Exhibit 99.1
| Tapestry Pharmaceuticals, Inc. Presentation to Investors |
| Forward Looking Statements Any statements in this presentation that are not historical facts are “forward-looking statements” that involve risks and uncertainties. Such forward-looking statements include the statements regarding the potential for TPI 287 in the treatment of disease, the Company’s intention to commence additional clinical trials for TPI 287, the timing of commencement or of clinical trials for TPI 287 and that TPI 287 may be active in combination with other chemotherapeutic drugs. Such statements involve known and unknown risks, uncertainties and other factors that may cause our actual results, performance or achievements to be materially different from the results, performance or achievements expressed or implied by such forward-looking statements. Such risks, uncertainties and other factors include risks that clinical trials for TPI 287 will be delayed due to institutional approvals, patient recruitment, formulation and manufacturing difficulties, delays in finalizing and receiving approval of Phase 2 protocols, negotiations with regulatory agencies or other factors; and that human clinical trials may show that TPI 287 is unsafe and/or ineffective in treating cancer in humans. General implementation risks associated with development of TPI 287 include those that we are blocked or limited in the development of TPI 287 because of the intellectual property rights of third parties; that we are limited in our ability to obtain, maintain and enforce our own intellectual property; that development of TPI 287 is delayed or terminated because the costs of further development exceed its value; and that the Company’s resources will be insufficient to continue development. Additional risks, uncertainties and other factors are identified under the captions “Risk Factors” and “Special Note Regarding Forward-Looking Statements” in the Company’s reports filed from time to time with the Securities and Exchange Commission (the “SEC”), including its Annual Report on Form 10-K for the year ended December 27, 2006 filed with the SEC on March 7, 2007 as well as an amendment thereto on Form 10-K/A filed with the SEC on April 26, 2007. The Company disclaims any intention or obligation to update publicly or revise any forward-looking statements, whether as a result of new or additional information, future events or otherwise. |
