Exhibit 99.1
| Tapestry Pharmaceuticals, Inc. Presentation to Investors |
| Forward-Looking Statements Statements in this presentation that are not historical facts are "forward-looking statements" that involve risks and uncertainties. Forward-looking statements can be identified by the use of words such as "may," "will," "should," "anticipates," "expects" or comparable terminology or by discussions of future events. Such forward looking statements include statements relating to the clinical development program for TPI 287, the anticipated activity of TPI 287 in taxanes resistant tumors, the safety and efficacy of TPI 287, the time tables for future development of TPI 287, and the company’s ability to continue to fund its development programs, either through existing resources or through new funding sources. Such statements involve known and unknown risks, uncertainties and other factors that may cause our actual results, performance or achievements to be materially different from the results, performance or achievements expressed or implied by such forward-looking statements. Such risks, uncertainties and other factors include risks that clinical trials for TPI 287 will be delayed due to institutional approvals, patient recruitment, formulation and manufacturing difficulties or other factors; and that human clinical trials may show that TPI 287 is unsafe and/or ineffective in treating cancer in human patients. General implementation risks associated with development of any of our products include the risk that we will be blocked or limited in the development or marketing because of the intellectual property rights of third parties; that we are limited in our ability to obtain, maintain and enforce our own intellectual property rights; that development of our product candidates is delayed or terminated because the costs of further development exceed the value of such candidates; and that the Company's resources are insufficient to continue development and the Company will be unable to raise sufficient additional capital to continue operations and development. Additional