset Sale (see pages 51-52)
The asset sale is subject to review by the Antitrust Division of the U.S. Department of Justice and the U.S. Federal Trade Commission under the HSR Act. You should be aware that:
- •
- under the HSR Act, the Company and Faulding are required to make pre-acquisition notification filings with the United States Federal Trade Commission and the Antitrust Division of the Department of Justice and to await the expiration or early termination of the statutory waiting period of 30 days prior to completing the acquisition;
- •
- either the United States Federal Trade Commission or the Antitrust Division of the United States Department of Justice could challenge or block the acquisition under the antitrust laws or request additional information; and
- •
- in some jurisdictions, a competitor, customer or other third party could initiate a private action under the antitrust laws challenging or seeking to enjoin the acquisition.
Other than applicable antitrust laws:
- •
- we are not aware of any other regulatory requirements or governmental approvals or actions that may be required to consummate the acquisition, except for compliance with the applicable regulations of the Securities and Exchange Commission in connection with this Proxy Statement and the Delaware General Corporation Law in connection with the asset sale;
- •
- should any such approval or action be required, it is contemplated that such approval or action would be sought; and
11
- •
- there can be no assurance, however, that any such approval or action, if needed, could be obtained, or if obtained, would not be conditioned in a manner that would cause the parties to abandon the acquisition.
Use of Proceeds; Operations After Asset Sale; Risks (see pages 52-56)
Proceeds from the asset sale are expected to be used to:
- •
- fund development and commercialization of products based on our proprietary gene editing and oncology platforms;
- •
- for general corporate purposes; and
- •
- at closing, approximately $21.7 million of the proceeds of the purchase price will be paid to Abbott or its designee to retire all outstanding debt and payables we owe to Abbott.
No final decision has been made as to which other purposes the proceeds will be applied.
After the asset sale, we intend to focus our future efforts on the development and commercialization of therapeutic candidates, primarily in two distinct classes of therapeutics:
- •
- Oncology Platform. In the field of oncology, we are developing several compounds that we believe have potential as anti-cancer agents to treat a variety of cancers including small cell lung, prostate, pancreatic and colorectal cancers and squamous cell head and neck carcinomas; and
- •
- Gene Editing. In the field of genomics and hereditary diseases, we are engaged in the discovery, patenting, development and commercialization of a range of products using our proprietary gene editing technology. Currently, we are engaged in developing therapeutic programs to treat Sickle Cell disease and Huntington's disease.
If we complete the asset sale, we will be exposed to risks related to the operation of this business, including the risk that the asset sale could adversely affect our ability to achieve profitability because the asset sale would transfer a significant portion of our total assets and current operations. If we are unable to complete the asset sale, we will face other risks including the risk that our development of other products or product candidates, ability to advance other businesses or ability to retire our debt may be delayed or impaired.
A COPY OF THE ASSET PURCHASE AGREEMENT IS INCLUDED IN THIS PROXY STATEMENT AS ANNEX A. YOU ARE STRONGLY ENCOURAGED TO READ IT CAREFULLY AND IN ITS ENTIRETY.
Other Agreements Related to the Asset Sale (see pages 42-44)
In connection with the asset sale, we have entered or will enter into certain other agreements with Faulding or Abbott. In addition, Faulding and Abbott have entered into an asset purchase agreement pursuant to which Abbott has agreed to sell to Faulding, and Faulding has agreed to purchase from Abbott, among other things, certain assets and rights, and Faulding will assume from Abbott certain liabilities, associated with the development agreement between the Company and Abbott (the "Abbott-Faulding Transaction"). These agreements between the Company and Faulding or Abbott include:
- •
- Intellectual Property Agreement (see page 42). To be entered into at the closing, this agreement will govern the parties' rights to use certain intellectual property rights relating to the research, development, manufacture, use and sale of paclitaxel products.
- •
- Transition Services Agreement (see page 42). The transition services agreement to be entered into by the Company and Faulding at the closing will set forth the Company's and Faulding's obligations to provide certain services to the other to assist in the transition of the Company's
12
- •
- Faulding Termination Agreement (see pages 42-43). We agreed to terminate certain of our existing agreements with Faulding and certain of our existing agreements with Abbott. In addition, we agreed to settle certain remaining delivery and payment issues under our agreements with Faulding, including, at our option, delivering to Faulding bulk paclitaxel or cash. In addition to the foregoing, Faulding will pay us for certain bulk paclitaxel. The parties also agreed to release each other from claims relating to the agreements terminated. The Faulding Termination Agreement is effective upon the later to occur of the closing of the Abbott-Faulding Transaction and the asset sale by us to Faulding.
- •
- Abbott Termination Agreement (see pages 43-44). We entered into a mutual release and settlement agreement with Abbott dated as of August 25, 2003 pursuant to which we and Abbott agreed to terminate certain existing agreements between the parties and acknowledged the assignment of certain agreements to Faulding. In addition, Abbott agreed to pay us for certain outstanding invoices and the parties agreed to release each other from claims relating to the agreements terminated. The Abbott Termination Agreement is effective upon the later to occur of the closing of the Abbott-Faulding Transaction and the asset sale by us to Faulding.
- •
- Voting Agreements (see page 44). Leonard P. Shaykin, the Company's Chairman and Chief Executive Officer, and Dr. Sterling K. Ainsworth, the Company's Vice Chairman, who together hold an aggregate of approximately 5.1% of the Company's total outstanding shares of common stock as of the record date, have agreed to vote their shares of the Company common stock in favor of the asset sale. The voting agreements with Messrs. Shaykin and Ainsworth are attached as Annex E and Annex F, respectively.
paclitaxel business to Faulding and the closing of the Company's former manufacturing facility in Boulder, Colorado, known as the "Gunbarrel Facility."
In addition, Abbott and Faulding have agreed to pay all accounts receivable owed to us at closing. We estimate that these accounts receivable will be approximately $1.2 million at the closing but may decrease prior to closing.
INFORMATION CONCERNING VOTING AND SOLICITATION OF PROXIES
The enclosed proxy is solicited by and on behalf of our board of directors for use at the special meeting of stockholders to be held on , 2003, at .m. local time at [location to be determined], and at any adjournment(s) or postponement(s) of that special meeting, for the purposes set forth in the accompanying Notice of Special Meeting. This Proxy Statement is being furnished to holders of our voting common stock, $0.0075 par value per share, as of , 2003, the record date.
Only stockholders of record at the close of business on the record date are entitled to notice of and to vote at the special meeting. As of the record date, there were [30,755,384] outstanding shares of our common stock. Each share is entitled to one vote, and there are no other classes of voting securities outstanding.
Holders of record of shares of our common stock can vote by completing, signing, dating and returning the enclosed proxy card in the postage-paid envelope or by appearing and voting in person by ballot at the special meeting.
Brokers or banks who hold shares of our common stock in "street name" for customers who are the beneficial owners of such shares may not give a proxy to vote those customers' shares in the
13
absence of specific instructions from those customers. The brokers and banks will provide their customers with directions on how to instruct the broker or bank to vote their shares. In addition, a large number of brokers and banks participate in the ADP Investor Communication Services online program. This program provides eligible stockholders who receive a paper copy of this Proxy Statement the opportunity to vote via the Internet or by telephone. If your broker or bank participates in ADP's program, your broker or bank will provide instructions.
Any stockholder giving a proxy has the power to revoke it any time before it is exercised. Proxies may be revoked by filing with our Secretary at our principal executive office, 4840 Pearl East Circle, Suite 300W, Boulder, Colorado, 80301, via mail received prior to the special meeting, a written notice of revocation, or a duly executed proxy bearing a later date. Proxies may also be revoked by attendance at the special meeting and an election to vote in person.
The persons designated as proxies in the enclosed proxy card will vote "FOR" the adoption of a resolution approving the asset sale and "FOR" the proposal to adjourn the special meeting to a later date or dates in certain circumstances, unless otherwise directed on the proxy card or the stockholder "ABSTAINS" from such vote. If any other matters are properly presented at the special meeting, proxies will be voted in accordance with the judgment of the proxy holders.
Proposal 1 to adopt a resolution to approve the proposed asset sale will require the affirmative vote of the holders of a majority of all outstanding shares of common stock. Broker non-votes and absentions with respect to Proposal 1 will have the same effect as votes "AGAINST" the proposal. "Broker non-votes" are shares represented at the special meeting that are held by brokers or nominees as to which instructions have not been received from the beneficial owners or persons entitled to vote those shares, and for which the brokers or nominees do not have discretionary voting power to vote those shares.
Proposal 2 to adjourn the special meeting to a later date or dates in certain circumstances requires the affirmative vote of a majority of the shares of common stock represented in person or by proxy at the special meeting and entitled to vote. Broker non-votes and abstentions with respect to Proposal 2 are not counted as votes "FOR" or "AGAINST" such proposals.
Except as may be provided in the Certificate of Incorporation or otherwise by the Delaware General Corporation Law, any other matter that may be submitted to a stockholder vote will require the affirmative vote of a majority of the shares of common stock represented in person or by proxy at the special meeting and entitled to vote. We are not aware of any other matters to be presented at the special meeting other than those described in this Proxy Statement.
Kai Larson, our Vice President and General Counsel, has been appointed by the board of directors to serve as inspector of election to determine the number of shares of common stock represented and voted at the special meeting.
A quorum of stockholders is necessary to hold a valid meeting. A quorum will be present if at least a majority of the outstanding shares are represented in person or by proxy at the special meeting. Abstentions and broker non-votes are counted for purposes of determining the presence or absence of a quorum. If a quorum is not present at the special meeting, the stockholders entitled to vote, present in person or represented by proxy, may adjourn the special meeting, without notice other than announcement at the special meeting, until a quorum is present or represented. If the special meeting
14
is convened and there are not sufficient votes to approve the asset sale, or if we need additional time to satisfy all conditions to the asset sale, we may move to adjourn the special meeting to a later date to solicit additional proxies or to satisfy any unsatisfied conditions. We have submitted Proposal 2 to our stockholders in order to allow proxies that we have received by the time of the special meeting to be voted for an adjournment, if necessary.
At the adjourned meeting at which a quorum is present or represented, any business may be transacted which might have been transacted at the special meeting as originally noticed. If the adjournment is for more than 30 days, or if after the adjournment a new record date is fixed for the adjourned meeting, a notice of the adjourned meeting will be given to each stockholder entitled to vote at the special meeting.
We will bear the entire cost of solicitation, including the preparation, assembly, printing, and mailing of this Proxy Statement, the form of proxy and any additional soliciting materials sent to stockholders. We may reimburse brokerage firms and other persons representing beneficial owners of shares for their expenses in forwarding solicitation materials to such beneficial owners. We have retained the services of MacKenzie Partners to aid in the solicitation of proxies, and to deliver proxy materials to brokers, nominees, fiduciaries, and other custodians for distribution to beneficial owners of stock and to solicit proxies. MacKenzie Partners will receive a fee of approximately $10,000 and reimbursement of all reasonable out-of-pocket expenses in connection with such solicitation. Proxies may also be solicited personally or by telephone by certain of our directors, officers and regular employees, without additional compensation.
15
PROPOSAL 1
SALE OF OUR PACLITAXEL BUSINESS TO FAULDING
The following is a description of the material aspects of the proposed asset sale and related transactions, including background information relating to the proposed sale and the terms of the asset purchase agreement and certain other agreements entered into (or to be entered into) in connection with the asset purchase agreement. While we believe that the following description covers the material terms of the asset sale, the asset purchase agreement and the related transactions and agreements, the description may not contain all of the information that is important to you. In particular, the following summary of the asset purchase agreement is not complete and is qualified in its entirety by reference to the copy of the asset purchase agreement attached to this Proxy Statement as Annex A and incorporated by reference herein. You should carefully read this Proxy Statement and the other documents to which we refer, including the asset purchase agreement attached as Annex A to this Proxy Statement, for a complete understanding of the terms of the asset sale and related transactions.
Background of the Proposed Asset Sale
Historically, the primary focus of our business has been the production and sale of paclitaxel, a naturally occurring chemotherapeutic anti-cancer agent found in certain species of yew, orTaxus, trees, and a majority of our resources have been devoted to this endeavor. Our strategy for advancing the development and commercialization of paclitaxel has been to form strategic alliances through long-term exclusive agreements with established pharmaceutical companies. We pursued this strategy principally through Faulding and its affiliates in substantially all of the world other than the U.S., Canada, Japan, Israel, the nations of the former Soviet Union and parts of Africa. Our strategic partner in the U.S. and Canada is Abbott Laboratories. Our paclitaxel is currently approved for commercial sale in the U.S., Australia, Israel and more than 25 other countries in the Middle East, Asia and Latin America.
For approximately 9 years prior to 2002 we devoted our efforts to securing FDA approval for the manufacture and sale of paclitaxel in the United States. During this period, we accumulated $100 million of losses principally through research and development activities, and the augmenting of our manufacturing capabilities so that we could supply bulk paclitaxel to our marketing partners, including to Abbott, to meet market demand once we secured FDA approval. These efforts culminated in the FDA's grant of an ANDA to Abbott and us in May 2002.
Our financial arrangement with Abbott included total potential funding of up to $118 million in the form of up to $30 million in development milestones, up to $57 million in marketing milestones, $20 million in secured debt (due in May 2004), and $11 million in equity investments. Marketing milestones were based upon certain annual sales levels. Through December 31, 2002, and excluding product sales, we received $40 million under the agreement, including $11 million in equity investments in exchange for 2 million shares of our common stock, $9 million in development milestones, and $20 million in secured debt. Of the $30 million of potential development-related milestone payments, we received $1 million upon execution of our agreement with Abbott in July 1999 and with the approval of our ANDA, and upon commencement of commercial sales, we received an $8 million payment in May 2002.
Since generic competition for paclitaxel was permitted by the FDA, the market price for the drug has decreased approximately 75% from pre-generic competition levels. We believe that this price erosion was largely brought about by generic paclitaxel competition, including by us, and the reaction to this competition by the established and dominant marketer of the drug, Bristol-Myers Squibb Company. This generic competition has substantially eliminated the prospects of our achieving any additional sales or development milestones that we had negotiated with Abbott. In addition, because our arrangement with Abbott presupposed that the excess cash flow from U.S. product sales combined with the milestone payments would service the $20 million of debt we owe Abbott, the generic competition and
16
Abbott's inability to dramatically increase market share raised concerns about how we would retire the debt when it came due. Furthermore, since Abbott was only able to reach an approximate 15% market share in the United States for paclitaxel, the volume of bulk drug required to meet Abbott's needs did not justify the infrastructure we developed to meet Abbott's anticipated needs. As a result in June 2002, our board authorized, among other restructuring provisions, the downsizing of the Company's paclitaxel manufacturing operations.
During the period immediately following the receipt of our ANDA, the market price of our shares increased to as much as $9.36. However, during the 16 months thereafter, the price declined to as low as $.27, reflecting, we believe, the market's reaction to the paclitaxel price erosion and the diminished commercial prospects for our generic paclitaxel in the United States.
In June 2001, Mayne (then known as Mayne Nickless Limited) made an unsolicited offer to purchase all of the stock of Faulding. The Company understands that, in response, Faulding commenced business combination discussions with a number of other parties. During the course of this the Company made an offer to purchase Faulding's injectable pharmaceutical business that was not accepted by Faulding. Mayne eventually acquired Faulding, and the Company continued its sales and distribution relationship with Faulding.
Beginning in the third quarter of 2002, our management began to develop further strategic responses to the Company's competitive and financial situation. Alternatives considered included terminating all of our development work in both oncology and gene editing, and radically downsizing the Company to live within the gross profit of the paclitaxel business, developing a broader generic oncology business through acquisition, and selling the paclitaxel business and focusing on our long term strategic development programs. Simultaneously, management began to explore the means available to the Company to retire, restructure or refinance its $20 million debt to Abbott. Our discussions on restructuring the debt with Abbott yielded a possible relaxation of repayment terms but did not address the Company's then-current need for capital. Management further investigated repaying the debt with the proceeds of one or more types of public or private financings. However, in the face of a difficult financial market for small pharmaceutical companies, coupled with the Company's declining stock price, all financing alternatives were eventually rejected as providing unattractive terms for the Company.
At the end of the second quarter of 2002, management and the board determined to engage an investment banking firm to assist the Company. After discussions with two banking firms, the board of directors approved the retention of Wells Fargo Securities as the Company's investment banker in June 2002. Wells Fargo Securities was formally engaged in September 2002. In connection with this engagement, Wells Fargo Securities provided investment banking services with respect to several possible acquisitions the Company was reviewing.
Throughout the third quarter of 2002 and thereafter, management reviewed a variety of possible acquisition and financing transactions that were designed to create a greater variety of strategic options for the Company. The possible transactions included acquisitions of pharmaceutical and biotechnology companies that had programs that complemented the Company's businesses or that held substantial amounts of cash that could be used to enhance the Company's financial condition. While the Company pursued several of these opportunities on a confidential basis, none of them became available to the Company on sufficiently attractive terms to take further action than exploratory discussions of possible terms.
While the Company was developing its strategic alternatives, in October 2002, an investment banking firm familiar with our management approached management and offered to arrange a meeting with a pharmaceutical company, referred to in this Proxy Statement as Company A, on the basis that Company A was interested in a business relationship with the Company. Management met with the management of Company A and from this meeting the possibility of Company A's interest in discussing the purchase of our paclitaxel business emerged.
17
At an October 2002 board of directors meeting during which a representative of the Company's outside counsel, Bartlit Beck Herman Palenchar & Scott, participated, the board of directors reviewed the development of the Company's strategic alternatives, including financing and the sale of the paclitaxel business.
Subsequent to the October 2002 board of directors meeting, one of our directors, Marc Ostro, who is also a partner in one of the Company's principal institutional investors, TL Ventures Funds, advised management of TL Ventures Funds' belief that a sale of the Company's paclitaxel business would give TL Ventures Funds the right to have its $8 million subordinated debt investment in the Company repaid. Management of the Company disputed this contention and commenced discussions with TL Ventures Funds regarding alternatives to a redemption that might benefit the Company and at the same time resolve TL Ventures Funds' concerns.
Following the October 2002 board of directors meeting and throughout the fourth quarter of 2002 and the first quarter of 2003, the Company pursued parallel strategies regarding the repayment or refinancing of the Abbott debt, the amendment of the Abbott agreement to make its terms more attractive to a potential purchaser of the Company's paclitaxel business should the Company decide to sell it, the sale of that business, and locating a suitable acquisition opportunity. At the same time, management continued its downsizing and cost reduction efforts by reducing overhead and outsourcing several operational and administrative functions. While the cost cutting efforts were generally quite effective in reducing cash burn, the Company was unable to either reach acceptable terms with Abbott or locate a suitable acquisition or financing opportunity.
In November 2002, the Company and Company A signed a non-disclosure agreement and the Company provided Company A with information about the Company's paclitaxel business over a several week period.
On January 21, 2003, the Company received a letter from Company A expressing its interest in acquiring the Company's polyethoxylated castor oil formulated injectable paclitaxel business. Company A's non-binding proposal contemplated a 90 day exclusive negotiating period, a purchase price of $50 million if no changes were made in the Company's marketing agreement with Abbott and $75 million if Abbott were willing to relinquish its marketing rights. Company A proposed that all liabilities of the paclitaxel business assumed by it, which would appear to include our obligations to Abbott, would reduce the purchase price. The proposed consideration was half cash and half Company A stock, was based on a variety of assumptions about the paclitaxel business being true, and was subject to the completion of due diligence.
On January 22 and 23, 2003, the board of directors met to discuss a broad range of strategic alternatives for the Company including the renegotiation of the Abbott debt, the strategy for a negotiation with TL Ventures Funds regarding its debt (during which Marc Ostro was not present), the closing of the Company's Colorado operations, the sale of all or a portion of the Company's genomics businesses, and the sale of the paclitaxel business. Participating in this meeting were representatives of Wells Fargo Securities and Bartlit Beck Herman Palenchar & Scott. Management advised the board of directors of its views that the optimal way to maximize value for the Company's stockholders was to sell the paclitaxel business and reinvest the proceeds of the sale in the Company's oncology and genomics businesses, a view with which the board of directors concurred. Management noted further that the sale of the paclitaxel business might become a practical and viable alternative given the interest of Company A. Finally, management recommended that the board of directors authorize Wells Fargo Securities to begin the work necessary to auction the paclitaxel business in order to maximize its value to the Company. The board of directors discussed with its legal and financial advisors a sales process and its impact on the board of directors' fiduciary duties and agreed that Wells Fargo Securities should be so engaged.
18
Wells Fargo Securities made a presentation to the board of directors that identified potential acquirers, marketing strategies, and the possible terms of a variety of approaches.
Management then discussed with the board of directors the proposal by Company A. The board of directors believed that the transaction was not sufficiently attractive to forego an auction of the business and that the due diligence contingency, when coupled with a request for exclusivity, could disadvantage the Company by excluding it from marketing the paclitaxel business. The board of directors determined that the Company should not offer Company A an exclusive arrangement unless Company A was willing to make a preemptive offer that exceeded $100 million and it was able to confirm that offer within a relatively short period of time.
Following the board of directors meeting, Company's management advised Company A that the Company would engage in three weeks of exclusive negotiations and due diligence if the purchase price was in excess of $100 million and its offer were 150% of the purchase price if Abbott relinquished its marketing rights. Company A did not respond to this proposal until May 2003, at which time it advised management that it would participate in the auction for the paclitaxel business. Company A did not address either the Company's concern about price or the contingencies to which the bid was subject.
Also following this board meeting, management approached a representative of Faulding and inquired whether Faulding would have an interest in acquiring the Company's paclitaxel business if the Company were interested in selling it. At that time, the representative of Faulding advised management that it would not be interested in becoming a bulk supplier of pharmaceuticals.
On February 11, 2003, the board of directors met to discuss, among other things, the status of discussions with TL Ventures Funds, as to which there remained no agreement. Marc Ostro was not present at the meeting during the discussions regarding TL Ventures Funds. The board instructed management to continue negotiations with TL Ventures Funds.
In early March 2003, the Company and Wells Fargo Securities formally amended the terms of Wells Fargo Securities' engagement to include the possible sale of the paclitaxel business. Throughout February and March 2003, the Company prepared marketing materials for the possible sale and began the necessary work to conduct an auction of the paclitaxel business. Also during this period, management confidentially advised Abbott of the possible sale of its paclitaxel business, and continued to attempt to amend the terms of its agreement with Abbott and to renegotiate the Abbott debt.
In mid-March a representative of Faulding approached management and requested a meeting to discuss the possibility of Faulding's interest in acquiring the Company's paclitaxel business. Management advised this representative confidentially that, because the Company's board of directors had determined to initiate an auction process, the Company would not be interested in discussions with Faulding at this time unless Faulding were willing to make a preemptive bid. Faulding's representative declined to do so and further advised management that Faulding was evaluating the possible acquisition of the Abbott's marketing relationship with the Company. Following this exchange, management contacted a representative of Abbott and determined that Abbott would be willing to consider a sale of its marketing relationship with the Company.
On April 9, 2003, the Company announced its offer to sell its worldwide generic polyethoxylated castor oil formulated injectable paclitaxel business and that proceeds from the sale would be used to accelerate the development and commercialization of therapeutic products based on the Company's gene editing and targeted oncology platforms, to advance the Company's reagent and services business, and to retire the Abbott debt. As announced, the contemplated sale would transfer all of the Company's assets related to the manufacture and sale of paclitaxel. The assets to be sold were to include the Company's bulk paclitaxel manufacturing facilities and related production equipment; plantations; the Company's contracts with Abbott, Faulding, Tzamal Pharma, and JCR Pharmaceuticals Co., Ltd.; regulatory filings; and patents and intellectual property relating to the manufacture and
19
formulation of paclitaxel in polyethoxylated castor oil. The Company said that it did not intend to further comment on the status of the sales effort prior to signing and announcing a definitive agreement.
On April 9, 2003, representatives of the Company and Faulding had a meeting at which Faulding's representatives advised management that Faulding was interested in participating in the Company's announced auction process and that it was also considering the acquisition of Abbott's right to market the Company's paclitaxel in North America.
Following the Company's announcement, Wells Fargo Securities, on behalf of the Company, began to contact a variety of potential bidders with respect to the business to be sold. Beginning in April 2003, and continuing through June 2003, Wells Fargo Securities, on behalf of the Company, invited a total of 49 potential bidders to participate in the bid process and began discussions regarding the timing of management presentations and a due diligence review. With the advice of Wells Fargo Securities, the Company established a minimum bid of $50 million as the point at which bidders would be invited to participate further in the auction process.
On April 28, 2003, the board of directors met, and a representative of Bartlit Beck Herman Palenchar & Scott participated. The board of directors held discussions with Wells Fargo Securities regarding the sales process, contacts with potential bidders and a timeline for completion of a successful sale. Management advised the board of directors of its belief that Abbott might attempt to offer for sale its contractual arrangements with the Company. The board of directors discussed the financial and legal aspects of that attempt and how such a decision might complicate the Company's sales process and dilute the value that the Company could realize for the sale of the paclitaxel business. The board of directors also discussed financing alternatives and the status of continuing discussion with TL Ventures Funds.
Following the board of directors meeting, management advised Abbott of the Company's position that Abbott was not entitled to transfer its contract with the Company without the Company's prior consent. Abbott advised the Company that it disagreed with the Company's interpretation of the agreement between them and that it was free to transfer the agreement on whatever terms it chose.
In mid-May 2003, Wells Fargo Securities, on behalf of the Company, contacted 24 of the potential bidders and informed them that non-binding indications of interest should be submitted no later than May 30, 2003.
In late May 2003, the Company and Abbott reached agreement that confidential information concerning both the Company's and Abbott's North American paclitaxel programs could be given to Faulding.
Of the potential bidders that Wells Fargo Securities contacted, two potential bidders elected to participate in the process, Faulding and a pharmaceutical company referred to as Company B. On May 30, 2003, the Company received indicative offers from Faulding and Company B.
Faulding submitted a non-binding indicative offer of between $50 to $60 million in cash for the assets and liabilities of the Company's paclitaxel business, including all physical assets used in the Company's paclitaxel business and all intellectual property related to the paclitaxel business. In addition, Faulding proposed to assume the Company's real estate leases, including the leases related to the yew plantations and biomass supply, and the marketing, development and supply agreements. Faulding's offer was conditioned upon negotiating an agreement for the acquisition of Abbott's paclitaxel operations. Faulding also proposed that the debt and payables the Company owed to Abbott would be paid off at the closing of Faulding's acquisition of the Company's paclitaxel business. This payoff amount would be deducted from the consideration paid to the Company. Faulding made a number of assumptions in calculating its proposed purchase price including that Faulding would be the
20
first to launch generic paclitaxel in the European market. Faulding proposed to negotiate a purchase agreement with the Company within three to four weeks of undertaking due diligence.
Company B submitted a non-binding indicative offer of $53 million for the assets and liabilities of the Company's paclitaxel business. Company B's offer was contingent on satisfactory completion of due diligence. Company B proposed that the Company would provide certain transition services after closing. After submitting its bid on May 30, 2003, Company B conducted due diligence but declined any further participation in the auction.
Neither offer contained a financing condition to closing.
While other potential bidders expressed varying degrees of interest in conducting due diligence, the Company received no other proposals to acquire the paclitaxel business.
In mid-May 2003, Company A contacted Wells Fargo Securities and noted that it would be difficult for it to meet the Company's May 30, 2003 deadline for submitting bids. At that time, Company A made a number of due diligence inquiries. After receiving these inquiries management contacted a representative of Company A to determine if Company A had an interest in participating in the bid process. The representative of Company A advised management that any interest of Company A would be contingent on its ability also to engage in negotiations with Abbott to acquire Abbott's North American distribution rights. Management contacted Abbott regarding this approach, and a representative of Abbott advised management that Abbott's discussions with Faulding were too advanced for it to commence discussions with any other party. Management advised the representative of Company A of Abbott's position. Company A did not approach the Company thereafter.
On June 5 and 27, 2003, the board of directors met to discuss, among other things, the progress of the auction of the paclitaxel business, in which a representative of Bartlit Beck Herman Palenchar & Scott participated.
In mid-June 2003, representatives of Faulding conducted due diligence review at the Company's offices in Boulder.
On June 17, 2003 the Company advised Faulding that, in the Company's opinion, Abbott could not transfer to Faulding the rights to market the Company's paclitaxel in North America without the Company's approval. Faulding responded that it understood that Abbott did not agree with the Company's position but that Faulding did not intend to consummate any transaction with Abbott unless it had a satisfactory transaction with the Company.
On June 19, 2003, Faulding confirmed to the Company that its primary diligence requests had been fulfilled and that it was preparing for a board of directors presentation regarding the proposed acquisition of the Company's paclitaxel business.
On July 22, 2003, Abbott filed a document with the Securities and Exchange Commission regarding a transaction it had entered into with Faulding for Faulding to acquire shares of the Company's common stock held by Abbott. Faulding's parent company, Mayne, then issued a press release stating that this agreement with Abbott was contingent upon Faulding entering into an agreement to acquire the Company's worldwide paclitaxel business. Abbott holds 2 million shares of the Company's common stock, or approximately 6.5% of the Company's total outstanding common stock.
On July 23, 2003, Faulding made a written offer to the Company to acquire the paclitaxel business. The offer contemplated the acquisition of all of the assets of the Company's paclitaxel assets and the assumption of only limited liabilities of that business for cash consideration of $68 million. The offer also contained a number of unacceptable terms and conditions to closing that made it uncertain from the Company's point of view.
21
On July 30, 2003, the board of directors held a meeting at which it reviewed Faulding's offer and a proposal by TL Ventures to restructure its subordinated debentures. A representative of Bartlit Beck Herman Palenchar & Scott participated at this meeting. After a presentation by management, the board of directors authorized management to negotiate a transaction with Faulding and to reject TL Ventures Funds' proposal. Mr. Ostro did not participate in the discussion regarding TL Ventures Funds.
On August 4, 2003, the Company advised Faulding of a series of issues raised by Faulding's bid that had to be resolved prior to any further discussions taking place. These included an increase in the purchase price, the assumption of the liabilities related to the business, the resolution of all outstanding commercial issues with Abbott and Faulding under their partnering agreements with the Company, and other contract matters. Through various discussions among representatives of the Company and its legal and financial advisors, and Faulding and its legal and financial advisors over a seven day period, the majority of these issues were resolved in a manner sufficient to cause the Company to believe that further discussions with Faulding would be fruitful. Among other things, Faulding indicated that it would increase its purchase price to $70 million in cash.
From August 19 through August 25, 2003, the Company and Faulding through their business, legal and financial representatives negotiated the asset purchase agreement and related agreements. Among other things, during this time, Faulding agreed to raise its purchase price to $71.7 million, subject to a closing date inventory adjustment, agreed to a termination and mutual release of the existing partner agreements with the Company, including the Abbott partner agreements. Faulding also agreed to pay all outstanding accounts receivable at closing, which, together with accounts receivable from Abbott which are to be repaid, were approximately $8.1 million as of July 2, 2003. Finally, Abbott and the Company reached an agreement terminating and releasing the remaining agreement between Abbott and the Company.
On August 25, 2003, the board of directors met to consider approval of the asset sale. Representatives of Wells Fargo Securities and Bartlit Beck Herman Palenchar & Scott participated in this meeting. At this meeting the board of directors reviewed and considered the asset purchase agreement that representatives of the Company and its legal and financial advisors had negotiated with Faulding. A representative of Bartlit Beck Herman Palenchar & Scott discussed with the board of directors the discharge of the directors' fiduciary duties in connection with the asset sale and made a presentation to the board of directors about the terms and conditions of the asset purchase agreement (that was provided to the board of directors), including the non-solicitation provision, the representations and warranties that the Company would be asked to make, conditions precedent to closing the asset sale, the termination fee potentially payable by the Company, certain covenants and the indemnification provisions and the consideration to be received by Company.
A representative of Wells Fargo Securities reviewed his firm's opinion regarding the fairness, from a financial point of view, of the consideration to be received by the Company in the transaction and supporting materials (that were provided to the board of directors) with the board of directors. In his presentation he reviewed and explained the procedures followed, the assumptions made, the matters considered, and the limitations on the reviews undertaken in his firm's analysis of the proposed asset sale. These considerations are set forth in more detail in the section entitled "—Opinion of the Company's Financial Advisor." Wells Fargo Securities further reviewed with the board of directors the auction process that had been conducted and advised the board of directors that it was Wells Fargo Securities' opinion that the consideration to be offered in the proposed asset sale was fair, as of that date and from a financial point of view, to the Company.
The board of directors concluded that, after conducting an auction process through which a variety of proposals to acquire the paclitaxel business were reviewed, and considering management's and Wells Fargo Securities' recommendations, Faulding was the purchaser candidate that proposed a transaction that was most likely to produce the highest value reasonably attainable for the Company's paclitaxel
22
business. All members of the Company's board of directors approved the asset sale and recommended that stockholders of the Company approve a resolution approving the asset sale.
On August 26, 2003, the Company and Faulding announced the signing of the asset purchase agreement.
On September 8, 2003, TL Ventures Funds informed the Company that it intends to pursue legal remedies if the parties cannot resolve promptly the issue regarding its subordinated debentures. The Company disputes TL Ventures Funds' position and will vigorously defend its legal position.
Consideration of the Asset Sale by the Company's Board of Directors
Factors Considered by the Company's Board of Directors
The Company's board of directors consulted with senior management and its financial and legal advisors and considered a number of factors, including the following, in evaluating the asset sale:
- •
- the board of directors' familiarity with and review of the Company's paclitaxel business, and the Company's prospects and financial condition;
- •
- the potential strategic benefits and potential negative factors associated with the asset sale;
- •
- the less attractive alternative terms of transactions in the current economic environment;
- •
- the current financial market conditions and historical market prices, volatility and trading information with respect to our common stock;
- •
- the terms of the asset purchase agreement and related agreements;
- •
- the fact that the board of directors conducted an extensive review of strategic alternatives and concluded that a sale of assets provided the best method of maximizing stockholder value;
- •
- the presentation of the Company's financial advisor, Wells Fargo Securities, and its fairness opinion (the full text of which is attached as Annex B to this Proxy Statement) setting forth the procedures followed, the assumptions made, the matters considered, and the limitations on the review undertaken;
- •
- the risk that the asset sale might not be consummated;
- •
- the potential impact of the asset sale on our customers, strategic partners and employees;
- •
- the strategic and financial options currently available to us, including the option to continue to operate the paclitaxel business;
- •
- the historical relationship with Faulding and its understanding of the paclitaxel marketplace; and
- •
- the Company's familiarity with Faulding.
The foregoing discussion of the information and factors considered by our board of directors, while not exhaustive, includes the material factors considered by the board of directors in its review of the proposed asset sale. In view of the variety of factors considered in connection with its evaluation of the asset sale, the Company's board of directors did not find it practicable to, and did not, quantify or otherwise assign relative or specific weight or values to any of these factors, and individual directors may have given different weights to different factors.
23
The Company's Reasons for the Asset Sale
In reaching its decision to approve the asset sale, our board of directors considered several potential strategic benefits and material factors pertaining to the asset sale, including the following:
- •
- the belief that, after reviewing the Company's financial condition, results of operations and business and earning prospects, the most promising long-term opportunities for the Company and its stockholders were likely to be created by selling the paclitaxel business and focusing on the development and commercialization of novel proprietary therapies that address specific clinical needs, rather than in continuing to pursue a generic pharmaceutical, manufacturing strategy with the Company's paclitaxel business;
- •
- the fact that we will retain our intellectual property not used in connection with the business to be sold after the asset sale;
- •
- our board of directors' belief that, after the consummation of the asset sale, the Company will have approximately $55 to $60 million of cash on hand (assuming no payments to TL Ventures in connection with its claim regarding redemption of its debentures), a controllable cash burn rate prior to incurring clinical expenses, a seasoned pharmaceutical research, development and clinical team, and promising therapeutic opportunities related to oncology and hereditary disease;
- •
- the conclusion of the sale should significantly increase the probability of successfully developing products based on our technology platforms in oncology and gene editing;
- •
- the extensive auction process conducted by the board of directors and the belief, after completion of this process, that the alternatives to the asset sale to Faulding were not reasonably likely to provide equal or greater value to the Company and our stockholders;
- •
- the board of directors' judgment, based in part on the advise of Wells Fargo Securities, that, despite offers that may be received from other parties, it was inadvisable to forgo the asset sale to Faulding and pursue such other alternatives because of delays, uncertainties and contingencies, and business, economic and market risks involved;
- •
- that the asset sale represents the most favorable alternative currently available to us to maximize stockholder value;
- •
- the uncertainties and volatility associated with the future market for generic paclitaxel;
- •
- the opinion of Wells Fargo Securities, the Company's financial advisor, to the effect that as of August 25, 2003, the consideration payable to the Company set forth in the asset purchase agreement was fair, from a financial point of view, to the Company;
- •
- the belief that, while no assurances can be given, the asset sale is likely to be completed because of the limited nature of the closing conditions included in the asset purchase agreement;
- •
- the belief that the strategic benefits to the Company contemplated in connection with the asset sale are likely to be achieved within a reasonable time frame;
- •
- the amount of the consideration to be paid in the asset sale;
- •
- the terms of the asset purchase agreement, including our ability to terminate the agreement in certain circumstances, including in connection with certain unsolicited third party superior offers, and the limitation on our potential indemnification obligations for breaches of certain of our representations and warranties to Faulding under the terms of the asset purchase agreement; and
24
- •
- the Company's opportunity to resolve outstanding issues it had with Abbott in the context of Faulding's recently negotiated transaction with Abbott, pursuant to which Faulding will acquire certain assets and assume certain liabilities from Abbott that relate to the Company's development, license and supply agreement with Abbott.
The Company's board of directors also considered a number of potentially negative factors in reaching its decision to approve the asset sale, including the following:
- •
- the risk that the potential strategic benefits of the asset sale may not be realized, in part or at all;
- •
- the resultant loss of sales and gross profit from the paclitaxel business including the reliance on current assets to fund product development;
- •
- the risk that the asset sale may not be consummated, including the risks associated with obtaining the necessary approval of our stockholders required to complete the asset sale, notwithstanding the voting agreements obtained from holders of approximately 5.1% of our common stock;
- •
- the risk of management and employee disruption associated with the asset sale and the transition services agreement, including the risk that key technical, marketing and management personnel might not remain employed by us after the asset sale;
- •
- the provision of the asset purchase agreement that permits Faulding to terminate the agreement if, among other things, the Company's board of directors withdraws its recommendation in favor of the asset sale or recommends another acquisition proposal;
- •
- the provision of the asset purchase agreement that requires us to pay liquidated damages to Faulding in the amount of $2 million if the asset purchase agreement is terminated under certain circumstances;
- •
- the interests of our directors and executive officers that may be different from, or in addition to, those of our stockholders generally, including the potential receipt of change of control payments by certain of our executive officers under severance agreements, the grant of stock options to senior management of the Company and the interests of TL Ventures Funds;
- •
- our obligations to provide services to and to rely on services from Faulding for a period of time following the closing pursuant to the terms of the transition services agreement;
- •
- the significant costs involved in consummating the asset sale;
- •
- the unlimited indemnification obligations under the terms of the asset purchase agreement with respect to particular matters, including the breach of certain of our representations and warranties;
- •
- the risk that the cash proceeds from the Faulding transaction, if realized, will not be sufficient to fund our strategic plans for the development of new proprietary therapeutic candidates;
- •
- the risk that the outcome of our dispute with TL Ventures Funds regarding redemption of their debentures will reduce the proceeds available to us from the Faulding transaction; and
- •
- the potential negative effect on our stock price as a result of the public announcement of the asset sale.
The Company board of directors noted that the voting agreements and the liquidated damage provisions of the asset purchase agreement could have the effect of discouraging alternative proposals for a business combination or asset sale between the Company and a third party. However, the board of directors concluded that the amount of the liquidated damages, payable in certain circumstances
25
upon termination of the asset purchase agreement, and the circumstances under which such liquidated damages may be payable, were necessary to induce Faulding to enter into the asset purchase agreement.
The Company's board of directors did not believe that the negative factors were sufficient, individually or in the aggregate, to outweigh the potential strategic benefits of the asset sale.
Recommendation of the Company's Board of Directors
THE COMPANY'S BOARD OF DIRECTORS HAS UNANIMOUSLY DETERMINED THAT THE SALE OF THE COMPANY'S PACLITAXEL BUSINESS TO FAULDING IS ADVISABLE AND IN THE BEST INTERESTS OF THE COMPANY'S STOCKHOLDERS. ACCORDINGLY, THE COMPANY'S BOARD OF DIRECTORS HAS UNANIMOUSLY APPROVED THE ASSET SALE AND RECOMMENDS THAT STOCKHOLDERS VOTE "FOR" APPROVAL OF PROPOSAL 1.
Opinion of the Company's Financial Advisor
Under a letter agreement dated September 3, 2002, as amended on March 5, 2003 and August 20, 2003, the Company engaged Wells Fargo Securities to provide financial advisory and investment banking services in connection with the transaction and to render an opinion as to the fairness, from a financial point of view, of the consideration to be received in the transaction. See Annex B for a copy of the full opinion. On August 25, 2003, at a meeting of the Company's board of directors held to evaluate the transaction, Wells Fargo Securities delivered to the Company's board of directors its oral opinion, subsequently confirmed in a written opinion dated as of August 25, 2003, that as of that date and based on the assumptions made, matters considered and the limitations on the review undertaken described in the written opinion, the consideration to be received by the Company in the transaction was fair, from a financial point of view, to the Company. The consideration to be received in the transaction was determined through negotiations between the respective managements of the Company and Faulding. Although Wells Fargo Securities did assist the management of the Company in those negotiations, Wells Fargo Securities was not asked by, and did not recommend to, the Company that any specific amount constituted the proper amount of consideration to be received in the transaction. Wells Fargo Securities has consented to the inclusion of its opinion, and this summary of its opinion, in this Proxy Statement.
You should consider the following when reading this discussion of the opinion of the Company's financial advisor:
- •
- We urge you to read carefully the entire opinion of Wells Fargo Securities, which is set forth in Annex B to this Proxy Statement and is incorporated by reference. The following description of the Wells Fargo Securities opinion is qualified by reference to the full opinion located in Annex B. The full opinion sets forth, among other things, the assumptions made, the matters considered and the limitations on the review undertaken by Wells Fargo Securities.
- •
- The Wells Fargo Securities opinion was prepared for the information of the Company's board of directors in connection with its evaluation of the transaction and is not intended to be and does not constitute a recommendation to any stockholder of the Company as to how that stockholder should vote, or take any other action, with respect to the transaction.
- •
- The Wells Fargo Securities opinion did not address the relative merits of the transaction and the other business strategies that the Company's board of directors has considered or may have been considering, nor did it address the decision of the Company's board of directors to proceed with the transaction.
26
- •
- The Wells Fargo Securities opinion was necessarily based upon market, economic and other conditions as in effect on, and information made available to Wells Fargo Securities as of, the date of the opinion. You should understand that subsequent developments may affect the conclusion expressed in the Wells Fargo Securities opinion, and that Wells Fargo Securities disclaims any undertaking or obligation to advise any person of any change in any matter affecting its opinion which may come or be brought to Wells Fargo Securities' attention after the date of its opinion.
- •
- The Wells Fargo Securities opinion was limited to the fairness, from a financial point of view and as of August 25, 2003, to the Company of the consideration to be received in the transaction.
- •
- The Wells Fargo Securities opinion does not address the value of the Company's remaining business after the transaction.
- •
- The Wells Fargo Securities opinion does not express any opinion regarding any payment which may be received by the Company pursuant to the asset purchase agreement in connection with the litigation involving Mylan Laboratories, Inc.
Opinion and Analysis of Wells Fargo Securities
In connection with the preparation of the Wells Fargo Securities opinion, Wells Fargo Securities:
- •
- reviewed certain publicly available financial statements and other business and financial information of the Company, the paclitaxel business and otherwise relating to the transaction;
- •
- reviewed certain internal financial statements and other financial and operating data, including certain financial forecasts and other forward looking information, concerning the paclitaxel business, prepared by the management of the Company;
- •
- held discussions with the management of the Company concerning the business, past and current operations, financial conditions and future prospects of the paclitaxel business;
- •
- reviewed the financial terms and conditions set forth in the asset purchase agreement;
- •
- reviewed the stock price and trading history of the Company's common stock;
- •
- compared the financial performance of the paclitaxel business with that of certain other publicly traded companies in businesses comparable with the paclitaxel business;
- •
- compared the financial terms of the transaction with the financial terms, to the extent publicly available, of other transactions Wells Fargo Securities deemed relevant;
- •
- prepared a discounted cash flow analysis of the paclitaxel business;
- •
- participated in discussions and negotiations among representatives of the Company and Faulding and their financial and legal advisors; and
- •
- made such other studies and inquiries, and took into account such other matters as Wells Fargo Securities deemed relevant, including Wells Fargo Securities' assessment of general economic, market and monetary conditions.
In its review and analysis, and in arriving at its opinion, Wells Fargo Securities assumed and relied upon the accuracy and completeness of all of the financial and other information provided to it, including information furnished to it orally or otherwise discussed with it by the Company's management, or publicly available, and neither attempted to verify, nor assumed responsibility for verifying, any of such information. Wells Fargo Securities relied upon the assurances of the Company's management that it was not aware of any facts that would make such information inaccurate or
27
misleading. Furthermore, Wells Fargo Securities did not obtain or make, or assume any responsibility for obtaining or making, any independent evaluation or appraisal of the properties, assets or liabilities, contingent or otherwise, of the Company or the paclitaxel business, nor was it furnished with any such evaluation or appraisal.
With respect to the financial forecasts and projections, and the assumptions and bases therefor, for the paclitaxel business that Wells Fargo Securities reviewed, Wells Fargo Securities has assumed that:
- •
- these forecasts and projections were reasonably prepared in good faith on the basis of reasonable assumptions;
- •
- these forecasts and projections reflect the best currently available estimates and judgments of the Company's management as to the future financial condition and performance of the paclitaxel business; and
- •
- these forecasts and projections will be realized in the amounts and in the time periods currently estimated.
In addition, Wells Fargo Securities assumed that:
- •
- the transaction will be consummated upon the terms set forth in the asset purchase agreement without material alteration thereof;
- •
- the historical financial statements of the Company reviewed by Wells Fargo Securities were prepared and fairly presented in accordance with U.S. generally accepted accounting principles consistently applied; and
- •
- the amount of the consideration to be received in the transaction will not be reduced as a result of indemnification, purchase price adjustment (other than the Inventory Adjustment Amount, as defined in the asset purchase agreement) or other provisions of the asset purchase agreement.
Wells Fargo Securities expressed no opinion as to:
- •
- any tax or other consequences that might result from the transaction; and
- •
- any legal or accounting matters, as to which Wells Fargo Securities understands that the Company obtained such advice as it deemed necessary from qualified professionals.
Wells Fargo Securities relied as to all legal matters relevant to rendering its opinion on the advice of its counsel. The following is a summary of the material financial analyses performed by Wells Fargo Securities in connection with rendering its opinion. This summary is not a complete description of all of the analyses performed by Wells Fargo Securities. Some of the information in this section is presented in tabular form.In order to better understand the financial analyses performed by Wells Fargo Securities, you must read the tables together with the text accompanying each table. The opinion is based upon the totality of the various analyses performed by Wells Fargo Securities and no particular portion of the analyses has any merit standing alone.
Comparable Companies Analysis
Using publicly available information, including Securities and Exchange Commission filings, press releases and Wall Street research, Wells Fargo Securities calculated the enterprise value, which Wells Fargo Securities defined as market capitalization plus total debt plus preferred stock plus minority interests minus cash and short term investments, as a multiple of estimated revenue and as a multiple of estimated earnings before interest, taxes, depreciation and amortization, or EBITDA, for the following companies:
- •
- Degussa AG
28
- •
- Clariant International Ltd
- •
- DSM N.V.
- •
- Rhodia Inc.
- •
- Lonza Group Ltd
- •
- Albemarle Corporation
- •
- Cambrex Corporation
- •
- Aceto Corporation
As set forth below, this analysis indicated the high, mean, median, "stripped mean," defined as the average of the data set excluding the highest and lowest data points, and low of multiples for the periods ending December 31, 2002, December 31, 2003, December 31, 2004 and the last twelve months, or LTM, defined as the twelve month period ending on June 30, 2003, and compared these multiples to those for the paclitaxel business for the same periods, with the exception of LTM, which refers to the twelve month period ending on July 2, 2003 when used in the context of the paclitaxel business:
| | Enterprise Value as a Multiple of | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| | 2002 | LTM Estimated | 2003 Estimated | 2004 Estimated | ||||||||||
| | Revenue | EBITDA | Revenue | Revenue | EBITDA | Revenue | EBITDA | |||||||
| High | 1.85x | 14.86x | 1.86x | 1.86x | 8.36x | 1.81x | 7.88x | |||||||
| Mean | 1.03x | 7.09x | 1.00x | 1.02x | 6.48x | 1.04x | 6.12x | |||||||
| Median | 0.84x | 6.45x | 0.79x | 0.76x | 6.67x | 1.05x | 6.74x | |||||||
| Stripped Mean | 0.99x | 6.26x | 0.93x | 0.96x | 6.65x | 1.01x | 6.50x | |||||||
| Low | 0.51x | 4.25x | 0.51x | 0.49x | 3.78x | 0.37x | 2.84x | |||||||
| Paclitaxel Business | 2.39x | 7.77x | 2.54x | 3.12x | 8.88x | 2.88x | 4.47x | |||||||
Wells Fargo Securities obtained revenue and earnings data for the paclitaxel business from the Company's management. Wells Fargo Securities then applied a range of selected multiples of revenue for 2002, LTM, 2003 and 2004 and of EBITDA for 2002, 2003 and 2004 derived from the selected companies listed above to the corresponding financial data of the paclitaxel business. The analysis of these periods indicated the following implied net asset value ranges for the paclitaxel business:
| | Implied Net Asset Value Range | |||||||
|---|---|---|---|---|---|---|---|---|
| | Relevant Range | Low | High | |||||
| | | (In Millions) | ||||||
| Revenue Multiples | ||||||||
| Total Enterprise Value / 2002 Revenues | 0.84x – 2.49x | $ | 28.6 | $ | 85.2 | |||
| Total Enterprise Value / LTM Revenues | 0.79x – 2.46x | $ | 25.4 | $ | 79.5 | |||
| Total Enterprise Value / 2003 Revenues | 0.76x – 2.46x | $ | 20.0 | $ | 64.6 | |||
| Total Enterprise Value / 2004 Revenues | 1.05x – 2.43x | $ | 29.8 | $ | 69.1 | |||
| EBITDA Multiples | ||||||||
| Total Enterprise Value / 2002 EBITDA | 6.45x – 7.34x | $ | 67.9 | $ | 77.4 | |||
| Total Enterprise Value / 2003 EBITDA | 6.67x – 9.36x | $ | 61.4 | $ | 86.2 | |||
| Total Enterprise Value / 2004 EBITDA | 6.74x – 8.88x | $ | 123.5 | $ | 162.7 | |||
| Adjusted Transaction Value | $81.8 | |||||||
The adjusted transaction value was calculated using the purchase price for the paclitaxel business of $71.7 million less assumed liabilities of $2.6 million plus accounts receivable, forgiven liabilities and revenue for delivered inventory of $12.8 million for a total adjusted transaction value of $81.8 million.
29
Selected Precedent Transactions
Using publicly available information, including Securities and Exchange Commission filings, press releases and Wall Street research, Wells Fargo Securities reviewed and analyzed, among other things, the transaction values and implied transaction multiples in the following selected transactions, listing the acquired company followed by the acquirer and with the date these transactions were publicly announced in parentheses, that Wells Fargo Securities believed to be reasonably comparable to the proposed transaction:
- •
- Solutia, Inc. (resins, additives, adhesives business)/UCB S.A. (December 3, 2002)
- •
- Marathon Biopharmaceuticals/Cambrex Corporation (October 30, 2001)
- •
- Martinswerk GmbH/Albemarle Corporation (April 27, 2001)
- •
- ChemFirst Inc. (fine chemicals business)/Albemarle Corporation (June 14, 2001)
- •
- Bio Science Contract Production Corp./Cambrex Corporation (April 30, 2001)
- •
- Hercules Incorporated (hydrocarbon and portions of rosins business)/Eastman Chemical Company (April 2, 2001)
- •
- Catalytica, Inc. (pharmaceutical business)/DSM N.V. (August 2, 2000)
- •
- ChiRex, Inc./Rodia SA (July 24, 2000)
- •
- Wyckoff Chemical Co./Catalytica, Inc. (July 15, 1999)
- •
- FMC Corporation (bioproducts business)/Cambrex Corporation (June 7, 1999)
- •
- NSC Technologies/Great Lakes Chemical (April 9, 1999)
- •
- Irotec Laboratories/Cambrex Corporation (March 12, 1999)
Wells Fargo Securities reviewed the transaction value at the announcement of the transaction in each of the selected transactions as a multiple of LTM revenue. Based on these calculations, Wells Fargo Securities noted the high, mean, median, stripped mean and low of the multiples in each of the selected acquisitions listed above as summarized in the following table and compared these multiples to those for the paclitaxel business:
| | Transaction Value at Announcement of Transaction as a Multiple of LTM | |
|---|---|---|
| | Revenue | |
| High | 5.56x | |
| Mean | 2.21x | |
| Median | 1.90x | |
| Stripped Mean | 2.06x | |
| Low | 0.44x | |
| Paclitaxel Business | 2.54x |
30
Wells Fargo Securities obtained LTM revenues for the paclitaxel business from the Company's management. Wells Fargo Securities then applied a range of selected multiples of LTM revenue from the selected transactions to the LTM revenue of the paclitaxel business.
| | Implied Net Asset Value Range | |||||||
|---|---|---|---|---|---|---|---|---|
| | Relevant Range | Low | High | |||||
| | | (In Millions) | ||||||
| Paclitaxel Business | 1.90x – 2.00x | $ | 61.3 | $ | 64.4 | |||
| Adjusted Transaction Value | $81.8 | |||||||
The adjusted transaction value was calculated using the purchase price for the paclitaxel business of $71.7 million less assumed liabilities of $2.6 million plus accounts receivable, forgiven liabilities and revenue for delivered inventory of $12.8 million for a total adjusted transaction value of $81.8 million.
Discounted Cash Flow Analysis
Using the Company's management estimates, Wells Fargo Securities performed a discounted cash flow analysis on the net cash flows of the paclitaxel business for calendar years 2003 through 2007. Wells Fargo Securities first discounted the net cash flows through the calendar year 2007 using discount rates ranging from 10.0% to 20.0%. Wells Fargo Securities then added the present value of these net cash flows to the terminal value of the paclitaxel business in the calendar year ending 2007, discounted back to the present at the same discount rates. Wells Fargo Securities computed the terminal value of the paclitaxel business in the calendar year ending 2007 under a revenue methodology, whereby estimated revenue for calendar year 2007 was multiplied by terminal revenue multiples ranging from 0.75x to 2.25x, and under an EBITDA methodology, whereby estimated revenue for calendar year 2007 was multiplied by terminal EBITDA multiples ranging from 6.0x to 8.0x. Applying the above ranges of discount rates, terminal revenue multiples and terminal EBITDA multiples to net cash flows of the paclitaxel business yielded the following implied net asset value ranges:
| | Implied Net Asset Value Range | |||||
|---|---|---|---|---|---|---|
| | Low | High | ||||
| | (In Millions) | |||||
| Revenue Methodology | $ | 71.5 | $ | 108.3 | ||
| EBITDA Methodology | $ | 189.8 | $ | 201.3 | ||
| Adjusted Transaction Value | $81.8 | |||||
As used above "net cash flows" means free cash flow to the Company. The adjusted transaction value was calculated using the purchase price for the paclitaxel business of $71.7 million less assumed liabilities of $2.6 million plus accounts receivable, forgiven liabilities and revenue for delivered inventory of $12.8 million for a total adjusted transaction value of $81.8 million.
Other Factors
No company, business or transaction compared in any of the above analyses is identical to the Company, Faulding or the transaction. Accordingly, an analysis of the results of the foregoing is not entirely mathematical. Rather, an analysis of the results of the foregoing involves complex considerations and judgments concerning differences in financial and operating characteristics and other factors that could affect the public trading, acquisition and other values of comparable companies, precedent transactions or the business segment, company or transaction to which they are being compared. In addition, various analyses performed by Wells Fargo Securities incorporate projections prepared by Wall Street analysts using only publicly available information. These projections may or may not be accurate.
31
While this summary describes the analyses and factors that Wells Fargo Securities deemed material in its presentation to the Company's board of directors, it is not a comprehensive description of all analyses and factors considered by Wells Fargo Securities. The preparation of a fairness opinion is a complex process that involves various determinations as to the most appropriate and relevant methods of financial analysis and the application of these methods to the particular circumstances. Therefore, a fairness opinion is not readily susceptible to partial analysis or summary description. In arriving at its opinion, Wells Fargo Securities did not attribute any particular weight to any analysis or factor considered by it, but rather made qualitative judgments as to the significance and relevance of each analysis and factor. Accordingly, Wells Fargo Securities believes that its analysis must be considered as a whole and that selecting portions of its analyses and of the factors considered by it, without considering all analyses and factors, could create a misleading or incomplete view of the evaluation process underlying its opinion. Several analytical methodologies were employed and no one method of analysis should be regarded as critical to the overall conclusion reached by Wells Fargo Securities. Each analytical technique has inherent strengths and weaknesses, and the nature of the available information may further affect the value of particular techniques. The conclusion reached by Wells Fargo Securities is based on all analyses and factors taken as a whole and also on the application of Wells Fargo Securities' own experience and judgment. This conclusion may involve significant elements of subjective judgment and qualitative analysis. Wells Fargo Securities expresses no opinion as to the value or merit standing alone of any one or more parts of the analyses it performed. In performing its analyses, Wells Fargo Securities made numerous assumptions with respect to industry performance, general business and other conditions and matters, many of which are beyond the control of the Company or Wells Fargo Securities. Any estimates contained in these analyses are not necessarily indicative of actual values or predictive of future results or values, which may be significantly more or less favorable than those suggested by these analyses. Accordingly, analyses relating to the value of businesses do not purport to be appraisals or to reflect the prices at which these businesses actually may be sold in the future, and these estimates are inherently subject to uncertainty.
As compensation for its services in connection with the transaction, the Company has paid Wells Fargo Securities a customary fee upon the delivery of the Wells Fargo Securities opinion, and the Company will pay Wells Fargo Securities an additional customary fee upon the closing of the transaction. In addition, the Company has agreed to indemnify Wells Fargo Securities for certain liabilities that may arise out of its engagement by the Company and the rendering of Wells Fargo Securities' opinion. Wells Fargo Securities has performed investment banking services for the Company in the past and has received customary fees for such services. In the ordinary course of business, Wells Fargo Securities may trade in the Company's securities for Wells Fargo Securities' own account and the account of its customers and, accordingly, may at any time hold a long or short position in the Company's securities.
Wells Fargo Securities was retained based on its reputation as an internationally recognized investment banking firm and based on its experience as a financial advisor in connection with mergers and acquisitions and in securities valuations generally. As part of its investment banking business, Wells Fargo Securities had been frequently engaged in the valuation of businesses and their securities in connection with mergers and acquisitions, negotiated underwritings, secondary distributions of securities, private placements and other purposes.
General
Under the terms of the asset purchase agreement, Faulding has agreed to purchase our paclitaxel business. We will sell the assets of the paclitaxel business for a purchase price of $71.7 million in cash minus an inventory adjustment, if any, to reflect the Company's actual inventory as of the closing, and Faulding will assume certain liabilities associated with the paclitaxel business. Approximately
32
$21.7 million of the proceeds of the purchase price will be paid to Abbott or its designee at closing to retire all outstanding debt and payables we owe to Abbott.
Assets to be Sold
We have agreed to sell to Faulding all of the assets of our paclitaxel business as of the closing, including:
- •
- certain specified patents, know-how, clinical trials data, paclitaxel data, and other intellectual property that we use in our paclitaxel business;
- •
- certain leases of real property related to our cultivation of yew trees and other leases of real property related to our paclitaxel business;
- •
- all of our tangible personal property that we use in our paclitaxel business, such as machinery, equipment, biomass, and inventories of raw materials, supplies, and finished goods;
- •
- certain contracts related to our paclitaxel business, including development and marketing agreements, manufacturing agreements, cultivation agreements, and certain license and other agreements related to our intellectual property;
- •
- all of our claims or causes of action relating to our paclitaxel business, including the claim filed by us and Abbott against Mylan Laboratories alleging patent infringement relating to certain paclitaxel patents;
- •
- all of our paclitaxel records and registrations;
- •
- the computer system related to our paclitaxel business; and
- •
- books and records related to our paclitaxel business.
Assets to be Retained
We are not selling to Faulding, and will retain, the following assets following the closing of the proposed asset sale:
- •
- documentation related to our corporate existence and qualifications to do business;
- •
- cash on hand as of the closing;
- •
- accounts receivable that accrue in accordance with generally accepted accounting principles prior to the closing;
- •
- all assets, including patents and other intellectual property, not related to or used in the operation of our paclitaxel business;
- •
- our rights under the asset purchase agreement, including the consideration to be paid to us pursuant to the asset purchase agreement, and any other agreements between us and Faulding on or after the date of the asset purchase agreement;
- •
- our insurance policies and insurance contracts;
- •
- employee benefit plans, policies, and contracts; and
- •
- certain retained inventory.
33
Liabilities to be Assumed
Faulding will be responsible for all liabilities associated with its ownership, operation and use of the acquired assets and the operation of the paclitaxel business after the closing. Faulding will assume only certain of our liabilities, which generally are limited to:
- •
- liabilities under the contracts assumed by Faulding, including the development and marketing agreements, cultivation agreements, certain intellectual property license agreements relating to, among other things, patents, certain computer hardware and software, certain purchase orders and manufacturing agreements, and certain other contracts related to our paclitaxel business;
- •
- Faulding will be responsible for obtaining and maintaining certain environmental permits related to our paclitaxel business from and after the closing; and
- •
- liabilities related to certain patent and product registration issues that we described to Faulding, including liabilities related to the claim filed by us and Abbott against Mylan Laboratories alleging patent infringement relating to certain paclitaxel patents.
Liabilities to be Retained
We will retain all liabilities not specifically assumed by Faulding following the closing of the asset sale, including, without limitation:
- •
- accounts payable and liabilities for materials and services connected to the manufacture, finishing, distribution, and sale of any product to the extent each arose or accrued prior to the closing;
- •
- liabilities arising out of or relating to any product liability, breach of warranty, product recall, product returns, or similar claim to the extent related to our paclitaxel business prior to the closing;
- •
- liabilities relating to employment, compensation, and benefits for our employees prior to the closing;
- •
- liabilities relating to contracts and purchase orders not assumed by Faulding;
- •
- liabilities relating to our operation of the paclitaxel business prior to the closing; and
- •
- liabilities relating to the asset purchase agreement and any agreement between us and Faulding entered into on or after the closing.
Purchase Price
The purchase price to be paid by Faulding is $71.7 million in cash, minus an inventory adjustment to reflect the amount, if any, by which our inventory as of the closing is different than its value as of May 30, 2003. Approximately $21.7 million of the proceeds of the purchase price will be paid to Abbott or its designee at closing to retire all outstanding debt and payables we owe to Abbott. In addition, if the resolution of the claims asserted against the adverse parties in the Mylan litigation, referred to above, results in the entry of a final order by any court that requires such parties to pay damages as part of the resolution of that litigation (including any settlement thereof), Faulding has agreed to pay us a portion of any cash payment received by Faulding from any adverse party in that litigation as and when received by Faulding. Faulding is not required to pursue the litigation, to resolve the litigation in a way that results in monetary damages, or to provide any benefit to us of any non-monetary resolution of the litigation.
34
Closing
The closing of the asset sale is expected to take place as soon as practicable after the special meeting is held (assuming approval of the asset sale by our stockholders) and all conditions to closing specified in the asset purchase agreement are satisfied or waived.
Representations and Warranties
We have made a number of customary representations and warranties, subject in some cases to customary qualifications, to Faulding in the asset purchase agreement and in other related documents and agreements regarding aspects of our paclitaxel business, financial condition, structure, contracts, intellectual property, and other facts pertinent to the asset sale, including, among other things, representations relating to (subject to certain specified exceptions):
- •
- the corporate organization, existence, good standing, and qualification to do business of the Company and NaPro BioTherapeutics, Ltd., which is our United Kingdom subsidiary;
- •
- our authorization to enter into the asset sale;
- •
- the absence of conflicts with existing agreements resulting from the consummation or performance of the asset sale;
- •
- our financial statements;
- •
- our right, title, and interest in our patents and other intellectual property and the enforceability and non-infringement of such intellectual property that are part of the acquired assets;
- •
- our real property;
- •
- the completeness of our records regarding our paclitaxel operations and manufacturing;
- •
- the fitness and merchantability of our inventory;
- •
- our notes related to the paclitaxel business;
- •
- the absence of undisclosed liabilities;
- •
- the absence of powers of attorneys pertaining to the paclitaxel business or assets;
- •
- the disclosure of litigation involving the paclitaxel business or assets;
- •
- the absence of material liabilities under product warranties;
- •
- the absence of any guarantees pertaining to the paclitaxel business;
- •
- our ownership of the assets being purchased by Faulding;
- •
- our compliance with applicable laws and regulations;
- •
- the absence of any orders or proceedings related to the asset sale;
- •
- the absence of any material adverse change and certain other material changes in our paclitaxel business or the purchased assets since December 31, 2002;
- •
- our payment of applicable taxes;
- •
- the enforceability of our contracts and leases;
- •
- the existence and enforceability of our insurance policies;
- •
- the absence of product liability claims;
- •
- the existence of confidentiality agreements with our employees;
35
- •
- the existence of our employee benefit plans;
- •
- the existence and status of our environmental permits;
- •
- our solvency;
- •
- the use of brokers and financial advisers in connection with the asset sale;
- •
- the sufficiency of the acquired assets for the conduct of our paclitaxel business in the manner in which we conduct it; and
- •
- the absence of any untrue statement of material fact or omission that would make any of our representations or warranties contained in the asset purchase agreement misleading.
Our representations and warranties relating to intellectual property and environmental matters will survive for a period of 3 years following the closing of the asset sale. All of our other representations and warranties will survive for a period of 18 months following the closing, except that our representations and warranties relating to our corporate organization, authorization of the transaction, the lack of violation of laws and other regulations applicable to the transaction, broker's fees, our title to the assets, and tax matters continue in full force and effect forever. In addition, if at any time prior to the end of these periods we receive notice from Faulding of a claim for indemnification, then that claim will survive, including any adverse consequences Faulding may suffer after the end of the survival period, until the claim is fully and finally resolved, despite the limitations outlined above.
Faulding has made a number of customary representations and warranties, subject in some cases to customary qualifications, to us in the asset purchase agreement regarding its corporate organization, its authority to enter into the asset sale, and other facts pertinent to the asset sale, including those relating to:
- •
- Faulding's corporate organization, existence and good standing;
- •
- Faulding's authority to enter into and perform its obligations under the asset purchase agreement;
- •
- the absence of conflicts with applicable laws and regulations and existing agreements resulting from the execution and delivery of the asset purchase agreement or the consummation of the asset sale;
- •
- the use of brokers and financial advisers in connection with the asset sale;
- •
- Faulding's ability to pay the purchase price;
- •
- Faulding's intention not to engage in a plant closing or mass layoff for purposes of the Worker Adjustment and Retraining Notification Act; and
- •
- the absence of any untrue statement of material fact or omission that would make any of Faulding's representations or warranties contained in the asset purchase agreement misleading.
All of Faulding's representations and warranties continue in full force and effect forever.
The representations and warranties contained in the asset purchase agreement are complicated and not easily summarized. You are urged to carefully read Sections 3 and 4 of the asset purchase agreement attached as Annex A to this Proxy Statement, entitled "Seller's Representation and Warranties" and "Buyer's Representations and Warranties," respectively.
36
Covenants and Agreements
Under the asset purchase agreement, we have agreed that, unless Faulding otherwise consents in writing, we will comply with certain restrictions relating to the operation of our paclitaxel business prior to the closing of the asset sale. These restrictions include provisions relating to the following matters:
- •
- operating our paclitaxel business in the ordinary course and preserving our business substantially intact;
- •
- obtaining consents from third parties and giving all required notices to, and making any filings with, governmental authorities as required;
- •
- permitting Faulding's representatives access to our business and personnel;
- •
- maintaining our leases and leased real property;
- •
- preparing and filing this Proxy Statement, soliciting proxies from our stockholders in favor of the adoption and approval of the asset purchase agreement, and holding the special meeting to which this Proxy Statement relates;
- •
- delivering to Faulding the fairness opinion of Wells Fargo Securities, LLC that is attached to this Proxy Statement as Annex B;
- •
- transferring assets of foreign subsidiaries that relate to the paclitaxel business to the Company;
- •
- assisting Faulding with certain European regulatory matters in connection with the asset sale; and
- •
- not amending certain agreements that are presently in place.
We have also agreed that we will not initiate or solicit any inquiries, or make any proposal or offer with respect to, (i) any merger, reorganization, share exchange, consolidation, business combination, recapitalization, liquidation, dissolution or similar transaction involving our Company or its affiliates, (ii) any purchase or sale of 33% or more of our consolidated assets; or (iii) any purchase or sale of, or tender or exchange offer for, our equity securities that, if consummated, would result in any person (or the stockholders of such person) beneficially owning more than 33% of the power to vote for the election of a majority of our directors. We have agreed that, with respect to such acquisition proposals, we will not have any discussion with, or provide any confidential information to, any person, we will not knowingly facilitate any effort or attempt to make or implement any such acquisition proposal, we will not approve or recommend, or propose publicly to approve or recommend, any such acquisition proposal, and we will not approve or recommend, or propose to approve or recommend, any letter of intent or agreement in principle relating to any such acquisition proposal. Notwithstanding these agreements, our board of directors may authorize discussions or negotiations about any acquisition proposal with any person, if our board of directors determines in the exercise of its fiduciary duties that such action is in the best interest of our stockholders. Following receipt of an acquisition proposal that is financially superior to the asset purchase agreement, as determined in good faith by our board of directors, our board of directors may withdraw, modify or not make its recommendation in favor of the asset purchase agreement, provided that our board of directors shall have concluded in good faith that such action is necessary in order for it to act in a manner that is consistent with its fiduciary obligations. We have agreed to notify Faulding if we receive any such acquisition proposal.
In addition, both we and Faulding have agreed to:
- •
- use our reasonable best efforts to consummate the asset sale;
- •
- give notice to the other of any material adverse development causing a breach of our or its own representations and warranties;
37
- •
- comply with the confidentiality provisions of the asset purchase agreement;
- •
- assist with the transfer of the computer system from us to Faulding; and
- •
- seek the prior approval of the other party prior to issuing any press release relating to the subject matter of the asset purchase agreement, except as required by law.
After the closing, both we and Faulding have agreed to:
- •
- take such further actions as may be reasonably requested by the other party to implement the transaction contemplated by the asset purchase agreement;
- •
- make available personnel and records in connection with certain litigation or other claims, at the cost of the other party, except that we will cooperate with Faulding with respect to the Mylan litigation at our own expense;
- •
- comply with the confidentiality provisions as set forth in the asset purchase agreement; and
- •
- negotiate in good faith the terms of a supply agreement to be entered into by us and Faulding pursuant to which we will buy from Faulding and Faulding will sell to us bulk active pharmaceutical ingredient paclitaxel.
After the closing, we have agreed that we will not take any action that is intended to have the effect of discouraging any lessor, licensor, customer, supplier or other business associate from maintaining the same business relationship with Faulding as we had prior to the closing.
We have also agreed not to compete in certain respects with Faulding. For five years after the closing, we have agreed not to manufacture, use, or sell bulk active pharmaceutical ingredient paclitaxel for use in formulation of paclitaxel formulated in polyethoxylated castor oil, including, without limitation, paclitaxel formulated in polyethoxylated castor oil and ethanol. For a period of three years after the closing, we have agreed not to manufacture or supply paclitaxel using the certain proprietary processes for commercial purposes.
Faulding has agreed to offer employment to substantially all of our employees who are actively employed in our paclitaxel business. However, nothing will limit Faulding's authority, at any time and for any reason, to terminate the employment of any person to whom Faulding has agreed to offer employment. We are responsible for any compensation and benefits payable to these employees prior to the closing; Faulding is responsible for any compensation and benefits payable to these employees after the closing.
We have granted Faulding a non-exclusive right to use our name to the extent necessary to manufacture, package, use, and sell the purchased inventory and finished goods existing as of the closing, for a period of two years or until the inventory is depleted, whichever is earlier.
We will continue to collect and have all rights to our accounts receivable that exist prior to the closing.
The covenants in the asset purchase agreement are complicated and not easily summarized. You are urged to carefully read Sections 5 and 6 of the asset purchase agreement attached as Annex A to this Proxy Statement, entitled "Pre-Closing Covenants" and "Post-Closing Covenants," respectively.
Conditions to Completion of the Asset Sale
Our obligations to complete the asset sale are subject to the satisfaction or waiver at or prior to the closing of each of the following conditions:
- •
- each of Faulding's representations and warranties in the asset purchase agreement must be accurate and true in all material respects as of the closing date;
38
- •
- Faulding shall have performed and complied with all of its covenants;
- •
- there shall not be any order or decree preventing consummation of the transactions contemplated by the asset purchase agreement;
- •
- all applicable waiting periods (and any extensions thereof) under the HSR Act shall have expired or otherwise been terminated;
- •
- we and Faulding shall have obtained all material authorizations, consents, and approvals of governments and governmental agencies as required;
- •
- the asset sale must have been approved by the requisite vote of our stockholders at the special meeting;
- •
- the fairness opinion of Wells Fargo Securities shall not have been withdrawn or modified in any material respect not acceptable to our board of directors;
- •
- we and Abbott shall have entered into the Abbott Termination Agreement;
- •
- we and Faulding shall have entered into the Faulding Termination Agreement;
- •
- either portions of the contract between us and ChromaDex, Inc. shall have been assigned to Faulding, or we, Faulding, and ChromaDex, Inc. shall have entered into an agreement or agreements mutually satisfactory to us and to Faulding regarding services to be provided by ChromaDex, Inc. to us and to Faulding after the closing; and
- •
- we and Faulding shall have entered into a Transition Services Agreement consistent with a term sheet agreed to between the parties.
Faulding's obligations to complete the asset sale are subject to the satisfaction or waiver at or prior to the closing of each of the following conditions:
- •
- each of our representations and warranties in the asset purchase agreement must be accurate and true in all material respects as of the closing date;
- •
- we shall have performed and complied with all of our covenants;
- •
- there shall not be any order or decree preventing consummation of the transactions contemplated by the asset purchase agreement;
- •
- all applicable waiting periods (and any extensions thereof) under the HSR Act shall have expired or otherwise been terminated;
- •
- we and Faulding shall have obtained all material authorizations, consent and approvals of governments and governmental agencies as required;
- •
- we shall have obtained all of the estoppel certificates and written consents to assignment of the leases, except where the failure to obtain such estoppel certificate or consent would not have a material adverse effect;
- •
- no damage or other change shall have occurred with respect to any leased real property that would materially impair the use or occupancy of the leased real property or the operation of the paclitaxel business;
- •
- we shall have paid all compensation due and owing to the employees to be offered employment by Faulding;
- •
- the asset sale must have been approved by the requisite vote of our stockholders at the special meeting;
39
- •
- either portions of the contract between us and ChromaDex, Inc. shall have been assigned to Faulding, or we, Faulding, and ChromaDex, Inc. shall have entered into an agreement or agreements mutually satisfactory to us and Faulding regarding services to be provided to each of us and Faulding after the closing; and
- •
- we and Faulding shall have entered into a Transition Services Agreement consistent with the term sheet.
Indemnification
We have agreed in the asset purchase agreement to indemnify Faulding for any adverse consequences arising from, as a result of, or in connection with:
- •
- any breach of a representation, warranty, or covenant we made in the asset purchase agreement (subject to the applicable survival periods, see the section of this Proxy Statement above entitled "—Representations and Warranties"); and
- •
- liabilities that are not assumed by Faulding.
Our indemnification obligation with respect to any breach of certain representations or warranties by us does not arise until the aggregate amount of adverse consequences to Faulding exceeds a certain minimum amount, and there is an aggregate ceiling on our obligation for such breaches of our representations and warranties. The representations and warranties subject to these limitations include all of our representations and warranties, except those with respect to our corporate organization, our authority to enter into the asset purchase agreement and to perform our obligations thereunder, the noncontravention of the asset purchase agreement and the asset sale with any applicable law or contract, broker's fees, and title to our assets of the paclitaxel business.
Faulding has agreed to indemnify us with respect to any breach of its representations, warranties, and covenants, as well as with respect to any third party claim regarding the Mylan litigation described above. Faulding's indemnification obligation with respect to any breach of representation or warranty contained in Section 4 of the asset purchase agreement does not arise until the aggregate amount of adverse consequences to us exceeds a certain minimum amount, but there is no aggregate ceiling on Faulding's obligations for such breaches of its representations and warranties.
Termination
The asset purchase agreement may be terminated in accordance with its terms at any time prior to the closing of the asset sale, whether before or after the approval of the asset sale by our stockholders at the special meeting:
- •
- by mutual written consent of us and Faulding;
- •
- by either us or Faulding if our stockholders do not approve the asset sale by the requisite vote at the special meeting;
- •
- by Faulding, if we have breached any material representation, warranty, or covenant in the asset purchase agreement in any material respect, Faulding has notified us of the breach, and the breach has continued without cure for a period of 30 days after the notice of breach;
- •
- by Faulding, if the closing shall not have occurred on or prior to December 23, 2003, by reason of the failure of any of the conditions to Faulding's obligation to close (see the section of this Proxy Statement above entitled "—Conditions to Completion of the Asset Sale")(unless the failure results primarily from Faulding's breach of any representation, warranty, or covenant contained in the asset purchase agreement);
40
- •
- by us, if Faulding has breached any material representation, warranty, or covenant in the asset purchase agreement in any material respect, we have notified Faulding of the breach, and the breach has continued without cure for a period of 30 days after the notice of breach;
- •
- by us, if the closing shall not have occurred on or prior to December 23, 2003, by reason of the failure of any of the conditions to our obligation to close (see the section of this Proxy Statement above entitled "—Conditions to Completion of the Asset Sale")(unless the failure results primarily from our breach of any representation, warranty, or covenant contained in the asset purchase agreement);
- •
- by Faulding, (i) if our board of directors shall have withdrawn or modified or amended, in a manner adverse in any material respect to Faulding, its approval of the asset purchase agreement or its recommendation that our stockholders approve the asset sale; (ii) if our board of directors shall have approved, recommended or endorsed any acquisition proposal other than the asset sale contemplated by the asset purchase agreement; (iii) if we fail to call the special meeting within a reasonable time after completion of the Securities and Exchange Commission review process or shall have failed within a reasonable time thereafter to mail this Proxy Statement to our stockholders; or (iv) if we fail to include in this Proxy Statement the recommendation of our board of directors that our stockholders approve the asset sale; and
- •
- by us, if (i) our board of directors determines in good faith that an acquisition proposal is financially superior to the asset sale contemplated by the asset purchase agreement and is reasonably capable of being financed, (ii) we have notified Faulding as required by the asset purchase agreement and have terminated the asset purchase agreement, and (iii) concurrently with such termination, we enter into a definitive agreement to effect such acquisition proposal.
