Searchable text section of graphics shown above
Tapestry Pharmaceuticals, Inc.
Rodman & Renshaw 3rd Annual Global Healthcare
Conference
05/15/06
[LOGO]
1
Statements in this presentation that are not historical facts are “forward-looking statements” that involve risks and uncertainties. Forward-looking statements can be identified by the use of words such as “may,” “will,” “should,” “anticipates,” “expects” or comparable terminology or by discussions of future events. Such forward looking statements include statements relating to the clinical development program for TPI 287, the anticipated activity of TPI 287 in taxanes resistant tumors, the safety and efficacy of TPI 287, the time tables for future development of TPI 287, and the company’s ability to continue to fund its development programs, either through existing resources or through new funding sources. Such statements involve known and unknown risks, uncertainties and other factors that may cause our actual results, performance or achievements to be materially different from the results, performance or achievements expressed or implied by such forwardlooking statements. Such risks, uncertainties and other factors include risks that clinical trials for TPI 287 will be delayed due to institutional approvals, patient recruitment, formulation and manufacturing difficulties or other factors; and that human clinical trials may show that TPI 287 is unsafe and/or ineffective in treating cancer in human patients. General implementation risks associated with development of any of our products include the risk that we will be blocked or limited in the development or marketing because of the intellectual property rights of third parties; that we are limited in our ability to obtain, maintain and enforce our own intellectual property rights; that development of our product candidates is delayed or terminated because the costs of further development exceed the value of such candidates; and that the Company’s resources are insufficient to continue development and the Company will be unable to raise sufficient additional capital to continue operations and development. Additional risks, uncertainties and other information is contained in the Company’s reports filed from time to time with the Securities and Exchange Commission. Particular attention should be paid to information in such SEC reports under the captions “Risk Factors” and “Special Note Regarding Forward-Looking Statements.” The Company disclaims any intention or obligation to update publicly or revise any forward-looking statements contained in this presentation or any other disclosure, whether as a result of new or additional information, future events or otherwise.
2
• Tapestry Overview
• TPI 287 Program
• Design and Structure
• Pre-clinical Biology Studies
• Q7D Phase I Study
• Q21D Phase I Study
• Oral Program
• Intellectual Property
• Board of Directors
• Scientific Advisory Board
3
• Founded in 1991
• Located in Boulder, Colorado
• Clinical stage biopharmaceutical company focused on developing novel oncology compounds
• Experienced pre-clinical & clinical oncology development team
• Leading clinical oncology advisory team
• Novel third generation taxane (TPI 287-IV) active in a broad range of solid tumors…currently in multiple Phase I clinical trials
• Compound also orally bioavailable (TPI 287-Oral) in animals
4
As of 5/09/2006
• Price: $3.60
• 52 Week High/Low: $2.10 - $7.00
• Market Value: $58.4M
Quarter Ended 3/29/2006
• Cash and Short Term Investments: $34.6M*
• Net Operating Loss: $4.4
• Number of Shares Outstanding: 16,235,433*
* Adjusted to reflect the financing completed on April 6, 2006
5
• Leonard Shaykin, Chairman and CEO
• Biotechnology entrepreneur, Venture capital, Private equity
• Martin Batt, COO
• Operations expertise in multiple industries including biotechnology, aerospace, computer software
• Gordon Link, CFO
• CPA plus over 15 years of biotechnology operations
• Michael Kurman, MD, CMO
• Senior clinical research positions at Janssen Research Foundation, U.S. Biosciences Inc., and Quintiles Transnational Corp.’s Oncology Therapeutics Division.
• David Emerson, VP of Cancer Biology
• 16 years in biopharmaceuticals; OSI, Gilead and GSK
• James McChesney, CSO
• Natural Products Chemistry specialist and former Director of Research Institute of Pharmaceutical Sciences and former Chairman of the Department of Pharmacognosy at the University of Mississippi
• Gilles Tapolsky, VP of Product Development
• 16 years in biopharmaceuticals; FeRx, ViroTex, Rhone Poulenc Rorer and Flamel Technologies
6
• 35 FTEs
• 27 Full time
• 8 Part time
• 3 Consultants
• 2 MDs, 7 PhDs
• 10 in clinical development and regulatory
7
• Achieved
• Initial Public Offering in 1994
• Generic paclitaxel approved in US & 25 other countries, then…
• Paclitaxel business sold to Mayne Pharma for $70MM in 2003
• Filed first proprietary IND for TPI 287 in 2004
• Commenced Phase I clinical trial for TPI 287 (Q7D) in 2005
• Evidence of oral bioavailability of TPI 287
• Commenced Phase I clinical trial for TPI 287 (Q21D) in 2006
• Upcoming
• Completion of Phase I clinical trials for TPI 287 (in 7 and 21 day dosing)
• Commencement of Phase II clinical trials for TPI 287
8
TPI 287: A Third Generation Taxane
• Pre-clinical data shows that TPI 287 has more activity than paclitaxel or docetaxel in a variety of taxane-resistant human tumor cell lines
• TPI 287 has the potential to:
• circumvent expression of the drug efflux transporter, MDR-1 protein, seen in breast, lung and prostate cancers
• be active in tumor types currently not responsive to paclitaxel, (i.e., colon cancer)
• be active in tumor cells that express mutant tubulin
10
TPI 287: Tubulin Binding Model
Computational modeling suggests that TPI 287 binds to wild-type tubulin through classic hydrogen bonds that contribute to a higher affinity than paclitaxel. In addition, TPI 287 appears to move closer and bind tighter to mutant tubulin by moving deeper into the binding pocket of mutant β-tubulin.