risks, uncertainties and other information is contained in the Company's reports filed from time to time with the Securities and Exchange Commission. Particular attention should be paid to information in such SEC reports under the captions "Risk Factors" and "Special Note Regarding Forward-Looking Statements." The Company disclaims any intention or obligation to update publicly or revise any forward-looking statements contained in this presentation or any other disclosure, whether as a result of new or additional information, future events or otherwise. |
| Tapestry Overview Proprietary next generation taxane, TPI 287 Potential for best in class Entering multiple Phase II trials Experienced oncology drug development team Large commercial opportunity Strong cash position Modest burn rate |
| TPI 287: The Next Generation Taxane |
| TPI 287: The Next Generation Taxane Proprietary new chemical entity More potent than paclitaxel or docetaxel in some preclinical tumor models Overcomes major mechanism for taxane resistance (the multiple drug resistance pump) Both intravenous and oral administration |
| TPI 287: The Next Generation Taxane TPI 287 has the potential to: Demonstrate activity in multiple tumor types Show improved activity in a variety of cancers resistant to other taxanes Be combined with other anti-cancer drugs |
| TPI 287 Clinical Development Phase I Clinical trials IV Q21 day schedule - completed IV Q7 day x 3 q 4 week schedule - ongoing Oral Phase I/II - planned 2Q07 Phase II Clinical trials Prostate Cancer Lung Cancer Other tumor types |
| TPI 287: Clinical Development |
| Phase I development Two Phase I studies exploring different regimens Study 01: dosing once each week for three weeks (Q7D x3) repeated every four weeks Continuing to evaluate this schedule for maximum tolerated dose (MTD) Study 02: dosing once every three weeks (Q21D) TPI 287 well tolerated Trial completed and closed to enrollment MTD determined for this schedule Data has been submitted to a major medical conference for presentation this year Phase I study to evaluate oral dosing in preparation TPI 287 Clinical Development |