If either we or Faulding terminate the asset purchase agreement pursuant to either of the two preceding paragraphs, we have agreed to pay Faulding the sum of $2 million as liquidated damages within ten business days after the earlier of the termination of the asset purchase agreement or the execution of an agreement with respect to an acquisition proposal. We and Faulding have acknowledged that these damages are liquidated damages and not a penalty, and that the liquidated damages are necessary because actual damages arising from the loss of opportunity would not be determinable with any degree of certainty.
Expenses
Except with respect to termination fees payable under certain circumstances in the section above entitled "—Termination," we and Faulding have agreed that we will each pay our own expenses and costs incurred in connection with the asset sale, except that we have agreed to pay certain fees, such as transfer and documentary fees and charges, incurred in connection with the consummation of the asset sale.
41
Other Agreements Related to the Asset Sale
In connection with the asset sale, we have entered or will enter into certain other agreements with Faulding or Abbott. These agreements include:
Intellectual Property Agreement
Pursuant to the asset purchase agreement, we will transfer certain intellectual property rights to Faulding relating to our paclitaxel business. At the closing of the asset sale, we will enter into an intellectual property agreement with Faulding pursuant to which:
- •
- Faulding will permit us to use these intellectual property rights to research, develop, make, use, and sell products other than those which contain, or which are incorporated into or are used in the manufacture of other products which contain, polyethoxylated castor oil;
- •
- we will permit Faulding to use our intellectual property rights relating to bulk paclitaxel and/or polyethoxylated castor oil which currently exist or which we develop in the future; and
- •
- if we request, Faulding will seek to obtain the consent of Bryn Mawr College to sublicense certain technology to us under a license agreement between us and Bryn Mawr.
At the closing of the asset sale, we will enter into a transition services agreement with Faulding. Pursuant to the transition services agreement, each party will provide certain services to the other to assist in the transition of our paclitaxel business to Faulding and the closing of our former manufacturing facility in Boulder, Colorado, known as the "Gunbarrel Facility." Specifically:
- •
- we will provide litigation support, finance and accounting assistance, technical support, medical information assistance, intellectual property assistance, regulatory services, assistance in assigning certain contracts to Faulding, payroll support and technology support;
- •
- Faulding will help us coordinate contractors hired to restore the Gunbarrel Facility to warehouse space, and provide general information and assistance;
- •
- these services will be provided for an hourly fee based on 150% of the normal compensation level of the employee providing such transition services, plus direct expenses;
- •
- our litigation support to Faulding related to the Mylan litigation will be provided without charge;
- •
- the agreement will expire six months after the closing of the asset sale, subject to an extension for an additional six months upon mutual agreement of the parties;
- •
- Faulding may terminate the agreement three months after the closing upon 10 days notice; and
- •
- all litigation support services relating to the Mylan litigation will continue until resolution of the litigation.
Faulding Termination Agreement
Faulding and Abbott have entered into a separate purchase agreement pursuant to which Abbott will sell to Faulding, among other things, certain assets and rights, and Faulding will assume from Abbott certain liabilities, associated with a development, license and supply agreement between the Company and Abbott. In connection with the asset sale and Faulding's and Abbott's separate purchase
42
agreement, we entered into a termination, mutual release and settlement agreement dated as of August 25, 2003 with Faulding pursuant to which the parties agreed that:
- •
- certain existing agreements between Faulding and the Company or Abbott and the Company will terminate as of the later of the closing of the asset sale or the Faulding/Abbott sale, including:
- •
- the second amended and restated master agreement (relating to the exclusive sales of our paclitaxel to Faulding in most of Asia (excluding Japan and Russia), central and South America and parts of Africa);
- •
- a European agreement between us and Central Laboratories, Limited, a Faulding affiliate (relating to our license to Faulding of exclusive rights to sell and distribute our paclitaxel in Europe for a license fee);
- •
- the development, license and supply agreement between Abbott and us (relating to our 20-year exclusive collaborative agreement with Abbott to develop and commercialize one or more formulations of paclitaxel covering United States and Canada); and
- •
- a loan and security agreement between Abbott and us (relating to Abbott's secured loan of $20 million to the Company).
- •
- we will pay Faulding:
- •
- $20 million principal amount plus accrued interest for amounts owed under the loan agreement with Abbott; and
- •
- $1,668,898 to reimburse Faulding for assuming Abbott's expenses related to the qualification of Abbott's formulation facility;
- •
- Faulding will pay us for outstanding trade receivables for bulk active pharmaceutical ingredient under the second amended and restated master agreement and European agreement, which we estimate will be fully paid prior to closing;
- •
- we would settle final deliveries and cash payments with Faulding to fulfill remaining obligations under the second amended and restated master agreement, including, at our option, delivering to Faulding bulk paclitaxel or cash as of the later of the closing of the asset sale or the Faulding/Abbott sale. In addition to the foregoing, Faulding will pay us for certain bulk paclitaxel as of the later of the closing of the asset sale or the Faulding/Abbott sale;
- •
- the parties would release each other from claims relating to the agreements terminated; and
- •
- the termination agreement will terminate if the asset sale and the Faulding/Abbott sale are not closed by December 31, 2003.
In addition to the Faulding termination agreement, we entered into a mutual release and settlement agreement with Abbott dated August 25, 2003 pursuant to which the parties agreed that:
- •
- certain existing agreements between Faulding and the Company or Abbott and the Company will terminate or will be assigned to Faulding as of the later of the closing of the asset sale or the Faulding/Abbott sale, including:
- •
- the termination of a stock purchase agreement between Abbott and us (related to Abbott's purchase of our common stock, Abbott's registration rights and obligations to purchase additional shares);
- •
- the assignment by Abbott of the development, license and supply agreement between Abbott and us to Faulding; and
43
- •
- the assignment by Abbott of the loan and security agreement between Abbott and us to Faulding.
- •
- Abbott will pay us for all outstanding invoices for bulk paclitaxel that is in Abbott's possession, which we estimate will be $1.2 million at closing. These accounts receivable may decrease prior to closing;
- •
- the parties would release each other from claims relating to the agreements terminated; and
- •
- the termination agreement will terminate if the asset sale and the Faulding/Abbott sale are not closed by December 31, 2003.
In connection with the asset purchase agreement, Leonard P. Shaykin, our Chairman and Chief Executive Officer, and Dr. Sterling K. Ainsworth, our Vice Chairman, who together hold an aggregate of 1,535,828 outstanding shares of our common stock (approximately 5.1% of the outstanding shares entitled to vote at the special meeting on the record date), have entered into voting agreements with Faulding pursuant to which they have agreed to vote their shares of our common stock in favor of the asset sale. Each such stockholder also agreed not to (i) enter into any other voting agreement or arrangement with respect to the voting of his shares or (ii) transfer his shares except to certain permitted transfers until the termination of the asset purchase agreement. The voting agreements with Messrs. Shaykin and Ainsworth are attached as Annex E and Annex F, respectively.
Interests of Our Directors and Executive Officers in the Asset Sale
In considering the recommendation of our board of directors to vote for the proposal to adopt a resolution to approve the asset sale, you should be aware that certain of our directors and executive officers have personal interests in the asset sale that are, or may be, different from, or in addition to, those of our stockholders generally. The Company's board of directors was aware of these interests and approved the asset sale. These interests are as follows:
Senior Executive Employment Agreements
The Company does not intend to take any action that would result in any change of control payments being made under the following employment agreements.
Effective October 1, 2001, the Company entered into employment agreements with Leonard Shaykin, Dr. Sterling Ainsworth, and Dr. Patricia A. Pilia, the Company's Executive Vice President, Secretary and a Director. Each of these employment agreements provides for an initial three-year employment term. These employment agreements provide for initial annual base salaries for Mr. Shaykin and Drs. Ainsworth and Pilia of$270,000,$260,000 and$210,000 respectively. Under the terms of these employment agreements, in the event a change of control occurs or is anticipated (including the sale of substantially all of the assets of the Company, as those terms are used in the employment agreements) and an executive's employment is terminated by the Company without cause (as defined in the applicable employment agreement) or by the executive for good reason (as defined in the applicable employment agreement), such executive is to be granted (i) a payment equal to the greater of 100% of their prior year's bonus or 75% of his or her base annual salary, (ii) a payment equal to a percentage of such executive's base annual salary (300% for Mr. Shaykin, 200% for Dr. Pilia, and 150% for Dr. Ainsworth) and (iii) a payment equal to accrued, unpaid salary and bonus through the date of termination. As defined in these agreements, "good reason" includes, along with other events, the board of directors' failure to grant, in each calendar year after a change of control occurs or is anticipated, a minimum annual bonus at least equal to the average of the three years' prior annual bonuses, if such failure is in anticipation of or following a change of control.
44
In addition, if the executive's employment is terminated by the Company without cause or by the executive for good reason, each executive would be entitled to receive, subject to certain limitations, (i) a lump sum of accrued, unpaid salary and bonus, if any, through the termination date, (ii) health and welfare benefits as in effect immediately prior to termination for a maximum of 18 months following termination, (iii) full vesting for all outstanding Company stock options owned by the executive that were granted prior to October 1, 2001, and (iv) a bonus payment in an amount determined by whether or not a change of control of the company (as defined in the employment agreements) is occurring or anticipated at the time of the termination of the executive's employment. If a change of control occurs, the executive is entitled to receive a payment equal to a percentage of the individual executive's base salary, according to the terms set forth above for each named individual. If a change of control does not occur, the executive is entitled to receive a payment equal to 100% of his or her base salary for the preceding 12 months. The foregoing benefits would be limited by the amount deductible for income tax purposes under the Internal Revenue Code of 1986, as amended.
Following the board of directors' approval of the proposed sale of the paclitaxel business, each of the executives advised the Company that he or she has waived any requirement that a minimum annual bonus be paid him or her insofar as the sale of the paclitaxel business could be construed to constitute a change of control pursuant to the agreements.
Other Employment Agreements
The Company does not intend to take any action that would result in any change of control payments being made under the following employment agreements.
Effective October 1, 2001, we entered into employment agreements with Gordon H. Link, Jr., currently the Company's Senior Vice President and Chief Financial Officer and Kai Larson, Vice President and General Counsel, two of our executive officers. Each of these employment agreements provides for an initial two-year employment term. The employment agreements provide for initial annual base salaries for Mr. Link and Mr. Larson of$210,000 and$180,000, respectively. Under the employment agreements, in the event a change of control occurs or is anticipated (including the sale of substantially all of the assets of the Company, as those terms are used in the employment agreements) and an executive's employment is terminated by the Company without cause or by the executive for good reason, such executive is to be granted (i) a payment equal to the greater of 100% of his prior year's bonus or 75% of his base annual salary and (ii) a payment equal to 200% of his base annual salary. As defined in these agreements, "good reason" includes, along with other events, the board of directors' failure to grant, in each year after a change of control occurs or is anticipated, a minimum annual bonus at least equal to the average of the three years' prior annual bonuses, if such failure is in anticipation of or following a change of control.
In addition, if the executive's employment is terminated by the Company without cause or by the executive for good reason, each executive would be entitled to receive, subject to certain limitations, (i) a lump sum of accrued, unpaid salary and bonus, if any, through the termination date, (ii) health and welfare benefits as in effect immediately prior to termination for a maximum of 18 months following termination, (iii) full vesting for all outstanding Company stock options owned by the executive that were granted prior to October 1, 2001, and (iv) a bonus payment in an amount determined by whether or not a change of control of the company (as defined in the employment agreements)is occurring or anticipated at the time of the termination of the executive's employment. If a change of control occurs, the executive is entitled to receive a payment equal to a percentage of the individual executive's base salary, according to the terms set forth above for each named individual. If a change of control does not occur, the executive is entitled to receive a payment equal to 100% of his or her base salary for the preceding 12 months. The foregoing benefits would be limited by the amount deductible for income tax purposes under the Internal Revenue Code of 1986, as amended.
45
Following the board of directors' approval of the proposed sale of the paclitaxel business, each of the executives advised the Company that he has waived any requirement that a minimum annual bonus be paid him insofar as the sale of the paclitaxel business could be construed to constitute a change of control pursuant to the agreements.
Grant of Stock Options
On August 22, 2003, the compensation committee of the board of directors approved a grant of stock options to senior management of the Company to be made on the fifth business day after the announcement of a transaction reached with Faulding to sell the paclitaxel business. These grants were made on September 2, 2003 to Leonard P. Shaykin (250,000 shares); Kai P. Larson, Gordon H. Link and Dr. Patricia A. Pilia (110,000 shares each); Brian J. McCudden, the Company's Vice President, Operations (80,000 shares); Martin Batt, the Company's Vice President–Chief Information Officer and E. Hope Liebke, the Vice President and General Counsel of the Company's Genomics Division (60,000 shares each); Dr. Sterling Ainsworth (50,000 shares); Jeffrey White, the President of the Company's Genomics Division (60,000 shares) and an aggregate of 360,000 shares to non-executive officers of the Company. The options have an exercise price of $1.55 per share and expire on September 2, 2013. The options fully vest and become exercisable on September 2, 2008. The options may vest earlier if our stock price, on a rolling 20 day average, exceeds $1.55. If the 20-day rolling average stock price exceeds $1.55 by 30%, then 16.67% of the options vest. Likewise, if the 20-day rolling average stock price exceeds $1.55 by 60%, 90%, 120%, 150% and 200%, then in each case an additional 16.67% of the options vest. The compensation committee determined to make the grants upon the request of management in light of the facts that no options had been granted to senior management for over two years, with the exception of sign-on or promotion grants, and that the proposed sale of the paclitaxel business to Faulding warranted special recognition.
TL Ventures Funds
TL Ventures Funds, one of our principal institutional investors, has advised us that it believes that the completion of the asset sale will entitle it to have its $8 million of Company subordinated debentures redeemed. The Company disputed this position and engaged in discussions with TL Ventures Funds regarding this matter. On September 8, 2003, TL Ventures Funds reasserted its position and informed us that, if we could not resolve this issue promptly, it intends to pursue legal remedies. We will vigorously defend our legal position. One of our directors, Marc Ostro, is also a partner in TL Ventures Funds.
Employment Agreement
Brian McCudden, currently the Company's Vice President, Operations, will enter into an arrangement with Faulding to become an employee of Faulding upon consummation of the asset sale. Under the terms of the employment agreement with Faulding, Mr. McCudden will become Vice President of Operations for Faulding and will be paid a salary in the first year of $195,000, a sign on bonus of $40,000 and a retention bonus of $60,000 if he remains employed by Faulding for 12 months. Mr. McCudden's employment arrangement will not take effect unless and until the Company completes the asset sale.
In accordance with the Delaware General Corporation Law, holders of our common stock will not have appraisal or dissenters' rights in connection with the sale of our paclitaxel business.
46
Material U.S. Federal Income Tax Consequences to the Company
The asset sale is a taxable event to us. The Company will recognize gain or loss in an amount equal to the cash received, plus liabilities assumed in exchange for the assets and the amount paid to Abbott at closing to retire all outstanding debt and payables we owe to Abbott, less the Company's adjusted tax basis in the purchased assets. The Company's gain will be offset to the extent of current year losses from operations plus available net operating loss carryforwards and general business carryforwards, subject to applicable limitations under the ownership changes rules under Internal Revenue Code section 382 and the Alternative Minimum Tax rules. Under Section 382, where an ownership change occurs, the annual utilization of the net operating loss (NOL) carryforwards maybe restricted. Additionally, to the extent any gain on the sale of assets exceeds the current year loss from operations, an alternative minimum tax may be due on the excess. Any tax liabilities generated as a result of the asset sale are not expected to exceed $1 million.
Accounting Treatment of the Asset Sale
If the asset sale is approved by the Company's stockholders as described herein, the Company will record the asset sale in accordance with accounting principles generally accepted in the United States. Upon the completion of the asset sale, the Company will recognize a financial reporting gain, if any, equal to the net proceeds (the sum of the purchase price received less the estimated $1,258,000 in expenses relating to the asset sale) less the net book value of the assets purchased and the fair value of the indemnification liability retained.
Unaudited Pro Forma Financial Statements
The following unaudited pro forma condensed consolidated financial statements are presented to illustrate the effects of the sale of our paclitaxel business to Faulding on our historical financial position and operating results. The following unaudited pro forma condensed consolidated balance sheet of the Company at July 2, 2003 gives effect to the asset sale as if it had occurred on that date. The unaudited pro forma condensed consolidated statements of operations for the six months ended July 2, 2003 and for the year ended December 31, 2002 give effect to the asset sale as if it had occurred as of the beginning of the earliest period presented.
The unaudited pro forma condensed consolidated financial statements have been derived from, and should be read in conjunction with, our historical consolidated financial statements, including the accompanying notes. Those financial statements are included in our Quarterly Report on Form 10-Q for the three months ended July 2, 2003 and our Annual Report on Form 10-K for the year ended December 31, 2002, as amended, included with this Proxy Statement as Annexes C and D, respectively.
The unaudited pro forma condensed consolidated financial statements are presented only for informational purposes. As a result, the accompanying unaudited pro forma condensed consolidated financial statements are not necessarily indicative of our financial position or results of operations that would have occurred had the asset sale been consummated as of the dates indicated.
47
NaPro BioTherapeutics, Inc.
Unaudited Pro Forma Condensed Balance Sheet
As of July 2, 2003
(In thousands)
| | As Reported | Adjustments | Notes | Pro Forma Balance Sheet | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ASSETS | |||||||||||||
| Current assets: | |||||||||||||
| Cash and cash equivalents | $ | 5,836 | $ | 53,600 | (1 | ) | $ | 59,436 | |||||
| Accounts receivable, net of allowance for doubtful accounts | 8,097 | (8,097 | ) | (2 | ) | — | |||||||
| Inventory | 6,871 | (6,871 | ) | (3 | ) | — | |||||||
| Prepaid expense and other current assets | 1,530 | (140 | ) | (4 | ) | 1,390 | |||||||
| Total current assets | 22,334 | 38,492 | 60,826 | ||||||||||
Property, plant and equipment, net | 11,154 | (9,451 | ) | (5 | ) | 1,703 | |||||||
| Inventory—raw materials | 6,106 | (6,106 | ) | (3 | ) | — | |||||||
| Other assets | 3,097 | (32 | ) | (6 | ) | 3,065 | |||||||
| Total assets | $ | 42,691 | $ | 22,903 | $ | 65,594 | |||||||
LIABILITIES AND STOCKHOLDERS' EQUITY | |||||||||||||
| Current liabilities: | |||||||||||||
| Accounts payable and accrued liabilities | $ | 6,653 | $ | (4,528 | ) | (7 | ) | $ | 2,125 | ||||
| Accrued payroll and payroll taxes | 1,265 | (248 | ) | (8 | ) | 1,017 | |||||||
| Notes payable—current portion | 20,188 | (19,923 | ) | (9 | ) | 265 | |||||||
| Deferred income | 1,250 | (1,120 | ) | (10 | ) | 130 | |||||||
| Total current liabilities | 29,356 | (25,819 | ) | 3,537 | |||||||||
Notes payable—long term | 81 | — | 81 | ||||||||||
| Deferred income—long term | 5,327 | (5,327 | ) | (10 | ) | — | |||||||
| Convertible debentures | 5,418 | — | 5,418 | ||||||||||
Stockholders' equity: | |||||||||||||
| Preferred stock, $.001 par value; 2,000,000 shares authorized; none issued | — | — | — | ||||||||||
| Common stock, $.0075 par value; 64,000,000 shares authorized; 30,762,631 shares issued at July 2, 2003 | 231 | — | 231 | ||||||||||
| Additional paid-in capital | 111,017 | — | 111,017 | ||||||||||
| Accumulated deficit | (108,558 | ) | 54,049 | (11 | ) | (54,509 | ) | ||||||
| Treasury stock, 54,306 shares at cost at July 2, 2003 | (181 | ) | — | (181 | ) | ||||||||
| Total stockholders' equity | 2,509 | 54,049 | 56,558 | ||||||||||
| Total liabilities and stockholders' equity | $ | 42,691 | $ | 22,903 | $ | 65,594 | |||||||
48
NaPro BioTherapeutics, Inc.
Unaudited Pro Forma Condensed Statement of Operations
For the Six Months Ended July 2, 2003
(In thousands, except per share data)
| | As Reported | Adjustments | Notes | Pro Forma | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Product sales | $ | 14,449 | $ | (14,446 | ) | (12 | ) | $ | 3 | ||||
Expenses: | |||||||||||||
| Cost of sales | 7,441 | (7,107 | ) | (13 | ) | 334 | |||||||
| Research and development | 5,490 | (704 | ) | (14 | ) | 4,786 | |||||||
| General and administrative | 5,819 | (665 | ) | (15 | ) | 5,154 | |||||||
| 18,750 | (8,476 | ) | 10,274 | ||||||||||
| Operating (loss) income | (4,301 | ) | (5,970 | ) | (10,271 | ) | |||||||
Other income (expense): | |||||||||||||
| Gain on sale of paclitaxel business | — | 55,535 | (16 | ) | 55,535 | ||||||||
| License fee income | 560 | (560 | ) | (17 | ) | — | |||||||
| Interest income | 47 | 353 | (18 | ) | 400 | ||||||||
| Interest expense | (1,186 | ) | 733 | (19 | ) | (453 | ) | ||||||
| (Loss) income before income tax | (4,880 | ) | 50,091 | 45,211 | |||||||||
| Provision for income tax | — | 764 | (20 | ) | 764 | ||||||||
| Net (loss) income | $ | (4,880 | ) | $ | 49,327 | $ | 44,447 | ||||||
Basic (loss) income per share | $ | (0.16 | ) | $ | 1.47 | ||||||||
| Diluted (loss) income per share | $ | (0.16 | ) | $ | 1.47 | ||||||||
Basic and weighted average shares outstanding | 30,221 | (21 | ) | 30,221 | |||||||||
| Diluted weighted average shares outstanding | 30,221 | (21 | ) | 30,236 | |||||||||
49
NaPro BioTherapeutics, Inc.
Unaudited Pro Forma Condensed Statement of Operations
For the Twelve Months Ended December 31, 2002
(In thousands, except per share data)
| | As Reported | Adjustments | Notes | Pro Forma | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Product sales | $ | 34,193 | $ | (34,193 | ) | (12 | ) | $ | — | ||||
Expenses: | |||||||||||||
| Cost of sales | 23,976 | (23,976 | ) | (13 | ) | — | |||||||
| Research and development | 15,879 | (5,687 | ) | (14 | ) | 10,192 | |||||||
| General and administrative | 10,109 | (590 | ) | (15 | ) | 9,519 | |||||||
| 49,964 | (30,253 | ) | 19,711 | ||||||||||
| Operating (loss) income | (15,771 | ) | (3,940 | ) | (19,711 | ) | |||||||
Other income (expense): | |||||||||||||
| Gain on sale of paclitaxel business | — | 54,455 | (16 | ) | 54,455 | ||||||||
| License fee income | 8,902 | (8,902 | ) | (17 | ) | — | |||||||
| Interest income | 267 | 914 | (18 | ) | 1,181 | ||||||||
| Interest expense | (2,063 | ) | 1,340 | (19 | ) | (723 | ) | ||||||
| (Loss) income before income tax | (8,665 | ) | 43,867 | 35,202 | |||||||||
| Provision for income tax | — | 546 | (20 | ) | 546 | ||||||||
| Net (loss) income | $ | (8,665 | ) | $ | 43,321 | $ | 34,656 | ||||||
Basic (loss) income per share | $ | (0.29 | ) | $ | 1.17 | ||||||||
| Diluted (loss) income per share | $ | (0.29 | ) | $ | 1.11 | ||||||||
Basic weighted average shares outstanding | 29,606 | (21 | ) | 29,606 | |||||||||
| Diluted weighted average shares outstanding | 29,606 | (21 | ) | 31,268 | |||||||||
Notes:
The adjustments represent the sale of the Company's paclitaxel business and the elimination of the assets, liabilities and operations related to the paclitaxel business. The balance sheet assumes the transaction occured on July 2, 2003; the income statements assume the transaction occurred on January 1, 2003 and January 1, 2002.
- (1)
- Cash proceeds primarily from the sale of the paclitaxel business and the collection of accounts receivable, less the repayment of debt associated with paclitaxel operations (see Notes 7, 8 and 9).
- (2)
- Collection of accounts receivable related to the paclitaxel business.
- (3)
- Sale of finished goods, work in process and raw materials used in the manufacture of paclitaxel. The Company has the right to retain certain finished goods inventory, however, these statements reflect no retainage. Such a retainage could result in a negative inventory adjustment to the sales price which would reduce the proceeds received from the sale.
- (4)
- Sale of prepaid expense and other current assets that directly relate to the paclitaxel business.
- (5)
- Sale of property, plant and equipment used in the manufacturing of paclitaxel and equipment and leasehold improvements to be abandoned or disposed of.
50
- (6)
- Deposits for facilities sold with the paclitaxel business.
- (7)
- Elimination of obligations specifically identified with the paclitaxel business.
- (8)
- Accrued wages, fringe benefits and payroll taxes for personnel associated with paclitaxel manufacturing operations.
- (9)
- Note due to Abbott Laboratories, net of unamortized issuance costs, retired with the proceeds from the sale of the paclitaxel business.
- (10)
- Deferred income from license fees that is recognized with the sale of the paclitaxel business.
- (11)
- Net income derived primarily from the gain on the sale of the paclitaxel business.
- (12)
- Sales of paclitaxel.
- (13)
- Cost of paclitaxel sold and acceleration of vesting on restricted stock issued as a retention bonus to certain employees in the paclitaxel business.
- (14)
- Research and development expenses allocated to the paclitaxel business primarily based upon labor costs charged to paclitaxel projects.
- (15)
- General and administrative expenses allocated to the paclitaxel business consists of wages, fringe benefits and payroll taxes of personnel that will transfer with the sale of the paclitaxel business, and for the 2003 period includes accelerated depreciation of leasehold improvements to be abandoned or disposed of.
- (16)
- Gain on sale of the paclitaxel business represents the gain on the sale of the business (the net proceeds, or the sum of the purchase price received less the estimated $1,258,000 in expenses relating to the asset sale, less the net book value of the assets purchased and the fair value of the indemnification liability retained), the recognition of all deferred income related to paclitaxel, reduced by the loss on paclitaxel related assets sold or abandoned and the loss associated with unamortized debt issuance costs related to the retired Abbott note.
- (17)
- License fee income consists of deferred income recognized with the sale of the paclitaxel business and for the period ended December 31, 2002 also an elimination of an $8.0 million milestone payment.
- (18)
- Interest income earned on additional cash received from the sale.
- (19)
- Interest expense accrued and/or paid on the note due to Abbott.
- (20)
- Tax expense is due to the estimated alternative minimum tax.
- (21)
- In connection with the sale of the paclitaxel business, our Board of Directors granted a total of 1,250,000 stock options to officers of the Company under the 1994 Stock Option Plan at an exercise price of $1.55. These options vest no later than September 2, 2008. Vesting would be accelerated if the 20-day average closing price of our common stock exceeds $2.02 and vesting would be fully accelerated if the 20-day average closing price exceeds $4.65. The Company does not record expense for options granted in accordance with Accounting Principles Board Opinion No. 25. The additional total compensation expense that would be related to the sale transaction if the options had been expensed under the fair value method would have been $1,700,000 over the next five years.
Regulatory Approvals Required to Complete the Asset Sale
The asset sale is subject to review by the United States Federal Trade Commission or the Antitrust Division of the United States Department of Justice under the HSR Act. Under the HSR Act, the Company and Faulding are required to make pre-acquisition notification filings and to await the
51
expiration or early termination of the statutory waiting period prior to completing the acquisition. On September 11, 2003, the parties filed the required information with the Antitrust Division of the U.S. Department of Justice and the U.S. Federal Trade Commission. The statutory waiting period under the HSR Act will expire on October 14, 2003.
Despite expiration of the statutory waiting period, and even after completion of the acquisition, either the Antitrust Division of the U.S. Department of Justice or the U.S. Federal Trade Commission could challenge, seek to block or block the acquisition under the antitrust laws or request additional information, as it deems necessary or desirable in the public interest. In addition, in some jurisdictions, a competitor, customer or other third party could initiate a private action under the antitrust laws challenging or seeking to enjoin the acquisition, before or after it is completed. We cannot be sure that a challenge to the acquisition will not be made or that, if a challenge is made, that the Company and Faulding will prevail.
Other than applicable antitrust laws, neither we nor Faulding are aware of any other regulatory requirements or governmental approvals or actions that may be required to consummate the acquisition, except for compliance with the applicable regulations of the Securities and Exchange Commission in connection with this Proxy Statement and the Delaware General Corporation Law in connection with the asset sale. Should any such approval or action be required, it is presently contemplated that such approval or action would be sought. There can be no assurance, however, that any such approval or action, if needed, could be obtained and would not be conditioned in a manner that would cause the parties to abandon the acquisition.
Use of Proceeds; Operations after Asset Sale; Risks
If the asset sale is approved by our stockholders and the other conditions to closing of the asset sale are satisfied or waived, we will sell our paclitaxel business to Faulding. Upon consummation of the asset sale, we will exit the generic paclitaxel business and our development agreements with Abbott and Faulding will terminate. At closing, approximately $21.7 million of the proceeds of the purchase price will be paid to Abbott or its designee to retire all outstanding debt and payables we owe to Abbott.
The assets to be sold to Faulding will include paclitaxel manufacturing assets, yew plantations, domestic and international issued and pending paclitaxel patents, a worldwide registration dossier, worldwide development and supply agreements and inventories. Our paclitaxel business operations generated $34.2 million in product sales and $10.2 million in gross profit in 2002. We will retain all our intellectual property not used in connection with the business being sold.
We currently plan to use the proceeds from the asset sale to fund the development and commercialization of products based on our proprietary gene editing and oncology platforms and for general corporate purposes. However, no final decision has been made as to which of these purposes the proceeds will be applied. As discussed under the caption "—Background of the Proposed Asset Sale," TL Ventures Funds has advised the Company that completion of the asset sale will entitle it to have its $8 million of Company subordinated debentures redeemed. On September 8, 2003, TL Ventures Funds informed us that it intends to pursue legal remedies if we cannot resolve the issue promptly. The Company disputes TL Ventures Funds' position and will vigorously defend its legal position. Were TL Ventures Funds to assert this claim and prevail with respect to it, the proceeds available to the Company from the asset sale would be reduced accordingly.
52
After the asset sale, we intend to focus our future efforts on the development and commercialization of therapeutic candidates, primarily in two distinct classes of therapeutics currently under development:
Oncology Platform
In the field of oncology, we are developing several targeted and non-targeted compounds that we believe have potential as anti-cancer agents to treat a variety of cancers including small cell lung, prostate, pancreatic colorectal cancers and squamous cell head and neck carcinomas. When referred to as "targeted" these compounds consist of conjugates of a cytotoxic agent coupled to a peptide, which selectively and specifically target certain types of tumor cells. Targeted delivery is designed to lower toxicity in normal tissue while increasing efficacy in tumor tissue by taking advantage of specific receptors on the tumor cells. We are testing these conjugates in various in-vitro and animal models, and are currently undertaking the development work necessary to introduce these agents into human clinical trials. It is our belief that coupling cytotoxic agents with targeting agents will lead to greater safety and efficacy when compared with the untargeted cytotoxic chemotherapy agents currently available on the market. These programs are being performed in collaboration with academic researchers under the direction of our drug development team.
Gene Editing
In the field of genomics, we are engaged in the discovery, patenting, development and commercialization of a range of products using our proprietary gene editing technology. Employing this technology, we are developing products that specifically and precisely edit genes. By making small, highly controlled modifications to genes, we believe we can contribute to the prevention and treatment of many different types of disease, both inherited and acquired.
In the area of therapeutics, our efforts to date have been directed primarily to treatments for Sickle Cell disease and Huntington's disease. We are currently developing an ex vivo therapy for Sickle Cell disease based upon our proprietary gene editing technology which employs proprietary oligonucleotides that are able, among other things, to rapidly locate and specifically modify a chosen single base pair in a targeted gene of choice. This therapy consists of treating hematopoietic progenitor cells, isolated from Sickle Cell patients which are then treated ex vivo, with a proprietary oligonucleotide that corrects the defective base pair in the hemoglobin gene. The resulting corrected progenitor cells, when re-introduced into the patient, would then begin producing normal, rather than sickle, hemoglobin. The therapy addresses the underlying genetic basis of the disease.
We are also developing an in vivo oligonucleotide therapy for Huntington's disease. Huntington's disease is a neurodegenerative disease that is specifically characterized by fatal aggregate formation and cell death in neural tissue. There is no current treatment to halt the progression of the disease, which leads to death after ten to twenty-five years of decline. We have patented proprietary oligonucleotides that may be effective in addressing the treatment for this disease. Both the Sickle Cell and the Huntington's program are in pre-clinical development and will require significant additional work before entering the clinic.
In addition to gene editing and oncology research and development activities, we are also evaluating the in-licensing or purchase of potential new products and/or technologies, whether or not those products or technologies are derived from natural products, are chemotherapeutic agents, or complement our gene editing technology. Our evaluation of new products and technologies may involve the examination of individual molecules, classes of compounds, or platform technologies, in both cancer and other therapeutic areas. Acquisitions of new products or technologies may involve the purchase of such products or technologies, the acquisition of, or merger with, other companies, or the incurrence of debt or equity financing that is dilutive to existing stockholders.
53
Risks Associated with Our Sale of the Paclitaxel Business
The proceeds from the asset sale would provide a portion of the resources we will need to fund the most successful of these therapies into the clinic. However, we cannot assure you that the proceeds from the asset sale, if realized, will be sufficient to fund our strategic plans to develop and commercialize additional products. The amount of proceeds available to the Company may also be reduced depending on the outcome of the Company's dispute with TL Ventures regarding redemption of their debentures. If the proceeds from the asset sale are not sufficient, we may be required to seek additional financing that could be dilutive to our existing stockholders. In addition, if we complete the asset sale, our ability to generate sales sufficient to support our operations would depend primarily on developing and commercializing gene editing, oncology and other new products and technologies. You should carefully consider these risks and the following additional risks related to our other potential products and technologies, in addition to the other information included in this Proxy Statement:
- •
- the sale of our paclitaxel business could adversely affect our ability to achieve profitability because the asset sale would transfer a significant portion of the Company's total assets and current operations;
- •
- our product candidates and technologies are in an early state of development and there is a high risk that they may never be commercialized because of the costs of continuing development or for other reasons;
- •
- we may be required to rely on strategic partners for the development, marketing and manufacturing of future products and technologies which may delay or impair our ability to generate significant revenue and may otherwise adversely affect our profitability;
- •
- our efforts in developing our gene editing technology may be unsuccessful and could adversely affect our financial position;
- •
- our efforts in researching and developing a broad range of new potential products and technologies may divert available capital or management's attention from other portions of our business and could adversely affect our financial position;
- •
- technological change may make our potential products and technologies less attractive or obsolete;
- •
- adverse events in the field of gene editing may negatively impact regulatory approval or public perception of our potential products and technologies resulting in decreased demand;
- •
- testing of our potential products and technologies relies heavily on the voluntary participation of patients in our clinical trials, which is not within our control, and could substantially delay or prevent us from completing development of such products and technologies;
- •
- our potential products and technologies must undergo rigorous clinical testing and regulatory approvals, which could substantially delay or prevent us from marketing any products;
- •
- our gene editing and oncology technologies are still new and developing; very little clinical data results exist and new information may arise which may cause us delays in designing our protocols, submitting applications that satisfy all necessary regulatory review requirements, and ultimately completing the clinical trials of our products;
- •
- we may not be successful in obtaining required foreign regulatory approvals, which would prevent us from marketing our products internationally;
- •
- we have no experience in manufacturing our potential therapeutic products, which raises uncertainty about our ability to manufacture our potential therapeutic products cost-effectively;
54
- •
- we have no experience in directly marketing or selling our potential therapeutic products and technologies, which raises uncertainty about our ability to commercialize our potential therapeutic products cost-effectively.
- •
- we may be required to obtain rights to proprietary technologies to further develop our business, which may not be available or may be costly; and
- •
- our use of hazardous materials exposes us to the risk of material environmental liabilities, and we may incur substantial additional costs to comply with environmental laws in connection with the operation of our research and manufacturing facilities.
Risks Associated with the Company's Current Operation of the Paclitaxel Business
We are currently operating our paclitaxel business until the consummation of the asset sale. If the asset sale is not approved by our stockholders, or if we are otherwise unable to complete the asset sale to Faulding, we would continue to operate this business for the foreseeable future. However, if we had to continue to operate our paclitaxel business, the Company would be exposed to various risks including:
- •
- the Company's ability to develop other products or product candidates, advance our other businesses or retire our debt to Abbott and TL Ventures may be delayed or impaired. Until we retire our debt currently owed to Abbott, the majority of our paclitaxel inventories and specific assets which relate to the manufacture of paclitaxel will remain pledged to Abbott as collateral;
- •
- political, economic or regulatory events in foreign countries may limit our ability to seek marketing authority or to generate revenue and may otherwise adversely affect our profitability;
- •
- our dependence on strategic partners for the development and marketing of paclitaxel may delay or impair our ability to generate significant revenue and may otherwise adversely affect our profitability;
- •
- if we or our strategic partners fail to obtain regulatory approval for the sale of paclitaxel in the primary world markets outside the United States, our ability to generate significant revenue and become profitable may be impaired;
- •
- if we or our strategic partners fail to maintain regulatory approval for the sale of paclitaxel in the United States or any other primary world market in which our paclitaxel may be approved for commercial sale, our business and financial condition could be materially harmed;
- •
- if we are unable to maintain access to a stable, long term supply of yew trees, yew biomass and crude paclitaxel extract for use in the production of paclitaxel, our business and financial condition would be materially harmed;
- •
- if we or our manufacturing contractors are unable to produce paclitaxel in sufficient quantities, on a timely basis and at an acceptable cost, we may be unable to meet demand for our paclitaxel and lose potential revenue;
- •
- increased competition which has resulted in recent declines in the price of paclitaxel and which declines, or continuing future price declines, could adversely affect our ability to generate significant revenue and our strategic partners' incentive to compete;
- •
- if we and our strategic partners fail to market our paclitaxel successfully against our competitors' paclitaxel, we may be unable to secure or maintain our market share;
- •
- new cost effective methods of harvesting or manufacturing paclitaxel or other technological developments could also result in our paclitaxel becoming less attractive or obsolete; and
55
- •
- if we are unable or fail to adequately protect our patents and trade secrets related to our paclitaxel, third parties may be able to use our technology, which could impair our ability to compete in the paclitaxel market.
These and other risks related to the Company's business are described in more detail under the caption "Risk Factors" or "Special Note Regarding Forward-Looking Statements" in the Company's filings with the Securities and Exchange Commission.
Vote Required and Board of Directors Recommendation
The approval of the asset sale to Faulding under the terms of the asset purchase agreement, requires the affirmative vote of a majority of the outstanding shares of our common stock. In connection with the asset purchase agreement, Leonard P. Shaykin, the Company's Chairman and Chief Executive Officer, and Dr. Sterling K. Ainsworth, the Company's Vice Chairman, who together hold an aggregate of 1,535,828 outstanding shares of our common stock (approximately 5.1% of the outstanding shares entitled to vote at the special meeting as of September 12, 2003), have entered into voting agreements pursuant to which they have agreed to vote their shares of the Company common stock in favor of the asset sale. In addition, Abbott holds 2 million shares of our common stock, or approximately 6.5% of our total outstanding common stock. Abbott has agreed to sell these shares to Faulding.
THE BOARD OF DIRECTORS OF THE COMPANY BELIEVES THAT THE PROPOSED ASSET SALE IS ADVISABLE AND IN THE BEST INTERESTS OF THE COMPANY AND OUR STOCKHOLDERS AND UNANIMOUSLY RECOMMENDS A VOTE "FOR" THE APPROVAL OF THE ASSET SALE.
56
SECURITY OWNERSHIP BY CERTAIN PERSONS
The following table sets forth certain information concerning the beneficial ownership of our common stock, as of September 12, 2003, by each person known by the Company to own beneficially more than 5% of the outstanding common stock, each individual serving as the Company's Chief Executive Officer in the last fiscal year, each of the other executive officers for whom disclosure is required, each director of the Company, and all directors and executive officers as a group. Except where otherwise indicated, the address for each of the persons listed in the table is: NaPro BioTherapeutics, Inc., 4840 Pearl East Circle, Suite 300W, Boulder, Colorado 80301.
| Name of Director, Officer or Owner(1) | Number of Beneficial Shares of Common Stock | Percent of Class | |||
|---|---|---|---|---|---|
| Leonard P. Shaykin | 1,286,322 | (2) | 4.09 | % | |
Sterling K. Ainsworth | 1,457,782 | (3) | 4.66 | % | |
Edward L. Erickson | 50,000 | (4) | * | ||
George M. Gould | 10,000 | (5) | * | ||
Arthur H. Hayes, Jr. | 90,000 | (6) | * | ||
Marc J. Ostro | 130,500 | (7) | * | ||
The Honorable Richard N. Perle | 90,000 | (8) | * | ||
Patricia A. Pilia | 629,294 | (9) | 2.02 | % | |
Robert E. Pollack | 65,500 | (10) | * | ||
Kai P. Larson | 127,287 | (11) | * | ||
Gordon H. Link, Jr. | 408,080 | (12) | 1.31 | % | |
Brian McCudden | 19,161 | (13) | * | ||
Hope Liebke | 55,922 | (14) | * | ||
Jeffrey White | 36,150 | (15) | * | ||
Martin Batt | 28,870 | (16) | * | ||
All Directors and Executive Officers as a Group (15 persons) | 4,484,868 | (17) | 13.50 | % | |
Abbott Laboratories 100 Abbott Park Road Abbott Park, IL 60064 | 2,000,000 | (18) | 6.50 | % |
- *
- Less than 1%
- (1)
- Unless otherwise noted, we believe that all persons named in the table have sole voting and investment power with respect to all shares of common stock beneficially owned by them. Percentage of beneficial ownership is based on 30,755,384 shares of common stock outstanding as of September 12, 2003, as adjusted as required by the rules promulgated by the Securities and Exchange Commission Beneficial ownership is determined in accordance with the rules of the Securities and Exchange Commission and generally includes voting or investment power with respect to securities. Shares of stock subject to options or warrants currently exercisable or exercisable within 60 days of September 12, 2003 are deemed outstanding for computing the
57
percentage of the person or entity holding such securities, but not outstanding for purposes of computing the percentage of any other person or entity.
- (2)
- Includes 663,768 shares of common stock issuable upon exercise of options granted to Mr. Shaykin under the 1994 Stock Option Plan (the "1994 Plan") and 37,937 shares of common stock beneficially owned through our 401(k) and Employee Stock Option Plan as of September 12, 2003.
- (3)
- Includes 544,509 shares of common stock issuable upon exercise of 1994 Plan options; 36,019 shares of common stock beneficially owned through the Company's 401(k) and ESOP plans as of September 12, 2003; and 42,550 shares of common stock gifted by Dr. Ainsworth to relatives and certain other persons, which Dr. Ainsworth may be deemed to beneficially own by virtue of holding powers of attorney to vote and take certain other actions with respect to such shares. Does not include shares held by Dr. Pilia, who is engaged to be married to Dr. Ainsworth, and Dr. Ainsworth disclaims beneficial ownership of the shares of common stock beneficially owned by Dr. Pilia as well as of the gifted shares over which Dr. Ainsworth holds powers of attorney. See note (9) below.
- (4)
- Includes 50,000 shares of common stock issuable upon exercise of options granted to Mr. Erickson under the 1994 Plan.
- (5)
- Includes 10,000 shares of common stock issuable upon exercise of options granted to Mr. Gould under the 1994 Plan.
- (6)
- Includes 90,000 shares of common stock issuable upon exercise of options granted to Dr. Hayes under the 1994 Plan.
- (7)
- Includes 90,500 shares of common stock issuable upon exercise of options granted to Dr. Ostro under the 1994 Plan. Does not include 1,470,195 shares of common stock (4.70%) or the outstanding shares as of September 12, 2003 beneficially owned by TL Ventures. Dr. Ostro is a managing director of an affiliate of the investment manager of the TL Venture Funds, and a voting limited partner in the TL Venture Funds, but he disclaims beneficial ownership of such shares.
- (8)
- Includes 80,000 shares of common stock issuable upon exercise of options granted to Mr. Perle under the 1994 Plan.
- (9)
- Includes 386,002 shares of common stock issuable upon exercise of 1994 Plan options; 37,786 shares of common stock beneficially owned through the Company's 401(k) and ESOP plans as of September 12, 2003; 2,000 shares owned in joint tenancy with a relative for which Dr. Pilia shares voting rights and 10,800 shares of common stock gifted by Dr. Pilia to relatives and certain other persons which Dr. Pilia may be deemed to beneficially own by virtue of holding powers of attorney to vote and take certain other actions with respect to such shares. Does not include shares held by Dr. Ainsworth, who is engaged to be married to Dr. Pilia. Dr. Pilia disclaims beneficial ownership of shares of common stock beneficially owned by Dr. Ainsworth and the gifted shares over which Dr. Pilia holds powers of attorney. See note (3) above.
- (10)
- Includes 65,500 shares of common stock issuable upon exercise of options granted to Dr. Pollack under the 1994 Plan.
- (11)
- Includes 94,166 shares of common stock issuable upon the exercise of options granted to Mr. Larson under the 1994 Plan and 33,121 shares beneficially owned through our 401(k) and ESOP plans as of September 12, 2003.
- (12)
- Includes 322,513 shares of common stock issuable upon the exercise of options granted to Mr. Link under the 1994 Plan and 38,035 shares of common stock beneficially owned through our 401(k) and ESOP plans as of September 12, 2003.
58
- (13)
- Includes 12,500 shares of common stock issuable upon exercise of options granted to Mr. McCudden under the 1994 Plan and 2,161 shares beneficially owned through our ESOP plan as of September 12, 2003.
- (14)
- Includes 25,000 shares of common stock issuable upon exercise of options granted to Ms. Liebke under the 1994 Plan and 922 shares beneficially owned through our ESOP plan as of September 12, 2003.
- (15)
- Includes 17,400 held in the name of Mr. White's spouse and 18,750 shares of common stock issuable upon exercise of options granted to Mr. White under the 1994 Plan.
- (16)
- Includes 18,750 shares of common stock issuable upon exercise of options granted to Mr. Batt under the 1994 Plan.
- (17)
- Includes an aggregate of 2,471,958 shares of common stock issuable upon exercise of outstanding stock options held by such persons.
- (18)
- Information in the table as to beneficial ownership of common stock by Abbott Laboratories is based upon Amendment No. 1 to Schedule 13D filed by Abbott Laboratories on July 22, 2003. Based on such Amendment No. 1 to Schedule 13D, Abbott disclosed that it has entered into a stock purchase agreement with Faulding dated July 16, 2003 pursuant to which Abbott agreed to sell Faulding the 2,000,000 shares of the Company's common stock Abbott holds. We understand that such sale will be consummated simultaneously with, and only upon, the closing of the Faulding/Abbott sale. For more information on the Faulding/Abbott sale, see "Other Agreements Related to the Asset Sale—Faulding Termination Agreement" above.
59
PROPOSAL 2
ADJOURNMENT OF THE SPECIAL MEETING
If the special meeting is convened and a quorum is present, but there are not sufficient votes to approve the asset sale at that time, or if we need additional time to satisfy all conditions to the asset sale, we may move to adjourn the special meeting to a later date to solicit additional proxies or to satisfy any unsatisfied conditions. In order to allow proxies that we have received by the time of the special meeting to be voted for an adjournment, if necessary, the Company has submitted the question of adjournment to its stockholders as a separate matter for their consideration. If it is necessary to adjourn the special meeting, no notice of the adjourned meeting is required to be given to stockholders, other than an announcement at the special meeting of the time and place to which the special meeting is adjourned, so long as the special meeting is adjourned for 30 days or less and no new record date is fixed for the adjourned meeting. At the adjourned meeting the Company may transact any business which might have been transacted at the original meeting.
OUR BOARD OF DIRECTORS UNANIMOUSLY RECOMMENDS THAT YOU VOTE "FOR" APPROVAL OF PROPOSAL 2.
CAUTIONARY NOTE REGARDING FORWARD-LOOKING STATEMENTS
This Proxy Statement and the documents incorporated by reference into this Proxy Statement contain forward-looking statements (within the meaning of Section 27A of the Securities Act of 1933, as amended, and Section 21E of the Securities Exchange Act of 1934, as amended) about the asset sale and the Company. These forward-looking statements represent management's beliefs and assumptions as of the date such statement was made, based on available information. Forward-looking statements can be identified by the use of words such as "believe," "intend," "estimate," "may," "will," "should," "anticipated," "expected," "trusts" or comparable terminology or by discussions of strategy. Although the Company believes that the expectations reflected in such forward-looking statements are reasonable, it cannot assure that these expectations will prove to be correct. Such statements involve risks and uncertainties as to which there can be no assurance, including, but not limited to the risk that the transaction announced with Faulding may not close because it is not approved by stockholders or otherwise or may be materially delayed, the costs of continuing development of any or all of the Company's development programs, the timing of when those programs will enter the clinic, the successful commercialization of the Company's platform technologies, the ability to obtain, maintain and enforce patents relating to the Company's technologies, the risk that developments in the Company's business will require new and unanticipated investment, the risk that the cash proceeds from the Faulding transaction, if realized, will not be sufficient to fund the Company's strategic plans for the development of new proprietary therapeutic candidates, the risk that the outcome of the Company's dispute with TL Ventures regarding redemption of their debentures will reduce the proceeds available to us from the Faulding transaction and other factors as described under the caption "Use of Proceeds; Operations after the Asset Sale; Risks" in this Proxy Statement, and under the caption "Risk Factors" or "Special Note Regarding Forward-Looking Statements" in the Company's filings with the Securities and Exchange Commission. These risks, uncertainties and other factors may cause the Company's actual results, performance or achievements to be materially different from any future results, performance or achievements expressed or implied by such forward-looking statements. The Company disclaims any intention or obligation to update publicly or revise such statements whether as a result of new information, future events or otherwise.
INCORPORATION OF CERTAIN DOCUMENTS BY REFERENCE
We file annual, quarterly and current reports, proxy statements and other information with the Securities and Exchange Commission. You may read and copy any document we file with the Securities and Exchange Commission at its facilities located at Judiciary Plaza, Room 1024, 450 Fifth Street,
60
N.W., Washington, D.C. 20549 or at the offices of the National Association of Securities Dealers, Inc. located at 1735 K Street, N.W., Washington, D.C. 20006. Please call the Securities and Exchange Commission at 1-800-SEC-0330 for further information on the SEC's public reference rooms. Our Securities and Exchange Commission filings also are available to the public at the its website athttp://www.sec.gov.
In conjunction with this Proxy Statement, we are sending you a copy of our annual report on Form 10-K for the year ended December 31, 2002 as amended by a Form 10-K/A on April 30, 2003 and a Form 10 K/A on August 8, 2003, attached as Annex C to this Proxy Statement, and our quarterly report on Form 10-Q for the quarter ended July 2, 2003, attached as Annex D to this Proxy Statement.
The following documents filed with the Securities and Exchange Commission are incorporated by reference in this Proxy Statement:
- 1.
- Our annual report on Form 10-K for the year ended December 31, 2002, as amended by a Form 10-K/A on April 30, 2003 and a Form 10 K/A on August 8, 2003;
- 2.
- Our quarterly reports on Form 10-Q for the quarters ended April 2, 2003 and July 2, 2003; and
- 3.
- Our current reports on Form 8-K dated January 23, 2003, February 12, 2003, March 26, 2003, April 9, 2003, May 15, 2003, August 7, 2003, August 26, 2003 and August 28, 2003.
All documents filed by the Company pursuant to Sections 13(a), 13(c), 14 or 15(d) of the Securities Exchange Act of 1934, as amended, subsequent to the date hereof and prior to the date of the special meeting or any adjournment or postponement thereof shall be deemed to be incorporated by reference herein and made a part hereof from the date of the filing of such documents. Any statement contained in a document incorporated or deemed to be incorporated by reference herein shall be deemed to be modified or superseded for purposes of this Proxy Statement to the extent that a statement contained herein or in any other document subsequently filed with the Securities and Exchange Commission which also is deemed to be incorporated by reference herein modified or supersedes such statement. Any such statement so modified or superseded shall not be deemed, except as so modified or superseded, to constitute a part of this Proxy Statement.
The Company will provide without charge to each person to whom a copy of this Proxy Statement is delivered, upon the written or oral request of such person, a copy of any or all of the documents incorporated by reference herein (not including the exhibits to such documents, unless such exhibits are specifically incorporated by reference in such documents). Requests for such copies should be directed to NaPro BioTherapeutics, Inc., 4840 Pearl East Circle, Suite 300W, Boulder, Colorado 80301, Attention: Kai Larson, Vice President and General Counsel.
THIS PROXY STATEMENT DOES NOT CONSTITUTE THE SOLICITATION OF A PROXY IN ANY JURISDICTION TO OR FROM ANY PERSON TO WHOM OR FROM WHOM IT IS UNLAWFUL TO MAKE SUCH PROXY SOLICITATION IN SUCH JURISDICTION. YOU SHOULD RELY ONLY ON THE INFORMATION CONTAINED IN THIS PROXY STATEMENT TO VOTE YOUR SHARES AT THE SPECIAL MEETING. WE HAVE NOT AUTHORIZED ANYONE TO PROVIDE YOU WITH INFORMATION THAT IS DIFFERENT FROM WHAT IS CONTAINED IN THIS PROXY STATEMENT. THIS PROXY STATEMENT IS DATED , 2003. YOU SHOULD NOT ASSUME THAT THE INFORMATION CONTAINED IN THIS PROXY STATEMENT IS ACCURATE AS OF ANY DATE OTHER THAN THAT DATE, AND THE MAILING OF THIS PROXY STATEMENT TO STOCKHOLDERS DOES NOT CREATE ANY IMPLICATION TO THE CONTRARY.
61
STOCKHOLDER MEETINGS AND PROPOSALS
We intend to hold our 2004 Annual Meeting of Stockholders on June 24, 2004. Stockholder proposals that are intended to be presented at that meeting and included in the Company's proxy statement pursuant to the rules of the Securities and Exchange Commission must be received by our Secretary at our principal executive office, 4840 Pearl East Circle, Suite 300W, Boulder, Colorado 80301, no later than January 29, 2004. That date is the 120th day prior to May 29, which is the date on which we first mailed the proxy statement in connection with our 2003 Annual Meeting of Stockholders, as required by applicable regulations of the Securities and Exchange Commission. The deadline for receipt of stockholder proposals previously stated in the proxy statement in connection with our 2003 Annual Meeting of Stockholders was not correctly computed. If a stockholder wishes to submit a proposal or director nomination pursuant to our bylaws that is not to be included in the Company's proxy statement for the 2004 Annual Meeting, the stockholder must do so not less than 50 days nor more than 75 days prior to the meeting. If, however, less than 60 days notice or prior public disclosure of the date of the meeting is given or made to stockholders, notice by the stockholder must be received not later than the close of business on the tenth day following the day on which such notice of the date of the meeting was mailed or such public disclosure was made, whichever first occurs. Stockholders are also advised to review our bylaws, which contain additional requirements with respect to advance notice of stockholder proposals and director nominations.
The board of directors knows of no other business to be presented at the special meeting, but if other matters do properly come before the special meeting, the persons designated as agents in the enclosed proxy card will vote on those matters in accordance with their best judgment.
The board of directors encourages you to vote your shares by signing and returning the enclosed proxy card. The fact that you will have returned your proxy in advance will not affect your right to vote in person should you decide to attend. However, by signing and returning the proxy, you have assured your representation at the special meeting. Thank you for your cooperation.
By Order of the Board of Directors,
Patricia A. Pilia, Ph. D.
Secretary
Boulder, Colorado
September , 2003
62
* Portions denoted with an asterisk have been omitted and filed separately with the Securities & Exchange Commission pursuant to a request for confidential treatment.
Asset Purchase Agreement
Between
Faulding Pharmaceutical Co.
and
NaPro BioTherapeutics, Inc.
August 25, 2003
A-i
A-ii
| §10. Miscellaneous | A-43 | |||
| (a) | Press Releases and Public Announcements | A-43 | ||
| (b) | No Third-Party Beneficiaries | A-43 | ||
| (c) | Entire Agreement. | A-43 | ||
| (d) | Succession and Assignment | A-43 | ||
| (e) | Counterparts | A-43 | ||
| (f) | Headings | A-43 | ||
| (g) | Notices | A-43 | ||
| (h) | Governing Law | A-44 | ||
| (i) | Amendments and Waivers | A-44 | ||
| (j) | Severability | A-44 | ||
| (k) | Expenses | A-45 | ||
| (l) | Construction | A-45 | ||
| (m) | Incorporation of Exhibits and Schedules | A-45 | ||
| (n) | Bulk Transfer Laws | A-45 | ||
| (o) | Arbitration | A-45 | ||
| (p) | Limitations on Representations and Warranties | A-45 | ||
| (q) | Mylan Litigation | A-46 | ||
Exhibits and Schedules
- (a)
- Exhibits
- (b)
- Schedules
Exhibit A—Form of Intellectual Property Agreement
Exhibit B—Term Sheet for Transition Services Agreement
Exhibit C—[intentionally omitted]
Exhibit D—Financial Statements
Exhibit E—List of Gunbarrel Assets
Exhibit F—Abbott Termination Agreement
Exhibit G—Faulding Termination Agreement
Schedule A—Definition of Paclitaxel
Disclosure Schedule—Supplemental Information and exceptions to representations and warranties
[The Registrant shall provide supplementally a copy of any omitted exhibit or schedule to the
Securities and Exchange Commission upon request.]
A-iii
This Asset Purchase Agreement (this "Agreement") is entered into on August 25, 2003, between Faulding Pharmaceutical Co., a Delaware corporation ("Buyer"), and NaPro BioTherapeutics, Inc., a Delaware corporation ("Seller"). Buyer and Seller are each referred to in this Agreement as a "Party" and collectively as the "Parties."
This Agreement contemplates a transaction in which Buyer will purchase all assets (and assume specified liabilities) of Seller's business of the manufacture of bulk active pharmaceutical ingredient Paclitaxel according to Seller's drug master files and Seller's intellectual property, in each case related to the right to make, use or sell Paclitaxel formulated in polyethoxylated castor oil and ethanol in exchange for cash.
Now, therefore, in consideration of the premises and the mutual promises made in this Agreement, and in consideration of the representations, warranties, and covenants contained in this Agreement, the Parties agree as follows.
§1. Definitions. Unless otherwise expressly provided in this Agreement:
(a) The following terms, as used in this Agreement, have the following meanings:
"Abbott Agreement" means the Development, License, and Supply Agreement, dated as of July 23, 1999, by and between Abbott Laboratories and Seller, and all amendments thereto.
"Abbott Loan Agreement" means that certain Loan and Security Agreement, dated as of July 23, 1999, by and between Seller and Abbott Laboratories.
"Abbott Termination Agreement" means the Agreement attached hereto as Exhibit F.
"Abbott Note" means that certain promissory note of the Seller payable to Abbott Laboratories made pursuant to the Abbott Loan Agreement.
"Acquired Assets" means all of Seller's right, title, and interest in and to all of the assets of the Business as of the Closing Date, including (a) the Specified Patents, Specified Know-how, clinical trials data, Paclitaxel data, and all Intellectual Property owned or licensed to or by Seller and/or now or formerly used in the Business (including, without limitation, all Intellectual Property owned or licensed to or by Seller that is subject to the Bryn Mawr Agreement and the Polypro Process and the Semi-synthetic Process), (b) the Leased Real Property, (c) tangible personal property (such as machinery, equipment, inventories of raw materials and supplies, biomass, manufactured and purchased parts, goods in process and finished goods, and furniture) used in the Business wherever located, (d) Seller's rights in the Assumed Contracts, (e) claims, counterclaims, credits, deposits, prepayments, refunds, causes of action, choses in action, rights of recovery, rights of set off, and rights of recoupment (including any such item relating to the payment of Taxes), including, but not limited to the Mylan Litigation and all rights of Seller under the Confidentiality Agreements, (f) all Product Registrations, Regulatory Documentation, and Paclitaxel Records (subject to Seller's right to retain a copy of such documents for regulatory purposes), (g) the assets of the Cayman Subsidiary related to the Business, (h) books, records, ledgers, files, documents, correspondence, lists, plats, architectural plans, drawings, and specifications, market research data, sales information, training materials, marketing materials, product literature, promotional materials, creative materials, studies, reports, and other printed or written materials, (i) either all of the outstanding stock or assets of the UK Subsidiary, and (j) the Ross Computer ERP system;provided, however, that the Acquired Assets shall not include (i) Seller's corporate charter, qualifications to conduct business as a foreign corporation, arrangements with registered agents relating to foreign qualifications, taxpayer and other identification numbers, seals, minute books, stock transfer books, blank stock certificates, and other documents relating to the
A-1
organization, maintenance, and existence of Seller as a corporation; (ii) any Cash of Seller on hand immediately prior to Closing and the deposit on the Gunbarrel Facility lease; (iii) accounts receivable of Seller that accrue in accordance with GAAP prior to the Closing Date; (iv) all assets, including, but not limited to, Intellectual Property, of Seller that are not related to or used in the operation of the Business; (v) any of the rights of Seller under this Agreement (or under any other agreement between Seller on the one hand and Buyer on the other hand entered into on or after the date of this Agreement); (vi) all of the outstanding capital stock of the UK Subsidiary (unless stock rather than assets are acquired by Buyer); or (vii) the Retained Inventory.
"Adverse Consequences" means all actions, suits, proceedings, hearings, investigations, charges, complaints, claims, demands, injunctions, judgments, orders, decrees, rulings, damages, dues, penalties, fines, costs, reasonable amounts paid in settlement, liabilities, obligations, taxes, liens, losses, expenses, and fees, including court costs and reasonable attorneys' fees and expenses.
"Affiliate" has the meaning set forth in Rule 12b-2 of the regulations promulgated under the Securities Exchange Act. In the case of Seller, the term "Affiliate" shall include, without limitation, the UK Subsidiary and the Cayman Subsidiary.
"Affiliated Group" means any affiliated group within the meaning of Code §1504(a) or any similar group defined under a similar provision of state, local, or foreign law.
"Applicable Rate" means the Prime rate of interest published in the "Monday Rates" column of the United States edition ofThe Wall Street Journal.
"Assumed Contracts" means the Partner Agreements, the Cultivation Agreements, the Bryn Mawr Agreement, the BMS Agreement, the Ross Agreement, the Leases, and the other agreements, contracts, indentures, mortgages, instruments, Liens and guaranties set forth on §1A of the Disclosure Schedule.
"Assumed Liabilities" means (A) the liabilities and obligations of Seller related to the Business that appear on Schedule 1B of the Disclosure Schedule;provided, however, that the Assumed Liabilities shall not include unless otherwise disclosed in the Disclosure Schedule or expressly assumed by Buyer elsewhere in this Agreement: (s) any account payable and any liability, obligation or commitment of Seller and its Affiliates for materials and services connected to the manufacture, finishing, distribution and sale of any product to the extent such account payable, liability, obligations or commitment arose or accrued prior to the Closing Date; (t) any liability arising out of or relating to any product liability, breach of warranty, product recall or similar claim to the extent related to the Business or the manufacture or sale of Paclitaxel prior to the Closing Date by or on behalf of Seller or its Affiliates or otherwise related thereto (including all proceedings relating to any such liabilities); (u) any liability, obligation or commitment of any kind arising out of or relating to employment, compensation or benefits for the employees of Seller or its Affiliates prior to the Closing Date; (v) any liability, obligation or commitment of any kind arising out of or relating to any contract, agreement, lease, license, commitment, sale and purchase order and other instrument; (w) any liability, obligation or commitment of whatever kind and nature arising as a result of bulk active pharmaceutical ingredient Paclitaxel that is sold by Seller prior to the Closing and that is returned for any reason whatsoever; (x) any liability, obligation or commitment of whatever kind and nature, primary or secondary, to the extent arising out of or relating to, directly or indirectly, the conduct of the Business by Seller but, in each case, only to the extent related to any period prior to the Closing Date; or (y) any liability or obligation of Seller under this Agreement (or under any other agreement between Seller on the one hand and Buyer on the other hand entered into on or after the date of this Agreement); and (B) all liabilities associated with Buyer's ownership, operation and use of the Acquired Assets and the operation of the Business.
A-2
"Avoca Agreement" means that certain Agreement, dated August 25, 2003, by and between Seller and Avoca, Inc.
"Basis" means any past or present fact, situation, circumstance, status, condition, activity, practice, plan, occurrence, event, incident, action, failure to act, or transaction that forms or could form the basis for any specified consequence.
"BMS Agreement" means the Release and License Agreement, dated November 28, 2001, by and among Bristol-Myers Squibb Company, Seller and Abbott Laboratories.
"Bryn Mawr Agreement" means the License Agreement, dated November 3, 1993, by and between Seller and Bryn Mawr College relating to the license of certain technology.
"Business" means the research, development, manufacture, use, marketing or sale throughout the world of bulk active pharmaceutical ingredient Paclitaxel, including without limitation (a) Seller's Drug Master Files as described in the §1E of the Disclosure Schedule, (b) the right to make, have made, use, sell, offer for sale and import, bulk active pharmaceutical ingredient Paclitaxel and Paclitaxel formulated in polyethoxylated castor oil and ethanol for injection, (c) the Polypro Process, (d) the Semi-synthetic Process, and (e) Intellectual Property related to each of the foregoing; [*]
"Business Day" means any day other than a Saturday, a Sunday or a United States federal or New York State banking holiday.
"Cash" means cash and cash equivalents (including marketable securities and short term investments) calculated in accordance with GAAP applied on a basis consistent with the preparation of the Financial Statements.
"Cayman Subsidiary" means NBT, Ltd., an exempted company incorporated under the laws of the Cayman Islands.
"ChromaDex Contract" means that certain Master Agreement, dated as of April 8, 2003, by and between Seller and ChromaDex, Inc.
"Claim for Indemnification" means a written notice by Buyer or Seller to the other asserting with reasonable particularity (including an estimate of the extent of the dollar amount of Adverse Consequences) a claim under §8 delivered in accordance with §10(g).
"COBRA" means the requirements of Part 6 of Subtitle B of Title I of ERISA and Code §4980B and of any similar state law.
"Code" means the Internal Revenue Code of 1986, as amended, and the rules and regulations thereunder.
"Confidential Information" means any information concerning the businesses and affairs of the Business that is not already generally available to the public, including, but not limited to, the Paclitaxel Records, as well as the terms and conditions of this Agreement to the extent not otherwise required to be disclosed under Laws.
"Controlled Group" has the meaning set forth in Code §1563.
"Cultivation Agreements" mean all of the leases and services agreements related to cultivation of Yew trees, specified on §3(p)(xv) of the Disclosure Schedule.
"Delaware General Corporation Law" means Title 8 of the Delaware Code.
"Drug Master File (Australia)" means all documents filed (in the past, as of the date of this Agreement and as of the Closing Date) (both the open and closed portions) with the Therapeutic Goods Administration related to Seller's manufacture of bulk active pharmaceutical ingredient Paclitaxel.
A-3
"Drug Master File (Europe)" means all documents filed (in the past, as of the date of this Agreement and as of the Closing Date) (both the open and closed portions) with any European regulatory authority including the EMEA related to Seller's manufacture of bulk active pharmaceutical ingredient Paclitaxel.
"Drug Master File (United States)" means all documents filed (in the past, as of the date of this Agreement and as of the Closing Date) (both the open and closed portions) with the FDA related to Seller's manufacture of bulk active pharmaceutical ingredient Paclitaxel.
"EMEA" means the European Agency for the Evaluation of Medicinal Products.
"Employee Benefit Plan" means any "employee benefit plan" (as such term is defined in ERISA §3(3)) and any other material employee benefit plan, program or arrangement of any kind.
"Employee Pension Benefit Plan" has the meaning set forth in ERISA §3(2).
"Employee Welfare Benefit Plan" has the meaning set forth in ERISA §3(1).
"Environmental, Health, and Safety Requirements" shall mean all federal, state, local, and foreign statutes, regulations, ordinances, and similar provisions having the force or effect of law, all judicial and administrative orders and determinations, and all common law concerning public health and safety, worker health and safety, and pollution or protection of the environment, including all those relating to the presence, use, production, generation, handling, transportation, treatment, storage, disposal, distribution, labeling, testing, processing, discharge, release, threatened release, control, or cleanup of any hazardous materials, substances, or wastes, chemical substances or mixtures, pesticides, pollutants, contaminants, toxic chemicals, petroleum products or byproducts, asbestos, polychlorinated biphenyls, noise, or radiation.
"ERISA" means the Employee Retirement Income Security Act of 1974, as amended, and the rules and regulations thereunder.
"ERISA Affiliate" means each entity that is treated as a single employer with Seller for purposes of Code §414.
"Faulding Agreements" means the Second Amended and Restated Master Agreement, dated as of June 5, 2000, by and between Seller and F.H. Faulding & Co., Limited, and the European Agreement, dated as of March 2, 2001, by and between Seller and Central Laboratories, Limited.
"Faulding Termination Agreement" means the Agreement attached hereto as Exhibit G.
"FDA" means the United States Food and Drug Administration.
"FDCA" means the United States Food, Drug and Cosmetic Act of 1938, as amended, and the rules and regulations thereunder.
"Fiduciary" has the meaning set forth in ERISA §3(21).
"GAAP" means United States generally accepted accounting principles as in effect from time to time consistently applied.
"Governmental Authority" means any agency, authority, board, bureau, commission, court, legislative body, tribunal, department, office or instrumentality of any nature whatsoever or any governmental unit, whether federal, state, county, district, city, other political subdivision, or taxing district, foreign or otherwise, and whether now or hereafter in existence, or any officer or official thereof acting in an official capacity.
"Gunbarrel Assets" means the assets (other than Inventory) that were included on the balance sheet of Seller as of the Most Recent Fiscal Year End, that were located at the Gunbarrel Facility and
A-4
that have been or will be sold since the Most Recent Fiscal Year End in connection with the closure of the Gunbarrel Facility, including, without limitation, the assets set forth on Exhibit E hereto.
"Gunbarrel Facility" means that certain former manufacturing facility of Seller located at 6304 Spine Road, Unit A, Boulder, Colorado.
"Hart-Scott-Rodino Act" means the United States Hart-Scott-Rodino Antitrust Improvements Act of 1976, as amended.
"Income Tax" means any federal, state, local, or foreign income tax, including any interest, penalty, or addition thereto, whether disputed or not.
"Income Tax Return" means any return, declaration, report, claim for refund, or information return or statement relating to Income Taxes, including any schedule or attachment thereto, and including any amendment thereof.
"Intellectual Property" means all of the following in any jurisdiction throughout the world: (a) all inventions (whether patentable or unpatentable and whether or not reduced to practice), all improvements thereto as of the Closing Date, and all patents, patent applications, and patent disclosures, together with all reissuances, continuations, continuations-in-part, divisionals, revisions, extensions, and reexaminations thereof, and all patents and patent applications with a common priority in all countries, (b) all copyrightable works, all copyrights, and all applications, registrations, and renewals in connection therewith, (c) all trade secrets and confidential information (including ideas, research and development, know-how, formulas, compositions, manufacturing and production processes and techniques, technical data, designs, drawings, specifications, customer and supplier lists, pricing and cost information, and business and marketing plans and proposals), and (d) all computerized data, programmable logic controllers (PLCs), software and software licenses.
"Inventory" means all raw materials and supplies, co-products and by-products, work in process, and finished goods relating to the Business, including, without limitation, the bulk active pharmaceutical ingredient generically known as Paclitaxel.
"Knowledge" means, in the case of Seller, the actual knowledge, after due inquiry of the representatives of Seller listed on §1C of the Disclosure Schedule and, in the case of Buyer, the actual knowledge, after due inquiry, of the representatives of Buyer listed on §1D of the Disclosure Schedule.
"Law" means any applicable statute, code, constitution, ordinance, principle of common law, decree, ruling, plan, injunction, judgment, charge, order or regulation of any applicable Governmental Authority and any treaty or other international agreement or accord. All references to "Laws," "Law," or specific reference to statutory or regulatory sections are referenced to those laws, that law, or those specific references to such statutory or regulatory sections as of the date hereof and as of the Closing Date.
"Leased Real Property" means all leasehold or subleasehold estates and other rights to use or occupy any land, buildings, structures, improvements, fixtures, or other interest in real property held by Seller for use in the Business, including, but not limited to, a 27,000 square feet manufacturing facility located at the Sterling Drive Facility, together with all Leased Real Property Subleases and the real property interests subject to the Cultivation Agreements, including the right to all security deposits and other amounts and instruments deposited by or on behalf of Seller thereunder.
"Leased Real Property Subleases" means all subleases, licenses or other agreements pursuant to which Seller conveys or grants to any Person a subleasehold estate in, or the right to use or occupy, any real property, including, without limitation, the Leased Real Property or any portion thereof.
A-5
"Leases" means all leases, subleases, licenses, concessions and other agreements (written or oral), including all amendments, extensions, renewals, guaranties, and other agreements with respect thereto,, including the Cultivation Agreements, pursuant to which Seller holds any Leased Real Property.
"Lien" means any mortgage, pledge, lien, encumbrance, charge, claim, option, restriction, financing statement or other security interest of any kind or nature whatsoever, including any conditional sale or other title retention agreement and any lease having substantially the same effect as any of the foregoing and any assignment or deposit arrangement in the nature of a security devise.
"Material Adverse Effect" or "Material Adverse Change" means any effect or change that would be (or could reasonably be expected to be) materially adverse to the business, assets, condition (financial or otherwise), operating results, operations, or business prospects of the Business taken as a whole;provided,however, that in determining whether a Material Adverse Effect or Material Adverse Change has occurred, the following changes or effects shall not be considered and shall be deemed not to constitute a Material Adverse Effect or Material Adverse Change: (i) changes in the oncology industry generally (including, but not limited to, the competitive or regulatory environment affecting injectable Paclitaxel in any market, the sale price of injectable Paclitaxel and the costs associated with the manufacture or sale of injectable Paclitaxel); (ii) changes in economic conditions in the United States or globally or in the financial markets in general; (iii) changes of which Buyer has Knowledge that result from actions taken or to be taken in connection with the transactions contemplated by this Agreement; (iv) changes in Law that apply generally to similarly situated persons; (v) any change or effect to the extent constituting or involving only assets other than Acquired Assets or only liabilities other than Assumed Liabilities; and (vi) any change in or effect on the Acquired Assets that is cured (including by payment of money) before the earlier of the Closing and the termination of this Agreement pursuant to §9.
"MHRA" means the Medicines and Healthcare Products Regulatory Agency.
"Most Recent Balance Sheet" means the balance sheet contained within the Most Recent Financial Statements.
"Multiemployer Plan" has the meaning set forth in ERISA §3(37).
"Mylan Litigation" means the patent infringement action filed in June 2001 by Seller and Abbott in the United States District Court for the Western District of Pennsylvania against Mylan Laboratories, Inc., alleging infringement of U.S. Patent numbers 5,733,888, 5,972,992, 5,977,164, 6,140,359 and 6,306,894, all of which relate to Paclitaxel.
"Names" means any logos, trademarks, trade names or service marks of Seller or any of its Affiliates other than those included in the Acquired Assets.
"Ordinary Course of Business" means the ordinary course of business consistent with Seller's past custom and practice (including with respect to quantity and frequency).
"Paclitaxel" means the chemical entity of the formula set forth in Schedule A.
"Paclitaxel Records" means all documents owned by or in the possession of Seller related to Seller's process for manufacturing bulk active pharmaceutical ingredient Paclitaxel, including batch records, analytical methods, analytical results, Drug Master File (Australia), Drug Master File (Europe), Drug Master File (United States), pending amendments to the Drug Master Files, the Abridged Market Authorisation Application (AMAA) in Europe, and Seller's standard operating procedures for the manufacture and testing of bulk active pharmaceutical ingredient Paclitaxel.
"Partner Agreements" means those agreements listed on §3(p)(xiv) of the Disclosure Schedule.
"PBGC" means the Pension Benefit Guaranty Corporation.
A-6
"Permitted Liens" means with respect to any Acquired Asset: (a) Taxes, assessments and other governmental levies, fees, or charges imposed that are not due and payable as of the Closing Date (b) mechanics' liens and similar liens for labor, materials, or supplies incurred in the Ordinary Course of Business for amounts that are not due and payable as of the Closing Date; (c) zoning, building codes, and other land use laws regulating use or occupancy of any real property or the activities conducted thereon that are imposed by any Governmental Authority that are not violated by the current use or occupancy of such real property or the operation of the Business as currently conducted thereon; (d) easements, covenants, conditions, restrictions, and other similar matters of record affecting title to real property which do not or would not impair the use or occupancy of such real property in the operation of the Business as currently conducted thereon; (e) restrictions on transfer imposed by Law; and (f) liens securing rental payments under capital lease arrangements.
"Person" means an individual, a partnership, a corporation, a limited liability company, an association, a joint stock company, a trust, a joint venture, an unincorporated organization, any other business entity or a governmental entity (or any department, agency, or political subdivision thereof).
"Polypro Process" means the process relating to, among other things, the extraction of biomass and the concentration of extract, which process is described with particularity in the protocol heretofore or simultaneously delivered to Buyer.
"Prohibited Transaction" has the meaning set forth in ERISA §406 and Code §4975.
"Product Registrations" shall mean the approvals, registrations, applications, licenses, permits, franchises, orders, certificates, variances, and similar rights (including but not limited to any ANDA) related to or used in the operation of the Business which have been prepared, fixed and/or received (in preliminary and/or draft form, including without limitation those relating to any semi-synthetic process) in order to manufacture, market or sell bulk active pharmaceutical ingredient Paclitaxel (and related submissions to and correspondence with the regulatory authority responsible for the Product Registrations), in each case held in Seller's or any Affiliate of Seller's name, prior to the Closing Date to, from or with a Governmental Authority, and which are set forth on §1E of the Disclosure Schedule.
"Proxy Statement" means the proxy statement to be prepared by Seller and distributed to Seller Stockholders in connection with the Special Meeting.
"Regulatory Documentation" shall mean all (a) regulatory filings and supporting documents, chemistry, manufacturing and controls data and documentation, preclinical and clinical studies and tests; (b) records maintained under record keeping or reporting requirements of the FDA, EMEA, MHRA or any other Governmental Authority including without limitation the Drug Master File (Australia), the Drug Master File (Europe) and the Drug Master File (United States); and (c) any complaint, adverse event and medical inquiry filings with respect to the Business; in each case held in Seller's name or in any of its Affiliates' names and related to bulk active pharmaceutical ingredient Paclitaxel and/or Paclitaxel formulated in polyethoxylated castor oil and ethanol.
"Reportable Event" has the meaning set forth in ERISA §4043.
"Requisite Stockholder Approval" means the affirmative vote of the holders of a majority of the outstanding Seller Shares in favor of this Agreement.
[*]
"Retained Inventory" means any Inventory that has been committed to, and paid for by, Abbott Laboratories or any other Person who is not a party to this Agreement, [*].
"Ross Agreement" means the Software License and Professional Services Agreement, dated as of June 27, 2002, by and between Seller and Ross Systems, Inc.
A-7
"Ross Computer ERP system "means the primary server (NAPRO_ERP), an FRX Reporter server (NAPRO_SQL1), an Optio Forms Software server (NAPRO-OPTIO), all software and software licenses required to operate the Ross Computer ERP system on these servers in the Seller's operation of the Business (including, without limitation, the Oracle software), and all back-up hardware and data relating to each of the foregoing.
