Exhibit 99.1

Link to searchable text of slide shown above

Link to searchable text of slide shown above

Link to searchable text of slide shown above

Link to searchable text of slide shown above

Link to searchable text of slide shown above

Link to searchable text of slide shown above

Link to searchable text of slide shown above

Link to searchable text of slide shown above

Link to searchable text of slide shown above

Link to searchable text of slide shown above
Link to searchable text of slide shown above

Link to searchable text of slide shown above
Link to searchable text of slide shown above
Link to searchable text of slide shown above
Link to searchable text of slide shown above
Link to searchable text of slide shown above
Link to searchable text of slide shown above

Link to searchable text of slide shown above

Link to searchable text of slide shown above

Link to searchable text of slide shown above
Link to searchable text of slide shown above
Link to searchable text of slide shown above

Link to searchable text of slide shown above

Link to searchable text of slide shown above
Link to searchable text of slide shown above
Link to searchable text of slide shown above
Link to searchable text of slide shown above
Link to searchable text of slide shown above

Link to searchable text of slide shown above

Link to searchable text of slide shown above

Link to searchable text of slide shown above

Link to searchable text of slide shown above

Link to searchable text of slide shown above

Link to searchable text of slide shown above
Searchable text section of graphics shown above
Rodman & Renshaw Techvest Conference
Investor Presentation – Oncology Focus
October 27, 2004
[LOGO]
Forward-Looking Statements