11
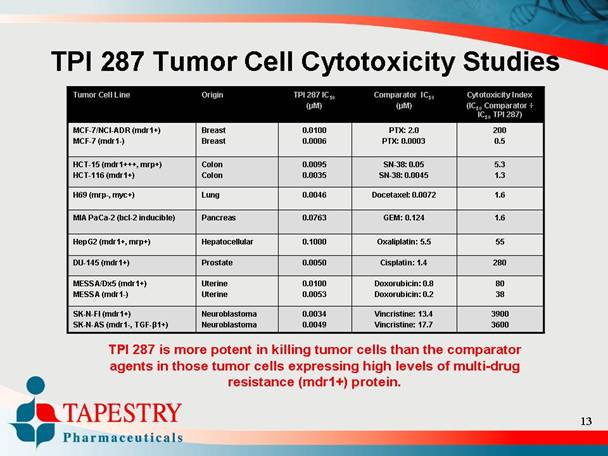
TPI 287 Tumor Cell Cytotoxicity Studies
Tumor Cell Line |
| Origin |
| TPI 287 IC50 |
| Comparator IC50 |
| Cytotoxicity Index |
|
MCF-7/NCI-ADR (mdr1+) |
| Breast |
| 0.0100 |
| PTX: 2.0 |
| 200 |
|
MCF-7 (mdr1-) |
| Breast |
| 0.0006 |
| PTX: 0.0003 |
| 0.5 |
|
HCT-15 (mdr1+++, mrp+) |
| Colon |
| 0.0095 |
| SN-38: 0.05 |
| 5.3 |
|
HCT-116 (mdr1+) |
| Colon |
| 0.0035 |
| SN-38: 0.0045 |
| 1.3 |
|
H69 (mrp-, myc+) |
| Lung |
| 0.0046 |
| Docetaxel: 0.0072 |
| 1.6 |
|
MIA PaCa-2 (bcl-2 inducible) |
| Pancreas |
| 0.0763 |
| GEM: 0.124 |
| 1.6 |
|
HepG2 (mdr1+, mrp+) |
| Hepatocellular |
| 0.1000 |
| Oxaliplatin: 5.5 |
| 55 |
|
DU-145 (mdr1+) |
| Prostate |
| 0.0050 |
| Cisplatin: 1.4 |
| 280 |
|
MESSA/Dx5 (mdr1+) |
| Uterine |
| 0.0100 |
| Doxorubicin: 0.8 |
| 80 |
|
MESSA (mdr1-) |
| Uterine |
| 0.0053 |
| Doxorubicin: 0.2 |
| 38 |
|
SK-N-FI (mdr1+) |
| Neuroblastoma |
| 0.0034 |
| Vincristine: 13.4 |
| 3900 |
|
SK-N-AS (mdr1-, TGF-β1+) |
| Neuroblastoma |
| 0.0049 |
| Vincristine: 17.7 |
| 3600 |
|
TPI 287 is more potent in killing tumor cells than the comparator
agents in those tumor cells expressing high levels of multi-drug
resistance (mdr1+) protein.
13
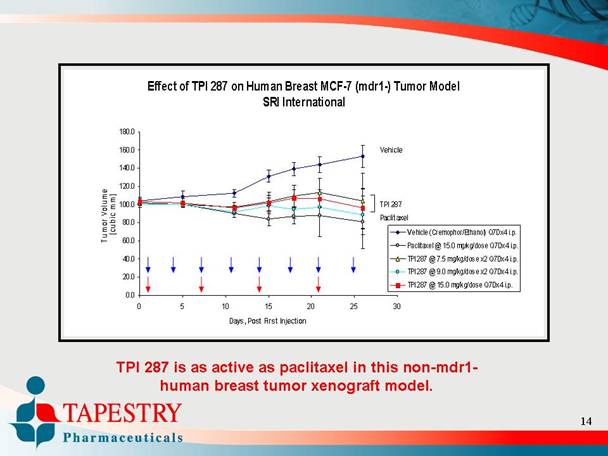
Effect of TPI 287 on Human Breast MCF-7 (mdr1-) Tumor Model
SRI International
[CHART]
TPI 287 is as active as paclitaxel in this non-mdr1-
human breast tumor xenograft model.