"SEC" means the U.S. Securities and Exchange Commission.
"Securities Exchange Act" means the Securities Exchange Act of 1934, as amended, and the rules and regulations thereunder.
"Seller Share" means any share of the common stock, par value US$0.0075 per share, of Seller.
"Seller Stockholder" means a holder of Seller Shares.
"Semi-synthetic Process" means all processes for the synthesis of Paclitaxel from 10- deacetylbaccatin III relating to and/or generally described in United States Drug Master File 16439 and/or the patents listed in §§3(l)(ii) and 3(l)(iii)(A) of the Disclosure Schedule and any modifications thereto to scale-up such process.
"Specified Know-how" means all trade secrets, inventions, confidential business information and other information (including SOPs, PLCs, flow diagrams, chemical, pharmacological, toxicological, pharmacological, safety, quality assurance, ideas, research and development, know-how, formulas, compositions, manufacturing and production processes and techniques, technical data, designs, drawings, specifications, customer and supplier lists, pricing and cost information, and business and marketing plans and proposals) related to the Business.
"Specified Patents" means the patents and registrations, pending patent applications or applications for registration specified on §3(l)(ii) of the Disclosure Schedule and any patent disclosures thereto (including all reissuances, continuations, continuations-in-part, divisionals, revisions, extensions and reexaminations), and all patents and patent applications with a common priority in all countries, owned by, or licensed to, Seller. For the avoidance of doubt, any patents and registrations, pending patent applications or applications for registration that are not listed on §3(l)(ii)of the Disclosure Schedule and any patent disclosures thereto (including all reissuances, continuations, continuations-in-part, divisionals, revisions, extensions and reexaminations), and all related patents and patent applications with a common priority in all countries, owned by, or licensed to, Seller, are not included in the Acquired Assets.
"Sterling Drive Facility" means that certain manufacturing facility located on Sterling Drive in Boulder, Colorado.
"Subsidiary" means, with respect to any Person, any corporation, limited liability company partnership, association, or other business entity of which (i) if a corporation, a majority of the total voting power of shares of stock entitled (without regard to the occurrence of any contingency) to vote in the election of directors, managers, or trustees thereof is at the time owned or controlled, directly or indirectly, by that Person or one or more of the other Subsidiaries of that Person or a combination thereof or (ii) if a limited liability company, partnership, association, or other business entity (other than a corporation), a majority of partnership or other similar ownership interest hereof is at the time owned or controlled, directly or indirectly, by that Person or one or more Subsidiaries of that Person or a combination thereof and for this purpose, a Person or Persons owns a majority ownership interest in such a business entity (other than a corporation) if such Person or Persons shall be allocated a majority of such business entity's gains or losses or shall be or control any managing director or general partner of such business entity (other than a corporation). The term "Subsidiary" shall include all Subsidiaries of such Subsidiary.
A-8
"Tax" or "Taxes" means any federal, state, local, or foreign income, gross receipts, license, payroll, employment, excise, severance, stamp, occupation, premium, windfall profits, environmental (including taxes under Code §59A), customs duties, capital stock, franchise, profits, withholding, social security (or similar), unemployment, disability, real property, personal property, sales, use, transfer, registration, value added, alternative or add-on minimum, estimated, or other tax, levy or other like assessment of any kind whatsoever, whether computed on a separate or consolidated, unitary or combined basis or in any other manner, including any interest, penalty, or addition thereto, whether disputed or not.
"Tax Return" means any return, declaration, report, claim for refund, or information return or statement relating to Taxes, including any schedule or attachment thereto, and including any amendment thereof.
"Third Party Claim" means any complaint, action, lawsuit, claim, proceeding, condemnation, investigation, audit or other legal proceeding by or before any Governmental Authority or any arbitration or other alternative dispute resolution proceeding made or brought by any Person who is not a Party or an Affiliate of a Party.
"Transaction Documents" means all agreements, instruments, certificates and other documents executed and/or delivered in connection with this Agreement, including, without limitation, the instruments of Assignment discussed in §2(e) hereof, the instruments of assignment and assumption delivered at Closing, the Intellectual Property Agreement, the Transition Services Agreement, the Abbott Termination Agreement, and the Faulding Termination Agreement.
"UK Subsidiary" means NaPro BioTherapeutics, Ltd., organized under the laws of the United Kingdom.
"USP Monograph" means the monograph for Paclitaxel adopted by The United States Pharmacopeial Convention, Inc., and as published in the U.S. Pharmacopeia as USP #26 effective January 1, 2003, subject to the issuance of the required reference standard.
"WARN Act" means the Worker Adjustment and Retraining Notification Act.
A-9
(b) Each of the following terms is defined in the location set forth opposite such term:
| Term | Location | |
|---|---|---|
| AAA | §10(o) | |
| Abbott Payment | §2(c) | |
| Agreement | Preface | |
| Base Inventory | §2(f)(i) | |
| Buyer | Preface | |
| CERCLA | §3(z) | |
| Closing | §2(e) | |
| Closing Date | §2(e) | |
| Closing Inventory | §3(f)(ii) | |
| Closing Payment | §2(c) | |
| Confidentiality Agreements | §3(w) | |
| Disclosure Schedule | §3 | |
| Employees | §6(g) | |
| Estoppel Certificates | §7(a) | |
| Fairness Opinion | §5(l) | |
| Final Inventory Measurement Date | §2(f)(ii) | |
| Financial Statements | §3(f) | |
| Former Seller Employee | §6(g)(i) | |
| Improvements | §3(k) | |
| Indemnified Party | §8(d) | |
| Indemnifying Party | §8(d) | |
| Inventory Adjustment Amount | §2(f)(vi) | |
| Inventory Notice | §2(f)(ii) | |
| Landlord Leases | §3(k) | |
| Lease Consents | §7(a) | |
| Letter | §6(i) | |
| Minimum | §8(b)(ii) | |
| Most Recent Financial Statements | §3(f) | |
| Most Recent Fiscal Month End | §3(f) | |
| Most Recent Fiscal Year End | §3(f) | |
| Party or Parties | Preface | |
| Purchase Price | §2(c) | |
| Real Property Laws | §3(k) | |
| Records | §6(f) | |
| Seller | Preface | |
| Seller Employee | §6(g)(i) | |
| Seller Pharmaceutical Products | §3(i) | |
| Seller Receivables Account | §6(i) | |
| Special Meeting | §5(k) | |
| Specified Patents | §3(l)(ii) | |
| Supply Agreement | §6(j) | |
| Transferred Employee | §6(g)(ii) |
(a) Purchase and Sale of Assets. On the terms and subject to the conditions of this Agreement, Buyer agrees to purchase from Seller, and Seller agrees to sell, transfer, convey, and deliver to Buyer, all of the Acquired Assets at the Closing for the consideration specified below in this §2.
A-10
(b) Assumption of Liabilities. On terms and subject to the conditions of this Agreement, Buyer agrees to assume and become responsible for all of the Assumed Liabilities at the Closing. Buyer will not assume or have any responsibility, however, with respect to any other obligation or liability of Seller not included within the definition of Assumed Liabilities.
(c) Closing Payment. Buyer agrees to pay at the Closing (i) US$71,700,000 minus the Inventory Adjustment Amount, if any, as determined in accordance with §2(f) of the Disclosure Schedule (such difference being hereinafter referred to as the "Base Amount"), of which (a) US$21,668,898, plus the amount of interest then due and payable under the loan to Seller from Abbott Laboratories under the Loan and Security Agreement, dated as of July 23, 1999, by and between Seller and Abbott Laboratories (the "Abbott Payment"), shall be paid by delivery of cash payable by transfer or delivery of other immediately available funds to the account designated by Abbott Laboratories or its designee, and (b) the Base Amount less the Abbott Payment (the amount computed in accordance with this subsection (b) is hereinafter referred to as the "Closing Payment") shall be paid by delivery of cash in the amount of the Closing Payment payable by wire transfer or delivery of other immediately available funds to the account designated by the Seller. The sum of the Abbott Payment, the Closing Payment, and the amount of the Assumed Liabilities is hereinafter referred to as the Purchase Price.
(d) The Closing. The closing of the transactions contemplated by this Agreement (the "Closing") shall take place via facsimile or electronic delivery of documents and receipt of documents previously sent by overnight courier service commencing at 10:00 a.m. (Mountain time) on the second Business Day following the satisfaction or waiver of all conditions to the obligations of the Parties to consummate the transactions contemplated by this Agreement (other than conditions with respect to actions each Party will take at the Closing itself), or such other date as the Parties may mutually determine (the "Closing Date");provided, however, that the Closing Date shall be no earlier than the date forty-five (45) days after the date of this Agreement. All of the deliveries of documents that are contemplated by this Agreement to be made at the Closing shall be delivered to the applicable Party by (i) overnight courier service for delivery on the Closing Date or (ii), if delivery by overnight courier service on the Closing Date is not practicable, then by facsimile on the Closing Date, with original executed documents delivered on the next succeeding Business Day. Any documents to be delivered to a Party on the Closing Date will be delivered and held in escrow until the Parties communicate via telephone to confirm delivery of all documents and consummation of all other actions contemplated by this §2.
(e) Deliveries at the Closing. At the Closing, (i) Seller will deliver to Buyer the various certificates, instruments, and documents referred to in §7(a); (ii) Buyer will deliver to Seller the various certificates, instruments, and documents referred to in §7(b); (iii) Seller will execute, acknowledge (if appropriate), and deliver to Buyer (A) instruments of assignment (including bills of sale, Real Property and Intellectual Property transfer documents), transfer, conveyance, and assignment as Buyer and its counsel reasonably may request; (iv) Buyer will execute, acknowledge (if appropriate) and deliver to Seller such instruments of assumption as Seller and its counsel reasonably request; (v) Buyer will execute, acknowledge (if appropriate), and deliver to Seller, and Seller will execute, acknowledge (if appropriate), and deliver to Buyer (A) an Intellectual Property Agreement in the form attached hereto as Exhibit A and (B) a Transition Services Agreement consistent with the term sheet attached hereto as Exhibit B; (vi) Buyer will deliver to Seller the Closing Payment specified in §2(c); (vii) Buyer will deliver to Abbott Laboratories or its designee the Abbott Payment specified in §2(c), and Buyer will deliver or cause to be delivered to Seller a copy of the Abbott Note marked "paid in full."
(i) The Inventory as of May 30, 2003 (the "Base Inventory") and the methodology for determining the amount of Inventory and calculating the value of Inventory are set forth on §2(f) of the Disclosure Schedule.
A-11
(ii) Not less than ten (10) Business Days prior to the Closing Date, Seller shall notify Buyer of the date on which Seller will take a final physical inventory count (the "Final Inventory Measurement Date"), which final physical inventory count date shall be at least 24 hours, but in no event more than 72 hours, prior to the Closing. Buyer shall have the right (but not the obligation) to observe such count in its sole discretion. On the Final Inventory Measurement Date, a physical inventory count will be conducted by Seller in accordance with the methodology set forth on §2(f) of the Disclosure Schedule or as agreed by Buyer and Seller prior to the Final Inventory Measurement Date in order to determine the Inventory as of the Final Inventory Measurement Date and will exclude the Retained Inventory (the "Closing Inventory"). Promptly upon completion of the final physical inventory count, Seller shall determine the value of such Inventory in accordance with the methodology set forth on §2(f) of the Disclosure Schedule and shall notify Buyer of the amount of Closing Inventory and the value of such Inventory (such notice being referred to as the "Inventory Notice").
(iii) In the event the Buyer agrees with the amount of the Inventory and the Inventory value calculation set forth in the Inventory Notice, the amount of, and value of such, Inventory shall constitute the Closing Inventory and the Inventory Adjustment Amount shall be calculated as set forth below. In the event Buyer does not agree with the amount of the Inventory and/or the Inventory value calculation set forth in the Inventory Notice, the Inventory Adjustment Amount for purposes of the Closing shall be zero;provided that Buyer and Seller shall negotiate in good faith to determine the Closing Inventory and the value of the Closing Inventory in accordance with §2(f)(iv) below.
(iv) In the event Buyer does not agree with the amount of the Inventory set forth in the Inventory Notice or believes that the principles set forth on §2(f) of the Disclosure Schedule have not been adhered to, Seller and Buyer shall undertake in good faith to agree upon the amount of the Inventory as of the Final Inventory Measurement Date in accordance with the methodology set forth on §2(f) of the Disclosure Schedule within five (5) Business Days after the Closing Date. If Buyer and Seller are unable to agree upon the Closing Inventory, as the case may be, within five (5) Business Days after the Closing Date, then Buyer and Seller hereby agree to submit such dispute to a internationally recognized independent auditing firm that is not then engaged by either Party for assurance services (the "Auditor"). The Auditor shall determine the Closing Inventory and apply the methodology specified on §2(f) of the Disclosure Schedule in determining the value of the Closing Inventory, and such determination shall be binding and final.
(v) To the extent the value of the Closing Inventory as established by the Auditor results in a positive Inventory Adjustment Amount, Seller shall immediately make payment to Buyer of the full Inventory Adjustment Amount. To the extent the value of the Closing Inventory as established by the Auditor results in a negative Inventory Adjustment Amount, Buyer shall immediately make payment to Seller of the full Inventory Adjustment Amount.
(vi) The "Inventory Adjustment Amount" shall be equal to (i) the value of the Base Inventory minus (ii) the value of the Closing Inventory, each as determined in accordance with the methodology set forth on §2(f) of the Disclosure Schedule and either agreed by Buyer and Seller or determined by the Auditor. Seller agrees that no sale or other disposition of Inventory shall occur between the Final Inventory Measurement Date and the Closing, without the express written consent of Buyer.
(g) Delivery After the Closing of the Mylan Litigation Payments. If resolution of the claims asserted against the adverse parties in the Mylan Litigation results in the entry of a final order by any court that requires such parties to pay damages as part of the resolution of such litigation (including any settlement thereof), Buyer agrees to pay Seller [*] of any cash payments received by Buyer from any adverse party in the Mylan Litigation pursuant to any judgment or settlement thereof, including
A-12
any royalties, as and when received by Buyer, as promptly as practicable following such resolution or settlement. For purposes of clarification, Buyer shall not be obligated to resolve the Mylan Litigation in any manner that requires any party to pay monetary damages, and Buyer shall not be required to provide to Seller the benefit, if any, which may result from any non-monetary resolution of the Mylan Litigation. Seller's failure to receive any payment pursuant to this section shall not constitute an Adverse Consequence;provided, however, that Buyer's failure to make payments as required by this section shall constitute an Adverse Consequence.
(h) Allocation. The Parties agree to allocate the Purchase Price among the Acquired Assets for all purposes (including financial accounting and tax purposes) in accordance with the allocation schedule delivered by Buyer to Seller pursuant to §5(r).
(i) Attempted Assignment of Acquired Assets or Assumption of Assumed Liabilities. If any attempted assignment of an Acquired Asset or assumption of any Assumed Liability pursuant to this Agreement would (i) constitute a breach or default under any contract, (ii) violate any Law or (iii) adversely affect the rights, or increase the obligations of Buyer, so that Buyer would not, in fact, receive all such rights, or assume the obligations, of Seller with respect thereto as they exist prior to such attempted assignment or assumption, then Buyer and Seller, without having to incur or suffer any Adverse Consequences, shall enter into such arrangements as may be reasonably acceptable to both Buyer and Seller to provide for or impose upon Buyer the benefits of such Acquired Assets or the obligations of such Assumed Liabilities, as the case may be, and any transfer or assignment to Buyer by Seller of any such Acquired Asset, or any assumption by Buyer of any such Assumed Liabilities, which shall require such consent or authorization of a third party that is not obtained, shall be made subject to such consent or authorization being obtained.
§3. Seller's Representations and Warranties. Subject to §10(p), Seller represents and warrants to Buyer that the statements contained in this §3 are correct and complete as of the date of this Agreement and will be correct and complete as of the Closing Date (as though made then and as though the Closing Date were substituted for the date of this Agreement throughout this §3), except as set forth in the disclosure schedule accompanying this Agreement and initialed by the Parties (the "Disclosure Schedule"). That portion of the Disclosure Schedule that pertains to Seller will be arranged in paragraphs corresponding to the lettered and numbered paragraphs contained in this §3.
(a) Organization of Seller. Seller is a corporation duly organized, validly existing, and in good standing under the laws of the jurisdiction of its incorporation. Seller has been duly qualified as a foreign corporation for the transaction of business, and is in good standing under the laws of each jurisdiction, including without limitation the State of Colorado, where the nature of its business makes such qualification necessary. Seller has all requisite power and authority to own, lease and operate the Acquired Assets and to carry on the Business as presently conducted.
(b) Authorization of Transaction. Seller has full power and authority (including full corporate or other entity power and authority) to execute and deliver this Agreement and the Transaction Documents and to perform its obligations hereunder and thereunder;provided,however, that Seller cannot consummate the transactions contemplated by this Agreement and the Transaction Documents unless and until it receives the Requisite Stockholder Approval. The board of directors of Seller has duly authorized the execution, delivery, and, subject to receiving the Requisite Stockholder Approval, performance of this Agreement and the Transaction Documents by Seller. This Agreement and the Transaction Documents constitute the valid and legally binding obligations of Seller, enforceable in accordance with their respective terms and conditions subject, however, to the effects of bankruptcy, insolvency, reorganization, moratorium or similar laws affecting creditors' rights generally, and to general principles of equity (regardless of whether such enforceability is considered in a proceeding in equity or at law).
A-13
(i) Neither the execution and the delivery of this Agreement, nor the consummation of the transactions contemplated hereby (including the assignments and assumptions referred to in §2 above), will (a) violate any Law to which Seller is subject or any provision of the charter or bylaws of Seller or any of its Subsidiaries or (b) conflict with, result in a breach of, constitute a default under, result in the acceleration of, create in any party the right to accelerate, terminate, modify, or cancel, or require any notice under any agreement, contract, lease, license, instrument, or other arrangement to which Seller or any of its Subsidiaries is a party or by which it is bound or to which any of its assets is subject (or result in the imposition of any Lien upon any of its assets other than Permitted Liens), except where the violation, conflict, breach, default, acceleration, termination, modification, cancellation, failure to give notice, or Lien would not have a Material Adverse Effect, or the required notice has been given or the required consent has been obtained.
(ii) Other than in connection with the provisions of the Hart-Scott-Rodino Act, the Delaware General Corporation Law, and the Securities Exchange Act, or as contemplated by §3(c) of the Disclosure Schedule, neither Seller nor any of its Subsidiaries needs to give any notice to, make any filing with, or obtain any authorization, consent, or approval of any Governmental Authority in order for the Parties to consummate the transactions contemplated by this Agreement (including the assignments and assumptions referred to in §2 above), except where the failure to give notice, to file, or to obtain any authorization, consent, or approval would not have a Material Adverse Effect.
(d) Brokers' Fees. Seller has no liability or obligation to pay any fees or commissions to any broker, finder, or agent with respect to the transactions contemplated by this Agreement for which Buyer could become liable or obligated.
(e) Title to Assets. As it pertains to the Business, except as set forth in §3(e) of the Disclosure Schedule, Seller has good and marketable title to, or a valid leasehold interest in, the properties and assets used by it, located on its premises, or shown on the Most Recent Balance Sheet or acquired after the date thereof, free and clear of all Liens (other than Permitted Liens), except for properties and assets disposed of in the Ordinary Course of Business since the date of the Most Recent Balance Sheet. Without limiting the generality of the foregoing, Seller has good and marketable title to all of the Acquired Assets, free and clear of any Lien or restriction on transfer other than Permitted Liens.
(f) Financial Statements. Attached hereto as Exhibit D are the following financial statements (collectively the "Financial Statements") (i) audited consolidated balance sheet and statement of operations, changes in stockholder's equity and cash flow as of and for the fiscal years ended December 31, 2001 and December 31, 2002 (the "Most Recent Fiscal Year End") with respect to the Company; and (ii) unaudited consolidated balance sheets and statements of income, as of and for the fiscal years ended December 31, 2001 and December 31, 2002 with respect to the Business; and (iii) unaudited consolidated balance sheets and statements of income (the "Most Recent Financial Statements") as of and for the seven months ended July 31, 2003 (the "Most Recent Fiscal Month End") with respect to the Business. The Financial Statements (including the notes thereto) have been prepared in accordance with GAAP applied on a consistent basis throughout the periods covered thereby. The financial statements referred to in clauses (ii) and (iii) above present fairly the financial condition of the Business as of such dates and the results of operations of the Business for such periods;provided, however, that the financial statements referred to in clauses (ii) and (iii) above lack footnotes and other presentation items and the financial statements referred to in clause (iii) above are subject to normal year-end adjustments (which will not be material individually or in the aggregate).
(g) Events Subsequent to Most Recent Fiscal Year End. Except as set forth in §3(g) of the Disclosure Schedule, since the Most Recent Fiscal Year End, there has not been any Material Adverse
A-14
Change with respect to the Business or the Acquired Assets. Without limiting the generality of the foregoing, since that date, with respect to the Business and the Acquired Assets:
(i) Seller has not sold, leased, transferred, or assigned any asset or group of assets, tangible or intangible, other than for fair consideration in the Ordinary Course of Business;
(ii) Seller has not entered into any agreement, contract, lease, or license (or series of agreements, contracts, leases or licenses which in the aggregate are) outside the Ordinary Course of Business;
(iii) no Person (including Seller) has accelerated, terminated, made modifications to, or cancelled any material agreement, contract, lease, or license to which Seller is a party or by which it is bound;
(iv) Seller has not imposed any Lien other than Permitted Liens upon any of its assets, tangible or intangible;
(v) Seller has not made any capital expenditures outside the Ordinary Course of Business;
(vi) Seller has not made any capital investment in, or any loan to, any other Person outside the Ordinary Course of Business;
(vii) Seller has not created, incurred, assumed, or guaranteed more than US$50,000 in aggregate indebtedness for borrowed money and capitalized lease obligations;
(viii) Seller has not transferred, assigned, or granted any license or sublicense of any material rights under or with respect to any Intellectual Property;
(ix) Seller has not experienced any material damage, destruction, or loss (whether or not covered by insurance) to its property;
(x) Seller has not (a) made any loan to any of the directors, officers, and employees of Seller or any of its Affiliates other than loans made in connection with payroll advances, initiation of employment or as permitted by employee benefit plans, or (b) entered into any other transaction outside the Ordinary Course of Business with any of the directors, officers, and employees of Seller or any of its Affiliates;
(xi) Seller has not entered into any employment contract or collective bargaining agreement, written or oral, or modified the terms of (i) any such existing contract or agreement or (ii) any employment policy;
(xii) Seller has not granted any increase in the base compensation of any of the directors, officers, and employees of Seller or any of its Affiliates outside the Ordinary Course of Business;
(xiii) Seller has not adopted, amended, modified, or terminated any bonus, profit sharing, incentive, severance, or other plan, contract, or commitment for the benefit of any employees of Seller involved with the Business, or taken any such action with respect to any other Employee Benefit Plan;
(xiv) Seller has not made any other material change in employment terms, including without limitation compensation and benefits for any of the employees involved with the Business outside the Ordinary Course of Business;
(xv) Seller has not changed its normal business practices or taken any other action outside the Ordinary Course of Business;
(xvi) Seller has not made any loans or advances of money; and
(xvii) Seller has not agreed, consented to or committed to do any of the foregoing.
A-15
(h) Undisclosed Liabilities. As it pertains to the Business, the liabilities set forth on the face of the Most Recent Balance Sheet (rather than in any notes thereto), the liabilities that have arisen after the Most Recent Fiscal Month End in the Ordinary Course of Business, and the liabilities set forth in §3(h) of the Disclosure Schedule are the only material liabilities (whether known or unknown, whether asserted or unasserted, whether absolute or contingent, whether accrued or unaccrued, whether liquidated or unliquidated, and whether due or to become due, including any liability for taxes) of Seller.
(i) Legal Compliance. As it pertains to the Business, Seller has complied with all Laws, and no action, suit, proceeding, hearing, investigation, charge, complaint, claim, demand, or notice has been filed or commenced against it alleging any failure so to comply, except where the failure to comply would not, individually or in the aggregate, have a Material Adverse Effect. Without limiting the generality of the foregoing:
(i) Seller holds all licenses from, and has submitted notices to, all Governmental Authorities (including, without limitation, all authorizations under the FDCA and all regulations of the FDA, the EMEA and all other applicable Governmental Authorities necessary for the lawful conduct of the Business);
(ii) to Seller's Knowledge, all intermediaries and drug products being manufactured, distributed, or developed by or for the benefit of Seller, including without limitation those being manufactured, distributed or developed by Avoca, Inc., ("Seller Pharmaceutical Products") that are subject to the jurisdiction of any Governmental Authority (including the FDA) are being manufactured, labeled, stored, tested, distributed, and marketed in compliance with all applicable requirements under the Laws and implementing regulations (including, as applicable, the FDCA and the Public Health Service Act, and their applicable implementing regulations);
(iii) to Seller's Knowledge, all clinical trials conducted by or for the benefit of Seller have been, and are being, conducted in material compliance with the applicable requirements of "Good Clinical Practice", informed consent, and all applicable requirements relating to protection of human subjects contained in 21 C.F.R. Parts 50, 54, 56 and 312 and all applicable Laws, including the privacy requirements of the Health Insurance Portability and Accountability Act and its implementing regulations;
(iv) all manufacturing operations conducted by or for the benefit of Seller relating to drugs for human use have been and are being conducted in compliance with the FDA's applicable current "Good Manufacturing Practice" regulations for drug products, to the extent applicable;
(v) Seller is in compliance in all respects with all applicable registration and listing requirements set forth in 21 U.S.C. Section 360 and 21 C.F.R. Part 207 and all similar Laws;
(vi) no Seller Pharmaceutical Product has been recalled, suspended or discontinued as a result of any action by the FDA or any other similar foreign governmental entity by Seller or, to the Knowledge of Seller, any licensee, distributor or marketer of any Seller Pharmaceutical Product, in the United States or outside of the United States;
(vii) Seller has not received any written notice that the FDA or any other Governmental Authority has commenced, or threatened to initiate, any action to withdraw approval, place marketing or sale restrictions, or request the recall of any Seller Pharmaceutical Product, or commenced, or threatened to initiate, any action to enjoin or place restrictions on the production, sale, marketing or reimbursement of any Seller Pharmaceutical Products;
(viii) Seller has not committed any act, made any statement or failed to make any statement that would reasonably be expected to provide a basis for the FDA to invoke its policy with respect
A-16
to "Fraud, Untrue Statements of Material Facts, Bribery, and Illegal Gratuities" set forth in 56 Fed. Reg. 46191 (September 10, 1991) and any amendments thereto;
(ix) neither Seller, nor any officer or employee nor, to the Knowledge of Seller, any agent of Seller has been convicted of any crime or engaged in any conduct that would reasonably be expected to result in (A) debarment under 21 U.S.C. Section 335a or any similar state law or regulation or (B) exclusion under 42 U.S.C. Section 1320a-7 or any similar state law or regulation; and
(x) neither Seller, nor to the Knowledge of Seller, any director, officer, manager, advisor, agent, or employee of Seller, nor any other Person associated with or acting for or on behalf of Seller, has directly or indirectly in connection with the operation of the Business (i) made or promised to make any contribution, gift, bribe, rebate, payoff, influence payment, kickback, or other payment to any Person, private or public, regardless of form, whether in money, property, or services, or otherwise taken any similar action, (A) to obtain favorable treatment in securing business, (B) to pay for favorable treatment for business secured, (C) to obtain special concessions or for special concessions already obtained for or in respect of Seller or any Affiliate of Seller, or (D) in material violation of any Law, or (b) established or maintained any fund or asset that has not been recorded in the books and records of Seller.
Seller makes no representations or warranties in this §3(i) with respect to Taxes, Environmental, Health, and Safety Requirements, Intellectual Property, or Laws respecting employment, employment practices, wage payment, health or safety, for which the sole representations and warranties of Seller are set forth in §§3(j), 3(z), 3(l) and 3(x) and (z), respectively.
(i) Seller is not currently the beneficiary of any extension of time within which to file any Income Tax Return. Seller has timely filed all Tax Returns that it was required to file. All such Tax Returns are correct and complete in all material respects. All Taxes owed by Seller (whether or not shown on any Tax Return) have been paid. As it pertains to the Business, Seller has withheld and paid all Taxes required to have been withheld and paid in connection with amounts paid or owing to any employee, independent contractor, creditor, stockholder, or other third party, and all Forms W-2 and 1099 required with respect thereto have been properly completed and timely filed. Seller has complied in all material respects with filing requirements related to tax returns in each jurisdiction in which it may be subject to taxation.
(ii) There is no dispute or claim concerning any Tax liability either (A) claimed or raised by any authority in writing or (B) as to which Seller has Knowledge.
(iii) §3(j) of the Disclosure Schedule lists all federal, state, local, and foreign Income Tax Returns filed with respect to the Business for taxable periods ended on or after December 31, 1999, indicates those Income Tax Returns that have been audited, and indicates those Income Tax Returns that currently are the subject of audit. Seller has delivered to Buyer correct and complete copies of all federal Income Tax Returns, examination reports, and statements of deficiencies assessed against or agreed to by Seller since December 31, 1999. As it pertains to the Business, Seller has not waived any statute of limitations in respect of Income Taxes or agreed to any extension of time with respect to an Income Tax assessment or deficiency.
(iv) As it pertains to the Business, Seller (A) has not made any material payments, is not obligated to make any material payments, and is not a party to any agreement that under certain circumstances could obligate it to make any material payments that will not be deductible under Code §280G, and (B) is not a party to any Tax allocation or sharing agreement. Neither Seller nor any Subsidiary (1) has been a member of an Affiliated Group filing a consolidated federal Income Tax Return (other than a group the common parent of which was Seller) or (2) has any liability for
A-17
the taxes of any Person (other than Seller or any Subsidiary of Seller) under Reg. §1.1502-6 (or any similar provision of state, local, or foreign law), as a transferee or successor, by contract, or otherwise.
(v) As it pertains to the Business, the unpaid Taxes of Seller (A) did not, as of the Most Recent Fiscal Month End, exceed by any material amount the reserve for Tax liability (rather than any reserve for deferred Taxes established to reflect timing differences between book and Tax income) set forth on the face of the Most Recent Balance Sheet (rather than in any notes thereto) and (B) will not exceed by any material amount that reserve as adjusted for operations and transactions through the Closing Date in accordance with the past custom and practice of Seller in filing its Tax Returns.
(i) Seller does not own any real property that is used in the Business.
(ii) The Leased Real Property identified in §3(k)(ii) of the Disclosure Schedule (collectively, the "Leased Real Property") comprises all of the real property used or intended to be used in the Business; and Seller is not a party to any agreement or option related to the Business to purchase any additional real property or interest therein. §3(k)(ii) of the Disclosure Schedule sets forth the address of each parcel of Leased Real Property, and a true and complete list of all Leases for each such Leased Real Property (including the date and name of the parties to such Lease document). Seller has delivered to Buyer a true and complete copy of each such Lease document, and in the case of any oral Lease, a written summary of the material terms of such Lease.
(iii) Except as set forth on §3(k)(iii) of the Disclosure Schedule, with respect to each of the Leases:
(A) such Lease is legal, valid, binding, enforceable and in full force and effect;
(B) the transaction contemplated by this Agreement does not require the consent of any other party to such Lease, will not result in a breach of or default under such Lease, and will not otherwise cause such Lease to cease to be legal, valid, binding, enforceable and in full force and effect on identical terms following the Closing;
(C) Seller's possession and quiet enjoyment of the Leased Real Property under each Lease with respect to Leased Real Property has not been disturbed and, to the Knowledge of Seller, there are no disputes with respect to any Lease;
(D) to the Knowledge of Seller, neither Seller nor any other party to the Lease is in breach or default under such Lease; and, to the Knowledge of Seller, no event has occurred or circumstance exists which, with the delivery of notice, the passage of time or both, would constitute such a breach or default, or permit the termination, modification or acceleration of rent under such Lease;
(E) no security deposit or portion thereof deposited with respect to such Lease has been applied in respect of a breach or default under such Lease which has not been redeposited in full;
(F) Seller does not owe, and will not owe in the future, any brokerage commissions or finder's fees with respect to such Lease;
(G) the other party to such Lease is not an Affiliate of, and otherwise does not have any economic interest in, Seller;
(H) Seller has not subleased, licensed or otherwise granted any Person the right to use or occupy any Leased Real Property or any portion thereof; and
A-18
(I) Seller has not collaterally assigned or granted any other Lien in such Lease or any interest therein.
(iv) §3(k) of the Disclosure Schedule sets forth a true and complete list of all Leased Real Property Subleases (including all amendments, extensions, renewals, guaranties and other agreements with respect thereto) (collectively, the "Landlord Leases"), including the date and name of the parties to such Landlord Lease document. Seller has delivered to Buyer a true and complete copy of each such Landlord Lease document, and in the case of any oral agreement, a written summary of the material terms of such agreement. Except as set forth in §3(k)(iv) of the Disclosure Schedule, with respect to each of the Landlord Leases: (i) such Landlord Lease is legal, valid, binding, enforceable and in full force and effect; (ii) none of Seller or, to the Knowledge of Seller, any other party to such Landlord Lease, is in breach or default thereunder, and, to the Knowledge of Seller, no event has occurred or circumstance exists which, with the delivery of notice, the passage of time or both, would constitute such a breach or default thereunder; (iii) no security deposit or portion thereof deposited with respect to such Landlord Lease has been applied in respect of a breach or default under such Landlord Lease which has not been redeposited in full; (iv) Seller does not owe, and will not owe in the future, any brokerage commissions or finder's fees with respect to such Landlord Lease; and (v) the other party to such Landlord Lease is not an Affiliate of, and otherwise does not have any economic interest in, Seller.
(v) All buildings, structures, fixtures, building systems and equipment, and all components thereof, included in the Leased Real Property (the "Improvements") are in good condition and repair and sufficient for their current use in the Business. There are no facts or conditions affecting any of the Improvements which would, individually or in the aggregate, interfere in any material respect with the use or occupancy of the Improvements or any portion thereof in the operation of the Business as currently conducted thereon.
(vi) There is no injunction, decree, order, writ or judgment outstanding, nor any claims, litigation, administrative actions or similar proceedings, pending or, to the Knowledge of Seller, threatened, relating to the ownership, lease, use or occupancy of the Leased Real Property or any portion thereof, or the operation of the Business as currently conducted thereon.
(vii) To the Knowledge of Seller, the Leased Real Property is in material compliance with all applicable building, zoning, subdivision, health and safety and other land use laws, including the Americans with Disabilities Act of 1990, as amended, and all insurance requirements affecting the Leased Real Property (collectively, the "Real Property Laws"). Seller has not received any notice of violation of any Real Property Law and, to the Knowledge of Seller, there is no Basis for the issuance of any such notice or the taking of any action for such violation.
(viii) Seller's use or occupancy of the Leased Real Property or any portion thereof and the operation of the Business as currently conducted is not dependent on a "permitted non-conforming use" or "permitted non-conforming structure" or similar variance, exemption or approval from any Governmental Authority.
(ix) To the Knowledge of Seller, none of the Leased Real Property or any portion thereof is located in a flood hazard area (as defined by the Federal Emergency Management Agency).
(l) Intellectual Property. With respect to the Specified Patents, the Specified Know-how, the Paclitaxel Records, the Intellectual Property used in the Business (including the Polypro Process and the Semi-synthetic Process), the Ross Computer ERP, and all other software used in the Business:
(i) To the Knowledge of Seller, except as set forth on §3(l)(i)(A) of the Disclosure Schedule, Seller has not infringed upon, misappropriated, or violated any material Intellectual Property rights of third parties, and Seller has not received any charge, complaint, claim, demand, or notice alleging any such infringement, misappropriation, or violation (including any claim that Seller must
A-19
license or refrain from using any Intellectual Property rights of any third party). To the Knowledge of Seller, except as set forth on §3(l)(i)(B) of the Disclosure Schedule, no third party has infringed upon, misappropriated, or violated any Intellectual Property rights of Seller in any respect.
(ii) §3(l)(ii) of the Disclosure Schedule identifies each of the Specified Patents, and identifies each material license, agreement, or other permission which Seller has granted to any third party with respect to any of its Intellectual Property (together with any exceptions). Seller has delivered to Buyer correct and complete copies of all such patents, registrations, applications, licenses, agreements, and permissions (as amended to date). §3(l)(ii) of the Disclosure Schedule also identifies each material trade name or unregistered trademark, service mark, corporate name, Internet domain name, copyright and all software used by Seller in connection with the Business.
(iii) Except as set forth on §3(l)(iii) of the Disclosure Schedule, with respect to each item of Intellectual Property required to be identified in §3(l)(ii) of the Disclosure Schedule:
(A) Seller possesses all right, title, and interest in and to the item, free and clear of any Lien, license, or other restriction;
(B) the item is not subject to any outstanding injunction, judgment, order, decree, ruling, or charge;
(C) no action, suit, proceeding, hearing, investigation, charge, complaint, claim, or demand is pending or, to the Knowledge of Seller, is threatened which challenges the legality, validity, enforceability, use, or ownership of the item;
(D) Seller has not agreed to indemnify any Person for or against any infringement, misappropriation, or other conflict with respect to the item; and
(E) no loss or expiration of the item is threatened, pending, or reasonably foreseeable, except for patents expiring at the end of their statutory terms (and not as a result of any act or omission by Seller, including, without limitation, a failure by Seller to pay any required maintenance fees).
(iv) §3(l)(iv) of the Disclosure Schedule identifies each material item of Intellectual Property that any third party owns and that Seller uses pursuant to license, sublicense, agreement, or permission in connection with the Business. Seller has delivered to Buyer correct and complete copies of all such licenses, sublicenses, agreements, and permissions (as amended to date).
(v) Except as set forth on §3(l)(v) of the Disclosure Schedule, with respect to each item of Intellectual Property required to be identified in §3(l)(iv) of the Disclosure Schedule:
(A) the license, sublicense, agreement, or permission covering the item is legal, valid, binding, enforceable, and in full force and effect in all material respects;
(B) no party to the license, sublicense, agreement, or permission is in material breach or default, and no event has occurred which with notice or lapse of time would constitute a material breach or default or permit termination, modification, or acceleration thereunder;
(C) no party to the license, sublicense, agreement, or permission has repudiated any material provision thereof; and
(D) Seller has not granted any sublicense or similar right with respect to the license, sublicense, agreement, or permission.
(vi) §3(l)(vi) of the Disclosure Schedule identifies the Specified Patents related to the Semi-synthetic Process owned or used by Seller.
A-20
(vii) The Intellectual Property licensed by Seller under the Bryn Mawr Agreement is used by Seller in the Semi-synthetic Process.
(viii) The Acquired Assets include all Intellectual Property used by Seller in the Business.
(m) Paclitaxel Records. The Paclitaxel Records completely and correctly reflect Seller's operations and manufacturing processes in accordance with requirements of all Governmental Authorities.
(n) Assets. Except for the Gunbarrel Assets, Seller owns, leases or licenses all buildings, machinery, equipment, other tangible assets, patents, copyrights, know-how, and other intangible assets necessary or desirable for the conduct of the Business as presently conducted and as conducted since January 1, 2003, and all such assets are included in the Acquired Assets to be conveyed to Buyer hereunder. Without limitation of the foregoing, no computer hardware or other equipment (other than that which is included in the Acquired Assets) is needed to operate the Ross Computer ERP system. Except as set forth in §3(n) of the Disclosure Schedule, there have been no dispositions of any material assets (singly or in the aggregate) since May 30, 2003. Without limitation of the foregoing, none of the Gunbarrel Assets are necessary or desirable for the conduct of the Business. Seller owns no trademarks necessary or desirable for the conduct of the Business. The buildings, machinery, equipment, and other tangible assets included in the Acquired Assets are free from material defects, have been maintained in accordance with normal industry practice, and are in good operating condition and repair (subject to normal wear and tear), and each is suitable for the purposes for which it is presently used.
(o) Inventory. All of the Inventory of Seller included in the Acquired Assets is merchantable and fit for the purpose for which it was procured or manufactured and conforms with the specifications required under the applicable contract(s) pursuant to which it will be delivered, to the extent that the Inventory is identified to such contract(s), and none of such Inventory is damaged or defective, subject only to the reserve for inventory write-down set forth in §2(f) the Disclosure Schedule as adjusted for operations and transactions through the Closing Date in accordance with GAAP and the past custom and practice of Seller.
(p) Contracts. §3(p) of the Disclosure Schedule lists the following contracts and other agreements to which Seller is a party that pertain to the Business or the Acquired Assets:
(i) any agreement (or group of related agreements) for the lease of personal property to or from any Person providing for lease payments in excess of US$50,000 per annum;
(ii) any agreement (or group of related agreements) for the purchase or sale of raw materials, commodities, supplies, products, or other personal property, or for the furnishing or receipt of services, the performance of which will extend over a period of more than one year, or involve consideration in excess of US$50,000;
(iii) any agreement concerning a strategic alliance, partnership or joint venture;
(iv) any agreement (or group of related agreements) under which Seller has created, incurred, assumed, or guaranteed any indebtedness for borrowed money, or any capitalized lease obligation, in excess of US$50,000 or under which it has imposed a Lien on any of its assets, tangible or intangible;
(v) any agreement concerning non-competition, or any material agreement concerning confidentiality, other than the Partner Agreements and the Confidentiality Agreements;
(vi) any material agreement involving any Affiliate of Seller (including any Subsidiaries);
(vii) any profit sharing, deferred compensation, severance, or other plan for the benefit of the officers and employees of Seller or any of its Affiliates;
A-21
(viii) any collective bargaining agreement;
(ix) any agreement for the employment of any individual on a full-time basis; and any agreement for the employment of any individual on a part-time, consulting, or other basis, in excess of US$50,000 per year or not terminable on 30 days' notice or less or providing material severance benefits;
(x) any agreement under which it has advanced or loaned any amount to any of the directors, officers, and employees of Seller or any of its Affiliates;
(xi) any agreement under which the consequences of a default or termination could have a Material Adverse Effect;
(xii) any agreement under which Seller has advanced or loaned any other Person amounts in the aggregate exceeding US$10,000;
(xiii) any agreement (or group of related agreements) not otherwise referred to in this §3(p) the performance of which involves consideration in excess of US$50,000;
(xiv) the Partner Agreements; and
(xv) the Cultivation Agreements.
Seller has delivered to Buyer a correct and complete copy of each written agreement listed in §3(p) of the Disclosure Schedule (as amended to date). With respect to each such agreement: (A) the agreement is legal, valid, binding, enforceable, and in full force and effect in all material respects; (B) no party is in material breach or default, and to the Knowledge of Seller, no event has occurred which with notice or lapse of time would constitute a material breach or default, or permit termination, modification, or acceleration, under the agreement; and (C) neither Seller nor, to the Knowledge of Seller, any other party has repudiated any material provision of the agreement.
(q) Notes. All notes of Seller related to the Business or included in the Acquired Assets are reflected properly on Seller's books and records, are valid and subject to no setoffs or counterclaims, are current and collectible, and will be collected in accordance with their terms at their recorded amounts.
(r) Powers of Attorney. There are no outstanding powers of attorney executed on behalf of Seller that pertain to the Business or the Acquired Assets.
(s) Insurance. §3(s) of the Disclosure Schedule sets forth the following information with respect to each material insurance policy (including policies providing property, casualty, liability, and workers' compensation coverage and bond and surety arrangements) with respect to which Seller is a party, a named insured, or otherwise the beneficiary of coverage affecting the Business or the Acquired Assets:
(i) the name, address, and telephone number of the agent;
(ii) the name of the insurer, the name of the policyholder, and the name of each covered insured;
(iii) the policy number and the period of coverage;
(iv) the scope (including an indication of whether the coverage is on a claims made, occurrence, or other basis) and amount (including a description of how deductibles and ceilings are calculated and operate) of coverage; and
(v) a description of any retroactive premium adjustments or other material loss sharing arrangements.
A-22
With respect to each such insurance policy: (A) the policy is legal, valid, binding, and in full force and effect in all material respects; (B) neither Seller nor any other party to the policy is in material breach or default (including with respect to the payment of premiums or the giving of notices), and no event has occurred which, with notice or the lapse of time, would constitute such a material breach or default, or permit termination, modification, or acceleration, under the policy; and (C) no party to the policy has repudiated any material provision thereof. §3(s) of the Disclosure Schedule describes any material self-insurance arrangements affecting Seller.
(t) Litigation. §3(t) of the Disclosure Schedule sets forth each instance in which Seller (i) is subject to any outstanding injunction, judgment, order, decree, ruling, or charge affecting the Business or the Acquired Assets, or (ii) is a party or, to the Knowledge of Seller, is threatened to be made a party to any action, suit, proceeding, hearing, or investigation of, in, or before any court or quasi-judicial or administrative agency of any federal, state, local, or foreign jurisdiction or before any arbitrator with respect to the Business or the Acquired Assets.
(u) Product Warranty. All of the products manufactured, sold, and delivered by Seller in connection with the Business have conformed in all material respects with all applicable contractual commitments and all express and implied warranties, and Seller does not have any material liability (whether known or unknown, whether asserted or unasserted, whether absolute or contingent, whether accrued or unaccrued, whether liquidated or unliquidated, and whether due or to become due) for replacement or repair thereof or other damages in connection therewith, subject only to the reserve for product warranty claims set forth on the face of the Most Recent Balance Sheet (rather than in any notes thereto) as adjusted for operations and transactions through the Closing Date in accordance with the past custom and practice of Seller. All of the products manufactured, sold, or delivered by Seller as part of the Business are subject to standard terms and conditions of sale as set forth in the Partner Agreements.
(v) Product Liability. To the Knowledge of Seller, Seller does not have any material liability (whether known or unknown, whether asserted or unasserted, whether absolute or contingent, whether accrued or unaccrued, whether liquidated or unliquidated, and whether due or to become due) arising out of any injury to individuals or property as a result of the ownership, possession, or use of any product manufactured, sold, leased, or delivered by Seller as part of the Business.
(w) Employees. To the Knowledge of Seller, no executive, officer or employee named on §6(g) of the Disclosure Schedule plans to terminate employment with Seller during the next 12 months, except as required to facilitate the transactions contemplated by this Agreement. As it pertains to the Business, Seller is not a party to or bound by any collective bargaining agreement, nor has it experienced any strike or material grievance, claim of unfair labor practices, or other collective bargaining dispute within the past three years before the NLRB or otherwise. Seller has not committed any material unfair labor practice in connection with the operation of the Business. Seller has no Knowledge of any organizational effort presently being made or threatened by or on behalf of any labor union with respect to employees of Seller involved with the Business. Seller has paid in full all amounts owed to the Employees (whether for salary, bonus or otherwise). Except as set forth on §3(w) of the Disclosure Schedule, each of the Employees and Former Seller Employees is, and as of the Closing Date, shall be, party to a valid and enforceable confidentiality agreement with Seller (each a "Confidentiality Agreement" and, collectively, the "Confidentiality Agreements"). True and correct copies of each of the Confidentiality Agreements, together with a list identifying (A) the persons party to such Confidentiality Agreements and (B) with respect to each of the Former Seller Employees, his/her respective last date of employment, have been delivered to Buyer. No bonus or bonuses are payable to any of the Seller Employees with respect to services provided prior to the Closing date except for retention bonuses and bonuses that are contingent upon the success of the transactions contemplated by this Agreement.
A-23
(i) §3(x)(i) of the Disclosure Schedule lists each Employee Benefit Plan that Seller maintains or to which Seller contributes or has any obligation to contribute in connection with the operation of the Business.
(A) Each such Employee Benefit Plan (and each related trust, insurance contract, or fund) has been maintained, funded and administered substantially in accordance with the terms of such Employee Benefit Plan and complies in form and in operation in all material respects with the applicable requirements of ERISA, the Code, and other applicable laws.
(B) All required reports and descriptions (including Form 5500 annual reports), summary annual reports, and summary plan descriptions) have been timely filed and/or distributed in accordance with the applicable requirements of ERISA and the Code with respect to each such Employee Benefit Plan. The requirements of COBRA have been met in all material respects with respect to each such Employee Benefit Plan and any Employee Benefit Plan maintained by an Seller or an ERISA Affiliate that is an Employee Welfare Benefit Plan subject to COBRA.
(C) All contributions (including all employer contributions and employee salary reduction contributions) that are due have been made within the time periods prescribed by ERISA and the Code to each such Employee Benefit Plan that is an Employee Pension Benefit Plan and all contributions for any period ending on or before the Closing Date which are not yet due have been made to each such Employee Pension Benefit Plan or accrued in accordance with the past custom and practice of Seller. All premiums or other payments for all periods ending on or before the Closing Date have been paid with respect to each such Employee Benefit Plan that is an Employee Welfare Benefit Plan.
(D) Each such Employee Benefit Plan that is intended to meet the requirements of a "qualified plan" under Code §401(a) has received a determination from the Internal Revenue Service that such Employee Benefit Plan is so qualified, and Seller is not aware of any facts or circumstances that could adversely affect the qualified status of any such Employee Benefit Plan.
(E) There have been no Prohibited Transactions with respect to any Employee Benefit Plan maintained by Seller or an ERISA Affiliate. No Fiduciary has any liability for material breach of fiduciary duty or any other material failure to act or comply in connection with the administration or investment of the assets of any such Employee Benefit Plan. No action, suit, proceeding, hearing, or investigation with respect to the administration or the investment of the assets of any such Employee Benefit Plan (other than routine claims for benefits) is pending or, to the Knowledge of Seller, threatened.
(F) Seller has delivered to Buyer correct and complete copies of the plan documents and summary plan descriptions, the most recent determination letter received from the Internal Revenue Service, the most recent annual report (IRS Form 5500, with all applicable attachments), and all related trust agreements, insurance contracts, and other funding arrangements which implement each such Employee Benefit Plan.
(ii) Neither Seller nor any Subsidiary of Seller, nor any ERISA Affiliate contributes to, has any obligation to contribute to, or has any Liability under or with respect to any Employee Pension Benefit Plan that is a "defined benefit plan" (as defined in ERISA §3(35)).
(iii) None of Seller, its Subsidiaries, or any ERISA Affiliate contributes to, has any obligation to contribute to, or has any material liability (including withdrawal liability as defined in ERISA §4201) under or with respect to any Multiemployer Plan.
A-24
(iv) Seller does not maintain, contribute to or have an obligation to contribute to, or have any material liability or potential liability with respect to, any Employee Welfare Benefit Plan providing health or life insurance or other welfare-type benefits for current or future retired or terminated employees of Seller (or any spouse or other dependent thereof) other than in accordance with COBRA.
(y) Guaranties. Seller, as it pertains to the Business, neither is a guarantor nor otherwise is responsible for any liability or obligation (including indebtedness) of any other Person.
(z) Environmental, Health, and Safety Matters. As it pertains to the Business or the Acquired Assets:
(i) each of Seller and its predecessors and Affiliates has complied and is in compliance, in each case in all material respects, with all Environmental, Health, and Safety Requirements;
(ii) without limiting the generality of the foregoing, each of Seller and its Affiliates, has obtained, has complied, and is in compliance with, in all material respects, all material permits, licenses and other authorizations that are required pursuant to Environmental, Health, and Safety Requirements for the occupation of its facilities and the operation of its business; a list of all such material permits, licenses and other authorizations is set forth in §3(z)(ii) of the Disclosure Schedule;
(iii) neither Seller nor its Affiliates has received any written or oral notice, report or other information regarding any actual or alleged violation of Environmental, Health, and Safety Requirements, or any liabilities or potential liabilities (whether accrued, absolute, contingent, unliquidated or otherwise), including any investigatory, remedial or corrective obligations, relating to any of them or its facilities arising under Environmental, Health, and Safety Requirements;
(iv) none of the following exists at any property or facility owned or operated by Seller: (1) underground storage tanks, (2) asbestos-containing material in any friable and damaged form or condition, (3) materials or equipment, including electric transformers, containing polychlorinated biphenyls, (4) landfills, surface impoundments, or disposal areas; (5) leach fields, dry wells and sumps, (6) waste water treatment, collection and disposal systems, including, without limitation, septic systems, seepage pits, lagoons, and dry wells, (7) landfarms, sprayfields, and incinerators, (8) non-contact cooling water discharge areas, (9) areas that may have received floodwater or stormwater runoff from potentially contaminated areas, (10) materials or equipment containing urea formaldehyde, (11) any contaminant, hazardous substance or hazardous material migrating from an off-site source, through the subsurface, including the soil and groundwater, onto Seller's property or facility, or (12) areas containing historical fill material including non-indigenous material, deposited to raise the topographic elevation of the site, which was contaminated prior to emplacement, and is in no way connected with the operations at the location of emplacement and which includes, without limitation, construction debris, dredge spoils, incinerator residue, demolition debris, fly ash, or non-hazardous solid waste.
(v) neither Seller nor any of its predecessors or Affiliates has treated, stored, disposed of, arranged for or permitted the disposal of, transported, handled, or released any substance, including without limitation any hazardous substance, or owned or operated any property or facility (and no such property or facility is contaminated by any such substance) in a manner that has given or would give rise to liabilities, including any liability for response costs, remedial or corrective action costs, personal injury, property damage, natural resources damages or attorney fees, pursuant to the Comprehensive Environmental Response, Compensation and Liability Act of 1980, as amended ("CERCLA") or the Solid Waste Disposal Act, as amended or any other Environmental, Health, and Safety Requirements; and
A-25
(vi) neither this Agreement nor the consummation of the transaction that is the subject of this Agreement will result in any obligations for site investigation or cleanup, or notification to or consent of government agencies or third parties, pursuant to any of the so-called "transaction-triggered" or "responsible property transfer" Environmental, Health, and Safety Requirements.
(aa) Certain Business Relationships with Seller's Affiliates. None of Seller's Affiliates, directors, officers, or employees has been involved in any material business arrangement or relationship regarding the Business or the Acquired Assets with Seller within the past 12 months, and none of Seller's Affiliates, directors, officers, or employees owns any material asset, tangible or intangible, which is used in the Business.
(bb) Solvency. As of the date of this Agreement, Seller is neither insolvent nor unable to pay its debts nor has made a general assignment with or for the benefit of its creditors, and no proceeding under any bankruptcy, insolvency or reorganization law has been commenced by or with respect to Seller. After consummation of the transactions contemplated by this Agreement, Seller will not be insolvent or unable to pay its debts.
(cc) UK Subsidiary. Seller represents and warrants to Buyer that the representations to Buyer with respect to Seller in §§3(a)-(e), §3(j)(i)-(ii), §3(t), and §3(bb) are true and correct with respect to the UK Subsidiary as if the UK Subsidiary had been named therein in place of Seller.
(dd) Disclosure. The definitive form of the Proxy Statement will comply with the Securities Exchange Act in all material respects. The definitive form of the Proxy Statement will not contain any untrue statement of a material fact or omit to state a material fact necessary in order to make the statements made therein, in the light of the circumstances under which they will be made, not misleading;provided,however, that Seller makes no representation or warranty with respect to any information that Buyer will supply specifically for use in the Proxy Statement.
(ee) Representations and Warranties of Buyer. The representations and warranties set forth in this Agreement and the Transaction Documents constitute the sole and exclusive representations and warranties of Buyer in connection with the transactions contemplated hereby. Except for the representations and warranties expressly set forth in this Agreement and the Transaction Documents, Seller disclaims reliance on any representations, warranties or guarantees, either express or implied, by or on behalf of Buyer including, without limitation, any oral, written or electronic response to any information request provided to Seller and acknowledges that any such reliance would be unreasonable.
§4. Buyer's Representations and Warranties. Buyer represents and warrants to Seller that the statements contained in this §4 are correct and complete as of the date of this Agreement and will be correct and complete as of the Closing Date (as though made then and as though the Closing Date were substituted for the date of this Agreement throughout this §4), except as set forth in the Disclosure Schedule. That portion of the Disclosure Schedule that pertains to Buyer will be arranged in paragraphs corresponding to the lettered and numbered paragraphs contained in this §4.
(a) Organization of Buyer. Buyer is a corporation duly organized, validly existing, and in good standing under the laws of the jurisdiction of its incorporation.
(b) Authorization of Transaction. Buyer has full power and authority (including full corporate or other entity power and authority) to execute and deliver this Agreement and to perform its obligations hereunder. This Agreement constitutes the valid and legally binding obligation of Buyer, enforceable in accordance with its terms and conditions subject, however, to the effects of bankruptcy, insolvency, reorganization, moratorium or similar laws affecting creditors' rights generally, and to general principles of equity (regardless of whether such enforceability is considered in a proceeding in equity or at law). The execution, delivery and performance of this Agreement and all other agreements contemplated hereby have been duly authorized by Buyer.
A-26
(c) Noncontravention. Neither the execution and the delivery of this Agreement, nor the consummation of the transactions contemplated hereby (including the assignments and assumptions referred to in §2 above), will (i) violate any constitution, statute, regulation, rule, injunction, judgment, order, decree, ruling, charge, or other restriction of any Governmental Authority, or court to which Buyer is subject or any provision of its charter, bylaws, or other governing documents or (ii) conflict with, result in a breach of, constitute a default under, result in the acceleration of, create in any party the right to accelerate, terminate, modify, or cancel, or require any notice under any agreement, contract, lease, license, instrument, or other arrangement to which Buyer is a party or by which it is bound or to which any of its assets is subject, except where the violation, conflict, breach, default, acceleration, termination, modification, cancellation, failure to give notice, or Lien would not have a Material Adverse Effect. Other than in connection with the provisions of the Hart-Scott-Rodino Act, the Delaware General Corporation Law, and the Securities Exchange Act or as contemplated by §4(c) of the Disclosure Schedule, Buyer does not need to give any notice to, make any filing with, or obtain any authorization, consent, or approval of any government or governmental agency in order for the Parties to consummate the transactions contemplated by this Agreement (including the assignments and assumptions referred to in §2 above), except where the failure to give notice, to file, or to obtain any authorization, consent, or approval would not have a Material Adverse Effect.
(d) Brokers' Fees. Buyer has no liability or obligation to pay any fees or commissions to any broker, finder, or agent with respect to the transactions contemplated by this Agreement for which Seller could become liable or obligated.
(e) Financing. Buyer has available to it cash or cash equivalents to provide Buyer with all of the financing required in pay the Closing Payment and the Abbott Payment and all related transaction expenses. Buyer expressly acknowledges that, in executing this Agreement, Seller is relying on Buyer's representation with regard to the availability of the necessary funds for the payment of the Closing Payment and the Abbott Payment upon Closing and that there is no condition precedent under this Agreement with regard to Buyer's ability to obtain financing.
(f) WARN Act. Buyer does not intend to engage within 60 days of the Closing Date in a "plant closing" or "mass layoff" as such terms are defined in the WARN Act.
(g) Disclosure. No information that Buyer will supply specifically for use in the Proxy Statement will contain any untrue statement of a material fact or omit to state a material fact necessary in order to make the statements made therein, in the light of the circumstances under which they will be made, not misleading.
(h) Representations and Warranties by Seller. The representations and warranties set forth in this Agreement and the Transaction Documents constitute the sole and exclusive representations and warranties of Seller in connection with the transactions contemplated hereby. There are no representations, warranties, covenants, understandings or agreements among the Parties regarding the Acquired Assets, the Assumed Liabilities or the Business or their transfer other than those contained in this Agreement and the Transaction Documents. Except for the representations and warranties expressly set forth in this Agreement and the Transaction Documents, Buyer disclaims reliance on any representations, warranties or guarantees, either express or implied, by or on behalf of Seller including, without limitation, any oral, written or electronic response to any information request provided to Buyer and acknowledges that any such reliance would be unreasonable. PRIOR TO THE EXECUTION OF THIS AGREEMENT, BUYER HAS CONDUCTED TO ITS SATISFACTION ALL NECESSARY AND SUFFICIENT EXAMINATION OF THE ACQUIRED ASSETS, ASSUMED LIABILITIES AND THE BUSINESS, AND IS RELYING ON ITS OWN EXAMINATION AND IS NOT RELYING ON ANY REPRESENTATION OR WARRANTY MADE BY SELLER (OTHER THAN THOSE SET FORTH IN THIS AGREEMENT AND THE
A-27
TRANSACTION DOCUMENTS) OR ANY PERSON PURPORTING TO ACT ON BEHALF OF SELLER.
§5. Pre-Closing Covenants. The Parties agree as follows with respect to the period between the execution of this Agreement and the Closing.
(a) General. Each of the Parties will use its reasonable best efforts to take all action and to do all things necessary in order to consummate and make effective the transactions contemplated by this Agreement (including satisfaction, but not waiver, of the Closing conditions set forth in §7), subject in the case of Seller to the exercise of the fiduciary duties of its board of directors.
(b) Notices and Consents. Seller will give any notices to third parties, and each of Seller and Buyer will use its commercially reasonable efforts to obtain any third party consents referred to in §3(c) above, the Lease Consents, and the items set forth in §5(b) of the Disclosure Schedule. Each of the Parties will give any notices to, make any filings with, and use its commercially reasonable efforts to obtain any authorizations, consents, and approvals of Governmental Authorities in connection with the matters referred to in §3(c) and §4(c) above. Without limiting the generality of the foregoing, each of the Parties will file any Notification and Report Forms and related material that it may be required to file with the Federal Trade Commission and the Antitrust Division of the United States Department of Justice under the Hart-Scott-Rodino Act, will use its commercially reasonable efforts to obtain a waiver from the applicable waiting period, and will make any further filings pursuant thereto that may be necessary in connection therewith.
(c) Operation of Business. Seller will not engage in any practice, take any action, or enter into any transaction outside the Ordinary Course of Business. Without limiting the generality of the foregoing, Seller will not with respect to the operation of the Business (i) pay any amount to any third party with respect to any liability or obligation (including any costs and expenses Seller or any of its Subsidiaries have incurred or may incur in connection with this Agreement and the transactions contemplated hereby) which would not constitute an Assumed Liability if in existence as of the Closing, or (ii) otherwise engage in any practice, take any action, or enter into any transaction of the sort described in §3(g) above.
(d) Preservation of Business. Seller will keep its business and properties substantially intact, including its present operations, physical facilities, working conditions, insurance policies and relationships with lessors, licensors, suppliers, customers, and employees. Seller shall not sell, distribute or otherwise dispose of any of the Acquired Assets, except for the sale of bulk active pharmaceutical ingredient Paclitaxel to Abbott in accordance with the terms and conditions of the Abbott Agreement, and to Buyer's Affiliates. Seller shall not sell or make any other disposition of its Inventory after the Final Inventory Measurement Date without the express written consent of Buyer.
(e) Full Access. Seller will permit representatives of Buyer (including legal counsel and accountants) to have full access at all reasonable times and with 24 hours prior written notice, and in a manner so as not to interfere with the normal business operations of Seller, to all premises, properties, personnel, books, records (including Tax records), contracts, and documents of or pertaining to Seller. Buyer will treat and hold as such any Confidential Information it receives from any of Seller and its Subsidiaries in the course of the reviews contemplated by this §5(e), will not use any of the Confidential Information except in connection with this Agreement, and, if this Agreement is terminated for any reason whatsoever, will return to Seller and its Subsidiaries all tangible embodiments (and all copies) of the Confidential Information which are in its possession.
(f) Notice of Developments. Each Party will give prompt notice to the other Party of any material adverse development causing a breach of any of its own representations and warranties in §3 and §4 above. Unless the Party receiving notice has the right to terminate this Agreement pursuant to §9(a) by reason of the development and exercises that right within the period of 10 Business Days referred to in §
A-28
9(a), the written notice pursuant to this §5(f) will amend the Disclosure Schedule to have qualified the representations and warranties contained in §3 and §4 above, and to have cured any misrepresentations or breach of warranty that otherwise might have existed hereunder by reason of the development.
(i) Seller agrees that neither it nor any of its Affiliates nor any of the officers and directors of it or any of its Affiliates will, and that it will use its best efforts to cause its and its Affiliates' employees, agents and representatives (including any investment banker, attorney or accountant retained by it or any of its Affiliates) not to, directly or indirectly (A) initiate or solicit any inquiries, or the making of any proposal or offer with respect to, (1) a merger, reorganization, share exchange, consolidation, business combination, recapitalization, liquidation, dissolution or similar transaction involving Seller or any of its Affiliates, (2) any purchase or sale of 33% or more of the consolidated assets (including, without limitation, stock or assets of Seller's Affiliates) of Seller and its Affiliates, taken as a whole, or (3) any purchase or sale of, or tender or exchange offer for, Seller's equity securities that, if consummated, would result in any Person (or the stockholders of such Person) beneficially owning securities representing in excess of 33% of the power to vote for the election of a majority of directors of Seller (any such proposal, offer or transaction (other than a proposal or offer made by Buyer or an Affiliate thereof) being referred to in this Agreement as an "Acquisition Proposal"), (B) have any discussion with or provide any confidential information or data to any Person relating to an Acquisition Proposal, (C) knowingly facilitate any effort or attempt to make or implement an Acquisition Proposal, (D) approve or recommend, or propose publicly to approve or recommend, any Acquisition Proposal, or (E) approve or recommend, or propose to approve or recommend, or execute or enter into, any letter of intent, agreement in principle, merger agreement, acquisition agreement, option agreement or other similar agreement or propose publicly or agree to do any of the foregoing related to any Acquisition Proposal.
(ii) Notwithstanding anything in this §5(g)(ii) to the contrary, (A) nothing in this Agreement shall restrict Seller from filing a Current Report on Form 8-K describing this Agreement and the transactions contemplated by this Agreement and by any other agreements being entered into by Seller on the date of this Agreement (which filing may include this Agreement as an exhibit) promptly after the date of this Agreement or from complying with its obligations under the Securities Act of 1933, as amended, the Securities Exchange Act and any other Law;provided, however, that such filing of Form 8-K shall comply with §6(d); (B) Seller's board of directors may authorize Seller to engage in discussions or negotiations with any Person who (without any solicitation or initiation, directly or indirectly, by Seller or any officer, director, employee, agent or representative of Seller (collectively, the "Representatives") after the date of this Agreement) seeks to initiate such discussions or negotiations and may furnish such Person information concerning and access to Seller and its Subsidiaries and their respective businesses, properties and assets, and Seller's board of directors may direct its Representatives to cooperate with and be available to consult with any such Person;provided that in the case of this clause (B), Seller's board of directors shall have determined in the exercise of its fiduciary duties that such action is in the best interests of Seller's Stockholders; (C) following receipt of an Acquisition Proposal that is financially superior to this Agreement (as determined in good faith by Seller's board of directors), Seller's board of directors may withdraw, modify or not make its recommendation in favor of this Agreement;provided that in the case of this clause (C), Seller's board of directors shall have concluded in good faith that such action is necessary in order for it to act in a manner that is consistent with its fiduciary obligations; and (D) Seller's board of directors may take and disclose to Seller Stockholders any position required under the Securities Exchange Act;provided that, in each case referred to in the foregoing clauses (B), (C) and (D), Seller shall not engage in negotiations with, or disclose any nonpublic information to, any Person unless it receives from such
A-29
Person an executed confidentiality agreement on terms and conditions deemed to be appropriate by Seller's board of directors and its counsel and financial advisors. Seller shall immediately cease and cause to be terminated any existing solicitation of, and any discussion or negotiation conducted prior to the date of this Agreement by it or any of its Representatives with respect to any Acquisition Proposal. Except to the extent Seller's board of directors deems it necessary not to do so in the exercise of its fiduciary obligations, Seller will promptly notify Buyer of the receipt of any Acquisition Proposal, including the identity of the Person or group making such Acquisition Proposal and the material terms and conditions of such Acquisition Proposal. Nothing in this §5(g)(ii) shall (x) permit Seller to terminate this Agreement (except as specifically provided in §9 of this Agreement) or (y) affect any other obligation of Seller under this Agreement.
(h) Maintenance of Leased Real Property. Seller will maintain the Leased Real Property, including all of the Improvements, in substantially the same condition as of the date of this Agreement, ordinary wear and tear excepted, and shall not demolish or remove any of the existing Improvements, or erect material improvements on the Leased Real Property or any portion thereof, without the prior written consent of Buyer.
(i) Leases. Seller will not cause or permit any Lease to be amended, modified, extended, renewed or terminated; nor shall Seller enter into any new lease, sublease, license or other agreement for the use or occupancy of any real property without the prior written consent of Buyer.
(i) Seller will as promptly as is reasonably practicable prepare and file with the SEC a preliminary Proxy Statement and related soliciting materials under the Securities Exchange Act relating to the Special Meeting; provided, however, prior to filing or mailing of the Proxy Statement, any supplement or amendment thereto, and any correspondence with the SEC, Seller will use its best efforts to respond to the comments of SEC thereon and will make any further filings (including amendments and supplements) in connection therewith that may be necessary. Buyer will provide Seller with whatever information and assistance in connection with the foregoing filings that Seller reasonably may request. Seller shall cause the Proxy Statement and related materials to comply as to form and substance in all material respects with Law, including without limitation the Securities Exchange Act, and the rules and regulations of Nasdaq.
(ii) Seller will notify Buyer promptly upon the receipt of any comments from the SEC or its staff or any other government officials in connection with any filing made pursuant to this §5(j) and of any request by the SEC or its staff or any other government officials for amendments, revisions or supplements to the Proxy Statement or other SEC filings, or for additional information made pursuant hereto. Seller will supply Buyer with copies of all correspondence between Seller or any of its representatives, on the one hand, and the SEC, or its staff or any other government officials, on the other hand, with respect to Seller's Proxy Statement, the transactions contemplated by this Agreement, the Transactional Documents, or any other filing with the SEC related to the transactions contemplated by this Agreement. Seller will cause all documents that it is filing with the SEC or other regulatory authorities under this §5(j) to comply in all material respects with all Law.
(k) Special Stockholders' Meeting. Promptly after the date of this Agreement, Seller will take all action necessary in accordance with Law, Seller's certificate of incorporation and bylaws, each as amended to date, and all contracts binding on Seller to give notice of a special meeting of its stockholders (the "Special Meeting"), as soon as reasonably practicable and in any event (to the extent permissible under Law) within 45 days after the clearance by the SEC of the preliminary Proxy Statement in order that the Seller Stockholders may consider and vote upon the adoption of a resolution approving the transactions contemplated by this Agreement in accordance with the Delaware General Corporation Law. Seller will mail the Proxy Statement to its stockholders as soon as
A-30
reasonably practicable. Subject to the fiduciary duty of Seller's directors and Law, Seller will use its reasonable best efforts to solicit from its stockholders proxies in favor of the adoption and approval of this Agreement and the approval of the transactions contemplated hereby, and will take all other action necessary to secure the approval of its stockholders (by vote or consent) required by the rules of the Nasdaq Stock Market and Law, Seller's certificate of incorporation and bylaws, each as amended to date, and all Contracts binding on Seller. The Proxy Statement will contain the affirmative recommendation of the board of directors of Seller in favor of the adoption of a resolution approving the transactions contemplated by this Agreement;provided,however, that no director or officer of Seller shall be required to violate any fiduciary duty or other requirement imposed by law in connection therewith. Seller will convene the Special Meeting as set forth in the Proxy Statement as soon as practicable in accordance with Seller's bylaws and the rules of the Nasdaq Stock Market and Law.
(l) Fairness Opinion. Seller will deliver to Buyer on or before the date the Proxy Statement is mailed to the stockholders of Seller a copy of the opinion of Wells Fargo Securities, LLC, dated as of the date of this Agreement and addressed to the Board of Directors of Seller, as to the fairness of the Purchase Price to Seller from a financial point of view (the "Fairness Opinion").
(m) Transfer of Assets of Cayman Subsidiary. Seller shall take all actions necessary to cause the Cayman Subsidiary to transfer all assets that are used in the Business and owned or otherwise held by the Cayman Subsidiary to be sold or transferred, as the case may be, to Seller prior to the Closing.
(n) Confidentiality. The Parties will comply in all respects with the terms of Section 6(d).
(o) Transfer of UK Subsidiary. Buyer will elect not less than thirty (30) days prior to the Closing Date whether to purchase the stock or assets of the UK Subsidiary.
(p) Assistance with European Regulatory Matters. Prior to the Closing, Seller shall from time to time and at the request of Buyer consult with Buyer in good faith with respect to Seller's European regulatory dossier, including distribution rights in European countries and the treatment of the Finnish market authorization.
(q) Information System Transition. Promptly after the date of this Agreement the Parties will begin taking the steps to accomplish the transfer of the Ross Computer ERP System and the information system from Seller to Buyer in an orderly manner, which shall include Seller dedicating time of its internal information technology professionals to separating and testing the separate servers. The steps to be taken will include (w) the transfer of the Ross Agreement from Seller to Buyer with consent of Ross Systems, Inc.; (x) configuring and implementing Seller's present production hardware, software and data for Buyer's use in running the Business after the Closing Date; (y) configuring and implementing the back-up server, software and data for Seller's use after the Closing; and (z) accomplishing appropriate testing and a cutover for both Buyer and Seller concurrent with Closing. Buyer, at its expense, will (i) direct Ross Systems, Inc. to perform the support tasks necessary to assist in accomplishing the described transfer, (ii) implement an e-mail system for its use after the Closing Date, (iii) upgrade the hardware and software of the Sterling Drive Facility's file server (NAPRO_STERLING) if required, (iv) implement its own Internet provider, (v) perform validation testing, (v) add another domain controller if desired, and (vi) implement its own FAX facility and appropriate network security.
(r) Allocation of Purchase Price. Not later than five (5) days before the Closing Date, Buyer shall provide to Seller an allocation of the Purchase Price among the Acquired Assets for all purposes (including financial accounting and tax purposes).
(s) Voting Assurances. Within five (5) Business Days after the date of this Agreement, Buyer and each of Leonard P. Shaykin and Sterling K. Ainsworth shall enter into a voting agreement substantially in the form heretofore provided to Buyer.
A-31
(t) Amendment of Certain Agreements. Seller shall not amend the Avoca Agreement or the Abbott Termination Agreement in any respect without the prior written consent of Buyer.
§6. Post-Closing Covenants. The Parties agree as follows with respect to the period following the Closing.
(a) General. In case at any time after the Closing any further action is necessary to carry out the purposes of this Agreement, each of the Parties will take such further action (including the execution and delivery of such further instruments and documents) as the other Party reasonably may request, all at the sole cost and expense of the requesting Party (unless the requesting Party is entitled to indemnification therefor under §8). Without limitation of the foregoing, upon the reasonable request of Buyer (or its assigns) and at Buyer's expense, Seller will promptly make available, and will direct its third party agents to make available, to Buyer (or its assigns) all pertinent facts and documents reasonably relating to the Intellectual Property transferred to Buyer pursuant to this Agreement as may be known and accessible to Seller, and Seller will promptly execute and deliver to Buyer or its legal representative any and all papers, instruments or affidavits required to apply for, obtain, maintain, issue, enforce and perfect the rights, title, and interest in the Intellectual Property transferred pursuant to this Agreement, in all countries.
(b) Litigation Support. In the event and for so long as any Party actively is contesting or defending against any action, suit, proceeding, hearing, investigation, charge, complaint, claim, or demand in connection with (i) any transaction contemplated under this Agreement or (ii) any fact, situation, circumstance, status, condition, activity, practice, plan, occurrence, event, incident, action, failure to act, or transaction on or prior to the Closing Date involving Seller, the other Party will cooperate with the contesting or defending Party and its counsel in the contest or defense, make available its personnel, and provide access to its books and records as shall be necessary in connection with the contest or defense, all at the sole cost and expense of the contesting or defending Party (unless the contesting or defending Party is entitled to indemnification therefore under §8). Notwithstanding anything to the contrary in this Agreement, Seller hereby acknowledges and agrees that Buyer is under no obligation to contest or defend against any action, suit, proceeding, hearing, investigation, charge, complaint, claim, or demand involving Seller, including, without limitation, the Mylan Litigation and the proceedings described on §3(t) of the Disclosure Schedule and that Seller shall have no recourse or claim if Buyer elects not to contest or defend any such claim,provided, however, that nothing in this §6(b) shall affect Seller's rights under §8(c)(ii).
(c) Transition. Seller will not take any action that is designed or intended to have the effect of discouraging any lessor, licensor, customer, supplier, or other business associate of Seller from maintaining the same business relationships with Buyer after the Closing as it maintained with Seller prior to the Closing and will use its commercially reasonable efforts to cooperate with Buyer to transition such relationship to Buyer, provided that Seller will not be obligated to bear any out of pocket expense associated with such transition.
(d) Confidentiality. Except as (i) expressly permitted under this Agreement or (ii) in connection with the operation of [*], Seller will treat and hold as such all of the Confidential Information, refrain from using any of the Confidential Information, and deliver promptly to Buyer or destroy, at the request and option of Buyer, all tangible embodiments (and all copies) of the Confidential Information which are in its possession;provided, however, that Seller shall obtain the prior written consent of Buyer, which shall not be unreasonably, withheld, conditioned or delayed, before disclosing the Polypro Process or the Semi-synthetic Process to any third party, andfurther provided that not such consent will be required if Seller obtains the agreement of any such third party to abide by the restrictions on use of such processes as they apply to Seller under this Agreement. In the event that Seller is requested or required (by oral question or request for information or documents in any legal proceeding, interrogatory, subpoena, civil investigative demand, or similar process) to disclose any Confidential
A-32
Information, Seller will notify Buyer promptly of the request or requirement so that Buyer may seek an appropriate protective order or waive compliance with the provisions of this §6(d). If, in the absence of a protective order or the receipt of a waiver hereunder, Seller is, on the advice of counsel, compelled to disclose any Confidential Information to any tribunal or else stand liable for contempt, Seller may disclose the Confidential Information to the tribunal;provided, however, that Seller shall use its reasonable best efforts to obtain, at the reasonable request of Buyer, an order or other assurance that confidential treatment will be accorded to such portion of the Confidential Information required to be disclosed as Buyer shall designate. Notwithstanding the foregoing, any Party and its employees, representatives and other agents are authorized to disclose the tax structure or tax consequences of the transaction contemplated by this Agreement. Buyer agrees, in connection with Seller's operation of [*], to submit a "letter of access" to the FDA and other Governmental Authorities designated by Seller granting Seller the right of access to Confidential Information transferred to Buyer as part of the transactions contemplated by this Agreement. Without limitation of the foregoing, each of Buyer and Seller agrees that the terms of this Agreement and the Transaction Documents shall not be disclosed or otherwise made available to the public and that copies of this Agreement and the Transaction Documents shall not be publicly filed or otherwise made available to the public, except where such disclosure, availability or filing is required by Law and only to the extent required by such Law. In the event that such disclosure, availability or filing is required by the Laws of the U.S. Securities and Exchange Commission (or other Governmental Authority), each of Buyer and Seller (as applicable) agrees to use commercially reasonable efforts to obtain "confidential treatment" of those portions of this Agreement and the Transaction Documents designated by the other as confidential with the U.S. Securities and Exchange Commission and any other applicable Governmental Authority (or the equivalent treatment by any other Governmental Entity) and to redact such terms of this Agreement and the Transaction Documents as the other party shall request. In furtherance of the foregoing, the parties agree to consult with each other in good faith in the preparation of any confidential treatment application and the redacted versions of this Agreement and any applicable Transaction Documents. In addition, notwithstanding anything to the contrary herein, either Party (and any employee, representative or other agent of such Party) may disclose to any and all Persons, without limitation of any kind, the tax treatment and tax structure of the transactions contemplated by this Agreement and all materials of any kind (including opinions or other tax analyses) that are provided to it relating to such tax treatment and tax structure. For this purpose, (i) tax treatment and tax structure shall not include the identity of any existing or future Party (or any affiliate of such Party) and (ii) disclosure of any information shall not be permitted to the extent that nondisclosure is necessary in order to comply with applicable securities laws.