| TPI 287 Clinical Development Phase II development Prostate Lung Multiple other tumors types based on preclinical models, MDR1 phenotype, as well as Phase I results Exploration of oral dosing in additional tumor types |
| TPI 287 Oral Program |
| Oral Program for TPI 287 TPI 287 is orally bioavailable in rodents Data presented at the AACR/NCI/EORTC International Conference on Molecular Targets and Cancer Therapeutics in Philadelphia in November 2005 Additional data has demonstrated efficacy of oral dosing in mouse xenografts First clinical study to evaluate oral dosing of TPI 287 to start 2Q07 |
| TPI 287 Pre-clinical Biology Studies |
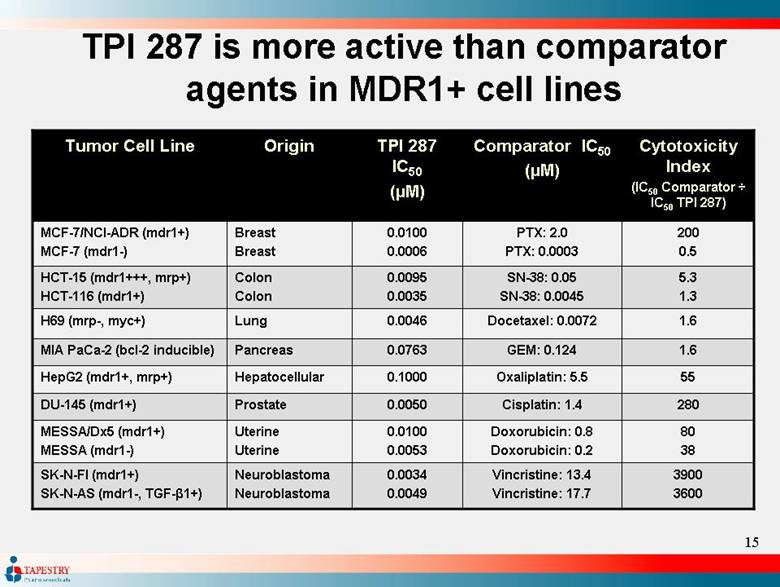
| TPI 287 Differentiation TPI 287 demonstrated greater efficacy in MDR1 positive tumors compared to docetaxel, paclitaxel, and Abraxane™ TPI 287 showed comparable activity in MDR1 negative tumor lines to the other taxanes Studies performed in MDCKII-MDR1 and Caco-2 cell systems demonstrate TPI 287 avoids MDR1 efflux TPI 287 has significant oral bioavailability in animals, and shows oral efficacy in xenograft models |
| TPI 287 is more active than comparator agents in MDR1+ cell lines 1.6 Docetaxel: 0.0072 0.0046 Lung H69 (mrp-, myc+) 1.6 GEM: 0.124 0.0763 Pancreas MIA PaCa-2 (bcl-2 inducible) 3900 3600 Vincristine: 13.4 Vincristine: 17.7 0.0034 0.0049 Neuroblastoma Neuroblastoma SK-N-FI (mdr1+) SK-N-AS (mdr1-, TGF-1+) Doxorubicin: 0.8 Doxorubicin: 0.2 Cisplatin: 1.4 Oxaliplatin: 5.5 SN-38: 0.05 SN-38: 0.0045 PTX: 2.0 PTX: 0.0003 Comparator IC50 (µM) 80 38 280 55 5.3 1.3 200 0.5 Cytotoxicity Index (IC50 Comparator ÷ IC50 TPI 287) 0.0100 0.0053 Uterine Uterine MESSA/Dx5 (mdr1+) MESSA (mdr1-) 0.0050 Prostate DU-145 (mdr1+) 0.1000 Hepatocellular HepG2 (mdr1+, mrp+) 0.0095 0.0035 Colon Colon HCT-15 (mdr1+++, mrp+) HCT-116 (mdr1+) 0.0100 0.0006 Breast Breast MCF-7/NCI-ADR (mdr1+) MCF-7 (mdr1-) TPI 287 IC50 (µM) Origin Tumor Cell Line |
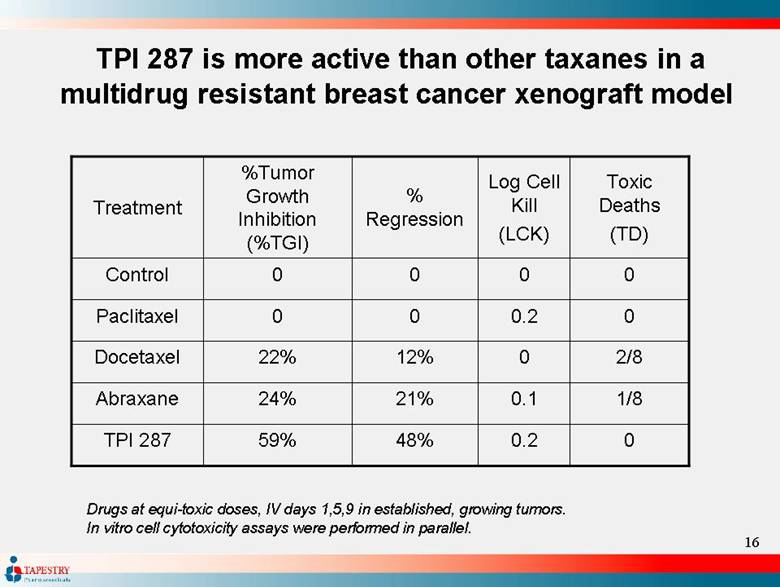