14
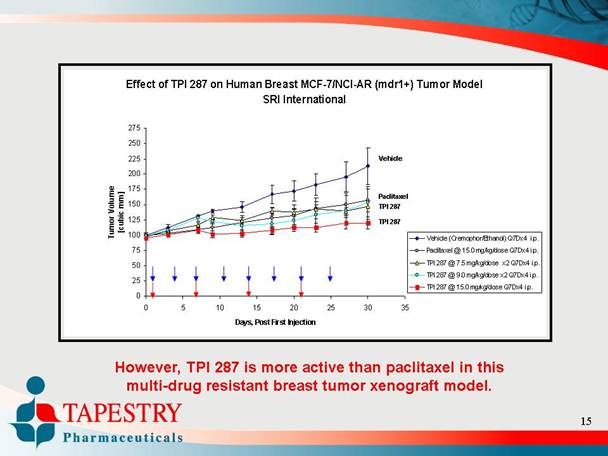
Effect of TPI 287 on Human Breast MCF-7/NCI-AR (mdr1+) Tumor Model
SRI International
[CHART]
However, TPI 287 is more active than paclitaxel in this
multi-drug resistant breast tumor xenograft model.
15
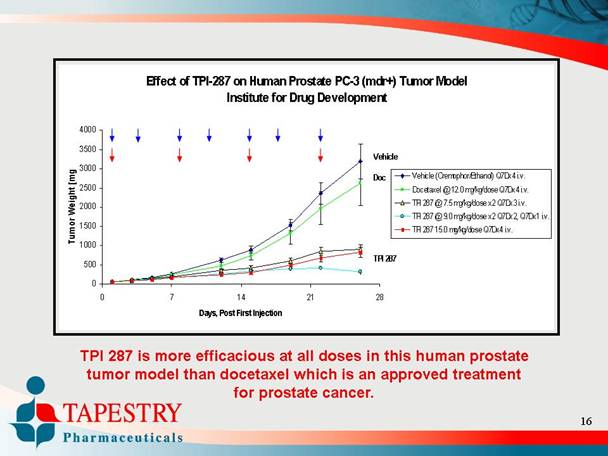
Effect of TPI-287 on Human Prostate PC-3 (mdr+) Tumor Model
Institute for Drug Development
[CHART]
TPI 287 is more efficacious at all doses in this human prostate
tumor model than docetaxel which is an approved treatment
for prostate cancer.
16
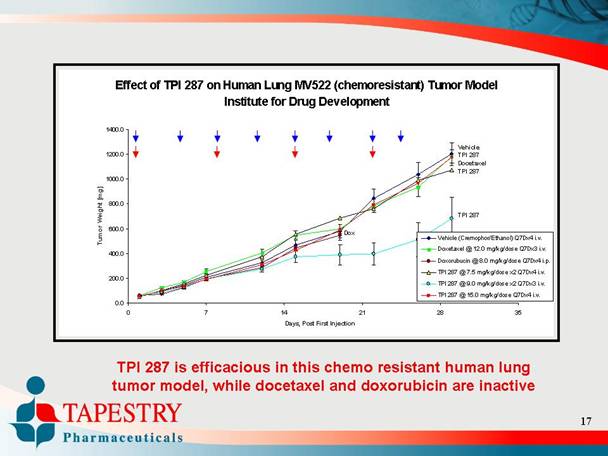
Effect of TPI 287 on Human Lung MV522 (chemoresistant) Tumor Model
Institute for Drug Development
[CHART]
TPI 287 is efficacious in this chemo resistant human lung
tumor model, while docetaxel and doxorubicin are inactive
17
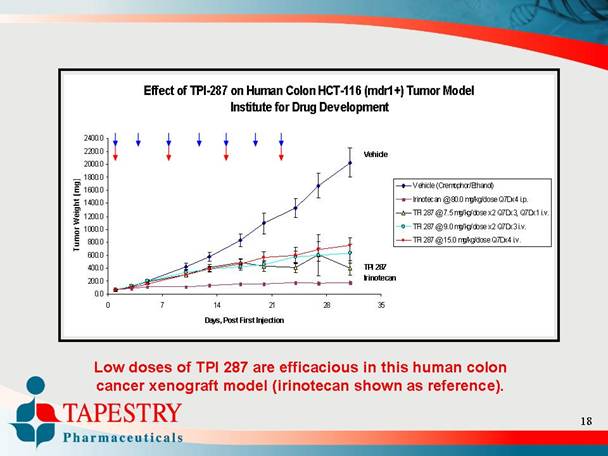
Effect of TPI-287 on Human Colon HCT-116 (mdr1+) Tumor Model
Institute for Drug Development
[CHART]
Low doses of TPI 287 are efficacious in this human colon
cancer xenograft model (irinotecan shown as reference).