(e) Covenant Not to Compete. For a period of five years from and after the Closing Date, Seller will not manufacture, use or sell bulk active pharmaceutical ingredient Paclitaxel for use in formulation of Paclitaxel formulated in polyethoxylated castor oil (whether including further components or not), including, without limitation, Paclitaxel formulated in polyethoxylated castor oil and ethanol; [*]. For a period of three years from and after the Closing Date, Seller will not manufacture and/or supply Paclitaxel using the Polypro Process or the Semi-synthetic Process for commercial purposes;provided, however, that Seller has the right to use the Polypro Process and the Semi-synthetic Process for non-commercial purposes in accordance with the Intellectual Property Agreement. With respect to this covenant not to compete, (i) no owner of less than 1% of the outstanding stock of any publicly traded corporation shall be deemed to engage solely by reason thereof in any of its businesses and (ii) no activity of Seller permitted or contemplated by Exhibit A shall be prohibited by this §6(e). If the final judgment of a court of competent jurisdiction declares that any term or provision of this §6(e) is invalid or unenforceable, the Parties agree that the court making the determination of invalidity or unenforceability shall have the power to reduce the scope, duration, or area of the term or provision, to delete specific words or phrases, or to replace any invalid or unenforceable term or provision with a term or provision that is valid and enforceable and that comes closest to expressing the intention of the
A-33
invalid or unenforceable term or provision, and this Agreement shall be enforceable as so modified after the expiration of the time within which the judgment may be appealed.
(f) Books and Records. On or before 15 days after the Closing Date, Seller will deliver or cause to be delivered to Buyer, at Buyer's request all files, records, information and data relating to the Business and the Acquired Assets that are in the possession or control of Seller and its Subsidiaries (the "Records"), except for such Records Seller is entitled to keep under the terms of Exhibit A. Buyer agrees (a) to hold the Records and not to destroy or dispose of any portion thereof for a period of four years from the Closing Date or such longer time as may be required by applicable law,provided that, if it desires to destroy or dispose of such Records during such period, it will first offer in writing at least 60 days before such destruction or disposition to surrender them to Seller and if Seller does not accept such offer within 20 days after receipt of such offer, Buyer may take such action and (b) following the Closing Date to afford Seller, its accountants, and counsel, during normal business hours, upon reasonable request, at any time, full access to the Records and to Buyer's employees at no cost to Seller (other than for reasonable out-of-pocket expenses);provided that such access will not be construed to require the disclosure of Records that would cause the waiver of any attorney-client, work product or like privilege;provided,further, that in the event of any litigation nothing in this Agreement shall limit any Party's rights of discovery under applicable law. Buyer agrees to provide Seller and its Affiliates reasonable access to, and the right to make copies of, the Records after the Closing Date in order for Seller to comply with its obligations under this Agreement, to prepare any tax returns and to prepare any filings with the SEC.
(i) §6(g)(i) of the Disclosure Schedule contains a true and correct list of the name, job title, classification, current base salary or hourly wage, date of hire, current vacation entitlement and assigned location of all employees of Seller (the "Employees") actively employed in the Business as of the date of this Agreement, including any such individual on short-term disability or approved leave of absence who was so employed immediately before such disability or absence, whose employment with Seller will terminate upon Closing. At the Closing, Seller shall provide to Buyer an updated §6(g)(i) of the Disclosure Schedule which shall disclose all the information required under the preceding sentence as of the day prior to Closing. All individuals included on the original §6(g)(i) of the Disclosure Schedule plus all individuals included on the updated §6(g)(i) of the Disclosure Schedule whose hiring was approved by Buyer, are herein referred to as the "Seller Employees." With respect to any Seller Employee who is not actively employed due to short-term disability or approved leave of absence, the updated §6(g)(i) of the Disclosure Schedule shall indicate the reason for such absence and the date such individual is reasonably expected to return to active employment. If a Seller Employee who is not actively employed at the time of Closing due to short-term disability or approved leave of absence is, in the reasonable opinion of Buyer, unfit to return to active employment with Buyer within 30 days of the expected return date indicated on the updated §6(g)(i) of the Disclosure Schedule or otherwise does not commence active employment with Buyer within such 30 day period, such individual shall be considered a "Former Seller Employee" for all purposes under this Agreement.
(ii) Effective as of 12:01 a.m. on the Closing Date, Seller shall cause the employment of all Seller Employees (other than those individuals who are not actively employed due to short-term disability or approved leave of absence, whose employment shall be terminated upon their return to active employment) to be terminated. Effective as of 12:01 a.m. on the Closing Date, Buyer intends to offer employment to all Seller Employees terminated in accordance with the preceding sentence (except that with respect to those individuals on short-term disability or an approved leave of absence, subject to their commencement of active employment with Buyer within 30 days of their expected return date as indicated on updated §6(g)(i) of the Disclosure Schedule, Buyer intends to offer employment to such individuals upon their return from short-term disability or
A-34
approved leave of absence) on terms and conditions, including salary and benefits, in conformance with Buyer's policies in effect for Buyer's employees at the Closing Date. All Seller Employees to whom Buyer offers employment and who accept such employment are herein referred to as the "Transferred Employees." Nothing in this §6(g) shall limit Buyer's authority to terminate the employment of any Transferred Employee at any time and for whatever reason. Beginning on the first Business Day following the execution of this Agreement, Seller shall provide or make available to Buyer, to the extent permitted by Law, such information regarding the Transferred Employees as is contained in Seller's personnel records, including without limitation information regarding accrued or incurred but unpaid liabilities for wages, vacations, deferred compensation, medical/dental/vision, workers' compensation, disability and other welfare benefit claims.
(iii) Seller shall be responsible for the payment of any Seller Employee benefits that are or become due to any Transferred Employees as a result of their termination by Seller in accordance with §6(g), including, without limitation, all accrued vacation, sick and severance pay, if any, as well as any amounts that may be payable to any Seller Employee under Seller's 401K plan. All such Transferred Employees shall receive credit for all prior service with Seller for purposes of participation in Buyer's employee benefit plans.
(iv) Seller shall be responsible for all legally mandated health care continuation coverage for Seller Employees and Former Seller Employees and their covered dependents who had or have a loss of coverage due to a "qualifying event" (within the meaning of Section 603 of ERISA) which occurred or occurs on or prior to the Closing Date including without limitation, any loss of coverage that results directly or indirectly from the transaction contemplated by this Agreement.
(v) Seller shall retain liability for payment of any long-term or short-term disability benefits to any Seller Employee or Former Seller Employee that relate to a disability which was first disclosed or arose prior to the Closing Date.
(vi) Seller shall retain liability under all health, medical, accidental death and disability life insurance and any other welfare and benefit plans, programs or policies of Seller accrued prior to the Closing Date.
(vii) Seller and Buyer hereby acknowledge and agree that in conformity with the Standard Procedure of IRS Revenue Procedure 96-60, 1996-2 C.B. 399, (i) Seller will be responsible for and perform all Tax withholding, payment and reporting duties with respect to any wages and other compensation paid by Seller to any Seller Employee in connection with employment on or prior to the Closing Date; and (ii) Buyer will be responsible for and perform all Tax withholding, payment, and reporting duties with respect to any wages and other compensation paid by Buyer to any Transferred Employee in connection with employment after the Closing Date.
(viii) Seller and Buyer hereby acknowledge and agree that (A) in accordance with §2101(b)(i) of the WARN Act, Seller will be responsible for any Seller plant closing or mass layoffs that take place prior to the Closing Date, and Buyer will be responsible for any Buyer plant closing or mass layoffs that take place on or after the Closing Date, and (B) all of Seller Employees as of the Closing Date (other than those individuals who are not actively employed on the Closing Date due to short-term disability or approved leave of absence) will be deemed to have become Transferred Employees of Buyer immediately on the Closing Date for purposes of the WARN Act. Buyer shall be liable for any WARN Act liability as to any Seller Employee not hired by Buyer.
(ix) Seller hereby covenants and agrees that during the three (3) year period following the Closing Date Seller shall not, and shall use its commercially reasonable efforts to cause its directors, officers and employees not to, directly or indirectly, either alone or as a stockholder, partner, consultant, LLC member, creditor, director, officer or LLC manager of any Person, or in
A-35
any other capacity, (A) solicit, induce or attempt to induce any employee of Buyer (including, without limitation, any Seller Employee) to leave the employ of Buyer; (B) interfere in any way with the relationship between Buyer and any of its employees; or (C) hire any person who is a Former Seller Employee or who is or was an employee of Buyer (including, without limitation, any Seller Employee), in each case unless six (6) months, shall have elapsed since the latest date such employee was an employee of Seller or Buyer, as the case may be, or unless the termination of such person's employment with Buyer was involuntary, without the prior written consent of Buyer, which consent shall not be unreasonably conditioned, withheld or delayed. Buyer hereby covenants and agrees that, except for the Transferred Employees, during the three (3) year period following the Closing Date, Buyer shall not, and shall use its commercially reasonable efforts to cause its directors, officers and employees, directly or indirectly, either alone or as a stockholder, partner, consultant, LLC member, creditor, director, officer or LLC manager of any Person, or in any other capacity, not to solicit, induce or attempt to induce any employee of Seller (including, without limitation, any Seller Employee) to leave the employ of Seller or (B) interfere in any way with the relationship between Seller and any of its employees, without the prior written consent of Seller, which consent shall not be unreasonably conditioned, withheld or delayed.
(h) Use of Names. Seller hereby grants to Buyer a non-exclusive, fully paid-up and royalty-free, right and license under the Names solely to the extent necessary to allow Buyer and its Affiliates and their designees to manufacture, package, use and sell the Inventory (including packaging used or available for use with the Inventory) and finished goods using the Inventory existing as of the Closing Date (whether in the possession of Buyer, a distributor or any other third party) and bearing the name or any logo, mark, sign or other label of Seller;provided, however, that such grant shall expire the earlier of: (i) the date two years after the Closing Date; or (ii) the date on which the Inventory (including packaging used or available for use with the Inventory) and finished goods using the Inventory bearing Seller's name, logo, mark, sign or other label has been depleted. Buyer will, and will cause its Affiliates to, use and sell the Inventory bearing the name or any logo, mark, sign, or other label of Seller prior to using or selling comparable inventory or product not bearing Seller's name, logo, mark, sign or other label.
(i) Collection of Accounts Receivable. Buyer shall, by letter prepared by Seller and reasonably acceptable to Buyer (the "Letter"), authorize, instruct and direct that the account parties of all accounts, receivables, and insurance proceeds (such parties, the "Account Parties") that exist immediately prior to the Closing shall make and deliver all payments relating thereto to such location, bank and account (the "Seller Receivables Account") as Seller shall specify. The Letter shall cover all such matters as Buyer and Seller shall reasonably determine. If, notwithstanding such Letter, any of the Account Parties remit payments on or after the Closing directly or indirectly to Buyer instead of to the Seller Receivables Account, Buyer shall promptly deliver all such payments (including but not limited to negotiable instruments which shall be duly endorsed by Buyer to the order of Seller) to Seller. Likewise, if Seller at any time receives any payment relating to accounts receivable arising on or after the Closing with respect to the Business, Seller shall promptly deliver all such payments (including but not limited to negotiable instruments which shall be duly endorsed by Seller to the order of Buyer) to Buyer.
(j) Supply Agreement. Buyer and Seller will negotiate in good faith the terms of a mutually acceptable supply agreement (the "Supply Agreement") to be entered into, if at all, effective as of the Closing Date that will provide for the purchase by Seller from Buyer and the sale by Buyer to Seller of bulk active pharmaceutical ingredient Paclitaxel on the prices and in the amounts set forth in the Supply Agreement, it being understood that neither Party is obligated to enter into such an agreement.
A-36
§7. Conditions to Obligation to Close.
(a) Conditions to Buyer's Obligation. The obligation of Buyer to consummate the transactions to be performed by it in connection with the Closing is subject to satisfaction of the following conditions:
(i) the representations and warranties set forth in §3 above shall be true and correct in all material respects at and as of the Closing Date, except to the extent that such representations and warranties are qualified by terms such as "material" and "Material Adverse Effect," in which case such representations and warranties shall be true and correct in all respects at and as of the Closing Date;
(ii) Seller shall have performed and complied with all of its covenants hereunder in all material respects through the Closing, except to the extent that such covenants are qualified by terms such as "material" and "Material Adverse Effect," in which case Seller shall have performed and complied with all of such covenants in all respects through the Closing;
(iii) Seller shall have procured all of the third party consents specified in §5(b) above;
(iv) there shall not be any injunction, judgment, order, decree, ruling, or charge in effect preventing consummation of any of the transactions contemplated by this Agreement;
(v) Seller shall have delivered to Buyer a certificate to the effect that each of the conditions specified above in §7(a)(i)-(v) is satisfied in all respects;
(vi) all applicable waiting periods (and any extensions thereof) under the Hart-Scott-Rodino Act shall have expired or otherwise been terminated, and Seller and Buyer shall have received all other material authorizations, consents, and approvals of governments and governmental agencies referred to in §3(c) and §4(c) above;
(vii) all actions to be taken by Seller in connection with consummation of the transactions contemplated hereby and all certificates, opinions, instruments, and other documents required to effect the transactions contemplated hereby will be reasonably satisfactory in form and substance to Buyer;
(viii) Seller shall have obtained and delivered to Buyer a written consent for the assignment of each of the Leases (the "Lease Consents"), in form and substance satisfactory to Buyer and Buyer's lender, except where the failure to obtain any such Lease Consent shall not have a Material Adverse Effect;
(ix) Seller shall have obtained and delivered to Buyer an estoppel certificate with respect to each of the Leases that relate to Leased Real Property, dated no more than 30 days prior to the Closing Date, from the other party to such Lease, in form and substance satisfactory to Buyer (the "Estoppel Certificates"), except where the failure to obtain any such Estoppel Certificate shall not have a Material Adverse Effect;
(x) no damage or destruction or other change has occurred with respect to any of the Leased Real Property or any portion thereof that, individually or in the aggregate, would materially impair the use or occupancy of the Leased Real Property or the operation of the Business;
(xi) Seller shall have delivered to Buyer a copy of its certificate of incorporation certified on or soon before the Closing Date by the Secretary of State of the State of Delaware;
(xii) Seller shall have delivered to Buyer a copy of a certificate of good standing issued on or soon before the Closing Date by the Secretary of State of the State of Delaware;
(xiii) Seller shall have delivered to Buyer a copy of a certificate of good standing issued on or soon before the Closing Date by the Secretary of State of the State of Colorado;
A-37
(xiv) Seller shall have paid all amounts accruing and/or owed to the Seller Employees that would become payable after the Closing Date with respect to services provided prior to the Closing Date, including, without limitation, any retention bonuses or bonuses that are contingent upon the success of the transactions contemplated by this Agreement;
(xv) neither the Avoca Agreement nor the Abbott Termination Agreement shall have been amended in any respect without the prior written consent of Buyer
(xvi) a resolution approving the transactions contemplated by this Agreement shall have been adopted by the Requisite Stockholder Approval;
(xvii) Seller shall have delivered to Buyer a certificate of the secretary or an assistant secretary of Seller, dated the Closing Date, in form and substance reasonably satisfactory to Buyer, as to (i) no amendments to the Certificate of Incorporation of Seller since the date specified in clause (xi) above; (ii) the bylaws of Seller; (iii) the resolutions of the board of directors (or a duly authorized committee thereof) of Seller authorizing the execution, delivery, and performance of this Agreement and the transactions contemplated hereby; and (iv) incumbency and signatures of the officers of Seller executing this Agreement or any other agreement contemplated by this Agreement; and
(xviii) if the portions of the ChromaDex Contract relating to the Business are not assigned to Buyer on terms and conditions satisfactory to Buyer, then Buyer and Seller shall have entered into an agreement or agreements mutually satisfactory to Buyer and Seller with respect to services to be provided by ChromaDex, Inc. to each of Buyer and Seller, respectively, after the Closing.
(xix) Buyer and Seller shall have entered into a Transition Services Agreement consistent with the term sheet attached hereto as Exhibit B.
Buyer may waive any condition specified in this §7(a) if it executes a writing so stating at or prior to the Closing.
(b) Conditions to Seller's Obligation. The obligation of Seller to consummate the transactions to be performed by it in connection with the Closing is subject to satisfaction of the following conditions:
(i) the representations and warranties set forth in §4 above shall be true and correct in all material respects at and as of the Closing Date, except to the extent that such representations and warranties are qualified by terms such as "material" and "Material Adverse Effect," in which case such representations and warranties shall be true and correct in all respects at and as of the Closing Date;
(ii) Buyer shall have performed and complied with all of its covenants hereunder in all material respects through the Closing, except to the extent that such covenants are qualified by terms such as "material" and "Material Adverse Effect," in which case Buyer shall have performed and complied with all of such covenants in all respects through the Closing;
(iii) there shall not be any injunction, judgment, order, decree, ruling, or charge in effect preventing consummation of any of the transactions contemplated by this Agreement;
(iv) Buyer shall have delivered to Seller a certificate to the effect that each of the conditions specified above in §7(b)(i)-(iii) is satisfied in all respects;
(v) a resolution approving the transactions contemplated by this Agreement shall have been adopted by the Requisite Stockholder Approval;
(vi) the Fairness Opinion shall not have been withdrawn or modified in any material respect not acceptable to the board of directors of Seller;
A-38
(vii) all applicable waiting periods (and any extensions thereof) under the Hart- Scott-Rodino Act shall have expired or otherwise been terminated and Seller and Buyer shall have received all other material authorizations, consents, and approvals of governments and governmental agencies referred to in §3(c) and §4(c) above;
(viii) Seller and Abbott shall have entered in the Abbott Termination Agreement;
(ix) Seller and Buyer shall have entered into the Faulding Termination Agreement;
(x) Buyer shall have delivered to Seller a copy of a certificate of good standing certified on or soon before the Closing Date by the Secretary of State (or comparable officer) of the jurisdiction of its incorporation;
(xi) Buyer shall have delivered to Seller a certificate of the secretary or an assistant secretary of Buyer, dated the Closing Date, in form and substance reasonably satisfactory to Seller, as to (A) the resolutions of the board of directors of Buyer authorizing the execution, delivery, and performance of this Agreement and the transactions contemplated hereby; and (B) incumbency and signatures of the officers of Buyer executing this Agreement or any other agreement contemplated by this Agreement;
(xii) if the portions of the ChromaDex Contract relating to the Business are not assigned to Buyer on terms and conditions satisfactory to Buyer, then Buyer and Seller shall have entered into an agreement or agreements mutually satisfactory to Buyer and Seller with respect to services to be provided by ChromaDex, Inc. to each of Buyer and Seller, respectively, after the Closing; and
(xiii) all actions to be taken by Buyer in connection with consummation of the transactions contemplated hereby and all certificates, opinions, instruments, and other documents required to effect the transactions contemplated hereby will be reasonably satisfactory in form and substance to Seller. Seller may waive any condition specified in this §7(b) if it executes a writing so stating at or prior to the Closing.
(ix) Buyer and Seller shall have entered into a Transition Services Agreement consistent with the term sheet attached hereto as Exhibit B.
§8. Remedies for Breaches of This Agreement.
(a) Survival of Representations and Warranties. All of the representations and warranties of Seller contained in §3(f)-(i) and §§3(k) and (m)-(cc) above shall survive the Closing and continue in full force and effect for a period of eighteen months thereafter;provided, however, that the representations and warranties contained in §§3(l) and (z) above shall survive the Closing and continue in full force and effect for a period of three years thereafter. All of the other representations and warranties of the Parties contained in this Agreement (including the representations and warranties of Seller contained in §3(a)-(e) and §3(j) above) shall survive the Closing and continue in full force and effect forever thereafter (subject to any applicable statutes of limitations).
(b) Indemnification Provisions for Buyer's Benefit.
(i) In the event Seller breaches any of its representations, warranties, and covenants contained in this Agreement within the survival period (if there is an applicable survival period pursuant to §8(a) above), and, provided that Buyer makes a written claim for indemnification against Seller pursuant to §10(g), then Seller agrees to indemnify and hold harmless Buyer from and against the entirety of any Adverse Consequences Buyer may suffer (including any Adverse Consequences Buyer may suffer after the end of any applicable survival period) resulting from, arising out of, relating to, in the nature of, or caused by Seller's breach;provided, however, that (A) Seller shall not have any obligation to indemnify or hold harmless Buyer from and against any Adverse Consequences resulting from, arising out of, relating to, in the nature of, or caused by
A-39
Seller's breach of any representation or warranty contained in §3(f)-(cc) above unless the aggregate of all such amounts exceeds [*] (the "Minimum") in the aggregate, and then only to the full extent of any such amount in excess of the Minimum; and (B) there will be an aggregate ceiling of [*] on the obligation of Seller to indemnify and hold harmless Buyer from and against Adverse Consequences resulting from, arising out of, relating to, in the nature of, or caused by breaches of the representations and warranties of Seller contained in §3(f)-(cc) above.
(ii) Seller agrees to indemnify Buyer from and against the entirety of any Adverse Consequences Buyer may suffer resulting from, arising out of, relating to, in the nature of, or caused by any liability of Seller which is not an Assumed Liability (including any liability of Seller that becomes a liability of Buyer under any bulk transfer law of any jurisdiction, under any common law doctrine of de facto merger or successor liability, or otherwise by operation of Law);
(c) Indemnification Provisions for Seller's Benefit.
(i) In the event Buyer breaches any of its representations, warranties, and covenants contained in this Agreement, and, provided that Seller makes a written claim for indemnification against Buyer pursuant to §10(g) within that survival period (if there is an applicable survival period pursuant to §8(a) above), then Buyer agrees to indemnify Seller from and against the entirety of any Adverse Consequences suffered (including any Adverse Consequences suffered after the end of any applicable survival period) resulting from, arising out of, relating to, in the nature of, or caused by the breach.
(ii) Buyer agrees to indemnify Seller from and against the entirety of (A) any Adverse Consequences suffered by Seller resulting from, arising out of, relating to, in the nature of, or caused by any Assumed Liability and (B) any Adverse Consequences suffered by Seller arising with respect to any third party claim regarding the Mylan Litigation. Except as provided by §10(q), Buyer shall not be responsible for any attorneys' fees or other costs incurred by Seller prior to the Closing Date. After the Closing Date, Buyer shall only be responsible for costs, including attorneys' fees, of maintaining the Mylan Litigation if the Closing occurs and if Buyer elects to continue prosecuting the Mylan Litigation in its sole discretion.
(iii) Notwithstanding anything to the contrary herein, Buyer shall not have any obligation to indemnify Seller from and against any Adverse Consequences resulting from, arising out of, relating to, in the nature of, or caused by Buyer's breach of any representation or warranty of Buyer contained in §4 unless the aggregate of all such amounts exceeds the Minimum in the aggregate and then only to the full extent any such amount is in excess of the Minimum.
(d) Matters Involving Third Parties.
(i) If any third party notifies either Party (the "Indemnified Party") with respect to a Third Party Claim which may give rise to a claim for indemnification by the Indemnified Party against the other Party (the "Indemnifying Party") under this §8, then the Indemnified Party shall promptly provide a written Claim for Indemnification to the Indemnifying Party;provided, however, that no delay on the part of the Indemnified Party in notifying the Indemnifying Party shall relieve the Indemnifying Party from any obligation hereunder but as a result of such delay, the Indemnified Party shall not be liable to the Indemnifying Party for the amount of damages caused by such delay.
(ii) The Indemnifying Party shall have the right to assume the defense of the Third Party Claim with counsel of its choice reasonably satisfactory to the Indemnified Party at any time within 15 days after the Indemnified Party has given notice of the Third Party Claim;provided, however, that the Indemnifying Party must conduct the defense of the Third Party Claim actively and diligently thereafter in order to preserve the Indemnified Party's rights in this regard; andprovided further that the Indemnified Party may retain separate co counsel at its sole cost and expense and
A-40
participate in the defense of the Third Party Claim. Failure to timely respond shall constitute a final and binding acceptance of the Claim for Indemnification by the Indemnifying Party, and the Claim for Indemnification shall be paid in accordance with §8(g).
(iii) So long as the Indemnifying Party has assumed and is conducting the defense of the Third Party Claim in good faith and in accordance with §8(d)(ii), (A) the Indemnifying Party will not consent to the entry of any judgment or enter into any settlement with respect to the Third Party Claim without the prior written consent of the Indemnified Party (not to be conditioned, withheld or delayed unreasonably); and (B) the Indemnified Party will not consent to the entry of any judgment or enter into any settlement with respect to the Third Party Claim without the prior written consent of the Indemnifying Party (not to be conditioned, withheld or delayed unreasonably).
(iv) In the event the Indemnifying Party does not assume and conduct the defense of the Third Party Claim in accordance with §8(d)(ii) above, however, (A) the indemnified Party may defend against, and consent to the entry of any judgment or enter into any settlement with respect to, the Third Party Claim in any manner it reasonably may deem appropriate (and the Indemnified Party need not consult with, or obtain any consent from, the Indemnifying Party in connection therewith) and (B) the Indemnifying Party will remain responsible for any Adverse Consequences the Indemnified Party may suffer resulting from, arising out of, relating to, in the nature of, or caused by the Third Party Claim to the fullest extent provided in this §8.
(e) Matters not Involving Third Party Claims. Buyer or Seller may make a claim for any matter that does not involve a Third Party Claim in any amount to which they may be entitled under this §8 by providing a Claim for Indemnification against the other promptly after such Indemnified Party has notice of any Adverse Consequence which may give rise to a claim for indemnification;provided,however, that no delay on the part of Buyer or Seller in notifying the other shall relieve the Indemnifying Party from any obligation hereunder unless (and then solely to the extent) the Indemnifying Party is actually prejudiced by such delay. The Indemnifying Party shall have 30 days to object to the Claim for Indemnification by delivery of a written notice of such objection to the Indemnified Party specifying in reasonable detail the basis for such objection. Failure to timely respond shall constitute a final and binding acceptance of the Claim for Indemnification by the Indemnifying Party, and the Claim for Indemnification shall be paid in accordance with §8(g). If an objection is timely interposed by the Indemnifying Party, then the Indemnified Party and the Indemnifying Party shall negotiate in good faith for a period of 20 days from the date the Indemnified Party receives such objection prior to commencing any arbitration pursuant to §10(o).
(f) Determination of Adverse Consequences. The Parties shall make appropriate adjustments for insurance coverage and take into account the time cost of money (using the Applicable Rate as the discount rate) in determining the Adverse Consequences for purposes of this §8.
(g) Exclusive Remedy. Buyer and Seller acknowledge and agree that the foregoing indemnification provisions in this §8 shall be the exclusive remedy of Buyer and Seller with respect to the Acquired Assets, the Assumed Liabilities, the Business, and the transactions contemplated by this Agreement.
(h) Payment of Claims. Upon Final Determination of the amount of a Claim for Indemnification, the Indemnifying Party shall pay the amount of such Claim for Indemnification within ten days of the date of such Final Determination. A "Final Determination" of a claim shall be (i) a judgment of any court determining the validity of a disputed claim, if no appeal is pending from such judgment or if the time to appeal therefrom has elapsed (it being understood that the Indemnified Party shall have no obligation to appeal); or (ii) an award of any arbitrator or arbitration panel determining the validity of such disputed claim, if there is not pending any motion to set aside such award or if the time within which to move to set such award aside has elapsed; or (iii) a written
A-41
termination of the dispute with respect to such claim signed by all of the parties thereto or their attorneys; or (iv) a written acknowledgment of the Indemnifying Party that it no longer disputes the validity of such claim; (v) the date on which an Indemnifying Party fails to respond to a Claim for Indemnification as specified in §8(d)(ii) or §8(e); or (v) such other evidence of final determination of a disputed claim as shall be reasonably acceptable to the Parties.
(a) Termination of Agreement. Certain of the Parties may terminate this Agreement at any time prior to Closing as provided below:
(i) Buyer and Seller may terminate this Agreement by mutual written consent;
(ii) Buyer may terminate this Agreement by giving written notice to Seller (A) in the event Seller has breached any material representation, warranty, or covenant contained in this Agreement in any material respect, Buyer has notified Seller of the breach, and the breach has continued without cure for a period of 30 days after the notice of breach or (B) if the Closing shall not have occurred on or before 120 days after signing, by reason of the failure of any condition precedent under §7(a) hereof (unless the failure results primarily from Buyer itself breaching any representation, warranty, or covenant contained in this Agreement);
(iii) Seller may terminate this Agreement by giving written notice to Buyer (A) in the event Buyer has breached any material representation, warranty, or covenant contained in this Agreement in any material respect, Seller has notified Buyer of the breach, and the breach has continued without cure for a period of 30 days after the notice of breach or (B) if the Closing shall not have occurred on or before 120 days after signing, by reason of the failure of any condition precedent under §7(b) hereof (unless the failure results primarily from Seller itself breach any representation, warranty, or covenant contained in this Agreement);
(iv) Buyer may terminate this Agreement by giving written notice to Seller (A)if Seller's board of directors shall have withdrawn or modified or amended, in a manner adverse in any material respect to Buyer, its approval of this Agreement or its recommendation set forth in §5(j)(ii), (B) if Seller's board of directors shall have approved, recommended or endorsed any Acquisition Proposal other than the transaction contemplated by this Agreement, (C) if Seller shall have failed to call the Special Meeting within a reasonable time after completion of the SEC review process or shall have failed within a reasonable time thereafter to mail the Proxy Statement to the Seller Stockholders, or (D) if Seller shall have failed to include in such Proxy Statement the recommendation set forth in §5(j)(ii); and
(v) Seller may terminate this Agreement by giving written notice to Buyer if (A) its board of directors determines in good faith that an Acquisition Proposal is financially superior to the transactions contemplated by this Agreement and is reasonably capable of being financed, (B) Seller has complied with the requirements of §5(g), (C) concurrently with such termination, Seller makes all payments required by §9(b)(ii), and (D) concurrently with such termination, Seller enters into a definitive agreement to effect such Acquisition Proposal, and
(vi) either Seller or Buyer may terminate this Agreement by giving written notice to the other if, at a duly held stockholders meeting of Seller or any adjournment thereof at which a resolution to approve the transactions contemplated by this Agreement is voted upon, the Requisite Stockholder Approval shall not have been obtained.
(i) Except for a termination pursuant to §9(a)(iv) or (v), if any Party terminates this Agreement pursuant to §9(a) above, all rights and obligations of the Parties hereunder shall terminate without any liability of any Party to the other Party (except for any liability of any Party then in breach).
A-42
(ii) If this Agreement is terminated as described in §9(a)(iv) or (v), all rights and obligations of the Parties hereunder shall terminate without any liability of any Party to any other Party (except for any liability of any Party then in breach) and Seller agrees to promptly pay to Buyer US$2,000,000 as liquidated damages within ten Business Days after the earlier of (A) termination of this Agreement, and (B) the execution of an agreement with respect to an Acquisition Proposal. Seller and Buyer agree that by agreeing to these liquidated damages, the Parties acknowledge that (X) such liquidate damages are an integral part of the transactions contemplated by this Agreement and constitute liquidated damages and not a penalty, and (Y) such liquidated damages are necessary because actual damages arising from the loss of opportunity would be not be determinable with any degree of certainty. If Seller fails to promptly pay the liquidated damages due under this §9(b)(ii), Seller shall pay the costs and expenses (including legal fees and expenses) in connection with any action in which Buyer prevails, including the filing of any lawsuit or other legal action, taken to collect payment, together with interest on the amount of any unpaid fee at the Applicable Rate from the date such fee was required to be paid.
(iii) Notwithstanding §9(b)(i) and (ii), the confidentiality provisions contained in §5(e) above shall survive termination of this Agreement.
(a) Press Releases and Public Announcements. No Party shall issue any press release or make any public disclosure or announcement relating to the subject matter of this Agreement without the prior written approval of the other Party, except as may be required by Law, including without limitation disclosure requirements of the SEC, NYSE, Nasdaq, or any stock exchange, in which event the other party shall use commercially reasonable efforts to give the other party reasonable advance notice and reasonable opportunity to review and comment upon any such disclosure.
(b) No Third-Party Beneficiaries. This Agreement shall not confer any rights or remedies upon any Person other than the Parties and their respective successors and permitted assigns.
(c) Entire Agreement. This Agreement and the Transaction Documents constitute the entire agreement between the Parties and supersede any prior understandings, agreements, or representations by or between the Parties, written or oral, to the extent they relate in any way to the subject matter hereof.
(d) Succession and Assignment. This Agreement shall be binding upon and inure to the benefit of the Parties named in this Agreement and their respective successors and permitted assigns. No Party may assign either this Agreement or any of its rights, interests, or obligations hereunder without the prior written approval of the other Party;provided, however, that Buyer may (i) assign any or all of its rights and interests hereunder to one or more of its wholly-owned Subsidiaries and (ii) designate one or more of its wholly-owned Subsidiaries to perform its obligations hereunder (in any or all of which cases Buyer nonetheless shall remain responsible for the performance of all of its obligations hereunder).
(e) Counterparts. This Agreement may be executed in one or more counterparts (including by means of facsimile), each of which shall be deemed an original but all of which together will constitute one and the same instrument.
(f) Headings. The section headings contained in this Agreement are inserted for convenience only and shall not affect in any way the meaning or interpretation of this Agreement.
(g) Notices. All notices, requests, demands, claims, and other communications hereunder will be in writing. Any notice, request, demand, claim, or other communication hereunder shall be deemed
A-43
duly given when delivered to the intended recipient as set forth below and signed for by a representative at that address:
| If to Seller: | Copy to: | |
NaPro BioTherapeutics, Inc. | Bartlit Beck Herman Palenchar & Scott | |
| 4840 Pearl East Circle | 1899 Wynkoop Street, Suite 800 | |
| Suite 300W | Denver, CO 80202 | |
| Boulder, CO 80301 | Attention: James L. Palenchar, Esq. | |
| Attention: Kai P. Larson | Fax: 303-592-3140 | |
| Fax: 303-530-1296 | ||
If to Buyer: | Copy to: | |
Faulding Pharmaceutical Co. | Reed Smith LLP | |
| Mack Cali Centre II | 599 Lexington Avenue | |
| 650 From Road, Second Floor | New York, NY 10022 | |
| Paramus, NJ 07652 | Attention: William R. Griffith, Esq. | |
| Attention: Stuart Hinchen | ||
And: | ||
Mayne Group Limited | Reed Smith LLP | |
| Level 22 | 136 Main Street | |
| 390 St. Kilda Road Melbourne | Suite 250 | |
| Victoria 3000 Australia | Princeton, New Jersey 08543 | |
| Attention: Company Secretary | Attention: Edward P. Bromley III, Esq. | |
| Attention: Chief Financial Officer |
Any Party may change the address to which notices, requests, demands, claims, and other communications hereunder are to be delivered by giving the other Party notice in the manner set forth in this Agreement.
(h) Governing Law. This Agreement shall be governed by and construed in accordance with the domestic laws of the State of Delaware without giving effect to any choice or conflict of law provision or rule (whether of the State of Delaware or any other jurisdiction) that would cause the application of the laws of any jurisdiction other than the State of Delaware.
(i) Amendments and Waivers. No amendment of any provision of this Agreement shall be valid unless the same shall be in a writing referring to this §10(i) of this Agreement and signed by Buyer and Seller. Seller may consent to any such amendment at any time prior to the Closing with the prior authorization of its board of directors;provided, however, that any amendment effected after Seller Stockholders have approved this Agreement will be subject to the restrictions contained in the DGCL. No waiver by any Party of any provision of this Agreement or any default, misrepresentation, or breach of warranty or covenant hereunder, whether intentional or not, shall be valid unless the same shall be in a writing referring to this §10(i) of this Agreement and signed by the Party making such waiver nor shall such waiver be deemed to extend to any prior or subsequent default, misrepresentation, or breach of warranty or covenant hereunder or affect in any way any rights arising by virtue of any prior or subsequent such occurrence.
(j) Severability. Any term or provision of this Agreement that is invalid or unenforceable in any situation in any jurisdiction shall not affect the validity or enforceability of the remaining terms and provisions hereof or the validity or enforceability of the offending term or provision in any other situation or in any other jurisdiction.
A-44
(k) Expenses. Each of Buyer and Seller will bear his, her, or its own costs and expenses (including legal fees and expenses) incurred in connection with this Agreement and the transactions contemplated hereby;provided, however, that all transfer, documentary, sales, use, stamp, registration and other such Taxes, and all conveyance fees, recording charges and other fees and charges (including any penalties and interest) incurred in connection with the consummation of the transactions contemplated by this Agreement shall be borne by Seller.
(l) Construction. The Parties have participated jointly in the negotiation and drafting of this Agreement. In the event an ambiguity or question of intent or interpretation arises, this Agreement shall be construed as if drafted jointly by the Parties and no presumption or burden of proof shall arise favoring or disfavoring any Party by virtue of the authorship of any of the provisions of this Agreement. Any reference to any federal, state, local, or foreign statute or law shall be deemed also to refer to all rules and regulations promulgated thereunder, unless the context requires otherwise. The word "including" shall mean including without limitation. All accounting terms used in this Agreement shall have the meanings given to them in accordance with GAAP. The singular shall mean the plural, the plural shall mean the singular, and the use of any gender shall include all genders; and all references to any particular party defined in this Agreement shall be deemed to refer to each and every Person defined in this Agreement as such party individually, and to all of them, collectively, jointly and severally, as though each were named wherever the applicable defined term is used. All references to "§" shall be deemed to refer to the provisions of this Agreement. All references to time in this Agreement shall mean Central Standard Time or Central Daylight Time, as then in effect. The words "this Agreement," "hereof," "hereunder," "herein," "hereby, or words of similar import shall refer to this Agreement as a whole and not to a particular section, subsection, clause or other subdivision of this Agreement, unless the context otherwise requires. The Parties intend that each representation, warranty, and covenant contained in this Agreement shall have independent significance.
(m) Incorporation of Exhibits and Schedules. The Exhibits, the Disclosure Schedule and Schedule A identified in this Agreement are incorporated in this Agreement by reference and made a part hereof.
(n) Bulk Transfer Laws. Buyer acknowledges that Seller will not comply with the provisions of any bulk transfer laws of any jurisdiction in connection with the transactions contemplated by this Agreement.
(o) Arbitration. Any dispute, controversy or claim arising out of or relating to this Agreement (a "Dispute") shall be settled by binding arbitration in accordance with the commercial arbitration rules of the American Arbitration Association ("AAA"). Any such Dispute shall be arbitrated on an individual basis, and shall not be consolidated in any arbitration with any dispute, claim or controversy of any other party. The arbitration shall be conducted in Denver, Colorado, and any court having jurisdiction thereof may immediately issue judgment on the arbitration award. The Parties agree that the arbitration provided for in this §10(o) shall be the exclusive means to resolve all Disputes.
(p) Limitations on Representations and Warranties. EXCEPT FOR THE REPRESENTATIONS AND WARRANTIES SET FORTH IN THIS AGREEMENT AND THE TRANSACTION DOCUMENTS, SELLER EXPRESSLY DISCLAIMS (A) ANY REPRESENTATIONS OR WARRANTIES OF ANY KIND OR NATURE, EXPRESS OR IMPLIED, AS TO LIABILITIES, OPERATIONS, TITLE, CONDITION, VALUE OR QUALITY OF THE ACQUIRED ASSETS OR THE PROSPECTS, FINANCIAL OR OTHERWISE, RISKS AND OTHER INCIDENTS OF THE BUSINESS AND (B) ANY REPRESENTATION OR WARRANTY OF MERCHANTABILITY, USAGE, OR SUITABILITY OR FITNESS FOR ANY PARTICULAR PURPOSE WITH RESPECT TO THE ACQUIRED ASSETS OR ANY PART THEREOF, OR AS TO THE WORKMANSHIP THEREOF, OR THE ABSENCE OF ANY DEFECTS THEREIN, WHETHER LATENT OR PATENT, OR COMPLIANCE WITH ENVIRONMENTAL REQUIREMENTS, OR WHETHER
A-45
SELLER POSSESSES SUFFICIENT REAL PROPERTY, PERSONAL PROPERTY OR INTELLECTUAL PROPERTY TO OPERATE THE BUSINESS. OTHER THAN THE DISCLOSURE SCHEDULE AND THE TRANSACTION DOCUMENTS, NO SCHEDULE OR EXHIBIT TO THIS AGREEMENT OR ANY RELATED COMMUNICATIONS MADE BY SELLER OR ITS REPRESENTATIVE OR ANY ORAL, WRITTEN, OR ELECTRONIC RESPONSE TO ANY INFORMATION REQUEST PROVIDED TO BUYER, WILL CAUSE OR CREATE ANY REPRESENTATION OR WARRANTY, EXPRESS OR IMPLIED, OTHER THAN THOSE SET FORTH IN THIS AGREEMENT AND THE TRANSACTION DOCUMENTS AND BUYER ACKNOWLEDGES THAT ANY RELIANCE BY BUYER UPON ANY SUCH COMMUNICATIONS OR RESPONSE IS UNREASONABLE.
(q) Mylan Litigation. Notwithstanding anything to the contrary herein, from and after the date of this Agreement, Seller shall prosecute the Mylan Litigation as directed by and in cooperation with Buyer until the Closing Date, after which time Buyer shall direct the prosecution of (or settle or otherwise resolve) the Mylan Litigation in the exercise of its sole discretion. Buyer shall be responsible for all costs, including attorney's fees, (but not including NaPro's internal personnel and other administrative costs) of maintaining the Mylan Litigation from the date of this Agreement forward.
* * * * *
A-46
IN WITNESS WHEREOF, the Parties hereto have executed this Asset Purchase Agreement as of the date first above written.
Faulding Pharmaceutical Co. | ||||
By: | /s/ STUART JAMES | |||
| Title: | Director | |||
NaPro BioTherapeutics, Inc. | ||||
By: | /s/ KAI P. LARSON | |||
| Title: | Vice President, General Counsel | |||
A-47
EXHIBIT A TO ASSET PURCHASE AGREEMENT
FORM OF INTELLECTUAL PROPERTY AGREEMENT
This Intellectual Property Agreement (this "Agreement") is entered into by and between NaPro BioTherapeutics, Inc. ("NaPro") and Faulding Pharmaceutical Co. ("Faulding") as of , 2003.
WHEREAS, NaPro and Faulding (each a "Party" and collectively, the "Parties") have entered into an Asset Purchase Agreement and related agreements, whereby Faulding has purchased from NaPro certain assets (including intellectual property) relating to the manufacture, use, and sale of bulk paclitaxel and paclitaxel formulated in polyethoxylated castor oil;
WHEREAS, NaPro will continue its activities in development of products other than polyethoxylated castor oil formulations and requires freedom to operate in such endeavors; and
WHEREAS, Faulding wishes to have freedom to operate in regards to any NaPro intellectual property developed by NaPro.
NOW, THEREFORE, for good and valuable consideration, the receipt of which is hereby acknowledged, the parties agree as follows:
"Asset Purchase Agreement" shall mean that agreement, entitled, "Asset Purchase Agreement" entered into between NaPro and Faulding as of August 25, 2003, including the schedules thereto.
"Bryn Mawr Agreement" shall mean the License Agreement, dated November 3, 1993, by and between NaPro and Bryn Mawr College relating to the license of certain technology.
"NaPro Improvements" shall mean any and all intellectual property rights relating to bulk paclitaxel and/or polyethoxylated castor oil which now exist or are developed by NaPro following execution of this Agreement, including, without limitation, inventions, patents, trade secrets, copyrights, confidential information, and other proprietary rights.
"NaPro Permitted Use" shall mean researching, developing, making, using and selling products other than products which contain polyethoxylated castor oil, or which are incorporated into any product which contains polyethoxylated castor oil or which are used in the manufacture of any product which contains polyethoxylated castor oil.
"Faulding Permitted Use" shall mean researching, developing, making, using, selling, offering for sale or importing bulk paclitaxel for any use and/or researching, developing, making, using, selling, offering for sale or importing paclitaxel formulated in polyethoxylated castor oil and ethanol for injection (the product currently marketed by Faulding.)
"Paclitaxel Intellectual Property" shall mean any and all intellectual property rights transferred to Faulding by NaPro pursuant to the Asset Purchase Agreement, including, without limitation, inventions, patents, trade secrets, confidential information, and other proprietary rights, and all patent or patent applications which are based in whole upon and claim priority to any of the patents or patent applications transferred pursuant to the Asset Purchase Agreement (including, without limitation, to any and all continuations, divisions, continuations-in-part, substitutes, reissues, renewals, reexaminations or extensions thereof.) but not including any rights of Faulding under the Bryn Mawr Agreement.
"Person" shall mean any person, corporation, or entity.
1
Faulding's Covenant Not to Sue:
Faulding shall not bring any action against NaPro or any of NaPro's Permitted Assignees or Collaborators arising out of or relating to the use of the Paclitaxel Intellectual Property for any NaPro Permitted Use anywhere in the world. This covenant not to sue set forth in this Agreement shall not, however, eliminate or reduce Faulding's right to enforce the covenant not to compete found in Section 6(e) of the Asset Purchase Agreement. Faulding shall require any person or entity to which Faulding may assign or license with right to enforce any portion of the Paclitaxel Intellectual Property as a condition of such assignment or grant of such right to agree to and comply with the covenants set forth in this section, including, without limitation, the obligation set forth in this sentence to bind any subsequent assignee or licensee. Faulding acknowledges that any assignment or license of Paclitaxel Intellectual Property inconsistent with this section shall be void.
Faulding agrees that a breach of the foregoing may cause irreparable harm to NaPro, the extent of which would be difficult to ascertain and for which damages would not be an adequate remedy. Therefore, in addition to its rights and remedies otherwise available at law, including, without limitation, the recovery of damages for breach of this Agreement, NaPro may be entitled to (a) immediate equitable relief, specifically including, but not limited to, both interim and permanent restraining orders and injunctions and (b) to such other and further equitable relief as the court may deem proper under the circumstances. NaPro may not be required to prove actual damages in any such action for such specific performance.
NaPro shall not bring any action against Faulding or any of Faulding's Permitted Assignees or Collaborators arising out of or relating to the use of the NaPro Improvements for any Faulding Permitted Use anywhere in the world. NaPro shall require any person or entity to which NaPro may assign or license with right to enforce any portion of the NaPro Improvements as a condition of such assignment or grant of such right to agree to and comply with the covenants set forth in this section, including, without limitation, the obligation set forth in this sentence to bind any subsequent assignee or licensee. NaPro acknowledges that any assignment or license of NaPro Improvements inconsistent with this section shall be void.
NaPro agrees that a breach of the foregoing may cause irreparable harm to Faulding, the extent of which would be difficult to ascertain and for which damages would not be an adequate remedy. Therefore, in addition to its rights and remedies otherwise available at law, including, without limitation, the recovery of damages for breach of this Agreement, Faulding may be entitled to (a) immediate equitable relief, specifically including, but not limited to, both interim and permanent restraining orders and injunctions and (b) to such other and further equitable relief as the court may deem proper under the circumstances. Faulding may not be required to prove actual damages in any such action for such specific performance.
Neither Party shall have any rights granted by virtue of this Intellectual Property Agreement to enforce any intellectual property rights of the other Party or to require any such enforcement by the other Party. Neither Party shall be obligated by virtue of this Intellectual Property Agreement to disclose to the other Party any confidential information or other intellectual property, including, without limitation, any NaPro Improvements or any intellectual property developed by Faulding. No rights or obligations with respect to any intellectual property are to be implied or assumed which go beyond those explicitly enumerated herein.
2
Permitted Assignees and Collaborators:
NaPro may assign this Agreement (as a whole) only to a successor or purchaser of all or a substantial part of NaPro's business relating to taxanes, including, without limitation, the NaPro Improvements, if any (a "Permitted Assignee"). Faulding may assign this agreement (as a whole) only to a successor or purchaser of all or a substantial part of Faulding's business relating to paclitaxel, including, without limitation, the Paclitaxel Intellectual Property (a "Permitted Assignee"). Any attempted assignment by NaPro or Faulding that is inconsistent with the foregoing shall be void.
NaPro may grant the benefits of the covenant not to sue contained herein to any person or any number of persons who assist or collaborate with NaPro in the research, development, manufacture, or marketing of a product, or to a person that purchases product developed or under development by NaPro directly or indirectly from NaPro or to a person who purchases rights to research, develop, manufacture such a product from NaPro;provided, however, that NaPro may not grant such benefits to any other person that would allow such other person to do what NaPro is constrained from doing by the covenant not to compete in Section 6(e) of the Asset Purchase Agreement. For purposes of clarification, it is the intent of the Parties that NaPro shall use this covenant not to sue in connection with its own research, and development and commercial efforts related to products (other than Paclitaxel formulated in polyethoxylated castor oil whether including further components or not) in which NaPro possesses proprietary rights and agreements with third parties regarding such efforts, and NaPro shall not simply grant third parties protection afforded by this Agreement absent some substantial cooperation or collaboration between NaPro and such third party, or absent the purchase by a third party of substantial intellectual property rights of NaPro in addition to the protection afforded by this covenant not to sue. Faulding may grant the benefits of the covenant not to sue contained herein to any person or any number of persons who assist or collaborate with Faulding in the research, development, manufacture, or marketing of a product, or to a person who purchases rights to research, develop, manufacture a product from it or to a person that purchases product directly or indirectly from it.
The covenants not to sue contained herein shall be applicable to and binding on any subsequent owner, licensee, grantee, or other successor in interest (in whole or in part) to the NaPro Improvements and Paclitaxel Intellectual Property, whether through acquisition, contract, bankruptcy, or operation of law.
Upon written request from NaPro, Faulding will use commercially reasonable efforts to seek the consent of Bryn Mawr College to allow Faulding to sublicense NaPro (at no cost to NaPro other than required payments to Bryn Mawr under the Bryn Mawr Agreement) under the Bryn Mawr Agreement for use solely as a NaPro Permitted Use; provided, that NaPro agrees to be bound by all terms and conditions of Bryn Mawr Agreement applicable to sublicensees and; provided further that NaPro holds Faulding harmless from all costs, expenses and license fees related thereto.
This Agreement shall be governed by and construed in accordance with the domestic laws of the State of Delaware without giving effect to any choice or conflict of law provision or rule (whether of the State of Delaware or any other jurisdiction) that would cause the application of the laws of any jurisdiction other than the State of Delaware.
This Agreement shall have a term which is equal to the last to expire of intellectual property contained in the Paclitaxel Intellectual Property or NaPro Improvements.
* * * * *
3
IN WITNESS WHEREOF, each of the parties hereto has executed this Agreement as of the date first above written.
| Faulding Pharmaceutical Co. | ||||
By: | ||||
| Title: | ||||
| NaPro BioTherapeutics, Inc. | ||||
By: | ||||
| Title: | ||||
4
EXHIBIT F TO ASSET PURCHASE AGREEMENT
ABBOTT TERMINATION AGREEMENT
MUTUAL RELEASE AND SETTLEMENT AGREEMENT
THIS MUTUAL RELEASE AND SETTLEMENT AGREEMENT (this "Agreement") dated as of August 25, 2003, by and between Abbott Laboratories, an Illinois corporation ("Abbott"), and NaPro BioTherapeutics, Inc., a Delaware corporation ("NaPro").
A. Abbott and NaPro are parties to a Development, License & Supply Agreement, dated as of July 23, 1999, as amended by an Amendment to Development, License & Supply Agreement, dated as of June 27, 2000, as amended by a Second Amendment to Development, License & Supply Agreement, dated as of September 14, 2001, and as further modified by a Side Letter Agreement to Second Amendment to the Development, License & Supply Agreement, dated as of November 27, 2001 (collectively, the "Development Agreement").
B. Abbott and NaPro are parties to a Loan and Security Agreement, dated as of July 23, 1999 (the "Loan Agreement").
C. Abbott and NaPro are parties to a Stock Purchase Agreement, dated as of July 23, 1999, and amended by an Amendment to Stock Purchase Agreement dated as of June 23, 2000 (collectively, the "Stock Purchase Agreement").
D. Abbott and Faulding Pharmaceutical Co., a Delaware corporation ("Faulding"), have entered into an asset purchase agreement pursuant to which Abbott shall sell to Faulding, and Faulding shall purchase from Abbott, among other things, certain assets and rights, and Faulding shall assume from Abbott certain liabilities, associated with the Development Agreement (the "Abbott-Faulding Transaction").
E. NaPro and Faulding have this date entered into an Asset Purchase Agreement pursuant to which NaPro shall sell to Faulding, and Faulding shall purchase from NaPro, among other things, certain assets and rights, and Faulding shall assume from NaPro certain liabilities, associated with the Development Agreement (the "NaPro-Faulding Transaction").
F. As of the closing under the later of the Abbott-Faulding Transaction and the NaPro-Faulding Transaction, Abbott and NaPro desire to terminate the Stock Purchase Agreement and Abbott shall have assigned to Faulding the Development Agreement and the Loan Agreement (collectively, the Development Agreement, the Loan Agreement and the Stock Purchase Agreement shall be herein referred to as the "Subject Agreements") on the terms and subject to the conditions set forth in this Agreement.
G. Capitalized terms not otherwise defined in this Agreement have the meanings given them in the Development Agreement.
NOW, THEREFORE, NaPro and Abbott, for good and valuable consideration the receipt of which is hereby acknowledged, agree as follows:
1. Agreements. On the terms and subject to the conditions of this Agreement, coincident with the last to occur of: (i)the closing of the Abbott-Faulding Transaction and (ii) the NaPro-Faulding Transaction (the "Effective Date"), the Stock Purchase Agreement shall be terminated by mutual agreement of Abbott and NaPro without any further liability of NaPro to Abbott or Abbott to NaPro except as otherwise provided in this Agreement.
1
3. Final Payments by Abbott. At and as of the Effective Date, Abbott shall pay or cause to be paid to NaPro all outstanding invoices for Bulk Drug that is in Abbott's possession, whether or not the time for payment has occurred pursuant to the terms of the Development Agreement.
4. Release.
4.1 Definition of Released Subject Matters. The term "Released Subject Matters" means and includes any and all obligations or liabilities under the Subject Agreements and any and all matters to the extent relating or pertaining to or arising out of the Subject Agreements;provided, that the term Released Subject Matters shall not include the obligations imposed by this Agreement.
4.2 Release by Abbott. At and as of the Effective Date, Abbott, on behalf of itself, and its parents, subsidiaries, affiliates, successors and assigns, and any person claiming by, through or under or in concert with, Abbott hereby irrevocably and unconditionally releases, acquits and forever discharges NaPro and its present and former agents, directors, officers, employees, representatives, attorneys, affiliates, parents, stockholders, and subsidiaries, and all persons acting by, through, under or in concert with NaPro (the "NaPro Released Parties"), from any and all charges, complaints, claims, liabilities, obligations, promises, agreements, controversies, damages, actions, causes of action, suits, rights, demands, costs, losses, debts and expenses (including reasonable attorneys' fees and costs actually incurred) of any nature whatsoever, known or unknown, suspected or unsuspected, which have arisen out of, or which may arise with respect to the Released Subject Matter, including, but not limited to, claims arising out of alleged violations of any contracts, express or implied, any covenant of good faith and fair dealing, express or implied, or any tort, or the violation of any federal, state or other governmental statute, regulation, or ordinance, which Abbott and its parents, subsidiaries, affiliates, stockholders, successors and assigns at any time hereinafter may have, own or hold, or claim to have, own or hold against each or any of the NaPro Released Parties.
4.3 Release by NaPro. At and as of the Effective Date, NaPro, on behalf of itself, and its parents, subsidiaries, affiliates, successors and assigns, and any person claiming by, through or under or in concert with, NaPro hereby irrevocably and unconditionally releases, acquits and forever discharges Abbott and each of Abbott's present and former agents, directors, officers, employees, representatives, attorneys, affiliates, parents, stockholders, and subsidiaries, and all persons acting by, through, under or in concert with Abbott (the "Abbott Released Parties"), from any and all charges, complaints, claims, liabilities, obligations, promises, agreements, controversies, damages, actions, causes of action, suits, rights, demands, costs, losses, debts and expenses (including reasonable attorneys' fees and costs actually incurred) of any nature whatsoever, known or unknown, suspected or unsuspected, which have arisen out of, or which may arise with respect to, the Released Subject Matter, including, but not limited to, claims arising out of alleged violations of any contracts, express or implied, any covenant of good faith and fair dealing, express or implied, or any tort, or the violation of any federal, state or other governmental statute, regulation, or ordinance, which NaPro and its parents, subsidiaries, affiliates, stockholders, successors and assigns at any time hereinafter may have, own or hold, or claim to have, own or hold against each or any of Abbott Released Parties.
4.4 Release Supported by Consideration. Abbott and NaPro expressly acknowledge and agree that the releases granted by this Agreement are supported by valid consideration bargained for at arm's length and shall be a complete bar to any and all claims, demands or causes of action that the Abbott Released Parties might have had, have or will have against any of the NaPro Released Parties as to the Released Subject Matters or that any of the NaPro Released Parties might have had, have or will have against any of the Abbott Release Parties as to the Released Subject Matters.
2
5. Covenants Not to Sue.
5.1 From and after the Effective Date, Abbott covenants not to institute, either on its own behalf or on behalf of any other person or entity, any action or suit at law or in equity, nor to institute, to prosecute or in any way to aid in the institution or prosecution of any claim, demand, action, or cause of action for damages, costs, expenses, or compensation for or on account of any damage, loss or injury either to person or property, or both, whether developed or undeveloped, resulting or to result, known or unknown, past, present or future, against the NaPro Released Parties arising out of the Released Subject Matter.
5.2 From and after the Effective Date, NaPro covenants not to institute, either in its individual capacity or on behalf of any other person or entity, any action or suit at law or in equity, nor to institute, to prosecute or in any way to aid in the institution or prosecution of any claim, demand, action, or cause of action for damages, costs, expenses, or compensation for or on account of any damage, loss or injury either to person or property, or both, whether developed or undeveloped, resulting or to result, known or unknown, past, present or future, against the Abbott Released Parties arising out of the Released Subject Matter.
6. Indemnification. Following the Effective Date, NaPro agrees to indemnify and hold harmless the Abbott Released Parties from and against the full amount of any liability, loss, damage, or expense (including costs and reasonable attorneys' fees incurred by Abbott Released Parties and any judgment required to be paid) for any breach by NaPro of the provisions of this Agreement. Following the Effective Date, Abbott agrees to indemnify and hold harmless the NaPro Released Parties from and against the full amount of any liability, loss, damage, or expense (including costs and reasonable attorneys' fees incurred by the NaPro Released Parties and any judgment required to be paid) for any breach by Abbott of the provisions of this Agreement.
7. No Admission. Neither any undertaking made nor any action taken hereunder nor the fact that this Agreement was executed by the parties shall be construed as an admission of any fact which might otherwise give rise to liability on the part of any party. Each party hereby expressly denies any such liability.
8. Consultation with Counsel. Each of Abbott and NaPro acknowledges that it has read this Agreement and understands all of its terms, and that this Agreement is executed voluntarily, without duress, and with full knowledge of its legal significance. Each of Abbott and NaPro has received independent legal advice from an attorney with respect to the legal consequences of making the settlement and release and with respect to the execution of this Agreement.
9. Entire Agreement. This Agreement contains the entire agreement between the parties and fully supersedes any and all prior agreements or understandings between the parties pertaining to the subject matter hereof.
10. Severability. The provisions of this Agreement are severable. If any provision is found to be unenforceable, all other provisions shall remain in full force and effect. This Agreement shall survive the termination of any arrangements contained herein.
11. Counterparts. This Agreement may be executed in counterparts, each of which shall be deemed an original for purposes of authentication, evidentiary validity, and governance of the parties.
12. Continuing Assistance. The parties agree, without further consideration, to sign and/or cause to be signed, and to deliver to the appropriate person or entity any other documents and to take any other action as may be reasonably necessary to fulfill their respective obligations under this Agreement.
13. Governing Law. This Agreement shall in all respects be interpreted, enforced and governed under the laws of the State of Illinois, without respect to conflict of laws provisions.
3
14. Alternative Dispute Resolution. Any controversy, dispute or claim arising out of or relating in any way to this Agreement shall be resolved pursuant to the alternative dispute resolution procedures set forth inExhibit A.
15. Proper Construction. The language of all parts of this Agreement shall in all cases be construed as a whole according to its fair meaning, and not strictly for or against either of the parties. The section headings used in this Agreement are intended solely for convenience of reference and shall not in any manner amplify, limit, modify or otherwise be used in the interpretation of any of the provisions hereof.
16. Amendments; Successors and Assigns. This Agreement may be modified, or any provision waived, only by a writing executed by Abbott and NaPro. This Agreement is binding upon and for the benefit of the respective successors and assigns of the parties.
16. Injunctive Relief. The parties confirm and acknowledge to each other that money damages might not be a sufficient remedy for any breach of this Agreement and that a party seeking to enforce this Agreement shall be entitled to seek to enforce this Agreement by injunctive relief as a remedy for such breach or any threatened breach of this Agreement, but that such remedy shall not be deemed the exclusive remedy for breach of this Agreement.
17. Representations. Each party represents and warrants that: (a) such party has the requisite power and authority to make, execute, deliver and perform this Agreement, (b) the execution, delivery and performance of this Agreement by such party has been duly authorized by all necessary action on the part of such party and (c) this Agreement has been duly executed and delivered by such party and constitutes the legal, valid and binding obligation of such party, enforceable against such party in accordance with its terms.
18. Termination. If the Effective Date has not occurred on or before December 31, 2003, this Agreement shall terminate and be of no further force or effect.
* * * * *
4
In WITNESS WHEREOF, the parties have executed this Agreement as of the date first set forth above:
Abbott Laboratories | |||||
By: | |||||
Name: | |||||
Title: | |||||
NaPro BioTherapeutics, Inc. | |||||
By: | |||||
Name: | Kai P. Larson | ||||
Title: | Vice President, General Counsel | ||||
5
ALTERNATIVE DISPUTE RESOLUTION
The parties recognize that from time to time a dispute may arise relating to either party's rights or obligations under this Agreement. The parties agree that any such dispute shall be resolved by the Alternative Dispute Resolution ("ADR") provisions set forth in this Exhibit, the result of which shall be binding upon the parties.
To begin the ADR process, a party first must send written notice of the dispute to the other party in accordance with the terms of the Agreement for attempted resolution by good faith negotiations between their respective presidents (or their designees) of the affected subsidiaries, divisions, or business units within twenty-eight (28) days after such notice is received (all references to "days" in this ADR provision are to calendar days). If the matter has not been resolved within twenty-eight (28) days of the notice of dispute, or if the parties fail to meet within such twenty-eight (28) days, either party may initiate an ADR proceeding as provided herein. The parties shall have the right to be represented by counsel in such a proceeding.
1. To begin an ADR proceeding, a party shall provide written notice to the other party of the issues to be resolved by ADR in accordance with the terms of the Agreement. Within fourteen (14) days after its receipt of such notice, the other party may, by written notice to the party initiating the ADR, add additional issues to be resolved within the same ADR.
2. Within twenty-one (21) days following receipt of the original ADR notice, the parties shall select a mutually acceptable neutral to preside in the resolution of any disputes in this ADR proceeding. If the parties are unable to agree on a mutually acceptable neutral within such period, either party may request the President of the CPR Institute for Dispute Resolution ("CPR"), 366 Madison Avenue, 14th Floor, New York, New York 10017, to select a neutral pursuant to the following procedures:
(a) The CPR shall submit to the parties a list of not less than five (5) candidates within fourteen (14) days after receipt of the request, along with aCurriculum Vitae for each candidate. No candidate shall be an employee, director, or shareholder of either party or any of their subsidiaries or affiliates.
(b) Such list shall include a statement of disclosure by each candidate of any circumstances likely to affect his or her impartiality.
(c) Each party shall number the candidates in order of preference (with the number one (1) signifying the greatest preference) and shall deliver the list to the CPR within seven (7) days following receipt of the list of candidates. If a party believes a conflict of interest exists regarding any of the candidates, that party shall provide a written explanation of the conflict to the CPR along with its list showing its order of preference for the candidates. Any party failing to return a list of preferences on time shall be deemed to have no order of preference.
(d) If the parties collectively have identified fewer than three (3) candidates deemed to have conflicts, the CPR immediately shall designate as the neutral the candidate for whom the parties collectively have indicated the greatest preference. If a tie should result between two candidates, the CPR may designate either candidate. If the parties collectively have identified three (3) or more candidates deemed to have conflicts, the CPR shall review the explanations regarding conflicts and, in its sole discretion, may either (i) immediately designate as the neutral the candidate for whom the parties collectively have indicated the greatest preference, or (ii) issue a new list of not less than five (5) candidates, in which case the procedures set forth in subparagraphs 2(a)—2(d) shall be repeated.
1
3. No earlier than twenty-eight (28) days or later than fifty-six (56) days after selection, the neutral shall hold a hearing to resolve each of the issues identified by the parties. The ADR proceeding shall take place at a location agreed upon by the parties. If the parties cannot agree, the neutral shall designate a location other than the principal place of business of either party or any of their subsidiaries or affiliates.
4. At least seven (7) days prior to the hearing, each party shall submit the following to the other party and the neutral:
(a) a copy of all exhibits on which such party intends to rely in any oral or written presentation to the neutral;
(b) a list of any witnesses such party intends to call at the hearing, and a short summary of the anticipated testimony of each witness;
(c) a proposed ruling on each issue to be resolved, together with a request for a specific damage award or other remedy for each issue. The proposed rulings and remedies shall not contain any recitation of the facts or any legal arguments and shall not exceed one (1) page per issue. The parties agree that neither side shall seek as part of its remedy any punitive damages.
(d) a brief in support of such party's proposed rulings and remedies, provided that the brief shall not exceed twenty (20) pages. This page limitation shall apply regardless of the number of issues raised in the ADR proceeding.
Except as expressly set forth in subparagraphs 4(a)-4(d), no discovery shall be required or permitted by any means, including depositions, interrogatories, requests for admissions, or production of documents.
5. The hearing shall be conducted on two (2) consecutive days and shall be governed by the following rules:
(a) Each party shall be entitled to five (5) hours of hearing time to present its case. The neutral shall determine whether each party has had the five (5) hours to which it is entitled.
(b) Each party shall be entitled, but not required, to make an opening statement, to present regular and rebuttal testimony, documents or other evidence, to cross-examine witnesses, and to make a closing argument. Cross-examination of witnesses shall occur immediately after their direct testimony, and cross-examination time shall be charged against the party conducting the cross-examination.
(c) The party initiating the ADR shall begin the hearing and, if it chooses to make an opening statement, shall address not only issues it raised but also any issues raised by the responding party. The responding party, if it chooses to make an opening statement, also shall address all issues raised in the ADR. Thereafter, the presentation of regular and rebuttal testimony and documents, other evidence, and closing arguments shall proceed in the same sequence.
(d) Except when testifying, witnesses shall be excluded from the hearing until closing arguments.
(e) Settlement negotiations, including any statements made therein, shall not be admissible under any circumstances. Affidavits prepared for purposes of the ADR hearing also shall not be admissible. As to all other matters, the neutral shall have sole discretion regarding the admissibility of any evidence.
6. Within seven (7) days following completion of the hearing, each party may submit to the other party and the neutral a post-hearing brief in support of its proposed rulings and remedies, provided
2
that such brief shall not contain or discuss any new evidence and shall not exceed ten (10) pages. This page limitation shall apply regardless of the number of issues raised in the ADR proceeding.
7. The neutral shall rule on each disputed issue within fourteen (14) days following completion of the hearing. Such ruling shall adopt in its entirety the proposed ruling and remedy of one of the parties on each disputed issue but may adopt one party's proposed rulings and remedies on some issues and the other party's proposed rulings and remedies on other issues. The neutral shall not issue any written opinion or otherwise explain the basis of the ruling.
8. The neutral shall be paid a reasonable fee plus expenses. These fees and expenses, along with the reasonable legal fees and expenses of the prevailing party (including all expert witness fees and expenses), the fees and expenses of a court reporter, and any expenses for a hearing room, shall be paid as follows:
(a) If the neutral rules in favor of one party on all disputed issues in the ADR, the losing party shall pay 100% of such fees and expenses.
(b) If the neutral rules in favor of one party on some issues and the other party on other issues, the neutral shall issue with the rulings a written determination as to how such fees and expenses shall be allocated between the parties. The neutral shall allocate fees and expenses in a way that bears a reasonable relationship to the outcome of the ADR, with the party prevailing on more issues, or on issues of greater value or gravity, recovering a relatively larger share of its legal fees and expenses.
9. The rulings of the neutral and the allocation of fees and expenses shall be binding, non-reviewable, and non-appealable, and may be entered as a final judgment in any court having jurisdiction.
10. Except as provided in paragraph 9 or as required by law, the existence of the dispute, any settlement negotiations, the ADR hearing, any submissions (including exhibits, testimony, proposed rulings, and briefs), and the rulings shall be deemed Confidential Information. The neutral shall have the authority to impose sanctions for unauthorized disclosure of Confidential Information.
11. All ADR hearings shall be conducted in the English language.
3
* Portions denoted with an asterisk have been omitted and filed separately with the Securities & Exchange Commission pursuant to a request for confidential treatment.
EXHIBIT G TO ASSET PURCHASE AGREEMENT
FAULDING TERMINATION AGREEMENT
TERMINATION, MUTUAL RELEASE AND SETTLEMENT AGREEMENT
This Termination, Mutual Release and Settlement Agreement (this "Agreement") dated as of August 25, 2003, by and between Faulding Pharmaceutical Co., a Delaware corporation ("Faulding"), F.H. Faulding & Co., Limited, a company organized under the laws of Australia, and Central Laboratories, Limited, a corporation organized under the laws of Ireland, and NaPro BioTherapeutics, Inc., a Delaware corporation ("NaPro").
A. F. H. Faulding & Co., Limited, a Faulding affiliate, and NaPro are parties to a Second Amended and Restated Master Agreement, dated as of June 5, 2000 (the "Second Amended and Restated Master Agreement") and Central Laboratories, Limited, a Faulding affiliate, and NaPro are parties to a European Agreement, dated as of March 2, 2001 (the "European Agreement" and, together with the Second Amended and Restated Master Agreement, the "Faulding Agreements").
B. Abbott Laboratories ("Abbott") and NaPro are parties to a Development, License and Supply Agreement, dated as of July 23, 1999, as amended by an Amendment to Development, License & Supply Agreement, dated as of June 27, 2000, as amended by a Second Amendment to Development, License & Supply Agreement, dated as of September 14, 2001, and as further modified by a Side Letter Agreement to the Second Amendment to the Development, License & Supply Agreement, dated as of November 27, 2001 (collectively, the "Development Agreement").
C. Abbott and NaPro are parties to a Loan and Security Agreement, dated as of July 23, 1999 (the "Loan Agreement").
D. Abbott and Faulding have entered into an asset purchase agreement pursuant to which Abbott has agreed to sell to Faulding, and Faulding has agreed to purchase from Abbott, among other things, certain assets and rights, and Faulding shall assume from Abbott certain liabilities, associated with the Development Agreement (the "Abbott-Faulding Transaction").
E. NaPro and Faulding have this date entered into an Asset Purchase Agreement pursuant to which NaPro has agreed to sell to Faulding, and Faulding has agreed to purchase from NaPro, among other things, certain assets and rights, and Faulding shall assume from Abbott certain liabilities, associated with the Development Agreement (the "NaPro-Faulding Transaction").
F. Upon the closing under the later of the Abbott-Faulding Transaction and the NaPro-Faulding Transaction, Abbott shall have assigned the Development Agreement and the Loan Agreement to Faulding. Faulding and NaPro desire to terminate the Faulding Agreements, the Development Agreement and the Loan Agreement (the Faulding Agreements, the Development Agreement and the Loan Agreement shall collectively be hereinafter referred to as the "Subject Agreements") on the terms and subject to the conditions set forth in this Agreement.
G. Capitalized terms not defined in this Agreement have the meanings given them in Asset Purchase Agreement dated as of August 25, 2003 between NaPro and Faulding.
1
NOW, THEREFORE, NaPro and Faulding, for good and valuable consideration the receipt of which is hereby acknowledged, agree as follows:
1. Termination of Agreements. On the terms and subject to the conditions of this Agreement, coincident with the last to occur of the closing of the Abbott-Faulding Transaction and the NaPro-Faulding Transaction (the "Effective Date"), the Subject Agreements shall be terminated by mutual agreement of NaPro and Faulding, in each case, without any further liability of NaPro to Faulding or Faulding to NaPro except as otherwise provided in this Agreement.
2. Final Payments by NaPro. On and as of the Effective Date, NaPro shall pay or cause to be paid to Faulding (a) $20,000,000 principal amount plus accrued interest through the close of business on the Effective Date owing in respect of the Loan Agreement; and (b) $1,668,898 to reimburse Faulding for the assumption of Abbott's incurred expenses in qualification of Abbott's McPherson formulation facility, which payments shall be for the benefit of Abbott or its assignee.
3. Final Payments by Faulding. Faulding shall pay NaPro for any outstanding trade receivables for bulk active pharmaceutical ingredient under the Faulding Agreements irrespective of whether the time for payment has occurred pursuant to the terms of the Faulding Agreements.
4. End-Use Reconciliation. The Parties agree to reconcile the Second Amended and Restated Master Agreement and the European Agreement. The Parties further agree that (a) any end-use reconciliation amount payable by NaPro to Faulding, or Faulding to NaPro, or their respective designees, as the case may be, pursuant to Section 4.7.3 of the Second Amended and Restated Master Agreement with respect to the closing of the contract year as of March 30, 2003 shall be made on or before the Effective Date, and (b) any end-use reconciliation amount payable by NaPro to Faulding, or Faulding to NaPro, or their respective designees, as the case may be, pursuant to Section 3.10.3 of the European Agreement with respect to the closing of the contract year as of March 30, 2003 shall be made on or before the Effective Date. The Parties further agree that the end-use reconciliation amount under each of the Second Amended and Restated Master Agreement and the European Agreement shall be determined for the period from March 31, 2003, through the date that is 14 days prior to the closing of the NaPro-Faulding Transaction, in a manner consistent with the procedures outlined in Section 4.7.3 of the Second Amended and Restated Master Agreement or Section 3.10.3 of the European Agreement, as the case may be, and such resulting end-use reconciliation amount payable by NaPro to Faulding, or Faulding to NaPro, or their respective designees, as the case may be, shall be made on the Effective Date.
5. Bulk Paclitaxel. At NaPro's option, NaPro shall, on the Effective Date, deliver to Faulding [*] of bulk active pharmaceutical ingredient Paclitaxel manufactured and conforming to the specifications included in the Second Amended and Restated Master Agreement or, in lieu of such delivery, pay Faulding [*] on the Effective Date. In addition to the foregoing, on and as of the Effective Date, Faulding shall pay or cause to be paid to NaPro [*] as payment for [*] of bulk active pharmaceutical ingredient Paclitaxel manufactured and conforming to the specifications included in the Second Amended and Restated Master Agreement to be delivered by NaPro to Faulding on the Effective Date.
6. Release.
6.1 Definition of Released Subject Matters. The term "Released Subject Matters" means and includes any and all obligations or liabilities under the Subject Agreements and any and all matters to the extent relating or pertaining to or arising out of the Subject Agreements;provided, that the term Released Subject Matters shall not include the obligations imposed by this Agreement.
6.2 Release by Faulding. At and as of the Effective Date, Faulding, on behalf of itself, and its parents, subsidiaries, affiliates, successors and assigns, and any person claiming by, through or under or in concert with, Faulding hereby irrevocably and unconditionally releases, acquits and forever discharges NaPro and its present and former agents, directors, officers, employees,
2
representatives, attorneys, affiliates, parents, stockholders, and subsidiaries, and all persons acting by, through, under or in concert with NaPro (the "NaPro Released Parties"), from any and all charges, complaints, claims, liabilities, obligations, promises, agreements, controversies, damages, actions, causes of action, suits, rights, demands, costs, losses, debts and expenses (including reasonable attorneys' fees and costs actually incurred) of any nature whatsoever, known or unknown, suspected or unsuspected, which have arisen out of, or which may arise with respect to, the Released Subject Matter, including, but not limited to, claims arising out of alleged violations of any contracts, express or implied, any covenant of good faith and fair dealing, express or implied, or any tort, or the violation of any federal, state or other governmental statute, regulation, or ordinance, which Faulding and its parents, subsidiaries, affiliates, stockholders, successors and assigns at any time hereinafter may have, own or hold, or claim to have, own or hold against each or any of the NaPro Released Parties.
6.2(a) Exception to Release by Faulding. Faulding does not release any claim related to [*] of bulk paclitaxel delivered previously by NaPro to Faulding, [*].
6.3 Release by NaPro. At and as of the Effective Date, NaPro, on behalf of itself, and its parents, subsidiaries, affiliates, successors and assigns, and any person claiming by, through or under or in concert with, NaPro hereby irrevocably and unconditionally releases, acquits and forever discharges Faulding and each of Faulding's present and former agents, directors, officers, employees, representatives, attorneys, affiliates, parents, stockholders, and subsidiaries, and all persons acting by, through, under or in concert with Faulding (the "Faulding Released Parties"), from any and all charges, complaints, claims, liabilities, obligations, promises, agreements, controversies, damages, actions, causes of action, suits, rights, demands, costs, losses, debts and expenses (including reasonable attorneys' fees and costs actually incurred) of any nature whatsoever, known or unknown, suspected or unsuspected, which have arisen out of, or which may arise with respect to, the Released Subject Matter, including, but not limited to, claims arising out of alleged violations of any contracts, express or implied, any covenant of good faith and fair dealing, express or implied, or any tort, or the violation of any federal, state or other governmental statute, regulation, or ordinance, which NaPro and its parents, subsidiaries, affiliates, stockholders, successors and assigns at any time hereinafter may have, own or hold, or claim to have, own or hold against each or any of Faulding Released Parties.
6.4 Release Supported by Consideration. Faulding and NaPro expressly acknowledge and agree that the releases granted by this Agreement are supported by valid consideration bargained for at arm's length and shall be a complete bar to any and all claims, demands or causes of action that the Faulding Released Parties might have had, have or will have had against any of the NaPro Released Parties as to the Released Subject Matters or that any of the NaPro Released Parties might have had, have or will have against any of the Faulding Release Parties as to the Released Subject Matters.
7. Covenants Not to Sue.
7.1 Faulding's Covenant Not to Sue. From and after the Effective Date, Faulding covenants not to institute, either on its own behalf or on behalf of any other person or entity, any action or suit at law or in equity, nor to institute, to prosecute or in any way to aid in the institution or prosecution of any claim, demand, action, or cause of action for damages, costs, expenses, or compensation for or on account of any damage, loss or injury either to person or property, or both, whether developed or undeveloped, resulting or to result, known or unknown, past, present or future, against the NaPro Released Parties arising out of the Released Subject Matter.
7.2 NaPro's Covenant Not to Sue. From and after the date of this Agreement, NaPro covenants not to institute, either in its individual capacity or on behalf of any other person or entity, any action or suit at law or in equity, nor to institute, to prosecute or in any way to aid in
3
the institution or prosecution of any claim, demand, action, or cause of action for damages, costs, expenses, or compensation for or on account of any damage, loss or injury either to person or property, or both, whether developed or undeveloped, resulting or to result, known or unknown, past, present or future, against the Faulding Released Parties arising out of the Released Subject Matter.