| Tapestry: Advancing the Development of TPI 297 Proprietary next generation taxane Potential for best in class Multiple Phase 2 programs Experienced oncology drug development team Large commercial opportunity Strong cash position Modest burn rate |
| TPI 287: Proprietary Next Generation Taxane |
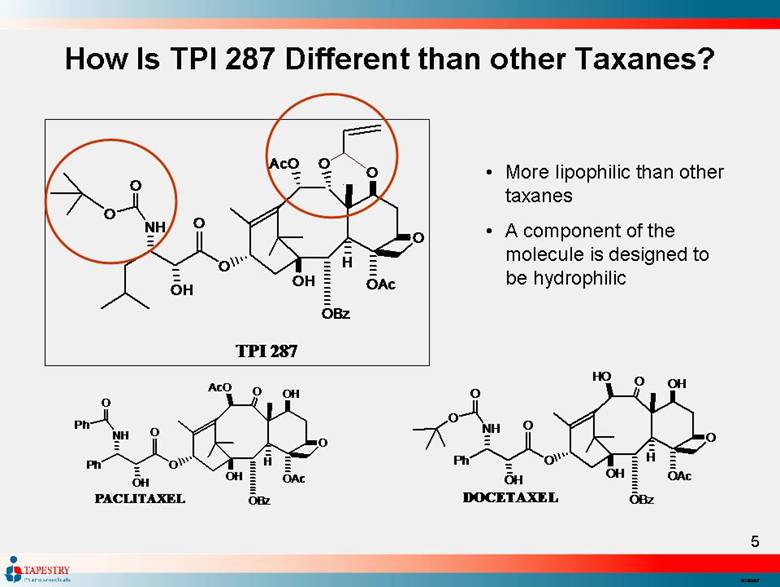
| More lipophilic than other taxanes A component of the molecule is designed to be hydrophilic How Is TPI 287 Different than other Taxanes? O H O O O H A c O O B z H O A c P h O O O H N H P h O P A C L I T A X E L O H O O O H H O O B z H O A c P h O O O H N H O O D O C E T A X E L O O O H A c O O B z H O A c O O O H N H O O T P I 2 8 7 O |
| TPI 287: Significantly Differentiated from Currently Marketed Taxanes TPI 287 has demonstrated the potential for: Differentially higher activity than taxanes in resistant tumors (mdr1) Both innately resistant tumors and those which develop resistance Oral activity and bioavailability Enables flexibility of dosing and scheduling for optimal combination therapy with other anti-cancer drugs Crossing the blood-brain barrier CNS tumors (primary or metastatic) Activity in tumors not currently approved for treatment by taxanes |
| TPI 287 Pre-clinical Biology Studies |
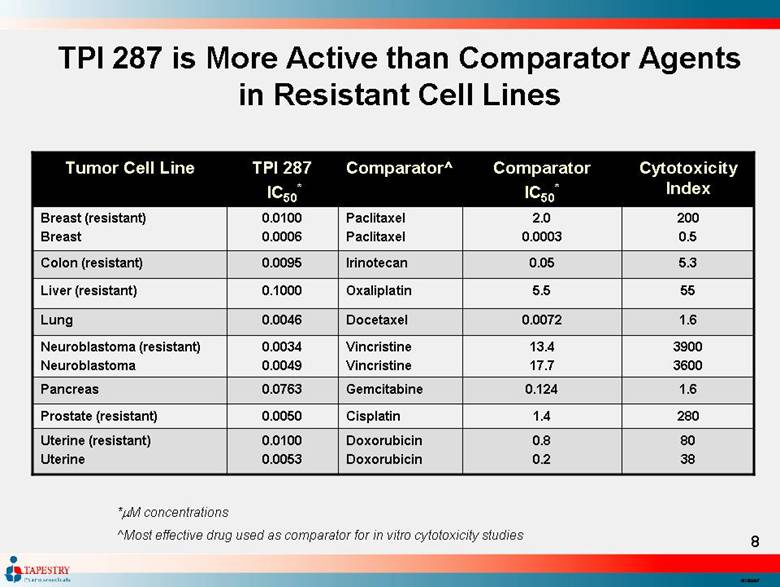
| TPI 287 is More Active than Comparator Agents in Resistant Cell Lines 55 5.5 Oxaliplatin 0.1000 Liver (resistant) 1.6 0.0072 Docetaxel 0.0046 Lung 80 38 0.8 0.2 Doxorubicin Doxorubicin 0.0100 0.0053 Uterine (resistant) Uterine 1.4 0.124 13.4 17.7 0.05 2.0 0.0003 Comparator IC50* 280 1.6 3900 3600 5.3 200 0.5 Cytotoxicity Index Cisplatin 0.0050 Prostate (resistant) Gemcitabine 0.0763 Pancreas Vincristine Vincristine 0.0034 0.0049 Neuroblastoma (resistant) Neuroblastoma Irinotecan 0.0095 Colon (resistant) Paclitaxel Paclitaxel 0.0100 0.0006 Breast (resistant) Breast Comparator^ TPI 287 IC50* Tumor Cell Line *M concentrations ^Most effective drug used as comparator for in vitro cytotoxicity studies |
| Unlike other Taxanes, TPI 287 is not Actively Pumped Out of Cells by mdr1 Efflux Pump 17.40 9.31 paclitaxel 6.17 TPI 287 B→A A→B Compound 27.9 paclitaxel 15.1 22.8 TPI 287 B→A A→B Compound MDCKII-mdr1 permeability rate Caco-2 permeability rate Conclusions: TPI 287 is not a P-gp (mdr1) substrate TPI 287 may be orally bioavailable 4.9 8.39 |