Statements in this presentation regarding Tapestry Pharmaceuticals, Inc.’s business that are not historical facts are “forward-looking statements” that involve risks and uncertainties. Forward-looking statements can be identified by the use of words such as “potential,” “believes,” “intends,” “estimates,” “may,” “will,” “should,” “anticipates,” “expects” or comparable terminology or by discussions of strategy. Such forward looking statements include information regarding the preclinical and clinical development of our product candidates, particularly about the potential for efficacy of such product candidates and the timing and outcome of such development. Actual results might differ materially from those projected in the forward-looking statements. Such forward-looking statements involve risks and uncertainties including: implementation risks associated with development of our oncology and targeted oncology businesses; risks that our product candidates will not work in treating disease or are no more effective in treating disease than new or existing competitive treatments; risks that we may be blocked or limited in the development of our product candidates because of the intellectual property rights of third parties; risks that we may be limited in ability to obtain, maintain and enforce our own intellectual property; risks that development of our product candidates may be delayed or not be completed because the costs of further development may exceed the value of our product candidates; risks that the entry into the clinic of our product candidates may be delayed because of regulatory or other factors; risks the Company’s business will require substantial additional capital that the Company has not secured; risks that the Company’s resources may be insufficient to fund the Company’s plans for the development of its product candidates; and all other factors identified under the captions “Risk Factors,” “Special Note Regarding Forward Looking Statements” and “Cautionary Note Regarding Forward Looking Statements” in the Company’s documents filed from time to time with the SEC, including the Company’s Current Report on Form 8-K, as amended, filed February 11, 2004, Annual Report on Form 10-K/A for the year ending December 31, 2003 filed May 5, 2004, Quarterly Report on Form 10-Q for the quarter ended June 30, 2004, and any report filed by the Company with the Securities and Exchange Commission thereafter.