18
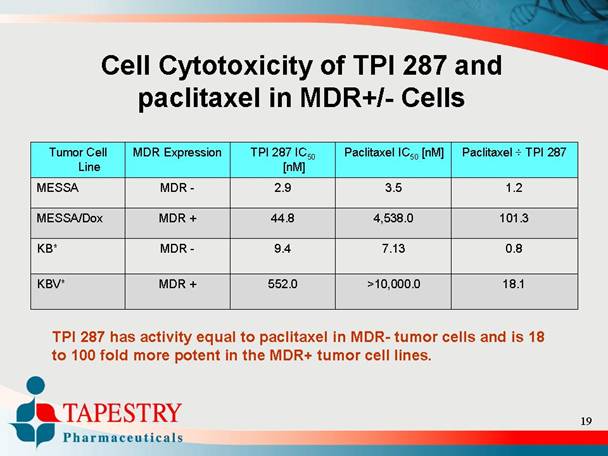
Cell Cytotoxicity of TPI 287 and
paclitaxel in MDR+/- Cells
Tumor Cell Line |
| MDR Expression |
| TPI 287 IC50 [nM] |
| Paclitaxel IC50 [nM] |
| Paclitaxel ÷ TPI 287 |
|
MESSA |
| MDR - |
| 2.9 |
| 3.5 |
| 1.2 |
|
MESSA/Dox |
| MDR + |
| 44.8 |
| 4,538.0 |
| 101.3 |
|
KB* |
| MDR - |
| 9.4 |
| 7.13 |
| 0.8 |
|
KBV* |
| MDR + |
| 552.0 |
| >10,000.0 |
| 18.1 |
|
TPI 287 has activity equal to paclitaxel in MDR- tumor cells and is 18
to 100 fold more potent in the MDR+ tumor cell lines.
19
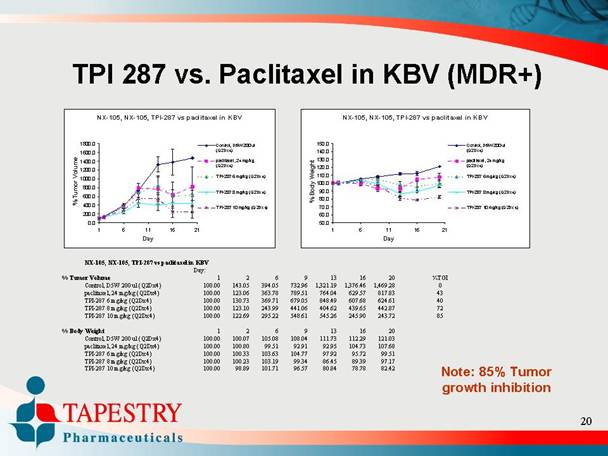
TPI 287 vs. Paclitaxel in KBV (MDR+)
[CHARTS]
NX-105, NX-105, TPI-287 vs paclitaxel in KBV
Day: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
% Tumor Volume |
| 1 |
| 2 |
| 6 |
| 9 |
| 13 |
| 16 |
| 20 |
| %TGI |
|
Control, D5W 200 ul (Q2Dx4) |
| 100.00 |
| 143.05 |
| 394.05 |
| 732.96 |
| 1,321.19 |
| 1,376.46 |
| 1,469.28 |
| 0 |
|
paclitaxel, 24 mg/kg (Q2Dx4) |
| 100.00 |
| 123.06 |
| 363.78 |
| 789.51 |
| 764.04 |
| 629.57 |
| 817.83 |
| 43 |
|
TPI-287 6 mg/kg (Q2Dx4) |
| 100.00 |
| 130.73 |
| 369.71 |
| 679.05 |
| 848.49 |
| 607.68 |
| 624.61 |
| 40 |
|
TPI-287 8 mg/kg (Q2Dx4) |
| 100.00 |
| 123.10 |
| 243.99 |
| 441.06 |
| 404.62 |
| 439.65 |
| 442.87 |
| 72 |
|
TPI-287 10 mg/kg (Q2Dx4) |
| 100.00 |
| 122.69 |
| 295.22 |
| 548.61 |
| 545.26 |
| 245.90 |
| 243.72 |
| 85 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
% Body Weight |
| 1 |
| 2 |
| 6 |
| 9 |
| 13 |
| 16 |
| 20 |
|
|
|
Control, D5W 200 ul (Q2Dx4) |
| 100.00 |
| 100.07 |
| 105.08 |
| 108.04 |
| 111.73 |
| 112.29 |
| 121.03 |
|
|
|
paclitaxel, 24 mg/kg (Q2Dx4) |
| 100.00 |
| 100.80 |
| 99.51 |
| 92.91 |
| 92.95 |
| 104.73 |
| 107.68 |
|
|
|
TPI-287 6 mg/kg (Q2Dx4) |
| 100.00 |
| 100.33 |
| 103.63 |
| 104.77 |
| 97.92 |
| 95.72 |
| 99.51 |
|
|
|
TPI-287 8 mg/kg (Q2Dx4) |
| 100.00 |
| 100.23 |
| 103.19 |
| 99.34 |
| 86.45 |