| TPI 287 is more active than other taxanes in a multidrug resistant breast cancer xenograft model 0.2 0.1 0 0.2 0 Log Cell Kill (LCK) TPI 287 Abraxane Docetaxel Paclitaxel Control Treatment 48% 21% 12% 0 0 % Regression 0 59% 1/8 24% 2/8 22% 0 0 0 0 Toxic Deaths (TD) % Tumor Growth Inhibition (% TGI) Drugs at equi-toxic doses, IV days 1,5,9 in established, growing tumors. In vitro cell cytotoxicity assays were performed in parallel. |
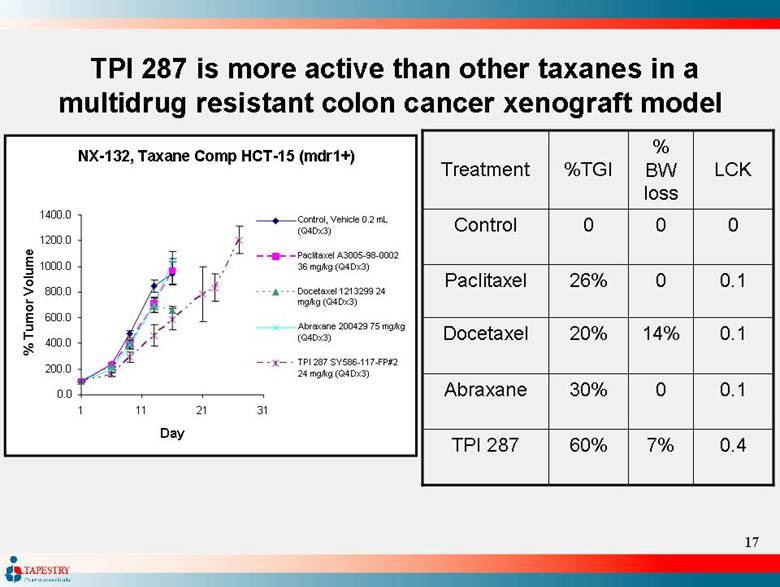
| TPI 287 is more active than other taxanes in a multidrug resistant colon cancer xenograft model TPI 287 Abraxane Docetaxel Paclitaxel Control Treatment 0.4 7% 60% 0.1 0 30% 0.1 14% 20% 0.1 0 26% 0 0 0 LCK % BW loss %TGI NX-132, Taxane Comp HCT-15 (mdr1+) 0.0 200.0 400.0 600.0 800.0 1000.0 1200.0 1400.0 1 11 21 31 Day % Tumor Volume Control, Vehicle 0.2 mL (Q4Dx3) Paclitaxel A3005-98-0002 36 mg/kg (Q4Dx3) Docetaxel 1213299 24 mg/kg (Q4Dx3) Abraxane 200429 75 mg/kg (Q4Dx3) TPI 287 SY586-117-FP#2 24 mg/kg (Q4Dx3) |
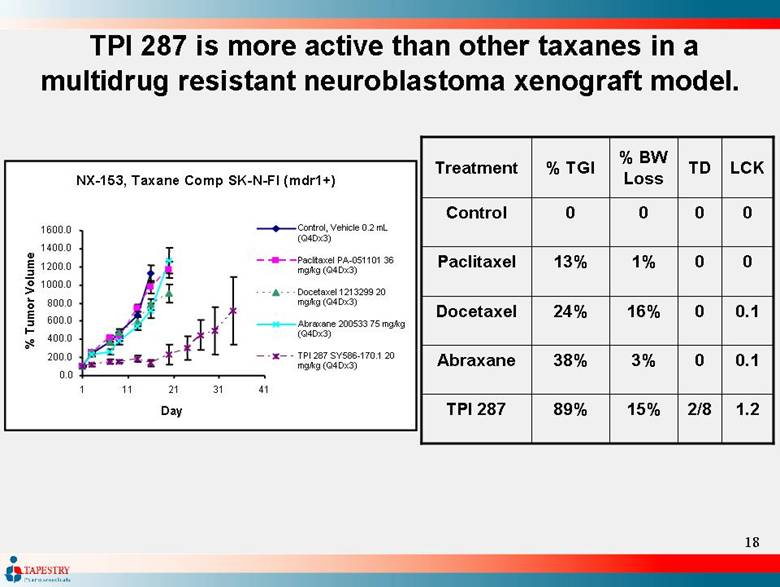
| TPI 287 is more active than other taxanes in a multidrug resistant neuroblastoma xenograft model. TPI 287 Abraxane Docetaxel Paclitaxel Control Treatment 1.2 2/8 15% 89% 0.1 0 3% 38% 0.1 0 16% 24% 0 0 1% 13% 0 0 0 0 LCK TD % BW Loss % TGI NX-153, Taxane Comp SK-N-FI (mdr1+) 0.0 200.0 400.0 600.0 800.0 1000.0 1200.0 1400.0 1600.0 1 11 21 31 41 Day % Tumor Volume Control, Vehicle 0.2 mL (Q4Dx3) Paclitaxel PA-051101 36 mg/kg (Q4Dx3) Docetaxel 1213299 20 mg/kg (Q4Dx3) Abraxane 200533 75 mg/kg (Q4Dx3) TPI 287 SY586-170.1 20 mg/kg (Q4Dx3) |
| Unlike other taxanes, TPI 287 is not actively pumped out of cells by MDR1 efflux pump 17.40 9.31 paclitaxel 6.17 TPI 287 B A A B Compound 27.9 paclitaxel 15.1 22.8 TPI 287 B A A B Compound MDCKII-MDR permeability rate Caco-2 permeability rate Conclusions: TPI 287 is not a P-gp (MDR1) substrate TPI 287 may be orally bioavailable 4.9 8.39 |
| TPI 287: Rodent Oral Bioavailability Oral administration of TPI 287 shows 35% – 50% bioavailability TPI 287 has anti-tumor activity for a variety of different tumor types when dosed orally in mouse xenograft tumor models |
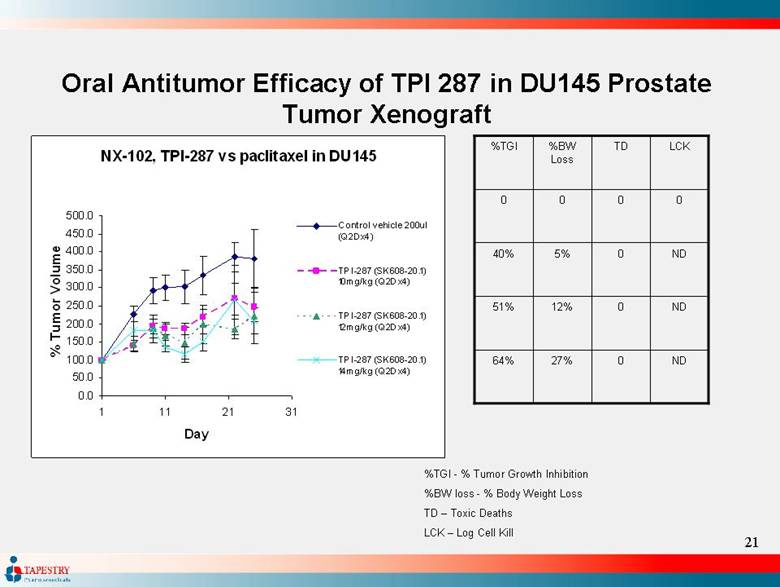