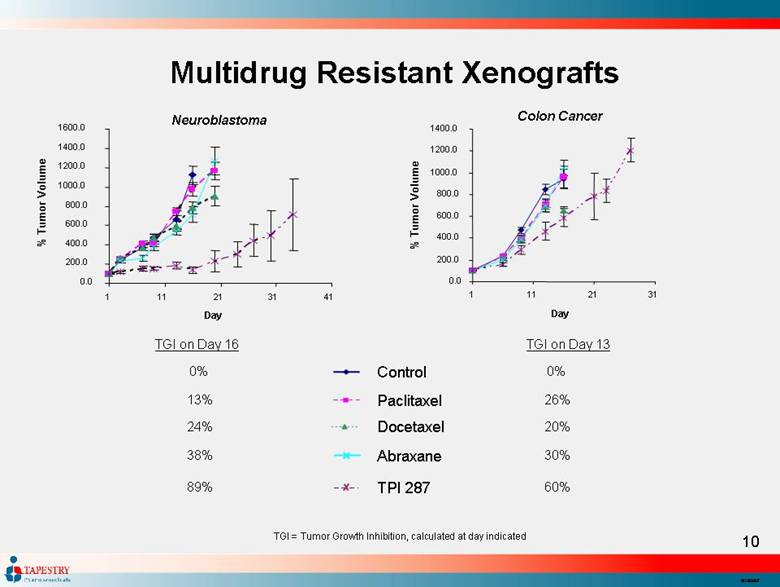
| Multidrug Resistant Xenografts 0.0 200.0 400.0 600.0 800.0 1000.0 1200.0 1400.0 1600.0 1 11 21 31 41 Day % Tumor Volume TGI = Tumor Growth Inhibition, calculated at day indicated TGI on Day 13 0% 26% 20% 30% 60% Control Paclitaxel Docetaxel Abraxane TPI 287 0.0 200.0 400.0 600.0 800.0 1000.0 1200.0 1400.0 1 11 21 31 Day % Tumor Volume TGI on Day 16 0% 13% 24% 38% 89% Neuroblastoma Colon Cancer |
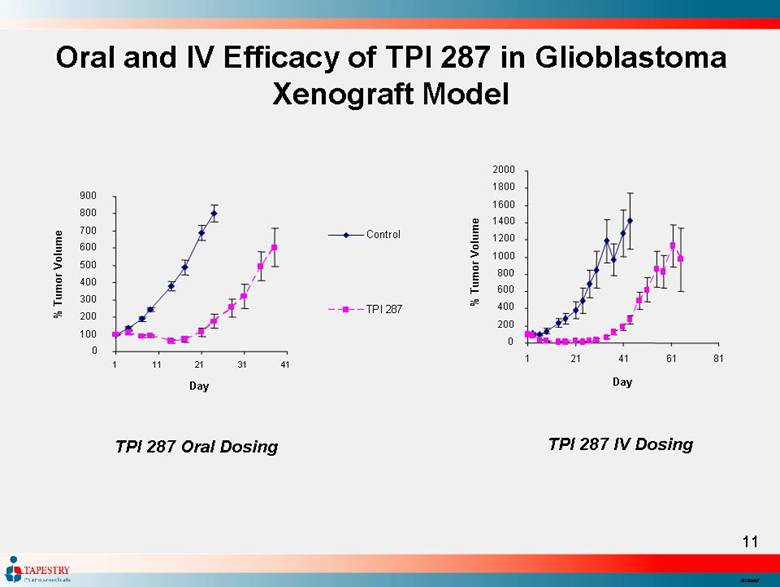
| Oral and IV Efficacy of TPI 287 in Glioblastoma Xenograft Model TPI 287 IV Dosing 0 200 400 600 800 1000 1200 1400 1600 1800 2000 1 21 41 61 81 Day % Tumor Volume TPI 287 Oral Dosing 0 100 200 300 400 500 600 700 800 900 1 11 21 31 41 Day % Tumor Volume Control TPI 287 |
| Intracranial Mouse GBM Xenograft Study ILS = Increased Life Span; measured at the median survival time (50% of mice alive) ILS 0% 45% 380% 650% Intracranial Mouse GBM Xenograft Study 0 10 20 30 40 50 60 70 80 90 100 0 20 40 60 80 100 Days % Survival Control TPI 287 Temozolamide Temozolamide + TPI 287 |
| TPI 287: Pre-clinical Summary In vitro cell lines: Extremely potent activity in multiple cell lines, including tumors of the breast, colon, lung, pancreas, & prostate Greater activity (up to 3000-fold) compared to standard agents in resistant (mdr1 gene expressing) cell lines Tumor Xenograft Studies Greater activity than other taxanes in breast, colon, lung, and prostate tumors models that express mdr1 (resistant) Tumor Orthotopic Xenograft Studies Shows significant antitumor and survival benefit in human glioblastoma intracranially Combination with temozolamide (approved for human therapy of glioblastoma) shows significant additivity Oral bioavailability and efficacy demonstrated Potential for novel scheduling, combinations and strategies |
| TPI 287 Program Overview Preclinical Data Extremely potent activity in multiple in vitro cell lines, including tumors of the breast, colon, lung, pancreas, & prostate Greater activity (up to 3000-fold) compared to standard agents in resistant (mdr1 gene expressing) cell lines Oral bioavailability and efficacy demonstrated potential for novel scheduling, combinations and strategies Completed Phase 1 Program One Phase 1 clinical trial completed and a second nearing completion Data presented at ASCO 2007 Enrolling Phase 2 Plan Enrolling prostate cancer trial Initiating multiple Phase 2 clinical trials in 2007 |