For further information, please contact L. Robert Cohen, Vice President, Investor Relations of Tapestry Pharmaceuticals, Inc., 212 218 8715.
A company focused on proprietary therapeutics for the treatment of cancer and hereditary disease
Development Pipeline
Oncology Products |
| Potential Indications |
|
|
|
• TPI 287 |
| Breast Cancer |
|
|
|
• TPI 273 |
| Breast Cancer |
|
|
|
• TPI 284 |
| Head and Neck Cancer |
|
|
|
Genomic Products |
| Indications |
|
|
|
• Oligo Therapy |
| Huntington’s Disease |
Therapeutic Development:
Oncology
Oncology Portfolio
• Applying our oncology expertise in natural products to advance our portfolio of cancer therapeutics
• Significant IP in all programs
• Third Generation Taxane: TPI 287
• Designed to overcome resistance caused by MDR-1
• Proprietary Quassinoid: TPI 273
• Protein synthesis inhibitor
• Downregulates c-myc overexpression in tumors
• Targeted Oncology Program: TPI 284
• Paclitaxel linked to proprietary peptide ligands that target specific tumors
• Potential for improved efficacy and toxicity profile
TPI 287:
A Third Generation Taxane
TPI 287: A Third Generation Taxane
Taxanes are highly active chemotherapeutic agents in the treatment of advanced common malignancies such as NSCLC and cancers of the breast, ovary, prostate and head and neck.
However, these neoplasms ultimately become resistant to the effects of currently available taxanes. A taxane that could overcome this resistance would be a valuable addition to the oncologic therapeutic armamentarium.
Rationale for TPI 287
• Leveraged extensive paclitaxel chemistry experience
• Optimized for desired attributes (e.g., MDR independence)
TPI 287: A Third Generation Taxane
• More active than paclitaxel or docetaxel in a variety of taxane resistant human tumor cell lines