| Oral Antitumor Efficacy of TPI 287 in DU145 Prostate Tumor Xenograft ND 0 27% 64% ND 0 12% 51% ND 0 5% 40% 0 0 0 0 LCK TD %BW Loss %TGI %TGI - % Tumor Growth Inhibition %BW loss - % Body Weight Loss TD – Toxic Deaths LCK – Log Cell Kill NX-102, TPI-287 vs paclitaxel in DU145 0.0 50.0 100.0 150.0 200.0 250.0 300.0 350.0 400.0 450.0 500.0 1 11 21 31 Day % Tumor Volume Control vehicle 200ul (Q2Dx4) TPI-287 (SK608-20.1) 10mg/kg (Q2Dx4) TPI-287 (SK608-20.1) 12mg/kg (Q2Dx4) TPI-287 (SK608-20.1) 14mg/kg (Q2Dx4) |
| TPI 287: Preclinical Summary In Vitro Studies More active than standard therapies in multiple cell lines expressing MDR1 Tumor cell lines included breast, colon, lung, pancreas, prostate In Vivo Studies TPI 287 shows oral bioavailability in rodents Tumor Xenograft Studies Greater activity than other taxanes in breast, colon, lung, and prostate tumors models expressing MDR1 Oral administration of TPI 287 results in anti-tumor activity in a variety of tumor types |
| Corporate Overview |
| Milestones Begin 1st Phase II Trial in Prostate Cancer 1Q07 Determine MTD in Q7Dx3 Phase I Trial 2Q07 Begin oral Phase I/II 2Q07 Begin 1st Phase II Trial in NSCLC 3Q07 Present data at upcoming scientific meetings |
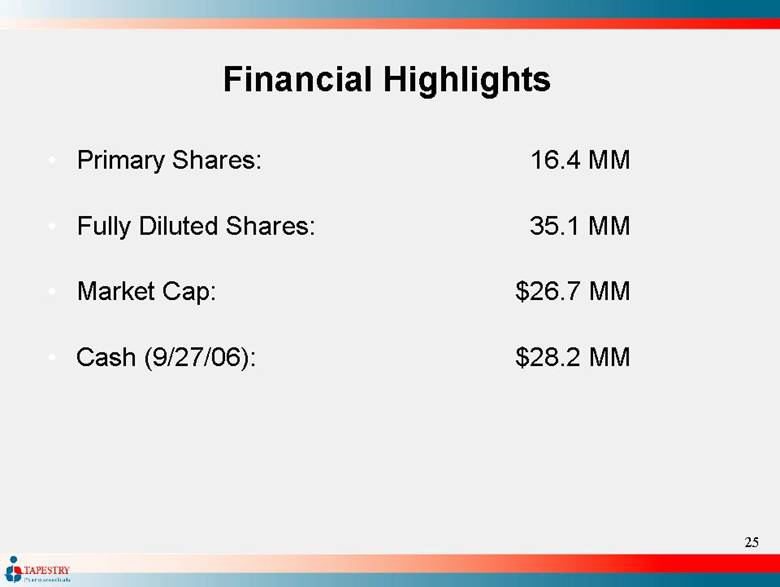
| Financial Highlights Primary Shares: 16.4 MM Fully Diluted Shares: 35.1 MM Market Cap: $26.7 MM Cash (9/27/06): $28.2 MM |
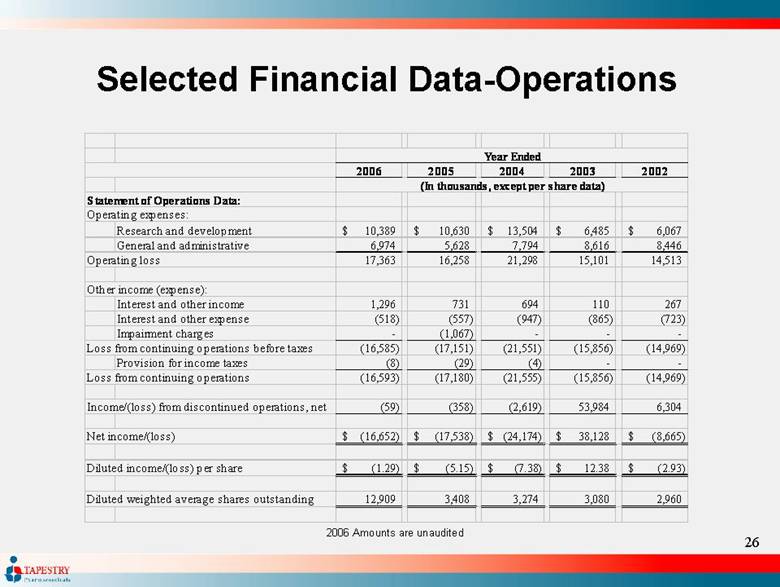
| Selected Financial Data-Operations 2006 Amounts are unaudited 2006 2005 2004 2003 2002 Statement of Operations Data: Operating expenses: Research and development 10,389 $ 10,630 $ 13,504 $ 6,485 $ 6,067 $ General and administrative 6,974 5,628 7,794 8,616 8,446 Operating loss 17,363 16,258 21,298 15,101 14,513 Other income (expense): Interest and other income 1,296 731 694 110 267 Interest and other expense (518) (557) (947) (865) (723) Impairment charges - (1,067) - - - Loss from continuing operations before taxes (16,585) (17,151) (21,551) (15,856) (14,969) Provision for income taxes (8) (29) (4) - - Loss from continuing operations (16,593) (17,180) (21,555) (15,856) (14,969) Income/(loss) from discontinued operations, net (59) (358) (2,619) 53,984 6,304 Net income/(loss) (16,652) $ (17,538) $ (24,174) $ 38,128 $ (8,665) $ Diluted income/(loss) per share (1.29) $ (5.15) $ (7.38) $ 12.38 $ (2.93) $ Diluted weighted average shares outstanding 12,909 3,408 3,274 3,080 2,960 Year Ended (In thousands, except per share data) |
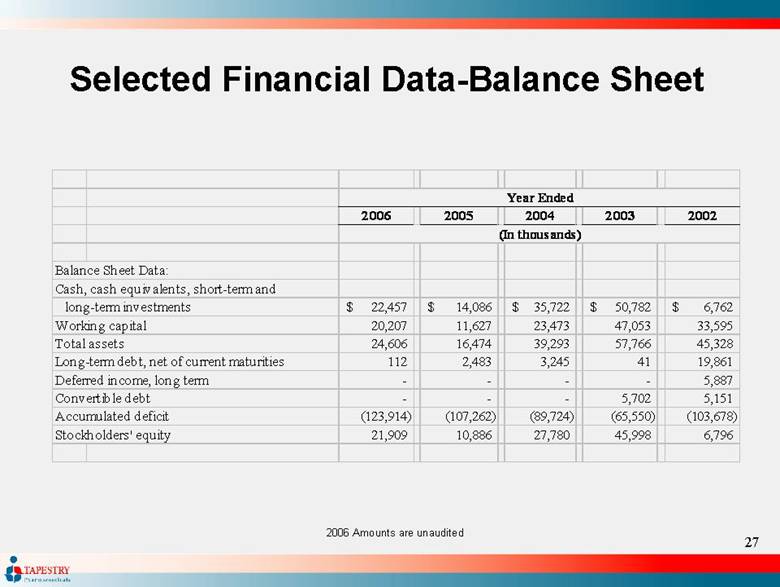
| Selected Financial Data-Balance Sheet 2006 Amounts are unaudited 2006 2005 2004 2003 2002 Balance Sheet Data: Cash, cash equivalents, short-term and 22,457 $ 14,086 $ 35,722 $ 50,782 $ 6,762 $ Working capital 20,207 11,627 23,473 47,053 33,595 Total assets 24,606 16,474 39,293 57,766 45,328 Long-term debt, net of current maturities 112 2,483 3,245 41 19,861 Deferred income, long term - - - - 5,887 Convertible debt - - - 5,702 5,151 Accumulated deficit (123,914) (107,262) (89,724) (65,550) (103,678) Stockholders' equity 21,909 10,886 27,780 45,998 6,796 Year Ended (In thousands) long-term investments |
| Management Team Leonard Shaykin Chairman and CEO Donald Picker, Ph.D. President – Genta (Genasense®), Johnson Matthey (Carboplatin, Satraplatin and Picoplatin) Martin Batt COO – Grisanti, Galef & Goldress Gordon Link CFO – Synergen, Deloitte & Touche Sandra Silberman, M.D., Ph.D. CMO – Eisai (Halichondrin), Novartis (GleevecTM), Pfizer (TarcevaTM) |
| Management Team James McChesney, Ph.D. CSO – Natural Products Chemistry - Director of Research Institute of Pharmaceutical Sciences and former Chairman of the Department of Pharmacognosy, University of Mississippi David Emerson, Ph.D. VP - Cancer Biology - OSI, Gilead and GSK Gilles Tapolsky, Ph.D. VP - Product Development - Rhone Poulenc Rorer, Flamel Technologies |
| Leonard P. Shaykin Chairman of the Board, Chief Executive Officer Stephen K. Carter, M.D. Former Senior Vice President, Worldwide Clinical Research and Development, Bristol-Myers Squibb and Deputy Director, Division of Cancer Treatment, National Cancer Institute George M. Gould, Esq. Attorney, Of Counsel, Gibbons, Del