| TPI 287: Phase 1 Clinical Trials |
| Phase 1 Clinical Trials (Based on June, 2007 ASCO presentation) TPI 287-01 Weekly x 3 with one week rest (28 day cycle) Assessment of safety and efficacy after 30 patients enrolled (29 evaluable) Median age 60.5 years (24 – 86) 17 males/13 females TPI 287-02 Once every 3 weeks (21 day cycle) Assessment of safety and efficacy after 21 patients Median age 58 years (35 – 78) 7 males/14 females |
| Summary of Safety – Weekly Dosing (Based on June, 2007 ASCO presentation) Treatment with 150 mg/m2 well tolerated (Phase 2 recommended dose) with DLTs at 185 mg/m2 (neuropathy) Most patients experience Grades 1 or 2 severity adverse events (more at higher doses) fatigue (52% of patients) constipation (34% of patients) dyspnea (31% of patients) nausea (31% of patients) Severe (Grade 3) adverse events in 17 patients (57%) and Grade 4 in 3 patients (10%) Three patients had Grade 3 neuropathy which proved to be dose-limiting (2 had baseline neuropathy) There were no Grade 3 or 4 myelosuppressive events |
| Summary of Safety – 21-Day Dosing (Based on June, 2007 ASCO presentation) The MTD of TPI 287 at 160 mg/m2 is the recommended Phase 2 dose with DLTs at 185 mg/m2 (neuropathy and neutropenia) All patients experienced at least one adverse event, most frequently: Anemia (43% of patients) Neuropathy (43% of patients) Diarrhea, nausea, and vomiting (33% of patients) Grade 3 adverse events in 13 patients (62%) and Grade 4 adverse events in 7 patients (33%) Four patients had Grade 3 neuropathy (one had neuropathy at baseline) Three patients had neutropenia (one Grade 3 and two Grade 4) One patient had Grade 3 anemia |
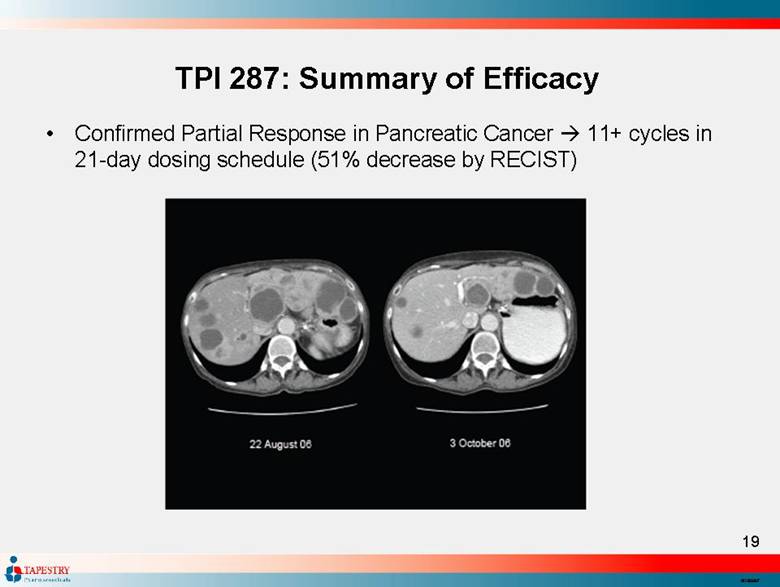
| TPI 287: Summary of Efficacy Confirmed Partial Response in Pancreatic Cancer 11+ cycles in 21-day dosing schedule (51% decrease by RECIST) |
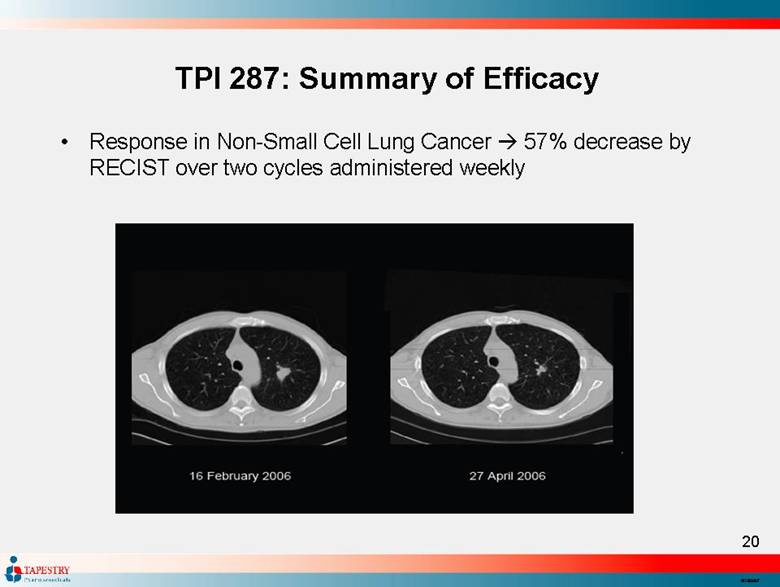
| TPI 287: Summary of Efficacy Response in Non-Small Cell Lung Cancer 57% decrease by RECIST over two cycles administered weekly |