8. Indemnification. From and after the Effective Date, NaPro agrees to indemnify and hold harmless the Faulding Released Parties from and against the full amount of any liability, loss, damage, or expense (including costs and reasonable attorneys' fees incurred by Faulding Released Parties and any judgment required to be paid) for any breach by NaPro of the provisions of this Agreement. From and after the Effective Date, Faulding agrees to indemnify and hold harmless the NaPro Released Parties from and against the full amount of any liability, loss, damage, or expense (including costs and reasonable attorneys' fees incurred by the NaPro Released Parties and any judgment required to be paid) for any breach by Faulding of the provisions of this Agreement.
9. No Admission. Neither any undertaking made nor any action taken hereunder nor the fact that this Agreement was executed by the Parties shall be construed as an admission of any fact which might otherwise give rise to liability on the part of any Party. Each Party hereby expressly denies any such liability.
10. Consultation with Counsel. Each of Faulding and NaPro acknowledges that it has read this Agreement and understands all of its terms, and that this Agreement is executed voluntarily, without duress, and with full knowledge of its legal significance. Each of Faulding and NaPro has received independent legal advice from an attorney with respect to the legal consequences of making the settlement and release and with respect to the execution of this Agreement.
11. Entire Agreement. This Agreement contains the entire agreement between the Parties and fully supersedes any and all prior agreements or understandings between the Parties pertaining to the subject matter hereof.
12. Severability. The provisions of this Agreement are severable. If any provision is found to be unenforceable, all other provisions shall remain in full force and effect. This Agreement shall survive the termination of any arrangements contained herein.
13. Counterparts. This Agreement may be executed in counterparts, each of which shall be deemed an original for purposes of authentication, evidentiary validity, and governance of the Parties.
14. Continuing Assistance. The Parties agree, without further consideration, to sign and/or cause to be signed, and to deliver to the appropriate person or entity any other documents and to take any other action as may be reasonably necessary to fulfill their respective obligations under this Agreement.
15. Governing Law; Jurisdiction. This Agreement shall in all respects be interpreted, enforced and governed under the laws of the State of Delaware, without respect to conflict of laws provisions. Faulding and NaPro hereby irrevocably submit to the exclusive jurisdiction of any court of the State of Delaware or the United States District Court for the District of Delaware for the purpose of any suit, action or other proceeding arising out of or related to this Agreement or the transactions contemplated hereby, which is brought by or for Faulding or NaPro.
16. Proper Construction. The language of all parts of this Agreement shall in all cases be construed as a whole according to its fair meaning, and not strictly for or against either of the Parties. The section headings used in this Agreement are intended solely for convenience of reference and shall not in any manner amplify, limit, modify or otherwise be used in the interpretation of any of the provisions hereof.
4
17. Amendments; Successors and Assigns. This Agreement may be modified, or any provision waived, only by a writing executed by Faulding and NaPro. This Agreement is binding upon and for the benefit of the respective successors and assigns of the Parties.
18. Injunctive Relief. The Parties confirm and acknowledge to each other that money damages might not be a sufficient remedy for any breach of this Agreement and that a Party seeking to enforce this Agreement shall be entitled to seek to enforce this Agreement by injunctive relief as a remedy for such breach or any threatened breach of this Agreement, but that such remedy shall not be deemed the exclusive remedy for breach of this Agreement.
19. Representations. Each Party represents and warrants that: (a) such Party has the requisite power and authority to make, execute, deliver and perform this Agreement, (b) the execution, delivery and performance of this Agreement by such Party has been duly authorized by all necessary action on the part of such Party and (c) this Agreement has been duly executed and delivered by such Party and constitutes the legal, valid and binding obligation of such Party, enforceable against such Party in accordance with its terms.
20. Termination. If the Effective Date has not occurred on or before December 31, 2003, this Agreement shall terminate and be of no further force or effect.
* * * * *
5
In WITNESS WHEREOF, the Parties have executed this Agreement as of the date first set forth above:
| Faulding Pharmaceutical Co. | |||
By: | |||
| Name: | |||
| Title: | |||
Central Laboratories, Limited | |||
By: | |||
| Name: | |||
| Title: | |||
F.H. Faulding & Co., Limited | |||
By: | |||
| Name: | |||
| Title: | |||
NaPro BioTherapeutics, Inc. | |||
By: | |||
| Name: | |||
| Title: | |||
6
August 25, 2003
Board of Directors
NaPro BioTherapeutics, Inc.
4840 Pearl East Circle
Suite 300W
Boulder, CO 80301
Members of the Board:
We understand that NaPro BioTherapeutics, Inc. (the "Company") and Faulding Pharmaceutical Co. ("Acquiror") are proposing to enter into an Asset Purchase Agreement (the "Agreement") which will provide, among other things, for the purchase (the "Transaction") by Acquiror of specified assets (and the assumption of specified liabilities) of the Company's bulk Paclitaxel manufacturing business and the Company's intellectual property related to the right to make, use or sell Paclitaxel formulated in polyethoxylated castor oil and ethanol (collectively, the "Business"). Pursuant to the Agreement, on the closing date of the Transaction Acquiror shall pay cash consideration for the Business of $71.7 million plus certain accounts receivables accruing prior to the closing and minus the Inventory Adjustment Amount (as defined in the Agreement) (such net amount, the "Consideration"), of which approximately $21.7 million shall be paid to Abbott Laboratories, or its assignee, to satisfy certain obligations owed by the Company to Abbott Laboratories and the balance shall be paid to the Company. The terms and conditions of the Transaction are set out more fully in the Agreement.
You have asked us whether, in our opinion, the Consideration is fair from a financial point of view and as of the date hereof to the Company.
For purposes of this opinion we have, among other things:
(i) reviewed certain publicly available financial statements and other business and financial information of the Company, the Business and otherwise relating to the Transaction;
(ii) reviewed certain internal financial statements and other financial and operating data, including certain financial forecasts and other forward looking information, concerning the Business prepared by the management of the Company;
(iii) held discussions with the management of the Company concerning the business, past and current operations, financial condition and future prospects of the Business;
(iv) reviewed the financial terms and conditions set forth in the Agreement;
(v) reviewed the stock price and trading history of the Company's common stock;
(vi) compared the financial performance of the Business with that of certain other publicly traded companies comparable with the Business;
(vii) compared the financial terms of the Transaction with the financial terms, to the extent publicly available, of other transactions that we deemed relevant;
(viii) prepared a discounted cash flow analysis of the Business;
(ix) participated in discussions and negotiations among representatives of the Company and Acquiror and their financial and legal advisors; and
(x) made such other studies and inquiries, and took into account such other matters as we deemed relevant, including our assessment of general economic, market and monetary conditions.
In our review and analysis, and in arriving at our opinion, we have assumed and relied upon the accuracy and completeness of all of the financial and other information provided to us (including information furnished to us orally or otherwise discussed with us by the Company's management) or publicly available and have neither attempted to verify, nor assumed responsibility for verifying, any of
such information. We have relied upon the assurances of the Company's management that it is not aware of any facts that would make such information inaccurate or misleading. Furthermore, we did not obtain or make, or assume any responsibility for obtaining or making, any independent evaluation or appraisal of the properties, assets or liabilities (contingent or otherwise) of the Company or the Business, nor were we furnished with any such evaluation or appraisal. With respect to the financial forecasts and projections (and the assumptions and bases therefor) for the Business that we have reviewed, we have assumed that such forecasts and projections have been reasonably prepared in good faith on the basis of reasonable assumptions and reflect the best currently available estimates and judgments of the Company's management as to the future financial condition and performance of the Business, and we have further assumed that such projections and forecasts will be realized in the amounts and in the time periods currently estimated. We have assumed that the Transaction will be consummated upon the terms set forth in the Agreement without material alteration thereof. In addition, we have assumed that the historical financial statements of the Company reviewed by us have been prepared and fairly presented in accordance with U.S. generally accepted accounting principles consistently applied. We have also assumed that the amount of the Consideration will not be reduced as a result of indemnification, purchase price adjustment (other than the Inventory Adjustment Amount) or other provisions of the Agreement.
This opinion is necessarily based upon market, economic and other conditions as in effect on, and information made available to us as of, the date hereof. It should be understood that subsequent developments may affect the conclusion expressed in this opinion and that we disclaim any undertaking or obligation to advise any person of any change in any matter affecting this opinion which may come or be brought to our attention after the date of this opinion. Our opinion is limited to the fairness, from a financial point of view and as to the date hereof, to the Company of the Consideration. We do not express any opinion as to any tax or other consequences that might result from the Transaction. Our opinion does not address the relative merits of the Transaction and the other business strategies that the Company's board of directors has considered or may be considering, nor does it address the decision of the Company's board of directors to proceed with the Transaction. Neither does our opinion address any legal or accounting matters, as to which we understand that the Company obtained such advice as it deemed necessary from qualified professionals. Furthermore, our opinion does not address the value of the Company's remaining business after the Transaction. In addition, we are not expressing any opinion regarding any payment which may be received by the Company pursuant to the Agreement in connection with the litigation involving Mylan Laboratories, Inc.
We are acting as financial advisor to the Company in connection with the Transaction and will receive a fee upon the delivery of this opinion and an additional fee contingent upon the consummation of the Transaction. In addition, the Company has agreed to indemnify us for certain liabilities that may arise out of our engagement. Further, we acted as the financial advisor to the Company in connection with the Company's purchase of the genomics business of Pangene Corporation in January of 2003. In the ordinary course of business, we may trade in the Company's securities for our own account and the account of our customers and, accordingly, may at any time hold a long or short position in the Company's securities.
Our opinion expressed herein is provided for the information of the board of directors of the Company in connection with its evaluation of the Transaction. Our opinion is not intended to be and does not constitute a recommendation to any stockholder of the Company as to how such stockholder should vote, or take any other action, with respect to the Transaction. This opinion may not be summarized, described or referred to or furnished to any party except with our express prior written consent.
Based upon and subject to the foregoing considerations, it is our opinion that, as of the date hereof, the Consideration is fair to the Company from a financial point of view.
Very truly yours, | ||
WELLS FARGO SECURITIES, LLC |
B-2
U.S. SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
Form 10-K
Annual Report Pursuant to Section 13 or 15(d)
of the Securities Exchange Act of 1934
Year Ended December 31, 2002
Commission File Number 0-24320
NAPRO BIOTHERAPEUTICS, INC.
(Exact name of Registrant as specified in its charter)
| Incorporated in Delaware | IRS ID No. 84-1187753 |
6304 Spine Road, Unit A
Boulder, Colorado 80301
(303) 516-8500
Securities registered pursuant to Section 12(b) of the Act: None
Securities registered pursuant to Section 12(g) of the Act:
Common Stock, $0.0075 par value; Preferred Stock Purchase Rights
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. ý Yeso No
Indicate by check mark if disclosure of delinquent filers pursuant to Item 405 of Regulation S-K is not contained herein, and will not be contained, to the best of registrant's knowledge, in definitive proxy or information statements incorporated by reference in Part III of this Form 10-K or any amendment to this Form 10-K. o
Indicate by check mark whether the registrant is an accelerated filer (as defined in Rule 12b-2 of the Act). o Yesý No
As of March 21, 2003, approximately 29,939,486 shares of the Registrant's common stock, $0.0075 par value per share, were outstanding. The aggregate market value of the voting stock held by non-affiliates of the Registrant was approximately $19,461,000 (based upon the closing price for shares of the Registrant's Common Stock as reported by the Nasdaq National Market for the last trading date prior to that date).
DOCUMENTS INCORPORATED BY REFERENCE
Portions of the Registrant's Proxy Statement for the 2003 Annual Meeting of Stockholders to be held on or about June 30, 2003 are incorporated by reference into Part III of this Form 10-K.
| | | Page | |||
|---|---|---|---|---|---|
| Part I | C-3 | ||||
Item 1 | Business | C-3 | |||
Item 2 | Properties | C-16 | |||
Item 3 | Legal Proceedings | C-17 | |||
Item 4 | Submission of Matters to Vote of Security Holders | C-17 | |||
Part II | C-18 | ||||
Item 5 | Market for Registrant's Common Equity and Related Stockholder Matters | C-18 | |||
Item 6 | Selected Financial Data | C-20 | |||
Item 7 | Management's Discussion and Analysis of Financial Condition and Results of Operations | C-21 | |||
Item 7A | Quantitative and Qualitative Disclosures about Market Risk | C-30 | |||
Item 8 | Financial Statements and Supplementary Data | C-30 | |||
Item 9 | Changes in and Disagreements with Accountants on Accounting and Financial Disclosure | C-30 | |||
Part III | C-31 | ||||
Item 10 | Directors and Executive Officers of the Registrant | C-31 | |||
Item 11 | Executive Compensation | C-31 | |||
Item 12 | Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters | C-31 | |||
Item 13 | Certain Relationships and Related Transactions | C-31 | |||
Item 14 | Controls and Procedures | C-31 | |||
Part IV | C-32 | ||||
Item 15 | Exhibits, Financial Statement Schedules, and Reports on Form 8-K | C-32 | |||
C-2
General
NaPro BioTherapeutics, Inc. (NaPro) is a pharmaceutical company focused in three distinct areas:
- •
- the production and sale of paclitaxel, an approved cancer drug;
- •
- the development of novel, targeted anti-cancer agents (which we refer to as Targeted Oncology), and
- •
- the development of novel genomic technologies, primarily in the area of gene editing for applications in human therapeutics, diagnostics, agribiotechnology and pharmacogenomics (which we refer to as Gene Editing).
Historically, the primary focus of our business has been the production and sale of paclitaxel, a naturally occurring chemotherapeutic anti-cancer agent found in certain species of yew, orTaxus, trees, and a majority of the NaPro's resources have been devoted to this endeavor. We have entered into marketing agreements with established pharmaceutical companies to assist us in gaining regulatory approvals, formulating, and marketing our paclitaxel. Our paclitaxel is currently approved for commercial sale in the U.S., Australia, Israel and more than 25 other countries in the Middle East, Asia and Latin America.
In the field of genomics, we are engaged in the discovery, patenting, development and commercialization of a broad range of products using our proprietary Gene Editing technology. Employing this technology, we are developing products that specifically and precisely edit genes. By making small, highly controlled modifications to genes, we believe we can contribute to the prevention and treatment of many different types of disease. In the area of therapeutics, our efforts to date have been directed primarily to treatments for Sickle Cell Disease and Huntington's Disease. Both projects are currently in the pre-clinical stages of development. In addition, we are developing gene alteration strategies for animals, plants, viruses and other microbes. Our proprietary Gene Editing technology also has application in the research reagent, genomics service and molecular diagnostic fields. We are currently assembling a portfolio of products and services, which we intend to offer to potential customers in these market segments.
In December 2002, we acquired the genomics business of Pangene Corporation. The acquisition consisted primarily of patents and intellectual property relating to Pangene's homologous recombination technology and gene isolation and service business, physical assets, instrumentation, software, customer relationships and third-party licenses. In connection with this agreement, we acquired additional patents in January 2003.
In the field of Targeted Oncology, we are developing several compounds that we believe have potential as new anti-cancer agents. Generally, these compounds consist of conjugates of a cytotoxic agent coupled to a peptide, which selectively and specifically targets certain types of tumor cells. We are testing these conjugates in various in-vitro and animal models, and are currently undertaking the development work necessary to introduce these agents into human clinical trials. It is our belief that coupled cytotoxic agents with a targeting agent will lead to greater safety and efficacy when compared with the cytotoxic chemotherapy agents currently available on the market.
In addition to Gene Editing and Targeted Oncology research and development activities, we are also actively engaged in evaluating the in-licensing or purchase of potential new products and/or technologies, whether or not those products or technologies are derived from natural products, are chemotherapeutic agents, or complement our Gene Editing technology. Our evaluation of new products
C-3
and technologies may involve the examination of individual molecules, classes of compounds, or platform technologies, in both cancer and other therapeutic areas. Acquisitions of new products or technologies may involve the purchase of such products or technologies, or the acquisition of, or merger with, other companies. In the event we enter into any such relationships or transactions, we may consider using available cash, issuing equity securities or increasing our debt. Such transactions could materially affect our capital structure.
NaPro Products: Commercial or under Development
The following chart identifies our therapeutic products, both commercial and under development.
| Product | Indication(s) | Development Status | ||
|---|---|---|---|---|
| Commercial Oncology Products | ||||
| ANZATAX™ (Mayne Pharma) | Breast, Ovarian and Non-Small Cell Lung Cancer | Commercialized in Australia, Asia, South America and the Mideast; in development in Europe | ||
| Biotax® (Tzamal Pharma) | Breast, Ovarian and Non-Small Cell Lung Cancer | Commercialized in Israel | ||
| NaPro Paclitaxel for injection (Abbott Laboratories) | Breast and Ovarian Cancer | Commercialized in the U.S. | ||
Targeted Oncology Products | ||||
| Targeted cancer therapeutic | Small Cell Lung, Prostate, Pancreatic and Colorectal Cancers | Pre-clinical research | ||
| Targeted cancer therapeutic | Head and Neck Cancer | Pre-clinical research | ||
Gene Editing Products | ||||
| Ex-vivo cell therapy | Sickle Cell Disease | Pre-clinical research | ||
| Oligonucleotide therapeutic | Huntington's Disease | Pre-clinical research |
Paclitaxel
To date, the majority of our resources have been directed toward the development and manufacture of paclitaxel. The market for paclitaxel is dominated by Bristol-Myers Squibb Company (Bristol). Bristol's paclitaxel is widely used in the treatment of breast and ovarian cancers, Kaposi's sarcoma, and non-small cell lung cancer when used in combination with cisplatin. Our proprietary manufacturing technology includes the extraction, isolation, and purification of paclitaxel, as well as the semisynthesis of the drug substance. We continue to develop renewable sources of paclitaxel biomass. We have alliances with established pharmaceutical companies who assist us in marketing these products. We believe the combination of our proprietary manufacturing technology, biomass capability, and pharmaceutical alliances has allowed us to participate significantly in the broad paclitaxel market.
Scientific Background. In 1963, the National Cancer Institute (NCI) recognized that the natural product paclitaxel killed leukemia cells and inhibited the development of a variety of tumors. Over the next two decades, researchers working under grants from the NCI conducted studies to determine paclitaxel's structure and its mechanism of action. The NCI studies indicated that paclitaxel inhibits the normal action of microtubules in cancer cell division. Microtubules, located in the cytoplasm of cells, play a vital role in cellular division. Paclitaxel promotes microtubule assembly and blocks normal microtubule disassembly in cells, which stops cell division and causes the cancer cell to die. This cytoplasmic mechanism of action contrasts with the nuclear mechanism of action of the majority of cell-killing drugs that kill the cell by attacking nuclear components such as DNA.
C-4
In June 1991, the NCI formalized a Collaborative Research and Development Agreement (CRADA) for development of paclitaxel with Bristol. Bristol assumed development of paclitaxel, including completion of the necessary clinical trials and manufacturing scale-up. Bristol submitted a New Drug Application (NDA) to the United States Food and Drug Administration (FDA) in June 1992, and received approval for the sale of paclitaxel as a treatment for refractory ovarian cancer in December 1992. Since then, Bristol has received approval for the sale of paclitaxel as a treatment for other cancers.
Paclitaxel is one of a family of compounds, commonly referred to as taxanes, that share a specific chemical structure. Taxanes are found naturally in many parts of various species of yew trees. The concentration of individual taxanes in yew trees is very small, generally less than 0.1%, and accordingly, the process of extracting taxanes from yew biomass is complicated and challenging. Several production approaches can be used to produce paclitaxel. We believe the two most prevalent processes used today are conventional biomass extraction and semisynthesis.
With conventional biomass extraction, the manufacturing process is designed to extract, isolate and purify paclitaxel from yew biomass leaving behind other components, including non-paclitaxel taxanes. However, the extraction, isolation and purification processes are complicated because there are more than 100 different taxanes present in yew biomass. In a semisynthesis process, the initial extraction, isolation and purification is similar to that of the conventional biomass extraction process, except that the process not only isolates paclitaxel, but also other taxanes (which would otherwise be waste byproducts) and converts these other taxanes into paclitaxel. By converting other taxanes into paclitaxel, the semisynthesis process increases the yield of paclitaxel from the same quantity of biomass. Regardless of which process is used, the final product must meet acceptable purity and drug substance criteria.
Other companies have developed taxane products that are similar, but not identical, to paclitaxel. For example, Aventis S.A., a large international pharmaceutical company, has developed docetaxel which is being marketed in various parts of the world under the trademark Taxotere®. Taxotere® has a different toxicity profile than paclitaxel and has side effects not observed with paclitaxel. The FDA approved Taxotere® for treatment of anthracycline-resistant breast cancer in patients without impaired liver function, and for non-small cell lung cancer after failure of a prior platinum-based chemotherapy.
Paclitaxel Strategic Alliances. Our strategy for advancing the development and commercialization of paclitaxel has been to form strategic alliances through long-term exclusive agreements with established pharmaceutical companies.
Abbott Laboratories. In July 1999, we entered into a 20-year exclusive collaborative agreement with Abbott Laboratories (Abbott) for the development and commercialization in the U.S. and Canada of one or more formulations of paclitaxel for treatment of a variety of cancer indications. Abbott is a large, multinational, diversified health care company. Pursuant to the agreement, we are responsible for supplying bulk drug. Clinical trials are conducted jointly with Abbott. We are not currently conducting any clinical trials with Abbott. Abbott is responsible for finishing, regulatory filings, marketing and sale of the finished drug product. Most primary decisions related to the paclitaxel development program are made by a joint Abbott-NaPro development committee. In May 2002, we and Abbott received FDA approval to market and sell paclitaxel in the U.S. Pursuant to the agreement, we receive a fixed price payment of $500 per gram, upon delivery of paclitaxel to Abbott, plus a quarterly royalty payment, if any. The royalty payment is a percentage of sales recorded by Abbott less any product payment already paid to NaPro. The percentage of sales varies each year based upon total sales in that year recorded by Abbott. We do not anticipate receiving significant royalty payments given the current price of the drug.
In connection with the Abbott agreement, we may receive total funding of up to $118.0 million in the form of up to $30.0 million in development milestones, up to $57.0 million in marketing milestones, $20.0 million in secured debt, and $11.0 million in equity investments. Marketing milestones are based
C-5
upon certain annual sales levels, the majority of which are unlikely to be achieved given the current pricing of paclitaxel. Through December 31, 2002, and excluding product sales, we have received $40.0 million under the agreement, including $11.0 million in equity investments in exchange for 2,000,000 shares of our common stock, $9.0 million in development milestones, and $20.0 million in secured debt.
Of the $30.0 million of potential development-related milestone payments, we received $1.0 million upon execution of its agreement with Abbott in July 1999. With the approval of NaPro's ANDA, and upon commencement of commercial sales, we received an $8.0 million payment in May 2002. Thus, $21.0 million in possible development milestones remain. Additional development milestones will only be available should NaPro or Abbott undertake and successfully complete new development work, which is not currently anticipated.
Our indebtedness to Abbott bears a primary interest rate of 6.5% and is due in full on the earlier of: (i) the second anniversary of the first sale of finished product by Abbott to a wholesaler or end-user customer following approval of finished product by the FDA (May 8, 2004); (ii) the termination of the Abbott agreement; or (iii) January 1, 2007. The debt is limited to a borrowing base of collateralized assets, recomputed monthly. The majority of our paclitaxel inventories, and specific assets, which relate to the manufacture of paclitaxel, are collateralized as security for the debt. Abbott's only recourse in the event of our default on the debt is to these specific assets.
Contingent upon receiving regulatory approval and achieving certain commercial sales thresholds over several years, we may receive additional milestone payments from Abbott of up to $57.0 million. We cannot assure that regulatory approval or sales thresholds necessary to trigger any of the future milestone payments will be achieved. Given current market conditions, we believe it is unlikely that any such milestones will be triggered. In addition, Abbott may terminate the agreement at any time with or without cause. Should Abbott terminate without cause, it is obligated to make certain payments to us and to give us twelve months prior written notice.
The Abbott agreement grants Abbott the exclusive right to develop and market our paclitaxel in the U.S. and Canada. Abbott is required to purchase all of its requirements for paclitaxel from us, except in certain circumstances if we become unable to supply Abbott's requirements. Except for limited instances where termination is due to specific breaches of the agreement by us, we retain exclusive rights following termination to any clinical data generated during the course of the agreement. We are required to indemnify Abbott for defects in our paclitaxel that is shipped to Abbott, breaches of our warranties or obligations under the agreement, harm caused by inappropriate co-marketing activities, and some intellectual property and product liability claims. Abbott is required to indemnify us for defects in a finished product containing our paclitaxel manufactured by Abbott, breaches of Abbott's representations and warranties, harm caused by inappropriate marketing activities, and some intellectual property and product liability claims. We have granted an exclusive license to Abbott for our paclitaxel-related patents for intravenous and oral paclitaxel formulations.
In November 2001, we and Abbott entered into a non-exclusive cross license with Bristol relating to paclitaxel which grants us a license under Bristol patents to market injectable paclitaxel, pursuant to an Abbreviated New Drug Application (ANDA) approval. We have the right under the agreement to sublicense to Abbott. The agreement grants Bristol a license to our patents relating to stable paclitaxel formulations. The scope of both licenses is limited to the polyethoxylated caster oil and ethanol formulation currently approved by the FDA.
Mayne Pharma. In 1992, we entered into a 20-year exclusive agreement with F.H. Faulding & Co., Ltd. (Faulding), a large Australian pharmaceutical company, for the clinical development, sale, marketing and distribution of our paclitaxel. In October 2001, Faulding was acquired by Mayne-Nickless Limited, an Australian based health care provider and logistics operator. As a result, Faulding is now known as Mayne Pharma. Mayne Pharma actively markets anti-cancer pharmaceuticals and other
C-6
health care products in Europe, Australia, Asia, South America, and other regions throughout the world. In 2000, we amended the Mayne Pharma agreement to, among other things, add additional countries to Mayne Pharma's exclusive territory. In 2001, we entered into a separate 12-year agreement with Mayne Pharma covering development and sale of our paclitaxel in Europe. With the new agreement, the Mayne Pharma territory now includes substantially all of the world other than the U.S., Canada, Japan, Israel, the nations of the former Soviet Union and parts of Africa. Mayne Pharma has received marketing approval for, and is selling our paclitaxel under the tradename ANZATAX™ in Australia and more than 25 other countries in the Middle East, Latin America and Asia.
In Europe, we are responsible for regulatory filings and for funding development. We are also responsible for supplying paclitaxel raw material exclusively to Mayne Pharma to formulate and finish the product. Although Bristol's European exclusivity period ends in 2003, we cannot assure that we will receive regulatory approval in the major markets in Europe. Should we receive approval, Mayne Pharma will then market and sell the final proprietary paclitaxel formulation in Europe. Under the agreement, Mayne Pharma paid an up-front licensing fee to us of $7.5 million. We will share equally the net sales of the product in Europe.
For the Mayne Pharma territories outside of Europe, Mayne Pharma is responsible for funding and, with our input, undertaking the development work required to obtain any necessary regulatory approvals for the commercialization of paclitaxel in such territories. We are responsible for supplying Mayne Pharma with our paclitaxel and Mayne Pharma is required to purchase all of its paclitaxel requirements from us. Once received, Mayne Pharma is responsible for formulating the paclitaxel into its commercial drug product, ANZATAX™. Mayne Pharma pays us a substantial share of its gross proceeds from sales of paclitaxel. We cannot assure, however, that Mayne Pharma will succeed in obtaining further regulatory approvals to market our paclitaxel within its territory. Furthermore, if such approvals are received, we cannot assure that Mayne Pharma will market our paclitaxel successfully in these additional countries.
Mayne Pharma may terminate the agreements: (i) upon our reorganization or insolvency; (ii) if Mayne Pharma becomes controlled by a pharmaceutical company that sells paclitaxel in the Mayne Pharma territory; (iii) if we are controlled by Ivax Corporation or Bristol; (iv) if we are purchased by a pharmaceutical company that sells paclitaxel in the Mayne Pharma territory and that company refuses to be bound by the terms of the Mayne Pharma agreement; (v) if we are unable to meet the paclitaxel supply requirements of Mayne Pharma; or (vi) for material, uncured breach. We may terminate the agreement: (i) upon the reorganization or insolvency of Mayne Pharma; (ii) in certain circumstances, upon a change in control of Mayne Pharma; or (iii) for material, uncured breach.
We are required to indemnify Mayne Pharma for any defect in our paclitaxel that is shipped to Mayne Pharma and for uncured breaches of our warranties or obligations. Mayne Pharma is required to indemnify us against all losses (i) resulting from a defect in a product manufactured by Mayne Pharma containing our paclitaxel except if the defect is our fault, (ii) resulting from a product containing our paclitaxel formulated, stored, handled, promoted, distributed, registered or sold by Mayne Pharma and (iii) for uncured breaches of Mayne Pharma's representations and warranties under the Mayne Pharma agreement.
Tzamal Pharma. We have established an exclusive supply and distribution agreement with Tzamal Pharma (Tzamal) for the development and distribution of paclitaxel in Israel. Tzamal is an Israeli pharmaceutical company, active and well established in the Israeli pharmaceutical market and specializing, among other areas, in ethical products for the treatment of cancer, metabolic diseases, neurological disorders and gynecology/fertility. In January 2001, we received approval in Israel to sell paclitaxel under the trade name Biotax®. The Israeli Ministry of Health has approved Biotax® for use in a variety of cancers and Tzamal is currently selling Biotax® in Israel.
C-7
JCR Pharmaceuticals Co., Ltd. In June 2001, we entered into a mutually exclusive agreement with JCR Pharmaceuticals Co., Ltd. (JCR) for the development, supply and distribution of paclitaxel in Japan. JCR, headquartered in Ashiya, Japan, is a research and development oriented pharmaceutical manufacturer, focusing on the research of bioactive substances such as enzymes, hormones, enzyme inhibitors, growth factors and stimulating factors. It is also engaged in the development and research of bioengineered products such as recombinant human growth hormone. Under the agreement, we will be responsible for manufacturing and supplying the finished drug. Both companies will jointly be responsible for the clinical and regulatory program which will be necessary for seeking approval to market paclitaxel in Japan. JCR will fund this clinical and regulatory program. JCR will also be responsible for the sales and distribution of the product in Japan upon its approval. We cannot assure, however, that we will receive regulatory approval of paclitaxel in Japan, or that we and our strategic partner will successfully market it.
Biomass. Paclitaxel and other taxanes we use in the production of paclitaxel are present in many parts of various species of yew trees. Our technology is designed to allow extraction and purification of paclitaxel and extraction of other taxanes from renewable sources of biomass such as needles and limbstock harvested from yew trees. Taxanes other than paclitaxel can be chemically converted into paclitaxel.
We believe we may be able to reduce our raw material cost while increasing our yield of paclitaxel by growing a reliable and renewable biomass source. In order to have a supply of biomass for use in the production of paclitaxel, we have agreements with various North American commercial nurseries. In 2002, substantially all of our production for Abbott was extracted from cultivated biomass. We regularly conduct other research related to enhancing paclitaxel production in yew trees.
Paclitaxel Manufacturing. The manufacture of paclitaxel occurs in three steps. First, a crude paclitaxel is extracted from yew trees. Second, the extracted crude paclitaxel mixture is isolated and purified. In the final step, the resulting active drug substance is formulated for final packaging. Mayne Pharma and Abbott are each responsible for formulating and final packaging of paclitaxel that they market.
The first step in the manufacture of paclitaxel requires the extraction of crude paclitaxel from the raw biomass materials we cultivate from our yew trees or purchase from others. We currently have contracts with third party manufacturers to process crude paclitaxel from raw biomass materials. While we own and have operated large-scale extraction facilities in Boulder, Colorado, we believe that our manufacturing partners increase our operating flexibility and lower our cost of manufacturing. As a result, we have suspended our extraction operations and rely upon our manufacturing partners to supply all of our requirements for crude paclitaxel extract. Any failure by our manufacturing partners to supply our requirements on a cost-effective basis would jeopardize our ability to supply our strategic partners' commercial needs on a timely and competitive basis. In addition, only a limited number of contract manufacturers are both capable of processing crude paclitaxel extract from raw biomass materials and complying with current federal and state good manufacturing practice regulations. Accordingly, if our manufacturing partners fail to supply our requirements, we may not be able to enter into manufacturing agreements with alternative suppliers of crude paclitaxel extract on commercially acceptable terms and, even if we do, any manufacturer with which we contract may not be able to deliver crude paclitaxel extract in appropriate quantity.
We perform the second manufacturing step, isolation and purification, in our facilities in Boulder, Colorado. Our manufacturing facilities are subject to inspection by the regulatory agencies in the countries in which our paclitaxel is sold. In the past, our manufacturing facilities have been inspected by the FDA, the Australian Therapeutic Goods Administration (TGA) and the European Medicines Evaluation Agency (EMEA). Those agencies have found our facilities to be acceptable for the manufacture of bulk paclitaxel. In 2001 and 2002, we expanded the manufacturing capacity of our
C-8
Boulder, Colorado facilities in anticipation of U.S. approval, which we received in May 2002. We believe that our Boulder, Colorado facilities have adequate capacity to meet commercial requirements for the near future, but we cannot assure such capacity. We also cannot assure that we will have the sales volume to utilize our current capacity.
In order to diversify our supply options and increase our manufacturing capacity, we are developing, and have applied for patent protection for, a semisynthesis process for manufacturing paclitaxel from other taxanes contained in renewable biomass sources. We own, or have licensed, several patents relating to this process and have applied for others. We have manufactured crude paclitaxel with the semisynthesis process in a pilot-scale contracted facility. This crude paclitaxel is then purified at our manufacturing facility in Boulder, Colorado. We continue additional development and testing with a contract manufacturer. The use of semisynthetic paclitaxel will require regulatory approvals, which cannot be assured. Furthermore, we cannot assure our semisynthesis process will perform as expected or that we will be able to effectively adapt the process to commercial-scale manufacturing.
Targeted Oncology Development
We are currently selecting lead compounds from two series of constructs designed to treat either small cell lung, prostate, pancreatic and colorectal cancers or squamous cell head and neck carcinomas. Both potential products have emerged from our targeted oncology program. The lead compounds use specific peptide ligands to deliver a cytotoxic agent to the tumor. Targeted delivery is designed to lower toxicity in normal tissue while increasing efficacy in tumor tissue by taking advantage of specific receptors on the tumor cells. We believe that this approach will result in increased effective concentrations of the cytotoxic agent at the site of the cancer. These programs are being performed in collaboration with academic researchers under the direction of NaPro's Drug Development Team. Despite the generation of positive results in animal and tissue studies, we cannot determine when, if ever, any of these prospects will be successful.
Genomics
In November 2000, we in-licensed Gene Editing technology from the University of Delaware and Thomas Jefferson University. The license agreement grants us exclusive, worldwide rights to intellectual property including patent applications relating to the use of proprietary molecules designed to precisely alter genes in humans, animals, plants, viruses and microbes. We have agreed to provide research and patent funding, as well as an ongoing license fee paid in our common stock. As of December 31, 2002, we have issued 300,000 shares under the license. Assuming we do not cancel the license, we will issue an additional 900,000 shares in 100,000 share-per-year increments on the license anniversaries. We may, at our option, accelerate the issuance dates. Certain milestones may also accelerate the issuance dates. The license is terminable at our option. If terminated, no further shares will be issued. We have committed to fund at least $300,000 in research at the University of Delaware during 2003.
In December 2002, we acquired the genomics business of Pangene Corporation. The acquisition consisted primarily of patents and intellectual property relating to Pangene's homologous recombination technology and gene isolation and service business, physical assets, instrumentation, software, customer relationships and third-party licenses. In connection with this agreement, we acquired additional patents in January 2003.
C-9
One of the technologies licensed to us allows us to use proprietary oligonucleotides to make small, specifically targeted modifications in the chromosomes of a target animal or plant. With this technology, we are attempting to develop products and processes that may allow us to:
- •
- Effectively treat certain human genetic disorders;
- •
- Improve plant traits without inserting foreign DNA into those plants; and/or
- •
- Develop processes to detect genetic variations in a patient's genes that may indicate which patients will better respond to specific medication or experience fewer, or less severe side effects.
We may also be able to manufacture diagnostics, reagents, cell lines and animal models that can be used by the scientific and medical research community to further their own research. This technology may permit specific control over changes in any genome under examination. Such control should help determine the function of genes and the consequences of natural variations in chromosomes in research, therapeutic, and agricultural applications.
Among the disorders we are researching are Sickle Cell Disease and Huntington's Disease. Sickle Cell Disease is a hereditary blood disorder caused by a single point mutation of the betaglobin gene. It is characterized by a defined change in the composition of hemoglobin, the protein used to carry oxygen in the blood. The sickle hemoglobin molecules polymerize into long fibers within red blood cells when deoxygenated, causing the cell to become deformed (sickled), rigid and adhesive. The sickled red blood cells block oxygen flow to the tissues leading to organ damage, stroke and joint pain. The majority of current treatments for Sickle Cell Disease address only the symptoms of the disease. These treatments include the use of pain medications and blood transfusions. While all of these treatments are used to manage the disease, breakthrough crises occur in most patients. We are developing anex-vivo technique aimed at treating this disorder. We are in the early stages of this development and our prospects for success, if any, cannot be measured at this time.
Huntington's Disease is a progressive, neurological disorder, resulting in degeneration of nerve cells in the brain. It is specifically characterized by lethal aggregate formation in neural tissue. Eventually, the patient suffers dementia, uncontrolled movements, and death. There is no known cure for this rare disease. Symptoms usually appear between the ages of 35 and 50, although younger people can also develop the disease. The disease affects five in every one million people. We are developing an oligonucleotide, which may potentially allow the cells to survive. We are in the early stages of this program and we can provide no assurance that we will be successful in this development effort.
We have formed a business around the sale of genomics related reagents, molecular diagnostic tools and services using our Gene Editing technology. This market may include the provision of reagents, tools and diagnostics that can predict predisposition, disease identification, disease progression and dose effectiveness, as well as toxicity.
Patents and Proprietary Technology
Our success depends in part on our ability to obtain and enforce patent protection for our products, both in the U.S. and other countries, and operate without infringing the proprietary rights of third parties. The scope and extent of patent protection for paclitaxel and our genomics and Targeted Oncology product candidates involves complex legal and factual questions and as a consequence is always uncertain. We cannot predict the breadth of claims that will be allowed and issued to us for patents related to biotechnology or pharmaceutical applications. Once patents have been issued, we cannot predict how the claims will be construed or enforced. In addition, statutory differences between countries may limit the protection we can obtain on some of our inventions outside of the U.S. For example, methods of treating humans are not patentable in many countries outside of the U.S.
C-10
We rely on patent and other intellectual property protection to prevent our competitors from developing, manufacturing and marketing products related to our technology. Our patents may not be enforceable and they may not afford us protection against competitors, especially since there is a lengthy time between when a patent application is filed and when it is actually issued. Additionally, there are hundreds of genomics, pharmaceutical and chemical patents being issued every week throughout the world. Many of these have patent claims that are difficult to categorize and interpret. Because of this, we may infringe on intellectual property rights of others without being aware of the infringement. If a patent holder believes that one of our product candidates infringes on their patent, they may sue us even if we have received patent protection for our technology. If another person claims we are infringing their technology, we could face a number of issues, including the following:
- •
- defending a lawsuit, which is very expensive and time consuming;
- •
- paying a large sum for damages if we are found to be infringing;
- •
- being prohibited from selling or licensing our products or product candidates until we obtain a license from the patent holder, who may refuse to grant us a license or will only agree to do so on unfavorable terms. Even if we are granted a license, we may have to pay substantial royalties or grant cross-licenses to our patents; and
- •
- redesigning our products or product candidates so they do not infringe on the patent holder's technology if we are unable to obtain a license. This may not be possible and, even if possible, it could require substantial additional capital and could delay commercialization.
The coverage claimed in a patent application can be significantly narrowed before a patent is issued, either in the U.S. or abroad. We do not know whether any of our pending or future patent applications will result in the issuance of patents. To the extent patents have been issued or will be issued, some of these patents are subject to further proceedings that may limit their scope. It is not possible to determine which patents may provide significant proprietary protection or competitive advantage, or which patents may be circumvented or invalidated. Furthermore, patents already issued to us, or patents that may issue on our pending applications, may become subject to dispute, including interference proceedings in the U.S. to determine priority of invention. We are currently involved in opposition proceedings in Europe and other countries contesting the validity of issued patents. If our currently issued patents are invalidated or if the claims of those patents are narrowed, our ability to prevent competitors from marketing products that are currently protected by those patents could be reduced or eliminated. We could then face increased competition resulting in reduced market share, prices and profit.
We have filed and have in-licensed a number of patents relating to our genomics and Targeted Oncology activities. However, there are numerous patents and pending applications in the fields of genomics and Targeted Oncology which are owned and controlled by third parties. Although we have aggressively worked to protect our proprietary technologies through the patenting process, there is no assurance that we will have freedom to operate in the fields where we are developing commercial products.
Paclitaxel is an unpatentable, naturally-occurring compound. Various compositions containing paclitaxel, and also various processes and other technologies, including those relating to extracting paclitaxel and preparing the drug for finished formulation, are or may be patented. In addition, some methods of administering paclitaxel are or may be patented.
In September 2000, we and Abbott filed a patent infringement suit in the U.S. District Court for the District of Colorado against Bristol alleging infringement by Bristol of two patents we own: U.S. Patent numbers 5,972,992 and 5,977,164 which relate to paclitaxel formulation. In November 2001, we and Abbott settled this suit with Bristol. In connection with this settlement, the parties entered into a non-exclusive cross license agreement relating to paclitaxel. The agreement grants us a license under
C-11
Bristol patents to market injectable paclitaxel, pursuant to an ANDA approval. We have the right under the agreement to sublicense to Abbott. The settlement agreement grants Bristol a license to NaPro's patents relating to stable paclitaxel formulations. The scope of both licenses is limited to the polyethoxylated castor oil and ethanol formulation currently approved by the FDA.
We are aware of competitors and potential competitors who are pursuing patent protection in various areas of genomics, Targeted Oncology, and for the extraction, preparation, formulation, administration and production of natural and semisynthetic paclitaxel. If our technology, products or activities are deemed to infringe the other companies' rights, we could be subject to damages or prevented from using the technology that is infringing other companies' rights, or we could be required to obtain licenses to use that technology. We cannot be sure that we would be able to obtain those licenses on terms acceptable to us, or at all. If we were unable to obtain those licenses or were prevented from using our technology, we could encounter significant delays in product market introductions while we attempt to design around the patents or rights infringed, or we could find the development, manufacture or sale of products to be impossible, any of which would have a material adverse effect on us. In addition, we could experience a loss of sales and incur substantial cost in defending ourselves and indemnifying our partners and collaborators in patent infringement or proprietary rights violation actions brought against them. We could also incur substantial cost if we find it necessary to assert claims against third parties to prevent the infringement of our patents and proprietary rights by others. Participation in such infringement proceedings could have a material adverse effect on us, even if the eventual outcome was favorable.
We also rely on trade secrets, know-how and continuing technological advancement to maintain our competitive position with our technology, some of which is not patented. In addition, our success will depend in part on our ability to protect our trade secrets related to extracting, isolating and purifying paclitaxel, as well as our Gene Editing and Targeted Oncology programs. Although we have entered into confidentiality agreements with employees, consultants and collaborators, which contain assignment of invention provisions, we cannot assure that others will not gain access to these trade secrets, that such agreements will be honored or that we will be able to effectively protect our rights to unpatented trade secrets. Moreover, we cannot assure that others will not independently develop substantially equivalent proprietary information and techniques or otherwise gain access to our trade secrets.
In June 2001, we and Abbott filed a patent infringement suit in the United States District Court for the Western District of Pennsylvania against Mylan Laboratories, Inc. The suit alleges infringement of U.S. Patent numbers 5,733,888, 5,972,992, 5,977,164, 6,140,359, and 6,306,894 which relate to paclitaxel. Mylan has asserted defenses that if successful, would result in the invalidity or unenforceability of the patents. A finding of invalidity or unenforceability of the patents could have a material adverse affect on us.
Government Regulation and Product Approvals
Our paclitaxel is currently approved for commercial sale in the U.S., Australia, Israel and more than 25 other countries in the Middle East, Asia and Latin America. We are currently working to obtain clearance to market our paclitaxel in Europe. Although we initially received approval to market our product in Finland in 2002, that approval has been contested by Bristol on the grounds of data exclusivity, and is currently not enforceable. We believe that we will be approved to sell in the major European markets in 2004. Together with JCR, we are also planning to obtain approval to sell our paclitaxel in Japan.
Even in those countries where we have already obtained approval for a pharmaceutical product, we must also comply with numerous government regulations. For example, our manufacturing systems must conform to the FDA's regulations on current Good Manufacturing Practices. In complying with these
C-12
regulations, manufacturers must continue to expend time and resources in order to ensure compliance. Thus, even though we have regulatory approval for paclitaxel, our current and any future facilities are subject to periodic review and inspections by the FDA or other regulatory authorities. The FDA, the Australian TGA and the European EMEA have inspected our paclitaxel manufacturing facilities and have found them to be in compliance with applicable regulations and to be acceptable for the manufacture of bulk paclitaxel. Subsequent discovery of previously unknown problems with a product or our manufacturing facilities may result in restrictions, including withdrawal of the product from the market.
We have filed Drug Master Files (DMFs) and other documents describing portions of our proprietary manufacturing processes with regulatory agencies in the U.S., Australia and Europe, relating to the manufacture of our paclitaxel. Abbott and Mayne Pharma, referring to our DMFs, have received marketing approval for paclitaxel in the United States, Australia and other countries.
In addition to regulations regarding manufacture, we are subject to U.S. statutes and regulations applicable to exporting drugs. Those laws authorize the export of a drug without marketing approval in the U.S. to any country only if certain conditions are met.
The adoption by federal, state or local governments of significant new laws or regulations or a change in the interpretation or implementation of existing laws or regulations relating to environmental or other regulatory matters could increase the cost of producing products, delay regulatory approval or otherwise adversely affect our ability to produce or sell our paclitaxel or other products.
In addition to regulations enforced by the FDA, we are also subject to, among others, the regulations of the European Union, the U.S. Environmental Protection Agency, the Department of Interior (U.S. Fish and Wildlife Services and the Bureau of Land Management), the Department of Agriculture (U.S. Forest Service) and other countries and regulatory agencies. We are also subject to regulation under the Occupational Safety and Health Act, the Environmental Protection Act, the Toxic Substances Control Act, the Resource Conservation and Recovery Act and other present and future federal, state or local regulations. Our research and development activities involve the controlled use of hazardous materials, chemicals and various radioactive compounds.
Our manufacturing partners are also subject to such laws and regulations, both in the U.S. and in any other countries in which they operate. Failure by our manufacturing partners to comply with such laws and regulations could lead to an interruption in our supply of necessary raw materials.
Paclitaxel is our only therapeutic product which has been approved for commercial sale. All of our other product candidates, including our Sickle Cell Disease, Huntington's Disease, and Targeted Oncology drugs will need to undergo extensive regulatory review prior to commercial approvals. We are in the early stages of these programs and we can provide no assurance that we will be successful in our development efforts, nor can we provide assurance that we will be successful in obtaining regulatory approvals for these products.
Research, preclinical development, clinical trials, manufacturing and marketing activities are subject to regulation for safety, efficacy and quality by governmental authorities in the U.S. and other countries. Regulations govern, among other things, the testing, manufacture, safety, efficacy, labeling, storage, record keeping, approval, advertising and promotion of our products and product candidates. Product development and approval within this regulatory framework take a number of years and involve the expenditure of substantial resources.
The steps required before a pharmaceutical agent may be marketed in the U.S. include preclinical laboratory tests, animal pharmacology and toxicology studies and formulation studies, the submission of an Investigational New Drug Application (IND) to the FDA for human clinical testing, the carrying out of adequate and well-controlled human clinical trials to establish the safety and efficacy, the submission of an NDA to the FDA, and FDA approval of the NDA. In addition to obtaining FDA approval for
C-13
each product, each domestic drug manufacturing establishment must be registered with the FDA. Domestic drug manufacturing establishments are subject to regular inspections by the FDA and must comply with FDA regulations.
Preclinical studies include the laboratory evaluation ofin vitro pharmacology, product chemistry and formulation, as well as animal studies to assess safety. Preclinical safety tests must be conducted by laboratories that comply with FDA regulations. The results of some of the preclinical tests form a part of an IND along with the proposed clinical study, and chemistry and manufacturing information. The IND process may be costly.
Clinical trials are typically conducted in three sequential phases. In Phase I, the initial introduction of the drug into a small number of healthy volunteers is undertaken. The drug is evaluated for safety. The Phase I trial must also provide pharmacological data that is sufficient to design the Phase II trials. For certain drugs such as cancer drugs Phase I trials may be conducted in patients rather than in healthy volunteers. Clinical trials must be sponsored and conducted in accordance with good clinical practice.
Phase II trials involve studies in a limited patient population in order to obtain initial indications of the efficacy of the drug for specific, targeted indications, determine dosage tolerance and optimal dosage, and identify possible adverse affects and safety risks. When a compound is determined preliminarily to be effective and to have an acceptable safety profile in Phase II evaluation, Phase III trials can be undertaken to evaluate safety and efficacy further in expanded patient populations at geographically diverse clinical trial sites. Positive results in Phase II are no guarantee of positive results in Phase III.
The results of the clinical trials and manufacturing, toxicology and pharmacology information are submitted to the FDA in the form of an NDA. The approval of an NDA permits commercial-scale manufacturing, marketing, distribution, and sale of the drug in the U.S. The FDA may deny a new drug application filed by us or our collaborators if the applicable scientific and regulatory criteria are not satisfied. The FDA may require additional testing or information, and may require post-approval testing, surveillance and reporting to monitor the products. The FDA may ultimately decide that an NDA filed by us or our collaborators does not meet the applicable agency standards, and even if approval is granted, it can be limited or revoked.
The regulatory pathway for our therapeutic products, including our therapy for Sickle Cell Disease and any other therapeutic products and processes involving our Gene Editing technology, may involve additional regulatory review and requirements not outlined above.
Outside the U.S., our ability to market a product is contingent upon receiving a marketing authorization from the appropriate regulatory authority. This foreign regulatory approval process includes many of the same steps and uncertainties associated with FDA approval described above.
Marketing and Sales
Paclitaxel
Currently, paclitaxel is our only pharmaceutical product approved for commercial sale. Marketing and sales of paclitaxel in the U.S. are handled by Abbott, which will also conduct marketing and sales of paclitaxel in Canada, if approved. Marketing and sales in territories covered by our agreements with Mayne Pharma are conducted by Mayne Pharma and in Israel by Tzamal. Anticipated marketing and sales, if any, in Japan will be conducted by JCR. Currently, we have no sales force and have only limited marketing capabilities. Sales to Mayne Pharma and Abbott account for a substantial portion of our revenue. As a result, the loss of Mayne Pharma or Abbott as a customer or the failure of Mayne Pharma or Abbott to successfully market our paclitaxel could have a material adverse effect on us in the absence of a comparable alternative strategic alliance arrangement.
C-14
Competition
The biopharmaceutical industry is an expanding and rapidly changing industry characterized by intense competition for product sales, financing, executive talent and intellectual property. We compete with all entities developing and producing therapeutic agents, including those for cancer treatment, many of whom have much greater capital resources and research and development capabilities.
Within the paclitaxel segment of the cancer treatment industry, competitors' success in entering the paclitaxel market may reduce our market share and reduce the price we can charge for our paclitaxel, which could have a material adverse effect upon us. In addition, marketing is being handled exclusively by Abbott, Mayne Pharma and Tzamal within their territories. Regulatory approvals are being handled by Abbott in its territory and by Mayne Pharma in its non-European territory. Although we believe Abbott and Mayne Pharma have capable drug development and marketing abilities, we cannot assure that they will be capable or effective in gaining additional regulatory approvals on a timely basis, if at all, or be able to compete effectively with existing or new competitors within their territories.
Bristol is marketing paclitaxel commercially in the U.S., Australia, Canada, Europe and other territories. In addition, Aventis has developed a proprietary analog of paclitaxel, docetaxel, which is marketed under the trademark Taxotere®. Taxotere® is approved in the U.S., the European Union, Australia, Canada and other countries. Taxotere® is approved in the U.S. for treatment of patients with locally advanced or metastatic breast cancer after failure of prior chemotherapy, and patients with locally advanced or metastatic non-small cell lung cancer after failure of prior platinum-based chemotherapy. While treatment with Taxotere® may cause certain side effects not observed with paclitaxel, Taxotere® competes with paclitaxel, and thereby may reduce overall paclitaxel sales.
The FDA has approved several generic versions of paclitaxel. Currently Ivax Corporation, Mylan Laboratories, Inc. and Bedford Laboratories have approval to sell generic paclitaxel in the U.S. Other generic manufacturers may enter the U.S. market. In most cases, a European exclusivity period will end in 2003, ten years after Bristol's initial approval. However, Ivax is marketing paclitaxel for Kaposi's Sarcoma in Europe. We are aware of several other pharmaceutical companies that are in the process of developing generic paclitaxel in the U.S., Europe and elsewhere. Finally, academic and research organizations and pharmaceutical and biotechnology companies are pursuing, among other things, genetically engineered drugs, chemical synthesis and cell-tissue culture that may compete with our products or technology. In addition, certain companies are pursuing the production of paclitaxel and other taxanes from natural product extraction techniques.
Our competitors, most notably Bristol and Aventis, have substantially greater capital resources, research and development capabilities, manufacturing and marketing resources and experience than we do. We expect Bristol to compete intensely to maintain its dominance of the paclitaxel market. Generic competition has already caused a decline in the average price of paclitaxel, and if more competitors enter the market, the price of paclitaxel may erode even further. Our competitors may succeed in developing products that are more effective or less costly than any that may be developed by us and may receive regulatory approval before we do. Many companies and research institutions are also seeking means to obtain paclitaxel and taxanes from renewable biomass components of yew trees and other sources in order to increase paclitaxel yields, avoid environmental concerns and reduce the cost of biomass. In addition, we are aware of several potential competitors that have developed and patented or are developing various processes for producing paclitaxel and paclitaxel-related substances semisynthetically, which may allow such competitors to produce a lower-cost paclitaxel. The development by a third party of a cost-effective means to fully synthesize paclitaxel in commercial quantities or the manufacture of taxane derivatives or analogs that are more effective than paclitaxel in treating cancer could have a material adverse effect on us.
C-15
Research and Development
During the years ended December 31, 2002, 2001 and 2000, we spent approximately $16.1 million, $12.3 million, and $8.4 million respectively, on research and development activity for both paclitaxel and other product candidates. Research and development is expected to remain a significant expense of our business. Our research and development is expected to concentrate on the development of novel, targeted anti-cancer agents, and the development of novel genomic technologies, primarily in the area of Gene Editing, for applications in human therapeutics, diagnostics, agribiotechnology and pharmacogenomics. We anticipate bringing our Targeted Oncology program to the clinic during the first half of 2004. Our Sickle Cell Disease and Huntington's Disease programs are expected to go into the clinic during 2004 or the beginning of 2005. However, there can be no assurance that we will be able to achieve the timing of these programs. We also cannot estimate the cost of the effort necessary to complete the programs or the timing of commencement of material net cash inflows from these programs.
Foreign and Domestic Operations; Export Sales
The following table sets forth, for the past three years, sales, profitability (operating income (loss)), and identifiable assets attributable to our U.S. and foreign operations (in thousands):
| | Year Ended December 31, | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| | 2002 | 2001 | 2000 | ||||||||
| U.S. Sales to Unaffiliated Customers(1) | $ | 34,193 | $ | 15,711 | $ | 8,148 | |||||
| Operating Income (Loss) | |||||||||||
| U.S. | $ | (15,784 | ) | $ | (26,444 | ) | $ | (16,168 | ) | ||
| Canada | 13 | (73 | ) | (79 | ) | ||||||
| Identifiable Assets | |||||||||||
| U.S | $ | 42,435 | $ | 34,161 | $ | 34,959 | |||||
| Canada | 2,893 | 2,900 | 3,042 | ||||||||
- (1)
- Includes export sales into Australia of $7,330,000 in 2002, $7,576,000 in 2001 and $7,720,000 in 2000, as well as sales into Israel of $1,881,000 in 2002, $1,954,000 in 2001 and $288,000 in 2000.
Sales of our paclitaxel into foreign markets accounted for approximately 27% of our 2002 revenue, 60% of our 2001 revenue and 98% of our 2000 revenue.
A substantial portion of our sales and operations will continue to be subject to the risks associated with foreign business, including economic or political instability, shipping delays, fluctuations in foreign currency exchange rates and various trade restrictions, all of which could have a significant impact on our ability to deliver products on a competitive and timely basis. Future imposition of, or significant increases in, the level of customs duties, export quotas, drug regulatory restrictions or other regulatory or trade restrictions could have a material adverse effect on us.
Employees
As of February 28, 2003, we had 151 full-time equivalent employees. 21 of these employees hold Ph.D. or M.D. degrees. 52 employees were engaged in drug development, 10 in quality assurance, 35 in manufacturing, 46 in administration and finance, 5 in regulatory affairs, and 3 in legal. We believe that our relations with our employees are good.
We lease 54,000 square feet of space in Boulder, Colorado, used for executive offices, research and development and commercial manufacturing. We will be terminating this lease in two phases during
C-16
2003 and in turn, we expect to lease an additional 7,000 to 10,000 square feet of administrative space by mid-2003. We lease an additional 27,000 square feet of space in Boulder, used for manufacturing, and 20,000 square feet of warehouse space, the lease for which expires in April 2003 in Weld County, Colorado. We own five acres of undeveloped land in Longmont, Colorado. We lease 5,600 square feet in Newark, Delaware, used for research and development, 1,100 square feet in Allentown, Pennsylvania, used for research and development, and 2,100 square feet of office space in New York City used for administration.
In June 2001, we and Abbott filed a patent infringement suit in the United States District Court for the Western District of Pennsylvania against Mylan Laboratories, Inc. The suit alleges infringement of U.S. Patent numbers 5,733,888, 5,972,992, 5,977,164, 6,140,359 and 6,306,894, all of which relate to paclitaxel. Mylan has asserted defenses that, if successful, would result in the invalidity or unenforceability of the patents. A finding of invalidity or unenforceability of the patents could have a material adverse affect on us.
Item 4 Submission of Matters to Vote of Security Holders
None.
C-17
Item 5 Market for Registrant's Common Equity and Related Stockholder Matters
Market Information
During 2002 and prior, our common stock was traded in the Nasdaq National Market under the symbol "NPRO." The following table sets forth, for the periods indicated, the high and low closing sale prices for our common stock.
| | High | Low | |||||
|---|---|---|---|---|---|---|---|
| 2002 | |||||||
| Fourth Quarter | $ | 2.09 | $ | 0.61 | |||
| Third Quarter | 6.18 | 1.12 | |||||
| Second Quarter | 9.23 | 5.04 | |||||
| First Quarter | 12.10 | 8.48 | |||||
2001 | |||||||
| Fourth Quarter | $ | 13.00 | $ | 6.11 | |||
| Third Quarter | 10.62 | 6.00 | |||||
| Second Quarter | 11.88 | 6.44 | |||||
| First Quarter | 8.25 | 5.53 | |||||
On February 12, 2003, we announced that we had applied to transfer our common stock listing to the Nasdaq SmallCap Market. The reason for this action is that we received on February 6, 2003 a determination letter from Nasdaq Staff that we no longer met the $10.0 million shareholders' equity requirements for continued listing on the Nasdaq National Market. On February 28, 2003, the Nasdaq staff notified us that our SmallCap Market application was approved. Our common stock continues to trade under the current symbol, NPRO.
In addition, on January 28, 2003, the Nasdaq Staff notified us that the bid price of our common stock had closed below the required $1.00 per share for 30 consecutive trading days, and, accordingly, that we did not comply with Marketplace Rule 4450(a)(5). We will be provided until July 28, 2003, to regain compliance with this requirement.
Stockholders
As of December 31, 2002, we had 264 stockholders of record.
Dividends
To date, we have not paid any dividends on our common stock. We intend to retain future earnings, if any, to finance the operation and expansion of our business and, therefore, we do not anticipate paying any cash dividends on our common stock in the foreseeable future, if at all.
Sales of Unregistered Securities
During 2002, we issued 200,000 shares of common stock to the University of Delaware associated with a 20-year Gene Editing technology license. The license provides for research and patent funding commitments and payments in common stock. To date, we have issued 300,000 shares under the license agreement. Assuming we do not cancel the license, we will issue an additional 900,000 shares in 100,000 share-per-year increments on the license anniversaries. We may, at our option, accelerate the issuance dates. Certain milestones may also accelerate the issuance dates. The license is terminable at NaPro's option. If terminated, no further shares would be issued.
C-18
In February 2002, we sold 888,889 shares of common stock and $8.0 million principal amount of five-year 4% debentures convertible into common stock at $15 per share. As issuances to accredited investors not involving any public offering, the sales were exempt from registration under Section��4(2) of the Securities Act and Regulation D thereunder. In August 2002, we issued 47,973 shares of common stock in lieu of cash interest payments.
In 2001, we issued 24,618 shares of common stock upon exercise of stock options held by consultants, which included one of our former directors. As issuances to sophisticated investors not involving any public offering, the sales were exempt from registration under Section 4(2) of the Securities Act.
In April 2001, we sold 888,889 shares of common stock to Abbott upon achievement of a development milestone. As an issuance to an accredited investor not involving any public offering, the sale of common stock to Abbott was exempt from registration under Section 4(2) of the Securities Act and Regulation D thereunder.
C-19
Item 6 Selected Financial Data
The selected financial data presented below for each year in the five years ended December 31, 2002, are derived from our financial statements, which have been audited by Ernst & Young LLP, independent auditors, and are qualified by reference to such Financial Statements and Notes thereto. The data presented below should be read in conjunction with our consolidated financial statements at December 31, 2002 and 2001 and for each of the three years in the period ended December 31, 2002, and the related Notes thereto, "Management's Discussion and Analysis of Financial Condition and Results of Operations" and other financial information included elsewhere in this report.
| | Year Ended December 31, | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| | 2002 | 2001 | 2000 | 1999 | 1998 | ||||||||||||
| | (In thousands, except per share data) | ||||||||||||||||
| Statement of Operations Data: | |||||||||||||||||
| Product sales | $ | 34,193 | $ | 15,711 | $ | 8,148 | $ | 7,592 | $ | 4,952 | |||||||
| Expenses: | |||||||||||||||||
| Cost of sales | 23,976 | 19,354 | 6,835 | 6,320 | 6,263 | ||||||||||||
| Research and development | 16,112 | 12,312 | 8,356 | 6,014 | 3,898 | ||||||||||||
| General and administrative | 9,876 | 8,596 | 6,959 | 5,901 | 6,270 | ||||||||||||
| Asset writedowns | — | 1,966 | 2,245 | 146 | 1,899 | ||||||||||||
| 49,964 | 42,228 | 24,395 | 18,381 | 18,330 | |||||||||||||
| Operating loss | (15,771 | ) | (26,517 | ) | (16,247 | ) | (10,789 | ) | (13,378 | ) | |||||||
| Other income (expense): | |||||||||||||||||
| License fee income | 8,902 | 1,092 | — | 2,320 | 11,110 | ||||||||||||
| Interest income | 267 | 793 | 372 | 309 | 550 | ||||||||||||
| Interest expense | (2,063 | ) | (1,136 | ) | (750 | ) | (842 | ) | (902 | ) | |||||||
| Net loss | $ | (8,665 | ) | $ | (25,768 | ) | $ | (16,625 | ) | $ | (9,002 | ) | $ | (2,620 | ) | ||
| Net loss attributable to common stockholders | $ | (8,665 | ) | $ | (25,768 | ) | $ | (16,625 | ) | $ | (10,213 | ) | $ | (3,212 | ) | ||
| Basic and diluted net loss per common share | $ | (0.29 | ) | $ | (0.93 | ) | $ | (0.69 | ) | $ | (0.50 | ) | $ | (0.22 | ) | ||
| Weighted average common shares outstanding | 29,606 | 27,585 | 23,924 | 20,554 | 14,642 | ||||||||||||
Year Ended December 31, | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| | 2002 | 2001 | 2000 | 1999 | 1998 | |||||||||||
| | (In thousands) | |||||||||||||||
| Balance Sheet Data: | ||||||||||||||||
| Cash, cash equivalents and short-term securities | $ | 6,762 | $ | 10,144 | $ | 18,982 | $ | 1,937 | $ | 7,441 | ||||||
| Working capital | 18,843 | 13,582 | 23,168 | 2,915 | 7,121 | |||||||||||
| Total assets | 45,328 | 37,061 | 38,001 | 19,257 | 25,666 | |||||||||||
| Long term debt, net of current maturities | 19,861 | 19,846 | 14,953 | 4,723 | 80 | |||||||||||
| Deferred income, long term | 5,887 | 6,508 | — | — | — | |||||||||||
| Senior convertible debt, long term portion | — | — | — | — | 5,176 | |||||||||||
| Senior convertible redeemable preferred stock | — | — | — | — | 3,805 | |||||||||||
| Convertible debt | 5,151 | — | — | — | — | |||||||||||
| Minority interest | — | — | — | 622 | 622 | |||||||||||
| Accumulated deficit | (103,678 | ) | (95,013 | ) | (69,245 | ) | (52,620 | ) | (43,618 | ) | ||||||
| Stockholders' equity | 6,796 | 1,137 | 18,587 | 11,133 | 10,884 | |||||||||||
C-20
Item 7 Management's Discussion and Analysis of Financial Condition and Results of Operations
The following discussion and analysis provides information that management believes is relevant to an assessment and understanding of the results of operations of NaPro BioTherapeutics, Inc. You should read this discussion in conjunction with the Financial Statements and Notes included elsewhere in this report. Certain statements set forth below constitute "forward-looking statements" within the meaning of the Private Securities Litigation Reform Act of 1995, referred to as the "Reform Act." See "Special Note Regarding Forward Looking Statements."
General
We are a pharmaceutical company focused in three distinct areas: the production and sale of paclitaxel, an approved cancer drug; the development of Targeted Oncology products, and the development of novel genomic technologies, primarily in the area of Gene Editing for applications in human therapeutics, diagnostics, agribiotechnology and pharmacogenomics. Our first and only commercialized product is paclitaxel, a naturally occurring chemotherapeutic anti-cancer agent found in certain species of yew, orTaxus trees. In addition to our efforts with paclitaxel and genomics, we are also working on several types of compounds that we believe have promising activity as anti-cancer agents. We believe some of these agents function by novel mechanisms, which may increase their likelihood of success as new chemotherapeutics. We are also actively engaged in evaluating the in-licensing or purchase of potential new products and/or technologies, whether or not those products or technologies are derived from natural products. Our evaluation of new products and technologies may involve the examination of individual molecules, classes of compounds, or platform technologies, in both cancer and other therapeutic areas. Acquisitions of new products or technologies may involve the purchase of such products or technologies, or the acquisition of, or merger with, other companies.
We continue to incur substantial research and development expense related to the development of novel, targeted anti-cancer agents, and the development of novel genomic technologies, primarily in the area of Gene Editing, for applications in human therapeutics, diagnostics, agribiotechnology and pharmacogenomics,as well as the improvement of our paclitaxel yield, the reduction of our long-term cost of product and the development of our semisynthesis process. Accordingly, we have incurred significant losses, including losses of $8.7 million, $25.8 million and $16.6 million for the years ended December 31, 2002, 2001 and 2000, respectively. Our accumulated deficit was $103.7 million as of December 31, 2002. We anticipate that losses may continue until such time, if ever, as we are able to generate sufficient sales to support our development operations, including the research and development activity mentioned above.
Our ability to generate sufficient sales to support our operations currently depends primarily upon the successful commercialization of our worldwide paclitaxel program. Our strategy for that program has been to form strategic alliances through long-term exclusive agreements with established pharmaceutical companies. In 1999, we entered into an exclusive collaborative agreement of up to 20 years covering the U.S. and Canada with Abbott Laboratories (Abbott) to develop and commercialize one or more formulations of paclitaxel for the treatment of a variety of cancers. Under our agreement with Abbott, we are responsible for supply of bulk drug; development activity is conducted jointly. Abbott is responsible for finishing, regulatory filings, marketing, and sale of the finished drug product. Most primary decisions related to the paclitaxel development program are made by a joint Abbott-NaPro Development Committee. In March 2001, we and Abbott filed an ANDA with the FDA for paclitaxel. The FDA granted approval of this ANDA on May 8, 2002. We received an approval for a second ANDA on August 1, 2002, which provides for greater manufacturing flexibility for the generic paclitaxel product.
In connection with the Abbott agreement, we may receive total funding of up to $118.0 million in the form of up to $30.0 million in development milestones, up to $57.0 million in marketing milestones,
C-21
$20.0 million in secured debt, and $11.0 million in equity investments. Marketing milestones are based upon certain annual sales levels, the majority of which are unlikely to be achieved given the current pricing of paclitaxel. Through December 31, 2002, and excluding product sales, we received $40.0 million under the agreement, including $11.0 million in equity investments in exchange for 2,000,000 shares of our common stock, $9.0 million in development milestones, and $20.0 million in secured debt.
Of the $30.0 million of potential development-related milestone payments, we received $1.0 million upon execution of our agreement with Abbott in July 1999. With the approval of our ANDA, and upon commencement of commercial sales, we received an $8.0 million payment in May 2002. Thus, $21.0 million in possible development milestones remain. Additional development milestones will only be available should NaPro or Abbott undertake and successfully complete new development work, which is not currently anticipated.
We have received $20.0 million of secured debt from Abbott. The debt bears a primary interest rate of 6.5% and is due in full on the earlier of: (i) the second anniversary of the first sale of finished product by Abbott to a wholesaler or end-user customer following approval of finished product by the FDA (May 8, 2004); (ii) the termination of the Abbott agreement; or (iii) January 1, 2007. The debt is limited to a borrowing base of collateralized assets, recomputed monthly. The majority of our paclitaxel inventories, and specific assets which relate to the manufacture of paclitaxel, are collateralized as security for the debt. Abbott's only recourse in the event of our default on the debt is to these specific assets.
Contingent upon achieving certain commercial sales thresholds for all approved formulations of paclitaxel over several years, we may receive marketing milestone payments from Abbott of up to $57.0 million. Given the current pricing of paclitaxel, we believe that the majority of these annual sales thresholds are unlikely to be achieved.
Under terms of the agreement, Abbott purchases bulk drug from us. Abbott may terminate the agreement at any time with or without cause. Should Abbott terminate without cause, it is obligated to make certain payments to us and to give us twelve months prior written notice. Since the approval of generic paclitaxel in mid-2001, the selling price of paclitaxel has fallen significantly, and may continue to do so. We receive a fixed price payment of $500 per gram, upon delivery of paclitaxel to Abbott, plus a quarterly royalty payment, if any. The royalty payment is a percentage of sales recorded by Abbott less any product payment already paid to us. The percentage of sales varies each year based upon total sales in that year recorded by Abbott. We do not anticipate receiving significant royalty payments given the current price of the drug.
In 1992, we entered into a 20-year exclusive agreement with F.H. Faulding & Co., Ltd. (Faulding), a large Australian pharmaceutical company, for the clinical development, sale, marketing and distribution of paclitaxel. In October 2001, Faulding was acquired by Mayne-Nickless Limited, an Australian based health care provider and logistics operator. As a result, Faulding is now known as Mayne Pharma. Mayne Pharma actively markets anti-cancer pharmaceuticals and other health care products in Europe, Australia, Asia, South America, and other regions throughout the world. In 2000, we amended the Mayne Pharma agreement to, among other things, add additional countries to Mayne Pharma's exclusive territory. In 2001, we entered into a separate agreement with Mayne Pharma covering development and sale of paclitaxel in Europe. We cannot assure that we will receive regulatory approval in any of the major markets in Europe. Should we receive approval, Mayne Pharma will then market and sell our paclitaxel in Europe. We and Mayne Pharma will share equally the net sales of the product in Europe. Including the new agreement for Europe, the Mayne Pharma territory includes substantially all of the world other than the U.S., Canada, Japan, Israel, the former Soviet Union and parts of Africa. Mayne Pharma has received marketing approval for, and is selling paclitaxel as ANZATAX™ in more than 25 countries.
C-22
In July 2002, the European Patent Office, Opposition Division, ruled that NaPro's currently issued paclitaxel formulation patent in the European Patent Convention is invalid. We have appealed this ruling and expect the appeal to be heard within the next 36 months. We do not believe this decision will have any effect outside of the jurisdiction of the European Patent Office; nor will it affect our ability to enter the market in Europe as regulatory approvals are received. We also do not believe this patent decision will affect our revenues or earnings through calendar year 2003.
In January 2001, we received approval in Israel to sell paclitaxel under the trade name Biotax™. We have established an exclusive supply and distribution agreement with Tzamal Pharma (Tzamal) for the development and distribution of paclitaxel in Israel. The Israeli Ministry of Health has approved Biotax for use in treating a variety of cancers.
In June 2001, we and JCR Pharmaceuticals Co., Ltd. (JCR) entered into a mutually exclusive development, supply and distribution agreement for paclitaxel in Japan. Under the agreement, we will be responsible for manufacturing and supplying the finished drug. We and JCR will jointly be responsible for the clinical and regulatory program necessary for seeking approval to market paclitaxel in Japan. JCR will be responsible for the sales and distribution of the product in Japan upon its approval. We cannot assure, however, that we will receive regulatory approval of paclitaxel in Japan, or that we and our strategic partner will successfully market it.
Research and Development
Research and development are major activities for us. We discussed the nature and status of our research and development in depth in this Form 10-K annual report in Item 1-Business, in the General, Paclitaxel, Paclitaxel Strategic Alliances, Biomass, Paclitaxel Manufacturing, Targeted Oncology, Genomics, Patents and Proprietary Technology, Government Regulation and Product Approvals, Competition and Research and Development sections, and in the General section of this Management's Discussion and Analysis.
We have incurred the following expense on research and development projects, including process improvements in our paclitaxel business (in thousands):
| | 2002 | 2001 | 2000 | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Oncology | $ | 9,933 | $ | 10,186 | $ | 8,252 | |||
| Genomics | 6,179 | 2,126 | 104 | ||||||
| $ | 16,112 | $ | 12,312 | $ | 8,356 | ||||
Research and development is expected to remain a significant expense of our business. Our research and development activity is subject to change as we develop a better understanding of our projects and their prospects. We currently expect to concentrate on the development of novel, targeted anti-cancer agents, and the development of novel genomic technologies, primarily in the area of Gene Editing, for applications in human therapeutics, diagnostics, agribiotechnology and pharmacogenomics. We anticipate bringing our Targeted Oncology program to the clinic during the first half of 2004. Our Sickle Cell Disease and Huntington's Disease programs are expected to go into the clinic during 2004 or the beginning of 2005. However, there can be no assurance that we will be able to achieve the timing of these programs. We also cannot estimate the cost of the effort necessary to complete the programs or the timing of commencement of material net cash inflows from these programs. Continued development of these programs is dependent upon additional capital. We cannot assure that we will be able to obtain such capital on terms, which are acceptable to us. We have included a number of the risks and uncertainties associated with completing development on schedule in the Special Note Regarding Forward-looking Statements, below.
C-23
Year Ended December 31, 2002 Compared to Year Ended December 31, 2001
Product Sales. Product sales for 2002 were $34.2 million, an increase of $18.5 million from the prior year. The increase during 2002 was primarily attributable to shipments to Abbott as a result of our FDA approval on May 8, 2002. Sales to Abbott for the current year were $24.9 million, an increase of $18.8 million from 2001. Sales to Mayne Pharma for the current year were $7.3 million, a decrease of $200,000 from the prior year. Sales to Tzamal for the current year were $1.9 million, a decrease of $100,000 from the prior year. Future sales are difficult to predict and are expected to vary from year to year depending upon numerous demand and competitive factors. Among these factors are: the relationship of competitors with their customers, the relationship of Abbott, Mayne Pharma and Tzamal with its customers and the amount of competitor product in the distribution channel.
Cost of Sales. Cost of sales for 2002 was $24.0 million, an increase of $4.6 million from the prior year. The increase was primarily attributable to an increase in volume produced, which was partially offset by a decrease in unit production cost due to improved yields and increased productivity. As a result, the gross margin increased to 29.9% in 2002 from a negative 23.2% in the prior year. We continue to look for improvements and cost saving measures to improve our gross margin in future periods.
Research and Development Expense. Research and development expense for 2002 was $16.1 million, an increase of $3.8 million from 2001. Genomics research and development increased $4.1 million from 2001, while oncology research and development decreased $300,000 from the prior year. The increase was primarily due to increased staffing ($2.1 million), legal costs associated with patent development ($1.4 million) and occupancy costs associated with development facilities ($400,000). These amounts were partially offset by a reduction in supplies expenses ($400,000), as well as a one-time charge for development costs, which we incurred in the prior year.
General and Administrative Expense. General and administrative expense for 2002 was $9.9 million, an increase of $1.3 million from 2001. The increase was primarily due to increased staffing ($1.2 million), general legal expenses ($900,000), insurance ($200,000) and services provided by third parties ($200,000). These amounts were partially offset by reductions in a number of areas, including supplies and recruiting expenses.
License Fee Income. License fee income for 2002 was $8.9 million, an increase of $7.8 million from 2001. The increase was attributable to our receipt of a one-time milestone payment of $8.0 million from Abbott upon commencement of commercial sales in the United States.
Interest Income. Interest income for 2002 was $300,000, a decrease of $500,000 from the prior year. The decrease was attributable to lower average balances of interest-bearing investments, as well as lower interest rates.
Interest Expense. Interest expense for 2002 was $2.1 million, an increase of $1.0 million from the prior year. The increase was primarily attributable to the issuance of $8.0 million in convertible debt during the first quarter of 2002, including amortization of the discount attributable to the conversion feature.
Year Ended December 31, 2001 Compared to Year Ended December 31, 2000
Product Sales. Product sales for 2001 were $15.7 million, an increase of $7.6 million from the prior year. Sales to Abbott for 2001 were $6.1 million, an increase of $6.1 million from 2000. Sales to Mayne Pharma for 2001 were $7.6 million, a decrease of $100,000 from 2000. Sales to Tzamal for 2001 were $2.0 million, an increase of $1.7 million from 2000.
C-24
Cost of Sales. Cost of sales for 2001 was $19.4 million, an increase of $12.6 million from 2000. The increase was primarily attributable to a 141% increase in volume ($9.6 million) and an increase in unit production cost ($1.6 million). The unit production cost increased as we rapidly expanded capacity by modifying equipment for increased volume and by adding shifts, resulting in training and other start-up cost.
Research and Development Expense. Research and development expense for 2001 was $12.3 million, an increase of $3.9 million from 2000. Genomics research and development increased $2.0 million from 2000, while oncology research and development increased $1.9 million from the prior year.
General and Administrative Expense. General and administrative expense for 2001 was $8.6 million, an increase of $1.6 million from 2000. The increase is primarily attributable to increased staffing ($1.0 million) and increased consulting expense ($600,000).
Asset Writedowns. Asset writedowns for 2001 were $1.9 million, a decrease of $300,000 from 2000. In 2001 we recorded a $1.0 million writedown of production equipment in the Boulder extraction facility related to operations that we transferred to a lower cost third party vendor. We expensed preliminary engineering and design cost of a considered new production facility when we determined that we could expand capacity in our existing facilities sufficiently to meet near-term demand ($900,000).
The 2000 amount was attributable to prior construction cost that was not applicable in the redesign of the manufacturing facilities in Boulder, Colorado.
License Fee Income. License fee income for 2001 was $1.1 million. There was no similar amount in 2000. Although license fees and milestone payments are unusual and may be non-recurring, $7.5 million was received under the Mayne Pharma European agreement and $1.5 million under the JCR Japanese agreement. The 2001 income represents amortization of these payments.