• May circumvent MDR-1 seen in breast cancer, non-small cell lung cancer and prostate cancer
• May circumvent mutant tubulin present in ovarian cancer
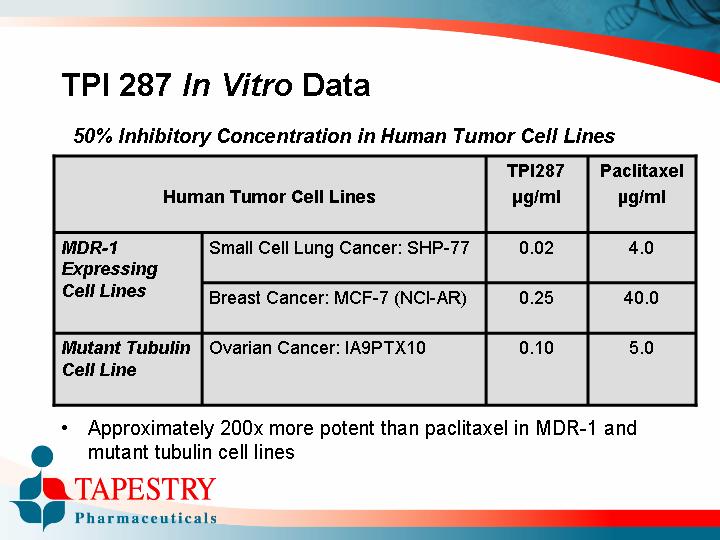
TPI 287 In Vitro Data
50% Inhibitory Concentration in Human Tumor Cell Lines
Human Tumor Cell Lines |
| TPI287 |
| Paclitaxel |
| ||
MDR-1 Expressing Cell Lines |
| Small Cell Lung Cancer: SHP-77 |
| 0.02 |
| 4.0 |
|
|
| Breast Cancer: MCF-7 (NCI-AR) |
| 0.25 |
| 40.0 |
|
|
|
|
|
|
|
|
|
Mutant Tubulin Cell Line |
| Ovarian Cancer: IA9PTX10 |
| 0.10 |
| 5.0 |
|
• Approximately 200x more potent than paclitaxel in MDR-1 and mutant tubulin cell lines
TPI 287 Clinical Opportunities
• Potential for taxane-resistant and taxane-naïve patients
• Breast, ovarian, non-small cell lung, and prostate cancer
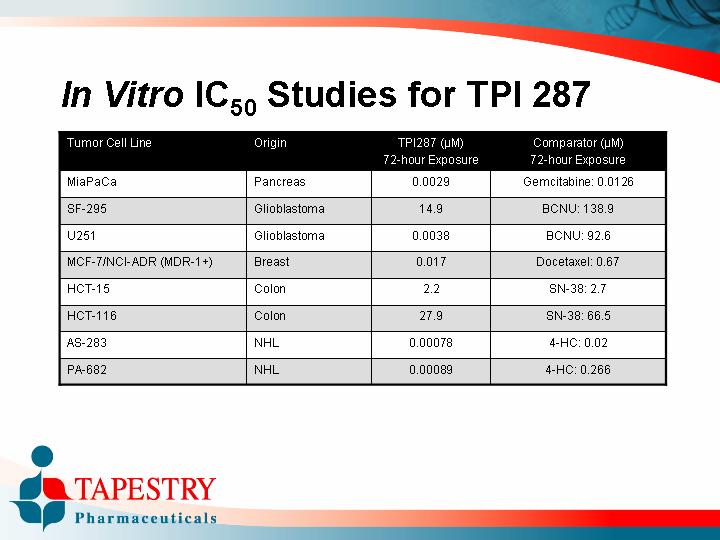
In Vitro IC50 Studies for TPI 287
Tumor Cell Line |
| Origin |
| TPI287 (µM) |
| Comparator (µM) |
|
MiaPaCa |
| Pancreas |
| 0.0029 |
| Gemcitabine: 0.0126 |
|
SF-295 |
| Glioblastoma |
| 14.9 |
| BCNU: 138.9 |
|
U251 |
| Glioblastoma |
| 0.0038 |
| BCNU: 92.6 |
|
MCF-7/NCI-ADR (MDR-1+) |
| Breast |
| 0.017 |
| Docetaxel: 0.67 |
|
HCT-15 |
| Colon |
| 2.2 |
| SN-38: 2.7 |
|
HCT-116 |
| Colon |
| 27.9 |
| SN-38: 66.5 |
|
AS-283 |
| NHL |
| 0.00078 |
| 4-HC: 0.02 |
|
PA-682 |
| NHL |
| 0.00089 |
| 4-HC: 0.266 |
|
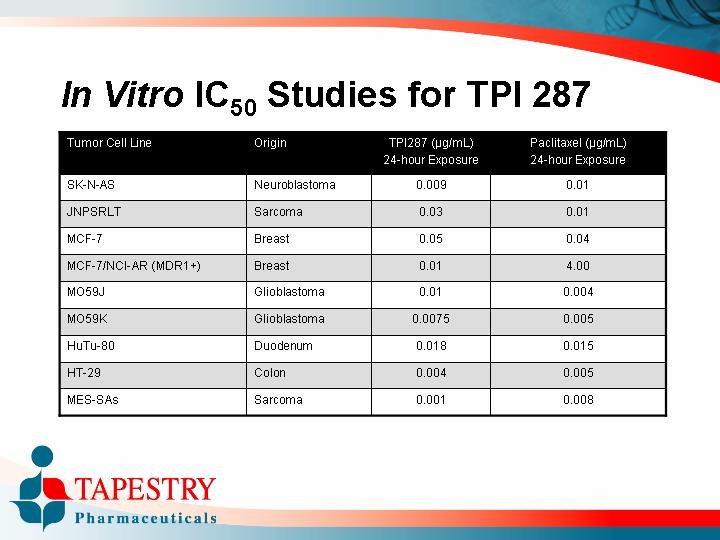
Tumor Cell Line |
| Origin |
| TPI287 (µg/mL) |
| Paclitaxel (µg/mL) |
|
SK-N-AS |
| Neuroblastoma |
| 0.009 |
| 0.01 |
|
JNPSRLT |
| Sarcoma |
| 0.03 |
| 0.01 |
|
MCF-7 |
| Breast |
| 0.05 |
| 0.04 |
|
MCF-7/NCI-AR (MDR1+) |
| Breast |
| 0.01 |
| 4.00 |
|
MO59J |
| Glioblastoma |
| 0.01 |
| 0.004 |
|
MO59K |
| Glioblastoma |
| 0.0075 |
| 0.005 |
|
HuTu-80 |
| Duodenum |
| 0.018 |
| 0.015 |
|
HT-29 |
| Colon |
| 0.004 |
| 0.005 |
|
MES-SAs |
| Sarcoma |
| 0.001 |
| 0.008 |
|
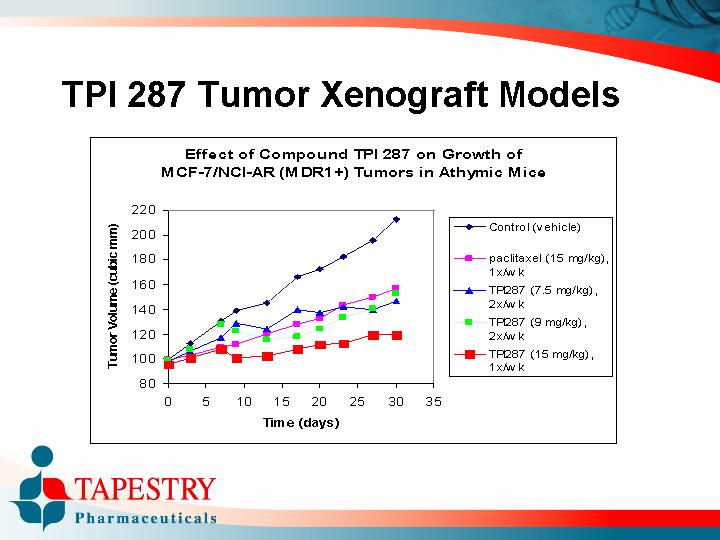
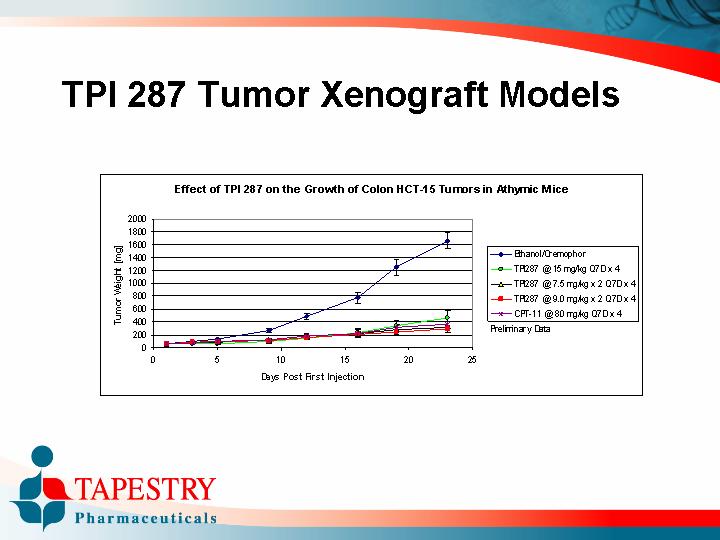