| TPI 287: Summary of Efficacy Stable Disease Breast cancer Therapy over 4 previous months with progressive disease Stable disease over 7 cycles (21-day dosing) Pancreatic cancer Over 3 cycles in weekly dosing regimen 15% decrease by RECIST |
| TPI 287: Interesting Activity and Profile Data from Phase 1 Clinical Trials Presented at ASCO 2007 Two Phase 1 Clinical Trials Completed Weekly (3 out of 4 weeks) and once every 3 weeks Well tolerated in heavily pretreated patient population Achieved dose recommended for Phase 2 Side Effect profile Few serious myelosuppressive events Dose limiting toxicity neuropathy, usually exacerbation of prior neuropathy Efficacy Four patients showed clinical benefit Tumors that had been exposed to taxanes (lung, breast) Tumors that are not generally sensitive to taxanes (pancreatic) |
| TPI 287: Phase 2 Clinical Trial Designs |
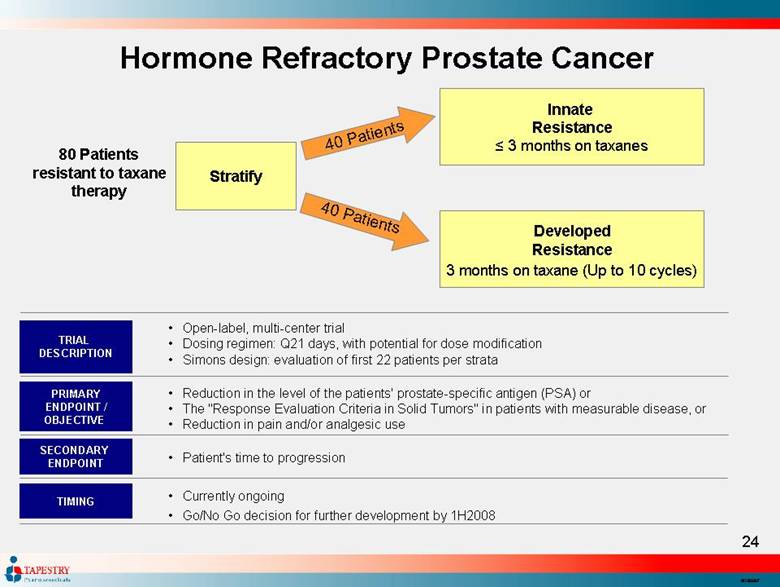
| Hormone Refractory Prostate Cancer 80 Patients resistant to taxane therapy Stratify 40 Patients 40 Patients Innate Resistance ≤ 3 months on taxanes Developed Resistance 3 months on taxane (Up to 10 cycles) Open-label, multi-center trial Dosing regimen: Q21 days, with potential for dose modification Simons design: evaluation of first 22 patients per strata TRIAL DESCRIPTION PRIMARY ENDPOINT / OBJECTIVE SECONDARY ENDPOINT Reduction in the level of the patients' prostate-specific antigen (PSA) or The "Response Evaluation Criteria in Solid Tumors" in patients with measurable disease, or Reduction in pain and/or analgesic use Patient's time to progression Currently ongoing Go/No Go decision for further development by 1H2008 TIMING |
| Glioblastoma Multiforme / Primary CNS Tumors Open-label, multi-center trial Up to 50 patients Patients with GBM that has recurred or progressed following prior radiation and temozolomide therapy Dosing regimen: Q7 days x 3 Q28 days Go/No Go based on 6 month PFS Six-month progression free survival (PFS) Evaluate any response to treatment including adverse events Initiate in 2H 2007 Open-label, multi-center dose escalation trial Up to 60 patients Patients whose condition has recurred or progressed following prior temozolamide therapy Determine MTD of the combination of TPI 287 and temozolomide Response to treatment Pharmacokinetics of both TPI 287 and temozolomide Initiate in 2H2008 PHASE 2 TRIAL: AS SINGLE AGENT IN GBM PHASE 1b/2 TRIAL: IN COMBINATION WITH TEMOZOLAMIDE FOR PRIMARY CNS TUMORS TRIAL DESCRIPTION PRIMARY ENDPOINT / OBJECTIVE SECONDARY ENDPOINT TIMING |