Interest Income. Interest income for 2001 was $800,000, an increase of $400,000 from 2000. The increase was primarily attributable to higher overall balances of interest bearing investments.
Interest Expense. Interest expense for 2001 was $1.1 million, an increase of $300,000 from 2000. The increase was primarily attributable to a higher balance on the Abbott loan.
Liquidity and Capital Resources
Our capital requirements have been, and will continue to be, significant. As of December 31, 2002, we had a working capital balance of $18.8 million compared to a working capital balance of $13.6 million as of December 31, 2001. To date, we have funded our capital requirements primarily with the net proceeds of public offerings of common stock of approximately $21.1 million, with private placements of equity securities of approximately $62.6 million, with the exercise of warrants and options of $8.0 million and with net borrowing of $27.8 million.
In February 2002, we sold privately $8.0 million of common stock issued at $9 per share and $8.0 million principal of five-year 4% debentures convertible into common stock at $15 per share to TL Ventures V, L.P. and one of its affiliated funds. No placement agent was involved in the transaction. The net proceeds were $15.6 million. As part of this transaction, we recorded a discount attributable to the conversion feature of the convertible debentures in the amount of $3.1 million, which is being amortized over the term of the debentures. During the first quarter of 2002, we filed a registration statement with the Securities and Exchange Commission to register the resale of the common stock, common stock issuable upon conversion of the debentures and common stock issuable in lieu of cash interest on the debentures under the Securities Act of 1933. In July 2002, the registration statement
C-25
was declared effective by the SEC. NaPro may pay the debenture interest in cash or common stock at its option. The debentures automatically convert into common stock during the first two years if the average price of the common stock for 15 trading days exceeds $22, and during the last three years if the average price for 30 trading days exceeds $16. If we have insufficient funds to repay the debentures in full at maturity or upon acceleration, the holders of the debentures may elect to convert the outstanding principal balance and any unpaid accrued interest on the debentures into shares of common stock at a conversion price equal to the then current 20 day average trading price of the common stock. The conversion price of the common stock in such a circumstance could be materially below the price at which we otherwise would be willing to issue our common stock. Issuance of common stock at such a price could result in substantial dilution to our other stockholders and could have a material adverse effect on the market price of our common stock.
In December 2002, we acquired the genomics business of Pangene Corporation for $1.3 million in cash and assumption of debt obligations totaling approximately $65,000. The acquisition consisted primarily of patents and intellectual property relating to Pangene's homologous recombination technology and gene isolation and service business, physical assets, instrumentation, software, customer relationships and third-party licenses. The purchase price was allocated to intangible assets totaling $987,000, fixed assets and software of $183,000, and other current assets of $44,000. In process research and development of $151,000 was expensed at the time of the acquisition. We will amortize intangible assets over 5 to 15 years, and fixed assets and software over 3 years. In connection with this agreement, we acquired additional patents in January 2003 in the amount of $400,000.
In November 2000, we entered into a 20-year Gene Editing technology license with the University of Delaware and Thomas Jefferson University relating to the use of proprietary oligonucleotides (DNA fragments) designed to precisely alter genes in humans, animals, plants, viruses and microbes. The license provides for research and patent funding commitments and payments in common stock. To date, we have issued 300,000 shares under the license agreement. Assuming we do not cancel the license, we will issue an additional 900,000 shares in 100,000 share-per-year increments on the license anniversaries. We may, at our option, accelerate the issuance dates. Certain milestones may also accelerate the issuance dates. The license is terminable at NaPro's option. If terminated, no further shares would be issued. We have committed to fund at least $300,000 in research under this agreement during 2003.
We believe existing capital and projected 2003 sales, as well as the conversion of current inventory and accounts receivable, will provide adequate capital to fund our operations and capital expenditures in 2003. However, pharmaceutical development is a costly and time consuming process. We may in-license or purchase new products or technologies. The cost of acquiring and developing such resources, and related capital expenditures, may be very large. As a result, we may seek additional capital. We cannot assure that we will be able to obtain such capital on terms, which are acceptable to us.
Working Capital and Cash Flow Cash and cash equivalents decreased $3.4 million to $6.8 million for the year ended December 31, 2002 from $10.1 million at December 31, 2001. During 2002, net cash used in operating activities of $12.9 million and in investing activities of $6.1 million was partially offset with net cash provided by financing activities of $15.6 million.
Inventory was $13.2 million at December 31, 2002, of which $3.8 million is classified as non-current. Non-current inventory consists of raw materials which are not expected to be utilized during 2003, due to the timing of biomass harvests and anticipated future changes to our production process. The amount of inventory is dependent on a number of factors, including the shipping requirements of our strategic partners, our production planning for meeting those needs, and the timing of seasonal biomass harvests. Inventory balances may vary significantly during product development, launch periods and during seasonal harvests.
C-26
Accounts receivable was $9.3 million at December 31, 2002. We anticipate that the level of our accounts receivable will decrease significantly during 2003.
Capital Expenditures We spent $4.8 million during 2002 for capital projects. These expenditures primarily included manufacturing capacity expansion, plantation cost and laboratory equipment purchases.
The amount and timing of future capital expenditures will depend upon numerous factors, including:
- •
- the development of new products;
- •
- the acquisition of new products or technologies;
- •
- the cost of manufacturing resources for new products;
- •
- the nature of our relationship with our strategic partners;
- •
- the progress of our research and development programs;
- •
- changes in manufacturing processes;
- •
- the magnitude and scope of these activities;
- •
- competing technological and marketing developments; and
- •
- changes in or terminations of existing strategic relationships.
We anticipate a significant reduction of capital expenditures during 2003. The primary focus of capital spending during 2003 is expected to be in our research and development areas. Although we may seek additional long-term financing to fund capital expenditures, we cannot assure that we can obtain such financing on terms, which are acceptable to us.
Net Operating Loss Carryforwards As of December 31, 2002, we had approximately $87.0 million of net operating loss carryforwards to offset future taxable income. Tax law provides limits on the utilization of net operating loss carryforwards if there has been a "change of ownership" as described in Section 382 of the Internal Revenue Code. Such a change of ownership may limit our utilization of our net operating loss carryforwards, and could be triggered by sales of securities by us or our stockholders.
Business Development Activities In the normal course of our business, we investigate, evaluate, and discuss licensing relationships, acquisitions, and other business combination opportunities in the pharmaceutical and genomics businesses. In the event we enter into any such relationships or transactions, we may consider using available cash, issuing equity securities or increasing our debt. Such transactions could materially affect our capital structure.
Critical Accounting Policies
We have identified certain accounting policies as critical to our business operations and the understanding of our results of operations. The impact and any associated risks related to these policies on our business operations is discussed throughout this Item 7 where such policies affect our reported and expected financial results. For a detailed discussion on the application of these and other accounting policies, see Note 1 in the Notes to our Consolidated Financial Statements.
Use of Estimates Policy: The preparation of financial statements in conformity with accounting principles generally accepted in the United States requires management to make estimates and assumptions that affect the amounts reported in the financial statements and accompanying notes. Actual results may differ from those estimates.
C-27
Inventory Policy: We state inventory at the lower of cost or market using the first-in, first-out method. Inventory, which is not expected to be utilized during 2003, has been classified as a non-current asset. We periodically review the composition of our inventory for slow-moving and obsolete items. If deemed appropriate, we record a reserve against those inventory items.
Plantation Cost Policy: We have determined the cultivation of renewable sources of biomass to be used in the manufacture of paclitaxel is a technically feasible business strategy. Paclitaxel is a naturally occurring cancer-fighting compound found in certain species of yew (Taxus) trees. We capitalize plantation expenditures incurred prior to the first commercial harvest on each planting of trees. Plantation expenditures include the acquisition cost of trees and the related cost of planting and growing. We deplete such cost using estimated units of production over the expected number of annual harvests. The number and timing of harvests can vary depending on product demand.
Long-Lived Assets Policy: In accordance with Statement of Financial Accounting Standards No. 144 (SFAS 144), Accounting for the Impairment of Long-Lived Assets, we review the carrying amount of long-lived assets when facts and circumstances suggest they may be impaired. If this review indicates long-lived assets will not be recoverable as determined based on the undiscounted cash flow estimated to be generated by these assets, we reduce the carrying amount of these long-lived assets to estimated fair value or discounted cash flow, as appropriate.
Comment on Inventory, Plantation Cost and other Long-Lived Assets Policies: We have invested substantial amounts of capital in these assets. Our accurate measurement and evaluation of these assets is important to the fair presentation of our financial position and results of operations. We follow very specific and detailed guidelines regarding capitalization of cost, market valuation, and potential impairment of these assets, and provide any necessary allowances as required.
The table below summarizes our future contractual obligations (in thousands):
| | Total | Less than 1 year | 1-3 years | 4-5 years | After 5 years | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Notes Payable | $ | 20,122 | $ | 102 | $ | 20,020 | $ | — | $ | — | |||||
| Convertible Debentures | 8,000 | — | — | 8,000 | — | ||||||||||
| Operating leases | 3,844 | 1,445 | 2,163 | 151 | 85 | ||||||||||
| Total | $ | 31,966 | $ | 1,547 | $ | 22,183 | $ | 8,151 | $ | 85 | |||||
Special Note Regarding Forward-looking Statements
The statements in this report that are not historical facts are forward-looking statements that represent management's beliefs and assumptions based on currently available information. Forward-looking statements can be identified by the use of words such as "believes", "intends", "estimates", "may", "will", "should", "anticipated", "expected" or comparable terminology or by discussions of strategy. Such forward-looking statements may include, among others:
- •
- statements concerning our plans, objectives and future economic prospects, such as matters relative to developing new products;
- •
- the timing and amount of sales of paclitaxel to our strategic partners;
- •
- the availability of patent and other protection for our intellectual property;
- •
- the completion of preclinical studies, clinical trials and regulatory filings;
- •
- the prospects for, and timing of, regulatory approvals;
- •
- the need and plans for, and availability of, additional capital;
C-28
- •
- the amount and timing of capital expenditures;
- •
- the timing of product introductions and sales;
- •
- the availability of raw materials;
- •
- prospects for future operations; and
- •
- other statements of expectations, beliefs, future plans and strategies, anticipated events or trends and similar expressions concerning matters that are not historical facts.
Although we believe that the expectations reflected in such forward-looking statements are reasonable, we cannot assure you that these expectations will prove to be correct. Such forward-looking statements involve known and unknown risks, uncertainties and other factors, which may cause our actual results, performance or achievements, or industry results, to be materially different from any future results, performance or achievements expressed or implied by such forward-looking statements. Such factors include, among other things, the following:
- •
- competition from Bristol-Myers Squibb Company, IVAX Corporation, Aventis S.A., Mylan Laboratories, Inc., and other producers of paclitaxel and other drugs;
- •
- the ability to obtain European or Japanese regulatory approvals for paclitaxel or a delay in such approvals;
- •
- technological advances in cancer treatment and drug development that may make paclitaxel obsolete;
- •
- the risks associated with patent litigation;
- •
- the ability to obtain rights to technology;
- •
- competition in the field of genomics therapeutics, reagents, services and diagnostics;
- •
- the ability to obtain, maintain and enforce patents;
- •
- the ability to maintain trade secrets;
- •
- the ability to obtain raw materials and commercialize manufacturing processes;
- •
- the effectiveness of other pharmaceuticals we develop in treating disease;
- •
- the results of preclinical and clinical studies for ourselves and our competitors;
- •
- the ability to fund preclinical and clinical studies;
- •
- the results of research and development activities;
- •
- the ability to fund research and development activities;
- •
- the ability to purchase or license new products;
- •
- the successful development of new products;
- •
- the business abilities and judgment of our management and other personnel;
- •
- the ability to hire and retain skilled personnel to perform research and development and to run our manufacturing operations;
- •
- the ability of contract manufacturers to perform adequately under anticipated contracts;
- •
- the decision-making processes of regulatory agencies;
- •
- changes in and compliance with governmental regulations;
C-29
- •
- the effect of capital market conditions and other factors on capital availability;
- •
- the ability of Abbott, Mayne Pharma, Tzamal and JCR to perform their obligations under their existing agreements with us;
- •
- our ability to perform our obligations under our existing and future agreements;
- •
- our limited relevant operating history upon which an evaluation of our prospects can be made;
- •
- the effect on sales, cash flow and earnings from foreign exchange rate fluctuations;
- •
- our ability to meet all applicable Nasdaq or Nasdaq SmallCap requirements for continued listing of our common stock;
- •
- adverse economic and general business conditions; and
- •
- other factors referenced in this report.
Should one or more of the risks mentioned above materialize (or the consequences of such a development worsen), or should the assumptions underlying our forward-looking statements prove incorrect, actual results could differ materially from those forecasted or expected. These factors are not intended to be an all-inclusive enumeration of the business risks we face. Reference is also made to the risk factors discussed in Amendment No. 4 to our Registration Statement (No. 333-82810) filed with the Securities and Exchange Commission on July 1, 2002. The forward-looking statements included in this report represent our view as of the date of this report. The reader should not assume that the statements made herein remain accurate at any future date. We do not intend to update these statements whether as a result of new information, future events or otherwise and undertake no duty to any person to make any update under any circumstance.
Item 7A Quantitative and Qualitative Disclosures about Market Risk.
During 2002, we sold a significant amount of paclitaxel to Mayne Pharma. Under the Mayne Pharma agreement, Mayne Pharma pays us a fixed percentage of their sales price for paclitaxel. Each year, Mayne Pharma estimates the sales price it will receive in the upcoming year, and, based upon that estimate, we determine the price we will charge Mayne Pharma. We recognize the corresponding sales at the time of shipment to Mayne Pharma. However, Mayne Pharma's actual selling price may differ from that estimated. Pursuant to the agreement, Mayne Pharma is obligated to provide us with their calculation of the actual sales price for sales made during the preceding year, and an adjustment is calculated that may increase or decrease our sales of products to Mayne Pharma during that year.
Mayne Pharma's sales are made in the currencies of each of the countries in which it sells paclitaxel. As a result, our sales are affected by fluctuations in the value of these various foreign currencies relative to the U.S. dollar. In the past, currency fluctuations were a significant factor in reductions in the price we charged Mayne Pharma. If changes in foreign currency markets continue to cause a decrease in the price per gram we receive from Mayne Pharma, there could be a material adverse effect on our earnings and cash flow. We currently record a reserve for potential Mayne Pharma pricing adjustments based upon historical experience and periodic reporting provided by Mayne Pharma. Our sales to JCR, if and when they occur, will be subject to the same risk.
Certain statements set forth in Item 7A may constitute "forward-looking statements." See "Special Note Regarding Forward-looking Statements."
Item 8 Financial Statements and Supplementary Data
The information required by this item begins at Page F-1.
Item 9 Changes in and Disagreements with Accountants on Accounting and Financial Disclosures
Not Applicable.
C-30
Item 10 Directors and Executive Officers of the Registrant
The information concerning our directors and executive officers is incorporated by reference to the section entitled "Election of Directors" in our definitive Proxy Statement with respect to our 2003 Annual Meeting of Stockholders to be filed with the SEC pursuant to Regulation 14A within 120 days after the end of the fiscal year covered by this report (the Proxy Statement).
Item 11 Executive Compensation
The section entitled "Executive Compensation" appearing in our Proxy Statement is incorporated herein by reference, except for such information as need not be incorporated by reference under rules promulgated by the SEC.
Item 12 Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters
The section labeled "Security Ownership of Directors and Executive Officers and Certain Beneficial Owners" appearing in our Proxy Statement is incorporated herein by reference.
Item 13 Certain Relationships and Related Transactions
The section labeled "Certain Relationships and Related Transactions" appearing in our Proxy Statement is incorporated herein by reference.
Item 14 Controls and Procedures
(a) The Company maintains a system of disclosure controls and procedures. The term "disclosure controls and procedures," as defined by regulations of the SEC, means controls and other procedures that are designed to ensure that information required to be disclosed in the reports that the Company files or submits to the SEC under the Securities Exchange Act of 1934, as amended (the Act), is recorded, processed, summarized and reported, within the time periods specified in the SEC's rules and forms. Disclosure controls and procedures include, without limitation, controls and procedures designed to ensure that information required to be disclosed by the Company in the reports that it files or submits to the SEC under the Act is accumulated and communicated to the Company's management, including its principal executive officer and its principal financial officer, as appropriate to allow timely decisions to be made regarding required disclosure. Within the 90 days prior to the date of this report, the Company carried out an evaluation, under the supervision and with the participation of the Company's management, including the Company's Chief Executive Officer and Chief Financial Officer, of the effectiveness of the design and operation of the Company's disclosure controls and procedures. Based upon that evaluation, the Company's Chief Executive Officer and Chief Financial Officer concluded that the Company's disclosure controls and procedures are effective in timely alerting them to material information relating to the Company required to be included in the Company's periodic SEC filings.
(b) The Company also maintains a system of internal controls. The term "internal controls," as defined by the American Institute of Certified Public Accountants' Codification of Statement on Auditing Standards, AU Section 319, means controls and other procedures designed to provide reasonable assurance regarding the achievement of objectives in the reliability of the Company's financial reporting, the effectiveness and efficiency of the Company's operations and the Company's compliance with applicable laws and regulations. There were no significant changes in our internal
C-31
controls or to our knowledge, in other factors that could significantly affect our internal controls and procedures subsequent to the date of such evaluation.
It should be noted that any system of controls, however well designed and operated, can provide only reasonable, and not absolute, assurance that the objectives of the system are met. In addition, the design of any control system is based in part upon certain assumptions about the likelihood of future events. Because of these and other inherent limitations of control systems, there can be no assurance that any design will succeed in achieving its stated goals under all potential future conditions, regardless of how remote.
Item 15 Exhibits, Financial Statement Schedules, and Reports on Form 8-K
Financial Statements
The Financial Statement Index is on Page F-1.
Financial Statement Schedules
All schedules are omitted because they are not applicable or not required or because the information is included in the consolidated financial statements or the notes thereto.
Exhibits and Reports on Form 8-K
We filed a Current Report on Form 8-K dated January 24, 2003 reporting the change in our fiscal year.
We filed a Current Report on Form 8-K dated February 13, 2003 reporting the application for the transfer of our common stock to the Nasdaq SmallCap Market.
| Exhibit Number | Description of Exhibit | |
|---|---|---|
| 3.1 | Amended and Restated Certificate of Incorporation of the Company, as amended August 2, 1996(1) | |
3.2 | Certificate of Amendment dated September 29, 1998 to the Amended and Restated Certificate of Incorporation of the Company(2) | |
3.3 | Certificate of Amendment dated September 13, 2000 to the Amended and Restated Certificate of Incorporation of the Company(3) | |
3.4 | Bylaws of the Company as amended through December 2000(4) | |
3.5 | Certificate of Designation for Convertible Preferred Stock, Series A(5) | |
3.6 | Certificate of Designation for Series B Junior Participating Preferred Stock(6) | |
3.7 | Certificate of Designation for Series C Senior Convertible Preferred Stock(7) | |
4.1 | Common Stock Certificate(8) | |
4.2 | Amended and Restated Rights Agreement dated September 25, 2001 between the Company and American Stock Transfer and Trust Company, as Rights Agent(9) | |
4.3 | Form of 4% Convertible Subordinated Debenture due 2007(10) | |
6 | The Certificate of Incorporation and Bylaws of the Company are included as Exhibits 3.1 - 3.4 | |
C-32
10.1 | * | Company's 1993 Stock Option Plan(8) |
10.2 | * | Amendment dated December 11, 2000 to the Company's 1993 Stock Option Plan(4) |
10.3 | * | Company's 1994 Long-Term Performance Incentive Plan, as amended and restated effective June 21, 2001(11) |
10.4 | Company's 1998 Stock Incentive Plan as amended and restated effective September 25, 2001(11) | |
10.5 | * | Employment Agreement effective October 1, 2001 between the Company and Leonard Shaykin(16) |
10.6 | * | Employment Agreement effective October 1, 2001 between the Company and Sterling Ainsworth(16) |
10.7 | * | Employment Agreement effective October 1, 2001 between the Company and Patricia Pilia(16) |
10.8 | * | Employment Agreement effective October 1, 2001 between the Company and Gordon Link(16) |
10.9 | * | Employment Agreement effective October 1, 2001 between the Company and Kai Larson(16) |
10.10 | * | Employment Agreement effective October 1, 2001 between the Company and David Denny(16) |
10.11 | * | Employment Agreement effective October 1, 2001 between the Company and James McChesney(16) |
10.12 | * | Employment Agreement effective October 1, 2001 between the Company and Steve Bannister(16) |
10.13 | * | Employment Agreement effective October 1, 2001 between the Company and Brenda Fielding(16) |
10.14 | Second Amended and Restated Master Agreement dated June 5, 2000 between the Company and F.H. Faulding & Co., Ltd.(12) | |
10.15 | European Agreement dated March 2, 2001 between the Company and F.H. Faulding & Co., Ltd.(4) | |
10.16 | Development, License and Supply Agreement dated July 23, 1999 by and between the Company and Abbott Laboratories(14) | |
10.17 | Loan and Security Agreement dated July 23, 1999 by and between the Company and Abbott Laboratories(14) | |
10.18 | Stock Purchase Agreement dated July 23, 1999 by and between the Company and Abbott Laboratories(14) | |
10.19 | Amendment dated June 23, 2000 to Development, License and Supply Agreement by and between the Company and Abbott Laboratories(15) | |
10.20 | Amendment dated June 23, 2000 to Stock Purchase Agreement by and between the Company and Abbott Laboratories(15) | |
10.21 | Amendment dated November 28, 2001 to Development, License and Supply Agreement by and between the Company and Abbott Laboratories(16) | |
C-33
10.22 | License Agreement dated November 21, 2000 by and between the Company and The University of Delaware and Thomas Jefferson University(4) | |
10.23 | + | Release and License Agreement dated November 28, 2001 by and between Bristol-Meyers Squibb Co. and the Company(16) |
10.24 | Lease dated October 16, 1995 between the Company and Gunbarrel Facility L.L.C.(13) | |
10.25 | First Amendment to Lease November 27, 1995 between the Company and Gunbarrel Facility L.L.C.(13) | |
10.26 | Form of Subscription Agreement including Registration Rights(4) | |
10.27 | Securities Purchase Agreement dated February 13, 2002 between T/L Ventures, Inc. and the Company(10) | |
10.28 | Registration Rights Agreement dated February 13, 2002 between T/L Ventures, Inc. and the Company(10) | |
10.29 | Agreement dated November 7, 2001 between R. J. Reynolds Tobacco Company and the Company(16) | |
10.30 | Agreement dated December 31, 2002 between Pangene Corporation and the Company | |
10.31 | Fourth Amendment to lease between the Company and Gunbarrel Facility L.L.C. | |
21.1 | List of Subsidiaries(16) | |
23.1 | Consent of Ernst & Young LLP | |
99 | Certification of Chief Executive Officer and Chief Financial Officer pursuant to Section 906 of the Public Company Accounting Reform and Investor Protection Act of 2002 |
- *
- A management compensation plan.
- +
- Portions have been omitted pursuant to a request for confidential treatment.
- (1)
- Incorporated herein by reference to the Company's Annual Report on Form 10-K filed with the Commission for the year ended December 31, 1996 (File No. 0-24320).
- (2)
- Incorporated herein by reference to the Company's Annual Report on Form 10-K for the year ended December 31, 1998 (File No. 0-24320).
- (3)
- Incorporated herein by reference to the Company's Quarterly Report on Form 10-Q for the period ending September 30, 2000 (File No. 0-24320).
- (4)
- Incorporated herein by reference to the Company's Annual Report on Form 10-K for the year ended December 31, 2000 (File No. 0-24320).
- (5)
- Incorporated herein by reference to the Company's Quarterly Report on Form 10-Q for the quarter ending June 20, 1995 (File No. 0-24320)
- (6)
- Incorporated herein by reference to the Company's Current Report on Form 8-K dated November 8, 1996 (File No. 0-24320).
- (7)
- Incorporated herein by reference to the Company's Registration Statement on Form S-3 filed on December 16, 1997 (File No. 333-42419)
- (8)
- Incorporated herein by reference to the Registration Statement on Form S-1 of the Company, filed with the Commission on July 24, 1994 (File No. 33-78016).
C-34
- (9)
- Incorporated herein by reference to the Registration Statement on Form 8-A12G/A of the Company, filed with the Commission on October 23, 2001 (File No. 0-24320)
- (10)
- Incorporated herein by reference to the Company's Current Report on Form 8-K, dated February 13, 2002 (File No. 0-24320).
- (11)
- Incorporated herein by reference to the Company's Quarterly Report on Form 10-Q for the period ending September 30, 2001 (File No. 0-24320).
- (12)
- Incorporated herein by reference to the Company's Quarterly Report on Form 10-Q for the period ending June 30, 2000 (File No. 0-24320).
- (13)
- Incorporated herein by reference to the Company's Annual Report on Form 10-K for the year ended December 31, 1995 (File No. 0-24320).
- (14)
- Incorporated herein by reference to the Company's Current Report on Form 8-K, dated July 23, 1999 (File No. 0-24320).
- (15)
- Incorporated herein by reference to the Company's Quarterly Report on Form 10-Q for the period ending June 30, 2000 (File No. 0-24320).
- (16)
- Incorporated herein by reference to the Company's Annual Report on Form 10-K for the year ended December 31, 2001 (File No. 0-24320).
C-35
Pursuant to Section 13 of the Securities Exchange Act of 1934, NaPro caused this report on Form 10-K to be signed on its behalf.
| NAPRO BIOTHERAPEUTICS, INC. | ||
| March 26, 2003 | ||
| By: /s/ LEONARD P. SHAYKIN Leonard P. Shaykin Chairman of the Board of Directors, Chief Executive Officer |
Pursuant to the Exchange Act, this report has been signed on behalf of NaPro BioTherapeutics, Inc. and in the capacities indicated.
| /s/ LEONARD P. SHAYKIN Leonard P. Shaykin | Chairman of the Board of Directors, Chief Executive Officer | March 26, 2003 | ||
/s/ STERLING K. AINSWORTH Sterling K. Ainsworth, Ph.D. | Vice Chairman, President, Chief Scientific Officer, Director | March 26, 2003 | ||
/s/ PATRICIA A. PILIA Patricia A. Pilia, Ph.D. | Executive Vice President, Director | March 26, 2003 | ||
/s/ GORDON LINK Gordon Link | Senior Vice President, Chief Financial Officer (Principal Financial Officer) | March 26, 2003 | ||
/s/ SANFORD D. GOLDBERG Sanford D. Goldberg | Controller (Principal Accounting Officer) | March 26, 2003 | ||
/s/ EDWARD L. ERICKSON Edward L. Erickson | Director | March 26, 2003 | ||
/s/ GEORGE M. GOULD George M. Gould | Director | March 26, 2003 | ||
Arthur H. Hayes, Jr., M.D. | Director | March 26, 2003 | ||
/s/ MARC J. OSTRO Marc J. Ostro, Ph.D. | Director | March 26, 2003 | ||
/s/ RICHARD N. PERLE The Honorable Richard N. Perle | Director | March 26, 2003 | ||
/s/ ROBERT E. POLLACK Robert E. Pollack, Ph.D. | Director | March 26, 2003 |
C-36
I, Leonard P. Shaykin, certify that:
- 1.
- I have reviewed this annual report on Form 10-K of NaPro BioTherapeutics, Inc.;
- 2.
- Based on my knowledge, this annual report does not contain any untrue statement of a material fact or omit to state a material fact necessary to make the statements made, in light of the circumstances under which such statements were made, not misleading with respect to the period covered by this annual report;
- 3.
- Based on my knowledge, the financial statements, and other financial information included in this annual report, fairly present in all material respects the financial condition, results of operations and cash flows of the registrant as of, and for, the periods presented in this annual report;
- 4.
- The registrant's other certifying officers and I are responsible for establishing and maintaining disclosure controls and procedures (as defined in Exchange Act Rules 13a-14 and 15d-14) for the registrant and we have:
- a.
- designed such disclosure controls and procedures to ensure that material information relating to the registrant, including its consolidated subsidiaries, is made known to us by others within those entities, particularly during the period in which this annual report is being prepared;
- b.
- evaluated the effectiveness of the registrant's disclosure controls and procedures as of a date within 90 days prior to the filing date of this annual report (the Evaluation Date); and
- c.
- presented in this annual report our conclusions about the effectiveness of the disclosure controls and procedures based on our evaluation as of the Evaluation Date;
- 5.
- The registrant's other certifying officers and I have disclosed, based on our most recent evaluation, to the registrant's auditors and the audit committee of registrant's board of directors (or persons performing the equivalent function):
- a.
- all significant deficiencies in the design or operation of internal controls which could adversely affect the registrant's ability to record, process, summarize and report financial data and have identified for the registrant's auditors any material weaknesses in internal controls; and
- b.
- any fraud, whether or not material, that involves management or other employees who have a significant role in the registrant's internal controls; and
- 6.
- The registrant's other certifying officers and I have indicated in this annual report whether or not there were significant changes in internal controls or in other factors that could significantly affect internal controls subsequent to the date of our most recent evaluation, including any corrective actions with regard to significant deficiencies and material weaknesses.
Date: March 26, 2003
| /s/ LEONARD P. SHAYKIN Leonard P. Shaykin Chairman of the Board of Directors Chief Executive Officer |
C-37
I, Gordon Link, certify that:
- 1.
- I have reviewed this annual report on Form 10-K of NaPro BioTherapeutics, Inc.;
- 2.
- Based on my knowledge, this annual report does not contain any untrue statement of a material fact or omit to state a material fact necessary to make the statements made, in light of the circumstances under which such statements were made, not misleading with respect to the period covered by this annual report;
- 3.
- Based on my knowledge, the financial statements, and other financial information included in this annual report, fairly present in all material respects the financial condition, results of operations and cash flows of the registrant as of, and for, the periods presented in this annual report;
- 4.
- The registrant's other certifying officers and I are responsible for establishing and maintaining disclosure controls and procedures (as defined in Exchange Act Rules 13a-14 and 15d-14) for the registrant and we have:
- a.
- designed such disclosure controls and procedures to ensure that material information relating to the registrant, including its consolidated subsidiaries, is made known to us by others within those entities, particularly during the period in which this annual report is being prepared;
- b.
- evaluated the effectiveness of the registrant's disclosure controls and procedures as of a date within 90 days prior to the filing date of this annual report (the Evaluation Date); and
- c.
- presented in this annual report our conclusions about the effectiveness of the disclosure controls and procedures based on our evaluation as of the Evaluation Date;
- 5.
- The registrant's other certifying officers and I have disclosed, based on our most recent evaluation, to the registrant's auditors and the audit committee of registrant's board of directors (or persons performing the equivalent function):
- a.
- all significant deficiencies in the design or operation of internal controls which could adversely affect the registrant's ability to record, process, summarize and report financial data and have identified for the registrant's auditors any material weaknesses in internal controls; and
- b.
- any fraud, whether or not material, that involves management or other employees who have a significant role in the registrant's internal controls; and
- 6.
- The registrant's other certifying officers and I have indicated in this annual report whether or not there were significant changes in internal controls or in other factors that could significantly affect internal controls subsequent to the date of our most recent evaluation, including any corrective actions with regard to significant deficiencies and material weaknesses.
Date: March 26, 2003
| /s/ GORDON LINK Gordon Link Senior Vice President and Chief Financial Officer |
C-38
NaPro BioTherapeutics, Inc. and Subsidiaries
Financial Statements
Index to Consolidated Financial Statements
| | Page | |
|---|---|---|
| Report of Independent Auditors | C-F-2 | |
Audited Consolidated Financial Statements: | ||
Consolidated Balance Sheets for the years ended December 31, 2002 and 2001 | C-F-3 | |
Consolidated Statements of Operations for the years ended December 31, 2002, 2001 and 2000 | C-F-4 | |
Consolidated Statements of Stockholders' Equity for the years ended December 31, 2002, 2001 and 2000 | C-F-5 | |
Consolidated Statements of Cash Flows for the years ended December 31, 2002, 2001 and 2000 | C-F-6 | |
Notes to Consolidated Financial Statements | C-F-7 |
C-F-1
Report of Independent Auditors
The Board of Directors and Stockholders
NaPro BioTherapeutics, Inc.
We have audited the accompanying consolidated balance sheets of NaPro BioTherapeutics, Inc. and subsidiaries as of December 31, 2002 and 2001, and the related consolidated statements of operations, stockholders' equity, and cash flows for each of the three years in the period ended December 31, 2002. These consolidated financial statements are the responsibility of the Company's management. Our responsibility is to express an opinion on these consolidated financial statements based on our audits.
We conducted our audits in accordance with auditing standards generally accepted in the United States. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement. An audit includes examining, on a test basis, evidence supporting the amounts and disclosures in the financial statements. An audit also includes assessing the accounting principles used and significant estimates made by management, as well as evaluating the overall financial statement presentation. We believe that our audits provide a reasonable basis for our opinion.
In our opinion, the financial statements referred to above present fairly, in all material respects, the consolidated financial position of NaPro BioTherapeutics, Inc. and subsidiaries as of December 31, 2002 and 2001, and the results of their operations and their cash flows for each of the three years in the period ended December 31, 2002 in conformity with accounting principles generally accepted in the United States.
| ERNST & YOUNG LLP | ||
Denver, Colorado February 21, 2003 |
C-F-2
NaPro BioTherapeutics, Inc. and Subsidiaries
Consolidated Balance Sheets
(Dollars in thousands)
| | December 31, | |||||||
|---|---|---|---|---|---|---|---|---|
| | 2002 | 2001 | ||||||
| ASSETS | ||||||||
| Current assets: | ||||||||
| Cash and cash equivalents | $ | 6,762 | $ | 10,144 | ||||
| Accounts receivable | 9,340 | 3,745 | ||||||
| Inventory | 9,397 | 8,098 | ||||||
| Prepaid expense and other current assets | 977 | 1,165 | ||||||
| Total current assets | 26,476 | 23,152 | ||||||
Property, plant and equipment, net | 13,731 | 13,454 | ||||||
| Inventory—raw materials | 3,781 | — | ||||||
| Other assets | 1,340 | 455 | ||||||
| Total assets | $ | 45,328 | $ | 37,061 | ||||
| LIABILITIES AND STOCKHOLDERS' EQUITY | ||||||||
| Current liabilities: | ||||||||
| Accounts payable and accrued liabilities | $ | 5,234 | $ | 6,513 | ||||
| Accrued payroll and payroll taxes | 1,147 | 1,473 | ||||||
| Notes payable—current portion | 102 | 184 | ||||||
| Deferred income | 1,150 | 1,400 | ||||||
| Total current liabilities | 7,633 | 9,570 | ||||||
Notes payable—long term | 19,861 | 19,846 | ||||||
| Deferred income—long term | 5,887 | 6,508 | ||||||
Convertible debentures—related party | 5,151 | — | ||||||
Commitments and contingencies | ||||||||
Stockholders' equity: | ||||||||
| Preferred stock, $.001 par value; 2,000,000 shares authorized; none issued | — | — | ||||||
| Common stock, $.0075 par value; 66,000,000 and 45,000,000 shares authorized in 2002 and 2001, respectively; 29,964,292 and 28,706,195 shares issued in 2002 and 2001, respectively | 225 | 215 | ||||||
| Additional paid-in capital | 110,430 | 96,776 | ||||||
| Accumulated deficit | (103,678 | ) | (95,013 | ) | ||||
| Treasury stock, 54,306 and 254,306 shares at cost in 2002 and 2001, respectively | (181 | ) | (841 | ) | ||||
| Total stockholders' equity | 6,796 | 1,137 | ||||||
| Total liabilities and stockholders' equity | $ | 45,328 | $ | 37,061 | ||||
See accompanying notes to Consolidated Financial Statements.
C-F-3
NaPro BioTherapeutics, Inc. and Subsidiaries
Consolidated Statements of Operations
(In thousands, except per share data)
| | Year Ended December 31, | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| | 2002 | 2001 | 2000 | ||||||||
| Product sales | $ | 34,193 | $ | 15,711 | $ | 8,148 | |||||
| Expenses: | |||||||||||
| Cost of sales | 23,976 | 19,354 | 6,835 | ||||||||
| Research and development | 16,112 | 12,312 | 8,356 | ||||||||
| General and administrative | 9,876 | 8,596 | 6,959 | ||||||||
| Asset writedowns | — | 1,966 | 2,245 | ||||||||
| 49,964 | 42,228 | 24,395 | |||||||||
| Operating loss | (15,771 | ) | (26,517 | ) | (16,247 | ) | |||||
| Other income (expense): | |||||||||||
| License fee income | 8,902 | 1,092 | — | ||||||||
| Interest income | 267 | 793 | 372 | ||||||||
| Interest expense | (2,063 | ) | (1,136 | ) | (750 | ) | |||||
| Net loss | $ | (8,665 | ) | $ | (25,768 | ) | $ | (16,625 | ) | ||
| Basic and diluted loss per share | $ | (0.29 | ) | $ | (0.93 | ) | $ | (0.69 | ) | ||
| Weighted average shares outstanding | 29,606 | 27,585 | 23,924 | ||||||||
See accompanying notes to Consolidated Financial Statements.
C-F-4
NaPro BioTherapeutics, Inc. and Subsidiaries
Consolidated Statements of Stockholders' Equity
(Dollars in thousands)
| | Nonvoting Common Stock | Common Stock | Number of Common Shares Issued | Additional Paid-in Capital | Accumulated Deficit | Treasury Stock | Total | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Balance at December 31, 1999 | $ | 3 | $ | 176 | 23,482,671 | $ | 65,358 | $ | (52,620 | ) | $ | (1,784 | ) | $ | 11,133 | ||||||
| Contribution of 133,982 shares of common stock from treasury at $6.44 per share to retirement plans | — | — | — | 420 | — | 442 | 862 | ||||||||||||||
| Issuance of common stock to retirement plan | — | — | 25,000 | 186 | — | — | 186 | ||||||||||||||
| Issuance of common stock upon completion of milestone | — | 5 | 711,111 | 3,995 | — | — | 4,000 | ||||||||||||||
| Issuance of common stock options in exchange for consulting services | — | — | — | 27 | — | — | 27 | ||||||||||||||
| Exercise of stock options and warrants | — | 2 | 301,646 | 565 | — | — | 567 | ||||||||||||||
| Private placement of common stock | — | 15 | 2,000,000 | 15,741 | — | — | 15,756 | ||||||||||||||
| Issuance of common stock for compensation | — | 1 | 60,444 | 472 | — | — | 473 | ||||||||||||||
| Issuance of common stock for in-licensing of genomics technology | — | 1 | 100,000 | 993 | — | — | 994 | ||||||||||||||
| Issuance of stock and warrants in connection with private placement | — | — | — | 592 | — | — | 592 | ||||||||||||||
| Exchange of subsidiary's preferred stock for common stock | — | 1 | 77,272 | 621 | — | — | 622 | ||||||||||||||
| Net loss | — | — | — | — | (16,625 | ) | — | (16,625 | ) | ||||||||||||
| Balance at December 31, 2000 | $ | 3 | $ | 201 | 26,758,144 | $ | 88,970 | $ | (69,245 | ) | $ | (1,342 | ) | $ | 18,587 | ||||||
| Balance at December 31, 2000 | $ | 3 | $ | 201 | 26,758,144 | $ | 88,970 | $ | (69,245 | ) | $ | (1,342 | ) | $ | 18,587 | ||||||
| Contribution of 151,579 shares of common stock from treasury at $9.50 per share to retirement plans | — | — | — | 939 | — | 501 | 1,440 | ||||||||||||||
| Issuance of common stock upon completion of milestone | — | 7 | 888,889 | 4,993 | — | — | 5,000 | ||||||||||||||
| Issuance of stock options in exchange for consulting services | — | — | — | 531 | — | — | 531 | ||||||||||||||
| Issuance of common stock for compensation | — | — | 4,636 | 29 | — | — | 29 | ||||||||||||||
| Exercise of stock options and warrants | — | 4 | 659,526 | 1,314 | — | — | 1,318 | ||||||||||||||
| Conversion of non-voting common stock to common stock | (3 | ) | 3 | 395,000 | — | — | — | — | |||||||||||||
| Net loss | — | — | — | — | (25,768 | ) | — | (25,768 | ) | ||||||||||||
| Balance at December 31, 2001 | — | 215 | 28,706,195 | 96,776 | (95,013 | ) | (841 | ) | 1,137 | ||||||||||||
| Contribution of 200,000 shares of common stock from treasury at $9.50 per share to retirement plans | — | — | — | 1,240 | — | 660 | 1,900 | ||||||||||||||
| Issuance of common stock for in-licensing of genomics technology | — | 2 | 200,000 | 961 | — | — | 963 | ||||||||||||||
| Issuance of stock options in exchange for consulting services | — | — | — | 93 | — | — | 93 | ||||||||||||||
| Issuance of common stock for compensation | — | — | 19,587 | 164 | — | — | 164 | ||||||||||||||
| Issuance of common stock for payment of interest expense | — | — | 47,973 | 148 | — | — | 148 | ||||||||||||||
| Exercise of stock options and warrants | — | 1 | 101,648 | 216 | — | — | 217 | ||||||||||||||
| Issuance of common stock in connection with private placement, net of issuance costs | — | 7 | 888,889 | 7,775 | — | — | 7,782 | ||||||||||||||
| Discount on convertible debentures issued in connection with private placement | — | — | — | 3,057 | — | — | 3,057 | ||||||||||||||
| Net loss | — | — | — | — | (8,665 | ) | — | (8,665 | ) | ||||||||||||
| Balance at December 31, 2002 | $ | — | $ | 225 | 29,964,292 | $ | 110,430 | (103,678 | ) | $ | (181 | ) | $ | 6,796 | |||||||
See accompanying notes to Consolidated Financial Statements.
C-F-5
NaPro BioTherapeutics, Inc. and Subsidiaries
Consolidated Statements of Cash Flows
(Dollars in thousands)
| | Year Ended December 31, | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| | 2002 | 2001 | 2000 | |||||||||
| Operating activities: | ||||||||||||
| Net loss | $ | (8,665 | ) | $ | (25,768 | ) | $ | (16,625 | ) | |||
| Adjustments to reconcile net loss to net cash used in operating activities: | ||||||||||||
| Depreciation | 2,267 | 1,559 | 1,805 | |||||||||
| Accretion of debt issue cost | 82 | 40 | 40 | |||||||||
| Amortization of debt discount | 386 | — | — | |||||||||
| Accretion of license fee income | (902 | ) | (1,092 | ) | — | |||||||
| In-process research and development expensed in connection with acquired assets of Pangene | 151 | — | — | |||||||||
| License fees paid with common stock | 718 | 1,014 | 83 | |||||||||
| Retirement contributions paid with common stock | 1,900 | 1,440 | 862 | |||||||||
| Compensation paid with common stock, options and warrants | 257 | 560 | 344 | |||||||||
| Interest expense paid with common stock | 148 | — | — | |||||||||
| Asset writedowns | — | 1,966 | 2,245 | |||||||||
| Biomass development cost | — | 453 | — | |||||||||
| Changes in operating assets and liabilities: | ||||||||||||
| Accounts receivable | (5,565 | ) | (2,332 | ) | 3 | |||||||
| Inventory | (2,755 | ) | (819 | ) | (968 | ) | ||||||
| Prepaid expense and other assets | 519 | (922 | ) | 110 | ||||||||
| Accounts payable | (1,139 | ) | 3,142 | 1,197 | ||||||||
| Accrued liabilities | (326 | ) | 518 | 398 | ||||||||
| Deferred income | — | 9,000 | — | |||||||||
| Net cash used in operating activities | (12,924 | ) | (11,241 | ) | (10,506 | ) | ||||||
| Investing activities: | ||||||||||||
| Additions to property and equipment | (4,760 | ) | (8,817 | ) | (3,641 | ) | ||||||
| Acquisition of assets from Pangene Corporation | (1,300 | ) | — | — | ||||||||
| Net cash used in investing activities | (6,060 | ) | (8,817 | ) | (3,641 | ) | ||||||
| Financing activities: | ||||||||||||
| Proceeds from convertible debentures and notes payable, net of issuance cost | 8,088 | 5,334 | 10,562 | |||||||||
| Payments of notes payable | (485 | ) | (432 | ) | (285 | ) | ||||||
| Proceeds from sale of common stock and the exercise of common stock options and warrants, net of issuance cost | 7,999 | 6,318 | 20,915 | |||||||||
| Net cash provided by financing activities | 15,602 | 11,220 | 31,192 | |||||||||
| Net (decrease) increase in cash and cash equivalents | (3,382 | ) | (8,838 | ) | 17,045 | |||||||
| Cash and cash equivalents at beginning of year | 10,144 | 18,982 | 1,937 | |||||||||
| Cash and cash equivalents at end of year | $ | 6,762 | $ | 10,144 | $ | 18,982 | ||||||
| Supplemental schedule of non-cash investing and financing activities: | ||||||||||||
| Plantation cost harvested to inventory | $ | 2,399 | $ | 1,022 | $ | 647 | ||||||
| Issuance of common stock to prepay license fee | 105 | — | 994 | |||||||||
| Insurance claim for damaged inventory | 100 | — | — | |||||||||
| Non-cash exercise of warrants | — | 145 | — | |||||||||
| Exchange of preferred shares of subsidiary for common stock of NaPro BioTherapeutics, Inc. | — | — | 622 | |||||||||
| Issuance of restricted common stock to prepay compensation | — | — | 342 | |||||||||
See accompanying notes to Consolidated Financial Statements.
C-F-6
NaPro BioTherapeutics, Inc.
Notes to Consolidated Financial Statements
Note 1: Summary of Significant Accounting Policies
Description of Business
NaPro BioTherapeutics, Inc. (referred to herein as NaPro or Company) together with its subsidiaries is a pharmaceutical company focused in three distinct areas: the production and sale of paclitaxel, an approved cancer drug; the development of novel, targeted anti-cancer agents, and the development of novel genomic technologies, primarily in the area of Gene Editing for applications in human therapeutics, diagnostics, agribiotechnology and pharmacogenomics. NaPro is a Delaware corporation, which was incorporated in 1991. In 1994, the Company completed an initial public offering of its common stock. The Company has incurred net losses since inception and may incur additional losses for the next several years. To date, all of NaPro's sales have been of one product, paclitaxel, which is derived from extractions from yew trees.
Capital Requirements and Management's Plans
The Company incurred a net loss of $8.7 million for the year ended December 31, 2002 and has an accumulated deficit of $103.7 million as of December 31, 2002. Development of the Company's Gene Editing and Targeted Oncology products will require a commitment of substantial additional funds to conduct the costly and time-consuming research, preclinical and clinical testing necessary to bring its proposed products to market and to establish manufacturing and marketing capabilities. The Company's future capital requirements will depend on many factors, including, among others, the progress of the Company's research, development and drug discovery efforts, the ability of the Company to establish collaborative arrangements for clinical testing, progress with preclinical studies and clinical trials, the time and costs involved in obtaining regulatory approvals, the costs involved in preparing, filing, prosecuting, maintaining, defending and enforcing patent claims, competing technological and market developments, changes in the Company's existing research relationships, determination as to the commercial potential of the Company's potential products, effective commercialization activities and arrangements, and the cost and availability of third-party financing for capital expenditures. Management intends to address the future capital requirements of the Company through strategic alliances, potential equity and debt financing and the management of current operations as required. Management believes that its operational plans will be sufficient to allow the Company to sustain its operations through December 31, 2003. However, there can be no assurance that the Company will be able to obtain additional capital on terms which are acceptable to it.
Principles of Consolidation
The consolidated financial statements include the accounts of the Company and its wholly-owned subsidiaries. All intercompany balances have been eliminated in consolidation.
Cash and Cash Equivalents
Cash and cash equivalents includes all highly liquid investments with maturities of three months or less when purchased.
Financial Instruments
Cash and cash equivalents, accounts receivable, accounts payable and notes payable (other than the note payable to Abbott Laboratories (Abbott)) are carried at cost, which approximates fair value. The fair value of notes payable, other than the Abbott note, is estimated using discounted cash flow analysis based on our estimated current borrowing rate for similar types of arrangements. The
C-F-7
Company has not estimated the fair value of the Abbott note due to a lack of quoted market price for such debt. The convertible debentures are carried at cost, which approximates fair market value, as NaPro recorded a discount attributable to the conversion feature (see Note 5).
Accounts Receivable and Provision for Doubtful Accounts
The Company evaluates the collectibility of its accounts receivable based on a combination of factors. In circumstances when the Company is aware of a specific customer's inability to meet its financial obligations, NaPro records a specific reserve for bad debts against amounts due. For all other instances, the Company reviews the historical collections experience for its customers in determining if a provision for doubtful accounts is deemed necessary. As the Company's receivables primarily relate to amounts due from its strategic partners and the collection of amounts due have historically been timely (see Note 12), the Company has not recorded a provision for doubtful accounts.
Inventory
Inventory is stated at the lower of cost or market using the first-in, first-out method. Inventory, which is not expected to be utilized during 2003, has been classified as a non-current asset.
Plantation Costs
The Company capitalizes plantation costs consisting of expenditures incurred prior to the first commercial harvest of its yew trees. Plantation expenditures include the acquisition cost of trees and the related cost of planting and growing. NaPro depletes such cost using estimated units of production over the expected number of annual harvests. The estimated units of production are based upon the estimated quantities of plant material obtained in the harvests, measured in dry weight kilograms. The depletion model anticipates eight annual harvests in which the trees in a given grove are trimmed, with the whole tree being harvested in the year after the eighth annual trimming harvest. The Company takes depletion of 10% of the initial value of the grove in the first year that the grove receives a trimming harvest. However, the number and timing of harvests can vary depending on product demand and alternative sources of plant material. A given grove may not be harvested each year, and we record no depletion for the grove in which there is no harvest. If an annual trimming is skipped, the depletion for a trimming harvest the following year would be 20%, reflecting the fact that the tree would have an additional year of growth to be trimmed. Alternatively, the whole trees in one or more groves may be harvested before the ninth harvest year in order to obtain more plant material, or to reduce expenditures necessary to grow and maintain the trees. In the year the whole trees in a given grove are harvested, the Company takes depletion of the full remaining value of the grove, whenever that may be, thereby fully depleting the grove. A grove that has five annual trimmings followed by a whole tree harvest would have 10% of its value depleted in each of the five years and 50% in the year of the whole tree harvest. The period over which depletion is taken is thus determined by the number of years between the initial trimming harvest and the whole tree harvest.
Long-Lived Assets
Long-lived assets consist primarily of property, plant and equipment, and inventory. In accordance with Statement of Financial Accounting Standards No. 144 (SFAS 144), Accounting for the Impairment or Disposal of Long-Lived Assets, the recoverability of the carrying value of long-lived assets to be held
C-F-8
and used is evaluated based upon changes in the business environment or other facts and circumstances that suggest they may be impaired. If this evaluation indicates the carrying value will not be recoverable, based on the undiscounted expected future cash flows estimated to be generated by these assets, the Company reduces the carrying amount to the estimated fair value.
Depreciation and Amortization
Depreciation of production and lab equipment, and furniture and fixtures is computed using the straight-line method over estimated useful lives ranging from three to seven years. Amortization of leasehold improvements is computed using the straight-line method over the lesser of the improvement's estimated useful life or remaining lease term. Maintenance and repairs that do not materially improve or extend the lives of the respective assets are expensed as incurred.
Stock Options
NaPro accounts for its stock options to employees and directors in accordance with Accounting Principles Board Opinion No. 25 (APB 25), Accounting for Stock Issued to Employees, and related interpretations. Pursuant to APB No. 25, compensation expense is recorded over the vesting period only if the fair value of the underlying stock exceeds the exercise price. Stock options granted to consultants are accounted for under the fair value method, in accordance with Statement of Financial Accounting Standards No. 123 (SFAS 123), Accounting for Stock-Based Compensation.
Pro forma information regarding net income and earnings per share is required by SFAS 123, which also requires that the information be determined as if the Company had accounted for employee stock options granted subsequent to December 31, 1994 under the fair value method of that statement. NaPro estimated the fair value for these options at the date of grant using a Black-Scholes option pricing model with the following weighted-average assumptions for 2002, 2001 and 2000, respectively: risk-free interest rate ranges of 2.51% to 5.07%, 3.75% to 5.10% and 5.01% to 5.16%; no expected dividend; volatility factor of 1.090 to 1.167, 0.974 to 0.984 and 0.984, and an estimated expected life range of three to six years.
For purposes of pro forma disclosures, the Company amortizes to expense the estimated fair value of the options over the options' vesting period. NaPro's pro forma information is as follows (in thousands, except per share amounts):
| | 2002 | 2001 | 2000 | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Net loss as reported | $ | (8,665 | ) | $ | (25,768 | ) | $ | (16,625 | ) | |
| Deduct: Total stock based employee compensation expense determined under fair value based method for all awards | (4,238 | ) | (2,829 | ) | (1,111 | ) | ||||
| Pro forma net loss | $ | (12,903 | ) | $ | (28,597 | ) | $ | (17,736 | ) | |
| Basic and diluted loss per share—as reported | $ | (0.29 | ) | $ | (0.93 | ) | $ | (0.69 | ) | |
| Pro forma basic and diluted loss per share | $ | (0.44 | ) | $ | (1.04 | ) | $ | (0.74 | ) | |
NaPro accounts for options issued to consultants using the provisions of SFAS 123. Expense recognized in 2002, 2001 and 2000 was $93,000, $531,000 and $27,000, respectively.
C-F-9
Revenue Recognition
Product sales: NaPro recognizes revenue from product sales at the time of shipment, as the title passes to the customer, and the customer assumes the risks and rewards of ownership. Payments received in advance against future sales are recorded as deferred revenue until earned. See also Note 12.
License fees: Prior to 2002, NaPro capitalized license fees and amortized them to income over the estimated economic life of the license. During the second quarter of 2002, the Company revised the period during which it amortizes deferred revenue from license fees, due to the potential decline in the price of paclitaxel in the European market. Since the second quarter of 2002, the amortization period consists of amortizing 80% of fees to income over the first five years of the license, and the remaining 20% of the fees to income over the remaining period of the license. The effect of this change in estimate is not material to the consolidated financial statements.
The Company recognizes income from development milestones when the milestone is achieved and the Company has no future obligation to perform additional work associated with the given milestone.
Foreign and Domestic Operations and Export Sales; Significant Customers
Domestic and foreign financial information is as follows (in thousands):
| | Year | United States | Canada | Eliminations | Total | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Net sales to affiliated and unaffiliated customers | 2002 | $ | 34,193 | $ | — | $ | — | $ | 34,193 | ||||||
| 2001 | 15,711 | — | — | 15,711 | |||||||||||
| 2000 | 8,148 | — | — | 8,148 | |||||||||||
| Operating income (loss) | 2002 | (15,784 | ) | 13 | — | (15,771 | ) | ||||||||
| 2001 | (26,444 | ) | (73 | ) | — | (26,517 | ) | ||||||||
| 2000 | (16,168 | ) | (79 | ) | — | (16,247 | ) | ||||||||
| Total identifiable assets December 31, | 2002 | 48,394 | 2,893 | (5,959 | ) | 45,328 | |||||||||
| 2001 | 40,120 | 2,900 | (5,959 | ) | 37,061 | ||||||||||
The Company is dependent on sales to its strategic partners, Abbott Laboratories, Mayne Pharma, and Tzamal Pharma. NaPro does not require collateral to secure accounts receivable. Substantially all of NaPro's accounts receivable at December 31, 2002 and 2001 were from these partners. Sales to these partners as a percent of total sales are as follows:
| | 2002 | 2001 | 2000 | ||||
|---|---|---|---|---|---|---|---|
| Abbott | 73 | % | 39 | % | — | ||
| Mayne Pharma | 21 | % | 48 | % | 95 | % | |
| Tzamal | 6 | % | 12 | % | — |
C-F-10
Export sales are as follows (in thousands):
| Sales | 2002 | 2001 | 2000 | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Australia | $ | 7,330 | $ | 7,576 | $ | 7,720 | |||
| Other Foreign | 1,881 | 1,954 | 288 | ||||||
| Total Foreign | 9,211 | 9,530 | 8,008 | ||||||
| United States | 24,982 | 6,181 | 140 | ||||||
| Total Sales | $ | 34,193 | $ | 15,711 | $ | 8,148 | |||
Research and Development
Research and development costs are expensed as they are incurred.
Patent Cost
All costs incurred in prosecuting and enforcing patents are expensed.
Asset Writedowns
In 2001, the Company recorded a $1.0 million writedown of production equipment in the Boulder extraction facility related to operations that were transferred to a lower cost third party vendor. The Company also expensed $900,000 related to preliminary engineering and design cost of a new production facility that was cancelled. In 2000, the Company expensed $2.2 million of previously capitalized construction costs incurred in connection with expanded production facilities deemed to be impaired.
Net Loss Per Share
Net loss per common share is computed using the weighted average number of shares of common stock outstanding. Common stock equivalents from stock options, warrants and convertible securities are excluded from the computation of diluted earnings per share as their effect is antidilutive.
The following table sets forth the computation of basic and diluted net loss per share (in thousands, except per share amounts):
| | 2002 | 2001 | 2000 | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Net loss | $ | (8,665 | ) | $ | (25,768 | ) | $ | (16,625 | ) | ||
| Denominator: | |||||||||||
| Weighted average common shares outstanding | 29,606 | 27,585 | 23,924 | ||||||||
| Basic and diluted net loss per common share | $ | (0.29 | ) | $ | (0.93 | ) | $ | (0.69 | ) | ||
Securities that could potentially dilute basic earnings per share in the future that were not included in the computation of diluted earnings per share, because to do so would be antidilutive for the periods presented, amounted to 6,181,800, 5,154,714 and 4,405,769 at December 31, 2002, 2001 and 2000, respectively.
C-F-11
Use of Estimates
In preparing financial statements in conformity with generally accepted accounting principles the Company uses estimates and assumptions. Actual results could vary from these estimates.
Recent Accounting Pronouncements
In December 2002, the FASB issued Statement of Financial Accounting Standards No.148 (SFAS 148), Accounting for Stock-Based Compensation—Transition and Disclosure. SFAS 148 amends SFAS 123 to provide three alternative methods of transition to SFAS 123's fair value method of accounting for stock-based employee compensation for companies that elect to adopt the provisions of SFAS 123. Transition to SFAS 123 is not required by SFAS 148. The Company has elected to use the intrinsic value method of accounting for stock compensation for options granted to employees and directors in accordance with APB 25.
SFAS 148 also amends the disclosure provisions of SFAS 123 and Accounting Principles Board Opinion No. 28 (APB 28), Interim Financial Reporting, to require disclosure in the summary of significant accounting policies of the effects of an entity's accounting policy with respect to stock based compensation on reported net income and earnings per share in annual and interim financial statements. The disclosure provisions of SFAS 148 are required to be adopted by all companies with stock-based employee compensation, regardless of whether they account for that compensation using the fair value method of SFAS 123 or the intrinsic value method of APB 25. The disclosure provisions of SFAS 148, effective for fiscal years ending after December 15, 2002, have been adopted by the Company, with the appropriate disclosures under Note 7, Common Stock Warrants and Options.
In June 2002, the FASB issued Statement of Financial Accounting Standards No. 146 (SFAS 146), Accounting for Costs Associated with Exit or Disposal Activities. SFAS 146 addresses financial accounting and reporting for costs associated with exit or disposal activities and nullifies Emerging Issues Task Force Issue No. 94-3 (EITF 94-3), Liability Recognition for Certain Employee Termination Benefits and Other Costs to Exit an Activity (including certain costs incurred in a restructuring). SFAS 146 requires recognition of a liability for a cost associated with an exit or disposal activity when the liability is incurred as opposed to when the entity commits to an exit plan under EITF 94-3. SFAS 146 is effective for exit or disposal activities initiated after December 31, 2002. The Company has not determined the effect of this new standard; however, the Company anticipates that the adoption of SFAS 146 may have a material effect on its financial position, results of operations or cash flows based upon the expected termination of leases, as well as the involuntary termination of employees during 2003.
In August 2001, the FASB issued SFAS 144, which modifies and expands the financial accounting and reporting for the impairment or disposal of long-lived assets other than goodwill, which is specifically addressed by SFAS 144.
SFAS 144 maintains the requirement that an impairment loss be recognized for a long-lived asset to be held and used if its carrying value is not recoverable from its undiscounted cash flows, with the recognized impairment being the difference between the carrying amount and the fair value of the asset. With respect to a long-lived asset to be disposed of other than by sale, SFAS 144 requires that the asset be considered held and used until it is actually disposed of, but requires that its depreciable life be revised in accordance with Accounting Principles Board Opinion No. 20, Accounting Changes.
C-F-12
SFAS 144 also requires that an impairment loss be recognized at the date a long-lived asset is exchanged for a similar productive asset or distributed to its owners in a spin-off if the carrying amount of the asset exceeds its fair value. With respect to a long-lived asset to be disposed of by sale, SFAS 144 requires that the asset classified as held for sale be measured at the lower of its carrying amount or fair value less selling costs, with no further depreciation or amortization. As such, discontinued operations are no longer measured on a net realizable value basis, and future operating losses are no longer recognized before they occur. SFAS 144 also broadens the previously existing income statement presentation requirements for discontinued operations to include a component of a business, that being the operations and cash flows that can be clearly distinguished, operationally and for financial reporting purposes, from the rest of the entity.
A component of an entity that is classified as held for sale, or has been disposed of, is presented as a discontinued operation if the operations and cash flows of the component have been or will be eliminated from the ongoing operations of the entity, and the entity will not have any significant continuing involvement in the operations of the component. As required, NaPro adopted SFAS 144 in its consolidated financial statements for the first quarter of 2002.
Reclassifications
Certain data in the prior years consolidated financial statements has been reclassified to conform to current year presentation.
Note 2. Inventory
Inventory consists of the following as of December 31 (in thousands):
| | 2002 | 2001 | |||||
|---|---|---|---|---|---|---|---|
| Raw materials | $ | 3,858 | $ | 4,685 | |||
| Work in process | 3,402 | 1,486 | |||||
| Finished goods | 2,137 | 1,927 | |||||
| Total current inventory | 9,397 | 8,098 | |||||
| Raw materials-long-term | 3,781 | — | |||||
| Total inventory | $ | 13,178 | $ | 8,098 | |||
Long-term inventory consists of raw materials which are not expected to be utilized during 2003, due to the timing of biomass harvests and anticipated future changes to our production process.
C-F-13
Note 3. Property, Plant and Equipment
Property and equipment consists of the following as of December 31 (in thousands):
| | 2002 | 2001 | |||||
|---|---|---|---|---|---|---|---|
| Land | $ | 718 | $ | 718 | |||
| Furniture, fixtures and office equipment | 987 | 569 | |||||
| Laboratory and production equipment | 9,140 | 6,967 | |||||
| Plantation costs | 4,650 | 5,916 | |||||
| Leasehold improvements | 3,911 | 3,057 | |||||
| Construction in progress | 568 | 386 | |||||
| 19,974 | 17,613 | ||||||
| Less accumulated depreciation and amortization | (6,243 | ) | (4,159 | ) | |||
| Property, plant and equipment, net | $ | 13,731 | $ | 13,454 | |||
Note 4. Notes Payable
Notes payable consists of the following as of December 31 (in thousands):
| | 2002 | 2001 | ||||
|---|---|---|---|---|---|---|
| Note payable, due in May 2004, interest at 6.5%, interest payable quarterly, net of unamortized issuance costs of $159,000 and $198,000 at December 31 2002 and 2001, respectively | $ | 19,841 | $ | 19,803 | ||
| Other | 122 | 227 | ||||
| 19,963 | 20,030 | |||||
| Less current portion | 102 | 184 | ||||
| Notes payable—long term | $ | 19,861 | $ | 19,846 | ||
The Company has $20.0 million of secured debt from Abbott. The debt bears a primary interest rate of 6.5% and is due in full on the earlier of: (i) the second anniversary of the first sale of finished product by Abbott to a wholesaler or end-user customer following approval of finished product by the FDA (May 8, 2004); (ii) the termination of the Abbott agreement (see Note 12); or (iii) January 1, 2007. The debt is limited to a borrowing base of collateralized assets, recomputed monthly. The majority of NaPro's paclitaxel inventories, and specific assets, which relate to the manufacture of paclitaxel, are collateralized as security for the debt. Abbott's only recourse in the event of NaPro's default on the debt is to these specific assets. Abbott may terminate the agreement at any time with or without cause. Should Abbott terminate without cause, it is obligated to make certain payments to NaPro and to give NaPro twelve months prior written notice.
In addition to the Company's $20.0 million secured debt with Abbott due in May 2004, notes payable have the following minimum future payments:
| 2003 | $ | 102 | |
| 2004 | 11 | ||
| 2005 | 9 | ||
| Total | $ | 122 | |
For the years ended December 31, 2002, 2001, and 2000, interest paid in cash was $1,651,000, $1,051,000, and $467,000, respectively.
C-F-14
Note 5. Convertible Debentures
In February 2002, the Company sold privately $8.0 million principal of five-year 4% debentures convertible into common stock at $15 per share to TL Ventures V, L.P. and one of its affiliated funds. No placement agent was involved in the transaction. The net proceeds were $7.8 million. As part of this transaction, NaPro recorded a discount attributable to the fair value of the conversion feature of the convertible debentures in the amount of $3.1 million, which is being amortized over the term of the debentures. During the first quarter of 2002, the Company filed a registration statement with the Securities and Exchange Commission to register the common stock issuable upon conversion of the debentures and common stock issuable in lieu of cash interest on the debentures under the Securities Act of 1933. In July 2002, the registration statement was declared effective by the SEC. The Company may pay the debenture interest in cash or common stock at its option. In 2002, the Company paid the interest in stock. The debentures automatically convert into common stock during the first two years if the average price of the common stock for 15 trading days exceeds $22, and during the last three years if the average price for 30 trading days exceeds $16. If NaPro has insufficient funds to repay the debentures in full at maturity or upon acceleration, the holders of the debentures may elect to convert the outstanding principal balance and any unpaid accrued interest on the debentures into shares of common stock at a conversion price equal to the then current 20 day average trading price of the common stock.
Note 6. Stockholders' Equity
Stockholder Rights Plan
In November 1996, the Company adopted a Stockholder Rights Plan and distributed a dividend of one Right to purchase one one-hundredth of a share of a new series of junior participating preferred stock, Series B, for each share of common stock. The Stockholder Rights Plan was amended and restated in September 2001. The objective of the Stockholder Rights Plan is to secure for stockholders the long term value of their investment and to protect stockholders from coercive takeover attempts by strongly encouraging anyone seeking to acquire us to negotiate with our Board of Directors. The adoption of the Stockholder Rights Plan was not in response to any hostile takeover proposal.
The Rights trade with common stock as a unit unless the Rights become exercisable upon the occurrence of certain triggering events relating to the acquisition of 20% or more of common stock. In certain events after the Rights become exercisable they will entitle each holder, other than the acquirer, to purchase, at the Rights' then current exercise price (currently set at $60), a number of shares of common stock having market value of twice the Right's exercise price or a number of the acquiring company's common shares having a market value at the time of twice the Rights' exercise price. For example, in the event of an acquisition of greater than 20% of our stock without approval of our Board of Directors, our stockholders (other than the 20% acquirer) would have the right to purchase $120 worth of stock for $60. A stockholder would have one such right for each share of stock held at the time the rights become exercisable.
The Company may amend the Rights, except in certain limited respects or redeem the Rights at $0.01 per Right, in each case at any time prior to the Rights becoming exercisable. The Rights will expire on November 8, 2006.
Private Placements
In February 2002, the Company sold privately $8.0 million of common stock issued at $9 per share to TL Ventures V, L.P. and one of its affiliated funds. No placement agent was involved in the transaction. The net proceeds were $7.8 million. See Note 5 for information concerning the convertible debentures.
C-F-15
In November 2000, the Company sold privately 2.0 million shares of common stock to a group of institutional investors for $17.5 million, receiving $16.3 million in proceeds net of issuance cost. The Company subsequently registered the resale of the shares under the Securities Act of 1933. The Company issued to the placement agent a three year warrant for 100,000 shares exercisable at $8.75 per share. The warrants were valued at $592,000 using the Black-Scholes option pricing model and credited that portion of the proceeds to Additional Paid-in Capital attributed to the warrants.
Authorized Shares
During the Annual Meeting of Stockholders in July 2002, the stockholders approved amendments to the Company's Amended and Restated Certificate of Incorporation, which increased the total number of authorized shares from 45.0 million to 66.0 million, as well as removing references to non-voting common stock from the Certificate of Incorporation. All of the Company's previously issued non-voting common stock was converted into common stock during 2001.
Stock Options and Warrants
The Company reserved 5,648,467 unissued, authorized shares of common stock as of December 31, 2002 for future issuance of common stock options and warrants.
Note 7. Common Stock Warrants and Options
The following summarizes warrant activity:
| | Warrants | Exercise Price | Expiration Dates | |||
|---|---|---|---|---|---|---|
| Outstanding at December 31, 2000 | 457,034 | $1.50 - $8.75 | 2001 - 2003 | |||
| Exercised | (327,700 | ) | 1.50 - 1.88 | 2001 - 2003 | ||
| Outstanding at December 31, 2001 | 129,334 | 1.88 - 8.75 | 2002 - 2003 | |||
| Expirations | (13,334 | ) | 1.88 | 2002 | ||
| Exercised | (5,000 | ) | 1.88 | 2003 | ||
| Outstanding at December 31, 2002 | 111,000 | $1.88 - $8.75 | 2003 | |||
In 2001, there was a cashless exercise of 112,375 warrants resulting in the issuance of 96,913 shares.
Nonplan Stock Options
In January 1994, the Company granted to four outside directors 27,000 nonplan options to purchase shares of common stock which were immediately exercisable at a price of $2.40 and which expire in January 2004. As a result of option exercises, 16,000 of these options remained outstanding as of December 31, 2002. In September 1997, The Company granted to its employees 20,075 nonplan options to purchase shares of common stock which vested over two years and which expire in September 2007. As of December 31, 2002, 750 of these options remained outstanding.
The 1993 Stock Option Plan
During 1993, the Board of Directors adopted the NaPro BioTherapeutics, Inc. 1993 Stock Option Plan (the Plan), to provide stock options to employees and other individuals as determined by the Board of Directors. The Plan provides for option grants designated as either nonqualified or incentive
C-F-16
stock options. The Plan provides for the issuance of up to 146,667 shares of common stock. The initial term of the Plan is ten years, which expires in September 2003, and the maximum option exercise period shall be no more than ten years from the date of grant. The term of options for 667 or more shares is eight years, and the term of options for fewer than 667 shares is five years. Options for 667 shares or more vest 25% after each anniversary date of the grant, and options for fewer than 667 shares vest 50% after each anniversary date of the grant. The exercise price for stock options issued under the Plan is equal to the fair market value of the Company's common stock on the date of grant.
1994 Long-Term Performance Incentive Plan
The Company has a Long-Term Performance Incentive Plan (the Incentive Plan) which was approved by stockholders in July 1994. The Incentive Plan initially authorized 375,000 shares for issuance. Stockholders subsequently approved increases in the number of authorized shares. There are currently 6,600,000 shares authorized for issuance under the Incentive Plan. The Incentive Plan provides for granting to employees and other key individuals who perform services for the Company the following types of incentive awards: stock options, stock appreciation rights, restricted stock, performance units, performance grants and other types of awards that the Compensation Committee deems to be consistent with the purposes of the Incentive Plan. In addition, each person who is not an employee and (i) who is elected or re-elected as a director by the stockholders at any annual meeting of stockholders, (ii) who continues as a director following an annual meeting of stockholders at which such director is not subject to re-election or (iii) is appointed as a director in accordance with Company bylaws other than at an annual meeting, upon such election or appointment, will receive, as of the business day following the date of each such election or appointment, a non-qualified option to purchase 10,000 shares of common stock. The Incentive Plan also provides for annual automatic grants of options to purchase 10,000 shares to the chairs of the Board of Directors' Audit, Compensation, Strategic Planning, and Research and Development committees. The Incentive Plan provides for an automatic grant of non-qualified stock options to purchase 7,500 shares of common stock to members of the Research and Development committee upon their initial appointment to the committee, and an automatic grant of non-qualified stock options to purchase 3,000 shares of common stock to a Research and Development committee member who continues to serve on the committee after an annual meeting of stockholders.
The 1998 Stock Option Plan
In 1998, the Board of Directors adopted the 1998 Stock Option Plan (the 1998 Plan) to provide stock options for employees and other individuals who perform services for the Company. Originally, 125,000 shares were authorized for issuance under the 1998 Plan. In 1999, 2000, 2001 and 2002 the Board of Directors approved increases in the number of authorized shares. There are currently 1,925,000 shares authorized for issuance under the 1998 Plan. Under the terms of the 1998 Plan, stock options cannot be granted to persons who are NaPro officers or directors.
C-F-17
The following summarizes stock option activity and balances:
| | Non Plan | 93 Plan | 94 Plan | 98 Plan | Total | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Authorized | 212,235 | 146,667 | 6,600,000 | 1,925,000 | 8,883,902 | ||||||
| Less: | |||||||||||
| Exercised | 195,085 | 133,232 | 427,349 | 99,344 | 855,010 | ||||||
| Stock grants | 400 | — | 174,956 | 87,558 | 262,914 | ||||||
| Issued and unexercised | 16,750 | — | 4,161,641 | 1,359,076 | 5,537,467 | ||||||
| Available to be issued | — | 13,435 | 1,836,054 | 379,022 | 2,228,511 | ||||||
| | Stock Options | Exercise Price | Weighted Average Exercise Price | |||||
|---|---|---|---|---|---|---|---|---|
| Outstanding at December 31, 1999 | 2,801,435 | $0.75 - 10.13 | $ | 2.00 | ||||
| Granted | 890,310 | 2.25 - 9.00 | 7.34 | |||||
| Forfeited | (6,364 | ) | 1.00 - 9.50 | 2.19 | ||||
| Exercised | (131,646 | ) | 1.00 - 10.13 | 1.71 | ||||
| Outstanding at December 31, 2000 | 3,553,735 | 0.75 - 10.13 | 3.35 | |||||
| Granted | 1,876,050 | 2.25 - 11.25 | 8.18 | |||||
| Forfeited | (56,587 | ) | 1.00 - 8.94 | 3.76 | ||||
| Expired | (530 | ) | 1.72 - 1.81 | 1.77 | ||||
| Exercised | (347,288 | ) | 0.75 - 8.00 | 2.86 | ||||
| Outstanding at December 31, 2001 | 5,025,380 | 1.00 - 11.25 | 5.18 | |||||
| Granted | 765,300 | 0.72 - 12.10 | 4.85 | |||||
| Forfeited | (139,897 | ) | 1.00 - 12.10 | 5.58 | ||||
| Expired | (16,668 | ) | 2.40 - 6.00 | 4.56 | ||||
| Exercised | (96,648 | ) | 1.00 - 3.65 | 6.37 | ||||
| Outstanding at December 31, 2002 | 5,537,467 | $0.72 - 12.10 | $ | 5.17 | ||||
The weighted-average fair value of options granted during 2002, 2001 and 2000 was $3.80, $6.49 and $5.84, respectively.
| | Outstanding Options | Exercisable Options | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Range of Exercise Price | Number Outstanding | Weighted-Average Remaining Contractual Life | Weighted-Average Exercise Price | Number Exercisable | Weighted-Average Exercise Price | ||||||||
| $0.72 - $1.79 | $ | 559,785 | 6.48 | $ | 1.04 | 459,785 | $ | 1.01 | |||||
| $1.80 - $1.99 | 683,212 | 5.33 | 1.82 | 679,712 | 1.82 | ||||||||
| $2.00 - $2.49 | 524,624 | 7.03 | 2.18 | 339,691 | 2.18 | ||||||||
| $2.50 - $4.95 | 910,000 | 7.38 | 2.87 | 622,500 | 2.55 | ||||||||
| $5.00 - $6.70 | 970,740 | 8.12 | 6.56 | 446,053 | 6.58 | ||||||||
| $6.75 - $8.95 | 959,606 | 8.07 | 7.96 | 507,729 | 8.01 | ||||||||
| $9.25 - $12.10 | 929,500 | 8.52 | 9.72 | 28,626 | 9.72 | ||||||||
| $0.72 - $12.10 | $ | 5,537,467 | 7.44 | $ | 5.17 | 3,084,096 | $ | 3.67 | |||||
C-F-18
The Company sponsors a defined contribution retirement plan (the Plan) for all eligible employees that allows participants to make contributions by salary deduction pursuant to Section 401(k) of the Internal Revenue Code. The Company may make discretionary matching contributions to the Plan on behalf of the participants in the form of cash or in shares of common stock. No matching contribution was made in 2002. In 2001 and 2000, the Company matched the first $2,000 in contributions of each participating employee with common stock, at a rate of 262% for 2001 and 284% for 2000. In 2001 and 2000, the Company contributed 25,263 and 44,661 shares of common stock to the Plan, valued at $240,000, and $287,000, respectively. In January 2003, the Board approved transferring NaPro common stock held in the Plan to the Employee Stock Ownership Plan (ESOP).
The Company adopted an ESOP for its employees, in accordance with the Internal Revenue Code. Under this plan, employees over the age of 17 are eligible to participate on the first day of the month immediately following the completion of six months of continuous service or 1,000 hours of service during a 12-continuous-month period. Participants make no contributions to the ESOP. The Company contributes common stock to the ESOP which is allocated to all eligible employees based on their allowable pay. For 2002, 2001 and 2000 the Company contributed 200,000, 126,316 and 114,321 shares to the ESOP, valued at $1.9 million, $1.2 million and $761,000, respectively. All shares held by the ESOP are treated as outstanding in computing earnings per share.
Note 9. Income Taxes
As of December 31, 2002, the Company had the following net operating loss carryforwards and research and development credits to offset future taxable income in the U.S. (in thousands):
| Expiring December 31, | Net Operating Losses | Research and Development Credits | ||||
|---|---|---|---|---|---|---|
| 2007 | $ | 1,746 | $ | 52 | ||
| 2008 | 3,328 | 54 | ||||
| 2009 | 4,713 | 38 | ||||
| 2010 | 4,960 | 15 | ||||
| 2011 | 7,389 | 49 | ||||
| 2012 | 12,043 | 140 | ||||
| 2018 | — | 205 | ||||
| 2019 | 8,445 | 230 | ||||
| 2020 | 15,899 | 340 | ||||
| 2021 | 19,781 | 112 | ||||
| 2022 | 9,083 | 566 | ||||
| $ | 87,387 | $ | 1,801 | |||
The Tax Reform Act of 1986 contains provisions that limit the utilization of net operating loss and tax credit carryforwards if there has been a "change of ownership" as described in Section 382 of the Internal Revenue Code. Such a change of ownership may limit the utilization of the Company's net operating loss and tax credit carryforwards, and could be triggered by sales of securities by NaPro or its stockholders.
In Canada, the Company had net operating loss carryforwards of approximately US $1.7 million, expiring in years ending in 2003 through 2009.