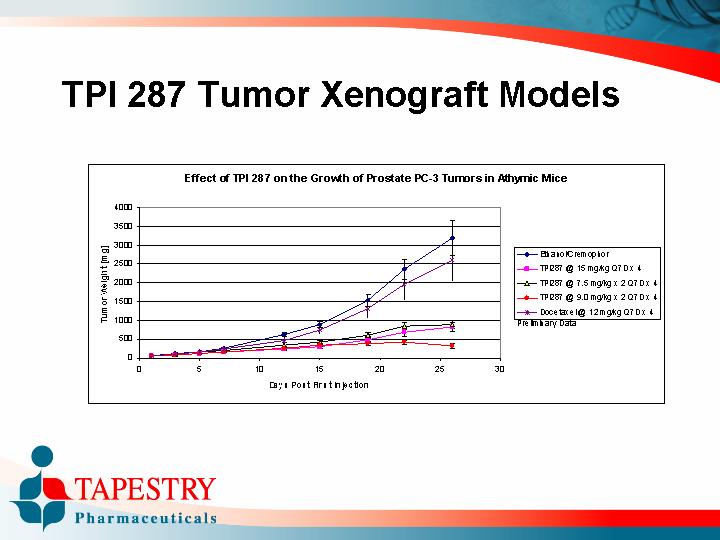
TPI 287 Tumor Xenograft Models
Effect of Compound TPI 287 on Growth of MCF-7/NCI-AR (MDR1+) Tumors in Athymic Mice
[CHART]
Effect of TPI 287 on the Growth of Colon HCT-15 Tumors in Athymic Mice
[CHART]
Effect of TPI 287 on the Growth of Prostate PC-3 Tumors in Athymic Mice
[CHART]
TPI 273:
Quassinoid
Quassinoids
The quassinoids are a class of organic compounds with growth inhibitory properties discovered in the early 1970’s.
The quassinoids were originally isolated from the Simaroubaceae family of plants indigenous to Guyana. Subsequently, quassinoids have been found in plants indigenous to Asia and North America.
Quassinoid Mechanism of Action
• Quassinoids inhibit protein synthesis in HeLa cells by 90% at a concentration of 2 µM.
• In a rabbit reticulocyte system, quassinoids inhibited protein synthesis in 79% at a concentration of 0.1 µM.
• Quassinoids are believed to act at the level of the ribosome, i.e., inhibiting the expression of myc protooncogene products.
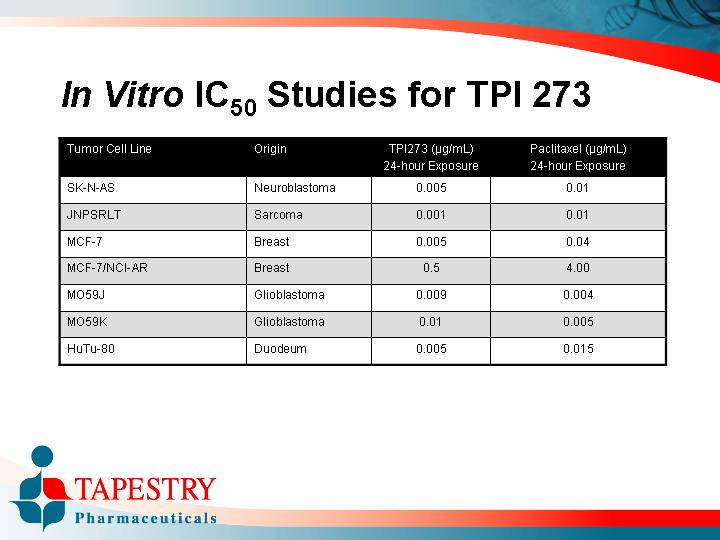
In Vitro IC50 Studies for TPI 273
Tumor Cell Line |
| Origin |
| TPI273 (µg/mL) |
| Paclitaxel (µg/mL) |
|
SK-N-AS |
| Neuroblastoma |
| 0.005 |
| 0.01 |
|
JNPSRLT |
| Sarcoma |
| 0.001 |
| 0.01 |
|
MCF-7 |
| Breast |
| 0.005 |
| 0.04 |
|
MCF-7/NCI-AR |
| Breast |
| 0.5 |
| 4.00 |
|
MO59J |
| Glioblastoma |
| 0.009 |
| 0.004 |
|
MO59K |
| Glioblastoma |
| 0.01 |
| 0.005 |
|
HuTu-80 |
| Duodeum |
| 0.005 |
| 0.015 |
|
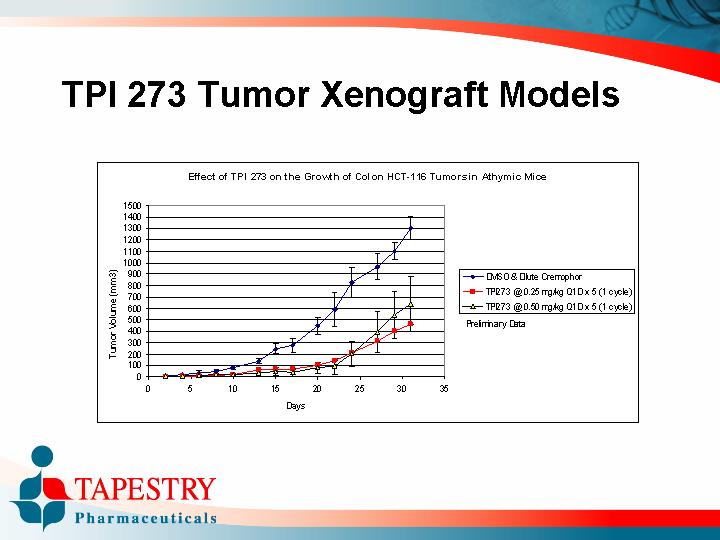
TPI 273 Tumor Xenograft Models
Effect of TPI 273 on the Growth of Colon HCT-116 Tumors in Athymic Mice
[CHART]
TPI 284
Head and Neck Tumor
Targeted Oncology Platform
Targeted Oncology Platform
• Cytotoxic agents chemically linked to proprietary peptides that target specific tumor receptors
• Preferential intracellular drug concentration
• Development program utilizing
• HN-1 Receptor Ligands
• In a collaboration with U.T. / M. D. Anderson Cancer Center