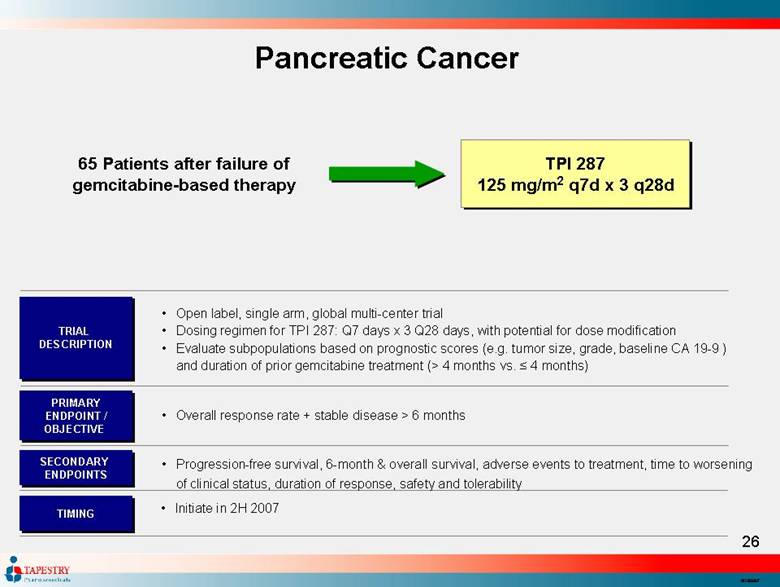
| Pancreatic Cancer 65 Patients after failure of gemcitabine-based therapy TPI 287 125 mg/m2 q7d x 3 q28d Open label, single arm, global multi-center trial Dosing regimen for TPI 287: Q7 days x 3 Q28 days, with potential for dose modification Evaluate subpopulations based on prognostic scores (e.g. tumor size, grade, baseline CA 19-9 ) and duration of prior gemcitabine treatment (> 4 months vs. ≤ 4 months) TRIAL DESCRIPTION PRIMARY ENDPOINT / OBJECTIVE SECONDARY ENDPOINTS Overall response rate + stable disease > 6 months Progression-free survival, 6-month & overall survival, adverse events to treatment, time to worsening of clinical status, duration of response, safety and tolerability Initiate in 2H 2007 TIMING |
| TPI 287 Clinical Development Plan Development Stage Dosage Form Indication Pre - Clinical Phase 1 Phase 2 Phase 3 Next Milestone / Timing Hormone Refractory Prostate Cancer Go/No Go by 1H 20 0 8 Glioblastoma Multiforme Initiate Ph ase 2 2H 2007 Primary C NS tumors (Combination with temozolamide) Initiate Phase 1b/2 2H 200 8 Pancreatic Cancer Initiate Phase 2 2H 2007 IV Exploratory Phase 2 (Additional Tumor Types) Initiate beginning 2 H 2008 Multiple Cancers Initiate Phase 1 b PK s tudy 2H 2007 Oral |
| Market and Corporate Overviews |
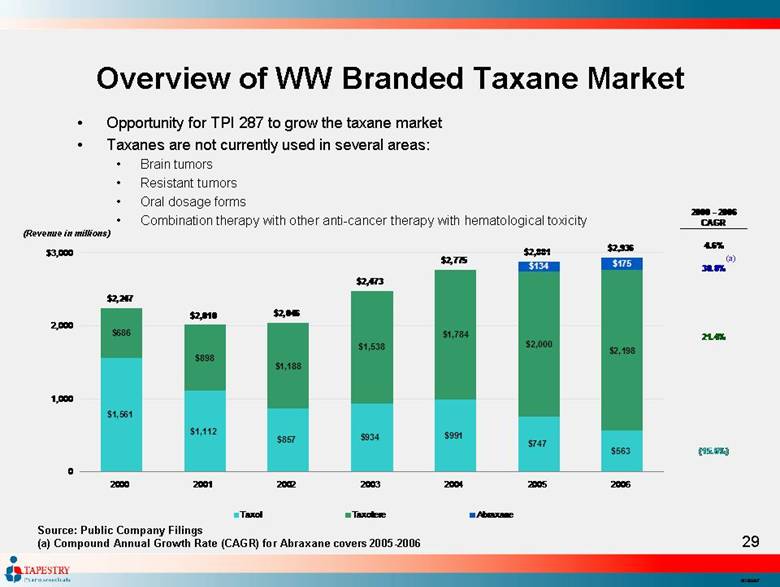
| Overview of WW Branded Taxane Market Source: Public Company Filings (a) Compound Annual Growth Rate (CAGR) for Abraxane covers 2005-2006 (Revenue in millions) (a) Opportunity for TPI 287 to grow the taxane market Taxanes are not currently used in several areas: Brain tumors Resistant tumors Oral dosage forms Combination therapy with other anti-cancer therapy with hematological toxicity 2000 - 2006 CAGR 4.6% 30.8% 21.4% (15.6%) $1,561 $1,112 $857 $934 $991 $747 $563 $686 $898 $1,188 $1,538 $1,784 $2,000 $2,198 $134 $175 $2,247 $2,936 $2,881 $2,775 $2,473 $2,046 $2,010 0 1,000 2,000 $3,000 2000 2001 2002 2003 2004 2005 2006 Taxol Taxotere Abraxane |
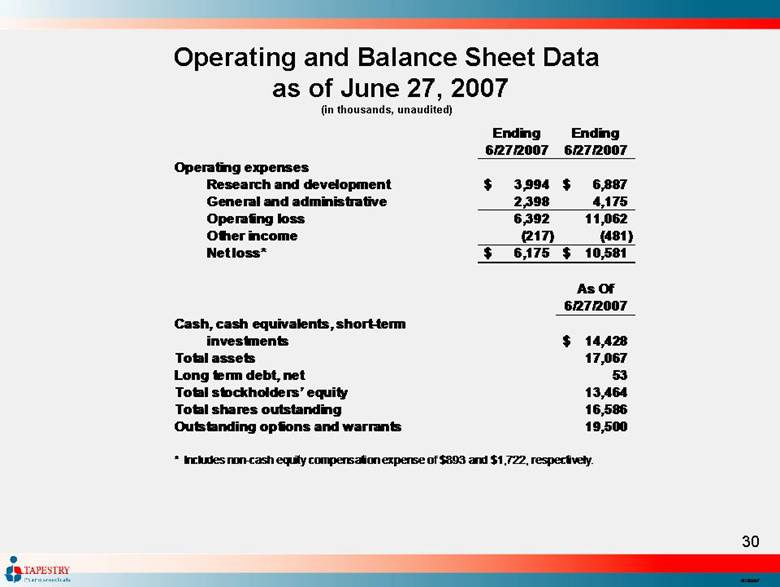