| 89.39 |
| 97.17 |
|
|
|
TPI-287 10 mg/kg (Q2Dx4) |
| 100.00 |
| 98.89 |
| 101.71 |
| 96.57 |
| 80.84 |
| 78.78 |
| 82.42 |
|
|
|
Note: 85% Tumor growth inhibition
20
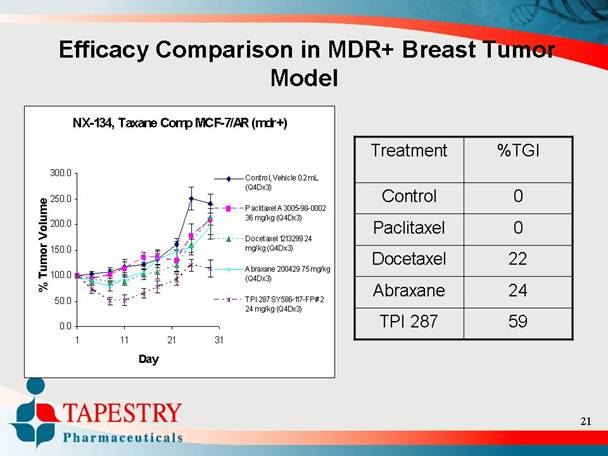
Efficacy Comparison in MDR+ Breast Tumor
Model
NX-134, Taxane Comp MCF-7/AR (mdr+)
[CHART]
Treatment |
| %TGI |
|
Control |
| 0 |
|
Paclitaxel |
| 0 |
|
Docetaxel |
| 22 |
|
Abraxane |
| 24 |
|
TPI 287 |
| 59 |
|
21
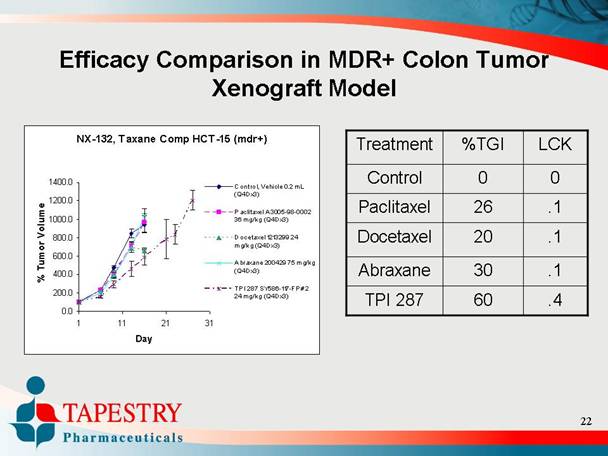
Efficacy Comparison in MDR+ Colon Tumor
Xenograft Model
NX-132, Taxane Comp HCT-15 (mdr+)
[CHART]
Treatment |
| %TGI |
| LCK |
|
Control |
| 0 |
| 0 |
|
Paclitaxel |
| 26 |
| .1 |
|
Docetaxel |
| 20 |
| .1 |
|
Abraxane |
| 30 |
| .1 |
|
TPI 287 |
| 60 |
| .4 |
|
22
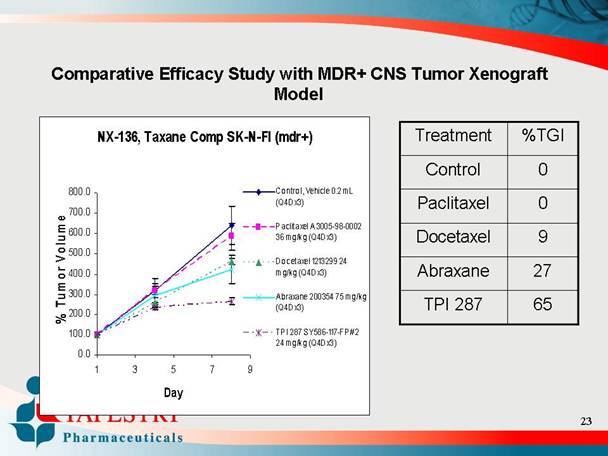
Comparative Efficacy Study with MDR+ CNS Tumor Xenograft
Model
NX-136, Taxane Comp SK-N-FI (mdr+)
[CHART]
Treatment |
| %TGI |
|
Control |
| 0 |
|
Paclitaxel |
| 0 |
|
Docetaxel |
| 9 |
|
Abraxane |
| 27 |
|
TPI 287 |
| 65 |
|
23
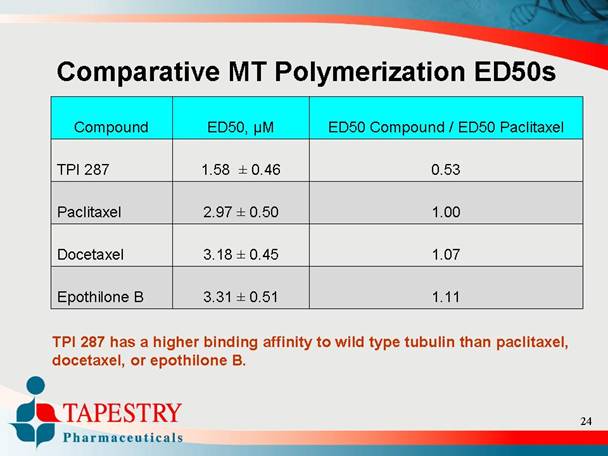
Comparative MT Polymerization ED50s
Compound |
| ED50, µM |
| ED50 Compound / ED50 Paclitaxel |
|
TPI 287 |
| 1.58 ± 0.46 |
| 0.53 |
|
Paclitaxel |
| 2.97 ± 0.50 |
| 1.00 |
|
Docetaxel |
| 3.18 ± 0.45 |
| 1.07 |
|
Epothilone B |
| 3.31 ± 0.51 |
| 1.11 |
|
TPI 287 has a higher binding affinity to wild type tubulin than paclitaxel, docetaxel, or epothilone B.
24
Paclitaxel is actively pumped out of cells by
MDR1 efflux pump, but TPI 287 is not
MDCKII-MDR permeability rate
Compound |
| A→B |
| B→A |
|
TPI 287 |
| 22.8 |
| 15.1 |
|
paclitaxel |
| 4.9 |
| 27.9 |
|
Caco-2 permeability rate
Compound |
| A→B |
| B→A |
|
TPI 287 |
| 6.17 |
| 8.39 |
|
paclitaxel |
| 9.31 |
| 17.40 |
|
Conclusions: TPI 287 is not a P-gp (MDR1) substrate
TPI 287 may be orally bioavailable
25
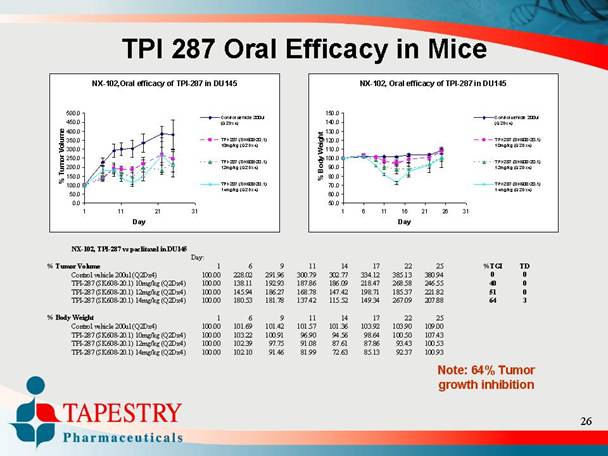
[CHARTS]
NX-102, TPI-287 vs paclitaxel in DU145