C-F-19
Significant components of our deferred tax assets are as follows (in thousands):
| | Year Ended December 31, | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| | 2002 | 2001 | 2000 | ||||||||
| Deferred tax assets: | |||||||||||
| Tax net operating loss carryforward | $ | 32,770 | $ | 30,738 | $ | 21,975 | |||||
| Deferred revenue | 1,878 | 2,216 | — | ||||||||
| Research and development credits | 1,801 | 1,813 | 1,330 | ||||||||
| Depreciation | 1,673 | 1,379 | 1,371 | ||||||||
| Other | 293 | 383 | 186 | ||||||||
| Total deferred tax assets | 38,415 | 36,529 | 24,862 | ||||||||
| Valuation allowance | (38,415 | ) | (36,529 | ) | (24,862 | ) | |||||
| Net deferred tax assets | $ | — | $ | — | $ | — | |||||
Variations from the federal statutory rate are as follows:
| | Year Ended December 31, | ||||||
|---|---|---|---|---|---|---|---|
| | 2002 | 2001 | 2000 | ||||
| Federal statutory income tax rate | 35.00 | % | 35.00 | % | 35.00 | % | |
| Effect of permanent differences | 5.54 | 0.30 | 0.35 | ||||
| State income tax rate net of federal benefit | (3.18 | ) | (2.94 | ) | (2.93 | ) | |
| Effect of foreign operations | 4.85 | 2.09 | 2.27 | ||||
| Valuation allowance | (42.21 | ) | (34.45 | ) | (34.69 | ) | |
| Effective income tax rate | 0.00 | % | 0.00 | % | 0.00 | % | |
Note 10. Restructuring Plan
In July 2002, NaPro initiated a company-wide restructuring plan involving a reduction of full-time staff by 30 employees, as well as the elimination of other operating expenses. Included in this reduction were 16 employees in manufacturing, 10 employees in research and development and 4 employees in general and administrative positions. The annual payroll cost savings is expected to be approximately $1.5 million. During the third quarter of 2002, the Company incurred restructuring charges of approximately $199,000, which were paid by December 31, 2002. NaPro will continue to focus on cost saving measures, which it believes will result in additional changes to its operations.
Note 11. Pangene Acquisition
In December 2002, NaPro acquired the genomics business of Pangene Corporation for $1.3 million in cash and assumption of debt obligations totaling approximately $65,000. The acquisition consisted primarily of patents and intellectual property relating to Pangene's homologous recombination technology and gene isolation and service business, physical assets, instrumentation, software, customer relationships and third-party licenses. The purchase price was allocated to intangible assets totaling $987,000, fixed assets and software of $183,000, and other current assets of $44,000. In process research and development of $151,000 was expensed at the time of the acquisition. The Company will amortize intangible assets over 5 to 15 years, and fixed assets and software over 3 years. In connection with this agreement, the Company acquired additional patents in January 2003 in the amount of $400,000.
C-F-20
Note 12. Strategic Alliances
In connection with the Company's paclitaxel business, NaPro has strategic alliances with four pharmaceutical companies: Abbott Laboratories, Mayne Pharma, Tzamal Pharma, and JCR Pharmaceuticals Co., Ltd.
Abbott Laboratories Agreement
In July 1999, the Company entered into a collaborative agreement of up to 20 years with Abbott to develop and commercialize one or more formulations of paclitaxel for the treatment of a variety of cancers. The exclusive agreement covers the U.S. and Canada. Under the agreement with Abbott, NaPro is responsible for supply of bulk drug; development activity is conducted jointly. Abbott is responsible for finishing, regulatory filings, marketing and sale of the finished drug product. Most primary decisions related to the development program are made by a joint Abbott-NaPro Development Committee. In March 2001, NaPro and Abbott filed an Abbreviated New Drug Application, or ANDA, with the U.S. Food and Drug Administration, or FDA, for paclitaxel. The FDA granted approval of this ANDA on May 8, 2002. The Company received an approval for a second ANDA on August 1, 2002, which provides for greater manufacturing flexibility for the generic paclitaxel product.
In connection with the Abbott agreement, NaPro may receive total funding of up to $118.0 million in the form of up to $30.0 million in development milestones, up to $57.0 million in marketing milestones, $20.0 million in secured debt, and $11.0 million in equity investments. Marketing milestones are based upon certain annual sales levels, the majority of which are unlikely to be achieved given the current pricing of paclitaxel. Through December 31, 2002, and excluding product sales, the Company has received $40.0 million under the agreement, including $11.0 million in equity investments in exchange for 2.0 million shares of our common stock, $9.0 million in development milestones, and $20.0 million in secured debt.
Of the $30.0 million of potential development-related milestone payments, the Company received $1.0 million upon execution of its agreement with Abbott in July 1999. With the approval of NaPro's ANDA, and upon commencement of commercial sales, the Company received an $8.0 million payment in May 2002. Thus, $21.0 million in possible development milestones remain. Additional development milestones will only be available should NaPro or Abbott undertake and successfully complete new development work, which is not currently anticipated.
NaPro has received $20.0 million of secured debt from Abbott. The debt bears a primary interest rate of 6.5% and is due in full on the earlier of: (i) the second anniversary of the first sale of finished product by Abbott to a wholesaler or end-user customer following approval of finished product by the FDA (May 8, 2004); (ii) the termination of the Abbott agreement; or (iii) January 1, 2007. The debt is limited to a borrowing base of collateralized assets, recomputed monthly. The majority of the Company's paclitaxel inventories, and specific assets, which relate to the manufacture of paclitaxel, are collateralized as security for the debt. Abbott's only recourse in the event of NaPro's default on the debt is to these specific assets.
Contingent upon achieving certain commercial sales thresholds for all approved formulations of paclitaxel over several years, NaPro may receive marketing milestone payments from Abbott of up to $57.0 million. Given the current pricing of paclitaxel, the Company believes that the majority of these annual sales thresholds are unlikely to be achieved.
Under terms of the agreement, Abbott purchases bulk drug from NaPro. Abbott may terminate the agreement at any time with or without cause. Should Abbott terminate without cause, it is obligated to make certain payments to NaPro and to give NaPro twelve months prior written notice. Since the approval of generic paclitaxel in mid-2001, the selling price of paclitaxel has fallen
C-F-21
significantly, and may continue to do so. NaPro receives a fixed price payment of $500 per gram, upon delivery of paclitaxel to Abbott, plus a quarterly royalty payment, if any. The royalty payment is a percentage of sales recorded by Abbott less any product payment already paid to NaPro. The percentage of sales varies each year based upon total sales in that year recorded by Abbott. The Company does not anticipate receiving significant royalty payments given the current price of the drug.
Mayne Pharma Agreement
In 1992, the Company entered into a 20-year exclusive agreement with F.H. Faulding & Co., Ltd. (Faulding), a large Australian pharmaceutical company for the clinical development, sale, marketing and distribution of paclitaxel. In October 2001, Faulding was acquired by Mayne Nickless Limited, an Australian based health care provider and logistics operator. As a result, Faulding is now known as Mayne Pharma. Mayne Pharma markets anti-cancer pharmaceuticals and other health care products in Europe, Australia, Asia, South America, and other regions throughout the world. In 2000, the Company amended the Mayne Pharma agreement to, among other things, add additional countries to Mayne Pharma's exclusive territory. In 2001, the Company entered into a separate agreement with Mayne Pharma covering development and sale of paclitaxel in Europe. Including the new agreement for Europe, the Mayne Pharma territory includes substantially all of the world other than the U.S., Canada, Japan, Israel, the former Soviet Union and parts of Africa. Mayne Pharma has received marketing approval for, and is selling, paclitaxel as ANZATAX™ in more than 25 countries.
In Europe, NaPro is responsible for regulatory filings and will supply paclitaxel exclusively to Mayne Pharma to formulate and finish the product. Should NaPro receive regulatory approval in Europe, Mayne Pharma will then market and sell the final proprietary paclitaxel formulation in Europe. NaPro will share equally the net sales of the product in Europe. Under the agreement, Mayne Pharma paid an up-front licensing fee to NaPro of $7.5 million. NaPro deferred the $7.5 million, $5.5 million of which is being amortized 80% over the first five years to license fee income and the remaining 20% over the remaining seven years of the license. NaPro has deferred $2.0 million until agreed upon events occur. NaPro may be obligated to return part or all of the $2.0 million should these events not occur.
Tzamal Pharma Agreement
In January 2001, the Company received approval to sell paclitaxel in Israel. NaPro established an exclusive supply and distribution agreement with Tzamal Pharma for the development and distribution of paclitaxel in Israel under the trade name Biotax™. The Israeli Ministry of Health has approved Biotax™ for use in a variety of cancers.
JCR Pharmaceuticals Co., Ltd. Agreement
In June 2001, NaPro and JCR Pharmaceuticals Co., Ltd. (JCR) entered into a mutually exclusive development, supply and distribution agreement for paclitaxel in Japan. Under the agreement, NaPro is responsible for manufacturing and supplying the finished drug. NaPro and JCR are jointly responsible for the clinical and regulatory program necessary for seeking approval to market paclitaxel in Japan. JCR will be responsible for the sales and distribution of the product in Japan upon its approval. NaPro cannot assure, however, such regulatory approval will be attained in Japan, or that JCR will successfully market paclitaxel in Japan. JCR paid an up-front licensing fee to NaPro of $1.5 million. NaPro deferred the $1.5 million, which is being amortized 80% over the first five years to license fee income and the remaining 20% over the remaining thirteen and one-half years of the license.
C-F-22
Gene Editing Technology License
In November 2000, the Company entered into a 20-year Gene Editing technology license, with the University of Delaware and Thomas Jefferson University, relating to the use of proprietary oligonucleotides (DNA fragments) designed to precisely alter genes in humans, animals, plants, viruses and microbes. The license provides for research and patent funding commitments and payments in common stock. To date, the Company has issued 300,000 shares under the license agreement. Assuming the Company does not cancel the license, it will issue an additional 900,000 shares in 100,000 share-per-year increments on the license anniversaries. The Company may, at its option, accelerate the issuance dates. Certain milestones may also accelerate the issuance dates. The license is terminable at NaPro's option. If terminated, no further shares would be issued. NaPro has committed to fund at least $300,000 in research under this agreement during 2003.
Note 13. Commitments and Contingencies
Operating Leases
The Company has executed noncancellable operating lease agreements for office, research and production facilities, equipment and for plantations. As of December 31, 2002, future minimum lease payments under noncancellable operating lease agreements are as follows (in thousands):
| 2003 | $ | 1,445 | |
| 2004 | 1,171 | ||
| 2005 | 504 | ||
| 2006 | 488 | ||
| 2007 | 94 | ||
| Thereafter | 142 | ||
| Total | $ | 3,844 | |
NaPro has renewal clauses in some of these leases which range from one to ten years. Rent expense for the years ended December 31, 2002, 2001, and 2000 was $1,528,000, $981,000 and $955,000, respectively.
Uncertainty Over the Selling Price Under the Mayne Pharma Agreement
Under the Mayne Pharma Agreement (see Note 12), Mayne Pharma pays NaPro a fixed percentage of their sales price. Title to the paclitaxel and risk of loss passes to Mayne Pharma when the product is shipped; the sales are unconditional other than for the failure of product to meet specifications. NaPro then recognizes revenue at the time of shipment, based upon Mayne Pharma's forecast of prices provided to NaPro. Mayne Pharma's actual selling price, however, may differ from the amounts originally estimated. Each year ended March 31, Mayne Pharma calculates and reports to NaPro the actual quantity of sales and the sales price per gram. NaPro then calculates a price adjustment, which may either increase or decrease NaPro's sales revenue during the prior twelve months. NaPro records the price adjustment and then either invoices or remits the adjustment to Mayne Pharma. As NaPro cannot control the price adjustments, it is possible that the adjustments could have a material effect on the Company's financial position, results of operations or cash flows. The Company currently records a reserve for potential Mayne Pharma price adjustments based upon historical experience and periodic reporting provided by Mayne Pharma.
C-F-23
Note 14. Research and Development
The Company has incurred the following expense on research and development projects for the years ended December 31, (in thousands):
| | 2002 | 2001 | 2000 | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Oncology | $ | 9,933 | $ | 10,186 | $ | 8,252 | |||
| Genomics | 6,179 | 2,126 | 104 | ||||||
| Total | $ | 16,112 | $ | 12,312 | $ | 8,356 | |||
Research and development is expected to remain a significant expense of the Company's business. NaPro's research and development activity is subject to change as it develops a better understanding of the feasibility of its projects. The Company currently expects to concentrate on the development of novel, targeted anti-cancer agents, and the development of novel genomic technologies, primarily in the area of Gene Editing, for applications in human therapeutics, diagnostics, agribiotechnology and pharmacogenomics. The Company anticipates bringing its Targeted Oncology program to the clinic in both the U.S. and Europe during the first half of 2004. The Sickle Cell Disease and Huntington's Disease programs are expected to go into the clinic by the second half of 2004 to the beginning of 2005. However, there can be no assurance that the Company will be able to achieve the timing of these programs. The Company also cannot estimate the cost of the effort necessary to complete the programs or the timing of commencement of material net cash inflows from these programs.
Note 15. Related Party Transactions
Marc Ostro, one of the Company's directors, is a managing director of an affiliate of the investment manager of the TL Venture Funds, and a voting limited partner in the TL Venture Funds, which purchased $8.0 million of common stock and $8.0 million principal of five-year 4% debentures convertible into common stock (see Notes 5 and 6).
Note 16. Quarterly Data (unaudited)
(In thousands, except per share amounts)
| Quarter Ended | Product Sales | Cost of Sales | Basic and Diluted Income (Loss) per Share | Net Income (Loss) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| March 31, 2002 | $ | 6,736 | $ | 7,120 | $ | (0.23 | ) | $ | (6,663 | ) | |||
| June 30, 2002 | 9,490 | 7,018 | $ | .10 | 3,055 | ||||||||
| September 30, 2002 | 8,320 | 5,551 | $ | (0.13 | ) | (3,944 | ) | ||||||
| December 31, 2002 | 9,647 | 4,287 | $ | (0.04 | ) | (1,113 | ) | ||||||
| 2002 | $ | 34,193 | $ | 23,976 | $ | (0.29 | ) | $ | (8,665 | ) | |||
| March 31, 2001 | $ | 3,931 | $ | 2,975 | $ | (0.08 | ) | $ | (2,101 | ) | |||
| June 30, 2001 | 2,158 | 4,665 | $ | (0.28 | ) | (7,701 | ) | ||||||
| September 30, 2001 | 3,872 | 3,458 | $ | (0.21 | ) | (5,901 | ) | ||||||
| December 31, 2001 | 5,750 | 8,256 | $ | (0.36 | ) | (10,065 | ) | ||||||
| 2001 | $ | 15,711 | $ | 19,354 | $ | (0.93 | ) | $ | (25,768 | ) | |||
C-F-24
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
Form 10-K/A
Amendment to Annual Report Pursuant to Section 13 or 15(d) of the
Securities Exchange Act of 1934
Year Ended December 31, 2002
Commission File Number 0-24320
NAPRO BIOTHERAPEUTICS, INC.
(Exact name of Registrant as specified in its charter)
| Incorporated in Delaware | IRS ID No. 84-1187753 |
6304 Spine Road, Unit A
Boulder, Colorado 80301
(303) 516-8500
Securities registered pursuant to Section 12(b) of the Act: None
Securities registered pursuant to Section 12(g) of the Act:
Common Stock, $0.0075 par value; Preferred Stock Purchase Rights
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. ý Yes o No
Indicate by check mark if disclosure of delinquent filers pursuant to Item 405 of Regulation S-K is not contained herein, and will not be contained, to the best of registrant's knowledge, in definitive proxy or information statements incorporated by reference in Part III of this Form 10-K or any amendment to this Form 10-K. o
Indicate by check mark whether the registrant is an accelerated filer (as defined in Rule 12b-2 of the Act). o Yes ý No
As of April 11, 2003, 29,939,486 shares of the Registrant's common stock were outstanding. The aggregate market value of the voting stock held by non-affiliates of the Registrant was approximately $16,766,000 on that date.
C-39
The registrant is filing this amendment to its 2002 Form 10-K in order to provide the information required by Part III of Form 10-K.
Item 10 Directors and Executive Officers of the Registrant
Directors
Our Board of Directors currently consists of nine members: Sterling Ainsworth, Ph.D. (Vice Chairman); Edward Erickson; George Gould, Esq.; Arthur Hayes, Jr., M.D.; Marc Ostro, Ph. D.; The Honorable Richard Perle; Patricia Pilia, Ph.D.; Robert Pollack, Ph.D., and Leonard Shaykin (Chairman). These directors are divided into three classes. Mr. Shaykin and Drs. Hayes and Pollack are Class I directors with terms of office expiring at the 2003 Annual Meeting. Dr. Pilia and Messrs. Erickson and Perle are Class II directors with terms of office expiring at the 2004 Annual Meeting. Drs. Ainsworth and Ostro and Mr. Gould are Class III directors with terms of office expiring at the 2005 Annual Meeting of Stockholders.
Sterling K. Ainsworth, Ph.D., 63, a co-founder of the Company, has served as an executive officer and director since our inception. He served as our Chief Executive Officer from November 1991 to August 1999, and has served as our President since October 1992 and as Vice Chairman of the Board and Chief Scientific Officer since August 1999. In 1990, he co-founded, with Dr. Pilia, Pacific Biotechnology, Inc. (a predecessor of ours) and served as Chairman and President of such company until our inception. From 1972 to 1990, Dr. Ainsworth held various levels of professorships of Pathology with tenure in the College of Medicine and Dental Medicine and Graduate Studies at the Medical University of South Carolina ("MUSC"), where he established, developed and directed MUSC's Immunopathology Diagnostic Laboratory. Dr. Ainsworth received a Bachelor's degree from the University of Mississippi in 1963. He received a Master's degree in medical microbiology in 1965 and a Doctoral degree in medical science in 1969 from the University of Mississippi Medical School. He completed his post-doctoral fellowship in the Department of Pathology at Harvard Medical School from 1970 to 1972. Dr. Ainsworth is engaged to be married to Dr. Pilia, a co-founder, director and executive officer of the Company.
Edward L. Erickson, 56, has served as a director since 2000. He is currently Chairman of the Board, President and Chief Executive Officer of Immunicon Corporation, a venture capital backed medical products company with technology for use in diagnostics, life science research, and therapeutic applications. He was appointed Chairman of Immunicon in April 1998 and Chief Executive Officer in September 1998. From 1993 to 1998, Mr. Erickson was President, Chief Executive Officer and a director of DepoTech Corporation, a biopharmaceutical company in the drug delivery field. Additionally, from 1995 to 1998, he served as a director of MegaBios Corporation, a gene therapy company. From 1991 to 1993, he was President, Chief Executive Officer and a director of Cholestech Corporation, a diagnostic products company in point-of-care cholesterol testing and screening. Mr. Erickson holds B.S. and M.S. degrees in mathematics from the Illinois Institute of Technology and an M.B.A. with high distinction from Harvard University.
George M. Gould, Esq., 65, has served as a director since January 2003. He has served as Of Counsel to the law firm Gibbons, Del Deo, Dolan, Griffinger & Vecchione since June 1996. Mr. Gould is also a director of Protein Design Labs, Inc. a biotechnology company engaged in the development of humanized monoclonal antibodies for the prevention and treatment of disease. Additionally, Mr. Gould is a director of Angiogenex, Inc., a privately-held biopharmaceutical company that develops therapeutic and diagnostic applications of Id gene and protein technologies as well as Supratek Pharma, a private Canadian biopharmaceuticals company developing novel block copolymer targeted drug formulations. From May 1996 to December 1996, Mr. Gould was a Senior Vice President of PharmaGenics, Inc.
C-40
Prior to that time Mr. Gould served as Vice President, Licensing & Corporate Development and Chief Patent Counsel for Hoffmann-La Roche Inc. from October 1989 to May 1996. Mr. Gould received a Bachelor of Arts degree in Organic Chemistry from The Johns Hopkins University, attended the New York University Graduate School of Chemistry, received a J.D. from Columbia University School of Law and an L.L.M. from New York University School of Law.
Arthur H. Hayes, Jr., M.D., 67, has served as a director since March 1996. He is currently President and Chief Operating Officer of MediScience Associates, Inc., a pharmaceutical consulting company, and is a Professor of Medicine at New York Medical College and Pennsylvania State University College of Medicine. From 1981 to 1983, Dr. Hayes served as Commissioner of the United States Food and Drug Administration. From 1986 to 1991, he was President and Chief Executive Officer of EM Pharmaceuticals, as well as a member of their board of directors. Dr. Hayes served as Provost & Dean at New York Medical College from 1983 to 1986, and served as the Director of the Institute of Human Values in Medical Ethics, International Health and Biomedical Sciences, the latter of which he also served as Chairman. Dr. Hayes has held several posts with Pennsylvania State University, which included Professor of Medicine and Pharmacology from 1977 to 1981, Dean of Admissions from 1976 to 1979 and Associate Professor of Medicine and Pharmacology and Director of the Division of Clinical Pharmacology from 1972 to 1977. Dr. Hayes currently serves on the board of directors of Myriad Genetics, Inc. (a genomic research and pharmaceutical company), Celgene Corporation (a pharmaceutical company), and Premier Research Worldwide. Dr. Hayes received his M.D. from Cornell University Medical College, and also attended Cornell's Graduate School of Medical Sciences, Department of Pharmacology. He undertook premedical studies, and attended medical school at Georgetown University. Dr. Hayes received his M.S. (philosophy, politics and economics) from Oxford University, where he was a Rhodes Scholar, and his B.A. (philosophy) from Santa Clara University.
Marc J. Ostro, Ph.D., 53, has served as a director since 2000. He has been a partner in TL Ventures, a Pennsylvania-based venture capital firm since January 1, 2002. Immediately prior to that, Dr. Ostro was a private consultant to the biotechnology industry since May 2000. From November 1997 to May 2000, he was Senior Managing Director and GroupLeader for KPMG Life Science Corporate Finance (Mergers and Acquisitions). From June 1997 to November 1997, Dr. Ostro was a Senior Vice President at Ross Financial Group engaged in portfolio management. From May 1994 to June 1997, he was Managing Director and Senior Biotechnology Analyst at UBS Securities. Prior to that, he was a Senior Vice President and Senior Biotechnology Analyst at Mabon Securities. In July 1981, he co-founded the Liposome Company (Princeton) where he held various positions in that company including President, Vice Chairman and Chief Scientific Officer until May 1993. Dr. Ostro received a B.A. in biology from Lehigh University, a Ph.D. in biochemistry from Syracuse University, and was a postdoctoral fellow and assistant professor at the University of Illinois Medical School.
The Honorable Richard N. Perle, 61, has served as a director since 2000. He is a fellow at the American Enterprise Institute. Additionally, Mr. Perle is a director of Hollinger International, Inc., a company that publishes English language newspapers in the United States, the United Kingdom, Canada and Israel. Mr. Perle is also a director of Autonomy, PLC, a company engaged in the development of various software applications, as well as a director of Morgan Crucible, PLC, a company specializing in the design, development, manufacture and marketing of engineered products for use in various industries. From 1981 to 1987, Mr. Perle was the United States Assistant Secretary of Defense for International Security Policy at the United States Department of Defense. Mr. Perle attended the London School of Economics with Honors Examinations, received a B.A. in international relations from the University of Southern California, an M.A. in politics from Princeton University, and completed various fellowships at Princeton University, the Ford Foundation and the American Council of Learned Societies.
C-41
Patricia A. Pilia, Ph.D., 54, a co-founder of the Company, has served as a director since our inception. She has served as our Secretary since November 1991, our Vice President of BioResearch and Toxicology since March 1993, and our Executive Vice President since October 1998. Additionally, in 2002, she was appointed Acting Head, Research and Development. In 1990, she co-founded, with Dr. Ainsworth, Pacific Biotechnology, Inc. (a predecessor of ours) and served as its Vice President and Director of Biotechnology. From 1983 to 1991, Dr. Pilia was an Assistant Professor of Pathology in the College of Medicine and Dental Medicine and the College of Graduate Studies at MUSC. Dr. Pilia served as the Assistant Director of the MUSC Immunopathology Diagnostic and Research Laboratories from 1985 to 1991. Since 1984 she has been a consultant on the design and development of biomedical devices and treatment modalities and the design and performance of clinical trials. Dr. Pilia received a Bachelor's degree from Boston University, a Master's degree in immunology/microbiology and a Doctoral degree in pathology from MUSC. Dr. Pilia is engaged to be married to Dr. Ainsworth, a co-founder, director and executive officer of the Company.
Robert E. Pollack, Ph.D., 61, has served as a director since 2000. He is currently Professor of Biological Sciences, Lecturer in Psychiatry at the Center for Psychoanalytic Training and Research, and Director of the Center for the Study of Science and Religion, at Columbia University. He has been a Professor of Biological Sciences at Columbia since 1978, and was Dean of Columbia College from 1982-1989. He received the Alexander Hamilton Medal from Columbia University, and has held a Guggenheim Fellowship. He currently serves on Advisory Boards of the John Templeton Foundation, California Newsreels, The Fred Friendly Seminars, the Program in Religion and Ecology of the Center for the Study of World Religions at Harvard University, and as a Senior Consultant for the Director, Program of Dialogue on Science, Ethics and Religion, American Association for the Advancement of Science. He is also currently a director and Chair of the Scientific Advisory Board of Nutrition 21, Inc., a company focusing on the development and marketing of proprietary nutritional products. Dr. Pollack graduated from Columbia University with a B.A. in physics, and received a Ph.D. in biology from Brandeis University.
Leonard P. Shaykin, 59, has served as our Chairman of the Board since June 1993, and our Chairman and Chief Executive Officer since August 1999. In 1995, Mr. Shaykin founded Shaykin & Co., LLC, a private investment and management company. Prior to founding Shaykin & Co., Mr. Shaykin was a managing partner of Adler & Shaykin, an investment partnership organized to sponsor management leveraged buyouts. Prior to that, Mr. Shaykin was Vice President, Director and a member of the Investment Committee of Citicorp Venture Capital, Ltd. and Citicorp Capital Investors, Inc., the venture capital and equity investment subsidiaries of Citicorp and Citibank. He is currently a Director of the Jerusalem Post, a subsidiary of Hollinger International, a Board Member of the Friends of Sheba Medical Center-Tel Hashomer, Israel, and a trustee of the Jackson Laboratories, a not-for-profit genetic research institute. Mr. Shaykin received a B.A. and an M.A. from the University of Chicago, and an M.B.A. from the University of Chicago Graduate School of Business.
Other Executive Officers
We have the following executive officers in addition to those who serve as directors:
Martin Batt, 60, has served as our Vice President, Chief Information Officer since September 2002. Prior to joining us, from 1986 to August 2002, he was a Partner in the consulting firm of Grisanti, Galef & Goldress specializing in the implementation of Information Technology solutions to complex business problems. Prior to that, Mr. Batt served in various Information Technology positions at U. S. Steel Corporation. Mr. Batt received a B.S. in Computer Sciences, Cum Laude, from Point Park College, Pittsburgh and has also taught Computer Sciences at the college level.
Kai P. Larson, Esq., 39, served as our Director of Legal Affairs from 1994 to December 1999, when his position was changed to Vice President and General Counsel. Prior to joining us, he worked
C-42
as an attorney in the New York office of Kirkland & Ellis. Mr. Larson received a B.A. from Brigham Young University, and a J.D. from Columbia University School of Law.
Hope Liebke, Ph.D., Esq., 57, has served as our Vice President, General Counsel, NaPro Genomics Division and Vice President, Business Development, NaPro Genomics Division since April 2002. Prior to joining us, from 1998 to 2002, she served as Of Counsel at the law firm of Fish and Neave. Previously, she held the position of Associate Attorney at the law firm of Pennie and Edmonds, LLP from 1989 to 1998. Dr. Liebke received a B.S. in Biology from Newcomb College of Tulane University and received a Ph.D. in Molecular and Cellular Biology from the University of Southern California. She performed research in the fields of biochemistry, genetics, physiology, nucleic acid sequencing and molecular biology at the University of Connecticut, Farmington and Storrs campuses and at Yale University, and received a J.D. from the University of Connecticut.
Gordon H. Link, Jr., 49, a certified public accountant and a certified management accountant, served as our Vice President and Chief Financial Officer from September 1993 to October 2002, when his position was changed to Senior Vice President and Chief Financial Officer. Prior to that, Mr. Link served concurrently as Corporate Controller of Synergen, Inc. and Treasurer of the Syntex-Synergen Neuroscience Joint Venture. From February 1991 to April 1993, Mr. Link was Treasurer of Synergen Development Corporation. From October 1983 to May 1990, Mr. Link practiced as a certified public accountant, including the position of Audit Manager with Deloitte & Touche. He attended the graduate school of the University of Denver and received undergraduate degrees in chemistry from Rensselaer Polytechnic Institute in 1976 and in accounting from Metropolitan State College in 1983.
Brian McCudden, 43, has served as our Vice President, Manufacturing since January 2002. Prior to joining us, from 1997 to December 2001, he served as Director, Manufacturing at Roche Colorado Corporation, a subsidiary of F. Hoffman-La Roche Ltd. Previously, he held the position of Director, Large Scale Manufacturing with Roche Colorado Corporation from 1995 to 1997. Mr. McCudden received a B.S. in chemical engineering, Cum Laude, from the University of Delaware and an M.B.A from Loyola College (Baltimore).
Jeffrey White, 41, joined us as President, NaPro Genomics Division in August 2002. Prior to that, Mr. White was VP/General Manager of BioChemistries and Services Division for the Life Sciences & Chemical Analysis Group at Agilent Technologies, Inc., where he was responsible for the Genomics and Proteomics businesses. Prior to this assignment Mr. White was employed at Hewlett Packard where he served in a number of senior management roles, including general management responsibility for worldwide businesses, as well as management positions in Research & Development, Marketing, Business Development, and Customer Support. He received a B.S. in Electrical Engineering/Computer Science from Worcester Polytech Institute in 1984 and an M.B.A. from Northeastern University Boston, MA, in 1989. Mr. White is an active member of the Delaware Biotech Community and serves on the board of directors for OVO, Inc., a privately-held manufacturer of ski wear.
Section 16(a) Beneficial Ownership Reporting Compliance
Under Section 16(a) of the Securities Exchange Act of 1934, our directors and certain of our officers, and persons holding more than ten percent of our common stock are required to file forms reporting their beneficial ownership of our common stock and subsequent changes in that ownership with the Securities and Exchange Commission. Such persons are also required to furnish us copies of forms so filed. Based solely upon a review of copies of such forms furnished to us, Gordon Link was late in filing a Form 4 in which he reported three transactions, Patricia Pilia, Hope Liebke and Arthur Hayes were each late in filing a Form 4 in which they reported one transaction each; and Richard Perle was late in filing a Form 5 in which he reported one transaction. No other directors or officers were late in filing any reports on Forms 3, 4 or 5.
C-43
Item 11 Executive Compensation
The following table shows for the years ended December 31, 2002, 2001 and 2000, compensation awarded or paid to, or earned by our chief executive and our four most highly compensated executive officers at December 31, 2002 (the Named Executive Officers):
| | | Annual Compensation | | | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Name and Principal Position | | Securities Underlying Options(#) | All Other Compensation(1) | ||||||||||
| Year | Salary | Bonus | |||||||||||
| Leonard P. Shaykin Chairman of the Board, Chief Executive Officer | 2002 2001 2000 | $ | 270,000 239,331 221,397 | $ | — 200,000 214,977 | — 375,000 120,000 | $ | 40,000 33,188 28,349 | |||||
Sterling K. Ainsworth Vice Chairman of the Board, President, Chief Scientific Officer | 2002 2001 2000 | 260,000 259,423 243,654 | — 100,000 203,453 | — 150,000 100,000 | 40,000 33,188 28,349 | ||||||||
Patricia A. Pilia Executive Vice President, Secretary | 2002 2001 2000 | 209,577 199,577 187,962 | — 125,000 156,114 | — 200,000 100,000 | 40,000 33,188 26,641 | ||||||||
Gordon H. Link, Jr. Senior Vice President, Chief Financial Officer | 2002 2001 2000 | 208,731 179,615 169,231 | 28,073 125,000 105,934 | (2) | — 250,000 80,000 | 40,000 33,188 28,349 | |||||||
Kai P. Larson Vice President, General Counsel | 2002 2001 2000 | 179,154 159,615 149,215 | 25,000 125,000 84,758 | — 200,000 80,000 | 40,000 33,188 28,349 | ||||||||
- (1)
- Represents our 401(k) plan and Employee Stock Ownership Plan (ESOP) contributions of common stock (valued at fair market value as of the date of the contribution) for each of the Named Executive Officers.
- (2)
- In light of an inadvertent expiration of certain options held by Mr. Link, the Board authorized a stock award to Mr. Link of 2,955 shares, with a market value of $28,073, which represented the economic value of the expired options as of the expiration date of the options.
In light of applicable regulations of the Securities and Exchange Commission, two executive officers, Hope Liebke and Brian McCudden, have been omitted from the Summary Compensation Table because the bonuses paid to each of them, which brought their total salary and bonus to levels that otherwise would have required them to be included among the four highest paid executive officers, were paid as an inducement to each of them to begin their employment with us and are not part of a recurring arrangement and are unlikely to continue.
In 2002, Ms. Liebke was paid a salary of $140,000 and a hiring bonus in April and September 2002 of $188,000, which consisted of a cash payment of $25,000 and a restricted grant of 20,000 shares of NaPro common stock at a fair market value of $163,000. During 2002, she received certain contributions of common stock under our Employee Stock Ownership Plan in the amount of $7,136. In addition, she received a grant of 100,000 stock options, which was 13.07% of the total options granted to employees during 2002, with an exercise price of $8.15 per share. The options expire on April 4, 2012. The potential realizable value of the options at an assumed annual rate of stock price appreciation for the option term of 5% is $512,549 and at an assumed annual rate of 10% is $1,298,900. Ms. Liebke did not acquire any shares upon exercise of options during 2002. At
C-44
December 31, 2002, Ms. Liebke held 100,000 stock options that were unexercisable as of that date and held no exercisable options. As of April 11, 2003, Ms. Liebke owned 55,000 shares of NaPro common stock, which includes 25,000 shares issuable upon exercise of options granted to her under the 1994 Plan.
In 2002, Mr. McCudden was paid a salary of $145,961 and a hiring bonus of $75,000, which was paid in January and September 2002. He also received during 2002 certain contributions of common stock under our Employee Stock Ownership Plan in the amount of $16,719. In addition, he received a grant during 2001 of 50,000 stock options with an exercise price of $11.25 per share. The options expire on December 13, 2011. The potential realizable value of the options at an assumed annual rate of stock price appreciation for the option term of 5% is $353,753 and at an assumed annual rate of 10% is $896,480. Mr. McCudden did not acquire any shares upon exercise of options during 2002. At December 31, 2002, Mr. McCudden held 12,500 options that were exercisable and 37,500 stock options that were unexercisable as of that date. As of April 11, 2003, Mr. McCudden owned 17,000 shares of NaPro common stock, which includes 12,500 shares issuable upon exercise of options granted to Mr. McCudden under the 1994 Plan.
No options were granted to any of the Named Executive Officers during the fiscal year ended December 31, 2002.
The following table reports information, as to each of the Named Executive Officers, concerning the number of shares subject to both exercisable and unexercisable stock options held as of December 31, 2002. Also reported are values for "in-the-money" options that represent the positive spread between the respective exercise prices of outstanding stock options and the fair market value of our common stock as of December 31, 2002:
Aggregated Options Exercises in Last Fiscal Year and Fiscal Year-End Option Values
| | | | Number of Securities Underlying Unexercised Options at Year End (#) | Value of Unexercised in-the-Money Options at Year End($) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| | Shares Acquired on Exercise (#) | | ||||||||||
| Name | Value Realized ($)(1) | |||||||||||
| Exercisable | Unexercisable | Exercisable | Unexercisable | |||||||||
| Leonard P. Shaykin | — | — | 633,768 | 341,232 | — | — | ||||||
| Sterling K. Ainsworth | — | — | 519,508 | 162,492 | — | — | ||||||
| Patricia A. Pilia | — | — | 361,001 | 199,999 | — | — | ||||||
| Gordon H. Link, Jr.(2) | 10,000 | 39,275 | 277,513 | 252,487 | — | — | ||||||
| Kai P. Larson | 10,000 | 66,675 | 61,666 | 202,500 | — | — | ||||||
- (1)
- Calculated on the basis of the closing price per share of our common stock on the date of exercise on the Nasdaq National Market, less the exercise price.
- (2)
- Mr. Link inadvertently permitted 3,334 options issued to him under the 1993 Plan to expire unexercised. In light of this inadvertent expiration, the Board authorized a stock award to Mr. Link under the 1994 Plan of 2,955 shares, with a market value of $28,073, which represented the economic value of the expired options as of the expiration date of the options.
Compensation of Directors
Pursuant to the 1994 Plan, each year non-employee directors are automatically granted, on the date of our annual meeting of stockholders, non-qualified options to purchase 10,000 shares of NaPro common stock. In addition, any non-employee director who is first appointed or elected other than at an annual meeting of stockholders automatically receives non-qualified options to purchase 10,000
C-45
shares of common stock upon such appointment or election. The 1994 Plan also provides for automatic annual grants of non-qualified stock options to purchase 10,000 shares of common stock to directors who serve as chair of the Audit, Compensation, Strategic Planning, and Research and Development committees of the Board of Directors. The 1994 Plan also provides for an automatic grant of non-qualified stock options to purchase 7,500 shares of common stock to members of the Research and Development Committee (the "RDC") upon their initial appointment to the committee, and an automatic grant of non-qualified stock options to purchase 3,000 shares of common stock to a RDC member who continues service on the RDC after an annual meeting of stockholders. In addition, the 1994 Plan permits the discretionary grant by the Board of Directors of non-qualified options to non-employee directors under certain circumstances. All such options are exercisable at an exercise price equal to the fair market value of the common stock on the date of grant and are subject to certain vesting schedules.
Directors are paid $3,000 for each meeting attended in person and $500 for each meeting attended by telephone. In addition, directors serving on committees of the Board of Directors are paid for attendance at each committee meeting as follows: $1,000 for the committee chairperson attending in person and $500 for non-chair committee members attending in person or any committee members attending by telephone. The RDC chairman receives $40,000 per year for service as chairman of the RDC. RDC members receive $2,500 for attendance at RDC meetings which are not held concurrently with regularly scheduled Board of Directors meetings. Directors are also reimbursed for their cost incurred in attending Board of Directors and committee meetings.
Employment Agreements and Termination of Employment Agreements
Effective October 1, 2001, we entered into employment agreements (collectively, the "Senior Executive Employment Agreements"), with Leonard Shaykin, Dr. Sterling Ainsworth, and Dr. Patricia Pilia (collectively, the "Senior Executives"). In addition, effective October 1, 2001, we entered into employment agreements (collectively, the "Employment Agreements") with Gordon Link and Kai Larson (collectively, the "Executive Officers"). The Senior Executive Employment Agreements, and the Employment Agreements are referred to together as the "Executive Agreements", and the Senior Executives and the Executive Officers are referred to together as the "Executives".
Each of the Senior Executive Employment Agreements provides for an initial three year employment term that expires on October 1, 2004 and is automatically renewed on each anniversary of the date of the agreement for an additional one-year term unless either party gives notice of termination to the other party at least 180 days prior to the commencement of any additional one-year term. No such notice of termination has been given by or to any of the Senior Executives. Each of the Employment Agreements provides for an initial two-year employment term that expires on October 1, 2003, and is automatically renewed on each anniversary of the date of the agreement for an additional one-year term unless either party gives notice of termination to the other party at least 180 days prior to the commencement of any additional one-year term. No such notice of termination has been given by or to any of the Executive Officers.
The Senior Executive Employment Agreements provide for initial annual base salaries for Mr. Shaykin and Drs. Ainsworth and Pilia of $270,000, $260,000 and $210,000 respectively. Under the Senior Executive Employment Agreements, in the event of a change of control, the Senior Executives are to be granted a payment equal to the greater of 100% of their prior year's bonus or 75% of their base annual salary and a payment equal to a percentage of the Senior Executive's base annual salary (300% for Mr. Shaykin, 200% for Dr. Pilia, and 150% for Dr. Ainsworth). Mr. Shaykin is a part-time employee, and is not required under his Senior Executive Employment Agreement to spend more than 32 hours in any week or 128 hours per month on NaPro affairs. However, since 1999, when Mr. Shaykin became Chief Executive Officer, he has spent a majority of his business time on NaPro's affairs.
C-46
The Employment Agreements provide for initial annual base salaries for Mr. Link and Mr. Larson of $210,000 and $180,000 respectively. Under the Employment Agreements, in the event of a change of control, the Executive Officers are to be granted a payment equal to the greater of 100% of their prior year's bonus or 75% of their base annual salary and a payment equal to 200% of the Executive Officer's base annual salary.
Under the Employment Agreements and Senior Executive Employment Agreements, Executives may receive an annual bonus in such amount, if any, as the Compensation Committee (or if the Board has no Compensation Committee at the time, then the Board), in its discretion, may award to Executives, based upon the Executive's and the Company's performance during each year of the Employment Period.
Each Executive Agreement provides for certain benefits if, prior to the end of the initial term or any additional one-year term, an Executive's employment is terminated either by us other than for Cause (as defined in the Executive Agreements) or by the Executive for good reason (as defined in the Executive Agreements). In general, each Executive would be entitled to receive, subject to certain limitations, (i) a continuance of their respective salary and bonus, if any, through the end of the initial term or the then current term, (ii) health and welfare benefits as in effect immediately prior to termination for a maximum of 18 months following termination, (iii) full vesting for all outstanding Company stock options owned by the Executive, which become immediately fully exercisable, and (iv) a bonus payment in an amount determined by whether or not a change of control of the company is occurring at the time of the termination of the Executive's employment. If a change of control occurs, the Executive is entitled to receive a payment equal to a percentage of the individual Executive's base salary, according to the terms set forth above for each named individual. If a change of control does not occur, the Executive is entitled to receive a payment equal to 100% of his or her base salary for the preceding 12 months. The foregoing benefits would be limited by the amount deductible for income tax purposes under the Internal Revenue Code of 1986, as amended (the Code).
The Executive Agreements also contain provisions (i) prohibiting disclosure of confidential information, (ii) granting to the Company rights to intellectual property developed by the Executives that relate to its business or are developed in the course of employment with NaPro, and (iii) prohibiting competition with NaPro under certain circumstances during and for five-years after the Executive's employment.
Compensation Committee Interlocks and Insider Participation
During the year ended December 31, 2002, our compensation committee consisted of Mr. Edward Erickson, The Honorable Richard Perle (Chair), and Dr. Robert Pollack. None of our executive officers serve as members of the board of directors or compensation committee of any entity that has one or more executive officers who serve on the Board of Directors or Compensation Committee.
Compensation Committee Report on Executive Compensation
The report of the Compensation Committee of the Board of Directors (the "Committee") shall not be deemed incorporated by reference by any general statement incorporating by reference this report into any filing under the Securities Act of 1933, or under the Securities Exchange Act of 1934, except to the extent that we specifically incorporate this information by reference, and shall not otherwise be deemed filed under such Acts.
The Compensation Committee implements our executive compensation policies. We are committed to executive compensation policies that promote and support our goals and that inspire executives to make a significant contribution to our financial success. Our overall compensation philosophy for executive officers has the following goals and objectives: (1) the attraction and retention of qualified personnel whose participation is important to our short-term and long-term success; and (2) the
C-47
creation of a mutual interest between executive officers and stockholders that permits executive officers to share in the risks and rewards of strategic decision-making. The Compensation Committee retained a compensation consultant to assist it in 2000. This same consultant was consulted in 2001 in connection with the Committee's review of the Employment Agreements described above.
The Committee has established the Company's executive compensation policies using the above objectives as its foundation. The Committee's practice is to review annually the compensation of each of the executive officers. The Committee also makes annual determinations of bonuses and equity-based incentive compensation including grants of stock options and restricted stock to executive officers. The following describes the three primary components of our current executive compensation program.
Base Salary. The existing base salary levels of our Chief Executive Officer, Mr. Shaykin, and the other senior executives named in the compensation table were set in connection with the signing of their Employment Agreements in October 2001 and they have not been increased since then. The decision to not increase their base salaries since that date has been driven by the Company's overriding need to conserve cash. In any event, the Compensation Committee believes that the current base salaries of our executive officers are generally below comparable companies.
Bonuses. Mr. Larson was paid a bonus in the amount of $25,000 during January 2002 relating to work he did on the Company's intellectual property portfolio in 2001. On February 11, 2002, Mr. Link was given a stock award with a value of $28,073 to replace options that he had inadvertently allowed to expire. Certain other executive officers were paid bonuses in 2002 in connection with their hiring. Ms. Liebke received a cash sign-on bonus of $25,000 and a restricted grant of 20,000 shares of NaPro common stock then valued at $163,000 upon her being hired. In 2002, Messrs. McCudden, White and Batt, respectively, received cash sign-on bonuses associated with their hiring of $75,000, $25,000 and $35,000. In each of these cases, the Compensation Committee believed that the bonuses were required to attract these employees to the Company. Because of the Company's need to conserve cash, however, the Compensation Committee awarded no other bonuses to executive officers in 2002.
Equity-based Incentives. Other than stock options granted to executive officers upon hiring, no other options were granted to executive officers during 2002. Factors considered in the decision not to grant stock options included the volatility of the Company's stock price and a desire to more fully develop measurable, long term strategic goals prior to granting of additional equity-based incentives. In this determination, the Compensation Committee took into account recommendations made to the Committee by the Chairman of the Board as well as the Committee's own evaluation of the circumstances. Although options were not granted in 2002 to existing executives, the Compensation Committee considers equity-based incentives to be an integral part of executive compensation. The grant of restricted stock awards, stock options and other awards pursuant to the 1994 Long-Term Performance Incentive Plan and the 1998 Stock Incentive Plan has been, and will continue to be, an effective method for the creation of a mutual interest between our employees and our stockholders. In granting equity-based incentives, the Committee takes into account an individual executive's performance and the performance of the Company as a whole, while taking into account the goals and the Company's overall compensation philosophy.
Compensation Committee
Richard Perle, Chair
Edward Erickson
Robert Pollack
April 29, 2003
C-48
Stock Price Performance Graph
The Stock Price Performance Graph shall not be deemed incorporated by reference by any general statement incorporating by reference this report into any filing under the Securities Act of 1933, or under the Securities Exchange Act of 1934, except to the extent that the Company specifically incorporates this information by reference, and shall not otherwise be deemed filed under such Acts.
The following graph compares the cumulative return of the Company's common stock against the Total Return Index for the NASDAQ Market (U.S.) and a peer group which is comprised of the companies listed on the NASDAQ Pharmaceutical Stock Index. The comparison assumes an initial investment of $100 on December 31, 1997 in NaPro's common stock and in each of the indices shown. All values assume the reinvestment of dividends by the companies included in these indices and are calculated as of December 31 of each year. The historical stock price performance on the graph below is not necessarily indicative of future stock price performance.
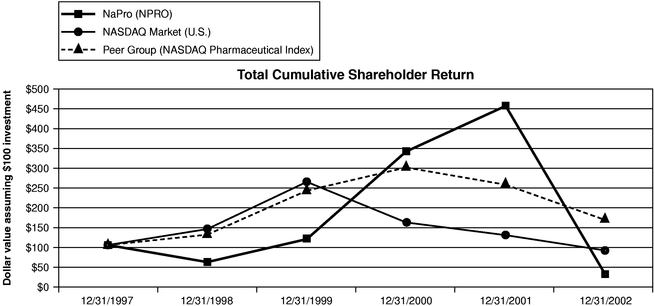
| | December 31, 1997 | December 31, 1998 | December 31, 1999 | December 31, 2000 | December 31, 2001 | December 31, 2002 | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| NaPro (NPRO) | $ | 100 | $ | 56.40 | $ | 115.20 | $ | 339.20 | $ | 456.00 | $ | 26.40 | ||||||
| NASDAQ Market (U.S.) | $ | 100 | $ | 140.99 | $ | 261.48 | $ | 157.77 | $ | 125.16 | $ | 86.53 | ||||||
| Peer Group (NASDAQ Pharmaceutical Index) | $ | 100 | $ | 126.94 | $ | 239.34 | $ | 298.54 | $ | 254.43 | $ | 164.38 | ||||||
C-49
Item 12 Security Ownership of Certain Beneficial Owners and Management
Equity Compensation Plan Information
The following table sets forth certain information as of December 31, 2002 concerning our common stock that may be issued upon the exercise of options or the purchases of restricted stock under all of our equity compensation plans approved by stockholders and equity compensation plans not approved by stockholders:
| Plan Category | (a) Number of securities to be issued upon exercise of outstanding options, warrants and rights | (b) Weighted average exercise price of outstanding options, warrants and rights | (c) Number of securities remaining available for future issuances under equity compensation plans (excluding securities reflected in column (a)) | ||||
|---|---|---|---|---|---|---|---|
| Equity compensation plans approved by security holders: | |||||||
| 1994 Long-Term Performance Incentive Plan | 4,161,641 | $ | 5.14 | 1,836,054 | |||
| 1993 Stock Option Plan | — | — | 13,435 | ||||
| Total Approved Plans | 4,161,641 | $ | 5.14 | 1,849,489 | |||
| Equity compensation plans not approved by security holders: | |||||||
| Non-plan | 16,750 | $ | 2.72 | — | |||
| 1998 Stock Option Plan | 1,359,076 | $ | 5.28 | 379,022 | |||
| Total Unapproved Plans | 1,375,826 | $ | 5.25 | 379,022 | |||
| Total Plans | 5,537,467 | $ | 5.17 | 2,228,511 | |||
Summary of Equity Compensation Plans Not Approved by Stockholders
Non-plan Stock Options
In January 1994, the Company granted to four outside directors 27,000 non-plan options to purchase shares of common stock which were immediately exercisable at a price of $2.40 and which expire in January 2004. As a result of option exercises, 16,000 of these options remained outstanding as of December 31, 2002. In September 1997, the Company granted to its employees 20,075 non-plan options to purchase shares of common stock which vested over two years and which expire in September 2007. As of December 31, 2002, 750 of these options remained outstanding.
The 1998 Stock Option Plan
In 1998, the Board of Directors adopted the 1998 Stock Option Plan (the "1998 Plan") to provide awards of stock options, stock appreciation rights, restricted stock, performance grants, or any other type of award deemed by the Board of Directors or its designated committee to employees and other individuals who perform services for the Company. The 1998 Plan provides for option grants designated as nonqualified stock options. Originally, 125,000 shares were authorized for issuance under the 1998 Plan. In 1999, 2000, 2001 and 2002 the Board of Directors approved increases in the number of authorized shares. There are currently 1,925,000 shares authorized for issuance under the 1998 Plan. Under the terms of the 1998 Plan, stock options cannot be granted to persons who are NaPro officers
C-50
subject to Section 16 of the Securities Exchange Act of 1934, as amended, or to NaPro directors. Options granted under the 1998 Plan typically vest 25% after each anniversary date of the grant, and expire ten years from the date of grant. The exercise price for stock options issued under the 1998 Plan is equal to the fair market value of the Company's common stock on the date of grant.
Security Ownership by Certain Persons
The following table sets forth certain information as of April 11, 2003 regarding the ownership of our common stock by (1) persons believed by us to be the beneficial owners of more than five percent of our outstanding common stock; (2) by each director and by each executive officer named in the Summary Compensation Table above; and (3) by all executive officers and directors as a group. Except where otherwise indicated, the address for each of the persons listed in the table is: NaPro BioTherapeutics, Inc., 6304 Spine Road, Unit A, Boulder, CO 80301.
| Name of Director, Officer or Beneficial Owner (1) | Number of Shares of Common Stock | Percent of Class | |||
|---|---|---|---|---|---|
| Leonard P. Shaykin | 1,256,322 | (2) | 4.11 | % | |
| Sterling K. Ainsworth | 1,432,782 | (3) | 4.70 | % | |
| Edward L. Erickson | 30,000 | (4) | * | ||
| George M. Gould | — | * | |||
| Arthur H. Hayes, Jr. | 80,000 | (5) | * | ||
| Marc J. Ostro | 107,500 | (6) | * | ||
| The Honorable Richard N. Perle | 70,000 | (7) | * | ||
| Patricia A. Pilia | 604,293 | (8) | 1.99 | % | |
| Robert E. Pollack | 45,500 | (9) | * | ||
| Kai P. Larson | 90,056 | (10) | * | ||
| Gordon H. Link, Jr. | 358,348 | (11) | 1.19 | % | |
| All Directors and Executive Officers as a Group (15 persons) | 4,174,322 | (12) | 13.00 | % | |
| Abbott Laboratories 100 Abbott Park Road Abbott Park, IL 60064 | 2,000,000 | (13) | 6.68 | % | |
| TL Ventures 700 Bldg., 435 Devon Park Dr. Wayne, PA 19087 | 936,862 | (14) | 3.13 | % |
- *
- Less than 1%
- (1)
- Unless otherwise noted, we believe that all persons named in the table have sole voting and investment power with respect to all shares of common stock beneficially owned by them. Percentage of beneficial ownership is based on 29,939,486 shares of common stock outstanding as of April 11, 2003, as adjusted as required by the rules promulgated by the SEC. Beneficial ownership is determined in accordance with the rules of the SEC and generally includes voting or investment power with respect to securities. Shares of stock subject to options or warrants currently exercisable or exercisable within 60 days of April 11, 2003 are deemed outstanding for computing the percentage of the person or entity holding such securities, but not outstanding for purposes of computing the percentage of any other person or entity.
- (2)
- Includes 633,768 shares of common stock issuable upon exercise of options granted to Mr. Shaykin under the 1994 Plan and 37,937 shares of common stock beneficially owned through our 401(k) and ESOP plans as of April 11,2003.
- (3)
- Includes 519,508 shares of common stock issuable upon exercise of 1994 Plan options; 36,019 shares of common stock beneficially owned through the Company's 401(k) and ESOP plans as of April 11, 2003; and 42,550 shares of common stock gifted by Dr. Ainsworth to relatives and certain other persons, which Dr. Ainsworth may be deemed to beneficially own by virtue of holding powers of attorney to vote and take certain other actions with respect to such shares. Does not
C-51
include shares held by Dr. Pilia, who is engaged to be married to Dr. Ainsworth, and Dr. Ainsworth disclaims beneficial ownership of the shares of common stock beneficially owned by Dr. Pilia as well as of the gifted shares over which Dr. Ainsworth holds powers of attorney. See note (8) below.
- (4)
- Includes 30,000 shares of common stock issuable upon exercise of options granted to Mr. Erickson under the 1994 Plan.
- (5)
- Includes 80,000 shares of common stock issuable upon exercise of options granted to Dr. Hayes under the 1994 Plan.
- (6)
- Includes 67,500 shares of common stock issuable upon exercise of options granted to Dr. Ostro under the 1994 Plan.
- (7)
- Includes 60,000 shares of common stock issuable upon exercise of options granted to Mr. Perle under the 1994 Plan.
- (8)
- Includes 361,001 shares of common stock issuable upon exercise of 1994 Plan options; 37,786 shares of common stock beneficially owned through the Company's 401(k) and ESOP plans as of April 11, 2003; and 10,800 shares of common stock gifted by Dr. Pilia to relatives and certain other persons which Dr. Pilia may be deemed to beneficially own by virtue of holding powers of attorney to vote and take certain other actions with respect to such shares. Does not include shares held by Dr. Ainsworth, who is engaged to be married to Dr. Pilia. Dr. Pilia disclaims beneficial ownership of shares of common stock beneficially owned by Dr. Ainsworth and the gifted shares over which Dr. Pilia holds powers of attorney. See note (3) above.
- (9)
- Includes 45,500 shares of common stock issuable upon exercise of options granted to Dr. Pollack under the 1994 Plan.
- (10)
- Includes 61,666 shares of common stock issuable upon the exercise of options granted to Mr. Larson under the 1994 Plan and 28,391 shares beneficially owned through our 401(k) and ESOP plans as of April 11, 2003.
- (11)
- Includes 277,513 shares of common stock issuable upon the exercise of options granted to Mr. Link under the 1994 Plan and 33,304 shares of common stock beneficially owned through our 401(k) and ESOP plans as of April 11, 2003.
- (12)
- Includes an aggregate of 2,173,956 shares of common stock issuable upon exercise of outstanding stock options held by such persons.
- (13)
- Information in the table as to beneficial ownership of common stock by Abbott Laboratories is based upon filings on Schedule 13D made by Abbott Laboratories on April 27, 2001.
- (14)
- Information in the table as to beneficial ownership of common stock by TL Ventures is based upon the registration statement that was declared effective in July 2002. This includes 47,973 shares of common stock issued in lieu of cash interest payments.
Item 13 Certain Relationships and Related Transactions
Arthur H. Hayes, Jr., M.D., provides certain consulting services to us. We are parties with MediScience Associates to a consulting agreement (the "MediScience Agreement") whereby Dr. Hayes, who is President and Chief Operating Officer of MediScience, provides us with consulting services in a variety of areas, including clinical research planning, strategic positioning and regulatory guidance. We make quarterly payments to MediScience under the MediScience Agreement in the amount of $12,500 for such services. Dr. Hayes is obligated to provide consulting services to us under the MediScience Agreement indefinitely, but the MediScience Agreement is terminable by us or MediScience at any time with 90 days' prior written notice.
C-52
In February 2002, we sold privately $8.0 million of our common stock issued at $9 per share and $8.0 million principal of five-year 4% debentures convertible into common stock at $15 per share to TL Ventures V, L.P. and one of its affiliated funds (collectively, the "TL Venture Funds"). Dr. Ostro, one of our directors, is a managing director of an affiliate of the investment manager of the TL Venture Funds, and a voting limited partner in the TL Venture Funds. No placement agent was involved in the transaction. The net proceeds of the transaction were $15.6 million. As part of this transaction, we recorded a discount attributable to the conversion feature of the convertible debentures in the amount of $3.1 million, which is being amortized over the term of the debentures. We maintain a registration statement with the Securities and Exchange Commission to register the resale of the common stock, common stock issuable upon conversion of the debentures and common stock issuable in lieu of cash interest on the debentures under the Securities Act of 1933. We may pay the debenture interest in cash or common stock at our option. The debentures automatically convert into common stock during the first two years if the average price of the common stock for 15 trading days exceeds $22, and during the last three years if the average price for 30 trading days exceeds $16. If we have insufficient funds to repay the debentures in full at maturity or upon acceleration, the holders of the debentures may elect to convert the outstanding principal balance and any unpaid accrued interest on the debentures into shares of common stock at a conversion price equal to the then current 20 day average trading price of the common stock.
Signatures
Pursuant to Section 13 of the Securities Exchange Act of 1934, we have caused this amendment to our report on Form 10-K to be signed on our behalf.
NAPRO BIOTHERAPEUTICS, INC.
| April 30, 2003 | ||
| /s/GORDON H. LINK, JR. Gordon H. Link, Jr. Senior Vice President, Chief Financial Officer (Principal Financial Officer) |
C-53
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 10-K/A
AMENDMENT NO. 2
TO ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(D) OF THE
SECURITIES EXCHANGE ACT OF 1934
| (Mark One) | |
ý | ANNUAL REPORT UNDER SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the fiscal year ended December 31, 2002 | |
o | TRANSITION REPORT UNDER SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the transition period from to | |
Commission File Number 0-24320
NaPro BioTherapeutics, Inc.
(Exact name of registrant as specified in its charter)
| Delaware (State or other jurisdiction of incorporation or organization) | 84-1187753 (I.R.S. Employer Identification No.) | |
4840 Pearl East Circle, Suite 300W Boulder, CO 80301 (303) 516-8500 (Address, including zip code, and telephone number, including area code of registrant's principal executive offices) | ||
Securities registered pursuant to Section 12(b) of the Act: None
Securities registered pursuant to Section 12(g) of the Act:
Common Stock, $0.0075 par value; Preferred Stock Purchase Rights
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. Yes ý No o
Indicate by check mark if disclosure of delinquent filers pursuant to Item 405 of Regulation S-K is not contained herein, and will not be contained, to the best of registrant's knowledge, in definitive proxy or information statements incorporated by reference in Part III of this Form 10-K or any amendment to this Form 10-K. o
Indicate by check mark whether the registrant is an accelerated filer (as defined in Rule 12b-2 of the Act). Yes ý No o
The approximate aggregate market value of the voting and non-voting common equity held by non-affiliates of the registrant was $151,191,000 as of June 28, 2002 (the last business day of the registrant's second fiscal quarter in 2002). For purposes of determining this number, 6,697,884 shares of common stock held by affiliates are excluded. For purposes of making this calculation, the registrant has defined affiliates as including all directors, executive officers and beneficial owners of more than ten percent of the common stock of the Company.
As of August 5, 2003, the Registrant had 30,762,631 shares of Common Stock outstanding.
C-54
NaPro BioTherapeutics, Inc. hereby amends its Form 10-K for the period from January 1, 2002 to December 31, 2002, as amended (the "Form 10-K"), as set forth in this Form 10-K/A (the "Form 10-K/A"). This Form 10-K/A is being amended to file Exhibits 3.8 and 10.38, to revise the Exhibit Index to add Exhibits 3.8 and 10.32 to 10.38, and to revise the Exhibit Index to show the proper date of the Certificate of Amendment listed as Exhibit 3.3. This Form 10-K/A does not reflect events occurring after the filing of the original Form 10-K, as previously amended, or modify or update the disclosures therein in any way other than as required to reflect the amendment set forth below.
C-55
Item 15. Exhibits, Financial Statement Schedules, and Reports on Form 8-K
Financial Statements
The Financial Statement Index is on Page F-1.
Financial Statement Schedules
All schedules are omitted because they are not applicable or not required or because the information is included in the consolidated financial statements or the notes thereto.
Exhibits and Reports on Form 8-K
We filed a Current Report on Form 8-K dated January 24, 2003 reporting the change in our fiscal year.
We filed a Current Report on Form 8-K dated February 13, 2003 reporting the application for the transfer of our common stock to the Nasdaq SmallCap Market.
| Exhibit Number | Description of Exhibit | |
|---|---|---|
| 3.1 | Amended and Restated Certificate of Incorporation of the Company, as amended August 2, 1996(1) | |
3.2 | Certificate of Amendment dated September 29, 1998 to the Amended and Restated Certificate of Incorporation of the Company(2) | |
3.3 | Certificate of Amendment dated October 31, 2000 to the Amended and Restated Certificate of Incorporation of the Company(3) | |
3.4 | Bylaws of the Company as amended through December 2000(4) | |
3.5 | Certificate of Designation for Convertible Preferred Stock, Series A(5) | |
3.6 | Certificate of Designation for Series B Junior Participating Preferred Stock(6) | |
3.7 | Certificate of Designation for Series C Senior Convertible Preferred Stock(7) | |
3.8 | Certificate of Amendment dated March 3, 2003 to the Amended and Restated Certificate of Incorporation of the Company | |
4.1 | Common Stock Certificate(8) | |
4.2 | Amended and Restated Rights Agreement dated September 25, 2001 between the Company and American Stock Transfer and Trust Company, as Rights Agent(9) | |
4.3 | Form of 4% Convertible Subordinated Debenture due 2007(10) | |
10.1* | Company's 1993 Stock Option Plan(8) | |
10.2* | Amendment dated December 11, 2000 to the Company's 1993 Stock Option Plan(4) | |
10.3* | Company's 1994 Long-Term Performance Incentive Plan, as amended and restated effective June 21, 2001(11) | |
10.4 | Company's 1998 Stock Incentive Plan as amended and restated effective September 25, 2001(11) | |
10.5* | Employment Agreement effective October 1, 2001 between the Company and Leonard Shaykin(16) | |
C-56
10.6* | Employment Agreement effective October 1, 2001 between the Company and Sterling Ainsworth(16) | |
10.7* | Employment Agreement effective October 1, 2001 between the Company and Patricia Pilia(16) | |
10.8* | Employment Agreement effective October 1, 2001 between the Company and Gordon Link(16) | |
10.9* | Employment Agreement effective October 1, 2001 between the Company and Kai Larson(16) | |
10.10* | Employment Agreement effective October 1, 2001 between the Company and David Denny(16) | |
10.11* | Employment Agreement effective October 1, 2001 between the Company and James McChesney(16) | |
10.12* | Employment Agreement effective October 1, 2001 between the Company and Steve Bannister(16) | |
10.13* | Employment Agreement effective October 1, 2001 between the Company and Brenda Fielding(16) | |
10.14 | Second Amended and Restated Master Agreement dated June 5, 2000 between the Company and F.H. Faulding & Co., Ltd.(12) | |
10.15 | European Agreement dated March 2, 2001 between the Company and F.H. Faulding & Co., Ltd.(4) | |
10.16 | Development, License and Supply Agreement dated July 23, 1999 by and between the Company and Abbott Laboratories(14) | |
10.17 | Loan and Security Agreement dated July 23, 1999 by and between the Company and Abbott Laboratories(14) | |
10.18 | Stock Purchase Agreement dated July 23, 1999 by and between the Company and Abbott Laboratories(14) | |
10.19 | Amendment dated June 23, 2000 to Development, License and Supply Agreement by and between the Company and Abbott Laboratories(15) | |
10.20 | Amendment dated June 23, 2000 to Stock Purchase Agreement by and between the Company and Abbott Laboratories(15) | |
10.21 | Amendment dated November 28, 2001 to Development, License and Supply Agreement by and between the Company and Abbott Laboratories(16) | |
10.22 | License Agreement dated November 21, 2000 by and between the Company and The University of Delaware and Thomas Jefferson University(4) | |
10.23+ | Release and License Agreement dated November 28, 2001 by and between Bristol-Meyers Squibb Co. and the Company(16) | |
10.24 | Lease dated October 16, 1995 between the Company and Gunbarrel Facility L.L.C.(13) | |
10.25 | First Amendment to Lease November 27, 1995 between the Company and Gunbarrel Facility L.L.C.(13) | |
10.26 | Form of Subscription Agreement including Registration Rights(4) | |
C-57
10.27 | Securities Purchase Agreement dated February 13, 2002 between T/L Ventures, Inc. and the Company(10) | |
10.28 | Registration Rights Agreement dated February 13, 2002 between T/L Ventures, Inc. and the Company(10) | |
10.29 | Agreement dated November 7, 2001 between R. J. Reynolds Tobacco Company and the Company(16) | |
10.30 | Agreement dated December 31, 2002 between Pangene Corporation and the Company** | |
10.31 | Fourth Amendment to lease between the Company and Gunbarrel Facility L.L.C.** | |
10.32 | Culture Agreement dated March 1, 1996 between Zelenka Nursery, Inc. and the Company(17) | |
10.33 | Agreement for Sale, Harvest and Storage of Nursery Stock dated May 1, 1996 between Zelenka Nursery, Inc. and the Company(17) | |
10.33 | Culture Agreement dated March 1, 1997 between Zelenka Nursery, Inc. and the Company(1) | |
10.34 | Lease Agreement dated March 1, 1997 between Zelenka Nursery, Inc. and the Company(1) | |
10.35 | Agreement for Sale, Harvest and Storage of Nursery Stock dated March 1, 1997 between Zelenka Nursery, Inc. and the Company(1) | |
10.36 | Culture Agreement dated July 26, 1997 between Cass-Mill, Inc. and the Company(18) | |
10.37 | Lease Agreement dated April 28, 2000 between Cass-Mill, Inc. and the Company(4) | |
10.38* | Form of Director and Officer Indemnification Agreement (together with Schedule required by Instruction 2 to Item 601 Regulation S-K) | |
21.1 | List of Subsidiaries(16) | |
23.1 | Consent of Ernst & Young LLP** | |
99 | Certification of Chief Executive Officer and Chief Financial Officer pursuant to Section 906 of the Public Company Accounting Reform and Investor Protection Act of 2002** |
- *
- A management compensation plan.
- **
- Previously filed.
- +
- Portions have been omitted pursuant to a request for confidential treatment.
- (1)
- Incorporated herein by reference to the Company's Annual Report on Form 10-K filed with the Commission for the year ended December 31, 1996 (File No. 0-24320).
- (2)
- Incorporated herein by reference to the Company's Annual Report on Form 10-K for the year ended December 31, 1998 (File No. 0-24320).
- (3)
- Incorporated herein by reference to the Company's Quarterly Report on Form 10-Q for the period ending September 30, 2000 (File No. 0-24320).
- (4)
- Incorporated herein by reference to the Company's Annual Report on Form 10-K for the year ended December 31, 2000 (File No. 0-24320).
- (5)
- Incorporated herein by reference to the Company's Quarterly Report on Form 10-Q for the quarter ending June 20, 1995 (File No. 0-24320)
C-58
- (6)
- Incorporated herein by reference to the Company's Current Report on Form 8-K dated November 8, 1996 (File No. 0-24320).
- (7)
- Incorporated herein by reference to the Company's Registration Statement on Form S-3 filed on December 16, 1997 (File No. 333-42419)
- (8)
- Incorporated herein by reference to the Registration Statement on Form S-1 of the Company, filed with the Commission on July 24, 1994 (File No. 33-78016).
- (9)
- Incorporated herein by reference to the Registration Statement on Form 8-A12G/A of the Company, filed with the Commission on October 23, 2001 (File No. 0-24320)
- (10)
- Incorporated herein by reference to the Company's Current Report on Form 8-K, dated February 13, 2002 (File No. 0-24320).
- (11)
- Incorporated herein by reference to the Company's Quarterly Report on Form 10-Q for the period ending September 30, 2001 (File No. 0-24320).
- (12)
- Incorporated herein by reference to the Company's Quarterly Report on Form 10-Q for the period ending June 30, 2000 (File No. 0-24320).
- (13)
- Incorporated herein by reference to the Company's Annual Report on Form 10-K for the year ended December 31, 1995 (File No. 0-24320).
- (14)
- Incorporated herein by reference to the Company's Current Report on Form 8-K, dated July 23, 1999 (File No. 0-24320).
- (15)
- Incorporated herein by reference to the Company's Quarterly Report on Form 10-Q for the period ending June 30, 2000 (File No. 0-24320).
- (16)
- Incorporated herein by reference to the Company's Annual Report on Form 10-K for the year ended December 31, 2001 (File No. 0-24320).
- (17)
- Incorporated herein by reference to the Company's Registration Statement on Form S-1 of the Company, filed with the Commission on August 1, 1996 (333-3051).
- (18)
- Incorporated herein by reference to the Company's Annual Report on Form 10-K for the year ended December 31, 1999 (File No. 0-24320).
C-59
Pursuant to the requirements of Section 13 of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
| NAPRO BIOTHERAPEUTICS, INC. | ||
/s/ GORDON H. LINK, JR. Gordon H. Link, Jr. Senior Vice President and Chief Financial Officer (Principal Financial Officer) | Date: August 8, 2003 |
C-60
I, Leonard P. Shaykin, certify that:
1. I have reviewed this annual report on Form 10-K/A of NaPro BioTherapeutics, Inc.;
2. Based on my knowledge, this annual report does not contain any untrue statement of a material fact or omit to state a material fact necessary to make the statements made, in light of the circumstances under which such statements were made, not misleading with respect to the period covered by this annual report;
3. Based on my knowledge, the financial statements, and other financial information included in this annual report, fairly present in all material respects the financial condition, results of operations and cash flows of the registrant as of, and for, the periods presented in this annual report;
4. The registrant's other certifying officer and I are responsible for establishing and maintaining disclosure controls and procedures (as defined in Exchange Act Rules 13a-14 and 15d-14) for the registrant, and we have:
a) designed such disclosure controls and procedures to ensure that material information relating to the registrant, including its consolidated subsidiaries, is made known to us by others, particularly during the period in which this annual report is being prepared;
b) evaluated the effectiveness of the registrant's disclosure controls and procedures as of a date within 90 days prior to the filing date of this annual report (the "Evaluation Date"); and
c) presented in this annual report our conclusions about the effectiveness of the disclosure controls and procedures based on our evaluation as of the Evaluation Date;
5. The registrant's other certifying officer and I have disclosed, based on our most recent evaluation, to the registrant's auditors and the audit committee of registrant's board of directors (or persons performing the equivalent function):
a) all significant deficiencies in the design or operation of internal controls which could adversely affect the registrant's ability to record, process, summarize and report financial data and have identified for the registrant's auditors any material weaknesses in internal controls; and
b) any fraud, whether or not material, that involves management or other employees who have a significant role in the registrant's internal controls; and
6. The registrant's other certifying officer and I have indicated in this annual report whether or not there were significant changes in internal controls or in other factors that could significantly affect internal controls subsequent to the date of our most recent evaluation, including any corrective actions with regard to significant deficiencies and material weaknesses.
| Date: August 8, 2003 | /s/ LEONARD P. SHAYKIN Leonard P. Shaykin Chairman of the Board of Directors Chief Executive Officer |
C-61
I, Gordon H. Link, Jr., certify that:
1. I have reviewed this annual report on Form 10-K/A of NaPro BioTherapeutics, Inc.;
2. Based on my knowledge, this annual report does not contain any untrue statement of a material fact or omit to state a material fact necessary to make the statements made, in light of the circumstances under which such statements were made, not misleading with respect to the period covered by this annual report;
3. Based on my knowledge, the financial statements, and other financial information included in this annual report, fairly present in all material respects the financial condition, results of operations and cash flows of the registrant as of, and for, the periods presented in this annual report;
4. The registrant's other certifying officer and I are responsible for establishing and maintaining disclosure controls and procedures (as defined in Exchange Act Rules 13a-14 and 15d-14) for the registrant and we have:
a) designed such disclosure controls and procedures to ensure that material information relating to the registrant, including its consolidated subsidiaries, is made known to us by others, particularly during the period in which this annual report is being prepared;
b) evaluated the effectiveness of the registrant's disclosure controls and procedures as of a date within 90 days prior to the filing date of this annual report (the "Evaluation Date"); and
c) presented in this annual report our conclusions about the effectiveness of the disclosure controls and procedures based on our evaluation as of the Evaluation Date;
5. The registrant's other certifying officer and I have disclosed, based on our most recent evaluation, to the registrant's auditors and the audit committee of registrant's board of directors (or persons performing the equivalent function):
a) all significant deficiencies in the design or operation of internal controls which could adversely affect the registrant's ability to record, process, summarize and report financial data and have identified for the registrant's auditors any material weaknesses in internal controls; and
b) any fraud, whether or not material, that involves management or other employees who have a significant role in the registrant's internal controls; and
6. The registrant's other certifying officer and I have indicated in this annual report whether or not there were significant changes in internal controls or in other factors that could significantly affect internal controls subsequent to the date of our most recent evaluation, including any corrective actions with regard to significant deficiencies and material weaknesses.
| Date: August 8, 2003 | /s/ GORDON H. LINK, JR. Gordon H. Link, Jr. Senior Vice President and Chief Financial Officer |
C-62
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
Form 10-Q
Quarterly Report Pursuant to Section 13 or 15(d) of the
Securities Exchange Act of 1934
For the quarterly period ended July 2, 2003
Commission File Number 0-24320
NAPRO BIOTHERAPEUTICS, INC.
(Exact name of Registrant as specified in its charter)
| Incorporated in Delaware | IRS ID No. 84-1187753 | |||
| 4840 Pearl East Circle, Suite 300W Boulder, Colorado 80301 (303) 516-8500 |
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. ý Yes o No
Indicate by check mark whether the registrant is an accelerated filer (as defined in Rule 12b-2 of the Act). o Yes ý No
As of July 23, 2003, the registrant had 30,762,631 shares of common stock, $0.0075 par value, outstanding.
NaPro BioTherapeutics, Inc. and Subsidiaries
Table of Contents
| | | | Page | |||
|---|---|---|---|---|---|---|
| Part I | Financial Information | D-3 | ||||
Item 1 | Financial Statements | D-3 | ||||
Item 2 | Management's Discussion and Analysis of Financial Condition and Results of Operations | D-11 | ||||
Item 3 | Quantitative and Qualitative Disclosures about Market Risk | D-21 | ||||
Item 4 | Controls and Procedures | D-21 | ||||
Part II | Other Information | D-26 | ||||
Item 1 | Legal Proceedings | D-26 | ||||
Item 4 | Submission of Matters to a Vote of Security Holders | D-27 | ||||
Item 6 | Exhibits and Reports on Form 8-K | D-27 | ||||
Signatures | D-28 | |||||
Exhibits
Exhibit 31.1 | Certification of Chief Executive Officer pursuant to Section 302(a) of the Sarbanes-Oxley Act of 2002. | |
Exhibit 31.2 | Certification of Chief Financial Officer pursuant to Section 302(a) of the Sarbanes-Oxley Act of 2002. | |
Exhibit 32.1 | Certification of Chief Executive Officer pursuant to Section 906 of the Sarbanes-Oxley Act of 2002 (18 U.S.C. Section 1350). | |
Exhibit 32.2 | Certification of Chief Financial Officer pursuant to Section 906 of the Sarbanes-Oxley Act of 2002 (18 U.S.C. Section 1350). |
D-2
NaPro BioTherapeutics, Inc. and Subsidiaries
Consolidated Condensed Balance Sheets
(In thousands, except per share data)
| | July 2, 2003 | December 31, 2002 | ||||||
|---|---|---|---|---|---|---|---|---|
| | (unaudited) | | ||||||
| ASSETS | ||||||||
| Current assets: | ||||||||
| Cash and cash equivalents | $ | 5,836 | $ | 6,762 | ||||
| Accounts receivable, net of allowance for doubtful accounts | 8,097 | 9,340 | ||||||
| Inventory | 6,871 | 9,397 | ||||||
| Prepaid expense and other current assets | 1,530 | 977 | ||||||
| Total current assets | 22,334 | 26,476 | ||||||
| Property, plant and equipment, net | 11,154 | 13,731 | ||||||
| Inventory—raw materials | 6,106 | 3,781 | ||||||
| Other assets | 3,097 | 1,340 | ||||||
| Total assets | $ | 42,691 | $ | 45,328 | ||||
| LIABILITIES AND STOCKHOLDERS' EQUITY | ||||||||
| Current liabilities: | ||||||||
| Accounts payable and accrued liabilities | $ | 6,653 | $ | 5,234 | ||||
| Accrued payroll and payroll taxes | 1,265 | 1,147 | ||||||
| Notes payable—current portion | 20,188 | 102 | ||||||
| Deferred income | 1,250 | 1,150 | ||||||
| Total current liabilities | 29,356 | 7,633 | ||||||
| Notes payable—long term | 81 | 19,861 | ||||||
| Deferred income—long term | 5,327 | 5,887 | ||||||
| Convertible debentures | 5,418 | 5,151 | ||||||
| Commitments and contingencies | ||||||||
| Stockholders' equity: | ||||||||
| Preferred stock, $.001 par value; 2,000,000 shares authorized; none issued | — | — | ||||||
| Common stock, $.0075 par value; 66,000,000 shares authorized; 30,762,631 and 29,964,292 shares issued at July 2, 2003 and December 31, 2002, respectively | 231 | 225 | ||||||
| Additional paid-in capital | 111,017 | 110,430 | ||||||
| Accumulated deficit | (108,558 | ) | (103,678 | ) | ||||
| Treasury stock, 54,306 shares at cost at July 2, 2003 and December 31, 2002 | (181 | ) | (181 | ) | ||||
| Total stockholders' equity | 2,509 | 6,796 | ||||||
| Total liabilities and stockholders' equity | $ | 42,691 | $ | 45,328 | ||||
See accompanying notes to Consolidated Condensed Financial Statements.