| Operating and Balance Sheet Data as of June 27, 2007 (in thousands, unaudited) Ending Ending 6/27/2007 6/27/2007 Operating expenses Research and development 3,994 $ 6,887 $ General and administrative 2,398 4,175 Operating loss 6,392 11,062 Other income (217) (481) Net loss* 6,175 $ 10,581 $ As Of 6/27/2007 Cash, cash equivalents, short-term investments 14,428 $ Total assets 17,067 Long term debt, net 53 Total stockholders’ equity 13,464 Total shares outstanding 16,586 Outstanding options and warrants 19,500 * Includes non-cash equity compensation expense of $893 and $1,722, respectively. |
| Leonard Shaykin Chairman and CEO Donald Picker, Ph.D. President – Genta (Genasense®), Johnson Matthey (Carboplatin, Satraplatin and Picoplatin), GSK Sandra Silberman, M.D., Ph.D. CMO – Eisai (Halichondrin), Novartis (GleevecTM), Pfizer (TarcevaTM) Gordon Link CFO – Synergen, Deloitte & Touche Martin Batt COO – Grisanti, Galef & Goldress James McChesney, Ph.D. CSO – Natural Products Chemistry - Director of Research Institute of Pharmaceutical Sciences and former Chairman of the Department of Pharmacognosy, University of Mississippi David Emerson, Ph.D. VP of Cancer Biology - OSI, Gilead and GSK Gilles Tapolsky, Ph.D. VP of Product Development - Rhone Poulenc Rorer, Flamel Technologies Management Team |
| Scientific Advisory Board Moshe Talpaz M.D. (Chair) Professor, Internal Medicine, Associate Director, Translational Research and Associate Chief of Hematologic Malignancies, University of Michigan Comprehensive Cancer Center Esteban Cvitkovic, M.D. Senior Medical and Scientific Consultant, AAIOncology (formely CAC). Board Member and Scientific Consultant, Access Pharmaceuticals, Inc. Chief Scientific Officer and Founder, OncoEthix. Scientific President, Fondation Nélia et Amadeo Barletta. Academic part-time practice. Susan Band Horwitz, Ph.D. Distinguished University Professor, Rose C. Falkenstein Professor of Cancer Research, and Co-Chair of the Department of Molecular Pharmacology at the Albert Einstein College of Medicine. Associate Director for Therapeutics at the Albert Einstein Cancer Center Patricia A. Pilia, Ph.D. Co-founder and Former Executive Vice President of Tapestry Pharmaceuticals, Former Assistant Professor of Pathology and Assistant Director of the Immunopathology Diagnostic and Research Laboratories in the