• Squamous cell carcinomas of the head & neck, lung and cervix
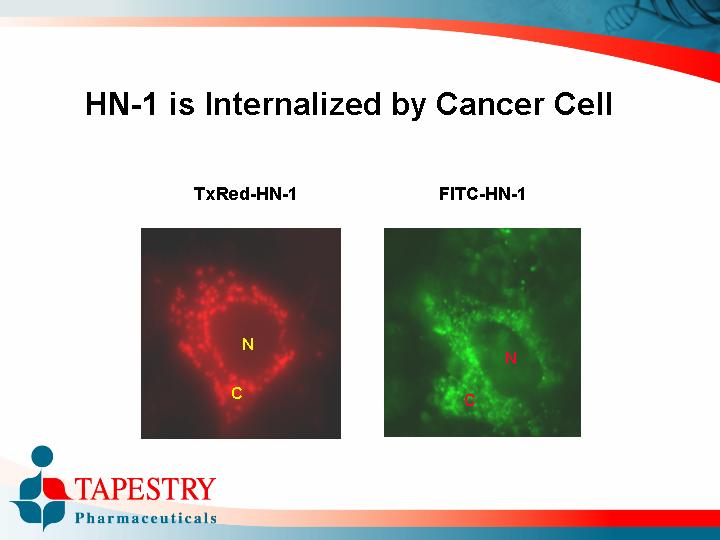
HN-1 is Internalized by Cancer Cell
TxRed-HN-1 | FITC-HN-1 |
|
|
[GRAPHIC] | [GRAPHIC] |
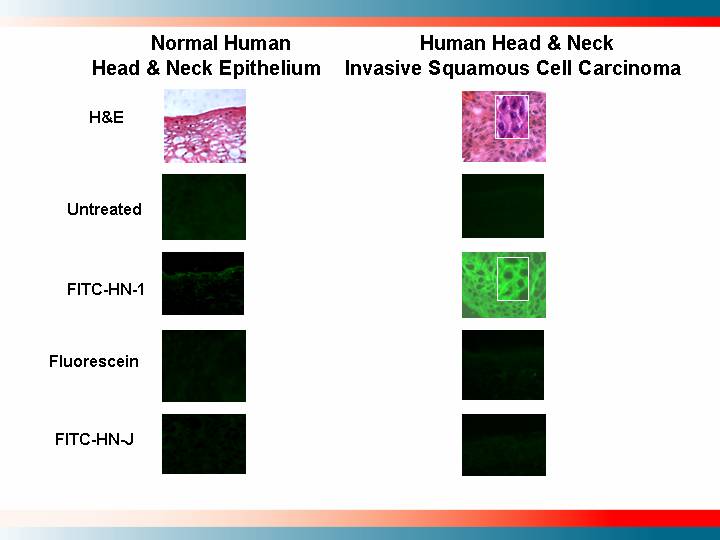
Normal Human |
| Human Head & Neck | |
|
|
| |
H&E | [GRAPHIC] |
| [GRAPHIC] |
Untreated | [GRAPHIC] |
| [GRAPHIC] |
FITC-HN-1 | [GRAPHIC] |
| [GRAPHIC] |
Fluorescein | [GRAPHIC] |
| [GRAPHIC] |
FITC-HN-J | [GRAPHIC] |
| [GRAPHIC] |
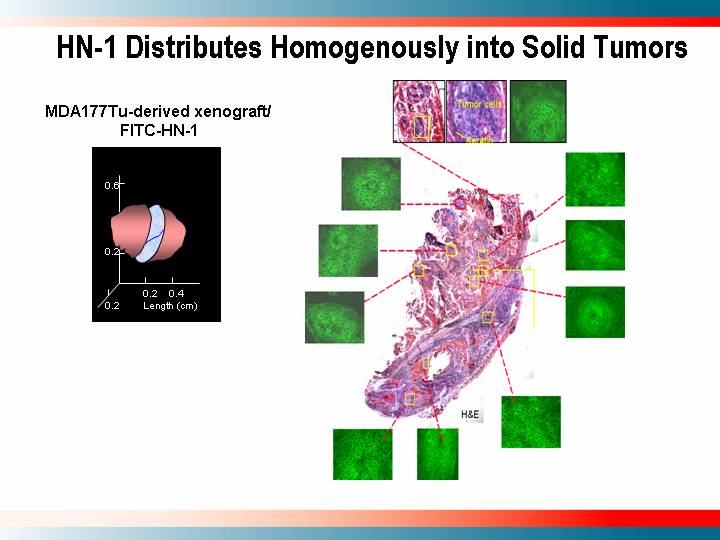
HN-1 Distributes Homogenously into Solid Tumors
MDA177Tu-derived xenograft/FITC-HN-1
[GRAPHIC]
[GRAPHIC]
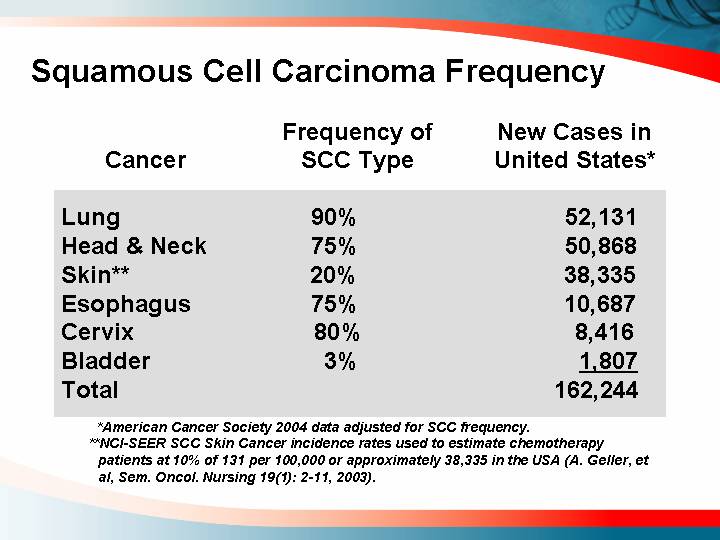
Squamous Cell Carcinoma Frequency
Cancer |
| Frequency of |
| New Cases in |
|
Lung |
| 90 | % | 52,131 |
|
Head & Neck |
| 75 | % | 50,868 |
|
Skin** |
| 20 | % | 38,335 |
|
Esophagus |
| 75 | % | 10,687 |
|
Cervix |
| 80 | % | 8,416 |
|
Bladder |
| 3 | % | 1,807 |
|
Total |
|
|
| 162,244 |
|
* American Cancer Society 2004 data adjusted for SCC frequency.
** NCI-SEER SCC Skin Cancer incidence rates used to estimate chemotherapy patients at 10% of 131 per 100,000 or approximately 38,335 in the USA (A. Geller, et al, Sem. Oncol. Nursing 19(1): 2-11, 2003).
Business Team
• Leonard Shaykin, Chairman and CEO
• Martin Batt, COO
• Gordon Link, SVP, CFO
• Kai Larson, VP, General Counsel
• Anne Bailey, VP & GM, Genomics Division
• Al Renzi, VP, Business Development
• L. Robert Cohen, VP, Investor Relations
Oncology Advisory Board
• Paul A. Bunn, Jr., M.D. (Chair)
Director of the University of Colorado Cancer Center, past president of American Society of Clinical Oncology (2002-2003), and former chairman of the Oncologic Drugs Advisory Committee (ODAC) for FDA
• S. Gail Eckhardt, M.D.
Professor of Medicine and Director of the Developmental Therapeutics and GI Malignancies Programs at the University of Colorado Health Sciences Center in Denver
• Eric K. Rowinsky, M.D.