Deo, Dolan, Giffinger & Vecchione; formerly Vice President and Chief Patent Counsel of Hoffman-La Roche Arthur Hull Hayes, Jr., M.D. President, MediScience Associates a pharmaceutical consulting company; Former FDA Commissioner The Honorable Richard N. Perle Former U.S. Assistant Secretary of Defense Patricia Pilia, Ph.D. Co-founder of Company Robert E. Pollack, Ph.D. Professor of Biological Sciences and Director of the Center for the Study of Science and Religion at Columbia University Elliot M. Maza, J.D., C.P.A. Chief Financial Officer of Intellect Neurosciences; formerly Partner at Ernst & Young and employed at Goldman Sachs & Co, and J.P. Morgan Securities Board of Directors |
| Scientific Advisory Board Paul A. Bunn, Jr., M.D. (Chair) Director of the University of Colorado Cancer Center, past president of American Society of Clinical Oncology (2002-2003), and former chairman of the Oncologic Drugs Advisory Committee (ODAC) for FDA S. Gail Eckhardt, M.D. Professor of Medicine and Director of the Developmental Therapeutics and GI Malignancies Programs at the University of Colorado Health Sciences Center in Denver Eric K. Rowinsky, M.D. CMO, Imclone, Former Director of the Institute for Drug Development, Cancer Therapy and Research Center and Clinical Professor of Medicine at the University of Medicine at the University of Texas Health Science Center at San Antonio Daniel D. Von Hoff, M.D. Professor of Medicine, Pathology, Molecular and Cellular Biology and the Director of the Arizona Health Sciences Center’s Cancer Therapeutics Program Susan Band Horwitz, Ph.D. Distinguished University Professor, Rose C. Falkenstein Professor of Cancer Research, and Co-Chair of the Department of Molecular Pharmacology at the Albert Einstein College of Medicine. Associate Director for Therapeutics at the Albert Einstein Cancer Center. |
| Investment Rationale Proprietary next generation taxane Potential for best in class Multiple Phase II programs Experienced oncology drug development team Large commercial opportunity Strong cash position Modest burn rate |
| Strategies for Life TM |