Colleges of Medicine, Dental Medicine and Graduate Studies at the Medical University of South Carolina Marcel Roczensweig M.D. CMO GPB-Biotech. Former Sr Vice President, Strategic Planning & Portfolio Management, Pharmaceutical Research Institute, Bristol-Myers Squibb Eric K. Rowinsky, M.D. CMO, Imclone. Former Director of the Institute for Drug Development, Cancer Therapy and Research Center and Clinical Professor of Medicine at the University of Medicine at the University of Texas Health Science Center at San Antonio Daniel D. Von Hoff, M.D. Professor of Medicine, Pathology, Molecular and Cellular Biology and the Director of the Arizona Health Sciences Center’s Cancer Therapeutics Program |
| Leonard P. Shaykin Chairman of the Board, Chief Executive Officer Stephen K. Carter, M.D. Former Senior Vice President, Worldwide Clinical Research and Development, Bristol-Myers Squibb and Deputy Director, Division of Cancer Treatment, National Cancer Institute George M. Gould, Esq. Attorney, Of Counsel, Gibbons, Del Deo, Dolan, Giffinger & Vecchione; formerly Vice President and Chief Patent Counsel of Hoffman-La Roche Arthur Hull Hayes, Jr., M.D. President, MediScience Associates a pharmaceutical consulting company; Former FDA Commissioner The Honorable Richard N. Perle Former U.S. Assistant Secretary of Defense Robert E. Pollack, Ph.D. Professor of Biological Sciences and Director of the Center for the Study of Science and Religion at Columbia University Elliot M. Maza, J.D., C.P.A. President and Chief Financial Officer of Intellect Neurosciences; formerly Partner at Ernst & Young and employed at Goldman Sachs & Co, and J.P. Morgan Securities Board of Directors |
| Upcoming Milestones IV Dosing Initiate Phase 2 Glioblastoma Multiforme 2H2007 Initiate Phase 2 Pancreatic Cancer 2H2007 Initiate Phase 1b/2 Primary CNS tumors 2H2008 Complete First Stage of Simons Design Phase 2 of HRPC 1H2008 Start Additional Exploratory Phase 2 Trials 2H2008 Oral Dosing Initiate Phase 1b PK study of Multiple Cancers 2H2007 |
| Tapestry: Advancing the Development of TPI 287 Proprietary next generation taxane Potential for best in class Multiple Phase 2 programs Experienced oncology drug development team Large commercial opportunity Strong cash position Modest burn rate |
| Strategies for Life TM |