Day: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
% Tumor Volume |
| 1 |
| 6 |
| 9 |
| 11 |
| 14 |
| 17 |
| 22 |
| 25 |
| %TGI |
| TD |
|
Control vehicle 200ul (Q2Dx4) |
| 100.00 |
| 228.02 |
| 291.96 |
| 300.79 |
| 302.77 |
| 334.12 |
| 385.13 |
| 380.94 |
| 0 |
| 0 |
|
TPI-287 (SK608-20.1) 10mg/kg (Q2Dx4) |
| 100.00 |
| 138.11 |
| 192.93 |
| 187.86 |
| 186.09 |
| 218.47 |
| 268.58 |
| 246.55 |
| 40 |
| 0 |
|
TPI-287 (SK608-20.1) 12mg/kg (Q2Dx4) |
| 100.00 |
| 145.94 |
| 186.27 |
| 168.78 |
| 147.42 |
| 198.71 |
| 185.37 |
| 221.82 |
| 51 |
| 0 |
|
TPI-287 (SK608-20.1) 14mg/kg (Q2Dx4) |
| 100.00 |
| 180.53 |
| 181.78 |
| 137.42 |
| 115.52 |
| 149.34 |
| 267.09 |
| 207.88 |
| 64 |
| 3 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
% Body Weight |
| 1 |
| 6 |
| 9 |
| 11 |
| 14 |
| 17 |
| 22 |
| 25 |
|
|
|
|
|
Control vehicle 200ul (Q2Dx4) |
| 100.00 |
| 101.69 |
| 101.42 |
| 101.57 |
| 101.36 |
| 103.92 |
| 103.90 |
| 109.00 |
|
|
|
|
|
TPI-287 (SK608-20.1) 10mg/kg (Q2Dx4) |
| 100.00 |
| 103.22 |
| 100.91 |
| 96.90 |
| 94.56 |
| 98.64 |
| 100.50 |
| 107.43 |
|
|
|
|
|
TPI-287 (SK608-20.1) 12mg/kg (Q2Dx4) |
| 100.00 |
| 102.39 |
| 97.75 |
| 91.08 |
| 87.61 |
| 87.86 |
| 93.43 |
| 100.53 |
|
|
|
|
|
TPI-287 (SK608-20.1) 14mg/kg (Q2Dx4) |
| 100.00 |
| 102.10 |
| 91.46 |
| 81.99 |
| 72.63 |
| 85.13 |
| 92.37 |
| 100.93 |
|
|
|
|
|
Note: 64% Tumor growth inhibition
26
TPI 287 Rat Oral Bioavailability
• Comparison of two experimental formulations dosed orally
• IV formulation (Cremophor El P/alcohol)
• Suspension of TPI 287 in aqueous medium
• Findings
• IV formulation has an oral bioavailability of about 45%
• Aqueous suspension has an oral bioavailability of about 35%
TPI 287 demonstrates significant bioavailability when
compared to paclitaxel (45% vs. less than 10%).
Non-optimized formulation of TPI 287 shows remarkable bioavailability.
27