D-3
NaPro BioTherapeutics, Inc. and Subsidiaries
Consolidated Condensed Statements of Operations
(In thousands, except per share data)
(Unaudited)
| | Quarter Ended | Six Months Ended | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| | July 2, 2003 | June 30, 2002 | July 2, 2003 | June 30, 2002 | ||||||||||
| Product sales | $ | 7,561 | $ | 9,490 | $ | 14,449 | $ | 16,226 | ||||||
| Expenses: | ||||||||||||||
| Cost of sales | 4,107 | 7,029 | 7,441 | 14,129 | ||||||||||
| Research and development | 2,495 | 4,035 | 5,490 | 8,328 | ||||||||||
| General and administrative | 2,755 | 2,869 | 5,819 | 4,913 | ||||||||||
| 9,357 | 13,933 | 18,750 | 27,370 | |||||||||||
| Operating loss | (1,796 | ) | (4,443 | ) | (4,301 | ) | (11,144 | ) | ||||||
| Other income (expense): | ||||||||||||||
| License fee income | 280 | 8,010 | 560 | 8,360 | ||||||||||
| Interest income | 18 | 82 | 47 | 151 | ||||||||||
| Interest expense | (632 | ) | (594 | ) | (1,186 | ) | (975 | ) | ||||||
| Net (loss) income | $ | (2,130 | ) | $ | 3,055 | $ | (4,880 | ) | $ | (3,608 | ) | |||
| Basic (loss) income per share | $ | (0.07 | ) | $ | 0.10 | $ | (0.16 | ) | $ | (0.12 | ) | |||
| Diluted (loss) income per share | $ | (0.07 | ) | $ | 0.10 | $ | (0.16 | ) | $ | (0.12 | ) | |||
| Basic weighted average shares outstanding | 30,507 | 29,724 | 30,221 | 29,408 | ||||||||||
| Diluted weighted average shares outstanding | 30,507 | 31,465 | 30,221 | 29,408 | ||||||||||
See accompanying notes to Consolidated Condensed Financial Statements.
D-4
NaPro BioTherapeutics, Inc. and Subsidiaries
Consolidated Condensed Statements of Cash Flows
(In thousands)
(Unaudited)
| | Six Months Ended | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| | July 2, 2003 | June 30, 2002 | |||||||
| Operating activities: | |||||||||
| Net loss | $ | (4,880 | ) | $ | (3,608 | ) | |||
| Adjustments to reconcile net loss to net cash used in operating activities: | |||||||||
| Depreciation and amortization | 1,682 | 1,030 | |||||||
| Accretion of debt issue cost | 103 | 32 | |||||||
| Amortization of debt discount | 246 | 159 | |||||||
| Accretion of license fee income | (560 | ) | (360 | ) | |||||
| License fees paid with common stock | — | 468 | |||||||
| Compensation paid with common stock, options and warrants | 88 | 230 | |||||||
| Retirement contributions paid with common stock | 218 | 950 | |||||||
| Asset writedown | 30 | — | |||||||
| Changes in operating assets and liabilities: | |||||||||
| Accounts receivable | 1,243 | (3,596 | ) | ||||||
| Inventory | 920 | (1,062 | ) | ||||||
| Prepaid expense and other assets | (195 | ) | (79 | ) | |||||
| Accounts payable and accrued liabilities | 1,419 | 421 | |||||||
| Accrued payroll and payroll taxes | 118 | 522 | |||||||
| Deferred income | 100 | — | |||||||
| Net cash provided by (used in) operating activities | 532 | (4,893 | ) | ||||||
Investing activities: | |||||||||
| Additions to property and equipment | (731 | ) | (3,798 | ) | |||||
| Acquisition of patents | (400 | ) | — | ||||||
| Investment in Chromadex | (554 | ) | — | ||||||
| Net cash used in investing activities | (1,685 | ) | (3,798 | ) | |||||
Financing activities: | |||||||||
| Proceeds from convertible debentures, net of issuance cost | — | 7,708 | |||||||
| Proceeds from notes payable | 486 | 302 | |||||||
| Payments of notes payable | (262 | ) | (291 | ) | |||||
| Proceeds from the sale of common stock, net of issuance cost | — | 7,823 | |||||||
| Proceeds from the exercise of common stock options and warrants | 3 | 183 | |||||||
| Net cash provided by financing activities | 227 | 15,725 | |||||||
| Net (decrease) increase in cash and cash equivalents | (926 | ) | 7,034 | ||||||
| Cash and cash equivalents at beginning of period | 6,762 | 10,144 | |||||||
| Cash and cash equivalents at end of period | $ | 5,836 | $ | 17,178 | |||||
See accompanying notes to Consolidated Condensed Financial Statements.
D-5
NaPro BioTherapeutics, Inc. and Subsidiaries
Consolidated Condensed Statements of Cash Flows (continued)
(In thousands)
(Unaudited)
| | Six Months Ended | ||||||
|---|---|---|---|---|---|---|---|
| | July 2, 2003 | June 30, 2002 | |||||
| Supplemental disclosure of cash flow information: | |||||||
| Interest paid | $ | 821 | $ | 699 | |||
| Non-cash investing and financing activities: | |||||||
| Issuance of common stock to prepay retirement plan contributions | 218 | 950 | |||||
| Transfer of fixed assets for investment in Chromadex | 946 | — | |||||
| Issuance of common stock to prepay license fees | — | 837 | |||||
| Depletion of plantation cost to inventory | 719 | 695 | |||||
| Issuance of restricted common stock to prepay retention bonus | 66 | — | |||||
See accompanying notes to Consolidated Condensed Financial Statements.
D-6
NaPro BioTherapeutics, Inc.
Notes to Consolidated Condensed Financial Statements
July 2, 2003
(Unaudited)
Note 1. Basis of Presentation
The accompanying financial statements are unaudited. However, in the opinion of management, the financial statements reflect all adjustments, consisting of only normal recurring adjustments, necessary for fair presentation. Interim results of operations are not indicative of results for the full year. These financial statements should be read in conjunction with the Company's Annual Report on Form 10-K and Form 10-K/A for the year ended December 31, 2002.
Effective January 1, 2003, the Company changed its fiscal year, from a calendar year ending on December 31 to a 52-53 week fiscal year ending on the Wednesday closest to December 31. As a result of this change, the Company's three fiscal quarters in 2003 end on April 2, July 2, and October 1, 2003, and its fiscal year will end on December 31, 2003. Thereafter, the Company will maintain a 52-53 week cycle ending on a Wednesday and the Company will add a 53rd week to every fifth or sixth fiscal year.
Note 2. Reclassifications
Certain data in the consolidated condensed financial statements of the prior year has been reclassified to conform to the current year presentation.
Note 3. Revenue Recognition
Product sales: NaPro recognizes revenue from product sales at the time of shipment, as the title passes to the customer, and the customer assumes the risks and rewards of ownership. Payments received in advance against future sales are recorded as deferred revenue until earned.
License fees: NaPro capitalizes license fees and amortizes them to income over the estimated economic life of the license. The amortization period consists of amortizing 80% of fees to income over the first five years of the license, and the remaining 20% of the fees to income over the remaining period of the license.
The Company recognizes income from development milestones when the milestone is achieved and the Company has no future obligation to perform additional work associated with the given milestone.
Note 4. Proposed Sale of Paclitaxel Business
In April 2003, the Company announced that it is offering to sell its worldwide polyethoxylated castor oil formulated injectable paclitaxel business, its first product approved for commercial sale. Polyethoxylated castor oil formulations are the only paclitaxel formulations currently approved by the United States Food and Drug Administration and other international regulatory agencies. Proceeds from the sale would be used to fund the development and commercialization of products based on NaPro's proprietary Gene Editing and Oncology platforms and to retire $20.0 million of debt owed to Abbott Laboratories. The contemplated sale would transfer all of the Company's assets related to the manufacture and sale of paclitaxel, which comprises a significant portion of the Company's total assets and its current operations. The assets to be sold include the Company's leased bulk paclitaxel manufacturing facilities and related production equipment; yew tree plantations (which includes related cultivation agreements, leases and yew trees owned by the Company); contracts with Abbott Laboratories (Abbott), Mayne Pharma, Tzamal Pharma (Tzamal), and JCR Pharmaceuticals Co., Ltd. (JCR) and other contracts related to the paclitaxel business; regulatory filings; and patents and
D-7
intellectual property relating to the manufacture and formulation of generic cremaphor injectible paclitaxel. The Company expects to retain rights for formulations other than polyethoxylated castor oil formulations. The timing and terms of sale of the paclitaxel business are uncertain at this time, and the Company can not give assurance that any transaction will occur. If the sale of the paclitaxel business is successful, NaPro will exit that business. If the transaction does not occur, the Company expects to remain in the paclitaxel business for the foreseeable future.
Note 5. Sale of Analytical Services group
In April 2003, the Company sold its technical and analytical services group to privately held ChromaDex, Inc. in exchange for approximately 15%, on a fully diluted basis, of the then outstanding common stock of ChromaDex, Inc. Ten Company employees transferred to ChromaDex in April and eight employees may transfer in September 2003 as part of this transaction. In exchange for the common stock received, the Company sold property and equipment valued at approximately $1.0 million, as well as provided rents and other subsidies of approximately $500,000, which included a payment of $300,000 in cash at the closing. ChromaDex assumed the lease for NaPro's research facility in Boulder, Colorado as part of this transaction. ChromaDex is a supplier of phytochemical reference standards for the nutraceutical, dietary supplement and functional food industries.
Note 6. Inventory
Inventory consists of the following (in thousands):
| | July 2, 2003 | December 31, 2002 | |||||
|---|---|---|---|---|---|---|---|
| Raw materials | $ | 1,966 | $ | 3,858 | |||
| Work in process | 2,724 | 3,402 | |||||
| Finished goods | 2,181 | 2,137 | |||||
| Total current inventory | 6,871 | 9,397 | |||||
| Raw materials-long-term | 6,106 | 3,781 | |||||
| Total inventory | $ | 12,977 | $ | 13,178 | |||
Long-term inventory consists of raw materials that are not expected to be utilized during the next twelve months, due to the timing of biomass harvests and anticipated future changes to our production process.
In May 2003, the Company recorded a loss of approximately $1.0 million resulting from the writedown of its current year harvest to the lower of cost or market. The writedown resulted from an early harvest of fields to reduce on-going maintenance costs.
D-8
Note 7. Property, Plant and Equipment
Property, plant and equipment consists of the following (in thousands):
| | July 2, 2003 | December 31, 2002 | |||||
|---|---|---|---|---|---|---|---|
| Land | $ | 718 | $ | 718 | |||
| Furniture, fixtures and office equipment | 1,578 | 987 | |||||
| Laboratory and production equipment | 7,610 | 9,140 | |||||
| Plantation costs | 4,129 | 4,650 | |||||
| Leasehold improvements | 3,783 | 3,911 | |||||
| Construction in progress | 266 | 568 | |||||
| 18,084 | 19,974 | ||||||
| Less accumulated depreciation and amortization | (6,930 | ) | (6,243 | ) | |||
| Property, plant and equipment, net | $ | 11,154 | $ | 13,731 | |||
Note 8. Stock Options
NaPro accounts for its stock options to employees and directors in accordance with Accounting Principles Board Opinion No. 25 (APB 25), Accounting for Stock Issued to Employees, and related interpretations. Pursuant to APB 25, compensation expense is recorded over the vesting period only if the fair value of the underlying stock exceeds the exercise price. Stock options granted to consultants are accounted for under the fair value method, in accordance with Statement of Financial Accounting Standards No. 123 (SFAS 123), Accounting for Stock-Based Compensation.
Pro forma information regarding net income and earnings per share is required by SFAS 123 and Statement of Financial Accounting Standards No. 148, Accounting for Stock-Based Compensation Transition and Disclosure, which requires that the information be determined as if the Company had accounted for employee stock options granted subsequent to December 31, 1994 under the fair value method of that statement. NaPro estimated the fair value for these options at the date of grant using a Black-Scholes option pricing model with the following weighted-average assumptions for the quarters ended July 2, 2003 and June 30, 2002, respectively: risk-free interest rates of 2.51% to 3.22% and 4.60% to 5.07%; no expected dividend; volatility factors of 1.194 and 1.090, and an estimated expected life range of three to six years. For the six months ended July 2, 2003 and June 30, 2002, respectively: NaPro estimated the fair value of these options with the following weighted average assumptions, risk-free interest rates of 2.49% to 3.22% and 4.12% to 5.07%; no expected dividend; volatility factors of 1.187 to 1.194 and 1.090 to 1.167, and an estimated expected life range of three to six years.
D-9
For purposes of pro forma disclosures, the Company amortizes to expense the estimated fair value of the options over the options' vesting period. NaPro's pro forma information is as follows (in thousands, except per share amounts):
| | Quarter Ended | Six Months Ended | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| | July 2, 2003 | June 30, 2002 | July 2, 2003 | June 30, 2002 | |||||||||
| Net (loss) income as reported | $ | (2,130 | ) | $ | 3,055 | $ | (4,880 | ) | $ | (3,608 | ) | ||
| Deduct: Total stock based employee compensation expense determined under fair value based method for all awards | (927 | ) | (1,231 | ) | (1,922 | ) | (2,295 | ) | |||||
| Pro forma net (loss) income | $ | (3,057 | ) | $ | 1,824 | $ | (6,802 | ) | $ | (5,903 | ) | ||
| Basic (loss) income per share—as reported | $ | (0.07 | ) | $ | 0.10 | $ | (0.16 | ) | $ | (0.12 | ) | ||
| Diluted (loss) income per share—as reported | $ | (0.07 | ) | $ | 0.10 | $ | (0.16 | ) | $ | (0.12 | ) | ||
| Pro forma basic (loss) income per share | $ | (0.10 | ) | $ | 0.06 | $ | (0.23 | ) | $ | (0.20 | ) | ||
| Pro forma diluted (loss) income per share | $ | (0.10 | ) | $ | 0.06 | $ | (0.23 | ) | $ | (0.20 | ) | ||
Note 9. Delaware Economic Development Authority Grant
In May 2003, the Company was awarded $130,000 of a grant that could total up to $1.0 million from the Delaware Economic Development Authority under the Delaware Strategic Fund Program. The grant award is based on maintaining a certain number of employees with an average salary above a minimum within the State of Delaware for four years or the Company is subject to defaulting on the grant. A default would require that the Company repay all or a portion of the grant that had been received. This grant award has been recorded as deferred income until the Company is past the default period. The Company does not expect to obtain any of the possible additional amounts under the grant. Application for the remaining awards, if any, must be submitted by May 2008.
Note 10. Restructuring Plan
During the first quarter of 2003, the Company incurred severance costs of $377,000 associated with the termination of the employment of two executive officers. During the second quarter of 2003 the Company incurred severance costs of approximately $26,000 resulting from the involuntary termination of the employment of six additional employees. The Company anticipates further personnel reductions as a result of the termination of a Boulder, Colorado lease (see Note 11) and expected production process changes during the remainder of 2003. However, NaPro is unable to estimate the financial impact of these potential charges at this time.
In June 2003, the Company offered certain of its paclitaxel manufacturing employees a retention bonus consisting of restricted stock grants, totaling 68,720 shares of the Company's common stock, valued at $77,000 plus related taxes of $25,000 to be paid by the Company. Restricted shares will vest if the employee is still employed by NaPro on the earlier of the sale of the paclitaxel business or January 5, 2004.
D-10
In May 2003, the Company negotiated a voluntary termination of the majority of its regulatory affairs personnel and their transfer to an independent, third party consulting company. NaPro contracts its regulatory affairs with that company on an as needed basis.
The sale of the Company's Analytical Services group, as discussed in Note 5, was also part of the Company's restructuring plan.
Note 11. Leases
In March 2003, the Company entered into an agreement to terminate its lease of 54,000 square feet of office, research and development and manufacturing space in Boulder, Colorado in two phases during 2003. In connection with terminating the lease, during the first quarter of 2003, NaPro paid to the lessor an early termination fee totaling $249,000 and anticipates paying estimated additional site remediation costs of between $300,000 and $500,000 during the remainder of 2003. As a result of this lease termination, the Company was relieved of future minimum lease obligation payments, totaling approximately $600,000, and other occupancy costs, totaling approximately $400,000, due during the remainder of 2003 and 2004.
In May 2003, NaPro entered into a new agreement to lease approximately 10,000 square feet of administrative space in Boulder, Colorado beginning June 1, 2003. The Company has the option to terminate all, or a portion of this lease in October 2003. The future minimum payments in connection with this lease total $37,000 from inception through October 2003. If the Company does not terminate this lease during the cancelable period, the lease becomes noncancelable over the remaining term, which goes through January 2008.
In connection with the sale of the Analytical Services group to Chromadex (see Note 5), Chromadex assumed the Company's lease obligation for one of its facilities in Boulder, Colorado. This lease is a different lease than the lease for 54,000 square feet of office, research and development and manufacturing space discussed above in this Note 11. As a result, the Company was relieved of future minimum lease obligation payments totaling approximately $863,000.
Note 12. Commitments and Contingencies
The Company has executed noncancelable operating lease agreements for office, research and production facilities, equipment and for plantations. As of July 2, 2003, future minimum lease payments under noncancelable operating lease agreements are as follows (in thousands):
| Remainder of 2003 | $ | 483 | |
| 2004 | 470 | ||
| 2005 | 266 | ||
| 2006 | 242 | ||
| 2007 | 74 | ||
| Thereafter | 142 | ||
| Total | $ | 1,677 | |
NaPro has renewal clauses in some of these leases that range from one to ten years.
D-11
Item 2. Management's Discussion and Analysis of Financial Condition and Results of Operations
The following discussion and analysis provides information that management believes is relevant to an assessment and understanding of the results of operations of NaPro BioTherapeutics, Inc. You should read this discussion in conjunction with the Financial Statements and Notes included elsewhere in this report. Certain statements set forth below constitute "forward-looking statements" within the meaning of the Private Securities Litigation Reform Act of 1995, referred to as the "Reform Act." See "Special Note Regarding Forward Looking Statements."
General
We are a pharmaceutical company focused in three distinct areas: the production and sale of paclitaxel, an approved cancer drug; the development of proprietary oncology products; and the development of novel genomic technologies, primarily in the area of Gene Editing for applications in human therapeutics, diagnostics, agribiotechnology and pharmacogenomics. Our first commercialized product is paclitaxel, a naturally occurring chemotherapeutic anti-cancer agent found in certain species of yew, orTaxus trees. We also may be engaged in evaluating the in-licensing or purchase of potential new products and/or technologies, whether or not those products or technologies are derived from natural products. Our evaluation of new products and technologies may involve the examination of individual molecules, classes of compounds, or platform technologies, in both cancer and other therapeutic areas. Acquisitions of new products or technologies may involve the purchase of such products or technologies, or the acquisition of, or merger with, other companies.
In April 2003, we announced that we are actively seeking to sell our worldwide polyethoxylated castor oil formulated injectable paclitaxel business. Polyethoxylated castor oil formulations are the only paclitaxel formulations currently approved by the United States Food and Drug Administration (FDA) and other international regulatory agencies. The contemplated sale would transfer all of our assets related to the manufacture and sale of paclitaxel, which comprises a significant portion of our total assets and our current operations. The assets to be sold include our bulk paclitaxel manufacturing facilities and related production equipment; yew tree plantations (which includes related cultivation agreements; leases and yew trees owned by NaPro); our contracts with Abbott Laboratories (Abbott), Mayne Pharma, Tzamal Pharma (Tzamal), and JCR Pharmaceuticals Co., Ltd. (JCR) and other contracts related to the generic cremaphor injectible paclitaxel business; regulatory filings; and patents and intellectual property relating to the manufacture and formulation of paclitaxel. We expect to retain rights for formulations other than polyethoxylated castor oil formulations. We have retained Wells Fargo Securities, LLC to act as our agent in this sale transaction. The timing and terms of sale of the paclitaxel business are uncertain at this time, and we can give no assurance that any transaction will occur. If the sale of our paclitaxel business is successful, we will exit that business and we intend to utilize the proceeds of the sale to fund the development and commercialization of products based on NaPro's proprietary Gene Editing and Oncology platforms and to retire $20.0 million of debt owed to Abbott. If the transaction does not occur, we would expect to remain in the paclitaxel business for the foreseeable future.
We continue to incur substantial research and development expense related to the development of novel, anti-cancer agents and the development of novel genomic technologies, primarily in the area of Gene Editing, for applications in human therapeutics, diagnostics, agribiotechnology and pharmacogenomics, as well as the improvement of our paclitaxel yield, the reduction of our long-term
D-12
cost of product and the development of our semisynthesis process. Accordingly, we have incurred significant losses, including losses of $8.7 million, $25.8 million and $16.6 million for the years ended December 31, 2002, 2001 and 2000, respectively. We incurred a loss of $4.9 million for the six months ended July 2, 2003, resulting in an accumulated deficit of $108.6 million as of July 2, 2003. We anticipate that losses may continue until such time, if ever, as we are able to generate sufficient sales to support our development operations, including the research and development activity mentioned above.
Our ability to generate sufficient sales to support our operations currently depends primarily upon the successful commercialization of our worldwide paclitaxel program or, if our paclitaxel business is sold, upon our successful development of Gene Editing and/or Oncology products. Our strategy for the paclitaxel program has been to form strategic alliances through long-term exclusive agreements with established pharmaceutical companies. In 1999, we entered into an exclusive collaborative agreement of up to 20 years covering the U.S. and Canada with Abbott to develop and commercialize one or more formulations of paclitaxel for the treatment of a variety of cancers. Under our agreement with Abbott, we are responsible for the supply of bulk drug; development activity is conducted jointly. Abbott is responsible for finishing, regulatory filings, marketing, and sale of the finished drug product. Most primary decisions related to the paclitaxel development program are made by a joint Abbott-NaPro Development Committee. In March 2001, we and Abbott filed an Abbreviated New Drug Application (ANDA) with the FDA for paclitaxel. The FDA granted approval of this ANDA on May 8, 2002. We received an approval for a second ANDA on August 1, 2002, which provides for greater manufacturing flexibility for the generic paclitaxel product.
In connection with the Abbott agreement, we could have received total funding of up to $118.0 million in the form of up to $30.0 million in development milestones, up to $57.0 million in marketing milestones, $20.0 million in secured debt, and $11.0 million in equity investments. Marketing milestones are based upon certain annual sales levels, the majority of which are unlikely to be achieved given the current pricing of paclitaxel. Through July 2, 2003, and excluding product sales, we received $40.0 million under the agreement, including $11.0 million in equity investments in exchange for 2,000,000 shares of our common stock, $9.0 million in development milestones, and $20.0 million in secured debt.
Of the $30.0 million of potential development-related milestone payments, we received $1.0 million upon execution of our agreement with Abbott in July 1999. With the approval of our ANDA, and upon commencement of commercial sales, we received an $8.0 million payment in May 2002. Additional development milestones will only be available should NaPro or Abbott undertake and successfully complete new development work, which is not currently anticipated. Therefore, it is highly unlikely that we will receive any further development milestone payments. If we were to sell our paclitaxel operations, it is probable that we would no longer be entitled to any future milestone payments from Abbott.
We have received $20.0 million of secured debt from Abbott. The debt bears a primary interest rate of 6.5% and is due in full on the earlier of: (i) the second anniversary of the first sale of finished product by Abbott to a wholesaler or end-user customer following approval of finished product by the FDA (May 8, 2004); (ii) the termination of the Abbott agreement; or (iii) January 1, 2007. The debt is limited to a borrowing base of collateralized assets, recomputed monthly. The majority of our paclitaxel inventories, and specific assets which relate to the manufacture and related sale of paclitaxel, are collateralized as security for the debt. Abbott's only recourse in the event of our default on the debt is
D-13
to these specific assets. We anticipate that we will use a portion of the proceeds from the sale of the paclitaxel business to retire the debt to Abbott.
Contingent upon achieving certain commercial sales thresholds for all approved formulations of paclitaxel over several years, we could have received marketing milestone payments from Abbott of up to $57.0 million. Given the current pricing of paclitaxel, we believe that the majority, if not all, of these annual sales thresholds are unlikely to be achieved.
Under terms of the agreement, Abbott purchases bulk drug from us. Abbott may terminate the agreement at any time with or without cause. Should Abbott terminate without cause, it is obligated to make certain payments to us and to give us twelve months prior written notice. Since the approval of generic paclitaxel in mid-2001, the selling price of paclitaxel has fallen significantly, and may continue to do so. We receive a fixed price payment of $500 per gram, upon delivery of paclitaxel to Abbott, plus a quarterly royalty payment on sales, if any. The royalty payment is a percentage of sales recorded by Abbott less any product payment already paid to us. The percentage of sales varies each year based upon total sales in that year recorded by Abbott. We do not anticipate receiving significant royalty payments given the current price of the drug.
In 1992, we entered into a 20-year exclusive agreement with F.H. Faulding & Co., Ltd. (Faulding), a large Australian pharmaceutical company, for the clinical development, finishing, sale, marketing and distribution of paclitaxel. In October 2001, Faulding was acquired by Mayne-Nickless Limited, an Australian based health care provider and logistics operator. As a result, Faulding is now known as Mayne Pharma. Mayne Pharma actively markets anti-cancer pharmaceuticals and other health care products in Europe, Australia, Asia, South America, and other regions throughout the world. In 2000, we amended the Mayne Pharma agreement to, among other things, add additional countries to Mayne Pharma's exclusive territory. In 2001, we entered into a separate agreement with Mayne Pharma covering development and sale of paclitaxel in Europe. We cannot assure that we will receive regulatory approval in any of the major markets in Europe. Should we receive approval, Mayne Pharma will then market and sell our paclitaxel in Europe. We and Mayne Pharma will share equally the net sales of the product in Europe. With the addition of the new agreement for Europe, the Mayne Pharma territory includes substantially all of the world other than the U.S., Canada, Japan, Israel, the former Soviet Union and parts of Africa. Mayne Pharma has received marketing approval for, and is selling paclitaxel as ANZATAX™ in more than 25 countries.
In July 2002, the European Patent Office, Opposition Division, ruled that NaPro's currently issued paclitaxel formulation patent in the European Patent Convention is invalid. We have appealed this ruling and expect the appeal to be heard within the next 24 to 36 months. We do not believe this decision will have any effect outside of the jurisdiction of the European Patent Office; nor will it affect our ability to enter the market in Europe as regulatory approvals are received. We also do not believe this patent decision will affect our revenues or earnings through calendar year 2003.
We have established an exclusive supply and distribution agreement with Tzamal for the development and distribution of paclitaxel in Israel. Tzamal is an Israeli pharmaceutical company, active and well established in the Israeli pharmaceutical market and specializing, among other areas, in ethical products for the treatment of cancer, metabolic diseases, neurological disorders and gynecology/fertility. In January 2001, Tzamal received approval in Israel to sell paclitaxel under the trade name Biotax™. The Israeli Ministry of Health has approved Biotax for use in treating a variety of cancers.
D-14
In June 2001, we and JCR entered into a mutually exclusive development, supply and distribution agreement for paclitaxel in Japan. JCR, headquartered in Ashiya, Japan, is a research and development oriented pharmaceutical manufacturer, focusing on the research of bioactive substances such as enzymes, hormones, enzyme inhibitors, growth factors and stimulating factors. Under the agreement, we will be responsible for manufacturing and supplying the finished drug. We and JCR will jointly be responsible for the clinical and regulatory program necessary for seeking approval to market paclitaxel in Japan. JCR will be responsible for the sales and distribution of the product in Japan upon its approval. We cannot assure, however, that we will receive regulatory approval of paclitaxel in Japan, or that we and our strategic partner will successfully market it.
Research and Development
Research and development are major activities for us. We discussed the nature and status of our research and development in depth in our Annual Report on Form 10-K for the year ended December 31, 2002, and in the General section of this Management's Discussion and Analysis. If the paclitaxel business is sold, we would expect to fund our near-term research and development efforts with a portion of the proceeds from the sale.
We have incurred the following expense on research and development projects, including process improvements in our paclitaxel business and legal expenses incurred in connection with patent applications and regulatory filings (in thousands):
| | Quarter Ended | Six Months Ended | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| | July 2, 2003 | June 30, 2002 | July 2, 2003 | June 30, 2002 | ||||||||
| Pharmaceutical | $ | 1,437 | $ | 2,532 | $ | 3,252 | $ | 5,770 | ||||
| Genomics | 1,058 | 1,503 | 2,238 | 2,558 | ||||||||
| $ | 2,495 | $ | 4,035 | $ | 5,490 | $ | 8,328 | |||||
Research and development is expected to remain a significant expense of our business. Our research and development activity is subject to change as we develop a better understanding of our projects and their prospects. We currently expect to concentrate on the development of novel anti-cancer agents, and the development of novel genomic technologies, primarily in the area of Gene Editing, for applications in human therapeutics, diagnostics, agribiotechnology and pharmacogenomics. We anticipate bringing our Targeted Oncology program to the clinic during 2004. Our Sickle Cell Disease and Huntington's Disease programs are expected to go into the clinic during 2004 or the beginning of 2005. However, there can be no assurance that we will be able to achieve the timing of these programs. We also cannot estimate the cost of the effort necessary to complete the programs or the timing of commencement of material net cash inflows from these programs. Continued development of these programs is dependent upon additional capital. We cannot assure that we will be able to obtain such capital on terms, which are acceptable to us. We have included a number of the risks and uncertainties associated with completing development on schedule in the Special Note Regarding Forward-looking Statements, below.
D-15
Quarter Ended July 2, 2003 Compared to the Quarter Ended June 30, 2002
Product Sales. Product sales for the quarter ended July 2, 2003 were $7.6 million, a decrease of approximately $1.9 million from the prior year period. The decrease during this quarter was primarily attributable to lower shipments to Abbott partially offset by an increase in sales to Mayne Pharma. Sales to Abbott for the quarter were $2.6 million, a decrease of $4.7 million from the same period in 2002. The larger quantities in 2002 corresponded with the launch of sales by Abbott following the May 2002 approval by the FDA. Sales to Mayne Pharma for the quarter were $4.9 million, an increase of $2.8 million from the prior year quarter. This increase was primarily due to shipping initial launch quantities for Europe at a higher per unit sales price ($1.4 million) and a net increase in our share of gross proceeds from sales price reconciliations ($1.1 million). Future sales are difficult to predict and are expected to vary from year to year depending upon numerous demand and competitive factors. Among these factors are: the relationship of competitors with their customers, the relationship of Abbott, Mayne Pharma and Tzamal with their respective customers and the amount of competitive product in the distribution channel.
Cost of Sales. Cost of sales for the current year quarter was $4.1 million, a decrease of $2.9 million from the prior year quarter. The decrease was attributable to lower quantities sold ($3.3 million) and lower per unit cost ($1.3 million). Lower per unit cost resulted primarily from our biomass strategic plan, process improvements and cost containment efforts. This was partially offset by a biomass inventory valuation writedown of $1.0 million. As a result of our lower cost of sales, our gross margin increased to 46% in the second quarter of 2003 compared with 26% in the prior year quarter. Gross margins may fluctuate in future periods depending on a number of factors, including biomass taxane content, production volumes and developments in our manufacturing process.
Research and Development Expense. Research and development expense for the current year quarter was $2.5 million, a decrease of $1.5 million from the prior year quarter. Genomics research and development decreased $400,000 from the 2002 quarter, while pharmaceutical research and development decreased $1.1 million from the prior year quarter. The decrease in 2003 was primarily attributable to reductions in our salaries and benefits ($900,000), consulting and contract services ($400,000) and supplies ($200,000). This overall decrease is primarily a result of our restructuring plan designed to control costs. In the 2002 quarter, a significant portion of our contract services expenditures were related to our semi-synthesis paclitaxel program and in-licensing of technology. Salaries and benefits declined in the 2003 quarter primarily due to the sale of our Analytical Services group to Chromadex and reduction of personnel in our oncology division.
General and Administrative Expense. General and administrative expense for the current year quarter was $2.8 million, a decrease of approximately $100,000 from the prior year quarter. The decrease was primarily due to lower legal expenses ($500,000) and outside services ($200,000). These decreases were partially offset by higher depreciation expense resulting from a shortened useful life of leasehold improvements at the Boulder, Colorado facility in which we terminated our lease early ($300,000), and the early termination fee paid on the lease ($200,000).
License Fee Income. License fee income for the current year quarter was $280,000, a decrease of $7.8 million from the prior year quarter. The decrease was attributable to a one-time milestone payment of $8.0 million received in 2002 from Abbott upon commencement of commercial sales in the United States.
D-16
Six Months Ended July 2, 2003 Compared to the Six Months Ended June 30, 2002
Product Sales. Product sales for the 2003 period were $14.4 million, a decrease of approximately $1.8 million from the 2002 period. The decrease during this period was primarily attributable to lower shipments to Abbott and Tzamal partially offset by an increase in shipments to Mayne Pharma. Sales to Abbott for the period were $6.7 million, a decrease of $4.6 million from the same period in 2002. The 2002 sales included significant quantities sold in anticipation of launch following the May 2002 approval by the FDA. There were no sales to Tzamal for the current year period, which resulted in a decrease of $500,000 from the prior year period. Sales to Mayne Pharma for the period were $7.6 million, an increase of $3.3 million from the prior year period. A large portion of the increase in sales to Mayne Pharma resulted from shipping initial launch quantities for Europe at a higher per unit sales price ($1.4 million) and a net increase in NaPro's share of gross proceeds from sales price reconciliations ($1.1 million). Future sales are difficult to predict and are expected to vary from year to year depending upon numerous demand and competitive factors. Among these factors are: the relationship of competitors with their customers, the relationship of Abbott, Mayne Pharma and Tzamal with their respective customers and the amount of competitive product in the distribution channel.
Cost of Sales. Cost of sales for the 2003 period was $7.4 million, a decrease of $6.7 million from the 2002 period. The decrease was attributable to lower quantities sold ($3.0 million) and lower per unit cost ($3.2 million). Lower per unit cost resulted primarily from increased production yields, implementation of our biomass strategic plan and results of our cost containment efforts. The 2002 cost per unit was impacted by costs associated with the start-up of larger scale production. As a result, the gross margin increased to 49% for the 2003 period compared with 13% in the prior year period. Gross margins may fluctuate in future periods depending on a number of factors, including biomass taxane content, production volumes and developments in our manufacturing process.
Research and Development Expense. Research and development expense for the current year period was $5.5 million, a decrease of $2.8 million from the prior year period. Genomics research and development decreased $300,000 from the 2002 period, while pharmaceutical research and development decreased $2.5 million from the prior year period. The decrease was primarily attributable to reductions in our consulting and contract services ($1.2 million), salaries and fringe benefits ($700,000) and supplies ($600,000). This overall decrease is primarily a result of our restructuring plan designed to control costs. In the prior year period, a significant portion of our research and development expenditures were related to our semi-synthesis paclitaxel program and in-licensing of technology. Salaries and benefits declined in the 2003 quarter due to the sale of our Analytical Services group to Chromadex and personnel reductions in our oncology division, partially offset by severance costs associated with an executive officer.
General and Administrative Expense. General and administrative expense for the current year period was $5.8 million, an increase of approximately $900,000 from the prior year period. The increase was primarily due to higher depreciation resulting from a shortened useful life of leasehold improvements at the Boulder, Colorado facility in which we terminated our lease early ($400,000), increased staffing at our genomics division ($300,000) and lease termination costs associated with our corporate facilities in Boulder, Colorado ($200,000). These increases were partially offset by lower legal costs ($300,000) and lower supplies expense ($300,000).
D-17
License Fee Income. License fee income for the current year period was $600,000, a decrease of $7.8 million from the prior year period. The decrease was primarily attributable to a to a one-time milestone payment of $8.0 million received in 2002 from Abbott upon commencement of commercial sales in the United States.
Interest Expense. Interest expense for the 2003 period was $1.2 million, an increase of $200,000 from the 2002 period. The increase was primarily attributable to the convertible debenture interest and amortization of the discount attributable to the conversion feature of the $8.0 million in convertible debt issued in February 2002 and higher amortization of debt issuance costs.
Liquidity and Capital Resources
Our capital requirements have been, and will continue to be, significant. As of July 2, 2003, we had a working capital deficit of $7.0 million compared to a working capital balance of $18.8 million as of December 31, 2002. We anticipate that we will be able to provide adequate capital to fund our operations and capital expenditures for at least the next twelve months. This would be accomplished through the sale of the paclitaxel business, renegotiation of debt, or raising additional capital, as well as projected sales, the reduction of current inventory and collection of accounts receivable. However, we cannot assure that we will be able to sell the paclitaxel business for a price acceptable to us or that we will be able to renegotiate the payment of the Abbott debt under terms acceptable to us. We also cannot assure that if we did seek additional capital we will be able to obtain such capital on terms that are acceptable to us.
To date, we have funded our capital requirements primarily with the net proceeds of public offerings of common stock of approximately $21.1 million, with private placements of equity securities of approximately $62.6 million, with the exercise of warrants and options of $8.0 million and with net borrowing of $28.3 million.
In February 2002, we sold privately $8.0 million of common stock issued at $9 per share and $8.0 million principal of five-year 4% debentures convertible into common stock at $15 per share to TL Ventures V L.P. and TL Ventures V Interfund L.P. No placement agent was involved in the transaction. The net proceeds were $15.6 million. As part of this transaction, we recorded a discount attributable to the conversion feature of the convertible debentures in the amount of $3.1 million, which is being amortized over the term of the debentures. During the first quarter of 2002, we filed a registration statement with the Securities and Exchange Commission to register under the Securities Act of 1933 the resale of common stock that was issuable upon conversion of the debentures and issuable in lieu of cash interest on the debentures. In July 2002, the registration statement was declared effective by the SEC. NaPro may pay the debenture interest in cash or common stock at its option. The debentures automatically convert into common stock during the first two years if the average price of the common stock for 15 trading days exceeds $22, and during the last three years if the average price for 30 trading days exceeds $16. If we have insufficient funds to repay the debentures in full at maturity or upon acceleration, the holders of the debentures may elect to convert the outstanding principal balance and any unpaid accrued interest on the debentures into shares of common stock at a conversion price equal to the then current 20 day average trading price of the common stock. The conversion price of the common stock in such a circumstance could be materially below the price at which we otherwise would be willing to issue our common stock. Issuance of common stock at such a price could result in
D-18
substantial dilution to our other stockholders and could have a material adverse effect on the market price of our common stock.
In February 2002, we sold privately $8.0 million of common stock and $8.0 million principal of five-year 4% debentures to TL Ventures V L.P. and TL Ventures V Interfund L.P. A representative of TL Ventures has recently asserted that TL Ventures will have the right to cause us to redeem its debentures if we complete the sale of our paclitaxel business. We do not agree with TL Venture's assertion and we are discussing with TL Ventures a resolution of this matter. However, if we are required to redeem these debentures with the proceeds of the sale of our paclitaxel business, the amount of the proceeds would be reduced accordingly and our ability to apply the proceeds to the benefit of our oncology and gene editing technologies would be reduced.
In December 2002, we acquired the genomics business of Pangene Corporation for $1.3 million in cash and assumption of debt obligations totaling approximately $65,000. The acquisition consisted primarily of patents and intellectual property relating to Pangene's homologous recombination technology and gene isolation and service business, physical assets, instrumentation, software, customer relationships and third-party licenses. The purchase price was allocated to intangible assets totaling $987,000, fixed assets and software of $183,000, and other current assets of $44,000. In process research and development of $151,000 was expensed at the time of the acquisition. We are amortizing the intangible assets over 5 to 15 years, and fixed assets and software over 3 years. In connection with this agreement, we acquired additional patents in January 2003 in the amount of $400,000.
In November 2000, we entered into a 20-year Gene Editing technology license with the University of Delaware and Thomas Jefferson University relating to the use of proprietary oligonucleotides (small, synthetic DNA polymers) designed to precisely alter genes in humans, animals, plants, viruses and microbes. The license provides for research and patent funding commitments and payments in common stock. To date, we have issued 300,000 shares under the license agreement. Assuming we do not cancel the license, we will issue an additional 900,000 shares in 100,000 share-per-year increments on the license anniversaries. We may, at our option, accelerate the issuance dates. Certain milestones may also accelerate the issuance dates. The license is terminable at NaPro's option. If terminated, no further shares would be issued. We have committed to fund at least $300,000 in research under this agreement during 2003.
Working Capital and Cash Flow
Cash and cash equivalents decreased $900,000 to $5.8 million for the period ended July 2, 2003 from $6.8 million as of December 31, 2002. During the 2003 period, net cash provided by operating activities was $500,000, net cash used in investing activities was $1.7 million and financing activities provided net cash of $200,000.
Inventory was $13.0 million at July 2, 2003, of which $6.1 million was classified as non-current. Non-current inventory consists of raw materials which are not expected to be utilized during the next twelve months, due to the timing of biomass harvests and anticipated future changes to our production process. The amount of inventory is dependent on a number of factors, including the shipping requirements of our strategic partners, our production planning for meeting those needs, and the timing of seasonal biomass harvests. Inventory balances may vary significantly during product development, launch periods and during seasonal harvests.
D-19
Accounts receivable was $8.1 million at July 2, 2003 and $9.3 million at December 31, 2002. We anticipate that the level of our accounts receivable will continue to decrease throughout the remainder of 2003. If we sell the paclitaxel business, we expect that our accounts receivable will be significantly reduced from our current levels for the foreseeable future.
Notes payable—current portion was $20.2 million at July 2, 2003, an increase of $20.1 million from December 31, 2002. We reclassified our note payable due to Abbott of $20.0 million, and related unamortized issuance costs to current as the note is due on May 8, 2004.
Capital Expenditures
We spent $700,000 on capital projects during the first six months of 2003. These capital project expenditures primarily consisted of manufacturing and laboratory equipment purchases, and plantation costs. We spent $600,000 for our investment in Chromadex and $400,000 for patents acquired in connection with the prior year acquisition of assets from Pangene Corporation.
The amount and timing of future capital expenditures will depend upon numerous factors, including:
- •
- the manufacturing cost of paclitaxel;
- •
- the development of new products and technologies;
- •
- the acquisition of new products and technologies;
- •
- the cost of manufacturing resources for new products and technologies;
- •
- the nature of our relationship with our strategic partners;
- •
- the progress of our research and development programs;
- •
- changes in manufacturing processes;
- •
- the magnitude and scope of these activities;
- •
- competing technological and marketing developments;
- •
- changes in or terminations of existing strategic relationships;
- •
- changes in facilities; and
- •
- changes in staffing levels.
We anticipate capital expenditures of approximately $1.7 million during 2003, which will be significantly reduced from the prior year. The primary focus of capital spending during the remainder of 2003 is expected to be in our manufacturing areas. Although we may seek additional long-term financing to fund capital expenditures, we cannot assure that we can obtain such financing on terms which are acceptable to us.
Net Operating Loss Carryforwards
As of December 31, 2002, we had approximately $87.0 million of net operating loss carryforwards to offset future taxable income. Tax law provides limits on the utilization of net operating loss
D-20
carryforwards if there has been a "change of ownership" as described in Section 382 of the Internal Revenue Code. Such a change of ownership may limit our utilization of our net operating loss carryforwards, and could be triggered by sales of securities by us or our stockholders. Any gain on a sale of the paclitaxel business would use a portion of these carryforwards.
Business Development Activities
In the normal course of our business, we investigate, evaluate, and discuss licensing relationships, acquisitions, and other business combination opportunities in the pharmaceutical and genomics businesses. In the event we enter into any such relationships or transactions, we may consider using available cash, issuing equity securities or increasing our debt. Such transactions could materially affect our capital structure.
Critical Accounting Policies
The Company's critical accounting policies are presented in its Annual Report on Form 10-K for the year ended December 31, 2002. There were no changes to these critical accounting policies during the six month period ended July 2, 2003.
Special Note Regarding Forward-looking Statements
The statements in this report that are not historical facts are forward-looking statements that represent management's beliefs and assumptions based on currently available information. Forward-looking statements can be identified by the use of words such as "believes", "intends", "estimates", "may", "will", "should", "anticipated", "expected" or comparable terminology or by discussions of strategy. Such forward-looking statements may include, among others:
- •
- statements concerning our plans, objectives and future economic prospects, such as matters relative to developing new products;
- •
- the timing and amount of sales of paclitaxel to our strategic partners;
- •
- the availability of patent and other protection for our intellectual property;
- •
- the completion of preclinical studies, clinical trials and regulatory filings;
- •
- the prospects for, and timing of, regulatory approvals;
- •
- the need and plans for, and availability of, additional capital;
- •
- the amount and timing of capital expenditures;
- •
- the timing of product introductions and sales;
- •
- the availability of raw materials;
- •
- the sale of the paclitaxel business;
- •
- the ability to outsource activities;
- •
- the future acquisition of products or business entities;
- •
- prospects for future operations; and
D-21
- •
- other statements of expectations, beliefs, future plans and strategies, anticipated events or trends and similar expressions concerning matters that are not historical facts.
Although we believe that the expectations reflected in such forward-looking statements are reasonable, we cannot assure you that these expectations will prove to be correct. Such forward-looking statements involve known and unknown risks, uncertainties and other factors, which may cause our actual results, performance or achievements, or industry results, to be materially different from any future results, performance or achievements expressed or implied by such forward-looking statements. Such factors include, among other things, the following:
- •
- our history of net losses;
- •
- our ability to obtain capital necessary to fund our operations when needed;
- •
- our ability to adequately protect our proprietary technologies;
- •
- litigation or third-party claims of intellectual property infringement;
- •
- success of any product liability lawsuits brought against us;
- •
- acceptance of our products in the marketplace;
- •
- reduction in revenues resulting from marketing practices such as returns, allowances and charge-backs and marketing programs adopted by wholesalers;
- •
- our ability to effectively price our products or obtain adequate reimbursement for sales of our products;
- •
- intense competition from our paciltaxel competitors;
- •
- our ability to retain our Chairman and Chief Executive Officer, Leonard P. Shaykin and other key executives;
- •
- our ability to attract and retain the qualified employees we need to be successful;
- •
- our reliance on independent, third-parties to perform certain services for us;
- •
- our ability to renew or extend the leases on our leased facilities;
- •
- any dilution resulting from future sales of common stock;
- •
- the volatility in our stock price;
- •
- our ability to continue to meet all applicable Nasdaq SmallCap requirements;
- •
- certain anti-takeover provisions we have implemented;
- •
- our ability to sell our paclitaxel business for an acceptable price;
- •
- any assertion by TL Ventures of a right to have its debentures redeemed;
- •
- political, economic or regulatory events in foreign countries that may limit our ability to generate revenue;
- •
- our dependence on strategic partners for the development and marketing of paclitaxel;
D-22
- •
- our ability to obtain regulatory approval for the sale of paclitaxel in the primary world markets outside the United States;
- •
- regulatory approvals for the sale of paclitaxel in the United States or any other primary world market in which our paclitaxel may be approved for commercial sale;
- •
- access to a stable, long term supply of yew trees, yew biomass and crude paclitaxel extract for use in the production of paclitaxel;
- •
- our ability to produce paclitaxel in sufficient quantities, on a timely basis and at an acceptable cost;
- •
- our ability to respond to recent declines in the price of paclitaxel resulting from increased competition;
- •
- our ability to market our paclitaxel successfully against our competitors' paclitaxel;
- •
- our ability to compete against new cost effective methods of harvesting or manufacturing paclitaxel or other technological developments;
- •
- adequate protection of our patents and trade secrets related to our paclitaxel;
- •
- our ability to replace the revenues lost if we sell our paclitaxel business;
- •
- our ability to commercialize our product candidates and technologies;
- •
- our reliance on strategic partners for the development, marketing and manufacturing of future products and technologies;
- •
- our efforts in developing our gene editing technology;
- •
- our ability to research and develop new potential products and technologies without diverting available capital or management's attention from other aspects of our business;
- •
- our ability to respond to technological changes that may make our potential products and technologies less attractive or obsolete;
- •
- adverse events in the field of gene alteration that could negatively impact regulatory approval or public perception of our potential products and technologies;
- •
- our reliance on the voluntary participation of patients in our clinical trials to test our potential products and technologies;
- •
- delays related to clinical testing of our potential products and technologies;
- •
- the limited clinical data relating to our gene editing and oncology technologies;
- •
- our ability to obtain foreign regulatory approvals;
- •
- our lack of experience in manufacturing our potential products;
- •
- our lack of experience in marketing or selling our potential products and technologies;
- •
- our ability to obtain rights to proprietary technologies to further develop our business; and
D-23
- •
- our exposure to material environmental liabilities related to our use of hazardous materials in connection with the operation of our research and manufacturing facilities.
Should one or more of the risks mentioned above materialize (or the consequences of such a development worsen), or should the assumptions underlying our forward-looking statements prove incorrect, actual results could differ materially from those forecasted or expected. These factors are not intended to be an all-inclusive enumeration of the business risks we face. Reference is also made to the risk factors discussed in Amendment No. 4 to our Registration Statement (No. 333-82810) filed with the Securities and Exchange Commission on July 1, 2002. The forward-looking statements included in this report represent our view as of the date of this report. The reader should not assume that the statements made herein remain accurate at any future date. We do not intend to update these statements whether as a result of new information, future events or otherwise and undertake no duty to any person to make any update under any circumstance.
Item 3. Quantitative and Qualitative Disclosures about Market Risk.
Under the Mayne Pharma agreement, Mayne Pharma pays us a fixed percentage of their sales price for paclitaxel. Each year, Mayne Pharma estimates the sales price it will receive in the upcoming year, and based upon that estimate, we determine the price we will charge Mayne Pharma. We recognize the corresponding sales at the time of shipment to Mayne Pharma. However, Mayne Pharma's actual selling price may differ from that estimated. Pursuant to the agreement, Mayne Pharma is obligated to provide us with their calculation of the actual quantity of sales and the sales price per gram as of each March 30 and an adjustment is calculated that may increase or decrease our sales of products to Mayne Pharma during that year.
Mayne Pharma's sales are made in the currencies of each of the countries in which it sells paclitaxel. As a result, our sales are affected by fluctuations in the value of these various foreign currencies relative to the U.S. dollar. In the past, currency fluctuations were a significant factor in reductions in the price we charged Mayne Pharma. If changes in foreign currency markets continue to cause a decrease in the price per gram we receive from Mayne Pharma, there could be a material adverse effect on our earnings and cash flow. We currently record a reserve for potential Mayne Pharma pricing adjustments based upon historical experience and periodic reporting provided by Mayne Pharma. Our sales to JCR, if and when they occur, will be subject to the same risk.
Certain statements set forth in this Item 3 may constitute "forward-looking statements." See "Special Note Regarding Forward-looking Statements."
Item 4. Controls and Procedures
(a) The Company maintains a system of disclosure controls and procedures. The term "disclosure controls and procedures," as defined by regulations of the SEC, means controls and other procedures that are designed to ensure that information required to be disclosed in the reports that the Company files or submits to the SEC under the Securities Exchange Act of 1934, as amended (the Act), is recorded, processed, summarized and reported, within the time periods specified in the SEC's rules and forms. Disclosure controls and procedures include, without limitation, controls and procedures designed to ensure that information required to be disclosed by the Company in the reports that it files or submits to the SEC under the Act is accumulated and communicated to the Company's management,
D-24
including its principal executive officer and its principal financial officer, as appropriate to allow timely decisions to be made regarding required disclosure. Within the 90 days prior to the date of this report, the Company carried out an evaluation, under the supervision and with the participation of the Company's management, including the Company's Chief Executive Officer and Chief Financial Officer, of the effectiveness of the design and operation of the Company's disclosure controls and procedures. Based upon that evaluation, the Company's Chief Executive Officer and Chief Financial Officer concluded that the Company's disclosure controls and procedures are effective in timely alerting them to material information relating to the Company required to be included in the Company's periodic SEC filings.
(b) The Company also maintains a system of internal controls. The term "internal controls," as defined by the American Institute of Certified Public Accountants' Codification of Statement on Auditing Standards, AU Section 319, means controls and other procedures designed to provide reasonable assurance regarding the achievement of objectives in the reliability of the Company's financial reporting, the effectiveness and efficiency of the Company's operations and the Company's compliance with applicable laws and regulations. There were no significant changes in our internal controls or to our knowledge, in other factors that could significantly affect our internal controls and procedures subsequent to the date of such evaluation.
It should be noted that any system of controls, however well designed and operated, can provide only reasonable, and not absolute, assurance that the objectives of the system are met. In addition, the design of any control system is based in part upon certain assumptions about the likelihood of future events. Because of these and other inherent limitations of control systems, there can be no assurance that any design will succeed in achieving its stated goals under all potential future conditions, regardless of how remote.
D-25
On April 28, 2003, NaPro filed a declaratory judgment action in the Federal District Court in Denver, and subsequently initiated legal action against Hande Tech Development Co. USA, Inc. (Hande Tech). Hande Tech is a supplier of bulk paclitaxel, who formerly supplied bulk paclitaxel feedstock to NaPro. This declaratory judgment filing was in response to allegations made by Hande Tech that NaPro had breached certain obligations to purchase paclitaxel feedstock from Hande Tech. Hande Tech has asserted that it was damaged by NaPro's failure to purchase paclitaxel feedstock valued at approximately $2.0 million. NaPro's position is that it had no obligation to purchase such feedstock and, therefore, owes Hande Tech nothing. This litigation is in the early stages, and no schedule has yet been set by the court.
Item 4. Submission of Matters to a Vote of Security Holders.
At NaPro's annual meeting of stockholders on June 27, 2003, the four matters listed below were submitted to a vote of NaPro's stockholders.
1. The election of three Class I directors to hold office until the 2006 Annual Meeting
| | Number of Shares | |||
|---|---|---|---|---|
| Nominee | ||||
| For | Withheld | |||
| Arthur H. Hayes, Jr. M.D. | 24,879,222 | 2,058,678 | ||
| Robert E. Pollack, Ph.D. | 25,191,528 | 1,746,372 | ||
| Leonard P. Shaykin | 24,214,061 | 2,723,839 | ||
2. Approval of amendment to the Company's Amended and Restated Certificate of Incorporation to effect a reverse stock split of the Company's outstanding common stock in the range of 1:5 to 1:40, as determined in the discretion of the Board of Directors.
| | Number of Shares | |
|---|---|---|
| For | 23,325,973 | |
| Against | 3,561,621 | |
| Abstain | 50,305 |
3. To ratify the selection by the Board of Directors of Ernst and Young LLP as our independent auditors for the year ending December 31, 2003:
| | Number of Shares | |
|---|---|---|
| For | 25,845,245 | |
| Against | 998,328 | |
| Abstain | 94,326 |
D-26
4. Shareholder proposal to urge the Board of Directors to significantly reduce executive compensation by eliminating bonuses, grants of new options to the Chief Executive Officer and the other four most highly compensated executive officers. In addition, to urge the Board of Directors to reduce salaries by 30% for the Chief Executive Officer and the other four most highly compensated executive officers.
| | Number of Shares | |
|---|---|---|
| For | 3,113,449 | |
| Against | 8,299,175 | |
| Abstain | 312,019 | |
| Unvoted | 15,213,257 |
Item 6. Exhibits and Reports on Form 8-K.
- (a)
- Exhibits:
| Exhibit Number | Description of Exhibit | |
|---|---|---|
| 10.1*+ | Employment Agreement effective May 22, 2003 between the Company and James McChesney. | |
31.1* | Certification of Chief Executive Officer pursuant to Section 302(a) of the Sarbanes-Oxley Act of 2002. | |
31.2* | Certification of Chief Financial Officer pursuant to Section 302(a) of the Sarbanes-Oxley Act of 2002. | |
32.1*# | Certification of Chief Executive Officer pursuant to Section 906 of the Sarbanes-Oxley Act of 2002 (18 U.S.C. Section 1350). | |
32.2*# | Certification of Chief Financial Officer pursuant to Section 906 of the Sarbanes-Oxley Act of 2002 (18 U.S.C. Section 1350). |
*Filed herewith.
# This certification shall not be deemed "filed" for purposes of Section 18 of the Exchange Act, or otherwise subject to liability pursuant to that section. Such certification shall not be deemed to be incorporated by reference into any filing under the Securities Act or the Exchange Act, except to the extent that the Company specifically incorporates it by reference.
+ A management compensation plan.
- (b)
- Reports on Form 8-K:
We filed a Current Report on Form 8-K dated April 9, 2003 reporting on Item 5, and attaching as an exhibit to the report a press release relating to the offering for sale of the Company's worldwide generic cremaphor injectable paclitaxel business.
We filed a Current Report on Form 8-K dated May 15, 2003 reporting on Item 9, Regulation FD Dislosure, and attaching as an exhibit to the report a press release announcing the Company's first quarter 2003 earnings.
D-27
Pursuant to the requirements of the Securities Exchange Act of 1934, NaPro has duly caused this report on Form 10-Q to be signed on its behalf by the undersigned thereunto duly authorized.
| NAPRO BIOTHERAPEUTICS, INC. | |||
August 8, 2003 | By: | /s/ LEONARD P. SHAYKIN | |
| Leonard P. Shaykin Chairman of the Board of Directors, Chief Executive Officer | |||
August 8, 2003 | By: | /s/ GORDON LINK | |
| Gordon Link Senior Vice President, Chief Financial Officer (Principal Financial Officer) | |||
August 8, 2003 | By: | /s/ ROGER NEWBERRY | |
| Roger Newberry Acting Controller (Principal Accounting Officer) | |||
D-28
VOTING AGREEMENT made this day of August, 2003 (the "Agreement"), between Leonard P. Shaykin (the "Stockholder"), in his capacity as a stockholder of NaPro Bio Therapeutics, Inc., a Delaware corporation (the "Company"), and Faulding Pharmaceutical Co., a Delaware corporation ("Buyer").
Concurrently with the execution of this Agreement, Buyer and the Company have entered into an Asset Purchase Agreement dated as of August 25, 2003 (the "Purchase Agreement") pursuant to which Buyer will acquire the Paclitaxel Business of the Company (the "Asset Purchase"). The transactions contemplated by the Purchase Agreement are collectively referred to as the "Transactions." Capitalized terms not defined in this Agreement have the meanings given them in the Purchase Agreement.
In order to induce Buyer to enter into the Purchase Agreement with the Company, Buyer has requested, and the Stockholder has agreed, that the Stockholder enter into this Agreement;
NOW, THEREFORE, in consideration of the foregoing and for other good and valuable consideration, the receipt and sufficiency of which is hereby acknowledged, the parties agree as follows:
The Stockholder hereby agrees with Buyer as follows:
SECTION 1.1. Voting of Shares. (a) At any meeting of the stockholders of the Company, however called, at every adjournment of any such meeting, and in connection with any written consent of the stockholders of the Company, the Stockholder will cause all of his Shares to be voted, during the term of this Agreement, in favor of (i) a resolution approving the transactions contemplated by the Purchase Agreement in accordance with the Delaware General Corporation Law, and (ii) all other Transactions as to which stockholders of the Company are called upon to vote.
For purposes of this Agreement, (i) "Person" shall mean an individual, partnership, limited liability company, corporation, joint stock company, trust, estate, joint venture, association or unincorporated organization, or any other entity or organization, including a government or political subdivision or any agency or instrumentality thereof, and (ii) "Shares" shall mean any and all shares of capital stock of the Company which are entitled to vote in any election of the board of directors of the Company or on any matter submitted to the stockholders of the Company now owned and/or subsequently acquired by the Stockholder through purchase, gift, stock splits, stock dividends and the exercise of stock options or other derivative security.
(b) The Stockholder agrees that during the term of this Agreement, the Stockholder shall attend or other wise participate in all duly called stockholder meetings and any adjournments of such meetings and in all actions by written consent of stockholders in which the Asset Purchase of any Transaction is being considered.
(c) The parties hereto agree and acknowledge that nothing in this Article I or any other part of this Agreement shall be construed as requiring the Stockholder to propose, endorse, approve or recommend the Purchase Agreement or the transactions contemplated thereby in the Stockholder's capacity as a directory or officer of the Company in any manner inconsistent with his fiduciary duties as director or officer.
E-1
SECTION 1.2. No Proxies or Encumbrances. The Stockholder shall not (i) grant any proxies or enter into any voting trust or other agreement or arrangement with respect to the voting of any of the Shares in a manner which would be inconsistent with the provisions of this Agreement, (ii) sell, assign, transfer, encumber or otherwise dispose of or enter into any contract, option or other arrangement or understanding with respect to, the direct or indirect sale, assignment, transfer, encumbrance or other disposition of any of his Shares or any interest therein except for Permitted Transfers to Permitted Transferees (as such terms are defined below) or (iii) seek or solicit any of the foregoing. For purposes of this Agreement, (i) "Permitted Transferee" means any Person controlled, directly or indirectly, by Stockholder, Stockholder's spouse and children, and any trust for the benefit of Stockholder, Stockholder's spouse of children, and (ii) each transfer to a Permitted Transferee shall constitute a "Permitted Transfer" only if it is a:
(i) transfer to a Permitted Transferee and, in the case of a Permitted Transferee, transfer to the Stockholder or to other Permitted Transferees of Stockholder;provided that, any such Permitted Transferee shall enter into a supplement to this Agreement, consented to in writing by Buyer, agreeing to be bound by the terms of this Agreement; or
(ii) pledge to a bank or securities firm of Stockholder's Shares securing a bona fide loan;provided that the pledge agreement with the pledgee shall provide that the Stockholder shall continue at all times to have the right, from time to time, to vote and to give consents, ratifications and waivers with respect to such pledged Shares; andprovided further that any pledge agreement that Stockholder enters into shall provide that the pledgee shall give written notice to Buyer at least 10 days prior to the date such pledgee takes any action to exercise any remedies with respect to such Shares;
provided that no such transfer is in violation of applicable federal or state securities laws.
ARTICLE 2
REPRESENTATIONS AND WARRANTIES
The Stockholder represents and warrants to Buyer as follows:
SECTION 2.1 Valid Title. The Stockholder is the owner of 100% of the Shares set forth next on the signature page to this Agreement, with full power to vote and dispose of such Shares, and there are no restrictions on the Stockholder's voting rights or rights of disposition pertaining to such Shares which would be inconsistent with this Agreement or interfere with Stockholder's performance of this Agreement.
SECTION 2.2 Non-Contravention. The execution, delivery and performance by the Stockholder of this Agreement and the consummation of the transactions contemplated hereby, do not and will not contravene or constitute a default under or give rise to a right of termination, cancellation or acceleration of any material right or obligation of the Stockholder or to a loss of any material benefit of the Stockholder under any provision of applicable law or regulation or of any agreement, judgment, injunction, order, decree or other instrument binding on the Stockholder.
SECTION 2.3 Authorization. The execution, delivery and performance by the Stockholder of this Agreement are within the Stockholder's powers and have been duly authorized by all necessary actions.
SECTION 2.4 Binding Effect. This Agreement constitutes a valid and binding agreement of the Stockholder, enforceable against the Stockholder in accordance with its terms, except as enforcement may be limited by bankruptcy, insolvency, moratorium or other similar laws related to creditors' rights generally.
E-2
SECTION 2.5 No Other Shares. The number of Shares set forth on the signatures page to thie Agreement are the only Shares owned by the Stockholder.
SECTION 3.1 Notices. All notices, requests and other communications to any party hereunder shall be deemed to have been duly given when delivered in person, by telegram, facsimile or by registered or certified mail (postage prepaid, return receipt requested) to such party as its address set forth on the signature page hereto.
SECTION 3.2 Amendments; No Waivers. (a) Any provisions of this Agreement may be amended of waived if, and only if, such amendment or waiver is in writing and signed, in the case of an amendment, by each of the parties hereto, or in the case of a waiver, by the party against whom the waiver is to be effective.
(b) No failure to delay by any party in the exercising any right, power or privilege hereunder shall operate as a waiver thereof nor shall any single or partial exercise thereof preclude any other or further exercise thereof or the exercise of any other right, power or privilege. The rights and remedies herein provided shall be cumulative and not exclusive of any rights or remedies provided by law.
SECTION 3.3 Termination. This Agreement shall automatically terminate upon termination of the Purchase Agreement in accordance with its terms.
SECTION 3.4 Severability. If any provision of this Agreement or the application of such provision to any party or set of circumstances shall, in any jurisdiction and to any extend, be finally held invalid or unenforceable, such term or provision shall only be ineffective as to such jurisdiction, and only to the extend of such invalidity or unenforceability, without invalidating or rendering unenforceable any other terms or provisions of this Agreement or under any other circumstances, and the parties shall negotiate in food faith a substitute provision which comes as close as possible to the invalidated or unenforceable term or provisions and which puts each party in a position as nearly as comparable as possible to the position it would have been in but for the finding of invalidity or unenforceability, while remaining valid and enforceable.
SECTION 3.5 Entire Agreement. This Agreement constitutes the entire Agreement among the parties hereto with respect to the subject matter hereof and supersedes all prior agreements and undertakings, both written and oral, among the parties with respect to the subject matter hereof.
SECTION 3.6 Successors and Assigns. The provisions of this Agreement shall be binding upon and inure to the benefit of the parties hereto and their respective successors and assigns (and, in the case of the Stockholder, the heirs and executors of the Stockholder); provided that, except as permitted by Section 1.2 or by will or intestacy, no party may assign, delegate or otherwise transfer all or any of his or its rights or obligations under this Agreement without the consent of the other party hereto.
SECTION 3.7. Counterparts; Effectiveness. This Agreement may be signed in any number of counterparts, each of which shall be an original, with the same effect as if the signatures thereto and hereto were upon the same instrument. This Agreement shall become effective when each party shall have received counterparts (or signature pages) hereof signed by all of the other parties.
SECTION 3.8 Governing Law; Specific Performance. The terms of this Agreement shall be construed in accordance with and governed by the law of the State of Delaware (without regard to principles of conflict of laws). Each of the parties acknowledges and agrees that the parties' respective remedies at law for a breach or threatened breach of any of the provisions of this Agreement would be
E-3
inadequate and, in recognition of that fact, each agrees that, in the event of a breach or threatened breach by any party of the provisions of this Agreement, in addition to any remedies oat law, each party, without posting any bond, shall be entitles to seek equitable relief in the form of specific performance, a temporary restraining order, a temporary or permanent injunction or any other equitable remedy which may then be available.
SECTION 3.9 Expenses. All costs and expenses incurred in connection with this Agreement shall be paid by the party incurring such cost or expense.
SECTION 3.10 Certain Events. The Stockholders agrees that this Agreement and the obligations hereunder shall attach to its Shares and shall be binding upon any person to which legal or beneficial ownership of such shares shall pass, whether by operation of law or otherwise.
SECTION 3.11 No Revocation. The voting agreements contained herein are coupled with an interest and may not be revoked prior to termination of this Agreement in accordance with Section 3.3, except by written consent of Buyer.
E-4
IN WITNESS WHEREOF, the parties herto have executed this Agreement, or caused this Agreement to be duly executed by their respective authorized officers or representatives, as of the day and year first above written.
| Faulding Pharmaceutical Co. | |||
By: | |||
| Its: | |||
Address: Mack Cali Centre II 650 From Road, Second Floor Paramus, NJ 07652 | |||
Stockholder: /s/ LEONARD P. SHAYKIN Leonard P. Shaykin | |||
Number of Shares Owned: 622,554 | |||
Address: c/o NaPro BioTherapeutics, Inc. 4840 Pearl East Circle, Suite 300W Boulder, Colorado 80301 | |||
E-5
VOTING AGREEMENT made this day of August, 2003 (the "Agreement"), between Sterling K. Ainsworth (the "Stockholder"), in his capacity as a stockholder of NaPro BioTherapeutics, Inc., a Delaware corporation (the "Company"), and Faulding Pharmaceutical Co., a Delaware corporation ("Buyer").
Concurrently with the execution of this Agreement, Buyer and the Company have entered into an Asset Purchase Agreement dated as of August 25, 2003 (the "Purchase Agreement") pursuant to which Buyer will acquire the Paclitaxel Business of the Company (the "Asset Purchase"). The transactions contemplated by the Purchase Agreement are collectively referred to as the "Transactions." Capitalized terms not defined in this Agreement have the meanings given them in the Purchase Agreement.
In order to induce Buyer to enter into the Purchase Agreement with the Company, Buyer has requested, and the Stockholder has agreed, that the Stockholder enter into this Agreement;
NOW, THEREFORE, in consideration of the foregoing and for other good and valuable consideration, the receipt and sufficiency of which is hereby acknowledged, the parties agree as follows:
The Stockholder hereby agrees with Buyer as follows:
SECTION 1.1. Voting of Shares. (a) At any meeting of the stockholders of the Company, however called, at every adjournment of any such meeting, and in connection with any written consent of the stockholders of the Company, the Stockholder will cause all of his Shares to be voted, during the term of this Agreement, in favor of (i) a resolution approving the transactions contemplated by the Purchase Agreement in accordance with the Delaware General Corporation Law, and (ii) all other Transactions as to which stockholders of the Company are called upon to vote.
For purposes of this Agreement, (i) "Person" shall mean an individual, partnership, limited liability company, corporation, joint stock company, trust, estate, joint venture, association or unincorporated organization, or any other entity or organization, including a government or political subdivision or any agency or instrumentality thereof, and (ii) "Shares" shall mean any and all shares of capital stock of the Company which are entitled to vote in any election of the board of directors of the Company or on any matter submitted to the stockholders of the Company now owned and/or subsequently acquired by the Stockholder through purchase, gift, stock splits, stock dividends and the exercise of stock options or other derivative security.
(b) The Stockholder agrees that during the term of this Agreement, the Stockholder shall attend or otherwise participate in all duly called stockholder meetings and any adjournments of such meetings and in all actions by written consent of stockholders in which the Asset Purchase or any Transaction is being considered.
(c) The parties hereto agree and acknowledge that nothing in this Article I or any other part of this Agreement shall be construed as requiring the Stockholder to propose, endorse, approve or recommend the Purchase Agreement or the transactions contemplated thereby in the Stockholder's capacity as a director or officer of the Company in any manner inconsistent with his fiduciary duties as director or officer.
F-1
SECTION 1.2. No Proxies or Encumbrances. The Stockholder shall not (i) grant any proxies or enter into any voting trust or other agreement or arrangement with respect to the voting of any of the Shares in a manner which would be inconsistent with the provisions of this Agreement, (ii) sell, assign, transfer, encumber or otherwise dispose of or enter into any contract, option or other arrangement or understanding with respect to, the direct or indirect sale, assignment, transfer, encumbrance or other disposition of any of his Shares or any interest therein except for Permitted Transfers to Permitted Transferees (as such terms are defined below) or (iii) seek or solicit any of the foregoing. For purposes of this Agreement, (i) "Permitted Transferee" means any Person controlled, directly or indirectly, by Stockholder, Stockholder's spouse and children, and any trust for the benefit of Stockholder, Stockholder's spouse or children, and (ii) each transfer to a Permitted Transferee shall constitute a "Permitted Transfer" only if it is a:
(i) transfer to a Permitted Transferee and, in the case of a Permitted Transferee, transfer to the Stockholder or to other Permitted Transferees of Stockholder;provided that, any such Permitted Transferee shall enter into a supplement to this Agreement, consented to in writing by Buyer, agreeing to be bound by the terms of this Agreement; or
(ii) pledge to a bank or securities firm of Stockholder's Shares securing a bona fide loan;provided that the pledge agreement with the pledgee shall provide that the Stockholder shall continue at all times to have the right, from time to time, to vote and to give consents, ratifications and waivers with respect to such pledged Shares; andprovided further that any pledge agreement that Stockholder enters into shall provide that the pledgee shall give written notice to Buyer at least 10 days prior to the date such pledgee takes any action to exercise any remedies with respect to such Shares;
provided that no such transfer is in violation of applicable federal or state securities laws.
ARTICLE 2
REPRESENTATIONS AND WARRANTIES
The Stockholder represents and warrants to Buyer as follows:
SECTION 2.1 Valid Title. The Stockholder is the owner of 100% of the Shares set forth next on the signature page to this Agreement, with full power to vote and dispose of such Shares, and there are no restrictions on the Stockholder's voting rights or rights of disposition pertaining to such Shares which would be inconsistent with this Agreement or interfere with Stockholder's performance of this Agreement.
SECTION 2.2 Non-Contravention. The execution, delivery and performance by the Stockholder of this Agreement and the consummation of the transactions contemplated hereby, do not and will not contravene or constitute a default under or give rise to a right of termination, cancellation or acceleration of any material right or obligation of the Stockholder or to a loss of any material benefit of the Stockholder under any provision of applicable law or regulation or of any agreement, judgment, injunction, order, decree or other instrument binding on the Stockholder.
SECTION 2.3 Authorization. The execution, delivery and performance by the Stockholder of this Agreement are within the Stockholder's powers and have been duly authorized by all necessary actions.
SECTION 2.4 Binding Effect. This Agreement constitutes a valid and binding agreement of the Stockholder, enforceable against the Stockholder in accordance with its terms, except as enforcement may be limited by bankruptcy, insolvency, moratorium or other similar laws related to creditors' rights generally.
F-2
SECTION 2.5 No Other Shares. The number of Shares set forth on the signatures page to thie Agreement are the only Shares owned by the Stockholder.
SECTION 3.1 Notices. All notices, requests and other communications to any party hereunder shall be deemed to have been duly given when delivered in person, by telegram, facsimile or by registered or certified mail (postage prepaid, return receipt requested) to such party as its address set forth on the signature page hereto.
SECTION 3.2 Amendments; No Waivers. (a) Any provisions of this Agreement may be amended of waived if, and only if, such amendment or waiver is in writing and signed, in the case of an amendment, by each of the parties hereto, or in the case of a waiver, by the party against whom the waiver is to be effective.
(b) No failure to delay by any party in the exercising any right, power or privilege hereunder shall operate as a waiver thereof nor shall any single or partial exercise thereof preclude any other or further exercise thereof or the exercise of any other right, power or privilege. The rights and remedies herein provided shall be cumulative and not exclusive of any rights or remedies provided by law.
SECTION 3.3 Termination. This Agreement shall automatically terminate upon termination of the Purchase Agreement in accordance with its terms.
SECTION 3.4 Severability. If any provision of this Agreement or the application of such provision to any party or set of circumstances shall, in any jurisdiction and to any extend, be finally held invalid or unenforceable, such term or provision shall only be ineffective as to such jurisdiction, and only to the extend of such invalidity or unenforceability, without invalidating or rendering unenforceable any other terms or provisions of this Agreement or under any other circumstances, and the parties shall negotiate in food faith a substitute provision which comes as close as possible to the invalidated or unenforceable term or provisions and which puts each party in a position as nearly as comparable as possible to the position it would have been in but for the finding of invalidity or unenforceability, while remaining valid and enforceable.
SECTION 3.5 Entire Agreement. This Agreement constitutes the entire Agreement among the parties hereto with respect to the subject matter hereof and supersedes all prior agreements and undertakings, both written and oral, among the parties with respect to the subject matter hereof.
SECTION 3.6 Successors and Assigns. The provisions of this Agreement shall be binding upon and inure to the benefit of the parties hereto and their respective successors and assigns (and, in the case of the Stockholder, the heirs and executors of the Stockholder); provided that, except as permitted by Section 1.2 or by will or intestacy, no party may assign, delegate or otherwise transfer all or any of his or its rights or obligations under this Agreement without the consent of the other party hereto.
SECTION 3.7. Counterparts; Effectiveness. This Agreement may be signed in any number of counterparts, each of which shall be an original, with the same effect as if the signatures thereto and hereto were upon the same instrument. This Agreement shall become effective when each party shall have received counterparts (or signature pages) hereof signed by all of the other parties.
SECTION 3.8 Governing Law; Specific Performance. The terms of this Agreement shall be construed in accordance with and governed by the law of the State of Delaware (without regard to principles of conflict of laws). Each of the parties acknowledges and agrees that the parties' respective remedies at law for a breach or threatened breach of any of the provisions of this Agreement would be
F-3
inadequate and, in recognition of that fact, each agrees that, in the event of a breach or threatened breach by any party of the provisions of this Agreement, in addition to any remedies oat law, each party, without posting any bond, shall be entitles to seek equitable relief in the form of specific performance, a temporary restraining order, a temporary or permanent injunction or any other equitable remedy which may then be available.
SECTION 3.9 Expenses. All costs and expenses incurred in connection with this Agreement shall be paid by the party incurring such cost or expense.
SECTION 3.10 Certain Events. The Stockholders agrees that this Agreement and the obligations hereunder shall attach to its Shares and shall be binding upon any person to which legal or beneficial ownership of such shares shall pass, whether by operation of law or otherwise.
SECTION 3.11 No Revocation. The voting agreements contained herein are coupled with an interest and may not be revoked prior to termination of this Agreement in accordance with Section 3.3, except by written consent of Buyer.
F-4
IN WITNESS WHEREOF, the parties herto have executed this Agreement, or caused this Agreement to be duly executed by their respective authorized officers or representatives, as of the day and year first above written.
| Faulding Pharmaceutical Co. | |||
By: | /s/ W. R. GRIFFITH William R. Griffith | ||
| Its: | Director | ||
Address: Mack Cali Centre II 650 From Road, Second Floor Paramus, NJ 07652 | |||
Stockholder: /s/ STERLING K. AINSWORTH Sterling K. Ainsworth | |||
Number of Shares Owned: 913,274 | |||
Address: c/o NaPro BioTherapeutics, Inc. 4840 Pearl East Circle, Suite 300W Boulder, Colorado 80301 | |||
F-5
NaPro BioTherapeutics, Inc.
Proxy Solicited by the Board of Directors
For the Special Meeting of Stockholders to Be Held on , 2003
The undersigned hereby appoints Leonard P. Shaykin, Dr. Sterling K. Ainsworth and Dr. Patricia A. Pilia, or any of them, with full power of substitution, as a proxy or proxies to represent the undersigned at the special meeting of stockholders of NaPro BioTherapeutics, Inc. (the "Company") to be held on , 2003, at local time at [location to be determined], and at any adjournment(s) or postponement(s) thereof (the "Special Meeting"), and to vote there all the shares of common stock, $.0075 par value per share, held of record by the undersigned at the close of business on , 2003, with all the power that the undersigned would possess if personally present, as designated on the reverse side on the matters set forth on the reverse side and described in more detail in the Notice of Special Meeting and Proxy Statement dated , 2003, receipt of which is hereby acknowledged.
Shares will be voted as specified.THE BOARD OF DIRECTORS RECOMMENDS A VOTE FOR AND APPROVAL OF PROPOSALS 1 AND 2. IF NO SPECIFICATION IS MADE, THIS PROXY WILL BE VOTED IN ACCORDANCE WITH THE RECOMMENDATIONS OF THE BOARD OF DIRECTORS.
The proxies or substitutes may vote accordingly in their discretion upon any other business that may properly come before the Special Meeting or any adjournments or postponements thereof. The undersigned hereby revokes any proxies as to said shares heretofore given by the undersigned, and ratifies and confirms all that said attorney and proxies may lawfully do by virtue hereof.
Please mark your votes as in this example: ý
1. To adopt a resolution approving the Company's sale of its paclitaxel business to Faulding Pharmaceutical Co. under the terms set forth in the asset purchase agreement between Faulding and the Company dated as of August 25, 2003, as described in more detail in the Proxy Statement:
| o FOR | o AGAINST | o ABSTAIN |
2. To adjourn the Special Meeting to a later date or dates, if necessary, to permit further solicitation of proxies in the event there are not sufficient votes to approve the asset sale at the time the special meeting is convened or to allow additional time for the satisfaction of conditions to the sale:
| o FOR | o AGAINST | o ABSTAIN |
3. The proxy is authorized to vote upon any other business as may properly come before the Special Meeting or any postponements or adjournments thereof.
THIS PROXY IS SOLICITED ON BEHALF OF THE BOARD OF DIRECTORS OF NAPRO BIOTHERAPEUTICS, INC. PLEASE DATE AND SIGN THIS PROXY AND RETURN IT IN THE ENCLOSED ENVELOPE. THE GIVING OF A PROXY WILL NOT AFFECT YOUR RIGHT TO VOTE IN PERSON IF YOU ATTEND THE SPECIAL MEETING.
| Signature(s): | Date: | |||
| Name (print): |
NOTE: Please sign this proxy as your name appears hereon, including the title "Executor," "Trustee," etc. if such is indicated. If joint account, each joint owner should each sign. If stock is held by a corporation, this proxy should be executed by a proper officer thereof.
THANK YOU FOR VOTING