Director of the Institute for Drug Development, Cancer Therapy and Research Center and Clinical Professor of Medicine at the University of Medicine at the University of Texas Health Science Center at San Antonio
• Daniel D. Von Hoff, M.D.
Professor of Medicine, Pathology, Molecular and Cellular Biology and the Director of the Arizona Health Sciences Center’s Cancer Therapeutics Program
Board of Directors
• Leonard P. Shaykin
Chairman of the Board, Chief Executive Officer
• Stephen K. Carter, M.D.
Formerly Senior Vice President, Worldwide Clinical Research and Development, Bristol-Myers Squibb and Deputy Director, Division of Cancer Treatment, National Cancer Institute
• Edward L. Erickson
Chairman of the Board, President, and Chief Executive Officer of Immunicon Corporation
• George M. Gould, Esq.
Attorney, Of Counsel, Gibbons, Del Deo, Dolan, Giffinger & Vecchione; formerly Vice President and Chief Patent Counsel of Hoffman-La Roche
• Arthur Hull Hayes, Jr., M.D.
President, MediScience Associates a pharmaceutical consulting company; Formerly FDA Commissioner
• The Honorable Richard N. Perle
A Director of Hollinger International; DigitalNet Holdings; Vital Spring Technologies; Autonomy, PLC; Formerly U.S. Assistant Secretary of Defense
• Patricia Pilia, Ph.D.
Executive VP, Secretary, Co-founder of Company
• Robert E. Pollack, Ph.D.
Professor of Biological Sciences and Director of the Center for the Study of Science and Religion at Columbia University
Investment Opportunity
• Experienced drug development team
• Experienced business development team
• Extensive intellectual property position in all compounds and technologies
• Four proprietary development programs
• Three in oncology and one in hereditary disease
• Distinguished scientific and clinical outside advisors
• Expect to file two INDs in Q4’04 and be prepared to enter the clinic pending regulatory & institutional approvals
• ~ $42 million cash resources available to advance development programs as of September 29, 2004
Strategies for Life TM