TPI 287 Additional Pre-clinical Studies
• Studies comparing TPI 287 to other approved taxanes
• Studies to determine efficacy of TPI 287 in brain tumors
• Studies designed to investigate combinations of TPI 287 with other anticancer drugs
• Studies designed to investigate sequencing of TPI 287 with other anticancer drugs
Tapestry is expanding its pre-clinical package in
anticipation of additional clinical studies
28
TPI 287 Single Dose Toxicology Studies
• Intravenous or Intraperitoneal dose tolerance and MTD
• Maximum Tolerated Dose (MTD)
• Mouse — 80 mg/kg or 240 mg/m2
• Rat — 48 mg/kg or 288 mg/m2
• Dog — 12.5 mg/kg or 221 mg/m2
The toxicology profile and the maximum tolerated dose of
TPI 287 are similar to other taxanes.
29
TPI 287 Repeat Dose Toxicology Studies
• Intravenous or Intraperitoneal dose tolerance and MTD
• Maximum Tolerated Dose
• Mouse — 12 mg/kg or 36 mg/m2
• Rat — 14 mg/kg or 84 mg/m2
• Dog — 4 mg/kg or 71 mg/m2 (basis for human dosing)
The toxicology profile and the maximum tolerated dose of
TPI 287 are similar to other taxanes.
30
• In Vitro Studies
• Evidence of activity in various cell lines including: breast, colon, lung, pancreas, prostate, and others
• Tumor Xenograft Studies
• Evidence of activity in breast, colon, lung, and prostate tumors models, including those tumors with expression of mdr-1 protein
• Toxicity
• Generally well-tolerated
• Key findings are GI in nature
• MTD based on dog repeat study is 4 mg/kg or 71 mg/m2
• Starting human dosing programs
• Based on 1/10 of MTD in dog
• Starting dose is 7 mg/m2
31
Q7D Trial Sites and Investigators
Lombardi Comprehensive Cancer Center at Georgetown University, Washington, D.C.
• John Marshall, M.D., Principal Investigator, Associate Professor of Oncology and Medicine at Georgetown University and Director of the Developmental Therapeutics program at the Lombardi Comprehensive Cancer Center.
• Westchester Medical Center, Valhalla, New York
• Tauseef Ahmed, M.D. Professor of Medicine and Chief of the Division of Oncology/Hematology at New York Medical College
• Rocky Mountain Cancer Center, Denver, Colorado
• Allen L. Cohn, M.D., Principal Investigator, Director Hematology/Oncology
33
Primary Objective:
• To determine the maximum tolerated dose of TPI 287
Secondary Objectives:
• To determine the safety of TPI 287
• To determine the antitumor activity of TPI 287
• To determine the pharmacokinetic profile of TPI 287
• To determine the pharmacodynamic profile of TPI 287
34
• Open-Label
• Multicenter Sequential groups
• Dose-escalation until MTD
• Anticipated number of patients: up to 48
35
• IND Submission Completed : December 2004
• First patient in Q7D Protocol: May 2005
• Expected Q7D Phase I trial MTD: 3Q06
36
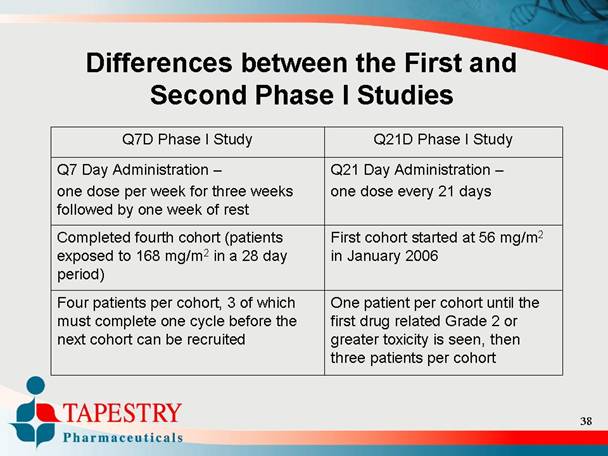
Differences between the First and
Second Phase I Studies
Q7D Phase I Study |
| Q21D Phase I Study |
Q7 Day Administration — one dose per week for three weeks followed by one week of rest |
| Q21 Day Administration — one dose every 21 days |
|
|
|
Completed fourth cohort (patients exposed to 168 mg/m2 in a 28 day period) |
| First cohort started at 56 mg/m2 in January 2006 |
|
|
|
Four patients per cohort, 3 of which must complete one cycle before the next cohort can be recruited |
| One patient per cohort until the first drug related Grade 2 or greater toxicity is seen, then three patients per cohort |
38
Q21D Trial Sites and Investigators
• Rocky Mountain Cancer Center, Denver, Colorado
• Allen L. Cohn, M.D., Principal Investigator, Director Hematology/Oncology
• Sheba Medical Center, Tel Hashomer, Israel
• Jacob Baram, M.D., Principal Investigator, Acting Director Inpatient Department
• Arizona Clinical Research Center, Tucson, Arizona
• Manuel R. Modiano, M.D., Principal Investigator, Medical Director
39
• Q21D Protocol Submission Completed : July 2005
• First patient in: January 2006
• Expected Q21D Phase I trial MTD: 3-4Q06
40
• Results of oral efficacy studies in mouse xenografts and oral bioavailability studies in rats were presented at the AACR/NCI/EORTC International Conference on Molecular Targets and Cancer Therapeutics in Philadelphia in November 2005
• These studies were performed using a non optimal formulation, thus on-going formulation work towards the development of a better formulation
• IND enabling studies early 2007 in support of a new IND to initiate the clinical development of TPI 287 orally administered
42
• Leonard P. Shaykin
Chairman of the Board, Chief Executive Officer
• Stephen K. Carter, M.D.
Former Senior Vice President, Worldwide Clinical Research and Development, Bristol-Myers Squibb and Deputy Director, Division of Cancer Treatment, National Cancer Institute
• George M. Gould, Esq.
Attorney, Of Counsel, Gibbons, Del Deo, Dolan, Giffinger & Vecchione; formerly Vice President and Chief Patent Counsel of Hoffman-La Roche
• Arthur Hull Hayes, Jr., M.D.
President, MediScience Associates a pharmaceutical consulting company; Former FDA Commissioner
• The Honorable Richard N. Perle
Former U.S. Assistant Secretary of Defense
• Patricia Pilia, Ph.D.
Co-founder of Company
• Robert E. Pollack, Ph.D.
Professor of Biological Sciences and Director of the Center for the Study of Science and Religion at Columbia University
• Elliot M. Maza, J.D., C.P.A.
Chief Financial Officer of Emisphere Technologies, Inc.
43
• Paul A. Bunn, Jr., M.D. (Chair)
Director of the University of Colorado Cancer Center, past president of American Society of Clinical Oncology (2002-2003), and former chairman of the Oncologic Drugs Advisory Committee (ODAC) for FDA
• S. Gail Eckhardt, M.D.
Professor of Medicine and Director of the Developmental Therapeutics and GI Malignancies Programs at the University of Colorado Health Sciences Center in Denver
• Robert E. Pollack, Ph.D.
Professor of Biological Sciences and Director of the Center for the Study of Science and Religion at Columbia University
• Eric K. Rowinsky, M.D.
Former Director of the Institute for Drug Development, Cancer Therapy and Research Center and Clinical Professor of Medicine at the University of Medicine at the University of Texas Health Science Center at San Antonio
• Daniel D. Von Hoff, M.D.
Professor of Medicine, Pathology, Molecular and Cellular Biology and the Director of the Arizona Health Sciences Center’s Cancer Therapeutics Program
44